Abstract
Objectives
Feline intracranial abscessation or empyema is infrequently reported in the veterinary literature. To date, the largest study is based on a population of 19 cats with otogenic infection. The aim of this study was to review a larger population of cats with intracranial empyema from multiple aetiologies and document their signalment, imaging findings, treatment protocols (including medical and/or surgical management) and to compare outcomes.
Methods
Cases presenting to a single referral centre over a 10 year period with compatible history, neurological signs and imaging findings consistent with intracranial abscessation and empyema were reviewed retrospectively.
Results
Twenty-three cats met the inclusion criteria. Advanced imaging (CT and/or MRI) was performed in 22/23 cats; one case was diagnosed via ultrasound. Ten cases underwent medical and surgical management combined, 10 underwent solely medical management and three were euthanased at the time of diagnosis. Short-term outcome showed that 90% of surgically managed and 80% of medically managed cats were alive at 48 h post-diagnosis. Long-term survival showed that surgically managed cases and medically managed cases had a median survival time of 730 days (range 1–3802 days) and 183 days (range 1–1216 days), respectively. No statistical significance in short- or long-term survival (P >0.05) was found between medically and surgically managed groups.
Conclusions and relevance
Feline intracranial abscessation and empyema are uncommon conditions that have historically been treated with combined surgical and medical management. This study documents that, in some cases, intracranial abscessation and empyema can also be successfully treated with medical management alone.
Introduction
Intracranial abscessation (IA; intra-axial collection of purulent material) and intracranial empyema (IE; suppuration within a pre-existing anatomical cavity) arise as a consequence of bacterial infection within the cranial cavity.1,2 IA and IE are infrequently seen in cats. Infection originating from local extension (eg, adjacent spread from the eyes, ears and sinuses), haematogenous spread, secondary to trauma (eg, skull fractures, penetrating foreign bodies) and iatrogenic infection are reported in the literature.3–7 These cases present with a wide range of neurological deficits due to the lesion location, the inflammatory response induced by the bacteria and/or secondary mass effect. Both aerobic and anaerobic bacteria have been isolated, with culture results typically yielding polymicrobial growth.3,7–9
IA and IE are severe and life-threatening diseases requiring emergency intervention; mortality rates as high as 100% have been reported.8,10 Treatment modalities consist of medical treatment with broad-spectrum antibiotics and supportive care, or combined medical and surgical intervention via craniectomy.3–5,11 Little information is currently available as to which treatment modality carries the most favourable prognosis.
The aim of this study is to describe medical and surgical treatment protocols for IA and IE and compare their effect on short- and long-term survival in feline patients.
Materials and methods
Ethical approval was granted by the Clinical Research Ethical Review Board at the Royal Veterinary College (RVC). Cats presenting to the RVC Queen Mother Hospital between April 2008 and August 2017 that had been diagnosed or treated for possible IA and/or IE were reviewed retrospectively. Terms entered into the search engine database included ‘intracranial empyema’, ‘intracranial abscess’, ‘meningoencephalitis’ and ‘otitis media and/or interna’.
Cats were included in the study if they had complete clinical records with magnetic resonance images, CT or ultrasound findings consistent with IA and/or IE. Data were collected regarding signalment, history and previous treatment prior to referral. All cases were required to have a documented neurological examination upon presentation. Pyrexia was defined by a rectal temperature over 39.2ºC. 12 Disease progression, imaging findings, additional diagnostics and therapeutics were recorded. Where available, ancillary test results such as feline immunodeficiency virus (FIV) and feline leukaemia virus (FeLV) status, cerebrospinal fluid (CSF) analysis and CSF and surgical swab culture and sensitivity results were recorded.
CT studies were performed with a Philips MX8000 16 MDCT unit, with 1.5 mm slice thickness for brain and 1–2 mm slice thickness for head sequences. Pre- and post-contrast exams were performed, with bone and soft tissue reconstructions. All magentic resonance images were obtained using a 1.5 Tesla Intera System (Philips Medical Systems). Each MRI study included a pre- and post-gadolinium contrast T1-weighted (T1W) series and T2-weighted (T2W) series in transverse and sagittal planes and transverse plane fluid-attenuated inversion recovery (FLAIR) sequence.
All images were independently reviewed by a board-certified veterinary neurologist and board-certified veterinary radiologist. A diagnosis of IA and/or IE was described further by location (intra-/extra-axial lesion and corresponding area of the brain), heterogenous or homogenous contrast enhancement, demarcation to surrounding tissues and secondary overlying soft tissue changes. Evidence of skull fractures and raised intracranial pressure such as midline shift, herniation through a craniotomy defect, caudal transtentorial, subfalcine and foramen magnum herniation and white matter oedema were recorded. 13
Cats were subsequently divided into solely medically treated or combined surgically and medically treated groups. Only those cases treated via craniectomy were defined as surgically treated; cases undergoing any other form of surgery (eg, ventral bulla osteotomy) were, therefore, categorised as medically managed.
Information regarding outcome was obtained via telephone consultation with the referring veterinarian and/or owner, combined with the referring vet clinical records and findings of re-examination appointments at the RVC. Short-term outcome was reviewed at 48 h and 1 month after diagnosis; longer-term outcome was assessed at 3 and 6 months. Outcome was classified as alive or dead, but, where available, survival was further categorised with a 1–3 grading system alongside neurological examination findings. Grade 1 was given if cats returned to being neurologically normal, grade 2 was defined as persistent mild neurological deficits that did not affect the normal ambulation and behaviour of the cat, and grade 3 was defined as persistent neurological abnormalities that significantly affected the patients’ ability to ambulate and display normal behaviours.
The two-tailed Fisher’s exact test was used to ascertain the significance of short- and long-term survival between surgically and medically managed groups. Other variables assessed included the effect of empyema location, use of steroids and the development of seizures upon outcome.
Results
A total of 27 cats presented to the Queen Mother Hospital for Animals between April 2008 and August 2017. One cat was excluded as initial diagnostics and surgery were performed at an alternative referral centre, and a second cat was excluded owing to a lack of preoperative imaging. A further two cats were excluded as they failed to fulfil the imaging criteria of IA and/or IE. Twenty-three cats were included in the study population.
The included cats had a median age of 7.4 years at presentation (range 7 months to 16 years) and had a male predominance, with 15 neutered males (65.2%), two entire males (8.7%), five neutered females (21.7%) and one entire female (4.3%). Male cats were over-represented in comparison to the hospital population over the same time period. Cats encompassed a range of breeds: domestic shorthair (n = 13), domestic mediumhair (n = 1), domestic longhair (n = 4), Bengal (n = 2), British Shorthair (n = 1), Siamese (n = 1) and exotic shorthair (n = 1).
Clinical signs at presentation
Cats had a variable duration of clinical signs prior to presentation reflecting the underlying aetiology and location of the IA and/or IE. Cats presenting with IE due to otitis media/interna (OMI) generally had a longer history (median duration 34 days [range 7–183 days] in 6/23 cats) compared with other aetiologies such as trauma (median duration 6 days [range 2–21days]). A history of cat bites and associated abscessation was the most common finding, reported in 13/23 cases, 11 of which were male.
Only two cats had normal clinical examination findings on presentation. Nine cats had visible wounds on their heads with two of these still actively discharging purulent material. Six cats had concurrent grade II–III/IV (n = 5) heart murmurs or a gallop rhythm (n = 1) on auscultation. These cats did not have a history of cardiac disease and no further cardiac assessment was performed at the time of presentation. Two cats had unilateral serous nasal discharge and one cat bilateral purulent nasal discharge with stertor. One cat had aural discharge and otitis externa noted on clinical examination, with concurrent Horner’s syndrome ipsilateral to the otitis.
Other clinical examination findings included tachycardia (n = 10), tachypnoea (n = 3), pale mucous membranes (n = 2) and a dull or quiet demeanour (n = 9). Rectal temperature was recorded in 21/23 cats; 2/21 cats were pyrexic at presentation; and a further 5/21 cats had pyrexia documented prior to referral but had recently received anti-inflammatory medication (non-steroidal anti-inflammatory drugs [NSAIDs] or anti-inflammatory doses of steroids).
Neurological examination findings at presentation
All 23 cats had a history of progressive, multifocal neurological dysfunction that reflected the location of their IA and/or IE. The most common neurolocalisation was to the forebrain in 12/23 cats; commonly described examination findings included altered mental status and an absent menace response (Table 1). Seizure activity was infrequently reported; 2/23 cats exhibited generalised seizures and 1/23 partial seizures prior to referral.
Summary of neurological examination findings

ICP = intracranial pressure; PLR = pupillary light reflex
Imaging
MRI studies were available for 21/23 cats (Table 2 and supplementary material). Fourteen cats had extra-axial lesions consistent with IE (Figure 1), four intra-axial IA (Figure 2) and three had findings compatible with IA and IE. Distribution was classified as forebrain (n = 13), brainstem (n = 4), cerebellar (n = 1) or multifocal (n = 3); and localisation further described by affected lobes of the cerebrum. Specifically, the temporal (n = 14) and frontal (n = 10) lobes were the most commonly affected. Solitary and well-defined lesions were the most common finding in 18/21 cats. Nine cats had concurrent overlying bite wounds; of these, seven had associated skull fractures. In 2/21 cases nasal infection with associated sinus pathology was visible.
MRI findings of 21 cats, including affected lobes of the cerebrum

Y = yes; N = no; Olf = olfactory; T = temporal including piriform; F = frontal; P = parietal; Occ = occipital; CTH = caudal transtentorial herniation; HTFM = herniation through foramen magnum; SFH = subfalcine herniation; MS = midline shift; OMI = otitis media/interna

Intracranial empyema in a 13-year-old male neutered domestic shorthair cat. (a) T2-weighted (W) and (b) T1W post-gadolinium transverse plane images show the left subarachnoid space is widened by a slightly heterogeneous layer of hyperintensity with mild peripheral contrast enhancement (white arrows). There is also evidence of midline shift (white arrowhead) away from the affected site

(a) T2-weighted (W) magnetic resonance transverse plane image of a 5-year-old female neutered domestic shorthair cat with an intracranial abscess in the frontal and temporal lobes (white arrow) that partially supresses on (b) fluid-attenuated inversion recovery (FLAIR) sequence (white arrow). There was increased T2W and FLAIR signal in the surrounding white matter (black arrows), the musculature overlying the calvarium (white arrowheads) and midline shift of the falx away from the lesion. (c) The T2W sagittal plane image shows signs of increased intracranial pressure with caudal transtentorial (black arrow) and caudal cerebellar herniation (white arrow)
Lesions had a mixed distribution of either homogeneous or heterogeneous appearance on T1W and T2W images. All lesions were hyperintense-to-normal grey matter on T2W series, and partially suppressed on FLAIR series. Post-gadolinium injection signal enhancement was present in all 21 cases and heterogeneous in all but 3/21 cases; 8/21 cats had evidence of rim enhancement. Changes consistent with mass effect suggestive of raised intracranial pressure were visible in all cases; 6/21 cases had extensive white matter oedema.
CT exams were available for 5/21 cases (supplementary material) and 4/5 had concurrent MRI (Figure 3). One case was not diagnostic for IE but was subsequently diagnosed with MRI. For the remaining 4/5 cats, lesion location included the forebrain (n = 1), cerebellum (n = 1) and brainstem (n = 2), and was further defined as extra-axial (n = 3) or mixed intra- and extra-axial (n = 1). All lesions were hypo-attenuating pre-contrast, with heterogeneous rim enhancement on post-contrast exam. Three of four cats had well-demarcated lesions with concurrent mass effect. Concurrent findings included otitis media (n = 2) and abscessated overlying soft tissues.
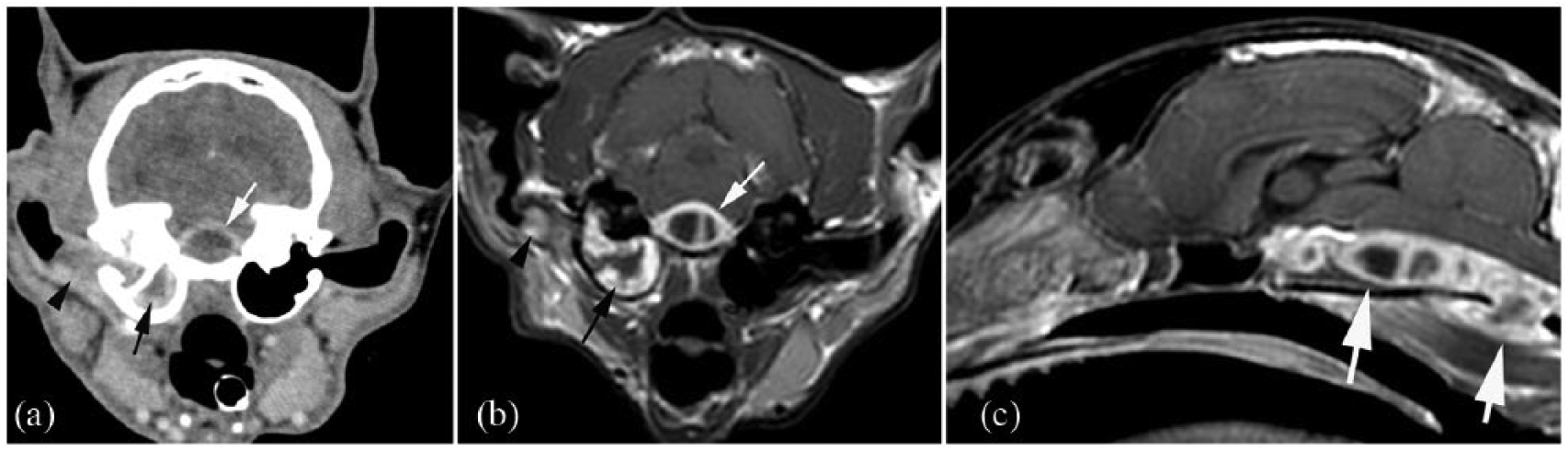
(a) Transverse plane post-intravenous contrast CT image of a 7-year-old male neutered domestic shorthair cat with an intracranial abscess. The lesion is located ventral to the brainstem and shows strong rim enhancement and absent contrast enhancement of the centre of the mass (white arrow). The right middle-ear cavity (black arrow) and the medial portion of the horizontal ear canal (black arrowhead) is filled with peripherally contrast-enhancing soft tissue-attenuating material, suggestive of otitis media and externa. (b) The corresponding T1-weighted (W) magnetic resonance post-gadolinium transverse plane image shows the same lesion location (white arrow) and right ear changes (black arrow and arrowhead in [b] as in [a]), with improved depiction of the soft tissues. (c) The sagittal plane T1W post-gadolinium view shows the extent and cavitated appearance of the lesion (white arrows) ventral to the brainstem
One cat was diagnosed via ultrasonography; flocculent material was observed passing between the subcutaneous space and the intracranial cavity via a defect in the overlying skull (Figure 4).

Ultrasound image of a 15-year-old female neutered domestic shorthair cat. Defect within the skull (white arrows), flocculent material communicating with the subcutaneous space and the intracranial cavity
Ancillary tests
Haematology results were available for 12/23 cats; only two had mild-to-moderate inflammatory neutrophilias with evidence of mild left shift and/or toxicity, six had leukograms most consistent with glucocorticoid response and four had values all within reference intervals. FIV and FeLV status was available for seven cats, all of which were negative for both diseases.
CSF analysis
Four of 23 cats had CSF analysis (three from the cerebellomedullary cistern and one from the lumbar cistern) performed, all of which demonstrated neutrophilic inflammation. Two of four cats had increased total nucleated cell count (1005/μl and 1310/μl, respectively) and total protein concentration (0.79 g/l and 2.21 g/l, respectively). Intracellular bacteria were identified on cytology in 2/4 cats (Figure 5). When cultured in enrichment medium, 1/4 cats had a growth of coagulase-negative Staphylococcus species. The second cat had an insufficient sample for culture; however, a polymicrobial infection with gram-positive organisms was identified from direct smears.

(a) Cytology image of cerebrospinal fluid from a 14-year-old male neutered domestic longhair cat with marked neutrophilic pleocytosis with bacterial sepsis. (b,c) Enlarged images of degenerate neutrophils containing filamentous bacteria (white arrows). Modified Wright’s stain
Culture results
CSF was submitted for bacterial culture in 3/4 cats. Bacterial culture yielded growth in 1/3 CSF samples and 6/9 samples taken at the time of craniectomy. Isolated bacteria encompassed both aerobic and anaerobic species, including Escherichia, Corynebacterium, Bacteroides, Streptococcus, Staphylococcus, Actinomyces, Nocardia and Enterococcus species. Polymicrobial growth occurred in two cases.
Treatment
Prior to referral, 13/23 cats had been treated with a range of both broad- and narrow-spectrum antibiotics for variable duration. Eight of which had also been given either NSAIDs, or anti-inflammatory or immunosuppressive doses of steroids (dexamethasone) but exhibited no improvement in their clinical signs.
Treatment groups consisted of solely medically treated (n = 10) or combined medically and surgically treated (n = 10) cats. Three cats were euthanased at the time of diagnosis but were included for descriptive purposes. Surgically managed cases and medically managed cases were hospitalised for a median duration of 6 and 5 days, respectively.
Antibiosis
All treated cases received broad-spectrum antibiosis; amoxicillin clavulanic acid (20 mg/kg q12h) was the most common agent used in 17/20 cats. Metronidazole (10–25 mg/kg q12h) was the most common second agent used and was administered to 10 cats. Antibiosis chosen was clinician-dependent or influenced by antibiosis given prior to referral, with a combination of up to four types of antibiotic used for a variable duration of 4–16 weeks.
Steroids
Twelve of 20 cats received dexamethasone ranging from 0.1–0.3 mg/kg intravenously (median dose 0.15 mg/kg) as either a single intraoperative dose or up to 3 days. 14
Antiepileptics
Seven of 23 cats received antiepileptic medication consisting of phenobarbital (2–3 mg/kg q12h) and/or levetiracetam (20 mg/kg q8h followed by 20mg/kg q12h dosing) postoperatively. Four of 23 cats were treated for seizure activity and three received prophylactic antiepileptic medication.
Outcome
Of the surgically managed group, most (n = 9/10) cats had a good short-term prognosis (survived 48 h postoperatively). One case failed to regain spontaneous ventilation after surgery and was euthanased. All 9/10 remaining cats were alive 6 months postoperatively and had a grade 1 neurological status. The median survival time for surgically managed cases was 730 days (range 1–3802 days). One cat was euthanased 7 years after diagnosis owing to the development of seizures and deterioration to status epilepticus; repeat imaging was not performed prior to euthanasia.
The medically managed group had a similar short-term prognosis, with 8/10 of cases surviving the first 48 h after diagnosis (Figure 6). Of the two cases that did not survive, one had cardiopulmonary arrest while receiving treatment and another was euthanased after 24 h of medical therapy due to financial constraints. Six month survival information was available for the eight remaining medically managed cats. Six of 8 cats had good long-term outcome, all of which were reported to be neurologically normal (grade 1). Medically managed cases had a median survival time of 183 days (range 1–1216 days).
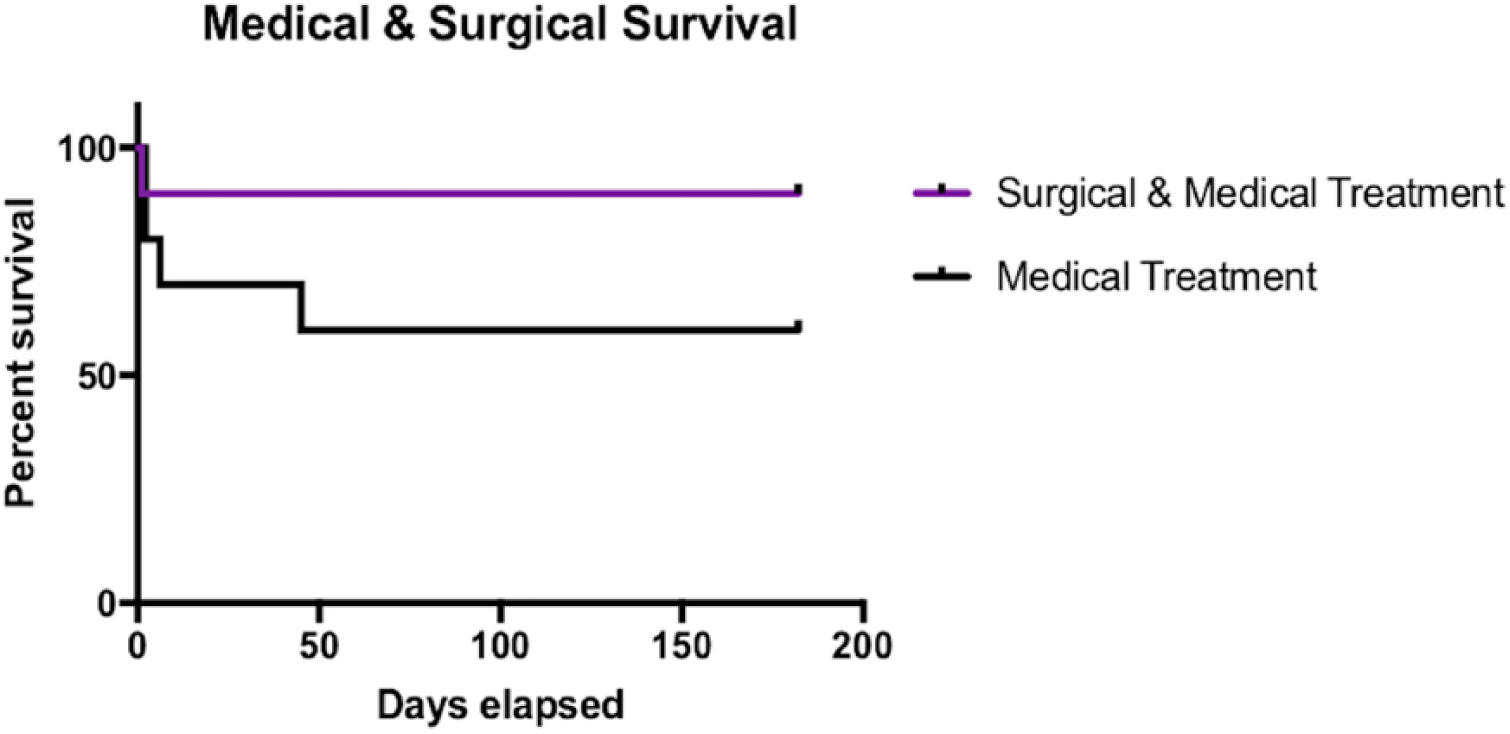
Kaplan–Meier curve depicting survival of surgically and medically managed cats (purple line) and solely medically treated cats (black line) over time
It is important to note that two cats, one of each treatment group, were still alive at the time of writing.
A two-tailed Fisher’s exact test demonstrated no statistical significance in short- or long-term survival (P = 0.237) between surgically and medically managed groups. The empyema location, use of steroids and the development of seizures were also not statistically significant when compared to outcome.
Discussion
To our knowledge, this study is the largest of its kind in cats. Six studies and four case reports within the literature describe IA and/or IE in 47 cats in total, with variable aetiologies, including cat bites, haematogenous spread, fungal infection and OMI. Of these, 11 cats were treated surgically and 35 medically with overall short-term mortality of 27% and 26%, respectively. The outcomes of each treatment group in this study demonstrate a lower mortality rate than that in the literature.3–11,15
Interestingly, in the human literature, the described treatment regimens for IA and IE typically involve a surgical approach. 16 Humans are treated via burr holes, a small craniectomy or craniotomy to facilitate drainage of pustular material and irrigation with antibiotics.17,18 Published reports evaluating solely medical management in humans do exist, with some reporting a favourable prognosis comparable with that of surgical intervention. 19 The over-representation of males in this study was also found to be a consistent finding in the human literature.16,17 Male cats typically presented with IA and/or IE secondary to cat bite abscesses overlying their calvarium; we hypothesised that this finding may be due to the increased likelihood of entire male cats to fight and roam larger areas.
The absence of prominent inflammatory leukogram changes (marked neutrophilia or neutropenia) on haematology, and the absence of pyrexia upon physical examination in the majority of cases is consistent with previous reports.4,6,7 This may reflect a lack of systemic response to intracranial infection; it is therefore important that IA and IE is still considered as a differential in normothermic cases without peripheral neutrophilia.
As with other case reports, CSF analysis was rarely undertaken, likely because of a greater risk of complications when performing this procedure in cases with increased intracranial pressure. 5 When performed, CSF results consistently demonstrated neutrophilic inflammation and were diagnostic (exhibited intracellular bacteria or had positive culture results) in two cases with empyema. Intracellular organisms are less frequently seen with intra-axial abscesses unless they have ruptured into the subdural space. 8 When available, CSF analysis provides valuable diagnostic information. It may be argued that these results are of more use in medically managed cases, as intraoperative findings and the ability to culture direct swabs taken at the time of craniectomy may make CSF findings redundant in surgical cases.
Reports of predisposition to both aerobic and anaerobic bacteria exist within the literature. 8 A mixed cohort of both aerobic and anaerobic isolates were identified in this study. This likely reflects the underlying aetiology of the IA and/or IE as both OMI and cat bite abscesses can yield polymicrobial growth.20,21 In this study only 33% of CSF samples and 67% of surgical swabs yielded positive culture results; this finding is consistent with the existing literature. 22 A likely explanation for the high incidence of negative culture results may be that many cases were exposed to antibiotics prior to referral. Additionally, species such as Actinomyces and Nocardia often require extended cultures and therefore may give false-negative results. 23
Potentiated amoxicillin and metronidazole were the two most commonly prescribed antibiotics while waiting for culture and sensitivity results. Combined, these broad-spectrum antimicrobials together provide activity against aerobic and anaerobic bacteria. When selecting antibiotics in these cases, consideration must be given to penetration of agents across the blood–brain barrier (BBB) to allow therapeutic concentrations to be reached within the CSF. Beta (β)-lactam antibiotics typically have poor penetration into the CSF; however, this penetration is enhanced in the face of meningeal inflammation and BBB dysfunction (an expected change secondary to both IA and IE). The use of higher intravenous doses of β-lactams has also been reported to increase their efficacy. Metronidazole readily penetrates the BBB; therefore, both metronidazole and β-lactams are suitable choices for intracranial infections, a likely explanation for their frequent use in this study. 24 Consideration should also be given to the duration of antibiotics prescribed, as our results showed a large variation from 4–16 week courses. A minimum course of 6–8 weeks is advised to treat IA within humans, alongside surgical drainage of the abscess.25,26
In this study, IA and/or IE was typically localised to the forebrain, likely owing to the overlying tissues being a common site of cat bite injury. Of the 11 cases that received treatment, eight were managed surgically. Perhaps, the comparative ease of a craniectomy to access the forebrain led to surgical treatment being the favoured choice.
Brainstem IE was infrequently documented and associated with infection spread from OMI or retrobulbar disease. Conversely, these cats were all managed medically, likely as a result of the challenging nature of a craniectomy at the skull base. Two of four cases died or were euthanased within 3 months. Our findings demonstrated a more favourable 50% mortality than those of Klopp et al 18 years previously, who found 100% mortality in two cats with brainstem abscessation. 10
Limitations of this retrospective study include a possible inherent bias between treatment groups as cases were not randomly allocated. Information regarding decision-making between different treatment modalities was not available for all cases. Those cases managed medically may have had other prognostic factors associated with their treatment choice, such as advanced disease, patient instability or financial limitations.
Analysing IA/IE secondary to multiple aetiologies may also make interpretation of results challenging, as analysis of one aetiology alone may have yielded different results. Furthermore, treatment regimens varied widely with inconsistent use of steroids, antiepileptic medication and type and duration of antibiotics. Follow-up examination was not always performed by a board-certified neurologist; therefore, return to a neurologically normal status was sometimes based upon referring veterinarian reports or owner communication. This means milder persisting neurological deficits (grade 2) may have been missed in some cats.
Our results found no significant difference between survival of those cases managed medically and those cases managed with combined surgical and medical therapy. These results are, however, based upon a relatively small sample size. A larger study population will increase the statistical power and may yield different results; therefore, future work in the form of a multi-centric retrospective study is required. A prospective, randomised control trial would provide the most reliable data; however, owing to the low prevalence of this disease it would be challenging to conduct.
Conclusions
IA/IE is an uncommon disease in cats and often presents a challenging diagnosis due to its non-specific and variable clinical signs. This study suggests that IA/IE can be successfully treated with medical management alone, and that the use of either solely medical, or combined medical and surgical treatment does not have a significant effect upon short- or long-term survival in feline patients.
Supplemental Material
Supplementary Material
CT data
Supplemental Material
Supplementary Material
MRI data
Footnotes
Author note
This paper was presented in part at the 2018 BSAVA conference.
Supplementary material
Supplementary data, including MRI and CT data.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
