Abstract
Clinical summary:
A tissue engineering approach was used to aid the surgical repair of a chronic oronasal fistula (ONF) in a 13-year-old cat. A three-dimensional (3D) printed mesh, tailored to the size and shape of the ONF, was created to support a soft tissue flap used to close the defect; and also to provide a matrix for mesenchymal stromal cells present in bone marrow aspirate and bioactive cytokines and growth factors present in platelet-rich fibrin harvested from the patient. A CT scan at day 75 after surgery revealed the formation of new tissue in the defect and the healing process was complete at follow-up 6 months after surgery.
Relevance and novel information:
Complications are frequently reported following surgical repair of ONFs and include dehiscence of the palatal suture line, flap necrosis due to damage to the greater palatine artery and maxillary osteomyelitis, mainly due to chronic infection and bone lysis. The case described here demonstrates how input from a multidisciplinary team and the use of a biomaterial, processed by sophisticated technologies, can create a precision regenerative medicine strategy adapted to the patient’s clinical needs; this provided a novel therapeutic solution for an otherwise hard to treat clinical problem.
Keywords
Introduction
Oronasal fistulas (ONFs) are abnormal epithelialised communications between the nasal and oral cavities. They can have a congenital or acquired aetiology.1–3 Acquired ONFs are generally related to oral pathologies (eg, secondary to severe periodontal disease, feline odontoclastic resorptive lesions or osteomyelitis) or trauma (road traffic accidents and accidental falls, generally associated with bone and/or teeth fractures); or are iatrogenic (resulting from surgical manipulation, such as mass excision and tooth extraction). 1
The abnormality leads to signs of upper respiratory tract pathology, such as ipsilateral mucopurulent or haemorrhagic nasal discharge, which is frequently associated with rhinitis, sneezing and discomfort, often manifested by pawing at the face; as well as regurgitation and/or vomiting due to the passage of liquid and food into the nasal cavity.3,4 The persistence of this condition leads to weight loss, malnutrition and the risk of aspiration pneumonia.5,6
Various techniques have been employed in human and veterinary medicine for the correction of ONFs. These include single- and double-layer mucoperiosteal flaps, buccal mucosal flaps, inverted periosteal flaps, free tissue grafts (auto- or allografts), prosthetic devices (usually removable), the application of synthetic (ceramic-based or composites) or natural bone grafts as regeneration-promoting materials (autologous or allogenic), and auricular cartilage grafts (from the conchal cartilage of the patient).4–13
However, complications can arise and there are many reports of unsuccessful surgical correction of large ONFs due to ischaemia and necrosis of the soft tissue flap. The approach to ONF closure should therefore also provide mechanical stability to avoid collapse of the soft tissue flap.1,2,4
A bone graft is one of the most commonly transplanted tissues in human medicine. Such grafts are used mostly in reconstructive orthopaedic surgery because of factors such as their volume and mechanical properties, and also their osteogenic and osteoinductive effects, dependent on the bone component utilised (cancellous bone, cortical bone or bone marrow [BM]).14,15 BM aspirates can be harvested using minimally invasive techniques, although this can be associated with some pain and donor site morbidity.10,15 BM has been employed in recent years as a regenerative strategy rich in mesenchymal stromal cells (MSCs), although few MSCs remain viable in BM aspirate transplantation.9,10,16
Platelet-rich fibrin (PRF) is a blood derivative that is used particularly in dentistry and oral and maxillofacial surgery. 17 PRF is obtained from whole blood and consists of leukocyte-platelet content, distributed in a fibrin matrix.18,19 It is a second-generation platelet-derived product proposed by Choukroun and colleagues 20 as an easier to prepare and completely autologous alternative to platelet-rich plasma.14,21 Platelets are rich in platelet-specific and non-platelet-specific proteins, such as growth factors, among other cytokines; the fibrin matrix acts as a net to contain these regulatory molecules, which help modulate wound healing and repair, promoting angiogenesis, mitogenesis and osteogenesis.17,18,22 PRF has also been suggested as a possible matrix for cell delivery in bone augmentation strategies, improving the volume of recently formed bone and also acting as an osteoblast carrier.18,23
However, BM and PRF do not have sufficient mechanical properties to support the soft tissue flaps produced for surgical repairs or to resist the forces created by mastication and movement of the tongue. This report describes the use of a polycaprolactone (PCL) scaffold as a reinforcing material in the repair of an ONF in a Persian cat. PCL is a biocompatible synthetic polymer, characterised by its slow degradation rate, which can support cell proliferation in the process of soft tissue healing. 24 PCL can be easily processed into different structures/shapes by high-resolution three-dimensional (3D) printing technology, and tailored to the specifications of a defect.25,26 PCL is used in different biomedical applications, such as suture material, and has also been considered for orthopaedic applications (bone repair). In tissue engineering settings, PCL scaffolds have been shown to provide structural support to guide tissue regeneration, aided by the biocompatibility of PCL and its ability to be manipulated during the surgical procedure.27,28
Case description
Clinical history
A 13-year-old, male neutered Persian cat, weighing 4.4 kg, with a 4/9 body condition score (WSAVA Global Nutrition Committee) was referred to the Vetlamacçãs Small Animal Clinic, Braga, Portugal, for clinical evaluation of a chronic oronasal defect. According to the owners the condition had arisen following the natural loss of a tooth 2 years previously. Since then, the cat had displayed frequent episodes of mucopurulent nasal discharge, predominantly from the right side, regurgitation, halitosis, sneezing, loss of appetite and lethargy. The animal had undergone two earlier surgical interventions in the previous 5 months, at another veterinary clinic, without success. The owners reported that the oronasal deformity had increased in size, and was also medially displaced, following these interventions. The cat had been monitored prior to these surgical interventions due to creatinine values being in the upper normal range in recent months (IRIS stage 2 chronic kidney disease).
The ONF measured approximately 7 mm in length (caudal to rostral) by 6 mm in width, and was located at the level of the first to third premolar teeth (Figure 1). The dentition of the animal was incomplete (with only incisors and third left maxillary premolar teeth present). There was no clinical evidence of gingival inflammation or periodontitis. The cat was negative for feline leukaemia virus and feline immunodeficiency virus. Considering the age of the cat and the serial anaesthetic/sedation procedures performed previously, this third intervention was thoroughly planned. A tailor-made implant, in combination with autologous PRF and BM aspirate, was used to support the soft tissue flap created to close the defect.

(a) Oronasal defect on the day of the cat’s surgery (day 0), with fibrotic tissue deposition surrounding the fistula in the mucosal epithelium. (b) Close-up view of the oronasal fistula (ONF)
The surgery was performed according to national guidelines, after approval by the Ethics Committee of the University of Trás-os-Montes and Alto Douro (UTAD), Vila Real, Portugal, and with the informed consent of the owners, in compliance with good clinical veterinary practice.
3D printed implant creation
The cat was mildly sedated with intravenous butorphanol (0.2 mg/kg) and propofol (2 mg/kg), with lactated Ringer’s solution administered intravenously, to allow a silicone putty impression mould of its maxillary dentition to be taken. A plaster mould was subsequently made (Figure 2).

Silicone putty mould (green colour) of the maxillary dentition of the cat, acquired under mild sedation. The respective plaster mould (greyish-yellow colour) allowed the production of a polycaprolactone (PCL) implant adapted to the oronasal defect
The PCL scaffold was produced by rapid prototyping. Briefly, PCL (average Mn 80,000; Sigma-Aldrich), containing <0.5% impurities (water), was loaded in a syringe and heated to 91°C to melt the polymer. A stainless steel deposition tip (21 G) was used to cap the syringe and a 15 x 15 mm square-shaped mesh was printed using a bioplotter (B10; Envision TEC). The construct was composed of three consecutive layers, designed to have 0.75 mm spacing between strands, each one oriented perpendicularly to the previous one. The implant was adapted to the dimensions of the defect using the maxillary impression mould from the cat.
The tailored mesh was sterilised using ethylene oxide (EtO) at 42°C, with 3 h of exposure and 10 h of aeration inside the equipment. An additional environmental aeration and removal of residual EtO concentrations was performed, according to the recommendations of the World Health Organization. The sterilisation process was confirmed by 1294 Rapid Readout Biological Indicator (3M Attest), according to the manufacturer’s instructions.
Implant characterisation
A scanning electron microscope (SEM) (JSM-6010 LV; JEOL) was used to obtain micrographs of the 3D printed PCL mesh microstructure. The microarchitecture and geometry of the mesh, namely its framework thickness, mean pore size and total porosity (Table 1), as well as a 3D reconstruction, were assessed by microcomputed tomography (Skyscan 1072; Skyscan). A series of about 400 two-dimensional projections, with a resolution of 6.13 [im, was acquired over a rotation range of 180°, with a rotation step of 0.45°, by cone-beam acquisition after irradiating the specimen with penetrative x-rays (source voltage 75.6 kV, current 131.9 [iA). The data were reconstructed using NRecon software (version 1.6.6.0; Bruker microCT), and analysed on a CT analyser (version 1.4; Skyscan). The region of interest (ROI) was defined as a circle comprising a representative section of the sample. Auto-interpolation of the manually defined ROI yielded a volume of interest with a shape of a disc, which was the essential basis for the quantitative analyses. The 3D models were generated using CTVox software (version 2.3.0 r810; SkyScan).
Principal microarchitecture features of the PCL scaffold

SEM analysis revealed that the 3D printed construct had an average thickness of 320 and that the different layers were fused (Figure 3a). Microcomputed tomography analysis showed that the scaffold had a well-defined interconnected structure composed of three layers of aligned PCL fibres (Figure 3b), with each consecutive layer rotated 90° relative to the previous layer.

Scaffold microstructure. (a) Micrograph of the PCL implant surface obtained by scanning electron microscopy at low magnification (x25); (b) 3D reconstruction of the PCL scaffold obtained by microcomputed tomography
Surgical intervention
The cat was premedicated with methadone (0.3 mg/kg IM) and acepromazine (0.01 mg/kg IV). To induce anaesthesia, propofol (2-4 mg/kg IV) was administered to effect, and general anaesthesia was maintained with isoflurane at 0.5-1.5% in oxygen. Lactated Ringer’s solution was administered during the surgery at a rate of 4 ml/kg/h.
The jugular region and the region of the greater tubercle of the right humerus were prepared under strict aseptic technique for whole blood collection and BM harvesting, respectively. After clipping, the skin of each area was scrubbed with 2% chlorhexidine gluconate solution and 70% ethanol. The oral mucosa was cleansed with a 0.12% chlor -hexidine gluconate solution.
After collecting 6 ml of blood into a sterile tube without anticoagulant, PRF was prepared according to Choukroun et al. 20 Briefly, the whole blood was centrifuged at 3000 rpm for 10 mins before the PRF clot was collected, observing strict aseptic technique, from the upper phase and the red blood fraction was discharged (Figure 4a,b). 18 For BM aspirate harvesting, a skin incision was made over the greater tubercle of the right humerus, before drilling the cortical bone to access the BM. The aspirate was harvested using an 18 G needle and a 10 ml syringe, coated with sodium heparin (5000 IU/ml) (Figure 4c). Some of the BM was injected into the PRF clot (Figure 4d), with some retained in the syringe for use later in the surgical procedure. Additionally, some BM was preserved in a sterilised closed Eppendorf tube for later harvesting of bone marrow MSCs.

Preparation of the platelet-rich fibrin (PRF) clot–bone marrow (BM) aspirate blend, under aseptic technique. (a) The PRF was produced by centrifugation of whole blood, without anticoagulant, at 3000 rpm for 10 mins; the clot produced has a jelly-like appearance, as can be seen in (b). (c) For the harvesting of BM aspirate, a skin incision over the greater tubercle of the right humerus was made, followed by drilling of cortical bone to access the BM. (d) Some of the BM aspirate was injected into the PRF clot

A single mucogingival pedicle flap was created in order to eliminate tension forces along the future suture line (Figure 5a). The fibrotic area surrounding the fistula was debrided to remove epithelialised tissue. Non-elastic palatal tissue was elevated, using a periosteal elevator, in order to avoid tension and retraction of suture edges (Figure 5b). The nasal cavity was irrigated with sterile 0.9% sodium chloride solution to eliminate foreign material, such as fur resulting from grooming. The PCL scaffold was implanted between the flap and subperiosteal space of the palatal bone (Figure 5c) and fixed with simple interrupted absorbable 4-0 sutures (Monosyn; B Braun), without the need for screws or plates (Figure 5d). The blended PRF clot-BM matrix was placed on to the mesh of the implant (Figure 5e); the remaining BM in the syringe was injected into the tissues and area around the PRF clot to fill the empty space between the mucogingival flap and PCL mesh before closure of the surgical site (Figure 5f,g). The flap was drawn over the implanted material and a simple interrupted 3-0 absorbable suture pattern was used to close the site (Monosyn; B Braun) (Figure 5h). The tongue spicules of the cat were slightly bluntened using a blade to minimise mechanical trauma over the surgical wound. An oesophageal feeding tube was placed to provide adequate nutritional support during the cat’s convalescence.
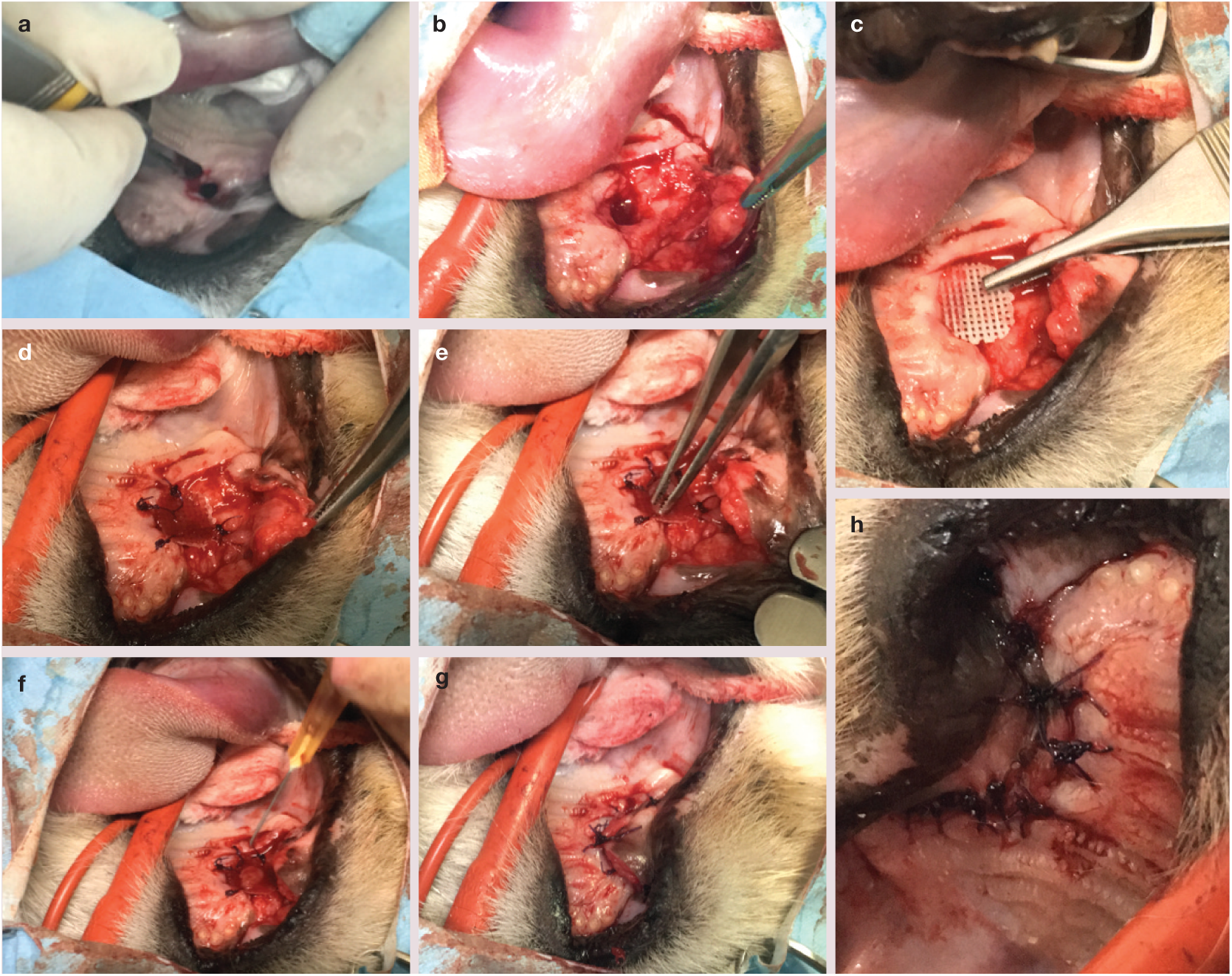
Surgical correction of the ONF. (a) With the patient in dorsal recumbency, thefibrotic tissue resulting from previous surgeries was removed and a single mucogingival pedicle flap was created. (b) Palatal tissue was elevated, in order to avoid tension and retraction of suture edges. (c) The 3D PCL implant was positioned and fixed (d) between the oral mucosa and subperiosteal space of the palatal bone. (e) The blended PRF clot–BM matrix was placed ventral to the implant. (f) BM aspirate was injected into the area, engulfing the PRF clot and reducing the empty space, while the flap covered the implanted material (g). The surgical site was closed using simple interrupted absorbable sutures (h)

The remaining, preserved BM was transported to a laboratory at ambient environmental temperature to assess for the presence of MSCs. It was placed in Dulbecco’s Modified Eagle’s low glucose culture medium; after an incubation period of 48 h, culture medium was replaced to remove non-adherent cells. Cells were cultured under standard conditions (at 37°C, 5% CO2 and 95% humidity) (Figure 6).

Feline bone marrow mesenchymal stromal cells (MSCs) in culture, with adherent and plastic cellular colony forming units, at day 7 after BM harvesting. The cells are in passage 0 (P0) and have the spindle-shaped appearance that is typical of MSCs
The cat was prescribed amoxicillin and clavulanic acid (20 mg/kg q12h SC) for 15 days after surgery. The use of antibiotics postoperatively, and the duration of their use, was felt to be justified by the failure of the two previous surgical attempts to repair the defect, which had been attributed to osteomyelitis, as well as by the presence of purulent nasal discharge at the time of surgery. Also, the cat had required multiple courses of antibiotics after its previous surgeries. The analgesic protocol included meloxicam (0.2 mg/kg for the first day and 0.05 mg/kg for the next 3 days q24h SC) and methadone (0.4 mg/kg q12h SC in the first 24 h after surgery), followed by buprenorphine (0.02 mg/kg q12h SC for 4 days, then tapered over 4 days before withdrawal).
A liquid diet (Hill’s a/d recovery Canine & Feline Liquid with water) was given until 6 days after surgery, when the feeding tube was removed under mild sedation and the cat started to eat (Hill’s Feline k/d canned food) and drink voluntarily. After 16 days, the cat was discharged from the clinic.
Clinical results and follow-up
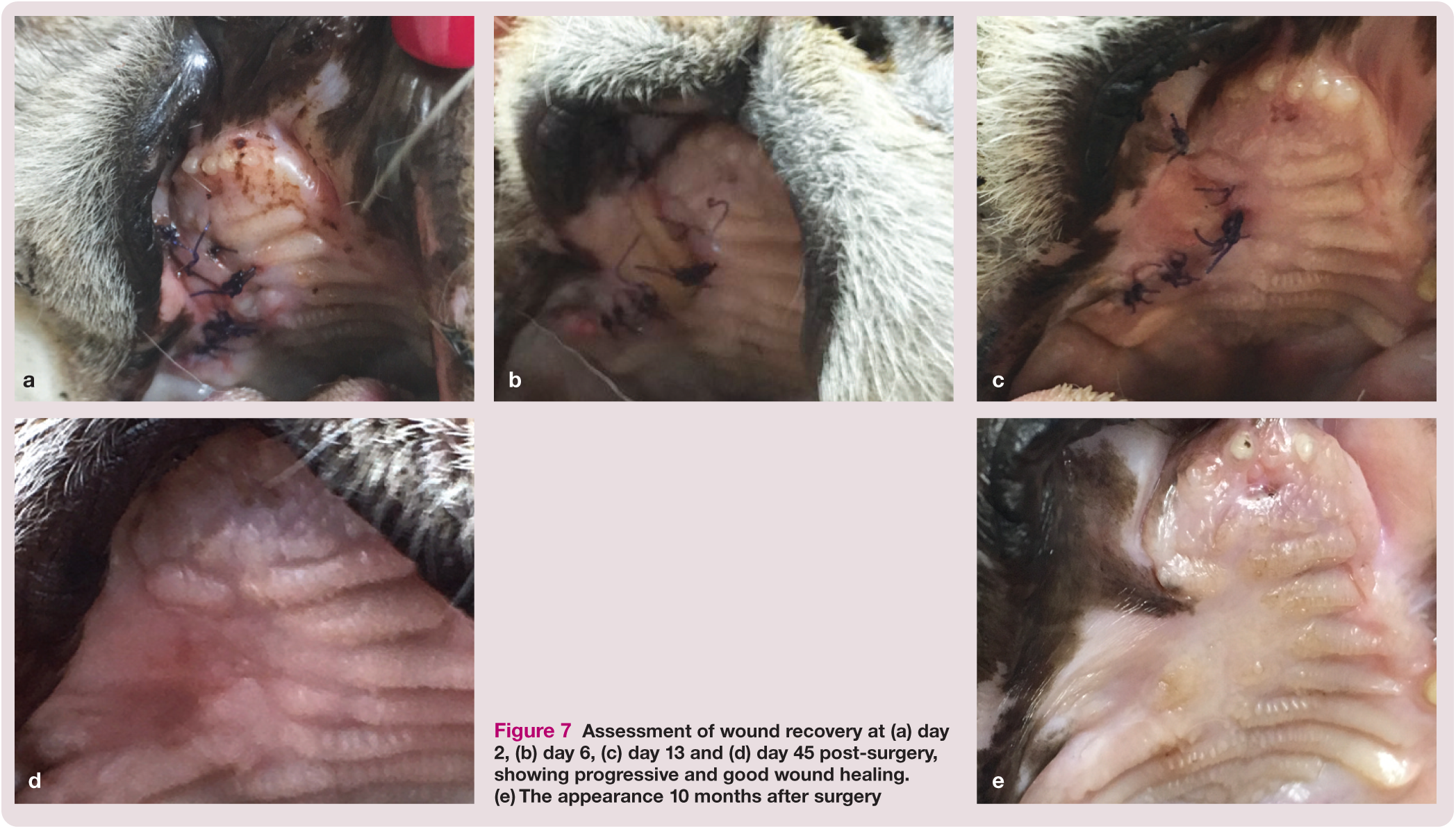
Six days after surgery, an oral examination revealed loosening of some suture knots. The right superior labial deviation seen after the surgery was no longer present. A complete blood count, serum biochemistry, electrolytes and urinalysis were performed 3 weeks after surgery, demonstrating that the cat remained IRIS stage 2 (creatinine 1.88 mg/dl) and was otherwise in good health. None of the suture material was removed and, at day 35, the suture knots were no longer visible. The wound margins were viable, without oedema, bleeding, infection or ischaemic signs. The cat was doing well and the owners described the reappearance of normal behaviour, such as interest in sniffing objects and food, and weight recovery. Postoperative wound healing was assessed and documented over 2 months and a last medical observation was performed at 10 months (Figure 7).

Assessment of wound recovery at (a) day 2, (b) day 6, (c) day 13 and (d) day 45 post-surgery, showing progressive and good wound healing. (e) The appearance 10 months after surgery
At day 75 after surgery, the patient underwent a CT scan (Brivo CT 325; General Electric) at the Veterinary Teaching Hospital of the UTAD. The scan showed homogeneous and regular layers of tissue covering both the nasal and oral aspects of the defect, with a similar appearance to non-compromised peripheral soft tissues. The implant was not observed and there was some limited evidence of bone healing (Figure 8). At follow-up appointments, 6 and 10 months after the surgery, the owners described the cat as showing a remarkable improvement in its quality of life.
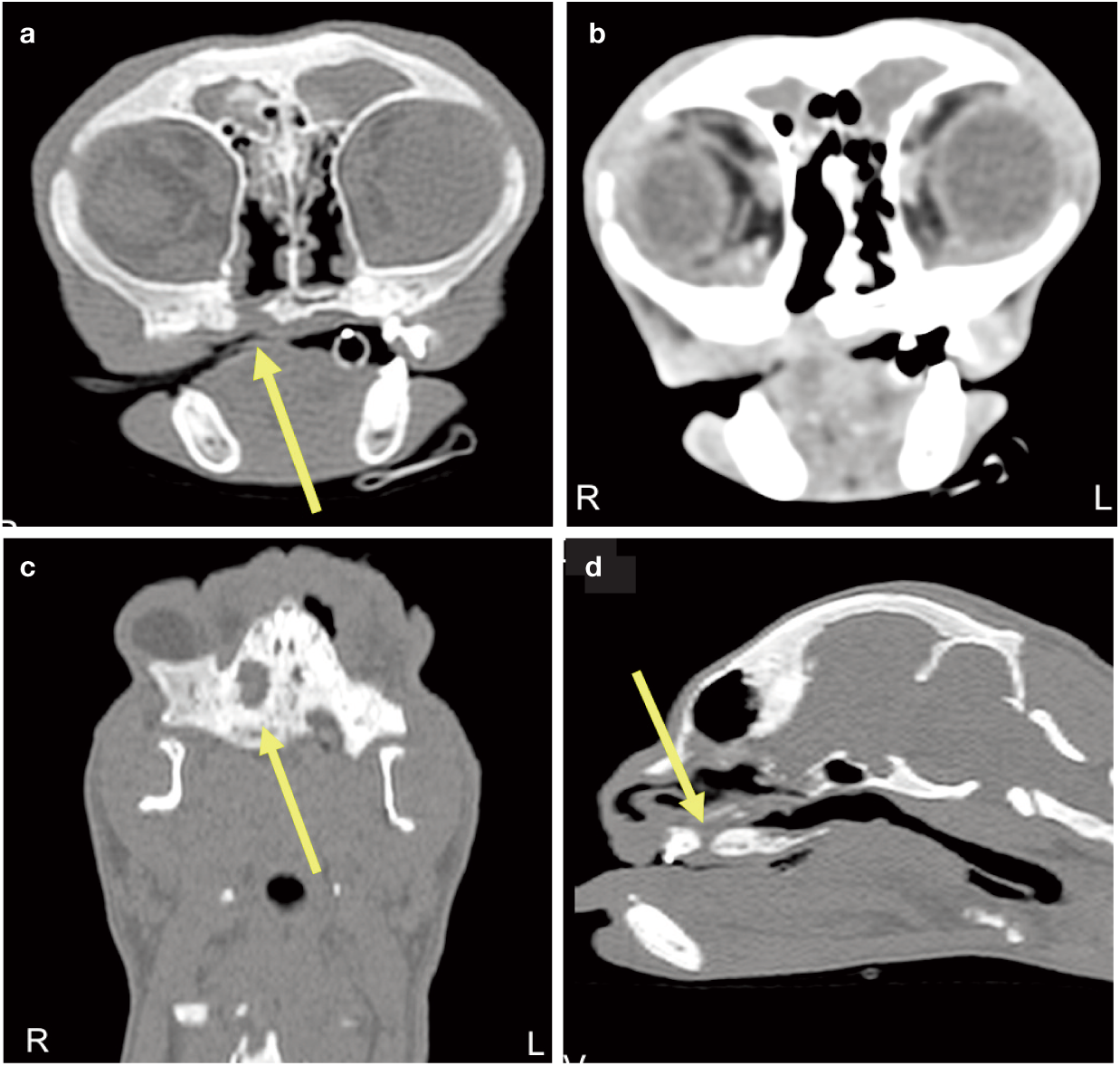
Transverse CT scan, performed 75 days after the surgical intervention (yellow arrow indicates the defect). (a) Non-contrast axial image and (b) contrast scan image illustrating the defect site, densely filled in both its nasal and oral aspects by homogeneous soft tissue; the PCL scaffold is not visible. (c) Non-contrast parasagittal scan and (d) non-contrast coronal section, showing the site and dimensions of the defect. Window width = 2000, window level = 200
Discussion
Some of the major complications associated with surgical correction of palatal defects include dehiscence of the palatal suture line, flap necrosis due to damage to the greater palatine artery and mandibular osteomyelitis. All of these sequelae had been reported following the previous unsuccessful surgeries performed on this cat and it had been referred as a result of this chronic situation. According to the cat’s clinical records, progressive compartments in the alveolar bone due to tooth loss had led to severe osteomyelitis, bone loss and, consequently, flap dehiscence in the earlier interventions.
BM aspirate has recently been used for alveolar cleft grafting in human patients, with clinical studies suggesting that PRF may enhance the regeneration capacity of stem cells when added to BM aspirate. 29 PRF may also support proliferation and growth of human-cultured alveolar bone-derived periosteal cells and stimulate local angio-genesis in the implantation site; 30 thus the combined use of bone-derived cells and PRF could be clinically applicable in bone regeneration therapy.
In the present case, the authors did not believe that a mesh and tension-free flap approach alone would be successful in resolving the ONF. The feline patient was geriatric and, although the size of the ONF was not considered critical, two previous surgical repairs had failed. The implantation of a PCL scaffold gave support to the tension-free soft tissue buccal flap created to close the defect. The scaffold also supported a bioactive matrix, rich in cytokines and growth factors, provided by PRF, which, in turn, retained proliferating MSCs present in the BM aspirate. This natural in situ matrix occupied the space around the mesh, integrating the scaffold and the surrounding tissues during the healing process, and contributing to the natural formation of a homogeneous and consistent layer of soft tissue covering the nasal aspect of the ONF, where there was no surgical flap.
Feeding assistance is strongly recommended in the healing period to simultaneously ensure adequate nutritional support of the animal and minimise mechanical trauma provoked by chewing and tongue movements.
PCL is characterised by its slow degradation rate (up to 3 years) and it is radiodense on CT scans;31,32 although, in the present case, the mesh was not visible 75 days after the surgery. In human patients, bone formation has been noted at 1.5 years after PCL implantation; but, in the present case, little bone formation was visible on the CT scan, probably because there had not been enough time for significant osteogenesis to occur. 33 However, given the animal’s life stage, IRIS classification and clinical history, involving a considerable number of recent anaesthetic procedures, the clinical team decided not to perform further CT scans.
Conclusions
The successful treatment of this case was achieved by a multidisciplinary team that brought together the experience and professional skills of veterinary clinicians, dentists and biologists, and allowed the translational application of novel biotechnology approaches tailored to the patient’s specific needs.
Three- and four-dimensional printing methodologies have been recently advanced as precision regenerative medicine and tissue engineering strategies, especially in a clinical context when surgical interventions are insufficient on their own. Autologous blood derivatives such as PRF, combined with BM, form a matrix naturally rich in bioactive cells and molecules that may promote wound healing, and could be used to support the surgical technique. Technical advances using PCL offer the potential to easily produce biodegradable and bioresorbable implants that are degraded by continuous hydrolysis. These give structural support for surgical reconstruction of large defects, and can be customised to the patient, because of their flexibility, avoiding the considerable disadvantages and complications associated with other materials, such as auto/allografts, ceramics and titanium.
Despite limited evidence of bone regeneration in the present case, a successful healing process and re-establishment of health and quality of life of the animal were achieved.
Finally, further clinical reports describing translational approaches from the laboratory to the veterinary clinic should be encouraged, alongside good manufacturing practices, to promote novel therapeutic solutions inspired by advances achieved in human medicine.
Footnotes
Acknowledgements
The authors are grateful to Dr Patricia Santos and Dr Ana Lucinda for the clinical care of the cat following its discharge from Vetlamacçãs Clinic. They would also like to thank Ana Goncalves and Paula Sol for their technical support and Professor Mario Ginja for assistance with the CT scan. The cat’s owners are gratefully acknowledged for their invaluable support.
Date accepted: 19 February 2018
Ethical approval and consent to participate
The cat’s owners provided written, informed consent for the clinical and diagnostic assessment and treatment, including clinical follow-ups. This study has been approved by the Ethics Committee of the University of Trás-os-Montes and Alto Douro (number 17/2017). Consent was obtained from the owners of the cat, authorising the publication of this case report with accompanying images.
Authors’ note
CSS and LCB took part in the clinical evaluation of the patient, provided medical assistance/monitoring and performed the surgical intervention. VS was responsible for the production of the maxillary moulding. CSS was responsible for the clinical literature review. MGF was responsible for programming the bioplotter, for the production of the implant and for the scanning electron microscopy analysis. PSB was responsible for the implant images acquisition and microcomputed tomography analysis. IRD was responsible for the clinical CT examination of the cat. RLR, PPC and MEG were responsible for the overall supervision of the study and critical review of the manuscript.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
FCT SFRH/BD/118871/2016 PhD grant, Norte-08-5369-FSE-000044 and FCT grant IF/00593/2015. This article is a result of the project ‘Accelerating tissue engineering and personalized medicine discoveries by the integration of key enabling nanotechnologies, marine-derived biomaterials and stem cells’, supported by Norte Portugal Regional Operational Programme (Norte 2020), under the Portugal 2020 Partnership Agreement, through the European Regional Development Fund. The authors acknowledge the financial support from the European Union Framework Programme for Research and Innovation Horizon 2020, under the TEAMING Grant agreement 739572 - The Discoveries CTR.
