Abstract
Background:
Terminal dyspnea is a distressing symptom in advanced cancer patients. Although opioids are standard treatment, their effectiveness may differ between opioid-naïve and -tolerant patients.
Objectives:
To evaluate responses to opioid titration for terminal dyspnea and assess differences in palliative care physicians’ perceptions of opioid effectiveness between opioid-naïve and -tolerant patients.
Design:
A pre-planned secondary analysis of a multicenter prospective cohort study.
Setting/Subjects:
Advanced cancer patients (n = 108) with moderate to severe terminal dyspnea and an Eastern Cooperative Oncology Group performance status of 3–4, receiving continuous parenteral opioid infusions, were included and categorized as opioid-naïve (n = 48) or opioid-tolerant (n = 60).
Measurements:
Opioid doses were titrated in four predefined levels within 48 hours, with effectiveness assessed 6 hours post-titration using the Clinician Global Impression-Improvement scale. Opioid ineffectiveness was defined as the need for continuous sedatives or death without dyspnea relief, with doses recorded as the parenteral morphine-equivalent daily dose.
Results:
The mean age was 72 years, and lung cancer was present in 43 patients (40%). Opioid-naïve patients showed a 100% response after 12 titrations, while opioid-tolerant patients exhibited a 68% response after 34 titrations. At death or one month after treatment, opioids were determined to be ineffective in 8.3% of opioid-naïve and 38% of opioid-tolerant patients (p < 0.001). Median doses at ineffectiveness were 15 mg (IQR = 7.5–23) and 36 mg (IQR = 14–200), respectively.
Conclusions:
Opioid-tolerant patients showed a lower-level response and were more likely to experience ineffectiveness than opioid-naïve patients. Larger studies are needed to confirm these findings and improve terminal dyspnea management.
Key Message
This secondary analysis of a multicenter cohort study highlights differences in opioid effectiveness for terminal dyspnea between opioid-naïve and -tolerant advanced cancer patients. Opioid-tolerant patients demonstrated lower response rates and higher rates of treatment ineffectiveness, emphasizing the need for tailored management strategies to address dyspnea in this population.
Introduction
Dyspnea is one of the most frequent and distressing symptoms experienced by cancer patients in the last weeks to days of life (terminal dyspnea).1–3 As death approaches, dyspnea typically worsens, becomes less responsive to palliative treatments, and often necessitates proportional sedation as the last resort.1,2,4–6 International guidelines recommend opioids as first-line pharmacological treatment for terminal dyspnea, frequently administered as continuous infusions to facilitate timely titration.7–9 However, real-world evidence from specialist palliative care settings indicates that continuous opioid infusion provides dyspnea relief in only about half of patients, leaving a significant proportion of individuals suffering from persistent dyspnea in their final days.10–12
The limited effectiveness of opioids for patients with terminal dyspnea contrasts with the relative success in managing chronic dyspnea, where low-dose oral morphine (10–30-mg-morphine-equivalent daily dose [MEDD]) is effective for most patients. 13 One possible explanation for this discrepancy is the inherent worsening of dyspnea as death approaches.1,2 Opportunities for opioid titration may be limited and should be weighed against risks and benefits, given the limited time remaining, decreased cognitive capacity, and worsening delirium as part of the natural dying process.2,14 Another plausible interpretation is that most advanced cancer patients with terminal dyspnea are already receiving opioids for cancer pain.11,15,16 In these cases, dyspnea that breaks through baseline opioid therapy may offer little scope for improvement with additional titration, particularly given the limited time for such adjustments to take effect. A nationwide survey of palliative care physicians in Japan revealed that approximately one-third believed that there were upper limits to the effectiveness of parenteral morphine for dyspnea relief. 17 These perceived upper limits were reported as median doses of 30 mg/day for opioid-naïve patients and a 50% increase from the baseline for opioid-tolerant patients.
As discussed, opioid-tolerant patients are already on opioids and may not derive sufficient additional benefit from further titration. The effectiveness and safety of opioids for terminal dyspnea may vary between opioid-naïve and -tolerant patients, but no empirical data have addressed the differences in responses to opioid titration between these groups. Furthermore, it remains unclear how far opioids are titrated for terminal dyspnea and whether the perceived upper limits of opioid effectiveness reported by physicians are observed in clinical practice for both opioid-naïve and -tolerant patients. Understanding these differences is critical for optimizing symptom management and determining when alternative treatments should be considered.
The aims of this study were to investigate responses to opioid titration for patients with terminal dyspnea and to explore how far opioids are titrated, as well as whether palliative care physicians perceive upper limits in the effectiveness of opioids for terminal dyspnea in opioid-naïve and -tolerant patients with advanced cancer.
Methods
Design
This was a pre-planned secondary analysis of a multicenter prospective cohort study. The study’s methodology has been reported in detail. 16 In brief, consecutive advanced cancer patients seen by palliative care physicians in inpatient consultation teams or inpatient hospices/palliative care units at five sites in Japan between February 2020 and June 2021 were enrolled. The study protocol was approved by institutional review boards (IRBs) at the principal site (Seirei Mikatahara General Hospital, No. 19-57) and all the other study sites. All assessments and interventions were conducted within routine practice. The study adhered to the ethical standards of the Helsinki Declaration and ethical guidelines for medical and health research involving human subjects presented by the Ministry of Health, Labour, and Welfare in Japan. 18 The requirement for written informed consent in this observational study was waived by IRBs in accordance with ethical guidelines. Instead, an opt-out method was used; all patients could receive information about the study through the instructions posted on the ward or institutional website, and they had the opportunity to decline participation.
Participants
Inclusion criteria included: age ≥18 years old; advanced cancer; Eastern Cooperative Oncology Group (ECOG) performance status (PS) ≥3; moderate-overwhelming dyspnea (Integrated Palliative care Outcome Scale score of 2–4); 19 maintained communication capacity (0 [voluntary and explicit communication of complex contents] − 2 [involuntary or inexplicit but meaningful communication] based on the Communication Capacity Scale [CCS], item 4); 20 and initiation or new dose escalation of parenteral opioids by continuous administration for dyspnea, depending on whether patients were already receiving opioid infusion for pain. Patients were excluded if they received treatment for dyspnea caused by etiologies unrelated to cancer (e.g., bronchodilators or corticosteroids for asthma attack or acute exacerbation of chronic obstructive pulmonary disease, and corticosteroids for drug-induced lung diseases), or if they were scheduled to undergo an intervention that would markedly influence dyspnea intensity (e.g., thoracentesis for pleural effusion). The patients were followed for 1 month or until death, whichever came first.
Treatment strategies
Morphine, oxycodone, and hydromorphone, the most commonly used opioids for dyspnea in Japan, were included.21,22 To reflect routine clinical practice across study sites, a pharmacological treatment algorithm was developed that allows visualization of how palliative care specialists typically use parenteral opioids by continuous administration, based on prior literature.7,15–17,23 The recommended initial dose of parenteral opioids was 6–12 mg/day (parenteral MEDD) for opioid-naïve patients and a 20%–50% increase in prior opioid doses for opioid-tolerant patients. 15 Thereafter, doses were maintained, increased by 20%, or decreased by 20%, as deemed clinically appropriate. 15 Opioid-tolerant patients were broadly defined as those who had been receiving around-the-clock opioids.10,11 Physicians were allowed to adjust doses based on organ function as well as patient and family preferences, and provide concurrent palliative treatment for dyspnea related to cancer such as supplemental oxygen and corticosteroids.
Measurements
Data collection has been described in detail elsewhere. 16 All data were collected by responsible physicians at the baseline, 6 hours after each opioid titration during the first 48 hours, and at one month or on death after treatment initiation. In this pre-planned secondary analysis, we report measurements at 6 hours after each opioid titration and at one month or on death, whichever came first. As many patients with terminal dyspnea were expected to become unable to communicate in the last days of life, we decided to utilize clinician-rated outcomes instead of patient-reported outcomes.2,11 The observation period was set to one month because, while the median survival period of cancer patients with terminal dyspnea requiring continuous opioid infusion is often less than one week, we aimed to sufficiently follow those who survived for longer. 11 Baseline data included patient demographics, etiologies of dyspnea, comorbidities, renal and hepatic functions, and prior opioid use.
Responses to opioid titration over 48 hours after treatment initiation were assessed 6 hours after each titration using the Clinician Global Impression-Improvement (CGI-I), a 7-point Likert scale (1 = very much improved, 2 = much improved, 3 = minimally improved, 4 = no change, 5 = minimally worse, 6 = worse, 7 = very much worse), as rated by palliative care physicians based on dyspnea improvement. 24 We also recorded the presence or absence of dose-limiting toxicities (DLTs), regarded as severe toxicities that restricted further opioid dose escalation, and evaluated six hours after each escalation. Titration levels were defined for both opioid-naïve and -tolerant patients based on clinical guidelines and prior literature.7,9,11,15,16,25 For opioid-naïve patients, the titration levels were defined as incremental increases in parenteral MEDD: Level 1 (6–12 → 13–18 mg/day), Level 2 (13–18 → 19–24 mg/day), Level 3 (19–24 → 25–36 mg/day), and Level 4 (25–36 → 37–48 mg/day). For opioid-tolerant patients, titration levels were defined as percentage increases in MEDD: Level 1 (0–25 → 26%–50%), Level 2 (26–50 → 51%–100%), Level 3 (51–100 → 101%–200%), and Level 4 (101–200 → 201%–300%). The evaluation time was set at six hours based on our prior study on the effectiveness and safety of continuous morphine infusion for terminal dyspnea, 11 which confirmed observable responses within this timeframe. A longer duration was avoided to minimize the influence of deterioration of the general condition or other concurrent treatments.
At one month or on death after treatment initiation (whichever came first), opioid doses and whether continuous opioid infusion was determined to be ineffective by physicians based on predefined criteria were recorded. “Ineffectiveness” was defined by any of the following: 1 the addition of continuous benzodiazepine infusion for dyspnea, 2 initiation of continuous sedation for dyspnea relief (defined as administration of ≥10 mg/day of midazolam, ≥25 mg/day of levomepromazine, or ≥20 mg/day of haloperidol), or 3 death without achieving dyspnea treatment goals. Achievement of dyspnea treatment goals was operationally defined as “the case where dyspnea is alleviated or is acceptable to the patient, and the patient does not wish for further adjustment of treatment for dyspnea” based on clinical experiences. 16 If the patient had difficulty communicating, and his or her family was present, the responsible physician asked the family if the patient looked comfortable and would not wish for further adjustment based on observation. If the patient had difficulty communicating and the family was not present, the responsible physician determined whether the treatment goals shared by the patient/family at the initiation of treatment had been achieved.
Statistical analyses
The primary outcome of this secondary analysis was the response to each opioid titration level at 6 hours, categorized as “improved” (CGI-I scores 1–3), “unchanged” (score 4), or “worse” (scores 5–7). Secondary outcomes included the presence of DLTs after each titration within 48 hours, opioid doses at the end of observation, and physician-perceived upper limits of opioid effectiveness. Descriptive statistics were used to summarize baseline data and outcomes. Chi-square tests compared the presence of perceived upper limits of opioids between groups. This secondary analysis was exploratory, as sample size calculation was based on the feasibility of the treatment algorithm as reported in the main paper rather than this analysis. A p value <0.05 (two-sided) was considered significant. Analyses were performed using The Statistical Package for the Social Sciences, version 25.0 (SPSS Inc., IBM, JAPAN).
Results
A total of 108 patients were eligible and enrolled (Table 1). The mean age was 72 years (standard deviation: 12). Lung cancer was present in 43 patients (40%), lung metastases in 59 patients (55%), and ECOG PS of 4 in 85 patients (79%). In total, 48 patients (44%) were opioid-naïve, while 60 patients (56%) had been using opioids prior to the baseline assessment (opioid-tolerant). The median parenteral MEDD of regular prior opioids in the opioid-tolerant group was 18 mg (interquartile range [IQR]: 8.0–30). The median overall survival was 6 days (95% confidence interval [CI]: 4.8–7.2), and 96 patients (89%) died within one month of starting opioid treatment.
Baseline Characteristics
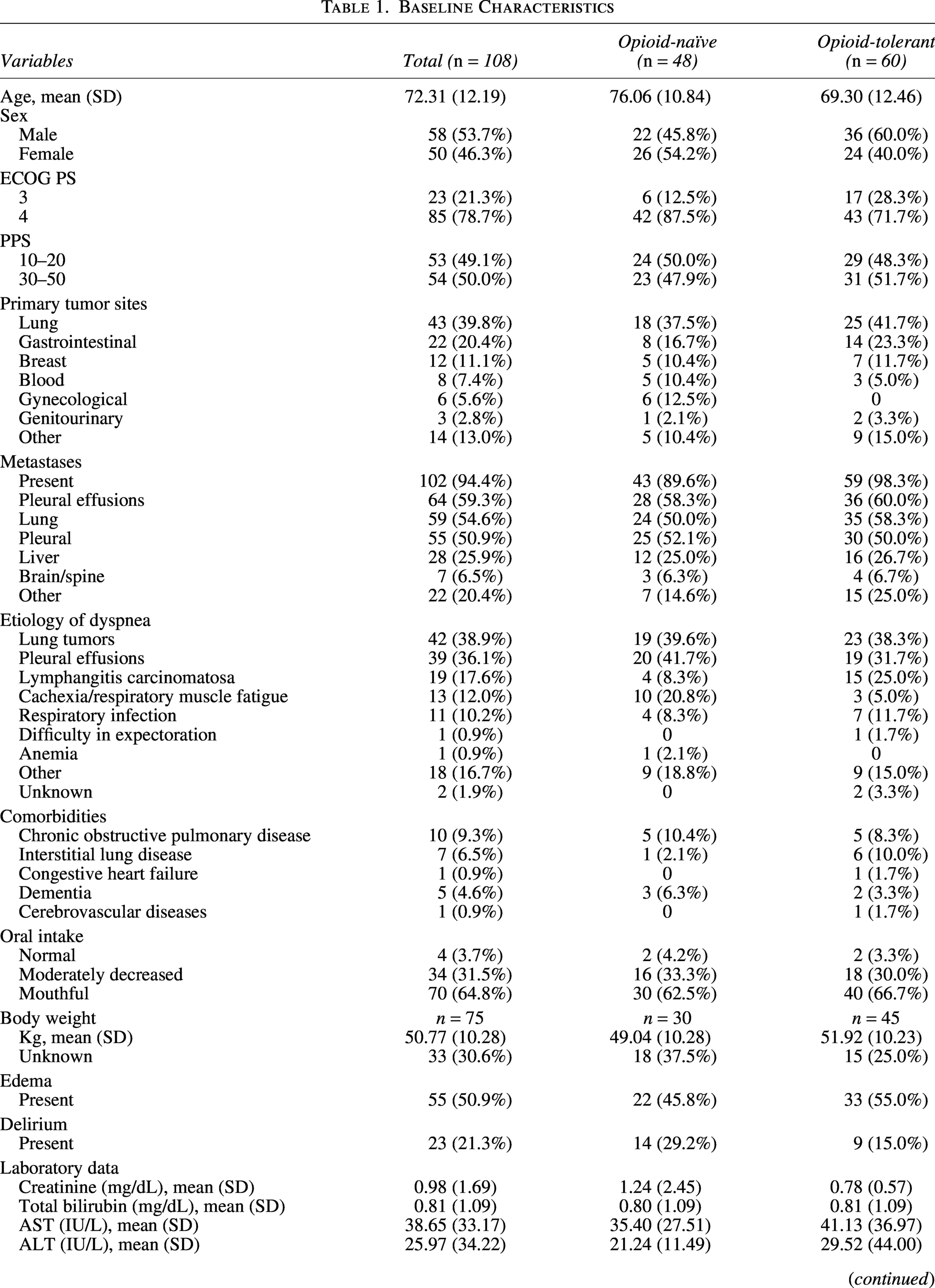
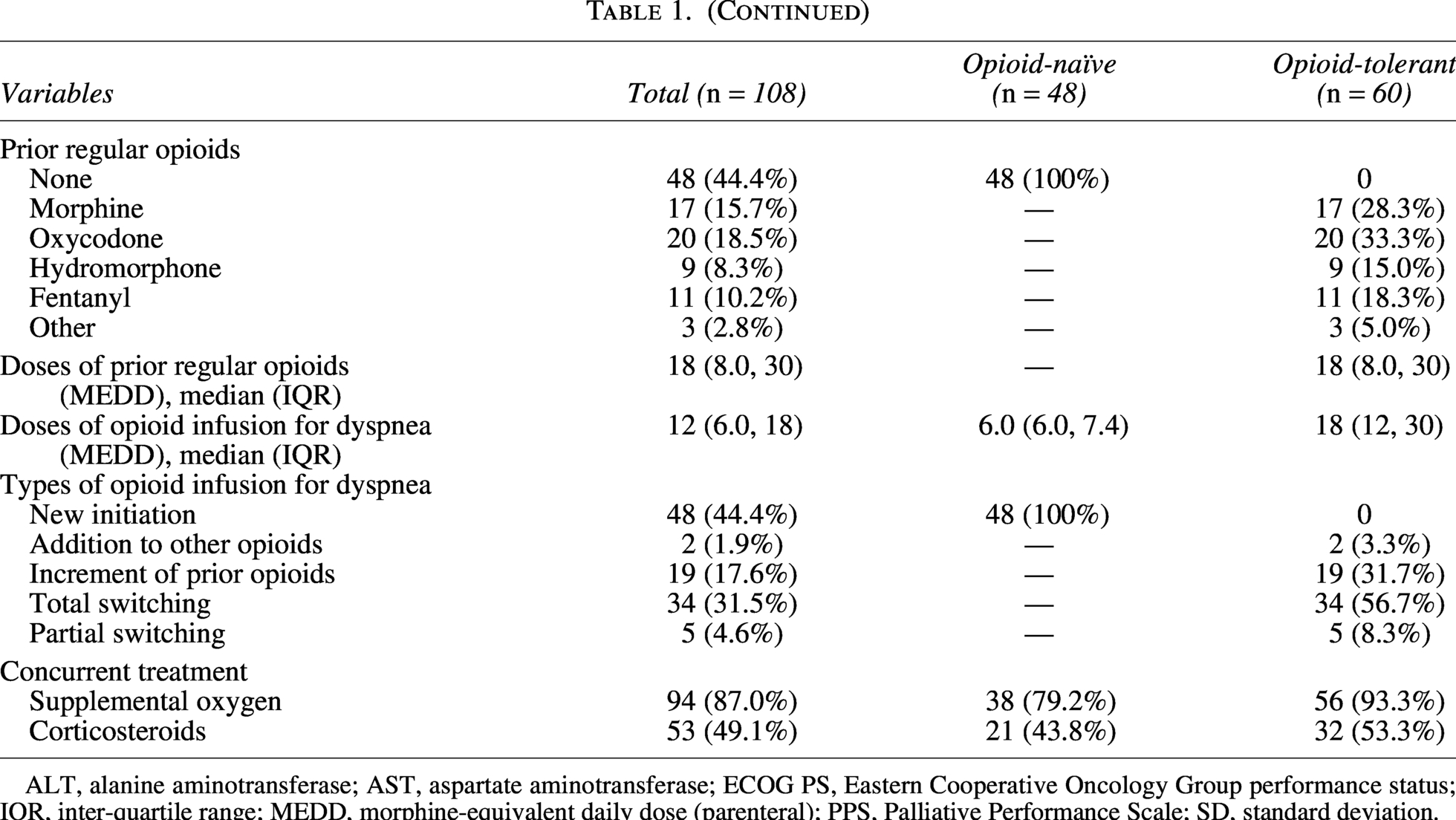
ALT, alanine aminotransferase; AST, aspartate aminotransferase; ECOG PS, Eastern Cooperative Oncology Group performance status; IQR, inter-quartile range; MEDD, morphine-equivalent daily dose (parenteral); PPS, Palliative Performance Scale; SD, standard deviation.
Outcomes of opioid titration within 48 hours
All 108 patients received algorithm-based treatment. For opioid-naïve patients, continuous opioid administration was initiated at a median parenteral MEDD of 6.0 mg/day. Over the subsequent 48 hours, a total of 12 dose titrations were performed (9 at level 1, 2 at level 2, 1 at level 3, and none at level 4), resulting in a 100% response rate (Table 2). One DLT (11%) occurred at level 1.
Outcomes after Opioid Titration Based on Clinician Global Impression-Improvement
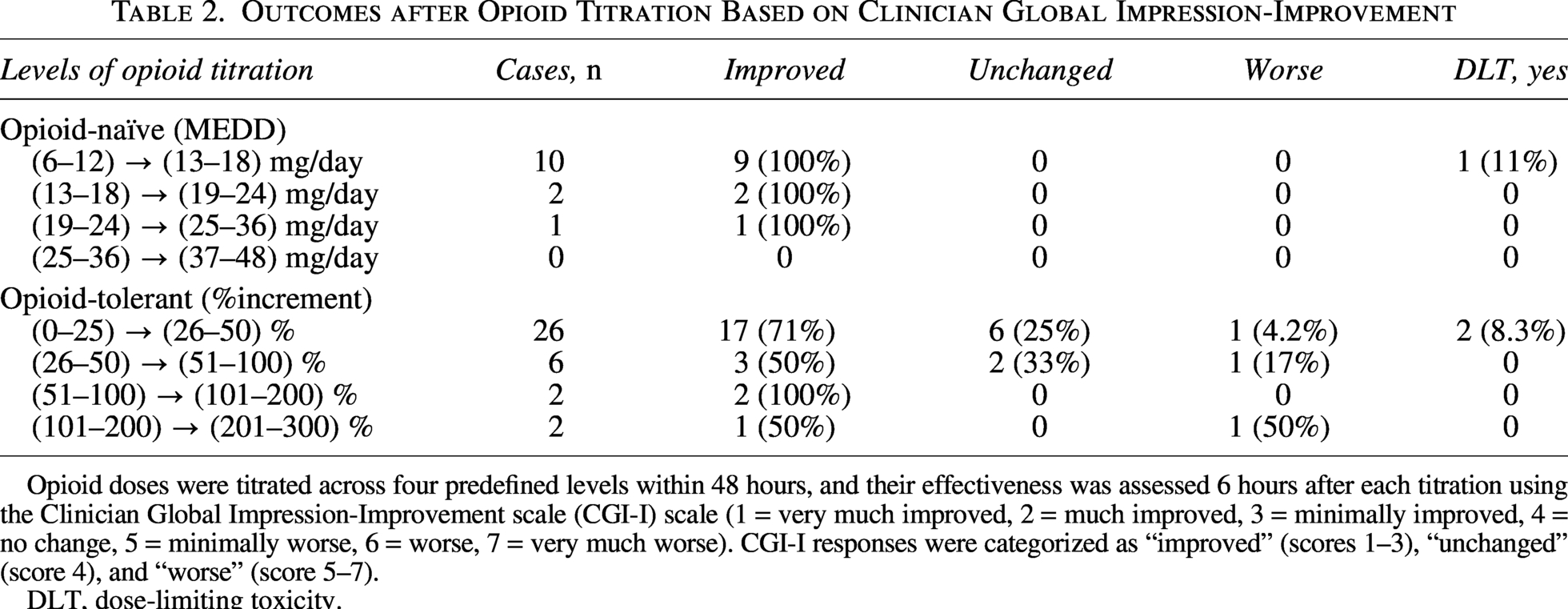
Opioid doses were titrated across four predefined levels within 48 hours, and their effectiveness was assessed 6 hours after each titration using the Clinician Global Impression-Improvement scale (CGI-I) scale (1 = very much improved, 2 = much improved, 3 = minimally improved, 4 = no change, 5 = minimally worse, 6 = worse, 7 = very much worse). CGI-I responses were categorized as “improved” (scores 1–3), “unchanged” (score 4), and “worse” (score 5–7).
DLT, dose-limiting toxicity.
For opioid-tolerant patients, continuous opioid administration was initiated at a median parenteral MEDD of 18 mg/day. Over 48 hours, 34 dose titrations were performed (24 at level 1, 6 at level 2, 2 at level 3, and 2 at level 4), resulting in an overall response rate of 68% (71% at level 1, 50% at level 2, 100% at level 3, and 50% at level 4) (Table 2). Two DLTs (8.3%) occurred, both at level 1.
Opioid dose at the end of observation
Retrospective analysis indicated that the median opioid doses (parenteral MEDD) at one month or on death after initiation were 12 mg (IQR: 6.0–18) for opioid-naïve patients and 33 mg (IQR: 21–48) for opioid-tolerant patients (Table 3). The corresponding median dose increases from the baseline were 100% (IQR: 0–200) for opioid-naïve patients and 50% (IQR: 0–175) for opioid-tolerant patients.
Opioid Dose at End of Observation
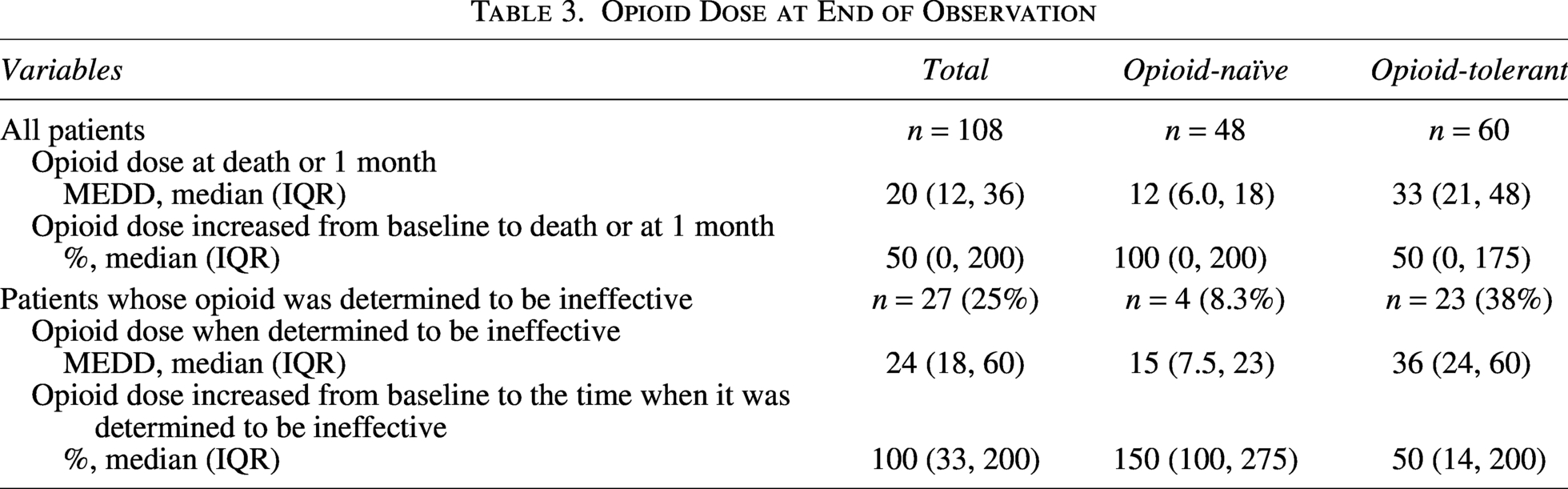
Physician-perceived upper limits of opioids
Opioids were retrospectively determined to be ineffective in 8.3% (4/48) of opioid-naïve patients and 38% (23/60) of opioid-tolerant patients (p < 0.001) (Table 3). The median parenteral MEDD at which opioids were considered ineffective was 15 mg (IQR: 7.5–23) for opioid-naïve patients and 36 mg (IQR: 24–60) for opioid-tolerant patients. These doses represented a median increase of 150% (IQR: 25–275) from the baseline in opioid-naïve patients and 50% (IQR: 24–200) in opioid-tolerant patients.
Discussion
To our knowledge, this is the first reported study to investigate responses to opioid titration for terminal dyspnea, overall opioid dose titration, and physician-perceived upper limits of opioid effectiveness in opioid-naïve and -tolerant patients with advanced cancer.
Key findings
The first important finding was that opioid-naïve patients demonstrated a more favorable response to opioid titration for terminal dyspnea than opioid-tolerant patients. This agrees with our hypothesis that prior opioid use may reduce responsiveness due to partial mu-opioid receptor occupancy. In addition, long-term opioid exposure may induce pharmacological adaptations, such as receptor downregulation and desensitization of intracellular signaling pathways, further attenuating the effect of opioid titration. 26 Opioid-tolerant patients may also represent a subgroup with more advanced disease burden, including extensive tumor infiltration, lymphangitic carcinomatosis, interstitial lung disease, heart failure, or associated hypoxemia, making dyspnea more refractory. This observation is concerning, as most advanced cancer patients who develop terminal dyspnea are already receiving opioids for pain and chronic dyspnea management.11,15,16,22 Further basic research is needed to disentangle pharmacological mechanisms from disease progression and comorbidities, and future intervention trials on opioid dose titration should validate these findings.
The second significant finding is the low incidence of DLTs. Only one DLT was observed in opioid-naïve patients and two in opioid-tolerant groups, all at the initial titration level. This is consistent with previous studies demonstrating the safety of opioid infusion with few serious adverse events.11,15,16,22 This may reflect careful titration by palliative care specialists, balancing effectiveness and safety regardless of prior opioid use. Our findings suggest that opioid titration is more constrained by limited effectiveness than by adverse effects.
Notably, opioid titration was modest overall. The median parenteral MEDD at the end of observation was 12 mg/day for opioid-naïve patients and reflected a 50% dose increase for opioid-tolerant patients. This agrees with studies on chronic dyspnea, where most opioid-naïve patients achieved symptom relief with less than 30 mg/day of oral morphine. 13 However, 38% of opioid-tolerant patients reached the perceived upper limit of opioid effectiveness, compared with less than 10% of opioid-naïve patients, which represents a significant and novel finding. A prior nationwide survey indicated that about one-third of palliative care physicians perceived an upper limit to parenteral opioid effectiveness for terminal dyspnea, which may predominantly apply to opioid-tolerant patients. 17 Our findings support this, with opioid-tolerant patients reaching a median 50% dose increase before opioids were determined to be ineffective, consistent with the survey results. 17 In contrast, opioid-naïve patients deemed to have reached upper limits received a median dose of 15 mg/day, being notably lower than the 30 mg/day reported in the survey. 17 This suggests that opioid-naïve patients may benefit more from lower opioid doses. Future research should explore effective second-line treatments for cancer patients with terminal dyspnea when opioid titration proves insufficient.
Strengths and limitations
The strengths of this study include its multicenter design, algorithm-based treatment approach in real-world settings, and use of validated outcome measures. However, several limitations should be acknowledged. First, this was a secondary analysis of an observational study rather than an interventional trial. Second, although CGI-I was used to assess the treatment response, patient-reported outcomes were not collected due to communication limitations of patients close to death.2,11 Furthermore, palliative care physicians who evaluated treatment response were not blinded to the intervention. Future studies should apply blinded assessments and identify the most suitable outcome measures for this population, including validated tools closer to patient assessment such as the Respiratory Distress Observation Scale. 27 Third, the median MEDD of prior opioids in opioid-tolerant patients was relatively low, which may limit the applicability of our findings to other international settings, partly because internationally accepted definitions of opioid tolerance (e.g., the US FDA definition) were not applied. 28 Future studies should adopt such definitions to enhance generalizability. Moreover, the initial MEDD values were lower than those recommended by international guidelines, 7 partly due to the smaller body weight of Japanese patients, although they were similar to those reported in our prior multicenter observational studies on terminal dyspnea.10,11 The most appropriate doses of opioids for terminal dyspnea should be established in future studies. Fourth, the potential influence of co-interventions was not assessed, making it difficult to determine whether the observed responses were specifically due to opioid titration or other treatments. Additionally, for patients who did not improve, it is possible that their lack of response was due to disease-related deterioration rather than treatment inefficacy. Fifth, as this secondary analysis captured only the presence or absence of DLTs without detailed categorization, we cannot exclude the possibility that phenomena such as somnolence, myoclonus, or respiratory depression reflected a mixture of opioid-induced neurotoxicity and natural decline in the last days of life. Information on adverse events (i.e., unfavorable events temporally associated with drug administration, including signs of neurotoxicity, regardless of causality) was systematically assessed using validated tools according to research policies for prospective dyspnea studies, 29 with details reported elsewhere. 16 Lastly, this study was conducted by palliative care specialists, which may limit generalizability to primary palliative care settings. Future work is needed to implement this treatment algorithm in primary palliative care and address the influence of co-interventions and disease progression on treatment outcomes.
Conclusion
In the management of terminal dyspnea, opioid-tolerant patients showed a lower response to opioid titration and a higher likelihood of their opioids being determined as ineffective compared with opioid-naïve patients. Larger prospective studies are warranted to confirm these findings and develop more effective treatment strategies for patients whose opioid titration provides suboptimal relief from terminal dyspnea.
Authors’ Contributions
All the authors contributed to the study conception and design. Material preparation, data collection, and analysis were conducted by M.M., T.Y., K.S., Y.M., R.M., H.W., T.I., Y.M., K.I., N.Y., S.M., T.Y., S.O., S.I., A.I., T.M., and E.S. The first draft of the article was written by M.M., and all the authors commented on previous versions of the article. All the authors read and approved the final article.
Footnotes
Acknowledgment
We are grateful to researchers involved in the Health, Labour and Welfare Sciences Research Grant (19EA1011, 22EA1004) and JSPS KAKENHI (Grant Number JP20K20618) for their useful advice.
Author Disclosure Statement
M.M. received honoraria from Daiichi-Sankyo, Takeda Pharmaceutical Co., Ltd., and Shionogi & Co., Ltd. T.Y. received honoraria from Shionogi & Co., Ltd, Daiichi-Sankyo, and Hisamitsu pharmaceutical Co., Inc. Y.M. received lectures fees from Hisamitsu Pharmaceutical Co., Inc., Fujimoto Pharmaceutical Corporation, Takeda Pharmaceutical Co., Ltd., Nippon Boehringer Ingelheim Co., Ltd., and Chugai Pharmaceutical Co., Ltd. Y.M. received honoraria from Shionogi & Co., Ltd, Daiichi-Sankyo, and Terumo Co. A.I. received honoraria from Daiichi-Sankyo and a donation from Shionogi & Co, Ltd. E.S. received honoraria from Shionogi & Co., Ltd, Daiichi-Sankyo, Hisamitsu pharmaceutical Co., Inc., Pfizer Co., Tsumura. Co., and Terumo Co. Other authors declare no relevant conflict of interest.
Funding Information
This work was supported in part by the Japan Hospice Palliative Care Foundation and Health, Labour, and Welfare Sciences Research Grant (19EA1011, 22EA1004) as well as JSPS KAKENHI (Grant Number JP20K20618).
Data Statement
The data of this study are available from M.M. upon reasonable request.
