Abstract
Magnesium deficiency is a common problem worldwide; however, existing magnesium dietary supplement sources require large doses to overcome low bioavailability. Previously research has established that seawater contains magnesium in addition to 72 other trace and ultratrace minerals, resulting in better bioavailability than traditional magnesium sources. The purpose of the present study was to compare the bioavailability of magnesium obtained from two different seawater processing methods (hydroxide vs. citrate). In a double-blind manner, healthy, young men and women (
INTRODUCTION
Magnesium is essential for normal biological function,1–4 and its deficiency leads to development of various negative health outcomes5–7 ; however, supplemental magnesium isn’t always effective due to low bioavailability. 8 Currently the primary means to counter low bioavailability is to increase the daily dose, which unfortunately can result in undesirable gastrointestinal side effects (i.e., increased Gastrointestinal (GI) transit time, etc.). 7 Worldwide, a variety of single-mineral magnesium forms (i.e., oxide, citrate, gluconate, glycinate, etc.) are included in many dietary supplements; however, recent evidence has suggested that North Atlantic seawater is a renewable, abundant source of natural magnesium in addition to 72 other trace and ultratrace minerals.3,9,10 Previous research has demonstrated the seawater-sourced minerals have better bioaccessibility and bioavailability than some traditional, single-mineral sources.9–11 Since there are different methods of processing seawater to generate a powder suitable for supplementation, we sought to compare the relative bioavailability between two common methods (i.e., magnesium citrate and magnesium hydroxide). To ensure that we compared the methods equally, we standardized the dosing based on the RDA for elemental magnesium (men = 480 mg/day and women = 360 mg/day). We hypothesized that regardless of the processing method, seawater-derived powder would result in those similar increases in serum/urine magnesium concentration.
Participants
We obtained approval from the Institutional Review Board at the University of North Texas (IRB # IRB-23-553; approved on April 24, 2024) to conduct this study. Twenty-two apparently healthy biological men (
Seawater Magnesium Doses

Dosing was based on the RDA for men and women. Seawater-derived magnesium citrate (Aquamin®-Mg) had a minimum of 10% elemental magnesium by weight. Seawater-derived magnesium hydroxide (Aquamin®-Mg) had a minimum of 33% elemental magnesium by weight.
Since the two seawater preparation methods (magnesium citrate and magnesium hydroxide) were known to have different elemental magnesium content, we adjusted total doses to ensure that both magnesium sources delivered similar absolute quantities of elemental magnesium.
Magnesium bioavailability trial
The testing procedures used in the present study were consistent with methods reported previously. 9 We used a standard, incremental 18-h urine magnesium test, verified by short-term serum magnesium concentration to calculate relative magnesium uptake. Briefly, participants reported to the laboratory in the morning (0500 and 1100 h) following an overnight fast (>8 h) and abstention from exercise (>24 h). Body weight and hydration (based on body water) were measured using a digital scale (SECA 554). Presupplement urine and venous blood samples (used to generate serum) were collected and frozen (−80°C) until analysis. Blood was collected into clotting evacuated tubes (Greiner SST VACUETTE), allowed to clot for 30 min at room temperature, centrifuged, aliquoted, and frozen (−80°C) until analysis. Participants were asked to collect all urine samples mid-flow consistent with previously published methods. 8 After the baseline sample collection, a low (<1 mg of Mg) magnesium meal (frozen breakfast sandwich and hashbrowns; ∼550 kcal) was provided and participants were given a maximum of 30 min to consume it along with six capsules containing their assigned supplement based on random trial order. After the food and capsules were fully consumed, a timer was set to allow for the collection of follow-up blood samples (1 and 2 h after the supplement ingestion). Subjects were also provided a cooler and a urine collection system (Greiner VACUETTE) to self-collect separate urine samples each time they urinated over the next 18 h. Participants returned between 7 and 13 individual urine samples based on the individual frequency of urination. Collected urine was returned to the laboratory and frozen (−20°C) until analysis. The procedure described above was repeated for all three test conditions in the study.
Magnesium analysis
Urine and serum samples were analyzed in triplicate using a commercially available kit (QuantiChrom Magnesium Kit; BioAssay Systems) on an automated analysis platform (Chemwell 2910; Awareness Technologies). Urine-specific gravity was measured by refractometry and in the normal range (1.005 to 1.030) for all samples collected and analyzed as part of this study. Consistent with previously published calcium research, 12 we calculated the AUC for urine magnesium excretion. This had the added benefit of normalizing variations in individual urine frequency between participants. We also calculated summated (i.e., absolute or total) magnesium excretion from baseline for each trial condition. The urine AUC and summated magnesium excretion responses were of similar magnitude. The 18-h excretion was subtracted from the RDA dose (amount absorbed) and then divided by the RDA dose to calculated percent absorbance or relative bioavailability. While we didn’t collect enough serum time points postingestion to accurately calculate AUC, we were able to calculate summated serum magnesium change over the 2-h postingestion period. These methods were appropriate for the comparison of different forms of seawater magnesium, although they don’t necessarily allow for the calculation of absolute bioavailability, which would have required the use of tracer techniques. To give a point of comparison, we also calculated the percentage change in magnesium concentration/excretion for magnesium citrate and hydroxide versus placebo.
Statistical analysis
After data were assessed to ensure the statistical assumptions of normality and constant error variance were met, key outcomes separately analyzed using a single-factor, repeated-measures Analysis of Variance (ANOVA). The single factor (condition) had three levels (placebo, magnesium citrate, and magnesium hydroxide). Condition order was included as a covariate to control for order effects in the analysis. A Bonferroni adjusted significance was set at
This short communication was designed to compare magnesium uptake between two seawater-derived preparation methods3,10,12 with different elemental magnesium content. Regardless of magnesium source, we observed a similar, significant increase in urine AUC (Fig. 1;
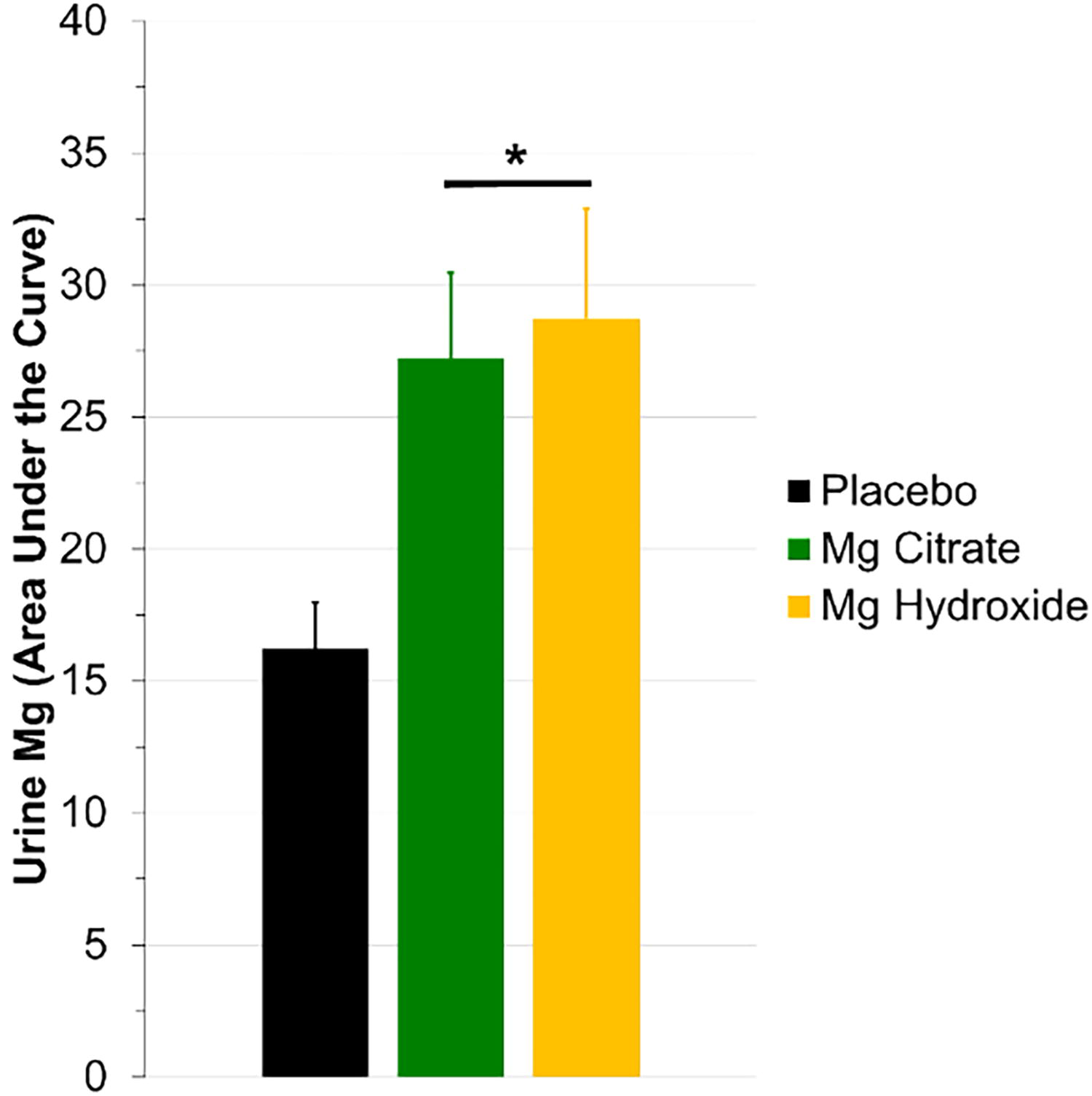
Urine magnesium area under the curve (AUC). After consuming a seawater-derived magnesium citrate (
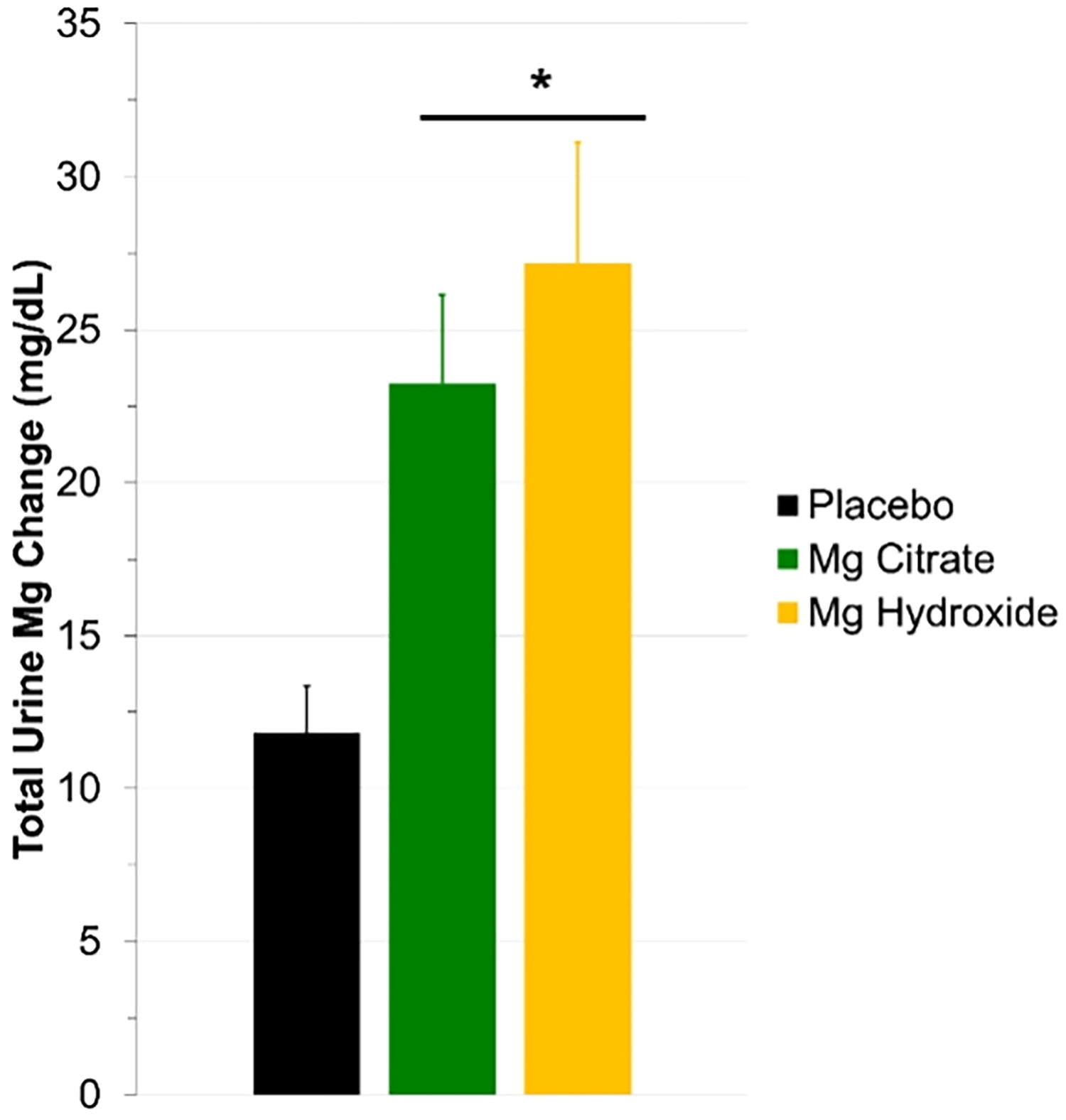
Total urine magnesium excretion. After consuming a seawater-derived magnesium citrate (
The percentage change in urine magnesium excretion compared with placebo for both magnesium citrate (97%) and magnesium hydroxide (130%) was consistent with what others have reported concerning seawater-derived magnesium supplements.3,10,12 The absolute serum magnesium concentration at baseline was similar across all three visits (placebo = 1.7 ± 0.04; Mg citrate = 1.6 ± 0.07; and Mg hydroxide = 1.6 ± 0.06), which all fall within the normal range. In support of our urine findings, summated change serum magnesium (Fig. 3;
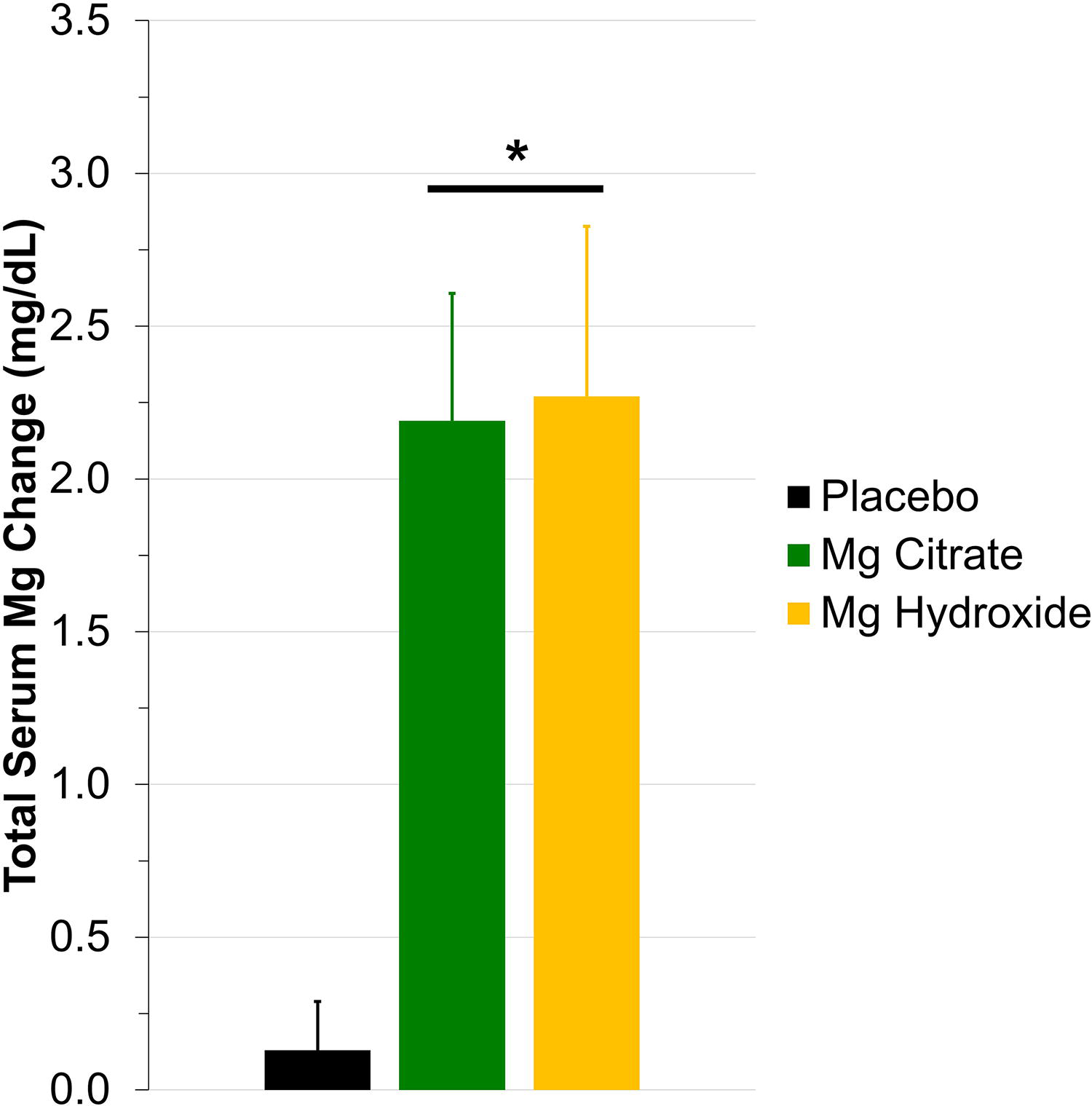
Total serum magnesium concentration. After consuming a seawater-derived magnesium citrate (
Magnesium and other minerals are important for maintenance of health, wellness, and athletic performance.2,3,5 Previous research has demonstrated that seawater-derived minerals absorb better than traditional, single-mineral supplements.9,10 The present short communication while not extensive in nature adds to the previous body of literature because we found that regardless of how you process the seawater to powder, a similar magnesium uptake is achieved. This finding will allow future research to use whichever method of processing that is most appropriate to the research design if dosing is standardized on elemental magnesium. Seawater-derived minerals are unique, in that it appears that the mix of 72 other trace and ultratrace minerals collectively improve uptake of all the containing minerals.9–11 An important aspect of the present study was our approach to standardizing the two seawater preparations on the RDA for elemental magnesium rather than total powder dose. Using this approach, it appears that the preparation method for the seawater-derived powder is less important than the elemental magnesium dose. While relative bioavailability was numerically greater with magnesium citrate, this difference did not reach statistical significance. The brief results of the present study fill an important gap in published literature concerning and may potentially give future researchers more flexibility when designing studies. Magnesium hydroxide has approximately 3.3 times more elemental magnesium than magnesium citrate by total weight.9,10 We speculate that had we designed the study based on similar total product weight, rather than elemental magnesium content, then the magnesium hydroxide preparation would have produced a greater increase in biological magnesium concentration in urine and serum.
Our finding of similar magnesium uptake was important because each of these forms has different patient-oriented supplement strategies. Formulating a magnesium containing beverage, where total product weight is not a limitation (but solubility is), then magnesium citrate may be the best option given its solubility. If formulating a capsule or tablet supplement, then magnesium hydroxide may be a better option since you can significantly reduce the total product weight (i.e., capsule size and/or number). Had we completed magnesium dosing based on total powder weight, it is plausible that serum/urine magnesium concentration levels would have been greater with magnesium hydroxide due to delivery of greater elemental magnesium dose. While the present study was brief in nature, we believe that our key findings support further investigation of seawater-derived magnesium in future, longer-term intervention studies. Consistent with what others have reported,9,10 we found that the unique composition of seawater and the 72 trace and ultratrace minerals enhanced the bioavailability of magnesium supplementation acutely. Future research should seek to further explore clinical and health application for seawater-derived trace and ultratrace mineral supplements.
AUTHORS’ CONTRIBUTIONS
B.M.F.: Funding acquisition, conceptualization, methodology, writing—reviewing and editing. A.L.P.: Methodology, writing—reviewing and editing. M.E.K.: Methodology, writing—reviewing and editing. S.M.W.: Methodology, writing—reviewing and editing.
Footnotes
AUTHOR DISCLOSURE STATEMENT
The present study was funded by grants and in-kind support to the University of North Texas from Marigot LLC (Cork, Ireland). The authors were not directly compensated, and the company was not involved with the interpretation of results or the preparation of this article. As a result, the authors report no conflict of interest associated with this work.
FUNDING INFORMATION
No funding was received for this article.
