Abstract
Background:
Guidelines for the treatment of intra-abdominal infections (IAIs) recommend a variety of potential antimicrobial regimens; however, there is a lack of real-world data documenting prescribing practices and incidence of multidrug-resistant organisms (MDROs).
Methods:
A single-center, retrospective study from June 1, 2021 to April 30, 2023, was conducted to evaluate patients with an IAI admitted to the surgical intensive care unit. Patients were categorized into four groups based on the primary gram-negative antibiotic initially prescribed for IAI treatment: piperacillin-tazobactam, cefepime, levofloxacin, and meropenem. The primary outcome was antibiotic prescribing practices for IAI. Regression analyses were conducted to evaluate risk factors for MDRO, duration of antibiotics, and mortality.
Results:
A total of 334 patients were included. Most patients received piperacillin-tazobactam (n = 227, 68%), followed by cefepime (n = 122, 36.5%), meropenem (n = 73, 21.9%), and levofloxacin (n = 61, 18.3%). There were 301 patients prescribed vancomycin for a median of 4 days (interquartile range [IQR]: 3–7). The median antibiotic duration was 8 days (IQR: 5–12). Variables associated with prolonged antibiotics were infectious disease consult (p < 0.001), multiple intra-abdominal procedures (p = 0.006), and exposure to more than one gram-negative antibiotic (p = 0.004). Predictors of MDRO were longer durations of antibiotics (odds ratio [OR]: 1.04, 95% confidence interval [CI]: 1.01–1.07) and meropenem exposure (OR: 2.11, 95% CI: 1.12–3.98). Older age (OR: 1.05, 95% CI: 1–1.08, p = 0.007) and septic shock (OR: 2.8, 95% CI: 1.3–6.1, p = 0.003) were risk factors for mortality.
Conclusions:
Piperacillin-tazobactam was the most frequently prescribed antibiotic for treatment of IAI in critically ill surgical patients. Prolonged durations of broad-spectrum antibiotics should be avoided due to the risk of developing an MDRO infection.
Background
Complicated intra-abdominal infections (IAIs) are a significant cause of sepsis in the critically ill and can be caused by a variety of organisms depending on the source of infection.1,2 IAIs are associated with significant morbidity and mortality in the critically ill.3,4 Early diagnosis, appropriate antimicrobial selection, and effective source control are the mainstays of treatment to reduce morbidity and mortality.
The Surgical Infections Society (SIS) and Infectious Diseases Society of America guidelines recommend prompt empirical antimicrobial therapy for the treatment of IAI.5,6 The selection of antimicrobial therapy depends on the infection severity, community versus hospital-associated infection, risk of multidrug-resistant organisms (MDROs), infection source, local resistance patterns, and individual patient risk factors. For healthcare-associated and severe infections, both guidelines recommend broad-spectrum antibiotic coverage targeting gram-positive, gram-negative, and anaerobic organisms. MDROs such as extended-spectrum beta-lactamase (ESBL)-producing Enterobacteriaceae, Pseudomonas aeruginosa, and vancomycin-resistant Enterococcus (VRE) spp. should be considered in critically ill patients with severe infections. Routine empirical vancomycin utilization for coverage against methicillin-resistant Staphylococcus aureus (MRSA) is not recommended and should be reserved for patients with known colonization or specific risk factors, such as prior colonization, previous treatment failure, immunocompromised status, and extensive healthcare exposures. 6
A variety of antibiotic regimens could be utilized to appropriately treat severe IAI. Piperacillin-tazobactam, ceftazidime or cefepime with metronidazole, levofloxacin or ciprofloxacin with metronidazole, and carbapenems are all potential empirical antibiotic regimens for critically ill patients with IAI; however, it is unknown which is the preferred regimen. 7 The use of inappropriate empirical antibiotic therapy increases unnecessary antibiotic exposure and is associated with treatment failure and poor outcomes.8–10 Due to the higher mortality associated with inappropriate antibiotics in critically ill patients with septic shock, it is important to ensure empirical antibiotic regimens are designed based on local antibiograms, patient-specific factors, and antibiotic stewardship principles.
Due to the variable antibiotic prescribing practices for treatment of IAI, this study was conducted to assess treatment of IAI at a large, tertiary, academic medical center. The purpose of this study was to provide a real-world description of antibiotic prescribing practices in critically ill surgical patients and assess risk factors for MDROs, mortality, and prolonged antibiotic durations.
Patients and Methods
This study was a single-center, retrospective study conducted at Vanderbilt University Medical Center (VUMC), a tertiary academic medical center in Nashville, Tennessee. This study was approved by the VUMC Institutional Review Board as an exempt study. All patients 18 years or older, admitted to the surgical intensive care unit (SICU), and prescribed antibiotics for an IAI between June 1, 2021 and April 30, 2023 were included. A manual chart review was performed to confirm diagnosis of IAI for all patients. If a patient had multiple independent instances of an IAI in a single admission, then each instance was treated as an independent event. Data were stored in research electronic data capture.
Demographic data collected included age, gender, race, and ethnicity. Other pertinent variables collected were hospital and ICU length of stay, hospital and ICU mortality, incidence of infectious disease (ID) consult, solid organ transplantation during the index admission, primary team, number of intra-abdominal surgical procedures, presence of an open abdomen, and septic shock. Septic shock was defined as requiring at least one vasopressor and a lactate >2 mmol/L. 11
The primary outcome was to describe antibiotic prescribing practices for IAI. Patients were categorized into four groups based on the primary gram-negative antibiotic initially prescribed for the treatment of the IAI. The four antibiotics were piperacillin-tazobactam, cefepime, levofloxacin, and meropenem. Patients with exposure to more than one of the four primary gram-negative antibiotics were documented as an exposure to each antibiotic. Antibiotic exposure was defined as at least one dose of the specified antibiotic. A day of antibiotic therapy was defined as administration of at least one dose of the antibiotic on a calendar day.
The SICU Antibiotic Stewardship Practice Management Guidelines at VUMC recommends using piperacillin-tazobactam first line for empirical treatment of IAI in critically ill surgical patients with options to use other broad-spectrum antibiotics in the case of a severe penicillin allergy or piperacillin-tazobactam resistance. It also discourages routine empirical use of vancomycin unless used in combination with cefepime or levofloxacin for enterococcal coverage or in patients with risk factors for MRSA infections. Risk factors for MRSA include immunocompromised state, previous history of MRSA infection, or extensive healthcare exposure in the past 90 days. Staphylococcus aureus nasal polymerase chain reaction (PCR) is not routinely utilized to assess MRSA risk. Our institution has a universal nasal decolonization protocol with administration of intra-nasal mupirocin on admission rendering the MRSA nasal PCR test unreliable.
Secondary outcomes included mortality, duration of antibiotic therapy for treatment of IAI, incidence of MDRO, incidence of being exposed to more than one antibiotic with gram-negative coverage, incidence of vancomycin exposure, and number of ID consults. The duration of antibiotic therapy for the treatment of IAI was defined as the total duration of antibiotic administration from initiation to discontinuation of therapy. The incidence of MDRO was defined as bacteria resistant to three or more classes of antibiotics (ceftriaxone or cefepime, fluoroquinolones, ampicillin-sulbactam or piperacillin-tazobactam, and aminoglycosides), ESBL-producing bacteria, VRE, or MRSA. Bacteria isolated from cultures could meet more than one MDRO criteria. For instance, an ESBL-producing Escherichia coli that was resistant to ceftriaxone, piperacillin-tazobactam, and a fluoroquinolone would be documented as both ESBL and resistant to three or more antibiotic classes. Patients may have had MDRO identified in more than one culture, including intra-abdominal, blood, urine, pulmonary, and tissue cultures.
Due to the high number of transplant patients at our institution, a priory subgroup analysis of solid organ transplant (SOT) patients was conducted. Variables included in this analysis were antibiotic exposures, duration of antibiotics, and incidence of MDRO.
All statistical analyses were completed using SPSS V29.0 (IBM Corporation, Armonk, NY). Descriptive statistics with medians and interquartile ranges (IQRs) were calculated for all variables. Categorical variables were compared using the χ2 test. The Mann–Whitney U test was used for nonparametric continuous variables. Binary logistic regression was performed to determine risk factors associated with MDRO and mortality. Multiple linear regression was used to examine factors associated with prolonged antibiotic use. For all analyses, p < 0.05 was considered to be significant.
Results
A total of 722 patients received broad-spectrum antibiotics in the SICU during the study period. Of those patients, 334 were prescribed antibiotics for the treatment of an IAI. Most patients were male (n = 189, 56.6%) with a median age of 60 years (IQR: 49–68) (Table 1). The admitting service with the highest number of patients included was the emergency general surgery service and accounted for about 45% (n = 151) of the patients. Twenty-four (7.2%) patients were on the liver transplant service, and a total of 25 patients (7.5%) had a SOT during their admission. Most of the patients (n = 244, 73%) required multiple intra-abdominal procedures with 146 (43%) having an open abdomen. Septic shock was common and occurred in 132 (39.5%) patients.
Baseline Demographics
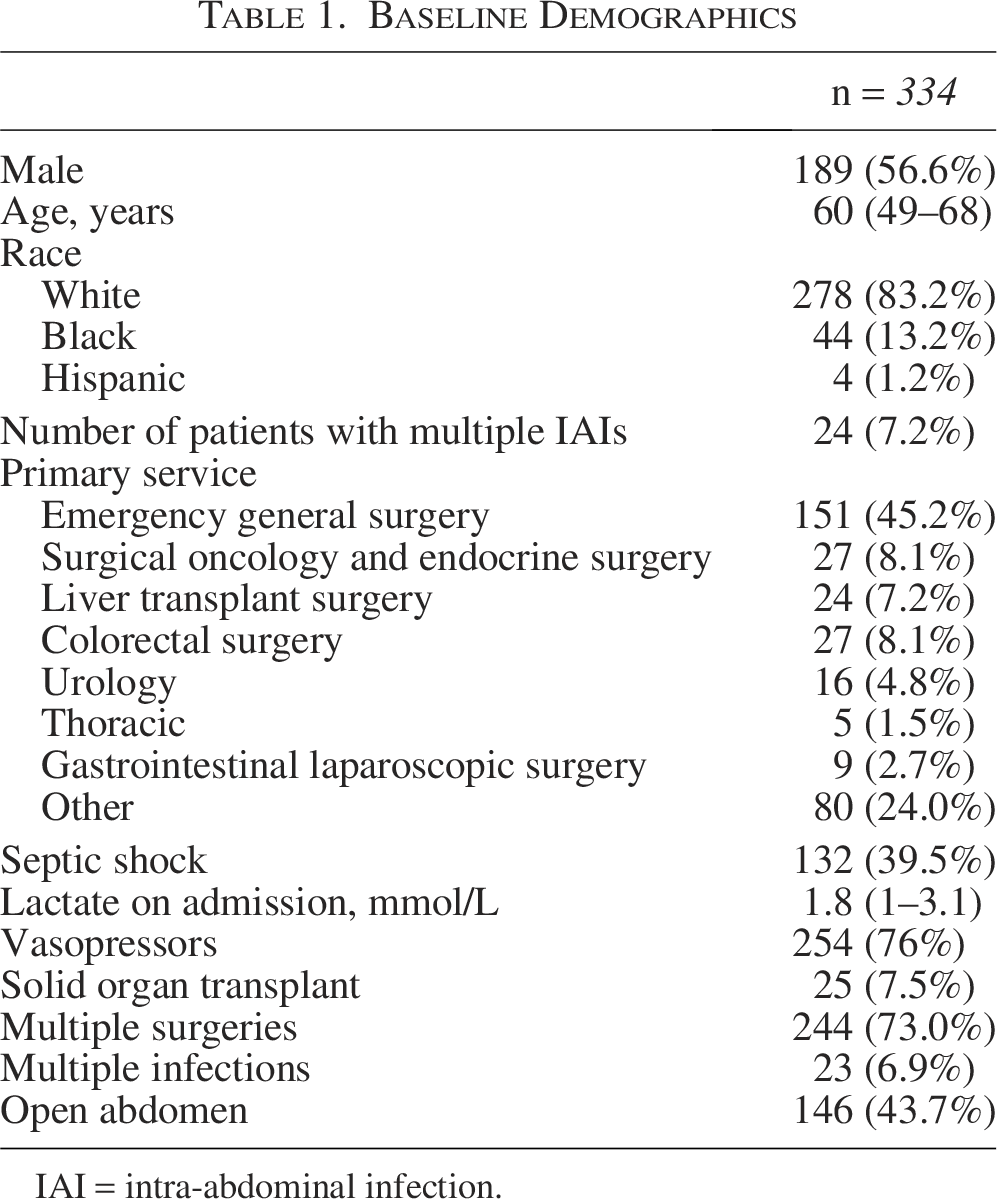
IAI = intra-abdominal infection.
The most prescribed antibiotic was piperacillin-tazobactam (n = 227, 68%) (Fig. 1). Cefepime was the second most prescribed antibiotic (n = 122, 36.8%), and meropenem the third most prescribed antibiotic (n = 73, 21.9%). Levofloxacin was the least encountered antibiotic (n = 61, 18.3%). One-hundred and twelve (33.5%) patients were exposed to two of the four primary gram-negative antibiotics, 19 (5.7%) were exposed to three of the four primary gram-negative antibiotics, and 2 (0.6%) were exposed to all four primary gram-negative antibiotics. Most of the patients (n = 238, 71.3%) were initially started on vancomycin empirically, and an additional 63 patients had vancomycin added later to their treatment regimen for IAI. When cefepime was used as the primary gram-negative antibiotic, vancomycin was prescribed along with cefepime in 96.2% (n = 113) of patients. A total of 301 patients (90.1%) had a vancomycin exposure.
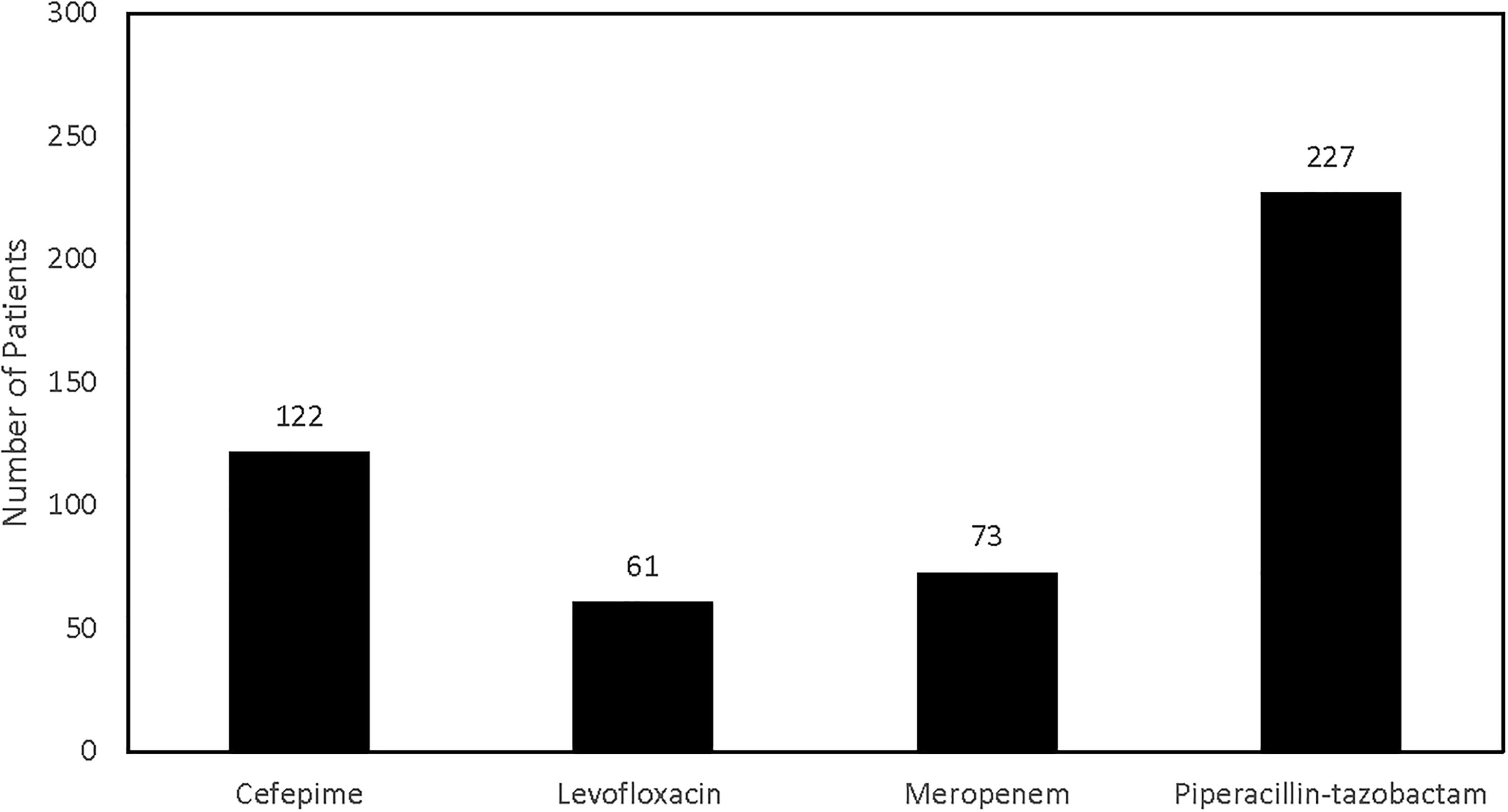
The number of patients prescribed the four antibiotics used for gram-negative coverage of IAI. The most common antibiotic was piperacillin-tazobactam. Patients could have exposure to more than one antibiotic during their treatment course. IAI, intra-abdominal infection.
The median duration of antibiotic therapy for the treatment of IAI was 8 days (IQR: 5–12; Table 2). Forty-nine percent (n = 165) of patients received an antibiotic course of 7 days or less with 30% (n = 49) of those patients receiving 4 days or less of antibiotics. About 20% (n = 66) of patients had a treatment course greater than 14 days with one patient receiving a course of therapy for 72 days. Although the overall vancomycin exposure rate was high (90.1%), the median duration of vancomycin therapy was only 4 days (IQR: 3–7). The presence of an open abdomen did not significantly change the duration of antibiotics (median 7 d vs. 8 d, p = 0.267). The median duration of antibiotic therapy was significantly longer in patients who had an ID consult (median 6 d vs. 10 d, p < 0.001), multiple intra-abdominal procedures (median 5 d vs. 8.5 d, p < 0.001), positive blood culture (median 7 d vs. 10 d, p = 0.011), positive intra-abdominal culture (median 6 d vs. 12 d, p < 0.001), and septic shock (median 7 d vs. 9.5 d, p < 0.001) compared with those without the specified risk factor (Table 3).
Secondary Outcomes
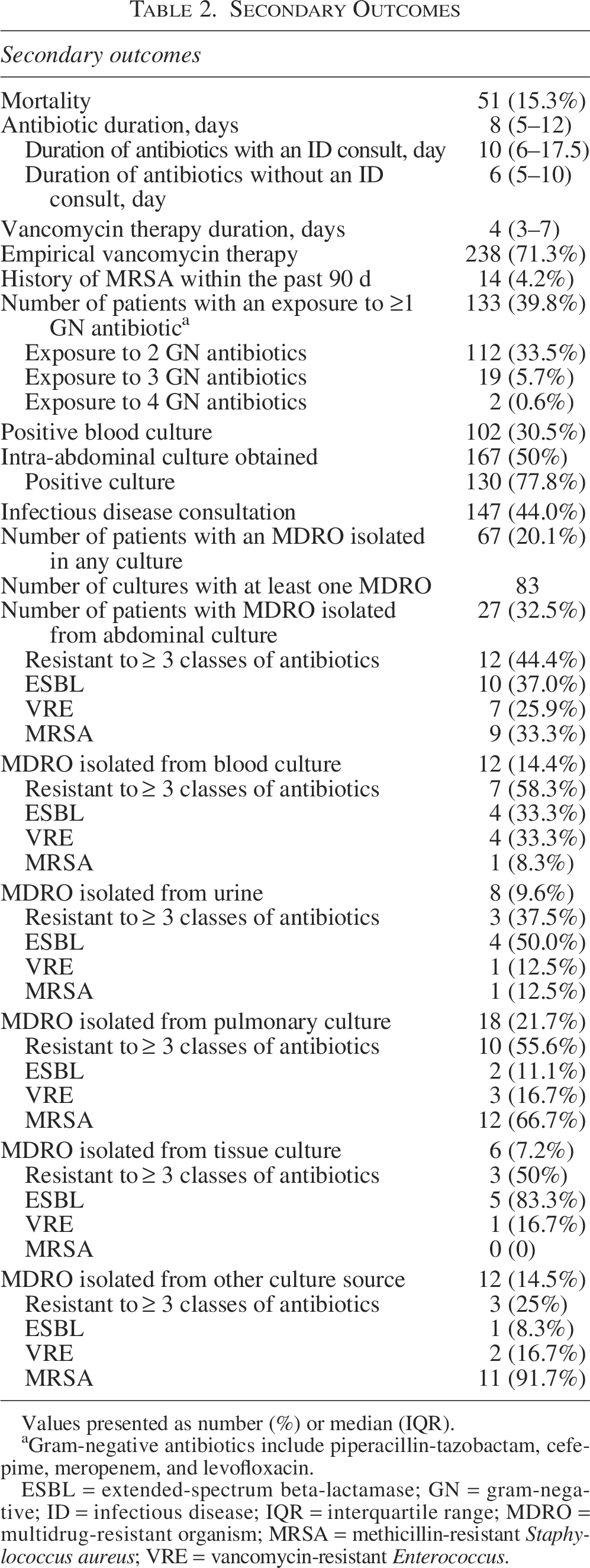
Values presented as number (%) or median (IQR).
Gram-negative antibiotics include piperacillin-tazobactam, cefepime, meropenem, and levofloxacin.
ESBL = extended-spectrum beta-lactamase; GN = gram-negative; ID = infectious disease; IQR = interquartile range; MDRO = multidrug-resistant organism; MRSA = methicillin-resistant Staphylococcus aureus; VRE = vancomycin-resistant Enterococcus.
Duration of Antibiotics
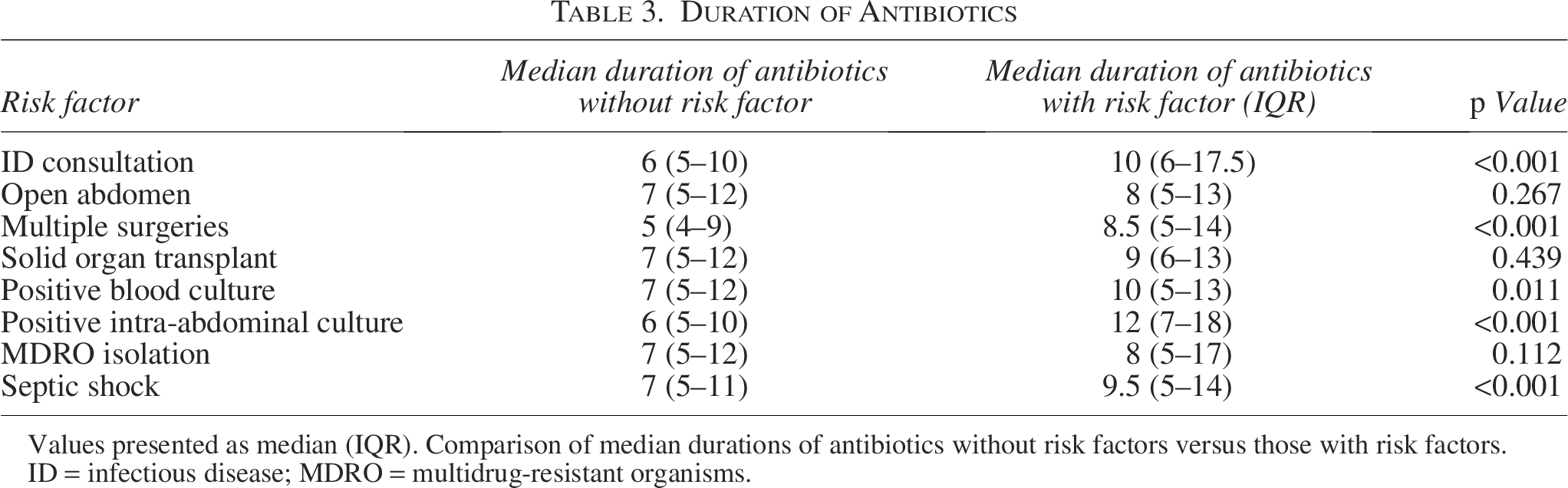
Values presented as median (IQR). Comparison of median durations of antibiotics without risk factors versus those with risk factors.
ID = infectious disease; MDRO = multidrug-resistant organisms.
Intra-abdominal cultures were obtained in 167 (50%) patients with 130 (77.8%) patients having a positive intra-abdominal culture. One-hundred and two patients (30.5%) had a positive blood culture. There were 37 cultures (22.1%) that resulted with no growth. E. coli was the most common organism isolated from intra-abdominal cultures (n = 37, 22.2%). Enterococcus spp. (n = 34, 20.4%), Klebsiella spp. (n = 26, 15.6%), and Streptococcus spp. (n = 21, 12.6%) were also common. Anaerobes only grew on 19 intra-abdominal cultures (Bacteroides spp., n = 10, 6.0%, and Clostridium spp., n = 9, 5.4%).
A total of 67 patients (20.1%) had at least one culture grow an MDRO (Table 2). There were 83 cultures that resulted with an MDRO in 67 (20%.1) patients. ESBL was isolated from at least one culture in 16 patients (4.8%), VRE in 13 patients (3.9%), and MRSA in 29 patients (8.7%). MDROs were most encountered in intra-abdominal cultures (n = 27, 32.5%) and pulmonary cultures (n = 18, 21.7%). Of the 27 patients who had positive intra-abdominal cultures, only 9 patients had MRSA isolated accounting for an overall incidence of 2.7%. Most of the MRSA positive cultures were from a pulmonary source (n = 12). For intra-abdominal cultures, there were 10 patients (3%) who had an ESBL isolated, and 7 patients (2.1%) had VRE.
When examining variables associated with MDRO, longer durations of antibiotics (odds ratio [OR]: 1.04, 95% confidence interval [CI]: 1.01–1.07) and meropenem exposure (OR: 2.11, 95% CI: 1.12–3.98) were independently associated with a positive MDRO culture (Table 4). Exposure to cefepime (OR: 0.71, 95% CI: 0.47–1.67), piperacillin-tazobactam (OR: 0.88, 95% CI: 0.46–1.70), and levofloxacin (OR: 0.92, 95% CI: 0.44–1.90) did not demonstrate a higher risk for MDRO isolation. Age (OR: 1.03, 95% CI: 1.01–1.06) and septic shock (OR: 2.59, 95% CI: 1.39–4.83) were found to be independent predictors of mortality. ID consultation (p < 0.001), multiple surgeries (p = 0.006), and exposure to multiple gram-negative antibiotics (p = 0.004) were all associated with prolonged antibiotic treatment courses (Table 4).
Factors Associated with Mortality, Prolonged Duration of Antibiotics, and MDRO
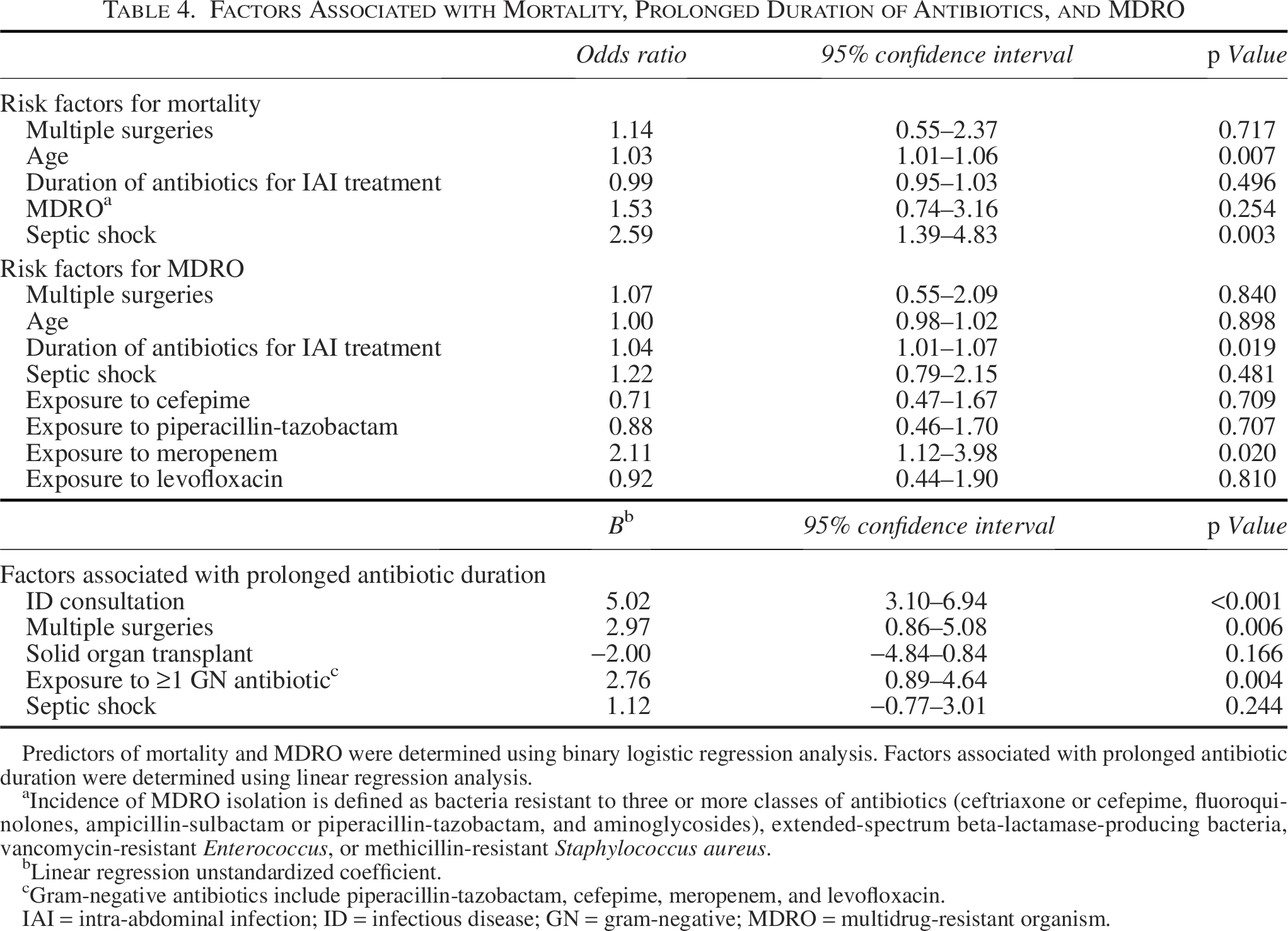

Predictors of mortality and MDRO were determined using binary logistic regression analysis. Factors associated with prolonged antibiotic duration were determined using linear regression analysis.
Incidence of MDRO isolation is defined as bacteria resistant to three or more classes of antibiotics (ceftriaxone or cefepime, fluoroquinolones, ampicillin-sulbactam or piperacillin-tazobactam, and aminoglycosides), extended-spectrum beta-lactamase-producing bacteria, vancomycin-resistant Enterococcus, or methicillin-resistant Staphylococcus aureus.
Linear regression unstandardized coefficient.
Gram-negative antibiotics include piperacillin-tazobactam, cefepime, meropenem, and levofloxacin.
IAI = intra-abdominal infection; ID = infectious disease; GN = gram-negative; MDRO = multidrug-resistant organism.
A subgroup analysis was conducted for SOT patients. Of the 25 SOT recipients, one patient (4%) experienced a second IAI during their admission. The antibiotic prescribing practices in this population were similar to the overall population: piperacillin-tazobactam (n = 17, 68%), cefepime (n = 7, 28%), meropenem (n = 8, 32%), and levofloxacin (n = 4, 16%). SOT recipients were more likely to be male (56.6% vs. 80%, p = 0.014), have septic shock (39.5% vs. 20%, p = 0.038), receive an ID consult (44% vs. 80%, p < 0.001), undergo multiple procedures (73% vs. 92%, p = 0.028), and have MDRO isolation from abdominal cultures (32.5% vs. 56%, p = 0.002) compared with non-SOT patients. There was no difference in duration of therapy between SOT recipients and non-SOT patients (median 9 vs. 7 d, p = 0.439). There were 20 patients (80%) who received an ID consult, 15 (60%) had an open abdomen, and 23 (92%) required multiple surgical procedures. Twenty-four percent of SOT patients (n = 6) had MDRO isolated from abdominal cultures compared with only 20% (n = 67) of non-SOT patients (p = 0.002). Of these patients, three patients had VRE, two had an ESBL, two had MRSA, and one had a bacteria resistant to three or more antibiotics.
Discussion
This study is a real-world description of antibiotic prescribing practices in critically ill surgical patients with IAI. We found that piperacillin-tazobactam was the most prescribed antibiotic utilized in 68% of patients. This aligns with current clinical guidelines, our institutional practice, and the most common causative bacteria for IAI. Meropenem was found to be associated with the presence of MDRO (OR: 2.11, 95% CI: 1.12–3.98). This was anticipated as meropenem is typically reserved for the treatment of MDRO. It was also not surprising that vancomycin was almost always used in combination with cefepime to provide Enterococcus spp. coverage. During the study period, the ACORN trial was being conducted. 12 It enrolled septic patients presenting from the emergency department and randomized them to either cefepime or piperacillin-tazobactam. Patients in our study could have been enrolled in the ACORN trial, and antibiotic prescribing practices could have been influenced by this trial.
Despite the low incidence of MRSA isolated in intra-abdominal cultures (2.7%), vancomycin exposure was notably high at 90.1% with 71.3% being empirical treatment. One explanation for this could be the high utilization of vancomycin with cefepime for empirical enterococcal coverage. There is limited evidence to support the routine use of adequate coverage against Enterococcus spp. in critically ill patients with IAIs.5,6,13 A post hoc analysis of the STOP-IT trial found no difference in treatment failure, recurrent infection, or mortality in patients who received empirical anti-enterococcal regimens compared with those who did not (OR: 1.53, 95% CI: 0.78–3.01, p = 0.22).14,15 A meta-analysis of 23 randomized controlled trials conducted by Zhang et al. demonstrated antibiotic regimens with enterococcal coverage had no significant improvement in treatment success compared with control regimens (relative risk: 0.99, 95% CI: 0.97–1.00, p = 0.15). 16 It may be reasonable to limit enterococcal coverage to select patient populations such as those with previous postoperative infections, cephalosporin exposure, immunocompromised, or exposure to other antibiotics selecting for Enterococcus spp. 5
Although the vancomycin exposure rate was high in our analysis, the median duration of therapy was only 4 days compared with 8 days for the IAI treatment course. This could have been due to the narrowing of antibiotics once culture results were finalized or antibiotic stewardship interventions.
The low incidence rate of MRSA IAI in this study is similar to rates published in the literature. 6 A study by Sanders et al. found no difference in the composite outcome of recurrent IAI or death at 30 days between patients who received vancomycin compared with those who did not. 15 In addition, the SIS guidelines recommend against routine empirical MRSA coverage with vancomycin unless the patient has known risk factors for a MRSA infection. 5 These results demonstrate that continued education of appropriate empirical vancomycin prescribing is needed.
Prolonged antibiotic duration was found to be a risk factor for MDRO isolation. This finding was also seen in a retrospective study by Smith et al. that examined short versus prolonged courses of antibiotics for IAI. 17 However, the two largest prospective, randomized controlled trials examining antibiotic duration for IAI found no difference in the incidence of MDRO infections. The DURAPOP trial compared 8 versus 15 days of antibiotics with similar rates of resistant infections in both groups (p = 0.28), and the STOP-IT trial compared 4 versus 8 days with no difference in MDRO infections (p = 0.29).14,18–23 Although there are conflicting results in recent studies, it is a core principle of antimicrobial stewardship to limit the duration of broad-spectrum antibiotics to prevent emergence of resistance bacteria.
In our study, the median duration of antibiotic therapy was 8 days (IQR: 5–12), with 64% of patients receiving therapy for 10 days or less. A limitation of this study is that the date of source control was not obtained due to the difficulty determining this in a retrospective review. Although the median duration of antibiotics in this study was longer than the recommended 4–7 days, the omission of the source control date makes it difficult to analyze whether the antibiotic durations were appropriate. However, the high number of patients with open abdomens and multiple procedures would indicate that a significant number of patients achieved source control several days into antibiotic therapy as antibiotics are initiated immediately upon diagnosis. Patients who underwent multiple procedures had longer antibiotic durations compared with those who had a single procedure (median 5 d vs. 8 d, p < 0.001). Our study also included patients who had IAI and bacteremia. Patients with positive blood cultures had significantly longer durations compared with those without bacteremia (median 10 d vs. 7 d, p < 0.011). The presence of an ID consult was also associated with longer antibiotic durations (median 10 d vs. 6 d, p < 0.001) indicating patients with complex infectious processes. Overall, our study population was complex with several patients having multiple sources of infection requiring longer durations of antibiotic therapy beyond the typical IAI treatment duration.
This study is unique because it included a variety of critically ill surgical patients, including septic shock (39.5%), open abdomen (43.7%), multiple surgical procedures (73.0%), multiple infections (6.9%), and SOT (7.5%). The inclusion of SOT patients is rare. The STOP-IT and DURAPOP studies excluded this patient population. SOT patients represent a high-risk population due to their immunocompromised state placing them at an increased risk for severe infections, hospital readmissions, and prolonged antimicrobial durations. Although our study demonstrated higher rates of MDRO in SOT patients, the overall duration of antibiotics was similar between SOT and non-SOT patients (median 9 vs. 7 d, p = 0.439).
The overall mortality of our population was about 15%, which is higher than the mortality rate seen in the DURAPOP (9.1–13.6%) and STOP-IT trials (0.8–1.2%). The increased mortality could be due to a higher severity of illness and the inclusion of all patients with IAI. When examining risk factors for mortality, age (OR: 1.03, 95% CI: 1.01–1.06, p = 0.007) and septic shock (OR: 2.59, 95% CI: 1.39–4.83, p = 0.003) were both independent predictors of mortality, which is consistent with previous literature.
This study has several limitations that should be considered. This was a retrospective, single-center study with data collection being limited to the information available in the electronic medical record. Due to the retrospective design, the rationale for the selection or duration of antibiotics could not be determined. Furthermore, patient identification was dependent on documentation of an IAI in the electronic medical record.
Conclusion
In conclusion, piperacillin-tazobactam was the most frequently prescribed antibiotic for the treatment of IAI in critically ill surgical patients. Treatment of IAI can be challenging as many patients develop secondary infections (e.g., bacteremia) and require multiple procedures to obtain source control. However, antibiotic stewardship principles of using shorter durations of antibiotics and reducing the use of unnecessary antibiotics were supported by our data. MRSA was rarely isolated, and routine empirical vancomycin should be avoided. Prolonged durations of broad-spectrum antibiotics should also be avoided due to the risk of developing an MDRO infection.
Authors’ Contributions
T.S.L., B.R.B., and K.A.R. designed the study. T.S.L. and B.R.B. collected the data. T.S.L., B.R.B., and K.A.R. interpreted the data and drafted the article. All authors have reviewed and approved this article prior to submission for publication and contributed to revisions.
Footnotes
Author Disclosure Statement
There are no conflicts of interest to disclose.
Funding Information
No funding was received for this article.
