Abstract
Pulmonary cystic lesions in infants are uncommon and can present diagnostic challenges due to overlapping radiologic features with other cystic lung conditions. We present 2 cases of left lung cystic lesions in infants. Initial high-resolution computed tomography (HRCT) suggested differential diagnoses, including pneumatocele, type I congenital pulmonary airway malformation (CPAM), or a solitary cystic lymphangioma. Definitive diagnosis was achieved through histopathological examination after left lower lobectomy and resection of the lesion. These cases highlight the challenges in accurately diagnosing pulmonary cystic lesions, given the limitations of imaging alone.
Introduction
Pulmonary cystic lesions in infants represent a critical area of neonatal care, with conditions like congenital pulmonary airway malformation (CPAM), Congenital Lobar Emphysema (CLE), bronchogenic cysts, and lymphatic malformations posing significant diagnostic and therapeutic challenges. 1 Early prenatal detection has transformed outcomes, yet these lesions vary widely—from benign to life-threatening (Table 1).2,3 Diagnosing these lesions is essential due to the potential impact on neonatal respiratory health, development, and long-term outcomes. This article highlights the importance of timely diagnosis and tailored management to improve respiratory function, reduce complications, and support long-term quality of life in affected infants.
Differential Diagnoses for Pulmonary Cystic Lesions in Neonates and Infants.

Case 1
The infant boy, born at 32 weeks following a pregnancy complicated by premature rupture of membranes and oligohydramnios, presented at birth with respiratory distress and cardiovascular instability. Treatment included surfactant, invasive ventilation, inhaled nitric oxide, and inotropic support. Bilateral pneumothoraces were resolved with chest tubes. After 10 days, the infant transitioned to non-invasive support. Chest X-rays (CXR; Figure 1) showed a cystic lesion in the left lower lobe, initially attributed to barotrauma. However, a high-resolution computed tomography (HRCT) scan (Figure 2) revealed a hypoplastic left lung with atelectasis, architectural distortion, and a posterior collection of cystic structures suggestive of pneumatoceles.
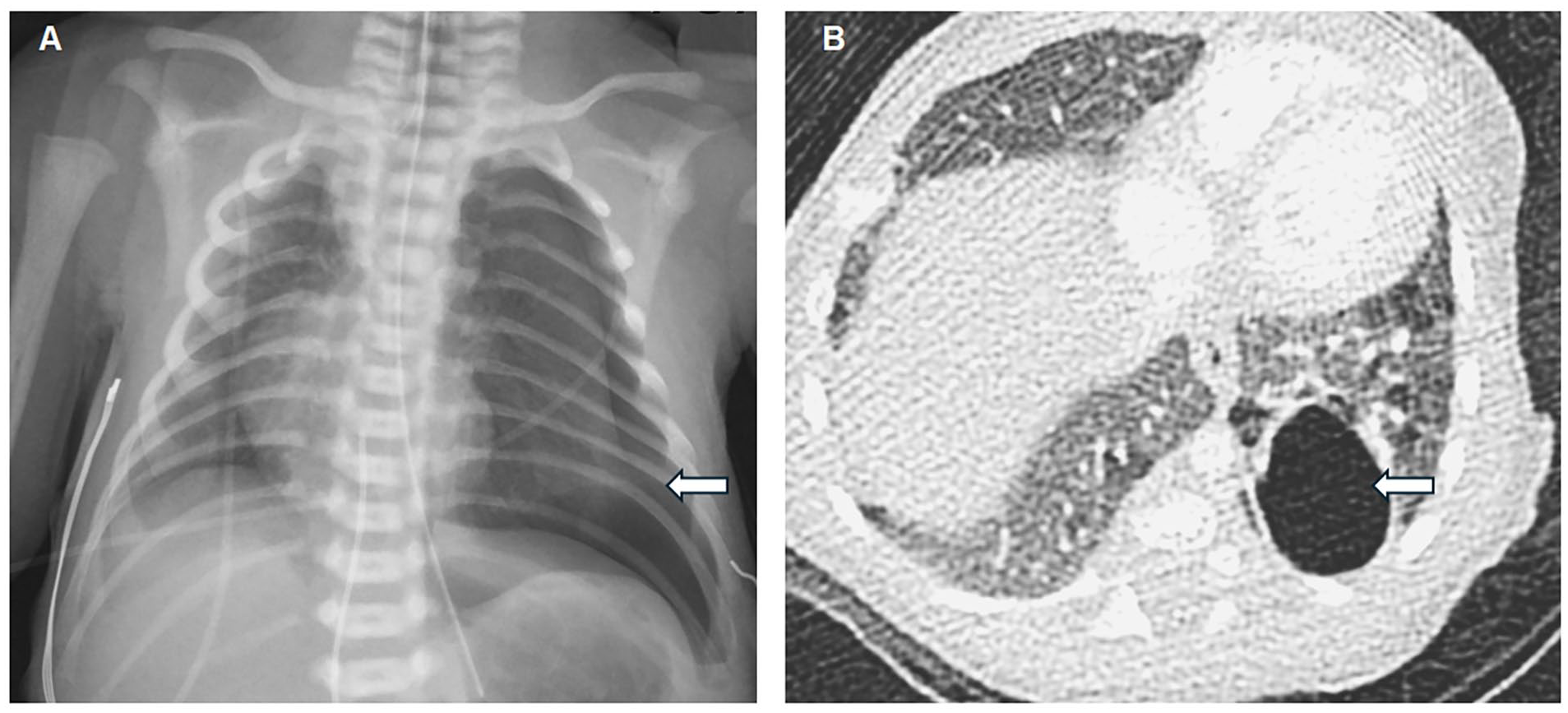
Case 1: (A) Initial CXR at 0 days of life showing large left pneumothorax and partial collapse of the left lung with mediastinal shift to the right, suggesting the pneumothorax is under tension. No focal cyst is seen in the left lung. (B) Contrast enhanced CT obtained at 2 months of age demonstrates hypoplasia of the left lung containing an elongated septated posterior cyst with adjacent areas of air trapping and architectural distortion. There was no aberrant arterial supply or venous drainage. Main differential considerations included complex pneumatocele and CPAM.
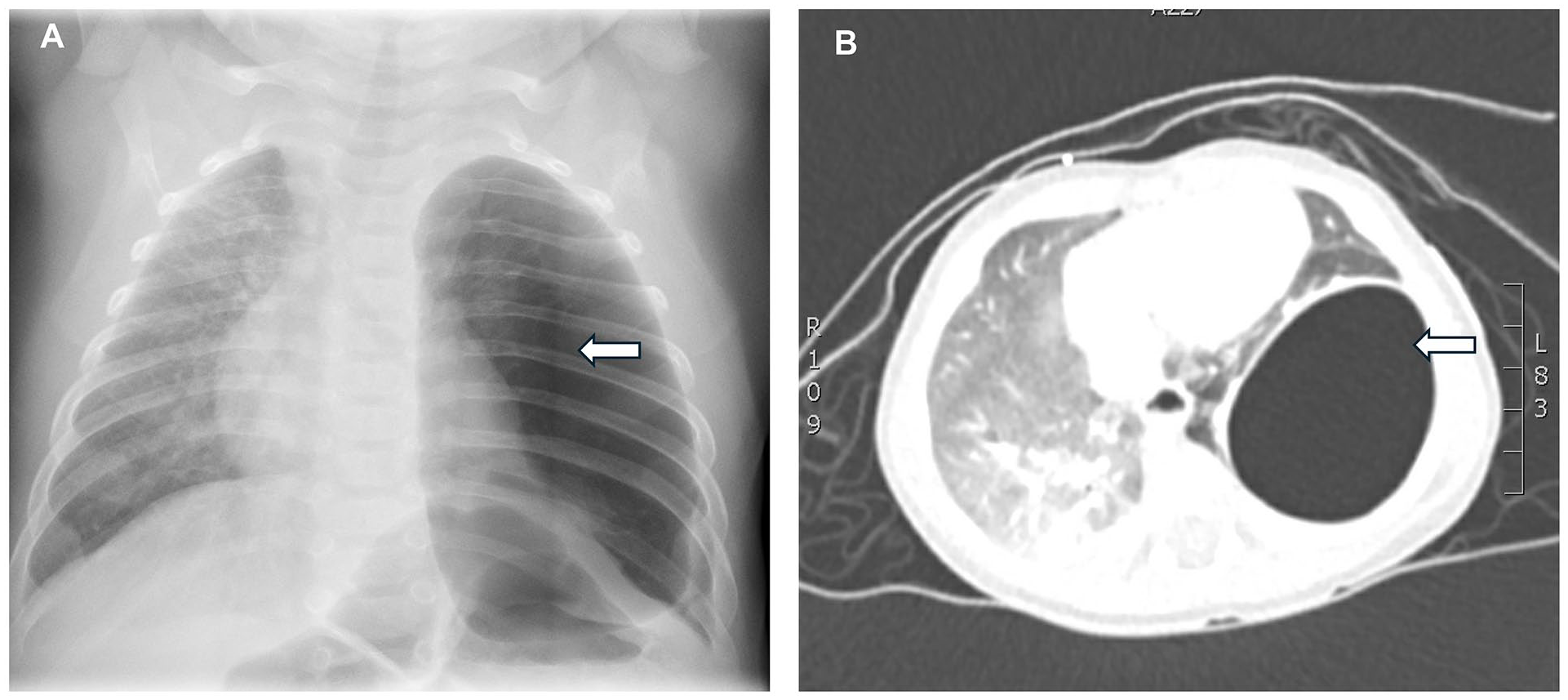
Case 2: (A) Initial CXR showing large left pneumothorax with mediastinal shift to the right, suggesting the pneumothorax is under tension. No focal cyst is seen in the left lung. (B) Contrast enhanced CT demonstrating thin-walled, air-filled, cystic lesion in the left lower lobe, situated posterolaterally. There was no evidence of communication with the bronchial tree. There is no evidence of systemic vascular supply. Differential diagnosis included pneumatocele, type I congenital pulmonary airway malformation, and pleuropulmonary blastoma if the patient has a DICER 1 mutation.
Given the patient’s history, barotrauma-associated localized persistent pulmonary interstitial emphysema (PPIE)/pneumatocele was most likely, however, type 1 CPAM or other less common causes (Table 1) could not be ruled out. A pediatric echocardiogram revealed a small patent foramen ovale, hypoplastic left upper pulmonary vein, and hypoplastic left pulmonary artery, with no pulmonary hypertension. After discharge at 40 weeks with a provisional diagnosis of barotrauma-related pneumatocele, subsequent short admissions for respiratory viral exacerbations showed increasing cyst size and evolving mass effect on the left lung. At 4 months, a thoracotomy performed revealed a large cyst with dense adhesions. Since it was contiguous with the left lower lobe, a lobectomy was performed. The left upper lobe was hypoplastic but otherwise normal. By 8 months, the patient had recovered, growing well without respiratory support.
Pathologic examination of the cystic lesion showed features that were most consistent with localized PPIE/pneumatocele (Figure 3). Grossly, a unilocular 5 cm cyst with a smooth lining had no pleural connection. Microscopically, it had a variably thick, hypocellular myofibroblastic wall (trichrome, SMA positive) without lining cells (CK, D2-40, Calretinin negative). No lymphoid aggregates or multinucleated giant cells were present. The adjacent lung showed reactive bronchial metaplasia and compression, with no vascular malformation or CPAM. The background lung had alveolar growth abnormalities with enlarged, simplified air spaces.
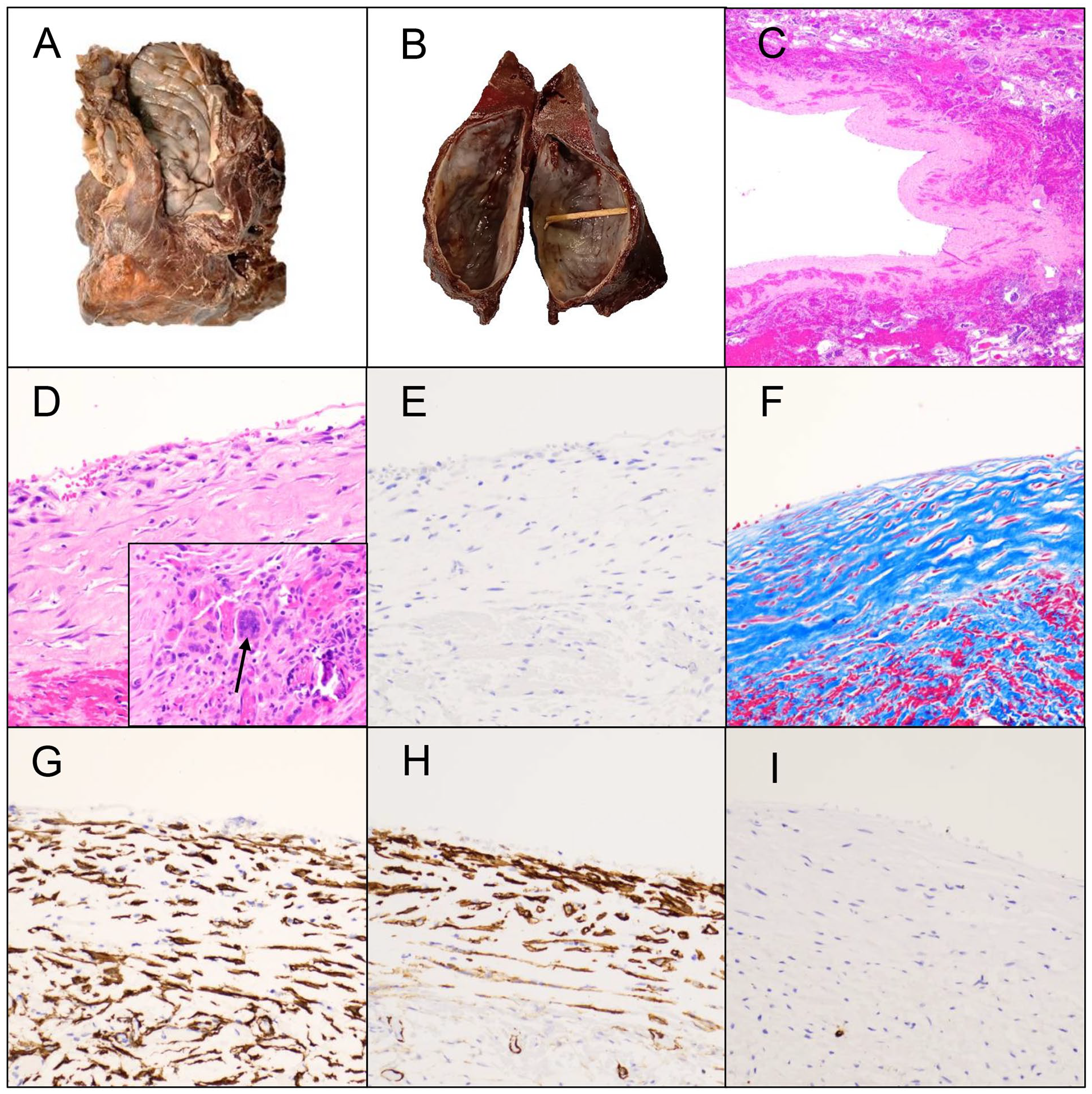
Uniloculated smooth walled cyst seen grossly for Case 1 (A) and Case 2 (B). The cyst showed variably thick walls (C: H&E 40X; D: H&E 100X) with no epithelial lining (E: CK 200X) and contained myofibroblastic tissue (F: Trichrome 200X; G: SMA 200x). Multinucleated giant cells were seen focally within the wall of Case 2 (D-arrow). There was aberrant expression of D2-40 in the myofibroblasts but no endothelial lining, confirmed with an additional vascular marker (H: D2-40 200X; I: CD34 200X). Total magnifications are listed.
At 19 months, the infant was well, but surveillance CXR revealed a new 1.8 cm left upper lobe cyst. The cyst remains static and proportionally smaller as he grows and remains asymptomatic at 2 years. A recent Ventilation-Perfusion (V/Q) scan showed minimal left lung perfusion, with the right lung accounting for 94%. Annual monitoring and imaging are planned, with pneumonectomy considered if significant growth or complications occur.
Case 2
The second infant boy was born at 35 weeks following a pregnancy complicated by premature rupture of membranes and associated oligohydramnios. His postnatal course was unremarkable, requiring only phototherapy for jaundice and no respiratory support. He exhibited subtle dysmorphic features, including micro-retrognathia and low-set ears. A Metabolic Genetics consultation attributed the micro-retrognathia to familial appearance, concluding no further genetic investigations were necessary. At 3 months, he was hospitalized for 3 weeks with community-acquired pneumonia, requiring a brief period of continuous positive airway pressure (CPAP) and antibiotics. His CXR revealed bilateral perihilar haziness and a small cystic lesion in the left lower region. A follow-up CXR at 6 months showed significant enlargement of the cystic lesion, now measuring 7.5 cm× 6.4 cm× 5.7 cm, favored to represent a pulmonary cyst. HRCT of the chest confirmed a large, thin-walled cystic lesion in the posterolateral left lower lobe. The lesion was air-filled, with no fluid or solid components, and showed no evidence of communication with the bronchial tree or systemic vascular supply. It displaced the pulmonary vasculature and abutted the pleura, though no deformity was noted in the adjacent ribs. The differential diagnosis included localized PPIE/pneumatocele, type I CPAM, and pleuropulmonary blastoma (PPB). Given the lesion’s stability and non-progressiveness, conservative management was chosen. However, a 3-month follow-up CXR showed persistence, prompting a left lower lobectomy. A large, densely adherent cyst was resected. At 10 months, he is doing well, growing, and no longer requiring respiratory support.
Pathologic examination of the cystic lesions showed features that were most consistent with localized PPIE/pneumatocele (Figure 3). Grossly, the lesion was a unilocular cyst (7.1 cm) with a smooth lining and no pleural connection. Microscopically, the cyst had a variably thick wall of hypocellular myofibroblastic tissue (SMA and trichrome positive), with no cells lining the inner surface (CK, D2-40, CD34 negative). Focal areas of multinucleated giant cells with a subset associated with small dystrophic calcifications were noted. Adjacent lymphoid aggregates and compressed lung parenchyma were present. No vascular malformation or CPAM features were seen, and the background lung showed no significant abnormality.
Discussion
Despite advances in antenatal screening, diagnosing cystic lung diseases in neonates and young children remains challenging due to the overlap in clinical and radiologic characteristics. Common congenital pulmonary cystic lesions include bronchogenic cysts, CPAM, and pulmonary sequestration, while conditions like CLE and bronchial atresia, though not true cystic lesions, can present with regional overinflation that mimics cystic changes on imaging.4,5
Primary cystic lung lesions in neonates and infants may be neoplastic or secondary to structural malformations, including lymphangioma, CPAM, Langerhans cell histiocytosis, respiratory papillomatosis, and pleuropulmonary blastoma (PPB). 6 Secondary causes include complications of infections, such as acute bacterial pneumonia leading to pneumatocele, cavitary necrosis, lung abscess, and bronchopleural fistula. 3 Air leaks, common in ventilated low-birth-weight preterm infants, are frequent mimics of cystic lung disease, with barotrauma increasing the risk of PPIE, especially in neonates requiring respiratory support. 7 Pulmonary infarcts can also result in necrotic formation of cysts. 8
Diagnosing pulmonary cystic lesions requires careful correlation of radiologic, clinical, and histopathologic findings. 9 Imaging modalities, including ultrasound, high-resolution computed tomography (HRCT), and magnetic resonance imaging (MRI), typically reveal well-circumscribed cystic lesions. Pulmonary cystic lesions on radioimaging have a broad differential. Localized PPIE/pneumatoceles, often caused by a bacterial infection or barotrauma, are thin-walled, air-filled cysts that usually resolve spontaneously. 10 CPAM may have solid components, bronchogenic cysts contain fluid, infectious abscesses show thick, irregular walls with air-fluid levels, and PPB exhibits aggressive growth with solid elements.11,12 Serial imaging is essential in distinguishing transient pneumatoceles from persistent localized PPIE, congenital lesions, or neoplastic lesions, with biopsy required when the diagnosis remains unclear.10,13 Despite the value of radiological imaging in neonates and young children, findings are often nonspecific and may require histological confirmation for a definitive diagnosis without the proper clinical context. 9
Additionally, in cases of uncertain radiological findings, surgery is crucial for histological examination to confirm the diagnosis, prevent infections, and mitigate the risk of malignant transformation, such as PPB. As noted in prior studies, histological evaluation provides definitive insights into the lesion’s nature and aids in planning further management strategies. 14
Surgical resection is preferred for both diagnostic and therapeutic purposes, with thoracoscopy or thoracotomy often required in young infants. Indications to resect are to eliminate risk of malignancy or infection.13,15 Incomplete resection risks recurrence, necessitating long-term postoperative monitoring. 16 In addition, due to potential for cystic lesion evolution and impact on lung growth, periodic follow-up with imaging is recommended to monitor residual lung changes. 17
Both infants presented with cystic lesions, likely localized PPIE/pneumatoceles, and were born to pregnancies complicated by oligohydramnios, which may impair pulmonary development. Case 1 showed imaging and histologic evidence of pulmonary hypoplasia, suggesting parenchymal fragility and susceptibility to interstitial disruption. Although no multinucleated giant cells were observed, alternative diagnoses were excluded morphologically and immunohistochemically. Given the lung’s overall susceptibility, an upper lobe cystic lesion may have formed similarly but was undetected until it enlarged. Alternatively, it could represent post-operative pulmonary infarction. Case 2 showed no lung disease or hypoplasia. Community-acquired pneumonia may have led to parenchymal destruction and air dissection into the interstitium due to CPAP ventilation.
In clinical practice, many pediatric surgeons recommend annual follow-up imaging once cysts stabilize in size, though this is based more on expert opinion than standardized protocols, with follow-up frequency and imaging modalities needing to be individualized. 1 Given the uncertainty of other lung lobe involvement in our patients, specifically in Case 1 with a hypoplastic lung and a new upper lobe cyst, both patients will undergo yearly CXR until age 5 to 6, then every 2 to 3 years to monitor lung changes during growth.
Conclusion
Cystic lung diseases in young children encompass a broad spectrum of clinical presentation and differential diagnosis, leading to a high potential for misdiagnosis. Accurate diagnosis requires a thorough clinical history, meticulous imaging, and, when indicated, surgical resection. This report highlights the challenges of relying solely on imaging for diagnosis, emphasizing the importance of a multidisciplinary approach. It also adds to the literature reporting these rare and atypical presentations of pulmonary cystic lesions, providing support for clinicians navigating similar cases in the future.
Footnotes
Data Availability
The datasets used and/or analyzed are available from the corresponding author on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval to report individual cases or case series when consent is provided by participants or, for pediatric patients, their legal guardians.
Informed Consent
The written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
