Abstract
Introduction:
Congenital cytomegalovirus (cCMV) is a common congenital viral infection. Testing for cCMV usually begins with assessing maternal CMV serology, specifically IgM and IgG antibodies. A negative maternal CMV IgM suggests a low risk of recent maternal CMV infection, thereby suggesting a low risk of cCMV in the fetus. Consequently, cCMV is often ruled out when maternal CMV IgM is negative.
Methods:
In our perinatal autopsy and placental pathology database, we identified 5 cases of cCMV despite negative maternal CMV IgM results in the second trimester.
Results:
In all 5 cases, fetal abnormalities were first detected by ultrasound in the second trimester, prompting maternal CMV testing. Since second trimester maternal CMV IgM was negative in all cases, cCMV was considered unlikely, thus precluding further prenatal CMV testing in 4 of these cases. The diagnosis of cCMV was subsequently made through placental and/or autopsy examinations. Following this diagnosis, retrospective CMV serology and IgG avidity testing was performed on stored frozen first-trimester maternal blood samples in 3 cases. Among these, the first-trimester samples in 2 cases were IgG+, IgM+, and exhibited low IgG avidity, suggesting a primary maternal CMV infection around the time of conception. In the third case, both first and second-trimester maternal blood samples were IgG+, IgM−, and showed high IgG avidity, suggesting a non-primary maternal CMV infection (i.e., reactivation or reinfection of CMV).
Conclusion:
A negative maternal CMV IgM in the second trimester cannot exclude cCMV infection. While CMV IgG avidity testing and analysis of stored frozen first-trimester maternal blood samples provide valuable insights, they have limitations. CMV PCR performed on amniotic fluid is a useful prenatal diagnostic tool. For cases of unexplained fetal abnormalities or death, autopsy and placental examination are recommended.
Keywords
Introduction
Congenital cytomegalovirus infection (cCMV) is one of the most common congenital viral infections worldwide,1-11 resulting from either primary or non-primary maternal CMV infection. A primary maternal CMV infection occurs when a pregnant woman with no prior exposure contracts the virus for the first time. In contrast, a non-primary infection occurs in a pregnant woman who has been previously exposed to the virus and contracts it again, either from the reactivation of a dormant strain or reinfection with a different strain. The risk of vertical CMV transmission to the fetus depends on the type of maternal infection. Primary maternal CMV infections carry a significantly higher risk, potentially affecting 30% to 40% of fetuses. 10 Meanwhile, non-primary infections have a much lower transmission rate, ranging from 1% to 3%.10,12
Infants with cCMV may exhibit a range of health complications, such as low birth weight, jaundice, hepatomegaly, splenomegaly, pancytopenia, pneumonia, seizures, hearing loss, and microcephaly. In its most severe manifestations, cCMV can be fatal.13-16 However, most infants born with cCMV show no symptoms at birth, although they may experience late sequelae, including neurocognitive delay, chorioretinitis, and delayed-onset hearing loss.1,15
Both the Society of Obstetricians and Gynecologists of Canada (SOGC) and the American College of Obstetricians and Gynecologists (ACOG) in the United States do not recommend routine maternal screening for CMV.13,17 Consequently, CMV screening is conspicuously absent from routine prenatal tests in both countries. For example, in Alberta, Canada, the recommended first-trimester screenings for expectant mothers include tests for human immunodeficiency virus (HIV), hepatitis B and C, syphilis, Neisseria gonorrhoeae, Chlamydia trachomatis, rubella immunity, and varicella immunity. 18 In the United States, the Centers for Disease Control and Prevention (CDC) recommend prenatal screenings for HIV, hepatitis B and C, and syphilis only. 19
Despite this, pregnant women may undergo CMV testing in certain clinical contexts.13,17 Testing may be considered for those presenting symptoms such as fever, cervical lymphadenopathy, sore throat, fatigue, and myalgia. Additionally, testing is advised if fetal anomalies—such as cerebral calcifications, microcephaly, cerebral ventriculomegaly, hepatomegaly, pericardial effusion, ascites, placentomegaly, fetal growth restriction, and hydrops—are observed during ultrasound examinations.20-24 SOGC recommends CMV testing for women considered to be at high risk, for instance, those with children under 3 years old living at home. However, this guideline is not always adhered to in practice.
While guidelines for maternal CMV laboratory testing vary by country, they share common elements. 25 Typically, maternal serum is screened for CMV IgG and IgM antibodies (referred to as CMV serology). If IgM is detected, CMV IgG avidity testing usually follows. When a woman tests positive for IgG but negative for IgM, further testing is often deemed unnecessary due to the low risk of recent or active CMV infection.26-29
CMV IgG avidity testing assesses the strength of the bond between CMV IgG antibodies and the virus. Low avidity indicates a recent or ongoing infection, while high avidity suggests a past infection. Although this test is generally performed when CMV IgM is positive to mitigate the risk of false positives, some health professionals advocate for performing CMV IgG avidity testing on all IgG-positive samples, regardless of IgM status.29,30 However, in clinical practice, CMV IgG avidity testing is not commonly carried out if IgM result is negative.
While most clinical guidelines suggest that a negative CMV IgM result in pregnant women generally rules out a recent or active maternal CMV infection, thus precluding a cCMV infection, several case reports have documented cCMV infections despite the absence of maternal CMV IgM antibodies.31-34 Our study adds to this body of literature by presenting 5 additional cases that demonstrate the same discrepancy. These cases underwent comprehensive prenatal assessments and postmortem fetal and/or placental examinations. We also explore the merits and limitations of CMV IgG avidity testing and CMV serologic testing of stored frozen first-trimester maternal blood samples.
Methods
This retrospective case series was approved by the University of Calgary Conjoint Health Research Ethics Board (REB14-2391).
Fetal and Neonatal Autopsies and Placental Examination
This case series is based on fetal and neonatal autopsies and placental pathologic examinations conducted at Alberta Children’s Hospital (Department of Pathology and Laboratory Medicine, University of Calgary and Alberta Precision Laboratories) between January 1, 2010, and May 31, 2023. The indications and protocols for autopsies and placental pathologic examinations at our institution are detailed in previously published works.35-38
Extraction of Fetal and Neonatal Autopsy Reports and Placental Pathology Reports
We retrieved fetal/neonatal autopsy and placental pathology reports within the aforementioned timeframe from the Cerner Millennium Laboratory Information System (North Kansas City, MO, USA).
To identify reports containing the word “cytomegalovirus” or “CMV,” a Perl script was used (performed by LdK). Reports identified through this process were then reviewed by a pediatric and perinatal pathologist (ESC) to confirm the diagnosis cCMV infection in the fetus/neonate or CMV infection in the placenta.
Maternal CMV Serology Data
We traced maternal CMV serology using the mothers’ Personal Health Numbers (PHNs) as listed on the fetal/neonatal autopsy reports or placental pathology reports. This study specifically focuses on cases where expectant mothers exhibited negative CMV IgM results during their second trimester.
Finally, we reviewed the clinical, radiological, and laboratory records of both the mothers and their fetuses/neonates. We assigned percentiles and normal ranges for placental weights using ranges previously published in the literature.39,40
Results
We identified 5 cases of cCMV infection where maternal CMV IgM tests were negative in the second trimester.
Case 1
A 20-year-old G2P0 woman, with a 10-year history of poorly controlled type 1 diabetes, presented for her first prenatal visit at 24 + 1 weeks of gestational age (GA). Serial prenatal ultrasounds from 24 to 30 weeks GA revealed progressive worsening of fetal and placental abnormalities. Sonographic findings from 24 to 26 weeks GA included intrauterine growth restriction, microcephaly, dolichocephaly, suspected corpus callosum agenesis, a small cerebellum, cardiomegaly with pericardial effusion, enlarged kidneys, persistent echogenic bowel, hepatomegaly, and a slightly thickened placenta. A fetal echocardiogram at 27 + 5 weeks GA demonstrated cardiomyopathy of undetermined etiology and moderate pericardial effusion, without apparent structural cardiac defects. By 29 + 1 weeks GA, the fetus exhibited cardiomegaly, an elevated cardiothoracic ratio, pericardial effusion, and hepatomegaly (Figure 1(a)-(c)). An MRI of the fetal brain at 29 + 5 weeks GA confirmed the agenesis of the corpus callosum and delayed cerebral sulcation. Fetal hydrops was also identified.
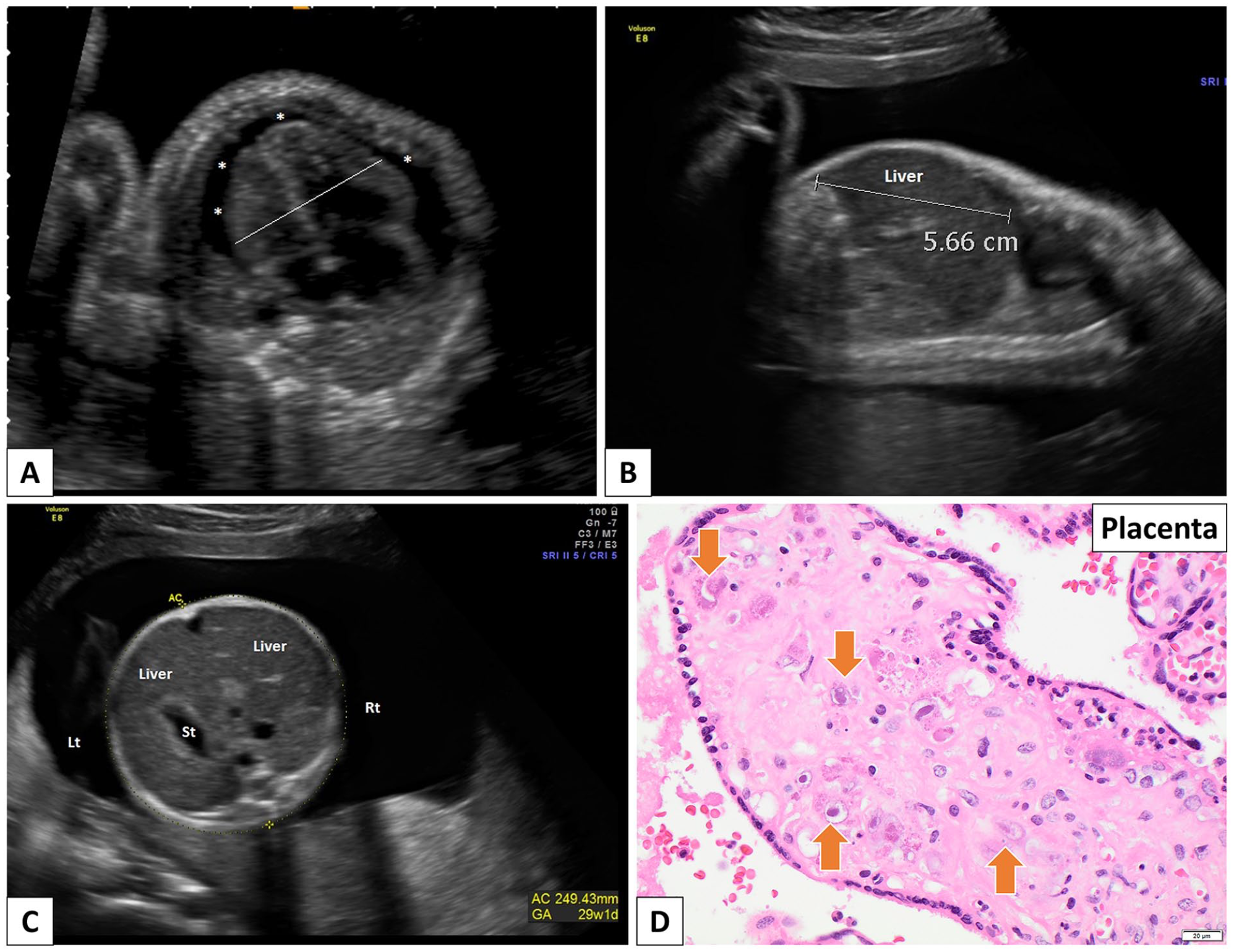
Case 1. (A-C) Antenatal ultrasound at 29 weeks 1 days GA. (A) Transverse view through the fetal chest. There is cardiomegaly present with a cardiothoracic ratio of 62%. A small pericardial effusion is present (*). (B) Parasagittal view through the fetal abdomen. There is hepatomegaly present. The right lobe of liver measures 5.66 cm (>95th percentile 53 ). (C) Transverse view through the fetal abdomen demonstrating the enlarged liver and no ascites. (D) Pathologic examination of the placenta following fetal demise at 30 weeks 3 days GA. Placental examination reveals many CMV inclusions (arrows) (hematoxylin and eosin stain).
After detecting these abnormalities via ultrasound, a maternal infectious workup was conducted at 25 weeks GA. This workup returned negative results. Specifically, maternal CMV IgG was found to be positive, while CMV IgM was negative. The mother received prenatal genetic counseling but declined invasive genetic testing. Given her history of suboptimal glucose control, diabetic embryopathy was considered as a possible cause of some of the fetal abnormalities. Fetal demise occurred at 30 + 3 weeks GA. Subsequently, the placenta was submitted for pathologic examination, but the parents did not consent to a fetal autopsy.
The placenta, weighing 609 g (>90th percentile), showed lymphoplasmacytic infiltrates in the chorionic villi (chronic villitis) and an increased number of nucleated red blood cells in the placental fetal capillaries. CMV-immunopositive inclusions were observed (Figure 1(d)), and polymerase chain reaction (PCR) confirmed CMV DNA in the placental tissue, leading to a diagnosis of cCMV infection.
Case 2
A 26-year-old G2P0 woman, with a history of 1 early spontaneous pregnancy loss and no significant medical history, underwent multiple prenatal ultrasounds between 11 and 23 weeks GA. The initial ultrasound at 11 + 1 weeks GA revealed no abnormalities. However, by 19 + 5 weeks GA, early signs of hydrops were noted, including skin edema, ascites, bilateral pleural effusions, and hepatomegaly (Figure 2(a) and (b)). Thoracentesis at 21 weeks GA revealed that the fetal pleural fluid was composed of 97% lymphocytes, suggesting a diagnosis of chylothorax. A fetal echocardiogram detected no abnormalities. By 21 + 6 weeks GA, the fetal hydrops had worsened (Figure 2(c) and (d)), leading to an amniocentesis and the placement of bilateral thoracoamniotic shunts. These interventions temporarily alleviated the pleural effusions. However, by 22 + 6 weeks GA, the left-sided shunt had displaced, resulting in a large left pleural effusion.
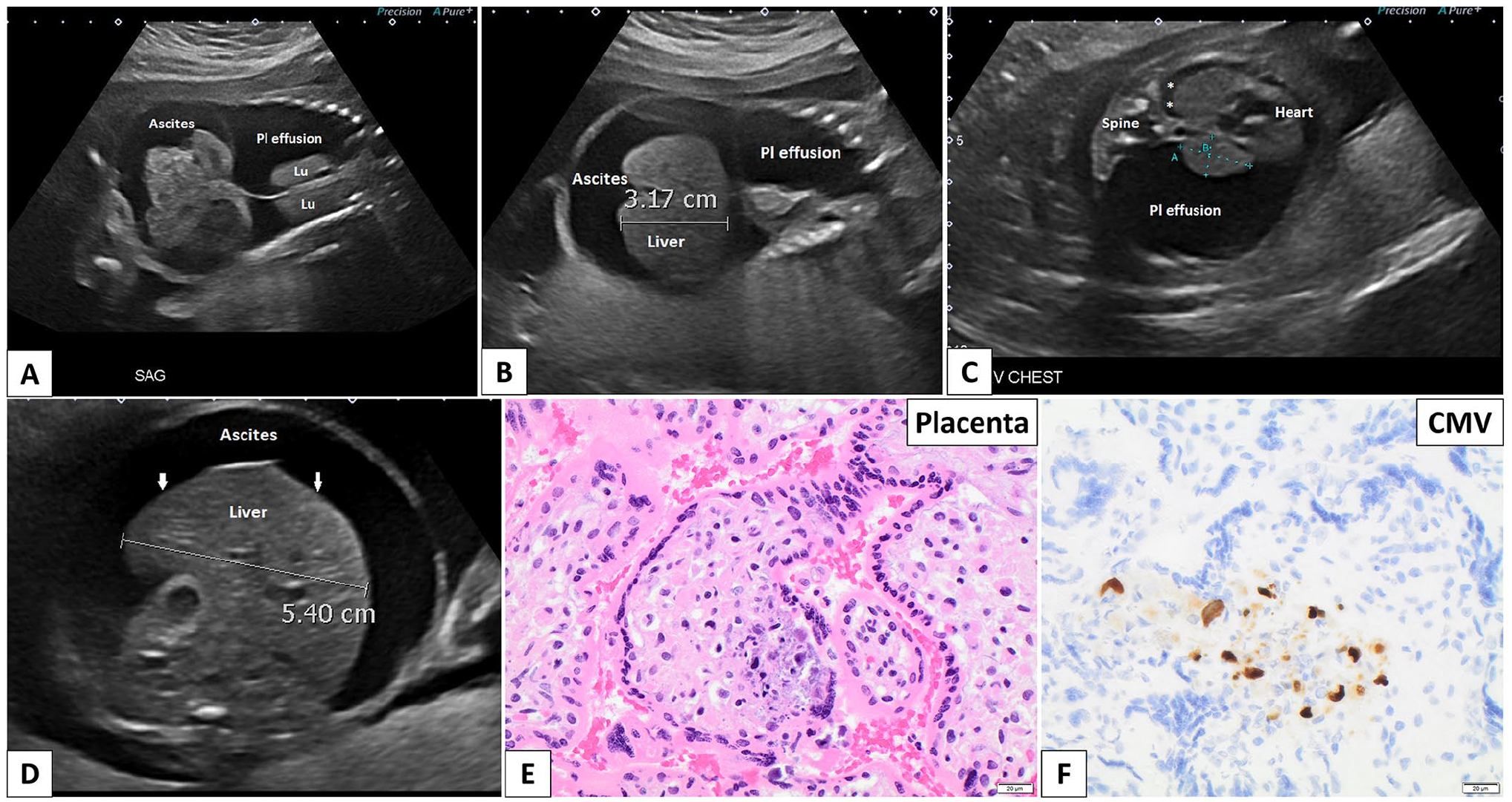
Case 2. (A and B) Antenatal ultrasound at 19 weeks 5 days. (A) Coronal image through the chest and abdomen demonstrating a large left and smaller right pleural effusion with secondary passive atelectasis of the lungs (Lu). Ascites is noted below the diaphragm. (B) Coronal image through the chest and abdomen demonstrating hepatomegaly—right lobe of liver measures 3.17 cm (95th percentile for current GA is 2.6 cm 53 ). A large left pleural effusion and ascites are present. (C and D) Antenatal ultrasound at 21 weeks 6 days GA. (C) Transverse view through the fetal chest demonstrating a large left pleural effusion, resulting in mediastinal shift of the heart and mediastinum to the right side of the chest. A small right pleural effusion is present (*). No pericardial effusion is present. (D) Transverse view through the fetal abdomen demonstrating an enlarged liver and ascites. Note the irregular liver surface (arrows). (E and F) Pathologic examination of the placenta following termination of pregnancy at 24 weeks 4 days GA. (E) The placenta reveals CMV inclusions (arrows) and calcifications (hematoxylin and eosin stain). (F) CMV immunohistochemistry highlights the viral inclusions (CMV immunostain).
At 21 + 1 weeks GA, the mother’s infectious workup revealed positive CMV IgG and negative IgM results. As part of the infectious workup, amniotic fluid collected at 21 + 6 weeks GA underwent CMV PCR testing, which returned a positive result. This was notable given the mother’s negative CMV IgM status. In light of these findings, CMV IgG avidity testing was performed on a blood sample collected from the mother at 21 + 1 weeks GA. The test showed low CMV IgG avidity, suggesting a recent or active maternal CMV infection.
Given the poor prognosis of cCMV infection in a fetus with multiple abnormalities, the parents elected to terminate the pregnancy at 24 + 4 weeks GA.
Pathologic examination of the placenta revealed that it was large (348 g; >90th percentile) and displayed areas of villous edema and patchy chronic (lymphoplasmacytic) villitis. The diagnosis of cCMV infection was confirmed with the detection of CMV-immunopositive inclusions in the placenta (Figure 2(e) and (f)). Rapid aneuploidy detection (RAD) performed on umbilical cord tissue found no evidence of aneuploidy for chromosomes 13, 18, 21, or the sex chromosomes. A fetal autopsy was not performed as no consent was provided.
Case 3
A 39-year-old G1P0 woman with hypothyroidism underwent routine prenatal ultrasounds. The dating ultrasound at 10 + 4 weeks GA and the first-trimester scan at 13 + 3 weeks GA showed no abnormalities. However, subsequent ultrasounds revealed concerning findings. At 19 + 2 weeks GA, mild ascites and an enlarged heart were observed in the fetus. By 19 + 3 weeks GA, the ascites had progressed to a moderate level, and an elevated middle cerebral artery peak systolic velocity (MCA-PSV)—suggestive of fetal anemia—was detected. The ultrasound at 20 + 2 weeks GA once again demonstrated moderate ascites, along with hepatomegaly and placentomegaly (Figure 3(a)-(c)), prompting an amniocentesis. A fetal echocardiogram at 21 + 1 weeks GA identified cardiomegaly but no structural cardiac abnormalities. At 21 + 2 weeks GA, an intrauterine transfusion (IUT) was performed with 15 mL of donor blood for fetal anemia, raising the fetal hemoglobin level from 76 L to 164 g/L. Additionally, thrombocytopenia was detected in the fetus, with a platelet count of 6.0 × 109/L. The fetal ascitic fluid exhibited a protein level of 14 g/L (reference range: <25 g/L). Imaging review revealed that an intracranial hemorrhage had occurred before the initiation of the IUT (Figure 3(d)).
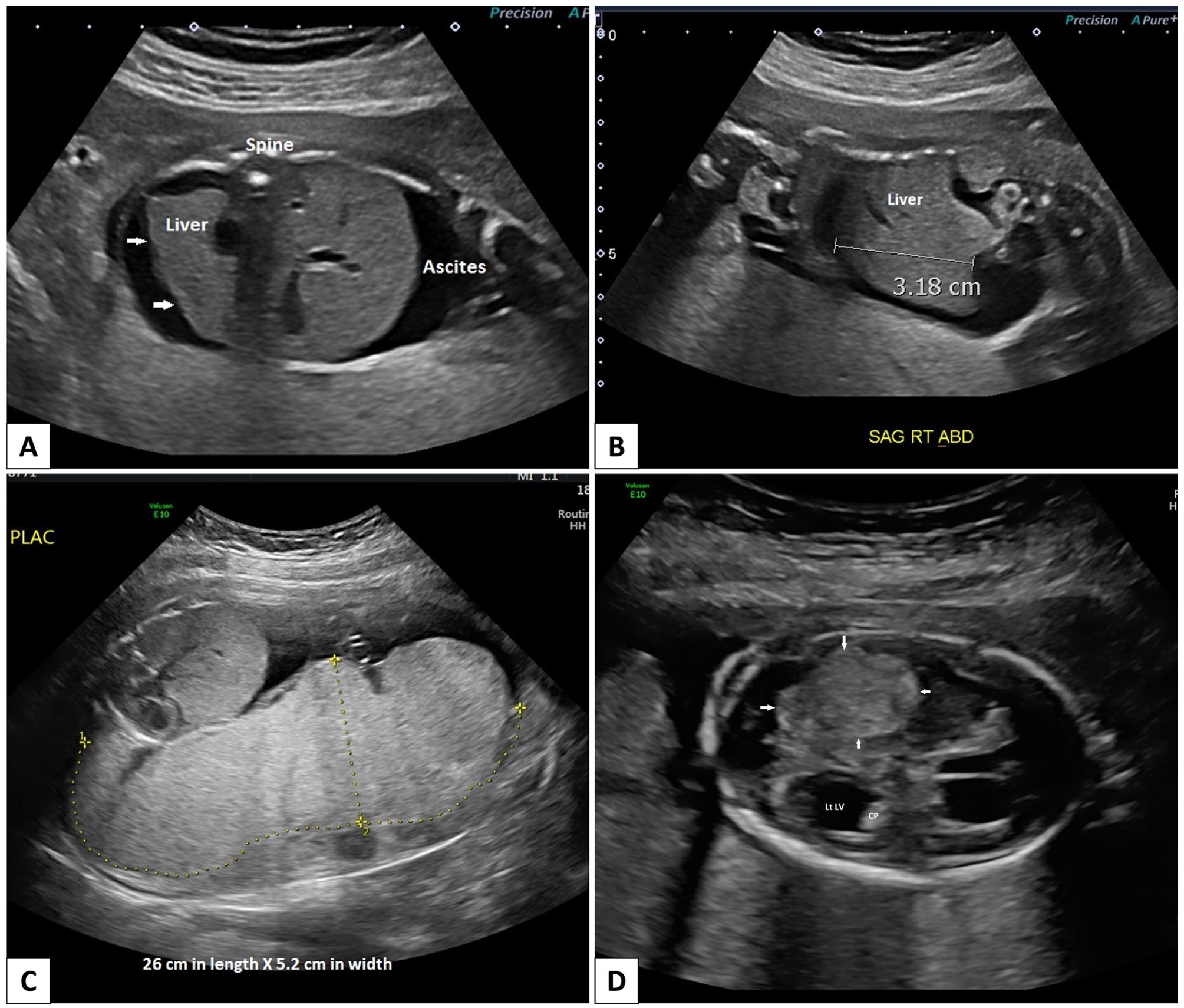
Case 3. (A-C) Antenatal ultrasound at 20 weeks 2 days GA. (A) Transverse view through the fetal abdomen demonstrating ascites. Note the irregular liver surface (arrows). (B) Parasagittal view through the fetal abdomen. There is no hepatomegaly present. Right lobe of liver measures 3.18 cm (95th percentile for current GA is 3.4 cm 53 ). (C) Placentomegaly is present. Sagittal view of the placenta measures 26 cm in length and 5.2 cm in width. The placental architecture is homogeneous. (D) Antenatal ultrasound at 21 weeks 2 days. Transverse view through the fetal central nervous system demonstrating a large echogenic mass in the right parietal lobe (between arrows) that is avascular and non-calcified consistent with a large acute hemorrhage. There is mild contralateral dilatation of the trigone and occipital horn of the left lateral ventricle (LtLV) with a dangling choroid plexus (CP).
Following the ultrasound abnormalities observed at 19 + 3 weeks GA, a comprehensive infectious workup was initiated, which included maternal CMV serology. The mother tested positive for CMV IgG but negative for CMV IgM. The rest of the infectious workup was also negative. Consequently, the expectant mother was referred for a Medical Genetics consultation. Genetic testing, including RAD and chromosomal microarray (CMA), conducted on the amniotic fluid, showed negative findings. Given the negative RAD and CMA results, whole exome sequencing with copy number variation detection was ordered to further investigate possible genetic causes of the fetal abnormalities. The amniotic fluid was not tested for infections.
Given the poor prognosis associated with fetal intracranial hemorrhage, the parents decided to terminate the pregnancy at 22 weeks GA. They consented to a complete fetal autopsy, which included a placental examination.
The autopsy revealed a moderately macerated, phenotypically male fetus with measurements corresponding to the stated gestational age of 22 weeks. Several external abnormalities were noted, including scalp edema, a distended abdomen (Figure 4(a)), and bilateral talipes equinovarus. Internal examination revealed peritoneal, pericardial, and pleural effusions. The heart appeared mildly globular but was otherwise anatomically normal. The cardiothoracic ratio was mildly elevated. Other findings included bilateral pulmonary hypoplasia, hepatomegaly, splenomegaly, and a Meckel’s diverticulum. Microscopically, an increased number of nucleated red blood cells in the fetal vasculature suggested fetal anemia. Numerous CMV-immunopositive viral inclusions were found in multiple fetal organs, including the thyroid glands, lungs, liver, adrenal glands, and kidneys (Figure 4(c)). CMV PCR testing of fetal kidney tissue was positive. Neuropathologic examination revealed CMV meningoencephalitis and an intraventricular hematoma in the right hemisphere.
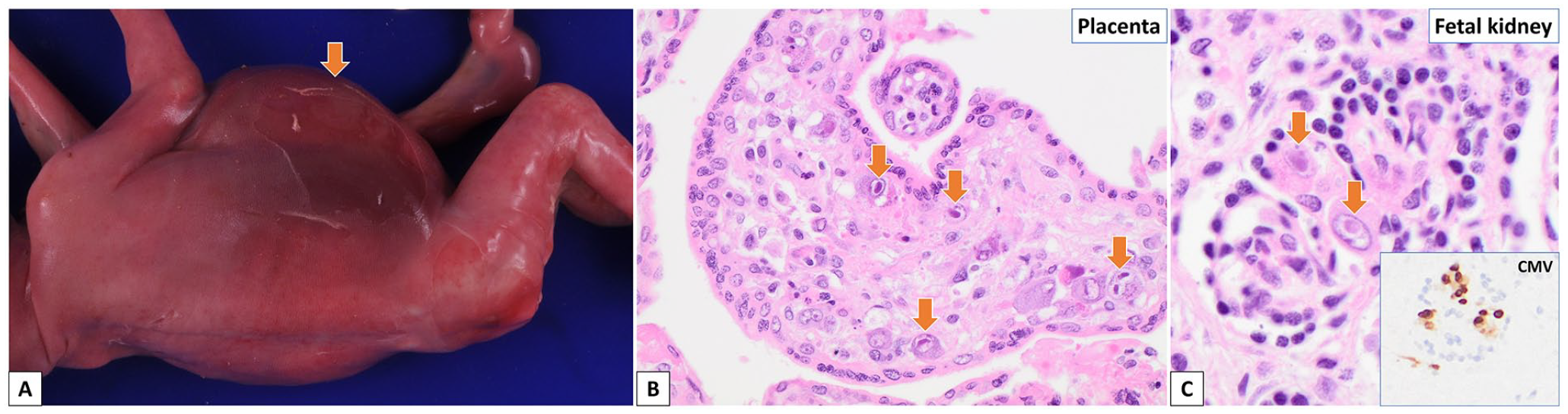
Case 3. (A-C) Fetal autopsy and placental examination at 22 weeks GA. (A) The fetus has a massively distended abdomen (arrow). (B) Many CMV inclusions (arrows) are noted in the chorionic villi of (hematoxylin and eosin). (C) CMV inclusions (arrows) are also identified in the fetal organs (hematoxylin and eosin). Insert shows CMV-immunopositive inclusions (CMV immunostain).
The placenta, weighing 316 g (>90th percentile), exhibited extensive villous edema, increased fetal nucleated red blood cells, and calcifications. Chronic (lymphoplasmacytic) villitis was observed, along with numerous CMV-immunopositive inclusions within the chorionic villi (Figure 4(b)). These autopsy and placental pathologic findings were consistent with cCMV infection.
The diagnosis of cCMV was unexpected, considering the negative maternal CMV IgM test at 19 + 3 weeks GA. This discrepancy led to additional maternal laboratory testing following the fetal autopsy. A blood sample from the mother collected at 19 + 3 weeks GA was tested for CMV IgG avidity, showing low avidity, indicative of a recent maternal CMV infection. Furthermore, a stored frozen blood sample from the mother, collected at 11 + 4 weeks GA, underwent CMV serology testing. The results were positive for both CMV IgG and IgM, with low IgG avidity. Of note, in Calgary, Alberta, Canada, first-trimester maternal blood samples are typically stored for 6 to 18 months. The paired (first-trimester and second-trimester) CMV serology results suggested that the mother likely acquired a primary CMV infection around the time of conception.
Case 4
A 33-year-old G4P2 woman, who had been receiving adequate prenatal care, presented at the hospital at 26 + 2 weeks GA with a breech presentation and suspected placental abruption. After receiving a dose of betamethasone, she underwent a cesarean section and delivered a baby girl. Of note, serial antenatal ultrasounds had shown no significant findings, except for a minor increase in fetal abdominal circumference at the detailed anatomy scan at 20 + 3 weeks GA.
The newborn weighed 0.86 kg (50th-90th percentile) and had a head circumference of 22.5 cm (10th-50th percentile). She exhibited signs of perinatal depression, with APGAR scores of 2, 5, and 5 at 1, 5, and 10 minutes, respectively. Diagnosed with acute liver failure and thrombocytopenia, she was transferred to the neonatal intensive care unit. Neonatal herpes simplex infection was considered a possible cause for the neonate’s symptoms, and prophylactic acyclovir was administered. Unfortunately, her condition continued to deteriorate, and imaging revealed cerebellar hemorrhage. She died on day 5 of life. Newborn metabolic screening and CMA were negative.
Extensive premortem laboratory tests, including blood cultures, serology, and nucleic acid tests for various infections, all yielded negative results. CMV serology was not performed, but the urine CMV PCR result was pending at the time of her death. The mother consented to a full autopsy. The placenta was not available for pathologic examination.
Postmortem examination revealed a jaundiced, premature female neonate with mild abdominal distension and yellow discoloration of most organs (Figure 5(a)). Both the liver and spleen were enlarged. The gallbladder, although normally formed, contained clear mucoid fluid without bile pigments, suggesting a possible biliary tract blockage. Cerebellar hemorrhage was observed.
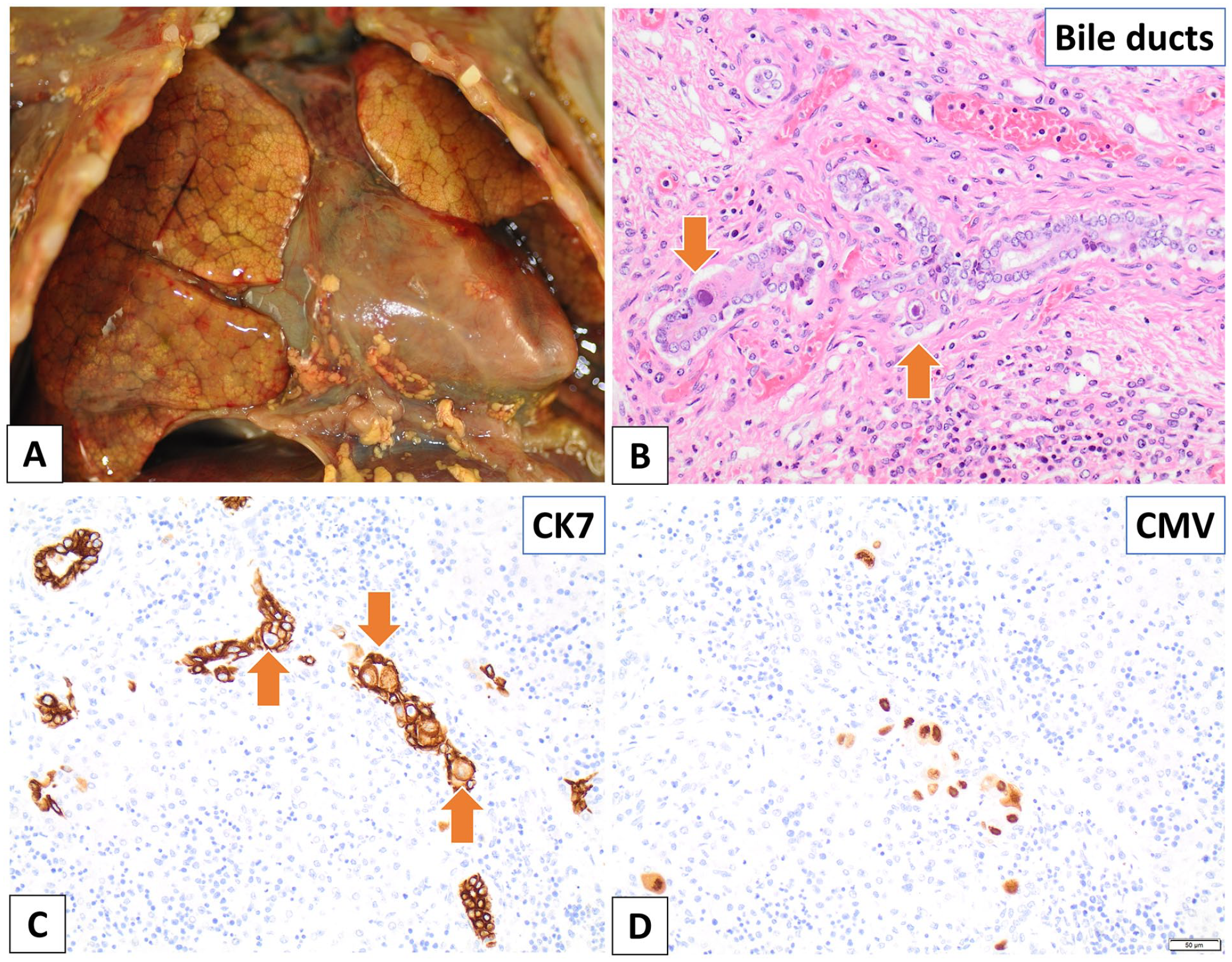
Case 4. (A-D) Autopsy of the premature neonate, who was born at 26 weeks 2 days GA and died on the fifth day of life. (A) The lungs and heart show yellow discoloration, consistent with jaundice. (B) CMV inclusions are noted in many organs, including within the bile duct epithelium (hematoxylin and eosin). (C) CK7 immunostain highlights the bile duct epithelium. Note the large viral inclusions (arrows). (D) CMV-immunopositive inclusions are found within the bile duct epithelium (CMV immunostain).
Microscopically, multiple organs, including the thyroid glands, thymus, heart, lungs, liver, spleen, adrenal glands, and kidneys, showed CMV-immunopositive inclusions (Figure 5(b)-(d)). CMV PCR performed on postmortem tissues (thymus, heart, lung, liver, and kidney) and premortem urine was positive. The findings were consistent with disseminated cCMV infection.
Following the autopsy’s diagnosis of disseminated cCMV infection, the mother’s blood sample (collected at 26 + 2 weeks GA) was tested for CMV serology, which showed positive IgG, negative IgM, and high IgG avidity. Additionally, the mother’s stored first-trimester blood sample (collected at 9 + 5 weeks GA) was analyzed, revealing positive CMV IgG, positive IgM, and low IgG avidity. These paired CMV serology results suggest that the mother likely acquired a primary CMV infection around the time of conception.
Please note that while this case has been previously published, the discussions in the previous publications did not specifically address cCMV infection in the context of a negative maternal CMV IgM result. Instead, the focuses of the earlier publications were on exploring cCMV as a potential cause of extrahepatic biliary atresia 41 and the association of cCMV with congenital rickets. 42
Case 5
A 29-year-old G1P0 healthy woman was diagnosed with an intrauterine fetal demise (IUFD) at 20 + 1 weeks GA. The dating ultrasound at 7 + 3 weeks GA and the first-trimester scan at 12 + 3 weeks GA revealed no fetal abnormalities, and screenings for trisomy 13, 18, and 21 indicated low risk. However, at 19 + 3 weeks GA, the fetus was found to be small for gestational age, with composite measurements corresponding to 18 + 2 weeks GA. The fetal head had a circumference 2.7 standard deviations below the mean and exhibited signs of dolichocephaly. Although the cerebellum was normally shaped, it was smaller than expected. The fetus also showed moderate to severe ascites and hepatomegaly (Figure 6(a)-(d)), and the placenta was notably thick. The MCA-PSV was 35 cm/s, or 1.41 MoM, indicating a low risk for moderate or severe anemia. IUFD was diagnosed at 20 + 1 weeks GA. An infectious workup yielded negative results, with maternal CMV IgG testing positive and CMV IgM negative. The mother consented to a complete fetal autopsy.
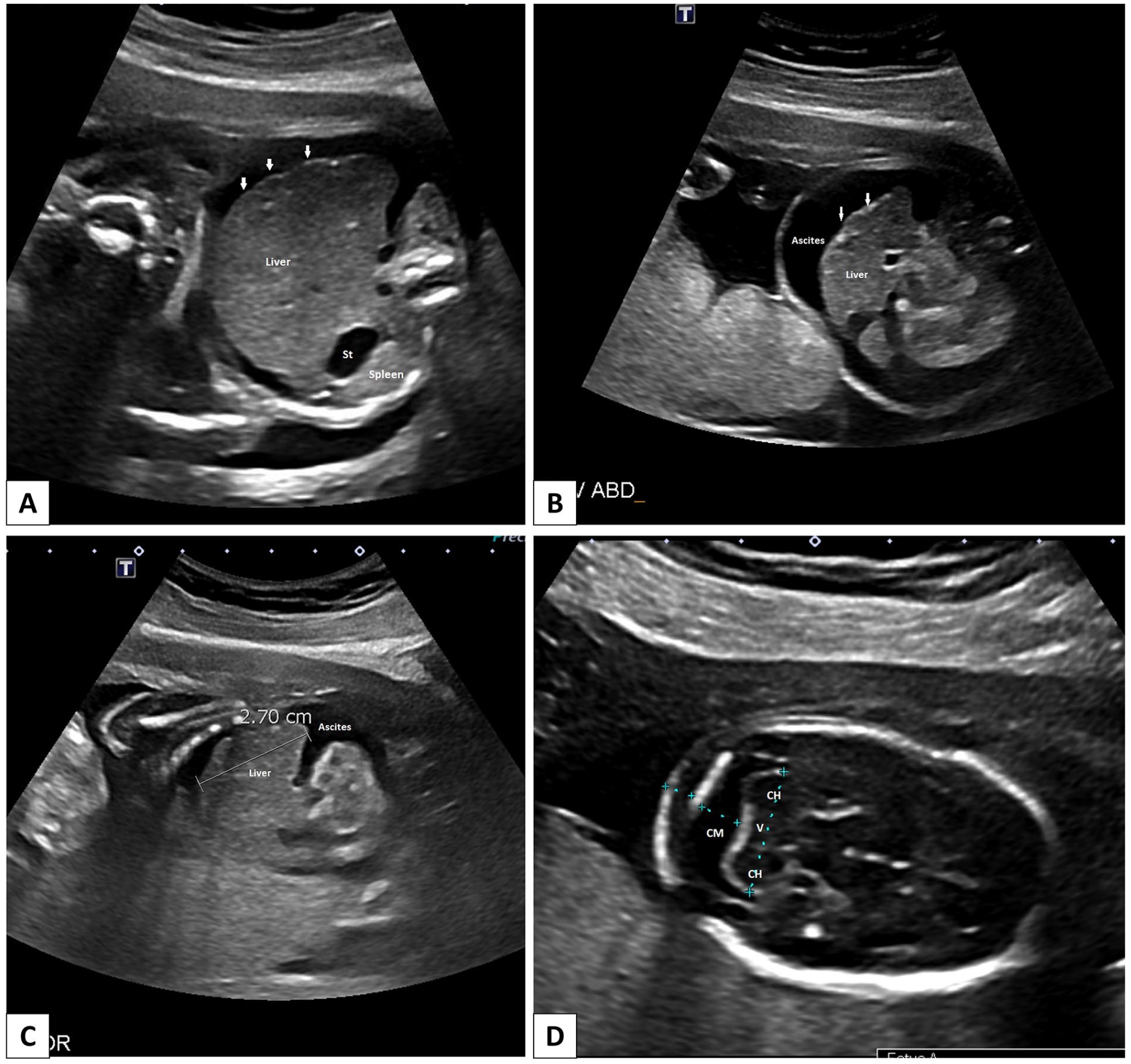
Case 5. (A-D) Antenatal ultrasound at 19 weeks 3 days GA. (A and B) Transverse view through the fetal abdomen demonstrating the enlarged liver with an irregular surface contour (arrows) and ascites. The spleen is not enlarged. (C) Parasagittal view through the fetal abdomen. There is hepatomegaly present (2.7 cm). (D) Transverse view through the fetal CNS at the level of the posterior fossa demonstrating a small cerebellum measuring 16.8 mm (below fifth percentile for current GA). The cerebellum appears morphologically normal.
The autopsy revealed a moderately macerated, phenotypically female fetus with intrauterine growth restriction and fetal hydrops. The fetus exhibited microcephaly, dolichocephaly, hypertelorism, a flat nose bridge with a flattened nose tip, and low-set ears. Internal examination revealed hepatomegaly with punctate calcifications (Figure 7(a)), splenomegaly, and a slightly globular and enlarged heart. Signs of fetal anemia were observed, evidenced by pale organs and an increased number of nucleated red blood cells in the fetal blood vessels. CMV-immunopositive inclusions were identified in nearly all organ tissues (Figure 7(b) and (c)), and CMV DNA was detected in the lungs, kidneys, liver, and spleen through PCR testing.
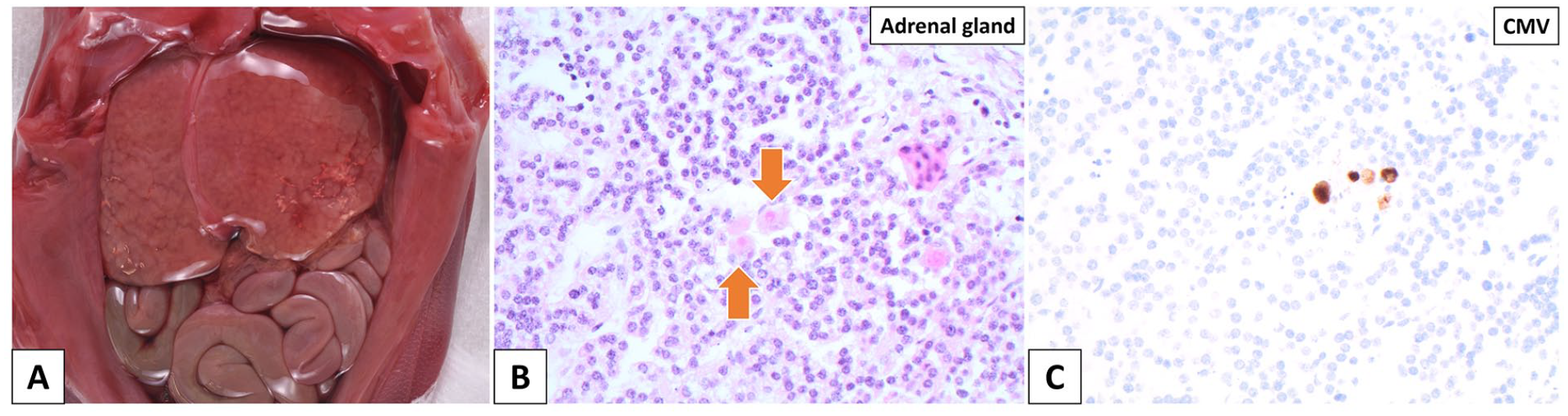
Case 5. Fetal autopsy—demise at 20 weeks 1 day GA. (A) The fetus has hepatomegaly with calcifications evident on the liver surface. (B) CMV inclusions are present in the adrenal gland (hematoxylin and eosin stain). (C) CMV-immunopositive inclusions identified in the adrenal gland (CMV immunostain).
RAD on the umbilical cord found no aneuploidy for chromosomes 13, 18, 21, or the sex chromosomes. The placenta weighed 168 g (reference range: 116.00 ± 22.98) and showed CMV-associated chronic villitis and intervillositis. There was also evidence of maternal vascular malperfusion, including retroplacental hemorrhage with associated hemosiderin deposition, as well as decidual arteriopathy, characterized by atherosis and fibrinoid necrosis.
A diagnosis of disseminated cCMV infection was rendered based on the autopsy and placental examinations. In light of these findings, further CMV serologic tests were conducted on the mother’s blood samples. The maternal blood sample collected at 20 + 1 weeks GA showed high CMV IgG avidity. Furthermore, a stored frozen maternal blood sample from 5 weeks GA tested positive for CMV IgG, negative for IgM, and exhibited high IgG avidity. The paired CMV serology results from the first and second trimesters suggested that the mother had a non-primary CMV infection during pregnancy.
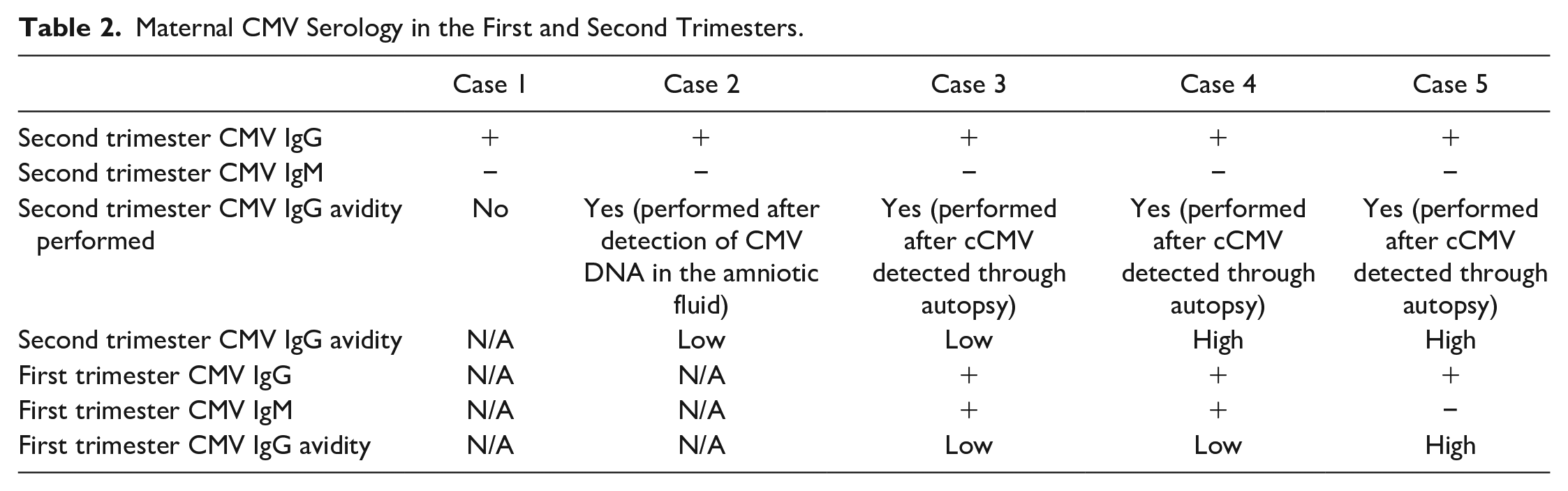
A summary of the findings from these 5 cases is presented in Table 1. A summary of maternal CMV serology is presented in Table 2.
Key Characteristics of cCMV Cases Analyzed in This Study.
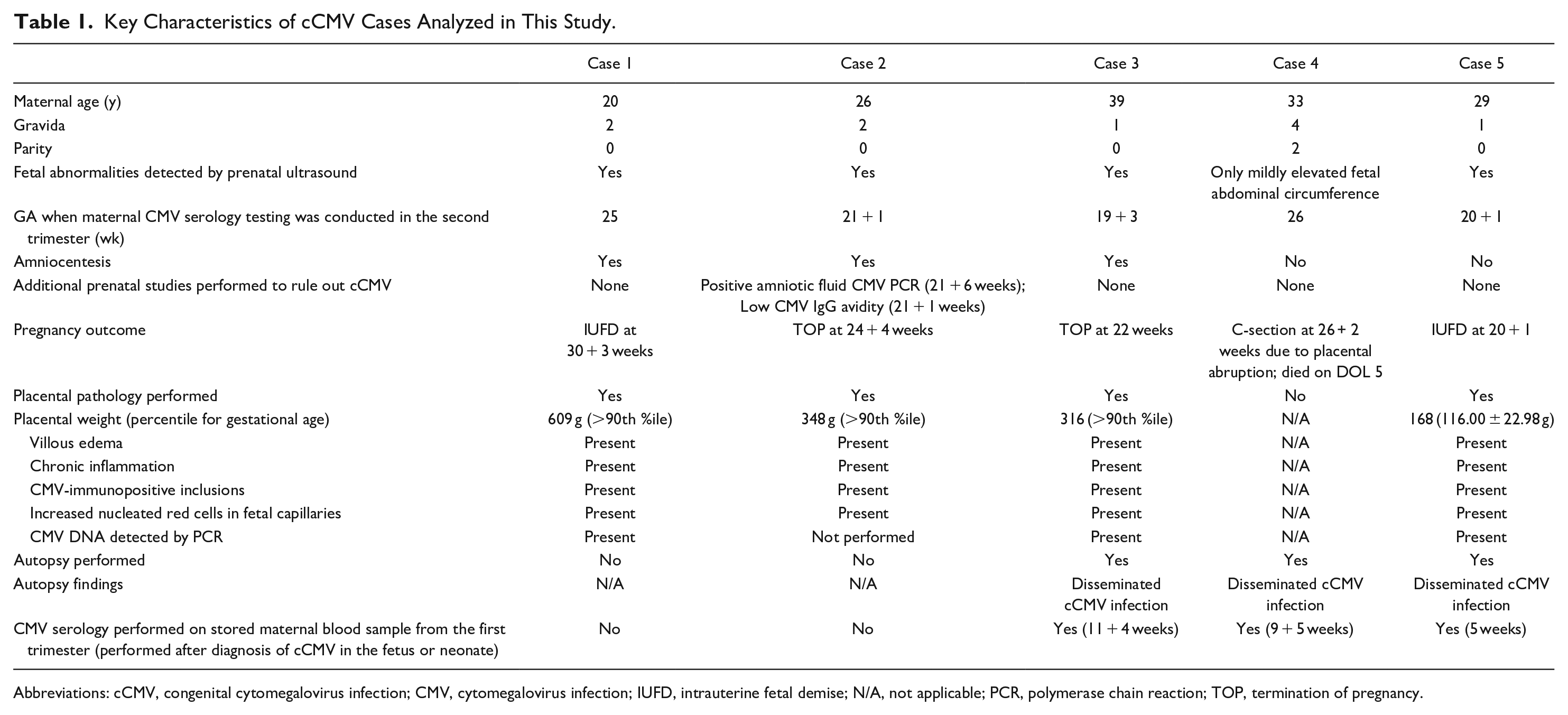
Abbreviations: cCMV, congenital cytomegalovirus infection; CMV, cytomegalovirus infection; IUFD, intrauterine fetal demise; N/A, not applicable; PCR, polymerase chain reaction; TOP, termination of pregnancy.
Maternal CMV Serology in the First and Second Trimesters.
Discussion
This study presents 5 cases of fetal and neonatal demise associated with congenital CMV (cCMV) infection, where the mothers tested negative for CMV IgM during the second trimester. Despite CMV being globally recognized as one of the most common congenital viral infections, routine screening for CMV among pregnant women is not a standard practice. The reasons for this include the high prevalence of prior CMV infections (over half of adults in US have been infected by age 40 years), challenges in interpreting serology results, limited treatment options, risks associated with unwarranted interventions, and concerns regarding the cost-effectiveness of universal screening.27,43-45
CMV infections during pregnancy can lead to adverse outcomes. A primary CMV infection in expectant mothers carries a significantly higher risk of vertical transmission compared to non-primary infections. Thus, accurately diagnosing primary CMV infection in pregnant women is essential. In clinical practice, maternal CMV testing is recommended when a pregnant woman exhibits symptoms such as fever, myalgia, and lymphadenopathy, or when fetal abnormalities are detected via ultrasound. The primary diagnostic approach involves CMV serologic assays, which include tests for CMV IgG and IgM, as well as an assessment of CMV IgG avidity.
A positive CMV IgG test in a pregnant woman indicates past exposure to CMV but does not specify the timing of the infection. Following a primary CMV infection, CMV IgM antibodies typically peak within 1 to 3 months and generally decline to undetectable levels within a year. Although a positive CMV IgM result often suggests a recent or ongoing primary CMV infection, it is not definitive; CMV IgM can appear during non-primary infections46,47 and may persist in some individuals long after the initial infection.48,49 Therefore, when a pregnant woman tests positive for CMV IgM, CMV IgG avidity testing is commonly performed to confirm a recent or active primary infection. CMV IgG avidity testing assesses the strength of the bond between IgG antibodies and the virus. Following a primary infection, IgG antibodies initially show low binding strength (low avidity), which transitions to high binding strength (high avidity) within 2 to 4 months50-52 (Figure 8).
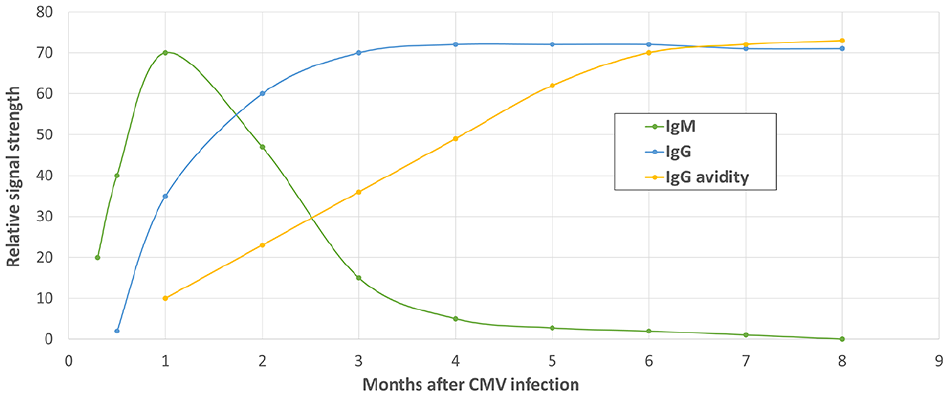
Changes in CMV IgM, IgG, and IgG avidity levels over time following a primary CMV infection. Upon a woman’s first exposure to CMV (primary infection), CMV IgM (green line) levels rise rapidly but also decline quickly. Imagine a situation where a woman acquires a primary CMV infection just before conception. In such a scenario, her IgM might be negative in the second trimester, even if vertical transmission of CMV from the mother to the fetus has already occurred. CMV IgG avidity (yellow line) testing can estimate the duration since the infection: a low-avidity result suggests a recent primary infection, while a high-avidity result indicates the primary infection was likely more than 6 months before.
When maternal CMV serology testing reveals positive IgG and negative IgM, this typically indicates that a recent or ongoing primary CMV infection in the mother is unlikely, thereby suggesting a low likelihood of cCMV infection in the fetus. However, instances of cCMV infection occurring in the fetus or neonate have been previously reported despite negative maternal CMV IgM results.31-34 One such case, 33 reported by an author (ESC) of this study, was identified at a different hospital and is therefore not included in this series. It is important to emphasize that CMV IgM levels can peak and then decline rapidly. Therefore, if a mother contracts CMV early in her pregnancy, her CMV IgM levels might become undetectable by the time she undergoes CMV serology testing in the second trimester, which is when fetal abnormalities associated with cCMV first become detectable by ultrasound.
In the current study, we searched our pathology database for reports of placentas and autopsies with cCMV infections. We then examined the mothers’ CMV serology results, specifically focusing on cases with negative maternal CMV IgM in the second trimester. Over a 13.5-year period, only 5 cases met our inclusion criteria, highlighting the rarity of such cases or the infrequency with which they are identified.
The mothers in our case series were aged between 20 and 39 years, with 3 being multigravida and 2 primigravida. For all cases where dating and first-trimester ultrasounds were performed, no fetal abnormalities were observed in these initial ultrasounds. However, by the time of the second trimester anatomy scan, fetal abnormalities were apparent in all 5 cases. Four of these cases presented significant fetal abnormalities, while 1 case (Case 4) showed a minor abnormality–a slightly increased abdominal circumference. In those 4 cases with notable ultrasound abnormalities, the mothers underwent an infectious disease workup, which included CMV serology testing.
Following the results of the second-trimester maternal CMV serology tests, which showed positive CMV IgG and negative CMV IgM in all 5 cases, additional prenatal CMV testing was undertaken in just 1 case (Case 2). Specifically, in Case 2, CMV PCR testing on the amniotic fluid revealed the presence of CMV DNA. For the remaining 4 cases in the current series, a diagnosis of cCMV infection was only established after the demise of the fetus or neonate, through either autopsy or pathologic examination of the placenta.
Additional maternal laboratory tests were conducted in 3 of the 5 cases (Cases 3, 4, and 5) after a diagnosis of cCMV was established through fetal/neonatal autopsy or placental examination. The mothers’ blood samples from the second trimester were analyzed for CMV IgG avidity. Furthermore, the mothers’ stored, frozen first-trimester blood samples underwent testing for CMV IgG, IgM, and CMV IgG avidity, as detailed in Table 1. Therefore, in these cases (Cases 3 to 5), we obtained paired maternal CMV serology results from the first and second trimesters.
In Case 3, the second-trimester maternal CMV serology results were as follows: positive IgG, negative IgM, and low IgG avidity. The first-trimester maternal CMV serology (tested on a stored frozen sample) revealed positive IgG, positive IgM, and low IgG avidity. These results suggest that the mother likely contracted primary CMV infection during the first trimester or preconception.
In Case 4, the second-trimester maternal CMV serology results were as follows: positive IgG, negative IgM, and high IgG avidity. The first trimester maternal CMV serology (tested on a stored frozen sample) showed positive IgG, positive IgM, and low IgG avidity. These findings suggest primary maternal CMV infection either in the first trimester or preconception.
In Case 5, the second-trimester maternal CMV serology showed positive IgG, negative IgM, and high IgG avidity. The first trimester maternal CMV serology (tested on a stored frozen sample) showed positive IgG, negative IgM, and high IgG avidity. These consistent findings across trimesters suggest the mother had a CMV infection prior to the pregnancy, implying a potential case of non-primary infection, as opposed to a primary maternal infection.
Despite the modest size of our dataset on first- and second-trimester CMV serology results, it offers significant insights. Current guidelines typically suggest performing the CMV IgG avidity test only when CMV IgM is positive, aiming to confirm a recent maternal CMV infection. Performing the CMV IgG avidity test is generally not recommended if the maternal CMV IgM result is negative. However, our findings suggest that the CMV IgG avidity test may have broader applicability. For instance, Cases 2 and 3 demonstrated low CMV IgG avidity despite negative CMV IgM tests in the second trimester. The low IgG avidity results could have alerted healthcare providers to a potential recent or active maternal CMV infection. Therefore, we recommend considering CMV IgG avidity testing for pregnant women whose second-trimester anatomy scans reveal fetal abnormalities, irrespective of maternal CMV IgM status.
However, it is important to acknowledge the limitations of CMV IgG avidity testing in the second trimester. While low IgG avidity may indicate a recent or active maternal CMV infection, high IgG avidity does not conclusively rule out a maternal CMV infection early in pregnancy. For example, if a mother contracts CMV around the time of conception, the fetus might be infected during the early stage of pregnancy. By the second trimester, when fetal anomalies are identified via ultrasound, maternal CMV IgG may already show high avidity to the virus, as observed in Case 4. Therefore, using high IgG avidity results from the second trimester alone to rule out cCMV could be misleading.
Moreover, Case 5 illustrates that high CMV IgG avidity, as expected in women with prior CMV exposure, does not preclude the possibility of a non-primary CMV infection in the mother or cCMV infection in the fetus.
Our findings highlight the value of analyzing stored first-trimester maternal blood samples. In Cases 3 and 4, these samples, when tested after fetal autopsies confirmed cCMV, showed positive CMV IgM and low CMV avidity, indicating a potential recent or active primary maternal CMV infection early in pregnancy. However, it is important to note that CMV serology testing on stored first-trimester maternal blood samples might not definitively exclude the possibility of cCMV infection, as demonstrated by Case 5 (a case of non-primary maternal CMV infection during pregnancy). In this instance, the first trimester maternal blood sample showed negative CMV IgM and high CMV avidity, which could have misleadingly suggested the absence of cCMV if solely relied upon for diagnosis.
In our case series, 3 mothers (Cases 1, 2, and 3) had amniocentesis for genetic testing after ultrasound detected fetal anomalies. In contrast, the mothers in Cases 4 and 5 did not undergo this procedure. The decision was due to no notable fetal anomalies in Case 4 and an IUFD shortly after sonographic detection of fetal anomalies in Case 5, leaving no time for further testing. Of note, while amniotic fluid was available in 3 cases (Cases 1, 2, and 3), only Case 2 had CMV PCR testing on the amniotic fluid. This was the only case in this case series with a prenatal cCMV diagnosis. For the other 4 cases, cCMV was diagnosed post-fetal/neonatal death through autopsy or placental examination. This emphasizes the value of CMV PCR testing on amniotic fluid when amniocentesis is done due to detected fetal anomalies.
This study highlights the limitations of CMV serology, including CMV IgM and IgG avidity, in diagnosing cCMV infection. The transient nature of the CMV IgM response, which can peak and decline rapidly to undetectable levels within a few months, means that a negative maternal CMV IgM result does not conclusively exclude a CMV infection during pregnancy. Consequently, if a woman contracts CMV just before or in the early stages of pregnancy, IgM levels might drop to undetectable by the time second-trimester prenatal ultrasounds detect fetal anomalies.
While CMV IgG avidity testing and the analysis of stored first-trimester maternal blood samples can offer additional insights in some instances, their inherent limitations must be acknowledged. Low CMV IgG avidity in the second trimester can suggest a recent or ongoing maternal infection, but high avidity does not definitively rule out a maternal CMV infection in early pregnancy that could have already been transmitted to the fetus. Moreover, high CMV IgG avidity cannot categorically exclude non-primary maternal infections, as women with previous CMV exposure would typically show high CMV IgG avidity.
The testing of stored first-trimester maternal samples can sometimes be informative. A positive CMV IgM result from the first trimester suggests an active or recent infection early in pregnancy. However, a negative CMV IgM result does not necessarily preclude a recent infection.
In conclusion, although maternal CMV serology is a standard test to screen for CMV infection during pregnancy, our case series underscores its limitations in diagnosing or ruling out cCMV infection. When fetal abnormalities like pleural or pericardial effusions, ascites, or hydrops are observed in the second trimester by ultrasound, clinicians should consider CMV PCR testing on amniotic fluid. Should fetal demise occur before amniotic fluid can be collected, both the fetus and placenta should undergo pathologic examination to determine the cause of death. Accurate diagnosis of cCMV is crucial to avoid unnecessary tests and procedures. Furthermore, women with active CMV infections are often recommended to wait 6 to 12 months before trying to conceive again.
Footnotes
Acknowledgements
The authors would like to thank the participants of the University of Calgary Maternal-Fetal Medicine, Pathology, and Genetics Rounds for their consistent engagement and contributions over the years. We also would like to thank Charlotte Monroe for her exceptional support as an administrative assistant.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
