Abstract
Objective
Congenital cytomegalovirus infection (cCMV) is a common, frequently unrecognized cause of childhood disability. The aim of the present study was to determine the symptoms that raise the suspicion of cCMV, define the neurodevelopmental outcomes, and assess their correlations.
Methods
This longitudinal observational study comprised 78 children with symptomatic cCMV who underwent neuropediatric follow-up for 4 to 17.9 years.
Results
Symptoms of central nervous system involvement, hearing/visual impairments, and hepatic involvement were mostly recognized. The average age of disease suspicion was 3.3 months. In terms of outcomes, 10.53% of the children developed complex minor neurological dysfunction and 23.68% developed cerebral palsy. Visual and hearing impairments occurred in 38.16% and 14.47% of patients, respectively. Intellectual disability was present in 30.26% of patients, and epilepsy in 21.05%. Microcephaly and hearing impairment was significantly associated with overall neurodevelopmental outcome. Microcephaly was also associated with poor motor outcomes, hearing impairment, and severe visual impairment. Furthermore, microcephaly and intrauterine growth restriction were significantly associated with poor cognitive outcomes.
Conclusion
Symptoms that raised the suspicion of cCMV—especially microcephaly, hearing impairment, and intrauterine growth restriction—were important parameters that were associated with outcomes; however, their recognition was often insufficient and/or late.
Keywords
Introduction
Congenital cytomegalovirus infection (cCMV) is the most common vertically transmitted infection, the leading cause of non-genetic sensorineural hearing loss (SNHL) in children, and a common cause of childhood disability.1–4 The prevalence of cCMV in newborns is 0.2% to 6.1% (in developed countries, it is approximately 0.7%), of whom nearly 10% are symptomatic at newborn age and 40% to 60% experience permanent neurodevelopmental sequelae.1–7 Furthermore, 10% to 15% of asymptomatic newborns develop permanent sequelae, mostly in the form of SNHL.1–3
Universal or selective newborn screening for cCMV is only performed in limited regions and countries; its diagnosis in the clinical setting is mostly established based on clinical symptoms, and is generally confirmed by isolation of the virus, specific serology, or polymerase chain reaction (PCR) of urine, blood, or saliva in the first 2 to 3 weeks of life. 8 Confirmation of cCMV after the neonatal period can also be obtained retrospectively by PCR of a dried blood sample from the neonatal period (e.g., a Guthrie card), although this method has lower sensitivity. 9 Nonetheless, a diagnosis of probable cCMV in the clinical setting is often established after newborn age, based on clinical symptoms and supported by specific cCMV neuroimaging findings.1,8,10,11
The symptoms and signs of cCMV in newborns differ among authors, but usually include microcephaly, birthweight small for gestational age, petechiae, thrombocytopenia, anemia, jaundice, hepatosplenomegaly, elevated transaminases, chorioretinitis, hearing loss, neurological disorders, and/or convulsions.1,6,8 The most severe and permanent disabilities include SNHL, intellectual disability, cerebral palsy (CP), visual impairment, epilepsy, and autism spectrum disorders (ASD).1–4,12–14
Clinical predictors of symptomatic cCMV (scCMV) outcomes have not yet been precisely defined.15–21 In the first studies of scCMV, children had multiple symptoms, a high rate of central nervous system (CNS) involvement, and high morbidity and mortality. 18 Furthermore, a recent study reported that significantly more children with symptoms of CNS involvement have intellectual disability and cumulative SNHL than children with transient non-CNS findings or petechial rash only; by contrast, the frequency of late-onset SNHL does not differ between these two groups. 21
The practical problem of recent studies, however, is that the recruitment of participants by newborn screening does not correspond to that of everyday clinical practice.7,22 Notably, a study by Dreher et al. revealed that the outcomes of children with cCMV detected by newborn screening differ significantly from those of children referred from clinical practice. 6
The aim of the present study was therefore to retrospectively determine the symptoms of cCMV that raise suspicion of cCMV, identify all other cCMV symptoms that each child had according to their medical history, and determine the timing of suspicion. We also aimed to describe the outcomes of children with scCMV and investigate the associations between symptoms and neurodevelopmental outcomes.
Patients and methods
The present longitudinal observational study comprised children with scCMV infection who underwent neuropediatric follow-up for between 4 and 17.9 years, to compare their initial symptoms and neurodevelopmental outcomes. Initially, the study included all consecutive patients with scCMV at the Children’s Hospital Zagreb, who were born between 1997 and 2009. A diagnosis of cCMV was based on clinical symptoms confirmed by laboratory proof of infection (viral isolation in urine, positive immunoglobulin M, or positive PCR in blood and urine) established by an infectious disease specialist and/or a neuropediatrician.
The research was approved by the Ethics Committee of the Children’s Hospital Zagreb (9 September 2010, approval number 01-23/5-10). All procedures were performed in accordance with the ethical standards of the responsible committees on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. We obtained informed consent from all subjects involved in the study and de-identified all patient details.
Data collection and patient grouping
The data on gestational age, birth weight, and head circumference were collected and classified according to Fenton charts for preterm infants or the World Health Organization (WHO) charts for full-term children.23,24 The symptoms of cCMV that were the immediate reason for CMV testing, and the age of each child at that moment, were established. The recorded symptoms were microcephaly, intrauterine growth restriction (IUGR), jaundice, hepatosplenomegaly, elevated transaminases, thrombocytopenia, petechiae, anemia, convulsions, neurological disorders, visual impairment with special emphasis on chorioretinitis, hearing impairments, and neonatal sepsis syndrome. Microcephaly was defined as a head circumference below the third percentile, and small for gestational age was defined as a birth weight below the 10th percentile for age and sex. Initial symptoms of neurological disorders encompassed delayed development of postural reactions, increased/decreased muscular tone, and newborn syndromes of irritability/apathy. 25 We also recorded all additional symptoms of cCMV in each patient’s medical history that were unrecognized at the time of suspicion.
Patients were grouped into children with confirmed scCMV (whose diagnosis was made within the first 3 weeks of life) and children with suspected scCMV (if the diagnosis was made later in life based on clinical symptoms and suggestive neuroimaging findings). 11
All children underwent neuropediatric follow-up until the age of at least 4 years of age. The outcome head circumference was assessed using WHO charts for children up to 5 years of age, and using Nellhaus charts for children after that age.24,26
Motor outcomes
Motor outcomes were categorized as normal, minor neurological dysfunction (MND) type 1 or 2, or CP based on the Surveillance of Cerebral Palsy in Europe classification.27,28 Furthermore, assessments of upper and lower limb function were conducted using the Bimanual Fine Motor Function (BFMF) and Gross Motor Function Classification System (GMFCS). 27
Visual and hearing impairments, epilepsy, cognitive development, and ASD
The presence of visual and hearing impairments was categorized as follows: no impairment, impairment (including all levels of impairment), and severe impairment only, with a special emphasis on chorioretinitis and progressive SNHL/cochlear implant. 27 The presence of epilepsy was also determined, with particular emphasis on intractable epilepsy. Cognitive development was stratified into the following categories: normal/borderline development (intelligence quotient [IQ] >70), mild intellectual disability (IQ 50–70), and moderate/severe intellectual disability (IQ < 50). 27 The presence of ASD was also documented.
CP
The severity of CP was classified according to the “impairment index” developed by Horber et al. 29 This classification includes mild CP (GMFCS I–II, no intellectual disability, no severe visual or hearing impairment, and no epilepsy), moderate CP (includes all levels of disability between mild and severe CP), and severe CP (GMFCS IV–V and/or severe intellectual disability with or without one or more of the following disabilities: severe visual or hearing impairment and active epilepsy). 29
Neurodevelopmental outcomes
Neurodevelopmental outcomes were stratified into the following four categories: 1) normal development (IQ ≥ 70, normal motor development or MND1, without severe visual or hearing impairment, without epilepsy, and without ASD); 2) permanent disability without CP (IQ 50–69, MND2, severe visual/hearing impairment, epilepsy, or ASD); 3) severe permanent disability without CP (IQ < 50 or IQ 50–69 with MND2, severe visual/hearing impairment, epilepsy, or ASD) or CP of a mild or moderate grade; and 4) severe CP according to the “impairment index” of Horber et al. 29
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics for Windows, version 25.0 (IBM Corp., Armonk, NY, USA). We described the sample using frequencies and proportions with nominal variables. The chi-square test or Fisher’s exact test was used to investigate differences in symptom incidence between groups. In all analyses, p < 0.05 was used to determine significance. The reporting of this study conforms to Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 30
Results
Of the 78 children (38 boys and 40 girls) with scCMV who were initially included in the present study, most were born at full-term (63 children; 80.77%). Of the 15 (19.23%) children who were born prematurely, eight were born at 35 to 36 weeks of gestation, six at 32 to 34 weeks of gestation, and one at 31 weeks of gestation. The distributions of the children according to percentiles for birth weights and head circumferences are shown in Table 1. There were 12 (15.38%) patients with IUGR and 12 (15.38%) patients with microcephaly.
Distributions of patients according to birth weights and head circumferences.

Of the 78 patients, 26 (33.33%) had confirmed scCMV (confirmed from samples obtained during the first 3 weeks of life); the other children were diagnosed with probable scCMV. The average age for the suspicion of cCMV was 3.3 months for all patients and 4.6 months for patients with probable scCMV. The symptoms of cCMV for both groups are listed in Table 2. In the 26 confirmed scCMV patients, 18 (69.23%) had two or more symptoms of infection, and in the 52 probable scCMV patients, 46 (88.46%) had two or more symptoms. At the initial evaluation, thrombocytopenia was more common in patients with confirmed scCMV (p = 0.040), whereas neurological disorders in early infancy were more common in patients with probable scCMV (p < 0.001). There were no significant differences in any other symptoms between patients with confirmed and probable scCMV. However, given the relatively small number of subjects with each symptom, all children were considered one group for further statistical analysis.
Comparison of symptoms of cCMV in children with confirmed and probable symptomatic cCMV.
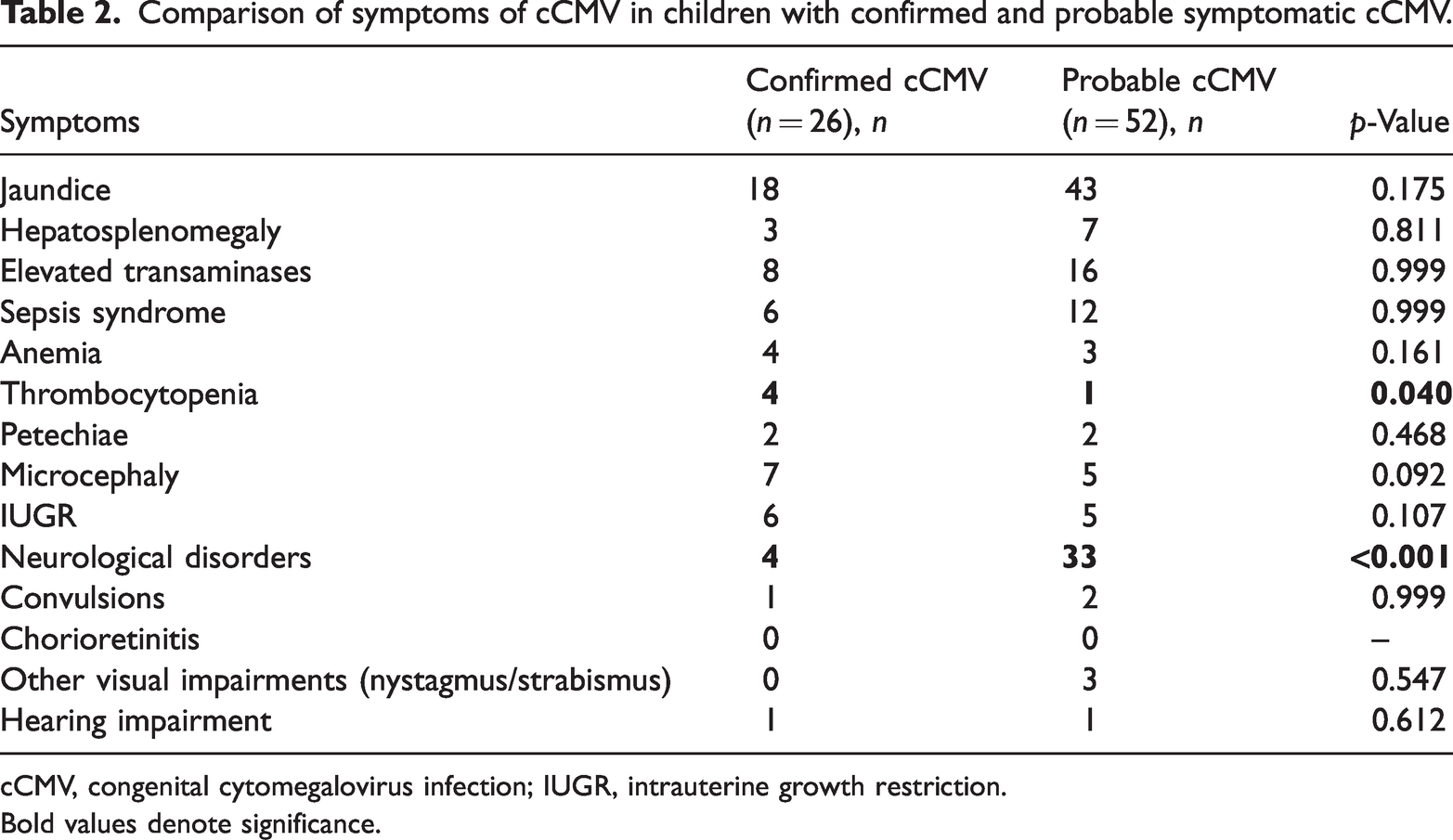
cCMV, congenital cytomegalovirus infection; IUGR, intrauterine growth restriction.
Bold values denote significance.
The clinically recognized symptoms of cCMV that raised suspicion of infection are shown in Table 3, alongside all symptoms noted in each child’s medical history. The most recognizable symptoms in our group were hepatosplenomegaly, elevated transaminases, microcephaly, neurological disorders, convulsions, and visual and hearing impairments.
Comparison of clinically recognized symptoms that raised the suspicion of symptomatic congenital cytomegalovirus infection, and all symptoms of congenital cytomegalovirus infection that the patients had (based on their medical history).

IUGR – intrauterine growth restriction.
Two patients were lost to follow-up before the age of 4 years. The average period of follow-up was 6.7 years (range, 4.0–17.9 years). Of the 76 children who underwent follow-up, 33 (43.42%) had normal motor development, 17 (22.37%) developed MND1, 8 (10.53%) had MND2, and 18 (23.68%) developed different types of CP. According to the “impairment index” of CP severity, all children with CP had moderate (5/18) or severe (13/18) types (Table 4). Furthermore, visual impairment was noted in 29 (38.16%) children and was severe in 5 (6.58%) children. Chorioretinitis typical of cCMV was detected in 11 (14.47%) children during follow-up, and atrophy of the optic nerve was identified in 15 (19.73%) children. Hearing impairment occurred in 11 (14.47%) children, and was severe in more than half (6/11; 54.54%) of these children. Unilateral hearing impairment and progressive/fluctuating SNHL were each detected in one (1.31%) child only. Four (5.26%) children underwent cochlear implantation. Epilepsy was diagnosed in 16 (21.05%) children, and was intractable in almost half (7/16; 43.75%) of these children. One (1.31%) child had West syndrome. Of the 18 children with CP, 11 (61.11%) had epilepsy. Cognitive development was normal/borderline in 53 (69.74%) children, whereas 9 (11.84%) had mild intellectual disability and 14 (18.42%) had moderate/severe intellectual disability. ASD was diagnosed in two (2.63%) children.
Classification of cerebral palsy and associated disabilities in children with symptomatic congenital cytomegalovirus infection.

BFMF, Bimanual Fine Motor Function; GMFCS, Gross Motor Function Classification System; IQ, intellectual quotient.
Considering multiple disabilities, we stratified the children into four neurodevelopmental categories. Of the 76 children, 43 (56.58%) had normal development, 14 (18.42%) had permanent disability without CP, 6 (7.89%) had severe permanent disability without CP or moderate CP, and 13 (17.11%) had severe CP.
The neuroimaging findings of two patients—one with probable scCMV and the other with confirmed scCMV that was diagnosed in the newborn period because of universal newborn hearing screening failure—are shown in Figures 1 and 2, along with their symptoms and outcome classifications.

Brain magnetic resonance imaging (MRI) in a child with suspected symptomatic congenital cytomegalovirus infection (symptoms: microcephaly, intrauterine growth restriction, neurological disorder, and sepsis syndrome). (a) MRI showing pachygyria, ventriculomegaly, and leukoencephalopathy and (b) MRI showing cysts of the temporal lobes and cerebellar hypoplasia. The MRI findings correspond to multiple extensive changes (score 4 according to Alarcon et al. 11 ); the child developed bilateral spastic cerebral palsy (Gross Motor Function Classification System: 5, Bimanual Fine Motor Function: 5) with epilepsy, severe visual and hearing impairments, severe intellectual disability, and microcephaly.

Brain magnetic resonance imaging (MRI) in a child with confirmed symptomatic congenital cytomegalovirus infection (symptom: universal newborn hearing screening failure). (a) MRI showing cysts of the temporal lobes. (b) MRI showing a periventricular cyst in the right occipital lobe and (c) MRI showing leukoencephalopathy. The MRI findings correspond to moderate changes (score 3 according to Alarcon et al. 11 ); the child had severe bilateral hearing loss, a cochlear implant, and impaired speech development, but had no other disabilities and received regular schooling.
We then examined the associations between symptoms of scCMV and overall neurodevelopmental outcomes. When only symptoms that led to CMV testing were considered, microcephaly (χ2 = 13.686, p = 0.003) and hearing impairment (χ2 = 9.097, p = 0.028) were significantly associated with overall neurodevelopmental outcomes (Table 5). Furthermore, when all symptoms, both recognized and unrecognized, were analyzed, both jaundice (χ2 = 14.558; p = 0.002) and petechiae (χ2 = 13.005; p = 0.005) were significantly associated with outcomes; however, they were not the sole symptoms in any of the patients. No significant differences in outcomes between sexes (χ2 = 0.652) or between patients with one or two or more symptoms were observed (χ2 = 6.679).
Associations between clinically recognized symptoms that raised the suspicion of scCMV and neurological outcomes.

IUGR, intrauterine growth restriction; scCMV, symptomatic congenital cytomegalovirus infection.
Bold values denote significance.
The associations between individual symptoms that prompted CMV testing and specific aspects of outcomes were also assessed. Both microcephaly (χ2 = 14.305; p = 0.003) and anemia (χ2 = 10.863; p = 0.012) were significantly associated with motor outcomes. Furthermore, severe visual impairment was associated with microcephaly (χ2 = 19.983; p < 0.001) and other visual impairments (nystagmus/strabismus) (χ2 = 18.348; p < 0.001). Overall hearing impairment was also associated with microcephaly (χ2 = 6.642; p = 0.010) as well as with sepsis (χ2 = 6.642; p = 0.010), petechiae (χ2 = 5.988; p = 0.014), and hepatosplenomegaly (χ2 = 3.830; p = 0.050). Notably, none of the symptoms related to epilepsy were recognized as symptoms of scCMV.
In addition, both microcephaly (χ2 =15.315; p < 0.001) and IUGR (χ2 = 10.483; p = 0.005) were significantly associated with poor cognitive outcomes. Moreover, microcephaly (χ2 = 24.429; p < 0.001) and IUGR (χ2 = 3.910; p = 0.048) as symptoms of cCMV were associated with microcephaly at the outcome, which was also significantly associated with poor overall neurodevelopmental outcomes (χ2 = 47.947; p < 0.001).
Discussion
In the present study, among the symptoms that raised a suspicion of scCMV, symptoms of CNS involvement (microcephaly, convulsions, and neurological disorders), visual/hearing impairment, and hepatic involvement were primarily recognized in clinical settings. Furthermore, microcephaly, hearing impairment, and IUGR were important outcome parameters. The presence of microcephaly, which indicates CNS involvement and has been identified as a reliable predictor of negative outcomes in previous studies,15,17 only led to suspicion of scCMV in approximately half of our patients. Nevertheless, microcephaly was significantly correlated with overall outcomes, motor and cognitive function, and hearing and severe visual impairments. We also observed that hearing impairments detected during initial evaluations were associated with unfavorable neurodevelopmental outcomes later in life. Most hearing loss data at birth were well-documented in our patients because of Croatia's comprehensive national hearing screening program, which has been in place since 2002 and has contributed to the early detection of scCMV in neonates. 31 According to a study by Noyola et al., 15 microcephaly is the most specific predictor of poor cognitive and motor outcomes. However, in contrast to our results, their study indicated that SNHL is only correlated with adverse cognitive outcomes when confirmed or detected after the neonatal period. 15
In our patients, hearing impairment was associated with microcephaly (as previously mentioned), sepsis-like syndrome, petechiae, and hepatosplenomegaly. On the basis of previous results, these may be considered signs of disseminated infection.16,19 Although Rivera et al. 19 reported that signs of CNS involvement, including microcephaly, are not predictive for SNHL, Pinninti et al. 21 reported a higher prevalence of SNHL and adverse cognitive outcomes in children with CNS involvement.
A study by Madden et al. 20 estimated the outcomes of children with scCMV and SNHL. They reported that 81% of participants had neurological and radiological complications. Interestingly, they also identified an inverse correlation between CP and SNHL. In the present study, however, 9 of 11 children with hearing impairments also had CP. Similarly, other studies have reported a relatively high rate of hearing impairments in children with CP caused by cCMV.4,14
Our findings also revealed that IUGR, which was a marker of poor cognitive outcome in our study, only raised a suspicion of scCMV in 27% of the children. This suggests that CMV placentitis, which can result in IUGR, 1 is often overlooked. None of the symptoms of cCMV in our patients were related to epilepsy, which aligns with the results of an earlier study on epilepsy in children with scCMV. 13 However, in the previous study, all children with epilepsy also developed CP, whereas in our cohort, 5 of the 16 children with epilepsy did not develop CP. 13 In addition, we did not identify any significant differences in the outcomes of patients with one or two and more symptoms, in contrast to a study showing that children with two or more symptoms had more adverse outcomes. 6
During our extended follow-up, we observed a bimodal distribution of the outcomes, as described previously. 17 More than half of the participants (56%) had normal outcomes, whereas approximately 15% had some permanent sequelae. All of the remaining children had poor outcomes in terms of moderate or severe CP and/or other moderate or severe permanent disorders. A recent comprehensive study investigated the neurodevelopmental outcomes of 753 children with cCMV. It revealed that 45.3% of the 266 children with symptomatic infection had normal outcomes, and the remaining children experienced mild, moderate, or severe neurodevelopmental disability, similar to our findings. 22 However, participants in the previous study were recruited through screening, and many specific outcomes were combined for asymptomatic and symptomatic infection, making comparisons with the present findings difficult. 22
It is worth noting that none of our patients had mild CP (as classified by the “impairment index” of Horber et al. 29 ). This result is consistent with the findings of previous studies indicating that children with cCMV tend to have severe CP, as opposed to the approximately 30% of children with CP in the Surveillance of CP in Europe common database who have mild CP.4,14,29
One important characteristic of the present study is its partially retrospective design. Once a diagnosis of scCMV was made, we thoroughly analyzed the medical history of each patient. This allowed us to identify any symptoms of scCMV that had gone unnoticed and assess how frequently scCMV symptoms were recognized in a clinical setting.
Limitations were also mostly determined by the partially retrospective design of our study. For example, the data on head circumference at birth were not available for approximately half of our patients; thus, one of the most important predictors of outcome was missed. Moreover, in the case of multiple symptoms in a single patient, we were unable to estimate the most indicative ones for diagnosing scCMV. We were also unable to determine typical clusters of symptoms because of the small number of participants relative to the number of symptoms. In addition, a further limitation was the possibility of early postneonatal cCMV in our patients with probable scCMV. Unfortunately, retrospective dried blood testing was not readily available. Another limitation of our study was possible selection bias, which may have resulted in the relatively low percentage of hearing impairments in our study. Our study comprised a population selected from neuropediatric patients, and children with isolated hearing impairments (which are a common isolated symptom of cCMV infection) are usually treated by otorhinolaryngologists and infectious disease specialists only.
The present findings suggest that symptoms that raise the suspicion of cCMV in a clinical setting—mainly microcephaly, hearing impairment, and IUGR—may be important outcome parameters. However, their recognition is often insufficient and/or late. Better screening of these symptoms during the newborn period may help to direct clinicians toward cCMV infections. One novelty of the current study is its long multidisciplinary follow-up, which was able to reveal progressive disabilities (e.g., progressive hearing loss) or disabilities developing in older childhood (e.g., epilepsy). However, more studies with larger numbers of participants are needed to compare the symptoms and outcomes of cCMV.
Footnotes
Acknowledgements
We acknowledge Associate Professor Vlasta Đuranović, MD, PhD, and Lucija Lujić, MD, MSc, from Children’s Hospital Zagreb, as the referring neuropediatricians for some of the patients from this study.
Author contributions
Conceptualization: Ivana Đaković, Vlatka Mejaški Bošnjak, Ivica Kostović; Methodology: Ivana Đaković, Vlatka Mejaški Bošnjak, Iva Prvčić, Katarina Vulin; Formal analysis and investigation: Ivana Đaković, Vlatka Mejaški Bošnjak, Iva Prvčić, Goran Tešović, Goran Krakar, Tomislav Gojmerac, Jadranka Sekelj Fureš; Writing – original draft preparation: Ivana Đaković, Vlatka Mejaški Bošnjak; Writing – review and editing: Ivica Kostović, Katarina Vulin, Iva Prvčić, Goran Tešović, Goran Krakar, Tomislav Gojmerac, Jadranka Sekelj Fureš; Funding acquisition: none; Resources: none; Supervision: Vlatka Mejaški Bošnjak, Ivica Kostović.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
