Abstract
We present 2 diagnostically challenging cases of pediatric/adolescent relapsed/refractory aggressive mature B-cell non-Hodgkin lymphoma (B-NHL) within the spectrum of Burkitt lymphoma and diffuse large B-cell lymphoma and illustrate the different therapeutic regimens that are employed for pediatric and adult cancer centers. Both cases displayed varying-sized lymphoma cells with occasional single prominent nucleoli and heterogeneous BCL2 expression. Cytogenetics revealed complex karyotypes with t(8:14)(q24.2;q32) and IGH::MYC rearrangement by FISH. Next generation sequencing revealed deleterious TP53 and MYC mutations. We concluded that both could be diagnosed as “DLBCL-NOS with MYC rearrangement” using the current pathologic classifications, 2022 International Consensus Classification (ICC) and World Health Organization Classifications of Haematolymphoid Tumors (WHO-HAEM5). This report illustrates diagnostic challenges and treatment dilemmas that may be encountered, particularly for adolescent and young adults (AYA).
Introduction
Diffuse large B-cell lymphoma (DLBCL) and Burkitt lymphoma (BL) are the most common aggressive mature B-cell non-Hodgkin lymphomas (B-NHLs) in childhood and adolescence. 1 The distinction between BL and DLBCL may be challenging due to overlapping histologic, cytogenetic, and molecular features. While similar treatment protocols are employed in pediatric DLBCL and BL per practice guidelines (PBCL-NCCN), 1 adolescent patients may receive adult-tailored protocols depending on treatment facility.
We present the clinicopathologic and molecular genomic profiles of 2 aggressive mature B-NHLs in 2 males aged 8 and 18 years. Targeted next-generation sequencing (NGS; Supplemental Table 1) was performed as previously desc-ribed.2 -4 The diagnostic challenges and treatment dilemmas, especially in AYAs, are presented.
Cases Presentations
Case #1
An 8-year-old male presented with fever, abdominal pain, and a lower anterior abdominal cavity mass measuring 12.3 cm × 9.7 cm × 7.3 cm with fluorodeoxyglucose-positron emission tomography (FDG-PET) avidity of standardized uptake value (SUV) 20.1. Biopsy was performed and the patient was diagnosed with Burkitt lymphoma versus diffuse large B-cell lymphoma, with the latter being favored. Histopathologic, karyotypic, and FISH findings are summarized in Tables 1 and 2. The intraabdominal disease and absence of CNS and bone marrow involvement warranted a Stage III classification per the International Pediatric Non-Hodgkin Lymphoma Staging System. A pediatric treatment regimen (Table 1) yielded a partial response with significant decrease of abdominal mass size at 2 months post-treatment. However, FDG-PET avidity (SUV 3.3) persisted leading to re-biopsy of the mass that confirmed relapsed/refractory (R/R) disease (Table 1, Figure 1(d)). Treatment was intensified, he achieved complete remission, and is disease-free at 23 months follow-up.
Summary of Clinical, Pathologic, and Immunophenotypic Findings.
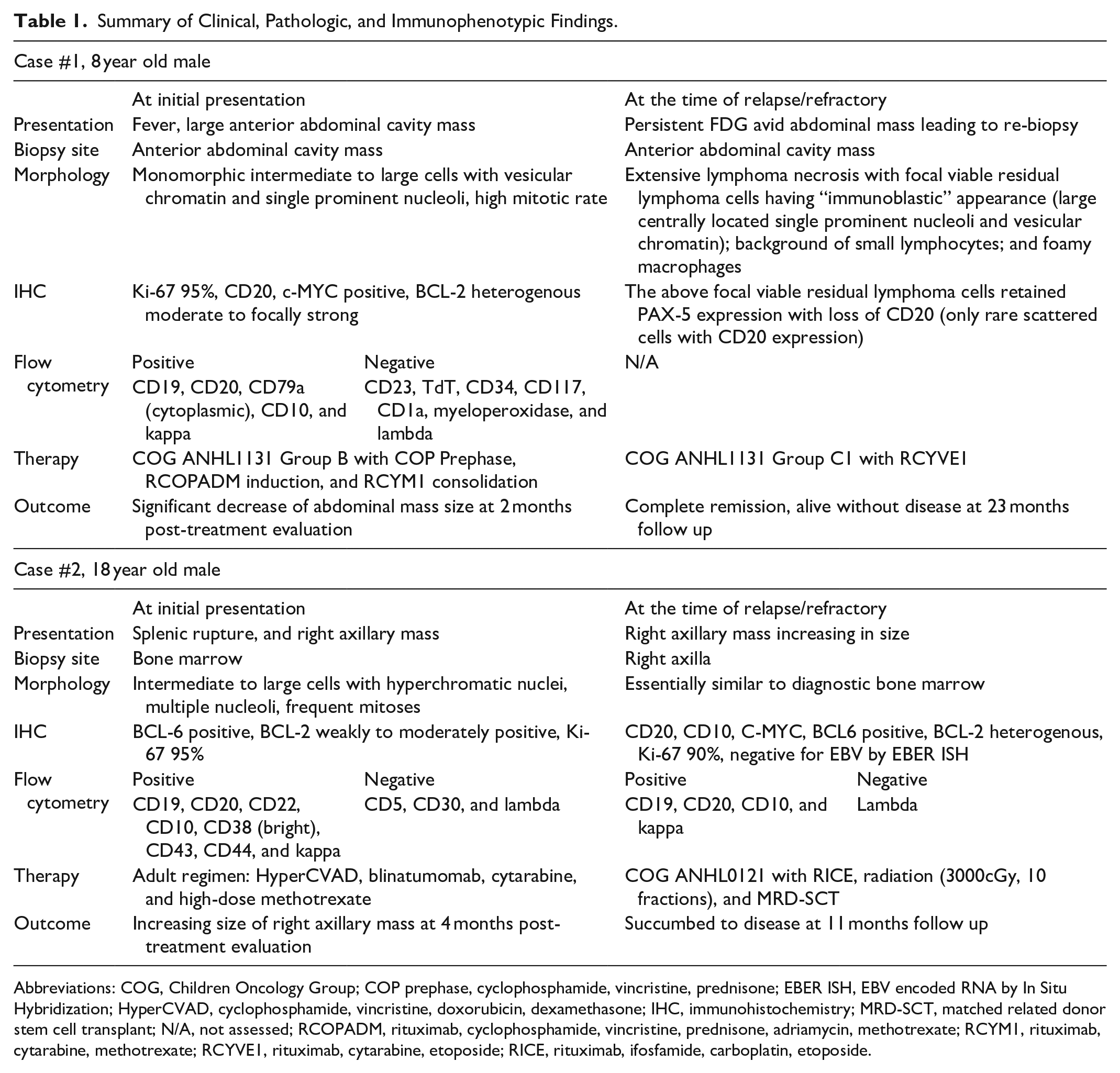
Abbreviations: COG, Children Oncology Group; COP prephase, cyclophosphamide, vincristine, prednisone; EBER ISH, EBV encoded RNA by In Situ Hybridization; HyperCVAD, cyclophosphamide, vincristine, doxorubicin, dexamethasone; IHC, immunohistochemistry; MRD-SCT, matched related donor stem cell transplant; N/A, not assessed; RCOPADM, rituximab, cyclophosphamide, vincristine, prednisone, adriamycin, methotrexate; RCYM1, rituximab, cytarabine, methotrexate; RCYVE1, rituximab, cytarabine, etoposide; RICE, rituximab, ifosfamide, carboplatin, etoposide.
Summary of Molecular and Cytogenetic Findings.

Abbreviations: BL, Burkitt lymphoma; CDS, coding sequence; DLBCL, diffuse large B-cell lymphoma; Dx, diagnosis; FL, follicular lymphoma; M, male; N/A, not assessed; NP, not provided; R/R, relapsed/refractory; VAF, variant allele fraction.
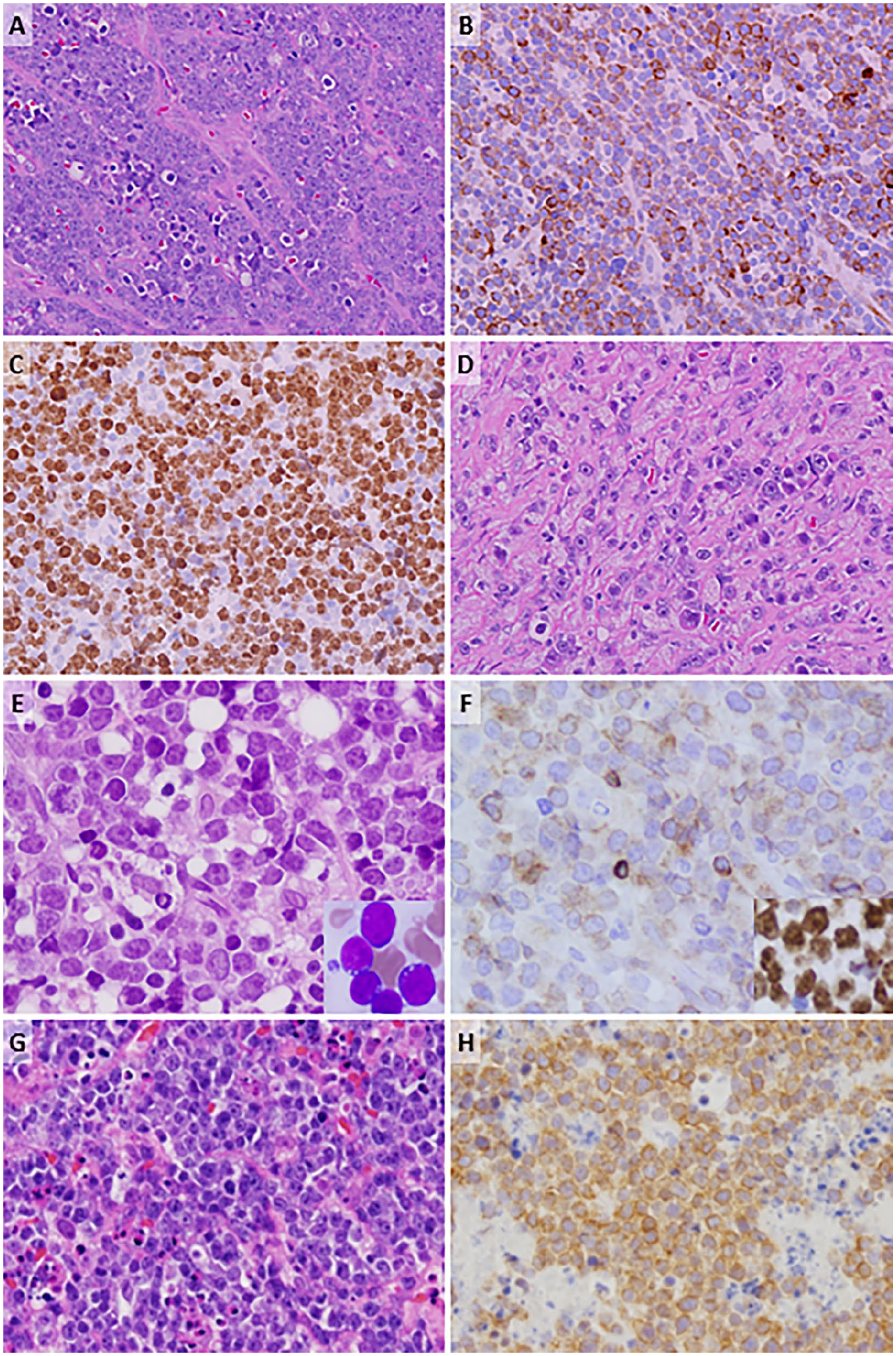
Pathologic findings of pediatric/adolescent aggressive mature B-NHLs with BCL2 expression. (A) Case #1, abdominal mass biopsy showed intermediate to large neoplastic cells with “large cell” appearance (vesicular chromatin and prominent nucleoli), hematoxylin and eosin (H&E) stain, (B) heterogeneous to strong focal positivity for BCL2, and (C) high proliferation index (Ki-67 ~95%). (D) Post-treatment abdominal mass biopsy demonstrates viable lymphoma cells (H&E). (E, F) Case #2, diagnostic bone marrow showed diffuse infiltration by intermediate-sized to large lymphoma cells on biopsy (H &E) (E) frequent cytoplasmic vacuoles on aspirate (Wright-Giemsa, E inset) with expression of BCL2 (focal, weak) (F) and diffuse C-MYC (F, inset). (G, H) Case #2, R/R right axillary mass biopsy with diffuse infiltration by lymphoma cells (H&E, G) showing heterogenous BCL2 expression (negative to focally strong positivity) (H). Olympus DP microscope, 400X magnification, CellSens imaging software.
Case #2
An 18-year-old male presented to an outside institution with pancytopenia, splenic rupture, and right axillary mass. Bone marrow biopsy and ancillary testing results are summarized in Tables 1 and 2. Notably, no BCL2 or BCL6 rearrangement was identified by karyotyping/FISH in the diagnostic bone marrow specimen.
The patient was initially diagnosed with BL and considered stage IV per IPNHLSS due to bone marrow involvement. The patient was treated with an adult regimen (Table 1) with disease progression (increasing mass size) by PET scan. Four months later, he presented to our institution and biopsy confirmed R/R disease (Table 1). Lymphoma cells showed heterogeneous BCL2 expression ranging from negative to diffusely positive. The patient was treated with chemotherapy, radiation therapy, and underwent matched related donor stem cell transplant with disease progression. Care shifted to palliative management; the patient passed from disease progression 11 months post-treatment initiation.
Molecular Findings
Both diagnostic and R/R samples underwent targeted NGS (Supplemental Table 1). Variants of established/potential and uncertain clinical significance (Tiers I-III) are summarized (Table 2, Supplemental Table 2). In addition to IGH::MYC rearrangements, both cases harbored pathogenic TP53 variants and at least 1 activating MYC missense variant. In case #2; however, the activating MYC p.P74S variant was only present in the R/R sample. Case #1 also harbored loss-of-function variants in ARID1A and BCL11B and copy loss of CDKN2A/B. Case #2 harbored additional Tier II variants in CCND3, PHF6, PTEN, and SMARCA4.
Discussion
Distinguishing among BL, DLBCL, or high-grade B-cell lymphoma (HGBCL) can be challenging even among experienced pathologists. 5 For children and adolescent patients treated at a pediatric centers, the treatment of the above entities are similar according to PBCL-NCCN. 1 However, for AYAs treated at adult oncology facilities, distinction and subsequent management depends on the adult classifications: WHO-HAEM5 and ICC.6,7 Our 2 cases highlight challenges rendering a definitive diagnosis where overlapping morphologic, immunophenotypic, and molecular features are encountered. Both cases showed shared histologic, cytogenetic, and molecular features along the spectrum of BL, DLBCL, and HGBCL: (a) morphology showed lymphoma cells with size variation, irregular nuclear contours, vesicular chromatin, variable prominent nucleoli, significant nuclear pleomorphism, and no obvious “starry sky” pattern; (b) ancillary studies showed heterogeneous BCL2 expression; (c) cytogenetics showed complex karyotypes with IGH::MYC translocations; and (d) genetic mutations were present that are identifiable in BL, DLBCL, and HGBCL.
The differential diagnosis for both cases included the following entities: BL, DLBCL-NOS (encompassing DLBCL with MYC rearrangement), and HGBCL-NOS, the latter of which was not supported due to the “large cell” cytology.8,9 While BCL2 expression is traditionally thought to preclude a diagnosis of BL in pediatric/adolescent patients, a few studies including considerable numbers of BLs with variable degrees of BCL2 expression have challenged this assumption.10,11 Furthermore, a Ki-67 proliferation index >95% and strong C-MYC positivity is generally not seen in DLBCL. 12 Both cases displayed t(8:14)(q24.2;q32) IGH::MYC translocation with a complex karyotype, which are common in DLBCL but have been reported in up to 27% of pediatric BLs. 13 Despite the clinical presentation of abdominal mass and IGH::MYC rearrangement making BL a high diagnostic consideration, complex karyotypes, “large cell” cytology, and heterogeneous BCL2 expression challenged the diagnosis of BL. We concluded that both cases could be diagnosed as “DLBCL-NOS with MYC rearrangement” using the WHO-HAEM5/ICC criteria.
The cytogenetic and molecular landscape of pediatric, AYA, and adult aggressive mature B-NHL entities share overlapping features although distinguishing features are discernable in pediatric/AYA and adult age groups. Chromosome abnormalities including +12q15, +19q13, and −6q are shared abnormalities among patients from both age groups, whereas −4p14, −19q13.32, and +16p11.2 are abnormalities enriched in pediatric/AYA age groups. 14 Likewise, clinical genomic profiling can provide informative diagnostic information. NGS detected pathogenic variants in ARID1A and BCL11B as well as CDKN2A/B loss in case #1, and case #2 harbored variants in CCND3, PHF6, PTEN, and SMARCA4 all of which have been variably reported in BL and DLBCL.14-17 Additionally, mutations in ARID1A and SMARCA4 are more frequently identified in pediatric Burkitt lymphomas while CCND3 and TP53 mutations are not age-specific. 18 Interestingly, targeted NGS revealed that both cases harbored Tier II MYC variants: MYC p.Pro72Ser and p.Thr73Asn (case #1) and a MYC p.Pro74Ser (case #2). N-terminal domain mutations in MYC result in increased MYC protein expression. 19 Recurrent MYC missense mutations are reported in HGBCLs,15,17 and the MYC p.Pro74Ser missense mutation is among the most frequently reported mutations in BL (40%-70%) but is rare in DLBCL.16,20,21 In patient #2, the MYC p.Pro74Ser mutation was detected at low levels only in the R/R sample. The variant allele fractions (VAFs) were increased in the R/R sample compared to the diagnostic sample suggesting clonal expansion and/or testing of a higher tumor percentage. Overall, the mutational profiles and the presence of MYC N-terminal activating mutations were more consistent with HGBCLs but did not favor one entity over another.
Adult and AYAs with DLBCL and BL typically have worse outcomes compared to children treated with similar pediatric protocols.22,23 A recent retrospective study suggested that AYA might benefit from a pediatric treatment protocols. 24 Among several cytogenetic aberrations, only +7q was significantly associated with worse overall survival in pediatric BL, 25 and pediatric B-NHL patients with TP53 abnormalities have significantly inferior survival outcomes compared to wild-type counterparts. 26 Interestingly, the TP53-mutant patients in our study experienced distinct outcomes. The prognostic impact of cytogenetic and molecular features of pediatric/adolescent R/R aggressive mature B-NHL with atypical features warrants investigation.
In conclusion, we present 2 diagnostically challenging cases of pediatric and adolescent R/R aggressive mature B-NHL and highlight the management dilemmas faced by both pediatric and adult facilities when treating AYA patients. Repeat tissue biopsy of R/R disease may provide additional mutational data distinct from the initial biopsies and potential therapeutic targets. Further investigations, particularly prospective studies, of the diagnostic and prognostic markers in pediatric/AYA aggressive mature B-NHLs are warranted to study outcomes in aggressive mature B-NHL when using pediatric and adult treatment protocols.
Supplemental Material
sj-xlsx-1-pdp-10.1177_10935266241230600 – Supplemental material for Childhood and Adolescent Relapsed/Refractory Aggressive B-Cell Lymphomas With t(8;14) and BCL2 Expression, Burkitt Lymphoma Versus Diffuse Large B-Cell Lymphoma: A Diagnostic Challenge
Supplemental material, sj-xlsx-1-pdp-10.1177_10935266241230600 for Childhood and Adolescent Relapsed/Refractory Aggressive B-Cell Lymphomas With t(8;14) and BCL2 Expression, Burkitt Lymphoma Versus Diffuse Large B-Cell Lymphoma: A Diagnostic Challenge by Fouad El Dana, Sofia Alexandra Garces Narvaez, Nader K. El-Mallawany, Jennifer E. Agrusa, ZoAnn E. Dreyer, Andrea N. Marcogliese, Mohamed Tarek Elghetany, Jyotinder N. Punia, Chi Young Ok, Keyur P. Patel, Dolores H. Lopez-Terrada, Kevin E. Fisher and Choladda V. Curry in Pediatric and Developmental Pathology
Supplemental Material
sj-xlsx-2-pdp-10.1177_10935266241230600 – Supplemental material for Childhood and Adolescent Relapsed/Refractory Aggressive B-Cell Lymphomas With t(8;14) and BCL2 Expression, Burkitt Lymphoma Versus Diffuse Large B-Cell Lymphoma: A Diagnostic Challenge
Supplemental material, sj-xlsx-2-pdp-10.1177_10935266241230600 for Childhood and Adolescent Relapsed/Refractory Aggressive B-Cell Lymphomas With t(8;14) and BCL2 Expression, Burkitt Lymphoma Versus Diffuse Large B-Cell Lymphoma: A Diagnostic Challenge by Fouad El Dana, Sofia Alexandra Garces Narvaez, Nader K. El-Mallawany, Jennifer E. Agrusa, ZoAnn E. Dreyer, Andrea N. Marcogliese, Mohamed Tarek Elghetany, Jyotinder N. Punia, Chi Young Ok, Keyur P. Patel, Dolores H. Lopez-Terrada, Kevin E. Fisher and Choladda V. Curry in Pediatric and Developmental Pathology
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
