Abstract
A 43-year-old female presented with blood loss and persistent abdominal pain at 14 weeks of gestation. Ultrasound examination and subsequent magnetic resonance imaging (MRI) revealed bilateral multicystic uterine adnexa. Exploratory laparotomy was performed at 17 weeks of gestation and bilateral serous ovarian adenocarcinoma FIGO stage IIIC was diagnosed. Complete cytoreductive surgery (CRS) was not feasible at that moment. Nine days after the exploratory laparotomy, immature rupture of membranes and contractions occurred and she delivered a premature boy after 19 weeks of gestation. Pathological examination of the placenta revealed that her ovarian cancer metastasized to the membranes. We describe the first case of ovarian cancer metastasized to the decidua of the placental membranes with histological, immunohistochemical, and molecular confirmation. This case highlights the importance of conscientious evaluation of placenta and membranes in pregnant women with ovarian cancer.
Introduction
Ovarian cancer affects 6.6 per 100 000 women globally with a peak age of 60–75 years and has the highest incidence of 10.7 per 100 000 women in Europe. 1 Ovarian cancer diagnosed during pregnancy is uncommon and affects only 1 out of 12 500–25 000 pregnant women. 2 In a large multicenter cohort study consisting of 215 patients with cancer during pregnancy, ovarian cancer occurred in only 8 patients (4%). 3 Due to this rarity, limited information is available regarding the incidence and location of ovarian cancer metastases, especially in the placenta. Only 3 cases of primary ovarian cancer metastasized to the placenta have been reported in literature.4–6 Noteworthy, metastases in the decidua of the placental membranes have not been previously reported.
Case report
A 43-year-old female, gravida 4, para 1, presented with blood loss and persistent abdominal pain at 14 weeks and 4 days of gestation. She was pregnant after frozen embryo transfer in a natural cycle. Years ago, she received in vitro fertilization (IVF) treatment due to tubal factor subfertility and a medical history of chlamydia with preservation of multiple embryos. Ultrasound examination revealed bilateral multicystic adnexa after which a subsequent magnetic resonance imaging (MRI) scan was planned to provide more accurate information regarding the characteristics of these cysts.
The MRI scan showed bilateral multicystic adnexa, measuring 12 cm on the right side and 9 cm on the left, with suspicion of more solid masses on the left ovary compared to the right (Figure 1). The adnexa were located adhesively on the uterus. No blood components nor endometrioma were detected and, therefore, further indication for deep infiltrating endometriosis was absent. Dermoid cysts were also ruled out due to the absence of fatty components. Lastly, the MRI scan showed no signs of peritoneal metastases, ascites, or enlarged lymph nodes and, thus, no secondary indication for malignancy was present.
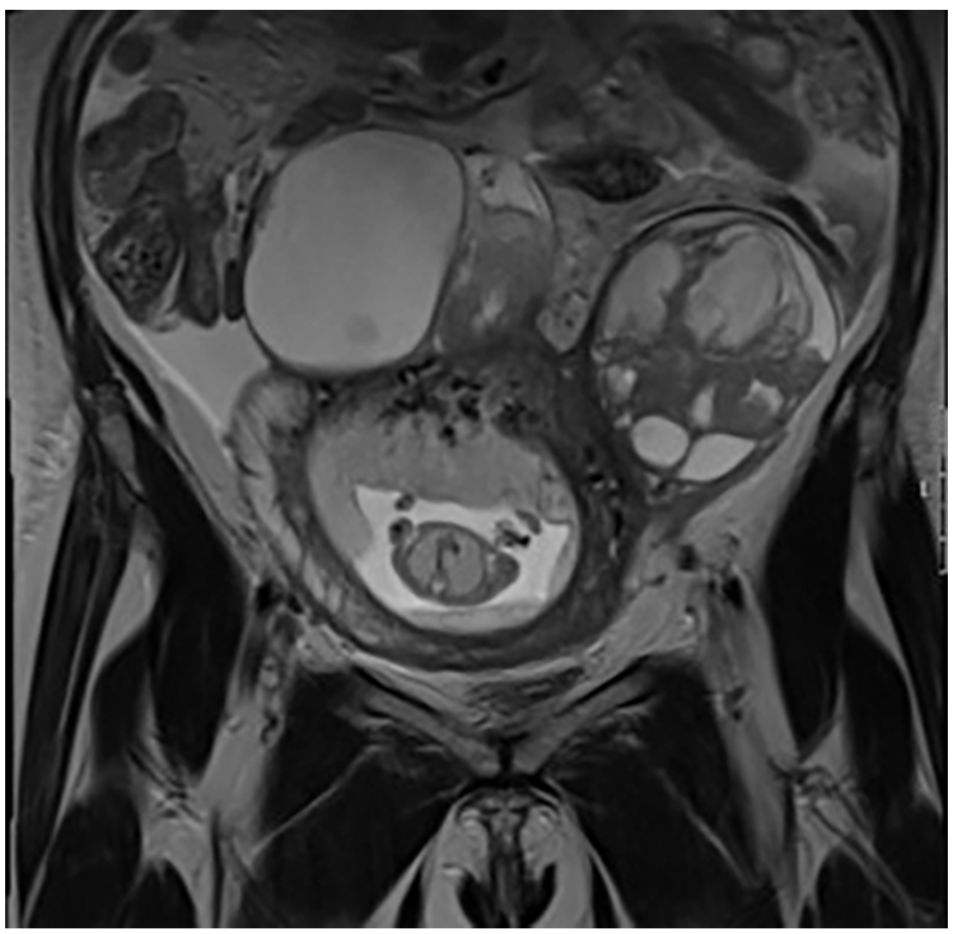
Coronal section of MRI performed before exploratory laparotomy with polycystic ovary and fetus.
During multidisciplinary consultation, a laparotomy with bilateral salpingo-oophorectomy with intraoperative frozen section examination was planned to be performed. Two and a half weeks after the MRI, exploratory laparotomy was initially performed at 18 weeks of gestation during which biopsies of the adnexa were performed. Metastasis to the pelvic lymph nodes was detected and bilateral high-grade serous ovarian adenocarcinoma FIGO stage IIIc was diagnosed. Contrary to the results of the MRI scan, ascites was now present and ascitic fluid cytology also indicated an adenocarcinoma. Biopsies of the peritoneum and omentum both showed metastatic high-grade serous adenocarcinoma of ovarian origin. Complete cytoreductive surgery (CRS) was not feasible at the time of the exploratory laparotomy and further surgery was omitted. Therefore, neoadjuvant chemotherapy was recommended.
Nine days after exploratory laparotomy, evident immature rupture of membranes and premature contractions occurred. Due to the dismal prognosis of the fetus and to prevent further delay of neoadjuvant chemotherapy, the delivery was actively induced by administering misoprostol. A male infant was born at 19 weeks of gestation, who died shortly after birth due to immaturity.
A week after delivery, the patient was admitted to our hospital due to symptoms consistent with an ileus. CT-scan showed progression of the ovarian cancer, since the number of metastases had increased in the peritoneum and omentum. On the same day, the first course of carboplatin monotherapy was administered. This chemotherapy regimen was chosen due to the poor condition of the patient. After her first course of chemotherapy, she improved clinically. Subsequent chemotherapy courses consisted of carboplatin and paclitaxel. Response evaluation after 2 courses of chemotherapy revealed a good clinical, radiological, and biochemical response.
A month after the third chemotherapy course, complete CRS combined with heated intraperitoneal chemoperfusion was performed. CRS consisted of a hysterectomy, bilateral salpingo-oophorectomy, omentectomy, and appendectomy. Additionally, metastases on the bowel mesentery and peritoneum on both pelvic walls were removed using the Plasma Jet surgical technique. A week after surgery, the patient was discharged from our center. After her last course of chemotherapy, she continued with poly ADP ribose polymerase (PARP) inhibitor niraparib. Unfortunately, she developed a platinum resistant recurrence of her ovarian cancer within 6 months after her last course of chemotherapy.
Histological examinations of the ovaries showed extensive localization of high-grade serous adenocarcinoma of ovarian origin using the WHO classification of female genital tumors (Figure 2(a) and (b)). 7 The tumor was bilaterally present in the adnexa with serosal involvement of the uterus. The sections showed ovarian tissue infiltrated by malignant tumor composed of cells arranged mainly in complex glandular and sheet pattern. The malignant cells displayed highly marked pleomorphism. The nuclei were enlarged, markedly atypical with prominent nucleoli and eosinophilic cytoplasm. Tumor giant cells were present, mitoses were easily seen and extensive areas of necrosis were present. The underlying stroma showed desmoplastic reaction with lymphoplasmacytic cells infiltration and focal lymphovascular invasion was noted. No serous tubal intraepithelial carcinoma (STIC) or mucosal involvement with in-situ lesions in the fallopian tubes were observed. Additionally, extensive localization was present in the omentum with minimal response to chemotherapy (CRS = 1). Biopsies of the abdominal wall, mesocolon, and serosa of the appendix were also positive for tumor.
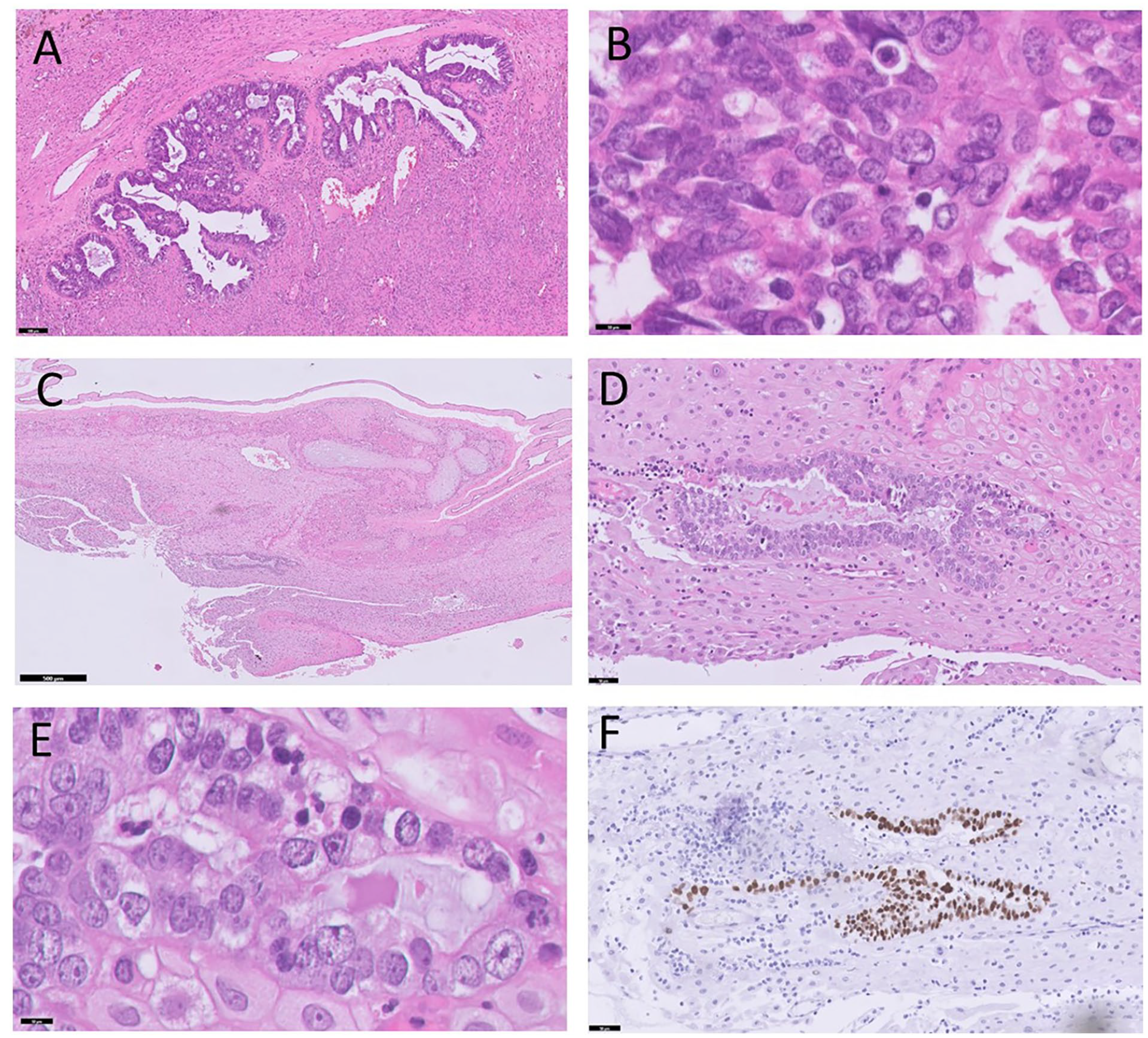
(A and B) Ovary with high-grade serous ovarian carcinoma, hematoxylin-eosin (HE) staining (100 and 10 µm). (C-E) Metastasis of high-grade serous ovarian carcinoma in membranes of placenta, hematoxylin-eosin (HE) staining (500, 50, and 10 µm). (F) Mutant P53 overexpression in metastasis in membranes of placenta (50 µm).
Immunohistochemistry for P53 detected a mutant overexpression in the lesion cells of the ovaries. Lesion cells in the ovaries were also positive for PAX8 and WT1. Furthermore, somatic mutations tested by next generation sequencing (NGS) analysis (custom AmpliSeq™ NGS HRDv2 panel) demonstrated class 5 pathogenic variation in TP53 (exon 5); NM_000546.5: c.535>T, p.(His179Tyr). The variant allele frequency was 0.87. Other class 3 variations with unknown pathogenicity were detected in CDK12 (exon 3); NM_016507.4: c.2079_2081delACC, p.(Pro694del). The corresponding variant allele frequency was 0.97.
This placenta was evaluated according to the Amsterdam Placental Consensus Statement 8 and had a normal placental weight conform P50. Histological examination was performed on 5 slides (membrane rolls and umbilical cord and 3 parenchyma) stained with hematoxylin and eosin (H&E). Gross examination and microscopy showed 2 arteries and 1 vein in the umbilical cord. Placental parenchyma demonstrated normal maturation for its gestation with minimal hydropic changes. There was no histological evidence of fetal vascular malperfusion or maternal vascular malperfusion in the placenta. A group of tumor cells consisting of atypical nuclei with multiple nucleoli and mitoses was present in the cross section of the membranes in the outer layer formed by decidua. A focal lymphocytic infiltrate surrounding the tumor cells was detected (Figure 2(c) and (e)). Similar to the ovarian cancer, the tumor cells presented with an overexpression of P53 (Figure 2(f)) in the outer layer of the membranes formed by the decidua. Additionally, the tumor cells were positive for PAX8 and WT1. In the placenta parenchyma there was no localization of ovarian carcinoma. The metastasis to the decidua of the membranes was confirmed by NGS and showed similar results of class 5 pathogenic variation in TP53 (exon 5) as the ovarian carcinoma. The variant allele frequency was 0.28.
Discussion
We describe the first molecularly confirmed case of ovarian cancer metastasized to the decidua of the placental membranes. Metastasis to the placenta most often occur in patients with a melanoma (22%), lung cancer (19%), and gastric cancer (14%). 9 Only 3 previous case reports have reported metastases of ovarian cancer to the decidua of the placental parenchyma, but none have found any metastases in the placental membranes.4 –6 This unique case prompts us to consider the pathophysiology and mechanism behind this pattern of metastasis.
A review by Tolar and Neglia, 10 describing 50 cases of maternal malignancies with metastasis to the placenta, only reported 14 cases with documented maternal-to-fetal metastases by transplacental transmission over a period of 136 years. All 14 neonates had a delayed presentation of cancer and only 3 neonates survived after treatment. The most common types of cancer were melanoma (5/14 cases) and acute lymphoblastic leukemia (2/14 cases). However, no cases of ovarian cancer were present in this review. In our case, the infant was born at 19 weeks of gestation and passed away shortly after birth due to immaturity. Stress from exploratory laparotomy and metastatic placental membranes may be the cause of preterm immature rupture of membranes and premature contractions.
Since ovarian cancer mainly spreads via the peritoneum, metastases to the placenta parenchyma, and membranes are very uncommon. 11 However, it is possible for ovarian cancer to spread hematogenous to the placenta, considering that uteroplacental blood flow represent 10% of maternal cardiac output.10,12 Tumor cells can infiltrate the placenta and may reach the fetal lungs via the ductus venosus and/or the fetal liver via the umbilical vein. 10 Nevertheless, fetal and maternal circulation is separated by a protective placental barrier consisting of trophoblasts, chorionic villi, and capillary endothelium. 10 This protective placental barrier in combination with the fetal immune response eliminating metastatic cells is in most cases efficient enough at preventing hematogenous spread of ovarian cancer to the placenta and fetus. 13
Another hypothesis could be that the ovarian cancer can spread to the placenta tissue through direct invasion by tumor cells. 11 Tumor cells can detach from the primary ovarian cancer and passively float in the peritoneal fluid, after which they can adhere to the cell surface of the myometrium and grow into the decidua. 11 However, the exact mechanism of metastasis of ovarian cancer to the placental membranes remains unknown.
In our case, the metastasis was present in the placental decidua of the membranes on the fetal-maternal barrier. Since perinatal demise occurred, and there was no consent for a perinatal autopsy, it was not possible for us to monitor the infant to evaluate the possibility of transplacental spread of the malignancy from the mother. Literature shows that transplacental tumor transmission from mother to child is very rare. Two out of the 3 cases of ovarian cancer with placental parenchymal metastasis had a live-born infant and have reported no evidence of disease in the neonate after 2 and 6 years of monitoring.4,6
The triple confirmation of the placental decidual membrane metastasis by histology, immunohistochemistry, and NGS of both the ovaries and placenta performed in our report provides confirmation of the origin of the metastasis in the decidua of the placental membranes. In general, in our institution placentas are sent for pathological examination for clinical purposes with a wide range of clinical indications such as pre-term birth (<37 weeks) including pre-term immature and premature rupture of membranes, fetal growth restriction, pre-eclampsia, pregnancy induced hypertension, gestational diabetes, malignancy in pregnancy, asphyxia of the new born, or intra uterine fetal demise (IUFD).
In conclusion, this is the first case of ovarian cancer metastasized to the decidua of the placental membranes and confirmed by histology, immunohistochemistry, and NGS. Clinicians should send placentas from women diagnosed with ovarian cancer during pregnancy to the pathologist for evaluation of metastases. Furthermore, immunohistochemistry and NGS can be performed to confirm this diagnosis made by histology. Lastly, clinicians should be aware that fetal metastases have a delayed presentation and that they should closely monitor neonates during the first 2 years of life for potential transplacental tumor transmission from mother to child.
Footnotes
Acknowledgements
We thank the patient for her consent for additional pathology analyses of the ovaries and placenta, and publication of the case report.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
