Abstract
In fetal circulation, oxygenated blood from the placenta flows through the umbilical vein into the ductus venosus (DV), then enters the inferior vena cava, and subsequently reaches the right atrium of the heart. The DV serves as a shunt, allowing this oxygen-rich blood to bypass the liver. The absence of the DV (ADV), also known as agenesis of the DV, is a rare congenital anomaly. Without a DV, blood from the umbilical vein must follow alternative routes to the heart. In ADV cases, blood from the umbilical vein must follow 1 of 2 primary drainage patterns: either an extrahepatic shunt or an intrahepatic shunt. This report details the antenatal ultrasound and postmortem findings of 2 fetuses diagnosed with ADV by prenatal imaging studies. The first case involved a fetus with a persistent right umbilical vein connected directly to the suprahepatic IVC, accompanied by early obliteration of the left umbilical vein and true agenesis of the DV. This fetus also had additional congenital anomalies. In contrast, the second case involved a fetus with a normal left umbilical vein that entered the liver. However, despite an ultrasound diagnosis of “absence” of the DV, a DV was present, though markedly hypoplastic and probably minimally functional or non-functional. In this case, blood from the umbilical vein likely followed an alternate intrahepatic route through the portal and hepatic veins, before reaching the heart (intrahepatic shunt). These contrasting cases emphasize the heterogeneity of vascular anomalies and embryologic origins captured by the term “ADV.” Additionally, the terminology of “absence” or “agenesis” may be misleading in some purported ADV cases. Specifically, in the second case, the DV was not absent; it was markedly hypoplastic instead. This also appears to be the first reported case of a hypoplastic DV in a fetus. Both cases underscore the importance of effective collaboration and clear communication between maternal-fetal medicine specialists and pathologists.
Keywords
Introduction
Intrauterine fetal circulation relies on 3 key physiological vascular shunts: the ductus venosus (DV), foramen ovale, and ductus arteriosus. These shunts are pivotal in directing oxygenated blood from the placenta to fetal tissues and organs, circumventing the developing liver and lungs. Together, they ensure the optimal oxygen and nutrient delivery necessary for proper fetal development.
Absence of the ductus venosus (ADV), alternatively known as absent ductus venosus or agenesis of the ductus venosus, refers to a rare congenital anomaly characterized by the absence of this vascular structure. This anomaly disrupts the regular pathway through which oxygenated blood from the placenta circumvents the fetal liver to enter the heart (Figure 1). While first documented in the 19th century, 1 it is in recent decades that modern ultrasound methods have facilitated the in utero identification of such cases.2 -18 Nowadays, the diagnostic process for ADV typically involves prenatal ultrasound with Doppler flow study. Prenatal ultrasound offers insight into the developing fetus and its circulatory system, while Doppler ultrasound gauges the velocity and direction of blood flow within vessels.
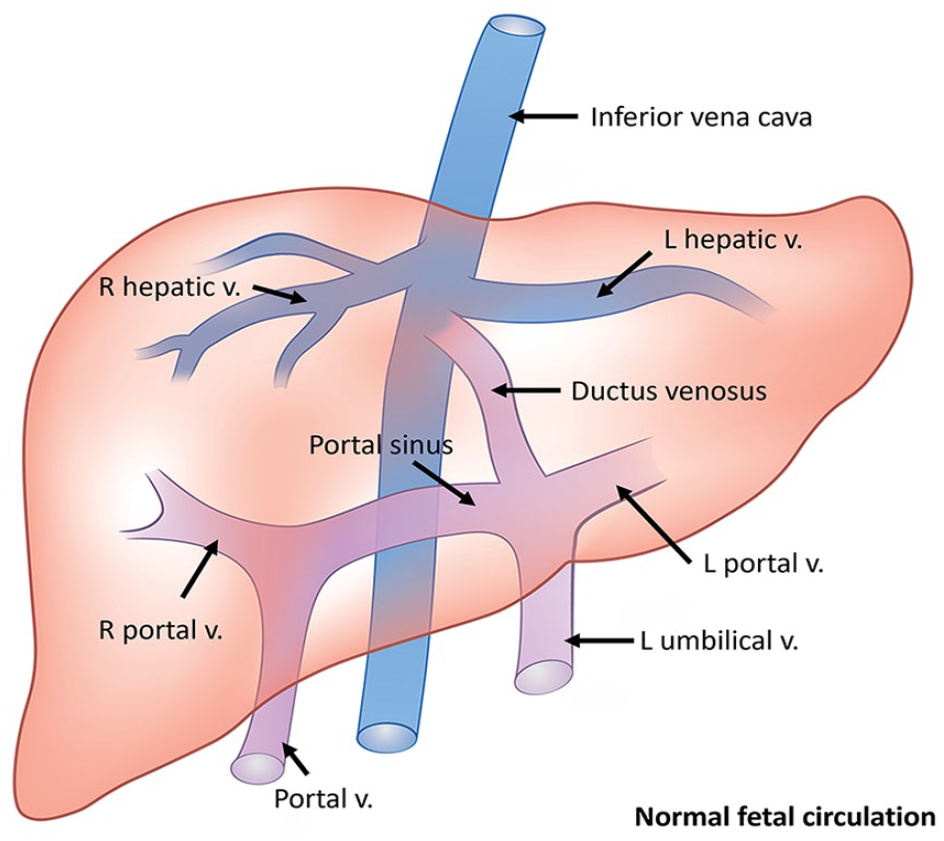
In fetal circulation, oxygenated and nutrient-rich blood from the placenta travels to the fetus via the umbilical vein. A portion of this blood bypasses the liver through the ductus venosus, merging with deoxygenated blood in the inferior vena cava (IVC). The mixed blood from the IVC then enters the right atrium of the fetal heart.
In ADV cases, 2 umbilical vein drainage variants are observed: 7
(1) Extrahepatic umbilical vein drainage with liver bypass: The umbilical vein connects directly to the systemic venous system, bypassing the liver. This can include connections to the iliac vein, inferior vena cava (IVC), renal vein, right atrium, and rarely, the left atrium or coronary sinus.
(2) Intrahepatic umbilical vein drainage without liver bypass: The umbilical vein enters the liver and connects to the portal sinus, but does not give rise to the DV.
The medical literature addressing ADV is primarily centered around fields such as obstetrics, maternal-fetal medicine, and radiology.4,5,19-27 In contrast, a relatively limited number of reports of ADV exist within the realm of pathology literature.28-31
In this report, we present 2 distinct autopsy cases involving fetuses diagnosed with ADV through antenatal imaging studies. Postmortem examinations revealed distinctive findings. In the first case, the fetus exhibited ADV associated with extrahepatic umbilical venous return and a spectrum of other congenital anomalies. Conversely, in the second case, the fetus had a markedly hypoplastic DV and an intrahepatic umbilical venous return. The embryologic differences underlying each case will be discussed.
Case 1
The first case involved the termination of pregnancy at 25 + 5 weeks of gestational age (GA), due to the detection of multiple fetal anomalies through prenatal imaging. The mother, a 20-year-old primigravida, had an unremarkable medical and family history.
The first trimester scan, performed at 12 + 6 weeks GA, revealed no fetal abnormalities. First trimester screening showed a low risk for trisomy 13, 18, and 21. However, the fetal detailed anatomical survey at 20 weeks GA demonstrated a number of fetal anomalies, including a cleft lip, rocker-bottom feet, and an abnormal curvature of the lumbosacral spine. At the 20 + 6 weeks GA follow-up ultrasound, multiple fetal abnormalities were again detected:
(1) Abnormal distal spine: Significant disorganization of the lumbosacral spine with multiple levels of vertebral abnormalities was observed. An anterior spinal defect was suspected, with an echogenic mass noted on the pelvic aspect of the sacral spine.
(2) Horseshoe kidney.
(3) Multiple intrabdominal echogenic foci (appeared to be intrahepatic).
(4) Bilateral rocker-bottom feet.
(5) Left-sided unilateral cleft lip and palate.
(6) Abnormal fetal vascular drainage/abnormal heart: Significantly enlarged right side of the heart was observed, characterized by a dilated right atrium, dilated right ventricle, and enlarged right ventricular outflow tract. Additionally, abnormal venous drainage was noted. A persistent right umbilical vein appeared to drain directly into the right atrium (Figure 2). The left umbilical vein was absent. A single umbilical artery was also noted.
(7) Small stomach.
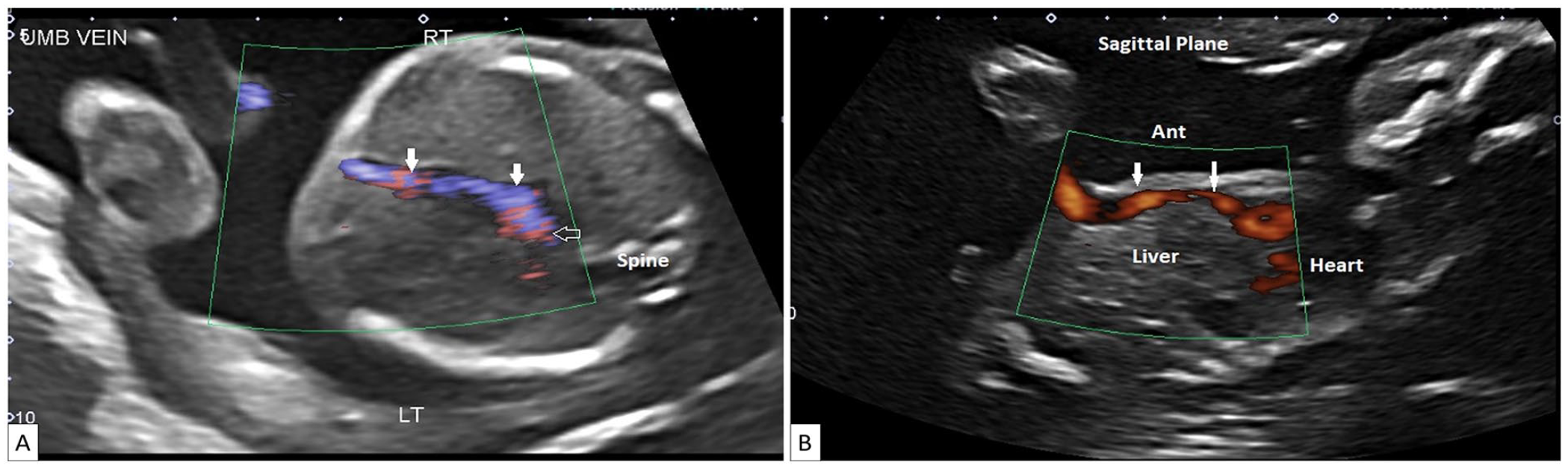
(A) Transverse view through the abdomen reveals a persistent right umbilical vein. The umbilical vein (arrows) drains towards rather than away from the stomach which is on the left side of the abdomen. (B) Parasagittal view of the abdomen and lower chest demonstrating the persistent right umbilical vein (arrows) draining into an enlarged right atrium.
At 21 + 1 weeks GA, an amniocentesis was performed, and cytogenetic analysis (including rapid aneuploidy detection and chromosomal microarray) of the amniotic fluid revealed no genetic abnormalities. Subsequently, a fetal echocardiogram conducted at 23 + 0 weeks GA confirmed the absence of the DV. The umbilical vein was enlarged and could be traced from the cord insertion into the right atrium. Notably, no focal narrowing was observed at the expected location of the DV.
In addition, the fetal echocardiogram revealed no structural cardiac malformations. However, there was global cardiomegaly, with an estimated cardiothoracic ratio of 65%. Moderate enlargement of the right atrium, mild enlargement of the right ventricle, and mild tricuspid insufficiency were observed. Furthermore, mild left axis deviation and mild bilateral ventricular hypertrophy were present, although systolic function remained preserved and normal. The estimated combined cardiac output was 653 ml/minute (140th percentile), significantly elevated compared to the normal range. The estimated median cardiac output for this GA would typically be around half this value.
The parents opted for termination of pregnancy at 25 + 5 weeks GA by induction and consented to a complete autopsy.
At autopsy, a phenotypically male fetus with mild maceration was identified. The fetal measurements were compatible with the stated GA of 25–26 weeks. Several external dysmorphic features were noted, including a cleft palate, a left cleft lip extending into the left nare, mild bilateral mi-crophthalmia, bilateral low-set ears, imperforate anus without a fistula, a skin tag at the location of the imperforate anus, and rocker-bottom feet.
Internal examination revealed the following findings. The heart was enlarged, with dilation of all 4 chambers and hypertrophy of the right ventricle. The absence of DV was confirmed, as the umbilical vein directly drained into the suprahepatic IVC, bypassing the liver (Figure 3). Ad-ditionally, a single right-sided umbilical artery was present. Other findings included a horseshoe kidney (Figure 4A), mild dilation of both ureters, partial malrotation of the small intestine and colon, and high rectal atresia (Figure 4B).
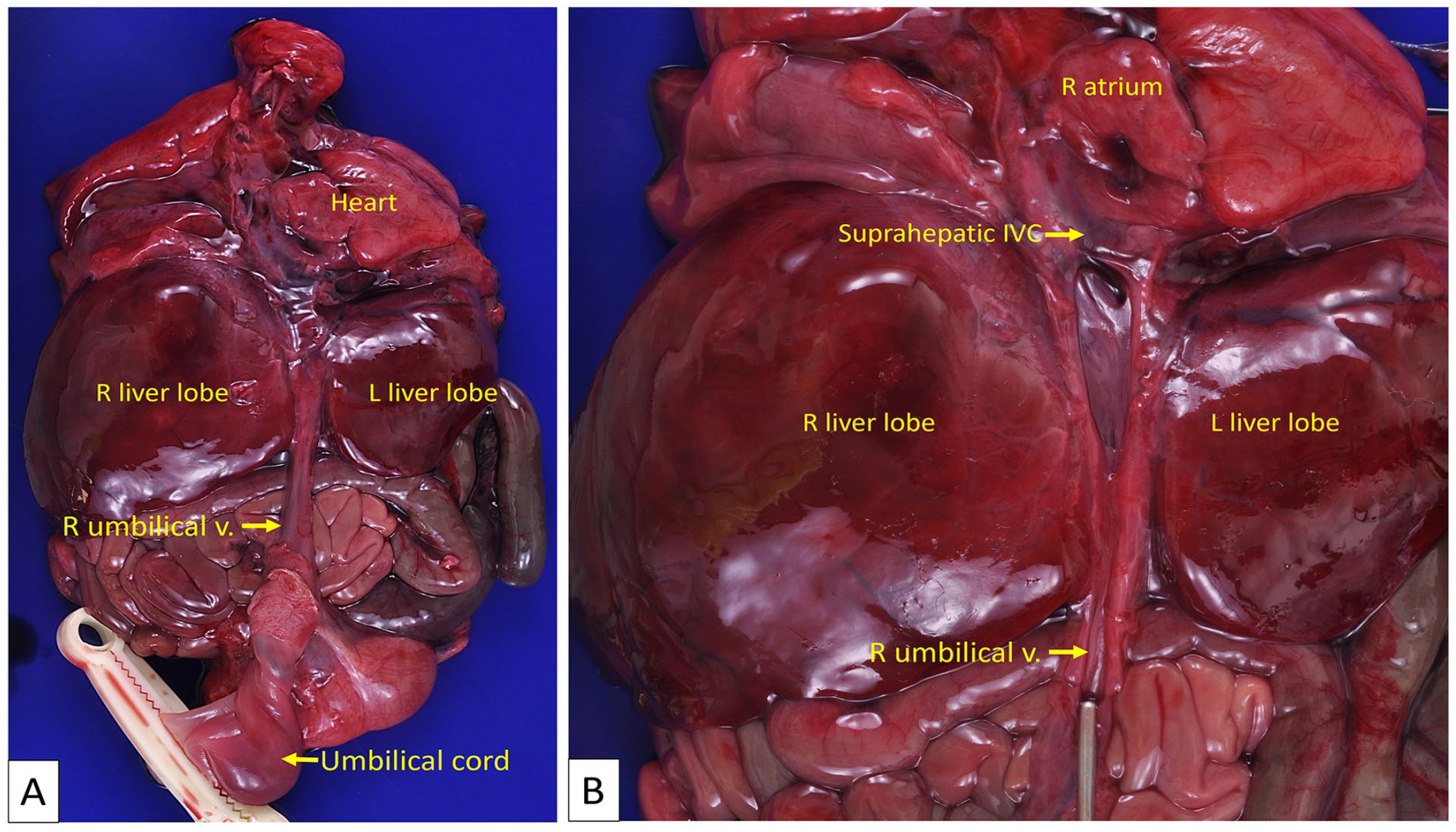
Case 1. (A) Anterior view of the fetal organ block. The right umbilical vein bypasses the liver, running along its external surface to directly connect with the suprahepatic portion of the inferior vena cava (IVC). (B) The umbilical vein is opened, revealing a patent lumen that connects with the suprahepatic IVC, which in turn drains into the right atrium.
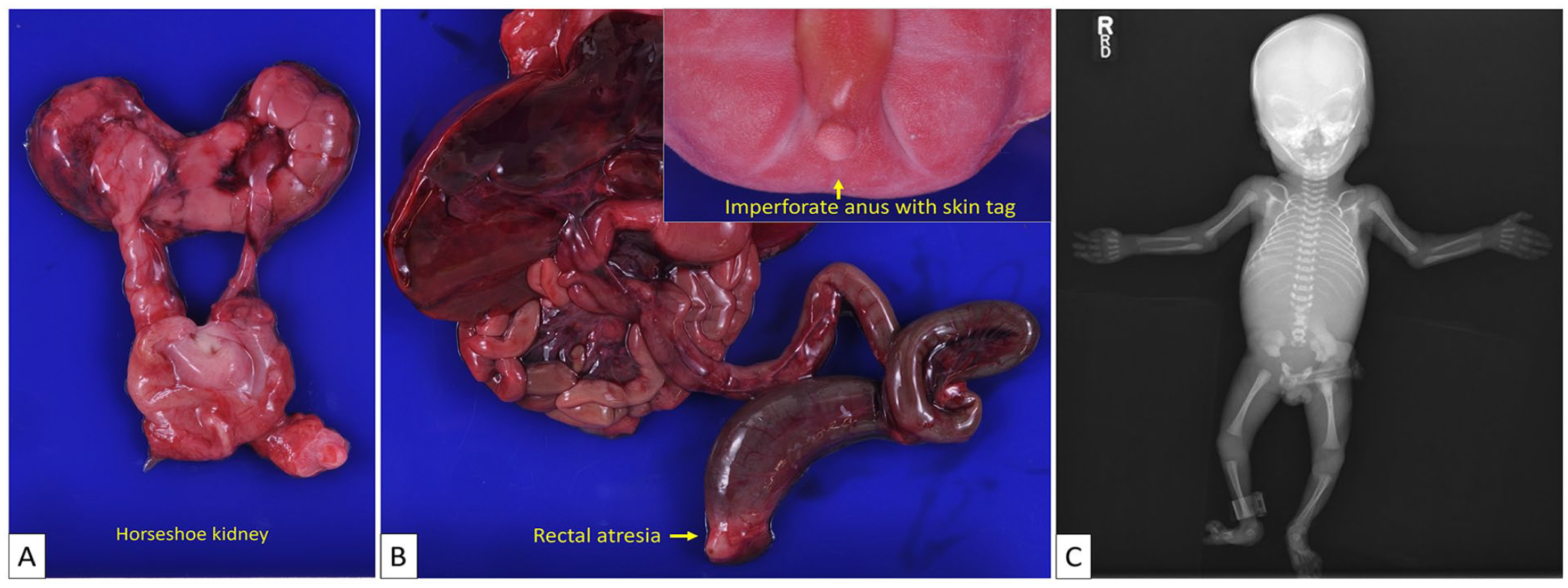
Case 1. (A) A horseshoe kidney with hydronephrosis is present. (B) The fetus has an imperforate anus accompanied by a skin tag (insert), as well as high rectal atresia. (C) A postmortem X-ray corroborates the prenatal ultrasound findings of anomalies in the lumbosacral spine.
In addition, postmortem X-ray revealed lumbosacral bony dysraphism and lumbar fusion anomalies (Figure 4C), which were confirmed by histologic examination. The spinal cord was intact and normally developed. The brain was mostly normal, but potential delayed myelination was noted, with faint myelin in the lumbosacral spinal cord and none in the brainstem.
The placenta was of normal size and morphology but had a 2-vessel umbilical cord; no histologic abnormalities were noted.
Case 2
The second case involved the termination of pregnancy at 24 weeks GA due to severe intrauterine growth restriction and abnormal Doppler findings. The mother, a 40-year-old G2P1, had an unremarkable medical and family history.
An 8 + 4 weeks GA dating ultrasound and a 13 weeks GA first trimester scan showed no fetal anomalies. The first trimester screen was positive for trisomy 21 and negative for trisomy 13 and 18. Subsequent non-invasive prenatal testing indicated a low risk for aneuploidy.
At the 19 + 4 weeks GA detailed anatomy ultrasound, the composite fetal measurements corresponded to 18 + 3 weeks GA. The color Doppler imaging revealed a 2-vessel umbilical cord. No other structural abnormalities were noted.
At the follow-up fetal ultrasound assessment at 23 weeks GA, the fetus measured small for gestational age, with the abdominal circumference in the third percentile. Interval growth had decreased since the last scan, but fetal movements and tone were normal. The umbilical artery showed increased pulsatility index (PI). The middle cerebral artery Doppler measurements showed increased end-diastolic velocity and decreased PI’s, consistent with cerebral redistribution. The DV was suspected to be absent. Doppler interrogation of the uterine arteries showed bilateral early diastolic notches and elevated PI’s. These findings indicated potential placental insufficiency and were associated with an increased risk of early-onset intrauterine growth restriction and pre-eclampsia. The placenta was normal in size and morphology. The amniotic fluid levels were within the normal range.
Fetal echocardiography showed no major heart anomalies but confirmed the absence of the DV. The umbilical vein was observed to drain into the hepatic veins, then into the IVC.
Amniocentesis was performed, and rapid aneuploidy detection on the amniotic fluid revealed rsa(X,13,18,21)x2—female fetus with no evidence of aneuploidy for chromosomes 13, 18, 21, or the sex chromosomes.
At 24 weeks GA, the parents chose to terminate the pregnancy via induction and agreed to a full autopsy.
The autopsy revealed a phenotypically female fetus with moderate autolysis, measuring smaller than expected for 24 weeks GA—more in line with 22 weeks GA in size. Aside from slightly prominent infraorbital creases, no external abnormalities were observed. Internally, a hypoplastic and atretic left umbilical artery was found, while the right umbilical artery appeared normal (Figure 5).
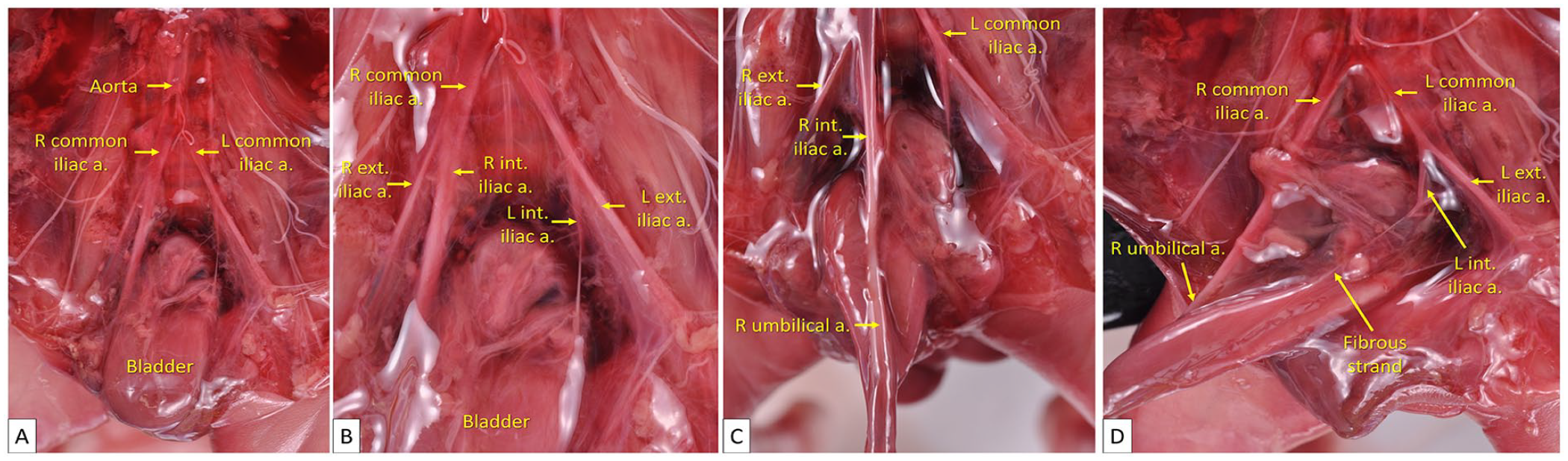
Case 2. (A) The abdominal aorta bifurcates into the right and left common iliac arteries. (B) The right and left common iliac arteries further divide into the external and internal iliac arteries. (C) On the right side, the internal iliac artery extends caudally to form the right umbilical artery. (D) In contrast, the left internal iliac artery is noticeably smaller in caliber. Instead of giving rise to a normally sized left umbilical artery, it becomes a thin fibrous strand.
During the autopsy, the umbilical vein was carefully dissected to trace its path. It entered the liver and drained into the portal sinus, which divided into the right and left portal veins. The umbilical vein continued cranially to a markedly hypoplastic DV, which then merged with the left hepatic vein and ultimately connected to the IVC (Figures 6 and 7).
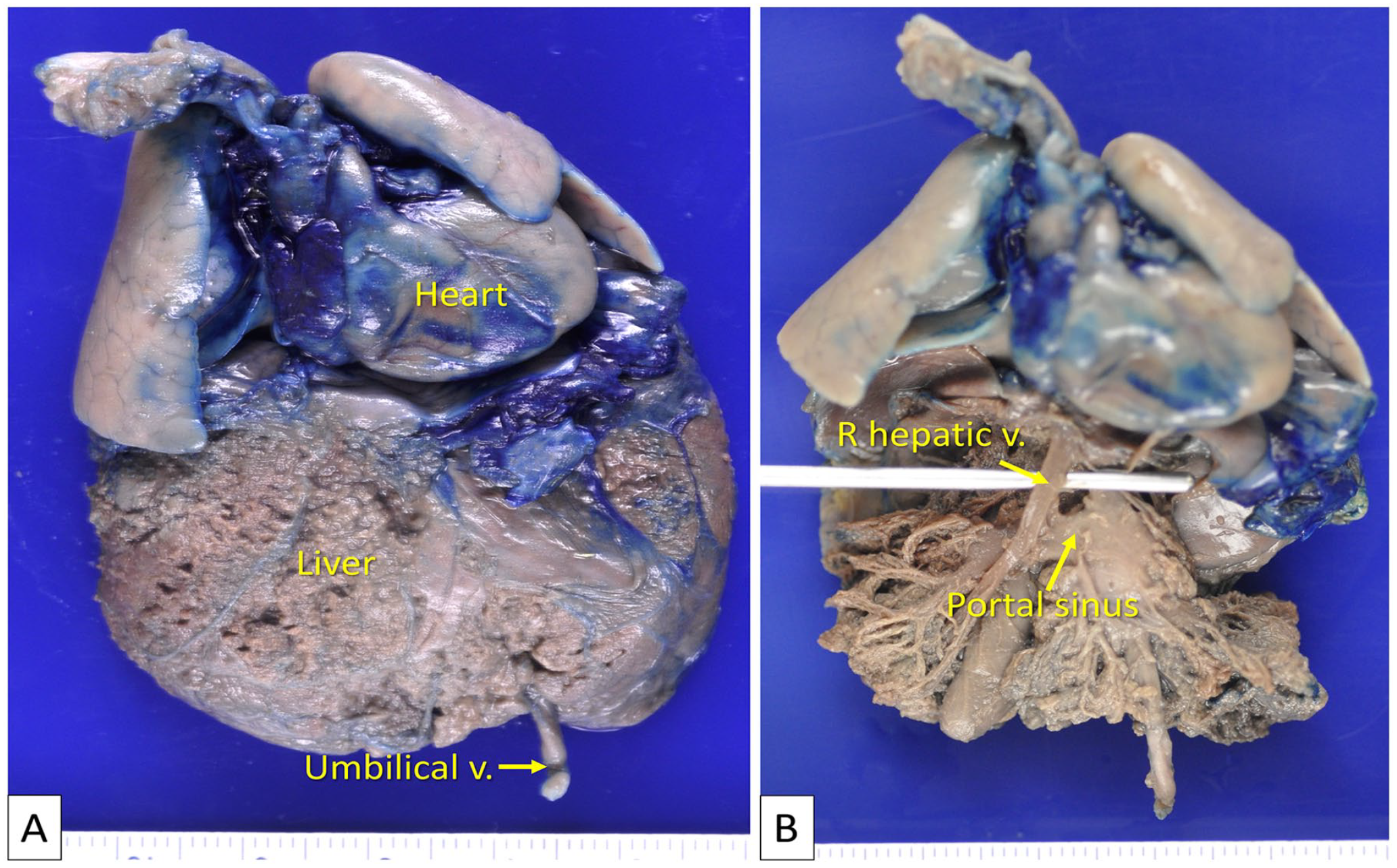
Case 2. (A) Before dissection, the block of organs, which includes the heart, lungs, and liver, is fixed in formalin. The umbilical vein enters the liver as per its usual course. Blue dye is injected into the umbilical vein following formalin fixation, in an attempt to better visualize the intrahepatic vasculature. However, excessive pressure during the dye injection causes the dye to leak, rendering it unhelpful. (B) The liver parenchyma is removed to reveal the underlying vasculature.

Case 2. (A) In this image, the umbilical vein, portal sinus, right and left portal veins, right and left hepatic veins, inferior vena cava (IVC), and the right atrium of the heart are visible. (B) Upon opening the umbilical vein, portal sinus, and the right and left portal veins, it is noted that their vascular lumens are patent and of normal size. The umbilical vein maintains its normal diameter until it reaches the level of the ductus venosus (DV), where it becomes markedly hypoplastic. Additionally, the DV is observed to connect with the left hepatic vein. (C) The diminutive DV opens into the left hepatic vein.
No cardiac abnormalities were noted.
Pathologic examination of the brain revealed a vertical sulcus in the right posterior temporal lobe, bilateral incomplete infolding rotation of the hippocampal formation, and deep cerebellar cortical heterotopias. The cause of these neuropathologic developmental abnormalities was unclear.
The placenta was small, and histology showed evidence of maternal vascular malperfusion, including accelerated villous maturation and decidual vasculopathy. These findings correlated with the abnormal uterine artery Doppler results and could provide an explanation for the intrauterine growth restriction observed in this case.
Discussion
During early human embryologic development, 2 umbilical veins—the right and left—travel through the umbilical cord into the embryo, entering the sinus venosus of the heart. Under normal circumstances, the right umbilical vein starts to atrophy around the fourth week of embryonic development and disappears completely by the seventh week. The segment of the left umbilical vein near the heart regresses too. In contrast, the segment of the left umbilical vein from the umbilicus to the liver remains and divides into the portal sinus and the DV. The DV, which begins to form between the fourth and eighth weeks GA, carries oxygenated blood directly into the IVC and then into the heart.32-36 If the left umbilical vein atrophies and degenerates, then the right umbilical vein persists.
Nasu and Arishima 37 studied the development of DV in fetal rat livers using scanning electron microscopy. They found that by the 13th day of gestation, the vascular system of the fetal rat liver had formed, with the left umbilical vein entering the liver and connecting to the IVC via a small DV.
On the 14th day, a distinct bulge appeared where the umbilical vein met the vitelline vein. This “bulging part,” as termed by the researchers, divided into 2 branches at its apex: a short, small DV connecting to the anterior portion of the IVC and another branch connecting with the vitelline vein.
Between the 15th and 20th days of gestation, the DV grew in both length and diameter. By the 20th day of gestation, some hepatic veins from the left liver lobe opened into the IVC near its connection to the DV. By the 22nd day of gestation, the DV began to show signs of degeneration.
In the present report, we detail prenatal ultrasound and autopsy findings for 2 fetuses diagnosed with ADV through antenatal imaging.
In the first case, prenatal ultrasound at 20 + 6 weeks GA showed a persistent right umbilical vein connected directly to the right atrium, regressed left umbilical vein, and absence of the DV. Autopsy confirmed that the umbilical vein bypassed the liver, connecting directly with the suprahepatic IVC (extrahepatic shunt). The heart was noticeably enlarged and dilated, likely adapting to accommodate the additional blood volume from the placenta, given the lack of partial diversion of placental blood to the fetal liver. Additionally, the fetus had multiple other anomalies, including lumbosacral bony dysraphism, horseshoe kidney, rectal atresia, imperforate anus, partial bowel malrotation, and a single umbilical artery. Interestingly, these anomalies were predominantly in the caudal trunk region, suggestive of a field effect phenomenon.
In a systematic review by Pacheco et al, 27 340 cases of DV agenesis with associated fetal malformations were identified. Among these cases, 82 (24.1%) had cardiomegaly, 24 (7.1%) had a single umbilical artery, 6 (1.8%) had spinal deformities, 5 (1.5%) showed persistent right UV, 4 (1.2%) had imperforate anus, 2 (0.6%) had horseshoe kidneys, and 2 (0.6%) had intestinal malrotation. This highlights the rarity of these fetal malformations in cases of DV agenesis. Additionally, DV agenesis has been reported in the context of various syndromes, including VACTERL, Beckwith-Wiedemann syndrome, Jacobsen syndrome, Pierre-Robin sequence, Smith-Lemli-Opitz syndrome, and Wolf-Hirschhorn syndrome. 27 However, the specific constellation of fetal anomalies observed in our first case has not been reported in a single fetus or neonate and does not correspond to any known syndrome.
Additionally, in a study by Jaeggi et al, 21 12 cases of DV agenesis with extrahepatic umbilical vein drainage were examined. Their findings suggest that most such fetuses did not have additional major malformations, leading to generally favorable postnatal outcomes. Similarly, a study by Moaddab et al 25 involving 259 fetuses with agenesis of DV found that those with extrahepatic shunts largely lacked other structural abnormalities. These observations suggest that the multiple congenital anomalies seen in our case (Case 1) may represent an exception rather than the norm within the population of fetuses with ADV and extrahepatic shunts.
The presence of a persistent right umbilical vein is rare, occurring in just 1 out of 889 births or 0.11%, according to a study by Kumar et al. 38 Most cases (91.3%) showed intrahepatic drainage, with only 8.7% exhibiting extrahepatic drainage. A persistent right umbilical vein can sometimes be associated with severe congenital anomalies. 38 Thus, its diagnosis warrants a comprehensive prenatal evaluation by Maternal-Fetal Medicine (MFM) specialists.
In the second case, the autopsy findings did not corroborate the prenatal imaging findings of an “absent” ductus venosus (DV). Postmortem examination revealed that the DV was present but markedly hypoplastic, likely rendering it non-functional or minimally functional. Instead, it appears that blood from the umbilical vein was channeled through an alternative intrahepatic route via the portal and hepatic veins before reaching the heart. Apart from the presence of a single umbilical artery, the fetus exhibited no other structural anomalies. Placental histology confirmed evidence of maternal vascular malperfusion, which correlated with abnormal uterine Doppler findings and provided a plausible explanation for the observed intrauterine growth restriction.
Additionally, in the second case, the umbilical vein appropriately gave rise to both the DV and the portal sinus. However, the DV appeared to either arrest in development or undergo premature closure, leaving it non-functional or significantly compromised. The presence of a discernible, albeit diminutive, DV implies that the developmental insult likely occurred post-formation of the DV—after fourth to eighth week GA. Additionally, the absence of a persistent right umbilical vein in this case provides further evidence that the left umbilical vein maintained a functional connection to the heart during early gestational stages. Had this not been the case, the right umbilical vein would have persisted, rather than undergoing the usual process of degeneration.
The presence of a DV in the second case implies that in situations where blood flow through the DV is undetectable by prenatal imaging, the use of terms like “agenesis” or “absence” may warrant reconsideration. In our second case, the autopsy revealed a hypoplastic DV, indicating limited functionality rather than a complete absence or agenesis of the structure.
The exact cause of ADV remains uncertain. There are hypotheses suggesting primary agenesis as well as functional or structural closure as the etiology of ADV. 7 While the first case in this report supports the idea of primary agenesis as the underlying cause, the second case introduces the possibility that functional or structural stenosis or atresia can also give rise to an “absent” ductus venosus by imaging. In addition, our second case appears to be the first reported case of a hypoplastic DV in a fetus.
Anomalies involving the DV are uncommon, rendering the insights gained from antenatal ultrasounds essential for pathologists during postmortem examinations. Without the diagnostic insights provided by MFM specialists, pathologists might overlook DV anomalies during autopsy. Thus, the cases presented here highlight the importance of effective communication between MFM specialists and pathologists.
These 2 cases also emphasize another important aspect: even though both fetuses were diagnosed with ADV through prenatal ultrasound and Doppler, they exhibited 2 distinct conditions likely stemming from distinct embryologic origins. The progress in prenatal imaging has greatly enhanced our ability to diagnose different conditions before birth. Nonetheless, these cases underscore the importance of careful assessment during fetal autopsy, which can reveal valuable insights and further add to our understanding of these uncommon congenital anomalies.
Footnotes
Acknowledgements
The authors would like to thank Charlotte Monroe for her invaluable administrative assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
