Abstract
Background:
Traditional open surgical repairs, including flap techniques, are associated with high recurrence rates and significant morbidity. Robotic-assisted techniques have emerged as promising alternatives to conventional methods due to improved visualization, precision, and reduced postoperative complications. The robotic transanal minimally invasive surgery (R-TAMIS) technique offers a new route for rectovaginal fistula (RVF) repair, providing enhanced dexterity and access through the transanal approach.
Methods:
An R-TAMIS technique was employed for same-day repair of RVF using the da Vinci Xi® system. After bowel preparation and prophylactic antibiotics, the patient was positioned in prone Jackknife. The GelPOINT Path was used for transanal access, and robotic ports were placed. The fistula tract was identified and dissected circumferentially. The vaginal wall was closed with absorbable barbed sutures, reinforced with fibrin sealant and acellular dermal mesh. The rectal wall was then closed, and patency was confirmed via endoscopic inspection.
Results:
The procedure was well tolerated, and the patient was discharged the next day. Minimal postoperative pain was reported but didn’t require analgesics. No major complications were observed in the immediate postoperative period. The robotic approach provided enhanced visualization and dexterity, facilitating precise dissection and suturing. The technique has already been successfully applied to benign RVFs and select rectourethral fistulas.
Conclusion:
Robotic-assisted transanal repair using the R-TAMIS technique is a feasible and effective minimally invasive option for selected patients with benign RVFs. With proper patient selection and dedicated surgical technique, this approach may reduce morbidity and improve outcomes compared to traditional open or laparoscopic methods.
Keywords
Introduction
Rectovaginal fistulas (RVFs) are abnormal epithelialized tracts between the rectum and vagina, resulting in fecal incontinence, recurrent infections, and significant psychological distress.1,2 These fistulas often arise from obstetric trauma, inflammatory bowel disease, pelvic radiation, surgical complications, or malignancy.1,2 The management of RVFs remains a surgical challenge due to their complex anatomical location, high recurrence rates, and the delicate nature of surrounding tissues. 2
Traditional surgical approaches—including transvaginal, transanal, transperineal, and transabdominal techniques often involve tissue flap interposition, such as Martius, gracilis, or rectal advancement flaps. 3 While these methods have demonstrated efficacy, they are associated with considerable morbidity and prolonged recovery.1,4 Moreover, radiation-induced and Crohn’s-related fistulas increase the level of complexity, often requiring diverted procedures. 5
Laparoscopic repair, with or without flap reinforcement, since the emergence of minimally invasive techniques as promising alternatives, has shown improved outcomes in selected cases. 6 However, limitations in instrument dexterity and visualization in deep pelvic spaces can hinder optimal repair. Robotic-assisted surgery addresses these limitations by offering enhanced three-dimensional visualization, tremor filtration, and wristed instrumentation, which facilitates precise dissection and suturing in confined anatomical regions.6,7
Robotic transanal minimally invasive surgery (R-TAMIS) represents a significant advancement in RVF management. Originally developed for excision of mitigating the technical challenges during excision of rectal neoplasia, 8 R-TAMIS has been adapted for fistula repair, offering ergonomic advantages and superior access to low rectal lesions. Studies have demonstrated its feasibility and safety in treating rectourethral and RVFs, with reduced complication rates and improved margin clearance compared with laparoscopic TAMIS.7–9
Recent literature supports the use of robotic platforms for complex pelvic fistulas, including simultaneous repairs involving vesicourethral or bladder neck structures.7–10 Additionally, endoscopic techniques such as over-scope clip systems and stenting are being explored for select cases, though their long-term efficacy remains under investigation. 11
This article presents our institutional methodology for same-day robotic-assisted transanal repair of benign RVFs using the da Vinci Xi® system. We detail patient selection criteria, preoperative preparation, surgical technique, and postoperative care. Our experience suggests that R-TAMIS offers a safe, effective, and minimally invasive alternative to traditional approaches, particularly for low-lying, non-radiation-induced fistulas.
Preoperative Considerations
Patient selection
Ideal candidates include those with: Low RVF (lower third of the rectum) Small to moderate fistula size No bowel necrosis or abscess Adequate rectal wall thickness No significant pelvic adhesions No prior pelvic irradiation
Bowel preparation
Two rectal enemas are standard, though full mechanical bowel prep (e.g., polyethylene glycol) is also acceptable.
Surgical technique
The patient is placed in the prone Jackknife position (as shown in Fig. 1). Consider other positions such as modified lithotomy (Lloyd Davis), as shown in the Figure 2, for obese patients when difficulties are anticipated with visualization. Caution is advised; however, due to more challenging positioning in these cases. Other positions such as right or left Decubitus positions (shown in Fig. 3) could be considered as well according to the surgeon’s discretion. Prep and drape. Dilate the anus, typically with GelPOINT Path (Index Figs. 4 and 5) with dilator with generous lubrication. TAMIS port to be placed and secured in place with sutures.
The anus is dilated using the GelPOINT Path with generous lubrication. The TAMIS port is inserted and secured with sutures. Robotic setup includes one bariatric 8 mm port and two standard ports, offset to minimize external collisions.
Three robotic ports are inserted (1 bariatric 8 mm, 2 standard). Instruments include a 30° scope, scissors, Maryland dissector, monopolar hook, and graspers. A Lone Star retractor may be used in obese patients to enhance visualization.
The fistula tract is identified with the robotic camera, grasped, and traction is applied. Dissection begins at the edges and proceeds circumferentially to create a full-thickness flap. Dissection continues through the rectal wall to mobilize perirectal fat in the anterior rectal region. Full separation of rectal and vaginal walls is not mandatory but should allow tension-open-access closure. Sharp and blunt dissection techniques are used to minimize collateral damage.
The vaginal defect is closed using absorbable barbed sutures in a running fashion. Interrupted sutures (3-0 barbed on RB1 needle) may be used as an alternative. Hemostasis is carefully maintained.
Fibrin sealant or similar biocompatible material is applied to the sutured vaginal wall. An acellular dermal mesh is placed to reinforce the repair. Equivalent materials may be used.
The rectal wall is then closed with absorbable sutures (barbed 3/0 RB1) in a continuous manner.
A scope is advanced beyond the repair site to confirm rectal patency. The surgical field is inspected for bleeding or complications. The transanal port is removed, and the rectum is digitally examined.
Patients are typically discharged the next day; same-day discharge is feasible depending on comorbidities and case complexity. Most patients tolerate the procedure well, often without the need for analgesics. Stool softeners are recommended to prevent constipation and rectal overdistension. Caution is advised for large fistulas. This technique may also be considered for select rectourethral fistulas.

Positioning of Patients: Jackknife position -preferred by most surgeons for non-obese patients.

Positioning of Patients: Modified Lithotomy (Lloyd Davis)—preferred by most surgeons for obese patients.
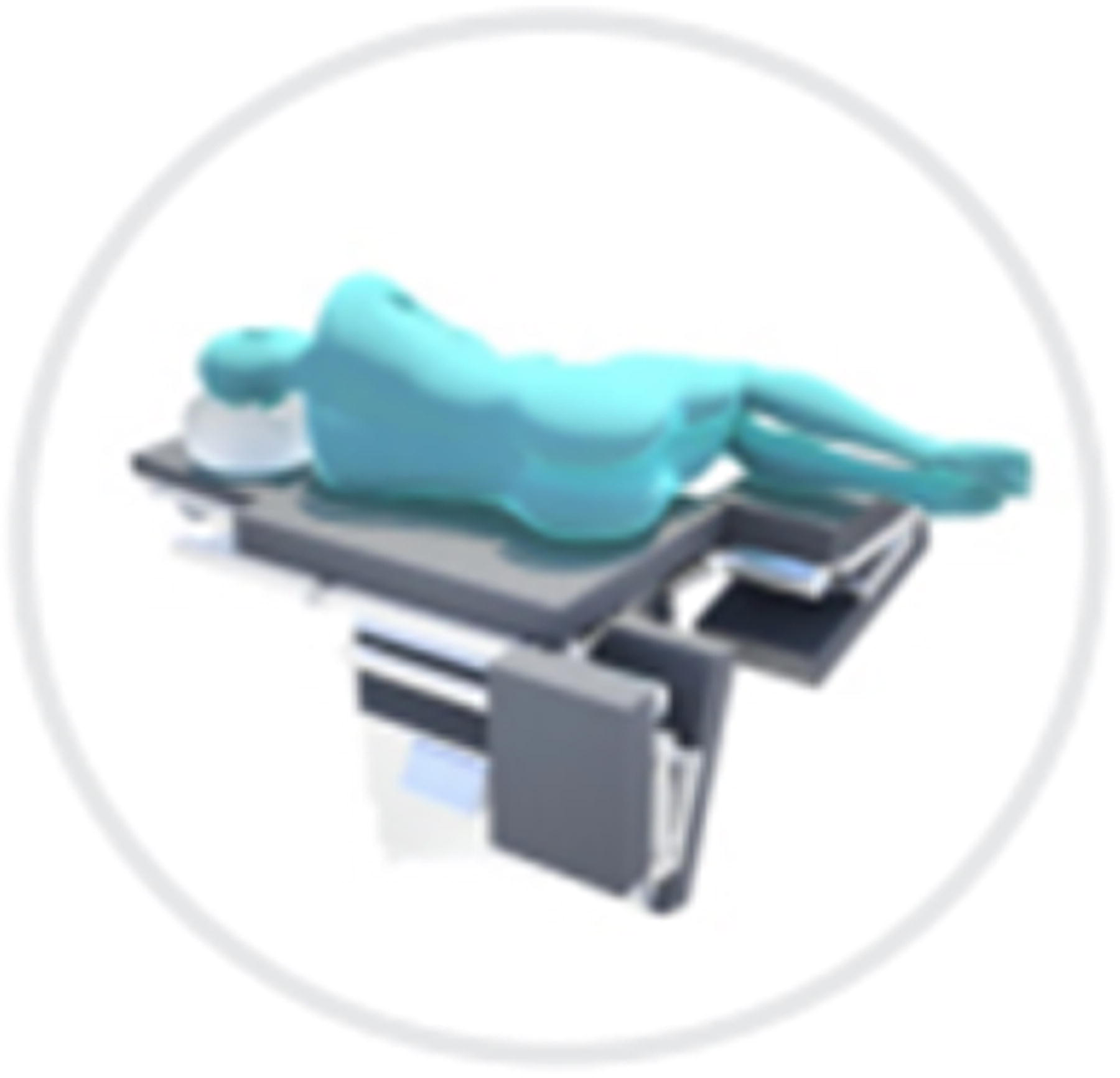
Positioning of Patients: Left Decubitus -an alternative that is preferred by some surgeons for both obese and non-obese patients.

GelPOINT Transanal Access Platform facilitates minimally invasive rectal procedures. Courtesy of www.appliedmedical.com.
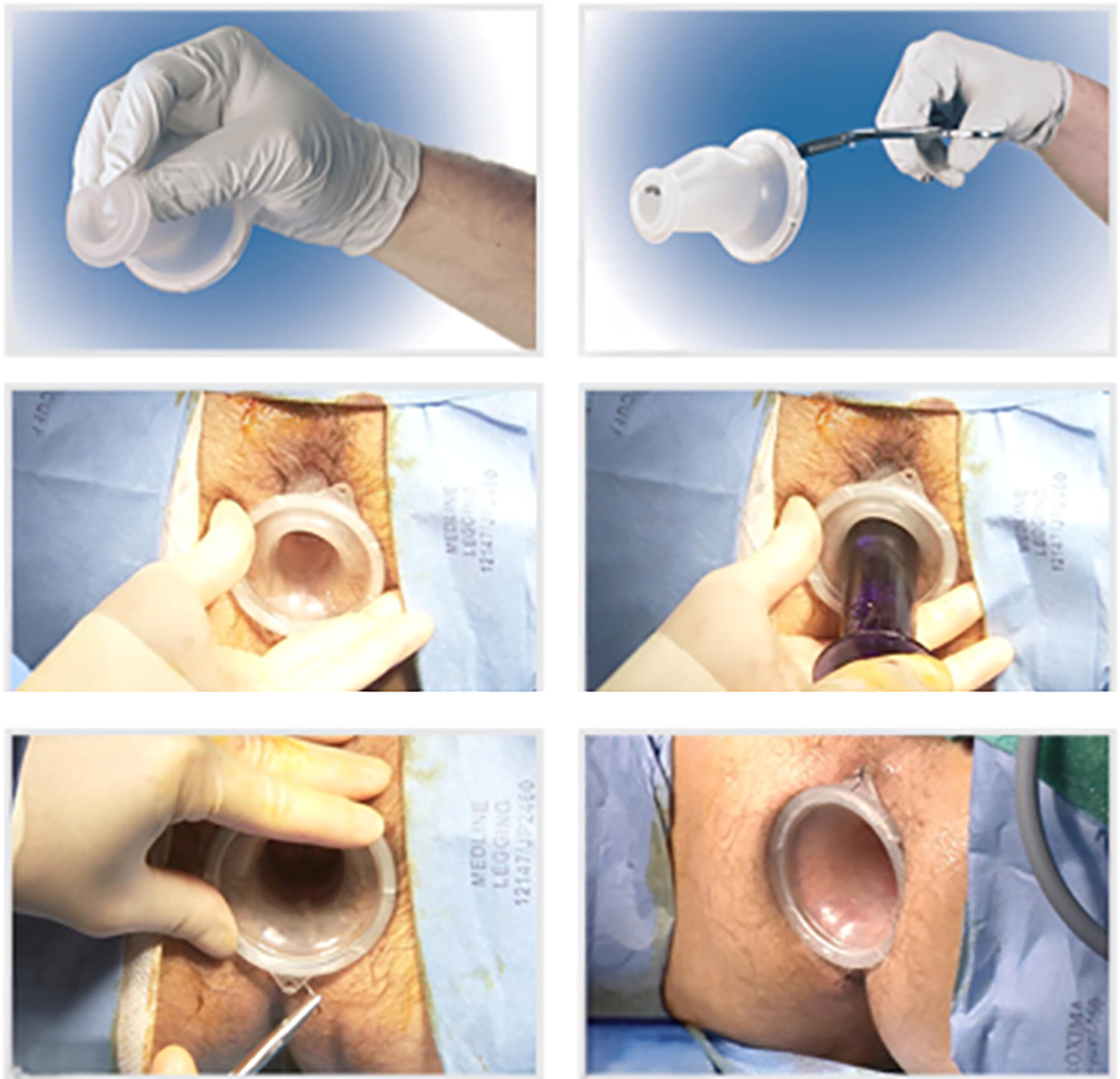
GelPOINT Steps of inserting and fixating the port. Courtesy of www.appliedmedical.com.
Advantages of Robotic-Assisted Approach
Enhanced Visualization: 3D high-definition imaging improves depth perception and magnification. Improved Dexterity: Wristed instruments allow precise dissection and suturing, especially near the fistula opening. Minimally Invasive: The transanal approach reduces surgical trauma, complications, and postoperative pain.
Limitations
Best suited for benign etiologies; not recommended for large, malignant, or radiation-induced fistulas. Learning Curve: Despite a faster learning curve than laparoscopic or open techniques, adoption is limited to specialized centers.
Conclusion
Robotic-assisted transanal repair of RVFs using the SP R-TAMIS technique offers a promising, minimally invasive option for selected patients. With appropriate patient selection and meticulous technique, this approach may improve outcomes and reduce morbidity compared to traditional methods.
Authors’ Contributions
S.M.S.: Conceptualization, methodology, investigation, writing—original draft, visualization, supervision. J.Z.: Surgical procedure, writing—review and editing. A.A.: Surgical procedure, supervision, validation. All authors have read and approved the final article and agree to be accountable for all aspects of the work.
Footnotes
Acknowledgments
The authors would like to thank the surgical team and staff at AdventHealth Tampa for their support and collaboration during the development and implementation of the R-TAMIS technique. Special appreciation is extended to the AdventHealth Research Institute for providing institutional resources and guidance throughout this project.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this study.
