Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of 14 alkyl amide MIPA ingredients as used in cosmetics. All of these ingredients are reported to function in cosmetics as a surfactant – foam booster and/or viscosity increasing agent. The Panel considered the available data, as well as data on read-across sources, and concluded these ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment when formulated to be non-irritating.
Keywords
Introduction
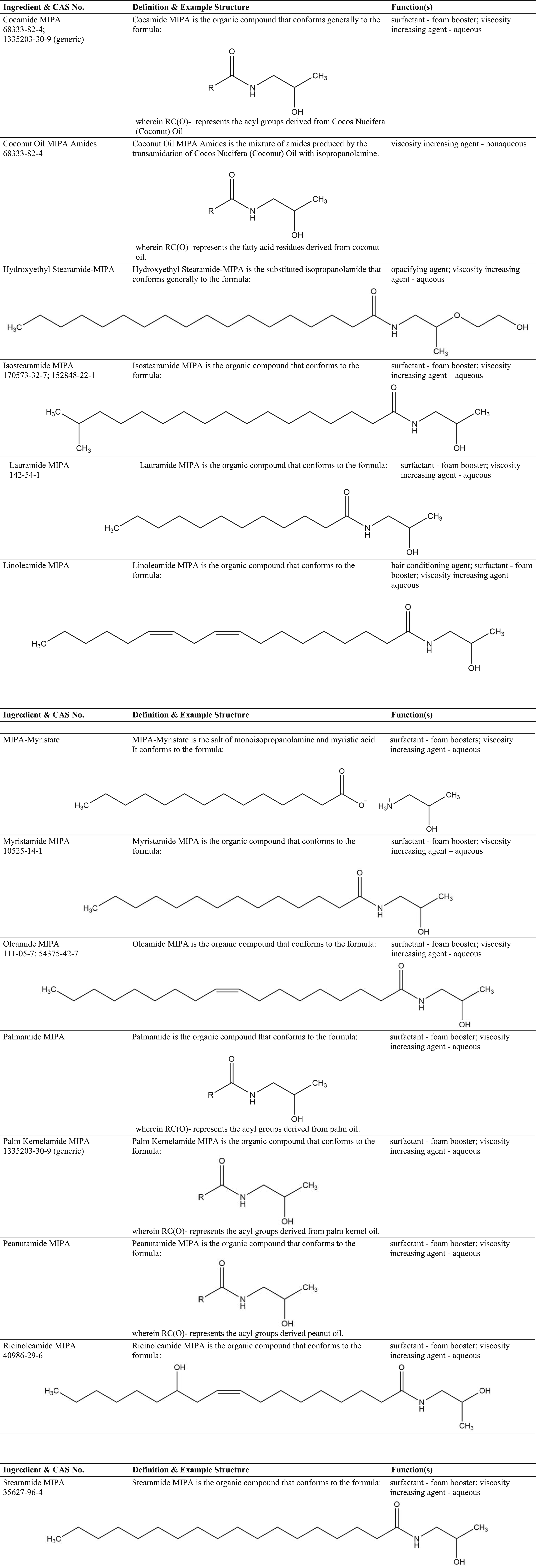
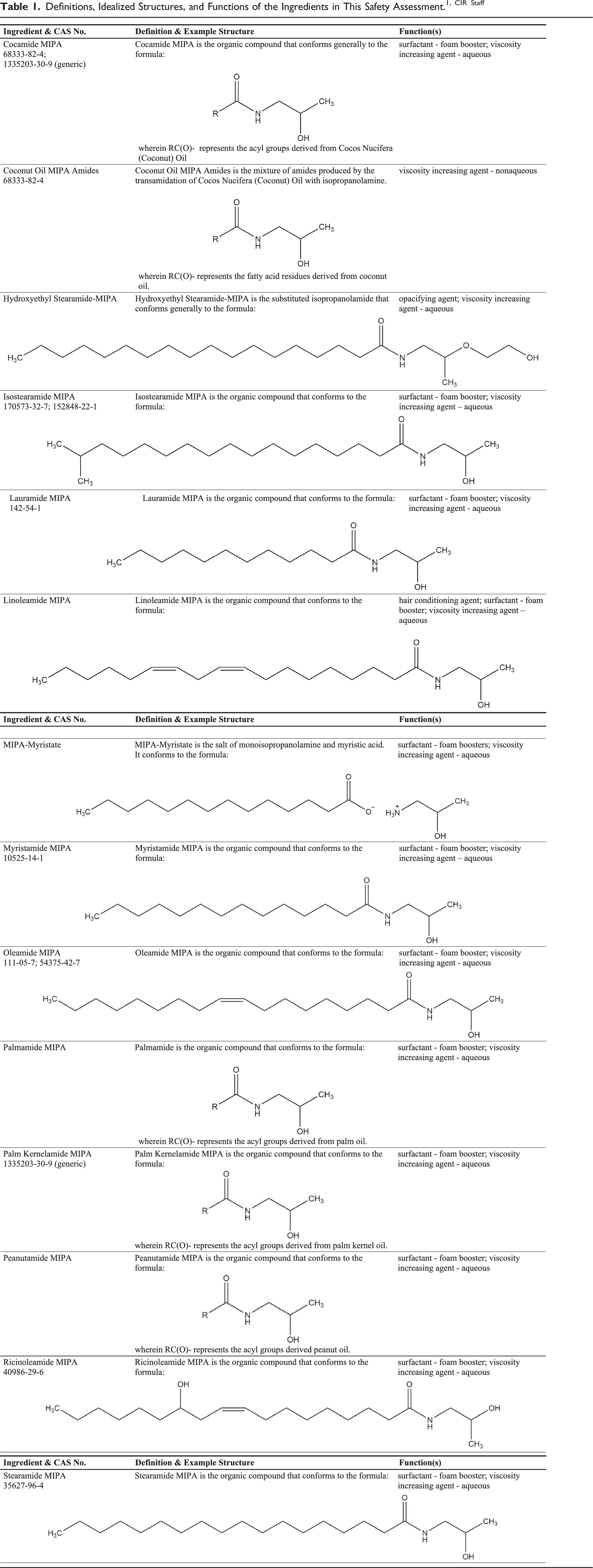
The safety of the following 14 alkyl amide MIPA ingredients as used in cosmetics is reviewed in this safety assessment:
Cocamide MIPA
Coconut Oil MIPA Amides
Hydroxyethyl Stearamide-MIPA
Isostearamide MIPA
Lauramide MIPA
Linoleamide MIPA
MIPA- Myristate
Myristamide MIPA
Oleamide MIPA
Palmamide MIPA
Palm Kernelamide MIPA
Peanutamide MIPA
Ricinoleamide MIPA
Stearamide MIPA
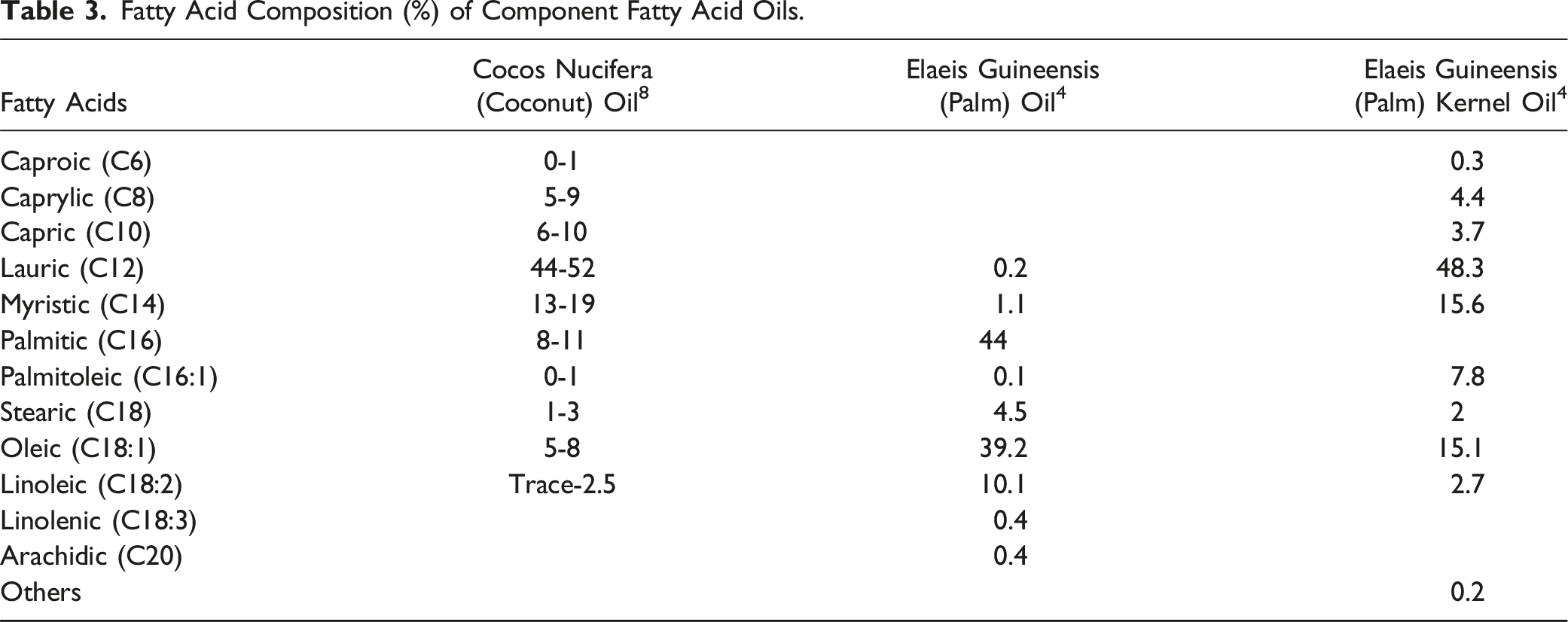
Definitions, Idealized Structures, and Functions of the Ingredients in This Safety Assessment.1, CIR Staff
The rationale for this grouping of alkyl amide MIPA ingredients stems from the fact that each of the ingredients is a mixture of isopropanolamides of a simple carboxylic acid. These ingredients are classic surfactants and viscosity increasing agents.
CIR Conclusions of Components of the Alkyl Amide MIPA Ingredients That Were Previously Reviewed.

The Panel also has reviewed the safety of another group of ingredients that are structurally similar to the alkyl amide MIPA ingredients. In 2013, the Panel published a safety assessment of diethanolamides as used in cosmetics; the Panel concluded that diethanolamides are safe in the present practices of use and concentration [as described in that safety assessment] when formulated to be non-irritating, and when the levels of free diethanolamine (DEA) in the diethanolamides do not exceed the present practices of use and concentration of DEA itself. 15 The Panel cautioned that diethanolamides should not be used in cosmetic products in which N-nitroso compounds can be formed.
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Much of the data in this report was obtained from robust summaries of data submitted to the European Chemical Agency (ECHA) as part of the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) chemical registration process.16–18 When appropriate, information from these summary documents has been included in this report and is cited to ECHA. It should be noted that some of the information pertains to similar compounds, and has been included for read-across to address gaps in information on the alkyl amide MIPA ingredients. Furthermore, results from a National Toxicology Program (NTP) technical report on coconut oil acid diethanolamine condensate (i.e., Cocamide DEA) 19 were included in an ECHA dossier, 18 and for those studies, ECHA and NTP are cited.
Chemistry
Definition and Structure
The ingredients reviewed in this report are the fatty amides resulting from the amidation of fatty acids with MIPA (Figures 1 and 2). MIPA. Alkyl amide MIPA ingredients (generic) and an example (Lauramide MIPA).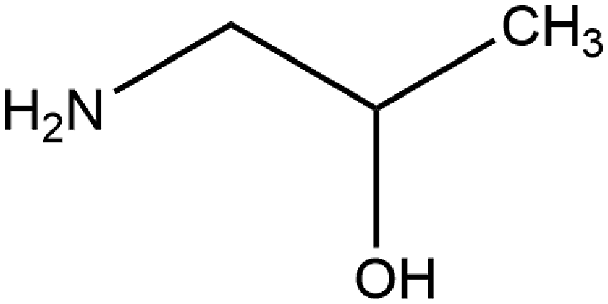
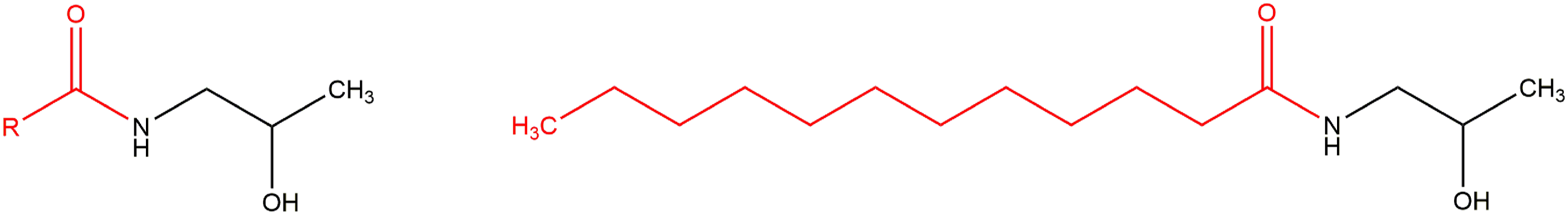
However, two ingredients in this group deviate from this structure pattern. One is further substituted at MIPA (Figure 3), while the other is the MIPA salt of a fatty acid (Figure 4). Specifically, Hydroxyethyl Stearamide-MIPA is substituted with 2-ethanol. MIPA-Myristate, on the other hand, is the MIPA salt of myristic acid. MIPA-Myristate would be the direct amidase metabolite of Myristamide MIPA. Hydroxyethyl Stearamide-MIPA. MIPA-Myristate.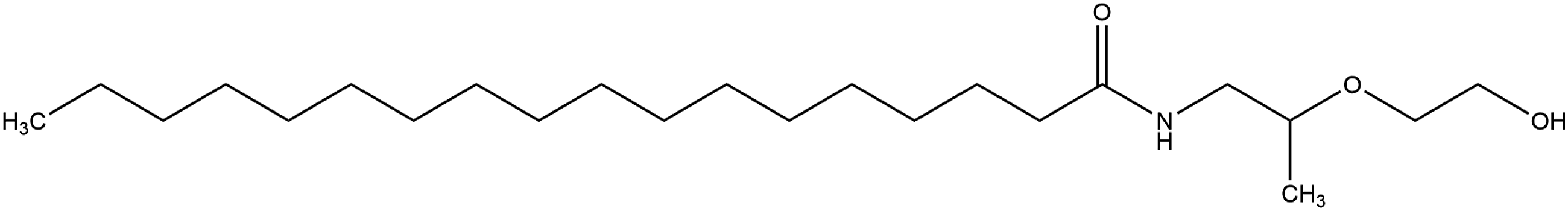
Chemical Properties
Physical and Chemical Properties.

Method of Manufacture
Alkyl amide MIPA ingredients are generally manufactured by the reaction of a fatty acid source (i.e., free fatty acids, fatty acid methyl esters, or triglycerides) with MIPA at elevated temperatures. 20 The fatty acid source determines the alkyl chain distribution. Given the natural origin of fatty acids, the alkyl chains are even-numbered.
Impurities
Typical impurities/residues contained in alkyl amide MIPA ingredients are free MIPA (≤2%) and free fatty acid source (≤5%). 20 Glycerol (≤5%) may be present if triglycerides are used in feedstock.
Cocamide MIPA
Cocamide MIPA (96% minimum) contains monoisopropylamine (2% max) and methanol (<1%). 21 In studies described later in this report, Cocamide MIPA (98.38% pure) was reported to contain 0.88% water and 0.74% free amine.17,18
Use
Cosmetic
The safety of the cosmetic ingredients addressed in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetic industry in response to a survey, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
Frequency and Concentration of Use Data for Alkyl Amide MIPA Ingredients.
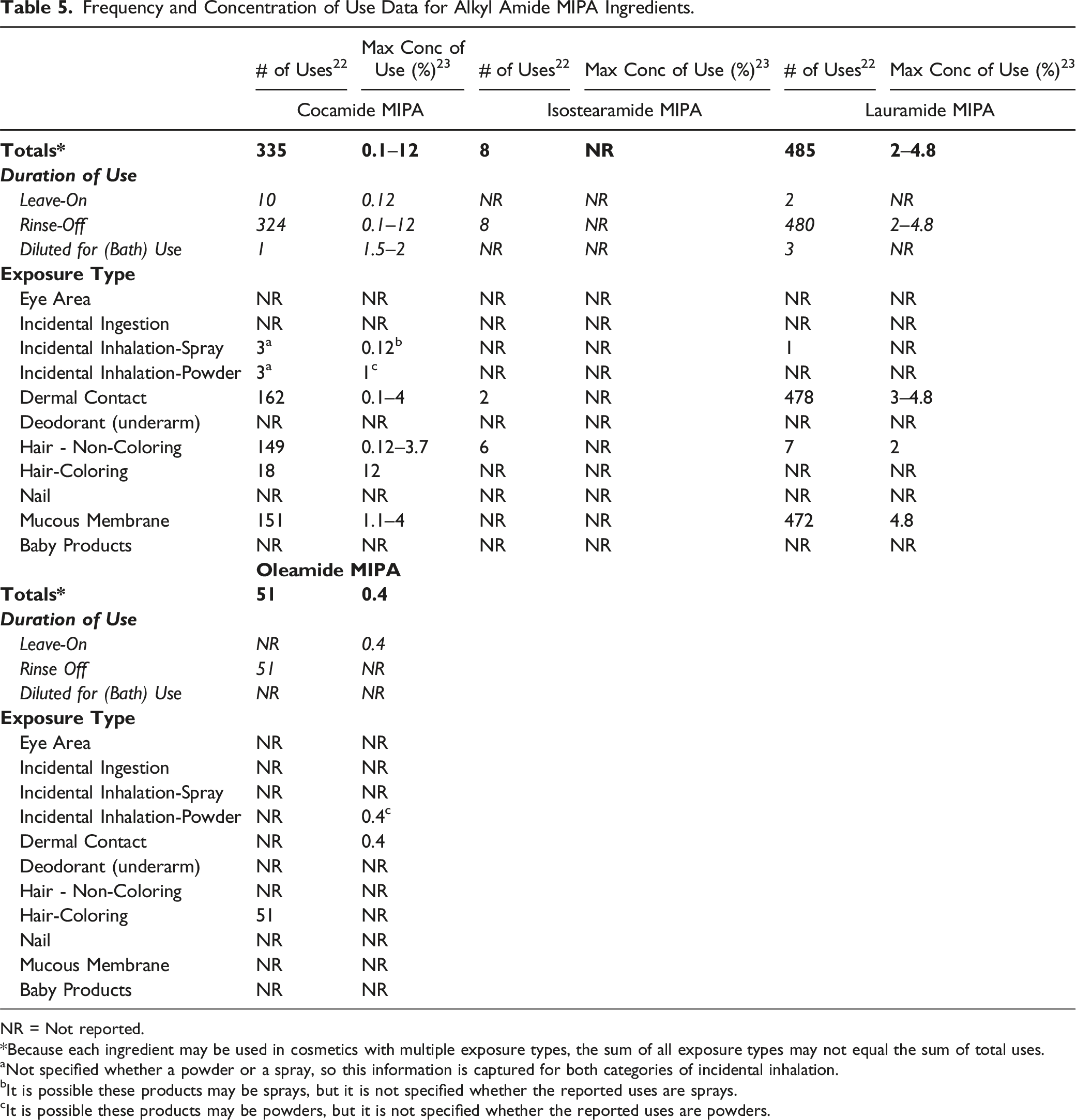
NR = Not reported.
*Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
aNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
bIt is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
cIt is possible these products may be powders, but it is not specified whether the reported uses are powders.
A few of the ingredients included in this safety assessment are reported to be used in products that come into contact with mucous membranes. For example, Lauramide MIPA is used in bath soaps and detergents at up to 4.8%, and Cocamide MIPA is used in bath soaps and detergents at up to 4%. 25
Of the 14 alkyl amide ingredients named in the report, 12 are not restricted from use in any way under the rules governing cosmetic products in the European Union. 26 MIPA-Myristate is included in Annex III, under the category “monoalkylamines, monoalkanolamines and their salts”; this category of ingredients is included in the list of substances which cosmetic products must not contain, except subject to the restrictions and conditions laid down. 27 Accordingly, monoalkylamines, monoalkanolamines and their salts are allowed a maximum secondary amine content of 0.5% in finished product; are not to be used with nitrosating agents; must have a minimum purity of 99%; a maximum secondary amine content of 0.5% in raw materials; and a maximum nitrosamine content of 50 μg/kg. Additionally, Peanutamide MIPA is associated with reference #306 in Annex III, as a peanut oil extract/derivative; the maximum concentration of peanut protein allowed in peanut oil is 0.5 ppm.
Non-Cosmetic
In the US, MIPA is allowed as an indirect food additive as a component of adhesives [21 CFR 175.105] and as a defoaming agent used in the manufacture of paper and paperboard [21CFR176.210].
Toxicokinetic Studies
Toxicokinetics studies were not found in the published literature. Unpublished data were not submitted.
Toxicological Studies
Acute Toxicity Studies
Dermal
Cocamide MIPA
The acute dermal LD50 of Cocamide MIPA was reported to be >2000 mg/kg in rabbits. 21 No further details were provided.
Isostearamide MIPA
The acute dermal toxicity of Isostearamide MIPA (100% pure) was determined using 5 male and 5 female HanIbm: WIST (SPF) rats in accordance with the Organization for Economic Cooperation and Development (OECD) test guideline (TG) 402. 17 Single semi-occlusive patches containing 2000 mg/kg Isostearamide MIPA (0.5 g/ml in PEG; 4 ml/kg) were applied for 24 h. No clinical signs were observed, and the LD50 was >2000 mg/kg.
Oleamide MIPA
The acute dermal toxicity of Oleamide MIPA was determined using five female and five male Sprague-Dawley rats. 16 Rats were dermally administered 2000 mg/kg of Oleamide MIPA. The application site was covered by a semi-occlusive dressing for 24 h. Each animal was observed for 15 d after treatment. In females, moderate to severe erythema was noted at the application site in 3/5 females on day 2. Well-defined erythema was observed in 5/5 females from day 2 or 3 until day 5, which turned into very slight erythema in 3/5 females on day 6 and in 2/5 females from day 6 until day 8. A slight dryness of the skin was also noted at the application site in 5/5 females from day 3 until day 6 or 7. In males, well-defined or very slight erythema was noted at the application site of all males, from day 2 up to day 6. No unscheduled deaths occurred during the study and no clinical signs indicative of systemic toxicity were observed in any animals. The dermal LD50 of the test article was >2000 mg/kg in rats.
Cocamide MIPA (Test Substance – Amides, C8-18 and C18-unsatd., N-(Hydroxyethyl), for Read-Across)
In a limit test that was performed in a manner similar to OECD TG 402, a single application of 2000 mg/kg amides, C8-18 and C18-unsatd., N-(hydroxyethyl) in polyethylene glycol (PEG) was made to 5 male and 5 female Hanlbm:WIST (SPF) rats. 18 Duration of the application and type of coverage was not stated. The LD50 was >2000 mg/kg.
Oral
Cocamide MIPA
The acute oral LD50 of Cocamide MIPA was reported to be >2000 mg/kg in rats. 21 No further details were provided.
Isostearamide MIPA
The acute toxicity of Isostearamide MIPA (94.1% pure) was determined according to OECD TG 401 using groups of 5 male and 5 female Sprague-Dawley rats. 17 The animals received a single dose of 2006 mg/kg bw by gavage (2.18 m/kg bw), and the oral LD50 was determined to be >2006 mg/kg bw.
Oleamide MIPA
An acute oral toxicity study was performed according to OECD TG 423. 16 Oleamide MIPA in corn oil was administered once by gavage to two groups of three female Sprague-Dawley rats at a dosage volume of 10 ml/kg. All animals were observed for 15 d after treatment. All animals survived until study termination. A lower body weight gain was noted in 1/6 females between days 1 and 8 and in 2/6 females between days 8 and 15. In addition, an overall lower body weight gain was observed in 1/6 females between days 1 and 15. There were no macroscopic post-mortem observations. No evidence of toxicity was observed. The oral LD50 of the test article was >2000 mg/kg.
Short-Term Toxicity Studies
Oral
Isostearamide MIPA
Groups of 5 male and 5 female Wistar rats were dosed by gavage with 0, 50, 200, or 1000 mg/kg bw/d Isostearamide MIPA in PEG 300 for 28 d in accordance with OECD TG 407. 17 An additional 5 rats/sex at the 0 and 1000 mg/kg bw/d were treated for 28 d, followed by a 14-d treatment-free recovery period to determine reversibility of effects. Clinical signs, food consumption, and body weights were recorded throughout the study. Functional observational battery, locomotor activity, and grip strength were performed during week 4. At the end of the dosing and the treatment-free recovery period, blood samples were withdrawn for hematology and plasma chemistry analyses. All animals were killed and necropsied; weights of several organs (including the testes) were determined. Microscopic examinations were performed on numerous organs (including the testes and ovaries) and tissues from all control and high dose animals, and on all gross lesions from all animals. Livers of animals of the low and mid-dose groups were examined to establish a no-effect level.
All animals survived until study termination. There were no effects on body weights. No test substance-related clinical signs were noted at any dose level, and no test substance-related clinical signs were evident in any animal of any group during the functional observational battery performed at week 4. Body weights and food consumption were unaffected by treatment. Salivation was noted in some of the high-dose animals; this finding was considered to be incidental. A statistically significant, test-article related, increase in absolute and relative liver weights of male and female high-dose animals was observed; this increase resolved after 2 wk of non-treatment. No treatment-related hematological findings were reported; some test article-related effects on clinical chemistry parameters were reported in the high-dose group. No gross lesions were reported at necropsy. Microscopically, test substance-related effects consisted of hepatocellular hypertrophy at minor degrees and hepatocellular cytoplasmic eosinophilia in both sexes treated with 1000 mg/kg bw/d; these effects were not observed in recovery animals. The no-observed-adverse-effect-level (NOAEL) was 200 mg/kg bw/d in male and female rats.
Cocamide MIPA (Test Substance – Amides, C12-18 and C18-unsatd. N-(Hydroxyethyl), for Read-Across)
A 28-d repeated dose study was performed in accordance with OECD TG 407 in which 0, 70, 250, and 750 (days 1–14)/1500 (days 15–28) mg/kg bw amides, C12-18 and C18-unsatd. N-(hydroxyethyl) in olive oil was administered by gavage 5 d/wk to groups of 10 male and 10 female Wistar rats. 18 Clinical signs, body weight, hematology, clinical chemistry, urinalysis, and gross and microscopic pathology were recorded. Additional groups of 5 male and 5 female rats were kept for a 4-mo recovery period. No mortalities were reported after dosing. No test article-related effects on organ weight were observed. Dose-independent, reversible local findings were found in the forestomach mucosa of the high dose group. Hyperplastic and cellular changes found in the forestomach were also found in controls. The NOAEL was considered to be >750 mg/kg bw.
Subchronic Toxicity Studies
Dermal
Cocamide MIPA and Isostearamide MIPA (Test Substance – Coconut Oil Acid Diethanolamine Condensate, for Read-Across)
Groups of 10 male and 10 female B6C3F1 mice were exposed to 0, 50, 100, 200, 400, or 800 mg/kg bw/d coconut oil acid diethanolamine condensate in ethanol by dermal application, 5 times/wk, for 14 wk.18,19 Mortality, clinical signs and body weights were recorded. At necropsy, gross effects were noted. Selected organs were weighed, and a complete histopathological evaluation was performed on animals of the 0 and 800 mg/kg groups. All mice survived until the end of the study. The only treatment-related clinical finding was irritation of the skin at the site of application in males and females administered 800 mg/kg bw/d. There were no effects on body weight. Liver and kidney weights in 800 mg/kg males and females, liver weights of 400 mg/kg females, and lung weights of 800 mg/kg females were significantly increased compared to the controls. Histopathologic lesions of the skin at the site of application included epidermal hyperplasia, sebaceous gland hyperplasia, chronic active inflammation, parakeratosis and ulcer; the incidences and severities of these skin lesions generally increased with increasing dose in males and females. The NOAEL was considered to be 200 mg/kg bw/d for systemic effects and 100 mg/kg bw/d for local effects.
In a 14-wk dermal study following a similar protocol, groups of 10 male and 10 female Fischer 344 rats were exposed 5 times/wk to 0, 25, 50, 100, 200, or 400 mg/kg bw/d coconut oil acid diethanolamine condensate in ethanol.18,19 All rats survived until the end of the study. Clinical findings included irritation of the skin at the site of application in males and females of the 100, 200, and 400 mg/kg dose groups. Final mean body weights and body weight gains of 200 and 400 mg/kg males and females were significantly lower than those of the controls. At week 14, a minimal microcytic, normochromic, non-responsive anemia occurred in the 100 and 200 mg/kg bw/d females and 400 mg/kg bw/d males and females. The anemia was also seen in the 400 mg/kg bw/d males and females on day 24. Increased segmented neutrophil counts occurred in 400 mg/kg bw/d males and females at week 14, and in 400 mg/kg bw/d females on day 24. Cholesterol concentrations were significantly decreased in 200 and 400 mg/kg bw/d males and in females administered 100 mg/kg or greater, and triglyceride concentrations were decreased in 200 and 400 mg/kg males. Histopathological lesions of the skin at the site of application included epidermal hyperplasia, sebaceous gland hyperplasia, chronic active inflammation, parakeratosis and ulcer; the incidence and severity of these skin lesions generally increased with increasing dose in males and females. The incidences of renal tubule regeneration in 100, 200, and 400 mg/kg bw/d females were significantly greater than in controls, and the severity in 200 and 400 mg/kg bw/d females was increased. The NOAELs for both systemic and local effects was 50 mg/kg bw/d in rats.
Oral
Oleamide MIPA
The subchronic toxicity of Oleamide MIPA was studied in a good laboratory practice (GLP)-compliant study performed in accordance with OECD TG 408.16,18 Oleamide MIPA diluted in corn oil was administered by gavage to groups of male and female Sprague-Dawley rats (10/sex/dose) at the dose levels of 0, 100, 300, 1000 mg/kg bw/d for 13 wk (at constant administration volume of 5 ml/kg bw). Mortality observed during the study was treatment-related. Five animals died during the study, specifically, two males of the 300 mg/kg group (days 59 and 88), and two males (days 59 and 80) and one female (day 91) of the 1000 mg/kg group. Additionally, one male of the 100 mg/kg group was killed on day 77. On the days before death, there were no particular clinical signs but on the day of the death, decedent animals treated with 300 mg/kg showed increased salivation (ptyalism) and absence of spontaneous locomotor activity in the males. In another male, there was blood around and in the mouth. At 1000 mg/kg, there were ptyalism, chromodacryorrhea, dyspnea, bradypnea, absence of locomotor activity in the males and ptyalism in female. At 100 mg/kg and at 300 mg/kg in females, there was no change in blood chemistry parameters. There was a higher creatinine level in the urine of males treated with the test article at 100 mg/kg. There were statistically significant higher plasma alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP) activities in the males treated with 300 and 1000 mg/kg and a statistically significant higher ALT activity in females treated with 1000 mg/kg. There were higher liver weights noted in males and females and higher adrenal gland weights/lower thymus weights in males treated with 1000 mg/kg of the test article. There was no other change in organ weight in animals treated with 100 or 300 mg/kg and no mortality in the control group. The NOAEL was not determined in males. In females, the NOAEL corresponds to 300 mg/kg.
Developmental and Reproductive Toxicity Studies
Dermal
Cocamide MIPA and Isostearamide MIPA (Test Substance – Amides, C8-18 and C18-unsatd., N,N-bis(Hydroxyethyl), for Read-across)
In a 14-wk dermal toxicity study described above in which groups of 10 male and 10 female B6C3F1 mice received open applications of 0–800 mg/kg bw amides, C8-18 and C18-unsatd., N,N-bis(hydroxyethyl) in ethanol, 5 d/wk for 14 wk, samples were collected at the end of the study for sperm motility or vaginal cytology from mice of 200, 400, and 800 mg/kg bw groups.17,18 The following sperm motility parameters were evaluated: spermatid heads per gram of testis, spermatid heads per testis, spermatid count, and epididymal spermatozoal motility and concentration. The left cauda epididymis, epididymis, and testis were weighed. Vaginal samples for cytology evaluations were collected for 12 consecutive days prior to the end of the studies from all female mice. The length of the estrous cycle and the length of time spent in each stage of the cycle were evaluated. Epididymal spermatozoal concentration was significantly increased in 800 mg/kg males. Estrous cycle lengths of dosed females were similar to that of the controls.
In the 14-wk dermal study described above in which groups of male and female Fischer 344 rats received open applications, 5 d/wk, of 0–400 mg/kg bw amides, C8-18 and C18-unsatd., N,N-bis(hydroxyethyl) in ethanol, sperm motility or vaginal cytology were collected at the end of the study from all rats receiving 100, 200, and 400 mg/kg bw of test material.17,18 Test material results were similar to those of the vehicle controls.
Oral
Oleamide MIPA
In an oral developmental toxicity study performed in accordance with OECD TG 414, Oleamide MIPA diluted in corn oil was administered by gavage to groups of mated female Sprague-Dawley rats (20 mated females/dose) at dose levels of 0, 100, 300, and 1000 mg/kg bw/d from days 6 to 19 of gestation. 16 On day 20 of gestation, all mated females were killed and necropsied, and all fetuses were examined. The clinical signs (ptyalism and chromodacryorrhea) observed were at low incidence and were not attributed to a toxicological effect of the test article. The test article did not induce any relevant changes in fetuses examined at skeletal and visceral examination. There was a statistically significant lower placenta weight in the group receiving 100 mg/kg of the test substance. This was low in amplitude and was not attributed to a toxicological effect of the test substance. The NOAEL for embryo fetal development was 1000 mg/kg bw/d.
Another oral reproductive study was performed in accordance with OECD guideline 422; Oleamide MIPA in corn oil was administered daily by gavage to groups of 10 male and 10 female Sprague-Dawley rats. 16 In males, the test article was administered 2 wk before mating, during the mating period, and until sacrificed (at least 5 wk in total). Females were treated 2 wk before mating, during the mating period (1 wk), during pregnancy, during lactation until day 5 post-partum (inclusive) and until sacrificed. Animals were treated at dose-levels of 0, 100, 300, or 1000 mg/kg/d. A constant dosage-volume of 5 ml/kg/d was used. At 100 mg/kg/d, the only finding was ptyalism in most test animals. At 300 mg/kg/d, ptyalism, hypoactivity, loud breathing, piloerection and/or round back was also noted with comparable incidence. At 1000 mg/kg/d, the main clinical sign noted was ptyalism in all test animals. Hypoactivity, loud breathing, piloerection and/or round back were also recorded transiently in a few animals. No effects in the study were considered to be adverse. The NOAEL for parental toxicity, reproductive performance (mating and fertility) and toxic effects on progeny was 1000 mg/kg/d.
Cocamide MIPA and Isostearamide MIPA (Test Substance – Amides, C12-18 (Even-Numbered) and C18-unsatd., N,N-bis(Hydroxyethyl), for Read-Across)
Groups of 30 gravid female Sprague-Dawley CD rats were dosed by gavage with 0, 100, 300, and 1000 mg/kg bw/d amides, C12-18 (even-numbered) and C18-unsatd., N,N-bis(hydroxyethyl), once daily on days 6–15 of gestation, in accordance with OECD TG 414.17,18 Control animals were given vehicle alone (arachis oil, DAB 9). Clinical condition and reaction to treatment were recorded daily, and body weights were determined on days 0, 6, 16, and 20 of gestation. All surviving females were sacrificed on day 20 of gestation, and the fetuses were removed by caesarean section. At necropsy, the females were examined macroscopically. Live fetuses were weighed, sexed and examined for visceral and skeletal abnormalities. No deaths or treatment-related changes in body weight gain and necropsy findings were observed in dams at any dose level. Treatment-related symptoms observed in all groups were salivation and propulsion of the head. The highest dose group showed severe salivation. Apart from the control (1 dead fetus) and the 100 mg/kg bw/d groups (7 dead fetuses), all females had viable fetuses. Pre-implantation loss and mean numbers of resorptions were not affected by treatment. The data for post-implantation loss, embryonic deaths and total fetuses showed some deviations, which were considered to be non-treatment-related. Mean placental and uterine weights were not affected by dosing. Fetal sex ratio was comparable in all groups. No treatment-related fetal abnormalities were found at necropsy. The examined fetuses showed no treatment-related visceral and skeletal abnormalities/variations. One fetus of the 300 mg/kg group had a stump tail and missing coccygeal vertebrae. Further, the data for skeletal ossifications showed some deviations in the two highest dose groups. However, it was stated that all these effects were assessed to be non-treatment-related. The NOAELs for parental toxicity and developmental toxicity were considered to be 1000 mg/kg bw/d.
Genotoxicity
Abbreviations: CMC – carboxymethylcellulose; DMSO – dimethyl sulfoxide; UDS – unscheduled DNA synthesis.
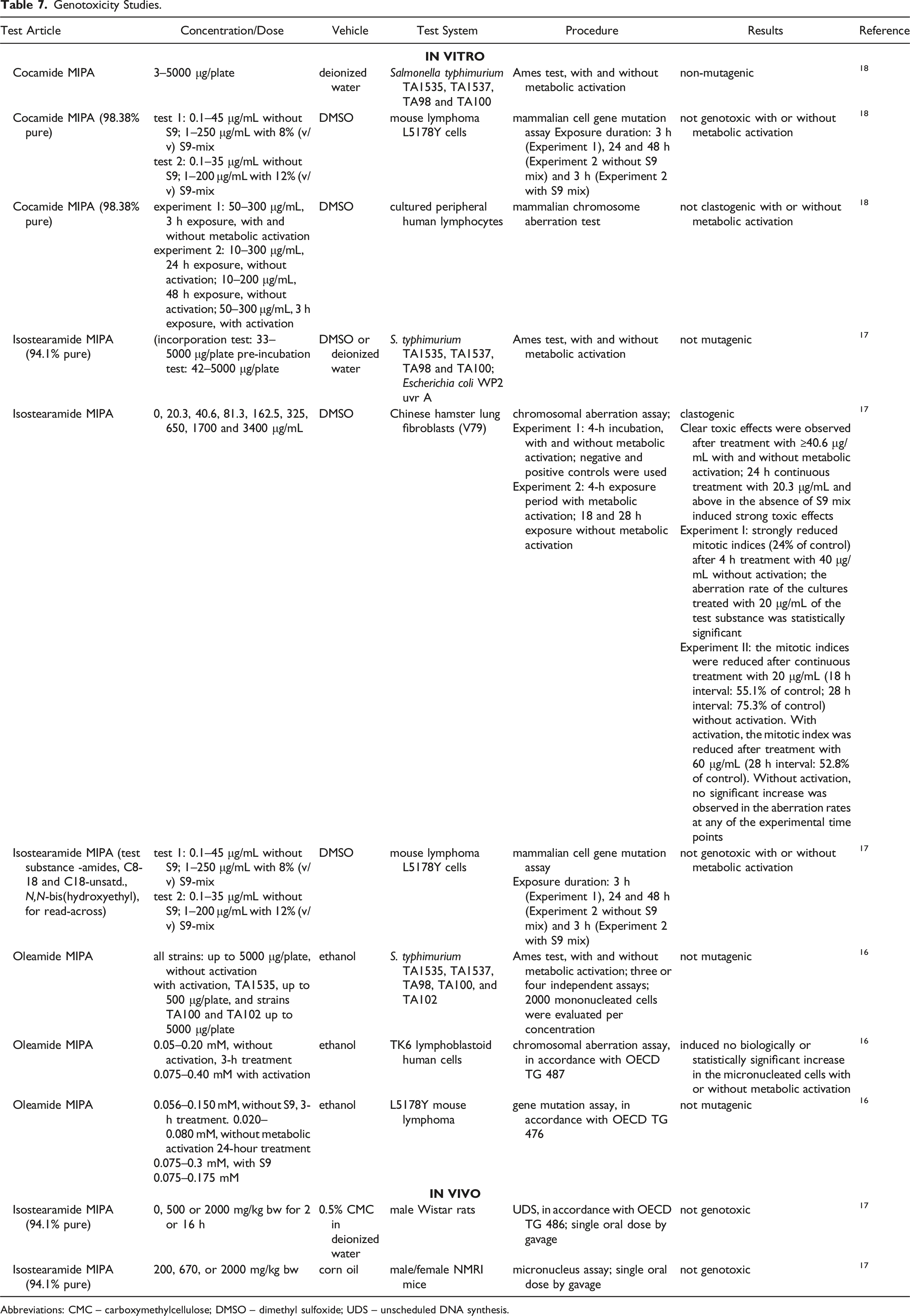
Cocamide MIPA, Isostearamide MIPA, and Oleamide MIPA were not mutagenic in the Ames test, and Oleamide MIPA and amides, C8-18 and C18-unsatd., N,N-bis(hydroxyethyl) (as read-across for Isostearamide MIPA) were not genotoxic in the mammalian cell gene mutation assay in L5178Y mouse lymphoma cells. Cocamide MIPA and Oleamide MIPA were not clastogenic in the chromosomal aberration assay. However, Isostearamide MIPA was clastogenic in the chromosomal aberration assay in Chinese hamster lung fibroblasts. In vivo, Isostearamide MIPA was not genotoxic in an unscheduled DNA synthesis (UDS) assay in male Wistar rats or a micronucleus test in NMRI mice.
Carcinogenicity Studies
Dermal
Cocamide MIPA and Isostearamide MIPA (Test Substance – Coconut Oil Acid Diethanolamine Condensate, for Read-Across)
Open applications of 0, 100, or 200 mg/kg bw of coconut oil acid diethanolamine condensate in ethanol were made 5 d/wk to shaved skin of groups 50 male and 50 female B6C3F1 mice for 104 wk.18,19 Survival of dosed males and 100 mg/kg bw females was similar to that of the vehicle controls; survival of the 200 mg/kg bw group of female mice was reduced compared to the vehicle control group, but the difference was not significant. Irritation was reported at the test site in males that received 200 mg/kg bw. Several non-neoplastic lesions of the skin at the application site were determined to be test article-related. Incidences of epidermal hyperplasia, sebaceous gland hyperplasia, and hyperkeratosis in all dosed groups of males and females were significantly greater than those in the vehicle control groups, and the incidences of ulceration in 200 mg/kg bw males and inflammation and parakeratosis in 200 mg/kg bw females were increased. In the thyroid gland, the incidences of follicular cell hyperplasia in all dosed groups of males (vehicle control, 11/50; 100 mg/kg bw, 20/50; 200 mg/kg bw, 23/50) and females (27/50, 36/50, 33/50) were significantly greater than those in the vehicle controls. Follicular cell hyperplasia consisted of focal areas of thyroid gland follicles lined with increased numbers of epithelial cells, which formed papillary projections in some instances.
Dosed male and female mice had significantly greater incidences of hepatic neoplasms (hepatocellular adenoma, hepatocellular carcinoma, and hepatoblastoma (males) than the vehicle controls. There was a morphologic continuum from adenoma to carcinoma, with less differentiation and typical trabecular formations in the carcinomas. Carcinomas were often a centimeter or more in diameter, whereas adenomas were generally smaller and more discrete. Carcinomas metastasized to the lung in a few males and females. Adenomas, carcinomas, and hepatoblastomas displaced normal liver parenchyma, and none contained normal lobular architecture. Hepatoblastomas were characterized by well-demarcated focal areas composed of bundles of deeply basophilic, spindle-shaped cells. The incidences of renal tubule adenoma (1/50, 1/50, 7/50) and of renal tubule adenoma or carcinoma (combined) (1/50, 1/50, 9/50) in 200 mg/kg bw males were significantly greater than those in the vehicle controls. Renal tubule hyperplasia, adenoma, and carcinoma formed a morphological continuum. Adenomas were focal, compressive masses approximately five or more tubules in diameter; carcinomas were morphologically similar to adenomas but were larger and often showed cellular debris and/or mineralization. Renal tubule neoplasms were located in the cortex or outer medulla. Focal proliferative masses less than five tubules in diameter were classified as focal hyperplasia. It was stated there was clear evidence of carcinogenic activity in male B6C3F1 mice based on increased incidences of hepatic and renal tubule neoplasms and in female B6C3F1 mice based on increased incidences of hepatic neoplasms. The lowest-observable-adverse-effect-level (LOAEL) for systemic and local effects was considered to be 100 mg/kg bw/d.
In a 104-wk dermal study in rats, groups of 50 male and 50 female Fischer rats were exposed 5 d/wk to 0, 50, or 100 mg/kg bw/d of coconut oil acid diethanolamine condensate in ethanol.18,19 Mortality, clinical signs and body weight were recorded throughout the study, and at necropsy, a gross macroscopic examination and complete histopathology were carried out. The survival rates of treated male and female rats were similar to those of controls. There were no significant differences in body weight throughout the groups. The only treatment-related clinical finding was irritation of the skin at the site of application in 100 mg/kg bw/d females. Non-neoplastic lesions of the skin at the site of application included epidermal hyperplasia, sebaceous gland hyperplasia, parakeratosis and hyperkeratosis; the incidences and severities of these lesions increased with increasing dose. There were marginal increases in the incidences of renal tubule adenoma or carcinoma (combined) in 50 mg/kg bw/day females. The severity of nephropathy increased with increasing dose in female rats. The incidences of chronic active inflammation, epithelial hyperplasia and epithelial ulcer of the forestomach increased with dose in female rats and the increases were significant in the 100 mg/kg bw/d group. There was no evidence of carcinogenic activity of the test substance in male rats at any dose; there was an equivocal evidence of carcinogenic activity in female rats based on a marginal increase in the incidences of renal tubule neoplasms. The NOAEL was considered to be 50 mg/kg bw/d in rats.
Dermal Irritation and Sensitization
Irritation
In Vitro
Oleamide MIPA
The primary skin irritation potential of Oleamide MIPA was evaluated using the EpiskinTM reconstructed human epidermis model based on OECD TG 439. 16 The test material (undiluted Oleamide MIPA; 10 mg) was applied to skin tissue. Oleamide MIPA was considered to be non-irritating to skin.
Animal
Isostearamide MIPA
Semi-occlusive patches containing 0.5 ml Isostearamide MIPA were applied for 4 h to a 6 cm2 area of shaved skin of 3 male New Zealand White rabbits. 18 Erythema (scores 1.7–2 out of 4 max) was present until day 5; no edema was observed. Erythema decreased after day 5, and was resolved by day 8. Undiluted Isostearamide MIPA was not considered to be irritating to rabbit skin.
Cocamide MIPA (Test Substance – Amides, C8-18 and C18-unsatd., N-(Hydroxyethyl), for Read Across)
Occlusive patches containing 0.5 g amides, C8-18 and C18-unsatd., N-(hydroxyethyl) with 0.5 ml water were applied for 4 h to a 6 cm2 area of shaved skin of 3 small white Russian rabbits. 18 Erythema, edema, and eschar were observed in all animals; the results were reversible within 14 d. The overall irritation score (24/48/72 h) was 3.67/8, and the test substance was considered to be moderately irritating.
Sensitization
Animal
Cocamide MIPA
A guinea pig maximization study was performed in accordance with OECD TG 406 to determine the sensitization potential of Cocamide MIPA. 18 Ten male Dunkin-Hartley guinea pigs were used in the test group, and 5 males were used as controls. Intradermal induction consisted of 3 injections: a 1:1 (v/v) mixture of Freund’s Complete Adjuvant (FCA) and physiological saline; two injections of 5% Cocamide MIPA in bi-distilled water. Epidermal induction was performed after 1 wk (on day 8); an occlusive patch (2 cm × 4 cm) with 25% of the test substance in bi-distilled water was applied for 48 h to the clipped and shaved flanks of the test animals. After a 2-wk non-treatment period, on day 22, the challenge was performed by applying 2 cm × 2 cm occlusive patches containing 0.1 ml of 5% test material in bi-distilled water for 24 h; the test sites were evaluated 24 and 48 h after patch removal. 2-Mercaptobenzothiazole was used as a positive control; 70% of the animals of the test group were observed with positive skin reactions after treatment with a non-irritant concentration of positive control (25% v/v). All animals survived, and no clinical signs of toxicity were reported. “Normal local symptoms” were observed in test and control animals following intradermal induction. No erythema or edema was observed following epidermal induction. No positive reactions were reported following the challenge; the test material was not a sensitizer.
Isostearamide MIPA
A guinea pig maximization study was performed in accordance with OECD TG 406 to determine the sensitization potential of Isostearamide MIPA. 17 Ten male albino Himalayan guinea pigs were used in the test group, and 5 males were used as controls. Intradermal induction consisted of 3 injections: a 1:1 (v/v) mixture of FCA and physiological saline; 5% Isostearamide MIPA in bi-distilled water; and 5% Isostearamide MIPA in a 1:1 (v/v) mixture of FCA and physiological saline. Epidermal induction was performed after 1 wk (on day 8); 4 occlusive patches (3 cm × 3 cm) with 25, 50, 75, or 100% of the test substance (0.3 ml) were applied for 24 h to the clipped and shaved flanks of the test animals. After a 2-wk non-treatment period, the challenge was performed by applying 3 cm × 3 cm occlusive patches containing 0.2 ml of the vehicle or 1% test material in bi-distilled water for 24 h; the test sites were evaluated 24 and 48 h after patch removal. 2-Mercaptobenzothiazole was used as a positive control.
One animal of the test group was found dead on test day 10; no findings were noted at necropsy, and the death was considered to be spontaneous and not treatment related. The “expected and common findings” were observed in the control and test group after the different applications using FCA intradermally (on test day 1) and consisted of erythema, edema, necrotizing dermatitis, encrustation, and exfoliation of encrustation. After epidermal induction on day 8, discrete/patchy erythema was observed in all surviving test animals (treated group) at the 24-h reading after treatment with the undiluted test substance; these effects persisted in 1 animal at the 48-h reading. No reactions were observed in the negative controls. Following challenge (day 22), no skin reactions were observed in the test or the vehicle-control groups. The test substance was not considered to be a skin sensitizer.
Oleamide MIPA
The sensitization potential of Oleamide MIPA was evaluated in a guinea pig maximization study. 16 The test group consisted of 10 male and 10 female Dunkin Hartley guinea pigs, and a group of 5 males and 5 females was used as the control group. For the test group, 10% Oleamide MIPA in corn oil was used for intradermal induction (day 1), and 75% Oleamide MIPA in ethanol/water was applied for the topical induction with an occlusive dressing for 48 h (day 8). On day 22, challenge consisted of a topical application of 50% Oleamide MIPA in acetone to the right flank and acetone to the left flank held in place by an occlusive dressing for 24 h. The control group was administered vehicle only. Oleamide MIPA induced delayed contact hypersensitivity in more than 30% of the animals.
Ocular Irritation Studies
In Vitro
Oleamide MIPA
The ocular irritation potential of Oleamide MIPA was evaluated in a bovine corneal opacity and permeability (BCOP) test performed in accordance with OECD TG 437. 16 The test material (750 μl) at a concentration of 10% (w/v) in the water was applied to three corneas for 10 min and rinsed following application. No notable opaque spots or irregularities were observed on corneas following the treatment. The in vitro irritancy score (IVIS) was calculated as 2.0, and Oleamide MIPA was not considered an ocular corrosive or severe eye irritant under the conditions of the test.
Animal
Isostearamide MIPA
Undiluted Isostearamide MIPA (94.1% pure; 0.1 ml) was instilled into the conjunctival sac of the left eye of 3 New Zealand White rabbits, and the contralateral eye served as an untreated control. 17 (Whether the eyes were rinsed was not stated.) Observations were made at 1, 24, 48, and 72 h. Some slight conjunctival reactions (chemosis with a score of ≤1 and enanthema with a score of 1 to 2) were observed in all rabbits after 1 h. Neither iris irritation nor corneal opacity was recorded. Reactions were fully reversible; no effects were seen at 24, 48, and 72 h. Under the study conditions, the test substance was not considered to be irritating to rabbit eye.
Oleamide MIPA
Three male New Zealand White rabbits were used to determine the ocular irritation potential of Oleamide MIPA. 16 A dosage volume of 0.1 ml of undiluted test article was instilled into the conjunctival sac of the left eye of each rabbit, and the eyes were not rinsed. The right eye remained untreated and served as control. The mean scores (calculated using the 24, 48, and 72-h scores for each animal) for the conjunctiva ranged from 0.3–1.0 for redness and 0–0.3 for chemosis. Corneal opacity and iridial inflammation were not observed. The test substance was not irritating to the eyes of rabbits.
Cocamide MIPA (Test Substance – Amides, C8-18 and C18-unsatd., N-(Hydroxyethyl), for Read-Across)
The ocular irritation potential of undiluted amides, C8-18 and C18-unsatd., N-(hydroxyethyl) was evaluated in 3 rabbits. 18 Ground test material (0.1 ml) was instilled into the conjunctival sac of the right eye; the contralateral eye served as a control. The mean overall score was 26.8/110, and the test substance was considered to be moderately irritating to rabbit eyes, and in one animal, irreversible effects (cornea, iris) occurred.
Summary
This is a safety assessment of 14 alkyl amide MIPA ingredients as used in cosmetics. All of these ingredients are reported to function in cosmetics as a surfactant – foam booster and/or viscosity increasing agent; some of the ingredients have other reported functions. In some instances, information on substances used for read-across is provided to address data needs for certain toxicological endpoints. Specifically, information on amides, C8-18 and C18-unsatd., N-(hydroxyethyl) and amides, C12-18 and C18-unsatd, N-(hydroxyethyl) was used for read-across to Cocamide MIPA, and information on coconut oil acid diethanolamine condensate; amides, C8-18 and C18-unsatd., N,N-bis(hydroxyethyl) and amides, C12-18 (even-numbered); and C18-unsatd., N,N-bis(hydroxyethyl) was used for read-across for Cocamide MIPA and Isostearamide MIPA.
Four of the 14 ingredients included in this assessment are reported to be in use, according to the VCRP and/or the results of a Council survey. According to 2019 VCRP data, Lauramide MIPA has the highest reported frequency of use (485 formulations), and Cocamide MIPA has the second greatest reported number of uses (335). The alkyl amide MIPA ingredients are primarily used in rinse-off formulations, and most of these reported uses are in some type of hair or skin cleansing formulations. Cocamide MIPA has the highest concentration of use, at 12% in hair bleaches. Lauramide MIPA has the next highest reported concentration of use; it is used at 4.8% in bath soaps and detergents. The only concentration of use reported resulting in leave-on dermal exposure is 0.4% Oleamide MIPA in face and neck preparations. Of the 14 alkyl amide ingredients named in the report, 12 are listed in the European Union inventory of cosmetic ingredients without restrictions; MIPA-Myristate is identified under the category monoalkylamines, monoalkanolamines and their salts, and restrictions regarding amine and nitrosamine content apply. For Peanutamide MIPA, as a peanut oil extract/derivative, the maximum concentration of peanut protein allowed in peanut oil is 0.5 ppm.
The dermal LD50 of Cocamide MIPA is >2000 mg/kg in rabbits (details not provided). In rats, the dermal LD50s of Isostearamide MIPA and Oleamide MIPA (both ingredients, 24-h semi-occlusive patch) and of amides, C8-18 and C18-unsatd., N-(hydroxyethyl) in PEG (type and duration of patch not provided) were reported to be >2000 mg/kg; these were the highest doses tested in each study. In acute oral studies in rats, the LD50s for Cocamide MIPA, Isostearamide MIPA, and Oleamide MIPA were all reported to be >2000 mg/kg; as with the dermal studies, these were the highest doses tested.
In a 28-d repeated-dose gavage study in rats with ≤1000 mg/kg bw/d Isostearamide MIPA, the NOAEL was 200 mg/kg bw, based on hepatic effects. Test substance-related effects consisted of hepatocellular hypertrophy at minor degrees and hepatocellular cytoplasmic eosinophilia in both sexes treated with 1000 mg/kg bw/d; these effects were not observed in 14-d recovery animals. In a 28-d study in which Wistar rats were dosed, 5 d/wk, with up to 1500 mg/kg amides, C12-18 and C18-unsatd. N-(hydroxyethyl) in olive oil by gavage, the NOAEL was considered to be >750 mg/kg. In a 13-wk oral toxicity study in which male and female Sprague-Dawley rats were given ≤1000 mg/kg bw/d Oleamide MIPA in corn oil by gavage, mortality, low food consumption, and low body weight gain were reported in males. There were slight changes in the liver and the bone marrow in animals treated with test article at 1000 mg/kg. The NOAEL in females was determined to be 300 mg/kg bw/d Oleamide MIPA; a NOAEL was not determined for males.
In 14-wk dermal studies of coconut oil acid diethanolamine condensate in ethanol, open applications were made 5 d/wk to mice (≤800 mg/kg bw/d) and rats (≤400 mg/kg bw/d). The NOAELs for local and systemic effects in mice were 100 and 200 mg/kg bw, respectively, and in rats, the systemic NOAEL was 50 mg/kg bw. In both rats and mice, microscopic lesions of the skin at application site included epidermal hyperplasia, sebaceous gland hyperplasia, chronic active inflammation, parakeratosis and ulcer, with incidences and severities of these skin lesions generally increased with increasing dose in males and females.
As part of the 14-wk dermal toxicity studies described above, samples were collected at the end of the study for sperm motility or vaginal cytology. Epididymal spermatozoal concentration was significantly increased in male mice of the 800 mg/kg group; estrous cycle lengths of dosed female rats and mice were similar to controls.
In an oral developmental toxicity test in which groups of 20 mated female rats were dosed by gavage with up to 1000 mg/kg/d Oleamide MIPA in corn oil from days 6 to 19 of gestation, the test article did not induce any relevant changes in fetuses examined at skeletal and visceral examination, and the NOAEL was considered to be 1000 mg/kg/d. In a study in which groups of 30 gravid female Sprague-Dawley CD rats were administered up to 1000 mg/kg bw/d amides, C12-18 (even-numbered) and C18-unsatd., N,N-bis(hydroxyethyl) by gavage on days 6–15 of gestation, the NOAELs for parental toxicity and developmental toxicity were considered to be 1000 mg/kg bw/d.
The reproductive toxicity of Oleamide MIPA was evaluated in groups of 10 male and 10 female Sprague-Dawley rats at dose levels of ≤1000 mg/kg/day. In males, test article was administered 2 wk before mating, during the mating period, and until sacrificed (at least 5 wk in total). Females were treated from 2 wk before mating until day 5 post-partum (inclusive). No treatment-related, adverse effects were observed, and the NOAELs for parental toxicity, reproductive performance (mating and fertility), and toxic effects on progeny were 1000 mg/kg/day.
Cocamide MIPA, Isostearamide MIPA, and Oleamide MIPA were not mutagenic in the Ames test, and Oleamide MIPA and amides, C8-18 and C18-unsatd., N,N-bis(hydroxyethyl) were not genotoxic in the mammalian cell gene mutation assay in L5178Y mouse lymphoma cells. Cocamide MIPA and Oleamide MIPA were not clastogenic in the chromosomal aberration assay. However, Isostearamide MIPA was clastogenic in the chromosomal aberration assay in Chinese hamster lung fibroblasts, but at cytotoxic concentrations. In vivo, Isostearamide MIPA was not genotoxic in a UDS assay in male Wistar rats or a micronucleus test in NMRI mice.
Open applications of up to 200 mg/kg bw coconut oil acid diethanolamine condensate in ethanol were made 5 d/wk to shaved skin of groups 50 male and 50 female B6C3F1 mice for 104 wk; there was clear evidence of carcinogenic activity in male mice based on increased incidences of hepatic and renal tubule neoplasms and in female mice based on increased incidences of hepatic neoplasms. The LOAEL for systemic and local effects was considered to be 100 mg/kg bw/d. In a 104-wk dermal study in which groups of 50 male and 50 female Fischer rats were exposed 5 d/wk to up to 100 mg/kg bw/d of coconut oil acid diethanolamine condensate in ethanol, there was no evidence of carcinogenic activity of the test substance in male rats at any dose; there was an equivocal evidence of carcinogenic activity in female rats based on a marginal increase in the incidences of renal tubule neoplasms. The NOAEL was considered to be 50 mg/kg bw/d in rats.
In an EpiskinTM reconstructed human epidermis assay, Oleamide MIPA was determined to be a non-irritant to skin. A 4-h semi-occlusive application of undiluted Isostearamide MIPA was not considered to be irritating to rabbit skin. However, in another study, a 4-h occlusive patch of amides, C8-18 and C18-unsatd., N-(hydroxyethyl) was moderately irritating to rabbit skin.
Neither Cocamide MIPA (epidermal induction and challenge with 5%) nor Isostearamide MIPA (epidermal induction with 25%–100%, challenge with 1%) were sensitizers in the guinea pig maximization test. However, in a guinea pig maximization test in which 10% Oleamide MIPA in corn oil was used for intradermal induction, 75% Oleamide MIPA in ethanol/water was used for topical induction, and 50% Oleamide MIPA was used at challenge, delayed contact hypersensitivity was reported in more than 30% of the 20 test animals.
The ocular irritation potential of Oleamide MIPA was evaluated in vitro using a BCOP study; an irritancy score of 2.0 was reported, and it was concluded that the Oleamide MIPA is not an ocular corrosive or severe irritant. In rabbits, undiluted Isostearamide MIPA and Oleamide MIPA were non-irritating to rabbit eyes, but amides, C8-18 and C18-unsatd., N-(hydroxyethyl) was a moderate ocular irritant.
Discussion
The ingredients in this group are fatty amides resulting from amidation with MIPA. Accordingly, the Panel specified that these ingredients should not be used in cosmetic products in which N-nitroso compounds can be formed.
The alkyl amide MIPA ingredients are primarily used in rinse-off formulations. However, leave-on uses are reported, with 0.4% Oleamide MIPA reported as the highest maximum concentration of use for leave-on dermal exposure. The Panel noted that delayed contact hypersensitivity was reported in a GPMT performed with high concentrations of Oleamide MIPA (75% for topical induction/50% at challenge), but not in GPMTs on Cocamide MIPA (25% at topical induction/5% at challenge) and Isostearamide MIPA (100% at topical induction/1% at challenge). The Panel stated that the sensitization observed with Oleamide MIPA was most likely a result of the high concentrations and a stressing of the system (as this method of testing utilizes a combination of exposures, including intradermal injections which bypass the stratum corneum). Because the Panel felt that it was appropriate to read-across from Cocamide MIPA and Isostearamide MIPA, concern that Oleamide MIPA would be a sensitizer in cosmetic formulations was mitigated. However, the Panel was concerned that the potential exists for dermal or ocular irritation with the use of products formulated with the ingredients named in this assessment. Therefore, the Panel specified that products containing alkyl amide MIPA ingredients must be formulated to be non-irritating.
Published studies were not found, and unpublished data were not submitted, for certain toxicological endpoints on the alkyl amide MIPA ingredients. The Panel determined that the data on similar substances provided in ECHA dossiers and in an NTP technical report could be used for read-across for the missing data endpoints. The Panel noted adverse effects observed in the 14-wk dermal studies in mice and rats on the read-across test substance (coconut oil acid diethanolamine condensate); however, the adverse effects reported were at concentrations above what is reported to be used in cosmetic formulations, thereby mitigating any concerns for dermal toxicity. The same reasoning was used to mitigate concerns for positive results that were observed in the dermal carcinogenicity study that same read-across test substance in mice; furthermore, positive results were not observed in a dermal carcinogenicity study on the same test compound in rats. Additionally, the Panel stated that the CIR safety assessment of diethanolamides also supported the safety of the alkyl amide MIPA ingredients because of the structural similarities between the ingredients.
The acyl groups (i.e., fatty acid chain residues) in Peanutamide MIPA are derived from peanut oil. The Panel has previously reviewed the safety of Arachis Hypogaea (Peanut) Oil as used in cosmetics, and discussed therein the relationship between food allergies and exposure to refined oils. Individuals who have food allergies to a plant protein rarely exhibit allergic reactions when exposed to refined oils of the same plant; proteins do not partition into the oil. Additionally, the Panel noted that aflatoxins, which could be associated with peanuts, do not partition into the oil. However, the Panel does caution manufacturers to make certain that Peanutamide MIPA is free from proteins and aflatoxins.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that the 14 alkyl amide MIPA ingredients listed below are safe in cosmetics in the present practices of use and concentration described in this safety assessment when formulated to be non-irritating.
Cocamide MIPA
Coconut Oil MIPA Amides*
Hydroxyethyl Stearamide-MIPA*
Isostearamide MIPA*
Lauramide MIPA
Linoleamide MIPA*
MIPA- Myristate*
Myristamide MIPA*
Oleamide MIPA
Palmamide MIPA*
Palm Kernelamide MIPA*
Peanutamide MIPA*
Ricinoleamide MIPA*
Stearamide MIPA*
*Use not reported in the VCRP and/or concentration of use survey. The expectation is that if used in cosmetic formulations, these ingredients would be used in product categories and at concentrations comparable to that reported for others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 555 13th Street, NW, Suite 300W, Washington, DC 20004, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.