Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of Ascorbyl Glucoside and Sodium Ascorbyl Glucoside in cosmetic products. These ingredients are reported to have the following functions in cosmetics: antioxidant, and skin-conditioning agent—miscellaneous. The Panel reviewed data relevant to the safety of these ingredients in cosmetic formulations, and concluded that Ascorbyl Glucoside and Sodium Ascorbyl Glucoside are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Keywords
Introduction
The safety of Ascorbyl Glucoside and Sodium Ascorbyl Glucoside, as used in cosmetics, is reviewed in this assessment. According to the web-based International Cosmetic Ingredient Dictionary and Handbook (Dictionary), Ascorbyl Glucoside is reported to have the following functions in cosmetics: antioxidant, skin bleaching agent, and skin-conditioning agent—miscellaneous 1 ; however, it should be noted that functioning as a skin bleaching agent is not a cosmetic use in the US, and, therefore, the Expert Panel for Cosmetic Ingredient Safety (Panel) did not evaluate safety for that use. Sodium Ascorbyl Glucoside is reported to function in cosmetics as a skin conditioning agent—humectant.
Because Ascorbyl Glucoside is a derivative of ascorbic acid, it should be noted that the Panel has evaluated the safety of ascorbic acid, and ethers and esters of ascorbic acid. The safety evaluations involve 3 separate ingredient reports, and the Panel’s conclusion in each report is that these ingredients are safe in the present practices of use and concentration, as described in the safety assessment. Those 3 final reports are: A final report on the safety assessment of Ascorbyl Palmitate, Ascorbyl Dipalmitate, and Ascorbyl Stearate was published in 1999. 2 The final report on the safety assessment of L-Ascorbic Acid, Calcium Ascorbate, Magnesium Ascorbate, Magnesium Ascorbyl Phosphate, Sodium Ascorbate, and Sodium Ascorbyl Phosphate was published in 2005. 3 Lastly, the final report on ethers and esters of ascorbic acid (Ascorbyl Dipalmitate, Ascorbyl Isostearate, Ascorbyl Linoleate, Ascorbyl Palmitate, Ascorbyl Stearate, Ascorbyl Tetraisopalmitate, and Tetrahexyldecyl Ascorbate) was issued in 2017. 4 The 3 ingredients evaluated in the 1999 published final report were included in the 2017 final report because the safety of these ingredients was reevaluated in 2017 due to the availability of new safety test data. All reports are available on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/ingredients).
The Panel has also evaluated the safety of glucose in a published report on monosaccharides, disaccharides, and related ingredients. 5 The conclusion in that report states that glucose and the other ingredients reviewed are safe in the present practices of use and concentration in cosmetics.
The published data in this document were identified by conducting an exhaustive search of the world’s literature. A list of the typical search engines and websites used, sources explored, and endpoints that the Panel evaluates, is available on the CIR website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data may be provided by the cosmetics industry, as well as by other interested parties. Much of the data included in this safety assessment was found at the National Industrial Chemicals Notification and Assessment Scheme (NICNAS) website. 6 Please note that this website provides summaries of information from other studies, and it is those summary data that are reported in this safety assessment when NICNAS is cited.
Chemistry
Definition
Ascorbyl Glucoside (CAS No. 129499-78-1; Figure 1) is defined as the product obtained by the condensation of ascorbic acid with glucose.
1
Sodium Ascorbyl Glucoside is defined as the sodium salt of Ascorbyl Glucoside. These 2 ingredients differ in structure only by the replacement of a proton with a sodium cation. Ascorbyl Glucoside.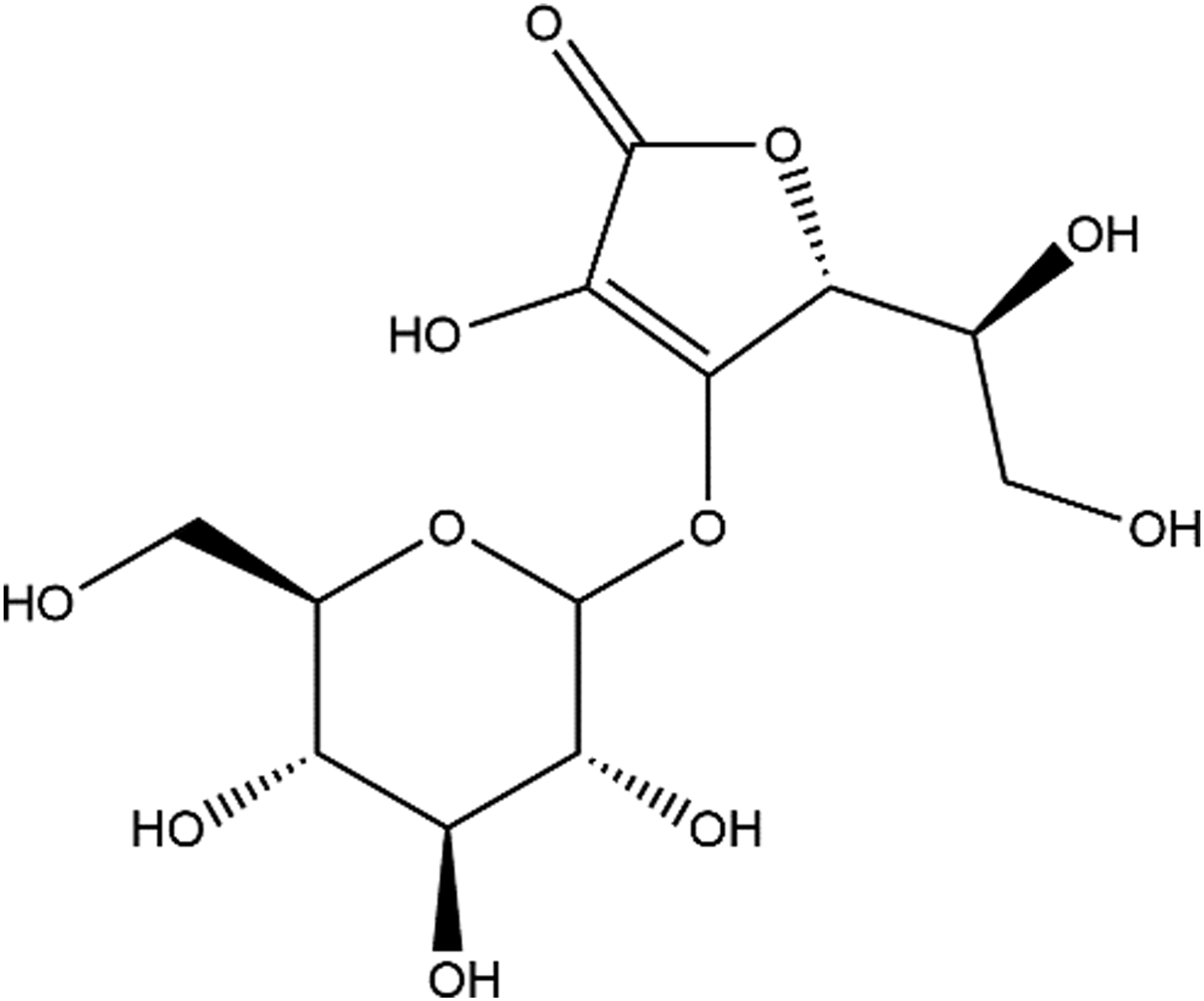
Chemicals Properties
Properties of Ascorbyl Glucoside and Sodium Ascorbyl Glucoside.
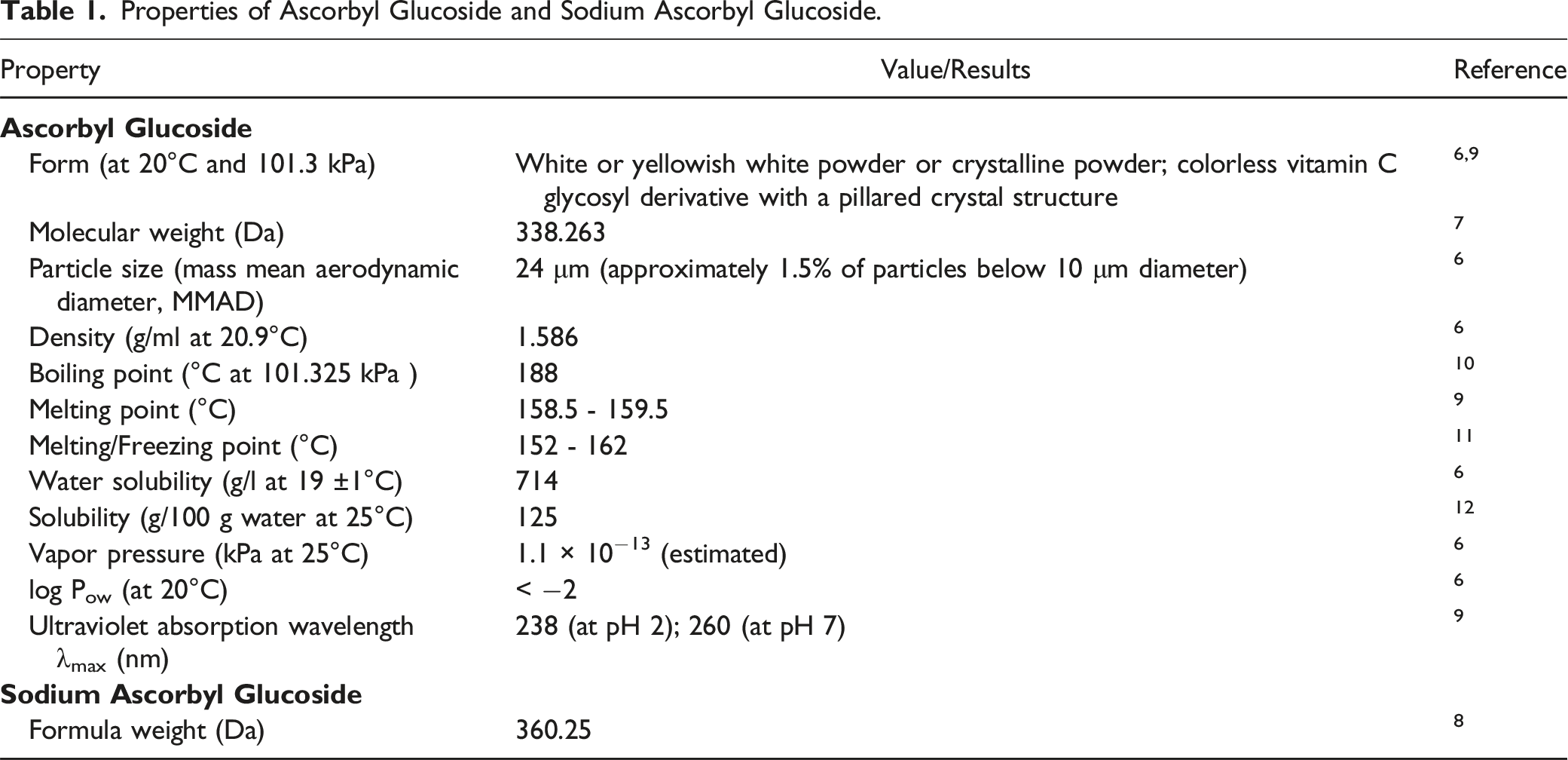
The optimal condition of retaining Ascorbyl Glucoside with the highest stability in cosmetic products has been determined to be 55.3°C and pH 6.4. 13 It has been noted that, generally, the pH of common cosmetic products is between 5.5 and 7.0. 14
Method of Manufacture
Ascorbyl Glucoside is synthesized by a biocatalytic transglucosylation in which starch-derived cyclic or linear oligosaccharides are reacted with l-ascorbic acid (i.e., vitamin C) by a glucanotransferase.15,16 More specifically, Ascorbyl Glucoside is prepared by transferring a glucosyl residue from α-1,4-glucan to the C-2 position of ascorbic acid and bound with α-1,2-linkage. 9 Enzymatic transglucosylation to synthesize a chemically stable form of l-ascorbic acid has been investigated by using commercially available enzymes. 17 Of the various glycosidases that were used, only rice seed α-glucosidase produced a nonreducing and stable glucoside of l-ascorbic acid, which was identified as Ascorbyl Glucoside.
According to another source, transglycosylation in the presence of the enzyme cyclodextrin glucanotransferase (CGTase, from Paenibacillus sp.) was used to produce Ascorbyl Glucoside. 18 The standard reaction mixture contained sodium ascorbate, dextrin or other saccharides, and CGTase. To hydrolyze ascorbic acid-2-oligosaccharides produced by CGTase, glucoamylase (from Rhizopus mold) was added to the reaction mixture, yielding Ascorbyl Glucoside.
Composition/Impurities
According to NICNAS, Ascorbyl Glucoside is 99.8% to 100% pure, and contains no hazardous impurities/residual monomers or non-hazardous impurities/residual monomers (>1% by weight). 6 It is also described as containing no additives/adjuvants.
Use
Cosmetic
The safety of these cosmetic ingredients is evaluated based, in part, on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of this ingredient in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. 19 Use data are submitted by the cosmetics industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category. 20
NR = Not Reported.
*Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
aIt is possible that these products may be sprays, but it is not specified whether the reported uses are sprays.
bNot specified these products are sprays or powders, but it is possible the use can be as a spray or powder, therefore the information is captured in both categories.
cIt is possible that these products may be powders, but it is not specified whether the reported uses are powders.
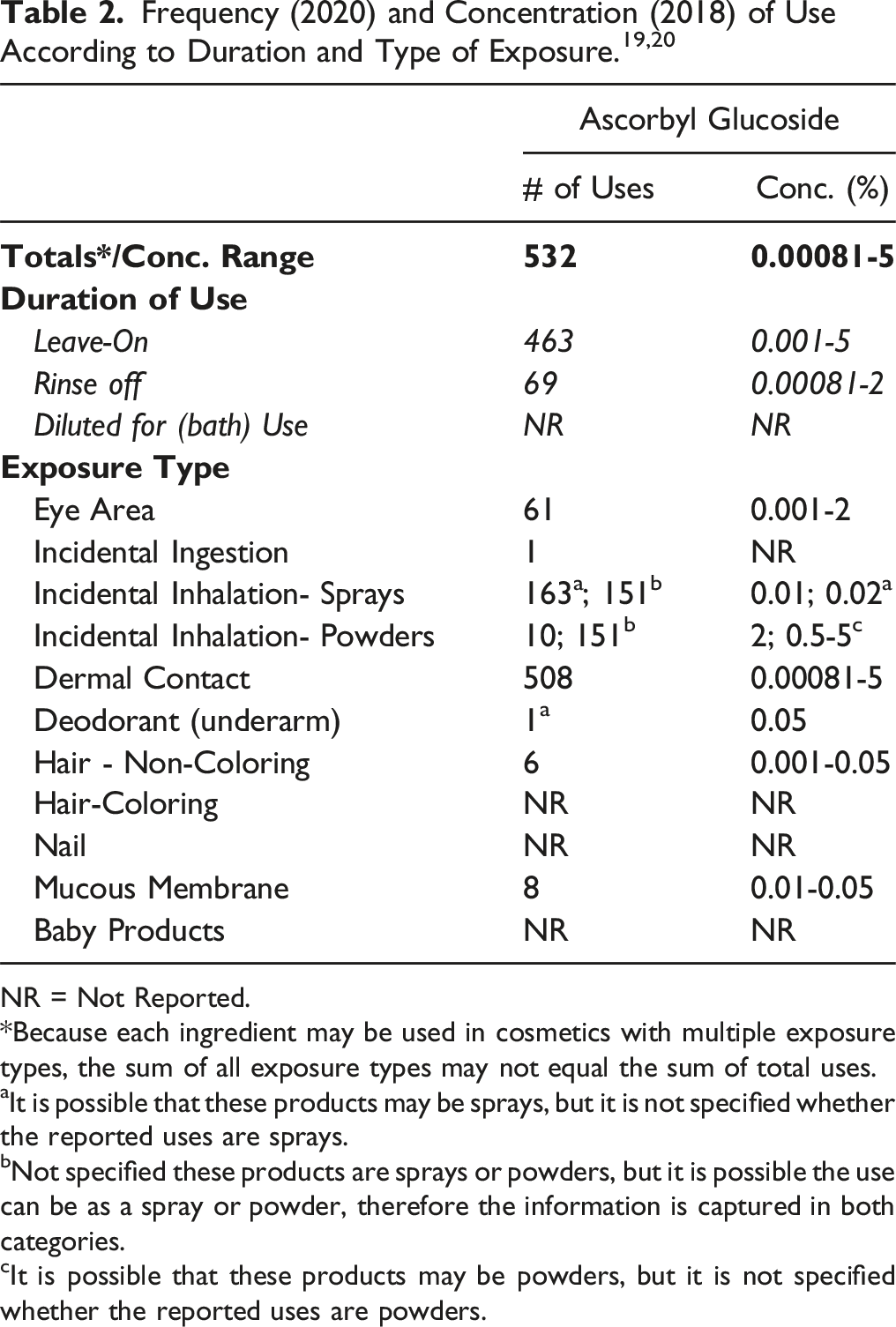
Cosmetic products containing Ascorbyl Glucoside may be applied to the skin at concentrations up to 5% (in face and neck skin care preparations) and may come in contact with the eyes during use of eye makeup preparations (highest reported maximum use concentration of 2% in eye lotions). Ascorbyl Glucoside also could be incidentally ingested during product use (e.g., use in lipsticks; concentrations not reported). Products containing Ascorbyl Glucoside may be applied as frequently as several times per day and may come in contact with the skin for variable periods following application. Daily or occasional use may extend over many years.
Ascorbyl Glucoside is reported to be used in both pump and aerosol hair sprays at concentrations up to 0.01%. 20 In practice, most droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles below 10 μm, compared with pump sprays.21–24 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.21,22 Ascorbyl Glucoside is reported to be used in face powders at concentrations up to 2%. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.25–27
Ascorbyl Glucoside and Sodium Ascorbyl Glucoside are not restricted from use in any way under the rules governing cosmetic products in the European Union. 28
Non-Cosmetic
The use of Ascorbyl Glucoside as a vitamin C supplement in foods has been reported. 15 Furthermore, hydrophilic ascorbic acid derivatives such as Ascorbyl Glucoside have been used not only as antioxidants, but also as food and pharmaceutical excipients. 29 However, it is important to note that Ascorbyl Glucoside does not appear on the FDA’s list of indirect additives used in food contact substances or in the FDA’s substances added to food inventory.30,31 Ascorbyl Glucoside also does not appear in the FDA’s inactive ingredient database for FDA-approved drug products. 32
Skin bleaching is a drug function, not a cosmetic function, in the US. Ascorbyl Glucoside has been identified as a bleaching agent in commercial bleaching cosmetics in Taiwan. 33 Commercial bleaching cosmetics contained additional ingredients such as kojic acid and niacinamide, which are associated with skin whitening effects. In commercial cosmetic bleaching creams (2 creams) and in a commercial cosmetic bleaching lotion, Ascorbyl Glucoside has been detected at concentrations of 1.89% and 2.05%, respectively, using high-performance liquid chromatography (HPLC). In South Korea, Ascorbyl Glucoside has been identified as one of the main ingredients used in cosmetics that are reported to function as skin whitening agents. 34
Toxicokinetic Studies
Dermal Penetration
In Vitro
A three-dimensional cultured human skin model (living skin equivalent) was used to study the percutaneous absorption of Ascorbyl Glucoside in vitro. 35 The skin model consisted of the following: stratum corneum and viable epidermis from human keratinocytes, and a collagen matrix composed of human dermal fibroblasts and a polycarbonate membrane. The skin model was mounted between the donor and receptor compartments of a modified Franz cell (effective area = 1.7 cm2). The donor compartment contained 1% Ascorbyl Glucoside in phosphate buffer (100 μl), and the receptor compartment contained phosphate buffer only. The donor solution containing the test substance was soaked into a wiping cloth that was set on the stratum corneum. At predetermined times, a 200 μl sample was withdrawn from the receptor solution and the donor was removed using wipes. Both surfaces of the stratum corneum side and the polycarbonate side of the skin model were washed with phosphate buffer, and the model was separated into the skin, collagen matrix, and polycarbonate membrane. The donor (wipes), skin, collagen matrix, and polycarbonate membrane were placed separately into sample vials with phosphate buffer and maintained for 24 h for chemical extraction. Chemical concentration was assayed by HPLC. The recovery rate of Ascorbyl Glucoside was 84.56%. The steady-state penetration flux of Ascorbyl Glucoside, evaluated by the linear portion of the penetration profile, was 0.91 ± 0.15 μg/cm2/h. Ascorbyl Glucoside only slightly penetrated through the hydrophobic layer stratum corneum, because it is a highly hydrophilic compound. The log Pow was not calculated because Ascorbyl Glucoside does not readily dissolve in octanol. The ratio of Ascorbyl Glucoside in the skin (defined as the percentage of Ascorbyl Glucoside in each section (μg) divided by the total amount of Ascorbyl Glucoside in the skin model) was 7.24%, and more than 90% of the Ascorbyl Glucoside remained in the wipe at 6 h.
The in vitro percutaneous absorption of Ascorbyl Glucoside was evaluated using excised skin (2 cm × 2 cm) from 8 pigs. 36 Each excised skin sample was mounted in a Franz diffusion assembly, with the stratum corneum facing the donor compartment. The donor compartment (0.5 ml) contained Ascorbyl Glucoside in a citrate-phosphate buffer (pH 7.4; quantity of this solution added to the compartment not stated). The receptor compartment (5.5 ml) was filled with citrate-phosphate buffer. The available area of the glass cylinder was 0.7854 cm2. At appropriate intervals, 300 μl aliquots of receptor medium were withdrawn and immediately replaced with an equal volume of fresh receptor solution. The length of the sampling period was 12 h, and the amount of Ascorbyl Glucoside in the receptor medium was determined using HPLC. Results indicated there was no flux of Ascorbyl Glucoside across the skin samples.
Human
An experiment was performed to evaluate the percutaneous absorption of Ascorbyl Glucoside. 37 To measure percutaneous absorption, a cream containing 2% Ascorbyl Glucoside (7.5 g) was applied to the legs of 5 male subjects. The area to which the test cream was applied was covered for 14 h with cling film and a bandage. Urine samples were taken every 2 h for 26 h. A cream containing no ascorbic acid derivative (placebo) was applied to one other male subject. Though the increase was not a sharp increase, the amount of ascorbic acid excreted in the urine was increased over the amount that was measured prior to cream application. Furthermore, the amount of ascorbic acid excreted into the urine was sustained at virtually the same level for 10 h and peaked at 14 h. Even after the cream was removed at 16 h post-application, the excretion of ascorbic acid into the urine continued up to 26 h. The increase in ascorbic acid concentration in the urine seemed to originate solely from the cream because the subjects were not allowed to take ascorbic acid orally. In the control subject, the amount of ascorbic acid excreted into the urine did not change.
The distribution of Ascorbyl Glucoside in the skin after application of the 2% Ascorbyl Glucoside cream to the forearm was also evaluated. To determine the distribution of ascorbic acid in the skin, the cream was applied to an area on the inside of the forearm. The area was then covered with cling film and a bandage for 12 h. Samples were taken from the area by punch biopsy at 12 h and 3 d after the bandage and cling film had been removed. The skin samples were fixed and prepared for electron microscopy. In the micrographs of skin treated with the cream, small black silver particles indicated the presence of ascorbic acid in the skin. Silver particles were observed between epidermal cells for as long as 3 d. The authors noted that these results indicate that Ascorbyl Glucoside releases ascorbic acid in the skin, that is, sustained release. These particles were not observed in micrographs of skin treated with a cream containing no ascorbic acid derivative. The authors noted that the results of the 2 experiments in this study combined indicate that Ascorbyl Glucoside (in cream) was absorbed percutaneously and converted to ascorbic acid through its metabolism in the skin and other parts of the body. It could also be stated that these results indicate that ascorbic acid from dermally applied Ascorbyl Glucoside was absorbed.
Absorption, Distribution, Metabolism, and Excretion (ADME)
In Situ
The intestinal absorption of Ascorbyl Glucoside in fasted Hartley guinea pigs was determined using the perfusion technique.38,39 Various concentrations of Ascorbyl Glucoside (0.2, 0.5, 1.0, or 5.0 mM) in isotonic phosphate buffer were perfused in the small intestine of the animals. At 2 h after perfusion, blood from the portal vein was collected to determine Ascorbyl Glucoside content, in that compounds absorbed from the small intestine accumulate primarily in the portal vein. Following perfusion, the amount of intact Ascorbyl Glucoside collected in the perfusate was less than the amount perfused, and an increase in ascorbic acid was observed. Intact Ascorbyl Glucoside was not detected in the plasma of the portal vein at 2 h after perfusion. The disappearance of Ascorbyl Glucoside from the perfusate was completely inhibited by the addition of castanospermine, a specific α-glucosidase inhibitor, or by carbohydrates such as maltose. These results indicate that ascorbic acid released from Ascorbyl Glucoside by α-glucosidase on the brush border membrane is effectively taken up across the intestinal ascorbate transport channels, into a serosal site, whereas Ascorbyl Glucoside permeation was poor via the passive transport system. Thus, Ascorbyl Glucoside administered orally could be hydrolyzed to ascorbic acid by α-glucosidase on brush border membranes, and absorbed as ascorbic acid from the small intestine in guinea pigs.
Animal
Oral
The in vivo formation of Ascorbyl Glucoside in guinea pigs and rats given ascorbic acid orally in combination with maltose was examined. 40 A metabolite of ascorbic acid that has the same HPLC retention characteristics as authentic Ascorbyl Glucoside was detected in the blood, urine and liver of guinea pigs 1 to 2 h after their administration. The metabolite was isolated from the urine by chromatographic procedures and identified as Ascorbyl Glucoside by its ultraviolet light (UV) spectrum, non-reducibility, susceptibility to α-glucosidase hydrolysis, HPLC profile, and elemental analysis. The same glucoside was also synthesized by rats and found in the urine, although it could not be determined qualitatively in the blood. The authors concluded that Ascorbyl Glucoside is a possible metabolite produced by enzymatic α-glucosidation after combined administration of ascorbic acid and maltose to guinea pigs and rats.
The ADME of Ascorbyl Glucoside was investigated using groups of male Hartley guinea pigs. 41 (Ascorbic acid activity (i.e., effect on body weight gain and serum alkaline phosphatase activity) was evaluated by comparing the effects of oral dosing with ascorbic acid versus Ascorbyl Glucoside, and these results are presented in the section on Short-Term Toxicity Studies.) Eight groups were fed ascorbic acid-deficient diet supplemented with either 0.96, 1.92, 9.6, or 192 mg/animal/d Ascorbyl Glucoside or 0.5, 1, 5, or 100 mg/animal/d ascorbic acid for 24 d; 5 animals were used per group, with the exception that n = 15 for the 5 mg/animal/d ascorbic acid group. The control group was fed ascorbic acid-deficient diet only, which was defined as follows: casein (20%), sucrose (12%), corn (20%), soybean oil (5%), soybean meal (5%), wheat bran (20%), alfalfa meal (10%), vitamin mixture (ascorbic acid-free, 1%), and mineral mixture (7%). In the Ascorbyl Glucoside dietary groups, Ascorbyl Glucoside was not detected in the liver, adrenal glands, or urine. However, ascorbic acid was detected (in the urine and in these organs), and the ascorbic acid concentration increased with increasing Ascorbyl Glucoside dosage. The ascorbic acid concentration in the tissues of each Ascorbyl Glucoside dose group was higher than that of the control group, and was similar to that of the groups fed ascorbic acid in the diet.
Animal and Human
Oral
A study was performed in which the bioavailability of Ascorbyl Glucoside as ascorbic acid in humans (8 subjects) was compared to that in rats (6 animals).13,42 The aim of the study was to clarify that Ascorbyl Glucoside is hydrolyzed by human intestinal maltase, and that oral ingestion of Ascorbyl Glucoside is physiologically utilized the same as orally administered ascorbic acid in human subjects. The hydrolyzing activities to Ascorbyl Glucoside by human or rat intestinal homogenates were measured using HPLC. Following an overnight fast, the subjects orally ingested 3.84 g of Ascorbyl Glucoside (2 g of ascorbic acid equivalent) dissolved in 100 ml of water. Venous blood (2 ml quantities) was drawn from the forearms prior to ingestion and at 1, 2, 3, and 4 h after ingestion. At least 1 wk later, ascorbic acid (2 g) was ingested and blood was collected according to the same method. In the rat study, 76.8 mg of Ascorbyl Glucoside (40 mg of ascorbic acid equivalent) were administered by stomach tube. Blood was collected from the tail vein prior to dosing and at 1, 2, and 3 h post-dosing. One week later, ascorbic acid (40 mg) was administered using the same method. Serum concentrations of ascorbic acid between humans and rats were compared.
In humans, Ascorbyl Glucoside was digested by intestinal maltase, and hydrolyzing activity was higher (but not statistically significantly different) when compared to the rat. The average concentration of serum ascorbic acid at basal levels in human subjects was 0.9 mg/100 ml (within normal range). The serum ascorbic acid concentrations increased from 0.9 mg/100 ml to 2.5 mg/100 ml within 2 h and were maintained at these levels until 4 h post-administration of Ascorbyl Glucoside in human subjects. In rats, the serum ascorbic acid concentration increased linearly from 1.1 mg/100 ml to 1.6 mg/100 ml within 3 h after dosing. Thus, the increment of the serum concentration of ascorbic acid in rats was less when compared to humans. These results suggest that the absorption of ascorbic acid in humans occurs at higher levels and at a faster rate when compared to rats. The authors concluded that Ascorbyl Glucoside is hydrolyzed by intestinal maltase and acts as ascorbic acid in humans.
Toxicological Studies
Acute Toxicity Studies
Dermal
The acute dermal toxicity of Ascorbyl Glucoside (99.8% to 100% pure) was evaluated using 10 Wistar rats (5 males, 5 females). 6 The study was performed in accordance with Organization for Economic Co-Operation and Development (OECD) Test Guideline (TG) 402. Each animal received a single 2000 mg/kg dose of the test substance in distilled water. The dose was administered under a semi-occlusive dressing. The dressing was removed on day 2. None of the animals died, but the test substance caused yellow discoloration of the skin (initially observed on day 2) at the application sites of all test animals. Skin discoloration was reported until termination of the study; however, the number of animals with skin discoloration at the end of the study was not stated. There were no signs of systemic toxicity or effect on organs. The authors concluded that Ascorbyl Glucoside was of low toxicity (LD50 > 2000 mg/kg) when administered dermally.
Oral
The acute oral toxicity of Ascorbyl Glucoside (99.8% to 100% pure) was evaluated using 3 groups of 10 rats (5 males and 5 females per group) of the Crj:CD strain. 6 The study was performed in accordance with OECD TG 401. A single dose of the test substance (in deionized water) was administered by gavage, at doses of 1000 mg/kg and 2000 mg/kg. The control group received deionized water only. On the day after dosing, soft stool or muddy stool was observed sporadically in test and control groups. None of the animals died, and no effects on the organs (not identified) of test or control animals were observed. The authors concluded that Ascorbyl Glucoside was of low toxicity (LD50 > 2000 mg/kg) when administered orally.
Short-Term Toxicity Studies
Oral
As stated in the ADME section, the ascorbic acid activity (i.e., effect on body weight gain and serum alkaline phosphatase activity) of Ascorbyl Glucoside in guinea pigs was investigated using 9 groups of male guinea pigs fed ascorbic acid-deficient diet supplemented with 0–192 mg/animal/d Ascorbyl Glucoside (5 groups), or fed 0.5–100 mg/animal/d ascorbic acid for 24 d (5 animals/group, with the exception that 15 animals were fed 5 mg/animal/d ascorbic acid in diet). 41 Study details are found in the ADME section. The body weight gain, serum alkaline phosphatase activity, and the concentration of ascorbic acid or Ascorbyl Glucoside in the liver, adrenal glands, and urine were measured at the end of the experimental period. Guinea pigs fed diet supplemented with Ascorbyl Glucoside had similar body weight gain when compared to guinea pigs fed diet supplemented with an equimolar amount of ascorbic acid. Serum alkaline phosphatase activity in Ascorbyl Glucoside and ascorbic acid dietary groups was statistically significantly (P < 0.05) higher when compared to the control group. However, there was no significant difference in serum alkaline phosphatase activity between the Ascorbyl Glucoside and ascorbic acid dietary groups. The authors concluded that these results, together with the distribution and excretion data in guinea pigs (as described in the ADME section), indicated that Ascorbyl Glucoside has the same effects as ascorbic acid on a molar basis.
Groups of 20 Wistar rats (10 males and 10 females per group) were dosed with Ascorbyl Glucoside (in distilled water) daily for 28 d. 6 There were 3 test groups and 1 control group. Ascorbyl Glucoside was administered by gavage at doses of 50 mg/kg, 200 mg/kg, and 1000 mg/kg, 7 d/wk, in accordance with OECD TG 407. The control group was dosed with distilled water. None of the animals died, and there were no clinical signs nor effects on food consumption or body weight gain. Laboratory findings relating to clinical chemistry, hematology, or urinalysis did not reveal any test substance-related changes. There also were no test substance-related changes in organ weights, or any test substance-related macroscopic or microscopic findings. The authors concluded that the no observed adverse effect level (NOAEL) for Ascorbyl Glucoside was 1000 mg/kg/d, the highest dose administered in this study.
Developmental and Reproductive Toxicity Studies
Data on the developmental and reproductive toxicity of Ascorbyl Glucoside and Sodium Ascorbyl Glucoside were neither found in the published literature, nor were these data submitted.
Genotoxicity Studies
In Vitro
The genotoxicity of Ascorbyl Glucoside (in distilled water) was evaluated in the Ames test using the following bacterial strains: Salmonella typhimurium strains TA98, TA100, TA1535, and TA1537, and Escherichia coli strain WP2 uvrA. 6 The test substance was evaluated at doses up to 5000 μg/plate, with and without metabolic activation. Dose-dependent increases in mutation frequency were not observed in any of the bacterial strains tested with Ascorbyl Glucoside. Both negative (distilled water) and positive controls (not stated) yielded appropriate responses. The authors concluded that Ascorbyl Glucoside was not genotoxic under the conditions of this test.
Ascorbyl Glucoside (in growth medium) was also evaluated for clastogenic potential using Chinese hamster Don cells. 6 The test substance was evaluated with and without metabolic activation at concentrations of 500 μg/ml, 1000 μg/ml, and 2000 μg/ml. The 50% growth inhibitory concentration, with or without metabolic activation, was ∼2000 μg/ml. When compared to control (not stated) levels, there was no increase in the frequency of cells with chromosomal aberrations either with or without metabolic activation. The positive controls (not stated) yielded the expected responses. The authors concluded that Ascorbyl Glucoside was not clastogenic to Chinese hamster Don cells under the conditions of this test.
The genotoxicity of Ascorbyl Glucoside (in growth medium) was evaluated in the mammalian chromosome aberration test (OECD TG 473) using Chinese hamster V79 cells. 6 The test substance was evaluated at concentrations up to 3400 μg/ml both with and without metabolic activation. There was no evidence of cytotoxicity over the range of concentrations tested, with or without metabolic activation. When compared to control (not stated) levels, no increase in the frequency of cells with chromosomal aberrations was observed either with or without metabolic activation. The positive controls (not stated) yielded the expected responses. The authors concluded that Ascorbyl Glucoside was not clastogenic to Chinese hamster V79 cells under the conditions of this test.
In Vivo
The genotoxicity of Ascorbyl Glucoside was evaluated in the micronucleus test using groups of 10 mice (5 males and 5 females per group) of the S1c:ICR strain. 6 The test substance (in physiological saline) was injected intraperitoneally (i.p.) at doses of 500 mg/kg, 1000 mg/kg, and 2000 mg/kg. The animals were killed at 24 h post-injection, and bone marrow cells were obtained and prepared for microscopic examination. Physiological saline and mitomycin C served as negative and positive controls, respectively. When compared to control levels, there were no increases in the frequency of micronucleated polychromatic erythrocytes at the administered doses. The authors concluded that Ascorbyl Glucoside was non-clastogenic under the conditions of this test. The positive control was genotoxic.
Carcinogenicity Studies
Data on the carcinogenicity of Ascorbyl Glucoside and Sodium Ascorbyl Glucoside were neither found in the published literature, nor were these data submitted.
Tumor Promoter Inhibition
The effect of Ascorbyl Glucoside (30 μM and 100 μM) at the promotion stage in the two-stage BALB/c 3T3 cell transformation assay was evaluated. 43 When BALB/c 3T3 cells were treated with 0.2 mg/ml 20-methylcholanthrene (MCA) as an initiator, and 100 ng/ml 12-O-tetradecanoylphorbol-13-acetate (TPA) as a promoter, the addition at the promotion stage of Ascorbyl Glucoside resulted in inhibition of transformation. When compared to the transformation control (treatment with MCA followed by TPA), the inhibition by 100 μM Ascorbyl Glucoside (31% inhibition) was statistically significant.
Other Relevant Studies
Effect on Melanin Synthesis
The effect of Ascorbyl Glucoside on melanin synthesis was studied using B16 melanoma cells. 37 Cells were cultured in the presence of Ascorbyl Glucoside (2 mmol/l). At 24 h before the cells were harvested, the cells were treated with theophylline and then trypsin. After the cells were harvested, the number of viable cells was determined, and the amount of melanin was measured. Melanoma cells to which no ascorbic acid derivative had been added served as the control. Ascorbyl Glucoside inhibited melanin synthesis. It caused a statistically significant (P < 0.01) reduction in melanin synthesis when compared to control values (baseline). This reduction was sustained over a period of ≥30 h. In another experiment in this study, B16 melanoma cells were cultured with Ascorbyl Glucoside (2 mmol/l) for 48 h, and changes in cell pigmentation were monitored. After 2 d of incubation with Ascorbyl Glucoside, a statistically significant decrease in melanin pigmentation was observed. Decreased melanin pigmentation was observed on days 1 and 2, indicating that Ascorbyl Glucoside had a sustained effect with respect to lightening the color of melanin.
Effect on Collagen Synthesis
The effect of Ascorbyl Glucoside on collagen synthesis was evaluated using cultured human skin fibroblasts.12,44 Ascorbyl Glucoside effectively stimulated collagen synthesis at concentrations of 0.1–0.5 mM. Continuous supplementation of Ascorbyl Glucoside (0.25 mM) to culture medium for 24 d enhanced cell growth to three-times that of control skin fibroblast cultures.
Cytotoxicity
Cultured human skin (abdominal) fibroblasts at different cell densities were used in a study evaluating the cytotoxicity of Ascorbyl Glucoside and ascorbic acid. 45 The fibroblasts were cultured for 24 h in medium containing Ascorbyl Glucoside (1 mM) or ascorbic acid (1 mM). Cell numbers (estimated by measuring uptake of neutral red) ranged from 0.625 × 104 cells per well to 10 × 104 cells per well. Results were expressed as the means of duplicate cultures. After 24 h of incubation of fibroblasts (cell density of 0.625 × 104 cells/well) with 1 mM Ascorbyl Glucoside, the cell number was 0.37 × 104 cells/well; the cell number in the control culture (same cell density) was 0.35 × 104 cells/well. After 24 h of incubation of fibroblasts at a higher cell density (cell density of 10 × 104 cells/well) with 1 mM Ascorbyl Glucoside, the cell number was 9.6 × 104 cells/well; the cell number in the control culture (same cell density) was 6.6 × 104 cells/well. These results indicate that Ascorbyl Glucoside was not cytotoxic to cell cultures of different densities. Similarly, Ascorbyl Glucoside was not cytotoxic to cultures at cell densities between 0.625 × 104 cells/well and 10 × 104 cells/well. However, ascorbic acid (1 mM) was cytotoxic in low density fibroblast cell cultures.
Effect on UVB-Induced Cytotoxicity
The inhibitory effect of Ascorbyl Glucoside on mid-wavelength ultraviolet light (UVB)-induced cytotoxicity was evaluated using HaCaT human keratinocytes, used as a skin model in this study. 46 The cytotoxicity assay (WST-1 assay) used is a modified 3-(4,5-dimethylthiazole-2-yl)-2,5-biphenyl tetrazolium bromide assay. After 12 h of incubation with Ascorbyl Glucoside, the cells were exposed to UVB and then cultured for 24 h. UVB irradiation of cell cultures was performed using 2 light sources (range: 280 to 360 nm). Results indicated that cell viability decreased significantly when cultures were exposed to UVB at 0.1 to 0.4 J/cm2, in a dose-dependent manner. Cell viability was ∼25% at a UVB dose of 0.4 J/cm2. Ascorbyl Glucoside dose-dependently (0.5 to 5 mM) suppressed UVB (0.25 J/cm2)-induced cytotoxicity in HaCaT cells. At a concentration of 5 mM Ascorbyl Glucoside, cell viability was 70.3% when compared to the control cultures.
Dermal Irritation and Sensitization Studies
Irritation
Animal
The skin irritation/corrosion potential of undiluted Ascorbyl Glucoside was evaluated in accordance with OECD TG 404 using 3 New Zealand White rabbits. 6 The test substance (0.5 g in distilled water) was applied for 4 h under a semi-occlusive dressing, and the test site was observed for 72 h. The area of application was ∼6 cm2. Neither erythema nor edema was observed in the animals tested. The authors concluded that Ascorbyl Glucoside was non-irritating to the skin of rabbits.
Sensitization
Animal
The skin sensitization potential of Ascorbyl Glucoside was evaluated in the maximization test (OECD TG 406) using guinea pigs (strain not stated). 6 Groups of 20 and 10 animals served as test and control groups, respectively. The induction phase involved intradermal injection of 1% Ascorbyl Glucoside and topical application of 75% Ascorbyl Glucoside. For topical application, a filter paper (2 cm × 4 cm) was fully-loaded with the test substance in a suitable vehicle (not stated) and applied under an occlusive dressing for 48 h. Control animals received a 48-h application of the vehicle, under an occlusive dressing. All test and control animals were pre-treated with 10% sodium lauryl sulfate (in liquid paraffin) at 1 day prior to topical induction. The challenge phase involved a 24-h topical application of Ascorbyl Glucoside (under occlusive dressing) to the flank at concentrations of 15, 25, 50, and 75%. At topical induction with the test substance for 24 h, slight erythema was observed in 1 animal. However, none of the animals (test or controls) exhibited erythema or edema at the challenge site. The authors concluded that there was no evidence of reactions to Ascorbyl Glucoside that were indicative of skin sensitization.
Human
A human repeated insult patch test (HRIPT) on a rinse-off product containing 0.1% Ascorbyl Glucoside was performed using 103 subjects. 47 Dilution of the product to a 2% aqueous solution (effective Ascorbyl Glucoside test concentration = 0.002%) was performed prior to testing. The diluted product (0.1 ml) was applied, under an occlusive patch, for 48 h and 72 h. The location of the patch test site and the dose per cm2 were not stated. Patch application was repeated over a 3-wk induction period, which consisted of 9 induction applications. Reactions (induction) were scored according to the following scale: 0 (no evidence of irritation) to 7 (strong reactions spreading beyond test site). The induction phase was followed by a 2-wk non-treatment period. A challenge patch was then applied to a new test site (not identified). Challenge reactions were scored at 48 h and 96 h according to the following scale: 0 (no evidence of erythema) to 3 (severe erythema (very intense redness)). Reactions with a grade of 0 or 1 were classified as low-level reactions. Reactions with a grade of 2 and above were classified as high-level reactions. One subject had a low-level reaction, and 2 subjects had a high-level reaction during induction. Neither low-level nor high-level reactions were observed during challenge. The authors noted that the diluted product did not induce an allergic response, and concluded that it did not induce dermal sensitization in any of the subjects tested.
A second HRIPT was performed on a leave-on product containing 2% Ascorbyl Glucoside (undiluted), and was performed using 113 subjects. 47 The test protocol is the same as that used in the preceding test, except for the following: amount applied (25 μl), only a 48-h patch application period, the scoring of challenge reactions at 30 min, 24 h, and 48 h, and the grading scale used. Locations of patch test sites and the dose per cm2 were not stated. The following International Contact Dermatitis Research Group (ICDRG) grading scale was used for the evaluation of induction and challenge reactions: 0 (no reaction) to 3 (extreme (bullous or ulcerative). The definitions of low-level and high-level reactions, same as stated in the preceding test were used. Two subjects had a low-level reaction during induction; there were no high-level induction reactions. One subject had a low-level reaction during challenge; there were no high-level challenge reactions. The authors noted that the undiluted product did not induce an allergic response, and concluded that it did not induce dermal sensitization in any of the subjects tested.
The skin irritation and sensitization (contact allergy) potential of Ascorbyl Glucoside (10% solution) was evaluated in an HRIPT involving 51 subjects. 48 The Fitzpatrick skin types distribution for these subjects was as follows: type 2 (burns easily, tans slightly—7 subjects), type 3 (burns moderately, tans progressively—39 subjects), and type 4 (burns a little, always tans—5 subjects). Occlusive patches containing the test material were affixed to skin of the dorsal intrascapular regions (to right or left of midline). The subjects were instructed to remove the patches 48 h after the first application, and after 24 h for the remainder of the study. Patch application “was repeated until a series of 9 consecutive, 24-h induction exposures had been made 3 times/wk for 3 consecutive weeks.” (The report includes scores for 9 induction applications, so it is uncertain if the first patch was actually applied for 24 h or 48 h.) The induction phase was followed by a 10- to 14-d non-treatment period. During the challenge phase, an occlusive patch containing the test material was applied to a new site. Reactions were scored at 48 h and 96 h post-application according to the following scale: 0 (no reaction) to 4 (erythema, induration, and bullae. No adverse reactions of any kind were reported during the course of the study. The authors concluded that, under the conditions of the study, there were no identifiable signs or symptoms of sensitization (contact allergy) after application of the 10% Ascorbyl Glucoside solution.
Ocular Irritation Studies
Animal
The ocular irritation potential of undiluted Ascorbyl Glucoside was evaluated in accordance with OECD TG 405 using 3 New Zealand White rabbits. 6 The test substance was instilled into the eye of each animal, and this was followed by a 7-d observation period. A mean ocular irritation score was calculated on the basis of scores at 24, 48, and 72 h for each animal. Discharge and slight effects (not described) on the iris were observed in one animal. The authors concluded that Ascorbyl Glucoside was slightly irritating to the eye.
Summary
The safety of Ascorbyl Glucoside and Sodium Ascorbyl Glucoside, as used in cosmetics, is reviewed in this safety assessment. According to the Dictionary, Ascorbyl Glucoside is reported to have the following functions in cosmetics: antioxidant, and skin-conditioning agent—miscellaneous.
Ascorbyl Glucoside is synthesized by a biocatalytic transglucosylation in which starch-derived cyclic or linear oligosaccharides are reacted with l-ascorbic acid. According to another source, transglycosylation in the presence of the enzyme cyclodextrin glucanotransferase (CGTase, from Paenibacillus sp.) has been used to produce Ascorbyl Glucoside.
According to 2020 VCRP data, Ascorbyl Glucoside is reported to be used in 532 cosmetic products (463 leave-on and 69 rinse-off). The results of a concentration of use survey conducted in 2018 indicate that Ascorbyl Glucoside is used at concentrations up to 5% (in face and neck skin care preparations, not spray), which is the highest reported maximum use concentration for leave-on formulations. In rinse-off products, Ascorbyl Glucoside is reported to be used at concentrations up to 2% (in paste masks and mud packs According to VCRP and Council survey data, Sodium Ascorbyl Glucoside is not being used in cosmetic products.
In vitro skin penetration data indicated slight (human skin) to no (pig skin) percutaneous absorption of Ascorbyl Glucoside. After application of a cream containing 2% Ascorbyl Glucoside to the legs and forearms of 5 male subjects, Ascorbyl Glucoside was absorbed percutaneously and converted to ascorbic acid through its metabolism in the skin and other parts of the body. It could also be stated that the results of this study indicate that ascorbic acid from dermally applied Ascorbyl Glucoside was absorbed.
The in vivo formation of Ascorbyl Glucoside in guinea pigs and rats was observed after oral administration of ascorbic acid in combination with maltose. The authors concluded that Ascorbyl Glucoside is a possible metabolite produced by enzymatic α-glucosidation after combined administration of ascorbic acid and maltose to guinea pigs and rats. In another study, groups of guinea pigs were fed an ascorbic acid-deficient diet supplemented with Ascorbyl Glucoside (0.96–192 mg/animal/d) or with equimolar amounts ascorbic acid (0.5–100 mg/animal/d). In the Ascorbyl Glucoside dietary groups, Ascorbyl Glucoside was not detected in the liver, adrenals, or urine. However, ascorbic acid was detected in the urine and in these organs, and the ascorbic acid concentration increased with increasing Ascorbyl Glucoside dosage. Thus, it was concluded that Ascorbyl Glucoside was metabolized to ascorbic acid.
Various concentrations of Ascorbyl Glucoside (0.2, 0.5, 1.0, or 5.0 mM) in isotonic phosphate buffer were perfused in the small intestine of fasted guinea pigs, and blood from the portal vein was collected. Following perfusion, the amount of intact Ascorbyl Glucoside collected in the perfusate was less than the amount perfused, and an increase in ascorbic acid was observed in portal vein blood. A study was performed that compared the bioavailability of Ascorbyl Glucoside as ascorbic acid between humans and rats. Serum ascorbic acid concentrations increased in both humans and rats after oral dosing, and the results suggested that the absorption of ascorbic acid in humans occurs at higher levels and at a faster rate when compared to rats.
The acute dermal toxicity of Ascorbyl Glucoside (concentration not stated) was evaluated using 10 Wistar rats, and an LD50 of >2000 mg/kg was reported. None of the animals died, and there were no signs of systemic toxicity or effect on organs. An LD50 of >2000 mg/kg was also reported in a study in which 3 groups of 10 rats of the Crj:CD strain were dosed orally with Ascorbyl Glucoside (concentration not stated). There were no animal deaths and no signs of systemic toxicity or organ effects.
Five groups of guinea pigs (mostly groups of 5) were fed an ascorbic acid-deficient diet supplemented with 0–192 mg/animal/d Ascorbyl Glucoside, and another 4 groups were fed an ascorbic acid-deficient diet supplemented with equimolar amounts of 0.5–100 mg/animal/d ascorbic acid. Guinea pigs fed diet supplemented with Ascorbyl Glucoside had similar body weight gain when compared to guinea pigs fed diet supplemented with an equimolar amount of ascorbic acid. Serum alkaline phosphatase activity in both Ascorbyl Glucoside and ascorbic acid dietary groups was statistically significantly (P < 0.05) higher when compared to the control group. However, there was no significant difference in serum alkaline phosphatase activity between the Ascorbyl Glucoside and ascorbic acid dietary groups.
In a study in which groups of 10 male and 10 female Wistar rats were dosed by gavage with up to 1000 mg/kg/d Ascorbyl Glucoside (in distilled water) for 28 d, no adverse effects were reported. The NOAEL was 1000 mg/kg/d.
In the Ames test, Ascorbyl Glucoside (doses up to 5000 μg/plate) was not genotoxic in the following bacterial strains, with and without metabolic activation: S typhimurium strains TA98, TA100, TA1535, and TA1537 and E coli strain WP2 uvrA. Ascorbyl Glucoside was not clastogenic to Chinese hamster Don cells (with and without metabolic activation) at concentrations up to 2000 μg/ml. The in vitro genotoxicity of Ascorbyl Glucoside was also evaluated in the mammalian chromosome aberration test using Chinese hamster V79 cells. At concentrations up to 3400 μg/ml, results were negative with and without metabolic activation. The in vivo genotoxicity of Ascorbyl Glucoside was evaluated in the micronucleus test using groups of 10 mice of the Slc:ICR strain injected i.p. with the test substance. The test substance was non-clastogenic at doses up to 2000 mg/kg.
The effect of Ascorbyl Glucoside (30 μM and 100 μM) at the promotion stage in the two-stage BALB/c 3T3 cell transformation assay was evaluated. When compared to the transformation control (treatment with MCA followed by TPA), the inhibition by 100 μM Ascorbyl Glucoside (31% inhibition of transformation) was statistically significant.
After 2 d of incubation of B16 melanoma cells with Ascorbyl Glucoside (2 mmol/l), a statistically significant decrease in melanin pigmentation was observed.
In an assay using cultured human skin fibroblasts, Ascorbyl Glucoside effectively stimulated collagen synthesis at concentrations of 0.1–0.5 mM.
Cultured human skin (abdominal) fibroblasts at different cell densities were used in a study evaluating the cytotoxicity of Ascorbyl Glucoside (1 mM). The test substance was not cytotoxic to cultures at cell densities between 0.625 × 104 cells/well and 10 × 104 cells/well. Ascorbyl Glucoside dose-dependently (0.5 to 5 mM) suppressed UVB-induced cytotoxicity in cultured HaCaT human keratinocytes.
In a study involving 3 New Zealand White rabbits, undiluted Ascorbyl Glucoside was non-irritating to the skin after 4 h of application under a semi-occlusive dressing.
The skin sensitization potential of Ascorbyl Glucoside was evaluated in the maximization test (OECD TG 406) using a group of 20 guinea pigs. The challenge phase involved a 24-h topical application of the test substance (under occlusive dressing) to the flank at concentrations of 15, 25, 50, and 75%. At topical induction, slight erythema was observed in 1 animal after a 24-h application. However, none of the animals (test or controls) exhibited erythema or edema at the challenge site.
An HRIPT on a rinse-off product containing 0.1% Ascorbyl Glucoside was performed using 103 subjects. Dilution of the product to a 2% aqueous solution (effective Ascorbyl Glucoside test concentration = 0.002%) was performed prior to testing. The diluted product did not induce dermal sensitization in any of the subjects tested. A second HRIPT in the same study was on a leave-on product containing 2% Ascorbyl Glucoside (undiluted), and was performed using 113 subjects. The undiluted product did not induce dermal sensitization in any of the subjects tested. The skin irritation and sensitization (contact allergy) potential of Ascorbyl Glucoside (10% solution) was evaluated in an HRIPT involving 51 subjects. There were no identifiable signs or symptoms of sensitization (contact allergy) after application of the 10% Ascorbyl Glucoside solution.
Undiluted Ascorbyl Glucoside was slightly irritating to the eyes of 3 rabbits.
Discussion
The results of a concentration of use survey conducted in 2018 indicate that Ascorbyl Glucoside is used at concentrations up to 5% (in face and neck skin care preparations, not spray), which is the highest reported maximum use concentration for leave-on formulations. Skin penetration data (in vitro and in vivo) support the percutaneous absorption of Ascorbyl Glucoside and its metabolism to ascorbic acid. Ascorbyl Glucoside has been identified in commercial bleaching products (that also contain kojic acid) in Asia, when used at concentrations of ∼2%. The Panel noted that skin lightening is considered to be a drug effect in the US, and should not occur during the use of cosmetic products, and cosmetic formulators should only use Ascorbyl Glucoside in products in a manner that does not cause depigmentation.
The Panel noted the absence of developmental and reproductive toxicity data on Ascorbyl Glucoside and Sodium Ascorbyl Glucoside. However, concern over the lack of these data were mitigated considering that Ascorbyl Glucoside is metabolized to ascorbic acid and glucose in the skin and would not be absorbed in an appreciable quantity. Additionally, concern was further mitigated because both of these substances are essential constituents of the body, and are not reproductive and developmental toxicants.
Finally, the Panel discussed the issue of incidental inhalation exposure from the use of Ascorbyl Glucoside in pump and aerosol hair spray formulations and in face powers; the maximum reported concentration of use in these types of products is 0.01% in hair sprays and 2% in face powders. The Panel noted that in aerosol products, most droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that Ascorbyl Glucoside and Sodium Ascorbyl Glucoside* are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
*Not reported to be in current use. Were this ingredient not in current use to be used in the future, the expectation is that it would be used in product categories and at concentrations comparable to Ascorbyl Glucoside.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 555 13th Street, NW, Suite 300W, Washington, DC 20004, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.