Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of 27 wheat-derived ingredients. Most of these ingredients are reported to function as skin conditioning agents in cosmetic products. The Panel reviewed the available data to determine the safety of these ingredients. Industry should continue to use good manufacturing practices to limit impurities that could be present in botanical ingredients. The Panel concluded that 21 wheat-derived ingredients are safe in cosmetics in the practices of use and concentration described in this safety assessment. However, the Panel also concluded that the available data are insufficient to make a determination of safety that the remaining six wheat-derived ingredients are safe under the intended conditions of use in cosmetic formulations.
Keywords
Introduction
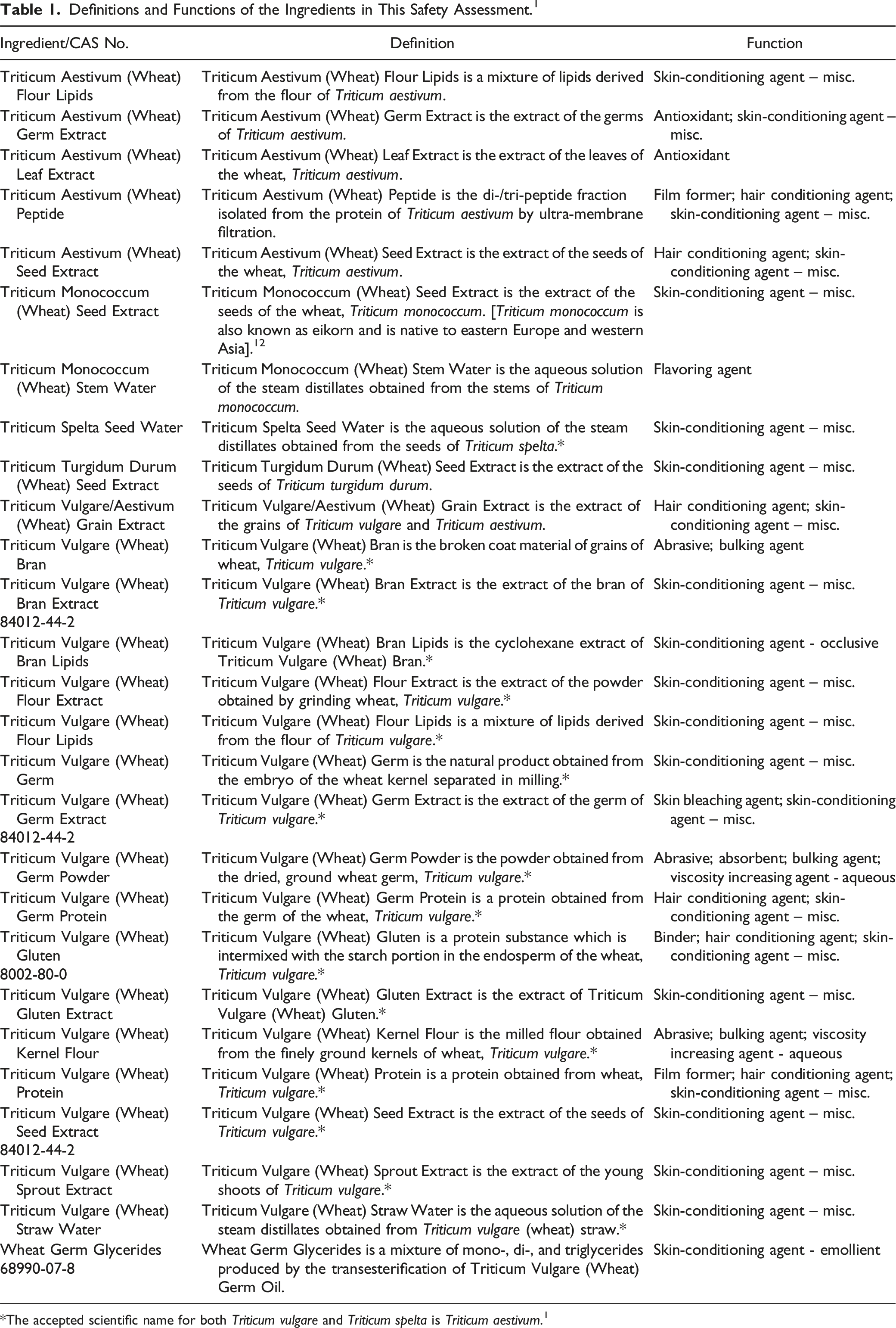
Definitions and Functions of the Ingredients in This Safety Assessment. 1
*The accepted scientific name for both Triticum vulgare and Triticum spelta is Triticum aestivum. 1
Triticum Aestivum (Wheat) Flour Lipids
Triticum Aestivum (Wheat) Germ Extract
Triticum Aestivum (Wheat) Leaf Extract
Triticum Aestivum (Wheat) Peptide
Triticum Aestivum (Wheat) Seed Extract
Triticum Monococcum (Wheat) Seed Extract
Triticum Monococcum (Wheat) Stem Water
Triticum Spelta Seed Water
Triticum Turgidum Durum (Wheat) Seed Extract
Triticum Vulgare/Aestivum (Wheat) Grain Extract
Triticum Vulgare (Wheat) Bran
Triticum Vulgare (Wheat) Bran Extract
Triticum Vulgare (Wheat) Bran Lipids
Triticum Vulgare (Wheat) Flour Extract
Triticum Vulgare (Wheat) Flour Lipids
Triticum Vulgare (Wheat) Germ
Triticum Vulgare (Wheat) Germ Extract
Triticum Vulgare (Wheat) Germ Powder
Triticum Vulgare (Wheat) Germ Protein
Triticum Vulgare (Wheat) Gluten*
Triticum Vulgare (Wheat) Gluten Extract
Triticum Vulgare (Wheat) Kernel Flour*
Triticum Vulgare (Wheat) Protein
Triticum Vulgare (Wheat) Seed Extract
Triticum Vulgare (Wheat) Sprout Extract
Triticum Vulgare (Wheat) Straw Water
Wheat Germ Glycerides*
*Previously reviewed ingredients
The safety of Wheat Flour, now known as Triticum Vulgare (Wheat) Kernel Flour, was previously reviewed by the Panel and the conclusion of “safe … in the present practices of use and concentration” was published in 1980, 2 and reaffirmed in a re-review that was published in 2003. 3 The safety of Triticum Vulgare (Wheat) Gluten and Wheat Germ Glycerides was also previously reviewed by the Panel and the conclusion of “safe when incorporated in cosmetic products and constitute no risk to the public in its present cosmetic use of these products” was published in 1980, 4 and reaffirmed in the re-review published in 2003. 3 Because it has been more than 15 years since the safety of Triticum Vulgare (Wheat) Gluten, Triticum Vulgare (Wheat) Kernel Flour, and Wheat Germ Glycerides was reviewed, these ingredients are included in this safety assessment for re-review.
The Panel has reviewed the safety of several additional wheat-derived ingredients including Wheat Amino Acids, 5 Triticum Vulgare (Wheat) Starch,2,3,6 Hydrolyzed Triticum Spelta Starch, 6 Hydrolyzed Wheat Starch, 6 Triticum Aestivum (Wheat) Germ Oil, 7 Triticum Vulgare (Wheat) Germ Oil,3,7,8 Triticum Vulgare (Wheat) Germ Oil Unsaponifiables, 7 Hydrogenated Wheat Germ Oil, 7 and Hydrogenated Wheat Germ Oil Unsaponifiables. 7 The Panel concluded that all of these ingredients are safe in the present practices of use and concentration in cosmetics. The Panel also reviewed Hydrolyzed Wheat Protein and Hydrolyzed Wheat Gluten, and concluded that these ingredients are safe for use in cosmetics when formulated to restrict peptides to an average molecular weight of 3500 Daltons (Da) or less 9 ; this conclusion is in response to reports of type 1 immediate hypersensitivity reactions that occurred in sensitized individuals following exposure to cosmetic products that contained one of these two ingredients with molecular weights greater than this limit. Polypeptides must be at least 30 amino acids long (i.e., molecular weights of about 3570 Da or more) to elicit type 1 hypersensitivity reactions (i.e., to interact with the two IgE-binding epitopes).
Most of the ingredients reviewed in this safety assessment may be consumed as food, and daily exposure from food use would result in much larger systemic exposures than those from use in cosmetic products. The primary focus of the safety assessment of these ingredients as used in cosmetics is on the potential for effects from topical exposure. Proteins from wheat in the diet, specifically wheat gluten, are associated with adverse health conditions (such as celiac disease and dermatitis herpetiformis) in a small portion of the general population. Since the maximum dose of gluten possible from cosmetics is low, there is little likelihood that cosmetic products could precipitate a flare-up of either gastrointestinal or cutaneous symptoms.4,10
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Note: In many of the published studies, it is not known how the substance being tested compares to the cosmetic ingredient. Therefore, if it is not known whether the substance being discussed is a cosmetic ingredient, the test substance will be identified as “wheat…” (e.g., wheat germ extract or wheat flour); if it is known that the substance is a cosmetic ingredient, the Dictionary nomenclature “Triticum Aestivum…” or “Triticum Vulgare…” (e.g., Triticum Aestivum (Wheat) Germ Extract or Triticum Vulgare (Wheat) Kernel Flour) will be used. Of additional note, the accepted scientific name for both Triticum vulgare and Triticum spelta is Triticum aestivum.1,11
Chemistry
Definition and Plant Identification
The definitions of the ingredients included in this review are provided in Table 1. 1 Triticum wheat species have been used as food staples for 10,000 years and originated in the Middle East. 12 Triticum aestivum L. is the most cultivated cereal grain in the world, making up about a third of total cereal grains. 13 Triticum monococcum is also known as eikorn and is native to eastern Europe and western Asia. 12 Triticum turgidum var. durum is also known as durum wheat, hard wheat, or macaroni or pasta wheat, and is commonly cultivated in the Mediterranean region.11,12 Triticum spelta is considered to be an ancient wheat grain that was once prominently grown in ancient Egypt and Italy and is now grown in northern and eastern European countries.12,14 The accepted scientific name for both Triticum vulgare and Triticum spelta is Triticum aestivum.1,11
Generic Plant Part Definitions as They Apply to Wheat-Derived Ingredients. 1

Triticum Vulgare (Wheat) Germ
Wheat germ is comprised of the embryonic axis and the scutellum of wheat seed and represents about 2.5%–3.8% of total seed weight. 16 It is a by-product of the flour milling industry.
Chemical Properties
Triticum Aestivum (Wheat) Leaf Extract
The pH of wheatgrass (the young leaves of wheat) is reported to be 7.4. 17
Triticum Vulgare (Wheat) Kernel Flour
The pH of refined wheat flours is approximately 6.0–6.1, while the pH of whole wheat flour is approximately 6.2. 13
Method of Manufacture
Triticum Monococcum (Wheat) Stem Water, Triticum Spelta Seed Water, and Triticum Vulgare (Wheat) Straw Water
In some cases, the definition of the ingredients, as given in the Dictionary, provides insight as to the method of manufacture. In general, botanical waters are prepared from the leaves, stems, flowers, bark, roots, or other parts of a plant or the whole plant. 1 The condensate from steam distillation produces two distinct fractions that contain the volatile ingredients from the plant. The water insoluble fraction contains the “oil.” The water-soluble fraction contains ingredients from the plant that are water soluble, and is identified by the term “Water” in the INCI name.
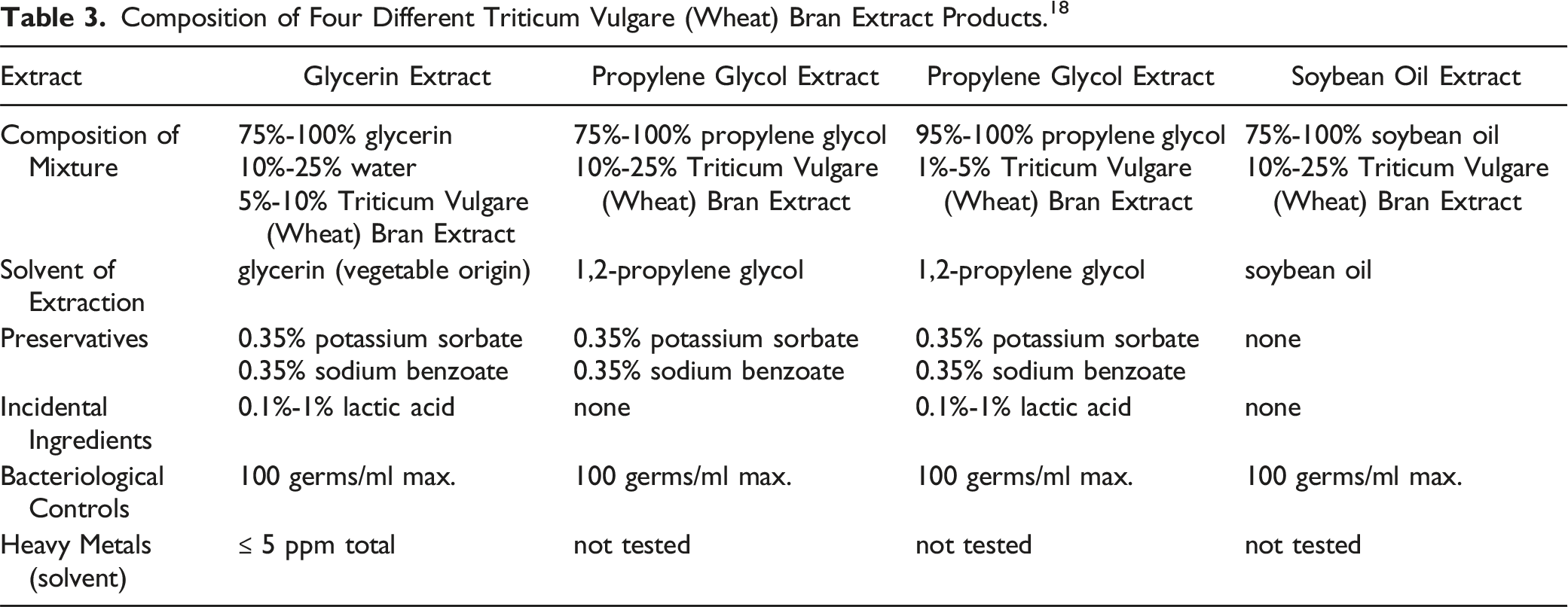
Triticum Vulgare (Wheat) Bran Extract
Composition of Four Different Triticum Vulgare (Wheat) Bran Extract Products. 18
Triticum Vulgare (Wheat) Germ
Wheat germ is a by-product of flour milling and is produced by mechanical separation of the germ from whole wheat. 16
Triticum Vulgare (Wheat) Germ Extract
A supplier reported that Triticum Vulgare (Wheat) Germ Extract is produced by solubilizing wheat germ in water. 19 The insoluble material is then removed and the extract is filtered.
Triticum Vulgare (Wheat) Gluten
Triticum Vulgare (Wheat) Gluten is prepared by water washing wheat flour then drying the insoluble matter with careful temperature control. 4 The cream-tan powder derived from this process is food grade.
Triticum Vulgare (Wheat) Kernel Flour
Wheat flour is produced by different forms of milling or grinding, of the grain’s endosperm.13,20 The different forms of compression and shear allow for producing different grain sizes for different end uses or flour types.
Wheat Germ Glycerides
Wheat Germ Glycerides are produced through the transesterification of wheat germ oil (from conventional milling processes) with glycerin. 4
Composition/Impurities
Yields of constituents in wheat have been found to be dependent on extraction methods and growing conditions such as soil composition, climate, duration of growth period, and height at harvest. 17 Additionally, different plant parts have different constituent compositions.
Triticum Aestivum (Wheat) Leaf Extract
At least 41 flavonoid derivatives have been identified in wheat leaf extract (extracted with 80% methanol) using liquid chromatography/mass spectrometry (LC/MS). 21 The identity of flavones, a subgroup of flavonoids, comprised therein were luteolin, tricetin, apigenin, chrysoeriol, and tricin.
Triticum Vulgare/Aestivum (Wheat) Grain Extract
Total phenolic content of wheat grain extract (as 80% methanol extracts) was 5.1–6.8 µg (±)-catechin/mg for endosperm and embryo and 16.0–16.7 µg (±)-catechin/mg for “pericarb” (pericarp) and testa. 22 Triticum aestivum grain may be contaminated with mycotoxins such as trichothecenes and zearaleonones from toxin-producing fungi and molds. 14
Triticum Vulgare (Wheat) Bran Extract
Descriptions of the compositions of four different Triticum Vulgare (Wheat) Bran Extract products are summarized in Table 3. 18
Triticum Vulgare (Wheat) Flour Extract
The yield of non-starch lipids, neutral lipids, glycolipids, and phospholipids of wheat flour extract was dependent on the solvent system used. 23 In a study that utilized chloroform-methanol, chloroform-methanol-hydrochloric acid, chloroform-methanol-sodium chloride, methanol, chloroform, water-saturated n-butanol, methanol-chloroform-hexane, hexane-isopropanol, and hexane, total lipids ranged from 6.30–9.76 g fatty acid/kg flour, neutral lipids ranged from 3.76–5.43 g fatty acid/kg flour, glycolipids ranged from 1.09–2.98 g fatty acid/kg flour, and phospholipids ranged from 0.78–1.85 g fatty acid/kg flour. The five major fatty acids present in the lipid classes extracted from wheat flour were palmitic acid, stearic acid, oleic acid, linoleic acid, and linolenic acid.
Triticum Vulgare (Wheat) Germ
Wheat germ contains about 10%–15% lipids, 26%–35% protein, 17% sugar, 1.5%–4.5% fiber, and about 4% minerals. 16 Bioactive constituents include tocopherols (300–740 mg/kg dry matter), phytosterols (24–50 mg/kg), policosanols (10 mg/kg), carotenoids (4–38 mg/kg), thiamin (15–23 mg/kg), and riboflavin (6–10 mg/kg).
Triticum Vulgare (Wheat) Germ Extract
Analyses of an aqueous mixture containing 3.5%–12% Triticum Vulgare (Wheat) Germ Extract determined that the heavy metals content was below the acceptability threshold of 0.5 ppm, with cobalt, mercury, and lead not quantifiable. 24 No pesticides were detected in this test material.
Triticum Vulgare (Wheat) Gluten
A supplier reported that a tradename mixture containing 4.7%–6.4% Triticum Vulgare (Wheat) Gluten also contained 7.0%–8.0% sodium laureth sulfate, <1.0% phenoxyethanol, <1.0% ethylparaben, <1.0% methylparaben, and water. 25 Microbial counts were <100 cfu/g and mold and yeast content was <10 cfu/g.
Triticum Vulgare (Wheat) Kernel Flour
There are four classes of protein in wheat flour: globulins, albumins, gliadins, and glutenins. 20 Gliadins and glutenins are components of gluten. Refined wheat flours may contain approximately 13 g/100 g dry weight protein, 0.8–0.9 g/100 g dry weight lipids, 85 g/100 g dry weight carbohydrates, and 9.5 g/100 g dry weight gluten. 13 Whole wheat flour contains approximately 15 g/100 g dry weight protein, 1.5 g/100 g dry weight lipids, 82 g/100 g dry weight carbohydrates, and 7 g/100 g dry weight gluten. Wheat flour may be contaminated with foodborne bacteria and fungi, including Salmonella spp., Escherichia coli, and Bacillus cereus. Wheat flour may also be contaminated with mycotoxins such as aflatoxins and ochratoxin A.
Triticum Vulgare (Wheat) Protein
Wheat proteins are classified into gluten and non-gluten proteins. 26 The gluten proteins are comprised of gliadins and high and low molecular weight glutenins. The non-gluten proteins are comprised of albumins and globulins.
Triticum Vulgare (Wheat) Sprout Extract
A hydroalcoholic extract of wheat sprouts was determined to be composed of hydrophilic compounds, such as peptides, and hydrophobic compounds, such as polyphenols. 27 Further extraction with 90% acetone resulted in the precipitation of 70% glutathione. Phospholipid classes identified from hydroalcoholic extract of wheat sprouts included phosphatidylethanolamine, phosphatidylcholine, phosphatidic acid, phosphatidylinositol, and lysophosphatidylcholine. 28 These phospholipids were characterized by a high content of essential fatty acids (α-linoleic acid and α-linolenic acid).
Wheatgrass is reported to contain chlorophyll, flavonoids, several varieties of vitamins including vitamins C and E, choline, minerals, enzymes, indoles, and a number of amino acids. 17
Wheat Germ Glycerides
Wheat Germ Glycerides contain 30% to 40% monoglycerides with di-and triglycerides of mixed fatty acids. 4
Use
Cosmetic
The safety of the cosmetic ingredients addressed in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetic industry in response to a survey, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
NR =not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIt is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
cNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
dIt is possible these products may be powders, but it is not specified whether the reported uses are powders.
g0.11% in a spray deodorant.
eListed with the nomenclature Triticum Aestivum in the VCRP database.
fVCRP data was listed generically as Wheat Germ Extract.
NR = not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bListed with the nomenclature Triticum Aestivum in the VCRP database.
cIt is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
dNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
eIt is possible these products may be powders, but it is not specified whether the reported uses are powders.
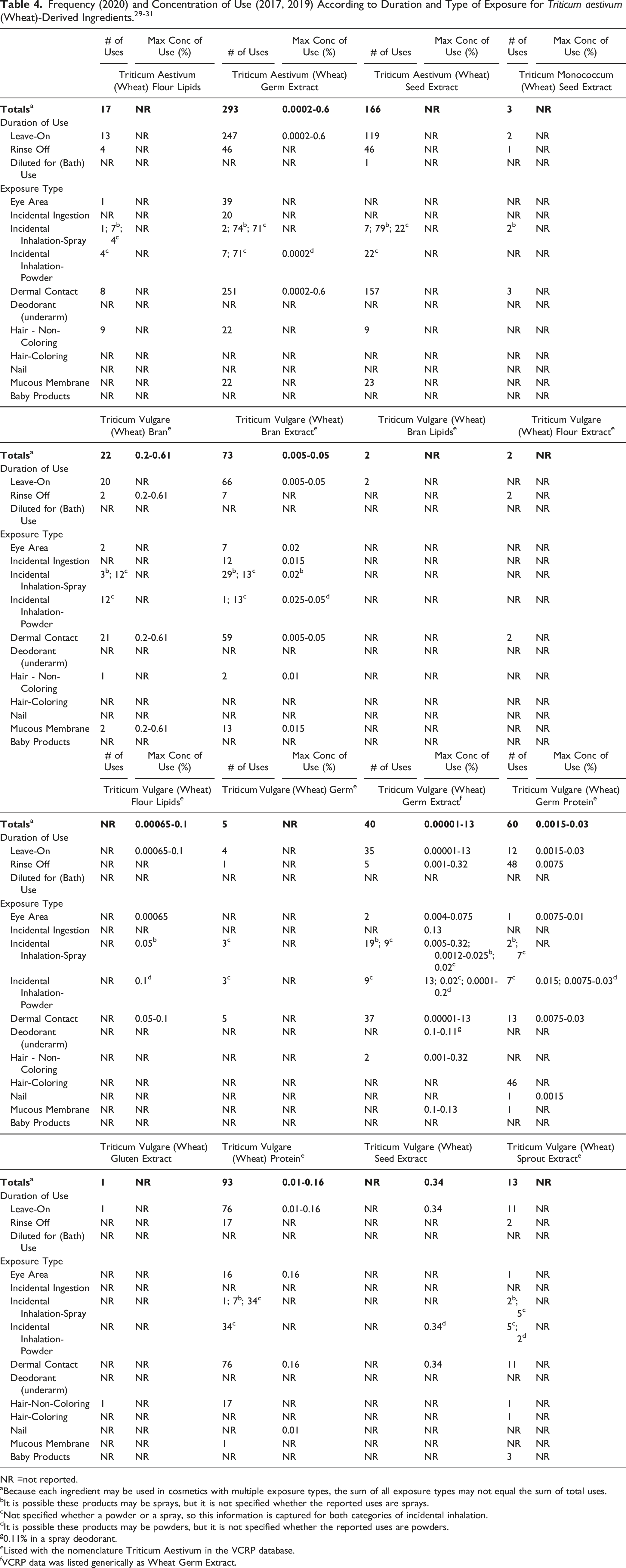
Wheat-derived ingredients may be used in products that can be incidentally ingested or come into contact with the eyes or mucous membranes; for example, Triticum Vulgare (Wheat) Germ Extract is reported to be used in lipstick at up to 0.13%, Triticum Vulgare (Wheat) Protein is reported to be used in eye lotion at up to 0.16%, and Triticum Vulgare (Wheat) Bran is reported to be used in “other” personal cleanliness products at up to 0.61%. 30
Additionally, some of the ingredients are used in cosmetic sprays and could possibly be inhaled; for example, Triticum Vulgare (Wheat) Germ Extract is reported to be used at up to 0.32% in hair spray and up to 0.11% in spray deodorant. 30 In practice, most droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared with pump sprays.32,33 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.34,35 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 34 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays. Triticum Vulgare (Wheat) Germ Extract is reportedly used in face powders at concentrations up to 13% and could possibly be inhaled. 30 Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.36-38
The wheat-derived ingredients described in this report are not restricted from use in any way under the rules governing cosmetic products in the European Union. 39
Non-Cosmetic
The FDA requires allergen labeling when major allergens, such as wheat, are included in food. 40 A major food allergen is an ingredient from a food or food group, such as wheat, that contains protein derived from the food.
Triticum vulgare is reported to be used extensively in traditional medicine to treat decubitus ulcers, burns, scarring delays, dystrophic diseases, and to accelerate tissue repair. 41 It has been investigated for anti-inflammatory properties.
Bran (source not specified) is an over-the-counter (OTC) laxative drug product. 42 Wheat germ is an active ingredient in weight control drug products; however, based on evidence currently available, there are inadequate data to establish general recognition of the safety and effectiveness of this ingredient for this specified use (21CFR§310.545). Wheat gluten is generally recognized as safe (GRAS) in the US for food (21CFR§184.1322).
Wheat leaf extract has been studied for therapeutic benefits for chronic fatigue syndrome, and for its immunological, anti-oxidative, and anti-cancer activities.17,43 Wheat sprout extract has been studied for its antioxidant content and potential use as a food ingredient and in cancer treatments.27,28,44 Wheat germ is used as a food supplement and an ingredient in several food products, 16 and wheat germ extract has also been studied for use in cancer prevention and treatment. 45
Toxicokinetic Studies
No relevant toxicokinetics studies on wheat-derived ingredients were found in the public literature, and unpublished data were not submitted. In general, toxicokinetics data are not expected to be found on botanical ingredients because each botanical ingredient is a complex mixture of constituents.
Toxicological Studies
Most of the wheat-derived ingredients that are reviewed in this safety assessment are found in the foods that are consumed daily, and daily exposure from food use would result in much larger systemic exposures than those from use in cosmetic products. The potential for systemic exposure from the absorption of these ingredients through the skin is much less than the potential for systemic exposure from absorption through oral exposures. This is because the rates of absorption and metabolism of these ingredients in the skin are expected to be negligible compared to the corresponding rates in the digestive tract. Thus, the potential for systemic effects, other than sensitization, is not discussed in detail in this report.
Developmental and Reproductive Toxicity (DART) Studies
Oral
Triticum Vulgare (Wheat) Sprout Extract
The effects of the water extract of wheat sprouts on spermatozoa were investigated using male BALB/c mice. 46 The mice were treated with benzo[a]pyrene (B[a]P) in corn oil (100 mg/kg bw) for 5 consecutive days via intraperitoneal injections. A control group of 19 mice only received corn oil (0.5 ml) and two groups of three mice only received wheat sprout extract (40 and 70 ml). Nineteen animals received the wheat sprout extract in parallel with the B[a]P treatment (3 mice received 40 ml extract, 10 mice received 70 ml extract, and 6 mice received 140 ml extract, in 18 equal doses by gavage) starting 3 days before the B[a]P treatment. Twenty-eight mice received just B[a]P. The sperm of the treated mice were examined 5 weeks after the B[a]P treatment. The mice treated only with B[a]P had an incidence of 61.1% for abnormally-shaped sperm heads. The corn oil control group only had sperm abnormalities observed in 1.93%. The wheat sprout extract alone did not enhance the level of sperm abnormalities in comparison with the corn oil controls. The simultaneous treatment of B[a]P and wheat sprout extract resulted in a decrease in the percentage of abnormally-shaped sperm heads when compared to the group that received just B[a]P, but not in a dose-dependent manner.
Genotoxicity Studies
In Vitro
Triticum Vulgare (Wheat) Bran Extract
The genotoxicity potential of a wheat bran extract that was highly-enriched in arabinoxylan-oligosaccharides was assessed in an Ames test using S. typhimurium strains TA98, TA100, TA1535, and TA1537 and E. coli strain WP2uvrA, with and without metabolic activation. 47 The extract was dissolved at 100 mg/ml in distilled water and was tested at 5, 15.8, 50, 158, 500, 1581, and 5000 µg/plate. Strain-specific positive and negative control chemicals were used and yielded expected results. Wheat bran extract was not mutagenic at up to 5000 µg/plate.
The same researchers assessed the clastogenic potential of the same wheat bran extract in a chromosome aberration assay using Chinese hamster lung fibroblast V79 cells. 47 The cells were treated for 3 h or 20 h with the extract at up to 5000 µg/ml, with and without metabolic activation. No biologically significant increases in the number of cells showing structural chromosome aberrations were observed. The wheat bran extract was not clastogenic in this assay.
Wheat Germ Glycerides
The genotoxicity potential of Wheat Germ Glycerides in ethanol was assessed in an Ames test using S. typhimurium strains TA98, TA100, TA102, TA1535, and TA1537, with and without metabolic activation. 48 Strain-specific positive and negative control chemicals were used and yielded expected results. Wheat Germ Glycerides was not mutagenic when tested at up to 5000 µg/plate.
Anti-Mutagenicity Studies
In Vitro
Triticum Vulgare (Wheat) Sprout Extract
The ability of the S-30 fraction (decanted, incubated supernatant fluid) of the water extract of wheat sprouts to inhibit mutagenicity was assessed in an Ames test using S. typhimurium strain TA 98, with metabolic activation and in the presence of 7,12-dimethyl benz[a]anthracene (DMBA; 50 µg/plate). 46 The S-30 fraction was prepared from thawed plants by squeezing out the juice and centrifuging it for 30 min at 30,000 g before removing proteins with heat, concentrating by vacuum, and sterilizing. A decrease in the number of revertants was observed. No further details were provided.
In Vivo
Triticum Vulgare (Wheat) Sprout Extract
The same research group from the above study investigated the effects of the S-30 fraction of the wheat sprout extract on the ability of BALB/c mouse skin enzymes to activate DMBA to mutagenic metabolites. 46 The S-9 fraction (added to in vitro systems to simulate metabolic capability) from the mouse skin that received the wheat extract subcutaneously for 4 days (0.1 ml/day) still activated DMBA (single painting; 100 µg/mouse) to mutagenic metabolites for the S. typhimurium strain TA 98, but the ability was 20% lower than that of the S-9 fraction from mice that did not receive the wheat sprout extract. No further details were provided.
Carcinogenicity Studies
Co-Carcinogenicity
Triticum Vulgare (Wheat) Bran
In a 31-week dietary study, a group of 12 male Sprague-Dawley rats received 20% wheat bran as a dietary supplement during and after administration of 1,2-dimethylhydrazine (a carcinogen) via 13 weekly subcutaneous injections. 49 Another group of 12 rats was fed the wheat bran in conjunction with administration of the carcinogen, and then switched to the control diet after 13 weeks. A third group of 11 rats received a fiber-free (control) diet during the administration of the carcinogen, and then switched to the wheat bran diet after 13 weeks. The control group (12 rats) received a fiber-free diet throughout the 31 weeks. In both groups that received wheat bran in conjunction with administration of the carcinogen, small intestinal or colonic tumor yield was significantly greater, with benign and malignant tumors incidences increased by 3.4-fold (P < .005), adenoma incidences increased by 3.5-fold (P < .025), and adenocarcinomas incidences increased by 3.25-fold (P < .05) when compared to rats that received a control diet. Rats that received wheat bran only after carcinogen exposure had a reduced number of benign adenomas (71.4%; P < .025) when compared to the rats that received control diet. The rats fed the wheat bran during carcinogen administration and then switched to the control diet had the highest tumor yield, with 4.5 times as many benign and malignant tumors as the control rats (P < .05), and at least six times as many adenomas as any of the other dietary groups (P < .05).
Tumor/Anti-Tumor Promotion
Triticum Aestivum (Wheat) Leaf Extract
The effects of wheatgrass leaf extract on skin papillomagenesis induced by DMBA and croton oil were investigated in male Swiss albino mice. 50 The mice were divided into five groups of 10: Group 1 was the control group that received a single dose of DMBA (100 µl/50 µl acetone) on shaved dorsal skin followed 2 weeks later by croton oil (1% in 100 µl acetone) three times a week for a total of 16 weeks; Group 2 (pre-group) received wheatgrass leaf extract orally (20 ml/kg bw) for 7 days prior to the application of DMBA and croton oil in the manner that Group 1 received these; Group 3 (peri-group) was treated with DMBA and then received wheatgrass leaf extract for 15 days prior to receiving the croton oil, as in Group 1; Group 4 (post-group) was treated with DMBA as in Group 1 and received wheatgrass leaf extract at the same time the croton oil treatment started; and Group 5 received the wheatgrass leaf extract 7 days prior to and after the treatment with DMBA and through the treatment of croton oil until experiment end (total number of exposure days not stated). Tumor incidence, yield, and burden in Groups 2–5 were significantly decreased as compared to Group 1. The average latent period was increased in Groups 2–5, with the longest latent period being observed in Group 5.
Triticum Vulgare (Wheat) Sprout Extract
The tumorigenic effects of wheat sprout extract (S-30 fraction) were investigated using 8-week-old male BALB/c mice. 46 Skin papillomas were initiated by painting the skin of mice with an acetone solution of DMBA (100 µg/mouse). Twice weekly treatments of croton oil dissolved in acetone (10 µl) were used for papilloma growth promotion. The mice received the promoter for 22 weeks. One group of mice (n = 38) received 10 successive subcutaneous injections of wheat sprout extract (0.1 ml) starting on day 3 before DMBA treatment) with the croton oil promoter, while another group (n = 8) received the extract during the promotion period without the croton oil promoter. Further groupings involved giving mice the wheat sprout extract (0.1 ml) subcutaneously twice a week for 22 weeks without the croton oil promoter during both the initiator and promoter phases (n = 13). Wheat sprout extract, when injected subcutaneously for 10 d during carcinogenesis initiation in mice, shortened the latency period from 9 to 4 weeks and increased the number of skin papillomas by 4-fold. When the extract was applied to mice treated with DMBA, but did not have croton oil promotion, only one mouse developed papillomas during the 24 weeks. Controls where the extract was injected alone without initiation or promotion did not produce skin papillomas (n = 17). The authors concluded that wheat sprout extract did not have initiating or promoting properties.
Dermal Irritation and Sensitization Studies
Irritation
Animal
Triticum Vulgare (Wheat) Germ Extract
In a cutaneous tolerance study of an aqueous solution containing 12% Triticum Vulgare (Wheat) Germ Extract, three rabbits received the test material as a single dose (0.5 ml) on the skin in accordance with Organization for Economic Co-operation and Development (OECD) test guideline (TG) 404. 24 A slight to clear erythema that was reversible within 72 h was observed in all animals. It was concluded that the test material was not an irritant to the skin. No additional details were provided.
Wheat Germ Glycerides
Mild irritation was observed on abraded rabbit skin following dermal application of different lots of Wheat Germ Glycerides. 4 Minimal skin irritation was observed in rabbits that received dermal applications of Wheat Germ Glycerides in several different cosmetic formulations. Concentrations were not provided for any of these studies.
Human
Triticum Vulgare (Wheat) Gluten
A mixture containing approximately 5% Triticum Vulgare (Wheat) Gluten and 7%–8% sodium laureth sulfate was tested for irritation potential in a single insult patch test using 20 subjects. 51 The test material (160 µl) was applied as supplied to the upper back once under a semi-occlusive TruMed® patch for 48 h. A control site using water was also prepared. The skin was examined 1 day before patching, on day 3 (15–30 min after patch removal), and on day 4 (24 h after patch removal). On day 3 and day 4, very slight erythema was observed in 4 (20%) of the test subjects. Based on the mean daily irritation score of 0.20, the study authors concluded the test material did not induce irritation and had good skin compatibility. The control site yielded expected results.
Sensitization
Animal
Wheat Germ Glycerides
Wheat Germ Glycerides (0.1% solution in olive oil) was evaluated for sensitization in a guinea pig sensitization study. 4 The treatment group comprised six guinea pigs that received an initial 0.05 ml intracutaneous injection and nine subsequent 0.1 ml injections during the induction phase followed by a 2-week rest period. The animals then received a 0.05 ml challenge injection. The test material was not a skin sensitizer.
Human
Triticum Vulgare (Wheat) Germ Extract
A human repeated insult patch test (HRIPT) of a face and body powder containing 13% Triticum Vulgare (Wheat) Germ Extract was conducted in 105 subjects. 52 The test material (200 mg) was applied to the test sites as supplied. The patches were partially occlusive and 2 cm2 in area. Nine sequential 24-h induction applications and two concurrent 24-h challenge applications were made, with the challenge patches on the initial induction site and a naïve site. Challenge sites were read at 24 and 48 h post-application. No irritation or sensitization was observed.
Triticum Vulgare (Wheat) Gluten and Wheat Germ Glycerides
Mascara-base products containing up to 1% Triticum Vulgare (Wheat) Gluten were not dermal irritants or sensitizers in HRIPTs of up to 202 subjects or in in-use studies (number of subjects not reported). 4 A lipstick base containing up 2% Wheat Germ Glycerides produced reactions in 8 out of 1154 subjects: reactions were considered to be irritation. Wheat Germ Glycerides were not dermal irritants or sensitizers in HRIPTs of lipstick products and blushers at concentrations up to 0.5%. No adverse reactions were reported in in-use studies of a lipstick base containing 2% Wheat Germ Glycerides in 149 subjects.
Ocular Irritation Studies
Animal
Triticum Vulgare (Wheat) Gluten and Wheat Germ Glycerides
Mild circumcorneal injection and mild discharge were noted after testing a mascara base containing 1% Triticum Vulgare (Wheat) Gluten in rabbit eyes: these changes were thought to be the results of foreign objects in the eye and drying of the formulation. 4 Complete recovery occurred within 72 h. Minimal to no ocular irritation was observed in rabbit studies with undiluted Wheat Germ Glycerides and in formulations containing up to 2% Wheat Germ Glyceride.
Triticum Vulgare (Wheat) Germ Extract
In an ocular tolerance study of an aqueous solution containing 12% Triticum Vulgare (Wheat) Germ Extract, three rabbits received the test material as a single dose (0.1 ml) instilled into the eye in accordance with OECD TG 405. 24 Mild irritation was observed that was reversible within 72 h. It was concluded that the test material was not an irritant to the rabbit eye. No additional details were provided.
Occupational Exposures
Work-related sensitization (IgE-mediated) to wheat flour and grain dusts has been reported in bakery workers.53-57 Commonly known as baker’s asthma, reactions are often preceded by rhinitis and other respiratory symptoms, with concomitant skin symptoms such as contact urticaria and hand eczema. Atopy and sensitization to flour and/or enzyme (e.g., α-amylase of fungal origin) occurs frequently.56,57 Aside from wheat, baker’s asthma may also be caused by molds, yeast, eggs, sesame seeds, nuts, and insects. Skin-prick testing, skin biopsies, and radioallergosorbent tests (RAST) have been utilized to identify and analyze the reactions observed in bakery workers.53-57
Epidemiology of Immune-Mediated Gluten and Wheat Reactions
Celiac disease affects approximately 1% of the population worldwide, including the US, with variations between countries.58-60 Wheat allergy affects between 0.5% to 9 % of the population globally, with about 0.4% of adults in the US diagnosed with the allergy.59,61,62 Children have a higher prevalence to food allergy to wheat compared to adults, and in Korean children, evidence of cross-reactivity or co-sensitization with barley has been found.63-65 Respiratory IgE mediated allergies to wheat (i.e., baker’s asthma or rhinitis) are often associated as occupational disease with the incidence in bakers ranging at 5%–10% for asthma and 15%–20% for rhinitis. 63 Bronchial reactivity to inhaled wheat proteins in adults with food allergy is very rare but has been documented. 66
Summary
Most of the 27 wheat-derived ingredients detailed in this safety assessment are reported to function in cosmetics as skin conditioning agents, while some are reported to have other functions, such as abrasives, absorbents, antioxidants, bulking agents, film formers, flavoring agent, hair conditioning agents and viscosity increasing agents. Functions such as skin bleaching agent (for Triticum Vulgare (Wheat) Germ Extract) are not considered cosmetic functions in the US and, therefore, are not addressed in this assessment.
Yields of constituents in wheat have been found to be dependent on extraction methods and growing conditions such as soil composition, climate, duration of growth period, and height at harvest. Additionally, different plant parts have different constituent compositions. Wheat grain may be contaminated by mycotoxins, such as aflatoxins, trichothecenes, and zearalenones. Wheat flour may be contaminated with mycotoxins and foodborne bacteria and fungi, such as Salmonella spp., E. coli, and B. cereus.
According to 2020 VCRP survey data, Triticum Aestivum (Wheat) Germ Extract has the most reported uses in cosmetic products with a total of 293 formulations; the majority of the uses are in leave-on skin care products. Triticum Aestivum (Wheat) Seed Extract has the second greatest reported number of uses in this safety assessment with 166 formulations; the majority of the uses are also in leave-on skin care products. All other in-use ingredients are reported to be used in significantly fewer formulations. The results of the concentration of use survey conducted by the Council indicate that Triticum Vulgare (Wheat) Germ Extract has the highest maximum concentration of use in a leave-on formulation; it is used at up to 13% in face powders. The concentrations of use for the remaining ingredients are much lower; the next highest maximum concentration of use reported for products resulting in leave-on dermal exposure is 0.6% in Triticum Aestivum (Wheat) Germ Extract in “other” skin care preparations.
Wheat is considered a major food allergen and is required by the FDA to be labeled as such when included in food. Triticum vulgare is reported to be used extensively in traditional medicine, and it has been investigated for anti-inflammatory properties. Bran (source not specified) is an OTC laxative drug product. Wheat leaf extract and wheat germ extract have been studied for various therapeutic effects and for use in food supplements.
Most of the wheat-derived ingredients that are reviewed in this safety assessment are found in foods consumed daily the world over. The potential for systemic exposure from the absorption of these ingredients through the skin is much less than the potential for systemic exposure from absorption through oral exposures. This is because the rates of absorption and metabolism of these ingredients in the skin are expected to be negligible compared to the corresponding rates in the digestive tract; and, the systemically available dose of these ingredients, even with theoretically complete absorption from cosmetic use, would be very small compared to that available from consumption.
Wheat sprout extract (40 and 70 ml) alone did not enhance the level of sperm abnormalities in mice in comparison with controls. Simultaneous treatment of B[a]P and wheat sprout extract resulted in a decrease in the percentage of abnormally-shaped sperm heads when compared to the group that received just B[a]P, but not in a dose-dependent manner.
Wheat bran extract was not mutagenic in an Ames test at up to 5000 µg/plate, nor was it clastogenic in Chinese hamster lung fibroblasts in a chromosome aberration assay at up to 5000 µg/ml. In anti-mutagenicity studies, a decrease in the number of revertants was observed following exposure to the S-30 fraction of a water extract of wheat sprouts in an Ames test with metabolic activation and DMBA. A decrease in mutagenic metabolites was observed in BALB/c mouse skin following subcutaneous exposure to the same wheat sprout extract (0.1 ml/day) and DMBA.
Tumor incidences were increased in a 31-week study of rats that received a dietary supplement containing 20% wheat bran during administration of a carcinogen for 13 weeks when compared to rats that received a control diet. Rats that received the wheat bran after carcinogen exposure had a reduced number for benign adenomas when compared to controls. Wheat sprout extract (0.1 ml) was not an initiator or a promoter in mice that received the extract subcutaneously, were induced with DMBA, and/or promoted with croton oil in a 24-week study. Anti-tumorigenic effects have been reported in an oral study with wheatgrass extract (20 ml/kg bw) in mice that were induced with DMBA and promoted with croton oil.
No irritation or sensitization was observed during an HRIPT of a face and body powder containing 13% Triticum Vulgare (Wheat) Germ Extract that was conducted in 105 subjects. No dermal or ocular irritation to 12% Triticum Vulgare (Wheat) Germ Extract was observed in rabbits in a cutaneous tolerance study and an ocular tolerance study, respectively.
Work-related sensitization has been reported in bakery workers. Symptoms include rhinitis, asthma, contact urticaria, and hand eczema. Celiac disease and wheat allergy affect populations worldwide. Children have a higher prevalence to food allergy to wheat compared to adults, and in Korean children, evidence of cross-reactivity or co-sensitization with barley has been found. Respiratory IgE mediated allergies to wheat (i.e., baker’s asthma or rhinitis) are often associated as occupational disease. Bronchial reactivity to inhaled wheat proteins in adults with food allergy is very rare but has been documented.
No relevant toxicokinetic studies were found in the published literature; however, in general, toxicokinetics data are not expected to be found on botanical ingredients because each botanical ingredient is a complex mixture of constituents.
Discussion
The Panel reviewed the botanical ingredients derived from the wheat plants Triticum aestivum, Triticum monococcum, Triticum spelta, Triticum turgidum durum, and Triticum vulgare. The accepted scientific name for both Triticum vulgare and Triticum spelta is Triticum aestivum. The Panel expressed concern about pesticide residues, heavy metals, and other plant species that may be present in botanical ingredients, and stressed that the cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit impurities.
While aflatoxin has been detected in wheat grain and flour, the Panel believes that aflatoxin should not be present in wheat-derived cosmetic ingredients that are derived from Triticum aestivum, Triticum monococcum, Triticum spelta, Triticum turgidum durum, or Triticum vulgare. The Panel has adopted the US Department of Agriculture (USDA) designation of ≤15 ppb as corresponding to “negative” aflatoxin content.
The Panel noted that it had previously concluded that Hydrolyzed Wheat Protein and Hydrolyzed Wheat Gluten were safe for use in cosmetics when formulated to restrict peptides to an average molecular weight of 3500 Da or less. This conclusion was in response to reports of type 1 (IgE-mediated) immediate hypersensitivity reactions that occurred in sensitized individuals following exposure to cosmetic products that contained one of these two ingredients with molecular weights greater than this limit. However, based on the available information, none of the wheat-derived ingredients in this report are hydrolyzed, and most are not even proteins. Coupled with lack of reports to the contrary or experience with such reactions to these ingredients in the clinical setting, concern over such reactions to these ingredients was mitigated. If the protein ingredients in this report are hydrolyzed in processing, then the Panel needs to be made aware of these methods of manufacturing to further assess the safety of these ingredients.
Some wheat-derived ingredients were reported to be used in spray and powder products that could possibly be inhaled. For example, Triticum Vulgare (Wheat) Germ Extract is reported to be used at up to 0.32% in hair spray, at up to 0.11% in spray deodorant, and at up to 13% in face powders. The Panel noted that in aerosol products, most droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns for these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
The Panel noted that detailed information was not provided for method of manufacturing for some of the individual ingredients, but the collective information provided across the group and reasonable inference between related ingredients support assessment of safety. Although the Panel found the remaining data on the wheat ingredients derived from the seed or components of the seed to be sufficient, the Panel determined that the data are insufficient to determination of safety for the following ingredients: Triticum Aestivum (Wheat) Leaf Extract, Triticum Aestivum (Wheat) Peptide, Triticum Monococcum (Wheat) Stem Water, Triticum Vulgare (Wheat) Protein, Triticum Vulgare (Wheat) Sprout Extract, and Triticum Vulgare (Wheat) Straw Water. The additional data needed to determine the safety of these ingredients as used in cosmetics are: • Composition and impurities data. ○ If significantly different from the ingredients considered safe: dermal irritation and sensitization data at maximum use concentration are needed.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that the following 21 wheat-derived ingredients are safe in cosmetics in the present practices of use and concentrations described in this safety assessment:
Triticum Aestivum (Wheat) Flour Lipids
Triticum Aestivum (Wheat) Germ Extract
Triticum Aestivum (Wheat) Seed Extract
Triticum Monococcum (Wheat) Seed Extract
Triticum Spelta Seed Water*
Triticum Turgidum Durum (Wheat) Seed Extract*
Triticum Vulgare/Aestivum (Wheat) Grain Extract*
Triticum Vulgare (Wheat) Bran
Triticum Vulgare (Wheat) Bran Extract
Triticum Vulgare (Wheat) Bran Lipids
Triticum Vulgare (Wheat) Flour Extract
Triticum Vulgare (Wheat) Flour Lipids
Triticum Vulgare (Wheat) Germ
Triticum Vulgare (Wheat) Germ Extract
Triticum Vulgare (Wheat) Germ Powder*
Triticum Vulgare (Wheat) Germ Protein
Triticum Vulgare (Wheat) Gluten
Triticum Vulgare (Wheat) Gluten Extract
Triticum Vulgare (Wheat) Kernel Flour
Triticum Vulgare (Wheat) Seed Extract
Wheat Germ Glycerides
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
However, the Panel also concluded that the available data are insufficient to make a determination that the following six wheat-derived ingredients are safe under the intended conditions of use in cosmetic formulations:
Triticum Aestivum (Wheat) Leaf Extract**
Triticum Aestivum (Wheat) Peptide**
Triticum Monococcum (Wheat) Stem Water**
Triticum Vulgare (Wheat) Protein
Triticum Vulgare (Wheat) Sprout Extract
Triticum Vulgare (Wheat) Straw Water**
**There are currently no uses reported for these ingredients.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 555 13th Street, NW, Suite 300W, Washington, DC 20004, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.