Abstract
Felzartamab is a recombinant fully human immunoglobulin G1 anti-CD38 monoclonal antibody under clinical investigation for immune-mediated diseases. In support of felzartamab clinical development, toxicology studies were conducted in marmoset monkeys, which was the most relevant species based on CD38 binding affinity, pharmacologic activity, and target expression. The felzartamab toxicology program included an enhanced prenatal and postnatal development (ePPND) study to identify potential reproductive and postnatal development risks. In this ePPND study, pregnant marmoset monkeys were randomized to receive vehicle (0 mg/kg) or felzartamab at two dose levels (15 mg/kg and 75 mg/kg) twice per week until parturition, and maternal animals and infants were evaluated for 6 months thereafter. Felzartamab exposure was confirmed in maternal animals and infants in both dosing groups. Overall, felzartamab was well tolerated by pregnant animals at the evaluated doses, with no effect on body weight or body weight gain during pregnancy. No felzartamab-related effects on pregnancy loss or stillbirth rate were observed, and litter counts and numbers of liveborn infants were similar between the vehicle and felzartamab groups. Among infants, there were no felzartamab-related malformations or variations in external anatomy or skeletal morphology and no felzartamab-related observations in histopathology, hematologic and immune cell development, or humoral immune response to vaccination. In conclusion, among pregnant marmoset monkeys dosed with felzartamab, the lack of reproductive toxicity and felzartamab-related effects on offspring supports the clinical evaluation of felzartamab in women of childbearing potential and further demonstrates the suitability of the marmoset monkey for ePPND studies.
Keywords
Introduction
Felzartamab is an investigational, fully human, IgG1 anti-CD38 monoclonal antibody that depletes plasmablasts and plasma cells through antibody-dependent cellular cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis (ADCP).1–3 Felzartamab is currently in clinical development for the treatment of multiple myeloma, primary membranous nephropathy, antibody-mediated rejection, IgA nephropathy, and lupus nephritis, which are diseases mediated by antibody-secreting cells (ASC), such as plasma cells and plasmablasts. ASC express CD38 at high levels but have little to no CD20 expression, making CD38 an attractive target for potential therapeutics for antibody-mediated chronic diseases.4-12
CD38 is a transmembrane glycoprotein in the ADP-ribosyl cyclase family and has multiple functions, including catalyzing the synthesis of ADP ribose, which is essential for intracellular calcium signaling, and is required for immune cell mobilization, adhesion, migration, activation, and regulation of cytokine release.13-19 CD38 is expressed at high levels on human plasmablasts and plasma cells, at low to intermediate levels on human hematopoietic cells that are important for inflammation and immunity (e.g., T cells, B cells, natural killer [NK], monocytes, dendritic cells, and neutrophils), and on platelets, erythrocytes, endothelial cells, prostatic epithelial cells, and osteoclasts.20–27 Furthermore, CD38 is required for the formation of an immune synapse between NK cells and virally infected cells, 28 and therefore, is important for maternal innate immunity.
The expression patterns and function of CD38 during embryogenesis and fetal development remain unclear. During human embryofetal development, CD38 is highly expressed by human cord blood pre-B and B cells and circulating plasmablasts, regulatory B cells, CD4+ T cells, and CD8+ T cells.29–31 Preclinical studies have shown that functional inhibition of the CD38 receptor increased osteoclast formation, reduced bone density, and reduced serum insulin after glucose stimulation.32,33 Mice deficient in CD38 ADP ribosyl cyclase activity had impaired humoral immunity to T-cell−dependent antigens by interfering with dendritic cell trafficking.34,35 Consequently, depletion of CD38 receptor positive cells or inhibition of CD38 receptor function may effect humoral immunity, skeletal development, and glucose metabolism.
In support of the clinical development of felzartamab, repeat-dose toxicity studies of intermediate and chronic duration were performed in marmoset monkeys (Callithrix jacchus), which were identified as the most relevant non-human primate species for toxicologic testing (for details, see Materials and Methods). There were no notable clinical or toxicologically relevant histopathological findings following intravenous administration of felzartamab when dosed up to 75 mg/kg twice weekly for 26 weeks. Decreases in platelet counts were observed and were considered to be a primarily species-specific pharmacologic effect due to the approximately three-fold greater expression of CD38 on marmoset monkey platelets relative to that of human platelets. The decreases in platelet counts were subclinical, with no correlating clinical observations (e.g., hemorrhage) and resolved by the end of the recovery period. Overall, the results of the non-clinical safety studies supported the clinical evaluation of felzartamab in chronic diseases that may require long-term treatment. To date, no reproductive development studies have been conducted for any of the approved anti-CD38 antibodies, and the effects of gestational exposure to anti-CD38 therapy on human embryofetal and early postnatal development are unclear. To fulfill the International Council for Harmonisation M3 (R2), S5 (R3), and S6 (R1) requirements for a reproductive development study in chronic diseases,36,37 we performed an enhanced prenatal and postnatal development (ePPND) study with felzartamab in marmoset monkeys to assess the effects on pregnancy, parturition, embryofetal and postnatal survival and development, as well as hematologic parameters, development of immune cell populations, and the induction of humoral immunity in infant marmoset monkeys. To our knowledge, this is the first published ePPND study in marmoset monkeys.
Materials and Methods
Species Selection
Mice and rats were not used for felzartamab toxicology testing because of the low amino acid sequence homology between human and rodent CD38, low binding affinity of felzartamab for rodent CD38, and downregulated or absent CD38 expression by mature plasma cells. 38 Cynomolgus monkeys (Macaca fascicularis) are typically selected for ePPND studies when pharmacologically relevant. 39 Despite the high sequence homology and binding affinity between cynomolgus monkey and human CD38 protein (data not shown), the marmoset monkey (Callithrix jacchus) was chosen for this ePPND study based on several unpublished observations. First, in a dose range finding study, cynomolgus monkeys developed anemia at felzartamab doses/exposures <1× of the intended clinical exposure. An exaggerated pharmacologic response was the cause of this clinical observation due to a 60-fold greater expression of CD38 on cynomolgus monkey red blood cells compared with human red blood cells. Therefore, determination of toxicities of doses at or above the clinical exposure would not have been feasible in cynomolgus monkey. Second, there is 81% amino acid sequence homology between human and marmoset monkey CD38. Third, felzartamab has a similar binding affinity (1−3.5×) for the human and marmoset CD38 receptor. Fourth, NK cells from marmoset monkeys exhibited a similar (3×) capacity to induce ADCC of target cells compared with those from humans. Lastly, adult marmoset monkeys, compared with humans, have similar levels of CD38 expression on lymphocytes, monocytes, NK cells, plasma cells, blood vessels in the cerebellum, cortex, spinal cord, and prostate glandular epithelium, and most notably, red blood cells.
Animals and Housing
One-year-old, female, purpose-bred marmoset monkeys of European origin weighing approximately 312–462 grams on gestational day (GD) 25 were used in this study. At the time of delivery, animals were provided an ultraviolet lamp for warmth and were acclimated for 2 weeks. Animals received tap water ad libitum, were fed twice daily with a certified laboratory diet (Ssniff Mar), and regularly received supplementary fresh fruit and vegetables. Animals were housed in a climate-controlled room with a minimum of eight air changes per hour, temperature 22–28°C, relative humidity 40%–70%, and artificial lighting controlled at 12 hours of light and 12 hours of dark.
Study Design
This study was conducted by Labcorp Early Development Services (Münster, Germany), an AAALAC International accredited program, and complied with Good Laboratory Practice, European regulations (EU directive 2010/63), and German Animal Welfare Act requirements. All procedures were approved by the local Institutional Animal Care and Use Committee (IACUC). The study met the criteria of the ARRIVE Guidelines. 40
This study design was based on a prior ePPND study conducted with a monoclonal antibody in cynomolgus monkeys.39,41,42 Forty-eight female marmoset monkeys were bred individually to naive, sexually mature male marmoset monkeys. Female marmoset monkeys do not have menstrual bleeding; therefore, ovulation was monitored and controlled by assessing blood progesterone levels and inducing luteal regression.43,44 Each female was paired with one male as soon as progesterone levels increased following the luteal phase of the menstrual cycle. The day after the start of mating was designated as GD 0. Beginning on GD 23, ultrasonic examination was performed to confirm pregnancy, defined as the uterus lumen being detectable as an echoless gap.
Upon pregnancy confirmation, females were randomized using a randomization table to be dosed with either vehicle (10 mM L-histidine, 260 mM sucrose, 0.1% polysorbate 20, pH 6.0; also referred to as “0 mg/kg”) or felzartamab (15 mg/kg or 75 mg/kg) intravenously twice weekly beginning on GD 25. Dosing continued until parturition (GD 145 ± 5; approximately 26 dose administrations). Each dose volume was adjusted based on body weight (maximum volume, 2 mL/kg). Postdose observations were performed approximately 2 hours after dosing. Selected dose levels were based on those of the unpublished 26-week, repeat-dose study of felzartamab in marmoset monkeys in which felzartamab was well tolerated without toxicologically relevant histopathology findings (data not shown). Although reduced platelet counts were detected in all animals at all felzartamab dose levels (3, 15, and 75 mg/kg/dose) in the 26-week study, there were no apparent clinical sequelae (i.e., bleeding).
During the gestation and lactation periods, a female was housed together with the male in an effort to prevent early postnatal infant death, given that all adult marmoset group members commonly participate in infant care. 45 This manner of “family housing” increased the possibility of a repeat pregnancy in lactating females, given the interbirth interval of 6 months for marmoset monkeys.46,47 To prevent second pregnancies, a gestagen-containing hormone stick (etonogestrel) was implanted subcutaneously on the backs of maternal animals.
Maternal Animal Endpoints
Clinical observations (i.e., health monitoring and clinical examination) were performed twice daily, and a detailed inspection (any changes while animal in-hand) was performed once weekly from GD 25 onward. Body weights were recorded once weekly as soon as pregnancies were confirmed on GD 25 until parturition (GD 145 ± 5). After parturition, a detailed inspection of the maternal animals was performed on lactational day (LD) 1 and 7 and once weekly thereafter until necropsy. Maternal body weights were recorded weekly starting on LD 0 until the end of the postnatal observation period (postnatal day [PND] 180). Due to the “family housing” of mating pairs, food consumption was not monitored during the study. The loss of a fetus, as determined by ultrasonography, was considered an abortion until GD 110 and a premature birth thereafter.
Due to the reduction of platelets observed in the 26-week repeat-dose study in marmoset monkeys, changes in hematology parameters (felzartamab-dosed versus control animals) were assessed in all pregnant females on GD 126, in pregnant females with an early abortion or postnatal infant loss, and in all remaining maternal animals on LD 180 ± 1 before necropsy.
In case of an early pregnancy loss, mortality of the maternal animal, or moribund euthanasia, organ weights and macroscopic and microscopic observations of gross lesions, heart, kidneys, lung, liver, spleen, lymph nodes (mandibular, mesenteric, and axillary), sternum with bone marrow, brain, thymus, uterus/cervix, ovaries, and placenta (if present) were performed. For maternal animals euthanized on LD 180 ± 1, only organ weights and macroscopic observations were recorded.
Infant Endpoints
Marmoset monkeys are one of a few anthropoid primates with multiple ovulations per menstrual cycle, 48 resulting in litter sizes of one to four infants. However, because maternal animals can nurse only two newborns, a higher number of infants per litter has been linked to a higher in utero abortion rate and death rate after birth. 47 To account for the expected high loss rate of animals in triplet litters in this study, one infant per litter received supplementary feeding on a daily rotation in an attempt to keep all three infants alive; however, this procedure still led to a marked body weight loss and increase in mortality of triplet infants in the first two to three litters. To prevent these postnatal deaths, one infant per triplet litter was euthanized as soon as one of the following euthanization criteria were met: (1) body weight loss for 4 days; (2) weight loss of several offspring for 4 days, as this was considered as a sign of stress and malaise of the entire litter; (3) physical weakness; or (4) inability to absorb milk.
After birth (PND 0), infants were sexed, weighed, and examined for external abnormalities on a weekly schedule beginning on PND 1, and external organs (head length and width circumference, distance between the eyes, crown–rump length, crown–heel length, tail length, chest circumference, length of upper and lower extremities, and anogenital distance) were measured. All infants had macroscopic and stereomicroscopic visual inspections for external abnormalities. Monitoring of appearance, behavior, and health parameters was performed twice daily beginning on PND 1; detailed inspections while holding the animal in hand were performed once weekly until PND 180. Body weights were recorded once weekly beginning on PND 1. Morphological measurements of extremities, the head, distance between the eyes, crown rump and heel, thorax circumference and anogenital distance were repeated on PND 28, 56, 84, and 168.
For the assessment of clinical pathology parameters, 2.9 mL of blood was withdrawn from the vena cephalica antebrachii or vena saphena for all infants on PND 98 and 171 for hematology, on PND 165 for coagulation, and on PND 180 for clinical chemistry. Given the small size and low body weight of marmoset monkey infants, blood collection volumes and the number of draws were limited. Blood draws with such volumes before PND 98 for clinical pathology parameters were considered not possible.
For infants that were stillborn, dead, or euthanized due to moribund condition from PND 0 to 7, macroscopic examinations of the GI tract, reproductive organs, kidney, urinary bladder, liver, spleen and gallbladder, secondary lymphoid organs, heart, lung, eyes, and brain were performed. Infants that died or were euthanized due to moribund condition from PND 8 to 180 received microscopic examinations in addition to the macroscopic examinations. Organ weights and a full histopathologic examination was performed for triplets euthanized due to reaching one endpoint of the termination criteria and for infants euthanized with ketamine hydrochloride (0.25 mL/kg of 100 mg/mL Ketamine) followed by an intravenous injection of sodium pentobarbitone (150 mg/kg) at the end of the postnatal observation period.
Skeletal Endpoints
Infants that died from PND 0 to 7 or that were euthanized in a moribund condition were assessed for skeletal developmental abnormalities using Alizarin red staining of the ossified femur, knee joints, skull, and carcass following an ethanol fixation after termination. Long-term fixation of skeletal preparations was performed in glycerin. To determine postnatal bone development, an X-ray examination was performed on infants on PND 100. Nonfasted infants were anesthetized using ketamine 0.1 mL/animal of 25 mg/mL S-Ketamine and 0.04 mL/animal of 1 mg/mL Medetomidine and examined to detect abnormalities and assess bone developmental stage.
Toxicokinetic and Immunogenicity Endpoints
Blood samples for the assessment of felzartamab concentrations were collected from maternal animals on GD 25, 28, 53, 81, 109, and 130 and on LD 14, 28, and 42 and from infants on PND 28 and 42. At each time point, plasma (maternal animals, ∼0.06 mL; infants, ∼0.02 mL) was obtained by centrifugation and stored frozen at −20°C before analysis. Samples were analyzed using a validated electrochemiluminescence ligand binding assay with a quantification limit of 100 ng/mL (MorphoSys AG, Planegg, Germany).
To minimize the amount of blood collected from pregnant and newborn marmoset monkeys, a population toxicokinetic (PopTK) approach was applied for toxicokinetic data analysis using Phoenix WinNonlin, version 8.3 (Certara USA Inc., New Jersey, USA). The PopTK model was previously established with data from the 26-week chronic toxicity study in male and non-pregnant female marmoset monkeys. Post hoc estimates of the model parameters were used to predict individual plasma concentration versus time profiles, which were subsequently utilized to determine felzartamab toxicokinetic parameters (i.e., area under the curve [AUC] and maximum observed plasma concentration [Cmax]) in the ePPND study and to assess the impact of pregnancy and the associated transplacental transfer of felzartamab over time.
Blood samples for the assessment of anti-felzartamab antibodies were collected from maternal animals on GD 25, 53, 81, 109, and 130 and LD 14, 28, 42, and 84 and from infants on PND 35 and 56. At each time point, plasma (maternal animals, ∼0.06 mL; infants, ∼0.02 mL) was obtained by centrifugation and stored frozen at −20°C before analysis. Samples were analyzed using a validated ELISA ligand binding assay (MorphoSys AG, Planegg, Germany).
Immunotoxicology Endpoints
The numbers of major peripheral blood immune cells in infants were measured on PND 98 and 171. Blood samples (600 μL) were collected from infants from the vena cephalica antebrachia in EDTA anticoagulant. Samples were gently mixed by hand and using a mixer. Immunophenotyping was performed by a validated flow cytometry method with monoclonal antibodies to detect CD3+ T cells, CD3+CD4+ T helper cells, CD3+CD8+ cytotoxic T cells, CD20+ B cells, CD14+ monocytes, and CD3−CD16+CD56+ natural killer (NK) cells. Total lymphocyte and leukocyte counts were determined by a hemoanalyzer on the same day. Data were analyzed using BD FACSDiva™ software.
To assess development of humoral immunity, all surviving infants were vaccinated with keyhole limpet hemocyanin (KLH) on PND 112 and PND 154 via subcutaneous injection into the lower right back (first and second immunization, 5 mg KLH in 0.5 mL). For the measurement of anti-KLH response with a T-cell–dependent antibody response (TDAR) assay, 0.5 mL blood was withdrawn from the vena cephalica antebrachia at 0, 7, 14, and 21 days after each vaccination using serum separator tubes. Following coagulation of blood for 30 minutes at room temperature, serum was separated by centrifugation at room temperature (1500×g for 10 minutes). Serum samples were diluted in a series (1/100, 1/500, 1/2500, 1/12500, and 1/62500) using 1% Blocker A loaded into the appropriate wells of two KLH-coated Meso Scale Discovery® 96-well plates. Anti-KLH IgM and IgG were detected by the addition of SULFO-TAG labeled goat anti-monkey IgG or IgM. After washing, both plates were loaded with read buffer and analyzed by electrochemiluminescence using the Meso Sector S600 microplate reader imager (Meso Scale Discovery, Rockville, MD, USA) and Watson® LIMS software. For samples above the upper limit of quantification, further dilution (1/62500) and analysis was performed. The amount of anti-KLH IgG and IgM (titration value) was calculated by multiplying the obtained results by the respective dilution factors. Values were reported positive if samples were above the lower limit of quantitation, which was obtained with KLH-naive marmoset monkey serum.
Statistics
Data from animals dosed with felzartamab were compared with those from animals dosed with vehicle using pairwise comparisons. Group data are reported using means and standard deviations. Tests were performed using a two-sided risk. P values were determined using analysis of variance (ANOVA) and the Dunnett test. The Mann–Whitney U test was used to compare control and felzartamab groups for immunophenotyping and TDAR assays. P values ≤0.05 were considered significant. Positive control groups were excluded from analyses. No analysis were performed when: the smallest nominal group size was <3; the mean of the nominal group size for the group/sex combination of interest was <3; the mean of the actual group size for a given parameter was ≤2.5 (i.e., the mean number of non-missing values for groups selected for analysis); or any group/sex combination with only one (or zero) non-missing value was excluded from the analysis. Statistical analyses were performed using SAS version 9.2 and GraphPad Prism software.
Results
Felzartamab Toxicokinetics and Immunogenicity
Forty-eight pregnant female marmoset monkeys, randomized equally into three groups, received twice weekly intravenous injections of either vehicle control (0 mg/kg) or felzartamab (15 mg/kg or 75 mg/kg). Felzartamab plasma concentrations were best described by a three-compartment model with first order elimination and dose as a covariate on typical clearance, clearance from the first peripheral compartment, and volume of distribution of the first peripheral compartment. The typical clearance and the central volume of distribution were estimated with acceptable precision of 0.469 mL/h and 11.5 mL, respectively. The observed data in maternal animals were well predicted by the marmoset monkey PopTK model and were consistent from the start of dosing (GD 25) until the end of dosing (parturition; Figure 1). Concentrations were also consistent with those observed in non-pregnant females in the 26-week chronic toxicity study (data not shown). After the end of felzartamab treatment (parturition), plasma felzartamab concentrations decreased steadily in the 15-mg/kg and 75-mg/kg dose groups. Mean felzartamab concentrations around parturition (GD 130) were 138,000 ng/mL in the 15-mg/kg group and 379,000 ng/mL in the 75-mg/kg group. Mean felzartamab concentrations at LD 14 (study day 159) ranged from 5050 ng/mL to 7710 ng/mL in the 15-mg/kg group and 19,900 ng/mL to 28,200 ng/mL in the 75-mg/kg group. Mean (±90% prediction interval) predicted versus observed plasma felzartamab concentration versus time profiles. Graph shows felzartamab concentrations during the gestation (GD (Study Day) 20–145; dosing period) and the postnatal observation period (PND 0–180 ± 1 (Study Day 146–180 ± 1)).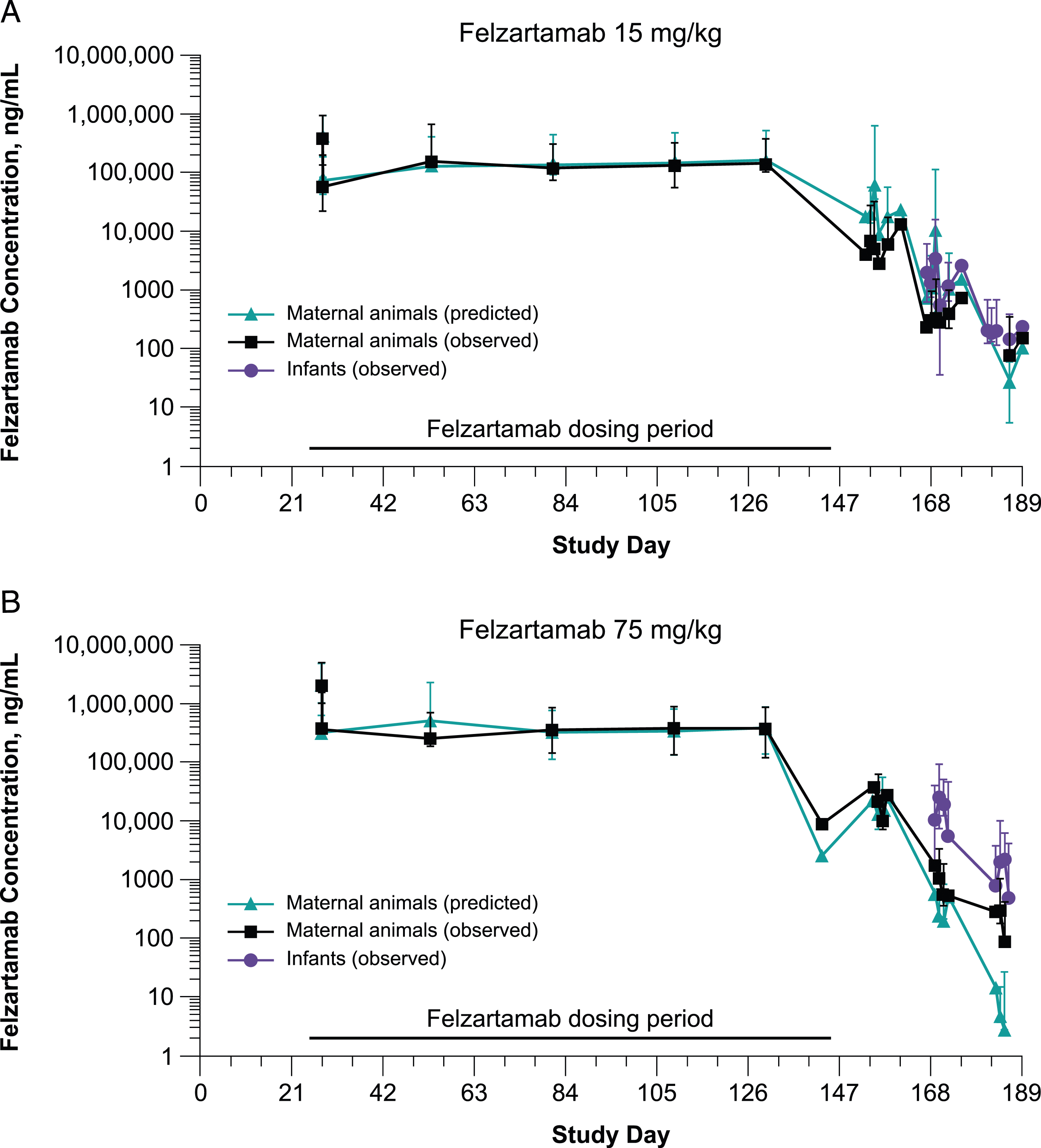
Felzartamab was detected in the plasma of newborn animals, indicating transplacental transfer of felzartamab. In the 15-mg/kg and 75-mg/kg groups, the mean plasma felzartamab concentrations of newborns were greater than those of mothers at PND 28 and 42 (study days 173 and 187, respectively) (Figure 1).
Post hoc estimates of felzartamab concentrations were used to simulate felzartamab concentration versus time profiles after the start of dosing for all maternal animals. The resulting concentration versus time profiles were then used to calculate toxicokinetic parameters. The median values of total exposure over the first week of treatment (i.e., AUC0−168h) were 1035 day*µg/mL for the 15-mg/kg group and 4629 day*µg/mL for the 75-mg/kg group. The median values of total exposure over the treatment period (i.e., AUCGD25−delivery) were 28,050 day*µg/mL for the 15-mg/kg group and 102,200 day*µg/mL for the 75-mg/kg group. The median values of highest felzartamab concentrations (i.e., Cmax) were 760 μg/mL for the 15-mg/kg group and 3463 μg/mL for the 75-mg/kg group. The mean effective elimination half-life was 77.5 hours for the 15-mg/kg group and 58.1 hours for the 75-mg/kg group.
Eight samples from eight animals had detectable anti-felzartamab antibodies, including seven maternal animals (15 mg/kg, n = 4; 75 mg/kg, n = 3) during gestation (15 mg/kg, n = 2; 75 mg/kg, n = 2) or lactation (15 mg/kg, n = 2; 75 mg/kg, n = 1) and one newborn animal (15-mg/kg). The anti-felzartamab titer was 4.1 for one maternal animal in the 15-mg/kg group but could not be determined (i.e., <1.0) in the other six felzartamab-dosed maternal animals and one felzartamab-exposed infant. Anti-felzartamab antibodies had no effect on felzartamab plasma concentrations in any of the eight animals.
Effect of Felzartamab Treatment on Pregnancy and Parturition
Pregnancies were confirmed in all 48 time-mated females within the 5-month mating period. Mean maternal body weight and body weight gain were as expected for all dose groups,
49
with mean body weights at GD 53 of 370 grams (0 mg/kg), 411 grams (15 mg/kg), and 400 grams (75 mg/kg) and mean body weight gain until end of gestation (GD 137) of 20% to 26% (Figure 2, Supplemental Table 1). No changes in other maternal health assessments or clinical observations (i.e., behavior) were observed during gestation in any of the dose groups (data not shown). Change in mean ( ± SEM) maternal body weight during gestation (gestational days 25–145). Data includes maternal animals of all dose groups.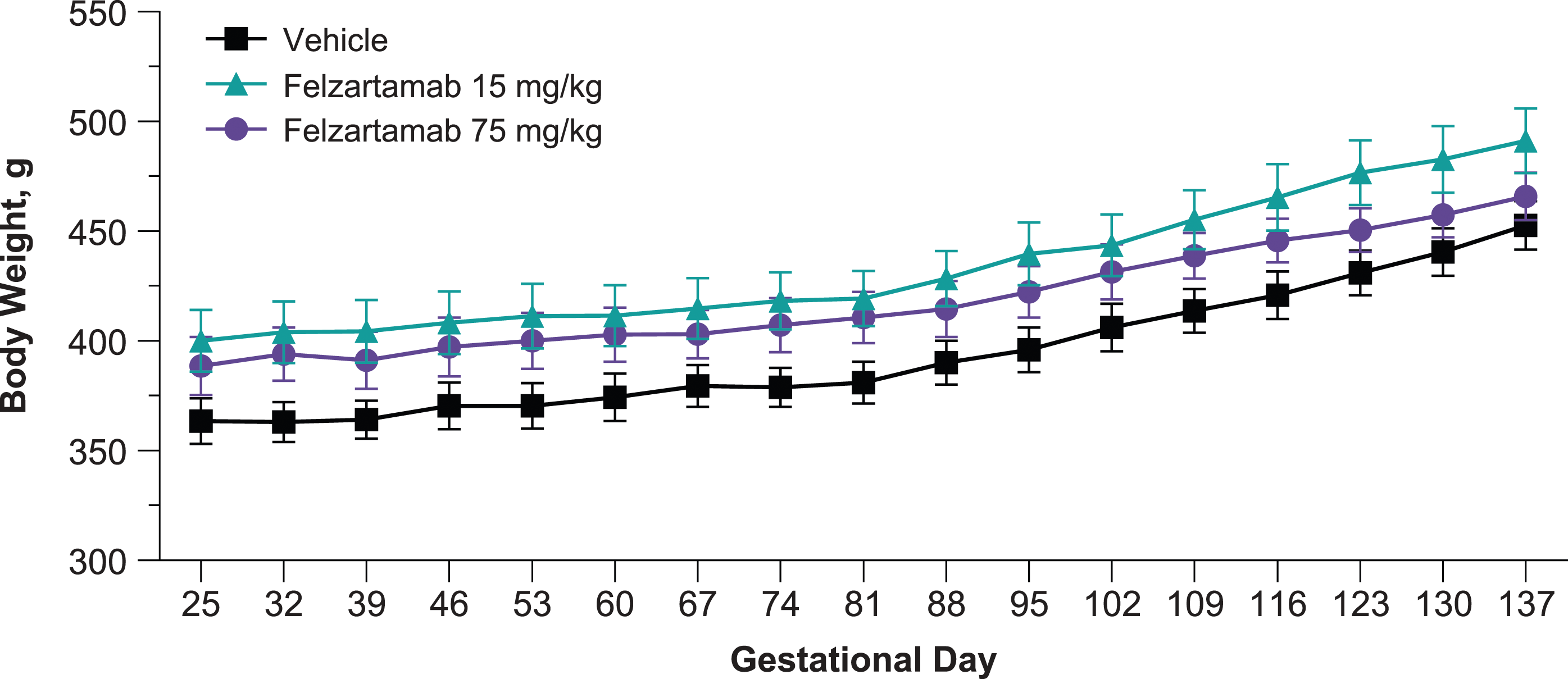
Pregnancy and Parturition.
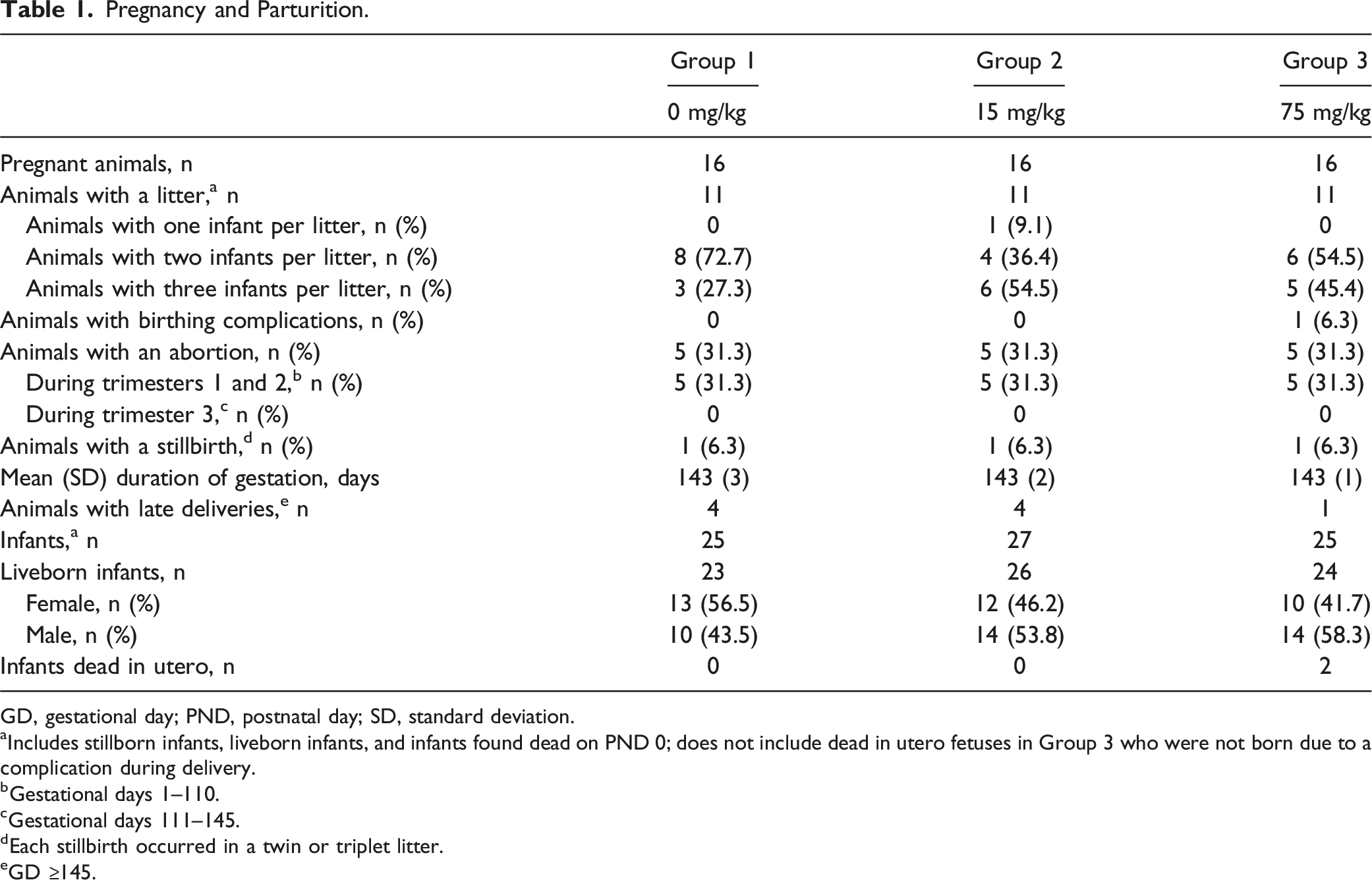
GD, gestational day; PND, postnatal day; SD, standard deviation.
aIncludes stillborn infants, liveborn infants, and infants found dead on PND 0; does not include dead in utero fetuses in Group 3 who were not born due to a complication during delivery.
bGestational days 1–110.
cGestational days 111–145.
dEach stillbirth occurred in a twin or triplet litter.
eGD ≥145.
The total rate of prenatal pregnancy loss (abortions plus stillbirths for each maternal animal) was 6 (37.5%) in each dose group (Table 1). Fetal abortions occurred in five (31.3%) maternal animals in each dose group, all within the first two trimesters (GD 1–110). No abortions occurred during the third trimester of pregnancy (GD 111–145). Abortions were more frequent early during the first two trimesters (GD 25−60; 0 mg/kg, n = 3 [60.0%]; 15 mg/kg, n = 3 [60.0%]; 75 mg/kg, n = 4 [80.0%]) than later (GD 61−110; 0 mg/kg, n = 2 [40.0%]; 15 mg/kg, n = 2 [40.0%]; 75 mg/kg, n = 1 [20.0%]).
Stillbirths occurred in one (6.3%) maternal animal in each dose group (Table 1). Necropsy of the stillborn infants revealed no notable findings for those in the 0-mg/kg and the 15-mg/kg groups. The stillborn infant in the 75-mg/kg group had marked enlargement of all four liver lobes, which was not considered to be felzartamab related as it was observed in a single animal.
One maternal animal in the 75-mg/kg group experienced a 12% body weight loss from GD 102 to 123 and subsequently had a breech delivery on GD 143, with one stillborn infant and two infants that died during birth. The maternal animal was euthanized in a moribund condition. Histopathologic examination revealed no findings that would explain the body weight loss or infant loss aside from those commonly reported in marmoset monkeys (i.e., hemorrhage, fibrin extravasation, and placental mineralization). The incorrect position of the first fetus and/or the premature death of the autolytic fetus may have contributed to the interrupted birth and the death of all three fetuses.
Effect of Felzartamab on Infant Survival
Survival of Liveborn Infants.
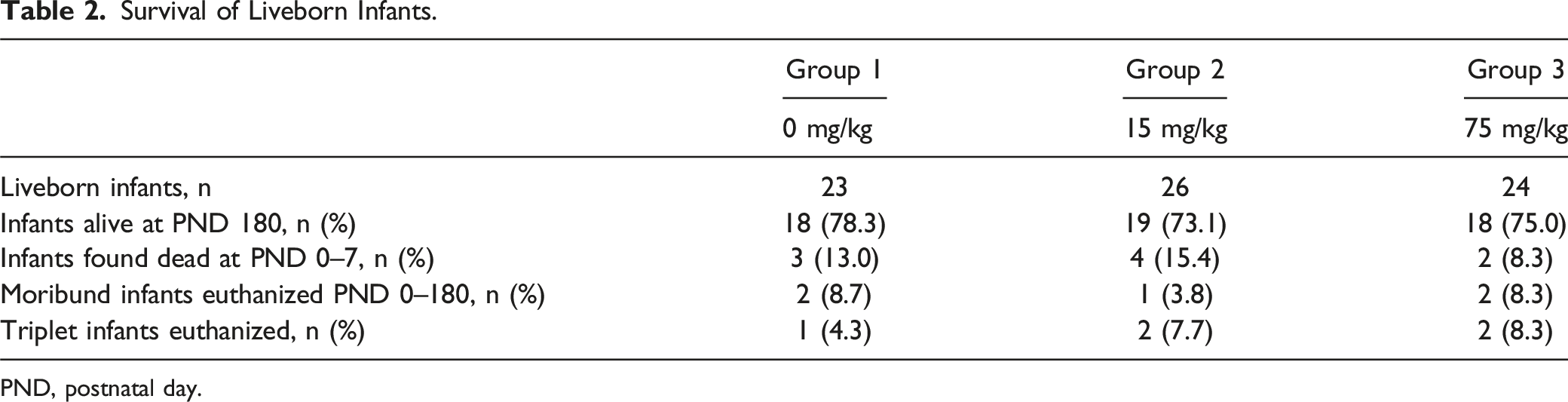
PND, postnatal day.
From PND 0 to 180, five infants (0 mg/kg, n = 2; 15 mg/kg, n = 1; 75 mg/kg, n = 2) were euthanized due to a moribund condition (Table 2). Among the two infants in the 0-mg/kg group, one presented in poor health on PND 14 and had no macroscopic or histopathologic findings at necropsy other than a decrease in cellularity in the spleen, mesenteric lymph node, and thymus, which were presumably related to poor health. For the second infant in the 0-mg/kg group, a fracture of the right femur was determined to be the cause of the deteriorating health observed on PND 119. For the infant in the 15-mg/kg group that was euthanized on PND 3, histopathologic assessment did not identify a cause for the moribund condition. For the infants in the 75-mg/kg group, one was found moribund on PND 4, and necropsy indicated abnormal content in the thoracic cavity with red discoloration of the occipital lobe, a red-discolored focus in the brain, multiple pale-yellow foci in the heart (including pericardium), red discoloration of the whole lung with multiple pale-yellow foci, and multiple pale-yellow foci in the thymus. The yellow foci in lung and thymus were negative for aerobic and anaerobic pathogens, and the cause of moribund condition was not determined. The second infant in the 75-mg/kg group presented moribund on PND 171 with abnormal mobility of the left knee that was determined at necropsy to be caused by a fracture of the left femur.
Given the increased risk of developing deteriorated health conditions for triplet-born infants,46,51,52 one infant born per triplet litter (0 mg/kg, n = 1; 15 mg/kg, n = 2; 75 mg/kg, n = 2) was euthanized before PND 7 based on predefined criteria to prevent the loss of all infants of triplet litters (Table 2; see Materials and Methods). Of the five triplet-born infants euthanized from PND 0 to 3, there were no histopathologic findings, suggesting that the euthanization criteria (i.e., body weight loss or physical weakness) were met early due to the lack of parental care rather than felzartamab-related impairment of physiologic conditions.
The proportion of live-born infants surviving to PND 180 was similar between the dose groups (0 mg/kg, n = 18 [78.3%]; 15 mg/kg, n = 19 [73.1%]; 75 mg/kg, n = 18 [75%]); Table 2). Given the lack of any observations in animals euthanized in a moribund condition and the similar rates of deteriorated health across the dose groups, all early euthanizations were considered incidental or due to the litter size but not felzartamab-related, indicating that felzartamab had no effect on postnatal survival.
Infant Growth and Morphological and Skeletal Development
No biologically significant felzartamab-related effects on infant birth weights were observed on PND 1 or during the postnatal observation period through PND 180 (Figure 3, Supplemental Table 2). Mean infant body weights in the felzartamab groups were consistent with those in the control group and were within the published historical control range.
53
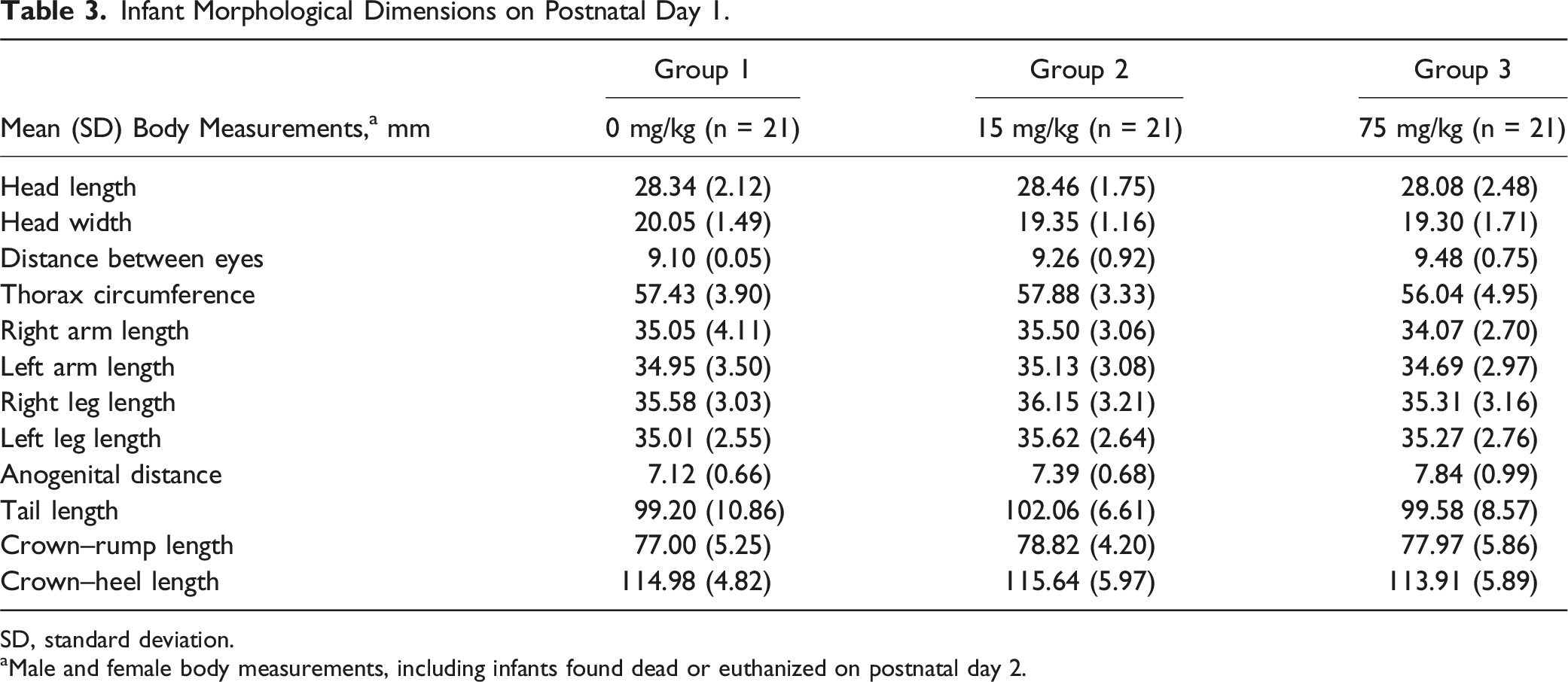
Likewise, morphological examinations on PND 1 did not show any differences in body measurements between infants in the felzartamab groups compared with infants in the vehicle group (Table 3). Head measurements were consistent within the reported historical control range.
54
Change in mean (± SEM) infant body weights (postnatal days 1–181). Data includes male and female animals. Infant Morphological Dimensions on Postnatal Day 1. SD, standard deviation. aMale and female body measurements, including infants found dead or euthanized on postnatal day 2.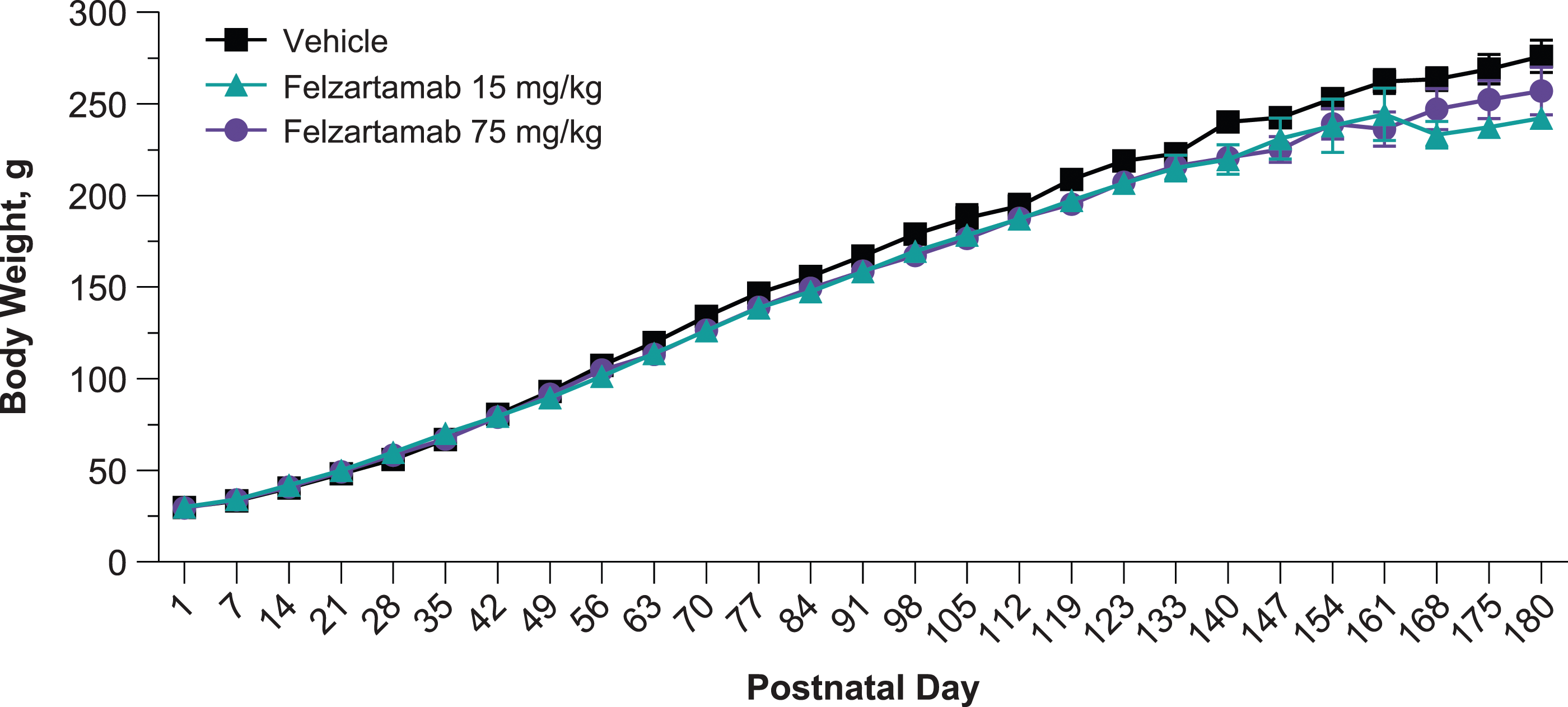
Infant Skeletal Variations and Malformations Among Infants With Early Death (Postnatal Days 0–7).
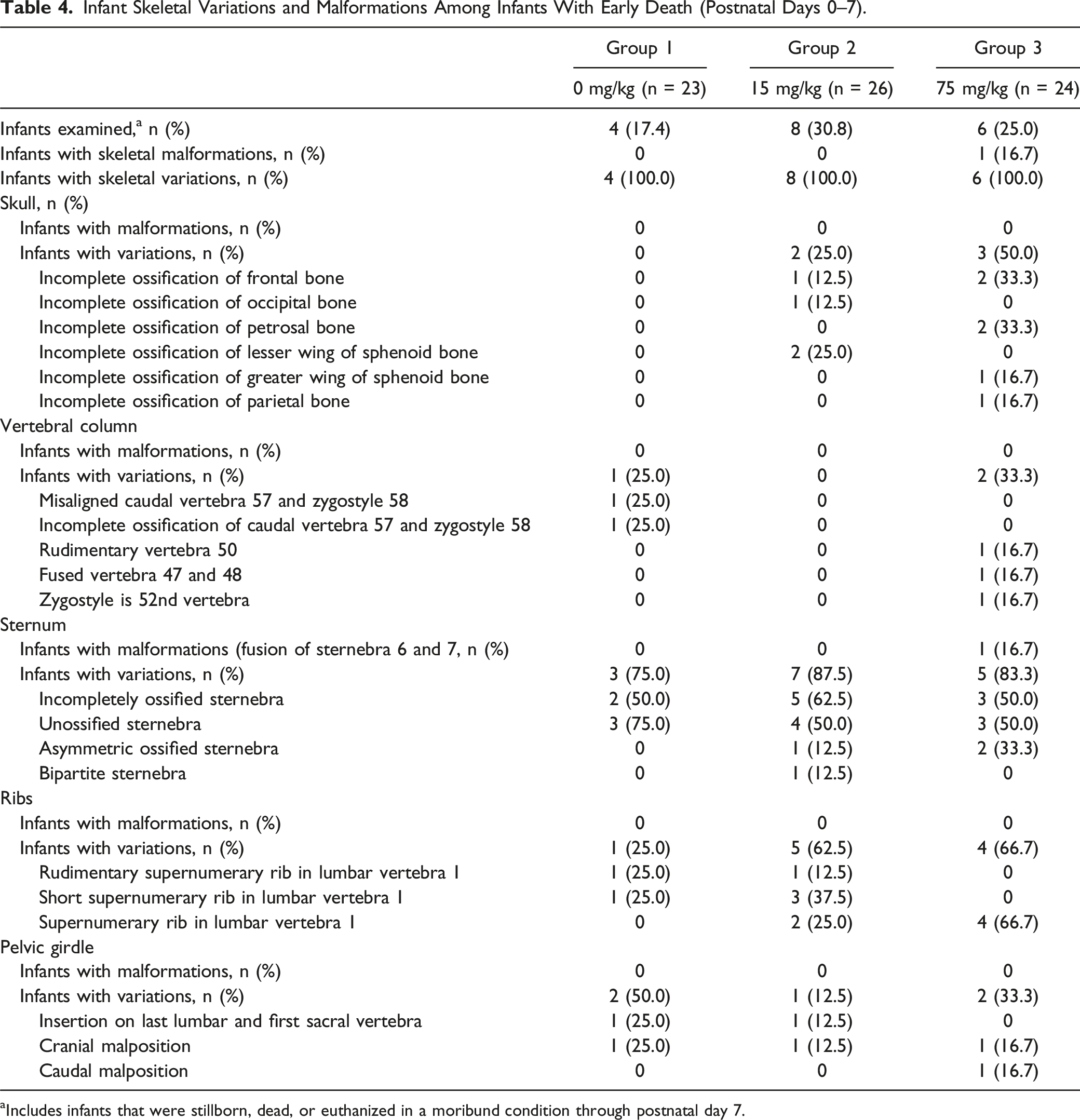
aIncludes infants that were stillborn, dead, or euthanized in a moribund condition through postnatal day 7.
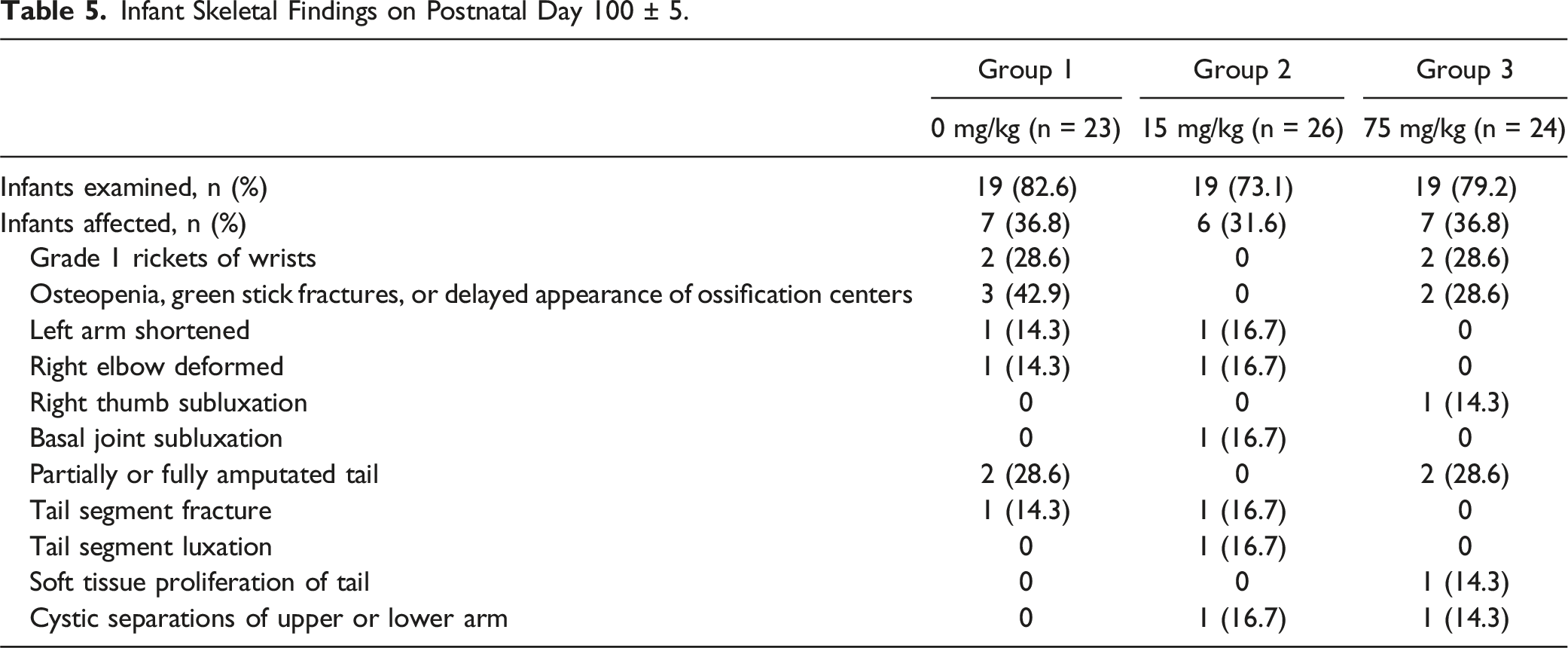
Infant Skeletal Findings on Postnatal Day 100 ± 5.
Infant Organ/Tissue Weights on Postnatal Day 180.
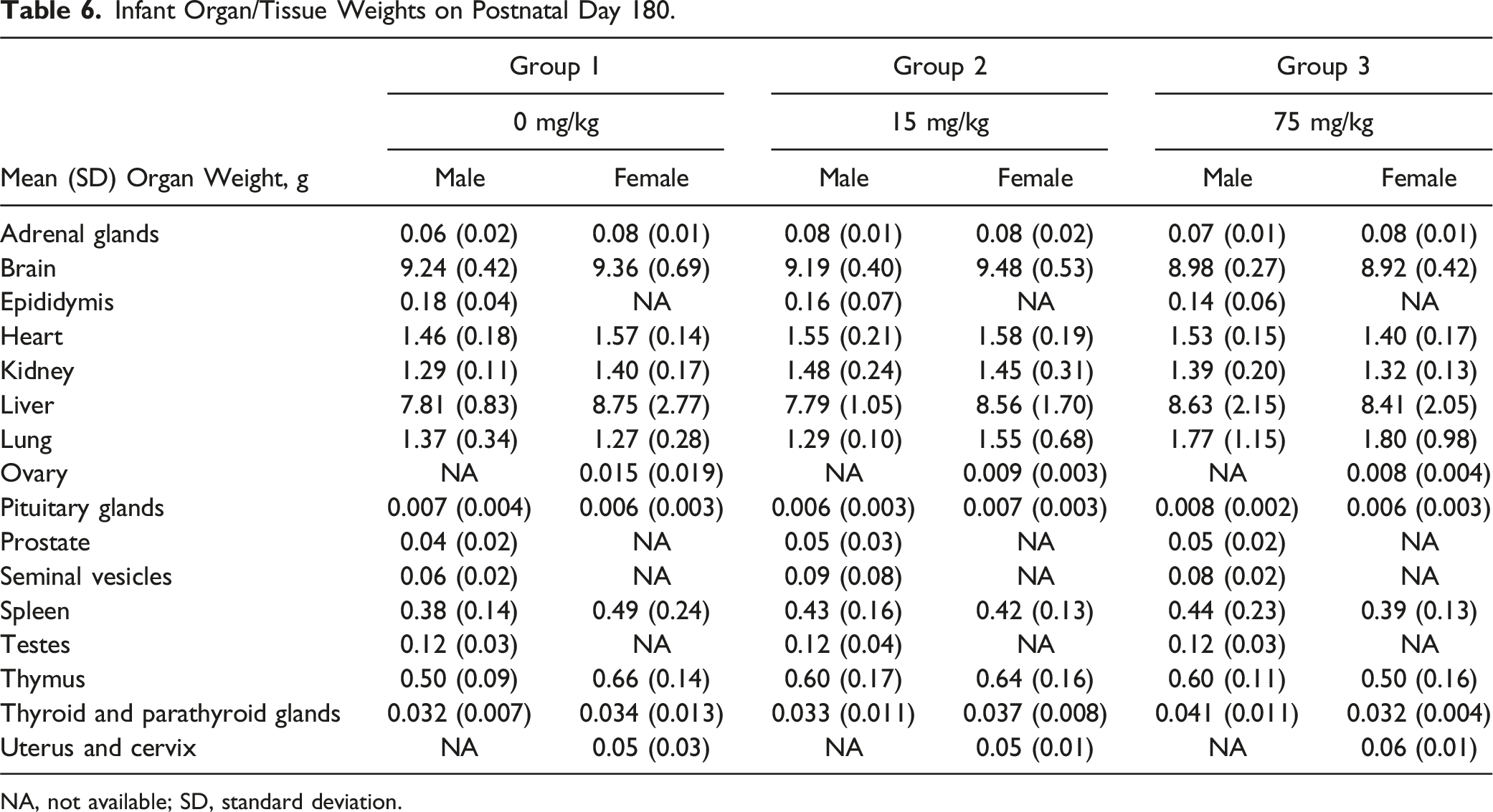
NA, not available; SD, standard deviation.
Effect of Felzartamab on Immune Cell Populations and Humoral Immunity in Infants
The effect of felzartamab on the development of B cells, T cells, NK cells, and monocytes was assessed in peripheral blood collected from infants on PND 98 and PND 171. At both time points, mean numbers of CD16+CD56+ NK cells, CD3+ T cells, and CD14+CD16− monocytes were similar between the vehicle and felzartamab groups and between the felzartamab 15-mg/kg and 75-mg/kg groups, except for mean CD20+ B cells in male infants on PND 98, which were more frequent in the 15-mg/kg and 75-mg/kg group (P<0.05) versus the 0-mg/kg group (Figure 4A, Supplemental Table 3). However, there was no increase in CD20+ B cells on PND 171, and the increase in B cells on PND 98 was not associated with an overall increase in lymphocyte count in blood. Immune cell counts and change over time in T cell–dependent antibody response. (A) Mean number of blood CD3+ T cells, CD20+ B cells, CD16+, CD56+ natural killer cells, and CD14+CD16− monocytes on postnatal day 98 in animals who received vehicle, felzartamab 15 mg/kg, and felzartamab 75 mg/kg. (B) Mean (±SEM) Serum IgG and IgM titer following the first (PND 112) and second (PND 154) immunizations with KLH in animals who received vehicle, felzartamab 15 mg/kg, and felzartamab 75 mg/kg. Data includes male and female animals. *P < .05, **P < .01, ***P < .001, and ns (not significant) using Mann–Whitney test.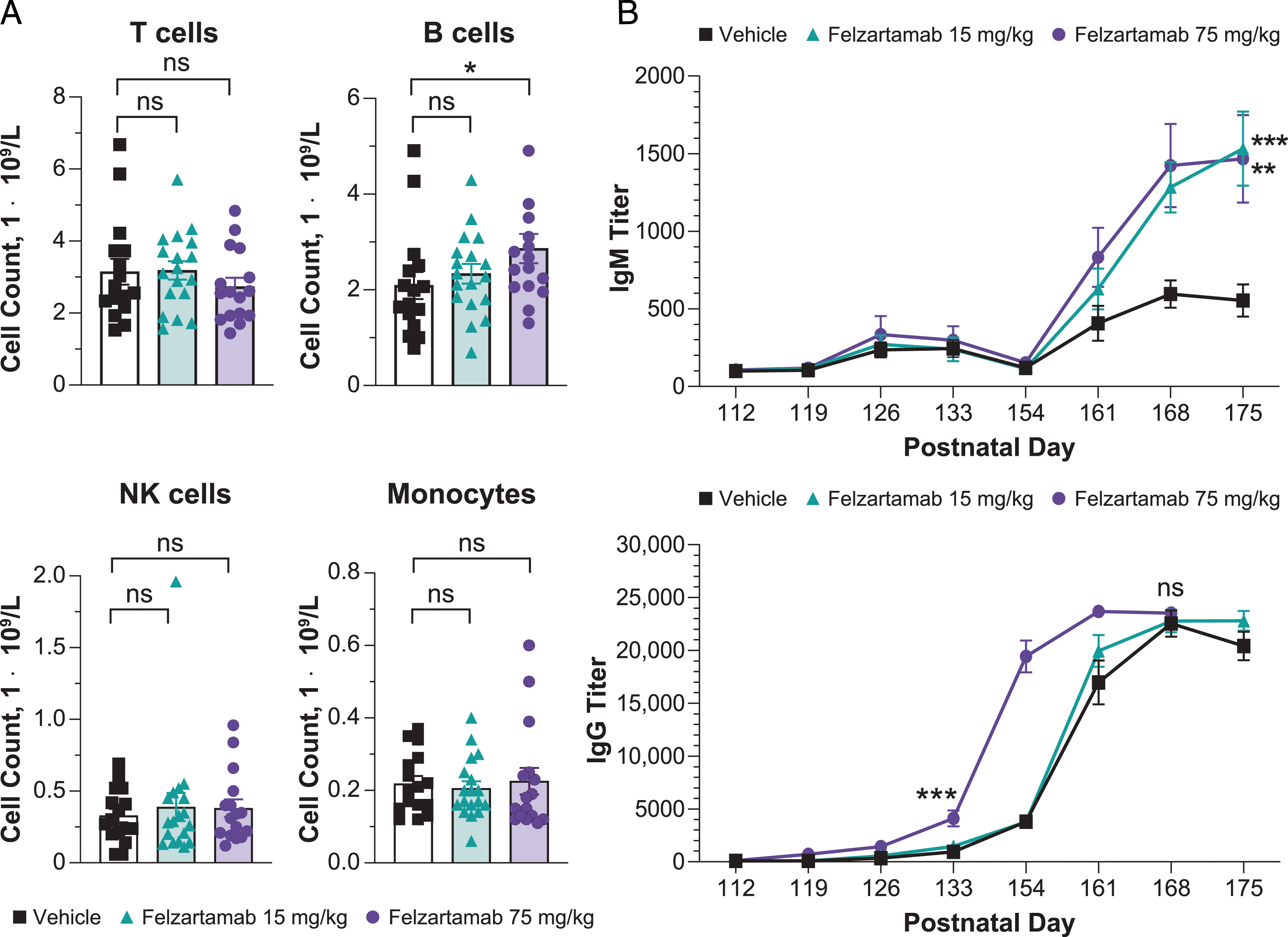
The effect of felzartamab dosing during pregnancy on the development of humoral immunity in the offspring was examined using a TDAR assay. Serum titers of anti-KLH IgG and IgM antibodies from infants in vehicle and felzartamab groups were evaluated following immunization with KLH on PND 112 and again on PND 154. A primary antibody response to KLH was observed within 3 weeks after the first immunization in all infants, with a high variability in IgM and IgG titers between animals of all dose groups (Supplemental Table 4). Although mean anti-KLH IgM titers in infants were similar between all groups after the first immunization (PND 126: 0 mg/kg, 235.4; 15 mg/kg, 272.9; 75 mg/kg, 335.2), mean IgM titers were significantly greater (P<0.01) in the felzartamab groups compared with the vehicle group 1 week after the second KLH immunization (PND 168: 0 mg/kg, 296.2; 15 mg/kg, 1282.6; 75 mg/kg, 1424.3) and reached maximum levels 3 weeks after the second immunization (Figure 4B). Mean anti-KLH IgG titers in infants were significantly greater (P<0.0001) 3 weeks after the first KLH immunization in the 75-mg/kg group (4103.5) compared with the two other groups (0 mg/kg, 942.6; 15 mg/kg, 1458.5) and continued to increase after the second immunization, with no differences in titers between groups detected 3 weeks after the second immunization (Figure 4B). Given that IgM and IgG titers in felzartamab-dosed groups were comparable or higher to control titers, felzartamab exposure during pregnancy had no negative effect on development of humoral immunity development by infants.
Effect of Felzartamab on Hematologic Parameters
Given the earlier observation of a felzartamab related dose-dependent decrease in platelets in a 26-week repeat-dose study in marmoset monkeys (data not shown), platelet counts in maternal animals were assessed on GD 126 (i.e., the end of the gestational period). Maternal animal mean platelet counts on GD 126, compared with the 0-mg/kg group, were decreased by 71.3% in the 15-mg/kg group and decreased by 75.7% in the 75-mg/kg group (P≤0.0001; Supplemental Table 5) but recovered rapidly between parturition and the end of the lactation period (Supplemental Figure 1A). In four maternal animals in the 15-mg/kg group and five maternal animals in the 75-mg/kg group, platelet counts decreased to below 100 × 109 cells/L on GD 126 but recovered by the end of the lactation period (LD 180). There were no apparent increases in pregnancy complications (i.e., placental hemorrhage with vaginal bleeding, premature delivery, and increased blood loss during parturition) resulting from the decreased platelet counts on GD 126 in the felzartamab groups.
Additional analysis of hematologic parameters in maternal animals on LD180 revealed no differences between the vehicle group and felzartamab groups in mean white blood cell count, lymphocyte count, neutrophil count, and red blood cell parameters (Supplemental Figure 2, Supplemental Tables 6). Similar results were observed in the prior 26-week study (data not shown), suggesting that felzartamab may not deplete CD38+ myeloid cell populations in the blood other than platelets.
Among infant animals on PND 98 and PND 171, there were no decreases in mean platelet count between the vehicle and felzartamab groups (Supplemental Figure 1B, Supplemental Table 7).
Discussion
The results of this ePPND study provide insight into potential effects of the anti-CD38 monoclonal antibody felzartamab on pre- and postnatal development, which have remained largely unclear. In pregnant marmoset monkeys, felzartamab was well tolerated at the evaluated doses, with no apparent effects on body weight, pregnancy loss, rates of stillbirth or abortion, litter counts, or the number of liveborn infants. Infant marmoset monkeys, exposed to felzartamab throughout development in utero, exhibited no felzartamab-related malformations or variations in external anatomy or morphology and no felzartamab-related changes in histopathology, hematologic and immune cell development, or humoral immune response to vaccination.
The use of marmoset monkeys for biomedical research has recently increased and spans multiple disciplines, including infectious disease, neuroscience, aging and toxicology.55–60 However, due to the lack of published marmoset control data required for evaluating in vivo observations, cynomolgus monkeys are typically selected as the non-human primate model when pharmacologically relevant. 39 To our knowledge, this is the first published ePPND study conducted in marmoset monkeys, which were selected as the pharmacologically relevant species for felzartamab toxicologic testing owing to a similar CD38 sequence homology, antibody binding affinity and functional activity as well as CD38 expression profile versus humans. The toxicokinetic, hematologic, and histopathologic observations collected during the gestation, parturition, and postnatal periods, as well as the data on anatomical, morphological, and immune system development, will provide important maternal and infant historical control data for future studies in marmoset monkeys.
Historically, there is a high rate of pregnancy loss among non-human primates. 50 Pregnancy loss (36.1% from GD 0 to 50), timing of early abortions, stillbirth, gestational length, and litter size in this study were consistent with those described in other studies of marmoset monkeys.45,61 A high pregnancy loss rate of 37.5% was observed in this ePPND study, with 31.3% occurring as spontaneous fetal abortions and 6.3% as stillbirths in all three dose groups. Furthermore, consistent with other reports,61,62 all abortions in this study occurred during the first two trimesters (predominantly GD 25 to 60). The proportion of maternal animals with liveborn infants was similar across the felzartamab dosing groups (0 mg/kg, 68.8%; 15 mg/kg, 68.8%; 75 mg/kg, 62.5%), and the mean duration of gestation in all three groups was 143 days, in agreement with other reports of gestational durations of 141 to 146 days.39,49-52 The consistency of these observations with published ranges and the similar frequencies between the control and felzartamab dosed groups suggest that felzartamab administered during pregnancy does not increase pregnancy loss as abortions or stillbirths in marmoset monkeys.
In contrast with macaques such as cynomolgus monkey, marmoset monkeys commonly ovulate multiple ova per estrous cycle and therefore often give birth to twins and triplets, whereas single infant births are infrequent and typically attributed to fetal reabsorption in response to maternal health or maternal caloric reserves.45,46,51,54,62,63 Likewise, most maternal animals in our study gave birth to twin (n = 18 [54.5%]) or triplet litters (n = 14 [42.4%]). Triplet litters have been associated with a higher rate of early infant mortality and/or moribundity (within the first week of life) than twin litters due to failure of marmoset families to provide proportionally increased care for larger litters.36,46,62,64 In this study, infants in triplet litters were placed on a rotational feeding schedule to prevent early mortality; however, this approach resulted in an increase in the number of moribund triplet infants. Consequently, five infants from triplet litters were euthanized as soon as weight loss in more than one infant in a triplet litter was detected. Notably, the rate of early infant death (during the first week of life) did not increase with felzartamab treatment (0 mg/kg, 13.0%; 15 mg/kg, 15.4%; 75 mg/kg, 8.3%), and the proportions of infants alive at the end of the study was similar across dosing groups (0 mg/kg, 78.3%; 15 mg/kg, 73.1%; 75 mg/kg, 75.0%). Likewise, the proportions of other non-triplet moribund infants euthanized throughout the study were similar between dosing groups (0 mg/kg, 8.7%; 15 mg/kg, 3.8%; 75 mg/kg, 8.3%). These results suggest that felzartamab does not affect postnatal infant survival.
The high frequency of twin and triplet litters among marmoset monkeys offers a greater chance of detecting morphological and visceral variations and malformations than in species with single-infant litters, such as the cynomolgus monkey. The observed lack of visceral variations and malformations in this study is consistent with the relatively low expression of CD38 on non-hematologic tissues in general.20–27 Furthermore, in non-human primates presumably including marmoset monkeys, higher levels of maternal antibodies are transferred across the placenta after organogenesis, at the end of the second trimester and during the third trimester of pregnancy, compared with the first trimester. 37
The single malformation (i.e., fusion of sternebra 6 and 7) observed in one infant in the 75-mg/kg group was considered not to be related to felzartamab due to the lack of similar observations in other infants. Skeletal variations observed in infants from PND 0 to 7 (i.e., those with early death), varied in frequency between the dosing groups, with some observed more frequently in the vehicle group than in the felzartamab groups (i.e., those in the pelvic girdle), some inconsistently between the vehicle and felzartamab groups (i.e., those in the vertebral column), and some more frequently in the felzartamab groups than in the vehicle group (i.e., those in the skull, sternum, and ribs). On PND 100, there were no differences in the incidence of skeletal alterations between the vehicle and felzartamab groups. The incomplete ossification findings among infants on PND 100 are not surprising given that ossification in the marmoset monkeys occurs later during gestation in marmoset monkeys than in other non-human primates. 54 Incomplete ossification can sometimes signal a developmental delay or toxicity if associated with reduced infant body weight or maternal toxicity; however, there were no observations of reduced mean infant body weight or maternal toxicity among felzartamab-dosed animals in this study. The observed short supernumerary ribs, which represent ossification centers lateral to the vertebrae, are usually not of developmental importance and typically resolve as the animal matures. 65 The low number of infants assessable from PND 0 to 7 in this study (considering early death), as well as the lack of historical data on skeletal variations in marmoset monkey infants in general, make it difficult to draw conclusions on the relatedness of skeletal variations to felzartamab among newborns.
Given the major similarities in immune system development between non-human primates and humans, marmoset monkeys are ideal for assessing the potential effects of immune modulating molecules on fetal immune system development. As in humans, development of the fetal immune system in non-human primates is a stepwise process during gestation, and although not specified, a similar developmental process is assumed for marmoset monkeys. The thymus (GD 35), spleen (GD 40), and lymph node tissues (GD 50) begin developing at the beginning of the first trimester. 66 From GD 100 to 145, the secondary lymphatic organs are populated with B cells, T cells, NK cells, dendritic cells, macrophages, and plasma cells and form a functional immune system at parturition. 66 Despite the embryofetal exposure to felzartamab throughout gestation, there were no differences between the vehicle and felzartamab groups in mean numbers of peripheral T cells, NK cells, and macrophages on PND 98, suggesting that felzartamab either does not result in measurable changes in these populations, or that any potential reduction had recovered by 3 months after parturition. Despite the observed increase in mean peripheral B cell numbers by flow cytometry analysis, the overall increase was small. Furthermore, the ability of all dosing groups to mount a robust IgM and IgG humoral immune response in the TDAR assay indicates that gestational exposure to felzartamab does not interfere with the development of a functional vaccination response 3 months after parturition in non-human primates. 66
The decrease in platelet counts among maternal animals dosed with felzartamab in this ePPND study and in the prior 26-week repeat-dose toxicology study appears to primarily be a pharmacologic effect specific to marmoset monkeys, which we observed to have three-fold greater expression of CD38 receptor on platelets compared with human platelets. Thrombocytopenia has not been reported as a safety concern for anti-CD38 therapeutics approved for the treatment of multiple myeloma, 66 or in clinical investigation for autoimmune and antibody-mediated diseases, such as primary immune thrombocytopenia, systemic lupus erythematosus, antibody-mediated rejection, and primary membranous nephropathy.67–70 Although thrombocytopenia has been observed with felzartamab in humans with multiple myeloma, the rates are low and could be attributed to the underlying disease and background treatments rather than felzartamab.71,72 Similarly, treatment with felzartamab did not result in an increased incidence of thrombocytopenia or decreased platelet counts in patients with antibody-mediated rejection or primary membranous nephropathy.70,73 Notably, despite the marked decrease in platelets below 100,000 cells/µL in felzartamab-dosed animals in this study, there were no observations of hemorrhage, vaginal bleeding, or blood in the urine or stool. At platelet counts above 30,000 cells/µL (the threshold for safe vaginal birth in humans 74 ), none of the maternal animals in this study had birthing complications despite a single breech birth with a dead litter. In addition, deaths of infants after birth from PND 0 to 7 were not associated with hemorrhage in microscopically evaluated tissues, supporting the assumption that the platelet decrease does not result in any adverse clinical effects. Interestingly, among infants, no platelet count reduction was identified 3 months following birth, suggesting a complete recovery from the initial decrease; however, due to the small size and blood volume of marmoset infants, earlier assessment of hematologic parameters was not possible.
There were no apparent changes in hematologic parameters (i.e., white blood cell count, lymphocyte count, neutrophil count, and red blood cell count) in females during gestation and at the end of the postnatal period, as numbers were within the published ranges for adult female marmoset monkeys. 75 The lack of impact (i.e., cytotoxicity) on these cellular populations may be due to their relatively lower surface expression of CD38 versus that on ASCs and NK cells.21,22,25,28
Frequent toxicokinetic sampling enabling noncompartmental data analysis is difficult with marmoset monkeys due to their low body weight, and consequently, overall limited blood volume. Therefore, this study used a sparse TK sampling protocol combined with a sensitive bioanalytical assay for the quantification of felzartamab in plasma. Felzartamab exposure in maternal animals of this ePPND study was estimated using a Good Laboratory Practice−compliant PopPK model that was initially developed with toxicokinetic data from the prior 26-week chronic toxicity study. The PopPK model predicted the observed data for both studies well, and the estimated toxicokinetic parameters were consistent with those generated by noncompartmental analysis in a prior 12-week chronic toxicity study (data not shown).
Although there were insufficient data to model plasma felzartamab concentrations in newborn animals in this ePPND study, observed plasma felzartamab concentrations in newborns were similar to or greater than those in maternal animals after parturition (PND 28), indicating that felzartamab is actively transferred across the placenta. Additionally, the observed similarity of felzartamab plasma concentrations between pregnant animals in the ePPND study and non-pregnant animals in the 26-week chronic toxicity study at the same dose levels suggest that transplacental transfer of felzartamab did not impact maternal felzartamab plasma concentrations. Although concentrations of felzartamab in breast milk were not assessed, systemic exposure to monoclonal antibodies via lactation was expected to be minimal due to a high molecular weight, which generally limits passage from the maternal blood into milk across the mammary epithelium, as well as systemic absorption from the infant’s gastrointestinal tract. 76 Consequently, transplacental transfer is hypothesized to be the primary mechanism for the plasma felzartamab concentrations detected in newborn animals in this study.
The lack of robust historical control data for marmoset monkeys was a limitation of this study. However, the data presented will inform future ePPND studies in marmoset monkeys and will serve as a historical reference. Furthermore, the inherent morbidity of triplet infants and the euthanasia of triplets upon signs of moribundity from PND 0 to 7 limited our ability to interpret whether observations in newborns were caused by felzartamab administration. The increased background morbidity also increased the proportion of infants that were euthanized moribund, which is common among marmoset monkey triplet litters. Other limitations considered during planning of this study were the use of family housing, which can lead to repeat pregnancies, and the small animal size, which restricted the volumes of vehicle and felzartamab that could be administered. Lastly, the transplacental transfer of felzartamab to infants was not characterized in a longitudinal manner such that timing of felzartamab exposure before parturition is unknown.
In conclusion, gestational dosing of felzartamab did not appear to affect rates of liveborn litters or pregnancy loss, including stillbirth and abortion, the timing of early abortions, or postnatal survival, as these were consistent with the known literature and were similar between dosing groups. Gestational felzartamab exposure did not affect infant development, including morphology and organogenesis. Despite CD38 expression on B cells, plasmablasts, NK cells, and T cells,20,22,27,28 no felzartamab related effects on peripheral immune cell counts or humoral immunity to vaccination were observed. Similarly, skeletal malformations were not detected despite prior observations indicating that bone density may decrease in CD38–deficient mice due to increased osteoclast activity. 32 Although the ability to assess the impact of felzartamab on early postnatal skeletal variations was limited due to small sample sizes, there was no evidence that felzartamab caused skeletal alterations later in postnatal development. Due to the high expression of CD38 on platelets, a transient decrease in platelet counts was observed in maternal animals but did not result in pregnancy complications or have any impact on the postnatal survival and development of infants. In addition, we have demonstrated that the marmoset monkey is a valid model for use in ePPND toxicologic studies and that a population pharmacokinetic model approach enables the characterization of toxicokinetics in a species with low blood volumes and a limited number of sampling time points. However, it is important to consider that management practices are limited in the ability to prevent early postnatal loss among triplet litters, which are more common among marmoset monkeys than other non-human primates. Overall, the outcome of this study warrants further development of the marmoset monkey ePPND model and supports the investigation of felzartamab as a treatment for chronic immune-mediated diseases driven by CD38+ cells.
Supplemental Material
Supplemental Material - Enhanced Prenatal and Postnatal Development Study in Marmoset Monkeys Following Administration of Felzartamab
Supplemental Material for Enhanced Prenatal and Postnatal Development Study in Marmoset Monkeys Following Administration of Felzartamab by Doris Mangelberger-Eberl, Mary Ellen Cosenza, Stefan Härtle, C. Marc Luetjens, Brian T. Welsh, Stefan Steidl, Donna L. Flesher, and Leslie W. Chinn in International Journal of Toxicology
Footnotes
Acknowledgments
Ben Scott, PhD (Scott Medical Communications, LLC), provided medical writing support funded by Human Immunology Biosciences, Inc.
Author Contributions
Mangelberger-Eberl, D. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Cosenza, M. contributed to acquisition and interpretation and critically revised manuscript; Härtle, S. contributed to conception and design, contributed to acquisition and analysis, and critically revised manuscript; Luetjens, C.M. contributed to acquisition and interpretation and critically revised manuscript; Welsh, B. contributed to acquisition and interpretation and critically revised manuscript; Steidl, S. contributed to conception and design, contributed to acquisition, and critically revised manuscript; Flesher, D. contributed to acquisition, analysis, and interpretation and critically revised manuscript; Chinn, L. drafted and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.M.-E., S.H., S.S., and B.T.W. are employees of and own stock in MorphoSys AG. M.E.C. is an independent toxicology consultant contracted by Human Immunology Biosciences. C.M.L. is employee of Labcorp Early Development Services GmbH. D.L.F. is an employee of Human Immunology Biosciences, Inc. and owns stock and/or options in Human Immunology Biosciences, Inc. and Amgen Inc. L.W.C. is an employee of Human Immunology Biosciences, Inc. and owns stock and/or options in Human Immunology Biosciences, Equillium, and Roche.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by MorphoSys AG and Human Immunology Biosciences, Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
