Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of 10 polyol phosphates. Some of the possible functions in cosmetics that are reported for this ingredient group are chelating agents, oral care agents, and skin conditioning agents. The Panel reviewed relevant data relating to the safety of these ingredients under the intended conditions of use in cosmetic formulations, and concluded that Sodium Phytate, Phytic Acid, Phytin, and Trisodium Inositol Triphosphate are safe in cosmetics in the present practices of use and concentration described in the safety assessment. The Panel also concluded that the data are insufficient to determine the safety of the following 6 ingredients as used in cosmetics: Disodium Glucose Phosphate, Manganese Fructose Diphosphate, Sodium Mannose Phosphate, Trisodium Fructose Diphosphate, Xylityl Phosphate, and Zinc Fructose Diphosphate.
Keywords
Introduction

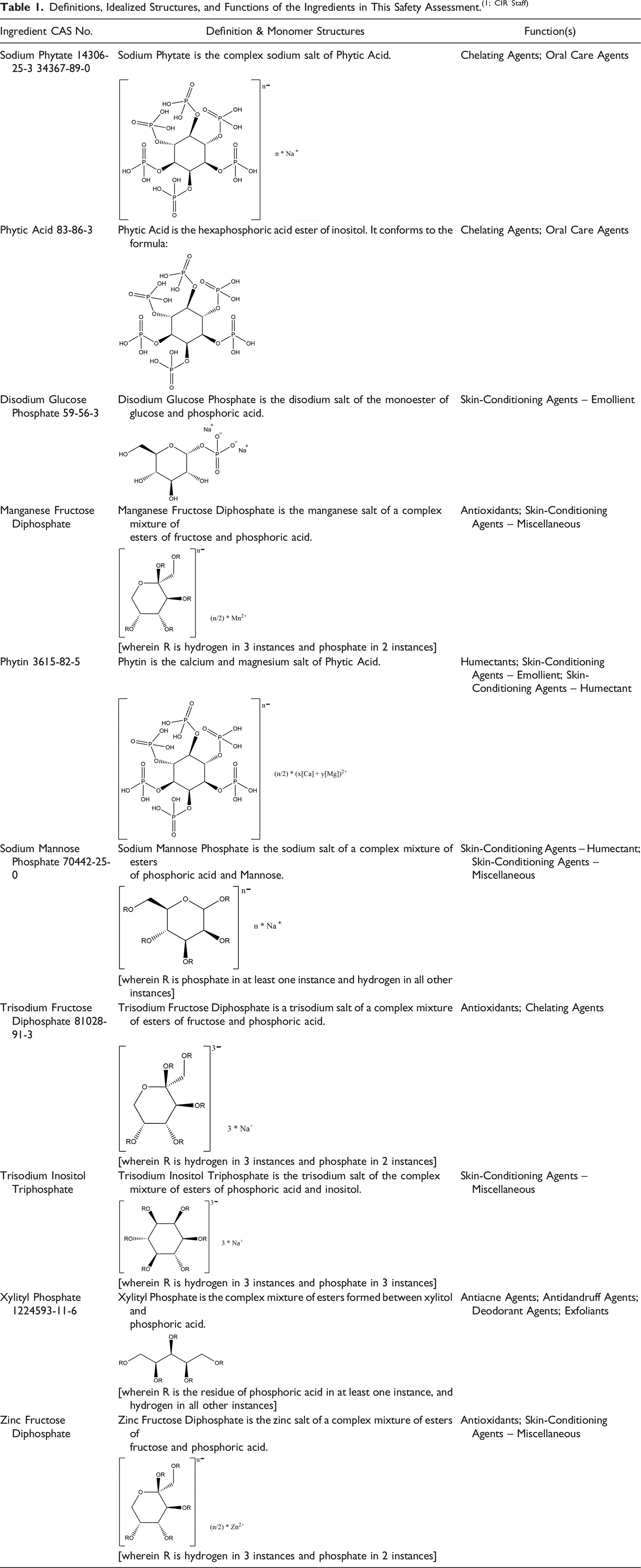
Definitions, Idealized Structures, and Functions of the Ingredients in This Safety Assessment.(1; CIR Staff)
This safety assessment includes relevant published and unpublished data for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A list of the typical search engines and websites used, sources explored, and endpoints that the Panel evaluates, is available on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
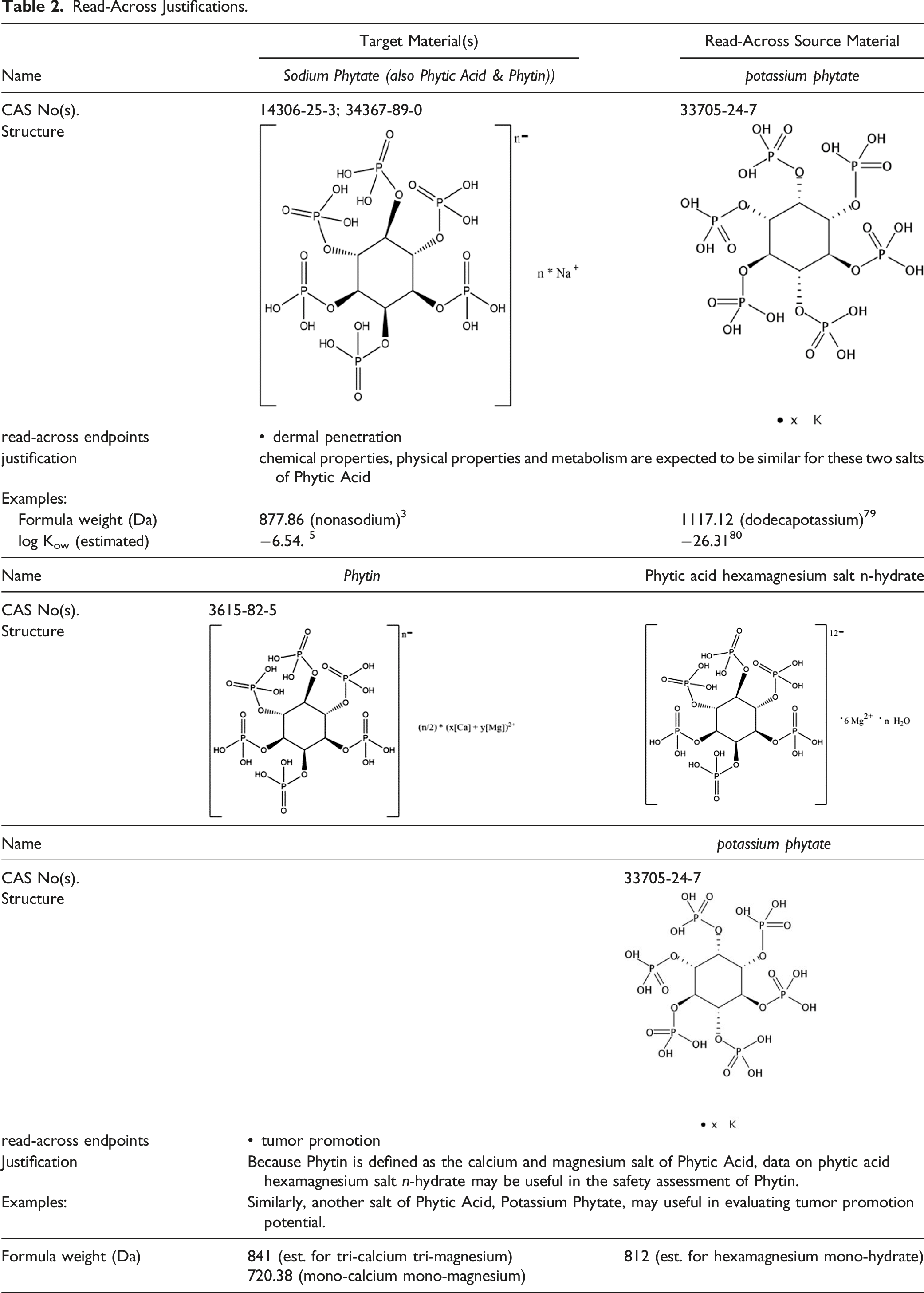
Read-Across Justifications.
Chemistry
Definition and General Characterization
The ingredients in this report are each the phosphate(s) of a carbohydrate (inositol or a monosaccharide or a “sugar alcohol”) or a salt thereof. One example of these polyol phosphate salts is Disodium Glucose Phosphate (Figure 1). Disodium Glucose Phosphate, example of a saccharide phosphate.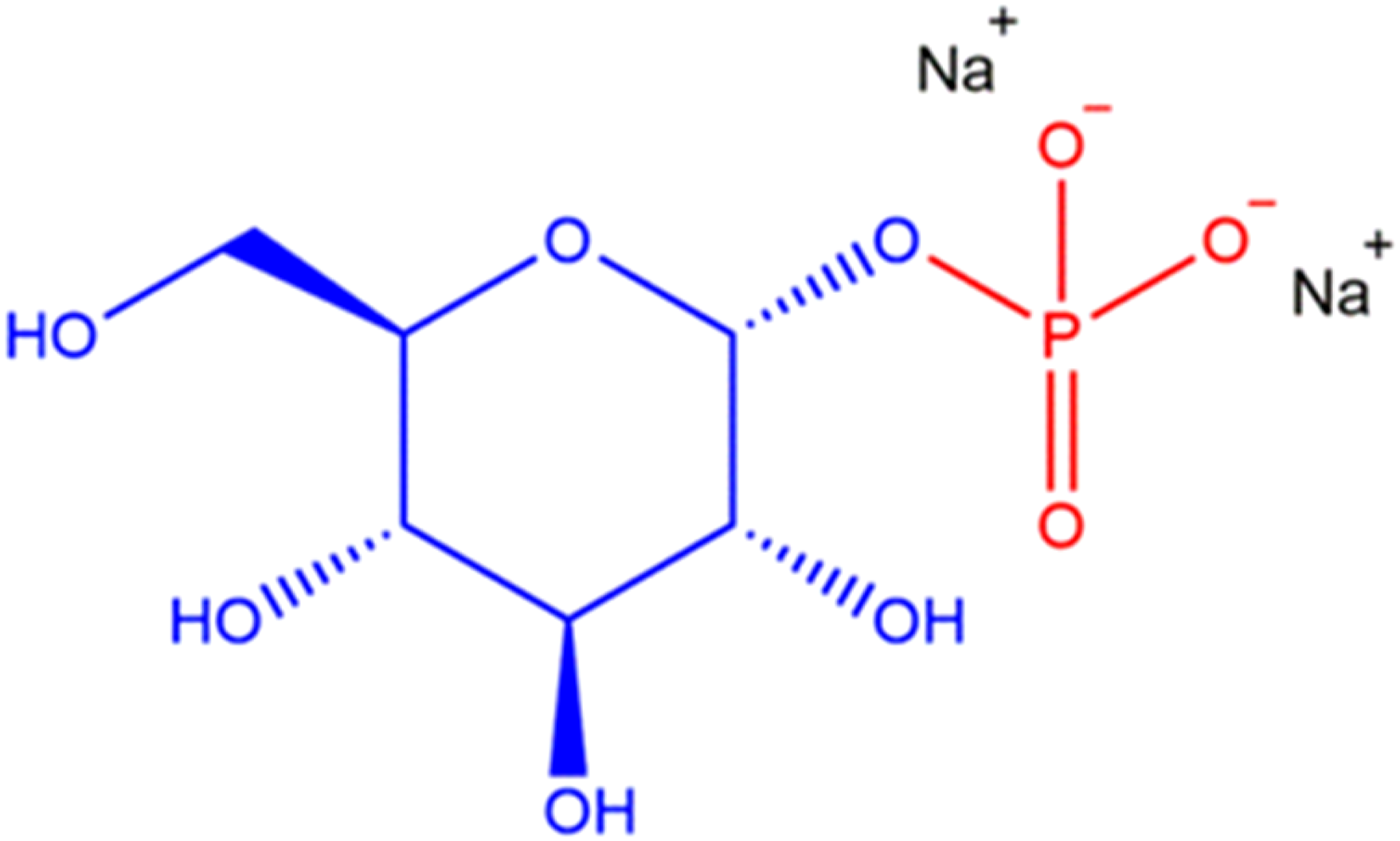
Some of these ingredients may exist in open chain, or cyclic furanose and/or pyranose forms, like many sugars do. Some of these ingredients are naturally occurring. Indeed, Phytic Acid and other particular inositol phosphates (Figure 2) are present in practically all mammalian cells.
2
Phytic Acid, example of an inositol phosphate.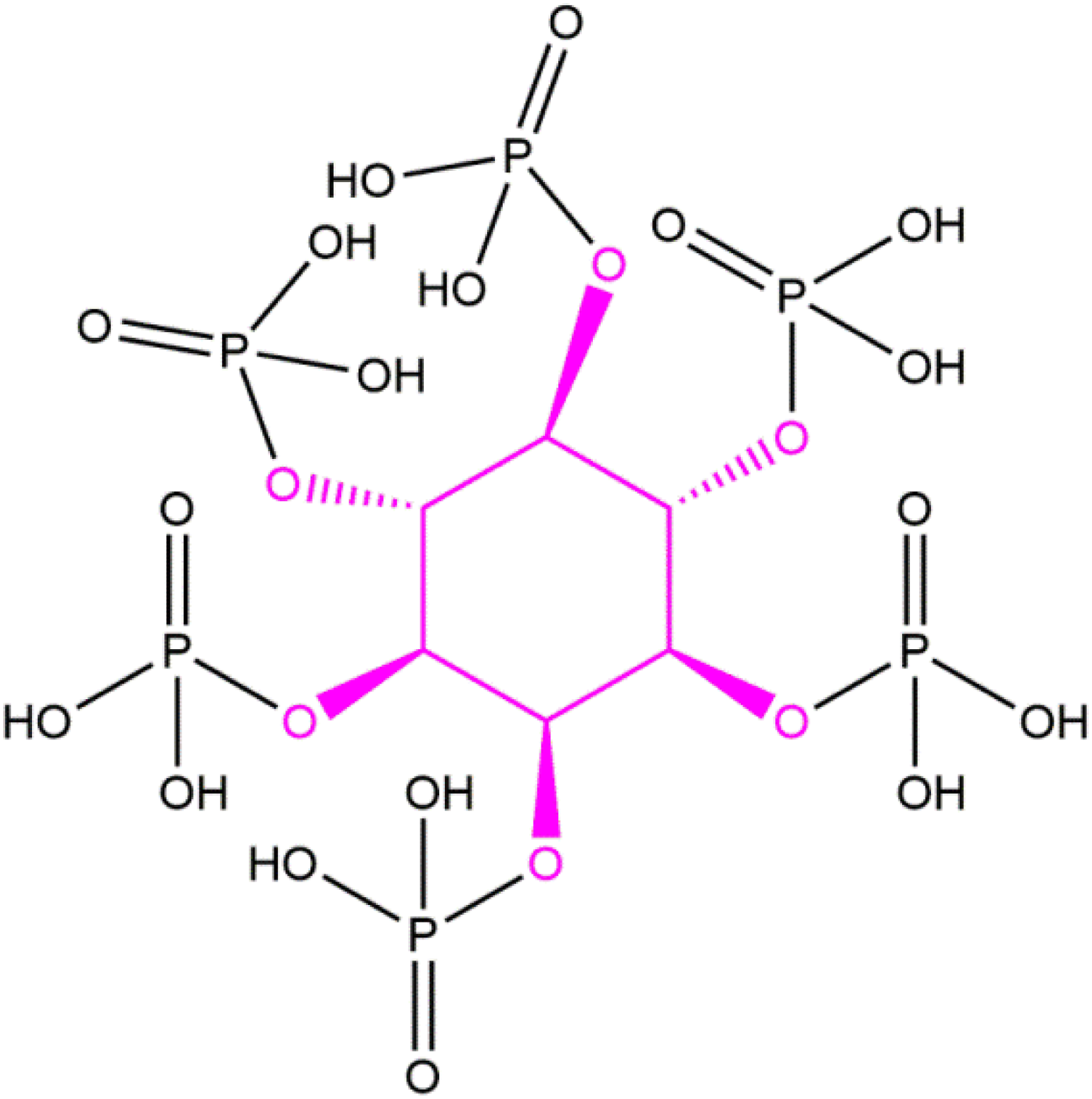
The definitions, structures, and functions in cosmetics of these ingredients are presented in Table 1.
Chemical and Physical Properties
Method of Manufacture
Phytic Acid
The methods for the production of Phytic Acid, summarized below, involve acid hydrolysis (e.g., sulfuric acid or hydrochloric acid) of one or more of the following plant materials: maize seed (kernels), defatted food-grade rice bran, rice bran, or rice husks (hulls).
According to one source, an aqueous solution of Phytic Acid (50% aqueous) for use in foods is obtained by acid hydrolysis of maize seed (kernels), rice bran, or rice husks (hulls). 8 The initial hydrolysis is followed by multiple processing steps that include: centrifugation, filtration, neutralization, dilution, decolorization, further hydrolysis and pH adjustment, ion-exchange, and concentration.
According to one foods manufacturer, the production of Phytic Acid (50% solution) involves the addition of diluted sulfuric acid to defatted food-grade rice bran to dissociate phytate from iron and protein complexes. 9 The solution then undergoes centrifugation, filtration to remove impurities, neutralization with sodium hydroxide, and dilution with water. Also, the diluted solution is decolorized, and sulfuric acid is added to dissociate the bound minerals from phytate to release Phytic Acid. The Phytic Acid-containing solution undergoes pH adjustment, ion-exchange, decolorization, and vacuum concentration to achieve a 50% concentration. Because rice bran is the source of Phytic Acid in this production method, it should be noted that one source indicates that the content of Phytic Acid in rice bran ranges from 0.22% to 2.22%. 10
Another reported method for the production of Phytic Acid begins with the hydrochloric acid leaching of bran, which is followed by filtration, neutralization with sodium hydroxide, and water scrubbing. 11 The resulting crude phytin paste is acidified and then subjected to positive ion exchange, condensation, and decolorization, yielding Phytic Acid.
Sodium Mannose Phosphate
Sodium Mannose Phosphate is manufactured by enzymatic reaction from pyrophosphate and mannose. 12 The reaction medium is then stabilized by denaturing the enzyme. This step is followed by purification of the medium.
Composition
Phytic Acid
According to a company’s food-grade chemical specification for Phytic Acid (50% solution), 48% to 52% is the range for Phytic Acid content and for water content. 9
Impurities
Phytic Acid
According to the United States Pharmacopeial (USP) Convention’s Food Ingredients Expert Committee, the acceptance criteria for Phytic Acid (aqueous solution) include: arsenic (not more than 3 mg/kg), calcium (not more than 0.02%), chloride (not more than 0.02%), inorganic phosphorus (not more than 0.2%), lead (not more than 1 mg/kg) and sulfate (not more than 0.02%). 8
Specifications for one manufacturer’s food-grade Phytic Acid (50% solution; as described above in Method of Manufacture) include: heavy metals (surmised via analysis of lead sulfide precipitate; <0.002%), lead (<0.0001%), arsenic (<0.0002%), total phosphorus (13.5% to 14.6%), inorganic phosphorus (not more than 1%), chloride (not more than 0.04%) and sulfate (not more than 0.071%). 9 Furthermore, because the raw material that is used in the production of Phytic Acid (50% solution) is defatted rice bran, there is the potential for presence of residual pesticides and herbicides.
An impurities analysis of 50% Phytic Acid (vehicle not stated) was provided. 13 Results indicated that the levels of the following heavy metals were below the detection limits (≤0.0004% to ≤0.0001%): mercury, cadmium, zinc, cobalt, copper, nickel, and lead. Determination of the level of arsenic was not possible because the 50% Phytic Acid preparation appeared to strongly interfere with the assay reagents. As expected, the negative control (distilled water) tested negative for arsenic.
Sodium Mannose Phosphate
Possible impurities (0.1% to 0.5%) of Sodium Mannose Phosphate are: phosphate, sodium salt; pyrophosphate, sodium salt; sodium chloride; and magnesium and ammonium ions. 12
Use
Cosmetic
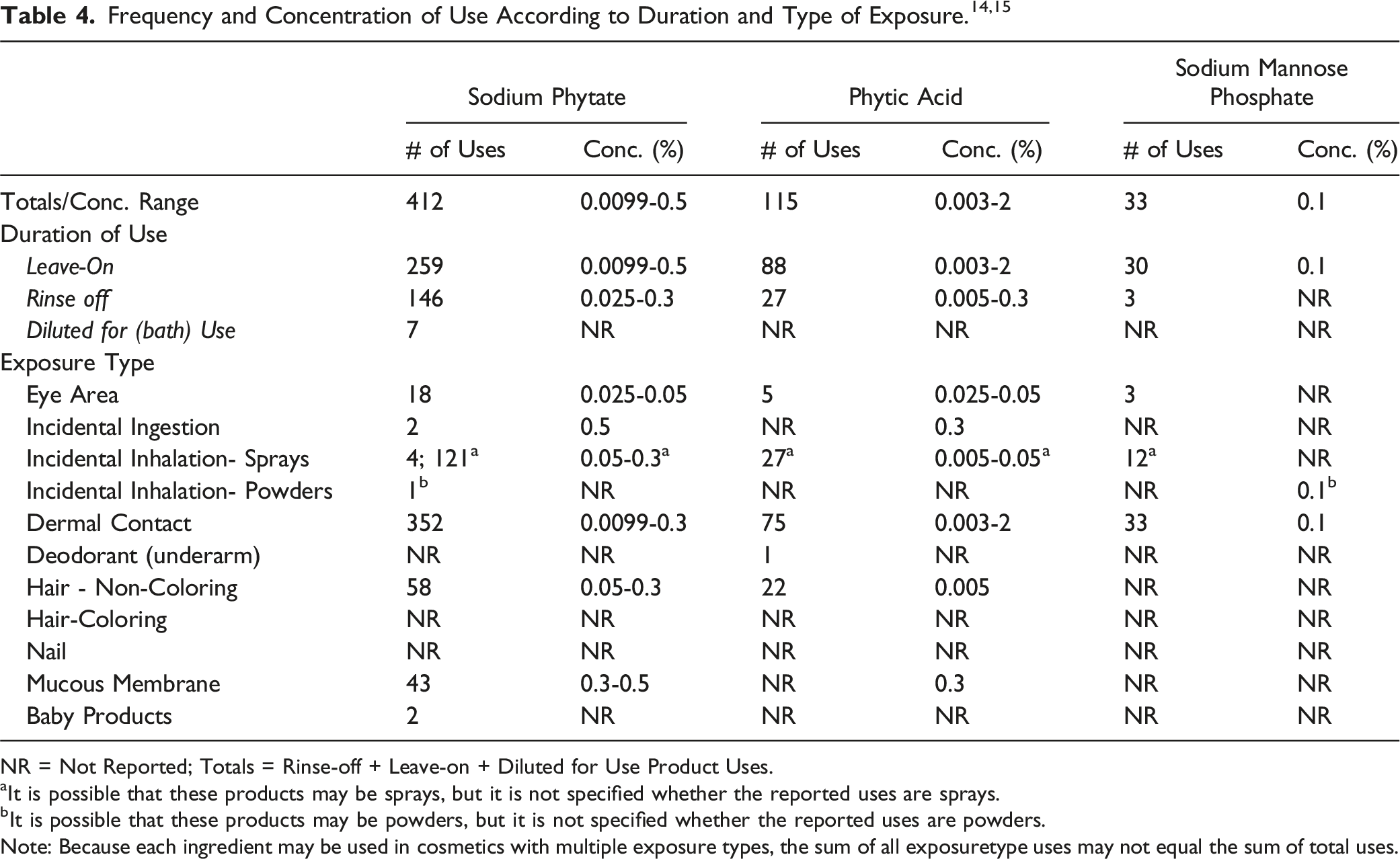
The safety of the polyol phosphates is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. 14 Use concentration data are submitted by the cosmetics industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category. 15
NR = Not Reported; Totals = Rinse-off + Leave-on + Diluted for Use Product Uses.
aIt is possible that these products may be sprays, but it is not specified whether the reported uses are sprays.
bIt is possible that these products may be powders, but it is not specified whether the reported uses are powders.
Note: Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposuretype uses may not equal the sum of total uses.
According to VCRP and Council survey data, the following 7 polyol phosphates are not used in cosmetic products in the US: Disodium Glucose Phosphate; Manganese Fructose Diphosphate; Phytin; Trisodium Fructose Diphosphate; Trisodium Inositol Triphosphate; Xylityl Phosphate; and Zinc Fructose Diphosphate.
Cosmetic products containing polyol phosphates may be applied to the skin and hair or, incidentally, may come in contact with the eyes (at maximum use concentrations up to 0.05% for Sodium Phytate and Phytic Acid in eye makeup removers and eye lotions, respectively) and mucous membranes (at maximum use concentrations up to 0.5% Sodium Phytate in lipstick). Ingredient use in lipstick products may result in incidental ingestion. Products containing polyol phosphates may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
Sodium Phytate is reported in the VCRP as being used in a perfume formulation, which may result in incidental inhalation exposure. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles below 10 μm, compared with pump sprays.16-19 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.16,17
The polyol phosphates reviewed in this safety assessment are not included on the European Union’s list of substances that are restricted or list of substances that are prohibited in cosmetic products. 20
Non-Cosmetic
Sodium Phytate
Sodium Phytate is used as a complexing agent for the removal of traces of heavy metal ions. 3 It is also used as the starting material in the manufacture of inositol.
Phytic Acid
After reviewing a GRAS exemption claim, the US FDA issued the following statement: “Based on the information provided … as well as other information available to FDA, the agency has no questions at this time regarding … [the submitted] conclusion that Phytic Acid is GRAS under the intended conditions of use. The agency has not, however, made its own determination regarding the GRAS status of the subject use of Phytic Acid.” 21
Reportedly, Phytic Acid (2% to 4%) has proven to be efficient in the treatment of epidermal melasma, especially when associated with glycolic acid or retinoic acid. 22 Furthermore, the Phytic Acid combination peel has been described as a proprietary peel that is a mixture of glycolic acid, lactic acid, mandelic acid, and Phytic Acid.
Phytic Acid is used in the chelation of heavy metals in processing of animal fats and vegetables, as a rust inhibitor, in the preparation of phytate salts, in metal cleaning, and in the treatment of hard water. 4
Toxicokinetic Studies
Absorption, Distribution, Metabolism, and Excretion Studies.

Dermal Penetration
Human
Potassium Phytate (read-across source for Sodium Phytate, Phytic Acid, and Phytin)
In a study involving 20 healthy volunteers on a Phytic Acid-poor diet, the urinary excretion of Phytic Acid increased by 54% following topical treatment with a standard moisturizing gel containing 4% potassium phytate. Thus, the test substance was absorbed through the epidermis and dermis, entered the blood, and was excreted into the urine. Urine samples were collected at day 7 of treatment. 23
Absorption, Distribution, Metabolism, and Excretion (ADME)
Animal
Dermal: Sodium Phytate and Phytin
Over a period of 16 days, groups of 6 female Wistar rats consumed a synthetic purified diet that resulted in undetectable urinary Phytic Acid. 24 The rats were then treated topically (once per day for 14 days) with 4 g of a standard moisturizing cream supplemented with Sodium Phytate (0.4%, 1.2%, or 2%) or 2.0% Phytin. Phytic Acid was absorbed through the skin layers (having crossed the epidermis and dermis), entered the bloodstream, and urinary excretion was increased.
Oral: Phytic Acid
When [14C]-Phytic Acid was administered orally (in distilled water, by gastric tube) to groups of 5 male Sprague-Dawley rats, ∼6% of the administered dose was recovered in the feces at 48 h post-dosing. 25 Following the oral administration of [3H]-Phytic Acid (by stomach tube) to 9 male Fisher 344 rats, absorption (79.0 ± 10.0% of total radioactivity) was described as rapid and, at 24 h, much of the radioactivity was distributed in the liver, kidneys, muscle, and skin. Also, at 24 h, the total radioactivity recovered in the feces was 14.1 ± 8.7% of the administered dose, and the overall radioactivity in the urine collected was 2.4 ± 1.6% (most due to presence of the metabolite, inositol (the core, non-phosphorylated carbohydrate of Phytic Acid), concentration not stated) of the total administered dose. 26
Groups of 12 female Wistar rats were fed Phytic Acid in the diet at doses of 11.6 g/kg dry matter (DM) and 9 g/kg DM for 12 weeks. 27 The highest Phytic Acid concentrations were detected in the brain (5.89 × 10−2 mg/g DM), and concentrations detected in other organs were 10-fold less. In another study, C.B-17 SCID female mice (specific pathogen-free, bearing MDA-MB-231 breast cancer xenografts; number not stated) were dosed orally with 0.01 ml/g [14C]-Phytic Acid and unlabeled Phytic Acid so that each mouse received 20 mg/kg Phytic Acid and 0.150 mCi/kg in phosphate-buffered saline. 28 The % of the administered dose that was excreted in the urine as inositol was 0.3%, and ∼10% of the administered dose was present in the feces, primarily as inositol.
Human
Oral: Phytic Acid
In human subjects (number not stated), 1% to 3% of the total amount of Phytic Acid administered (oral dosing method unknown) was excreted in the urine as Phytic Acid. 29 The results of another study indicated that 1% to 10% of the total amount of Phytic Acid ingested was excreted in the urine. 30
Sodium Phytate, Phytic Acid, and Phytin
In a study in which 7 volunteers received Phytic Acid, Sodium Phytate, or Phytin in the diet, urinary levels of Phytic Acid increased continuously until normal values were reached; the amount of Phytic Acid excreted was not affected by the type of Phytic Acid salt that was administered. 31 Because normal values for urinary Phytic Acid are not stated in this publication it should be noted that, according to another source, the amount of Phytic Acid that is usually present in human urine is 0.4 g/l. 30
Phytate (cation not declared; read-across source for Sodium Phytate, Phytic Acid, and Phytin)
Healthy women (15 young and 14 elderly) consumed low-phytate diets (young women: 682 mg phytate/day; elderly women: 782 mg phytate/day) or a high-phytate diet (young women: 1587 mg phytate/day; elderly women: 1723 mg phytate/day) for a period of 10 days. 32 Study results indicated that phytate degradation in the gastrointestinal tract was substantial and more variable in young women than in elderly women. In a similar study, healthy women (14 young and 14 elderly) consumed low-phytate diets (young women: 681 mg phytate/day; elderly women: 782 mg phytate/day) or a high-phytate diet (young women: 1584 mg phytate/day; elderly women: 1723 mg phytate/day) for a period of 10 days. A considerable amount of dietary phytate was degraded in the human gut. 33 The degradation rate of dietary phytate was approximately 77% for young women, which was significantly lower than that reported for elderly women (86%) (P < 0.05). Results relating to toxicity in these two oral feeding studies are included in the Other Clinical Reports section of this safety assessment.
The extent of dietary phytate degradation has been reported to vary from 40 to 75% in humans, and may occur throughout the whole gut.34,35 Phytate degradation may result from the activities of dietary phytase, intestinal mucosal phytase, or phytase that is produced by the small intestinal microflora. 32 Mucosal phytase in the human small intestine seems to play a minor role when compared to dietary phytase for phytate hydrolysis. 36 Phytate degradation is also thought to occur in the colon, due to the action of microbial phytase originating from colonic bacteria. 35
Toxicological Studies
Acute Toxicity Studies
Acute Toxicity Studies.

Oral
Phytic Acid
In an acute oral toxicity study involving Jcl:ICR mice (number not stated), LD50 values of 1150 mg/kg (females) and 900 mg/kg (males) were reported.9,37 LD50 values of 480 mg/kg (females) and 400 to 500 mg/kg (males) were reported in an acute oral toxicity study involving F344 rats (number not stated).9,38
Intravenous
Sodium Phytate
The intravenous (i.v.) administration of Sodium Phytate to groups of 10 NMRI mice at doses up to 0.56 mg/g (range of doses administered within 7 minutes) yielded an LD50 of ∼0.5 mg/g, and there were no detectable effects from infusion when the rate was not more than 0.02 mg/g/minute. 39 When Sodium Phytate was administered i.v. to rats at lower doses of 0.035 and 0.07 mg/g, there were no detectable signs when doses were administered at a rate requiring 40 minutes for administration of the total dose. Different infusion rates were used in this study, and whether or not mortalities were observed was dependent on the infusion rate.
Short-Term Toxicity Studies
Short-Term Oral Toxicity Studies.

Oral
Sodium Phytate
Groups of 5 male Wistar rats were fed Sodium Phytate at dietary concentrations ranging from 0.02% to 10% (in high-sucrose diet) for 14 to 15 days. 40 Statistically significant depression of food intake and growth was observed at dietary concentrations of 5% and 10% Sodium Phytate, but not at lower concentrations. There were no significant differences in food intake, body weight, and organ weights among groups of 10 diabetic KK mice fed Sodium Phytate in the diet (0.5% or 1%) for 8 weeks. 41
Phytic Acid
Three different concentrations of 50% Phytic Acid solution (equivalent to doses of 80, 155, or 315 mg/kg/day) were administered orally to groups of 21 to 24 pregnant female JcI:ICR mice on gestation days 7 to 15. There were no maternal mortalities in the control or 80 mg/kg/day group. Two of 22 dams in the 155 mg/kg/day group and 15 of 24 dams in the 315 mg/kg/day group died during the study. Statistically significant changes in organ weights were observed in all dose groups; however, there was no significant dose-response relationship for these findings and no statistically significant macroscopic findings were observed.9,42 Other study results are included in the section on Developmental and Reproductive Toxicity. Groups of 8 male Wistar rats were fed dietary concentrations of 0.1% to 1% Phytic Acid for 20 days. No effects on organ weight were noted, but the concentration of triiodothyronine (T3) in the serum was statistically significantly lower at all administered Phytic Acid concentrations. 43
In a 12-week dose range-finding study (for 108-week oral carcinogenicity study), groups of 20 F344 rats (10 males and 10 females) received Phytic Acid at concentrations up to 10% in drinking water. 44 All rats that received 10% Phytic Acid and all males and 1 female that received 5% Phytic Acid died before the end of the experiment. The 108-week oral carcinogenicity study is summarized in the ‘Carcinogenicity’ section of this safety assessment.
In another study, 10 female C7BL/6J mice received Phytic Acid (2% in distilled drinking water) for a 70-day period. Dosing with Phytic Acid was well tolerated. 45
Chronic Toxicity Study
In a chronic study, 8 female Tg2576 mice (Alzheimer’s mouse model) and 10 female C7BL/6J mice received Phytic Acid at a concentration of 2% in distilled water for 6 months. 45 Seven control female Tg2576 mice and 12 control female C7BL/6J mice received distilled drinking water for the same duration. Phytic Acid was well tolerated, as indicated by the observation that average weekly body weights (an indirect measurement of toxicity) were similar for vehicle and Phytic Acid-treated animals.
Developmental and Reproductive Toxicity Studies
Developmental and Reproductive Toxicity Studies.

Oral
Phytic Acid
Three different concentrations of 50% Phytic Acid solution (equivalent to doses of 80, 155, or 315 mg/kg/day) were administered orally to groups of 21 to 24 pregnant JcI:ICR mice on gestation days 7 to 18.9,42 No significant effects on the incidence of external or skeletal malformations were observed at any dose of Phytic Acid. There were also no significant effects on the following: number of live fetuses; number of corpora lutea; number of implantations; or incidence of early resorptions. 42 The treatment of groups of 30 male albino rats (Rattus norvegicus) with Phytic Acid had an ameliorative effect on the pathological and hormonal alterations induced by aflatoxin B1 injection. 46 Specifically, treatment with Phytic Acid had a marked regenerative effect upon the aflatoxin B1-induced histopathological changes in the seminiferous tubules (i.e., degeneration with absence of spermatozoa) and resulted in statistically significant (P < 0.05) amelioration of the reduced testosterone concentration induced by aflatoxin B1injection. 46
Genotoxicity Studies
Genotoxicity Studies.

In Vitro
Sodium Phytate
The genotoxicity of a Sodium Phytate trade name material consisting of 50% water, 1% ethanol, and approximately 49% Sodium Phytate was evaluated in the Ames test using the following Salmonella typhimurium strains: TA97a, TA98, TA100, TA102, and TA1535. 47 The test material, in deionized water, was evaluated at doses up to 4995 μg/plate with and without metabolic activation. Results were negative for genotoxicity. A second experiment (pre-incubation method, modification of Ames test) was performed to confirm the results of the first. The test material was evaluated at doses up to 5013 μg/plate, with and without metabolic activation. There were no signs of genotoxicity.
Phytic Acid
Phytic Acid (50% solution) was non-genotoxic in the Ames test, with or without metabolic activation, when tested at doses up to 10 mg/plate. 48 In the L5178Y TK+/− mouse lymphoma assay, Phytic Acid was non-genotoxic at concentrations up to 5000 μg/ml with or without metabolic activation. 49 Also, in chromosomal aberrations assays using Chinese hamster ovary (CHO) cells, 2 mg/ml Phytic Acid was non-genotoxic, 48 but at an unknown high concentration, it was genotoxic in CHO cells. 9
Sodium Mannose Phosphate
The genotoxicity of Sodium Mannose Phosphate was evaluated in the Ames test using S. typhimurium strains TA98, TA100, TA1535, and TA1537 and Escherichia coli strain WP2 uvrA. 50 Sodium Mannose Phosphate was tested at doses up to 5000 μg/plate, with and without metabolic activation. The test material was not genotoxic in any of the bacterial strains tested, with or without metabolic activation.
In Vivo
Phytic Acid
In the micronucleus test involving bone marrow cells (polychromatic erythrocytes) from ddY mice, Phytic Acid was non-genotoxic when administered intraperitoneally (i.p.) as 4 doses of 30 mg/kg or as a single 60 mg/kg dose. 9
Carcinogenicity Studies
Carcinogenicity Studies.

Phytic Acid
Phytic Acid was administered at a concentration of 1.25% or 2.5% in drinking water to groups of 60 male and 60 female F344 rats for 108 weeks. 44 Renal papillomas (related to calcification and necrosis of renal papillae) were observed in 3 male and 4 female rats treated with 2.5% Phytic Acid and in 3 female rats treated with 1.25% Phytic Acid. Many tumors developed in all groups, including the control group, and the organ distribution of tumor types (other than the renal tumors observed) did not differ significantly from those known to occur spontaneously in the F344 strain.
Tumor Promotion
Phytic Acid, Sodium Phytate, and hexamagnesium phytate hydrate (read-across source for Phytin)
Sodium Phytate (2% in diet) was classified as a promoter of urinary bladder carcinogenesis, after initiation by exposure to 0.05% N-butyl-N-(4-hydroxybutyl) nitrosamine, in a study involving groups of 15 to 16 male F344 rats. Sodium Phytate significantly increased the development of pre-neoplastic and neoplastic lesions of the urinary bladder. Potassium phytate brought about a tendency for increase in papillomas, whereas hexamagnesium phytate hydrate and Phytic Acid were without effect. 51 Both Sodium Phytate and potassium phytate caused an increase in urinary pH.
Anti-carcinogenicity Studies
Anti-Carcinogenicity Studies.
Dermal
Phytic Acid
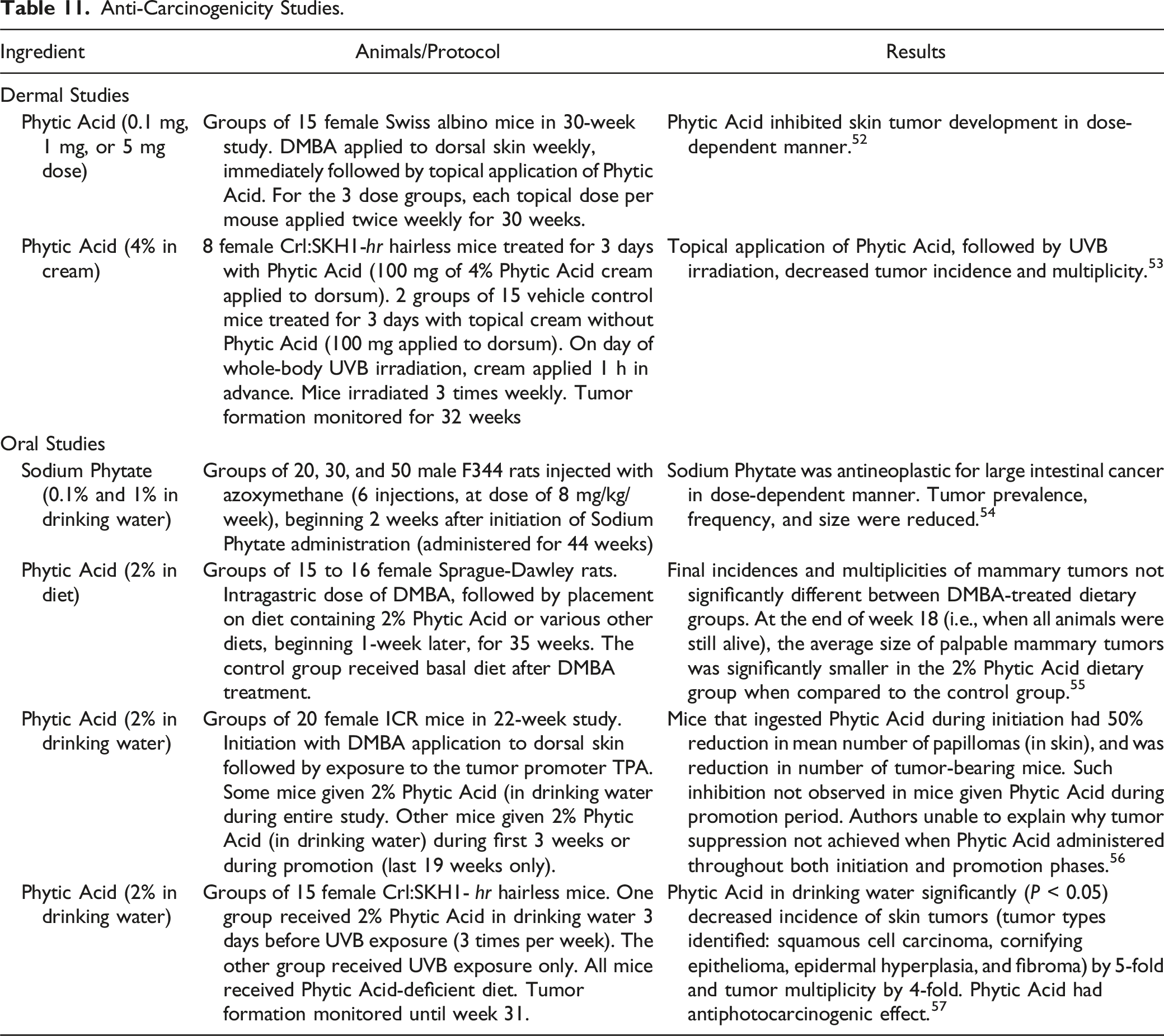
In a 30-week study involving groups of 15 female Swiss albino mice, Phytic Acid (0.1 mg, 1 mg, or 5 mg) was applied to the skin weekly after application of 7,12-dimethylbenz[a]anthracene (DMBA). Skin tumor development was inhibited in a dose-dependent manner. 52 When 8 female Crl:SKH1-hr hairless mice were treated with 4% Phytic Acid cream (100 mg applied to dorsum), followed by mid-wavelength ultraviolet light (UVB) irradiation, topical application of the 4% cream was found to decrease tumor incidence (monitored for 32 weeks) and multiplicity when compared to application of the cream without Phytic Acid. 53
Oral
Sodium Phytate
Sodium Phytate (0.1% or 1% in drinking water) was administered to groups of 20, 30, or 50 male F344 rats for 44 weeks after azoxymethane injection, and was found to be antineoplastic (reduction in tumor prevalence, frequency, and size) for large intestinal cancer in a dose-dependent manner. 54
Phytic Acid
In a study involving groups of 15 to 16 female Sprague-Dawley rats, feeding with 2% dietary Phytic Acid after dosing with DMBA resulted in significant reduction in the size of palpable mammary tumors, when compared to the control group, at the end of week 18. 55 In a 22-week study involving groups of 20 female ICR mice that received 2% Phytic Acid in drinking water, the animals were initiated with DMBA and then exposed to the tumor promoter 12-O-tetradecanoyl phorbol-13-acetate (TPA). Mice that ingested Phytic Acid during initiation had a 50% reduction in mean number of skin papillomas, but such inhibition was not observed when Phytic Acid was given during the promotion period or throughout both initiation and promotion phases. 56 Phytic Acid (2% in drinking water) was administered to 15 female Crl:SKH1-hr hairless mice prior to UVB exposure, and another group of 15 received UVB exposure only. Tumor formation was monitored until week 31, and concomitant administration of Phytic Acid during UVB exposure caused a statistically significant decrease in the skin tumor incidence, an anti-photocarcinogenic effect. 57
Other Relevant Studies
Anti-Inflammatory Activity
Phytic Acid
The anti-inflammatory activity of Phytic Acid in adult Swiss albino rats (groups of 6) was evaluated using the carrageenan-induced rat paw edema model. 58 The animals received oral doses (in water, given ad libitum) of Phytic Acid ranging from 30 to 150 mg/kg, and control animals were dosed with distilled water. At 1 h post dosing, the animals received a subplantar injection (left hind paw) of 1% carrageenan solution. The development of edema was the index of acute inflammatory changes, and differences in paw volume determined immediately after carrageenan injection versus 3 h post-injection were reported. Dosing with Phytic Acid caused a dose-dependent reduction in carrageenan-induced paw edema. The reduction in edema volume was statistically significant (P < 0.05) at doses ranging from 60 to 150 mg/kg, but not at a dose of 30 mg/kg. The maximum anti-inflammatory activity of Phytic Acid was observed at an oral dose of 150 mg/kg.
Cytotoxicity
Phytic Acid
The effect of Phytic Acid on cell growth was evaluated using a colorimetric assay for the quantification of cell proliferation and viability based on the cleavage of the WST-1 tetrazolium salt by mitochondrial dehydrogenases in viable cells. 59 The following cell lines were used: HL60 human promyelocytic leukemia cell line, chronic myelogenous leukemia cell lines K562, AR23, and RWLeu4, and the KG1 progenitor leukemia cell line. The WST-1 tetrazolium salt (10 μl) was added to well culture plates containing 100 μl of cell suspension. The plates were evaluated after 4 h of incubation. Phytic Acid had a clear cytotoxic effect on all of the tested cell lines, with an IC50 of 5 mmol/l after 72 h of culture.
Phytic Acid extracted from rice bran induced marked growth inhibition in ovary, breast, and liver cancer cells, with 50% growth inhibition concentration (IC50) values of 3.45, 3.78, and 1.66 mM, respectively. 60 Cells of a normal cell line (BALB/c 3T3 cells) exhibited no increased sensitivity towards Phytic Acid.
Effect on Nutrient Absorption
Phytate (cation not declared; read-across for Sodium Phytate, Phytic Acid, and Phytin)
In a study involving 717 pregnant women in rural Bangladesh, the mean dietary intake of phytate was found to be ∼695.1 mg/day. 61 Phytate inhibited iron absorption from the diet in all of the women, inhibited calcium absorption in 52% of the women, and inhibited zinc absorption in 12% of the women.
Dermal Irritation and Sensitization Studies
Skin Irritation and Sensitization Studies of Polyol Phosphates.
Irritation
In Vitro
Sodium Phytate
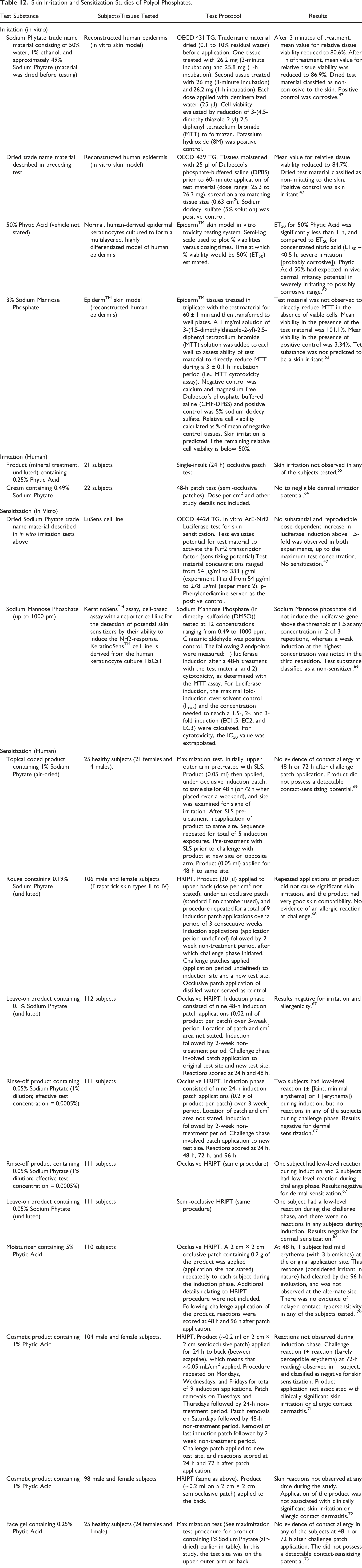
The skin corrosion potential of a Sodium Phytate trade name material consisting of 50% water, 1% ethanol, and approximately 49% Sodium Phytate was evaluated in an in vitro skin model (reconstructed human epidermis, EpiDermTM) test for skin corrosion. 47 The concentration of Sodium Phytate in the trade name material was not stated. Prior to testing, the trade name material was dried, yielding 0.1% to 10% residual water. After 3 minutes of treatment with the test material, the mean value of relative tissue viability was reduced to 80.6%, which is above the threshold for corrosion potential (50%). After 1 h of treatment, the mean value of relative tissue viability was reduced to 86.9%. The test material was classified as non-corrosive to the skin. Using the same skin model, the same test material was evaluated for skin irritation potential. At the end of the 60-minute application period, the mean value for relative tissue viability was reduced to 84.7%, above the threshold for skin irritation potential (50%). The test material was classified as non-irritating to the skin.
Phytic Acid
The skin irritation potential of 50% Phytic Acid (vehicle not stated) was evaluated using the EpiDermTM skin model in vitro toxicity testing system. 62 Phytic Acid (50%) elicited an ET50 that was significantly less than 1 h. The authors concluded that 50% Phytic Acid has an expected in vivo dermal irritancy potential in the severely irritating to possibly corrosive range.
Sodium Mannose Phosphate
The skin irritation potential of 3% Sodium Mannose Phosphate was evaluated using the EpidermTM skin model (reconstructed human epidermis). 63 EpidermTM tissues were treated in triplicate with the test material for 60 ± 1 min and then transferred to well plates. Test results indicated that the test substance was not predicted to be a skin irritant.
Human
Sodium Phytate
The skin irritation potential of a cream containing 0.49% Sodium Phytate was evaluated in a 48-h patch test (semi-occlusive patches) involving 22 subjects. 64 The dose per area and other study details are not included in this study summary. The conclusion for this study is stated as “no to negligible dermal irritation potential.”
Phytic Acid
A product (mineral treatment, undiluted) containing 0.25% Phytic Acid was evaluated for skin irritation potential in a single-insult (24 h) occlusive patch test involving 21 subjects. 65 Test results were negative.
Sensitization
In Vitro
Sodium Phytate
The skin sensitization potential of a dried Sodium Phytate trade name material (defined in skin irritation study on Sodium Phytate) was evaluated in the in vitro ArE-Nrf2 Luciferase test (Organization for Economic Co-operation and Development (OECD) 442d test guideline (TG), 2 experiments) for skin sensitization. 47 The dried test material was tested at concentrations ranging from 54 μg/ml to 333 μg/ml in the first experiment, and at concentrations ranging from 54 μg/ml to 278 μg/ml in the second experiment. It was concluded that the dried test material had no sensitization potential.
Sodium Mannose Phosphate
The sensitization potential of Sodium Mannose Phosphate was evaluated using the KeratinoSensTM assay. 66 Sodium Mannose Phosphate (in dimethyl sulfoxide (DMSO)) was tested at 12 concentrations ranging from 0.49 to 1000 ppm, and was classified as a non-sensitizer.
Human
Sodium Phytate
A rinse-off product containing 0.05% Sodium Phytate (1% dilution; effective test concentration = 0.0005%) produced negative results in an occlusive human repeated insult patch test (HRIPT) involving 111 subjects. 67 HRIPT results (using occlusive patches, unless otherwise stated) were also negative for another rinse-off product containing 0.05% Sodium Phytate (1% dilution; effective test concentration = 0.0005%) in a study involving 111 subjects. The following other negative HRIPT results for products containing Sodium Phytate have been reported: a leave-on product containing 0.05% Sodium Phytate (undiluted, semi-occlusive patches; 111 subjects), 67 a leave-on product containing 0.1% Sodium Phytate (undiluted, 112 subjects), 67 a rouge containing 0.19% Sodium Phytate (undiluted, 106 subjects), 68 and a topical coded product containing 1% Sodium Phytate (maximization test, 25 subjects). 69
Phytic Acid
A moisturizer containing 5% Phytic Acid was classified as a non-sensitizer in an HRIPT involving 110 subjects. 70 The skin irritation and sensitization potential of a cosmetic product containing 1% Phytic Acid was evaluated in an HRIPT using semi-occlusive patches involving 104 male and female subjects. 71 Application of the product was not associated with clinically significant skin irritation or allergic contact dermatitis. The same results were reported for another cosmetic product containing 1% Phytic Acid in an HRIPT (same procedure) involving 98 male and female subjects. 72 In a maximization test involving 25 subjects, a face gel containing 0.25% Phytic Acid produced negative results. 73
Photosensitization/Phototoxicity
A photosensitization HRIPT on a clear liquid containing 1% Sodium Phytate was performed using 25 subjects (21 females and 4 males). 74 During induction, the test substance (∼40 mg) was applied for 24 h, under an occlusive patch, to a 2 cm × 2 cm area on the lower back. After patch removal, the test site was irradiated with 3 minimal erythemal doses (MEDs) from a xenon arc solar simulator. This procedure was repeated for a total of 6 induction exposures over a 3-week period. The induction phase was followed by a 10- to 14-day non-treatment period. During the challenge phase, the test substance (∼40 mg) was applied, in duplicate, for 24 h to new sites (2 × 2 cm) on the opposite side of the lower back. The sites were then irradiated with ½ an MED +4 J/cm2 of long-wave ultraviolet light (UVA). Reactions were scored at 48 h and 72 h after irradiation. No reactions suggestive of photocontact allergy were observed in any of the subjects tested.
Ocular Irritation Studies
Ocular Irritation Studies.

In Vitro
Sodium Phytate
In the EpiOcularTM eye irritation test, negative results were reported for a cream containing 0.49% Sodium Phytate 64 and for a coded product containing 50% Sodium Phytate. 75 In a bovine corneal opacity and permeability (BCOP) test, results were negative for a dried Sodium Phytate (unknown concentration) trade name material and the same material at a concentration of 2% aqueous. 47 In the reconstructed human cornea-like epithelium (RhCE) test, the same dried Sodium Phytate trade name material was classified as non-irritating, 47 and a Sodium Phytate trade name material consisting of 50% water, 1% ethanol, and approximately 49% Sodium Phytate was classified as slightly irritating in the in vitro hen’s egg chorioallantoic membrane test (HET-CAM). 76
Phytic Acid
Phytic Acid (50%) (vehicle not stated) was evaluated for ocular irritation potential using the EpiOcularTM tissue model in vitro toxicity testing system. 77 The ET50 for Phytic Acid (50%) was ∼9 minutes (estimated Draize ocular irritation score of >25 (moderately irritating)).
Sodium Mannose Phosphate
The ocular irritation potential of 3% Sodium Mannose Phosphate was evaluated in the BCOP assay using excised corneas. 78 An aliquot (750 μl) of the test material was introduced into the anterior chamber of 5 corneas. The in vitro ocular irritation score was 0.
Clinical Studies
Other Clinical Reports
Phytate (cation not declared; read-across for Sodium Phytate, Phytic Acid, and Phytin)
Healthy women (15 young and 14 elderly) consumed low-phytate diets (young women: 682 mg phytate/day; elderly women: 782 mg phytate/day) or a high-phytate diet (young women: 1587 mg phytate/day; elderly women: 1723 mg phytate/day) for a period of 10 days. 32 No overt signs of toxicity were reported among the women in the study. In a similar study, healthy women (14 young and 14 elderly) consumed low-phytate diets (young women: 681 mg phytate/day; elderly women: 782 mg phytate/day) or a high-phytate diet (young women: 1584 mg phytate/day; elderly women: 1723 mg phytate/day) for a period of 10 days. Again, no overt signs of toxicity were reported for women in the study. 33
Summary
The safety of 10 polyol phosphates as used in cosmetics is reviewed in this safety assessment. According to the Dictionary, Sodium Phytate, Phytic Acid, and Trisodium Fructose Diphosphate are reported to function as chelating agents in cosmetic products. Sodium Phytate and Phytic Acid are also reported to function as oral care agents; and Trisodium Fructose Diphosphate and Manganese Fructose Diphosphate are reported to function as antioxidants in cosmetic products. The remaining ingredients have the skin conditioning agent function in common, except for Xylityl Phosphate, which is reported to function as an antiacne agent, antidandruff agent, deodorant agent, and exfoliant. Functioning as an antiacne or antidandruff agent is not a cosmetic use and, therefore, the Panel did not evaluate safety in relation to those uses.
An aqueous solution of Phytic Acid is obtained by acid hydrolysis of maize seed (kernels), rice bran, or rice husks (hulls). The production of Phytic Acid (50% solution) involves the addition of diluted sulfuric acid to defatted food-grade rice bran to dissociate phytate from iron and protein complexes. Sodium Mannose Phosphate is manufactured by enzymatic reaction from pyrophosphate and mannose.
The Food Chemicals Codex acceptance criteria for Phytic Acid solution (aqueous solution) include: arsenic (not more than 3 mg/kg), calcium (not more than 0.02%), chloride (not more than 0.02%), inorganic phosphorus (not more than 0.2%), lead (not more than 1 mg/kg) and sulfate (not more than 0.02%). The results of an impurities analysis on 50% Phytic Acid (vehicle not stated) indicated that the levels of heavy metals were lower than the detection level provided by the assay. Detection of a level of arsenic was not possible due to a problem with the assay that was described as strong interference of 50% Phytic Acid with the assay reagents. Possible impurities (0.1% to 0.5%) of Sodium Mannose Phosphate are: phosphate, sodium salt; pyrophosphate, sodium salt; sodium chloride; and magnesium and ammonium ions.
According to 2018 VCRP data, the greatest use frequency is reported for Sodium Phytate, which is reported to be used in 412 cosmetic products (259 leave-on, 146 rinse-off, and 7 diluted for bath use). The results of a concentration of use survey conducted in 2016-2017 indicate that Phytic Acid is being used at concentrations up to 2% in leave-on products (body and hand products [not spray]), which is the greatest use concentration that is being reported for the polyol phosphates reviewed in this safety assessment.
Following the topical treatment of Wistar rats with a cream supplemented with Sodium Phytate (up to 2%) or 2% Phytin, Phytic Acid was detected in the urine. Phytic Acid was also detected in the urine of human subjects on a Phytic Acid-poor diet after application of a moisturizing gel containing 4% potassium phytate.
Phytic Acid concentrations were detected in the brains of Wistar rats fed Phytic Acid in the diet for 12 weeks; concentrations detected in other organs were 10-fold less. When [14C]-Phytic Acid was administered orally to Sprague-Dawley rats, much of the radioactivity was distributed in the liver, kidneys, muscle, and skin at 24 h. Most of the radioactivity in the urine was due to the presence of inositol. In human subjects, 1% to 10% of administered Phytic Acid ingested was excreted in the urine. The feeding of Phytic Acid, Sodium Phytate, or Phytin in the diet resulted in a continuous increase in urinary levels of Phytic Acid until normal values were reached.
LD50 values of 480 mg/kg (females) and 400 to 500 mg/kg (males) were reported in an acute oral toxicity study involving F344 rats. In an acute oral toxicity study involving male and female Jcl:ICR mice, LD50 values of 1150 mg/kg (females) and 400 to 900 mg/kg (males) were reported.
There was no significant dose-response relationship regarding changes in organ weights and no statistically significant macroscopic findings in pregnant female JcI:ICR mice that received oral doses up to 315 mg/kg/day on gestation days 7 to 15. Groups of 10 male diabetic KK mice were fed dietary concentrations of 0.5% or 1% Sodium Phytate for 8 weeks. Concentrations of fasting and random blood glucose levels were statistically significantly lower (P < 0.05) only in the group fed 1% Sodium Phytate. Groups of 8 male Wistar rats were fed dietary concentrations of 0.1% to 1% Phytic Acid for 20 days. No effects on organ weight were noted, but the concentration of T3 in the serum was statistically significantly lower at all administered Phytic Acid concentrations. Dosing with Phytic Acid (2% in distilled drinking water) was well tolerated in female C7BL/6J mice treated for 70 days.
In a 12-week dose range-finding study, groups of 20 male and female F344 rats received Phytic Acid at concentrations up to 10% in drinking water. All rats that received 10% Phytic Acid and all males and 1 female that received 5% Phytic Acid died before the end of the experiment. There were no consistent differences in results for control vs test animals in a study in which 8 female Tg2576 mice (Alzheimer’s mouse model) and 10 C7BL/6J mice received Phytic Acid at a concentration of 2% in distilled water for 6 months.
Three different concentrations of 50% Phytic Acid solution (equivalent to doses of 80, 155, or 315 mg/kg/day) were administered orally to groups of 21 to 24 pregnant female JcI:ICR mice on gestation days 7 to 15. No significant effects on the incidence of external or skeletal malformations were observed at any dose of Phytic Acid. The treatment of groups of 30 male albino rats (Rattus norvegicus) with Phytic Acid had an ameliorative effect on the pathological and hormonal alterations induced by aflatoxin B1 injection.
In in vitro assays, Phytic Acid and Sodium Mannose Phosphate were non-genotoxic in the Ames test. Also, Phytic Acid was non-genotoxic in the L5178Y mouse lymphoma assay, but was genotoxic (at an unknown high concentration) in the chromosomal aberrations assay involving Chinese hamster ovary cells. Phytic Acid was also non-genotoxic in the in vivo micronucleus test involving bone marrow cells from mice that received four i.p. doses of 30 mg/kg or a single i.p. dose of 60 mg/kg.
The genotoxicity of a Sodium Phytate trade name material consisting of 50% water, 1% ethanol, and approximately 49% Sodium Phytate was evaluated in the Ames test using the following S. typhimurium strains: TA 97a, TA 98, TA 100, TA 102, and TA 1535. The test material, in deionized water, was evaluated at doses up to 4995 μg/plate with and without metabolic activation, and results were negative. A second experiment (pre-incubation method, modification of Ames test) was performed to confirm the results of the first. The test material was evaluated at doses up to 5013 μg/plate, with and without metabolic activation, and results were negative.
Renal papillomas (related to calcification and necrosis of renal papillae) were observed in a very small number of male and female F344 rats in groups of 120 animals treated orally with 1.25% or 2.5% Phytic Acid in drinking water. The organ distribution of other tumor types did not differ significantly from those known to occur in F344 rats. Sodium Phytate (2% in the diet) was classified as a promoter of urinary bladder carcinogenesis. The results of animal studies indicate that Phytic Acid is anti-photocarcinogenic (2% in drinking water [mice]) as well as anti-carcinogenic (doses up to 5 mg applied to skin [mice]; 4% in cream applied to skin [mice]; 2% in drinking water [mice]); 2% in diet [rats]), and that Sodium Phytate is anti-carcinogenic (up to 1% in drinking water [rats]). Anti-inflammatory activity (oral dose of 150 mg/kg in rats) and cytotoxicity (IC50 = 5 mmol/l, leukemia cell lines) have also been associated with Phytic Acid treatment.
A Sodium Phytate trade name material consisting of 50% water, 1% ethanol, and approximately 49% Sodium Phytate was evaluated in an in vitro skin model (reconstructed human epidermis, EpiDermTM) to determine its skin irritation and corrosive potential. Results were classified as negative for skin irritation and corrosion. Sodium Mannose Phosphate (3%) also was not predicted to be a skin irritant using the same model. Based on results from the EpiDermTM skin model in vitro toxicity testing system, Phytic Acid (50%) (vehicle not stated) has an expected in vivo dermal irritancy potential in the severely irritating to possibly corrosive range.
A cream containing 0.49% Sodium Phytate was classified as having no to negligible irritation potential in a 48-h semi-occlusive patch test involving 22 subjects. A product (mineral treatment, undiluted) containing 0.25% Phytic Acid was evaluated for skin irritation potential in a single-insult (24 h) occlusive patch test involving 21 subjects. Test results were negative.
The skin sensitization potential of a dried Sodium Phytate (concentration not stated) trade name material was evaluated in the in vitro ArE-Nrf2 Luciferase test. The test material was evaluated at concentrations ranging from 54 μg/ml to 333 μg/ml; the test material was classified as having no sensitizing potential. The sensitization potential of Sodium Mannose Phosphate (in DMSO) was evaluated at 12 concentrations (ranging from 0.49 to 1000 ppm) using the KeratinoSensTM assay. The test substance was classified as a non-sensitizer.
A topical coded product containing 1% Sodium Phytate did not cause skin sensitization in a maximization test involving 25 subjects, and a rouge containing 0.19% Sodium Phytate did not cause irritation or sensitization in an HRIPT involving 106 subjects. A leave-on product containing 0.1% Sodium Phytate (undiluted) was negative for irritation and allergenicity in an occlusive HRIPT involving 112 subjects. Two rinse-off products, each containing 0.05% Sodium Phytate (1% dilution; effective test concentration = 0.0005%) were evaluated in occlusive HRIPTs involving 111 subjects. Both products were classified as non-sensitizers. In another study, a leave-on product containing 0.05% Sodium Phytate (undiluted) was evaluated in a semi-occlusive HRIPT involving 111 subjects. The product did not induce dermal sensitization. There was no evidence of delayed contact hypersensitivity in the 110 subjects evaluated in an HRIPT on a moisturizer containing 5% Phytic Acid. The application of cosmetic products containing 1% Phytic Acid was not associated with clinically significant skin irritation or allergic contact dermatitis in a semi-occlusive HRIPTs involving 98 and 104 subjects. A face gel containing 0.25% Phytic Acid did not induce skin sensitization in groups of 25 subjects in maximization tests.
A clear liquid containing 1% Sodium Phytate did not induce photosensitization in a study involving 25 subjects.
A cream containing 0.49% Sodium Phytate was classified as having no ocular irritation potential in the in vitro EpiOcularTM eye irritation test. A product containing 50% Sodium Phytate was classified as a minimal to non-irritant and Phytic Acid (50%) was classified as moderately irritating in this test. The ocular irritation potential of a Sodium Phytate (concentration not stated) trade name material was also evaluated in the following in vitro assays: BCOP test, RhCE test, and HET-CAM assay. Test results indicated that the trade name material was non-irritating/non-corrosive to slightly irritating. Sodium Mannose Phosphate (3%) was a non-irritant in the in vitro BCOP assay using excised corneas.
A clinical study evaluated the effect of phytates in the diet. No overt signs of toxicity were reported when healthy women consumed a low-phytate diet (682 mg phytate/day) or a high-phytate diet (1723 mg phytate/day) for a period of 10 days.
Discussion
The Panel determined that the data were sufficient to conclude on the safety of four polyol phosphates, but additional data are needed for completion of the safety assessment of the following six polyol phosphates: Disodium Glucose Phosphate, Manganese Fructose Diphosphate, Sodium Mannose Phosphate, Trisodium Fructose Diphosphate, Xylityl Phosphate, and Zinc Fructose Diphosphate. Of these six ingredients, only Sodium Mannose Phosphate is reported to be in use. The complete list of data needs includes: • Method of manufacture (not needed for Sodium Mannose Phosphate) • Impurities (not needed for Sodium Mannose Phosphate) • ADME data
While method of manufacture and impurities data on Sodium Mannose Phosphate were received, no ADME data were submitted. The Panel agreed that absorption data on this ingredient are needed to conclude on safety. Additionally, the Panel previously requested skin sensitization data (animal or human) on Phytic Acid at the highest maximum use concentration of 2% or on a cosmetic product containing 2% Phytic Acid. A negative human maximization test on a product containing 1% Sodium Phytate, negative HRIPT data on products containing Sodium Phytate (up to 0.1%) and on a moisturizer containing 5% Phytic Acid (highest ingredient concentration tested), and negative human photosensitization data on a clear liquid containing 1% Sodium Phytate were among the data that were received in response to this request. The Panel agreed that the results of these studies indicate that these ingredients do not have discernible skin sensitization potential at cosmetic use concentrations.
The Panel discussed the issue of incidental inhalation exposure from perfumes. Sodium Phytate is reportedly used in a perfume formulation, which may result in incidental inhalation exposure. The Panel noted that most of the droplets/particles produced in cosmetic aerosols would not be respirable (would not enter the lungs) to any appreciable amount. However, the potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Conclusion

*Not reported to be in current use. Were the ingredients in this group not in current use to be used in the future, the expectation is that it would be used in product categories and at concentrations comparable to others in this group.

**Not reported to be in use.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 555 13th St., NW, Suite 300W, Washington, DC 20004, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.