Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of 10 alkanoyl lactyl lactate salts. These ingredients have the surfactant function in cosmetics in common. The Panel reviewed data relevant to the safety of these ingredients, and concluded that these 10 ingredients are safe in cosmetics in the present practices of use and concentration described in the safety assessment when formulated to be nonirritating and nonsensitizing, which may be based on a quantitative risk assessment (QRA) or other accepted methodologies.
Keywords
Introduction
Definitions, Idealized Structures, and Functions of the Ingredients in this Safety Assessment.1,CIR Staff
This safety assessment includes relevant published and unpublished data for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A list of the typical search engines and websites used, sources explored, and endpoints that the Panel evaluates, is available on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Chemistry
Definition and General Characterization
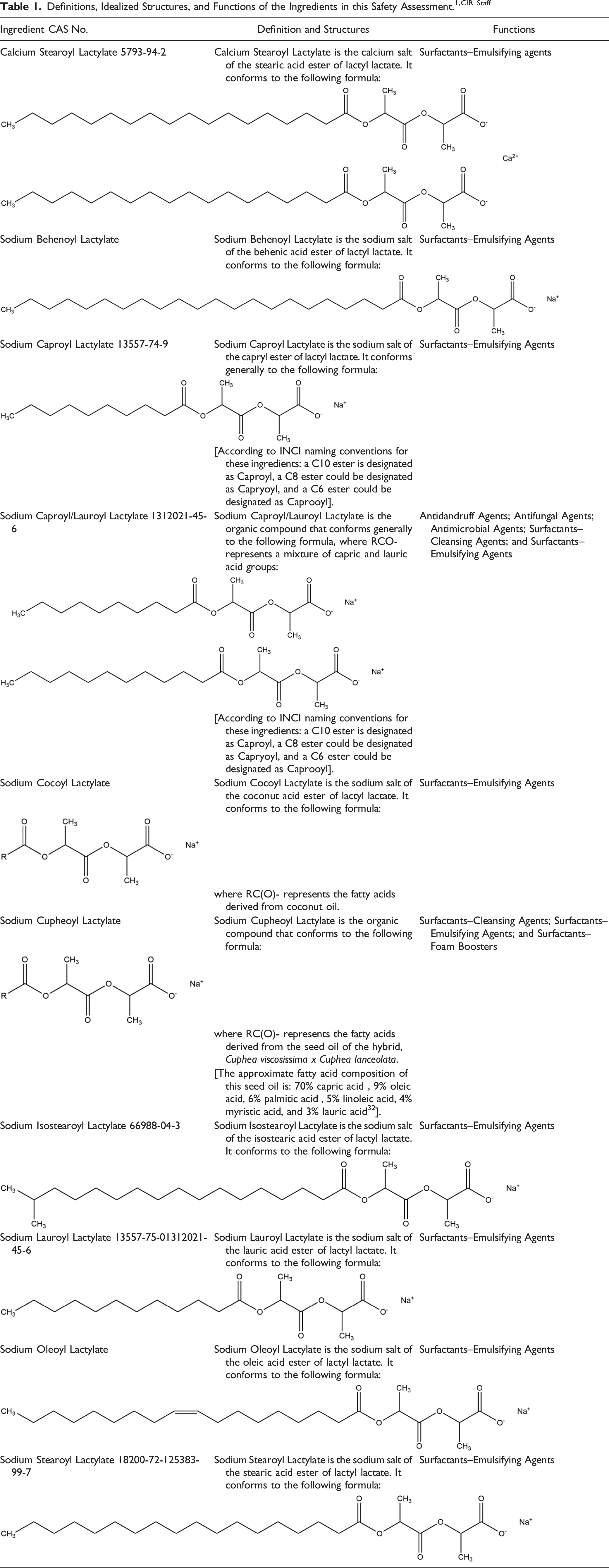
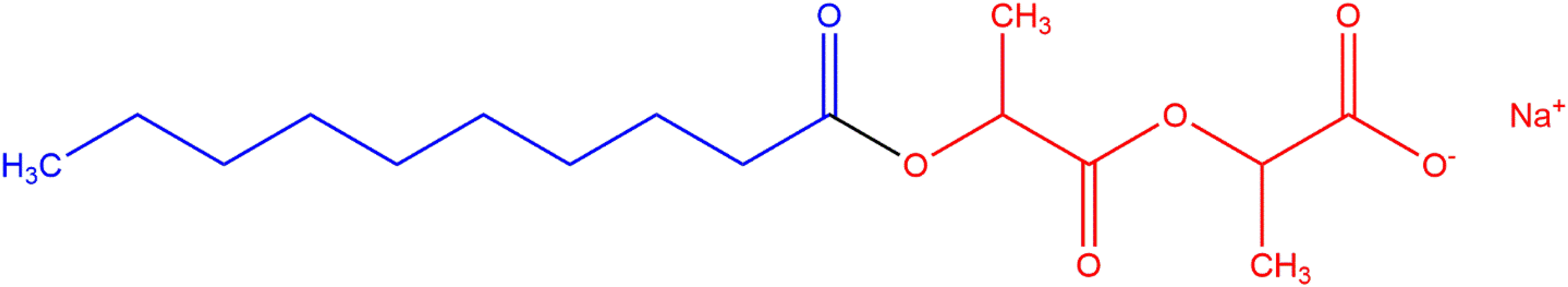
Alkanoyl lactyl lactates (i.e., alkanoyl lactylates) are the carboxylic acid salts of diesters that are formed between a fatty acid group and two equivalents of lactic acid.
2
The generic structure of alkanoyl lactyl lactates is presented below (Figures 1 and 2). Like other anionic emulsifiers/surfactants, the properties of these ingredients result from the diametrically opposed lipophilic (fatty acid) tail and the hydrophilic (lactylate) head. The definitions and structures of the alkanoyl lactyl lactate salts are presented in Table 1.
1
Generic structure of alkanoyl lactyl lactates. Example structure of an alkanoyl lactyl lactate, Sodium Caproyl Lactylate.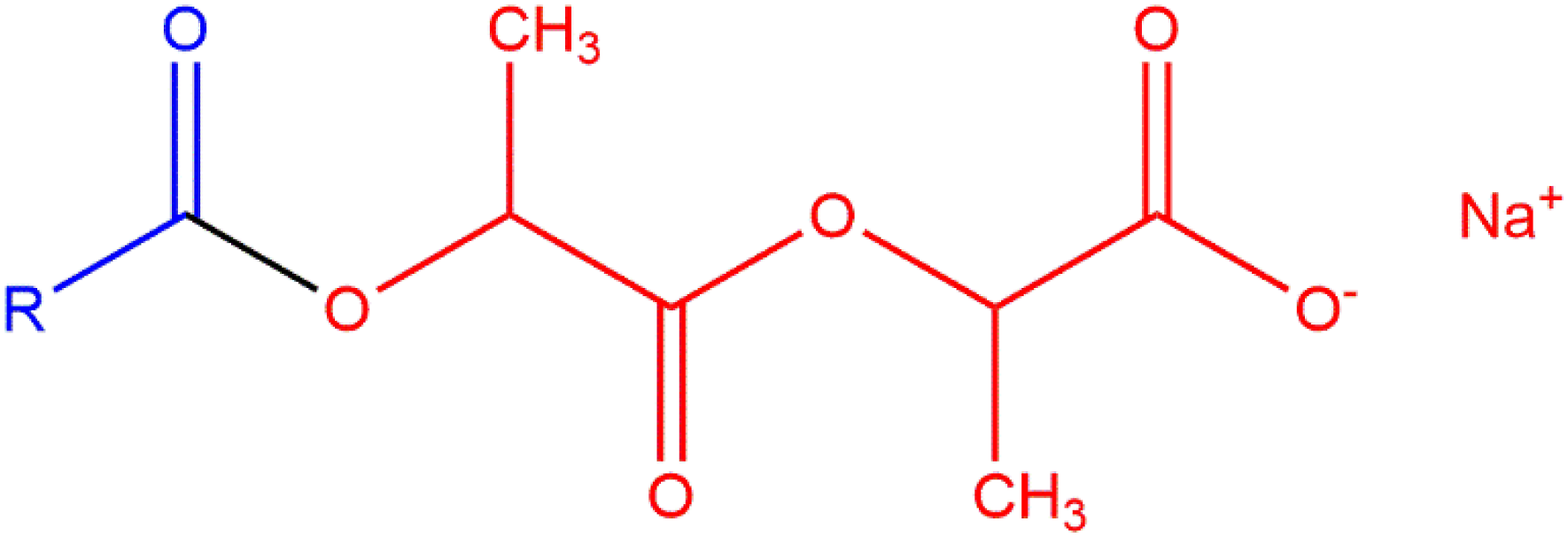
Chemical and Physical Properties
Chemical and Physical Properties of Alkanoyl Lactyl Lactate Salts.

Method of Manufacture
Calcium Stearoyl Lactylate
According to one food additive supplier, Calcium Stearoyl Lactylate (defined as a mixture of the calcium salts of stearoyl lactylic acids and its polymers and minor amounts of calcium salts of other related acids) is manufactured by base-catalyzed esterification of lactylic acid and commercial stearic acid. 4 However, the Dictionary describes Calcium Stearoyl Lactylate as the calcium salt of the stearic acid ester of lactyl lactate (i.e., no indication of polymers or other acids provided).
Sodium Isostearoyl Lactylate
According to one method, Sodium Isostearoyl Lactylate is the reaction product of isostearic acid with lactic acid in the presence of sodium hydroxide. 5
Sodium Stearoyl Lactylate
According to one food additive supplier, Sodium Stearoyl Lactylate (defined as a mixture of the sodium salts of stearoyl lactylic acids and its polymers and minor amounts of sodium salts of other related acids) is manufactured by base-catalyzed esterification of lactic acid and commercial stearic acid. 4 However, the Dictionary describes Sodium Stearoyl Lactylate as the sodium salt of the stearic acid ester of lactyl lactate (i.e., no indication of polymers or other acids).
Composition
Sodium Stearoyl Lactylate and Calcium Stearoyl Lactylate
As noted above, when used as food additives, Sodium Stearoyl Lactylate and Calcium Stearoyl Lactylate are manufactured using a base-catalyzed esterification process. 4 The distribution of the components in each is dependent upon the relative proportion of lactylic acid, fatty acid and the amount of sodium/calcium salt that is used in the neutralization process. The typical composition of the product of the neutralization process is approximately 50% stearoyl-mono-lactylate, 20% stearoyl-di-lactylate (equivalent to structure of Sodium Stearoyl Lactylate or Calcium Stearoyl Lactylate), 5% stearoyl-tri-lactylate, and trace amounts of stearoyl-tetra-lactylate. Other components may include sodium/calcium salts of fatty acids (depending on the ingredient, i.e., if the ingredient is Sodium Stearoyl Lactylate or Calcium Stearoyl Lactylate) or free fatty acids (15–20%), non-neutralized stearoyl lactylic acid, sodium/calcium lactate, and free lactic acid or polymers of lactic acid. Additionally, the actual fatty acid profile of Sodium Stearoyl Lactylate and Calcium Stearoyl Lactylate will depend upon the source of the fatty acids. However, the Dictionary describes Sodium Stearoyl Lactylate and Calcium Stearoyl Lactylate as the salts of the stearic acid ester of lactyl lactate (i.e., no indication of stearoyl-mono-lactylate, stearoyl-tri-lactylate, or stearoyl-tetra-lactylate).
Impurities
Calcium Stearoyl Lactylate
The Food Chemicals Codex specifications for Calcium Stearoyl Lactylate are as follows: lead (not more than 2 mg/kg), acid value (between 50 and 86), ester value (between 125 and 164), calcium content (between 4.2% and 5.2%), and total lactic acid (between 32% and 38%). 6 According to European Commission regulations, specifications relating to the purity of Calcium Stearoyl Lactylate are as follows: calcium (not less than 1% and not more than 5.2%), ester value (not less than 125 and not more than 190), acid value (not less than 50 and not more than 130), total lactic acid (not less than 15% and not more than 40%), arsenic (not more than 3 mg/kg), lead (not more than 2 mg/kg), mercury (not more than 1 mg/kg), and cadmium (not more than 1 mg/kg). 7
Sodium Stearoyl Lactylate
The Food Chemicals Codex specifications for Sodium Stearoyl Lactylate are as follows: lead (not more than 2 mg/kg), acid value (between 60 and 80), ester value (between 120 and 190), sodium content (between 3.5% and 5%), and total lactic acid (between 23% and 34%). 6 According to European Commission regulations, the following specifications relate to the purity of Sodium Stearoyl Lactylate: sodium (not less than 2.5% and not more than 5%), ester value (not less than 90 and not more than 190), acid value (not less than 60 and not more than 130), total lactic acid (not less than 15% and not more than 40%), arsenic (not more than 3 mg/kg), lead (not more than 2 mg/kg), mercury (not more than 1 mg/kg), and cadmium (not more than 1 mg/kg). 4
Use
Cosmetic
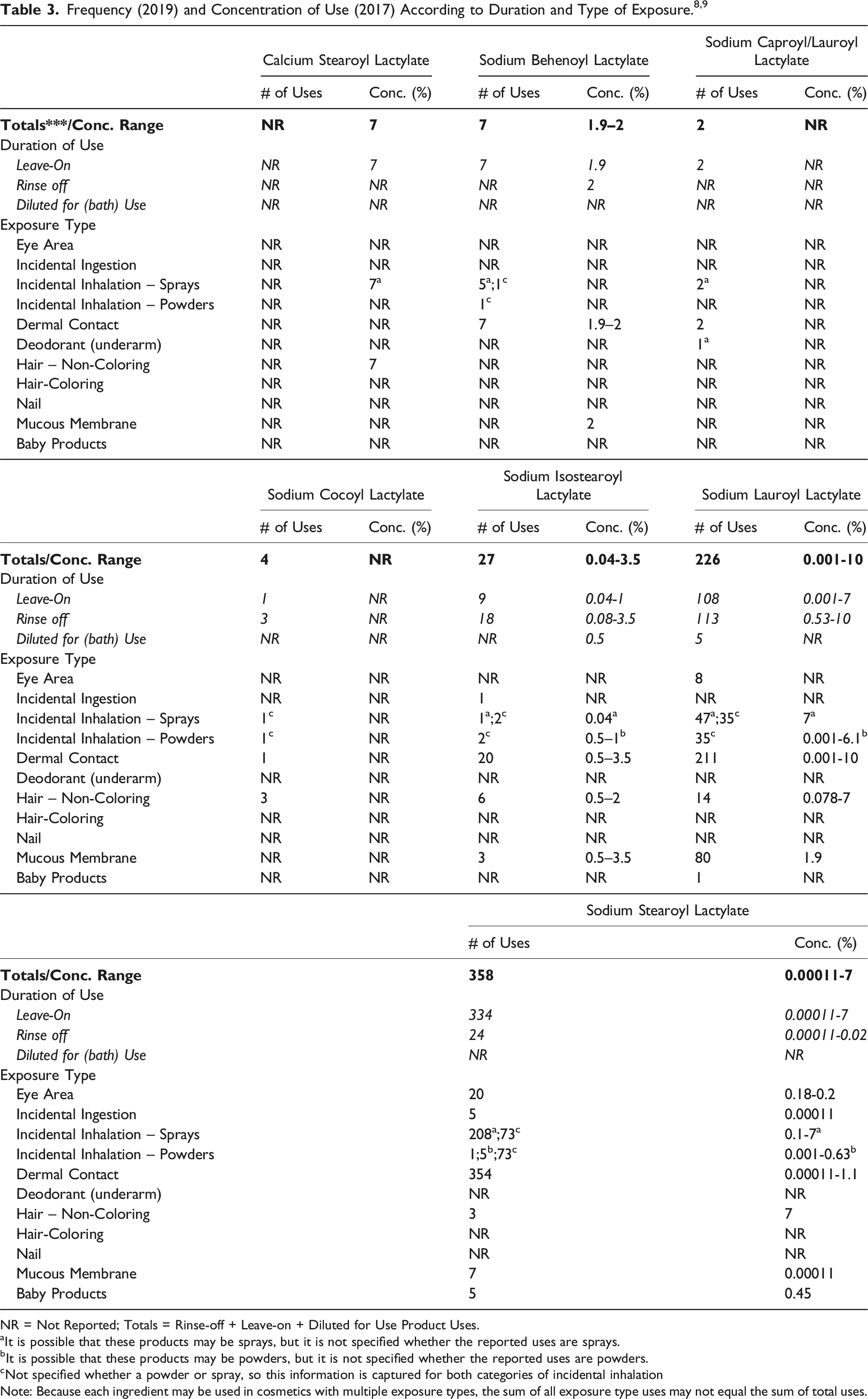
The safety of the alkanoyl lactyl lactate salts is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. 8 Use concentration data are submitted by the cosmetics industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category. 9
NR = Not Reported; Totals = Rinse-off + Leave-on + Diluted for Use Product Uses.
aIt is possible that these products may be sprays, but it is not specified whether the reported uses are sprays.
bIt is possible that these products may be powders, but it is not specified whether the reported uses are powders.
cNot specified whether a powder or spray, so this information is captured for both categories of incidental inhalation
Note: Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum of total uses.
Cosmetic products containing alkanoyl lactyl lactate salts may be applied to the skin or, incidentally, may come in contact with the eyes (at maximum use concentrations up to 0.2%, for Sodium Stearoyl Lactylate in eye lotions). Similarly, products containing these ingredients may incidentally come in contact with mucous membranes (at maximum use concentrations up to 3.5%, for Sodium Isostearoyl Lactylate in bath soaps and detergents). The highest maximum use concentration of alkanoyl lactyl lactate salts in products that may be incidentally ingested is 0.00011% (for Sodium Stearoyl Lactylate in lipsticks). Products containing alkanoyl lactyl lactate salts may be applied as frequently as several times per day and may come in contact with the skin for variable periods following application. Daily or occasional use may extend over many years.
The alkanoyl lactyl lactate salts reviewed in this safety assessment are not included on the European Union’s list of substances that are restricted or list of substances that are prohibited in cosmetic products. 10
Non-Cosmetic
Calcium Stearoyl Lactylate and Sodium Stearoyl Lactylate
Both Calcium Stearoyl Lactylate and Sodium Stearoyl Lactylate have been known to have a dough strengthening effect in the process of bread making (high-protein breads). 11 Both salts can form a complex with gluten to stabilize the gluten-network in dough. It has been noted that the dough strengthening effect of these salts may be due to formation of this complex.
The US FDA has determined that Calcium Stearoyl Lactylate (defined as a mixture of calcium salts of stearoyl lactylic acid and minor proportions of other calcium salts of related acids) may be used safely as a direct food additive, provided that the specifications defined in the Food Chemicals Codex are met [21 CFR 172.844]. Furthermore, the FDA has established limits for this ingredient in food ranging from 0.05% to 0.5%, depending on the food product type. The FDA has also established a limit of 0.5 parts for each 100 parts by weight of flour for Calcium Stearoyl Lactylate in yeast-leavened bakery products and prepared mixes for yeast-leavened bakery products.
The FDA has also determined that Sodium Stearoyl Lactylate (defined as a mixture of sodium salts of stearoyl lactylic acids and minor proportions of sodium salts of related acids) may be used safely as a direct food additive, provided that it meets the specifications of the Food Chemicals Codex [21 CFR 172.846]. (The Food Chemicals Codex specifications for Sodium Stearoyl Lactylate are stated in the section on Impurities). Furthermore, the FDA has established limits for this ingredient in food ranging from 0.2% to 0.5%, depending on the food product type. The FDA has also established a limit of 0.5 parts for each 100 parts by weight of flour for Sodium Stearoyl Lactylate in baked products, pancakes, and waffles.
Following a request by the European Commission, the Panel of Food Additives and Nutrient Sources added to Food (ANS) was asked to issue a scientific opinion on the safety of Sodium Stearoyl Lactylate and Calcium Stearoyl Lactylate when used as food additives. 4 The Panel concluded that based on the no-observed-adverse-effect-level (NOAEL) of 2200 mg/kg body weight/day Sodium Stearoyl Lactylate that was derived from a 1-year oral toxicity study in rats and an uncertainty factor of 100, an acceptable daily intake (ADI) of 22 mg/kg body weight/day for Sodium Stearoyl Lactylate and Calcium Stearoyl Lactylate, either singly or in combination, can be established. The 1-year oral study is summarized in the section on Chronic Toxicity.
Toxicokinetic Studies
Dermal Penetration
Data on the dermal penetration of the alkanoyl lactyl lactate salts reviewed in this safety assessment were not found in the published literature, nor were these data submitted.
Absorption, Distribution, Metabolism, and Excretion
In Vitro
Calcium Stearoyl Lactylate
The hydrolysis of [14C]-Calcium Stearoyl Lactylate, radiolabeled at the lactylate moiety, was demonstrated in vitro using liver homogenates from rats, mice, and guinea pigs. 12 [14C]-Calcium Stearoyl Lactylate was rapidly hydrolyzed to lactic acid and stearic acid. Hydrolysis was also demonstrated using whole blood from rats and mice, but no significant hydrolysis of Calcium Stearoyl Lactylate was detected using human blood. Also, in a single sample of human duodenal mucosa, [14C]-Calcium Stearoyl Lactylate was rapidly hydrolyzed to stearic acid and lactic acid.
Animal
Oral: Calcium Stearoyl Lactylate
The absorption, metabolism, tissue distribution, and excretion of Calcium Stearoyl Lactylate was studied using groups of 4 male Tuck TO mice and groups of 4 male Dunkin–Hartley guinea pigs. 12 A single oral dose of an aqueous suspension of [14C]-Calcium Stearoyl Lactylate (90 or 900 mg/kg body weight) was administered by gavage. Radioactivity was determined in exhaled air, urine, feces, liver, kidneys, heart, lungs, spleen, testes, and in the gastrointestinal tract. Following oral administration, rapid absorption of radioactivity from the gastrointestinal tract was observed in mice as well as in guinea pigs. More than 50% of the applied radioactivity was exhaled as 14CO2 within 7 h. In both species, ∼80% of the applied dose was exhaled as 14CO2 within 48 h. Most of the remaining radioactivity was excreted in the urine within 24 h after dosing. Only minor amounts were detected in the feces of both species. No relevant differences were detected between the 90 and 900 mg/kg doses of [14C]-Calcium Stearoyl Lactylate. Approximately 2 % (in mice) or 6 % (in guinea pigs) of the administered dose remained in the tissues, mainly in the liver and gastrointestinal tract. Only traces of radioactivity were found in other organs (kidneys, lungs, testes, spleen, and heart). Thin layer chromatography of the urine of mice and guinea pigs indicated that lactic acid is a metabolite of Calcium Stearoyl Lactylate. Furthermore, the authors suggested that the additional radioactivity in the urine of treated animals is lactylate (i.e., without the stearic acid residue).
Toxicological Studies
Acute Toxicity Studies
Oral
Calcium Stearoyl Lactylate
In an acute oral toxicity study involving male rats (strain not stated), an oral LD50 of 25 g/kg body weight was reported for Calcium Stearoyl Lactylate. 2 Details relating to the test protocol were not included.
Sodium Isostearoyl Lactylate
The acute oral toxicity of Sodium Isostearoyl Lactylate was evaluated using white rats (number not stated). 5 Administration of a single oral dose was followed by a 14-day observation period. An LD50 of > 6.1 g/kg was reported.
Sodium Lauroyl Lactylate
In an acute oral toxicity study in which male rats (number not stated) were dosed orally with Sodium Lauroyl Lactylate, the LD50 was 6.81 g/kg. 2 The test protocol was not stated.
The acute oral toxicity of Sodium Lauroyl Lactylate was also evaluated using male and female rats (numbers and strains not stated). 13 The doses administered orally (dosing method not stated) ranged from 2.4 g/kg to 6 g/kg. Additional details were not included. The oral LD50 was estimated to be 4.88 g/kg.
Sodium Stearoyl Lactylate
In an acute oral toxicity study involving male rats (strain not stated), an oral LD50 of 25 g/kg body weight was reported for Sodium Stearoyl Lactylate. 2 Details relating to the test protocol were not included.
Short-Term, Subchronic, and Chronic Toxicity Studies
Short-Term, Subchronic, and Chronic Toxicity Studies.

Sodium Caproyl/Lauroyl Lactylate (25 μl, in acetone-olive oil (AOO) vehicle) was applied to the ears of 4 mice of the CRL:NMRI BR strain at a concentration of 25% or 50% on 3 consecutive days. 13 There was no evidence of systemic toxicity. (Appropriately, results from this local lymph node assay (LLNA) are presented in the section on Sensitization). Calcium Stearoyl Lactylate was evaluated in short-term oral toxicity studies at dietary concentrations ranging from 0.5% to 12.5%. 14 Groups of up to 32 rats were tested. Increased liver weight was observed at concentrations of 2% and 12.5%, but not at 0.5% (43-day feeding study), 5% and 7.5% in the diet (1-month feeding study), and at 5% in the diet (4-week feeding study). However, in other feeding studies at 5% in the diet (27 days, 32 days, and duration unspecified), liver weight/histology was normal. Kidney histology was normal in a feeding study on 0.5% Calcium Stearoyl Lactylate in which the duration was not specified. In short-term feeding studies on Sodium Stearoyl Lactylate, a transient increase in liver weight was observed in 20 rats fed 5% in the diet for 28 days, and organ weights were normal in a dog fed up to 15% in the diet for 1 month.
The subchronic oral toxicity of Calcium Stearoyl Lactylate was evaluated in a study in which groups of 10 male and 10 female rats were fed dietary concentrations of 0.5%, 5%, and 12.5% for 98 days. 14 There was no evidence of histological abnormalities in internal organs, but lipogranulomata in adipose tissue were detected at the 12.5% concentration. Relative weights of the liver, spleen, and brain were also increased after feeding with 12.5% Calcium Stearoyl Lactylate. The Joint FAO/WHO Expert Committee on Food Additives noted that the appearance of lipogranulomata and increased liver weight are related to excessive intake of abnormal proportions of long-chain fatty acids. Groups of 10 male and 10 female rats (strain not stated) were fed Sodium Stearoyl Lactylate in the diet at concentrations of 0.5%, 5%, and 12.5% for 102 days. The results of gross and histopathological evaluations were normal.
In a chronic study, groups of 5 rats were maintained on diets containing 8 to 22% Calcium Stearoyl Lactylate for periods of up to 6 months. 14 Mortality was high (number of deaths not reported) at concentrations of ≥ 20%. Relative liver weights were normal at a saturated to unsaturated (S:U) fatty acid ratio of 0.6, but increased with higher ratios in the absence of histopathological abnormalities. When the experiment was repeated using 40 male and 40 female rats fed 25% Calcium Stearoyl Lactylate in the diet, all of the animals developed severe lipogranulomata. In a 1-year chronic oral toxicity study performed in accordance with Organization for Economic Co-operation and Development (OECD) Test Guideline (TG) 452, groups of 60 Wistar rats were fed a diet that yielded doses of Sodium Stearoyl Lactylate up to 2214 mg/kg/day (males) and 2641 mg/kg/day (females). 15 No treatment-related toxic effects were observed, and NOAELs of 2214 mg/kg/day and 2641 mg/kg/day were reported for males and females, respectively. Results relating to tumorigenicity in this study are included in the section on Carcinogenicity.
There were no test substance-related gross or microscopic changes in 4 Beagle dogs fed a diet containing 7.5% Calcium Stearoyl Lactylate for 2 years. 14
Developmental and Reproductive Toxicity
Data on the developmental and reproductive toxicity of the alkanoyl lactyl lactate salts reviewed in this safety assessment were not found in the published literature, and unpublished data were not submitted.
Genotoxicity Studies
In Vitro
Calcium Stearoyl Lactylate
The genotoxicity of Calcium Stearoyl Lactylate (in benzene), with and without metabolic activation, was evaluated in the Ames test using the following Salmonella typhimurium strains: TA92, TA94, TA98, TA100, TA1535, and TA1537. 16 Doses of the test substance at up to 300 μg/plate were tested. Cytotoxicity data on the test substance were not included. Calcium Stearoyl Lactylate was non-genotoxic with and without metabolic activation.
In a chromosome aberration test involving a Chinese hamster fibroblast cell line, the genotoxicity of Calcium Stearoyl Lactylate (in ethanol) was evaluated at concentrations up to 63 µg/ml (highest non-cytotoxic dose) without metabolic activation. 16 One hundred metaphases per concentration were analyzed for polyploid cells and structural chromosomal aberrations. Chromosome and chromatid gaps were included in the evaluation. Calcium Stearoyl Lactylate did not cause polyploidy or clastogenic effects.
Sodium Caproyl/Lauroyl Lactylate
The genotoxicity of Sodium Caproyl/Lauroyl Lactylate (in dimethyl sulfoxide (DMSO)) was evaluated in the Ames test using the following S. typhimurium strains, with and without metabolic activation: TA97a, TA98, TA100, TA102, and TA1535. 13 The test substance was tested at doses up to 502 µg/plate, considering that doses of 1500 and 5000 µg/plate were cytotoxic. Water and DMSO served as the negative and solvent controls, respectively. The positive controls were sodium azide, benzo[a]pyrene, 4-nitro-O-phenylenediamine, and 2-aminoanthracene. Sodium Caproyl/Lauroyl Lactylate was non-genotoxic in all of the bacterial strains that were tested. The positive controls were genotoxic.
In Vivo
In vivo genotoxicity data on the alkanoyl lactyl lactate salts reviewed in this safety assessment were not found in the published literature, and unpublished data were not submitted.
Carcinogenicity Studies
Sodium Stearoyl Lactylate
As described earlier, a 1-year chronic oral toxicity study was performed in accordance with OECD TG 452. 15 Three groups of 60 Wistar rats (30 males and 30 females per group) were fed a diet that yielded mean daily Sodium Stearoyl Lactylate intakes of 558, 1115, and 2214 mg/kg/day (males) and 670, 1339, and 2641 mg/kg/day (females); the corresponding diet concentrations were 1.25%, 2.5%, and 5%, respectively. The negative control group was fed diet only. At histopathological examination, the incidence of endometrial stromal polyps in the uterus was reported as follows: 1 control female rat, 2 female rats fed 1.25% Sodium Stearoyl Lactylate in the diet, 6 female rats fed 2.5% in the diet, and 6 female rats fed 5% in the diet. However, these data lack statistical significance, and there is an absence of biological evidence to suggest a mechanism for the slightly higher incidence in the groups fed 2.5% and 5%. Furthermore, a comparison of these data with historical incidences of this tumor type (up to 10% in control rats of 1-year studies in the laboratory conducting this study) demonstrated that endometrial stromal polyps are common in rats of this strain and age. It was concluded that the endometrial polyps observed in females fed Sodium Stearoyl Lactylate in the diet were not treatment-related.
Other Relevant Studies
Protein Binding
Sodium Stearoyl Lactylate
Sodium Stearoyl Lactylate was mixed with gluten (protein in wheat, barley, and rye) in the presence of water. 17 Approximately 49% of the Sodium Stearoyl Lactylate remained bound until it was released by protease digestion of the protein. Details relating to the protocol for this experiment were not included. However, using a Tissue Metabolism Simulator Skin Sensitization model (TIMES-SS), it was determined that Sodium Lauroyl Lactylate is a non-binder to skin proteins, despite being a weak sensitizer in the LLNA (see Sensitization section). 18 TIMES-SS is defined as an expert system describing structure-toxicity and structure–metabolism relationships through a number of transformations simulating skin metabolism and interaction of the generated reactive metabolites with skin proteins.
Dermal Irritation and Sensitization Studies
Skin Irritation and Sensitization Studies on Alkanoyl Lactyl Lactate Salts.

Irritation
Sodium Caproyl/Lauroyl Lactylate was considered noncorrosive when the irritation/corrosive potential of this ingredient was evaluated using a tissue model that consisted of human-derived epidermal keratinocytes (EpiDerm® tissue model). 13 The skin irritation potential of alkanoyl lactyl lactate salts has been evaluated in the following experiments involving albino rabbits: 2 undiluted Calcium Stearoyl Stearate (nonirritating), 10% Sodium Lauroyl Lactylate (nonirritating), and undiluted Sodium Stearoyl Lactylate (nonirritating; primary irritation index (PII) = 0.5 in 2 studies).2,19 Sodium Caproyl/Lauroyl Lactylate (in AOO vehicle) caused erythema and an increase in ear thickness in 4 mice (CRL:NMRI BR strain) when tested at concentrations of 25% and 50%. 13 In a skin irritation test on Sodium Isostearyl Lactylate involving 6 albino rabbits, the PII was 7.17 (severe irritation) for the undiluted ingredient and 1.13 (slight irritation) for 15% Sodium Isostearyl Lactylate. 5
The skin irritation potential of Sodium Stearoyl Lactylate was evaluated using 51 subjects. 20 Twenty-five and 26 subjects were patch tested with 2% and 5% Sodium Stearoyl Lactylate in petrolatum, respectively. Details relating to the test protocol were not included. Sodium Stearoyl Lactylate was classified as having skin irritation potential. A diluted hair styling product (Calcium Stearoyl Lactylate effective concentration = 2.5%) was classified as a skin irritant in a study involving 54 subjects. 21 In 4 separate skin irritation studies, each involving 50 subjects, a hair molding cream containing 7% Calcium Stearoyl Lactylate was classified as nonirritating to the skin.22-25
Sensitization
The skin toxicity of Sodium Stearoyl Lactylate was investigated using reconstructed human epidermis (RHE) and detection of the inflammation markers interleukin (IL)-1α and IL-8. 26 Sodium Stearoyl Lactylate was predicted to be an allergen based on the results of this assay. The LLNA was used to evaluate the sensitization potential of Sodium Caproyl/Lauroyl Lactylate and Sodium Lauroyl Lactylate using the following test concentrations (in AOO vehicle): 2.5%, 5%, 10%, 25%, or 50%. 13 Sodium Caproyl/Lauroyl Lactylate was classified as a weak-moderate sensitizer (EC3 = 12.4%; EC3 = 9.3%), 13 and Sodium Lauroyl Lactylate was classified as a weak sensitizer (EC3 = 15%).27,28 An EC3 of 15% was also reported for a Sodium Lauroyl Lactylate trade name material in the LLNA. 29 Sodium Lauroyl Lactylate (challenge concentration = 0.5%; injection and dermal induction doses not stated) was classified as a weak sensitizer in the guinea pig maximization test (10 guinea pigs). 27 A silicone antifoam emulsion containing Sodium Stearoyl Lactylate (75% dilution; effective test concentration = 1.5 %) Sodium Stearoyl Lactylate) was a nonsensitizer in guinea pigs. 30
Computational Analyses/Predictions
The modeling of skin sensitization data on a number of diverse compounds, including data on Sodium Stearoyl Lactylate, and calculated descriptors was performed to develop multiple predictive classification models. 31 The following 2 automated procedures were used to select significant and independent descriptors in order to build the models: (1) D-optimal design to select optimal members of the training and test sets and (2) k-Nearest Neighbor classification (kNN) method along with Genetic Algorithms (GA-kNN Classification). The EC3 values (from LLNAs) of the compounds were ranked quantitatively according to their potencies. Class 1 signified extreme/strong/moderate sensitizers (EC3 < 10%) and Class 2 signified weak/non-sensitizers (EC3 ≥ 10%). Sodium Stearoyl Lactylate was identified as a Class 2 sensitizer, and the LLNA data on this chemical are included in the preceding section. Of the 5 models developed, 4 placed Sodium Stearoyl Lactylate in Class 2, and 1 placed the chemical in Class 1. Thus, the consensus prediction based on the models 1–5 was Class 2.
Ocular Irritation Studies
In Vitro
Sodium Caproyl/Lauroyl Lactylate
The ocular irritation potential of Sodium Caproyl/Lauroyl Lactylate was evaluated using the bovine corneal opacity and permeability test method for identifying ocular corrosives and severe irritants (OECD TG 437). 13 Corneas were exposed to the test substance (750 µl; 10% solution diluted in 0.9% sodium chloride solution) for 10 min, and exposure was followed by rinsing with and without phenol red. Exposure was followed by a 2-h observation period. A mean in vitro irritation score (for cornea) of 46.308 for the 10% solution was reported, classifying the solution as non-corrosive.
Animal
Calcium Stearoyl Lactylate, Sodium Lauroyl Lactylate, Sodium Stearoyl Lactylate, and Sodium Isostearoyl Lactylate
In an ocular irritation study involving 6 albino rabbits, Sodium Isostearoyl Lactylate (undiluted or 15% concentration, 0.1 ml) was instilled into one eye. 5 Ocular reactions were scored at 24 h, 48 h, and 72 h post-instillation. The undiluted ingredient was irritating to the eyes, whereas 15% Sodium Isostearoyl Lactylate was not. When other alkanoyl lactyl lactate salts were tested according to the same procedure using groups of 6 albino rabbits, the results were as follows: undiluted Calcium Stearoyl Lactylate (nonirritating), 10% Sodium Lauroyl Lactylate (nonirritating), and undiluted Sodium Stearoyl Lactylate (nonirritating, 2 tests). 2 In another study, undiluted Sodium Lauroyl Lactylate (0.1 g) was applied to the right eye of each of 6 albino rabbits according to the same procedure. 19 Mild conjunctivitis was observed in 3 of 6 rabbits, and Sodium Lauroyl Lactylate was classified as a nonirritant in rabbit eyes.
Clinical Studies
Case Reports
Sodium Stearoyl Lactylate
A female patient with a 20-year history of palmoplantar pustulosis and chronic hand and foot dermatitis had a positive patch test reaction (score not stated) to 5% Sodium Stearoyl Lactylate in petrolatum. 20 The patient was patch tested with ingredients of the cosmetic products that she had been using, and Sodium Stearoyl Lactylate was the only ingredient that caused a positive reaction. When the patient was re-tested with a 2% Sodium Stearoyl Lactylate (in petrolatum) preparation, a + reaction was observed. A use test that involved 2 daily applications of 5% Sodium Stearoyl Lactylate in petrolatum to the lower arm was also performed. Small papules and itching resulted after a few days, and the reaction was clearly positive on day 18. The control groups consisted of 51 subjects patch tested with 2% or 5% Sodium Stearoyl Lactylate in petrolatum, and Sodium Stearoyl Lactylate was classified as having skin irritation potential in this skin irritation test. (This study was described in the Irritation and Sensitization section). The authors noted that the reproducible patch test and use test reactions are considered to be of an allergic nature, because of the clinical picture, patient history, and patch test results for the 51 controls. Furthermore, the authors noted that this patient seemingly belongs to a group of patients with sensitive, labile skin that easily contracts new allergies.
Summary
The safety of 10 alkanoyl lactyl lactate salts as used in cosmetics is reviewed in this safety assessment. According to the Dictionary, all of these ingredients are surfactants, while some have additional possible functions reported.
According to 2019 VCRP data, Sodium Stearoyl Lactylate is reported to be used in 358 cosmetic products (334 leave-on and 24 rinse-off products). Of the alkanoyl lactyl lactate salts that are reviewed in this safety assessment, this is the greatest reported use frequency. The results of a concentration of use survey conducted by the Council in 2017 indicate that Sodium Lauroyl Lactylate is being used at maximum use concentrations up to 10% in skin cleansing products (rinse-off products). 9 Calcium Stearoyl Lactylate, Sodium Lauroyl Lactate, and Sodium Stearoyl Lactylate are being used at maximum use concentrations up to 7% in leave-on products (tonics, dressings, and other hair grooming aids); this is the highest maximum use concentration in leave-on products that is being reported for the alkanoyl lactyl lactate salts. The highest maximum use concentration in leave-on cosmetic products that are applied directly to the skin is 6.1% for Sodium Lauroyl Lactylate in body and hand products that are not sprayed.
Sodium Stearoyl Lactylate, the most frequently used alkanoyl lactyl lactate salt in cosmetic products, can be manufactured by base-catalyzed esterification of lactic acid and stearic acid. Food Chemicals Codex and European Commission specifications on the composition of this ingredient as a food additive are available, and the same is true for Calcium Stearoyl Lactylate.
In a single sample of human duodenal mucosa in vitro, [14C]-Calcium Stearoyl Lactylate was rapidly hydrolyzed to stearic acid and lactic acid. In an oral dosing study on [14C]-Calcium Stearoyl Lactylate involving mice and guinea pigs, ∼80% of the administered dose was exhaled as 14CO2 within 48 h and most of the remaining radioactivity was excreted in the urine within 24 h post-dosing. Approximately 2% (in mice) and 6% (in guinea pigs) of the administered dose remained in the tissues, mainly in the liver and gastrointestinal tract.
In acute oral toxicity studies involving male rats, an oral LD50 of 25 g/kg body weight was reported for Sodium Stearoyl Lactylate and for Calcium Stearoyl Lactylate. The following other acute oral LD50 values have been reported: > 6.1 g/kg (Sodium Isostearoyl Lactylate, in rats), 6.81 g/kg (Sodium Lauroyl Lactylate, in male rats), and 4.88 g/kg (Sodium Lauroyl Lactylate, in male and female rats).
When Sodium Caproyl/Lauroyl Lactylate was applied to the ears of mice at concentrations of 25% and 50% (2 mice/group) on 3 consecutive days, none of the animals died, and there was no evidence of systemic toxicity.
Some of the findings reported in short-term feeding studies on Calcium Stearoyl Lactylate (rats) at various concentrations were: no deaths (at 12.5% in diet – 43 days), increased relative liver weight (at 5% and 7.5% in the diet – 1 month), no significant liver pathology (at 5% in diet – 27 days), increased liver weight (at 5% in diet – 4 weeks), relative liver weights normal (at 5% in diet – 32 days), liver histology normal (at 5% in diet – duration unknown), relative liver weights normal (at 5% in diet – duration unknown), and kidney histology normal (0.5% in diet – duration unknown). A transient increase in liver weights was observed in rats fed 5% Sodium Stearoyl Lactylate in the diet for 28 days, and organ weights were normal in a dog fed up to 15% Sodium Stearoyl Lactylate in the diet for 1 month.
In a subchronic oral toxicity study, groups of 20 male and female rats were fed Calcium Stearoyl Lactylate at dietary concentrations up to 12.5% for 98 days. Increased relative weights of major organs were observed after feeding with 12.5% Calcium Stearoyl Lactylate. There was no evidence of histological abnormalities in major organs, but lipogranulomata was observed in the adipose tissue of animals fed a dietary concentration of 12.5%. It has been noted that the appearance of lipogranulomata and the increased relative liver weight reported were related to the excessive intake of abnormal portions of long-chain fatty acids. The results of gross and histopathological evaluations of groups of 20 rats fed up to 12.5% Sodium Stearoyl Lactylate in the diet for 102 days were normal.
In a chronic oral toxicity study in which groups of 5 rats were fed diets containing 8% to 22% Calcium Stearoyl Lactylate for periods of up to 6 months, mortality was high (number of deaths not reported) at concentrations of ≥ 20%. Histopathological abnormalities were not observed in this study. Neither gross nor microscopic changes were observed in a 2-year study in which 4 Beagle dogs were fed 7.5% Calcium Stearoyl Lactylate in the diet. Liver weights were in the normal range. A 1-year chronic oral toxicity study involved groups of 60 male and female rats fed a diet containing Sodium Stearoyl Lactylate at concentrations up to 5%. The NOAELs for Sodium Stearoyl Lactylate were 2214 mg/kg/day for males and 2641 mg/kg/day for females, which were the highest doses tested).
Sodium Caproyl/Lauroyl Lactylate and Calcium Stearoyl Lactylate were not genotoxic to any of the S. typhimurium strains evaluated in the Ames test, with or without metabolic activation. Calcium Stearoyl Lactylate also was not genotoxic, with or without metabolic activation, in the chromosome aberrations test involving a Chinese hamster fibroblast cell line.
In a 1-year oral study, the occurrence of endometrial stromal polyps in the uterus of female animals was reported after groups of 30 male and 30 female Wistar rats were fed Sodium Stearoyl Lactylate in the diet at concentrations of 1.25%, 2.5%, and 5%. The incidence in treated rats was higher than that in concurrent controls, but was not statistically significant. Additionally, data on the historical incidences of this this tumor type at the laboratory where the study was performed demonstrated that endometrial stromal polyps are common in the rat strain that was tested. Therefore, this finding was not considered treatment-related.
The following results are from skin irritation studies involving albino rabbits (number of animals not stated): undiluted Calcium Stearoyl Stearate (nonirritating), 10% Sodium Lauroyl Lactylate (nonirritating), and undiluted Sodium Stearoyl Lactylate (PII = 0.5, 2 tests). Sodium Caproyl/Lauroyl Lactylate (in AOO vehicle) caused erythema and an increase in ear thickness in 4 mice (CRL:NMRI BR strain; 2/group) when tested at concentrations of 25% and 50%. In a skin irritation test on Sodium Isostearyl Lactylate involving 6 albino rabbits, the PII was 7.17 for the undiluted ingredient and 1.13 for 15% Sodium Isostearyl Lactylate. In a human skin irritation study, 25 and 26 subjects were patch tested with 2% and 5% Sodium Stearoyl Lactylate in petrolatum, respectively. The 2% concentration produced 10 reactions that were classified as doubtful (i.e., probably irritating), and the 5% concentration produced 14 reactions with the same classification. It was concluded that Sodium Stearoyl Lactylate has skin irritation potential. A diluted hair styling product (Calcium Stearoyl Lactylate effective concentration = 2.5%) was classified as a skin irritant in a study involving 54 subjects. In 4 separate skin irritation studies, each involving 50 subjects, a hair molding cream containing 7% Calcium Stearoyl Lactylate was classified as nonirritating to the skin.
In LLNAs of Sodium Caproyl/Lauroyl Lactylate and Sodium Lauroyl Lactylate at test concentrations up to 50%, Sodium Caproyl/Lauroyl Lactylate was classified as a weak-moderate skin sensitizer and Sodium Lauroyl Lactylate was classified as a weak skin sensitizer. A Sodium Lauroyl Lactylate trade name material was also classified as a weak sensitizer in the LLNA. Sodium Lauroyl Lactylate was also classified as a weak skin sensitizer in a guinea pig maximization test in which 10 animals were challenged with a test concentration of 0.5%. A silicone antifoam emulsion containing Sodium Stearoyl Lactylate (75% dilution; effective test concentration = 1.13% Sodium Stearoyl Lactylate) was a nonsensitizer in 20 guinea pigs.
An in vitro assay involving the RHE and detection of inflammation markers (IL-1α [released by injured cells] and IL-8 [secondary inflammatory cytokine]) was used to evaluate the skin toxicity of Sodium Stearoyl Lactylate. Sodium Stearoyl Lactylate was predicted to be an allergen based on the results of this assay. In a study on the modeling of skin sensitization data on a number of diverse compounds, EC3 values (from LLNAs) were ranked for these compounds quantitatively based on sensitization potency. Sodium Stearoyl Lactylate was classified as a Class 2 (weak/non-sensitizers) sensitizer in the ranking.
Caproyl/Lauroyl Lactylate (10% in saline solution) was classified as a non-corrosive substance in the in vitro bovine corneal opacity and permeability test. The results of ocular irritation tests on alkanoyl lactyl lactate salts involving groups of 6 albino rabbits were as follows: undiluted Calcium Stearoyl Lactylate (nonirritating), undiluted Sodium Isostearoyl Lactylate (irritating), 15% Sodium Isostearoyl Lactylate (nonirritating), 10% Sodium Lauroyl Lactylate (nonirritating), and undiluted Sodium Stearoyl Lactylate (nonirritating, 2 tests). In another study, undiluted Sodium Lauroyl Lactylate (0.1 g) was applied to the right eye of each of 6 albino rabbits. 19 Mild conjunctivitis was observed in 3 of 6 rabbits, and Sodium Lauroyl Lactylate was classified as a nonirritant.
A female patient with a 20-year history of hand and foot dermatitis had positive patch test reactions to Sodium Stearoyl Lactylate (2% and 5% in petrolatum) that were considered allergic in nature. A use test that involved 2 daily applications of 5% Sodium Stearoyl Lactylate in petrolatum to the lower arm of this patient was also performed. Small papules and itching resulted after a few days, and the reaction was clearly positive on day 18.
Discussion
The Panel noted that based on the preponderance of data included in this report, alkanoyl lactyl lactate salts may have the potential to induce skin sensitization. Although a silicone antifoam emulsion containing Sodium Stearoyl Lactylate (75% dilution; effective test concentration = 1.5% Sodium Stearoyl Lactylate) was a nonsensitizer in guinea pigs, LLNA results indicated weak or moderate sensitization potential. Specifically, in LLNAs of Sodium Caproyl/Lauroyl Lactylate and Sodium Lauroyl Lactylate at test concentrations up to 50%, Sodium Caproyl/Lauroyl Lactylate was classified as a weak or moderate skin sensitizer (EC3 = 12.4%; EC3 = 9.3%, respectively), and Sodium Lauroyl Lactylate was classified as a weak skin sensitizer (EC3 = 15%). A Sodium Lauroyl Lactylate trade name material was also classified as a weak sensitizer in the LLNA (EC3 = 15%). Additionally, Sodium Lauroyl Lactylate was classified as a weak skin sensitizer in a guinea pig maximization test in which animals were challenged with a test concentration of 0.5%. In a case report, a patient with a history of hand and foot dermatitis had positive patch test reactions to Sodium Stearoyl Lactylate (2% and 5% in petrolatum) that were considered allergic in nature. Furthermore, following daily applications of 5% Sodium Stearoyl Lactylate in petrolatum to this patient, a positive reaction was observed. After reviewing these sensitization data on alkanoyl lactyl lactate salts, the Panel noted that the potential for induction of skin sensitization varies depending on a number of factors, including the area of product application; thus, formulators should assess the potential for final formulations to induce sensitization using a QRA or other accepted methodologies.
DART data were lacking for these ingredients. However, the concern over any potential developmental or reproductive toxicities was mitigated by the consistently negative short-term, subchronic, and chronic toxicity data, and the lack of clinical reports associated with the long-term historical use of these ingredients as food additives.
The Panel was also concerned that the potential exists for dermal irritation with the use of products formulated using alkanoyl lactyl lactate salts. The Panel also specified that products containing these ingredients must be formulated to be nonirritating.
Food Chemicals Codex specifications and European Commission regulations relating to the following components/ impurities of Calcium Stearoyl Lactylate/Sodium Stearoyl Lactylate are available: arsenic, calcium, cadmium, lactic acid, lead, mercury, and sodium. The Panel stressed that the cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit impurities.
Conclusion
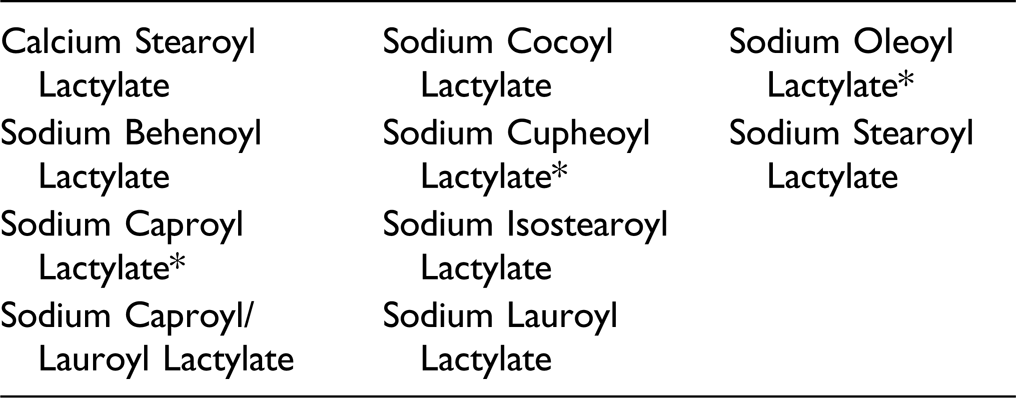
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 555 13th St., NW, Suite 300W, Washington, DC 20004, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.