Abstract
Streptococcus salivarius is a common, harmless, and prevalent member of the oral microbiota in humans. In the present study, the safety of S. salivarius UBSS-01 was evaluated using in silico methods and preclinical and clinical studies. In an acute toxicity study, rats were administered with 5 g/kg (500 × 109 CFU) S. salivarius UBSS-01. The changes in phenotypic behaviors and hematological, biochemical, electrolytes, and urine analyses were monitored. No toxicity was observed at 14 days post-treatment. The no observable effects limit (NOEL) of S. salivarius UBSS-01 was >5 g/kg in rats. In a 28-day repeat dose toxicity study, rats were administered S. salivarius UBSS-01 once daily at doses of 0.1, 0.5, and 1 g/kg (10, 50, and 100 billion CFU/kg, respectively) body weight. S. salivarius UBSS-01 did not influence any of the hematology parameters and clinical chemistry parameters in plasma and serum samples after 28-day repeated administration. No structural abnormality was observed in the histological examination of organs. Whole genome analysis revealed the absence of virulence factors or genes that may transmit antibiotic resistance. In the double-blind study with 60 human participants (aged 18–60 years), consumption of S. salivarius UBSS-01 for 30 days was found to be safe and results were comparable with placebo treatment These findings indicate that S. salivarius UBSS-01 may be safe for human consumption.
Keywords
Introduction
In 1974, Park coined the word “probiotics” to describe “microorganisms or substances that promote the gut homeostasis of host animals.” 1 Probiotics are now classified by the Food and Agriculture Organization (FAO) and World Health Organization (WHO) as living microorganisms that, whenever ingested in sufficient doses, enhance the health of the host. 2 The use of probiotics as an adjuvant to traditional therapies has been recommended for the treatment of several human ailments. Recently, probiotics have been recognized as one of the beneficial ingredients of various foods and dairy products. However, it is essential to have experimental evidence to support the safety of probiotics, submitted to regulatory agencies, who must allow a novel probiotic to enter the food supply. Thus, robust experimental evidence is needed to validate the safety of a new probiotic strain. In the United States, the U.S. Food and Drug Administration (FDA) provides a regulatory process to determine if an ingredient intended to be added to foods in the country meets the criteria and statutory requirements to be considered “Generally Recognized as Safe” (GRAS). Europe and other country’s regulatory agencies around the world carry out their own methods for assessment of the safety of a food ingredient before it is added to foods. While the utility of probiotics has been intended to gain benefits from intestinal flora, 3 dysbiosis of oral microbiota is believed to be responsible for dental caries, gingivitis, and halitosis.4,5 Approximately 800 species of microorganisms have been identified in the human oral cavity to date. 6 Streptococcus salivarius is one of the pioneering organisms that colonize the human buccal cavity from newborns and later contribute to nasopharyngeal and oral microorganism colonization.7-9 S. salivarius comprises 10%–30% of the total Streptococcal taxa, and adults and children’s saliva samples contain around 107 and 108 CFU/mL of S. salivarius.10,11 Normally, S. salivarius is mainly located on the dorsal surface of the tongue but not in high abundance in sub-gingival sites and other mucosal membranes. However, this bacterium was reported to reduce dental plaque by inhibiting inflammation and volatile sulphur compounds. 12
S. salivarius also serves as a starter in some conventional fermented milk products, such as European, African, and Colombian milk and cheeses.13,14 Ingestion of S. salivarius BP8, S. salivarius BP156, and S. salivarius BP160 was found to inhibit the proliferation of liver and breast cancer cells. 15 Additionally, it has been demonstrated that S. salivarius K12 and M18 contain a minor proteinaceous component that can inhibit the activation of primary human gingival fibroblasts and the production of cytokines interleukin-6 (IL-6) and IL-8 via periodontal disease pathogens in 20 participants of 20–60 years age. Furthermore, the significance of S. salivarius strains in the management of oral health has been established through clinical studies on dental caries, gingivitis, and oral candidiasis, 7 making it beneficial for enhancing oral health and addressing various human health conditions.
A major obstacle to bacteriotherapy for strains that do not already have a long history of use in conventional fermented items is safety concerns, even though they are generally regarded to be widespread and unharmful members of the typical human indigenous microbiota. Moreover, some recent studies have reported safety concerns with probiotic treatment. Probiotics such as Saccharomyces cerevisiae, Lactobacillus, and Bifidobacterium were suspected to translocate from intestinal mucosa to the bloodstream or critical organs, producing systemic or localized infections. However, these untoward effects of probiotics may be related to other concomitantly consumed medication-induced toxicity, hyperinflammation, leaky gut, and weakened immunity due to chronic diseases in study participants.16-18 Probiotic virulence factors and toxic metabolites may cause opportunistic infections and metabolic disorders. 19 Antibiotic-resistant bacteria may develop as a result of horizontal antibiotic resistance gene transfer from healthy gut microbiota to pathogenic intestinal microbes and vice versa.20,21 It is proposed that extensive comparative proteomics and functional genomics studies should be conducted to elucidate novel genes in probiotics related to antibiotic resistance. This is aimed at preventing the introduction of transient phenotypic strains into commerce that may confer antibiotic resistance. Such studies would better inform the medical community and consumers pertaining to the risks and benefits associated with the consumption of probiotic dietary supplements. Excessive immune response via provoked cytokine production may result in autoimmune illness or inflammation. 22 From a broader perspective, probiotics are considered safe, however as per recent regulatory guidelines each novel strain should undergo a safety assessment before it is available in the market for human consumption. S. salivarius UBSS-01, a proprietary strain of Unique Biotech Limited Hyderabad, India, has been completely characterized with probiotic properties established. Since the effects of probiotics are strain-specific, the present study aimed to establish the safety of S. salivarius UBSS-01 for human consumption through screening of the whole genome sequence for virulence factors, plasmids, and transferable antibiotic resistance genes, animal toxicity studies (acute and repeat dose in rats), and safety and tolerability in a randomized, double-blind, placebo-controlled trial in healthy subjects. Some other measures, such as hemolytic potential, lecithinase activity, and lack of biogenic amines (BA) production, were also evaluated.
In the preclinical study, we assessed the safety of S. salivarius UBSS-01 in male and female Sprague-Dawley rats through acute- and 28-day repeat dosage treatments in accordance with Organisation for Economic Co-operation and Development (OECD) criteria 423 and 407, respectively. Similar experimental design has been adopted for other probiotic safety assessments.23,24 For acute toxicity testing, we orally administered S. salivarius UBSS-01 at the dose of 5000 mg/kg (corresponding to 500 billion CFU/kg) of body weight. The signs of toxicity in animals included observations of altered fur quality, lacrimation, aberrant breathing patterns, and changes to their skin, nose, and tongue. Rats’ gait and body posture, reaction to handling or sensory stimuli, grip strength, and aberrant behavior like self-mutilation, walking backward, circling, or stereotypical behavior were noted at 30 min, and 1, 2, 3, and 4 h post-administration followed by twice daily from day 1 through day 14. For a 28-day repeat dose study of S. salivarius UBSS-01, rats received 0.1, 0.5, and 1 g/kg per day (equivalent to 10, 50, and 100 billion CFU/kg, respectively) probiotics. The reversal groups, i.e., vehicle control and high dose (1 g/kg), were observed for an additional 14 days to check for the appearance of any delayed symptoms. At the end of the study, animals were euthanized for biochemical and histopathological analyses. The hematological system is among the most vulnerable units to toxic elements and serves as a reliable biomarker of pathological and physiological conditions in animals and humans. Hence, hematological and serum biochemical analyses are crucial for assessing the toxicological effects. Here, we aim to address any safety concerns, such as the potential for toxigenicity and pathogenicity associated with ingesting the probiotic strain S. salivarius UBSS-01.
In the clinical study, administration of a high dose of S. salivarius UBSS-01 for 1 month was evaluated on vital parameters such as heart rate, systolic blood pressure (BP), diastolic BP, oral temperature, and respiration rate, hematological (WBC, RBC, neutrophils, lymphocytes, eosinophils, basophils, monocytes, platelets, PCV, and hemoglobin) and biochemical parameters (RBS, urea, creatinine, sodium, potassium, chloride, total bilirubin, direct bilirubin, indirect bilirubin, total protein, albumin, AST, ALT, and alkaline phosphatase). Moreover, the effect of S. salivarius UBSS-01 on the gastrointestinal system was also evaluated by the Gastrointestinal Symptom Rating Scale (GSRS).
Materials and Methods
Probiotic: Streptococcus salivarius UBSS-01
For the rat safety study, lyophilized S. salivarius UBSS-01 powder with a purity of 97.01% (Batch # UBL/SAM/22/03/092, obtained from fermented food) with a potency of 100 billion CFU/g was used. In the clinical study involving human participants, individuals received either placebo sachets or S. salivarius UBSS-01 sachets, each containing 10 billion CFU per sachet.
Bacterial Strain
16S rDNA sequencing and whole genome sequencing (WGS) of S. salivarius UBSS-01 were conducted and submitted to NCBI (https://www.ncbi.nlm.nih.gov/nuccore/VDCW00000000).
Hemolytic Activity of S. salivarius UBSS-01
The hemolytic ability of S. salivarius UBSS-01 was estimated using Columbia sheep blood agar. Briefly, the hemolytic activity of the strain was evaluated by incubating the plates for a period of 48 hours at 37°C, and red blood cell lysis in the media around the colonies was used to classify the strain. Green zones surrounding the colonies were regarded to be α-hemolysis, clear zones were interpreted as β-hemolysis, and no zones around colonies were considered to be γ-hemolysis. The γ-hemolysis-producing strains are regarded as safe.25,26
Lecithinase Activity
The lecithinase activity of S. salivarius UBSS-01 was assessed using MYP agar with egg yolk. An enzyme called lecithinase, which may be released by bacteria, leads to the hydrolysis of lecithin—the primary component of the cell membrane—resulting in cell lysis. After 48 hours of incubation at 37°C with the cultured bacterium, precipitate production was measured. The presence of precipitation suggests a positive indication of lecithinase activity in the culture.
Qualitative Assay for Biogenic Amine Production
S. salivarius UBSS-01 was streaked onto M17 agar medium, which consisted of M17 agar (pH 5.0) supplemented with 0.2% precursor amino acids and 0.006% bromocresol purple. The precursor amino acids employed were histidine, tyrosine, arginine, and phenylalanine, specifically chosen for the detection of histamine (HIS), tyramine (TYR), putrescine (PUT), and β-phenethylamine (PHE), respectively. The pH indicator included in the medium facilitated the phenotypic assessment of biogenic amine (BA) production; a more alkaline environment resulted in a distinct purple coloration of the medium. For comparison, Escherichia coli MTCC 1687 served as the positive control, and the S. salivarius K12 strain was used as a reference strain.
In Silico Analyses of S. salivarius UBSS-01 Genome
Genome Mining for Antibiotic Resistance Genes
The whole genome sequence (WGS) of S. salivarius UBSS-01 was analyzed to identify the genes involved in antibiotic resistance using the Comprehensive Antibiotic Resistance Database (CARD; https://card.mcmaster.ca/) as described by Carattoli et al. 27
Detection of Virulence Factors of S. salivarius UBSS-01
The genome of S. salivarius UBSS-01 was screened for possible virulence factors using the Virulence Factor Database (VFDB) described earlier. 28 The protein sequences of S. salivarius UBSS-01 were compared with the VFDB core database utilizing a local reciprocal blast p analysis using the BLAST + software. Criteria for selection were greater than 50% identity match and E-values under 10−5. Genes identified as hits in the VFDB were further investigated to assess their virulence potential. These hit genes were subjected to reciprocal blasting against the NCBI’s nr database to determine their expected roles and functions.
Experimental Animals
Seven to 8-week-old male (190 ± 20 g) and female (180 ± 20 g) Sprague-Dawley rats were purchased for acute and subacute toxicity studies. Both acute and subacute toxicity studies were approved by the NIPER Institutional Animal Ethics Committee (IAEC) (Approval number: NIP/12/2021/PC/441), and experiments were performed as per guidelines of the Committee for the Purpose of Control and Supervision of Experimentation on Animals (CPCSEA). Animals were housed under standard laboratory temperature (22 ± 2°C), relative humidity (45%–60%), and 12 h light-dark cycle. The animals were given unlimited access to a chow diet (Laboratory animal feed, VAK Nutritional Solutions, Hyderabad, India) and potable water.
Preclinical Acute Oral Toxicity Study (Single-Dose Toxicity Study)
The acute oral toxicity study was performed according to the OECD guideline 423 adopted on December 17, 2001, for the Testing of Chemicals as well as the United States FDA Redbook, 2000 (Chapter IV.B.1—Guidelines for Designing and Conducting Toxicity Studies). In this acute toxicity study, rats (of each sex) were randomly divided into three different groups (n = 3), including the S. salivarius UBSS-01 treated group (step 1 and step 2) and the vehicle group (Figure 1). In step 1, after 24 hours of fasting, group IIA (male n = 3) and group IIB (female n = 3) received a peroral single dose of S. salivarius; UBSS-01 (5 g/kg, equivalent to 500 billion CFU/kg) that was dissolved in deionized water (10 mL/kg). Group IA (male n = 3) and group IB (female n = 3) rats served as vehicle control and received deionized water (10 mL/kg). These controls were included to elucidate the broad safety range of probiotics as specific recommendations, particularly for probiotics toxicity screening, are unavailable.
29
Noteworthy, similar experimental designs have also been reported for other probiotic safety assessments in the published literature.30,31 The same process was repeated with the three rats in group IIIA (male n = 3) and group IIIB (female n = 3). After dosing, each animal was monitored for 14 days to examine for any clinical signs of toxicity and mortality. The food and water intake were measured once daily for 14 days. Schematic presentation of in vivo acute toxicity study.
Throughout the experiment (14 days), all animals were monitored twice daily for clinical symptoms (abnormal respiratory, autonomic (lacrimation and piloerection), circulatory, neurological activity, and mortality). Furthermore, toxicity symptoms such as skin changes, examinations of the fur, eyes, mucous membranes, and secretory occurrence were also noted, if any. The presence of clonic or tonic movement, changes in gait or posture in reaction to handling, and odd behaviors such as excessive grooming, circling, walking backward, and self-mutilation were also noted as additional clinical indicators of poisoning. Weekly records of the body weight, water intake, and food consumption were kept. At the end of 14 days, all the animals were euthanized in a carbon dioxide chamber. A gross morphological examination of vital organs like the brain, lungs, heart, liver, stomach, kidney, spleen, testis, and uterus was performed.
Preclinical 28-Day Repeat Dose Toxicity Study
The systemic toxic effect of S. salivarius UBSS-01 was evaluated through a repeat dose (28 days) study conducted according to OECD guideline 407. This study was conducted based on preliminary information gathered through acute toxicity testing. The 28-day repeat dose study helps understand the effects of daily consumption of probiotics following repeated oral exposure. In this study, 60 rats were randomly assigned to 6 groups, each having 5 males and 5 females. G1: Vehicle control (received deionized water, 5 mL/kg of body weight/day); G2: received low dose 0.1 g/kg body weight/day of S. salivarius UBSS-01 (10 billion CFU); G3: mid dose 0.5 g/kg body weight/day of S. salivarius UBSS-01 (50 billion CFU); G4: high dose 1 g/kg body weight/day of S. salivarius UBSS-01 (100 billion CFU); G1R: vehicle control reversal (deionized water, 5 mL/kg body weight/day); and G4R: high dose reversal 1 g/kg body weight/day of S. salivarius UBSS-01 (100 billion CFU). The highest dose for this repeated dose administration study was 1 g/kg body weight, which is the maximum quantity allowed per OECD guidelines.
The different doses of probiotics, i.e., low dose (0.1 g/kg in 5 mL/kg deionized water), mid-dose (0.5 g/kg in 5 mL/kg deionized water), and high dose (1 g/kg in 5 mL/kg deionized water), were freshly formulated in deionized water. Autoclaved water was used as vehicle control. For all groups, dosing was carried out through oral gavage at a dosage level of 5 mL/kg until the 28th day. After the 29th day of therapy, G1–G4 groups were euthanized, while the reversal group (G1R and G4R), had a thorough inspection for an extra 14 days without receiving any therapy to assess any potential delayed signs of toxicity (Figure 2). Study design for in vivo repeat dose toxicity testing of S. salivarius UBSS-01 for 28 days and additional 14-day probiotic-free observation in reversal groups.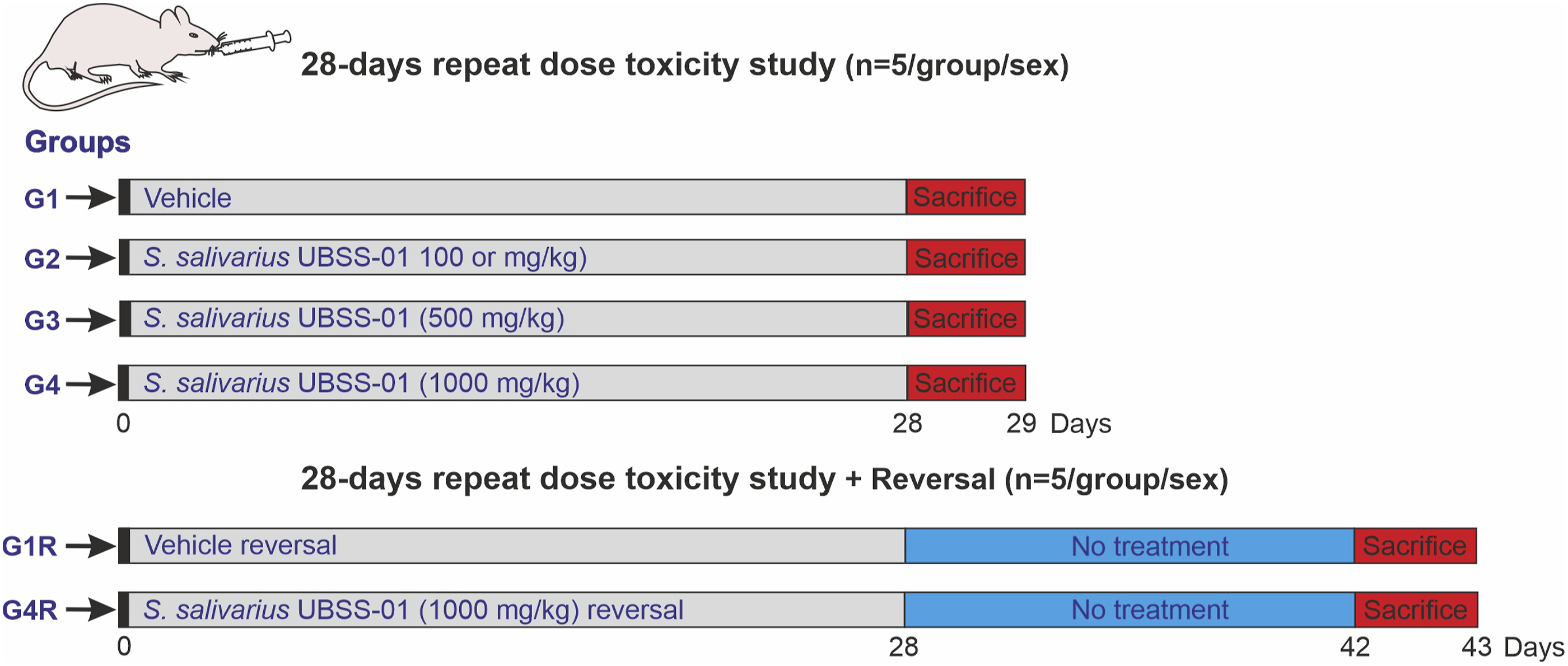
Clinical signs were observed once daily during the recovery period in all animals. The clinical signs as described above in the acute study were assessed during the 28-day repeated dose study like before dosing, at intervals of 30 min, 1 hour, 2 hours, 3 hours, and 4 hours after dosing on day 1, and then once daily until the 29th day for G1–G4 groups and additional 14 days for reversal groups (G1R and G4R). The animal’s body weight was recorded weekly once.
Hematology Analysis of Preclinical Samples
Under anesthesia, blood samples were drawn into a centrifuge tube containing dipotassium ethylenediaminetetraacetic acid (K2-EDTA) through a retro-orbital plexus micro-hematocrit heparinized glass capillary tube. Hematological parameters, including blood clotting time, hemoglobin, mean corpuscular volume, erythrocytes, mean corpuscular hemoglobin, leucocytes, mean corpuscular hemoglobin concentration, hematocrit, differential count, and platelets (PLT), were analyzed clinically using (Nihon Koheden Celltac G MEK-9100K) via scatter laser and flow cytometry. The DynaHelix flow cytometry technology was adopted for counting leucocytes, erythrocytes, and PLT. WBCs were analyzed and distinguished using the DynaScatter laser optical technique. We measured WBC size at a small forward angle (FSS), cell shape and nucleo-chromatin particle complexity at a large forward angle (FLS), and internal granularity and globularity at a side angle (SDS).
Biochemical and Urine Analysis of Preclinical Samples
Plasma samples were collected by centrifuging the blood at 10000 rpm at 4°C for 10 min and used to estimate the biochemical parameters like total protein, total bilirubin, alanine transaminase (ALT), triglycerides, aspartate aminotransferase (AST), alkaline phosphatase, urea nitrogen, glucose, total protein, albumin, and creatinine by using Accurex Kits. Serum electrolytes like Na+, K+, and Ca2+ were estimated using a SensaCore ST-200 CL electrolyte analyzer. The ST-200 CL electrolyte analyzer from Sensa Core was used for the analysis of whole blood, serum, plasma, and urine for various combinations of sodium, potassium, calcium, ionized calcium, and chloride.
Gross Morphology and Organ Weight
Following the collection of the blood, all rats were euthanized using a saturated CO2 chamber; vital organs such as the brain, lungs, heart, liver, and kidneys were removed and observed for gross morphology. All extra fat and tissue were removed from the organs, which were weighed, and stored in 10% (v/v) formalin for histological examination. The percent organ weight to body weight ratio was calculated using the formula organ weight/body weight × 100.
Histological Examination
The formalin-fixed vital organs from G1 and G4 were used to evaluate histological changes. Briefly, paraffin-embedded sections were cut (5 μm) using a microtome (Leica RM2145, Germany), mounted on a Mayers albumin-coated slide and dried overnight at RT. Tissue sections were deparaffinized in xylene, rehydrated with ethanol, stained with hematoxylin and eosin, and mounted with dibutyl phthalate polystyrene xylene (DPX). 32 The stained slides were observed under the microscope by an individual blind to the treatment conditions, for examining the histological changes.
Safety Studies in Humans with S. salivarius UBSS-01
Study Design
This randomized, double-blind, placebo-controlled study was conducted at Adichunchanagiri Hospital & Research Centre, Karnataka, India, according to Good Clinical Practice guidelines. Out of 80 people screened, 60 participants who satisfied the inclusion criteria were enrolled. These participants were divided into two groups of thirty subjects each (S. salivarius UBSS-01 and placebo-treated groups) using the block randomization method.
Inclusion Criteria and Exclusion Criteria
Demographic Data.
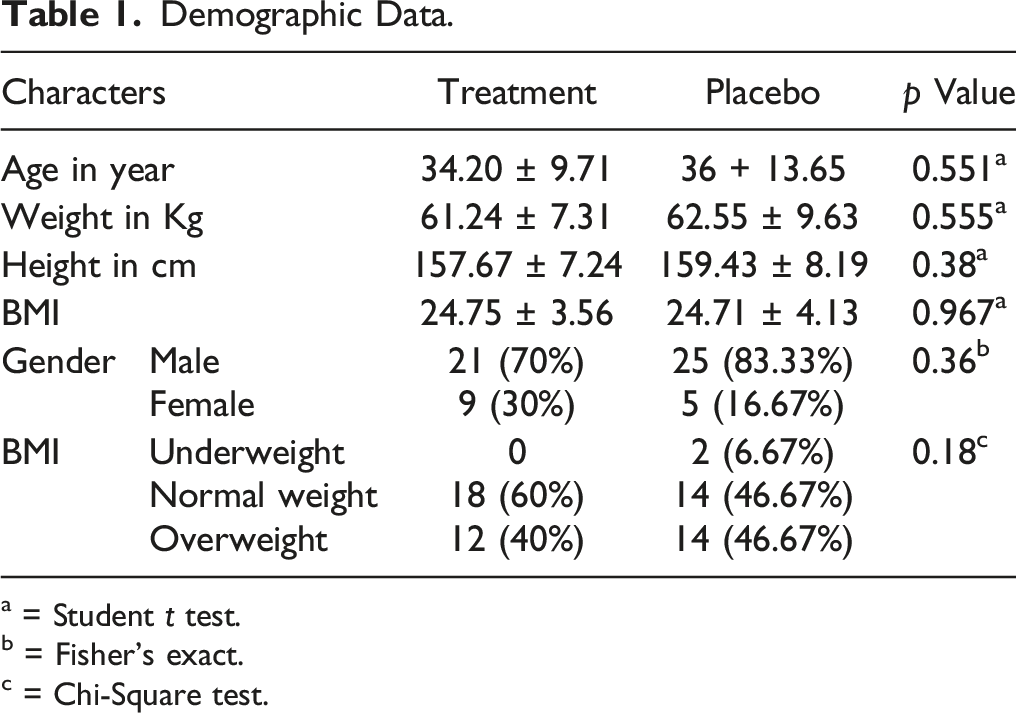
a = Student t test.
b = Fisher’s exact.
c = Chi-Square test.
The study excluded individuals with known food allergies or intolerances, those with a history of active or chronic dental or medical disorders, individuals susceptible to bloating, gas, or diarrhea, pregnant or attempting to conceive, nursing mothers, recent recipients of antibiotics, current users of nicotine, e-cigarettes, cannabis, or nicotine replacement drugs, and those with a history of regular alcohol consumption. Additionally, anyone who took over-the-counter laxatives, probiotic supplements, probiotic-rich foods like yogurt or kefir, or any other drugs, supplements, or items that may have affected the study’s outcomes were omitted from the analysis.
Probiotic and Placebo Samples
In this study, 46 male and 14 female subjects were randomized into two treatment groups to receive either S. salivarius UBSS-01 sachets (probiotic group, n = 30) or identical-looking placebo sachets [placebo group (n = 30)], once daily for 30 days (Figure 3). The probiotic samples were administered in the form of single-use sachets that contained 10 billion CFU of S. salivarius UBLSS-01. The placebo sachets had the same appearance as the probiotic sachets but contained only the excipient, lacking the active ingredient. For 30 days, the participants were advised to consume one probiotic or placebo sachet every night before bedtime. The participants recorded the daily intake of the sachets in their study diary and noted symptoms that were related to gastrointestinal health (abdominal discomfort, bloating, nausea, burping, gas, change in bowel movements and stool consistency, and heartburn), fever, or any adverse events. Compliance was assessed by reviewing the study diaries. Consort flow diagram representing the intention-to-treat (ITT) population.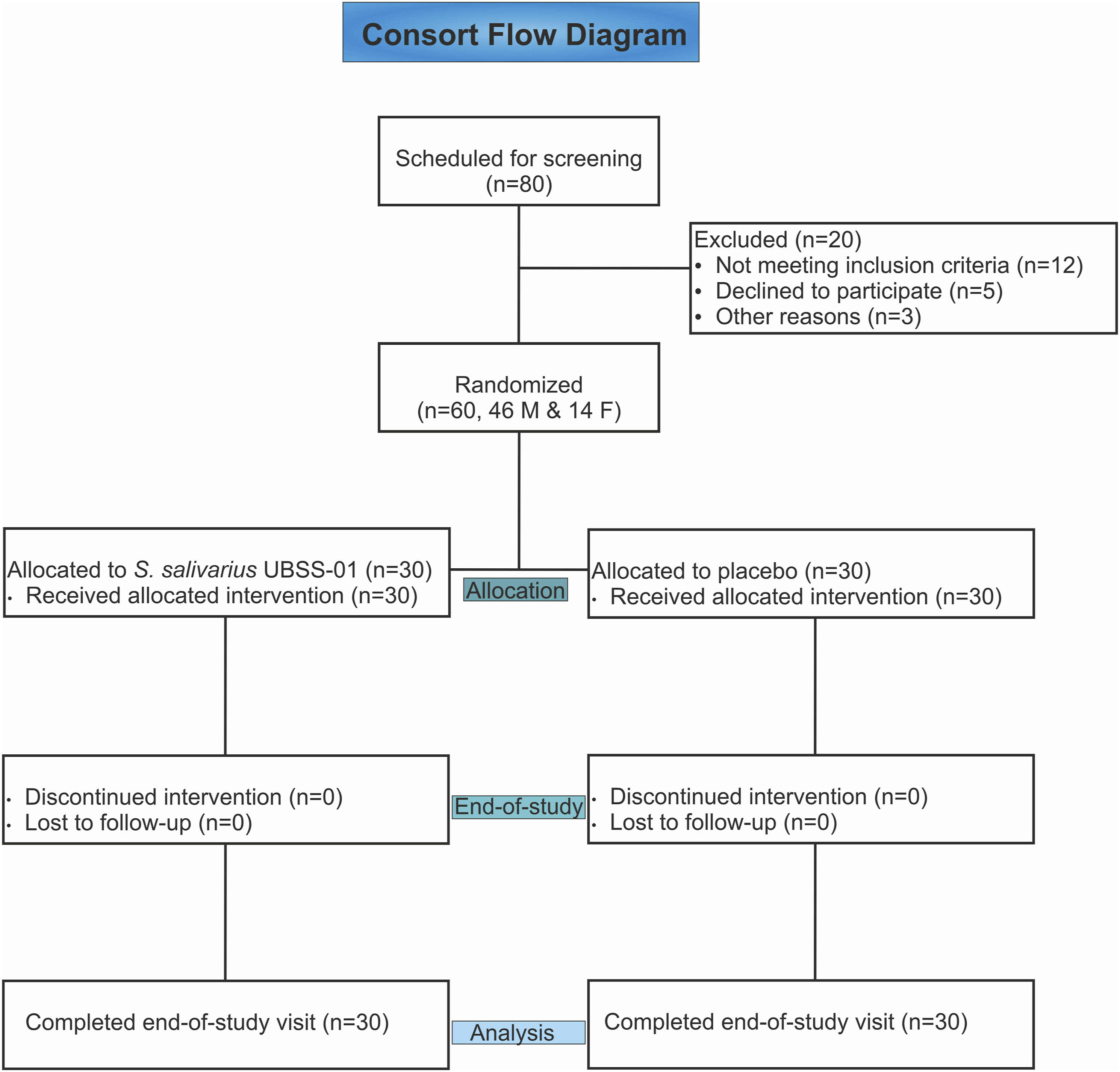
Study Outcome Measures
Participants were asked to report changes in their health and AEs, if any. The subjects were instructed to assess their own gastrointestinal health through the Gastrointestinal Symptom Rating Scale (GSRS) prior to the start of treatment and at the end of the study. The GSRS consisted of 15 questions covering five symptom parameters, including abdominal pain (stomach pain, nausea, and hunger pains), reflux disorder (acid regurgitation and heartburns), diarrhea condition (loose stools and urgent need for defecation), indigestion symptoms (borborygmus, abdominal distension, eructation, and increased flatus), and constipation syndrome (hard stools and feeling of incomplete evacuation). Every question was graded on a 7-point Likert scale with “1” indicating “no discomfort”, “2”—“minor discomfort”, “3”—“mild discomfort”, “4”—“moderate discomfort”, “5”—“moderately severe discomfort”, “6”—“severe discomfort”, and “7” indicating “very severe discomfort”. 33
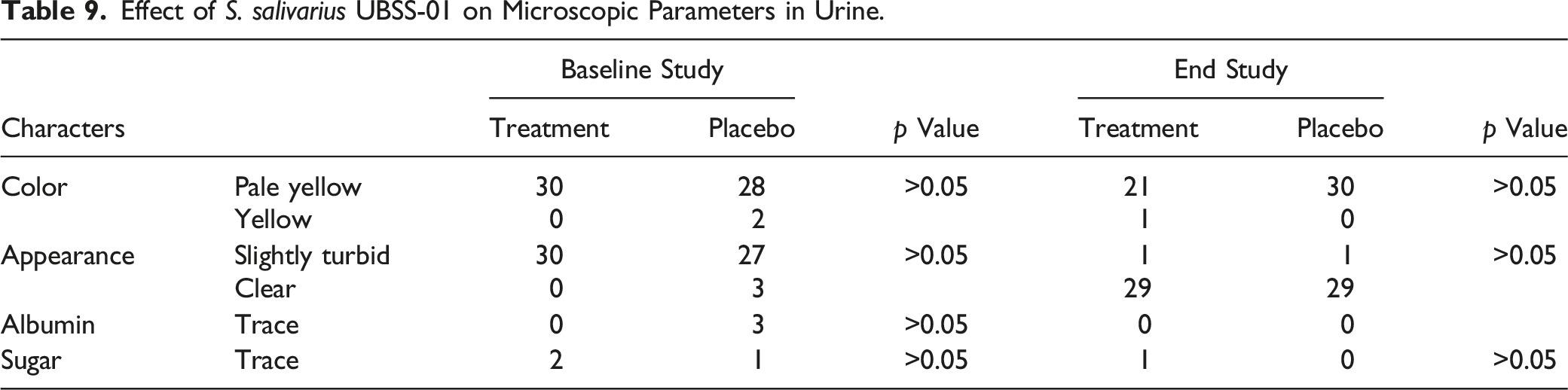
At the beginning and end of the study, fasting blood and urine samples were collected from each participant to analyze common laboratory markers. Complete blood count (CBC), leukocyte count, erythrocytes, hemoglobin, packed cell volume, platelet count, serum electrolytes (sodium, potassium, and chloride), serum chemistry (random blood sugar, blood urea nitrogen, and creatinine), and liver function tests (albumin globulin, bilirubin, alkaline phosphatase, and aspartate aminotransferase), were all performed on fasting blood samples. Urine samples were evaluated for color, appearance, specific gravity, pH, protein, occult blood and albumin, sugar, presence of pus and epithelial cells, RBC, cast, and crystals. The baseline and end of the study values were compared to assess the effect of S. salivarius UBSS-01 on various laboratory parameters for the evaluation of safety.
Statistical Analysis
All numerical data, including body weight, hematological parameters, clinical chemistry parameters, urine analysis, electrolyte, and percent organ-to-body weight ratio, were expressed as mean ± standard deviation (SD). The mean difference between the control and treatment groups was analyzed using GraphPad Prism (version 8) (GraphPad Software, Inc., CA, USA). The one-way analysis of variance (ANOVA) followed by post-hoc Tukey’s multiple comparison tests was used to derive the significance. For the clinical study, the Statistical Package for the Social Sciences (SPSS) version 26 was used to analyze data. The continuous numerical variables were represented as mean and standard deviation (S.D.), and the categorical variables were represented as frequency and percentage. Student’s t test was used to compare the continuous numerical variables, and Chi-Square or Fisher’s exact test was used to compare the categorical variables. p ≤ 0.05 was considered statistically significant.
Results
In Vitro Studies with S. salivarius UBSS-01
Hemolytic and Lecithinase Activity
The hemolytic activity of bacteria was phenotypically assessed by streaking them on blood agar plates. The majority of oral Streptococcus species, particularly those belonging to the Streptococci group, exhibit alpha hemolysis. However, in this study, no hemolytic activity of S. salivarius UBSS-01 was observed (Supplemental Figure 1A). Additionally, we investigated the lecithinase synthesis activity of S. salivarius UBSS-01 (Supplemental Figure 1B). No precipitation with S. salivarius UBBS-01 was observed, indicating that it is lecithinase negative.
Biogenic Amine (BA) Production
BAs are compounds containing one or more low-molecular-weight amine molecules. Despite having important functions in several physiological processes, eating foods high in BA may contribute to toxicological symptoms like hypertension, tachycardia, rashes, and migraines. 34 The decarboxylation of amino acids by bacteria is a source of BA. In this study, S. salivarius was streaked onto medium decarboxylase plates for 24 h to carry out a BA plate test. After 24 h of incubation at 37°C, the media turned into purple color in the presence of E coli (produces biogenic amines) but no color change was observed with S. salivarius UBSS-01 and K12 strains (Supplemental Figure 2), indicating that the strains did not produce biogenic amines. S. salivarius growth did not cause a pH color change, whereas E coli growth caused the decarboxylase media to change to a distinct purple color, indicating the rise of pH to more alkaline conditions due to the presence of BA
Preclinical Study
Acute Oral Toxicity of S. salivarius UBSS-01
No abnormal behavior in gait and posture, response to handling or sensory stimuli, grip strength, and aberrant behavior like self-mutilation, walking backward, circling, or stereotypical behavior was observed after 30 min of single oral administration of S. salivarius UBSS-01 at the dose of 5000 mg/kg (500 billion CFU/kg) body weight. No mortality, morbidity, or behavioral alterations were noted during the 14-day observation period. Moreover, we did not observe any change in body weight, food intake, water intake, or morphological phenotypes related to fur color, lacrimal secretion, skin allergy, nose toxicity, and swollen tongue (data not shown). On the 15th day, a gross necropsy of internal organs was executed, and no toxicity was detected in the organs like the heart, brain, lungs, liver, and kidneys. The U.S. Environmental Protection Agency (EPA) has classified S. salivarius UBSS-01 as a four-category concern due to its LD50 being larger than the 5000 mg/kg 35 category, which possesses a relatively low potential for acute toxicity hazard. No-observed-adverse-effect level (NOAEL) for S. salivarius UBSS-01 was found to be 5000 mg/kg (500 billion CFU).
28-Day Repeat Dose Oral Toxicity Testing of S. salivarius UBSS-01
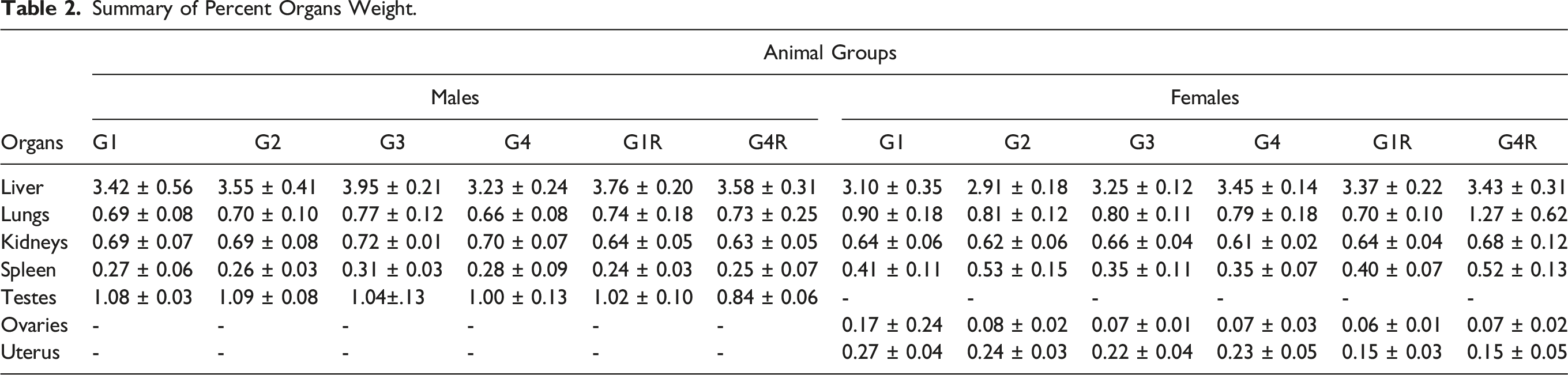
Summary of Percent Organs Weight.
Effect of 28-Day Repeat Dose S. salivarius UBSS-01 on Hematological Parameters
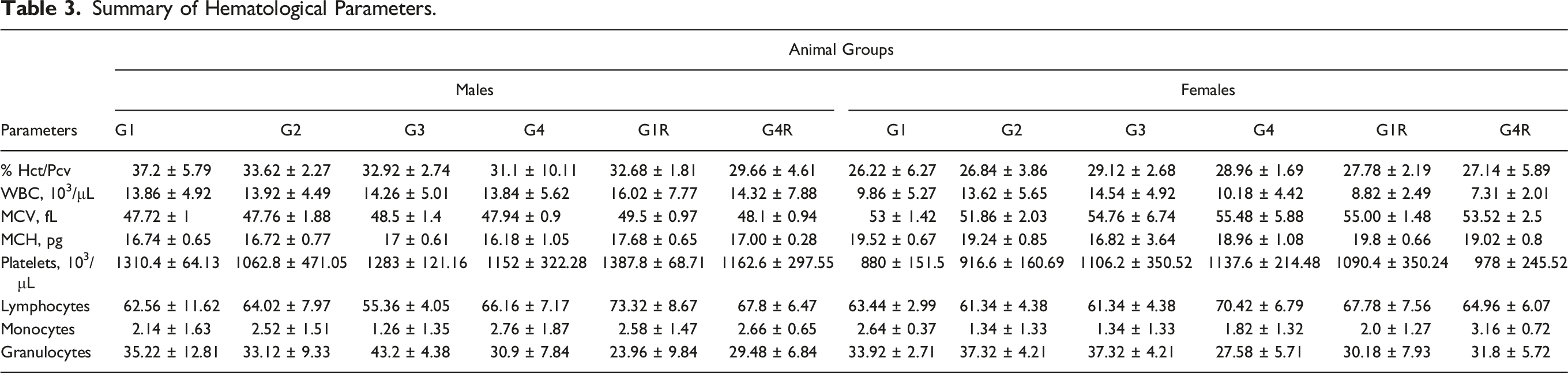
Summary of Hematological Parameters.
Effect of 28-Day Repeat Dose S. salivarius UBSS-01 on Biochemical Parameters and Electrolytes
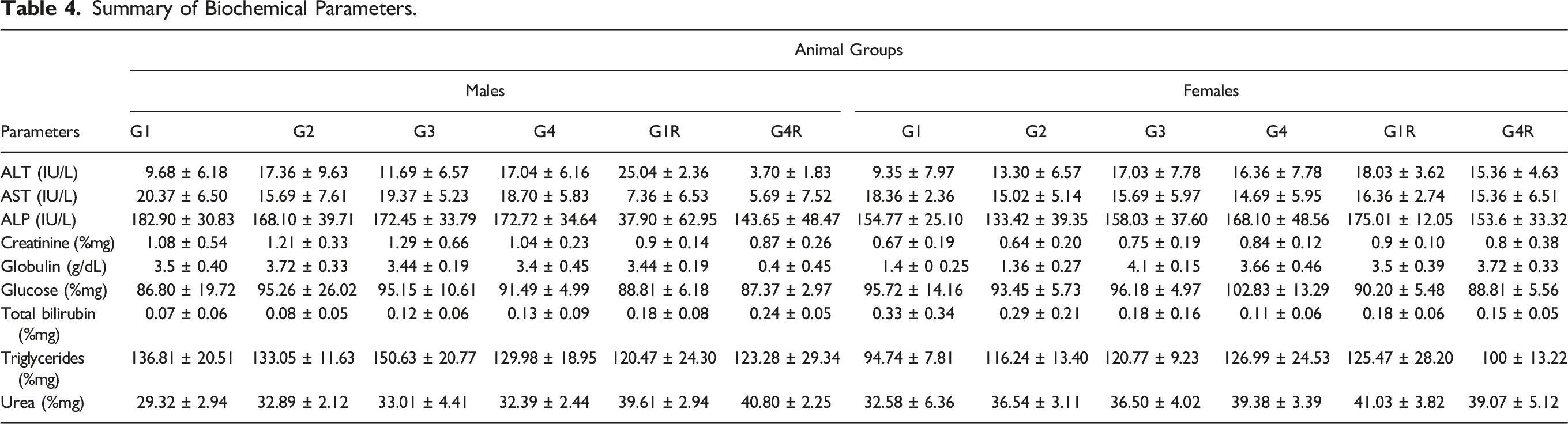
Summary of Biochemical Parameters.
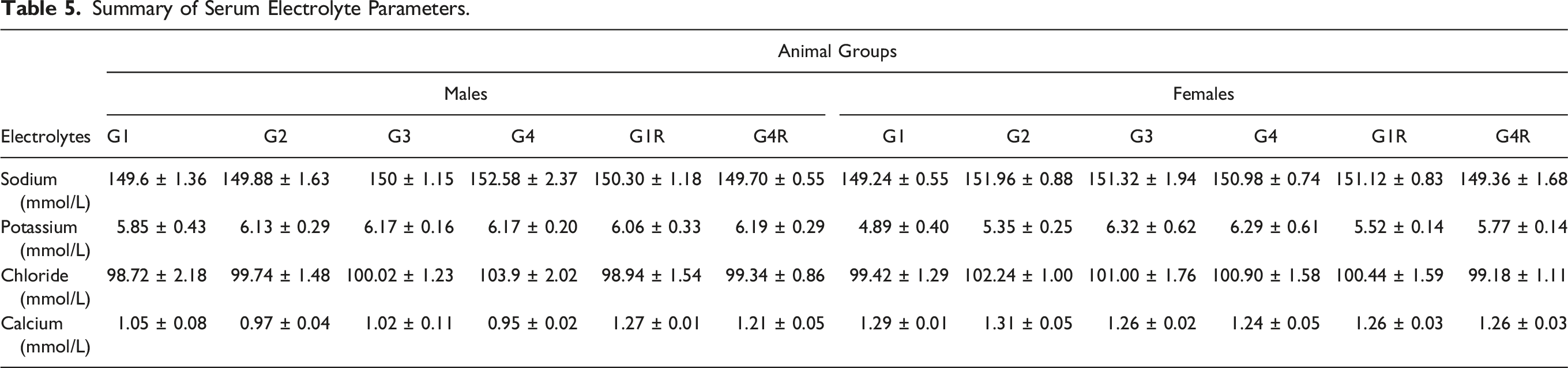
Summary of Serum Electrolyte Parameters.
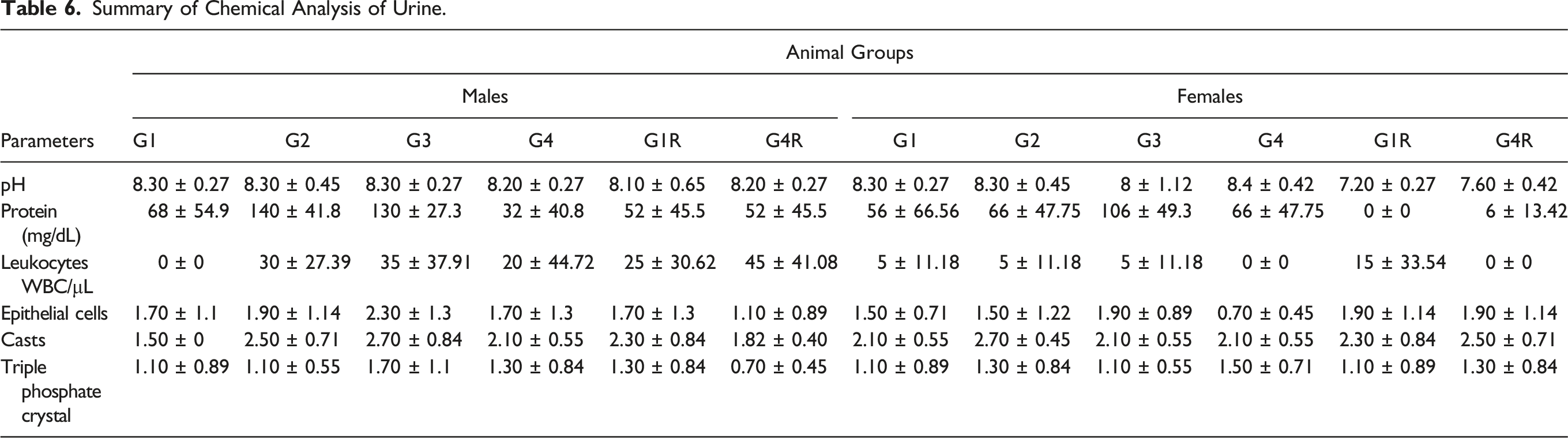
Summary of Chemical Analysis of Urine.
Histopathological Observation After Repeated S. salivarius UBSS-01 Administration
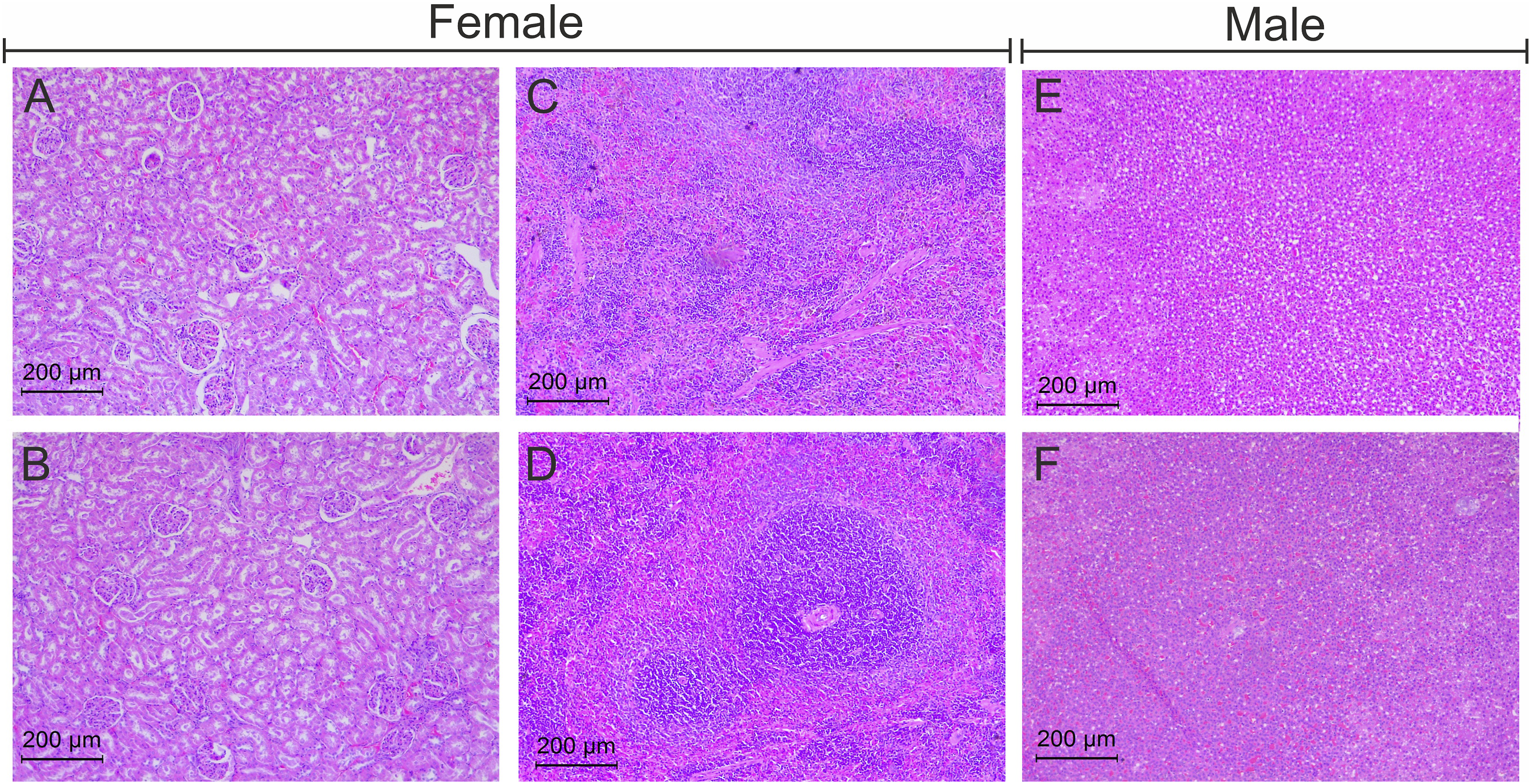
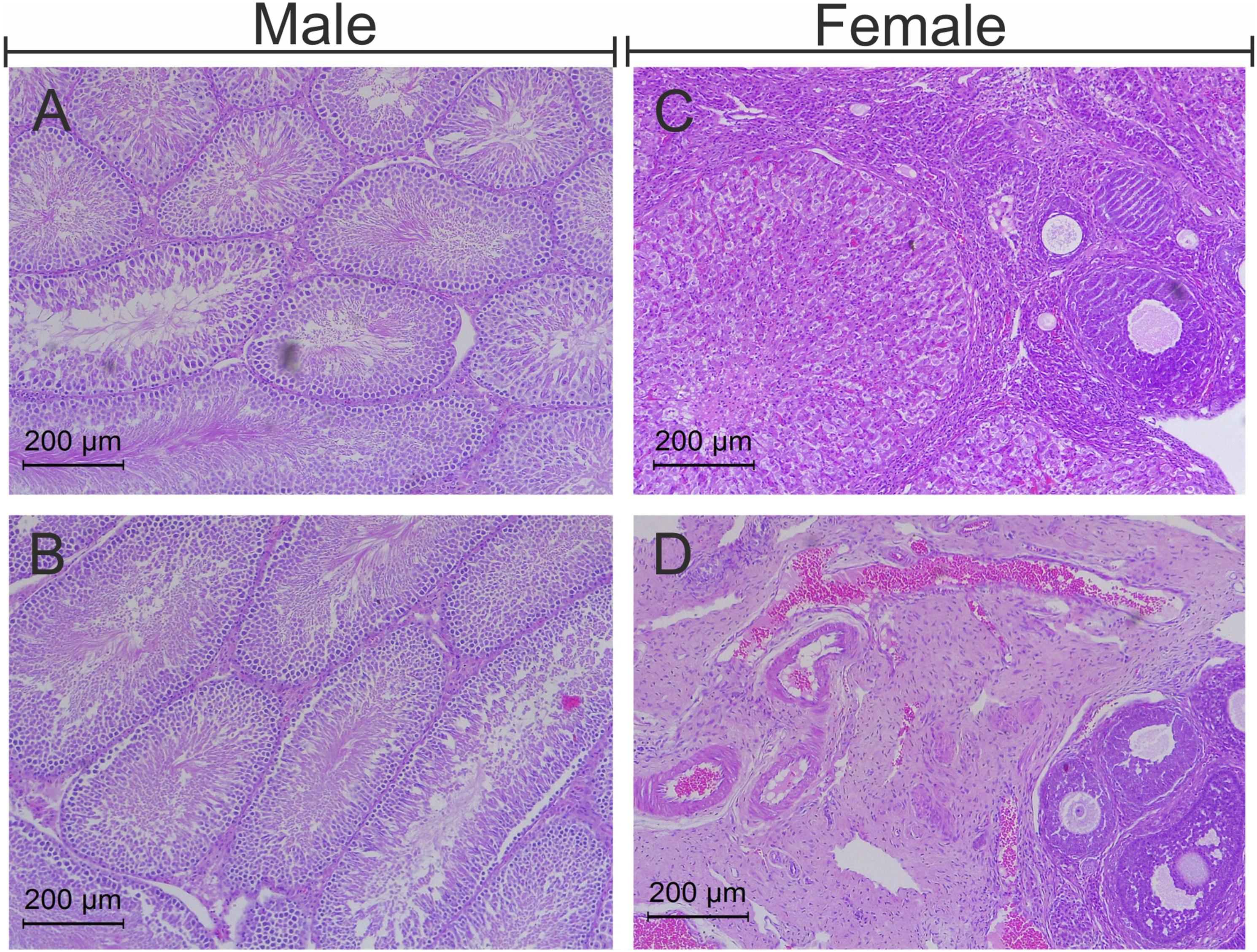
In the histological examination (by H&E staining) of vital organs, we observed no neuronal abnormality in the brain (Figure 4B), no hepatocellular damage in the liver (Figure 4D), no evidence of pulmonary edema and inflammation in the lungs (Figure 4F), and no sign of necrosis in the heart (Figure 4H) after high dose (1000 mg/kg) treatment with S. salivarius UBSS-01 as compared to the vehicle group (Figures 4A, C, E, and G). Moreover, no sign of toxicity was seen in the kidneys (Figure 5B), adrenal gland (Figure 5D), and spleen (Figure 5F) in the high-dose probiotic treatment group as compared to vehicle (Figures 5A, C, and E). No change was seen in reproductive organs such as the testis (Figure 6B) and ovary (Figure 6D) of probiotic animals as compared to control (Figures 6A and C). The representative H&E stained photomicrographs of the brain (A, B), liver (C, D), lungs (E, F), and heart (G, H) of vehicle-treated and high-dose, i.e, 1000 mg/kg S. salivarius UBSS-01 group, respectively. Scale bar = 200 μm and 500 μm. The representative H&E stained photomicrographs of kidney (A, B), spleen (C, D), and adrenal glands (E, F) of rats. Scale bar = 200 μm. Histology photomicrographs of the testis (A, B) and ovaries (C, D) stained with H&E of S. salivarius UBSS-01 high-dose group (1000 mg/kg). Scale bar = 200 μm.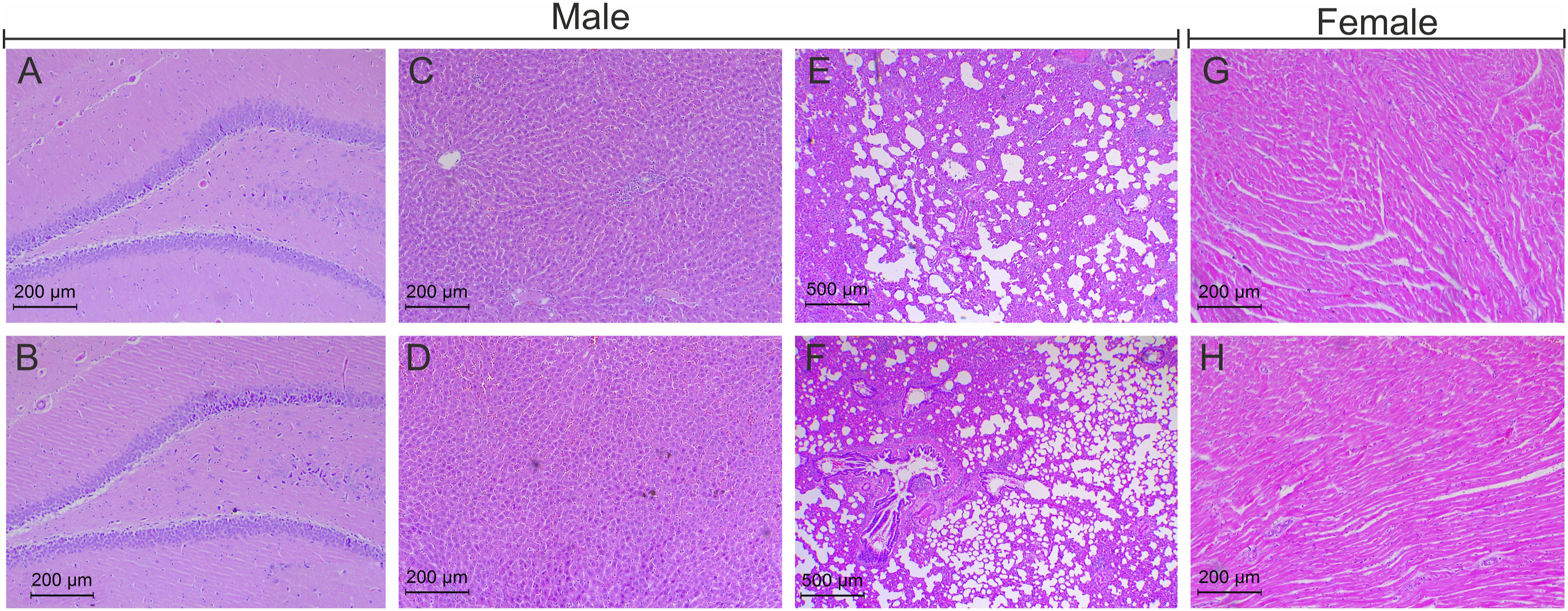
In Silico Study
Plasmid Detection
All the contigs were searched against nr database, and no significant hits were observed to the plasmid at an Evalue of 1e-5. Contigs were submitted to plasmid finder 1.3 which showed no plasmid availability.
Detection of Putative Virulence Factors
The putative virulence factors in the genome were determined by accessing the genome assembly available at the NCBI website against the Virulence Factor Database (VFDB) (Supplemental Table 2). Hemolysin BLB enterotoxin genes, non-hemolytic enterotoxin genes, and cytotoxic genes were found to be absent.
Clinical Study
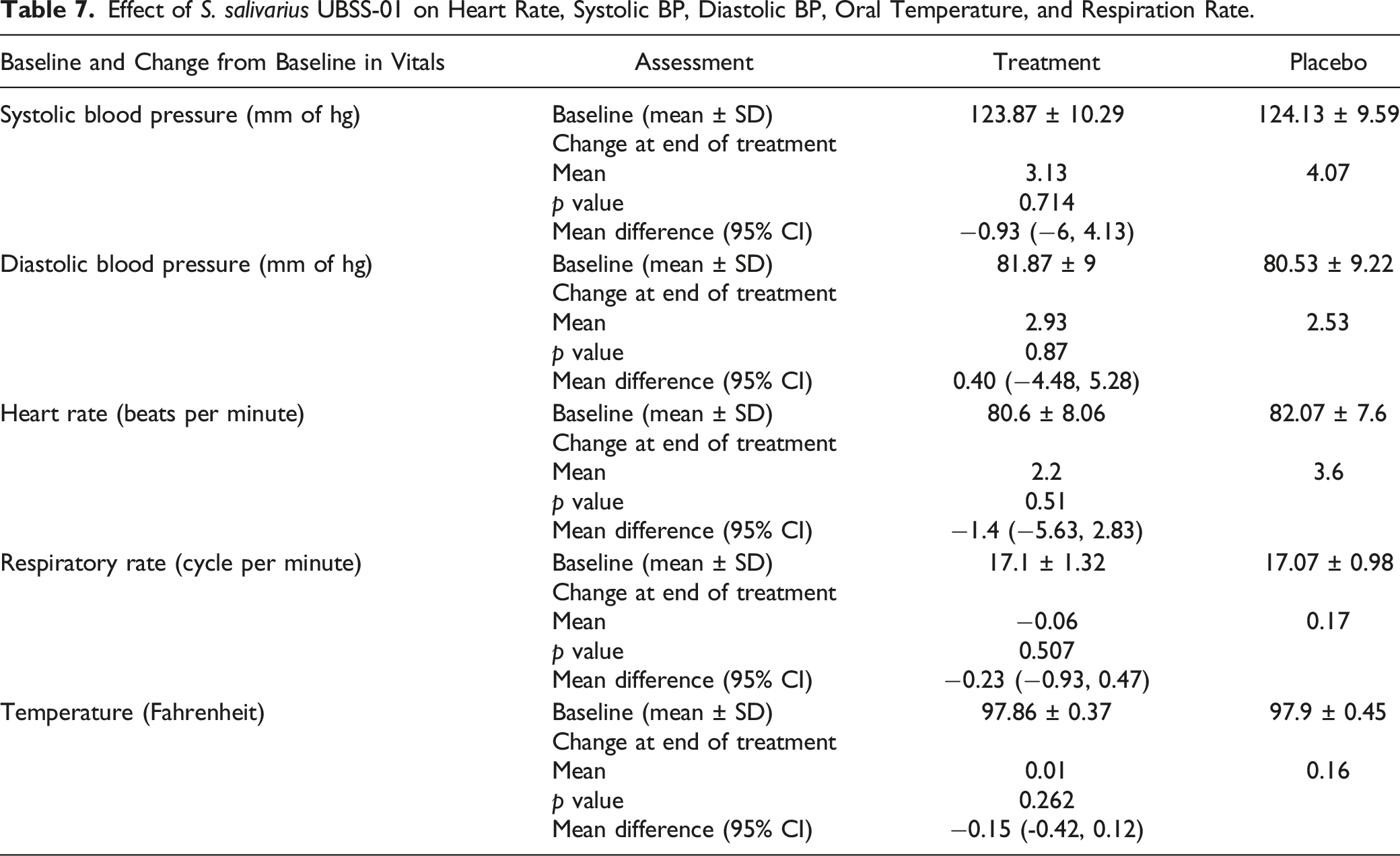
Effect of S. salivarius UBSS-01 Treatment on Vital Parameters
Effect of S. salivarius UBSS-01 on Heart Rate, Systolic BP, Diastolic BP, Oral Temperature, and Respiration Rate.
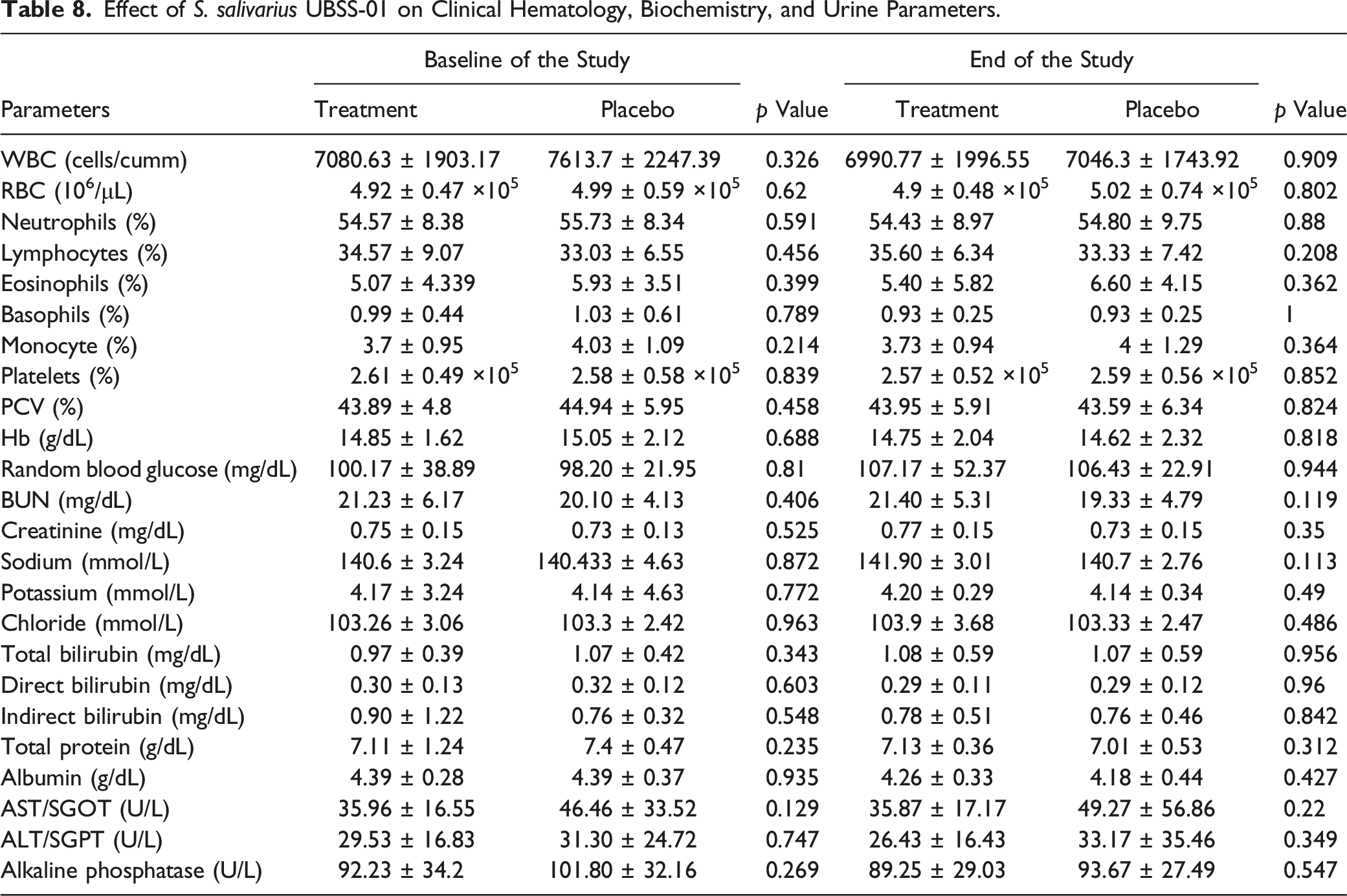
Effect of S. salivarius UBSS-01 treatment on clinical hematology, biochemistry, and urine parameters
Effect of S. salivarius UBSS-01 on Clinical Hematology, Biochemistry, and Urine Parameters.
Effect of S. salivarius UBSS-01 on Microscopic Parameters in Urine.
Adverse Events
Adverse events that were monitored were abdominal discomfort, nausea, burping, gas, change in bowel movements and stool consistency, heartburn, and fever. No serious adverse events were observed in either the probiotic or placebo group. Totally five subjects reported adverse events (AE), of which three subjects were from the placebo group (one subject reported abdominal pain for a day and two subjects reported loose stools for 1 day and 2 days, respectively) and two subjects from the probiotic-treated group (one subject had nausea for 1 day and the other subject reported loose stools for 1 day). The adverse events in both groups were not related to the treatment.
Effect of S. salivarius UBSS-01 Treatment on Gastrointestinal Symptoms (GSRS)
No significant changes (p > 0.05) were noted in the GSRS between S. salivarius UBSS-01 and placebo-treated groups. The mean GSRS score was found to be 0.7 ± 1.32 and 0.8 ± 1.29 in S. salivarius UBSS-01 and placebo groups, respectively, indicating the absence of gastrointestinal disturbances in both the groups.
Discussion
In this study, we evaluated the safety of the probiotic strain S. salivarius UBSS-01 through acute and 28-day repeat dose administration in rats, as recommended in OECD 423 and 407 toxicity guidelines, respectively. The aim of the acute toxicity study was to understand the preliminary toxicity, i.e. any morbidity and mortality, for selecting the correct dose for repeated dose toxicity study. In the acute study, a single-dose administration of S. salivarius UBSS-01, at 5000 mg/kg of body weight, did not produce any abnormal morphological changes with respect to skin color, and eye or mucous membrane secretions. The locomotor activity, animal gait, and body weight gain were similar in both vehicle- and probiotic-treated animals and no mortality was seen in the 15-day observation timeframe. Moreover, none of the male and female rats showed tremors, convulsions, drooling, diarrhea, or sleepiness. The lack of toxicity after a single administration of S. salivarius UBSS-01 indicates that NOEL is > 5 g/kg body weight in male and female rats. As stated in 2008 OECD guidelines, acute toxicity data is insufficient to portray the repeat-dose safety of investigational agents, therefore a repeat oral toxicity screening of S. salivarius UBSS-01 probiotic was performed for 28 days at the dose of 0.1, 0.5, and 1 g/kg per day.
No changes in the hematological parameters such as hemoglobin, Hct, RBC, WBC, MCV, MCHC, platelets count, clotting time, lymphocytes, monocytes, granulocytes, RDW-CV, RDW-SD, PCT, MPV, and PDW were observed in S. salivarius UBSS-01 treated animals at doses of 100, 500, and 1000 mg/kg as compared to control rats. These results indicate that S. salivarius UBSS-01 did not have any toxic effects based on the experimental model in the rats. Moreover, the number of biochemical markers such as total cholesterol, triglycerides, glucose, albumin, total protein, SGPT, SGOT, alkaline phosphatase, total bilirubin, creatinine, and urea, were also within the reference and baseline range. 36 Serum biochemical markers are used to identify organ-related issues, and hematological parameters are considered indications of inflammation and infection status. 37 We did not observe any alterations in the ALT and AST liver enzymes and bilirubin, which are key indicators of liver functions, 38 following 28-day S. salivarius UBSS-01 treatment and 14 days of recovery. Moreover, no discernible differences were observed in hematological and/or serum biochemical parameters of urea, creatinine, sodium, potassium, and chloride electrolytes in S. salivarius UBSS-01 treated rats compared to that in control groups. Thus, we may not expect any negative effects of S. salivarius UBSS-01 on renal functions, heart illness, muscle problems, nerve problems, and diabetes. 39 Similarly, in earlier studies, other strains of S. salivarius were found non-pathogenic and safe in humans and animals.11,40 Moreover, several bacteriocin-producing S. salivarius strains that inhibited the growth of pathogenic bacteria have been designated as safe probiotics and used in food products.41-44
In 28-day repeat dose oral toxicity studies, changes in organ weights can also indicate the harmful impacts of a test compound. 38 The relative weights of the brain, heart, lungs, liver, spleen, kidneys, adrenal glands, thymus, testes, epididymis, uterus, and ovaries in treated groups compared to control groups were not significantly different in this study. This demonstrated that S. salivarius UBSS-01 had no adverse effects on any organs. This was further confirmed by organ histological analyses, which showed no abnormalities or histopathological alterations in the organs. It was determined that S. salivarius UBSS-01 had no adverse effects on internal organ functions. Organ weight was not substantially different across the treatment groups. Over the course of the test, weight increase assessments showed that all male and female categories were highly equivalent. The repeat-dose oral toxicity investigation revealed that S. salivarius UBSS-01 did not show any toxicity when fed to rats at a concentration as high as 1000 mg/kg BW/day.
During the course of the 28-day repeat dose oral toxicity study, no appreciable differences between S. salivarius UBSS-01 treated groups and control in terms of body weight, general and behavioral conditions, relative organ weights, hematological and serum biochemical parameters, gross pathological and histopathological changes were observed. In an earlier study, S. salivarius K12 also showed no sign of toxicity in animals and humans with daily doses of 1 billion CFU. 45 The overall finding was that S. salivarius K12, when administered as a single bolus to rats, did not evoke any dose-related toxicity. Based on our results, we suggest that rats administered with acute and 28-day recurrent doses of S. salivarius UBSS-01 showed no toxicity. Moreover, as depicted in previous studies the investigational compounds rarely showed negative effects in a 90-day repeat dose study (OECD 408), if it is safe in the case of a 28-day repeat dose paradigm (OECD 407). 46
Apparently, genes for antibiotic resistance (ARGs) are known to be present in the microbes and can spread horizontally to pathogens, leading to the development of bacteria that are resistant to drugs.47,48 The preclinical and clinical data proposed that the ARGs can transfer from infections to commensal microbiota.49-51 Given the possibility that the S. salivarius UBSS-01 probiotic could transfer genes contributing to the development of antibiotic resistance, we conducted a comprehensive in silico genome mining for antibiotic resistance genes using two distinct strategies. The results indicated that S. salivarius UBSS-01 does not contain any antibiotic-resistance genes that could become mobile.
The propensity for toxigenicity and pathogenicity is another potential issue with live bacteriotherapy. Fifteen VFDB-positive genes were discovered by an in silico analysis of the S. salivarius UBSS-01 genome, and these genes’ putative virulence roles were further examined. The entire set of genes were examined such as heat shock proteins or genes involved in the manufacture of surface components. 8 S. salivarius UBSS-01 has no virulence potential, according to the evaluation of all other genes that have been positively identified using VFDB.
S. salivarius UBSS-01 did not produce any biogenic amines which are linked to toxicity. Certain probiotic strains possess specific decarboxylase enzymes that produce biogenic amines from food and dairy products which lead to toxic effects associated with food poison like rashes, hypertension, and difficulty in breathing. Additionally, in the hemolytic assay on Columbia blood agar plates, no zones around colonies indicated that it was γ-hemolysis, and strains exhibiting γ-hemolysis are considered as safe. Lecithin, the primary component of cell membranes, is hydrolyzed by the enzyme lecithinase found in pathogenic bacteria, resulting in cell lysis. In the present study, no precipitation was observed with S. salivarius UBSS-01 and hence it is lecithinase-negative and safe for consumption.
The safety and tolerability of S. salivarius UBSS-01 was also tested in healthy human volunteers. In the clinical study, consumption of 10 billion CFU of S. salivarius UBSS-01 every day for 30 days was well-tolerated in healthy persons. The consumption of S. salivarius UBSS-01 did not lead to any clinically significant health changes. There were no statistically notable differences in laboratory measurements observed between the intervention groups or visits, affirming its safety. These findings align with previous results concerning other strains of S. salivarius. No significant changes were noted in any of the parameters (vital signs, clinical chemistry and hematology parameters, gastrointestinal health, and urine analysis) between S. salivarius UBSS-01 (10 billion CFU daily for 30 days) and placebo-treated groups.
In conclusion, the results of the current study offer a piece of evidence for the safety and tolerability of S. salivarius UBSS-01 in preclinical studies and humans.
Limitations and Future Directions
While our in vivo experimental evidence of safety in the rat is limited to the use of acute and 28-day sub-chronic study models, studies of longer duration such as the 90-day sub-chronic or 180-day chronic study are warranted. Another study limitation is that the clinical study was carried out in a broad age range of 18–60 years, with a mean age of 35.10 ± 11.56 years. For vulnerable groups, such as infants, significantly more scientific experimental evidence of safety is needed than the limited toxicological studies.
Supplemental Material
Supplemental Material - Safety Assessment of Streptococcus salivarius UBSS-01 in Rats and Double-Blind Placebo-Controlled Study in Healthy Individuals
Supplemental Material for Safety Assessment of Streptococcus salivarius UBSS-01 in Rats and Double-Blind Placebo-Controlled Study in Healthy Individuals by Bindhu S. Jatoth, Ziaur Rahman, Manoj P. Dandekar, Rajesh Venkataraman, Ravi K. Shivalingegowda, and Gloriya G. Manuel in International Journal of Toxicology
Footnotes
Acknowledgments
The authors (BJ, ZR, and MPD) would like to thank NIPER Hyderabad, and the Ministry of Chemicals and Fertilizers, Government of India, for providing all the resources.
Author Contributions
Jatoth, B.S., contributed to design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Rahman, Z., contributed to acquisition, analysis, and interpretation and drafted manuscript; Dandekar, M.P., contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Venkataraman, R., contributed to conception and drafted manuscript; Shivalingegowda, R.K., contributed to conception and drafted manuscript; Manuel, G.G., contributed to conception and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Unique Biotech Ltd. Hyderabad, India funded the study.
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
