Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of 10 alkane diol ingredients as used in cosmetics. The alkane diols are structurally related to each other as small diols, and most are reported to function in cosmetics as solvents. The Panel reviewed the relevant data for these ingredients, and concluded that seven alkane diols are safe in cosmetics in the present practices of use and concentration described in this safety assessment, but that the available data are insufficient to make a determination of safety for three ingredients, namely 1,4-Butanediol, 2,3-Butanediol, and Octanediol.
Keywords
Introduction

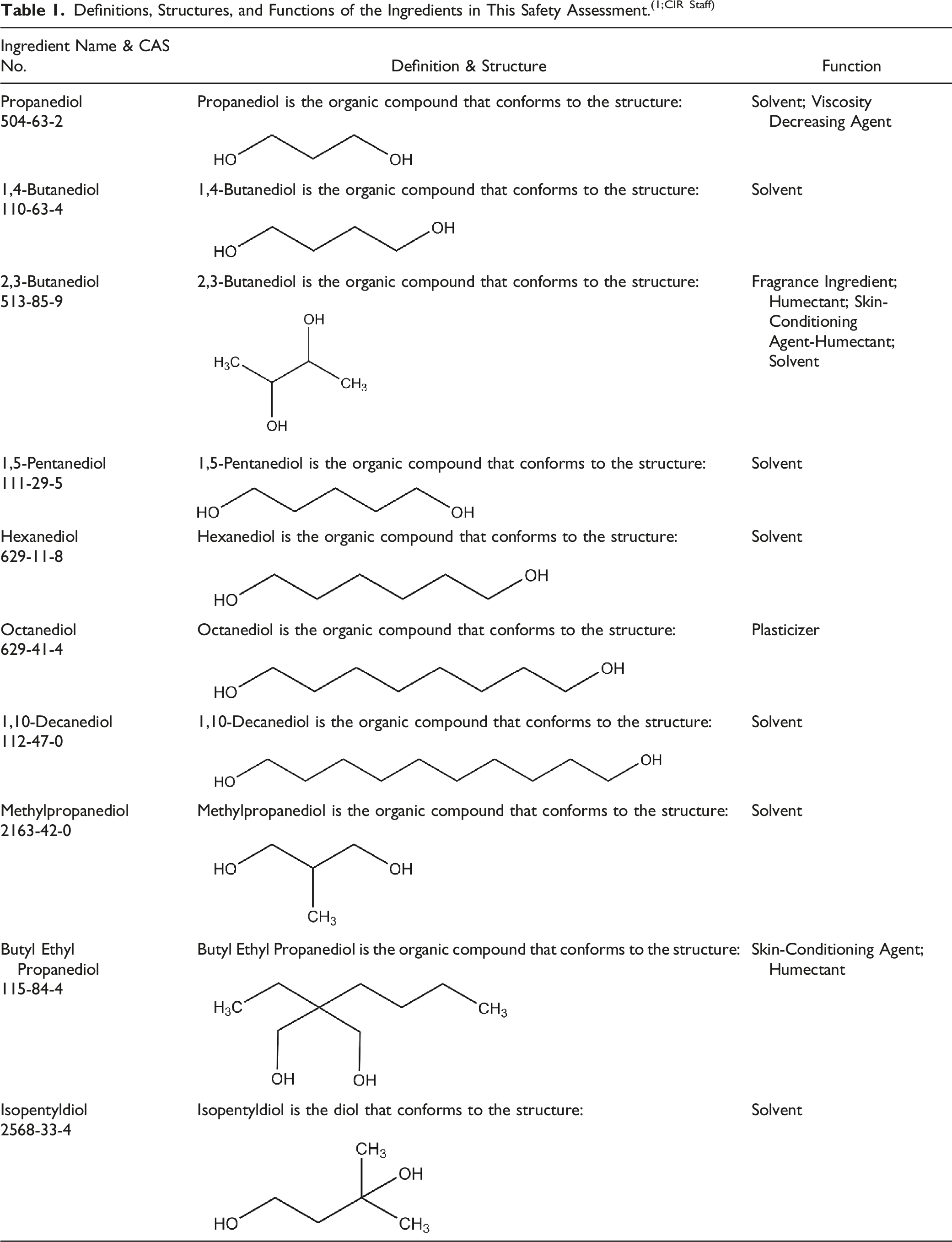
Definitions, Structures, and Functions of the Ingredients in This Safety Assessment.(1;CIR Staff)
Aliphatic Diols and Constituent Acids Previously Reviewed by the Panel.
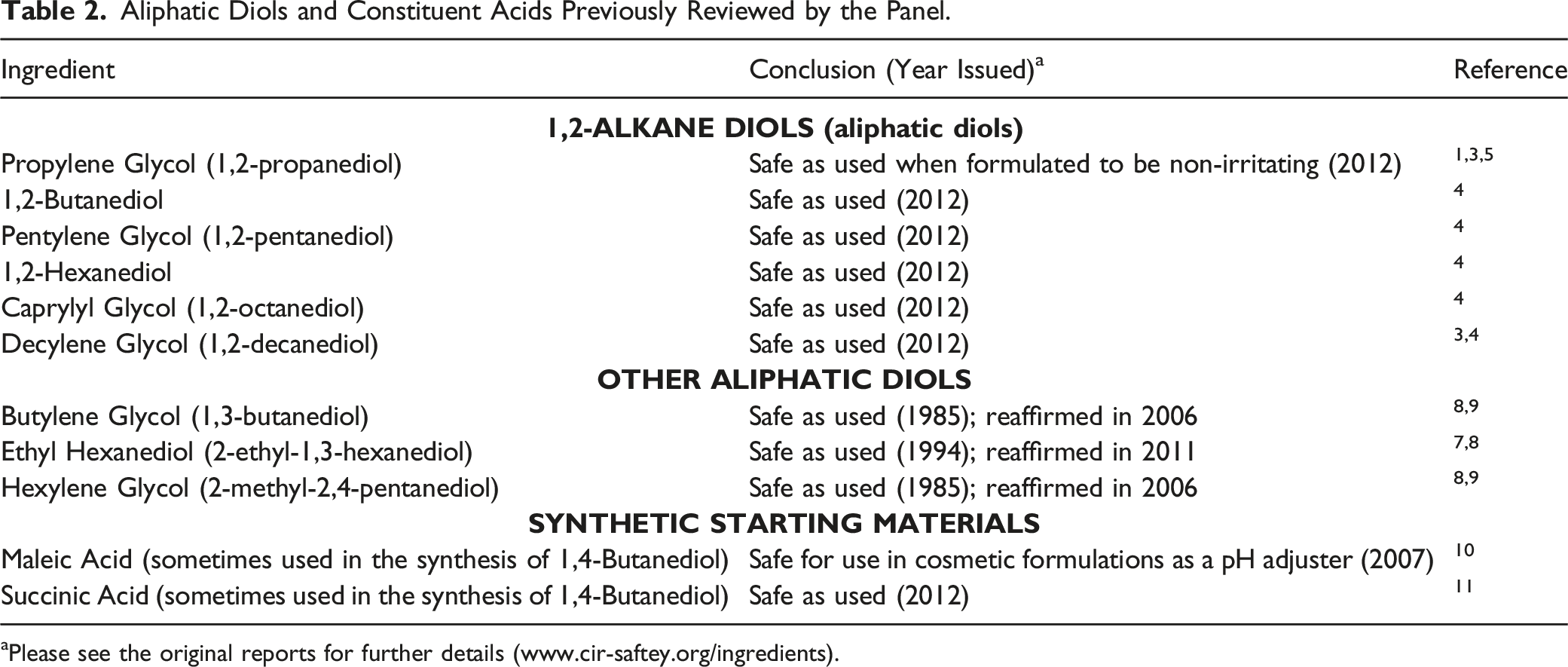
aPlease see the original reports for further details (www.cir-saftey.org/ingredients).
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
The European Chemicals Agency (ECHA)12-17 website and the Australian Government Department of Health National Industrial Chemicals Notification and Assessment Scheme (NICNAS)18-20 website provide summaries of data generated by industry, and ECHA and NICNAS are cited as the sources of the summary data in this safety assessment as appropriate. Also referenced in this safety assessment are summary data found in reports published by the World Health Organization (WHO), 21 the Organization for Economic Co-operation and Development Screening Information Data Sets (OECD SIDS), 22 and in reports made publicly available by the United States (US) Food and Drug Administration (FDA),23-27 the US Environmental Protection Agency (EPA),2,28-32 and through the National Technical Information Service (NTIS).33-37
Chemistry
Definition and Structure
All of the ingredients in this report are structurally related to each other as small diols (i.e., three to ten carbon alkyl diols). The ingredients in this report include regiochemistry other than 1,2-substitution. For example, 2,3-Butanediol is a vicinal diol with the first hydroxyl substitution at the 2-position and 1,4-Butanediol is a terminal diol with substitution at the 1- and 4-positions (Figure 1). 2,3-Butanediol and 1,4-Butanediol.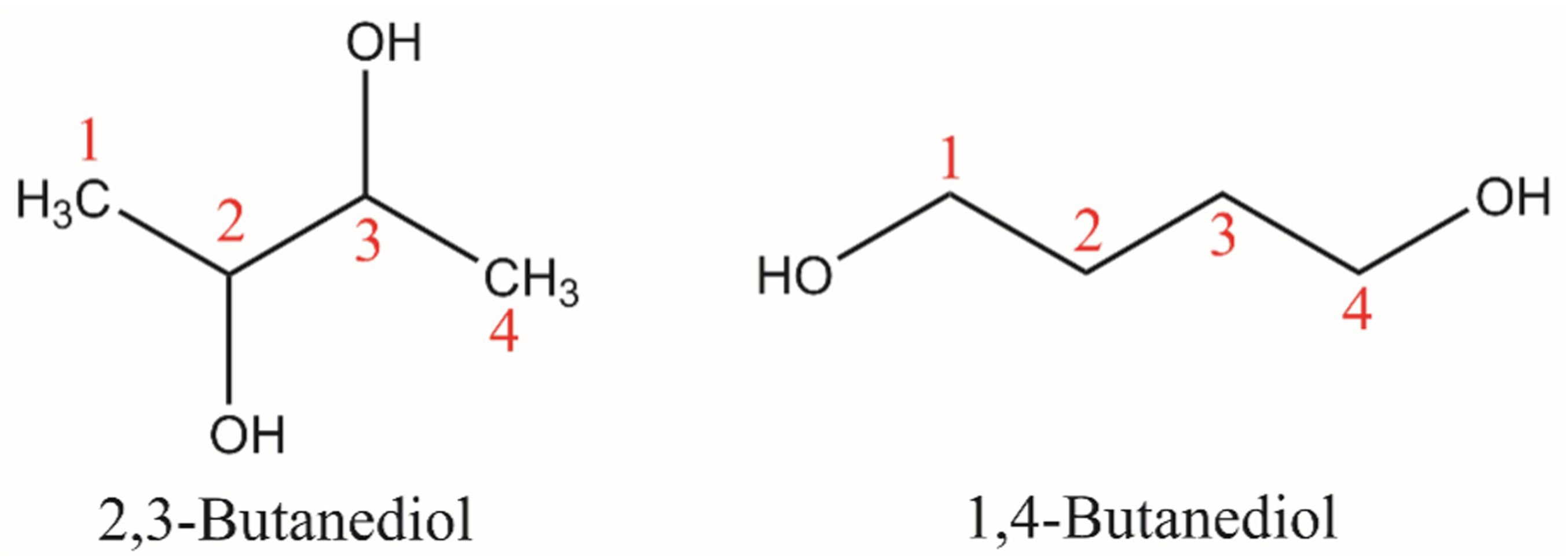
Variations in the regiochemistry of small alkane diols may lead to significant differences in toxicity. For example, 2,5-hexanediol, which is not a cosmetic ingredient, is known to be a neurotoxic metabolite of hexane.38,39 However, the structurally similar cosmetic ingredient, Hexanediol (i.e., 1,6-hexanediol), is not a neurotoxin.
Physical and Chemical Properties
Physical and Chemical Properties.

Method of Manufacture
Propanediol
Propanediol may be prepared by fermentation from corn-derived glucose using a biocatalyst (non-pathogenic strain of Escherichia coli K-12). 40 Propanediol can also be manufactured by heating γ,γ-dihydroxydipropyl ether with hydrobromic acid, followed by hydrolysis with sodium hydroxide. It is also reported to be obtained from plants that produce glycerol. 37
1,4-Butanediol
Some industrial chemical companies manufacture 1,4-Butanediol using cupric acetylide catalysts in the condensation reaction of acetylene with formaldehyde. 37 Some manufacturers convert propylene oxide to allyl alcohol, which is then hydroformylated to 4-hydroxybutyraldehyde, followed by reduction to the diol via hydrogenation. 21 Maleic acid and succinic acid can be used to manufacture 1,4-Butanediol via vapor phase hydrogenation of their corresponding esters and anhydrides. E. coli can be genetically engineered to metabolize sugar to 1,4-Butanediol. 41
2,3-Butanediol
2,3-Butanediol has been commercially produced by fermentation of molasses or sugar using Mesentericus, Aerobacter, Klebsiella, and Serratia bacteria; Bacillus polymyxa, Lactobacilli and Staphylococci strains and filamentous fungi (e.g., Rhizopus nigricans, Penicillium expansum) can also produce 2,3-Butanediol.
37
Fermentation of potatoes or wheat mash also yields 2,3-Butanediol. Mixtures of gases containing isobutylene and n-butenes, when combined with hydrogen peroxide and formic acid, yield a product containing 2,3-Butanediol, fractions of which are collected by distillation. The meso-form of 2,3-Butanediol can be prepared from trans-2,3-epoxybutane; the
1,5-Pentanediol
1,5-Pentanediol can be prepared in the presence of copper chromite via hydrogenolysis of tetrahydrofurfuryl alcohol. 42
1,10-Decanediol
1,10-Decanediol may be prepared by reducing diethyl or dimethyl sebacate with sodium metal in ethyl alcohol. It may also be prepared by catalytic hydrogenation of sebacic esters. 42
Methylpropanediol
On an industrial scale, carbon monoxide and hydrogen can be used to hydroformylate allyl alcohol to the intermediate, hydroxymethylpropionaldehyde, which is then hydrogenated to yield Methylpropanediol. 2
Impurities
Propanediol
The following Food Chemicals Codex acceptance criteria apply for Propanediol in relation to food preparation: cobalt (≤1.0 mg/kg or 1 ppm); lead (≤1.0 mg/kg or 1 ppm); nickel (≤1.0 mg/kg or 1 ppm).2,40 The purity of Propanediol should be ≥99.9% and water content should be ≤.1%. A manufacturer reported Propanediol to be 99.8% pure (impurities were not provided) and stated that the product did not contain added preservatives, animal by-products, or petroleum ingredients. 43 Propanediol was reported to be ≥99.98% pure; water was listed as an impurity, but no heavy metals, monomers, or amines were known to be present. 44
1,4-Butanediol
Maleic acid and succinic acid may be potential residual impurities of 1,4-Butanediol because they are sometimes used as starting materials in the manufacture of this ingredient, as mentioned above. 21 1,4-Butanediol has been reported to be 98% pure. 22
1,5-Pentanediol
1,5-Pentanediol was found to be 98.1% pure by gas chromatographic/mass-spectrometry analysis; a total of .28% unknown impurities (not diols, as stated by the study authors) were reported. 45 Contamination by water, 1,5-hexanediol, and 1,6-Hexanediol was found to be .02, 1.02, and .56%, respectively. Other diol impurities, including 1,4-Butanediol, 2,5-Hexanediol, and cyclic diols, were below the limit of detection (<.05%).
Hexanediol
Hexanediol has been reported to be >96% pure (impurities were not specified). 46
Methylpropanediol
Methylpropanediol has been reported to be 98% pure (maximum 2% impurities; maximum .1% water content, maximum .05% carbonyl content) by a manufacturer. 47
Isopentyldiol
Isopentyldiol has been reported to be 97% pure with 3% of impurities (no further details provided). 19 Isopentyldiol is >99% pure as reported by a cosmetics raw material supplier. 48
Natural Occurrence
2,3-Butanediol
2,3-Butanediol occurs naturally in certain foods, some examples include “.006 mg/kg in fish (lean), up to 90 mg/kg in cheddar cheese, up to 2.3 mg/kg in raspberries, up to 850 mg/kg in vinegar, 1.9 mg/kg in sherry, and up to 2900 mg/kg in various types of wine.” 49
Use
Cosmetic
The Panel evaluated the safety of the cosmetic ingredients included in this assessment based on the expected use of and potential exposure to the ingredients in cosmetics. The data received from the US FDA are collected from manufacturers through the FDA Voluntary Cosmetic Registration Program (VCRP), and include the use of individual ingredients in cosmetics by cosmetic product category. The data received from the cosmetic industry are collected by the Personal Care Products Council (Council) in response to a survey of the maximum reported use concentrations by product category.
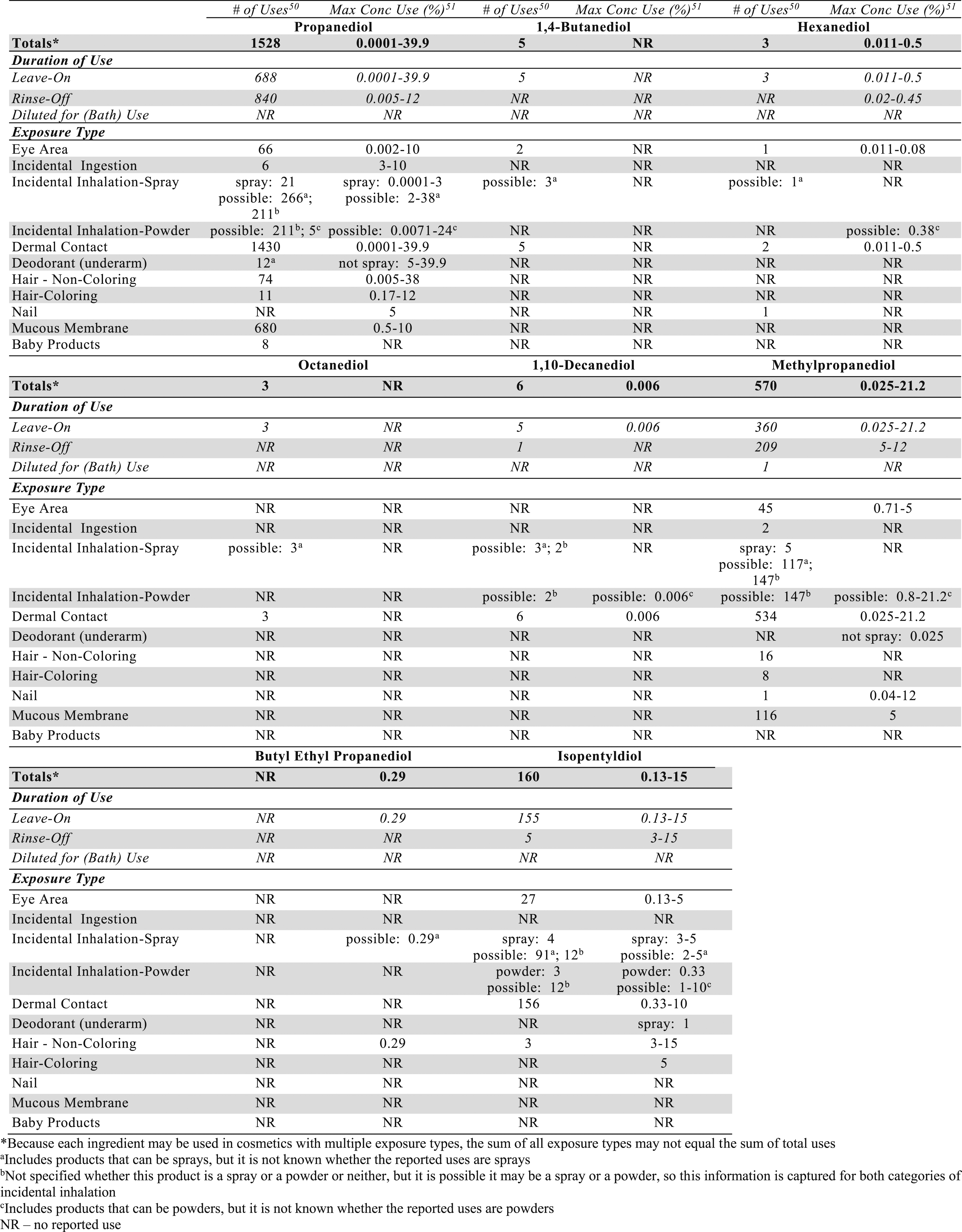
Frequency and Concentration of Use of Alkane Diols.
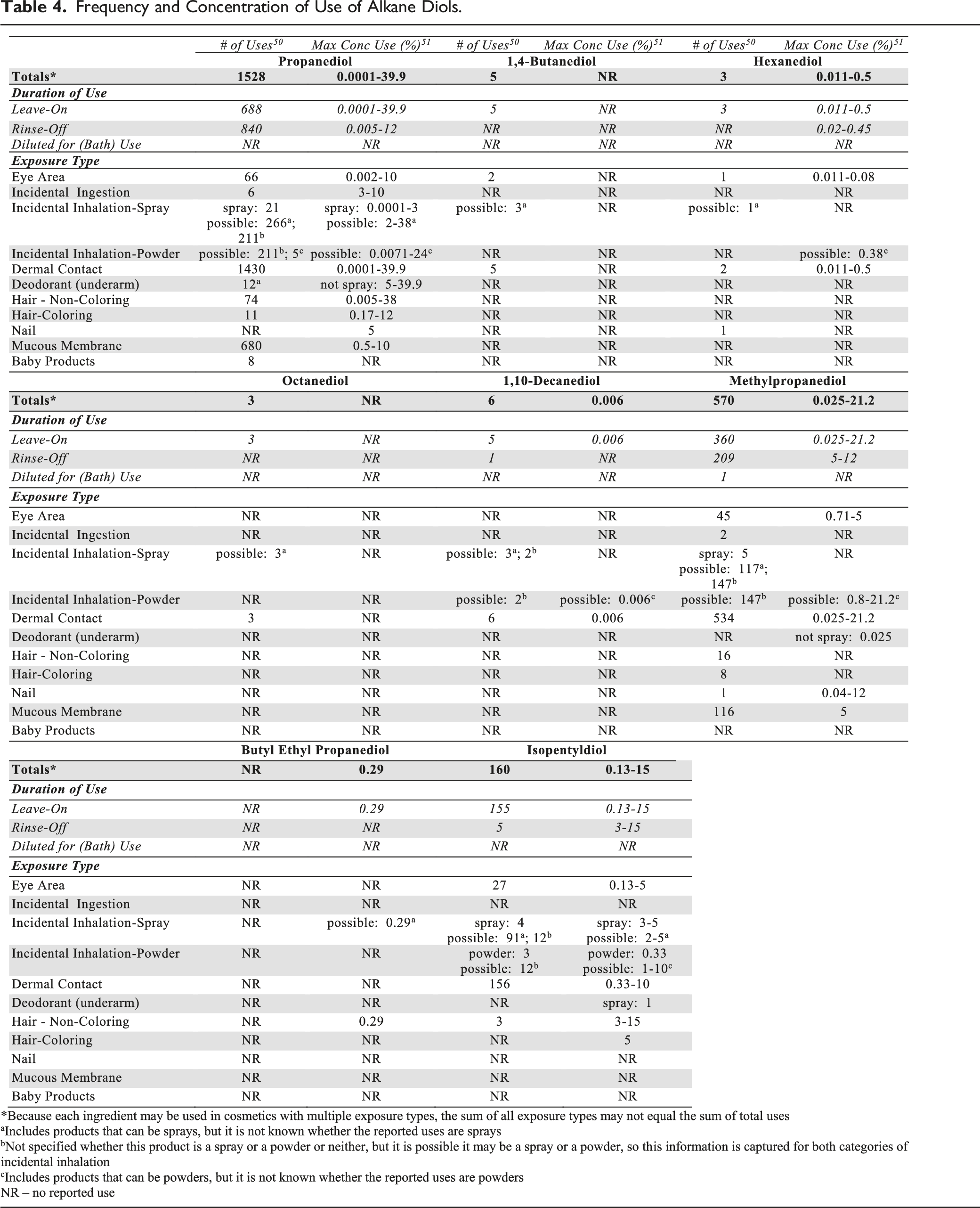
In some cases, uses of alkane diols were reported in the VCRP, but concentration of use data were not provided in the Council survey. For example, 1,4-Butanediol is reported to be used in 5 cosmetic formulations, but no use concentration data were reported. 50 Conversely, there was an instance in which no uses were reported in the VCRP, but use concentrations were provided in the industry survey; Butyl Ethyl Propanediol was not reported to be in use in the VCRP, but the Council survey indicated that it is used at concentrations of .29% in tonics, dressings and other hair grooming aids. 51 It should be presumed that there is at least one use in this category.
There are no frequency or concentration of use data reported for 2,3-Butanediol or 1,5-Pentanediol.50,51
Alkane diols were reported to be used in cosmetic sprays, including perfumes, hair sprays, and deodorants, and could potentially be incidentally inhaled. For example, Propanediol was reportedly used in aerosol and pump hair sprays at concentrations up to .12 and 1.5%, respectively, and it was used in face and neck sprays at concentrations up to 3%. 51 Isopentyldiol was reportedly used in perfumes and aerosol deodorants at concentrations up to 5% and up to 1%, respectively. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays.52-55 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.52,54 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 54 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays. Isopentyldiol was reportedly used in face powders at concentrations up to .33%, 51 and could possibly be inhaled. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.56-58
Some alkane diols were reported to be used in cosmetic formulations indicative of potential eye exposure (e.g., Propanediol is used at up to 10% in eye makeup removers) and possible mucous membrane exposure and ingestion (e.g., Propanediol at up to 10% in dentifrices). Propanediol was reported to be used in baby shampoos and baby lotions, oils, powders, and creams (concentrations of use were not reported).
None of the alkane diols named in this report are restricted from use in any way under the rules governing cosmetic products in the European Union. 59 In a NICNAS report, Isopentyldiol was determined not to be an unacceptable risk to public health in cosmetic products up to 10% (the highest use concentration reported in the NICNAS document). 19
Non-Cosmetic
U.S. Permitted Non-Cosmetic Uses.

1,4-Butanediol
1,4-Butanediol is known to be an illicit drug of abuse because of its conversion to gamma-hydroxybutyric acid (GHB) after oral administration. 60 GHB, occurring endogenously in mammals, is a neurotransmitter with a high affinity for pre- and postsynaptic neuron GHB-receptors.60,61 In 1999, the FDA issued a warning about products (i.e., dietary supplements advertised as a sleep aid) containing 1,4-Butanediol and gamma-butyrolactone because of reports linking these compounds to adverse health effects (eg, decreased respiration) and 3 deaths. In this warning, the FDA noted 1,4-Butanediol to be a Class I Health Hazard (potentially life-threatening risk). GHB has been used in dietary supplements because it can reportedly increase physiological concentrations of growth hormone, leading to an increase in lean muscle mass; weight control and sedation were other effects of GHB ingestion advertised by health food stores.28,61 In 1997, the FDA re-issued a warning for GHB used recreationally and in body building because it caused serious adverse health effects. 28 As of 2000, the Drug Enforcement Agency (DEA) reported GHB to be a Schedule I Controlled Substance and 1,4-Butanediol and gamma-butyrolactone to be controlled substance analogs if they are intended for human consumption pursuant to 21 USC §§802(32)(A) and 813.28,60 Sodium oxybate (the sodium salt form of GHB) is an FDA-approved prescription drug product (schedule III controlled substance) 60 used to treat attacks of muscle weakness and daytime sleepiness in narcolepsy patients.24,25 The warnings and regulatory actions listed above pertain to oral administration.
Pentylene Glycol
Pentylene Glycol is listed as an ingredient in a prescription hydrogel wound dressing (medical device classified under 21CFR878.4022), which was cleared by the FDA (Section 510(k)).27,30 Sources did not specify whether 1,2-Pentanediol or 1,5-Pentanediol was used or the concentration used.
1,5-Pentanediol
1,5-Pentanediol has been reported to have antimicrobial and antifungal properties in pharmaceutical applications.45,62-64 Additionally, 1,5-Pentanediol has reported uses in products for hair loss, cold sores, nail problems, dry and scaly feet, and eczema; it can be used as a moisturizing substance and solvent. 64 According to information submitted to CIR, 1-5 Pentanediol is used at concentrations of 5 - 25% in products used for baldness, dandruff, cold sores, nail fungus infections, and for the treatment of foot problems. 65
Toxicokinetic Studies
Dermal Penetration
In vitro
Propanediol
A dermal penetration study conducted using human cadaver skin evaluated the penetration of Propanediol. 12 The stratum corneum (abdominal region of human cadaver skin, n = 6 representing 3 donors) was mounted on an in vitro static diffusion cell (skin surface area .64 cm2). The experiment was conducted using Good Laboratory Practice (GLP) and in accordance with OECD Test Guideline (TG) 428 (Skin Absorption: in vitro Method). A solution containing 1.059 g/mL Propanediol (purity 99.953%) was applied to the skin (1200 µL/cm2, infinite dose) in the donor chamber (opening to chamber was occluded). The receptor fluid (.9% saline) was maintained at 32°C in a recirculating water bath and was sampled at time zero and every 4 - 6 hours up to 48 hours post-application. The permeability coefficient was calculated to be 1.50 × 10−5 cm/h, based on the slope at steady state (15.9 µg/cm2/h) and the concentration of Propanediol applied (test solution density 1,059,700 µg/cm3). The percentage of the applied Propanediol recovered from the receptor chamber 48 hours post-application was .12%.
Penetration Enhancement
In vitro
Penetration Enhancement Studies.
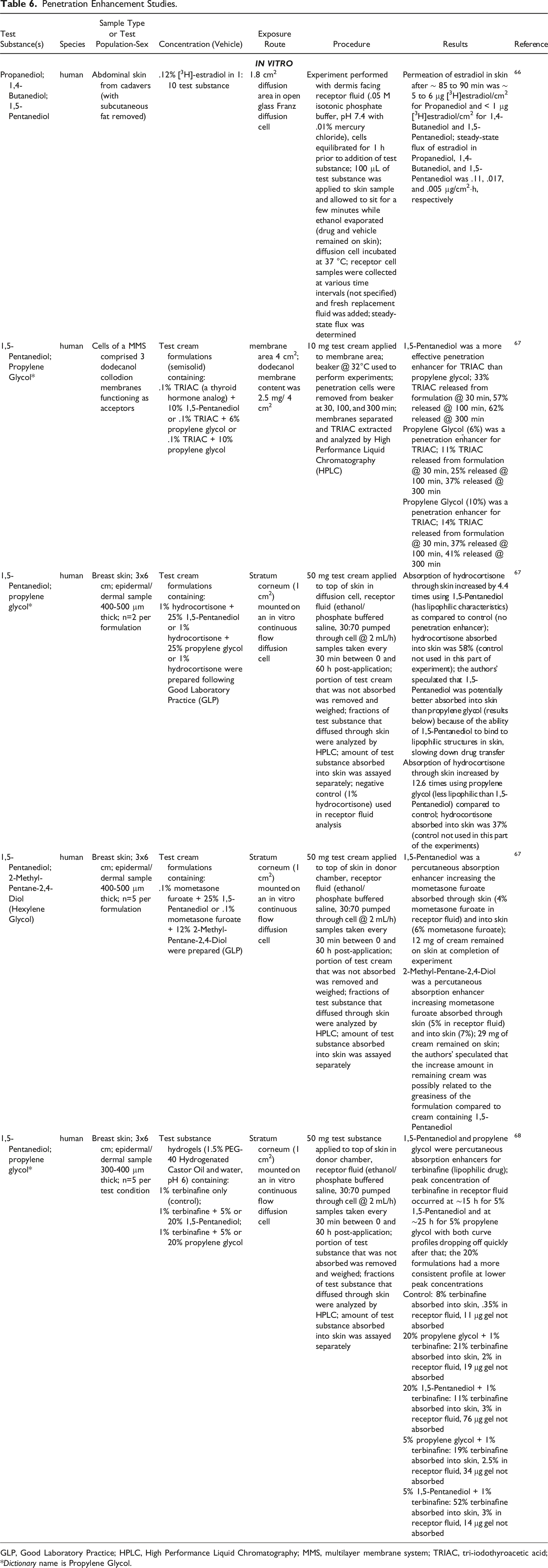
GLP, Good Laboratory Practice; HPLC, High Performance Liquid Chromatography; MMS, multilayer membrane system; TRIAC, tri-iodothyroacetic acid; *Dictionary name is Propylene Glycol.
The ability of Propanediol, 1,4-Butanediol, and 1,5-Pentanediol to enhance the penetration of the drug estradiol in human skin was evaluated in an in vitro experiment using a Franz diffusion cell; (.05 M isotonic phosphate buffer, pH 7.4 with .01% mercury chloride was used as the receptor fluid). 66 The test substance (100 µL of .12% [3H]-estradiol in 1:10 Propanediol, 1,4-Butanediol, or 1,5-Pentanediol/ethanol solution) was applied to the dermis, which faced the receptor side of the cell. Receptor fluid samples were collected at various time points. The steady-state flux of estradiol in Propanediol, 1,4-Butanediol, and 1,5-Pentanediol was determined to be .11, .017, and .005 µg/cm2/h, respectively, indicating a decrease in steady-state flux with increasing alkyl chain length. After ∼ 85 - 90 minutes the permeability of [3H]-estradiol in human skin was ∼ 5 - 6 µg/cm2 with Propanediol and <1 µg/cm2 with 1,4-Butanediol or 1,5-Pentanediol.
Penetration enhancement tests in vitro showed 1,5-Pentanediol to be a penetration enhancer for certain pharmaceutical drugs.67,68 Test cream formulations containing .1% triiodothyroacetic acid (TRIAC; a thyroid hormone analog) and either 1,5-Pentanediol (10%) or propylene glycol (10%) showed 1,5-Pentanediol to be a more effective penetration enhancer than propylene glycol for TRIAC in a multilayer membrane system (MMS) experiment. 67 Results for 1,5-Pentanediol indicated that 33% of the TRIAC (pharmacologically active agent) was released from the carrier vehicle, or formulation (in MMS), to enable TRIAC to contact the skin at the epidermal surface by 30 minutes post-application; 62% TRIAC was released from the formulation by 300 minutes. 67 In a separate experiment, test cream formulations containing 1% hydrocortisone and either 1,5-Pentanediol (25%) or propylene glycol (25%) were evaluated using human breast skin.
Both 1,5-Pentanediol (increased drug absorption 4-fold, compared to controls) and propylene glycol (increased drug absorption 13-fold, compared to controls) were shown to be penetration enhancers. 67 However, propylene glycol enhanced the transfer of the drug through the skin more effectively and 1,5-Pentanediol increased retention of the drug in the skin more effectively (receptor fluid [ethanol/phosphate buffered saline (PBS)] collected up to 60 hours post-application). Another experiment evaluating test cream formulations containing .1% mometasone furoate and either 1,5-Pentanediol (25%) or Hexylene Glycol (12%) revealed that both formulations were percutaneous absorption enhancers in human breast skin (receptor fluid [ethanol/PBS] collected up to 60 hours post-application). The absorption of .1% mometasone furoate into the skin was 6% using 1,5-Pentanediol and 7% using Hexylene Glycol as penetration enhancers.
1,5-Pentanediol (5% and 20%) and propylene glycol (5% and 20%) were also evaluated in an in vitro experiment investigating the penetration enhancement of 1% terbinafine, a lipophilic drug used to treat foot and nail fungus, in a hydrogel formulation. 68 Both alkane diols were found to be percutaneous absorption enhancers in human breast skin (receptor fluid [ethanol/PBS] collected up to 60 hours post-application). Results indicated that 21% and 11% terbinafine was absorbed into the skin with 20% propylene glycol or 20% 1,5-Pentanediol, respectively. The 5% propylene glycol or 5% 1,5-Pentanediol yielded 19 and 52% terbinafine absorption into skin, respectively. For comparison, the control (1% terbinafine in hydrogel without either alkane diol) resulted in 8% drug absorption into the skin.
Absorption, Distribution, Metabolism, Excretion
In mammals, 1,4-Butanediol is metabolized endogenously to gamma-hydroxybutyraldehyde by alcohol dehydrogenase and then by aldehyde dehydrogenase to GHB. 61 This metabolism has been reported to occur in rat brain and liver. 69 Ethanol, a competitive substrate for alcohol dehydrogenase, can inhibit 1,4-Butanediol metabolism.61,70 GHB is metabolized to succinic semialdehyde by GHB dehydrogenase, and then to succinic acid by succinic semialdehyde dehydrogenase; succinic acid then enters the Krebs cycle. 61 Alternatively, succinic semialdehyde can be metabolized by gamma-aminobutyric acid (GABA) transaminase to produce the neurotransmitter GABA.
Toxicokinetics Studies-Absorption, Distribution, Metabolism, Excretion (ADME).
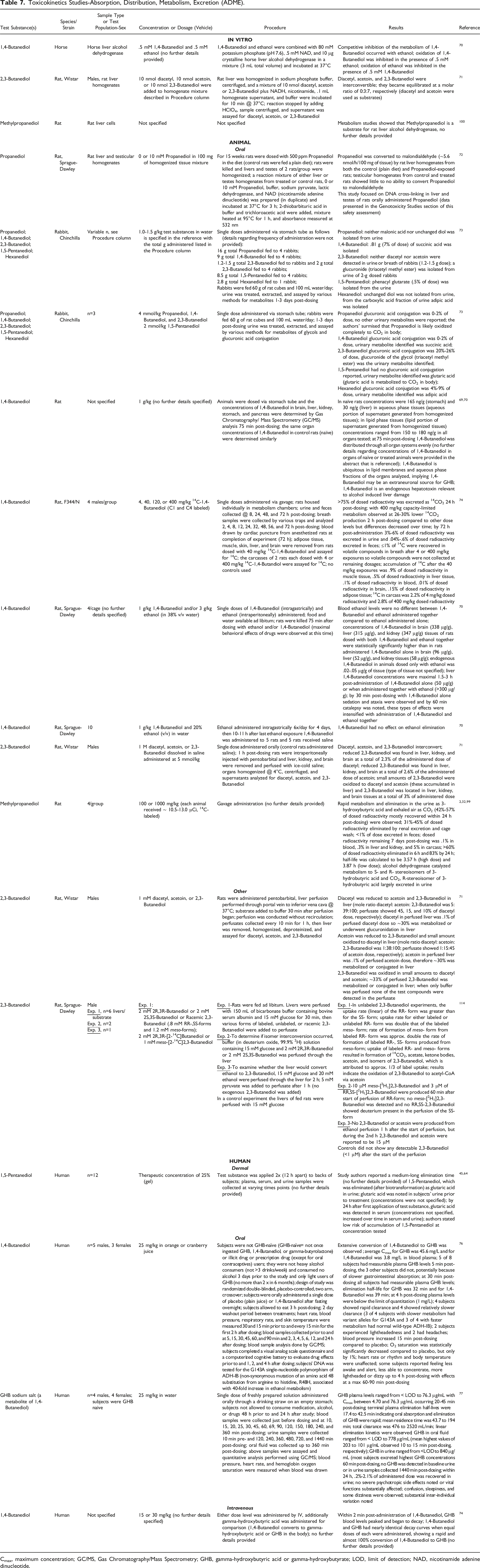
Cmax, maximum concentration; GC/MS, Gas Chromatography/Mass Spectrometry; GHB, gamma-hydroxybutyric acid or gamma-hydroxybutyrate; LOD, limit of detection; NAD, nicotinamide adenine dinucleotide.
In vitro
Competitive inhibition between 1,4-Butanediol (.5 mM) and ethanol (.5 mM) occurred in a test performed using horse liver alcohol dehydrogenase. 70 In rat liver homogenates, 10 nmol of diacetyl, acetoin, and 2,3-Butanediol were interconvertible with a molar equilibrium ratio of 0:3:7, respectively. 71 Methylpropanediol was a substrate for rat liver alcohol dehydrogenase. 2
Animal
Metabolism experiments conducted using liver homogenates from rats that were fed 500 ppm Propanediol in the diet for 15 weeks and control rats (fed a plain diet) revealed that Propanediol was converted to malondialdehyde (5.6 nmol/h/100 mg tissue) in the liver homogenates of Propanediol-exposed rats and controls, but little-to-no conversion occurred in the testicular homogenates of treated or control rats. 72 Experiments in rabbits administered single doses of alkane diols via stomach tube revealed metabolites isolated from the urine 1 to 3 days post-dosing. Propanediol glucuronic acid conjugation accounted for up to 2% of the administered dose (4 mmol/kg); 1,4-Butanediol (9 g) was metabolized to succinic acid (7% of administered dose); 2,3-Butanediol glucuronic acid conjugation accounted for up to 26% of the administered dose (4 mmol/kg); phenacyl glutarate (.5% of dose) was identified after 1,5-Pentanediol (8.5 g) administration; Hexanediol glucuronic acid conjugation accounted for up to 9% of the administered dose (2 mmol/kg) and adipic acid was detected. 73
Rats were intragastrically exposed to a single dose of 1 g/kg 1,4-Butanediol; 75 minutes post-dosing 96 µg/g were measured in the brain, 52 µg/g in the liver, and 58 µg/g in the kidney; endogenous levels of 1,4-Butanediol in rats dosed with ethanol were found to be .02 to .05 µg/g (type of tissue not specified), by comparison; 1,4-Butanediol levels in the liver peaked at 50 µg/g 1.5 to 3 hours post-dosing; sedation and ataxia were observed 30 minutes post-dosing and, by 60 minutes, catalepsy was noted (these effects were synergistically intensified when ethanol was concurrently administered). 70 In rats orally administered up to 400 mg/kg 1,4-Butanediol (radiolabels on C1 and C4), >75% of the radioactivity was excreted as [14C]-CO2 (by 24 hours post-administration), up to 6% of the radioactivity was excreted in urine (by 72 hours post-administration), and up to .6% of the radioactivity was excreted in feces (by 72 hours post-administration). 74 Endogenous concentrations of 1,4-Butanediol in rats were found to be 165 ng/g (stomach) and 30 ng/g (liver) in aqueous phase tissues (i.e., aqueous portion of supernatant of homogenized tissues) and in lipid phase tissues (i.e., lipid portion of supernatant of homogenized tissues) were 150 to 180 ng/g. 69
Experiments in rats orally administered 1 M diacetyl, acetoin or 2,3-Butanediol showed that these compounds interconvert. 71 Methylpropanediol orally administered to rats (100 or 1000 mg/kg, [14C]-labeled) was rapidly metabolized and eliminated in the urine as 3-hydroxybutyric acid (31% - 45% of dosed radioactivity), in the exhaled breath as CO2 (42 - 57% of dosed radioactivity), and in the feces (<1% of dosed radioactivity).32,75
In liver perfusion experiments in rats (in vivo), perfusion with 1 mM 2,3-Butanediol resulted in the oxidation of 2,3-Butanediol to small amounts of diacetyl and acetoin; 33% of the perfused 2,3-Butanediol was metabolized or conjugated in the liver.2,71
Human
In human subjects dermally exposed to 25% 1,5-Pentanediol (2 applications, 12 hours apart), increasing levels of glutaric acid were detected in urine and serum (no concentrations were provided). 64 The study authors reported that the risk of 1,5-Pentanediol accumulation at the concentration tested (therapeutic dose) was low.
Human subjects orally exposed to 1,4-Butanediol (single 25 mg/kg dosage) in fruit juice exhibited measurable plasma concentrations of GHB between 5 and 30 minutes post-dosing, indicating rapid conversion of 1,4-Butanediol to GHB; 4 hours post-dosing plasma levels were below the limit of quantitation (1 mg/L). 76 Clearance of 1,4-Butanediol was rapid in some subjects and relatively slow in subjects who were confirmed to have a genetic mutation of variant alleles (G143 A single nucleotide-polymorphism of ADH-1B). Lightheadedness, headaches, and increased blood pressure were observed 15 minutes post-dosing, and reports of subjects feeling dizzy or less alert were expressed for up to 4 hours post-dosing. A study in which human subjects were injected intravenously with 1,4-Butanediol (15 or 30 mg/kg) showed rapid and nearly 100% conversion of 1,4-Butanediol to GHB; 1,4-Butanediol and GHB had essentially the same decay curves when equal doses of each were administered. 74 In another study, human subjects were orally administered GHB (single 25 mg/kg dosage) in water; absorption and elimination (linear kinetics) of GHB were rapid. 77 Terminal plasma elimination half-life was 17.4 to 42.5 min. The majority of subjects showed the highest concentrations in urine 60 minutes post-dosing; by 24 hours post-dosing, up to 2% of the administered dose was recovered in the urine. Confusion, sleepiness, and dizziness were observed, with substantial variation among the subjects.
Toxicological Studies
Acute Toxicity
Acute Toxicity Studies.
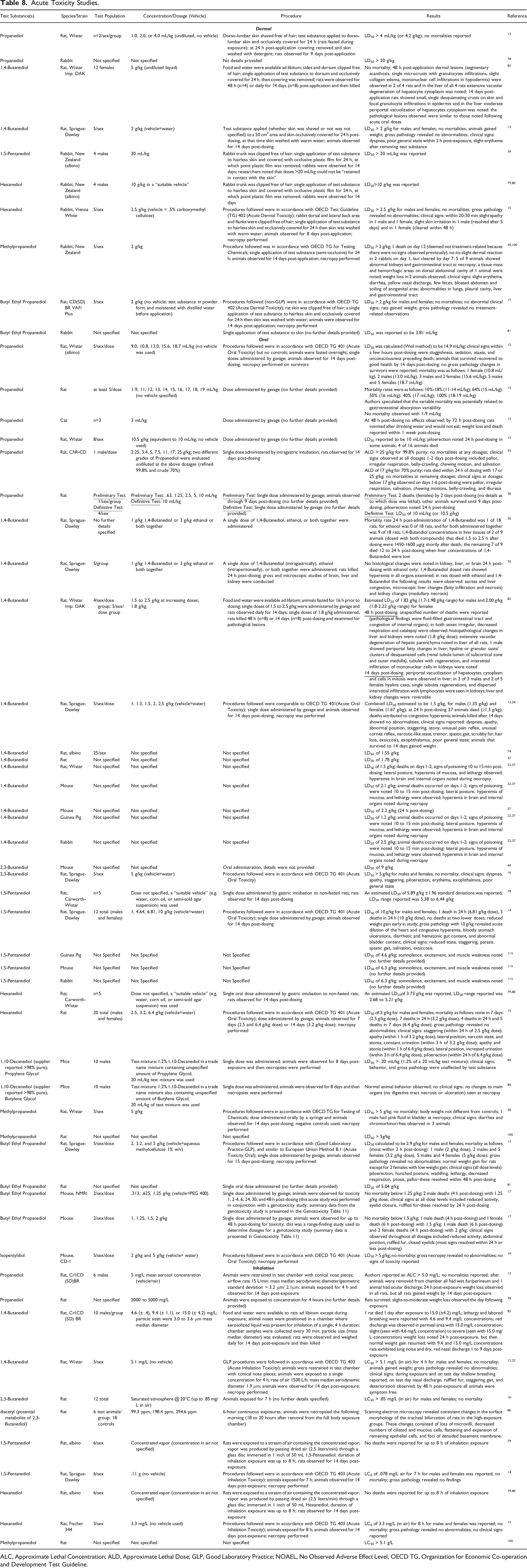
ALC, Approximate Lethal Concentration; ALD, Approximate Lethal Dose; GLP, Good Laboratory Practice; NOAEL, No Observed Adverse Effect Level; OECD TG, Organization for Economic Co-operation and Development Test Guideline.
Animal
Dermal
Dermal exposure animal studies evaluating the toxicity of the alkane diols indicated an LD50 > 20 g/kg in rats for Propanediol, 78 >20 mL/kg in rabbits for 1,5-Pentanediol, 79 >10 g/kg in rabbits for Hexanediol,79,80 and >2 g/kg in rabbits for Butyl Ethyl Propanediol. 81 The LD50s reported for 1,4-Butanediol and Methylpropanediol were >2 g/kg in dermally exposed rats 13 and rabbits. 20 After dermal exposure to 1,4-Butanediol (5 g/kg) in rats, findings included dermal lesions (48 h post-application) and abnormalities in the liver (14 days post-application), but no mortality. 82 Clinical signs observed in rats within 2 hours of exposure to 2 g/kg 1,4-Butanediol were dyspnea and poor general state; slight erythema was noted 24 hours post-exposure. 13 One source reported that 1,4-Butanediol was toxic on the skin, however the quality of the test material was questionable; the same source noted that there was no indication of absorption of acutely toxic quantities of 1,4-Butanediol in rabbit skin (no further details provided). 83 Clinical signs reported in rabbits following dermal exposure to 2 g/kg Methylpropanediol (time between exposure and appearance of signs not specified) were slight erythema, diarrhea, yellow nasal discharge, bloated abdomen, soiling of anogenital area, gastrointestinal tract abnormalities, and lung and liver abnormalities. 20 By 14 days post-application (2 g/kg Methylpropanediol), abnormalities in kidney and gastrointestinal tract of rabbits were reported at necropsy; there were no treatment-related mortalities.
Oral
Propanediol, 1,4-Butanediol, 2,3-Butanediol, 1,5-Pentanediol, Hexanediol, 1,10-Decanediol, Methylpropanediol, Butyl Ethyl Propanediol, and Isopentyldiol were evaluated for toxicity in acute oral exposure studies in animals. An approximate lethal dosage (ALD) of 17 g/kg (70% purity) and >25 g/kg (99.8% purity) and an LD50 of 14.9 mL/kg were reported in rats dosed with Propanediol; clinical effects noted were sluggishness, sedation, ataxia, irregular respiration, unconsciousness followed by the death of some of the animals.12,35 Various animal studies reported an LD50 between 1.2 and 2.5 g/kg for 1,4-Butanediol.13,22,34,37,74,82 Findings at necropsy in one rat study (animals killed 48 h post-dosing with 1.8 g/kg 1,4-Butanediol) were fluid-filled gastrointestinal tract and congestion of internal organs, histopathological changes in liver and kidneys, extensive vacuolar degeneration of hepatic parenchyma, granular clusters of desquamated cells, and interstitial infiltration of mononuclear kidney cells. 82 In another rat study, 14-days post dosing (1 to 2.5 g/kg 1,4-Butanediol), the animals that survived to necropsy showed no abnormal findings and an LD50 of 1.5 g/kg was reported. 13 Clinical signs observed after 1,4-Butanediol (1.35 to 2 g/kg dosage) administration in rats included irregular, decreased respiration and catalepsy, dyspnea, apathy, abnormal position, staggering, spastic gait, atony, and unusual pain reflex.13,82 For the following alkane diols, LD50s were reported as: >5 g/kg in rats16,34 and 9 g/kg49 in mice for 2,3-Butanediol; 10 g/kg 1,5-Pentanediol in rats; 14 3 g/kg Hexanediol in rats 15 ; >.20 mL/kg 1,10-Decanediol (1.2% in a 20 mL/kg trade name mixture also containing unspecified amounts of Propylene Glycol) in mice; 84 >5 g/kg Methylpropanediol in rats; 20 2.9 g/kg17 and 5 g/kg81 Butyl Ethyl Propanediol in rats; and >5 g/kg Isopentyldiol in mice. 19 Clinical signs reported in rats included staggering, dyspnea, piloerection, and erythema after dosing with 2,3-Butanediol, staggering, spastic gait, salivation, exsiccosis, paresis, and dyspnea after dosing with 1,5-Pentanediol, staggering, apathy, narcotic state, constant urination, and piloerection after dosing with Hexanediol, diarrhea, chromorhinorrhea, piloerection, and pallor after dosing with Methylpropanediol, and piloerection and pallor and dosing with Butyl Ethyl Propanediol.14-17,20 In rats dosed with 10 g/kg 1,5-Pentanediol, dilation of the heart and congestive hyperemia, bloody stomach ulcerations, and abnormal bladder content were observed at necropsy. 14 After dosing with Methylpropanediol (5 g/kg), 1 rat (n = 10) showed pink bladder fluid at necropsy. 20 There were no clinical signs reported in mice dosed with Isopentyldiol 19 ; at necropsy, rats dosed with Hexanediol 15 or Butyl Ethyl Propanediol 17 and mice dosed with 1,10-Decanediol 84 or Isopentyldiol 19 showed no abnormalities. In mice (n = 2/sex/dosage) dosed with Butyl Ethyl Propanediol, 2 deaths were reported at 1.25 g/kg; 2 deaths at 1.5 g/kg; 3 deaths at 2 g/kg. 17
Inhalation
Studies evaluating the toxicity of Propanediol, 1,4-Butanediol, 2,3-Butanediol, 1,5-Pentanediol, Hexanediol, and Methylpropanediol were conducted in rats exposed by inhalation. An approximate lethal concentration (ALC) was estimated by the authors to be >5 mg/L for Propanediol (4 h exposure time, 3.2 µm mass median aerodynamic diameter); clinical signs were wet fur/perineum and ocular discharge. 12 Rats survived a 4-hour exposure to 2000 to 5000 mg/L Propanediol. 78 Rats exposed to 1,4-Butanediol (4.6 to 15 mg/L) by inhalation showed lethargy, labored breathing, red discharge in perineal area, weight loss within 24 hours post-exposure, followed by resumption of normal weight gain, and lung noise/dry nasal discharge 1 to 9 days post-dosing; 1 death (15 mg/L) occurred 1 day post-dosing. 85 In a study in which groups of 6 rats were exposed for 6 hours to 99.3 ppm, 198.4 ppm, or 294.6 ppm diacetyl (potential metabolite of 2,3-Butanediol), and necropsied 18-20 hours after removal from the full body exposure chamber, consistent changes in the surface morphology of the tracheal bifurcation of rats in the high-exposure groups were observed. 86 In another rat study, an LC50 > 5.1 mg/L 1,4-Butanediol (4 hour exposure time) was reported; no mortality or abnormalities during gross pathology examination were reported and clinical signs, which resolved within 48 hours post-exposure, included shallow breathing, nasal discharge, ruffled fur, staggering gait, and deterioration.13,22 The results for other alkane diols evaluated were: no deaths after 7 to 8 hours of exposure to 2,3-Butanediol (up to .85 mg/L in air) 16 ; 1,5-Pentanediol (concentrated vapor), 79 Hexanediol (concentrated vapor),79,80 or an LC50 > 5.1 g/L was reported for inhalation of Methylpropanediol (duration of inhalation not specified). 2
Short-Term Toxicity
Short-Term and Subchronic Toxicity Studies.
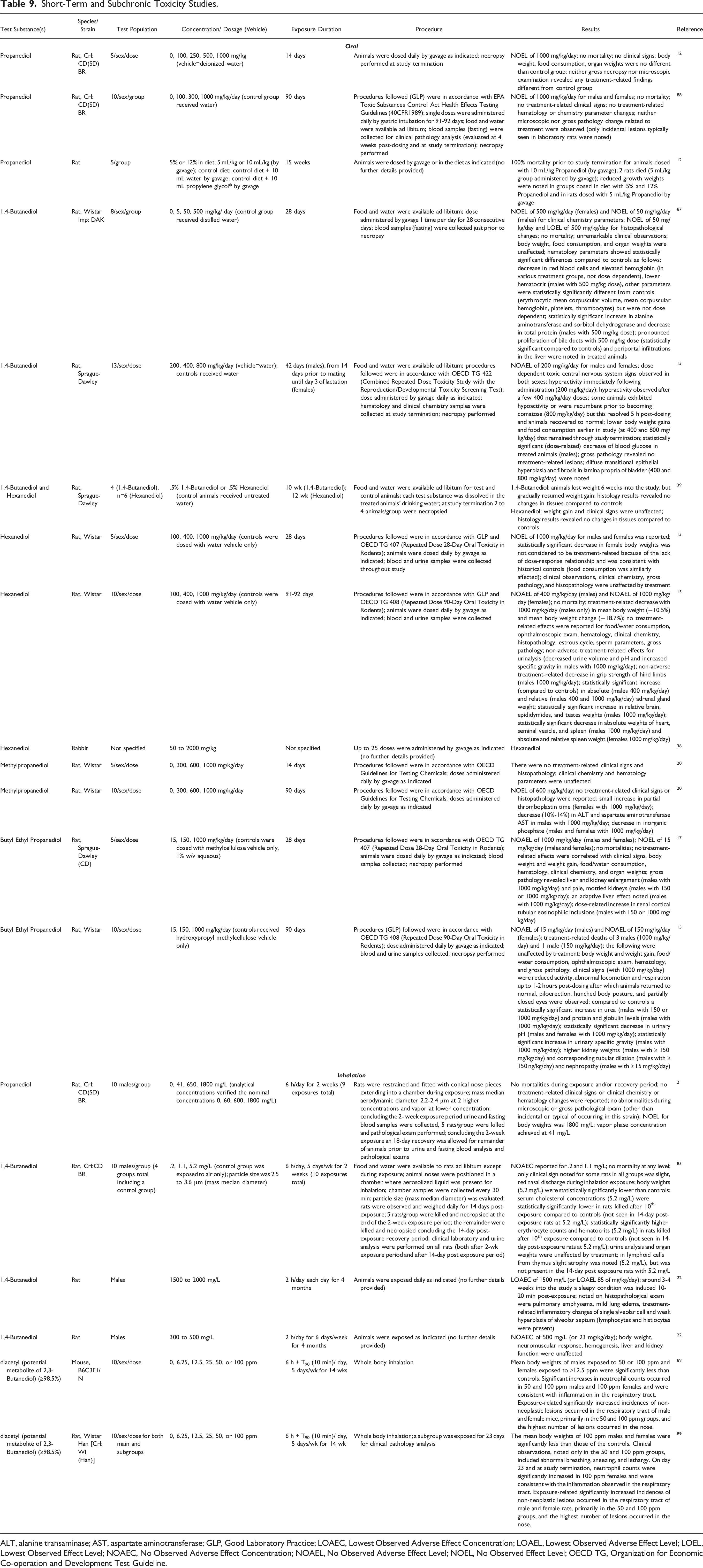
ALT, alanine transaminase; AST, aspartate aminotransferase; GLP, Good Laboratory Practice; LOAEC, Lowest Observed Adverse Effect Concentration; LOAEL, Lowest Observed Adverse Effect Level; LOEL, Lowest Observed Effect Level; NOAEC, No Observed Adverse Effect Concentration; NOAEL, No Observed Adverse Effect Level; NOEL, No Observed Effect Level; OECD TG, Organization for Economic Co-operation and Development Test Guideline.
Animal
Oral
Short-term oral exposure studies were conducted in animals to investigate the toxicity of Propanediol, 1,4-Butanediol, Hexanediol, Methylpropanediol, and Butyl Ethyl Propanediol. A no-observed-effect-level (NOEL) of 1000 mg/kg/day was reported for Propanediol in a 14-day rat study. 12 A 28-day experiment in rats evaluating the toxicity of 1,4-Butanediol revealed liver abnormalities; NOELs of 500 mg/kg/day (females) and 50 mg/kg/day (males) were reported. 87 Another rat study (approximately 42 days exposure duration) examining 1,4-Butanediol, showed lower body weight gains and food consumption (400 and 800 mg/kg/day), a statistically significant dose-related decrease of blood glucose (male treated animals), and bladder abnormalities (400 and 800 mg/kg/day); a no-observed-adverse-effect-level (NOAEL) of 200 mg/kg/day was reported. 13 The results of testing Hexanediol in rats (up to 1000 mg/kg/day for 28 days) 15 and rabbits (up to 2000 mg/kg for 25 doses, duration unknown) 36 yielded a reported NOEL of 1000 mg/kg/day for the rats 15 and observations of thrombosis and treatment-related effects (unspecified) on the liver and kidneys in the rabbits. 36 Results of testing Methylpropanediol in rats up to 1000 mg/kg/day for 14 days were reported to be unremarkable. 20 A NOAEL of 1000 mg/kg/day was reported for Butyl Ethyl Propanediol in a 28-day rat experiment; rats exhibited abnormalities of the liver (in males at 1000 mg/kg/day) and kidney (in males at 150 or 1000 mg/kg/day). 17
Inhalation
Short-term inhalation exposure studies were conducted in animals to evaluate the toxicity of Propanediol and 1,4-Butanediol. A rat study evaluating exposure to Propanediol, up to 1800 mg/L, 6 h/day for 2 weeks (9 exposures total), reported no remarkable results. 78 A study in which rats were exposed to 1,4-Butanediol (up to 5.2 mg/L), 6 h/day, 5 days/week for 2 weeks showed slight, red nasal discharge at all levels tested (.2, 1.1, 5.2 mg/L), lower body weights (at 5.2 mg/L only), and abnormal blood chemistry parameters (at 5.2 mg/L only); a no-observed-adverse-effect-concentration (NOAEC) of 1.1 mg/L was reported. 85
Subchronic Toxicity
Below is a synopsis of the subchronic toxicity studies that are presented in detail in Table 9.
Animal
Oral
Propanediol, Hexanediol, Methylpropanediol, and Butyl Ethyl Propanediol were evaluated for toxicity in subchronic (approximately 3-month) studies in rats with oral exposure. A NOEL of 1000 mg/kg/day was reported for Propanediol 88 ; another evaluation of 5 or 10 mL/kg of Propanediol resulted in 100% mortality (5 deaths) at 10 mL/kg and 2 deaths at 5 mL/kg. 12 NOAELs for Hexanediol were reported to be 400 mg/kg/day (males) and 1000 mg/kg/day (females); a treatment-related decrease (in males at 1000 mg/kg/day) in mean body weights and a statistically significant increase in relative adrenal gland weights (in males at 400 and 1000 mg/kg/day) and in relative weights of the brain, epididymides, and testes (in males at 1000 mg/kg/day) were observed. 15 A NOEL of 600 mg/kg/day was reported for Methylpropanediol; abnormalities seen were decreased liver enzymes and inorganic phosphate (at 1000 mg/kg/day). 20 NOAELs of 150 mg/kg/day (females) and 15 mg/kg/day (males) were reported for Butyl Ethyl Propanediol; there were 4 treatment-related deaths (males at 150 or 1000 mg/kg/day), abnormal locomotion and respiration 1 to 2 hours post-dosing (after which animals returned to normal), hunched body, and urinary (at 150 and 1000 mg/kg/day) and kidney abnormalities (at ≥15 mg/kg/day) reported. 17
Inhalation
In rat studies of 4-month durations (2 h/day exposure time) evaluating 1,4-Butanediol, a NOAEC of 500 mg/L (or NOAEL of 23 mg/kg/day) and a lowest-observed-adverse-effect-concentration (LOAEC) of 1500 mg/L (or lowest-observed-adverse-effect-level, LOAEL, of 85 mg/kg/day) were reported; observations in the study reporting the LOAEC of 1500 mg/L included a sleepy condition 20 minutes post-exposure, and histopathological exam revealed pulmonary emphysema, mild lung edema, treatment-related inflammatory changes of single alveolar cell and weak hyperplasia of alveolar septum. 22 In 14-wk studies of diacetyl (potential metabolite of 2,3-Butanediol) in mice and rats, significant increases in neutrophil counts consistent with inflammation in the respiratory tract were observed at 50 and 100 ppm (mice), and at 100 ppm (rats). 89 Significantly increased incidences of exposure-related, non-neoplastic lesions occurred in the respiratory tract of male and female rats and mice, primarily in the 50 and 100 ppm groups; the highest number of lesions occurred in the nose.
Chronic Toxicity
Oral
1,4-Butanediol
Experimental details for one chronic toxicity study found in the literature were limited.22,90 In this study male rats (n = 6/group) were orally exposed to .25, 3, or 30 mg/kg 1,4-Butanediol for 6 months. Controls were used (no further details). At the 30 mg/kg dosage, blood cholinesterase activity was reduced, the ratio of blood serum protein fractions changed, the thiol groups in whole blood and the brain decreased, liver glycogen and choline esterase activity decreased, vitamin C in organs decreased, and there was an increase in blood serum transaminases. A substantial increase in the auto-diffusion coefficient of tissue fluid (herein, owing to variation in the permeability of cell membranes) was found in the liver and brain with the 3 and 30 mg/kg dosages. Liminal morphological changes were noted with the 3 mg/kg dosage. At the 30 mg/kg dosage, the morphological changes observed were a reduction in Nissl bodies, glial element growth in cerebral tissue, fatty dystrophy, hyperemia in organs, and sclerotic growth in liver.
Developmental and Reproductive Toxicity (dart) Studies
Developmental and Reproductive Toxicity (DART) Studies.
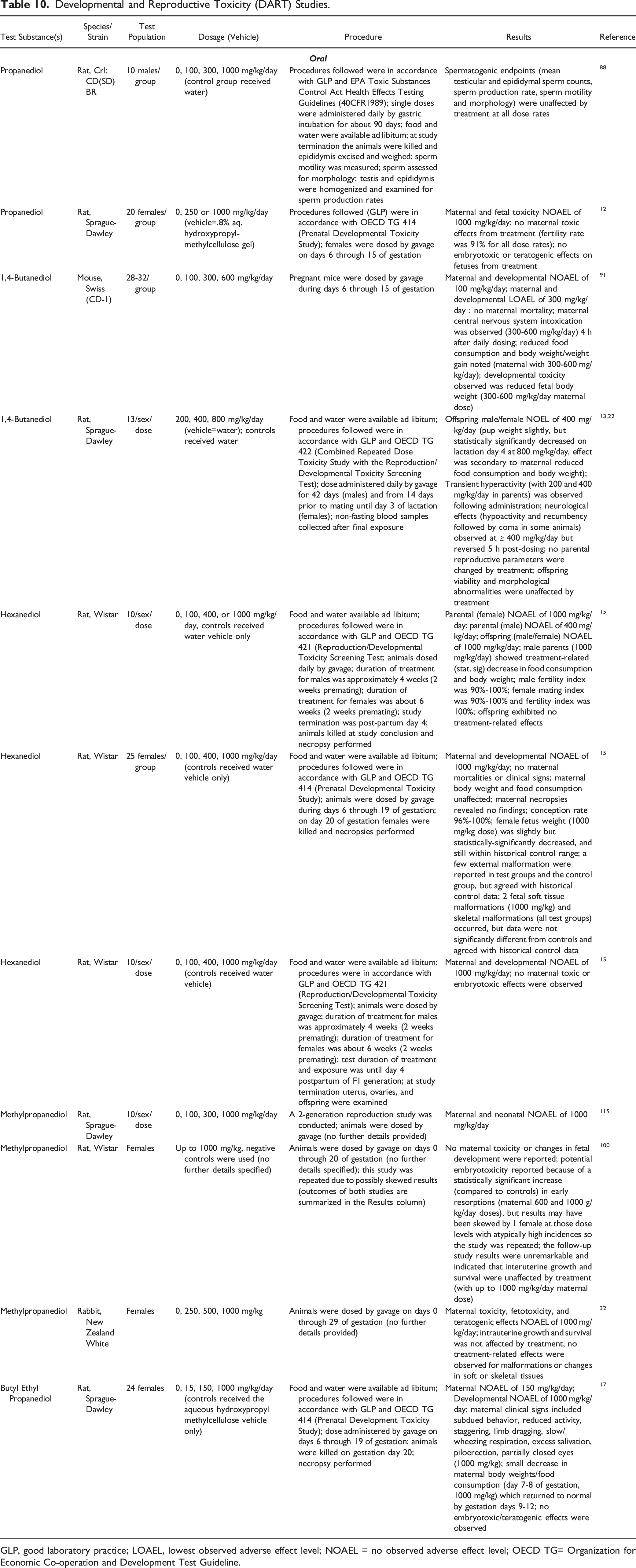
GLP, good laboratory practice; LOAEL, lowest observed adverse effect level; NOAEL = no observed adverse effect level; OECD TG= Organization for Economic Co-operation and Development Test Guideline.
Oral
Developmental and reproductive toxicity studies were conducted in animals that were orally exposed to Propanediol, 1,4-Butanediol, Hexanediol, Methylpropanediol, or Butyl Ethyl Propanediol. In rat studies evaluating Propanediol at dose rates up to 1000 mg/kg/day, spermatogenic endpoints were unaffected (90-day exposure duration) 88 and no maternal (dosing on days 6 - 15 of gestation) or fetal toxic effects were observed (maternal and fetal NOAEL 1000 mg/kg/day). 12 In a mouse study evaluating 1,4-Butanediol at up to 600 mg/kg/day (dosing on days 6 - 15 of gestation), a maternal and developmental NOAEL of 100 mg/kg/day and a LOAEL of 300 mg/kg/day were reported; maternal central nervous system intoxication (300-600 mg/kg/day) and maternal and fetal body weight reduction (maternal 300 - 600 mg/kg/day) were observed. 91 For male and female rats dosed with up to 800 mg/kg/day 1,4-Butanediol (14 days prior to mating and for females through day 3 of lactation), the following were reported: developmental NOEL of 400 mg/kg/day (pup weight slightly but statistically significantly decreased on lactation day 4 at 800 mg/kg/day, secondary to maternal reduction in body weight), parental transient hyperactivity (200 and 400 mg/kg/day) and reversible parental hypoactivity (≥400 mg/kg/day), but no parental reproductive parameters were changed by treatment.13,22 A maternal and developmental NOAEL of 1000 mg/kg/day was reported in animal studies on Hexanediol (rats dosed on days 6-19 of gestation) 15 and for Methylpropanediol (rats dosed on days 0 - 20 of gestation; rabbits on days 0 - 29).2,32 In a rat study evaluating Butyl Ethyl Propanediol (up to 1000 mg/kg/day on days 6 - 19 of gestation), a maternal NOAEL of 150 mg/kg/day (reduced activity, staggering, limb dragging, slow respiration, and reduced food consumption/body weight at 1000 mg/kg dose) and a developmental NOAEL of 1000 mg/kg/day were reported. 17
Genotoxicity
Genotoxicity Studies.
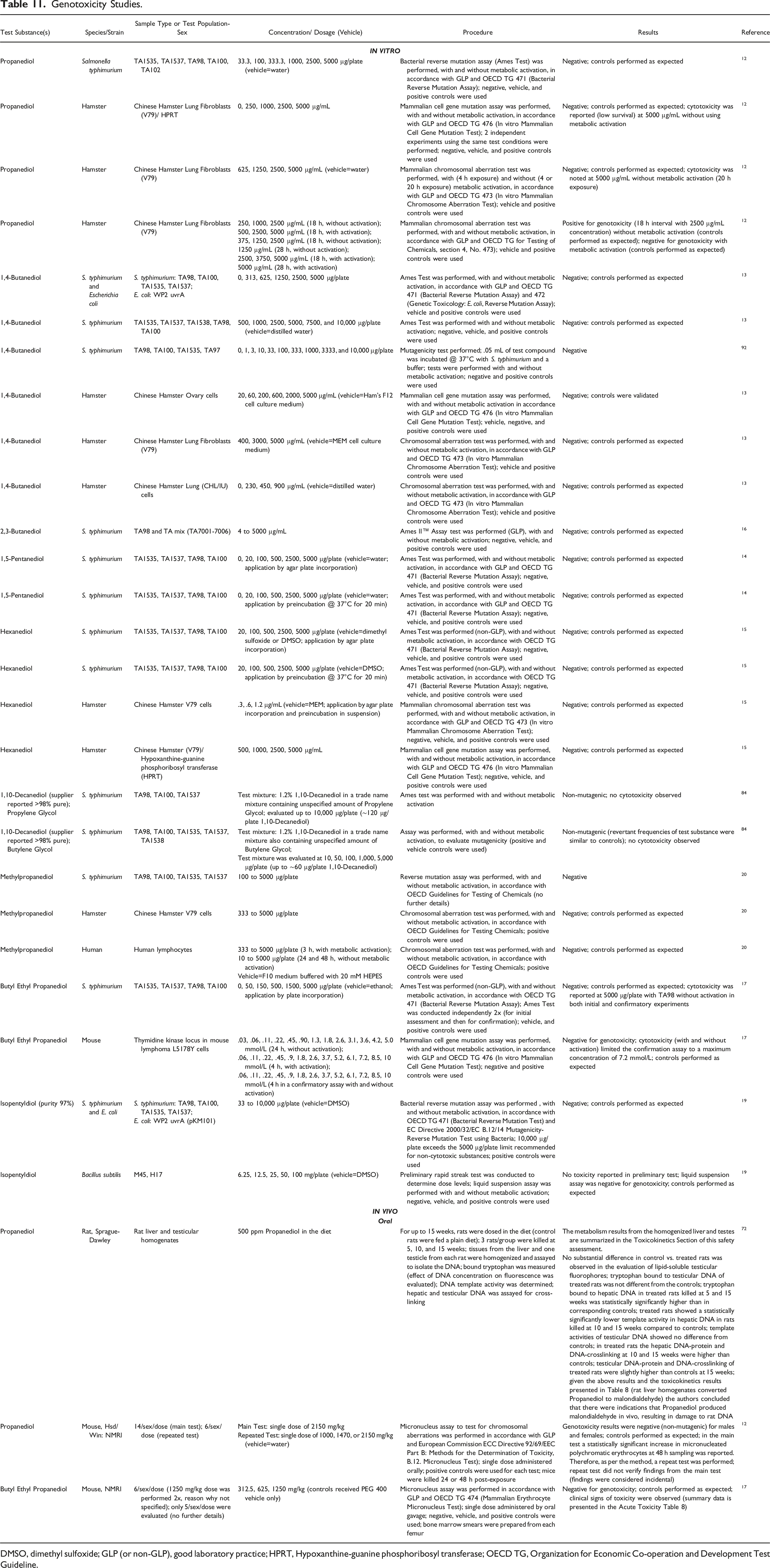
DMSO, dimethyl sulfoxide; GLP (or non-GLP), good laboratory practice; HPRT, Hypoxanthine-guanine phosphoribosyl transferase; OECD TG, Organization for Economic Co-operation and Development Test Guideline.
In Vitro
Genotoxicity data are available for Propanediol, 1,4-Butanediol, 2,3-Butanediol, 1,5-Pentanediol, Hexanediol, 1,10-Decanediol, Methylpropanediol, Butyl Ethyl Propanediol and Isopentyldiol. Experiments conducted in vitro evaluating Propanediol were negative for genotoxicity in a mammalian cell gene mutation assay (up to 5000 µg/mL), a chromosomal aberration test (up to 5000 µg/mL), and an Ames test (up to 5000 µg/plate). 12 A mammalian chromosomal aberration test (2500 µg/mL) evaluating Propanediol resulted in positive responses for genotoxicity without metabolic activation, but was negative with metabolic activation. 12 1,4-Butanediol was negative for genotoxicity in a Salmonella typhimurium mutagenicity test (up to 10,000 µg/plate), 92 in an Ames test (up to 10,000 µg/plate), 13 in a mammalian cell gene mutation assay (up to 5000 µg/mL), 13 and in a chromosomal aberration test (up to 5000 µg/mL). 13 2,3-Butanediol was negative in an Ames II™ test (up to 5000 µg/mL). 16 In an Ames test (up to 5000 µg/plate) 1,5-Pentanediol was negative for genotoxicity. 14 Hexanediol was negative for genotoxicity in an Ames test (up to 5000 µg/plate), in a mammalian chromosomal aberration test (up to 1.2 µg/mL), and in a mammalian cell gene mutation assay (up to 5000 µg/mL). 15 1,10-Decanediol (1.2% in a trade name mixture also containing unspecified amounts of Propylene Glycol or Butylene Glycol) was non-mutagenic in an Ames test (up to ∼ 120 µg/plate 1,10-Decanediol). 84 Methylpropanediol was negative in a reverse mutation assay (up to 5000 µg/plate) and in a chromosomal aberration test (up to 5000 µg/plate). 20 Butyl Ethyl Propanediol was negative for genotoxicity in an Ames test (up to 5000 µg/plate) and in a mammalian cell gene mutation assay (up to 7.2 mmol/L). 17 Isopentyldiol was negative for genotoxicity in an Ames test (up to 10,000 µg/plate) and in a liquid suspension assay (up to 100 mg/plate). 19
In Vivo
Oral
Tests performed in rat liver and testicular homogenates from rats that were fed 500 ppm Propanediol in the diet for 15 weeks (controls fed plain diet), showed that the DNA-protein and interstrand DNA-crosslinking in the hepatic DNA at 10 and 15 weeks were greater than in controls, and the DNA-protein and interstrand crosslinking in testicular DNA of treated rats were slightly greater than in controls at 15 weeks. 72 The study authors concluded that Propanediol was converted to malondialdehyde in vivo, causing damage to rat DNA. Mouse micronucleus tests conducted in vivo were negative for Propanediol (single oral dose of 2150 mg/kg) 12 and for Butyl Ethyl Propanediol (single oral dosage up to 1250 mg/kg). 17
Carcinogenicity Studies
Carcinogenicity studies data on alkane diol ingredients were not found in the published literature, and unpublished data were not submitted. A carcinogenicity study of diacetyl (potential metabolite of 2,3-Butanediol) in rats and mice is described. 89
Inhalation
Diacetyl (potential metabolite of 2,3-Butanediol)
The carcinogenic potential of diacetyl (≥98.5%) was evaluated by the National Toxicology Program (NTP) in inhalation studies in mice and rats. 89 Groups of 50 male and 50 female B6C3F1/N mice and Wistar Han [CRL:WI (Han)] rats were exposed to diacetyl vapor by whole body inhalation at concentrations of 0, 12.5, 25, or 50 ppm, for 6 h + T90 (12 min)/day, 5 days/wk, for 105 wk; T90 refers to the time to achieve 90% of the target concentration after the beginning of vapor generation.
In the mouse study, mean body weights of the 50 ppm groups were reduced to 65% (males) and 62% (females) of those of the respective chamber control groups. Clinical observations, which were most prominent in the 50 ppm groups, included abnormal breathing, thinness, sneezing, and eye abnormality in both males and females. Adenocarcinomas occurred in the nose of two female mice at the 50 ppm dose level; nasal adenocarcinomas have not been recorded historically in the NTP database. Statistically significant increases in several non-neoplastic lesions were observed in the nose and larynx (all test groups), the trachea (25 and 50 ppm groups), and the lungs (50 ppm group; most common bronchial lesion in both males and females was bronchus epithelium regeneration). Effects on the cornea of the eye were observed in the 25 and 50 ppm diacetyl groups. It was concluded that there was no evidence of carcinogenic activity of diacetyl in male B6C3F1/N mice exposed to 12.5, 25, or 50 ppm, but there was equivocal evidence of carcinogenic activity in female B6C3F1/N mice based on the occurrences of adenocarcinoma of the nose.
In the rat study, survival of males exposed to 50 ppm diacetyl was significantly less than that of the chamber control group, and survival was moderately reduced in females exposed to 25 ppm. At the end of the study, mean body weights of both sexes exposed to 50 ppm were decreased relative to the respective control groups, with more of an effect in males (81% of controls) than in females (91% of controls). Exposure-related clinical observations were reported and included thinness, abnormal breathing, eye abnormality, and nasal/eye discharge in males and eye abnormality and abnormal breathing in females. Three squamous cell carcinomas and one squamous cell papilloma of the nasal mucosa occurred in male rats exposed to 50 ppm, and three squamous cell carcinomas of the nasal mucosa occurred in females exposed to 50 ppm; no squamous cell carcinomas or papillomas of the nose occurred in the concurrent male or female controls, and none were recorded historically in the NTP database. Statistically significant increases in several non-neoplastic lesions were observed in the nose, larynx, the trachea, and the lungs of rats of the 25 and 50 ppm groups. Effects on the cornea of the eye were also observed in rats of the 25 and 50 ppm diacetyl groups. It was concluded that there was some evidence of carcinogenic activity of diacetyl in male and female Wistar Han rats based on the combined incidences of squamous cell papilloma and squamous cell carcinoma of the nose in males and incidences of squamous cell carcinoma of the nose of females.
Other Relevant Studies
Cytotoxicity
1,10-Decanediol
An agarose overlay test was performed by evaluating the diffusion in an agarose gel of a trade name mixture containing 1.2% of 1,10-Decanediol and an unspecified amount of Butylene Glycol. Average diameters (total score) were 1.075 cm; results indicated that cytotoxicity was low. No further details were provided. 84
Neurotoxicity
“Hexacarbon neurotoxicity” describes the ability of n-hexane and 2-hexanone (methyl n-butyl ketone; MnBK) to produce structural damage to the central and peripheral nervous system. 93 Both of these compounds are metabolized to similar metabolites, ie, 2-hexanol, 5-hydroxyl-2-hexanone, 2,5-hexanediol, and 2,5-hexanedione. 94 2,5-Hexanediol and 2,5-hexanedione are neurotoxic agents, and 2-hexanol and 5-hydroxyl-2-hexanone may be neurotoxic, because they are metabolized to 2,5-hexanedione.
Peripheral neuropathy develops in several species with exposure to these 6 metabolically-interrelated hexacarbons. 93 The relative neurotoxicity of these compounds in Charles River CD COBS rats (2,5- hexanedione > 5-hydroxy-2-hexanone > 2,5-hexanediol > MnBK > 2-hexanol > n-hexane) has been attributed to differential serum levels of the common metabolite, 2,5- hexanedione, and is in proportion to the peak serum value that each compound generates.
The specific molecular configuration of the hexacarbon compounds plays a role in neuropathy. 93 Neuropathy did not develop in Sprague-Dawley rats exposed to the diketones 2,4-hexanedione, 2,3-hexanedione, 3,5-heptanedione, or 2,6-heptanendione. In addition, the mono-ketone, 2-heptanone, did not produce neuropathy in rats, but 2,5-heptanedione and 3,6-octanedione produced neuropathological alterations. 95 These findings support the requirement for a diketone with γ-spacing to produce neuropathy. Additional studies suggested that although pyrrole formation is necessary for the pathogenesis of γ-diketone neuropathy, autoxidation to reactive intermediates, resulting in inter- or intraneurofilament crosslinking, is required for the neurotoxic effects of γ-diketones to be manifested. 96
1,4-Butanediol
Central nervous system effects have been reported for exposures to 1,4-Butanediol. 74 Central nervous system depression, anesthetic effect, loss of righting reflex, struggle response, and voluntary motor activity were documented in rats administered 496 mg/kg 1,4-Butanediol (no further details were provided). During oral, intraperitoneal, or intravenous exposure, neuropharmacologic responses have been reported. These effects were also observed after administration of GHB. Endogenous levels of GHB in the brain of mammals are in micromolar concentrations, while in the liver, heart, and kidneys concentrations are 5 to 10 times higher. Although 1,4-Butanediol can be converted to GHB in the brain, liver, kidney, and heart, the liver has the greatest capacity (per gram of tissue) to metabolize GHB. When GHB was administered at dosages exceeding 150 mg/kg in rats, a state of behavioral arrest was observed, with bilaterally synchronous electroencephalogram readings resembling those of humans undergoing seizures (non-epileptic).
Dermal Irritation and Sensitization Studies
Dermal Irritation, Sensitization, and Photoirritation/Photosensitization Studies.
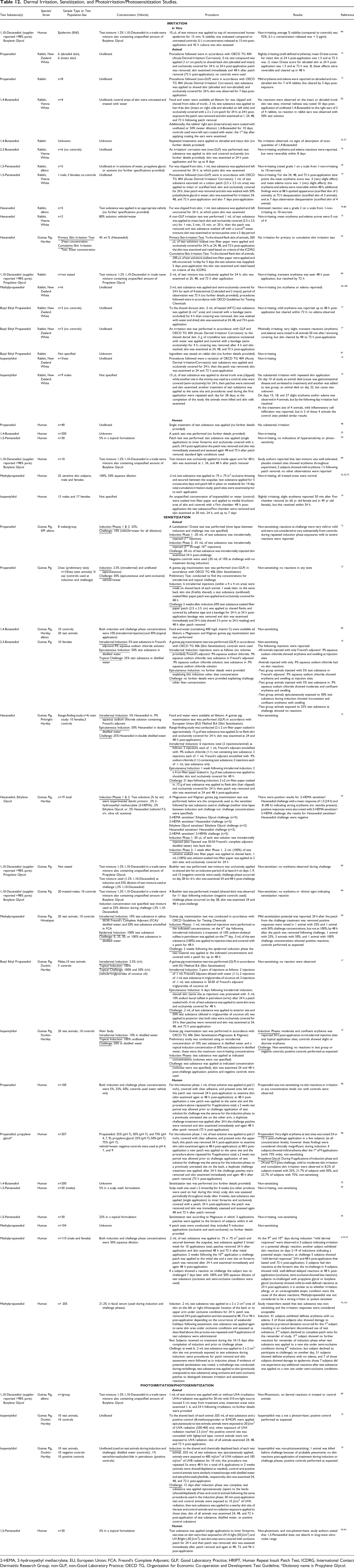
2-HEMA, 2-hydroxyethyl methacrylate; EU, European Union; FCA, Freund’s Complete Adjuvant; GLP, Good Laboratory Practice; HRIPT, Human Repeat Insult Patch Test; ICDRG, International Contact Dermatitis Research Group; non-GLP, non-Good Laboratory Practice; OECD TG, Organization for Economic Co-operation and Development Test Guideline; *Dictionary name is Propylene Glycol.
Irritation
In vitro
1,10-Decanediol (1.2% in a trade name mixture also containing an unspecified amount of Butylene Glycol) was non-irritating in an in vitro test evaluating the test substance on reconstructed human epidermis. 84
Animal
Skin irritation testing of Propanediol, 1,4-Butanediol, 2,3-Butanediol, 1,5-Pentanediol, Hexanediol, 1,10-Decanediol, Methylpropanediol, Butyl Ethyl Propanediol, and Isopentyldiol was conducted. Results indicated the following observations: Propanediol (undiluted) was mildly irritating to rabbit skin in 24-hour occlusive patch tests 12 ; 1,4-Butanediol (undiluted) caused only minimal redness after application to rabbit ears and no irritation was observed in a 24-hour occlusive patch test on intact and abraded rabbit skin; 82 2,3-Butanediol (undiluted) was non-irritating to rabbit skin in a 24-hour occlusive patch test; 16 1,5-Pentanediol (undiluted) was non-irritating to rabbit skin in both a 24-hour non-occlusive skin test 79 and a 20-hour occlusive patch test on intact and scarified skin; 14 Hexanediol (45 to 80%) was non-irritating to animal skin in both non-occlusive and occlusive tests performed with approximately 24-hour dermal exposure;15,79,80,97 1,10-Decanediol (1.2% in a trade name mixture also containing an unspecified amount of Propylene Glycol) was non-irritating to rabbit skin in a 24 h occlusive patch test; 84 Methylpropanediol (undiluted) was non-irritating to animal skin; 20 Butyl Ethyl Propanediol (undiluted) was non-to-minimally irritating to rabbit skin in 4-hour semi-occlusive patch tests;2,17 Isopentyldiol (undiluted) was non-to-slightly irritating to rabbit skin in 24-hour occlusive and semi-occlusive patch tests.
Human
Skin irritation testing of Propanediol, 1,4-Butanediol, 1,5-Pentanediol, 1,10-Decanediol, Methylpropanediol, and Isopentyldiol in human subjects showed the following: Propanediol (undiluted) was non-irritating after a single application of test substance (no further details provided);19,98 1,4-Butanediol (concentration not specified) was non-irritating in a patch test (no additional details provided); 22 1,5-Pentanediol (5%) was non-irritating in an occlusive patch test; 45 1,10-Decanediol (1.2% in a trade name mixture also containing an unspecified amount of Butylene Glycol) was well-tolerated, according to study authors (2 subjects showed mild erythema 1 h following patch removal), in a 48 h occlusive patch test; 84 Methylpropanediol (100%, 50% aqueous dilution) was non-irritating to subjects with sensitive skin in a 14-day cumulative irritation study; 99 Isopentyldiol (concentration not specified) was slightly irritating in a 48-hour Finn chamber skin test.2,19,100
Sensitization
Animal
Skin sensitization testing of Propanediol, 1,4-Butanediol, 2,3-Butanediol, Hexanediol, 1,10-Decanediol, Methylpropanediol, Butyl Ethyl Propanediol, and Isopentyldiol was performed in guinea pigs. Propanediol (2.5% intradermal and 100% epicutaneous concentrations applied at induction, 50% epicutaneous and semi-occlusive at challenge) was non-sensitizing; 12 1,4-Butanediol (10% intradermal and 30% topical concentrations applied at induction and challenge) was non-sensitizing. 82 2,3-Butanediol (5% intradermal and 50% epicutaneous concentrations applied at induction, 25% at challenge) was non-sensitizing, although during epicutaneous induction animals showed incrustation and confluent erythema with swelling. 16 Hexanediol (5% intradermal and 50% epicutaneous concentrations applied at induction, 25% at challenge) was non-sensitizing in one test. 15 In another test, strong erythema was reported with Hexanediol challenge (no concentration specified) following induction (sensitization) with another compound (.2% hydroxyethyl methacrylate). However no Hexanediol induction (.2%)/Hexanediol challenge (no concentration specified) tests showed a positive sensitization reaction. 97 1,10-Decanediol (1.2% in a trade name mixture containing an unspecified amount of Propylene Glycol or Butylene Glycol) was non-sensitizing in a Buehler test (1.2% 1,10-Decanediol in trade name mixture used at induction and .3% 1,10-Decanediol in trade name mixture used at challenge). 84 Methylpropanediol showed mild sensitization potential (10% intradermal to 100% epidermal concentrations applied at induction, up to 100% at challenge). 20 Butyl Ethyl Propanediol (2.5% intradermal and 100% topical concentrations applied at induction, 50% and 100% at challenge) was non-sensitizing. 17 Isopentyldiol (10% intradermal and 100% topical concentrations applied at induction, 50% at challenge) was non-sensitizing. However, during intradermal injection at induction and topical induction, moderate and confluent erythema were observed. 19 The alkane diols showed mild or no sensitization potential and some positive skin irritation reactions were observed during induction.
Human
Clinical skin sensitization studies of Propanediol, 1,4-Butanediol, 1,5-Pentanediol, and Methylpropanediol showed the following results: Propanediol was non-sensitizing (5% to 75% concentrations applied at induction and at challenge) with mild erythema reported in 4 subjects of 207 during induction (75% only) after the 1st of 9 applications; 98 1,4-Butanediol (concentration not specified) was non-sensitizing; 22 1,5-Pentanediol (5% and 25% in different tests) was non-sensitizing; 45 Methylpropanediol (concentration not specified) was non-sensitizing in one test; in another test Methylpropanediol (50% aqueous dilution applied at induction and challenge) showed mild skin sensitization potential, however the study authors concluded that it was unclear as to whether or not the skin reactions were caused by irritation, allergic response, or an atopic condition.2,99 An additional test showed that Methylpropanediol (21.2% applied at induction and challenge) caused erythema and damage to epidermis in some subjects during the induction phase. However, the reactions were not reproducible after a new skin site was tested on those subjects under semi-occlusive conditions; Methylpropanediol was non-sensitizing in this study. 101 The alkane diols evaluated were non-sensitizing in human skin.
Photoirritation/Photosensitization
Animal
1,10-Decanediol (1.2% in a trade name mixture also containing an unspecified amount of Butylene Glycol) was non-phototoxic in guinea pig skin. 84 Isopentyldiol (undiluted) was neither a photo-irritant nor a photo-sensitizer when tested in guinea pig skin; positive controls were used in both experiments and yielded expected results. 19
Human
1,5-Pentanediol (5%) was not phototoxic and not photosensitizing in a 24-hour occlusive patch test performed following long-wave ultraviolet light (UVA)/short-wave ultraviolet light (UVB) exposure to the treated skin. The study authors stated that it does not absorb in UVA range.45,64
Ocular Irritation
Ocular Irritation Studies.
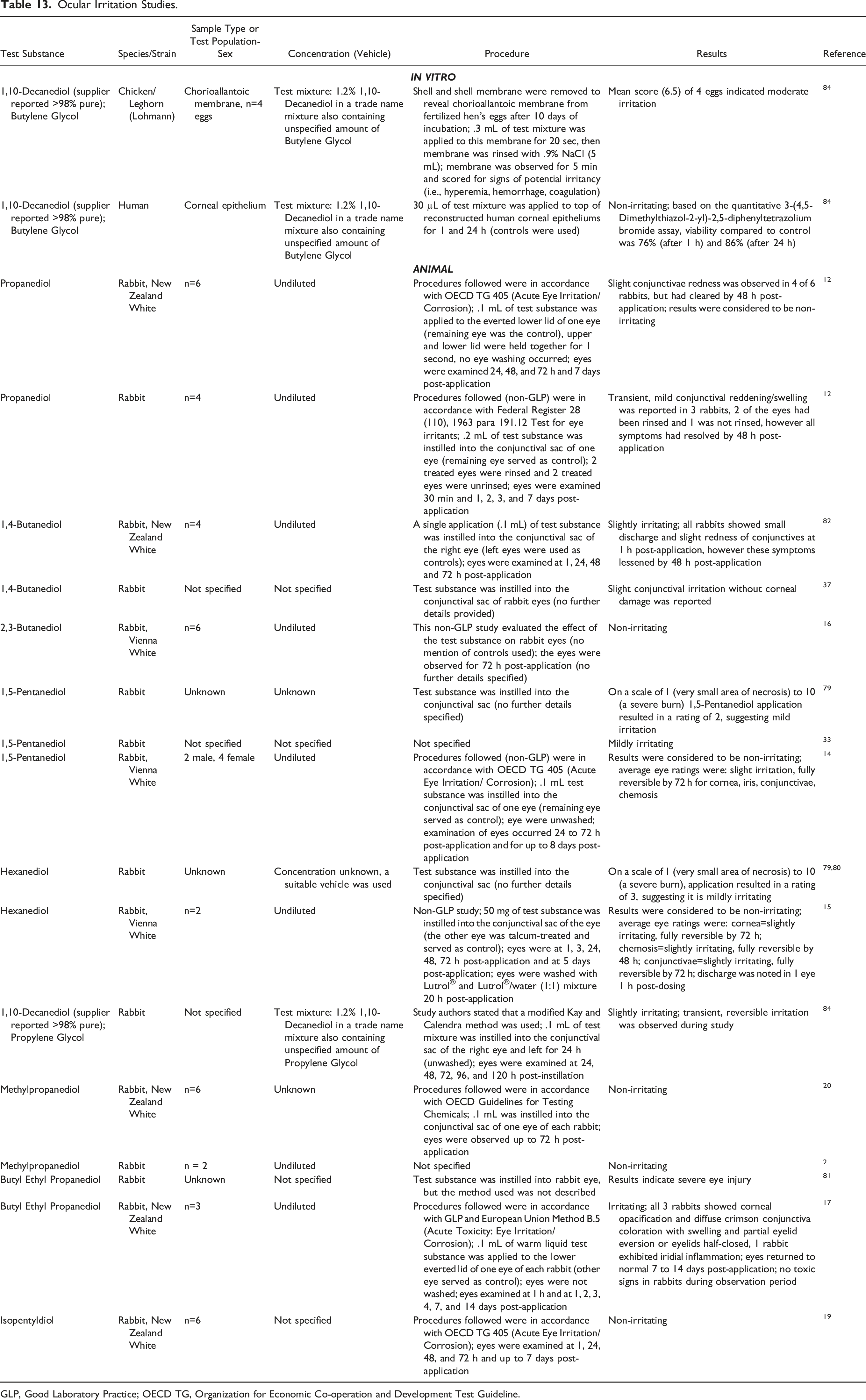
GLP, Good Laboratory Practice; OECD TG, Organization for Economic Co-operation and Development Test Guideline.
In Vitro
1,10-Decanediol (1.2% in a trade name mixture also containing an unspecified amount of Butylene Glycol) was evaluated in a hen’s egg experiment and found to have moderate irritation potential when tested on the chorioallantoic membrane. 84 The same 1,10-Decanediol test substance was also evaluated on reconstructed human corneal epithelium in vitro and found to be non-irritating.
Animal
Ocular irritation was evaluated in rabbit eyes for Propanediol, 1,4-Butanediol, 2,3-Butanediol, 1,5-Pentanediol, Hexanediol, 1,10-Decanediol, Methylpropanediol, Butyl Ethyl Propanediol, and Isopentyldiol. No-to-slight irritation (resolved within 48 hours post-application) was reported for undiluted Propanediol. 12 Undiluted 1,4-Butanediol was slightly irritating.37,82 Undiluted 2,3-Butanediol was non-irritating to rabbit eyes. 16 No-to-mild irritation was observed for undiluted 1,5-Pentanediol14,33,79 and undiluted Hexanediol.15,79,80 1,10-Decanediol (1.2% in a trade name mixture also containing an unspecified amount of Propylene Glycol) was slightly irritating. 84 Methylpropanediol (undiluted, n = 2) was non-irritating to rabbit eyes.20,100 Butyl Ethyl Propanediol (concentration not specified) resulted in severe eye injury in one test. 81 In another experiment, undiluted Butyl Ethyl Propanediol was considered to be irritating, with corneal opacification and diffuse crimson conjunctiva coloration, swelling, and partial eyelid eversion; the rabbit eyes returned to normal by 14 days post-application. 17 Isopentyldiol (concentration not specified) was non-irritating. 19 Generally, the alkane diols were non-to-mildly irritating, with the exception that Butyl Ethyl Propanediol was irritating.
Clinical Studies
1,5-Pentanediol
A controlled, double-blind comparative study was conducted to evaluate the treatment of atopic dermatitis with hydrocortisone and 1,5-Pentanediol. 102 Patients with atopic dermatitis were treated 2x/day with either 1% hydrocortisone (n = 31) or 1% hydrocortisone with 25% 1,5-Pentanediol (n = 32) in a cream formulation for 6 weeks. Quantitative bacteria cultures were taken for Staphylococcus aureus (commonly seen in the skin of atopic dermatitis patients) from the lesional skin prior to treatment and at weeks 2, 4, and 6 of treatment. The results indicated that the hydrocortisone-only formulation was effective for 68% of the patients in that test group; the hydrocortisone plus 1,5-Pentanediol formulation was effective for 69% in that group. There was a statistically significant reduction in S. aureus (baseline to week 2 and baseline to week 6) in the hydrocortisone plus 1,5-Pentanediol group, which was not observed in the hydrocortisone-only group. There were 2 instances in each treatment group of “slight burning sensation” following cream application. The study authors noted that bacteria are not likely to develop resistance to 1,5-Pentanediol because of the interaction of diols on membranes.
The therapeutic effect of 1,5-Pentanediol was investigated for the treatment of herpes simplex labialis (cold sore virus) in a placebo-controlled, randomized, double-blind clinical trial. 103 Patients included in the trial were those with known, frequent recurrences of herpes labialis. The treatment group (n = 53) received 25% 1,5-Pentanediol in a gel formulation, which was applied to both lips (.04 g total/day) during the 26-week prophylactic evaluation. The placebo group (n = 52) received the same gel formulation without 1,5-Pentanediol for 26 weeks. During the occurrence of herpes labialis episodes the treatment gel or placebo was applied to both lips (.16 g total/day) for 5 days and then the prophylactic treatment resumed until the next herpes episode. The herpes episodes reported during the trial were 109 for the treatment group and 120 for the placebo group. 1,5-Pentanediol did not demonstrate a prophylactic effect, compared to the placebo, in preventing the recurrence of herpes labialis. However, there was a statistically significant improvement in blistering, swelling, and pain for the therapeutic use of 1,5-Pentanediol as compared to the placebo. There were no treatment-related adverse events attributable to 1,5-Pentanediol or the placebo reported. In the treatment and placebo groups, body weight and temperature, heart rate, and clinical parameters were nearly unchanged.
Case Reports
Case Reports.
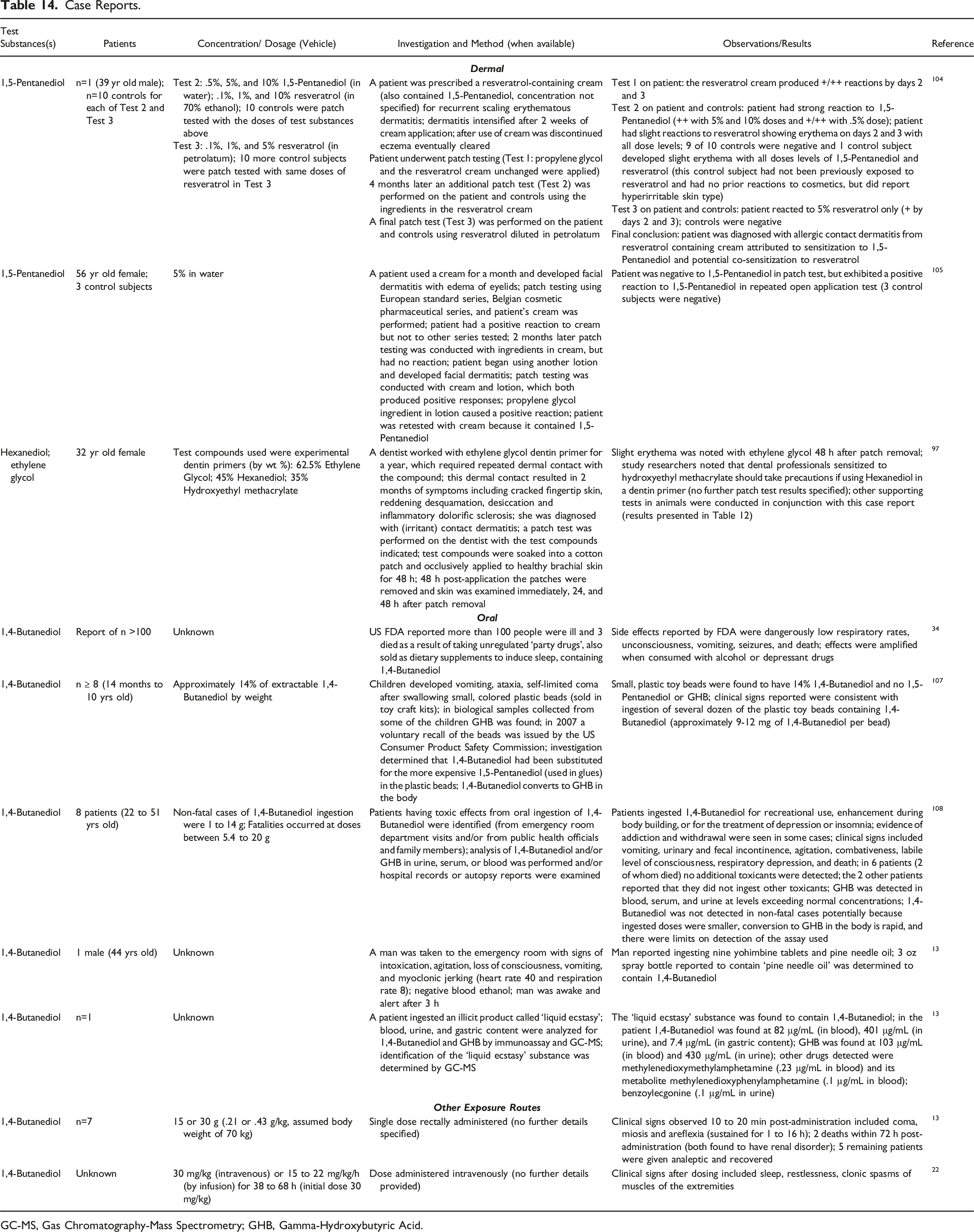
GC-MS, Gas Chromatography-Mass Spectrometry; GHB, Gamma-Hydroxybutyric Acid.
Information from case reports for the alkane diols included allergic contact dermatitis as a result of dermal exposure to 1,5-Pentanediol (.5 to 10%) in various creams,104,105 a recommendation by study researchers for dental professionals exposed to Hexanediol in dentin primers to take precautions because of the potential to cause contact dermatitis following repeated occupational exposure, 97 and adverse effects reported in adults (including death) and poisoning in children from oral exposure to 1,4-Butanediol (varying doses).13,22,106-108
Risk Assessment
Occupational Standards
1,4-Butanediol
In Germany, the occupational limit value for 1,4-Butanediol is 50 mL/m3 (ppm) or 200 mg/m3. 109
Diacetyl (potential metabolite of 2,3-Butanediol)
As of 2012, the American Conference of Governmental Industrial Hygienists (ACGIH) threshold limit value (TLV) for diacetyl is .01 ppm (.04 mg/m3) time-weighted average (TWA) and .02 ppm (.07 mg/m3) short-term exposure limit (STEL); diacetyl has an A4 classification (not classifiable as a human carcinogen). 110 A permissible exposure limit (PEL) has not been established by the US Occupational Safety and Health Administration (OSHA).
In 2016, the US National Institute for Occupational Safety and Health (NIOSH) established a recommended exposure limit (REL) of 5 ppb for diacetyl as a TWA for up to 8 h/day during a 40-h work week. 111 NIOSH recommends a STEL for diacetyl of 25 ppb for a 15-minute time period.
Summary
The 10 alkane diols included in this safety assessment reportedly function in cosmetics as solvents, humectants, and skin conditioning agents.
VCRP data received from the FDA in 2018 indicated that the highest reported uses are for Propanediol (1528 uses), and Methylpropanediol (570 uses). The Council industry survey data from 2015 indicated that the highest maximum use concentration in leave-on products was 39.9% Propanediol in non-spray deodorants.
1,4-Butanediol and Hexanediol are permitted as indirect food additives. The FDA has issued warnings about dietary supplements containing 1,4-Butanediol because of associated adverse health effects, including death. 1,4-Butanediol is considered to be a Class I Health Hazard by the FDA, as well as a Schedule I Controlled Substance Analog by the DEA, if illicit human consumption is intended.
A permeability coefficient of 1.50 × 10−5 cm/h was calculated for Propanediol after abdominal skin from human cadavers was exposed for 48 hours in a static diffusion cell to a 1.059 g/mL Propanediol solution (infinite dose, 99.953% purity).
The ability of Propanediol, 1,4-Butanediol, or 1,5-Pentanediol to enhance the penetration of the drug estradiol (.12% [3H]-estradiol in 1:10 alkane diol/ethanol solution) in human skin was evaluated in an in vitro experiment using a Franz diffusion cell. After ∼ 85 - 90 minutes the permeability of [3H]-estradiol in human skin was determined to be ∼ 5 - 6 µg/cm2 with Propanediol and <1 µg/cm2 with 1,4-Butanediol or 1,5-Pentanediol. In vitro tests of pharmaceutical formulations containing .1% mometasone furoate and 25% 1,5-Pentanediol or 1% hydrocortisone and 25% 1,5-Pentanediol or 1% terbinafine and either 5% or 20% 1,5-Pentanediol, showed that 1,5-Pentanediol was a penetration enhancer in human breast skin samples exposed to the formulations for 60 hours.
1,4-Butanediol was a competitive inhibitor of ethanol metabolism by alcohol dehydrogenase. Diacetyl, acetoin, and 2,3-Butanediol were interconvertible with a molar equilibrium ratio of 0:3:7, respectively, in rat liver homogenates. Methylpropanediol was demonstrated to be a substrate for alcohol dehydrogenase in vitro.
Rat liver homogenates metabolized Propanediol to yield malondialdehyde in treated rats (500 ppm in the diet for 15 weeks) and in control rats (plain diet). A single dose of Propanediol, 1,4-Butanediol, 2,3-Butanediol, or Hexanediol administered orally to rabbits yielded the corresponding glucuronic acid conjugates in the urine representing 2 to 26% of the administered dose. Orally administered 1,4-Butanediol and 1,5-Pentanediol produced succinic acid and phenacyl glutarate, respectively, in the urine.
Endogenous concentrations of 1,4-Butanediol in rats were 30 to 165 ng/g in aqueous phase tissues (aqueous portion of supernatant generated from homogenized tissues) and 150 to 180 ng/g in lipid phase tissues (lipid portion of supernatant generated from homogenized tissues). 1,4-Butanediol concentrations were 96 µg/g, 52 µg/g, and 58 µg/g in the brain, liver, and kidney, respectively, of rats 75 minutes after oral exposure to 1 g/kg 1,4-Butanediol. In rats orally exposed to up to 400 mg/kg 1,4-Butanediol (radiolabels on C1 and C4), >75% of the radioactivity was excreted as [14C]-CO2 by 24 hours post-dosing; up to 6% was eliminated in feces 72 hours post-dosing. Experiments in rats orally administered 1M diacetyl, acetoin, or 2,3-Butanediol showed interconversion among these compounds in vivo. Methylpropanediol (100 or 1000 mg/kg, [14C]-labeled) orally administered to rats was reported to be rapidly metabolized and eliminated as 3-hydroxybutyric acid in the urine (31% - 45% dosed radioactivity), as CO2 in exhaled breath (42% - 57%), and in the feces (<1% dosed radioactivity).
In human subjects dermally exposed to 25% 1,5-Pentanediol (2 applications, 12 hours apart), increasing levels of glutaric acid were detected in urine and serum (no concentrations were provided). Oral exposure to 25 mg/kg 1,4-Butanediol resulted in measurable plasma concentrations of GHB in human subjects within 5 to 30 minutes after exposure, indicating rapid conversion of 1,4-Butanediol to GHB; GHB concentrations were below the limit of quantitation within 4 hours. Clearance of 1,4-Butanediol was rapid in some subjects and relatively slow in others; the latter were confirmed to have a genetic mutation of variant alleles of ADH-1B. Nearly 100% of 1,4-Butanediol was rapidly converted to GHB in a study in which 15 or 30 mg/kg 1,4-Butanediol was intravenously injected into human subjects.
Dermal exposure animal studies evaluating the toxicity of the alkane diols indicated an LD50 >20 g/kg in rats for Propanediol, >20 mL/kg in rabbits for 1,5-Pentanediol, >10 g/kg in rabbits for Hexanediol, and >2 g/kg in rabbits for Butyl Ethyl Propanediol. A single dermal exposure to 5 g/kg 1,4-Butanediol caused dermal lesions within 48 hours and liver abnormalities within 14 days, but no mortalities in rats. In rabbits, a single 2 g/kg dermal application of Methylpropanediol caused kidney, lung, liver, and gastrointestinal tract abnormalities, among other effects, but no mortalities.
Acute oral LD50s reported in multiple studies of mammalian test species included 14.9 mL/kg Propanediol, 1.2 to 2.5 g/kg 1,4-Butanediol, 10 g/kg 1,5-Pentanediol, 3 g/kg Hexanediol, 3 to 5 g/kg Butyl Ethyl Propanediol, >.20 mL/kg 1,10-Decanediol (1.2% in a 20 mL/kg trade name mixture also containing unspecified amounts of Propylene Glycol), and ≥5 g/kg for 2,3-Butanediol, Methylpropanediol and Isopentyldiol.
A single, 4-hour inhalation exposure of 2000 to 5000 mg/L Propanediol caused moderate weight loss but no deaths in rats. A single 4.6 to 15 mg/L exposure to 1,4-Butanediol resulted in lethargy, labored breathing, and lung noise/dry nasal discharge in rats 1 to 9 days post-dosing, and 1 death at 15 mg/L 1 day post-dosing. Rats exposed for 4 hours to 5.1 mg/L 1,4-Butanediol exhibited shallow respiration that resolved within 48 hours post-exposure; gross pathology examination revealed no abnormalities. No deaths were reported after a single 7- to 8-hour inhalation exposure to 2,3-Butanediol (up to .85 mg/L in air), 1,5-Pentanediol (concentrated vapor), or Hexanediol (concentrated vapor). An LC50 > 5.1 g/L for inhalation (duration of inhalation not specified) was reported for Methylpropanediol.
Reported NOELs and NOAELs for short-term oral exposures in rats included 200 mg/kg/day 1,4-Butanediol (∼42 days), 500 mg/kg/day 1,4-Butanediol in females and 50 mg/kg/day in males (28 days), and 1000 mg/kg/day Propanediol and Methylpropanediol (14 days) or Hexanediol and Butyl Ethyl Propanediol (28 days). The 28-day experiment in rats evaluating the toxicity of 1,4-Butanediol revealed liver abnormalities in treated animals. The rat study (approximately 42 days exposure duration) examining 1,4-Butanediol, showed lower body weight gains and food consumption (400 and 800 mg/kg/day), a statistically significant dose-related decrease of blood glucose (male treated animals), and bladder abnormalities (400 and 800 mg/kg/day). The 28-day experiment evaluating oral exposure to Butyl Ethyl Propanediol in rats resulted in abnormalities in the liver (in males at 1000 mg/kg/day) and kidney (in males at 150 or 1000 mg/kg/day). Rabbits orally exposed to Hexanediol (up to 2000 mg/kg for 25 doses, duration unknown) exhibited thrombosis and treatment-related effects (unspecified) on the liver and kidneys.
Results were unremarkable in a study in which rats inhaled up to 1800 mg/L Propanediol, 6 h/day, for 2 weeks (9 total exposures). Rats exposed to up to 5.2 mg/L 1,4-Butanediol, 6 h/day, 5 days/week, for 2 weeks, showed slight red nasal discharge (at levels .2, 1.1, and 5.2 mg/L), lower body weights (at 5.2 mg/L only), and abnormal blood chemistry parameters (at 5.2 mg/L only); a 1.1 mg/L NOAEC was reported.
The NOAELs reported in subchronic oral exposure studies were 15 mg/kg/day and 150 mg/kg/day Butyl Ethyl Propanediol (90 days) in male and female rats, respectively. In 90-day studies, a NOAEL of 600 mg/kg/day was reported for Methylpropanediol and NOAELs of 1000 mg/kg/day were reported for Propanediol and Hexanediol (in females; 400 mg/kg/day NOAEL in males) in oral exposure studies in rats. An evaluation of oral exposure to 5 or 10 mL/kg Propanediol for 15 weeks in rats resulted in 100% mortality (5 deaths) at 10 mL/kg and 2 deaths at 5 mL/kg. In the male rats dosed with Hexanediol, mentioned above, a treatment-related decrease (in males at 1000 mg/kg/day) in mean body weights and a statistically significant increase in organ weights (in males at 400 and 1000 mg/kg/day) were observed. The rats dosed with Methylpropanediol showed decreased liver enzymes and inorganic phosphate (at 1000 mg/kg/day). In rats dosed with Butyl Ethyl Propanediol, there were 4 treatment-related deaths (males at 150 or 1000 mg/kg/day), abnormal respiration 1 to 2 hours post-dosing (after which animals returned to normal), and urinary (at 150 and 1000 mg/kg/day) and kidney abnormalities (at ≥15 mg/kg/day) reported.
In subchronic inhalation studies, rats were exposed to 1,4-Butanediol 2 hours/day for 4 months; a NOAEC of 500 mg/L (equivalent to approximately 23 mg/kg/day) and a LOAEC of 1500 mg/L (equivalent to about 85 mg/kg/day) were reported. Effects at the reported LOAEC included a sleepy condition 20 minutes after each exposure and a histopathological exam revealed pulmonary abnormalities. In 14-wk studies of diacetyl (potential metabolite of 2,3-Butanediol) in mice and rats, significant increases in neutrophil counts consistent with inflammation were observed at 50 and 100 ppm (mice) and at 100 ppm (rats). Significantly increased incidences of exposure-related, non-neoplastic lesions occurred in the respiratory tract of male and female rats and mice, primarily in the 50 and 100 ppm groups, and the highest number of lesions occurred in the nose.
In a chronic study, rats were orally exposed to .25, 3, or 30 mg/kg 1,4-Butanediol for 6 months. At the 30 mg/kg dosage, blood cholinesterase activity was reduced, the ratio of blood serum protein fractions changed, the –SH (thiol) groups in whole blood and the brain decreased, liver glycogen and choline esterase activity decreased, vitamin C in organs decreased, and there was an increase in blood serum transaminases. A substantial increase in the auto-diffusion coefficient of tissue fluid was found in the liver and brain with the 3 and 30 mg/kg dosages. At the 30 mg/kg dosage, the morphological changes were observed.
In rat studies evaluating oral Propanediol exposures up to 1000 mg/kg/day, spermatogenic endpoints were unaffected (90-day exposure) and no maternal or fetal toxic effects were observed (dosing on days 6-15 of gestation). A NOAEL of 100 mg/kg/day and a LOAEL of 300 mg/kg/day 1,4-Butanediol were reported for maternal (dosing on days 6-15 of gestation) and developmental toxicity in an oral exposure mouse study; maternal central nervous system intoxication and maternal and fetal body weight reduction were observed at the LOAEL. Results reported in male and female rats orally exposed to 1,4-Butanediol for 14 days before mating and, with dosing continuing in females through day 3 of lactation, included a developmental NOEL of 400 mg/kg/day (pup weight was slightly, but statistically significantly decreased on lactation day 4 at 800 mg/kg/day, effect was secondary to maternal reduction in body weight), parental transient hyperactivity (at 200 and 400 mg/kg/day) and reversible parental hypoactivity (≥400 mg/kg/day), but no parental reproductive parameters were changed by treatment. A NOAEL of 1000 mg/kg/day Hexanediol (dosing on days 6-19 of gestation) and Methylpropanediol (dosing on days 0-29 of gestation) was reported in oral exposure studies for maternal and developmental effects in rats. In another oral exposure study, the NOAEL for maternal effects was 150 mg/kg/day Butyl Ethyl Propanediol in rats (dosing on days 6-19 of gestation); 1000 mg/kg/day caused staggering, slow respiration, and reduced food consumption and body weights in the dams. The NOAEL for developmental effects was 1000 mg/kg/day Butyl Ethyl Propanediol in this study.
Genotoxicity experiments conducted in vitro evaluating Propanediol were negative in a mammalian cell gene mutation assay (up to 5000 µg/mL), a chromosomal aberration test (up to 5000 µg/mL), and an Ames test (up to 5000 µg/plate). Another mammalian chromosomal aberration test (2500 µg/mL, without metabolic activation) that evaluated Propanediol resulted in positive responses for genotoxicity, however the same test (up to 5000 µg/mL Propanediol) performed with metabolic activation yielded negative results. 1,4-Butanediol was negative for genotoxicity in a Salmonella typhimurium mutagenicity test (up to 10,000 µg/plate), in an Ames test (up to 10,000 µg/plate), in a mammalian cell gene mutation assay (up to 5000 µg/mL), and in a chromosomal aberration test (up to 5000 µg/mL). 2,3-Butanediol was negative in an Ames II™ test (up to 5000 µg/mL). In an Ames test (up to 5000 µg/plate) 1,5-Pentanediol was negative for genotoxicity. Hexanediol was negative for genotoxicity in an Ames test (up to 5000 µg/plate), in a mammalian chromosomal aberration test (up to 1.2 µg/mL), and in a mammalian cell gene mutation assay (up to 5000 µg/mL). 1,10-Decanediol (1.2% in a trade name mixture also containing unspecified amounts of Propylene Glycol or Butylene Glycol) was negative in an Ames test (up to ∼120 µg/plate 1,10-Decanediol). Methylpropanediol was negative in a reverse mutation assay (up to 5000 µg/plate) and in a chromosomal aberration test (up to 5000 µg/plate). Butyl Ethyl Propanediol was negative for genotoxicity in an Ames test (up to 5000 µg/plate) and in a mammalian cell gene mutation assay (up to 7.2 mmol/L); Isopentyldiol was negative for genotoxicity in an Ames test (up to 10,000 µg/plate) and in a liquid suspension assay (up to 100 mg/plate). Tests performed in rat liver and testicular homogenates from rats that were fed 500 ppm Propanediol in the diet for 15 weeks (controls fed plain diet), showed that the hepatic DNA-protein and DNA-crosslinking at 10 and 15 weeks were higher than controls, and the testicular DNA-protein and DNA-crosslinking of treated rats were slightly higher than controls at 15 weeks. The study authors concluded that Propanediol was converted to malondialdehyde in vivo, causing damage to rat DNA. Mouse micronucleus tests conducted in vivo were non-mutagenic for Propanediol (single dose of 2150 mg/kg bw) and for Butyl Ethyl Propanediol (single dose up to 1250 mg/kg).
Carcinogenicity studies data on alkane diol ingredients were not found in the published literature, and unpublished data were not submitted. An inhalation study was conducted by NTP in which B6C3F1/N mice and Wistar Han [CRL:WI (Han)] rats were exposed to diacetyl vapor by whole body inhalation at concentrations up to 50 ppm, for 6 hours and 12 min/day, 5 days/wk, for 105 wks. In mice, there was no evidence of carcinogenic activity of diacetyl in males, but there was equivocal evidence of carcinogenic activity in females, based on the occurrences of adenocarcinoma of the nose. In rats, there was some evidence of carcinogenic activity of diacetyl in males and females based on the combined incidences of squamous cell papilloma and squamous cell carcinoma of the nose in males and incidences of squamous cell carcinoma of the nose of females.
The degree of structural damage induced by certain hexacarbon compounds (6-carbon compounds) to the central and peripheral nervous system has been attributed to differential serum levels of the common metabolite, 2,5-hexanedione. The production of neuropathy is dependent on the specific molecular configuration of hexacarbon compounds, and a γ-spacing of the two keto groups is needed. Additionally, it is suggested that although pyrrole formation is necessary for the pathogenesis of γ-diketone neuropathy, autoxidation to reactive intermediates, resulting in inter- or intraneurofilament crosslinking, is required for the neurotoxic effects of γ-diketones to be manifested.
1,10-Decanediol (1.2% in a trade name mixture also containing an unspecified amount of Butylene Glycol) was non-irritating in an in vitro test evaluating the test substance on reconstructed human epidermis.
Undiluted Propanediol, 1,4-Butanediol, 2,3-Butanediol, 1,5-Pentanediol, or Isopentyldiol was non-irritating to slightly or minimally irritating to the skin of rabbits in 20-to 24-hour patch tests. Undiluted 1,4-Butanediol was minimally irritating when applied to rabbit ears. Hexanediol was non-irritating to guinea pig skin (45% test substance applied) and rabbit skin (80% test substance applied) in 24-hour patch tests. 1,10-Decanediol (1.2% in trade name mixture also containing an unspecified amount of Propylene Glycol) was non-irritating to rabbit skin in a 24 h occlusive patch test. Methylpropanediol (concentration not specified) was non-irritating to rabbit skin. Undiluted Butyl Ethyl Propanediol was non-to-mildly irritating to rabbit skin in 4-hour semi-occlusive patch tests.
A single, dermal application of undiluted Propanediol was non-irritating in human subjects (no further details). 1,4-Butanediol was non-irritating in a patch test on human subjects (concentration not specified). 1,5-Pentanediol (5%) was non-irritating in a 24-hour occlusive patch test in human subjects. 1,10-Decanediol (1.2% in trade name mixture also containing an unspecified amount of Butylene Glycol) was well-tolerated, according to study authors (2 subjects showed mild erythema 1 h following patch removal) in a 48-hour occlusive patch test. Methylpropanediol (100%, 50% aqueous dilution) was non-irritating to subjects with sensitive skin in a 14-day cumulative irritation study. Slight irritation was observed in a 48-hour Finn chamber skin test evaluating unspecified concentrations of Isopentyldiol. Generally, the alkane diols were non-to-slightly irritating in human skin.
The following treatments were negative in tests for the induction of dermal sensitization in guinea pigs: Propanediol (2.5% intradermal and 100% epicutaneous concentrations applied at induction, 50% at challenge), 1,4-Butanediol (10% intradermal and 30% topical concentrations applied at induction and challenge), 2,3-Butanediol (5% intradermal and 50% epicutaneous concentrations applied at induction, 25% at challenge), Hexanediol (5% intradermal and 50% epicutaneous concentrations applied at induction, 25% at challenge), 1,10-Decanediol (1.2% in a trade name mixture containing an unspecified amount of Propylene Glycol or Butylene Glycol) in a Buehler test (1.2% 1,10-Decanediol in trade name mixture used at induction and .3% 1,10-Decanediol in trade name mixture used at challenge), Butyl Ethyl Propanediol (2.5% intradermal and 100% topical concentrations applied at induction, 50% and 100% at challenge), and Isopentyldiol (10% intradermal and 100% topical concentrations applied at induction, 50% at challenge). In another test, strong erythema was reported in guinea pigs with Hexanediol challenge (no concentration specified) following induction (sensitization) with another compound (.2% hydroxyethyl methacrylate); however no Hexanediol induction (.2%)/Hexanediol challenge (no concentration specified) tests showed a positive sensitization reaction. Methylpropanediol showed mild sensitization potential in guinea pigs (10% intradermal to 100% epidermal concentrations applied at induction, up to 100% at challenge).
Propanediol (5 to 75% concentrations applied at induction and challenge) was non-sensitizing in human subjects; mild erythema was reported in 4 subjects during induction (75% only) after the 1st of 9 applications. 1,4-Butanediol (concentration not specified), and 1,5-Pentanediol (5% or 25% in different tests) were non-sensitizing in human subjects. Methylpropanediol (undiluted) was non-sensitizing in one test and showed mild skin sensitization potential in another test (50% aqueous dilution applied at induction and challenge). However, the study authors concluded that it was unclear as to whether or not the skin reactions were caused by irritation, allergy, or an atopic condition. An additional study showed that Methylpropanediol (21.2% applied at induction and challenge) induced erythema and damage to epidermis in some subjects during induction, however the reactions discontinued after a new skin site in those subjects was tested under semi-occlusive conditions; Methylpropanediol was non-sensitizing in that study. Overall, the alkane diols were non-sensitizing to human subjects.
1,10-Decanediol (1.2% in a trade name mixture also containing an unspecified amount of Butylene Glycol) was non-phototoxic in guinea pig skin. Undiluted Isopentyldiol was neither a photo-irritant nor a photo-sensitizer when tested in guinea pig skin.
Human subjects were treated with 1,5-Pentanediol (5%) on the forearms, followed by UVA/UVB exposure. Results from a 24-hour occlusive patch test to the treated skin revealed that the test substance was non-phototoxic and non-photosensitizing.
Experiments evaluating 1,10-Decanediol (1.2% in a trade name mixture also containing an unspecified amount of Butylene Glycol) performed in vitro showed moderate irritation potential in a hen’s egg test, and was non-irritating in a test on reconstructed human corneal epithelium.
Undiluted Propanediol, 1,4-Butanediol, 2,3-Butanediol, 1,5-Pentanediol, and Hexanediol were non-to-slightly irritating or mildly irritating in rabbit eyes. 1,10-Decanediol (1.2% in a trade name mixture also containing an unspecified amount of Propylene Glycol) was slightly irritating to rabbit eyes. Methylpropanediol (undiluted) was non-irritating to rabbit eyes. Isopentyldiol was also non-irritating to rabbit eyes (concentration not specified). In contrast, undiluted Butyl Ethyl Propanediol caused severe injury in rabbit eyes, including irritation, corneal opacification, partial eyelid eversion, all of which were reversible.
In a 6-week study investigating the therapeutic effect of 1,5-Pentanediol (25% in a cream formulation) plus hydrocortisone (1%) compared to only hydrocortisone (1%) on patients with atopic dermatitis, there were 2 instances in each treatment group of a slight skin burning sensation after application. In the group treated with hydrocortisone and 1,5-Pentanediol, a statistically significant decrease in S. aureus colonies at weeks 2 and 6 of treatment was observed, which was not seen with treatment of hydrocortisone alone.
In a 6-month clinical trial evaluating the therapeutic effect of 1,5-Pentanediol (25% in a gel formulation) on herpes labialis in patients with recurrent herpes episodes, there were no treatment-related adverse events reported; body weight and temperature, heart rate, and clinical parameters were nearly unchanged.
Information from case reports for the alkane diols included allergic contact dermatitis as a result of dermal exposure to 1,5-Pentanediol (.5 to 10%) in various creams; recommendations by study researchers for dental professionals exposed to Hexanediol in dentin primers to take precautions because of the potential to cause contact dermatitis following repeated occupational exposure; the adverse effects in adults (non-fatal cases occurred with doses between 1 to 14 g, fatalities occurred with 5.4 to 20 g doses) and poisoning in children (with 14% 1,4-Butanediol by weight) from oral exposure to 1,4-Butanediol.
Discussion
The Panel reviewed the safety of 10 alkane diols and determined that the data were sufficient to determine safety for seven of the ingredients, but insufficient to determine safety of the remaining three ingredients (i.e., 1,4-Butanediol, 2,3-Butanediol, and Octanediol). Maximum concentrations of use were reported for several of the ingredients, and these concentrations ranged from .006% (1,10-Decanediol) to 39.9% (Propanediol) in leave-on products. 1,4-Butanediol has uses reported in the VCRP, but concentrations of use were not reported in response to the industry survey. Therefore, because 1,4-Butanediol can be metabolized into gamma-hydroxybutyric acid (GHB), which is a controlled substance in the United States, and because of the wide range of use concentrations reported for the other ingredients, the Panel stated that concentration of use data are needed to determine safety of 1,4-Butanediol.
Concentration of use data were not available for 2,3-Butanediol (not reported to be in use) and Octanediol, and additional data were also lacking. The complete list of data needed to assess safety of these two ingredients includes: • Concentration of use; • 28-day dermal toxicity studies; • Dermal and reproductive toxicity data; and • Mammalian genotoxicity studies, if the ingredients are not used at low concentrations.
2,3-Butanediol can exist as 3 different stereoisomers. Data on the toxicities of these isomers would help to inform the safety assessment.
Variations in the regiochemistry of small alkane diols may lead to significant differences in toxicity. For example, 2,5-hexanediol, which is not a cosmetic ingredient, is known to be a neurotoxic metabolite of hexane. However, the structurally similar cosmetic ingredient, Hexanediol (i.e., 1,6-hexanediol), is not a neurotoxin. The Panel discussed whether there was concern that 2,5-hexanediol could be present as a significant impurity of Hexanediol (aka 1,6-hexanediol). The Panel determined that, based on the low maximum concentration of Hexanediol reported (.5% in leave-on dermal contact cosmetics) and the >96% purity reported for Hexanediol, the potential presence of 2,5-hexanediol would be toxicologically insignificant.
During the initial review of this safety assessment, the Panel requested neurotoxicity data for Isopentyldiol. No data were received in response to this request. However, because oral toxicity studies with Isopentyldiol reported no adverse clinical or histopathological changes, and due to the fact that bioactivation to a diketone similar to 2,5-hexanediol requires a very specific pathway and was not likely to occur, the Panel no longer felt these data were needed.
Although positive results were obtained in one mammalian chromosomal aberration test at one concentration of Propanediol (2500 µg/mL without metabolic activation), another mammalian chromosomal aberration test reported negative results at concentrations up to 5000 µg/mL Propanediol (with metabolic activation). Additionally, the genotoxicity data for the other alkane diols were largely negative, supporting the fact that genotoxicity was not a likely concern for Propanediol. Furthermore, the Panel noted that carcinogenicity data were absent, but because the genotoxicity data were largely negative, carcinogenicity data were not needed.
Alkane diols, especially lower molecular weight alkane diols such as 1,3-Propanediol, can enhance the penetration of other ingredients through the skin. The Panel cautioned that care should be taken in formulating cosmetic products that may contain these ingredients in combination with any ingredients whose safety was based on their lack of dermal absorption data, or when dermal absorption was a concern.
Some of the alkane diols used as cosmetic ingredients, such as Propanediol and 2,3-Butanediol, can be derived from plant sources. The Panel expressed concern about pesticide residues and heavy metals that may be present in botanically sourced ingredients, and they stressed that the cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit any potential impurities.
The Panel discussed the issue of incidental inhalation exposure from perfumes, hair sprays, deodorant sprays, and face powders. The data that are available from animal inhalation studies, including acute and short-term exposure data, suggest little potential for respiratory effects at relevant doses by the ingredients that are used in products that could incidentally be inhaled. Propanediol (up to 3%) and Isopentyldiol (up to 5%) are reportedly used in cosmetic products that may be aerosolized and Isopentyldiol is used up to .33% in face powder that may become airborne. The Panel noted that most of the droplets/particles produced in cosmetic aerosols and loose-powder cosmetic products would not be respirable to any appreciable amount. The potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Lastly, the Panel noted that for the most part, the alkane diols were not irritants. However, Butyl Ethyl Propanediol (undiluted in one study, concentration not specified in another) was irritating to rabbit eyes. Butyl Ethyl Propanediol is not reported to be used in formulations that are used in the eye area. However, if it were to be included in products used near the eye, those products must be formulated to be non-irritating.
Conclusion

*Not reported to be in current use. Were the ingredient in this group not in current use to be used in the future, the expectation is that it would be used in product categories and at concentrations comparable to others in this group.
The Panel also concluded that the available data are insufficient to make a determination that 1,4-Butanediol, 2,3-Butanediol, and Octanediol are safe under the intended conditions of use in cosmetic formulations.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.