Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of 13 Butyrospermum parkii (shea)-derived ingredients, which are most frequently reported to function in cosmetics as skin and hair conditioning agents. The Panel reviewed the available data to determine the safety of these ingredients. Because final product formulations may contain multiple botanicals, each containing similar constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may be hazardous to consumers. Industry should use good manufacturing practices to limit impurities that could be present in botanical ingredients. The Panel concluded that these ingredients are safe in the present practices of use and concentration when formulated to be non-sensitizing.
Keywords
Introduction
The Panel previously reviewed the safety of Butyrospermum Parkii (Shea) Oil, Butyrospermum Parkii (Shea) Butter, Butyrospermum Parkii (Shea) Butter Unsaponifiables, and Hydrogenated Shea Butter in the 2011 safety assessment of plant-derived fatty acid oils and found these ingredients to be safe as used in cosmetics. 2 Because data from the previous assessment may help to inform the safety of the ingredients listed in this current assessment, the relevant information has been summarized in this report.
Botanicals such as Butyrospermum parkii (shea)-derived ingredients may contain hundreds of constituents, some of which may have the potential to cause toxic effects. In this assessment, the Panel is reviewing the potential toxicity of each of the Butyrospermum parkii (shea)-derived ingredients as a whole, complex mixture.
The ingredient names, according to the Dictionary, are written as listed above, without italics and without abbreviations. When referring to the tree from which these ingredients are derived, the standard scientific practice of using italics will be followed (e.g., Butyrospermum parkii). The shea tree is also known taxonomically as Vitellaria paradoxa and is referred to as such by many references and by the US Food and Drug Administration (FDA).
Shea oleine (“oleine” is an oleate triglyceride) is listed as a cosmetic ingredient in the FDA Voluntary Cosmetic Registration Program (VCRP) database, but it is not an ingredient listed in the Dictionary.1,3 This chemical has been included in this report because use as a cosmetic ingredient may be demonstrated. (Triolein, the triester of glycerin and oleic acid, was previously reviewed by the Panel and was found safe as used in cosmetics). 4
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Chemistry
Definition and Plant Identification
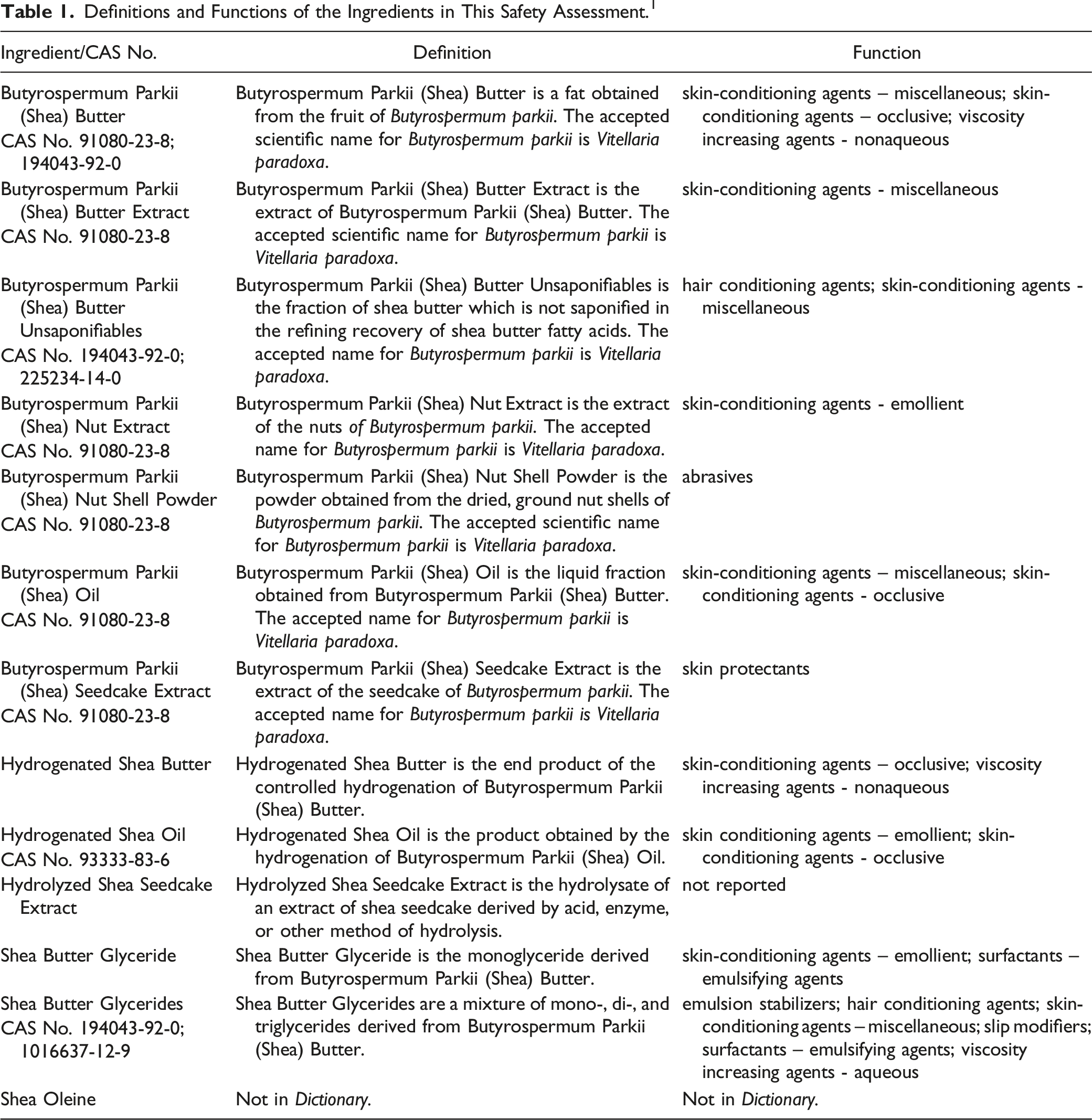
Definitions and Functions of the Ingredients in This Safety Assessment. 1
Physical and Chemical Properties
Butyrospermum Parkii (Shea) Butter, depending on level of refinement, is an off-white or grey to yellowish-cream tallow-like solid, with a specific gravity of .918 at 15°C and a melting point of 37.8°C (reported range: 28-46°C).8-13 Butyrospermum Parkii (Shea) Oil is a pale yellow liquid. 14 Shea Oleine is a clear yellow liquid, with a characteristic fatty odor and a density of .922-.928 at 20°C. 15
Method of Manufacture
The general description of the method of manufacturing of several Butyrospermum parkii (shea)-derived ingredients is described in Figure 1.
16
General description of the manufacturing of Butyrospermum parkii (shea)-derived ingredients.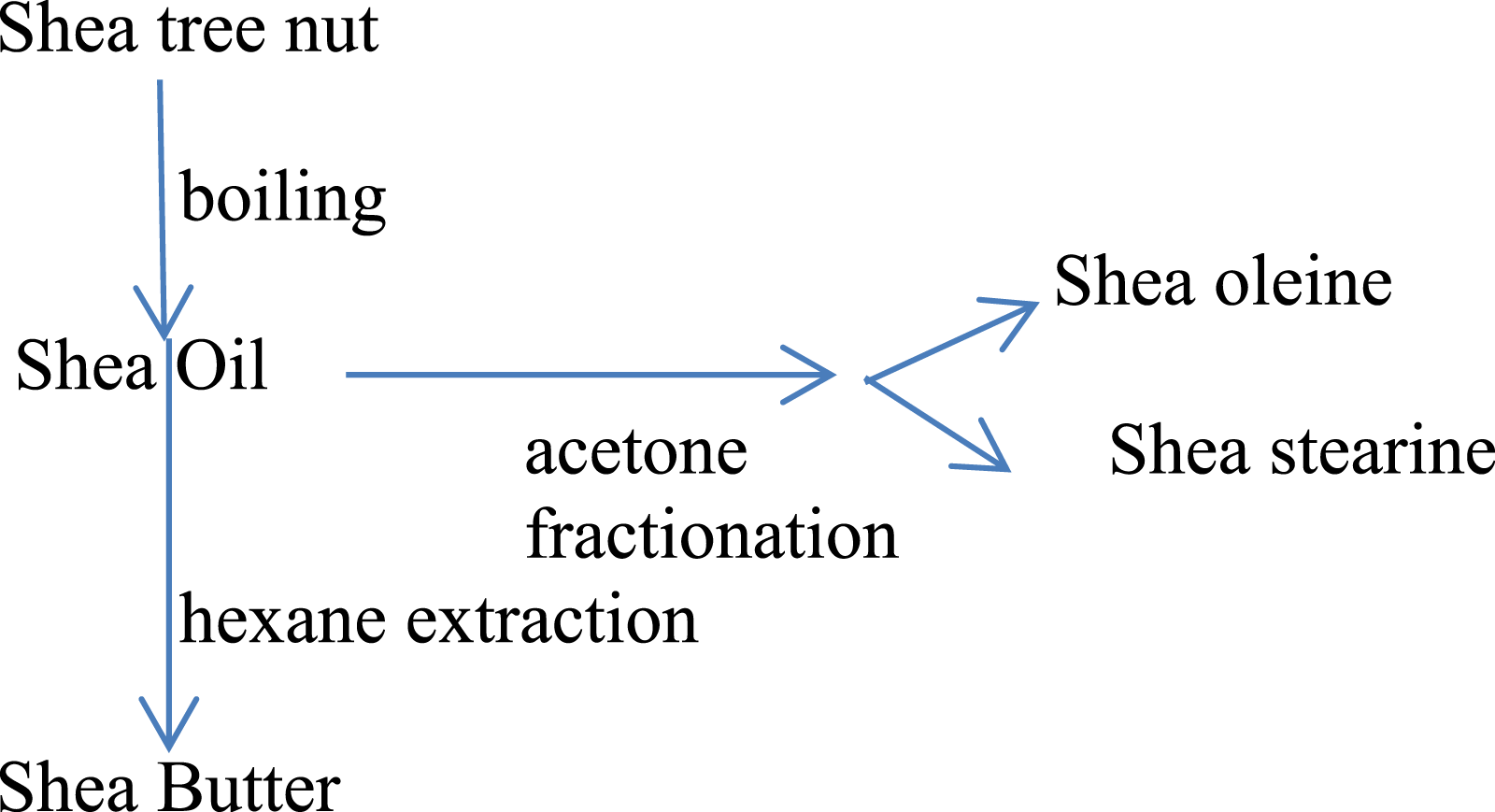
Butyrospermum Parkii (Shea) Butter Unsaponifiables
Butyrospermum Parkii (Shea) Butter Unsaponifiables is obtained by molecular distillation and supercritical carbon dioxide extraction of Butyrospermum Parkii (Shea) Butter. 17
Butyrospermum Parkii (Shea) Nut Shell Powder
According to a supplier, Butyrospermum Parkii (Shea) Nut Shell Powder is obtained by removing the shea seed from the shell and drying the shell in the sun, followed by grinding and sieving the resultant product. 18 The ground shell is then sterilized and examined by quality control.
Butyrospermum Parkii (Shea) Seedcake Extract
Butyrospermum Parkii (Shea) Seedcake Extract is produced by solubilizing the seedcake of Butyrospermum parkii in a mixture of water and butylene glycol (50%/50%, v/v), and then separating the soluble and insoluble phases, filtering, and sterilizing. 19
Composition/Impurities
Mean Concentrations of Tocopherols in 102 Butyrospermum Parkii (Shea) Butter Samples by HPLC Analysis (μg/g). 5

NR-Not reported.
A study of Butyrospermum Parkii (Shea) Butter (described as kernel fats; n-hexane extraction) from 36 samples from seven different countries found the principal triacylglycerols to be stearic-oleic-stearic (mean 31.2% of total triacylglycerols), stearic-oleic-oleic (27.7%), and oleic-oleic-oleic (10.8%).
20
Triterpene ester contents ranged from .5 to 6.5% and consisted of α-amyrin cinnamate (mean 29.3% of total triterpene esters), butyrospermol cinnamate (14.8%), α-amyrin acetate (14.1%), lupeol cinnamate (9.0%), β-amyrin cinnamate (7.6%), lupeol acetate (7.2%), butyrospermol acetate (5.8%), and β-amyrin acetate (4.9%) (Figure 2). Triterpene esters.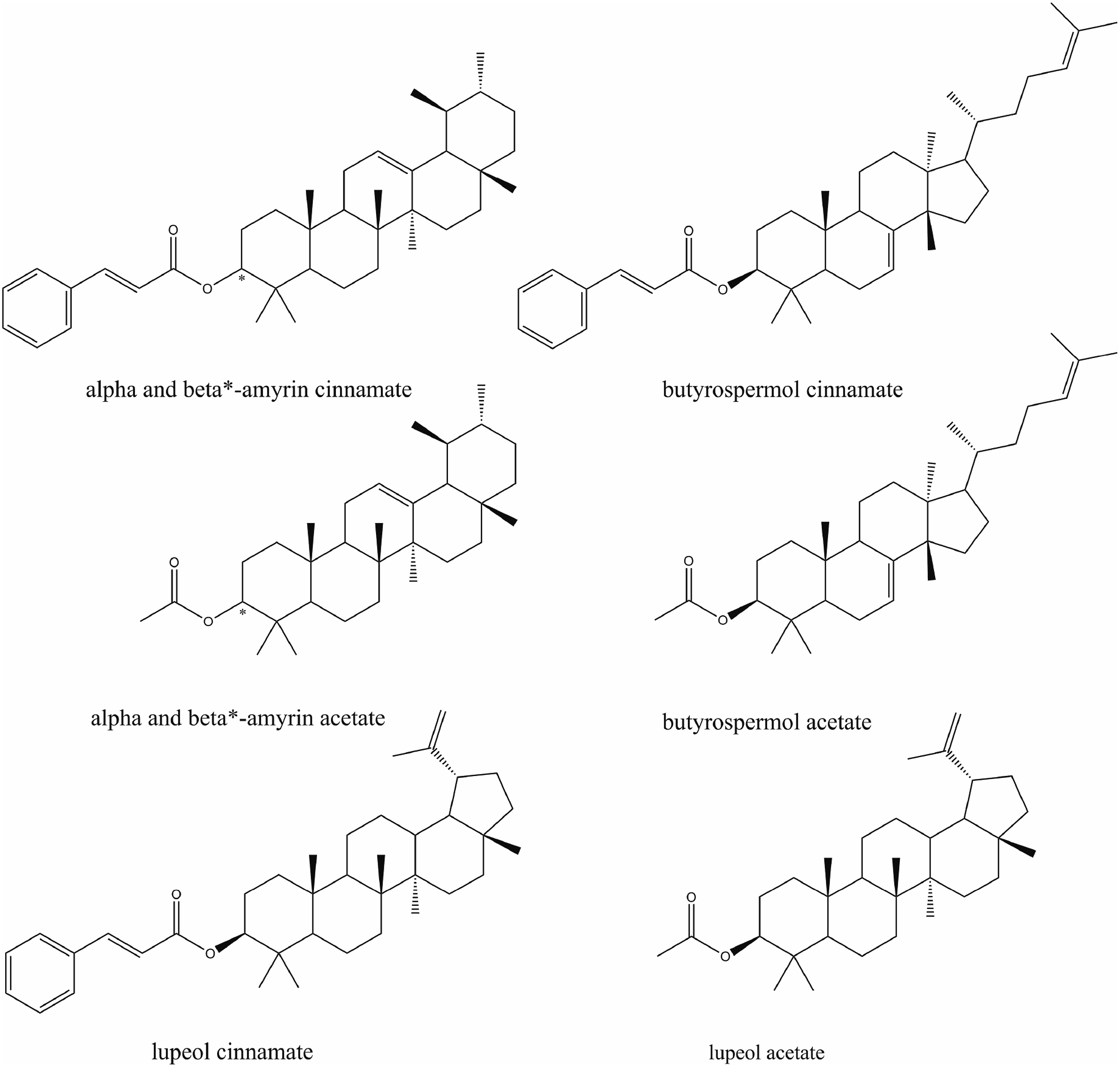
The same researchers identified the content and composition of triterpene alcohol fractions of the non-saponifiable lipids of Butyrospermum Parkii (Shea) Butter from 36 samples. 21 The shea kernels contained 30-54% fat, of which 2-12% were non-saponifiable lipids. Triterpene alcohol content in the non-saponifiable lipids was 22-72%. The triterpene alcohol fractions contained α-amyrin, β-amyrin, lupeol, and butyrospermol with minor or trace amounts of ψ-taraxasterol, taraxasterol, parkeol, 24-methylene-24-dihydroparkeol, 24-methylenecycloartanol, dammaradienol, and 24-methylenedammarenol.
An analysis of the phenolic constituents of shea kernels by liquid chromatography-mass spectrometry (LC-MS) identified the following catechin compounds: gallic acid, catechin, epicatechin, epicatechin gallate, gallocatechin, epigallocatechin, gallocatechin gallate, and epigallocatechin gallate. 7 Quercetin and trans-cinnamic acid were also identified. The mean kernel content of the catechin compounds was 4000 ppm with a range of 2100-9500 ppm.
Butyrospermum Parkii (Shea) Butter Unsaponifiables
Butyrospermum Parkii (Shea) Butter Unsaponifiables mainly contain terpene alcohols present in the butter in the form of cinnamic acid esters (including α- and β-amyrin, lupeol, butyrospermol, and cycloartenol) and phytosterols including α-spinasterol, Δ7-stigmasterol, and stigmasterol). 17
Butyrospermum Parkii (Shea) Nut Shell Powder
A supplier has reported that Butyrospermum Parkii (Shea) Nut Shell Powder does not contain any fragments or pieces of shea seed. 18 This ingredient consists of woody tissues and does not contain asbestos, free amines, antioxidants, formaldehyde, monomers, nitrosamines, ethylene oxide, triethanolamine, 1,4-dioxane, or volatile organic compounds.
Butyrospermum Parkii (Shea) Seedcake Extract
According to a supplier, Butyrospermum Parkii (Shea) Seedcake Extract is composed of 2.8% of the seedcake extract, 50.0% butylene glycol, and 47.2% water. 19 This supplier also reported that alkaloids were less than the limit of sensitivity (.05 g/L in Dragendorff reagent), heavy metal composition included trace levels of nickel (.049 ppm) and lead (.478 ppm), and the sum of the concentrations of aflatoxins B1, B2, G1, and G2 was less than 1.0 µg/kg. The supplier also reported that neither the 26 allergenic compounds regulated by the European Union nor any pesticides were detected in this ingredient.
Shea Oleine
The primary component of shea oleine is the oleate triglyceride (Figure 3). Oleate triglyceride.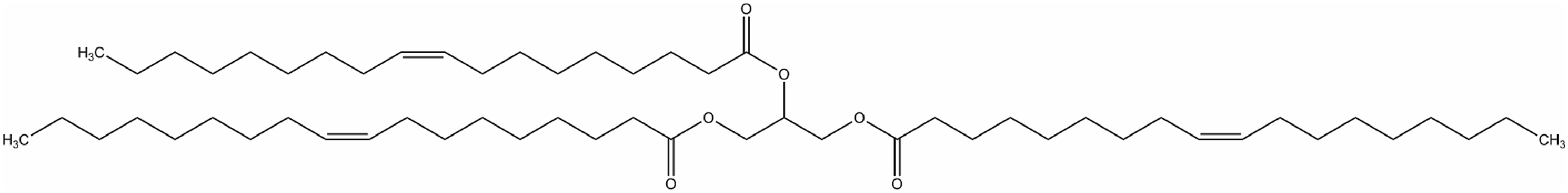
However, the sterol component of shea oleine is approximately 8% (w/w), of which approximately 97% is 4,4-dimethyl sterols (mostly as esters of cinnamic acid), 2% is 4-demethylsterols and .5% is 4-α-methylsterols. 16
Use
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the US FDA and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s VCRP database. Use concentration data are submitted by Industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
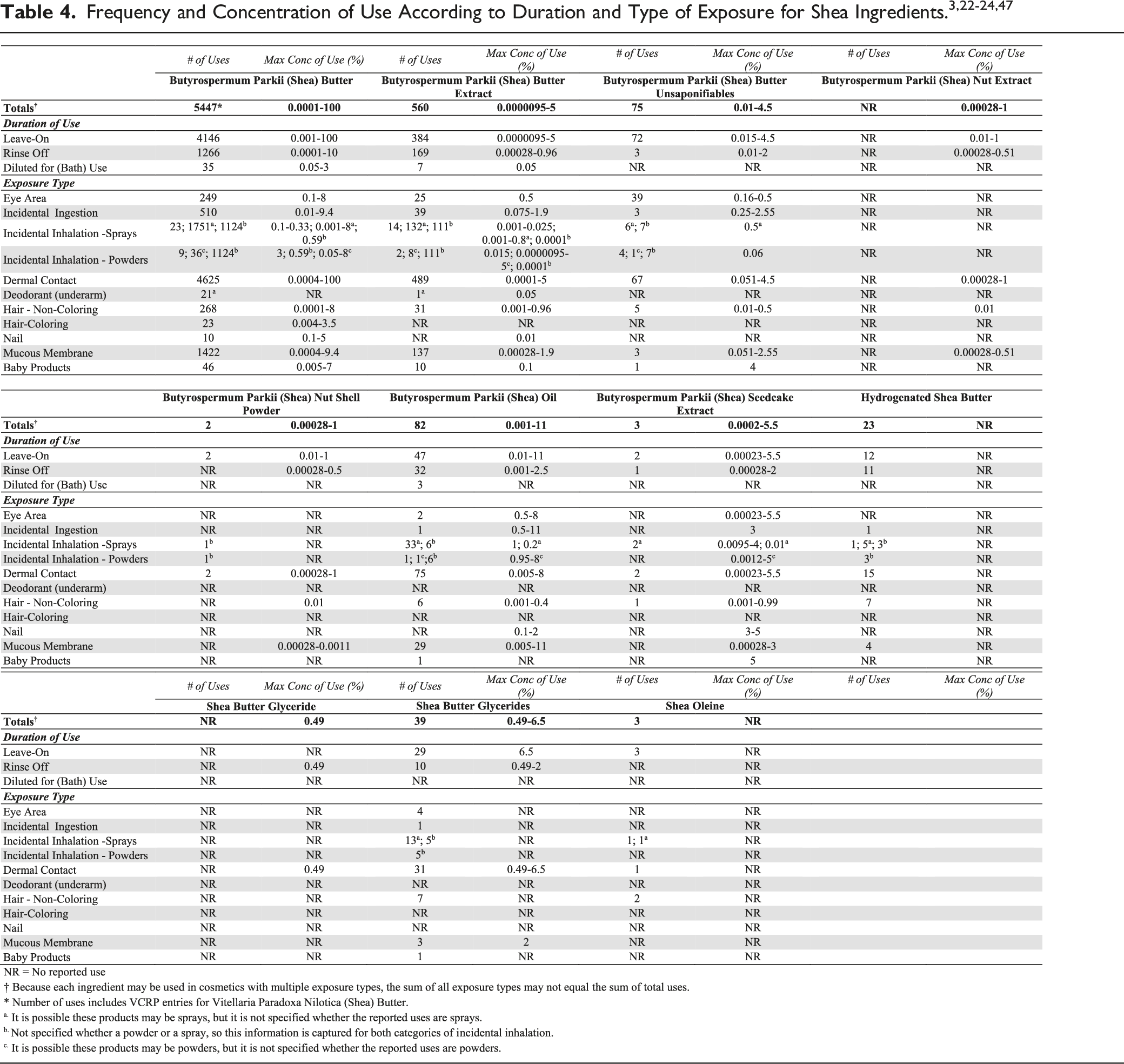
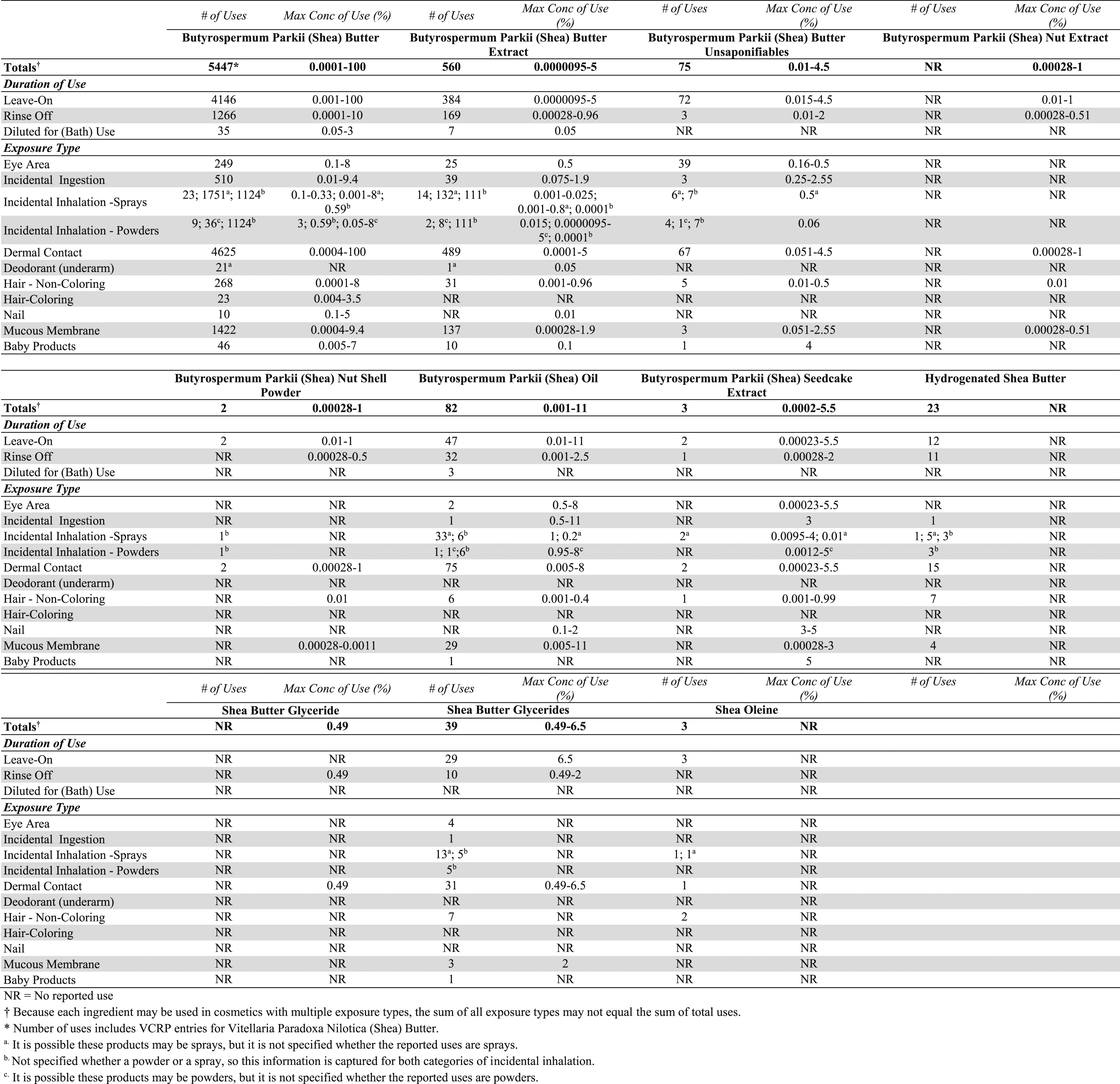
In some cases, reports of uses were received from the VCRP, but no concentration of use data were provided. For example, Hydrogenated Shea Butter is reported to be used in 23 formulations, but no use concentration data were provided. In other cases, no uses were reported to the VCRP, but a maximum use concentration was provided in the industry survey. For example, Shea Butter Glyceride was not reported in the VCRP database to be in use, but the industry survey indicated that it is used at concentrations up to .49%. It should be presumed that Shea Butter Glyceride is used in at least one cosmetic formulation.
Some of these ingredients may be used in products that can be incidentally ingested or come into contact with mucous membranes. For example, Butyrospermum Parkii (Shea) Oil is used in lipsticks at up to 11%. 24 Additionally, some of these ingredients were reported to be used in hair sprays, face powders, fragrances and body and hand sprays and could possibly be inhaled. For example, Butyrospermum Parkii (Shea) Seedcake Extract was reported to be used in fragrance preparations at a maximum concentration of 4% and Butyrospermum Parkii (Shea) Butter was reported to be used in a face powder at 3%. 23 In practice, 95 to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.25-27
The Butyrospermum parkii (shea)-derived ingredients described in this safety assessment are not restricted from use in any way under the rules governing cosmetic products in the European Union. 28
Non-Cosmetic
Butyrospermum Parkii (Shea) Oil (sheanut oil), from which many of the ingredients of this report are derived, is generally recognized as safe (GRAS) in the U.S. as a direct food additive (21CFR§184.1702). It is used in confections and frostings, coatings of soft candy, and sweet sauces and toppings.
Refined sheanut oil is described as a component of a mixture of oils used as a cocoa butter substitute, as a coating agent, and in margarine and shortening in the Food Chemicals Codex, a compendium of internationally recognized standards published by the United States Pharmacopeia (USP) for the purity and identity of food ingredients. 14
A triterpene-rich extract of Butyrospermum parkii has been reported to be used as a dietary supplement for the treatment of osteoarthritis. 29 Other studies have found that components of shea extracts potentially have anti-inflammatory, antioxidant, and anti-tumor effects.30-33
ToxicoKinetic Studies
Absorption, Distribution, Metabolism, and Excretion (ADME)
Animal
Oral
In an oral absorption and excretion study, groups of Colworth Wistar male rats were fed shea oleine in a semisynthetic diet. 34 In a low-dose experiment, groups of 24 rats received control feed, feed containing .5% shea oleine, or feed containing 5% shea oleine for 1 week, with control feed administered to all rats the week prior and the week following the exposure week. In a high-dose experiment, 2 groups of 15 male and 15 female rats received either 10 or 20% shea oleine in the feed for 3 weeks. In the first experiment, feces were collected and pooled weekly for each treatment group throughout weeks 2 and 3. In the second experiment, feces were collected and pooled for each treatment group in week 3 only. The dried fecal matter of the rats was then analyzed with thin-layer and gas-liquid chromatography for fecal lipid, total sterol, differential sterol levels, and, specifically, 4,4-dimethylsterols (the main sterol fraction (∼97%) of shea oleine). Excretion of 4,4-dimethylsterols increased with the consumption of shea oleine. Apparent absorption was 27 to 52% and was estimated from the disappearance of 4,4-dimethylsterols from the feces. The majority of the 4,4-dimethylsterols was excreted unchanged.
Human
Oral
The oral absorption and excretion of shea oleine was studied in 4 male volunteers. 34 On day 3 of an 8 day period, the subjects consumed a single 25 g portion (approximately .4 g/kg) of shea oleine in mayonnaise. No other vegetable fats were consumed during the course of the study. Feces were collected on days 3 to 8 inclusively, freeze-dried, and weighed. The dried fecal matter was analyzed in the manner described above. Excretion of 4,4-dimethylsterols increased with the consumption of shea oleine, with a marked increase from baseline on days 4 and 5 and a return to approximate baseline on day 8. Absorption of 4,4-dimethylsterols was estimated to be 13 to 49%. The majority of the 4,4-dimethylsterols was excreted unchanged.
Toxicological Studies
Acute Toxicity Studies
No relevant published acute toxicity studies on Butyrospermum parkii (shea)-derived ingredients were identified in a literature search for these ingredients, and no unpublished data were submitted.
Subchronic Toxicity Studies
Shea Oleine
In a 13-week rat feeding study, groups of 15 male and 15 female Colworth-Wistar rats received a diet containing 20% (w/w; 10 to 15 g/kg/day) shea oleine or hydrogenated shea oleine. 35 Additional groups of 15 male and 15 female rats were fed either 20% (w/w) palm oil, soy bean oil, or the hydrogenated equivalents. During the exposure period, body weight, food and water consumption, urine chemistry, and clinical pathology were assessed. Gross necropsy and microscopic examination of select tissues and organs were performed at study completion.
Results showed that shea oleine diets produced biological effects similar to those of palm oil and soy bean oil diets. Slightly reduced body weight gain was observed in rats fed either of the shea oleine diets when compared to diets with palm oil and soy bean oil. No significant differences in body weight gains were observed between rats fed hydrogenated shea oleine versus non-hydrogenated shea oleine. Slightly reduced cholesterol levels, increased aminotransferase levels, and lower triglyceride and alanine aminotransferase values were observed in rats fed non-hydrogenated diets, as were increased liver weights and reduced liver-lipid values. These changes were not considered to be biologically significant. Also considered biologically insignificant by the authors were raised alkaline phosphatase levels and increased food consumption in rats fed hydrogenated shea oleine. The authors concluded that all diets were well tolerated in the rats and considered none of the findings in this study to be adverse. 35
Chronic Toxicity Studies
Butyrospermum Parkii (Shea) Oil and Shea Oleine
See Carcinogenicity section below.
Developmental and Reproductive Toxicity (DART) Studies
Oral
Butyrospermum Parkii (Shea) Oil and Shea Oleine
The reproductive toxicity potential of shea oleine and hydrogenated shea oleine was assessed in two dietary studies in rats. 36 In study 1, groups of 20 male and 20 female Colworth-Wistar rats received 7% (w/w; 3.5 g/kg/day) of either type of shea oleine in their diet for 20 weeks (breeding began at week 12 and lasted for 2 weeks). In study 2, groups of 50 male and 50 female Colworth-Wistar rats received 15% (w/w; 7.5 g/kg/day) of either type of shea oleine or Butyrospermum Parkii (Shea) Oil in their diets for 10 weeks (breeding began at week 2 and lasted for 1 week). Both studies also evaluated other commercially available materials, such as palm oil and cocoa butter. The rats received the test materials during pre-mating, mating, pregnancy and offspring weaning. Reproduction was assessed by counting the number of litters, pups born, and pups surviving, and by measuring body weights at birth and at weaning on day 21. Skeletal evaluation using X-ray, clinical pathology and macroscopic examination were performed on F1 rats. Parental animal parameters assessed were body weight, food consumption, clinical pathology, organ weights and macroscopic examination. Fatty acids and hydrocarbon levels were measured, and various tissues were evaluated in F0 animals for lipogranulomata in Study 2.
Slightly decreased body weight gain, reduced cholesterol, and increased alkaline phosphatase activities were observed in rats treated with either shea oleine or hydrogenated shea oleine. No adverse effects on reproduction from any shea materials were observed in either study for any parameter. Results showed that shea oleine, hydrogenated shea oleine, and Butyrospermum Parkii (Shea) Oil were toxicologically comparable to the other commercially available materials used in this study. The authors concluded that there was no evidence of reproductive toxicity following dietary exposure to shea oleine, hydrogenated shea oleine, and Butyrospermum Parkii (Shea) Oil in rats at concentrations equating to 15% (7.5 g/kg/day). 36
Genotoxicity
In Vitro
Butyrospermum Parkii (Shea) Butter and Butyrospermum Parkii (Shea) Butter Unsaponifiables
A material containing Butyrospermum Parkii (Shea) Butter (70%) and Butyrospermum Parkii (Shea) Butter Unsaponifiables (30%) was not mutagenic in an Ames test. 17 The material was tested at 50 to 5000 µg/plate, with and without metabolic activation. No further details were provided.
Carcinogenicity
Oral
Butyrospermum Parkii (Shea) Oil and Shea Oleine
The carcinogenic potential of shea oleine and Butyrospermum Parkii (Shea) Oil were evaluated in a dietary study in Colworth-Wistar rats for 104 weeks. 16 The study also evaluated palm oil. Groups of 50 male and 50 female rats received diets containing 15% (w/w; approximately equivalent to 7.5 g/kg/day) shea oleine, 15% (w/w) Butyrospermum Parkii (Shea) Oil, or 15% (w/w) palm oil. The rats were the offspring of the animals used in the reproduction study described above (study 2) and the test diets began at weaning (21 days of age). The following parameters were assessed: mortality, clinical signs of toxicity, body weight, feed intake, clinical pathology, organ weights and macroscopic and histopathological changes plus tumor type and incidence evaluation.
Final mortality rates for both sexes for shea oleine and Butyrospermum Parkii (Shea) Oil were in the range of 28 to 30% each, while the mortality rates for both sexes exposed to palm oil was 40%. No clinical signs of toxicity were found after exposure to either shea test material. Decreased body weight gain and increased feed intake were observed in rats of both sexes fed either shea diets, while reduced cholesterol was observed in females fed the shea oleine diet. Increased alkaline phosphatase levels were observed in both sexes fed the Butyrospermum Parkii (Shea) Oil diet, but this value was only increased in females fed the shea oleine diet. Decreased heart weights and an increased incidence of pulmonary lipidosis were observed in rats of both sexes fed either shea diet. In females fed either shea diet, an increase in the number of hepatomas was observed, while in males fed shea oleine, increases in pancreatic exocrine adenomas and skin keratoacanthomas were observed. The increase in the incidence of hepatomas was thought to be related to the high fat content of the diets and were interpreted to be consistent with previous tumor data in high fat fed rats. The authors concluded that none of the findings in this study were adverse effects and that shea oleine and Butyrospermum Parkii (Shea) Oil showed no tumorigenic potential in the rat at 15% in the diet (7.5 g/kg/day). 16
Dermal Irritation and Sensitization Studies
Irritation
Dermal Irritation Studies for Butyrospermum parkii (Shea)-Derived Ingredients.
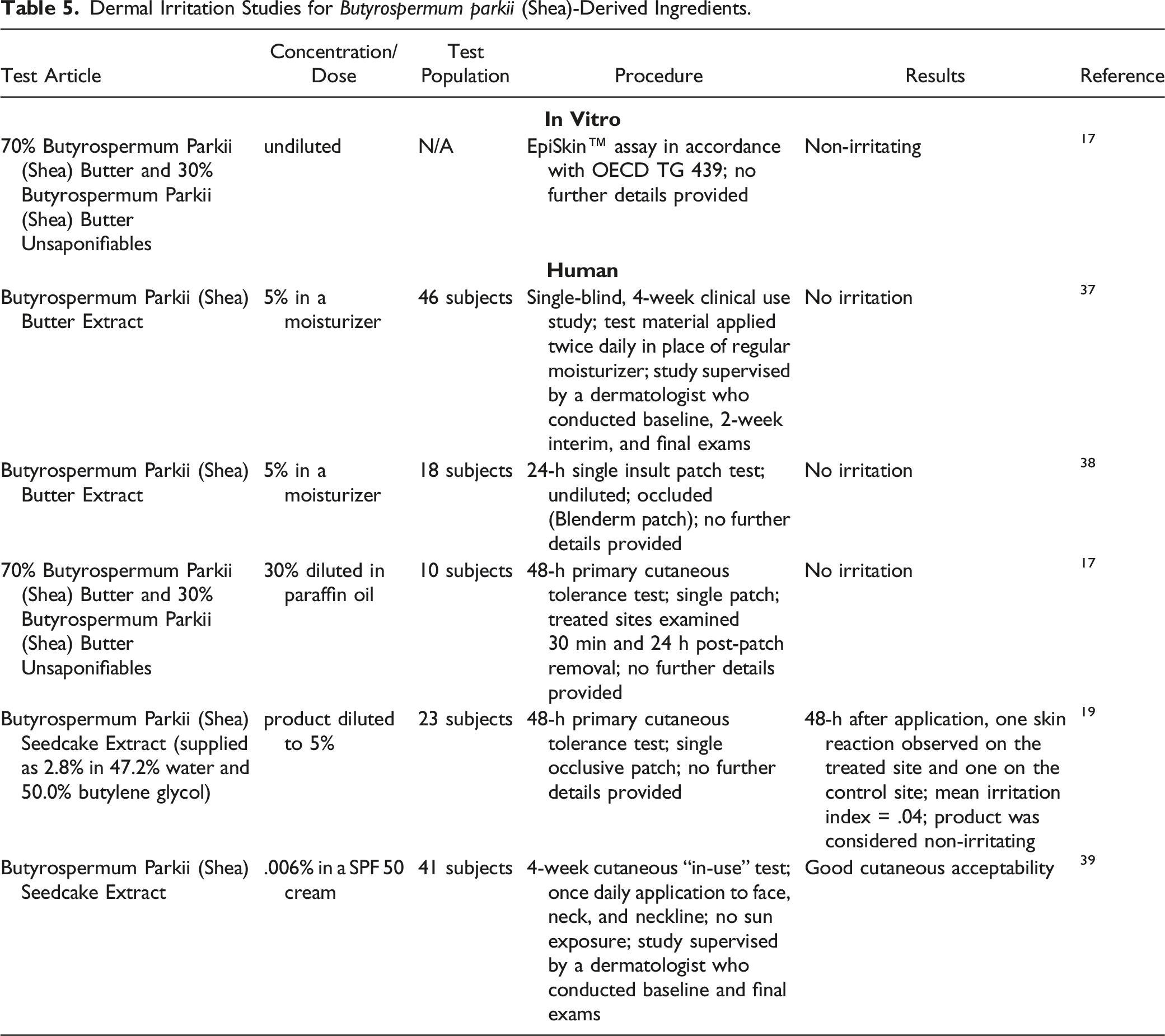
Butyrospermum Parkii (Shea) Butter
In an EpiSkin™ in vitro assay, 24.1% Butyrospermum Parkii (Shea) Butter in a lip wax was not an irritant. 2 In a rabbit study, Butyrospermum Parkii (Shea) Butter (concentration not reported) produced very slight erythema with or without edema in 2/3 rabbits exposed to the test material for 4 h in an irritation study utilizing occlusive patches. The erythema was resolved 3 or 4 days after patching. Butyrospermum Parkii (Shea) Butter did not cause primary cutaneous irritation when tested at up to 2%. No irritation to Butyrospermum Parkii (Shea) Butter was observed in human volunteers for in-use studies of lip gloss or body/hand massage oils at concentrations up to 45%.
Sensitization
Dermal Sensitization Studies for Butyrospermum Parkii (Shea)-Derived Ingredients.
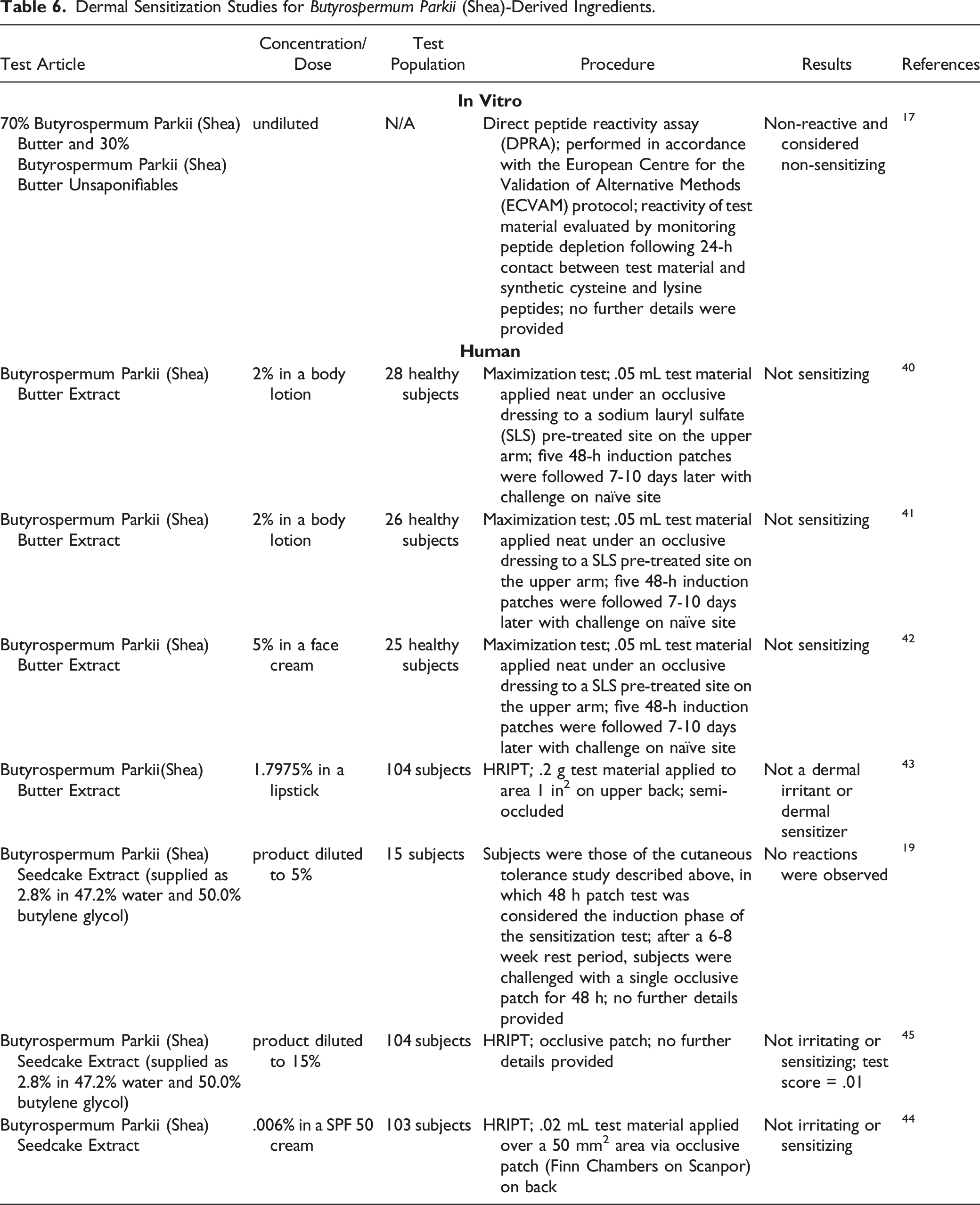
Butyrospermum Parkii (Shea) Butter
Butyrospermum Parkii (Shea) Butter was not sensitizing in a guinea pig maximization study. 2 The induction concentration was 75% and the challenge concentrations were 20 and 50%. No sensitization was observed in multiple HRIPTs with products containing Butyrospermum Parkii (Shea) Butter. Concentrations tested were up to 60%.
Phototoxicity and Photosensitization
In Vitro
Butyrospermum Parkii (Shea) Butter and Butyrospermum Parkii (Shea) Butter Unsaponifiables
A material containing Butyrospermum Parkii (Shea) Butter (70%) and Butyrospermum Parkii (Shea) Butter Unsaponifiables (30%) was considered non-phototoxic in a 3T3 NRU assay when tested at .005 to 1 mg/mL. 17 This study was performed in accordance to Organization for Economic Co-operation and Development’s (OECD) TG 432. No further details were provided.
Animal
Butyrospermum Parkii (Shea) Butter
Butyrospermum Parkii (Shea) Butter was not phototoxic in guinea pigs when tested at 10 and 20% in acetone. 2 The test sites were irradiated with UV-B light for 80 seconds followed by UV-A light for 80 min.
Ocular Irritation Studies
Animal
Butyrospermum Parkii (Shea) Butter
While mild conjunctival reactions were observed, undiluted Butyrospermum Parkii (Shea) Butter was considered not irritating when tested in the eyes of male rabbits. 2
Mucous Membrane Irritation Studies
In Vitro
Butyrospermum Parkii (Shea) Butter and Butyrospermum Parkii (Shea) Butter Unsaponifiables
A balm containing 1.5% of the mixture Butyrospermum Parkii (Shea) Butter (70%) and Butyrospermum Parkii (Shea) Butter Unsaponifiables (30%) was considered non-irritating in a SkinEthic™ reconstituted mucous membrane model. 17 Approximately 10 µl of the balm was applied undiluted for 24 h. The negative control was phosphate buffer saline and the positive control was .1 and 3% sodium dodecyl sulfate. No further details were provided.
Summary
The 13 Butyrospermum parkii (shea)-derived ingredients detailed in this report function mainly as skin and hair conditioning agents in personal care products. Studies on Butyrospermum Parkii (Shea) Butter and Butyrospermum Parkii (Shea) Butter Unsaponifiables included in the 2011 safety assessment of plant-derived fatty acid oils are not included this this summary. The original safety assessment found these ingredients to be safe as used in cosmetics.
According to 2017 VCRP data, Butyrospermum Parkii (Shea) Butter has the most reported uses of the ingredients listed in this safety assessment in cosmetic products, with a total of 5447; nearly three-fourths of the uses are in leave-on formulations. Butyrospermum Parkii (Shea) Butter Extract has the second greatest number of overall reported, with a total of 560; about two-thirds of the uses are in leave-on formulations. The results of the concentration of use survey conducted in 2016 by the Council indicate Butyrospermum Parkii (Shea) Butter has the highest reported maximum concentration of use; it is used at up to 100% in moisturizers. Butyrospermum Parkii (Shea) Oil is used at up to 11% in a lipstick. No uses were reported for Hydrogenated Shea Oil or Hydrolyzed Shea Seedcake Extract.
Butyrospermum Parkii (Shea) Oil is a GRAS direct food additive in the U.S. It is used as a cocoa butter substitute in confections and frostings, coatings of soft candy, and sweet sauces and toppings. It is also used as a margarine or shortening. Components of shea extracts reportedly have potential anti-inflammatory, antioxidant, and anti-tumor effects.
Oral absorption and excretion studies of rats fed up to 20% shea oleine in a semisynthetic diet found excretion of 4,4-dimethylsterols increased with the consumption of shea oleine. Apparent absorption of shea oleine was 27 to 52%, as measured by 4,4-dimethylsterols. The majority of the 4,4-dimethylsterols was excreted unchanged. The findings for the absorption and excretion of approximately .4 g/kg in a single dose study of human volunteers were similar, with the absorption of shea oleine estimated to be 13 to 49%, as measured by 4,4-dimethylsterols.
In a 13-week rat feeding study, shea oleine or hydrogenated shea oleine (20% w/w, equivalent to 10-15 g/kg/day, for both test materials) did not produced adverse effects. No reproductive effects were observed in rats fed shea oleine, hydrogenated shea oleine, or Butyrospermum Parkii (Shea) Oil (up to 15% w/w, equivalent to 7.5 g/kg/day, for all test materials) for up to 20 weeks. No tumorigenic potential or adverse effects to shea oleine or Butyrospermum Parkii (Shea) Oil (15% w/w, equivalent to 7.5 g/kg/day) were observed in a carcinogenicity study in the offspring of the rats from the reproductive study.
A material containing Butyrospermum Parkii (Shea) Butter (70%) and Butyrospermum Parkii (Shea) Butter Unsaponifiables (30%) was not mutagenic in an Ames test.
A material containing Butyrospermum Parkii (Shea) Butter (70%) and Butyrospermum Parkii (Shea) Butter Unsaponifiables (30%) was non-irritating in an EpiSkin™ assay when tested undiluted and in a human primary cutaneous tolerance tested at a 30% dilution in paraffin oil. Butyrospermum Parkii (Shea) Butter Extract at 5% in a moisturizer and Butyrospermum Parkii (Shea) Seedcake Extract at up to .14% were not irritating in human irritation studies.
A material containing Butyrospermum Parkii (Shea) Butter (70%) and Butyrospermum Parkii (Shea) Butter Unsaponifiables (30%) was considered non-sensitizing in a DPRA when tested undiluted. Butyrospermum Parkii (Shea) Butter Extract was non-sensitizing in human patch tests at up to 5% in formulation. Butyrospermum Parkii (Shea) Seedcake Extract did not produce sensitization reaction in humans in a non-standard sensitization study (at .14%) or in HRIPTs (at up to .42%).
A material containing Butyrospermum Parkii (Shea) Butter (70%) and Butyrospermum Parkii (Shea) Butter Unsaponifiables (30%) was considered non-phototoxic in a 3T3 NRU assay when tested at .005 to 1 mg/mL.
A balm containing 1.5% of the mixture Butyrospermum Parkii (Shea) Butter (70%) and Butyrospermum Parkii (Shea) Butter Unsaponifiables (30%) was considered non-irritating in a SkinEthic™ reconstituted mucous membrane model.
No relevant published acute toxicity or case reports on Butyrospermum parkii (shea)-derived ingredients were identified in a literature search for these ingredients and no unpublished data were submitted.
Discussion
There are no irritation or sensitization data for Butyrospermum Parkii (Shea) Nut Extract and Butyrospermum Parkii (Shea) Nut Shell Powder, and no irritation or sensitization data for Butyrospermum Parkii (Shea) Seedcake Extract and Butyrospermum Parkii (Shea) Butter at maximum use concentrations (5.5 and 100% in leave-on products, respectively). However, human repeated patch tests for Butyrospermum Parkii (Shea) Seedcake Extract and Butyrospermum Parkii (Shea) Butter were negative at lower concentrations (.42 and 70%, respectively). Based on the Panel’s clinical experience, the absence of adverse event reports, and the available negative safety test data (including negative results from DPRAs), the Panel does not expect dermal irritation or sensitization following exposure to these ingredients at reported use concentrations.
The Panel noted that, because botanical ingredients are complex mixtures, there is concern that multiple botanical ingredients in one formulation may each contribute to the final concentration of a single constituent. While there are no specific constituents of concern detailed for the Butyrospermum parkii (shea)-derived ingredients described in this safety assessment, not all constituents have been identified, especially for the nut shell and seedcake ingredients. Therefore, when formulating products, manufacturers should avoid reaching levels in final formulation of known botanical constituents that may cause sensitization or other adverse effects.
The Panel discussed the issue of incidental inhalation exposure from hair sprays, fragrance preparations, body and hand sprays, and face powders. There were no inhalation toxicity data available. The Panel noted that droplets/particles from spray and loose-powder cosmetic products would not be respirable to any appreciable amount. The potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
The Panel also expressed concern about pesticide residues, heavy metals, and other plant species that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit impurities.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that the following 13 ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment when formulated to be non-sensitizing.
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.