Abstract
This commentary discusses the implementation of fasting in nonclinical animal experimental subjects. The short-term removal of food from cages of experimental animals is in all respects innocuous. The term “stress” is ill-defined and the statutes and regulations governing animal research laboratories that exert their authority in the performance of their operations do so without substantive grounds to base compliance. The legislative and administrative history of the implementation of the Animal Welfare Act (AWA) has evolved into the development of laboratory management strategies that focus on the reduction of the biological cost of stress to the animals and the determination of when subclinical stress (eustress) becomes distress. Animal welfare is based on the tenet that in laboratories conducting animal research in compliance with Good Laboratory Practices (Title 21 USC, Chapter 13,§58), it is the study protocol and the study director that establish procedures and processes that are approved by each Institutional Animal Care and Use Committee to ensure the humane care and use of animals in research, teaching, and testing and to ensure compliance with guidelines and regulations. This approval process establishes the justification of eustress in the environment that do not rise to the threshold of distress under the AWA.
Introduction
Screening for the safety and efficacy of new molecular entities (NMEs) using purpose-bred laboratory animal species prior to drug licensure and release to the public has been mandated by International Conventions and by national (United States) statutes. Despite significant development and refinement in methodologies by the research community itself, contemporary animal research is still a requirement by federal regulations and is not amenable to exclusion.1,2 The welfare of laboratory animals is an obligate concern for researchers. The welfare of animals in research is generally under the purview of United States government oversight since 1966 (Animal Welfare Act [AWA] P.L. 89-544). 3 While Congress mandated the United States Department of Agriculture (USDA) to regulate and control the transportation, sale, and handling of dogs, cats, and certain other animals, the law excluded the governmental control of laboratory rats and mice. The exclusionary clause of the AWA regarding to rodents has been minimized and the standards of care have generally been voluntarily and universally applied to all sentient animals in research laboratories nationwide.
While it is desirable to replace the use of live animals by other reliable and scientifically valid methods, the use of live animals continues to be necessary to protect human and animal health and the environment (European Directive 2919/63/EU, 2010). The use of animals in research is stringently controlled and monitored by government agencies (US Food and Drug Administration [FDA], US Environmental Protection Agency [EPA], US Department of Agriculture ([USDA], National Institutes of Health [NIH], etc.) and professional organizations (American Veterinary Medical Association, Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC), etc.). In the assessment and evaluation of the possible toxic characteristics of an NME, single dose and repeated dose studies are initiated to provide information on the health risks and hazards to health and welfare of the patient population targeted for its use (refer to Redbook 2000, IV.B.1., General Federal guidelines for designing and conducting toxicology studies; FDA, 2007). In its initial steps to establish safety, a component of many of these studies is directed at assessing the dosage regimen to provide information on the mode of action related to the absorption, distribution, metabolism and excretion of the substance in the patient population.
Under the current animal research regulations, food and water should be provided ad libitum (unlimited access) and should meet the species-nutritional requirements for normal growth and reproduction. However, many situations exist whereby unrestricted access to food and regulatory-required experimental endpoints are directly opposed. For example, under the general FDA guidelines for designing and conducting toxicity studies (Redbook 2000, IV.B.1), FDA proposes a listing of parameters to be quantified and states that: Blood samples should be drawn at the end of the fasting time and before feeding. Fasting duration should be appropriate for the species and the analytical tests to be performed.
The following review summarizes some common related misconceptions and difficulties experienced in laboratory animal research for commercial development of NMEs. The term “fasting” is used within to refer to the intentional restriction of food or water intake for any period of time.4,5
Defining “Distress” Using Humane Endpoints and the Animal Welfare Act (“Welfarism”)
The National Research Council’s (US) Committee on Recognition and Alleviation of Distress in Laboratory Animals
6
has established a national policy on the topic and has established the following: Although clinical signs can be used to assess physical well-being, and behavioral studies can provide information about animals’ preferences and cognitive state…the Committee would like to emphasize that no physiologic measures exist to date with which to assess mental well-being directly. Nevertheless, discussions about animal welfare in the laboratory as well as in farm animal communities take into consideration a variety of criteria to assess an animal’s quality of life. It has been proposed that the most important consideration for the assessment of an animal’s welfare is its emotional state (Duncan 2005). Be that as it may, some of these criteria focus on the animals’ ability to experience pleasure and pain.
In 2000, the Committee on Animal Research and Ethics (CARE) of the American Psychological Association (APA) recommended to the Animal and Plant Inspection Service (APHIS) at the USDA to eliminate the term “distress” from the AWA regulations on the following grounds: • The extremely subjective and vague nature of the concept makes it impossible to define and compare among humans, much less across different species. • A global definition of “distress” in terms of animal use in research fails to take account of the objectives of animal use. • Any distress experienced by laboratory animals needs to be evaluated in the context of the experimental question and goals. • There is experimental evidence that animals can experience stress or some pain without being distressed (CARE, APA, 2015).
7
As an alternative to the current pain classification system, the APA has suggested that experimental procedures be classified by their degree of aversiveness, rather than by the distress they may cause. To refer to fasting as “distressful” is scientifically unsound. According to the APA, distress primarily means to “cause discomfort, anxiety, or suffering” and is defined purely by subjective response. This subjectivity makes it impossible to measure or apply it to non-verbal laboratory animals. According to the APA: Distress is likely to be manifested in very different ways not only between species but also among different individuals within a species. It is important to emphasize that distress should not be confused with stress, which can be related more easily to environmental stimuli that can be arranged, at least in nominal order, from mild to severe. Further, stress can be related to measurable biological responses. Life is full of stressors, and its function in behavior is complex. Most stressors have no long-term negative effects, and many of which serve important functions in normal homeostasis or survival. In rats, for example, pup separation from a dam is stressful to the pups (and the mother), but it is a normal part of the developmental process. Moreover, studies have shown that early mild stress reduces later stress vulnerability.
Subjective Effects of Fasting in Humans
Li et al
8
used the National Institutes of Health standard “Profile of Mood States” and found a fasting-induced
Yang et al 9 conducted a 10-day complete fasting study in healthy adults and reported that the prolonged fast induced a “U-shaped” and an “inverted U-shaped” curve in subjective sensation scores over the 10-day period. Yang et al 9 proposed that fasting typically results in a decrease in serum glucose, and in response to the loss of circulating glucose concentrations the brain compensates for the hypoglycemic state induced by fasting through the stronger recruitment of brain areas related to the given cognitive tasks of the study. This is consistent with the “selfish brain” theory10,11 which proposes that although serum glucose decreases during fasting, brain cognitive function can be maintained as the ketone body level increases to meet its metabolic needs. Some studies have revealed that cognitive performance, such as mental flexibility, does not change or improves after fasting 12 ; other studies have also shown no deteriorations in cognitive performance tests such as vigilance, choice reaction time, and grammatical reasoning.13-15
Yang et al 9 concluded that their 10-day fasting study data were consistent with previous studies conducted by Zhang et al 14 and van de Rest et al 15 showing that mood was susceptible to metabolic influences and the physiological indicators of energy metabolism shifts from glucose to ketone metabolism. Lieberman et al 12 have reported that a 48-hour near-total calorie-restricted diet in humans did not affect mood states and in an 18-hour fasting study there were no increases in major alterations in mood. 16 Additionally, there were no negative mood changes reported in a 2-day consecutive fasting study by Appleton and Baker, 17 and Michelsen et al 18 reported no significant mood changes induced by a 1-week modified fasting study.
Generalizing the Subjective Effects in Humans to Laboratory Animals
When assessing the subjective effects of an operational process or treatment to an animal, government regulators require the establishment of humane endpoints. If the condition is painful or distressful in humans it must be assumed that it is painful or distressful in less sentient animals. 19 According to Wolfle, 16 stress is complex and often idiosyncratic—not all agree on a single definition of stress. Animal care personnel need a clear recommendation regarding the many ramifications of stress as they are not normally experts in this complex subject. It is the Study Director and the IACUC that are expected by regulatory agencies to understand and recognize distress to ensure the well-being of the animals in their care. The goal in nonclinical safety assessment is not to eliminate all stress in laboratory purpose-bred animals being used in the analysis, and it is not a determination if standard fasting protocols meet the definition of “suffering.” As care-givers we must rather provide these animals with a suitable opportunity to adapt to new situations to avoid reaching the threshold of an unnecessary or excessive challenge to homeostasis called “distress.” Arnason and Clausen 20 have concluded that we are not interested in a balance between benefits and harm, but in imbalance, where the benefits outweigh the harms. There is a more significant problem with the metaphor of balancing or weighing, namely that the things to be weighed are not commensurable. The critical issue for the study director and the IACUC is not if fasting implies “suffering”…We are considering things that are so fundamentally different, that they cannot be weighed against one another, at least not as aggregates.
The challenges induced by multiple factors in a natural (free) and laboratory environment may also be viewed as positive sources of stress or eustress, since they present complex problems, that when solved successfully elicit a greater degree of behavioral plasticity and adaptability in animals. Structurally diverse landscapes require animals to display complex behaviors and exhibit adaptive capabilities, like building or finding shelter or finding a reliable water and food source, which ultimately lead to positive emotional states. Thus, maintaining or enhancing the diversity occurring in natural systems or laboratory conditions represent a management approach that can be used to improve welfare and prepare the animal for an efficient adaptation to future, and potentially unknown, environmental challenges. Within the contemporary CRO, fasting or calorie restriction of both short and long duration is not inherently negative. Fasting is approved by the Institute for Laboratory Animal Research21,22 and the NIMH.
23
In 2020, Bienerto-Vasku et al
24
may have summarized the current controversy the best: The adaptation reaction of an organism under stress is not intrinsically good or bad, and its effect on health or performance depends on a plethora of other interactions of the body with the environment as well as on the history of such interactions. The vagueness of the terms “eustress/distress” has historically led to vast differences in the perception and application of the terms across disciplines. While psychology or sociology perceive eustress as something inextricably linked to positive perception and enhanced cognition, biomedicine perceives eustress as generally associated with better survival, health, or increased longevity, no matter how the event is perceived.
Statement of the Issue on Animal Fasting
All species and strains of species do not respond to restricted calories alike. 25 Animals in a true free-access condition allow for self-regulated food intakes. With unlimited access to food, some animals will tend to overeat and as a consequence these animals are more sedentary, obese, and glucose intolerant compared to controlled diet conditions. 26 For these reasons, the suitability of restricted feeding to all laboratory-bred animals used in research has been called into question. 27 It is currently standard practice in University, Industry, and commercial research laboratories that conduct multispecies preclinical research for regulatory agency submission to feed non-human primates (NHP)28,29 and canines30,31 in a single daily meal feeding. This limited daily feeding provides the NHP or canine a fixed amount of food based on the body weights. The subjects may consume the meal quickly in which case they would remain without food for the rest of the day, a dietary condition often referred to as Intermittent Fasting or Limited Fasting. In these situations, these research species are standardly food restricted over the course of 24 hours. 32 With these facts in mind, the forthcoming discussion is focused on laboratory-bred research rodents: rats and mice, only.
As stated above, regulatory agencies require, hematologic analysis, hemostasis evaluation, clinical chemistry testing, and urinalysis as a minimal data base of clinical pathology testing used to evaluate therapeutic agents (new chemical entities, test articles, and biologics), pesticides, and industrial chemicals in nonclinical safety assessment studies. Results of clinical pathology testing are communicated to regulatory authorities and generally form the basis for development of a monitoring strategy for molecules in development. These factors that affect the optimal design of the clinical pathology component of protocols to optimize the opportunity to achieve study objectives. Additional tests may be included in study protocols to help characterize the mechanism of test article action, to assist in prioritization of candidate molecules, or to address requests by regulatory authorities; these ancillary tests include acute phase proteins, hormones, and exploratory and established biomarkers of renal, hepatic, cardiac, and vascular injury.33,34
In cases of protocol-driven clinical pathology blood collections that are required by both the European Union and United States Good Laboratory Practice Guidelines for nonclinical research. The same standardized protocols under the jurisdiction of the Norwegian Animal Research Authority have been put under extreme limits on the sudden fasting (acute food deprivation) of laboratory rodents that are accustomed to the ad libitum access to food. 5 These same restrictions are infiltrating other research laboratories. Under these regulations, rats are considered to be extremely intelligent animals, and they do notice the rapid removal of all food from its cage during these standard research driven functions.
The position of those who do not approve of fasting is that the sudden removal of the predictability of food, particularly when it affects factors related to animals’ basal needs, can be expected to be particularly distressing, and it has been demonstrated that unforeseen environmental changes can lead to an increase in cortisone release in a number of vertebrates.
35
This acute food removal procedure is posited to induce psychological stress. The degree of stress, in relation to fasting will depend upon the species (how often they eat, when they eat during the 24-hour period and whether periods of fasting are a normal part of the animal’s life history. The interpretation of legal phrases in the AWA such as
The current PREPARE Guidelines of the Royal Society for the Prevention of Cruelty to Animals (RSPCA; https://norecopa.no/prepare/)
37
prioritizes the need for sufficient attention that must be made to the animals’ instincts and needs from birth to death. Since most animals are more active in one phase of the 24-hour period, environmental influences to homeostasis may easily be overlooked,
36
such as acute food restrictions. Tannenbaum and Bennett
38
have argued that
In such circumstances, fear must become an acutely unpleasant state which, by human analog may be termed anxiety” (p 22, italics in original). Russell and Burch subsequently add to the list of kinds of
For Tennenbaum and Bennett, 38 it was not the use of animals in research that Russell and Burch found problematic, but the infliction on research animals of unnecessary or avoidable pain, fear, stress, anxiety, bodily discomfort, and other significantly unpleasant feelings. Under the present discussion, Tennenbaum and Bennett 38 are equating the subjective experience of the rat in just observing food removal and a period of food withholding, regardless of duration or weight loss, as distress and therefore is inhumane (refer to Juruena 39 ). According to Landys, Ramenofsky, and Wingfield, 40 these intervening episodes of distress in a laboratory animal may play specific and distinct roles in the regulation of life processes, and thus, proponents of animal research may argue that food withdrawals for standard blood tests in laboratory rats may participate in the promotion of different physiological states that by their nature should be considered inhumane. Claassen 41 has provided the reader to over 100 other references highlighting the influence of fasting in laboratory animals.
Monso, Benz-Schwarzburg, and Bremhorst 42 have stated the position that “animal welfare” has many different meanings that stem from debates in axiology, political philosophy, animal ethics, and contemporary animal welfare science. The current position regarding fasting is based on a position of “experiential welfare” or “subjective quality of life” in a hedonistic sense of these terms. 43 This view of animal welfare centers on the idea that hedonistic welfare, or hedonistic quality of life, is the only variable that matters when it comes to “well-being.” Welfare activists like Rollin 44 (2004, p. 16), Ryder 45 (1999, p. 36), and Duncan 46 (2004, p. 88) consider that animals are harmed by humans only in those cases in which our treatment of them generates pain or suffering, or removes opportunities for pleasure or satisfying experiences (ie, eating).
This means that increases in well-being are understood to correlate with an improvement in the hedonistic (pleasure/pain) aspects of an individual’s life related to the ad libitum access to food in an animal’s cage, and conversely, decreases in well-being are understood to correlate with a deterioration in these aspects when an animal observes the removal of food from the cage for acute, short-term fasting during a study. In this case the intrinsic negative hedonic state is psychological suffering, anxiety, inducted by the observation of food removal. It has been proposed that animals will notice changes in the supply of food rapidly, and this may lead to psychological stress. The interpretation of such legal phrases in the AWA such as
These critical concerns in conducting animal research may not apply to university-based research that may be considered “efficacy” studies or may not be conducted under the FDA regulations for NDA approvals, or are not usually conducted under GLPs. However, for the pharmaceutical industry and support businesses the issue of fasting is particularly problematic, especially for: (1) The Contract Research Organization (CRO) that must balance the conservative expectations of European sponsors contracting their nonclinical program here in the United States, or (2) The CRO that must balance the expectations of US Sponsors that are subsidiary companies of European or Japanese pharmaceutical companies (eg, Merck, Bayer, and Eisai), and/or (3) The CRO that may have both European and US locations that attempts to unify procedures and policies for all of its locations.
Stress Is Not Inherently Negative
In understanding the issues surrounding fasting, it must be made clear that the link between cortisol and negative affect is not as straightforward as presumed. 47 In humans, the process of just waking up in the morning produces a rise in cortisol48-51 and the voluntary expression of physical exercise, such as at a gym or a bout of local biking52,53 will also raise cortisol. Additionally, many experiences of the human patient that is associated with strong negative affect do not cause increases in cortisol. Alternatively, it is not surprising that manipulation of cortisol in healthy participants often does not alter affect.54-56 Changes in feeding conditions and cortisol concentrations have been reported.57-59 So if measured serum cortisol or administration of corticosterone to healthy human subjects do not predict the expression of negative emotions (affect), then why do we presume measured cortisol changes in rats have any foundation to conclude that a rat is in “distress” or use these data to establish a national animal welfare policy?
The Yerkes-Dodson Law is one of the “first principles” of modern psychology. First proposed in 1908 by Robert Yerkes and John Dodson to describe the relationship between shock intensity and the general locomotor response in Japanese Dancing mice
21
; it has been used to describe the functional relationship between performance and the intensity of perceived stress.
60
There is an empirical relationship between stress and performance and there is an optimal level of stress corresponding to an optimal level of performance. Stress is not inherently bad. All animals have evolved mechanisms to cope with the stresses of their lives such as the presence and absence of a stable food supply. According to modern psychology textbooks, stress has no defined etiology or prognosis. Moberg
61
has generally defined stress as “the biological response elicited when an individual perceives a threat to its homeostasis,” in which the threat is the “stressor.” The term “distress” helps to differentiate between a non-threatening stress response (often referred to as “good stress” or eustress) and a biological state where the stress response has a deleterious effect on the individual animal’s welfare (or “bad stress”). Fasting is not necessarily deleterious and in some cases can be beneficial (see below). Figure 1 shows the standard “inverted U-shaped functional relationship” describing the Yerkes-Dodson phenomenon: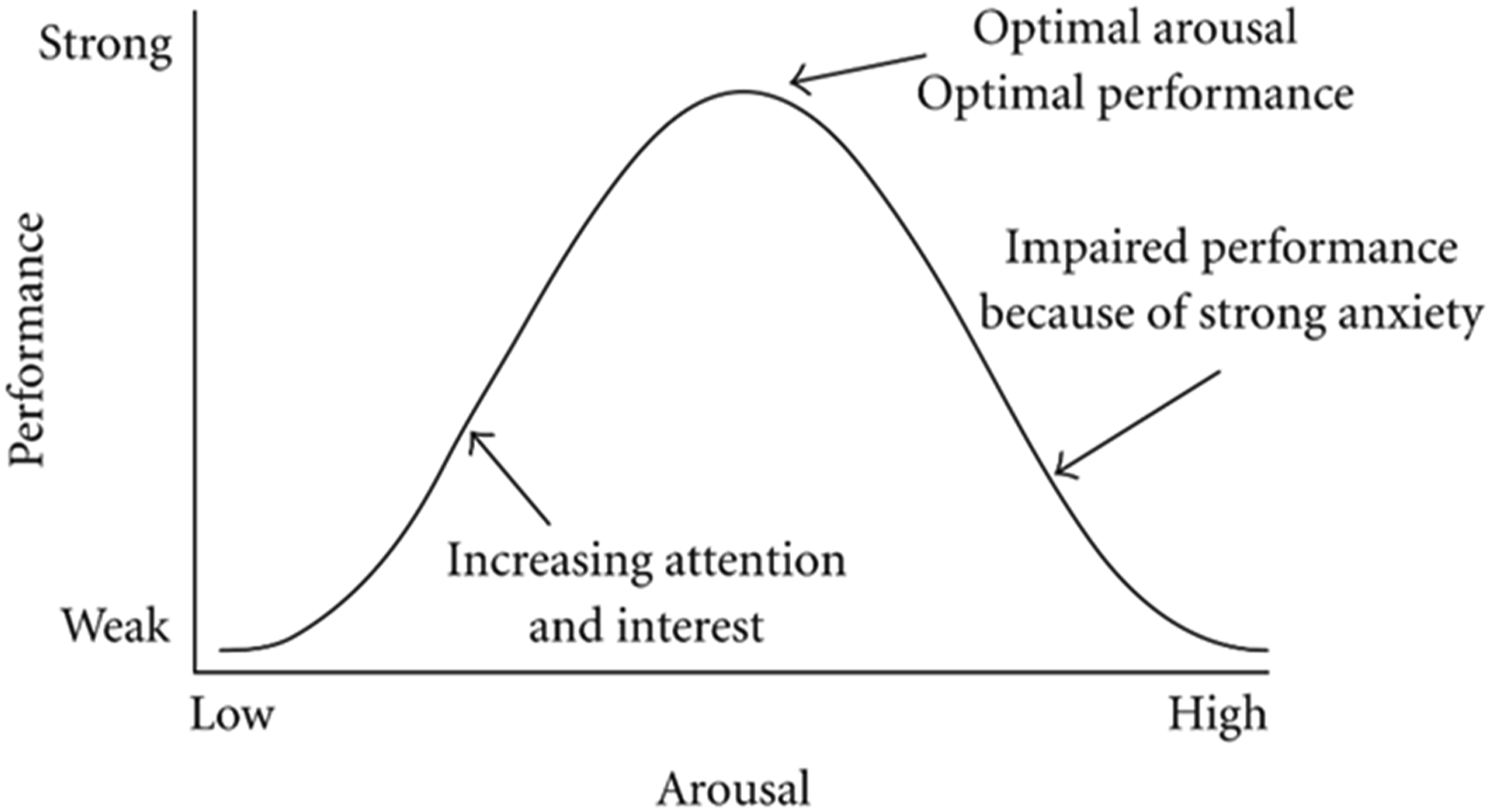
According to Moberg, 61 when we consider all the problems encountered in attempting to measure stress including what to measure, comparable biological responses to good and bad stress, difficulties in monitoring biologic responses and inter-animal variability, 62 it is tempting to conclude that measuring stress is too daunting a problem and make no distinction between stress and distress. However, the aim of nonclinical research to advance the health and welfare of human patients is too important to ignore this problem. Schreck 63 has suggested that any aversive event can be lumped into one category under the term “stress,” but in doing so, the term becomes a potentially dangerous unifying concept. Schreck 63 admonishes that when a label is placed on an event it is generally understood that the label provides further clarification and explanation of matters by using the term. When two or more events of different phenomena are lumped together into one category there is a tendency to overemphasize the similarities and ignore the differences. Because of these dangers, Schreck 63 and Moberg 61 suggest that the term “stress” be abandoned but under the AWA it is not possible to do away with such terminology; instead the focus must remain on the appropriate use of the term.
Suffering from stress may threaten an animal’s welfare, but it is a categorical error to assert that all stressors are inherently bad. An animal’s response to stress is a cascade of biological events, and the nature of the biological defenses used during stress is not a reliable approach to evaluating animal welfare. Contemporary struggles in the current research environment involve the problem of measuring stress and defining distress. Experimental goals are routinely weighed and prioritized against animal welfare. The IACUC’s challenge is to determine when stress becomes distress—the key to making this differentiation is to identify the biological cost of the stressor (eg, periods of limited resources or food scarcity). Short-term food restriction in a standard fasting protocol for clinical pathology, postmortem evaluations, or in-life electrophysiological recordings (eg, an auditory brainstem response) does not represent an impactful life event for animals that have evolved to forage. When a rat endures the presentation of electrical shock in a standard developmental and reproductive toxicology (DART) study, or 12-hour fast and subsequently returns to eat and drink or to socialize with its cage-mates, the cost of stress is not believed to be of consequence to the animal. In the shock presentation example, the catecholamines secreted during the shock presentation result in conversion of glycogen to readily utilizable glucose or other metabolic products required for glucogenesis. Once the stressor is alleviated, the glycogen stores are quickly replenished by glycogenesis to pre-shock levels. Burman et al 64 reported on the biological stress response induced by removal of individual rats from social groups. This is a standard operational practice in all developmental toxicology laboratories to maintain appropriate stocking densities in groups of rapidly growing young laboratory rats. Regulatory restrictions exist on housing conditions based on cage size and body mass (eg, age). Irrespective of stocking density, the rats remaining in the home cage significantly increased agonistic behavior, audible vocalizations, aggressive grooming, bar-chewing, and climbing behaviors following removal of their cage-mates. These behavioral changes were associated with a highly significant post-removal increase in their fecal corticosterone metabolite levels. Burman et al 64 concluded that the removal of individuals from groups of young laboratory rats during normal growth and development in the laboratory resulted in social stress, and thus an apparent impairment of their welfare. Despite this knowledge few laboratory managers would refuse to comply with the regulations of housing management based on the identity of the cage change as a stressor.
In another example, Hunt and Hambly 65 investigated how different housing conditions affected stress levels by measuring both corticosterone levels, using non-invasive fecal collections, and behavioral expressions of stress in mice. Groups of MF1 outbred mice were separated into five different housing conditions at the beginning of the study: (1) individually housed, floor area 490 cm2 per individual, (2) groups of three mice, floor area 163 cm2 per individual, (3) groups of three mice, floor area 320 cm2 per individual, (4) groups of six mice, floor area 160 cm2 per individual, and (5) groups of six mice, floor area 230 cm2 with extra height per individual to allow visual contact. Mice are “social animals”; however, Hunt and Hambly 64 reported that singly housed mice had reduced corticosterone levels over time after separation, and there was no significant difference in corticosterone levels between the different housing densities, with no differences for aggression or stereotypical behavior suggesting that housing conditions may not be a stressor for this strain and sex of mouse. Crockett et al 65 reported that a standard room change and cage changes from a soiled cage to a clean cage induced significant stress in cynomolgus monkeys as quantified by both cortisol secretion and observable behaviors over 24 hours. Most researchers would agree that pain is a stressor—analgesia assessments (eg, hot plate paw lick latencies) are stressful, but are part of standard nonclinical CNS safety assessment protocols (ICH S7A). Rectal temperature collection procedures are stressful64,65 but are included in standard veterinary examinations and research protocols. Catching, restraint, and bodyweight measurements of large animal species (NHPs, canines and swine) are stressful but are core requirements in collection of the requisite endpoints for each nonclinical safety protocol. Identifying stress is not the issue at hand—the Study Director must help the IACUC in determining when a stressor becomes distress.
Norway’s 3R Centre and National Consensus Platform for the Replacement, Reduction and Refinement of animal experiments (Norecopa) 5 has recommended that the food deprivation period should be during the daytime if possible, when rodents normally eat less food. Adequate emptying of the gastrointestinal tract may well be achieved after 6–8 hours’ fasting, enabling the experiment to start during the afternoon or evening of the same day, avoiding the need for overnight fasting. This recommendation may in fact avoid overnight fasting; however, this recommendation sets the stage for dramatic changes in operational staff workloads as well as the deleterious impact of photoperiod phase shifts in research facilities as a result of moving study conduct to late afternoon and into the “dark cycle” of the 12:12 light/dark cycle. 66
In its opposition to overnight fasting, Norecopa 5 recommended that some source of energy (eg, a sugar solution) should be given to the animals wherever possible in the food deprivation period, to satisfy the animals’ energy needs.67,68
In contrast to the Norecopa recommendation, Turner et al 69 examined groups of rats (5 males and 5 females/group) that were (1) fasted overnight; (2) fed standard chow; or (3) fed sucrose for a period of 17 hours prior to scheduled blood sampling, euthanasia and necropsy. Turner et al concluded that there was little cause for concern in fasting animals overnight prior to necropsy. The reference intervals for hematological and clinical chemistry parameters obtained from chow-fed and fasted rats overlap considerably, in which Turner et al interpreted as indicating that both fed and fasted animals had clinical findings consistent with normal physiology. Turner et al 69 further proposed that the similarity between fed and fasted groups may be due in part to marked residual digesta in the gastrointestinal tracts of ad libitum-fed rats after an overnight fast. 69 Turner et al concluded that overnight fasting of rats was not accompanied by alterations in plasma epinephrine or corticosterone concentrations, suggesting that any stress induced by fasting of rodents for 17 hours was minimal, which supported the previous conclusions of De Boer et al. 36 In addition to these conclusions regarding comparisons between fed and fasted animals in the Turner et al study, they also reported that the glucose-fed group experienced marked hepatic alterations after overnight feeding of sucrose to rats compared to those seen after feeding chow or fasting. The conclusion was just the opposite of the Norecopa recommendations; according to Turner et al based on the alterations in hepatic structure and function, the practice of feeding sucrose to rats prior to necropsy for toxicology studies or any studies examining hepatic function is not recommended.
Fasting Is Not Inherently Negative
Emerging findings from animal and human studies suggest that intermittent food restriction periods of as little as 16 h can improve health indicators and counteract disease processes. 70 For example, Wan et al 70 at the National Institutes of Health (NIH) reported a study in which 5-month-old male rats had either ad libitum access to food or were deprived of food every other day (intermittent food deprivation) for 6 months, during which time their heart rate, blood pressure, physical activity, and body temperature were measured by radiotelemetry under nonstress and stress (immobilization or cold-water swim) conditions. Wan et al 70 concluded that intermittent fasting improved both cardiovascular risk factors and cardiovascular/neuroendocrine stress adaptations in rats. Duffy et al71-73 also examined the effects of dietary restriction (10, 25, and 40%) on non-neoplastic diseases in rats (age 58 and 110 weeks), to determine whether low-level fasting could increase the survival rate and decrease variability in chronic bioassay studies. At 110 weeks of age, the severity of chronic heart and kidney diseases was significantly reduced in all food restricted rat groups, with significant fasting-dependent linear trends for these diseases. Moreover, fasting prevented the progression of skin irritation to foot ulcers, and reduced the age-related degeneration in the adrenal, lacrimal, and thymus glands, and the liver. Snorek et al 74 have demonstrated that 3-day fasting effectively protected rat hearts against two major endpoints of acute ischemic reperfusion injury (myocardial infarction and reperfusion ventricular arrhythmias). These data are representative of the literature clearly indicating that fasting is not inherently damaging or stressful to laboratory animals and that even short durations of fasting are effective in preventing or slowing the progression of non-neoplastic diseases. In a comprehensive review of the nonclinical safety assessment studies, de Cabo and Mattson 75 observed a consistent pattern of data showing a robust disease-modifying efficacy of intermittent fasting in animal models on a wide range of chronic disorders, including obesity, diabetes, cardiovascular disease, cancers, and neurodegenerative brain diseases.75-79
A working group of the National Centre for the Replacement, Refinement and Reduction of Animals in Research (NC3Rs) 3 has suggested that reliable and stable behavioral performance is crucial for the success of behavioral neuroscience experiments with animals. The FDA requires some animal research using standard operant conditioning paradigms to investigate learning and memory dynamics (behavioral toxicology) and for all new CNS-active compounds for their abuse potential. According to the working group, there may be many factors early in training that may lead to errors in the experimental task, such as lack of attention or misdirected attention, and too much or too little motivation. After training, when animals perform the experimental task reliably and consistently, errors in performance can reveal valuable information about exactly how the animal performs the task and which brain structures are functionally related to the task requirements. Taffe 80 concluded that consistent behavioral performance of monkeys on a range of cognitive tasks were detrimentally affected by short-term, ad libitum chow feeding, even when the food reinforcer is highly preferred or the tasks are well trained. Furthermore, Taffe 80 has shown that maintenance of weekly chow intake in the range of 70–85% of National Research Council (NRC) recommendations for metabolizable energy is necessary for consistent behavioral responding. The subjective experience of stress cannot be identified externally. The interoceptive experience of each animal in a contract research organization is sculpted and modified by their current and past behavioral, experimental, and drug histories.
Of the available animal models, the rhesus monkey (Macaca mulatta) is most useful in investigating human aging, as they share about 93% sequence identity with the human genome. 74 For this reason, non-human primates are considered important models for translational research findings.81,82 In non-human primates, the starting point of practicing caloric restriction is a key factor in determining the beneficial effect of aging. Calorie restriction has been shown to extend lifespan and reduce the incidence of age-related disease in short-lived laboratory animal species, 82 and the same may be true for macaques.53,80,83,84 Studies have shown that caloric restriction may reduce the risk for age-related morbidity by more than 2-fold, and like rodents, the incidence of cancer in monkeys is lower with caloric restriction.85,83 Given the obvious link between rhesus monkeys and humans, the beneficial effects of caloric restriction may also be observed in humans.
It has been suggested that the positive mechanisms of fasting involve a metabolic shift to fat metabolism and ketone production. Contrary to the notion that fasting is distressful, de Cabo and Mattson 74 suggest that the stimulation of adaptive cellular oxidative stress responses appear to prevent and repair molecular damage. Periodic flipping of the metabolic switch not only provides the ketones that are necessary to fuel cells during the fasting period but also elicits highly orchestrated systemic and cellular responses that carry over into the fed state to bolster mental and physical performance, as well as disease resistance.86,87 There is now strong and convincing nonclinical evidence that alternate-day fasting can delay the onset and progression of the disease processes in animal models of Alzheimer’s disease and Parkinson’s disease.86-88 Animal studies have shown that the amount and frequency of food bouts can have a major impact on brain health span and vulnerability to Alzheimer’s, Parkinson’s, and stroke. Findings from these studies show that feeding and exercise regimens that result in intermittent metabolic switching from liver-derived glucose to fat-derived ketones facilitates brain neuroplasticity and resilience.86 It is proposed that brains and the bodies they control have evolved to function optimally in the setting of intermittent food deprivation.
In a recent literature review Xu et al 89 concluded that there are overwhelming data that suggest that several types of calorie restrictions and intermittent fasts play a promising role in promoting post-traumatic brain injury recovery. This recovery is believed to be achieved by alleviating mitochondrial dysfunction, promoting hippocampal neurogenesis, inhibiting glial cell responses, shaping neural cell plasticity, as well as targeting apoptosis and autophagy.
General regulatory guidelines90,91 for designing and conducting drug safety assessment studies in animals recommend the number of animals and time intervals for evaluation of such physiological markers as clinical chemistries. Under ideal conditions, the same animals should be sampled at each scheduled collection time point on a given study and their blood “should be drawn at the end of the fasting time before feeding” (see Chapter IV.C.3-5. of “Redbook 2000,” 89 for example). Because food can either increase or decrease bioavailability, Sponsors must address the utility of food–drug interactions in human subjects early in drug development to provide the most rational dosing strategies for clinical trials to be used as a basis for FDA-approval. 90 In spite of this critical factor in safety assessment, there seems to be a contemporary movement to presume that fasting in nonclinical safety assessment laboratories is distressful to animals.
Endogenous Feeding Patterns and Fasting
There is a circadian rhythm to many biological functions including general activity, sleep, and food consumption. With respect to fasting in nonclinical safety assessment studies conducted with laboratory-bred animals, the duration of fasting is often linked to environmental controls. The vast majority of pre-study function fasting periods in a laboratory environment are set to correspond to the 12-hour light:dark cycle established as a standard environmental control.
Bruguerolle 92 proposed that many of the biological factors demonstrating a circadian rhythm are involved in drug kinetics, such as gastric emptying, gastric pH, gastrointestinal motility, cutaneous permeability, posture, blood flow, tissue perfusion, plasma protein binding, metabolizing enzymes activity, renal perfusion, glomerular filtration, and urinary pH. Though the oral route is the most common route of administration, the circadian fluctuations in systemic blood drug concentrations are not limited to the relationship between food consumption and oral administration of drug. Skin penetration of a eutectic mixture of lidocaine and prilocaine has been reported to be dependent on the time of administration; lidocaine plasma levels in children were higher after evening administration as opposed to morning.92-94
It seems intuitive that food content in the stomach will affect the absorption and distribution of an orally-administered drug.
95
Brueguerolle et al
92
reported an interaction between the time-of-day of dosing and feeding status in rats. Circadian variations in carbamazepine kinetics were examined following oral administration in adult male Wistar rats. 100 mg/kg carbamazepine was administered by oral gavage. Figure 2 shows the Cmax:Tmax ratios of carbamazepine concentrations quantified at five different times in the day for both fasting and nonfasting conditions. A circadian time-dependent variation in drug absorption was demonstrated in nonfasting (free feed) and fasting rats. However, at 2200 hrs, the Cmax:Tmax ratios were not determined by stomach contents alone. The Brueguerolle et al
92
data demonstrate that stomach content is not the only factor involved in the circadian change in drug absorption. Selection of the most therapeutic time-of-day in which the drug should be taken and for a more realistic view of chronopharmacology of carbamazepine the optimal time of dosing may be 2200 hrs under standard lighting conditions. Quantitative changes in carbamazepine plasma concentrations varied over the 24-hour day that was also modified by the fasting status of rats. These data point out the necessity to control and take into account multiple factors that interact in the normal response to a dose administration. Redrawn and modified from Bruguerolle.93,94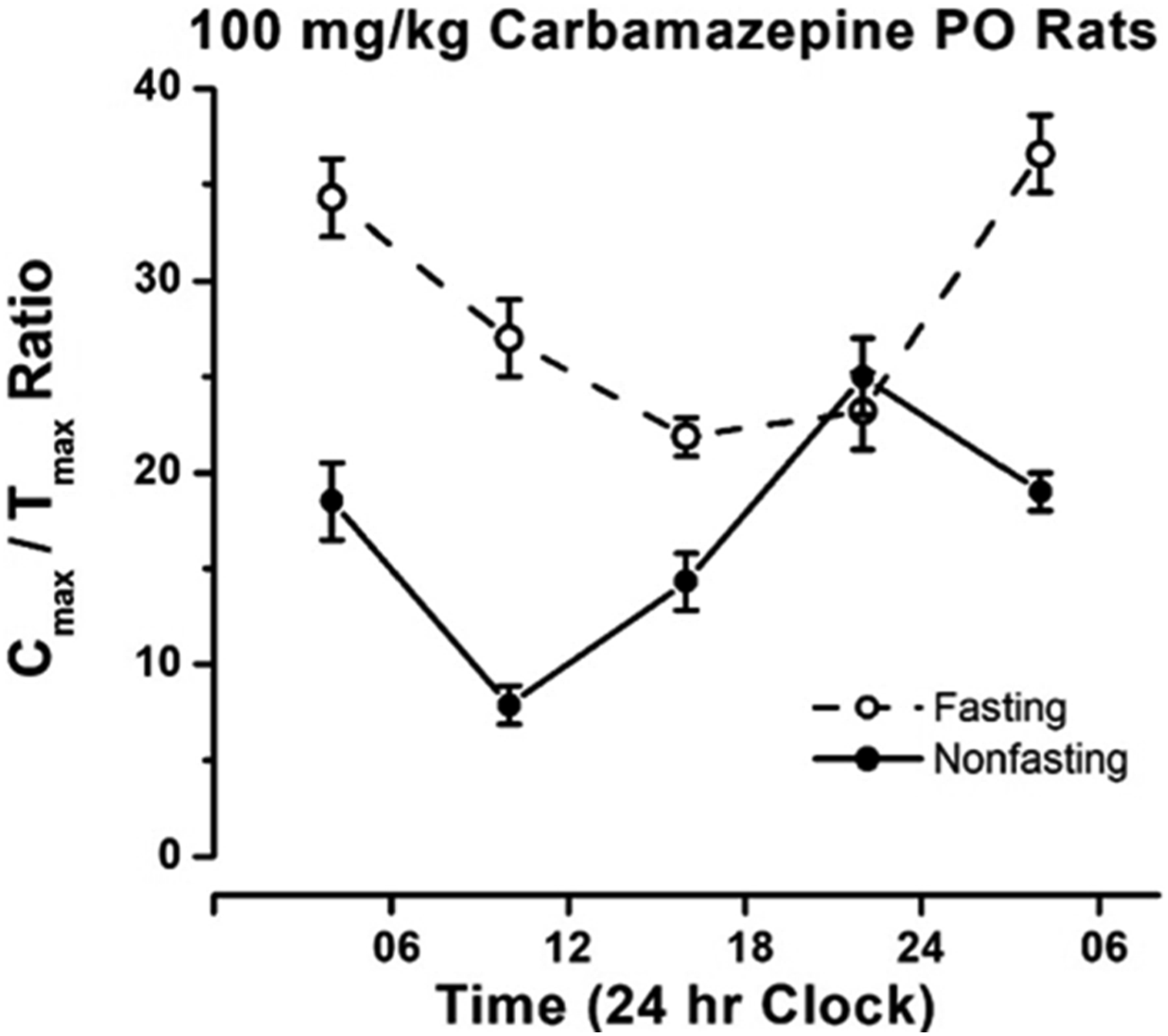
Changes in Kinetics of 4 Drugs From the Same Class of Therapeutics (Chemotherapy) Related to Food Access.

aNew Drug Application Number, FDA’s @drugs website.
For both operational management and those biological factors involved in fasting within the GLP guidelines, most fasting events occur over the 12 hours of “lights out” period in the vivarium. In a standard GLP-laboratory this 12-hour standard paradigm is not due to convenience of staff working hours. The 12 hours of food removal in a standard short-term fast can be documented with lights on (prior to 1800 hr) and prevents the need to turn lights on during the dark cycle just to ensure food is removed and documented in fasting time frames less than 12 hours.
Fasting and Clinical Pathology
Laboratory and diagnostic tests are tools used to gain information on the health status of the animals on study. A number of study-related functions and pre-analytical factors including anesthesia, stress, fasting (food intakes), fluid intakes, and light:dark illumination cycles are known to affect the electrolyte and related hormone measurement determinations in animals. 93 Using laboratory blood sample collections in drug safety evaluations as the basis for not allowing fasting has some merit in that fasting effects are known to engender decreases in alkaline phosphatase, changes in urea, glucose, creatine, and plasma proteins with hemoconcentrations. 94 However, these parameters are known to vary in both fasted and non-fasted conditions. By themselves, they should not be therapeutic determinants of the relevance, importance, reliability, or validity of fasting during nonclinical research safety assessment studies conducted for new drug approval. 92
As an integral part of the clinical pathologist’s evaluation of the general health status of the study population, the influence of fasting on specific protocol-driven test parameters in the animals should be interpreted based on the background, education, and experience of the clinical pathologist. Reviews of these tests are routinely conducted by Sponsors, and such interpretive value of these findings is not at the center of this controversy. Clinical chemistry data supports the conclusion that the standard fasting of 12- to 16-hour duration is a benign or “good” stress (eustress) that is brief in duration and is amenable to correction by normal homeostatic mechanisms.33,34
Under current regulatory guidelines that define stress and distress anthropomorphically, based on the fact the humans often report enhanced mood and positive subjective experiences with fasting, then animals subjected to intermittent fasting in research laboratories worldwide cannot be presumed to exist in a negative emotional state. What is done in nonclinical research laboratories around the globe is to administer novel compounds at novel doses to living organisms to test for safety and efficacy. Such testing is initiated by acute single dose administrations using dose escalation procedures to be able to identify early in the protocols when systemic toxicity is developing. When all is said and done fasting is transient and may be far less injurious than the test article being administered. The standard fasts of 12- to 16-hour duration in purpose-bred laboratory animals may provide a statistically significant change a parameter or two but the physiological meaning of such findings are generally minimal and more importantly are recoverable within a day.
De Cabo and Mattson 75 has defined a standard laboratory procedure referred to as “intermittent fasting” as an eating pattern that includes hours or days of no or minimal food consumption without deprivation of essential nutrients. These commonly employed regimens in GLP compliant studies include (1) alternate-day fasting, (2) 5:2 intermittent fasting (fasting two days each week), and (3) daily time-restricted feeding (such as eating only during a 6-hour window). De Cabo and Mattson 75 have concluded that hundreds of animal studies and scores of human clinical trials have shown that intermittent fasting can lead to improvements in health conditions or disorders in both animals and humans. In the final analysis, the prohibition of fasting laboratory animals does not seem to be based on the current scientific evidence.
Footnotes
Author Contributions
Gauvin, D. V. contributed to conception and design and contributed to acquisition, analysis, and interpretation; McComb, M. contributed to conception and design and contributed to interpretation; Farero, R. contributed to conception and design and contributed to interpretation. All authors drafted the manuscript, critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Work for hire. Publication is part of yearly evaluated performance requirements. Funding to all 3 from Charles River Laboratories, Inc.
