Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of 10
Keywords
Introduction

Definitions and Functions of the Ingredients in this Safety Assessment. 1

Because often in the published literature the information provided is not sufficient to determine how well the tested substance represents the cosmetic ingredient, the taxonomic name is used unless it is clear that the test substance is similar to a cosmetic ingredient. However, in the case of data on the extract of
Botanicals, such as
This safety assessment includes relevant published and unpublished data for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the CIR website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Chemistry
Definition and Plant Identification
The definitions and functions of the
Physical Properties
Methods of Manufacturing
Gingko Biloba Leaf Extract
A general description of manufacturing for “medicinal” GBE reported that the leaves of the
GBEs may be full extracts or standardized extracts.
4
Full extracts are prepared with alcohol and contain all constituents soluble in alcohol. Standardized extracts (one of which is referred to as EGb 761® in published literature) are more common, especially in herbal supplements, and are prepared in manufacturer-dependent multi-step processes (Figure 1). These processes may include additional steps in which some compounds, such as flavonoids and lactones, are enriched while others, such as ginkgolic acids, are removed. General manufacturing process of a standardized 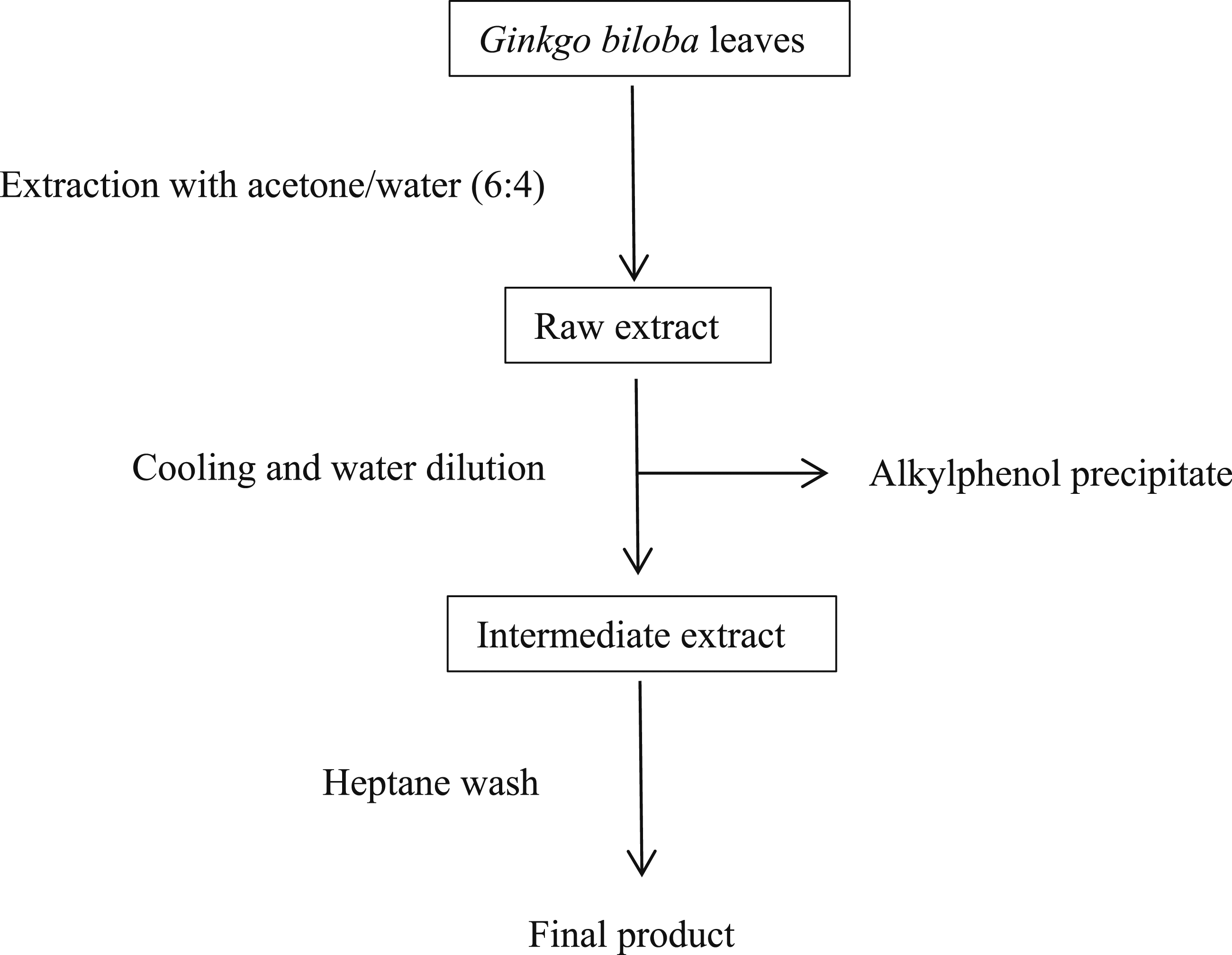
A manufacturer has reported that one Ginkgo Biloba Leaf Extract product is produced through extraction with an ethanol-water solution, while another product is produced through extraction with an ethanol-water solution before being evaporated and resolved in 50% butylene glycol. 6
Ginkgo Biloba Meristem Cell
Ginkgo Biloba Meristem Cell is produced by sterilizing cambium-containing tissue from the
Composition/Impurities
Ginkgo Biloba Leaf Extract
Major Constituents of GBEs (%). †
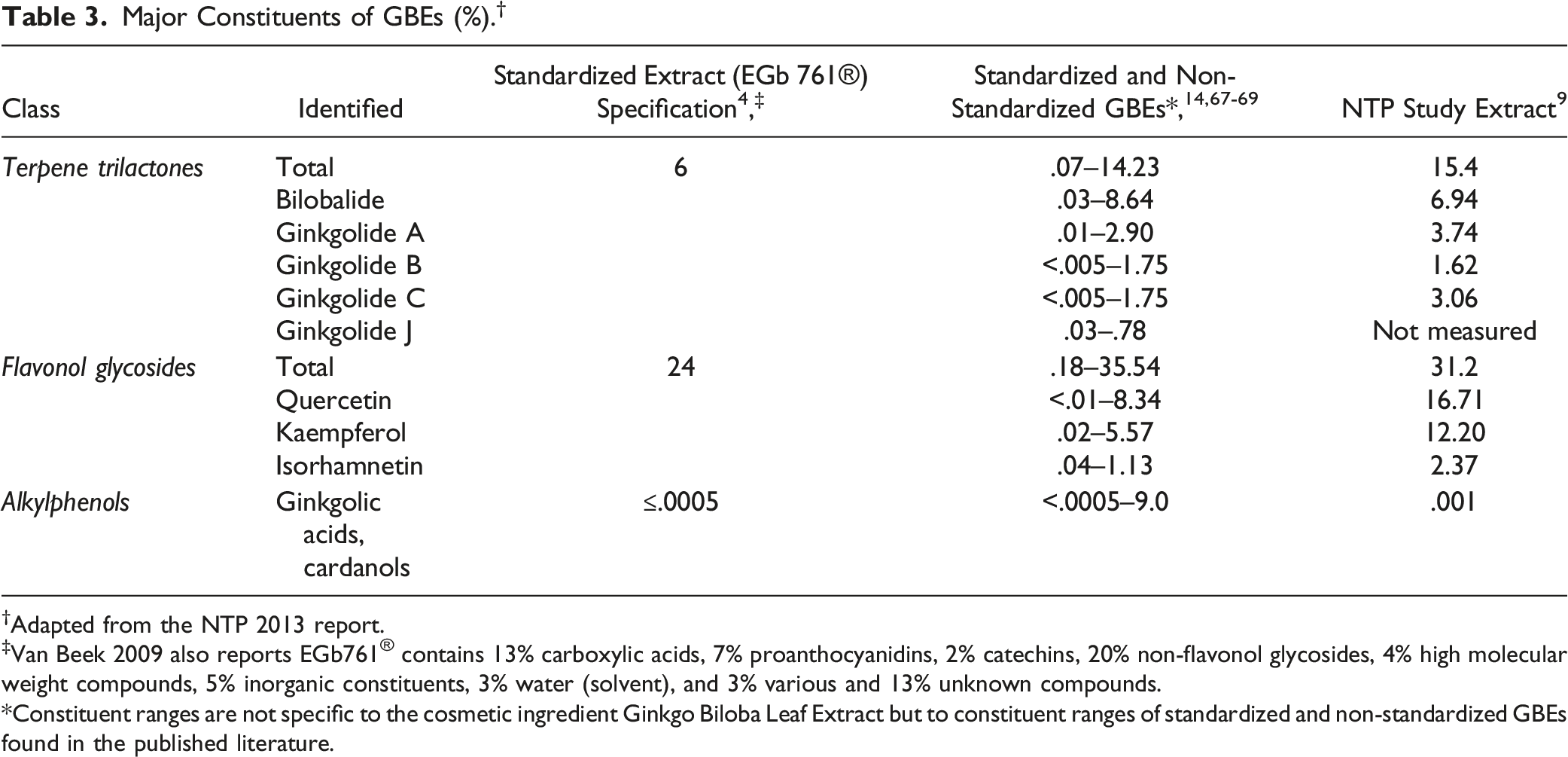
†Adapted from the NTP 2013 report.
‡Van Beek 2009 also reports EGb761® contains 13% carboxylic acids, 7% proanthocyanidins, 2% catechins, 20% non-flavonol glycosides, 4% high molecular weight compounds, 5% inorganic constituents, 3% water (solvent), and 3% various and 13% unknown compounds.
*Constituent ranges are not specific to the cosmetic ingredient Ginkgo Biloba Leaf Extract but to constituent ranges of standardized and non-standardized GBEs found in the published literature.
The target levels of the major constituents of the standardized GBE EGb 761® are reported to be: not less than 6% total terpene trilactone content, not less than 24% total flavonol glycosides, and not more than 5 ppm (.0005%) ginkgolic acids. 4 This extract is reported to be a brown powder with characteristic smell containing not more than 20 ppm heavy metals and not more than 2 ppm arsenic. The standardized extract used in National Toxicology Program (NTP) studies is reported to contain 15.4% terpene trilactones, 31.2% flavonol glycosides, and 10.45 ppm (.001%) ginkgolic acids. 9
According to an analysis of crude extracts of
General
The
The
An extraction with 60% w/w ethanol of dried green
For use as an herbal medicine in Germany, GBE must be extracted with acetone/water and contain 22%–27% flavone glycosides (quercetin and kaempferol) with a molar mass of 756.7 (quercetin glycoside) and 740.7 (kaempferol glycoside); 5%–7% terpene lactones of which 2.8%–3.4% consists of ginkgolides A, B, and C and 2.6%–3.2% bilobalide; and less than 5 ppm (.0005%) ginkgolic acids. 14
Ginkgolic acid is a salicylic acid derivative with a C15 side chain that is related to the pentadecylcatechols (i.e., urushiol) found in poison ivy.
15
One analysis found crude aqueous extracts of
A cosmetic ingredient supplier reported that a Ginkgo Biloba Leaf Extract produced with ethanol/water and sold in a tradename mixture with butylene glycol contains .51% flavonol glycosides, .16% terpene lactones (.08% bilobalide, .04% ginkgolide A, .02% ginkgolide B, and .02% ginkgolide C), .21% quercetin, and less than .1 ppm ginkgolic acid. 6
A certificate of analysis from a cosmetic ingredient supplier on a Ginkgo Biloba Leaf Extract (solvent not specified) described the sample as a light tan powder that contained 25.3% ginkgo flavonol, 6.4% ginkgolides (bilobalide, ginkgolide A, ginkgolide B, ginkgolide C), 2.3 ppm ginkgolic acid, 100 ppm free quercetin, 200 ppm free kaempferol, 200 ppm free isorhamnetin, and less than 20 ppm heavy metals. 8
A cosmetic ingredient supplier for a tradename mixture of Ginkgo Biloba Leaf Extract in an alcohol base reported that heavy metals were below reporting limits and no residual pesticides were detected. 16 This supplier also reported the 26 allergens defined by the seventh amendment to the EU Cosmetic Directive were below testing thresholds.
Ginkgo Biloba Meristem Cell
A supplier has reported that Ginkgo Biloba Meristem Cell is distinctly different from general GBEs, with major constituents being catechin, gallocatechin, epigallocatechin, and bilobalide.17,18
UV Absorption
Ginkgo Biloba Leaf Extract
In a spectral analysis provided by a supplier of a Ginkgo Biloba Leaf Extract (ethanol: water:butylene glycol extract), no maximum UV absorption peaks were observed in the 280 to 450 nm range. 19
Use
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetics industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
Frequency (2018) and Concentration of use (2014) According to Duration and Type of Exposure for
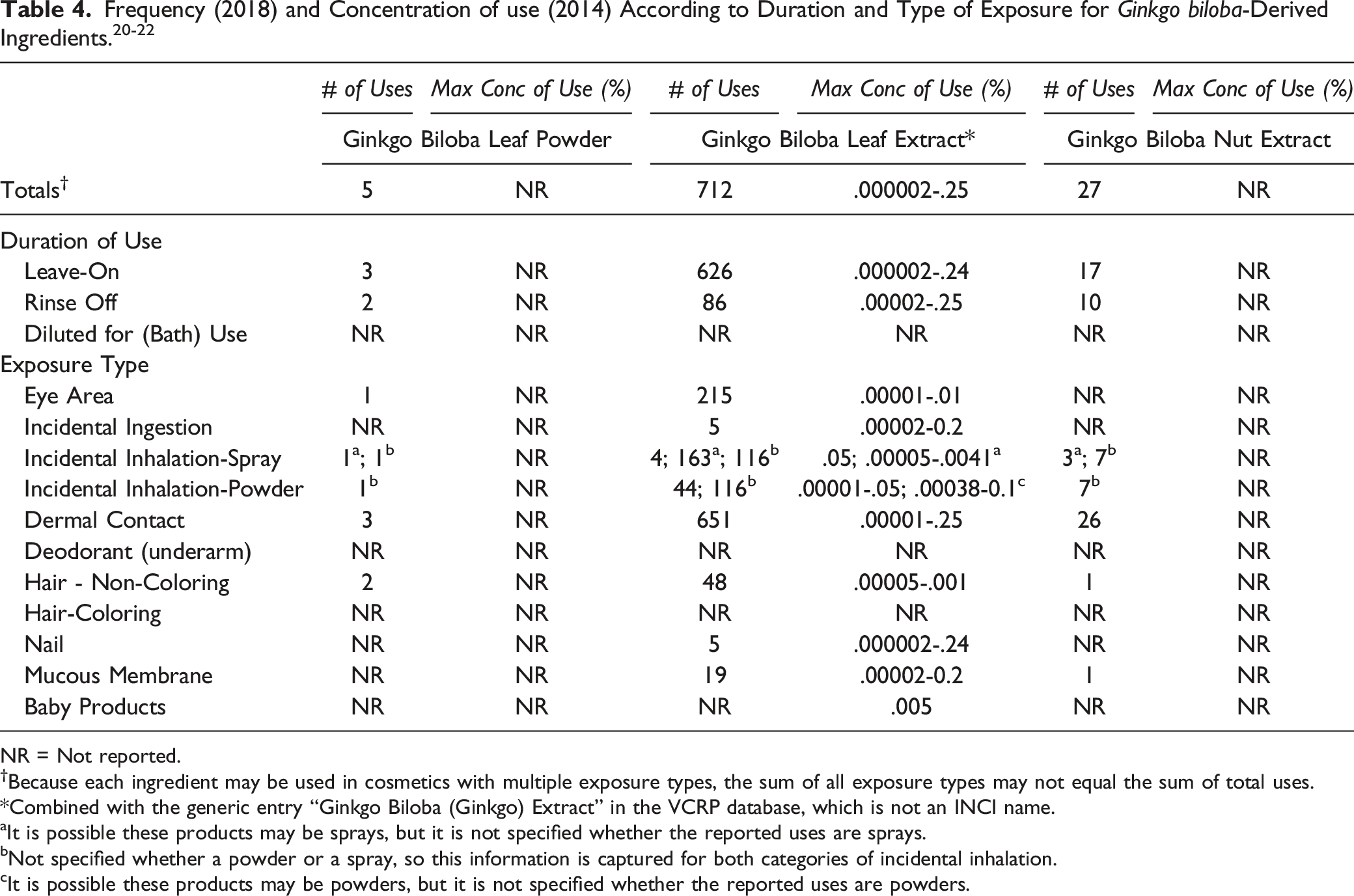
NR = Not reported.
†Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
*Combined with the generic entry “Ginkgo Biloba (Ginkgo) Extract” in the VCRP database, which is not an INCI name.
aIt is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
bNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
cIt is possible these products may be powders, but it is not specified whether the reported uses are powders.
Ginkgo Biloba Leaf Extract may be used in products that can be incidentally ingested or come into contact with mucous membranes; for example, use is reported in a lipstick at up to .2%.20,22 Additionally, Ginkgo Biloba Leaf Extract has been reported to be used in products that may come into contact with the eyes, such as eye shadows and eye lotions at up to .01%.20,22 Moreover, Ginkgo Biloba Leaf Extract was reported to be used in spray products that could possibly be inhaled, like pump spray suntan products at a maximum concentration of .05%. 22 In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles below 10 μm compared with pump spray.24-27 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.24,26 Ginkgo Biloba Leaf Extract is also used in powders, and these products could possibly be inhaled; for example, it is used in face powders at a maximum concentration of .05%. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.28-30
The
Non-Cosmetic
GBE is used extensively as an herbal supplement for anti-inflammatory, cognitive-promoting, antioxidant, and vascular effects at daily doses of 120 to 240 mg.2,3,32 In Germany, GBE is an approved herbal medicine for use for treatment of memory deficits, dementia, and other organic brain syndromes when extracted with acetone/water. 14 It is not approved when extracted with other solvents due to lack of supporting safety data.
Standardized GBEs and/or constituents of the extracts, such as bilobalide, kaempferol and ginkgetin, have also been studied for potential neuroprotective effects against Huntington’s disease, and for anti-inflammatory and analgesic effects on post-surgical incisions. Additionally, these extracts have been researched for their effects on diseases such as osteoarthritis and atopic dermatitis, for protective effects (antioxidant) against radiation and chemotherapy-induced toxicity, for anticancer effects, and for therapy for vitiligo.33-41
GBE as an herbal supplement may interact with pharmaceutical drugs and act as or enhance anticoagulants, anti-inflammatory agents, antihypertensives, and/or anesthetics which may lead to hemorrhage, apraxia, hematoma, hyphema, permanent neurological deficit, and death.42,43 The
The nuts of
Toxicokinetic Studies
In general, toxicokinetics data are not expected to be found on botanical ingredients because each botanical ingredient is a complex mixture of hundreds of constituents. However, there have been many pharmacokinetics studies on GBEs, specifically on some of the key constituents, which indicate GBE may be well absorbed after oral administration. 9
Dermal Penetration
The ability of the GBE constituent, quercetin, to penetrate the skin while in a cosmetic formulation was studied in vitro with human dermatomed skin.
44
The cosmetic formulation used in the study was an emulsion containing trilaureth-4 phosphate, ammonium acryloyldimethyltaurate/VP copolymer and emollients, sclerotium gum, humectants, preservatives, and water that was prepared and supplemented with 6.0% (w/w) tritiated
Absorption, Distribution, Metabolism, and Excretion (ADME)
Animal
The absorption, distribution, and elimination of a radiolabeled GBE were studied in male and female Sprague-Dawley rats.9,45 The rats received a single oral suspended dose (20 μCi; 380 mg/kg) of a radiolabeled GBE. The test material was obtained from
Human
The bioavailability and pharmacokinetics of
Blood samples (36 mL) were taken 30 min prior to administration and 15, 30, 45, 60, and 360 min after administration. The samples were centrifuged to separate the plasma and plasma was analyzed by LC-MS. The resulting maximum concentrations (median) of bilobalide, ginkgolide A and ginkgolide B in plasma after administration of the maximum daily dose of the different
Toxicological Studies
Acute Toxicity Studies
Oral
Ginkgo Biloba Leaf Extract
The LD50 of a standardized GBE (EGb 761®) administered orally to mice was reported to be 7730 mg/kg. 47
Ginkgo Biloba Meristem Cell
In a toxicity test to determine lethal dose, a single oral dose of 0 or 2000 mg/kg Ginkgo Biloba Meristem Cell was administered to 5 male and female Sprague-Dawley rats in each group (written as provided, no further details). 48 After a 14-d observation period, the animals were killed and underwent necropsy. No unscheduled deaths or treatment-related effects were observed during the observation period or at necropsy. The lethal dose for Ginkgo Biloba Meristem Cell was greater than 2000 mg/kg in this rat study.
In a single dose oral volume increase toxicity test, 2 male and female Beagle dogs (written as provided, no further details) received Ginkgo Biloba Meristem Cell at 250, 500, and 1000 mg/kg, respectively, for 4 d. 48 No unscheduled deaths were observed. All animals vomited after receiving 500 and 1000 mg/kg of the test material. Only 1 animal vomited after receiving the 250 mg/kg dose, but the effects were determined to be too slight a symptom to confirm treatment-related effects. No adverse effects were observed in body weights or at necropsy. The maximum tolerated dose for Ginkgo Biloba Meristem Cell was determined to be greater than 1000 mg/kg in this dog study.
Intravenous
Ginkgo Biloba Leaf Extract
The LD50 after intravenous administration of a standardized GBE (EGb 761®) was 1100 mg/kg for both rats and mice. 9
Short-Term Studies
Oral
Ginkgo Biloba Leaf Extract
The results of a combined liver comet assay (see Genotoxicity section) using male and female C3H-derived constitutive androstane receptor knockout (CARKO) and wild-type mice found no abnormal clinical signs and no treatment-related effects on body weight following oral exposure of up to 2000 mg/kg body weight/day of a GBE used by the NTP for 3 d in either mouse genotype. 49 Relative liver weights were significantly increased in male and female wild-type mice at all doses of a GBE in a dose-dependent manner. The liver weights in the CARKO mice were similar to the negative control group. The wild-type mice in all GBE-treated groups had dose-dependent slight-to-moderate hepatocellular hypertrophy in the centrilobular area: this effect was only observed in a single CARKO mouse in the highest dose group. No histopathological findings suggesting cytotoxicity in the liver was observed in any GBE-treated groups.
Ginkgo Biloba Meristem Cell
In a dose-range finding study for a 13-wk oral repeated dose toxicity test (see below), groups of male and female Sprague-Dawley rats received 500, 1000, or 2000 mg/kg Ginkgo Biloba Meristem Cell for 4 wk (number of rats/group and method of administration not described). 48 No unscheduled deaths or clinical signs of toxicity were observed during the treatment period. Additionally, no treatment-related changes in body weight gains, feed intake, hematological/biochemical measurements, or organ weights were observed. No adverse effects were noted at necropsy in any dose group.
Subchronic Toxicity Studies
Oral
Ginkgo Biloba Leaf Extract
The toxicity of a specific GBE was investigated in a 3-mo mouse study performed by the NTP. 9 Groups of 10 male and 10 female B6C3F1/N mice received 0, 125, 250, 500, 1000, or 2000 mg/kg body weight of the GBE in corn oil via gavage, 5 d/wk for 14 wk. Control groups received corn oil (5 mL/kg). Clinical findings and body weights were recorded initially, then weekly, and at the end of the study. Blood was collected at the end of the study from all animals for hematology analyses. Sperm motility and vaginal cytology evaluations were made on the mice in the 0, 500, 1000, and 2000 mg/kg dose groups. At the end of the study period, tissues from over 40 sites were examined for every animal, including ovaries and uteri in females and prostate gland and testes with epididymis and seminal vesicles in males.
One female mouse in the 1000 mg/kg group died of a dosing accident during week 11. Mean body weights of 2000 mg/kg females were significantly less than those of the vehicle control group. Ruffled fur was observed in two 1000 mg/kg males between weeks 7 and 8 and all 2000 mg/kg males between weeks 5 and 9. No treatment-related differences were observed in sperm parameters in males administered 500, 1000, or 2000 mg/kg or in the estrous cycle of females administered 500 or 1000 mg/kg when compared to controls. Female mice in the 2000 mg/kg group had a significantly higher probability of extended estrous than did the vehicle control females. Liver weights of males of the 250 mg/kg or greater dose groups and females of all dose groups were significantly greater than those of the vehicle control groups. Kidney weights of males of the 2000 mg/kg group were significantly less than those of the vehicle control group. Incidences of hepatocytic hypertrophy were significantly increased in males and females dosed with 250 mg/kg or greater. Significantly increased incidences of focal hepatocytic necrosis occurred in males of the 1000 and 2000 mg/kg dose groups. The incidences of hyaline droplet accumulation in the respiratory epithelium of the nose were significantly increased in males of the 500 mg/kg and females of the 1000 and 2000 mg/kg dose groups. In the olfactory epithelium of the nose, the incidences of hyaline droplet accumulation were significantly increased in the 125 (female only), 500, and 1000 mg/kg groups. Incidences of atrophy of the olfactory epithelium were significantly increased in the 1000 mg/kg groups. The incidences of pigment accumulation in macrophages in the olfactory epithelium were significantly increased in males in the 500 mg/kg or greater groups and in females in the 1000 and 2000 mg/kg dose groups. 9
The NTP also performed a 3-mo study of the same GBE used above in rats. 9 Groups of 10 male and 10 female F344/N rats received 0, 62.5, 125, 250, 500, or 1000 mg/kg body weight of the GBE in corn oil via gavage, 5 d/wk for 14 wk. Additional groups of 10 male and 10 female rats received the same doses for a clinical pathology study, 5 d/wk for 23 d. Control groups received corn oil (2.5 mL/kg). The same methods that were followed in the mouse study described above were used in the main study animals, while animals in the clinical pathology study had blood samples collected on days 4 and 23.
All rats survived to the end of the study. Mean body weights of all dosed groups were similar to those of the vehicle control groups. No treatment-related clinical findings were observed. Liver weights of all dosed groups of males and females were significantly greater than those of the vehicle control groups. Incidences of hepatocyte hypertrophy in all dosed groups of males and in 500 and 1000 mg/kg females were significantly greater than those in the vehicle control groups; there was a dose-related increase in severity of this lesion in males. “Hepatocyte fatty change” occurred in all dosed males. The incidences of thyroid gland follicular cell hypertrophy were significantly increased in 500 and 1000 mg/kg males and in 1000 mg/kg females. The incidences of pigmentation in the olfactory epithelium of the nose were significantly increased in 500 and 1000 mg/kg males and in females administered 125 mg/kg or greater. 9
Ginkgo Biloba Meristem Cell
In a 13-wk oral study, groups of 10 male and female Sprague-Dawley rats received 250, 500, or 1000 mg/kg Ginkgo Biloba Meristem Cell (further dosing details were not provided). 48 Observations made during the treatment period included clinical signs of toxicity, body weight and feed measurements, ophthalmology assessment, and urinalysis. At study end, necropsy, hematological/biochemical examinations of blood, organ weight measurement, microscopic examination, and histopathological examination were performed. No unscheduled deaths or adverse clinical signs of toxicity were observed during the treatment period in any dose group. No treatment-related adverse changes were reported in any of the measured parameters before or after necropsy. Based on the results of this study, the no-observed-adverse-effect-level (NOAEL) in rats for Ginkgo Biloba Meristem Cell was determined to exceed 1000 mg/kg.
Chronic Toxicity Studies
Oral
Ginkgo Biloba Leaf Extract
There was no evidence of organ damage or impairment of hepatic or renal function when a standardized GBE (EGb 761®) was administered orally over 27 wk to rats and mice at doses ranging from 100 to 1600 mg/kg. 47 No further details were provided.
The results of the NTP chronic toxicity bioassays are summarized in the Carcinogenicity section below.
Developmental and Reproductive Toxicity (DART) Studies
The reproductive and developmental toxicity of a standardized GBE (EGb 761®) was studied in mice. In one study, groups of 25 mated female CD-1 mice received 0, 100, 350, or 1225 mg/kg/d GBE in tap water via gavage (20 mL/kg) on days 6 through 15 of gestation. 50 The dams were observed daily for clinical signs of toxicity. Feed and water consumption were monitored during the study. Body weight was measured daily. On day 17 of gestation, the dams were killed and the ovaries, uteri, and the fetuses were removed. The internal organs and the placentae of the dams were examined macroscopically. The fetuses were examined for several parameters, including external and internal damages (malformations), sex, viability, and weight. The skeletal systems and soft tissues of the fetuses were also examined.
No clinical signs of toxicity were observed in the dams and there were no unscheduled deaths. No treatment related effects were observed in body weight gains or feed and water consumption. There were no pathological findings observed during necropsy. No embryotoxic effects were observed during external and internal examinations of the fetuses nor were any observed in skeletal or soft tissues. There were no increased incidences of malformation, variations, or retardations. The authors concluded the no-observed-effect-level (NOEL) was greater than 1225 mg/kg/d for both the dams and the fetuses in this study of a standardized GBE. 50
Another study examined the dose response and pathologic effects of a standardized GBE (EGb 761®) in saline on cycling female Swiss albino mice. 51 The test material was orally administered at doses of 0, 3.7, 7.4, or 14.8 mg/kg body weight/d for 28 d from the day of estrus phase (prior to mating), from day 1 to day 7 of gestation, or from day 10 to day 18 of gestation. A total of 200 cycling female mice were assigned for the experiments. There were 10 animals for each group used to study the effect of graded doses of GBE on anti-implantation and abortifacient activities and the remaining 120 animals were used to study the reproductive cycle (40 mice, 10 per group). Blood hormones of non-pregnant mice were measured on day 28. Kidneys, liver, brain, placenta, spleen and ovaries were quickly removed and weighed from all animals that were killed. Post-mortem evaluations included preparing ovaries for histological examinations, and counting ovarian follicles. Maternal toxicity, estrous cycle, reproductive hormones, ovarian follicle counts, resorption index, implantation index, fetal viability and fetuses, and placenta mean weights were also evaluated.
No signs of clinical toxicity such as depressed activities, respiratory distress, salivation, tremor, fasciculation, dull eyes, diarrhea, or change in fur appearance were observed in the dams during any of the treatments, and there were no unscheduled deaths. Statistically significant decreases in body weight gains were observed in the 14.8 mg/kg/d dose group treated for 28 d when compared to the controls. In comparison to body weight, there were no treatment-related differences in the relative weights of the liver, kidney, brain, spleen, ovary, and placenta, but there was a significant dose-dependent decrease in the relative weight of the gravid uterus in the 14.8 mg/kg/d dose group treated for 28 d when compared to controls. Ovarian follicle counts, resorption index, implantation index, and fetal viability were significantly reduced in 14.8 mg/kg/d dose group. Treatment with 14.8 mg/kg bw/d of this particular GBE induced disruption of estrous cycle and caused maternal toxicity, in addition to fetal toxicity. No adverse effects were observed in the 3.7 or 7.4 mg/kg bodyweight/d dose groups in any of the different test groups. The authors concluded that 14.8 mg/kg body weight/d of this GBE produced adverse effects on the estrous cycle, fertility, reproductive performance, and hormone levels of female mice and may cause adverse effects on ovarian function as an antifertility agent. The highest dose tested was based on the equivalent to the suggested supplement dose level for humans of three 260 mg capsules/d. 51
The effects of an aqueous GBE (similar to EGb 761®) on embryo-fetal development were investigated in pregnant Wistar rats. 52 Groups of 17 rats received 0, 3.5, 7, or 14 mg/kg/d of the test material during the tubal transit and implantation period of pregnancy. The dams were then killed on the 15th day of pregnancy. The following parameters were evaluated during the study: clinical symptoms of maternal toxicity; maternal body weight; feed and water intake; maternal liver, kidney, and ovary weights; number of corpora lutea; implants per group ratio; pre- and post-implantation loss per group ratio; live fetuses mean; dead fetuses percentage; fetus and placenta weight per offspring ratio; and fetal external malformation. No significant adverse effects were observed for any of the parameters in the dams or the embryos. The authors of this study concluded that the studied GBE did not produce adverse effects in maternal or embryonic rats.
Genotoxicity
In Vitro
Ginkgo Biloba Leaf Extract
The specific GBE tested by the NTP at up to 10,000 μg/plate was mutagenic in an Ames test using
The genotoxicity of the same GBE and eight of its constituents (quercetin; quercetin-3-β-D-glucoside; kaempferol; isorhamnetin; ginkgolide A; ginkgolide B; ginkgolide C; and bilobalide) were evaluated in mouse L5178Y cells using a lymphoma assay and a Comet assay.
53
The GBE (.2–1.2 mg/ml) and the eight constituents were tested in a dimethyl sulfoxide (DMSO) solution. A dose-dependent increase in mutant frequency was observed in the studied GBE, quercetin (10-100 μM), quercetin-3-β-
In a comparative review and analysis of published and unpublished data on the GBE herbal supplement EGb761®, the authors of the review concluded that the positive findings in some in vitro genotoxicity tests are associated with cytotoxic effects of the
Gingko Biloba Meristem Cell
Ginkgo Biloba Meristem Cell at up to 5000 μg/plate was not mutagenic in an Ames test in
Ginkgo Biloba Meristem Cell did not induce chromosomal aberrations in Chinese hamster lung cultured cells, with and without metabolic activation. 48 The cells were treated with 210.0 μg/ml without metabolic activation (short-time treatment), 333.6 μg/ml with metabolic activation (short-time treatment), and 202.2 μg/ml without metabolic activation (24 h continuous treatment). Short-time treatment was not defined.
In Vivo
Ginkgo Biloba Leaf Extract
In a micronucleus test in male and female B6C3F1/N mice performed by the NTP, no increase in the frequency of micronucleated erythrocytes was observed in peripheral blood of male mice administered 125 to 2000 mg/kg/d of a GBE orally for 3 mo.
9
Female mice that received the same doses had results that were deemed equivocal based on a significant trend test and due to no individual dose group being significantly elevated over the vehicle control group. A significant (
In a reporter gene mutation assay using male B6C3F1
This assay was performed in conjunction with a combined liver comet assay and bone marrow micronucleus assay using male and female CARKO and wild-type mice. The short-term toxicity effects were described in the Toxicological Studies section. In the micronucleus study, no significant alterations in the percentages of PCEs were observed in females of either genotype; however, a significant decrease in the percentage of PCEs were observed in both genotypes in males, indicating the studied GBE induced bone marrow toxicity in male mice. In the comet assay, there was no significant difference in the percent tail DNA in any of the GBE-treated groups in either mouse genotype. Heavily damaged cells called “hedgehogs” indicating cytotoxic effects were not detected in any animals. The researchers performing these 3 assays concluded that the studied GBE is not genotoxic. 49
Ginkgo Biloba Meristem Cell
In a micronucleus test, no increase in the frequency of micronucleated polychromatophilic erythrocytes in bone marrow was observed in male mice administered 500 to 2000 mg/kg/d Ginkgo Biloba Meristem Cell. 48 There was no significant difference in the ratio of polychromatophilic erythrocytes in total red blood cells when compared to the negative control. The positive control yielded expected results. No further details were provided.
Carcinogenicity
The carcinogenic potential of a GBE administered orally was studied by the NTP in male and female rats and mice. 9 In the study on mice, groups of 50 male and 50 female B6C3F1/N mice received 200, 600, or 2000 mg/kg of this GBE in corn oil 5 d/wk for 104 wk via gavage. In the study on rats, groups of 50 F344/N male and 50 female rats received 100, 300, or 1000 mg/kg body weight of this GBE for 104 (males) or 105 (females) wk via gavage. Control groups received corn oil (5 mL/kg in mice and 2.5 mL/kg in rats). In rats involved in what was deemed a “special study,” groups of 10 male and female rats received the same doses as in the main study; blood was collected from these rats on day 22 and at week 14 for thyroid hormone analyses and other analyses of the liver and thyroid gland. All animals were observed twice daily. Body weights were evaluated at study beginning and ending and at different intervals during the course of the study. At the end of the study period, tissues from over 40 sites were examined for every animal, including ovaries and uteri in females and prostate gland and testes with epididymis and seminal vesicles in males.
In mice, mortality was significantly higher in the 600 and 2000 mg/kg males than in the vehicle controls, with the most frequent cause of death being liver tumors. Survival in the 600 mg/kg females was significantly greater than that of the vehicle controls. Mean body weights in the mid- and high-dose group male mice were less than (10% or more) those of the vehicle controls after weeks 85 and 77, respectively. The mean body weights of the high-dose females were generally less than the vehicle controls between weeks 17 and 69 and after week 93.
In rats, mortality in the 1000 mg/kg males was significantly higher than that of the vehicle controls, with the most frequent cause of death being mononuclear cell leukemia. The survival of the treated groups of female rats was comparable to the vehicle control. In week 14, all dose group males and females of the 1000 mg/kg group in the special study had increased levels of thyroid stimulating hormone compared to the vehicle controls; the increase was dose-related in the male rats. Mean body weights in the mid- and high-dose male and female rats were less than (10% or more) those of the vehicle controls after weeks 93 and 89, respectively.
Lesions in the liver, thyroid gland, and nose were observed in all the studied GBE dose groups in mice and rats. These lesions included hypertrophy in the liver and thyroid gland in rats and mice, liver hyperplasia in male and female rats, and hyperplasia and atrophy of the epithelium in the nose of male and female rats. Inflammation, hyperplasia, hyperkeratosis, and ulcers were also observed in the forestomach of male and female mice. Additionally, increased incidences of cancers of the thyroid gland were observed in male and female rats and male mice and of liver cancers in male and female mice. The study concluded that the studied GBE caused cancers of the thyroid gland in male and female rats and male mice, and cancers of the liver in male and female mice. 9
In dietary carcinogenicity studies of a standardized GBE (EGb 761®) in mice (at up to 200 mg/kg/d) or rats (at up to 100 mg/kg/d), no neoplastic or pre-neoplastic effects were observed. 54 The rodents received the test material for up to 85 wk. No changes in body weight gain were reported. No further details are available.
The International Agency for Research on Cancer (IARC) has determined that GBEs are possibly carcinogenic to humans (group 2B) based on inadequate human carcinogenicity evidence and sufficient evidence in experimental animals. 55 The animal data used to reach this determination were from the NTP studies that are described above that used a specific GBE. IARC also reviewed the findings of a randomized control study, 4 nested case-control epidemiological studies researching the potential effects of the use of GBE dietary supplements in elderly patients, and a population based case-control epidemiological study in ovarian cancer patients. IARC suggested that the mechanisms for carcinogenicity associated with GBEs may be genotoxicity and/or topoisomerase inhibition that could be related to the constituents: quercetin, kaempferol, and/or rutin.
Other Relevant Studies
Immunotoxicity
In a popliteal lymph node assay (PLNA), the sensitization potential of a GBE was evaluated. 13 Groups of male C57BL/6 mice received subplantar injections of 10 μl DMSO (induction) followed by another injection of DMSO (negative control group), a crude ethanolic-aqueous GBE, heptane fraction of the crude GBE, or diphenylhydantoin (positive control group) at doses of 2 mg each. The negative control yielded small enlargement of the lymph nodes, while the crude ethanolic-aqueous GBE resulted in statistically significant lymphoproliferative reaction (LPR) in the ipsilateral popliteal lymph node. A massive lymph node hyperplasia that was almost comparable to the positive control was observed in the heptane solution fraction of the crude GBE. Chemical analyses of the crude extract and the heptane fraction found ginkgolic acid at 5.5% and 24.6%, respectively, which were theorized to be responsible for the LPR observed in this study.
Dermal Irritation and Sensitization Studies
Irritation
Human
No irritation was observed in a 24-h human patch test of a Ginkgo Biloba Leaf Extract (100%; ethanol:water: butylene glycol extract) in 20 subjects. 6 No further details were provided.
Sensitization
Animal
The sensitizing potential of ginkgolic acid and a GBE was studied in 10 female albino guinea pigs using a modified Freund’s complete adjuvant (FCA) technique.
56
The pure ginkgolic acid was extracted from
Human
Cross-Reactivity
Guinea pig sensitization studies of crude
Phototoxicity/Photosensitization
No phototoxicity or photosensitization was reported to a lip product containing .0072% Ginkgo Biloba Leaf Extract in a study of 29 subjects. 59 The test material was applied neat under semi-occlusive patches. No further details were provided.
Ocular Irritation Studies
In an EpiOcular in vitro assay of an eye product containing .013% Gingko Biloba Leaf Extract, it was predicted that the test substance had no potential for eye irritation. 59 No further details were provided.
Clinical Studies
Case Studies
The fruit pulp of the
A 66-yr-old woman presented with progressive erythematous eruption over the face, neck, trunk, and extremities that started approximately 1 week after the patient had ingested two 60 mg doses of a GBE supplement. 63 No other new medications or changes in behavior were reported. A physical examination, complete blood cell count, and chemistry panel were unremarkable. The authors of the report did not disclose if patch or skin prick tests were performed.
A 45-yr-old man developed acute generalized exanthematous pustulosis on his limbs and face 48 h after starting an oral GBE treatment for tinnitus. 64 The patient had not previously taken any GBEs before and was not taking any other medication. The patient had no history of adverse drug reactions or psoriasis. The rash cleared within 10 d of stopping the GBE treatment. The patient refused a follow-up cutaneous patch test.
In anecdotal accounts from Chinese medicine, consumption of fresh
Other Clinical Reports
No adverse effects were reported in a clinical study of two cosmetic formulations containing 1.5% GBE (glycolic extract standardized by quercetin concentrations) and other antioxidants in 45 volunteers (no further information provided on adverse effect testing). 65 One formulation contained sunscreen and was applied during the day, while the other formulation was without sunscreen and was applied at night. These formulations were applied daily for 90 d.
In another clinical study, no adverse effects were reported in 20 volunteers following use of a cosmetic formulation containing .30% GBE twice daily for 28 d. 66 No further details regarding the GBE used or on adverse effect testing were provided.
Numerous studies have investigated the efficacy and safety of GBEs in humans in the treatment of various afflictions. In a cross-matching review of much of this published toxicological and clinical data on GBEs (mainly the herbal supplement EGb 761®), the authors of the review evaluated the findings of 75 clinical studies with a total of 7115 patients treated orally with GBEs and found no specific or serious undesired reactions to GBEs. 54 Any adverse events observed frequently occurred at the same frequency as placebo treatments. Based on cross-matching data on the historic use by humans, large intake, toxicological and clinical studies, the authors concluded that GBEs are well tolerated and safe.
Summary
According to the
According to 2018 VCRP survey data, Ginkgo Biloba Leaf Extract has the most reported uses in cosmetic products, with a total of 712; the majority of the uses are in leave-on eye makeup preparations and skin care products. Two other
GBEs are used extensively as an herbal supplement for anti-inflammatory, cognitive-promoting, antioxidant, and vascular effects and are approved herbal medicines in Germany for use for treatment of memory deficits, dementia, and other organic brain syndromes when extracted with acetone/water. GBEs may interact with pharmaceutical drugs. Nuts from
In general, toxicokinetics data are not expected to be found on botanical ingredients because each botanical ingredient is a complex mixture of hundreds of constituents. However, there have been many pharmacokinetics studies on GBEs, specifically on some of the key constituents, which indicate GBEs may be well absorbed after oral administration. The GBE constituent, quercetin, was found to penetrate human dermatomed skin; however, quercetin was not present in the dermis or receptor fluid of this dermal penetration study. In an oral ADME study in rats, at least 60% of a radiolabeled GBE (flavonol glycosides and proanthocyanidins) was absorbed, with the main site of absorption likely in the upper gastrointestinal tract. Radioactivity was measured in exhalation and elimination products. In a human plasma study, ginkgolide A, ginkgolide B, and bilobalide were found to be bioavailable after single oral dosing of 3 different
The LD50 of a standardized GBE (EGb 761®) administered orally to mice was reported to be 7730 mg/kg, and the LD50 after intravenous administration with this standardized GBE was 1100 mg/kg for both rats and mice. The lethal dose for Ginkgo Biloba Meristem Cell was greater than 2000 mg/kg in rats and the maximum tolerated dose for this ingredient was greater than 1000 mg/kg in dogs.
In 3-mo studies by the NTP of a specific GBE at up to 2000 mg/kg/d in corn oil, increased liver weights, decreased kidney weights, increased incidences of hepatocytic hypertrophy and focal hepatocytic necrosis, and increased incidences hyaline droplet accumulation, atrophy and pigment accumulation in macrophages in the olfactory epithelium were observed in mice. In a similar NTP study of the same GBE test material in rats, increased liver weights, increased incidences of hepatocyte hypertrophy, increased incidences of thyroid gland follicular cell hypertrophy, and increased incidences of pigmentation in the olfactory epithelium of the nose were observed. There was no evidence of organ damage or impairment of hepatic or renal function when a GBE (EGb 761®) was administered orally over 27 wk to rats and mice at doses ranging from 100 to 1600 mg/kg. In a 4-wk oral repeated dose study, no adverse effects were observed in rats that received up to 2000 mg/kg Ginkgo Biloba Meristem Cell. In the follow-up 13-wk oral study, the NOAEL in rats for Ginkgo Biloba Meristem Cell was greater than 1000 mg/kg.
In an oral DART study in which mated female mice received standardized GBE (EGb 761®) on gestation days 6 through 15, the NOEL for dams and fetuses was greater than 1225 mg/kg/d. No maternal toxicity and no embryotoxic effects were observed. Another oral DART study investigated the effects of standardized GBE (EGb 761®) in female mice that received the test material during a 28-d period before mating, on gestation days 1 through 7, or on gestation days 10 through 18. The standardized GBE produced adverse effects at 14.8 mg/kg/d, including effects on the estrous cycle, fertility, reproductive performance, and hormone levels. The standardized GBE may also cause adverse effects on ovarian function as an antifertility agent. In an embryo-fetal development study, no adverse effects were observed in maternal or embryonic rats following dosing of an aqueous GBE similar to EGb 761® on gestation days 1 through 14, with doses up to 14 mg/kg/d.
The authors of a comparative review and analysis of published and unpublished data of the GBE herbal supplement EGb761® concluded that the positive findings in some in vitro genotoxicity tests are linked to cytotoxic effects of
In oral carcinogenicity studies of rats and mice conducted by the NTP, lesions in the liver, thyroid gland and nose were observed in all GBE dose groups (200–2000 mg/kg/d in corn oil, by gavage). Lesions included hypertrophy in the liver and thyroid gland in rats and mice, liver hyperplasia in male and female rats, and hyperplasia and atrophy of the epithelium in the nose of male and female rats. Inflammation, hyperplasia, hyperkeratosis, and ulcer were also observed in the forestomach of male and female mice. Additionally, increased incidences of cancers of the thyroid gland were observed in male and female rats and male mice, as were liver cancers in male and female mice. In dietary carcinogenicity studies of a standardized GBE (EGb 761®) in mice (at up to 200 mg/kg/d) or rats (at up to 100 mg/kg/d) for up to 85 wk, no neoplastic or pre-neoplastic effects were observed. IARC has determined that GBEs are possibly carcinogenic to humans (group 2B) based on data that included the NTP studies.
In a PLNA validation study, a GBE exposure yielded statistically significant lymphoproliferative reactions in the ipsilateral popliteal lymph node, which may have been caused by ginkgolic acid.
No irritation was observed in a 24-h human patch test of Ginkgo Biloba Leaf Extract (100%; ethanol:water:butylene glycol extract).
In a guinea pig study, sensitization was observed to ginkgolic acid at concentrations of .1% and 1%, but no sensitization was observed to a GBE that contained ∼1000 ppm (∼.1%) ginkgolic acid. No dermal sensitization was reported in HRIPTs of products containing up to .2% Ginkgo Biloba Leaf Extract.
Guinea pig sensitization studies of crude
The results of a phototoxicity and photosensitization study on a lip product containing .0072% Ginkgo Biloba Leaf Extract were negative.
An in vitro assay using an eye product containing .013% Ginkgo Biloba Leaf Extract predicted no ocular irritation.
Reports of contact dermatitis have been reported following exposure to the fruit pulp of
Discussion
This report assesses the safety of cosmetic ingredients derived from the plant
The Panel determined that the available safety test data, methods of manufacturing, and composition and impurities data on Ginkgo Biloba Leaf Extract are sufficient, and reasonable inferences to the safety of the 4 other leaf-derived ingredients can be made. The Panel considered the findings of the carcinogenicity studies performed by the NTP on a
The Panel also expressed concern about pesticide residues, heavy metals, and other plant species that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit impurities.
Ginkgo Biloba Leaf Extract was reported to be used in spray and powder products that could possibly be inhaled, such as pump spray suntan products at a maximum concentration of .05%, and face powders at a maximum concentration of .05%. There were no inhalation toxicity data available. Although the Panel noted that droplets/particles from spray and loose-powder cosmetic products would not be respirable to any appreciable amount, the potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
After reviewing this safety assessment, the Panel determined that although a conclusion of safety could be made for five • Method of manufacturing, composition, and impurities data for each of these ingredients, except Ginkgo Biloba Meristem Cell; • 28 Day dermal toxicity data for each of these ingredients, (a) Dependent on the results of these studies, additional data on other toxicological endpoints, such as developmental and reproductive toxicity and carcinogenicity, may be needed; • Dermal irritation and sensitization data at leave-on use concentrations; and • Ocular irritation data, if available.
Conclusion


Footnotes
Author's Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.



