Abstract
The Expert Panel for Cosmetic Ingredient Safety reviewed updated information that has become available since their original assessment from 2004, along with updated information regarding product types, and frequency and concentrations of use, and reaffirmed their original conclusion that Dioscorea Villosa (Wild Yam) Root Extract is safe as a cosmetic ingredient in the practices of use and concentration as described in this report.
The Expert Panel for Cosmetic Ingredient Safety (Panel) first published the Final Report of the Amended Safety Assessment of Dioscorea Villosa (Wild Yam) Root Extract in 2004. 1 The Panel concluded that based on the chemical and animal data included in the safety assessment, Dioscorea Villosa (Wild Yam) Root Extract is safe for use in cosmetic products. In the Discussion of that report, the Panel further clarified that this conclusion is valid only for extracts prepared in a manner that produces a similar chemical profile as that described in the safety assessment, particularly in regard to diosgenin (ie, an expected upper limit of 3.5%). Additionally, the Panel stated that extracts not prepared in a manner that produces a similar chemical profile would be considered safe if they have a similar safety test profile.
Frequency (2022/1998) and Concentration (2022/1999) of Use According to Likely Duration and Exposure by Product Category.
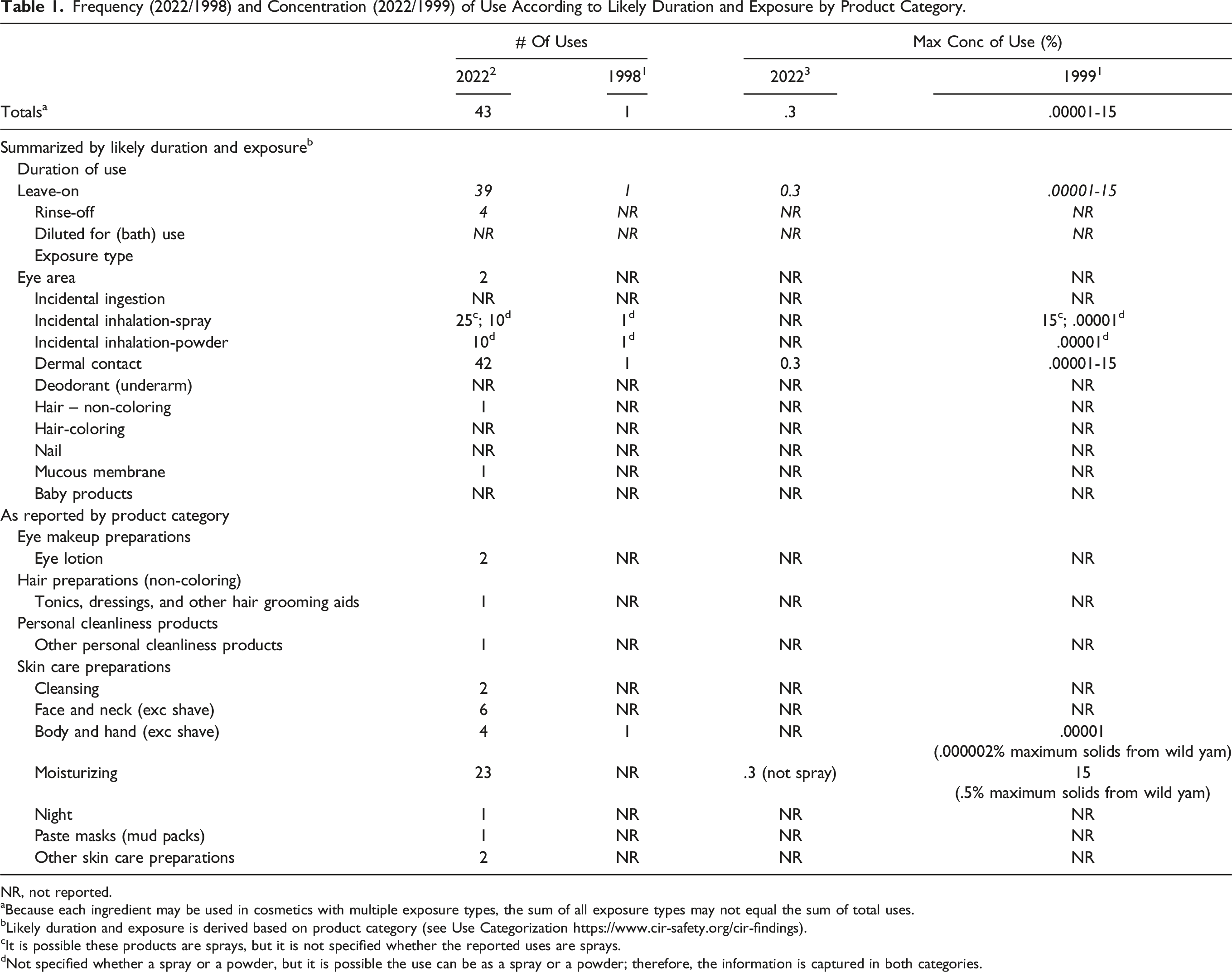
NR, not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bLikely duration and exposure is derived based on product category (see Use Categorization https://www.cir-safety.org/cir-findings).
cIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
dNot specified whether a spray or a powder, but it is possible the use can be as a spray or a powder; therefore, the information is captured in both categories.
In January 2023, an extensive search of the world’s literature was performed for studies dated 1999 forward, and new data were found.4-9 Notable findings include 2 short-term oral toxicity studies and a 13-week oral toxicity study in which the no-observed-adverse-effect-level (NOAEL) for rats of both sexes was determined to be the maximum received dose of 5000 mg/kg/d. Additionally, studies demonstrating the potential cytotoxicity of Dioscorea villosa (wild yam) root extract against breast cancer cell lines, a study of anti-inflammatory effects, and a clinical study in which no significant side effects or metabolic/endocrinal changes were seen in healthy premenopausal women following 3 mo of topical application of wild yam cream, were found.
In summary, the Panel reviewed 2022 frequency and concentration of use data, in addition to any new, available, relevant safety data. Considering this information, as well as the information provided in the original safety assessment, the Panel reaffirmed the 2004 conclusion for Dioscorea Villosa (Wild Yam) Root Extract. The Panel discussed the possibility for this ingredient to be used in cosmetic products which may be incidentally inhaled. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
