Abstract
Roginolisib (IOA-244) is a novel, non-ATP competitive phosphoinositide-3-kinase (PI3K) delta inhibitor that regulates Akt/mTOR signaling. Roginolisib was administered once daily to rats and dogs in dose-range finding (DRF) and 4-week GLP toxicology studies. Free plasma levels of roginolisib exceeded the cellular target engagement IC90 for PI3Kδ for ≥12 hours at doses of 5 mg/kg, the IC90 for PI3Kβ for ≥2 hours at doses ≥15 mg/kg, and the IC50 for PI3Kα for ≥2 hours at dose levels ≥45 mg/kg. Toxicity in rats occurred at doses ≥100 mg/kg. In dogs, we observed dose-dependent skin and gastrointestinal toxicity and doses ≥30 mg/kg had a greater incidence of mortality. Lymphoid tissue toxicity occurred in both species. Toxicities in dogs observed at the ≥15 mg/kg dose, affecting the digestive mucosa, liver, and skin, cleared after treatment cessation. Doses ≤75 mg/kg were tolerated in rats and the no-observed-adverse-effect-level (NOAEL) in rats was 15 mg/kg. Due to mainly epithelial lesions of the skin at 5 mg/kg and necrotizing damage of the intestinal epithelia at ≥15 mg/kg, no NOAEL was determined in dogs. However, the adverse effects observed in dogs at 5 mg/kg were considered monitorable and reversible in patients with advanced malignancies. Furthermore, the PK profile subsequently proved to be a decisive factor for achieving selective PI3Kδ inhibition without the toxicities observed in dogs. As the result of the unique PK profile of roginolisib, patients were able to take daily roginolisib without dose modification and showed pharmacodynamic PI3Kδ inhibition over several months without gastrointestinal or dermatologic toxicities.
Keywords
Introduction
Phosphoinositide 3-kinases (PI3Ks) are heterodimeric lipid kinases that regulate protein kinase B (Akt)/mammalian target of rapamycin (mTOR) signaling, which controls cell growth, motility, survival, and angiogenesis. 1 Aberrant Akt/mTOR signaling occurs in many treatment-resistant cancers.2,3 High expression of the PI3K isoform PI3KD, which encodes the p110 catalytic subunit of PI3Kδ (i.e., p110δ), promotes tumor growth and metastasis; 4 conversely, PI3Kδ inhibition impairs tumorigenesis.5,6 The PI3Kδ isoform is expressed in leukocytes and modulates B- and T-cell activation, B-cell-driven proliferation, and neutrophil inflammatory responses.7,8 Direct inactivation of PI3Kδ in regulatory T cells and myeloid-derived suppressor cells promote tumor regression by increasing effector T-cell immunosurveillance.9,10
Inhibitors of PI3K and mTOR are used to treat various malignancies, but the therapeutic window is narrow. Most PI3K and mTOR inhibitors exhibit limited efficacies as monotherapies and cause dose-limiting toxicities (e.g., fatigue, diarrhea, rash, and hyperglycemia). 11 Inhibiting PI3Kα, Akt and mTOR primarily affects insulin signaling and glucose metabolism, 12 the modulation of which is crucial in mitigating toxic adverse effects and increasing efficacy. 13 However, PI3K isoforms have specific functions and the impact of isoform specificity on the type of toxic adverse effects (AEs) experienced by patients remains to be fully elucidated. New isoform-selective PI3K inhibitors aim to improve efficacy and minimize the frequency and severity of AEs. For example, alpelisib is a PI3Kα inhibitor used in combination therapy to treat breast cancer 14 ; tenalisib is a dual PI3Kδ/γ inhibitor being investigated for treating relapsed/refractory T-cell lymphoma; 15 and idelalisib is a first-in-class PI3Kδ inhibitor that was originally approved by the United States Food and Drug Administration (FDA) for treating relapsed chronic lymphocytic leukemia.16,17 Recently, the FDA’s public review of idelalisib and similar agents recommended their withdrawal from use in multiple indications based on unfavorable benefit:risk ratios. 18 Thus, safer PI3Kδ inhibitors are needed to achieve their full therapeutic potential.
Roginolisib (IOA-244) is a novel, small molecule non-ATP competitive, and potentially a safer and more effective PI3Kδ inhibitor, that is selective for the PI3Kδ isoform. 19 Based on its potency, selectivity, and efficacy in non-clinical models,19,20 roginolisib may provide a therapeutic option for wide ranging malignancies, including solid and hematological tumors with dysregulated PI3K/Akt/mTOR signaling. The non-clinical toxicology studies in both rats and dogs reported here provide insights into the toxicological profile of roginolisib. These findings have advanced roginolisib into clinical investigation and preliminary clinical data suggests this drug is propitious for treating malignancies, including uveal melanoma and lymphoma. 21
Materials and Methods
Test Compound
A free-base or a hemi-fumarate salt form of roginolisib was used in these studies. The activity and pharmacokinetics (PK) of both salt forms were similar (data not shown).
Animals were administered with vehicle alone (.25% aqueous hydroxypropyl methylcellulose [Methocel® K4M Premium]) or specified doses of roginolisib (diluted in the vehicle solution). Throughout, samples of 2 dosing formulation preparations of roginolisib were collected at the beginning and end of usage, and sample concentrations were assessed using high performance liquid chromatography (HPLC). Vehicle or roginolisib was administered orally via gavage in accord with the intended administration route for roginolisib in humans. All treatment doses were defined as mg/kg of bodyweight and were administered once daily in a volume of 5 mL/kg bodyweight for rats and 10 mL/kg bodyweight for dogs.
Animals and Husbandry
Two hundred and seventy-four (137 male and 137 female) healthy Wistar rats aged approximately 8 weeks, whose body weights ranged from 145 to 254 g, were purchased from Charles River Laboratories (Germany and USA). Rats were housed in single-sex groups of 2-3 per cage with free access to food (Provimi Kliba 3433.0) and water, and the room was maintained at 22°C (±2°C) with a relative humidity of 35%-78%, and the day/night cycle was 12:12 hours.
Non-Clinical Toxicology Studies.
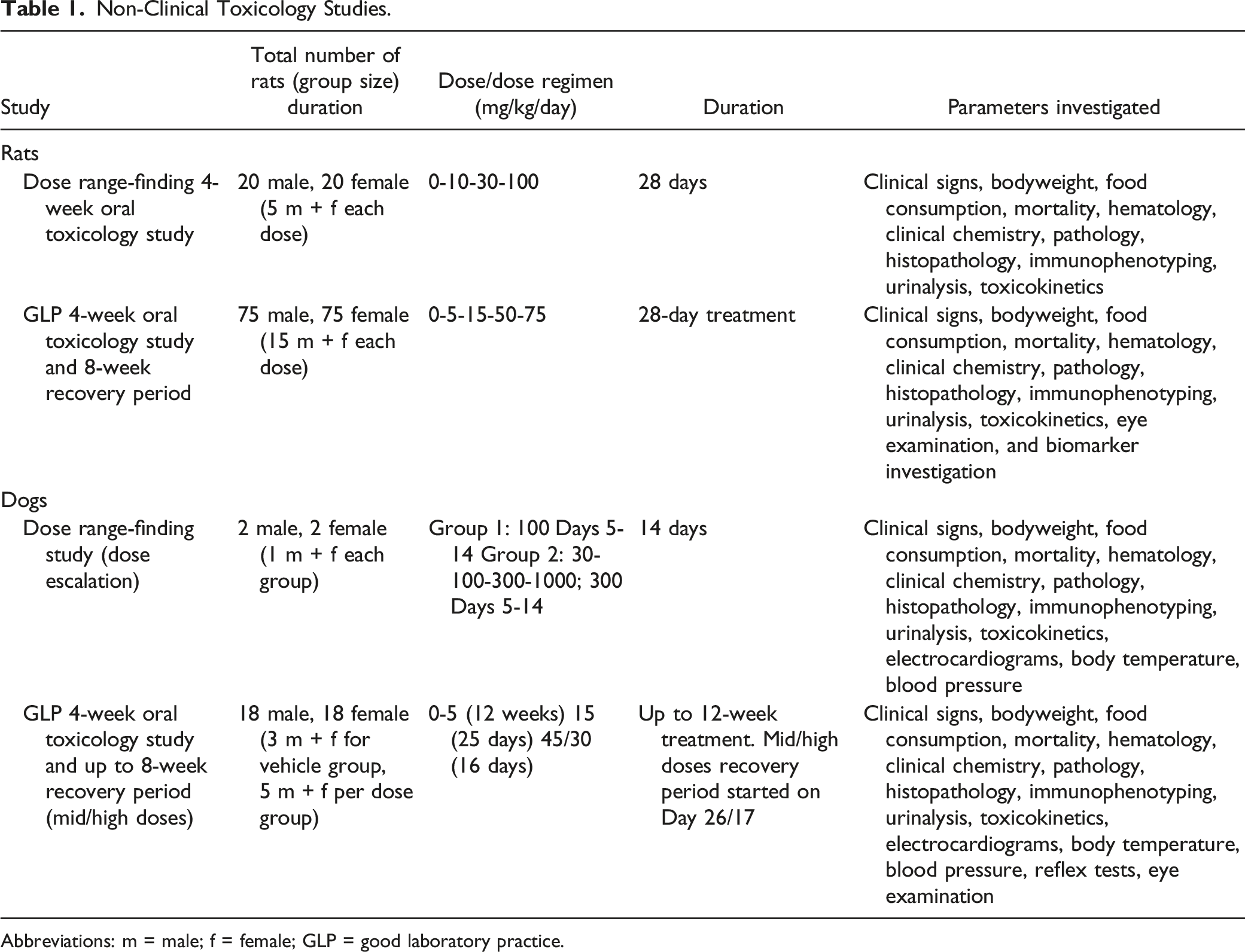
Abbreviations: m = male; f = female; GLP = good laboratory practice.
All studies were conducted in compliance with the Organization for Economic Co-operation and Development Principles of Good Laboratory Practice (GLP) (as revised in 1997), with the Principles of Good Laboratory Practice according to Annex 1 German Chemicals Act, and the Principles of Good Laboratory Practice of the European Union. The GLP standards applied also included the requirements of Japanese, USA, and other non-European guidelines.
Experimental Procedures
Rats
In the 4-week DRF study, 40 rats (20 male and 20 female) were assigned to 4 treatment groups, each consisting of 10 animals (5 male and 5 female). Each group was assigned either vehicle alone or a specific dose of the free-base of roginolisib (10, 30, and 100 mg/kg) that was administered daily for 28 days. Based on the high bioavailability of roginolisib in rat, 20 only 2 additional males and 2 additional females were assigned to each group as "satellite animals" for use in the toxicokinetic investigation. The middle dose of 30 mg/kg was considered a therapeutically effective dose based on the calculated ED50 value of 18 mg/kg in female Lewis rats with semi-established type II collagen arthritis (unpublished data).
In the 4-week oral toxicology study, 150 rats (75 male and 75 female) were assigned to 5 treatment groups, each consisting of 15 males and 15 females. Each group received hemi-fumarate salt roginolisib (0, 5, 15, 50 or 75 mg/kg) daily for 28 days and then the animals were monitored during an 8-week, treatment-free, recovery period. An additional 3-5 males and 3-5 females (68 animals [34 males, 34 females] in total) were assigned to each group as "satellite animals" for the toxicokinetic investigation.
Dogs
In the DRF study, 4 dogs (2 male and 2 female) were assigned to 2 treatment groups. In group 2, dosing started with 30 mg/kg of the free-base of roginolisib on Day 1, and then the roginolisib dose was increased to 100 mg/kg on Day 2, 300 mg/kg on Day 3, and 1000 mg/kg on Day 4 for both dogs. As the top dose was not tolerated in the group 2 female, the remaining group 2 male was treated with 300 mg/kg from Days 5 to 14. Group 1 dogs received a dose of 100 mg/kg of the free-base of roginolisib from Days 5 to 14.
In a 4-week oral toxicology study, 36 dogs (18 males and 18 females) were assigned to 4 treatment groups based on dose (0, 5, 15, 45 mg/kg roginolisib) daily. Each treatment group consisted of 10 dogs (5 male and 5 female), and the control group consisted of 6 dogs (3 males and 3 females). As there were no clinical signs of toxicity after 4 weeks of treatment, the treatment period for the vehicle and the low dose group (5 mg/kg) was extended from 4 to 12 weeks. However, dogs treated with 15 mg/kg (mid-dose group) were prematurely euthanized in Week 4 owing to severe clinical symptoms. Dogs treated with the high dose were initially administered 45 mg/kg for 14 days and then the dose was reduced to 30 mg/kg until Day 21. For the mid- and high-dose groups, 2 animals per sex were investigated during the 8-week recovery period. Based on the nature of the vehicle (.25% aqueous hydroxypropyl methylcellulose in water), only 1 control group was included for both the end of dosing as well as the recovery group assessment.
A minimum of 3 males and 3 females, including all recovery animals (with the exception of 1 female in the high-dose group that was euthanized during the recovery period), from each treatment group were used for the toxicokinetic investigation.
Toxicokinetic Investigations
For the toxicokinetic investigation, blood samples were collected from designated satellite animals, in their respective studies, on multiple days, including Days 1 and 28. Samples were taken before dosing and at 1, 3, 6, and 24 hours post-dose in rats and at .5, 1, 2, 4, 6, and 24 hours in dogs following roginolisib administration. Any variations from this dosing schedule in a study are reported alongside the results.
Whole blood samples were transferred into tubes containing a lithium-heparin anti-coagulation agent, centrifuged under cooled conditions (∼4°C), and subsequently stored at −20°C. Concentrations of roginolisib in the lithium-heparin plasma were determined using an Agilent 1100 Series HPLC system with a CTC Analytics HTC PAL Autosampler coupled to an AB Sciex API4000 mass spectrometer and bioanalytical methods validated according to the FDA Guidance for Industry: Bioanalytical Method Validation, May 2001. Toxicokinetic parameters assessed included the highest concentration value of roginolisib in blood plasma (Cmax), the time at which the Cmax was determined (tmax), and the area under the plasma concentration vs time curve from dosing until 24 hours post-dose (AUC0-24h). The AUC0-24h corresponded to an entire dosing interval and was calculated by the log-linear trapezoidal rule (linear up to the Cmax, logarithmic after the Cmax). To determine the rate of accumulation, an accumulation ratio (Rac AUC0-24h) was used to compare the AUC0-24h for the first dose and the AUC0-24h on subsequent sampling days. The formula used for this calculation was: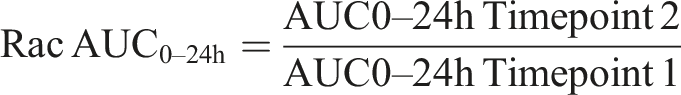
Clinical Symptoms, Bodyweight, and Food Consumption
General condition and behavior as well as food consumption were assessed daily. In addition, bodyweight was measured once per week.
Electrocardiography, Arterial Blood Pressure, and Reflex Tests
In dogs, electrocardiograms (ECGs) were recorded with a MAC 1200ST electrocardiograph (GE Medical Systems, USA). Standard leads I, II, III, and the Goldberger leads aVR, aVL, and aVF were recorded and then standard lead II was examined. Heart rates and interval data were quantified. Van de Water´s equation (V = QT − .087 × [(60/HR) − 1]) was used to dissociate the effects of heart rate on the QT-interval in standard lead II (Van de Water, 1989).
Systolic, diastolic, and mean arterial blood pressure values were measured indirectly using high definition oscillometry (Vet HDO Monitor, S + B medVet, Germany). The dogs were conscious during the measurement. The following reflexes in dogs were tested: pupillary light reflex, lid reflex, patellar reflex, flexor reflex, and anal reflex. All investigations were performed 3 weeks before starting the treatment and once in week 3 during the treatment period in all dogs before and 2 hours after dosing.
Ophthalmology
In the 4-week GLP oral toxicology study in rats, eye examinations (pupillary reflex, anterior parts, and fundus) and further examinations by slit lamp were performed in non-satellite rats before starting the treatment and at the end of treatment, in addition to at the end of the recovery period for rats in the control and the high-dose groups (75 mg/kg).
In the 4-week oral toxicology study in dogs, the anterior and posterior part of the eye was examined [handlamp (Heine Optotechnik, Germany), ophthalmoscope (Welch Allyn, USA), slit-lamp (Zeiss, Germany), fundus camera (KOWA Ltd, Japan)] 4 weeks before initiating treatment, and in weeks 5 and 10 during the treatment period and the recovery period, respectively.
Hematology and Immunophenotyping
Blood samples were collected according to the schedule of each individual study. The following indexes were examined: red blood cells, platelets, immune cells, hemoglobin, and mean cell volume. For the studies using dogs, coagulation parameters including erythrocyte sedimentation rate and prothrombin time were also investigated. All measurements were generated using either ADVIA 120 Hematology System (Siemens Medical Solutions GmbH), FACS Canto Flow Cytometer (BD), and recorded in the LIM-System; Westergren method (erythrocyte sedimentation); or Coasys Plus–Thrombolyzer (Disasys-Greiner, Germany). Samples were also examined visually via a light microscope (ZEISS).
For immune cell phenotyping to identify the absolute number of each lymphocyte population (e.g., CD4+ and CD8+ T cells) the lyse-no-wash method was used. 22 The relative numbers of lymphocytes subpopulations refer to the number of lymphocytes analyzed via Forward/Side scatter measured with a NAVIOS™ flow cytometer (Beckman Coulter GmbH, Germany). The absolute numbers were determined by using flow-count fluorospheres. Here, a known volume and concentration of flow-count fluorospheres were mixed with an identical volume of the biological sample. After analysis on the flow cytometer, the absolute number was calculated by the instrument software.
Clinical Chemistry
Blood and urine samples were collected according to the schedule of each individual study. The following clinical chemistry parameters were investigated in blood: electrolytes, phosphate, iron, glucose, urea, creatinine (CREA), bilirubin (BIL), cholesterol, triglycerides, protein, albumin, alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (AP), and glutamate dehydrogenase (GLDH). All appropriate tests were conducted using either ADVIA 1650 Autoanalyser (Siemens Medical Solutions Diagnostics GmbH), InfiniTE M200 EIA/ELISA-System (TECAN Deutschland GmbH), or COBRA II Gamma Counter (Canberra Packard).
Urinalysis focused on glucose, bilirubin, protein, urobilinogen, blood, ketone, sediment and pH value using Clinitek ADVANTUS Reflection Spectrophotometer (Siemens Medical Solutions Diagnostics GmbH) or Olympus BX40F Microscope (Olympus Optical CO, LTD).
Pharmacodynamic Biomarker
In the 4-week oral toxicology study in rats, the pharmacodynamic effects of roginolisib were evaluated by measuring Akt phosphorylation (pAkt) in whole blood B cells using flow cytometry (BD FACSCanto). For this purpose, blood samples were collected on Days 5, 20, and 28, as well as 1 hour after treatment. Anti-immunoglobulin M (IgM)-induced pAkt (Ser473; Cell signaling Technology, Danvers, MA, US) was measured as previously described [19B].
Pathology
Rat
On the day of necropsy, all rats were anesthetized with a carbon dioxide (CO2) air mixture and terminated via abdominal exsanguination on the scheduled last day of study or sampling (satellite animals). In the event of an animal’s death, from any cause, a necropsy was performed to assess gross pathological alterations, organ weight, and histopathology.
Dog
On the day of necropsy, all dogs were anesthetized by an intravenous injection with thiopental (initial phase: 10 mg/kg; maintaining phase: dose depended on the corneal and flexor reflexes) in the vena saphena or vena cephalica. Afterward, an approximately 10-cm skin incision was performed besides the trachea (mid-cervical region), and the external carotid arteries were prepared bluntly and were cut. In the event of an animal’s death, from any cause, a necropsy was performed to assess gross pathological alterations, organ weight, and histopathology were performed.
Statistical Analyses and Evaluation
In the rat studies, all parameters were analyzed separately for each time. To take the number of dose groups into account, all the test procedures maintained a multiple significance level of .05. Body weight, body weight gain, food consumption, and organ weights of the dose groups were compared with those of the control, using the multiple two-sided Dunnett's test.23,24 For the hematological, clinical chemistry, and immunophenotyping parameters, the Wilcoxon rank-sum test 25 was used to make pairwise comparisons with the control group. The correction for multiple testing was in accordance with Bonferroni-Holm. 26
Due to the small number of dogs used (3 males and 3 females per main group without recovery animals), no statistical test procedure was applied. Additionally, the animals of the mid- and high-dose groups were prematurely killed and the vehicle and low dose group were treated for 12 weeks. These factors further confound the relevance of formal statistical analysis. Hence descriptive statistical analysis was performed and mean values and standard deviations were calculated for all parameters per group for each sex and time point. Body weight, body weight gain, food consumption, hematological and clinical chemistry parameters, and ECG and blood pressure parameters, were recorded and evaluated using the Merck´s proprietary laboratory information management (LIM)-system. The LIM-system was also used to record pathological and histopathological findings.
Results
Dose Range-Finding Studies in Rats and Dogs
Toxicokinetic Investigations
Mean Toxicokinetic Parameters of Roginolisib After Single and Repeated p.o. Administration to Male and Female Rats and Dogs in the DRF and 4-Week GLP Toxicology Studies (Summary Table).
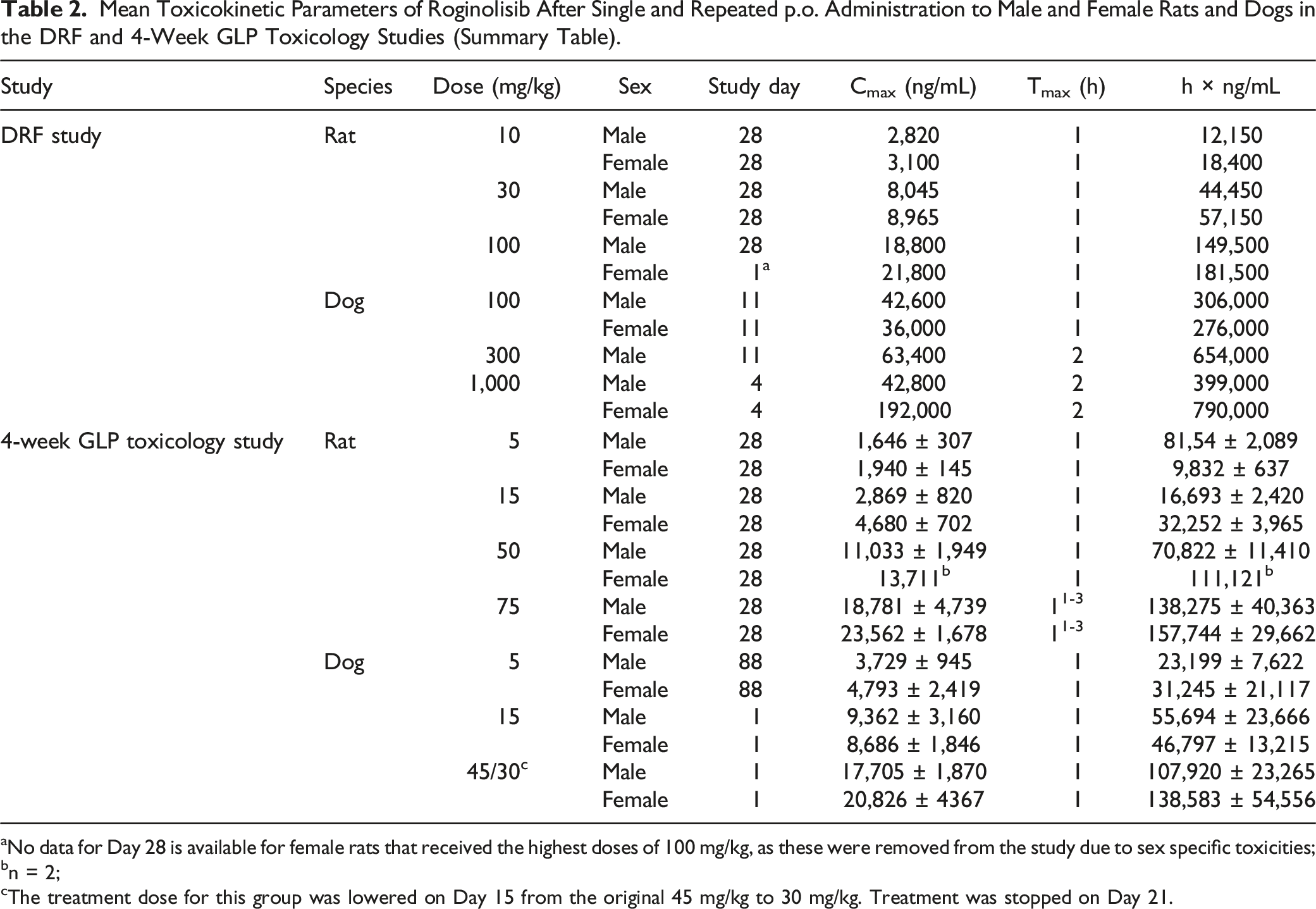
aNo data for Day 28 is available for female rats that received the highest doses of 100 mg/kg, as these were removed from the study due to sex specific toxicities;
bn = 2;
cThe treatment dose for this group was lowered on Day 15 from the original 45 mg/kg to 30 mg/kg. Treatment was stopped on Day 21.
Mortality and Clinical Symptoms
Mortality was seen in 4 of 5 female rats that received 100 mg/kg between Days 7 and 9 as well as 1 female that was prematurely sacrificed on Day 9. In contrast, all male rats treated with the same dose survived. No toxicokinetic samples were obtained from the diseased animals to enable correlation of mortality with compound exposure.
Female rats did not tolerate the highest tested dose of 100 mg/kg and showed clinical signs including incomplete/complete eyelid closure, pallor, lowered predose body temperature, piloerection, diarrhea, and reduced spontaneous activity.
In dogs, the female that received the highest tested dose of 1000 mg/kg was euthanized as a consequence of severe clinical symptoms on Day 4. The main clinical signs observed in at least one of the dogs that received a dose of ≥300 mg/kg included vomiting, stool changes, salivation, hyperesthesia, tremor, hypoactivity, and genital discharge. In addition, increased body temperature was seen in the male treated at 300 mg/kg and the female treated at 100 mg/kg on some days.
Body Weight and Food Consumption
In the DRF study, male rats administered 100 mg/kg showed a decrease in relative body weight gain of 16% on Day 28 compared with that of control male rats. Male rats exhibited slight changes in food consumption, but it was without any dose-dependency and therefore not treatment-related. However, in female rats treated with 100 mg/kg, a treatment-related decrease of 18% in food consumption was observed on Day 7.
In dogs, body weight was only slightly reduced compared to the pre-treatment values (range: −2.9 to −7.2%). Food consumption in all dogs was impaired, that is, marked reduction or no food intake during the treatment period.
Hematology
Immunophenotyping of Rats Treated with Roginolisib.
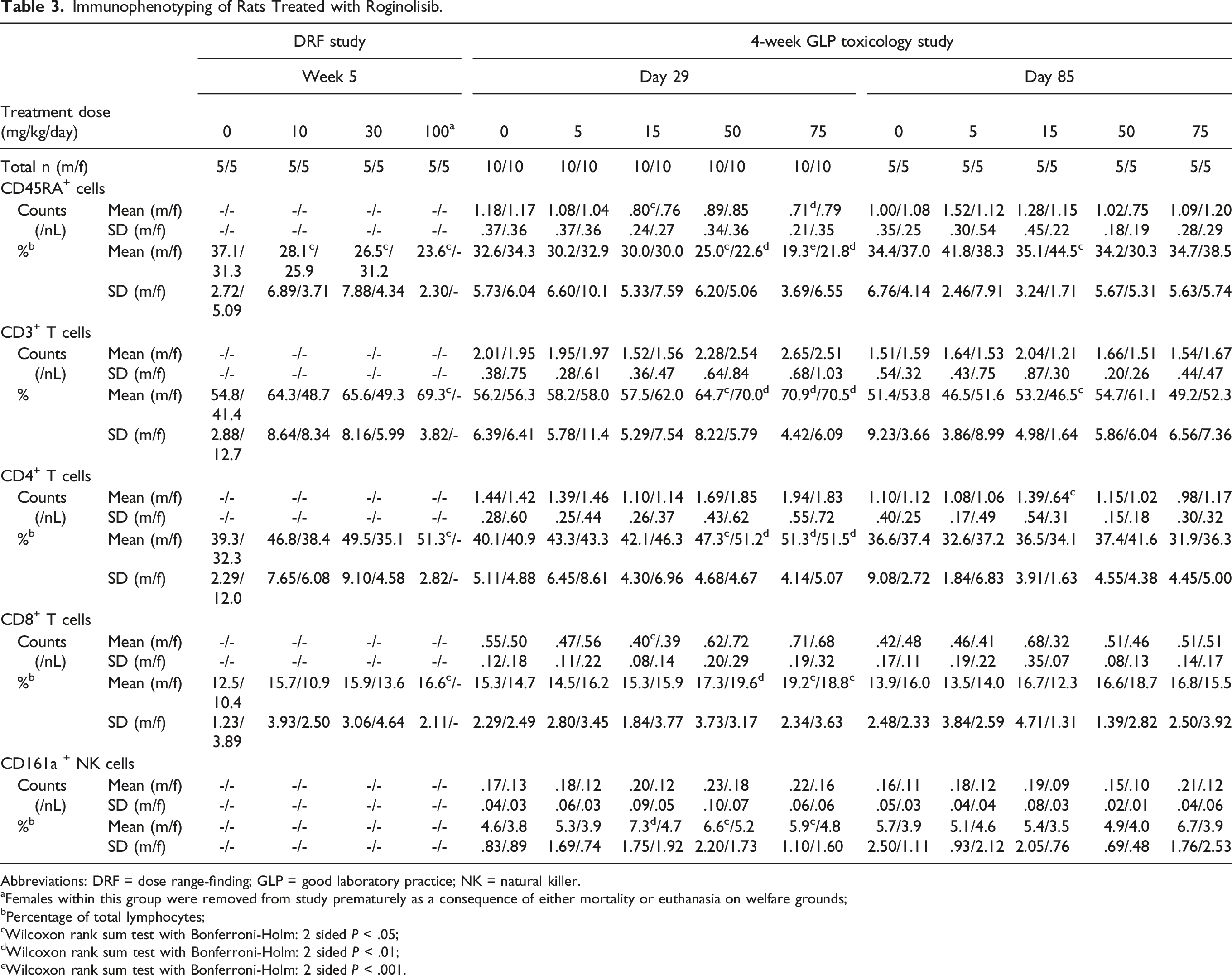
Abbreviations: DRF = dose range-finding; GLP = good laboratory practice; NK = natural killer.
aFemales within this group were removed from study prematurely as a consequence of either mortality or euthanasia on welfare grounds;
bPercentage of total lymphocytes;
cWilcoxon rank sum test with Bonferroni-Holm: 2 sided P < .05;
dWilcoxon rank sum test with Bonferroni-Holm: 2 sided P < .01;
eWilcoxon rank sum test with Bonferroni-Holm: 2 sided P < .001.
In the dog DRF study, the female dog that received the escalating dose had an increase in red blood cells at Day 4 (9.76/pL) compared with Day 5 (7.85/pL), as well as elevated hemoglobin levels (22.3 g/dL at Day 4 compared with 17.6 g/dL at Day 5). Immunophenotyping, including T cell subsets, did not indicate a global immune deficiency.
Clinical Chemistry
Clinical Chemistry Parameters in Rat Toxicology Studies Showing Significant Treatment-Related Changes Compared to Control.
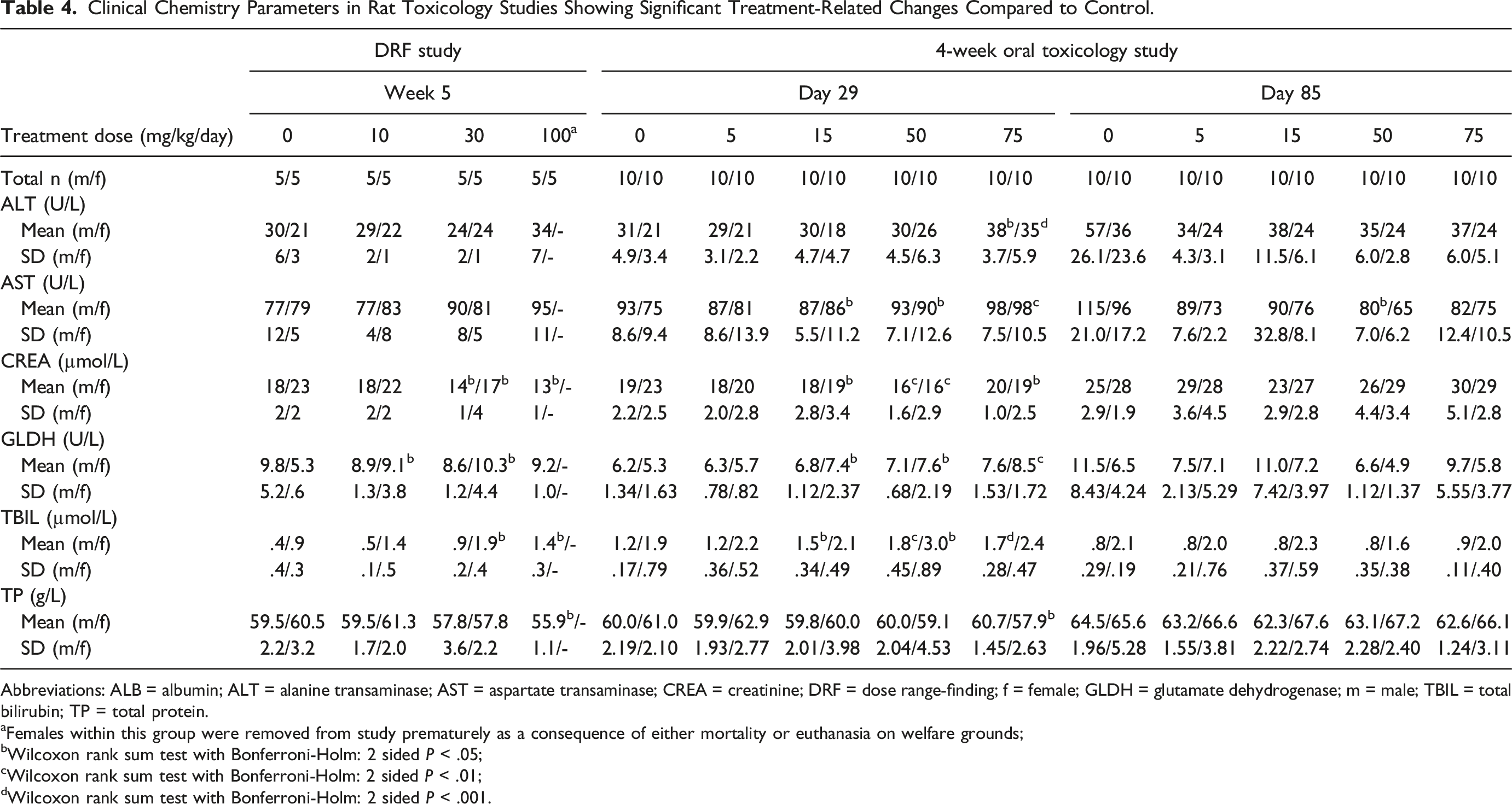
Abbreviations: ALB = albumin; ALT = alanine transaminase; AST = aspartate transaminase; CREA = creatinine; DRF = dose range-finding; f = female; GLDH = glutamate dehydrogenase; m = male; TBIL = total bilirubin; TP = total protein.
aFemales within this group were removed from study prematurely as a consequence of either mortality or euthanasia on welfare grounds;
bWilcoxon rank sum test with Bonferroni-Holm: 2 sided P < .05;
cWilcoxon rank sum test with Bonferroni-Holm: 2 sided P < .01;
dWilcoxon rank sum test with Bonferroni-Holm: 2 sided P < .001.
Clinical Chemistry Parameters in Dog Toxicology Studies Showing Treatment-Related Changes Compared to Control.
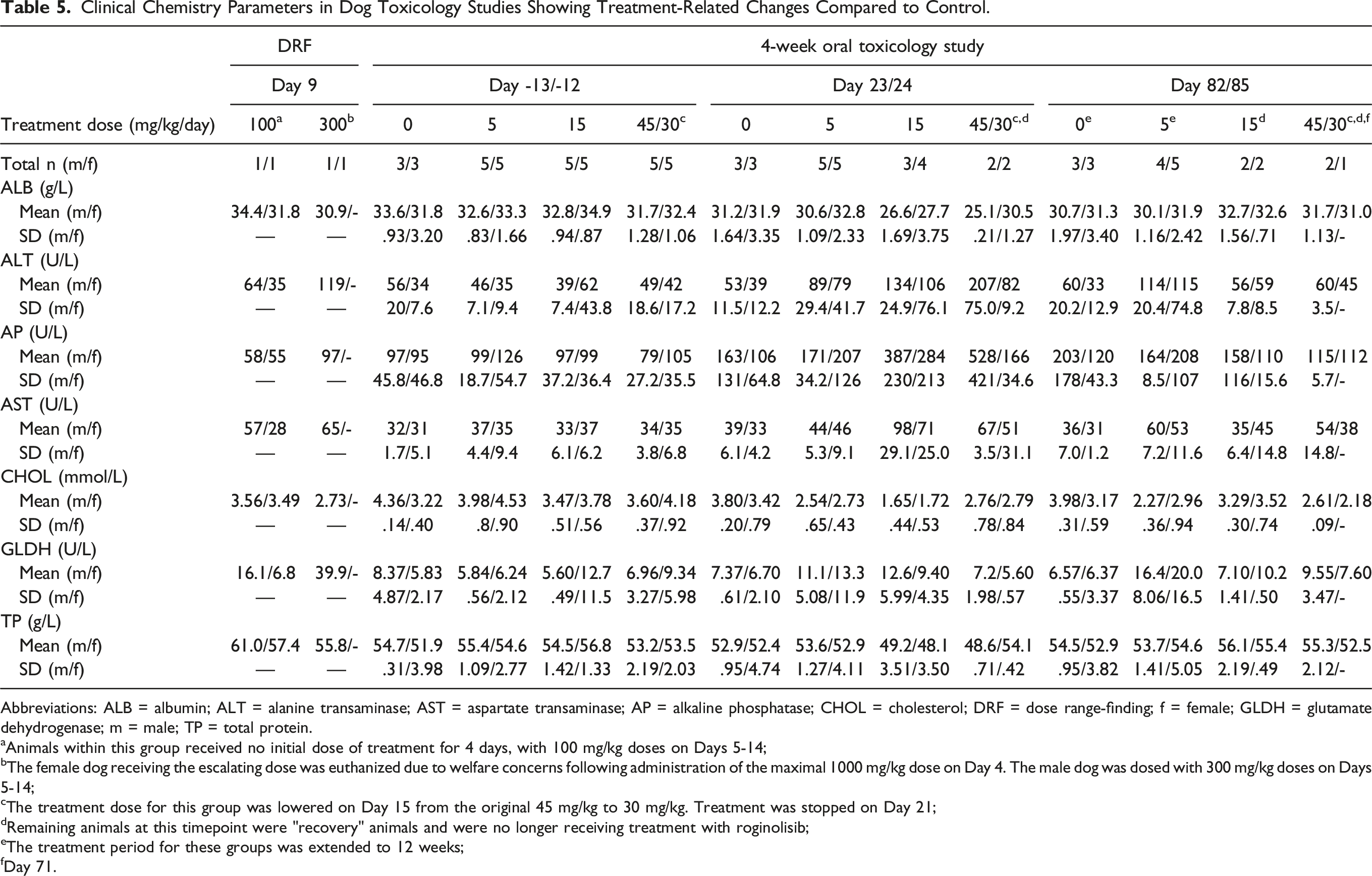
Abbreviations: ALB = albumin; ALT = alanine transaminase; AST = aspartate transaminase; AP = alkaline phosphatase; CHOL = cholesterol; DRF = dose range-finding; f = female; GLDH = glutamate dehydrogenase; m = male; TP = total protein.
aAnimals within this group received no initial dose of treatment for 4 days, with 100 mg/kg doses on Days 5-14;
bThe female dog receiving the escalating dose was euthanized due to welfare concerns following administration of the maximal 1000 mg/kg dose on Day 4. The male dog was dosed with 300 mg/kg doses on Days 5-14;
cThe treatment dose for this group was lowered on Day 15 from the original 45 mg/kg to 30 mg/kg. Treatment was stopped on Day 21;
dRemaining animals at this timepoint were "recovery" animals and were no longer receiving treatment with roginolisib;
eThe treatment period for these groups was extended to 12 weeks;
fDay 71.
Pathology
In the rat DRF study, gross pathology, and organ weights of all animals were inconspicuous. Histopathology of the deceased females (100 mg/kg) revealed depletion of the lymphatic system sometimes combined with cell necrosis, ulceration of the forestomach, necrosis of the intestinal mucosa, and skin hyperkeratosis. In female rats treated with 10 and 30 mg/kg as well as in male rats treated with 10, 30, and 100 mg/kg, morphological alterations were mainly restricted to the B-cell compartments of lymph nodes and the spleen, demonstrating a decrease of germinal center formation and a depletion of the marginal zone. The observed histopathological findings including the respective severity grade are given in the form of a heatmap in Figure 1 (increasing color intensity in red matches the degree in severity for males represented in upper triangle, females in lower triangle). Heat map shows the histopathological findings including the severity grades of sites of toxicity in rats in a 4-week DRF study. Females that received 100 mg/kg of roginolisib resulted in premature mortality due to either clinical symptoms or euthanasia due to welfare concerns on Days 7-9. Male findings are represented in the upper colored triangles and female findings in the lower colored triangles. Coloring is matched with the severity grade assigned to the finding with increasing intensity of red being associated with severe grade toxicity.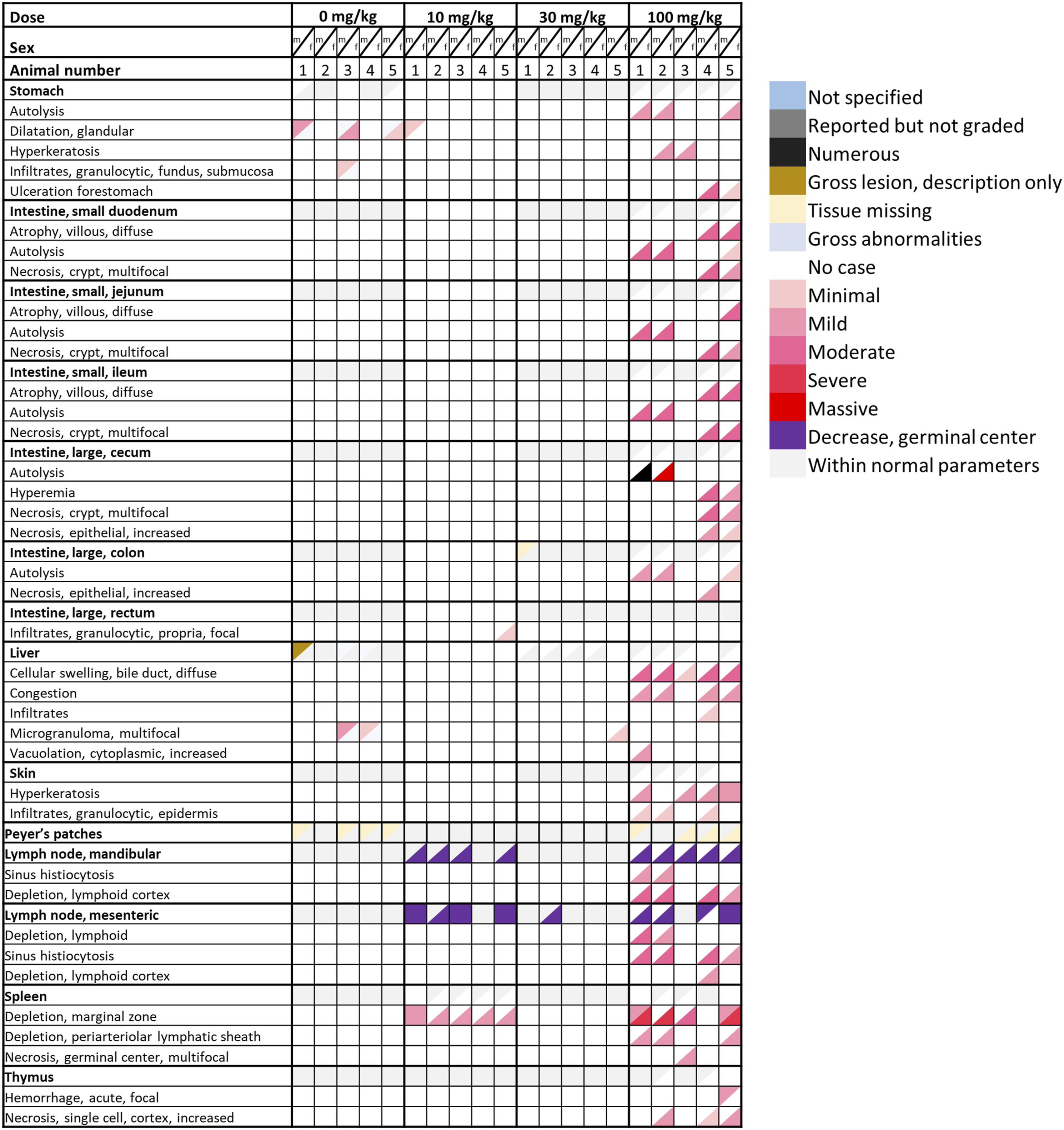
Gross pathology revealed a red and white discoloration of the intestinal mucosa in the female dog at 100 mg/kg. The female dog that was euthanized prematurely at 1000 mg/kg showed a completely empty lumen of the small and large intestine. Histopathology revealed a massive lymphoid depletion in all lymphatic tissues, in particular the thymus and spleen. Beside the pronounced pharmacodynamically mediated atrophy of the lymphatic system, secondary focal alterations were observed in the liver in almost all dogs (small necroses and microgranuloma). Single observations were additionally noted in the stomach (hypersecretion), intestine (hyperemia), adrenals (hemorrhage), bone marrow (necrosis), and prostate (inflammation).
Pivotal Oral Repeat Dose Toxicology Studies in Rats and Dogs
Toxicokinetic Investigations
In rats, Cmax and AUC0-24h increased approximately dose proportional and concentrations of roginolisib in the blood plasma were similar between repeated doses, suggesting effective clearance of the compound from the body (see Table 2). In general, females were slightly more exposed than males on both sampling occasions, the male/female AUC0-24h ratios being .6-.8 on Day 1 and .5-.9 on Day 28. A moderate accumulation was determined after repeated dosing at the higher dose levels, that is, the Rac AUC0-24h ranged from .9 at 15 mg/kg to 1.4 at 75 mg/kg in male rats and from 1.2 at 15 mg/kg to 1.3 at 75 mg/kg in female rats on Day 28.
In dogs, Cmax and AUC0-24h showed an approximately dose proportional increase in males and a less than dose proportional increase in females (see Table 2). A moderate accumulation was determined after repeated dosing, that is, the Rac AUC0-24h ranged from 1.5 at 5 mg/kg to 2.2 at 15 mg/kg in male dogs and from 1.3 at 5 mg/kg to 1.6 at 15 mg/kg in female dogs on Day 25.
Mortality and Clinical Signs
In contrast to the rat DRF study, no treatment-related mortality was detected in rats treated with 5, 15, 50, and 75 mg/kg in the 4-week oral toxicology study. Furthermore, no treatment-related clinical signs were observed in rats treated with doses of ≤50 mg/kg. Only short and transient episodes of salivation (<5 minutes) were seen in 8 rats receiving 75 mg/kg.
In the pivotal toxicology study in dogs, premature euthanasia was performed in dogs at 5 mg/kg (1 dog, Day 65), 15 mg/kg (2 dogs, Day 19), and 45/30 mg/kg (1 dog, Day 12; 4 dogs, Day 16; 1 dog, Day 36). In addition, mortality was seen in 1 dog at 15 mg/kg during the premature euthanasia on Day 15 and 1 dog at 45/30 mg/kg on Day 16. Dogs receiving 5 mg/kg had shown very few clinical symptoms up to Day 25, and therefore, it was decided to extend the treatment of both that dose group and the control group to 12 weeks.
At 5 to 45/30 mg/kg, epithelial lesions of the skin (whole body including paws) and mucosa as well as discharge from the eyes and genitals (all males, occurring around Days 9-11) were seen during the treatment period and/or recovery period (especially paw lesions). In addition, at 15 and 45/30 mg/kg gastrointestinal symptoms like vomiting, soft stool with yellowish-mucous admixtures, soft stool with bloody admixtures, and diarrhea as well as bloody urine, salivation, hypothermia, photophobia, tremor, hypoactivity, apathy, and lateral and prone positions were detected during the treatment period.
Body Weight and Food Consumption
In the pivotal toxicology study in rats, body weight did not differ between treatment and control males at the end of the treatment and recovery period. In females, body weight was slightly lower in group 4 (50 mg/kg, statistically significant) and group 5 (75 mg/kg) animals compared to control by 4.9 and 3.6%, respectively, on Day 28. The body weight gain was therefore slightly lower in these animals. No treatment-related changes of food consumption during the treatment or recovery period were observed in either sex.
In dogs at 15 mg/kg (1 female) and 45/30 mg/kg (2 females), significantly reduced body weight during the treatment period was related to bad clinical condition. All other roginolisib treated dogs showed only minor changes in bodyweight. Short incidences (1-4 days) of reduced or no food consumption were observed in some dogs of either sex that received ≥15 mg/kg.
Electrocardiography, Arterial Blood Pressure, and Reflex Tests
In dogs, the ECG parameters PQ-, QRS-, and QT-interval as well as heart rate showed no relevant change within or among the groups during the treatment period. In addition, the heart rate-corrected QT-intervals (according to Van de Water) did not change relevantly during treatment within 1 group, and no relevant differences between the groups were observed. No treatment-related effect was seen on mean-, systolic-, and diastolic arterial blood pressure. Furthermore, there were no changes in reflex tests which are considered to be related to the treatment.
Ophthalmology
In rats, ophthalmological examinations including pupillary reflex, and the examination of anterior and posterior eye segment did not reveal any treatment-related effects. However, in dogs a corneal erosion (ulcer) was seen in 1 animal at 5 mg/kg and central vitreous floater, hyper-reflectivity, and foci formation in the tapetal-non tapetal junction in a dog at 45/30 mg/kg in week 10. These findings were considered likely related to a spontaneous event (such as housing conditions or animal behavior) and not to roginolisib.
Hematology Parameters
A dose-dependent increase of neutrophils was observed in both male and female rats, accompanied by a decrease of relative lymphocyte count. Differentiation of lymphocyte populations by immunophenotyping identified a clear decrease of relative B-cell count (males: 75 mg/kg = 19.3%; control = 32.6%, P < .001; females: 75 mg/kg = 21.8%, control = 34.3%; P < .01) and an increase of relative T cells (males: 75 mg/kg: = 70.9%, control = 56.2%; P < .01; females 75 mg/kg = 70.5%, control = 56.3%; P < .01) consisting of T-helper and T-suppressor cells in both sexes at 50 and 75 mg/kg. The effects proved to be reversible. Data for immunophenotyping in rats is reported in Table 3.
Male and female dogs treated with 5 to 45/30 mg/kg showed a non-dose-dependent increase in platelet counts on Day 23/24. For example, the pre-test vs Day 23/24 platelet count in the 45/30 mg/kg group increased from 324 to 440/nL in male dogs, and from 290 to 473/nL in female dogs. Furthermore, an increase in WBC, mainly due to an increase in neutrophils, was seen at 15 mg/kg (WBC in males = 8.84/nL at pre-test vs 15.45/nL on Day 23; WBC in females = 9.16/nL at pre-test vs 17.24/nL on Day 24) and at 45/30 mg/kg (WBC in males = 10.15 at pre-test vs 20.45/nL on Day 23; WBC in females = 8.71/nL at pre-test vs 17.24/nL on Day 24). At the highest dose of 45/30 mg/kg males additionally showed a slight reduction in red blood cell counts, hemoglobin and hematocrit. Females showed slight decreased hemoglobin values only, and a concurrent slight increase of reticulocytes. Thrombin and prothrombin time were slightly prolonged in males as well. Box plots of platelet, WBC, neutrophil, and lymphocyte counts are shown in Figure 2. Observed hematology and clinical chemistry changes in 4-week dog toxicity study. Graphs show combined data for male and female animals. Data is presented as boxes with whiskers for min to max values. WBC = white blood cells; PLT = platelets; NEU = neutrophils; LYMPH = lymphocytes; ALAT = alanine aminotransferase; ASAT = aspartate aminotransferase; AP = alkaline phosphatase; GLDH = glutamate dehydrogenase; TP = total protein; ALB = albumin; CHOL = cholesterol.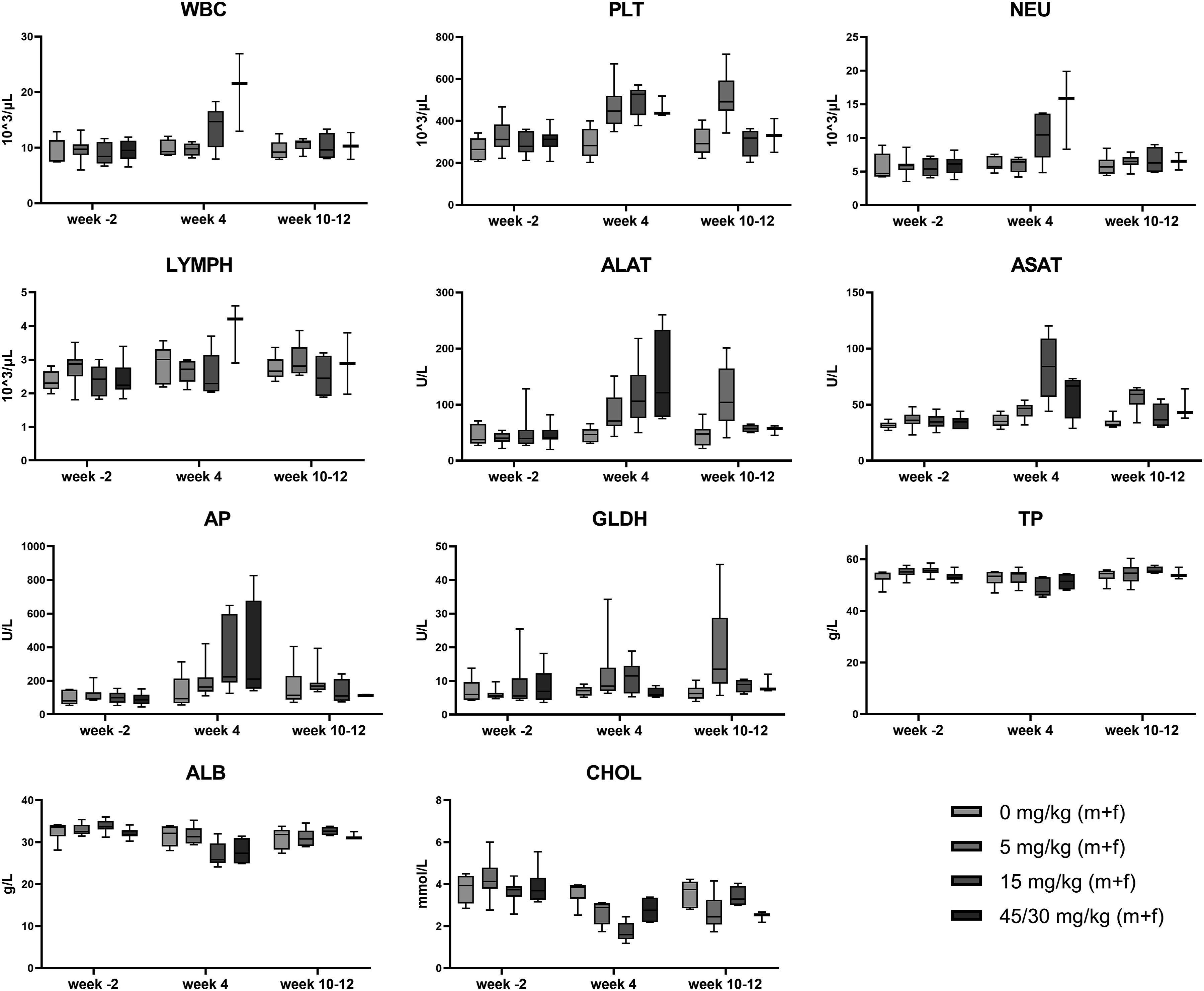
Immunophenotyping of Dogs Treated with Roginolisib.
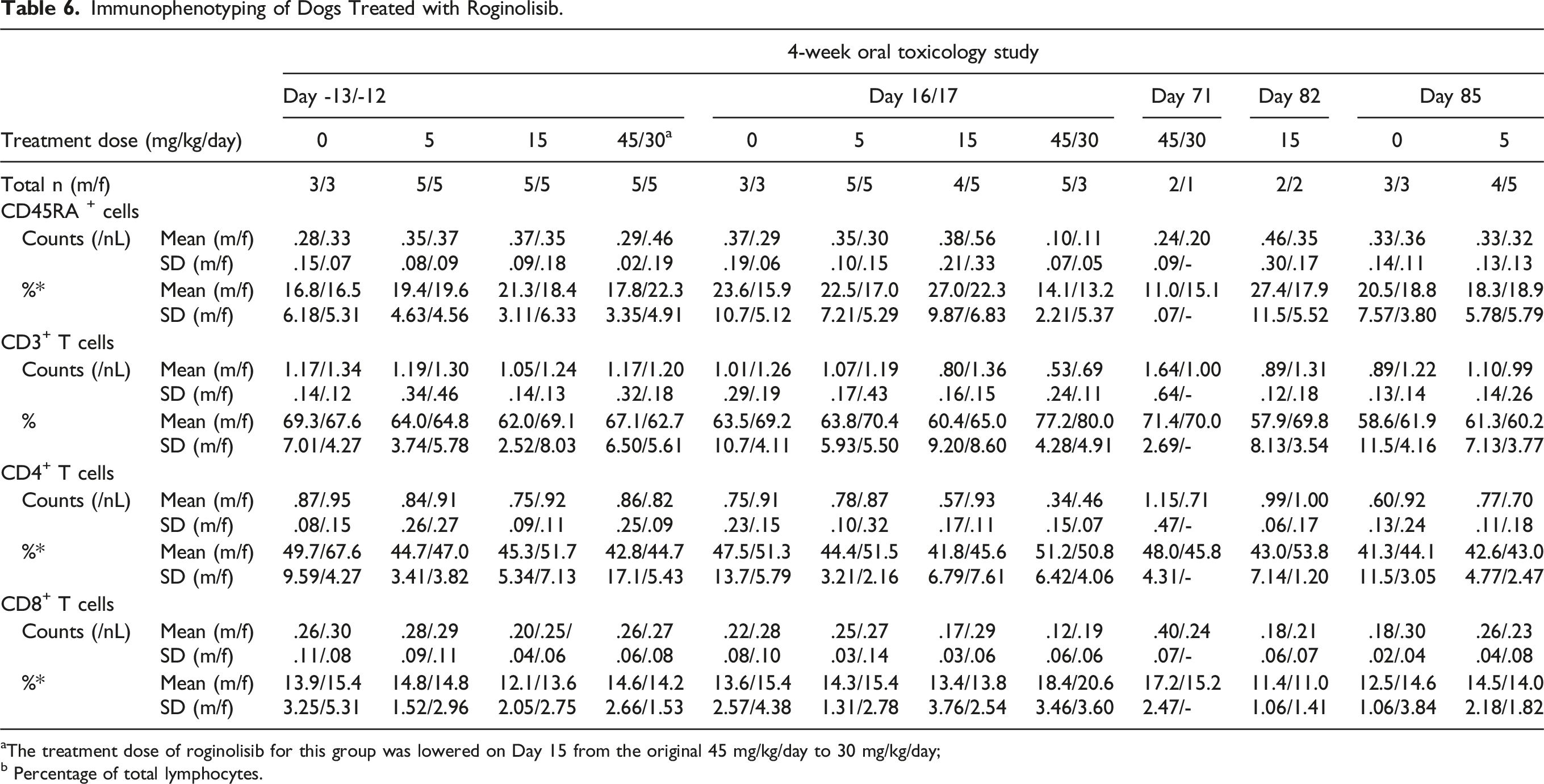
aThe treatment dose of roginolisib for this group was lowered on Day 15 from the original 45 mg/kg/day to 30 mg/kg/day;
b Percentage of total lymphocytes.
Clinical Chemistry
Only a slight dose-dependent and reversible increase in ALT, AST (females only), GLDH, TBIL, and TP values as well as a slight dose-dependent decrease in CREA were seen in male and female rats (see Table 2). However, within the pivotal toxicology study in dogs treated with 5 to 45/30 mg/kg, a mild to severe dose-dependent increase in the liver enzymes ALT, AST, AP, and GLDH as well as a slight to moderate decrease in the liver substrates CHOL, ALB, and TP were observed (see Table 5 and Figure 2). These findings indicate an impairment of liver cell function and liver cell damage; however, the effects proved to be reversible in the recovery animals.
Pharmacodynamic Biomarker
In rats, pAkt was inhibited by roginolisib at all dose levels on Days 5, 20, and 28 (see Figure 3). On Day 5, the range of inhibition was variable especially in female rats (between 15% and 73%) and no clear dose response could be observed. The pAkt inhibition was more consistent on Day 20 with approximately 65% of inhibition at the dose levels of 5 and 15 mg/kg and approximately 80% at the dose levels of 50 and 75 mg/kg. On Day 28 pAkt was inhibited >95% at all dose levels. Bar graph shows inhibition of pAkt in whole blood rat B cells 1 hour after treatment with roginolisib.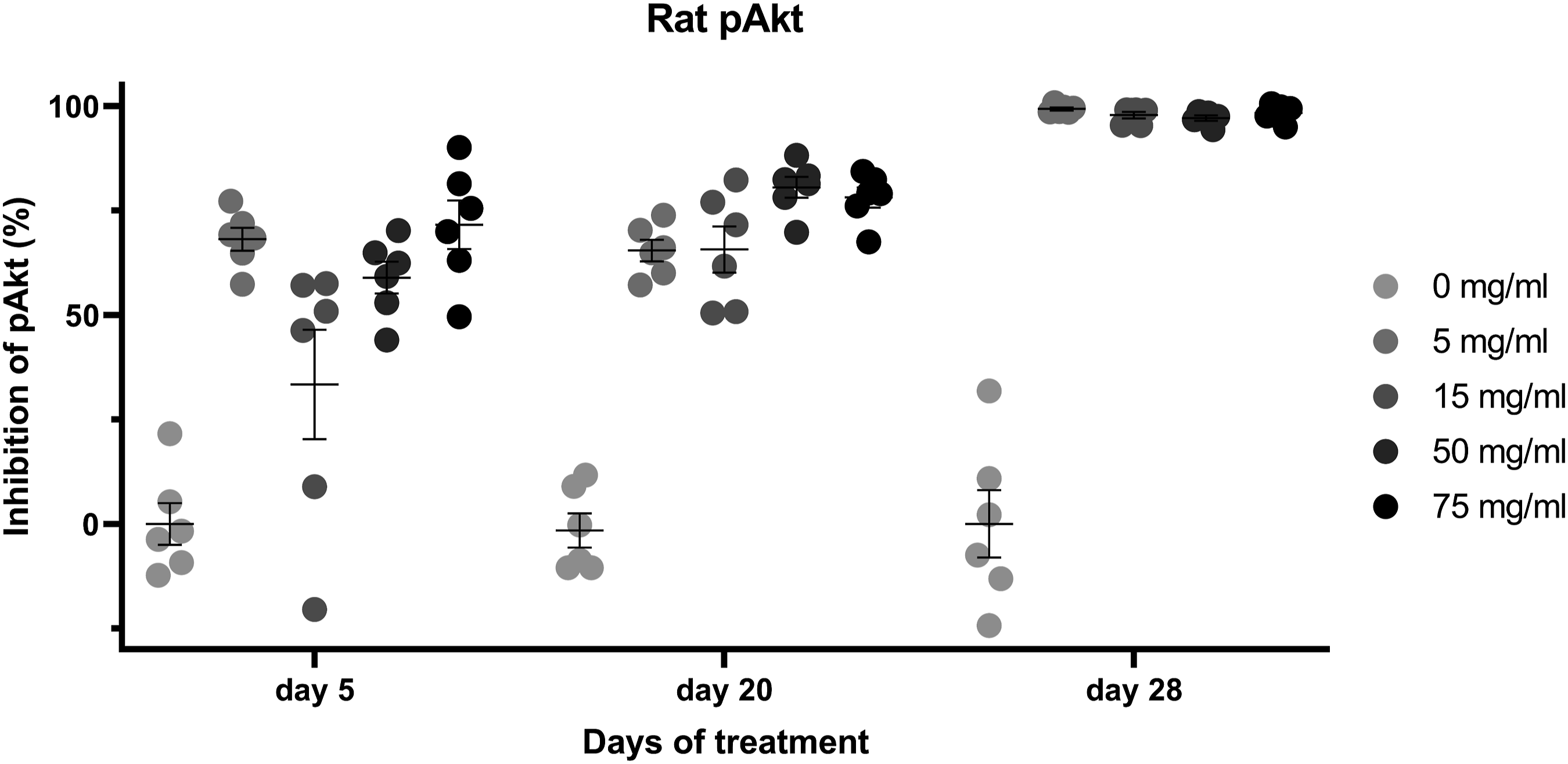
Pathology
In the pivotal toxicology study in rats, gross pathology and organ weight determination revealed several findings that were considered to be spontaneous. The spleen weight in females at 50 and 75 mg/kg were decreased, which might be caused by depletion of marginal zone cells.
Histopathology showed treatment-related effects in lymph nodes and spleen. The mandibular lymph node showed a decrease of germinal centers in some rats at 50 mg/kg and in most rats at 75 mg/kg. The doses of 5 and 15 mg/kg were not affected. In the mesenteric lymph node, the decrease of germinal centers was more prominent as almost all rats at 50 and 75 mg/kg were affected and additionally 2 females at 15 mg/kg. In the spleen, a minimal to moderate depletion of the marginal zone was seen at 50 and 75 mg/kg, and a minimal depletion in few males and females at 15 mg/kg and in 1 male already at 5 mg/kg. At ≤75 mg/kg doses of roginolisib in rats, only isolated abnormalities were found in the liver, with multi-focal microgranuloma of a minimal severity being the most frequent observed finding. All other organs were congruous with the controls. At the end of the 8-week recovery period no relevant histopathology findings were observed in rats. The observed histopathological findings in rats including the respective severity grade are given in the form of a heatmap in Figure 4. Heat map shows the histopathological findings including the severity grades in the liver and hematopoietic and lymphoid tissues of rats in a 4-week oral toxicology study. No histopathological findings were present in the digestive system or skin of any of the rats in this study. Male findings are represented in the upper colored triangles and female findings in the lower colored triangles. Coloring (i.e., increasing intensity of red) is matched with the severity grade assigned to the finding.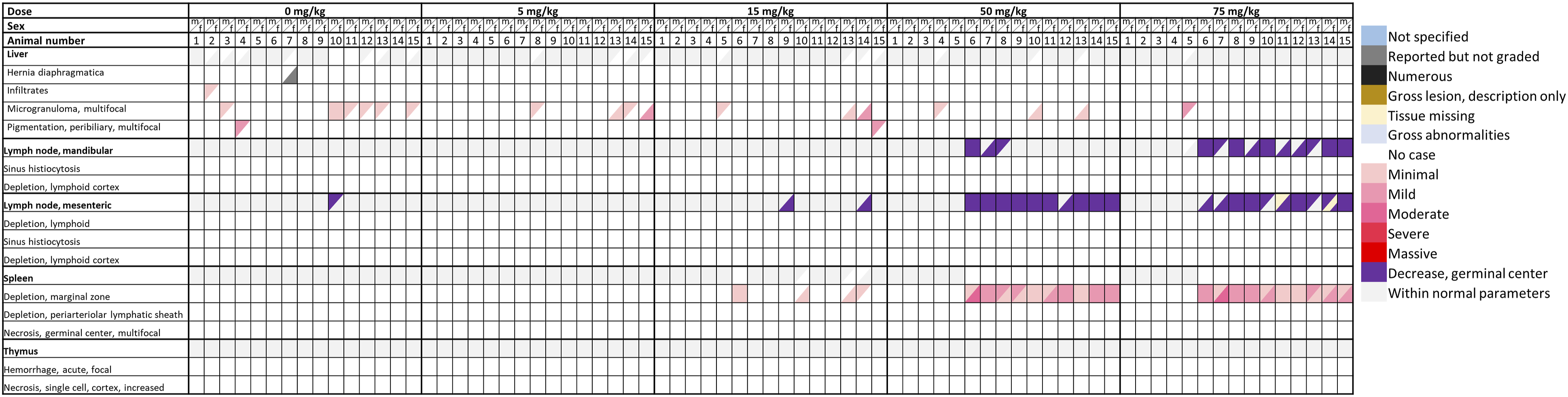
The pivotal toxicology study in dogs found gross pathological changes in the skin at 5 mg/kg and in the intestinal tract at 15 and 45/30 mg/kg. In addition, no organ weight changes were observed. Histopathology revealed pharmacologically mediated alterations in the lymphatic system at all dose levels. Administration of 5 mg/kg led to a minimal to mild and 15 and 45/30 mg/kg to a moderate to marked depletion of lymphocytes in spleen, thymus, lymph node and lymph follicles of the intestine. At 15 and 45/30 mg/kg, moderate bone marrow depletion, moderate to marked necrosis of the intestinal mucosa, focal necrosis of the gall bladder mucosa, mild to marked liver cell necrosis as well as moderate depletion of the spermatogenic epithelium of the testes were seen. The recovery animals did not exhibit any conspicuous findings except for 1 recovery female at 45/30 mg/kg which developed a dysplastic alteration in the skin of the hind paws within 20 days after treatment. The observed histopathological findings in dogs including the respective severity grade are given in the form of a heatmap in Figure 5. Heat map shows the nature and severity of histopathological findings in the digestive system, liver, skin, and hematopoietic and lymphoid tissues of dogs during a 12-week oral toxicology study. Male findings are represented in the upper colored triangles and female findings in the lower colored triangles. Coloring (i.e., increasing intensity of red) is matched with the severity grade assigned to the finding. aThe treatment dose for this group was lowered on Day 15 from the original 45 mg/kg to 30 mg/kg; bThese animals were "recovery" animals and were investigated after an 8-week, treatment-free recovery period; cThis animal died/was prematurely euthanized.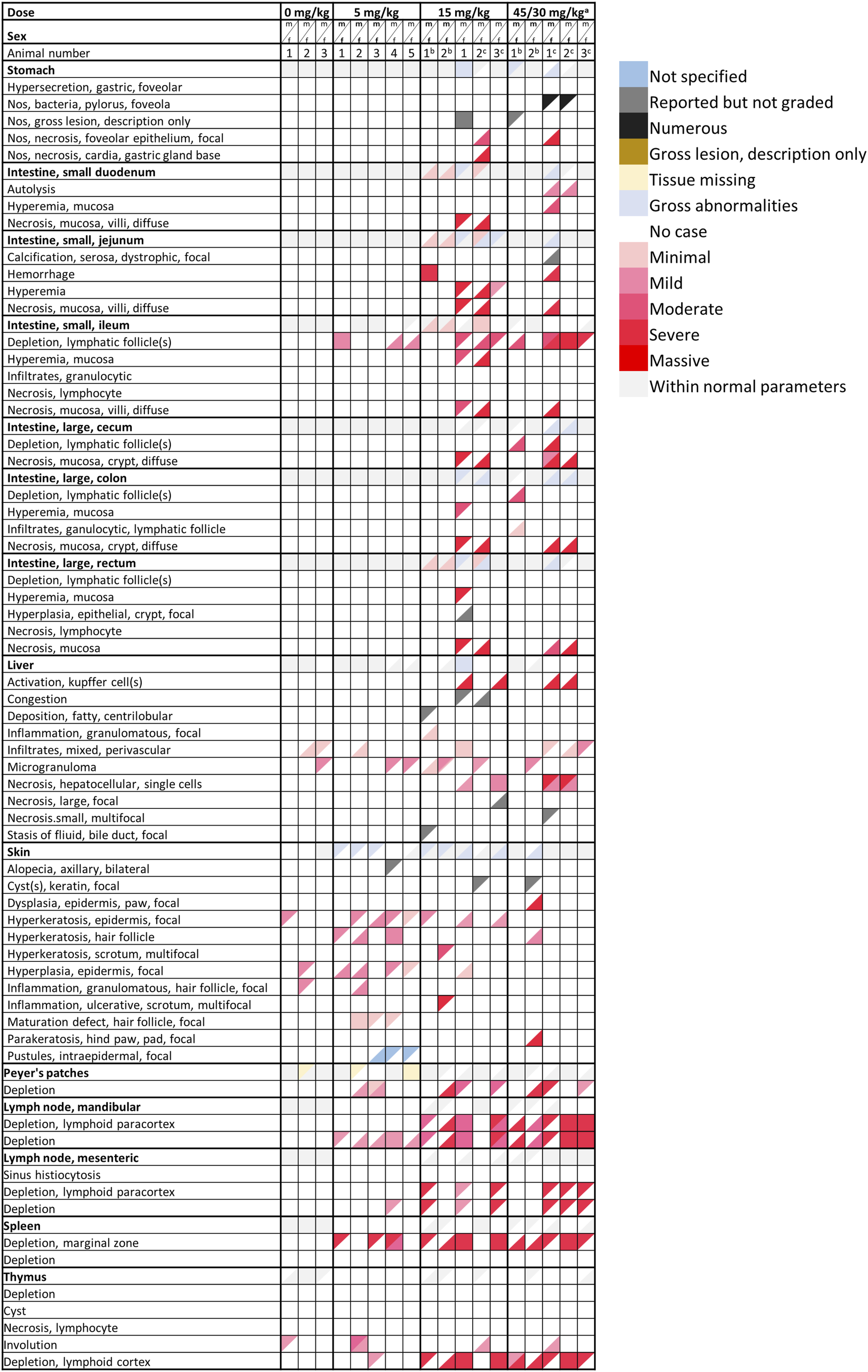
Overall, in the pivotal oral toxicology study in rats, roginolisib was clinically tolerated and the no observed adverse effect level (NOAEL) was determined at 15 mg/kg. However, in the pivotal toxicology study in dogs, roginolisib was not tolerated due to severe clinical symptoms, that is, mainly epithelial lesions of the skin at 5 mg/kg and necrotizing damage of the intestinal epithelia at 15 and 45/30 mg/kg, which were dose limiting and lethal. A NOAEL could be not established in dogs.
Discussion
Here, we report the non-clinical DRF and 4-week pivotal toxicology studies of roginolisib, a selective non-competitive PI3Kδ inhibitor. 19 The level of off-target inhibition, particularly driven by levels of unbound drug within the body, appears to be a key driver of roginolisib’s toxicity profile in rats and dogs. Hence, from being a well-tolerated, specific drug, roginolisib may become a poorly tolerated unspecific drug should free drug levels exceed the threshold for selective PI3Kδ inhibition.
The aim of the DRF studies was to evaluate the tolerability and the toxicity of roginolisib and to identify target organs after repeated daily dosing. In addition, the results were used to establish the doses for the 4-week pivotal studies. The aim of the latter studies was to define the starting dose for clinical development based on a no-observed-adverse-effect-level (NOAEL). The oral route was chosen because roginolisib is intended to be given orally in clinical studies, and the rats and dogs were used as species as their metabolic profiles match with that of human (data not reported).
Based on the non-clinical toxicity profile of idelalisib27,28 and the expression of PI3Kδ in human tissue, 29 the target organs for the expected toxicity were the hematopoietic and lymphoid systems, gastrointestinal tract and pancreas, lungs, heart, testes, and liver. The expected toxicities 30 include lymphoid depletion, reduced weight of spleen and thymus, thymic hemorrhage and necrosis, myeloid and granulopoietic hyperplasia (hematopoietic and lymphoid systems), inflammation, infiltration, hemorrhage and ulceration (gastrointestinal tract), increased liver enzymes, increased liver weight, inflammation, and hepatocellular necrosis (liver), myocardium infiltrate, fibrosis (heart; rat hearts were also expected to have increased weight), erythema, dryness, swelling, and redness (skin), and testicular seminiferous tubule degeneration and reduced testicular weight (male reproductive tissues).
Overall, the findings in the DRF studies and the 4-week pivotal studies were similar. Consistent with the expected toxicities, roginolisib, besides depletion of the lymphoid system, predominantly caused hepatotoxicities and gastrointestinal toxicities, especially at the mid and high doses in dogs and at the 100 mg/kg dose in female rats. These toxicities were also associated with poor clinical condition of the animals, decreases in body weight gain and food consumption, changes in liver enzymes, and mortality. The mortality observed in female rats at the highest dose level of 100 mg/kg may be explained by a disproportional increase in exposure due to compound accumulation. The gastrointestinal toxicities without a histopathological correlation were likely the result of gluconeogenesis as a result of either poor clinical condition, inhibition of the PI3Kα isoform or both. Contrary to idelalisib, however, no lung and heart toxicities were observed and testicular toxicities were only observed in dogs, not in rats. Importantly, in rats and dogs treated with roginolisib that were assessed at the end of the recovery period, histopathology and clinical pathology investigations were similar to control animals (rats) or animals treated with vehicle alone at Day 85 (dogs). These observations imply that the effects of roginolisib were reversible with no sequelae.
The degree of loss of lymphocytes in all lymphatic tissues observed in all studies, deemed to be related to inhibition of PI3Kδ, was mild at the low dose level and moderate-to-severe at the mid and high dose levels. Lymphoid depletion was most prominent in the B-cell-dependent regions with an almost complete depletion of lymphocytes in the B-cell-dependent marginal zone of the spleen at all dose levels in male dogs. Expectedly, B cells and some classes of T cells were also reduced in blood in response to PI3Kδ inhibition in rats and dogs in the 4-week studies. Other hematology parameters were also affected, with a dose-dependent increase observed in platelet and neutrophil counts in dogs. These changes did not progress over time beyond the treatment period, suggesting that roginolisib does not induce thrombocytopenia or neutropenia, 2 common side effects observed in patients receiving treatment with other PI3K inhibitors (e.g., copanlisib).30,31 Other significant hematological toxicities such as reduction of reticulocytes, monocytes, or eosinophils were also not observed. Contrary to the 4-week studies, the DRF study in dog did not reveal any changes in B and T cells, likely due to the shorter treatment duration in this study. Future animal toxicology studies may be designed to include a comprehensive immunophenotyping panel, including T regulatory cells, memory and exhausted CD8 T cells. Such immunophenotyping will help to inform clinical development on the need of using appropriate monitoring of immune-related adverse events.
The dermatological manifestations in dogs (e.g., hyperkeratosis, epidermal-hyperplasia, erythema, and maturity-onset alopecia), appearing a month after roginolisib treatment at 5 mg/kg, were considered PI3Kδ related. Dysregulation of PI3Kδ may have affected the maintenance of keratinocyte proliferation and migration as the PI3K/AKT signaling plays a role in maintaining skin-derived precursors in vitro 32 ; moreover, PI3Kδ specifically has been shown to be expressed by hyperproliferating keratinocytes in psoriatic plaques and implicated in the psoriatic inflammatory infiltrate. 33
Strong inhibition of the pharmacodynamic biomarker pAkt occurred in rats treated with 5 to 75 mg/kg 1 hour after dosing. There was a trend towards higher inhibition at higher dose levels and longer treatment duration. At Day 28, almost complete inhibition of pAkt was observed for all dose levels, indicating that the rat is a relevant species for the toxicological characterization of roginolisib. The therapeutically active dose level is ≥ 5 mg/kg, similar to the results observed in a rat collagen-induced arthritis (CIA) model (unpublished data). The efficacy data from the CIA model were subsequently confirmed in mouse models of cancer. 19
Toxicokinetics analysis showed that at 5 mg/kg, roginolisib free plasma levels exceeded the cellular target engagement IC90 for PI3Kδ (10 ×IC50 value of 10 ng/mL
19
) for ≥ 12 hours (Figure 6). In rats the dose of 5 mg/kg was well tolerated. In dogs, no NOAEL was established; however, the observed adverse effects at the 5 mg/kg dose level were considered monitorable and reversible. At the dose levels of ≥ 15 mg/kg free plasma levels exceeded the IC90 for PI3Kδ up to the complete dosing period and the IC50 for PI3Kβ for ≥ 12 hours (IC50 value of 226 ng/mL
19
). Additionally, the free Cmax approached the IC50 for PI3Kα (IC50 value of 5.3 µg/mL
19
). The dose level of 15 mg/kg was well tolerated in rats and defined as the NOAEL. By contrast, in dogs this dose level was associated with high mortality and severe side effects. Dose levels ≥ 45 mg/kg resulted in free plasma levels exceeding the IC50 for PI3Kα (>3 fold in both rat and dog at 100 mg/kg) and were associated with increasing toxicity in both species. These results indicate that roginolisib’s toxicities in rats and dogs are not driven exclusively by the level of PI3Kδ inhibition. Aggravated toxicity is observed, especially in dogs, at dose levels projected to also inhibit PI3Kβ and especially PI3Kα. Data from these and other studies indicates a complex association between isoform selectivity and the safety profiles of PI3K inhibitors. For example, a quantitative systems pharmacology model-based study concluded that co-inhibition of the PI3Kα isoform as a consequence of prolonged PI3Kδ inhibition exacerbates colitis in humans.
34
Similar to this observation in humans, unbound plasma levels of roginolisib at or in excess of the IC50 for PI3Kα resulted in gastrointestinal toxicities in dogs. Graphs show the free concentration of roginolisib (ng/mL) in plasma at each measured timepoint following daily administration, as calculated for rat A and dog B at escalating doses (5, 15, 50, and 75 mg/kg for rat and 5, 15, and 45/30 mg/kg for dog) based on plasma protein binding data (24% for rat and 56% for dog). IC50 thresholds for PI3Kα, PI3Kß, and PI3Kδ and IC90 threshold PI3Kδ, based on intracellular target engagement measured in Jurkat cell lysate (Kinativ®), are depicted with dotted lines. For comparison the free concentration of roginolisib as measured in patients at the 80 mg dose is depicted in Figure 6B as well.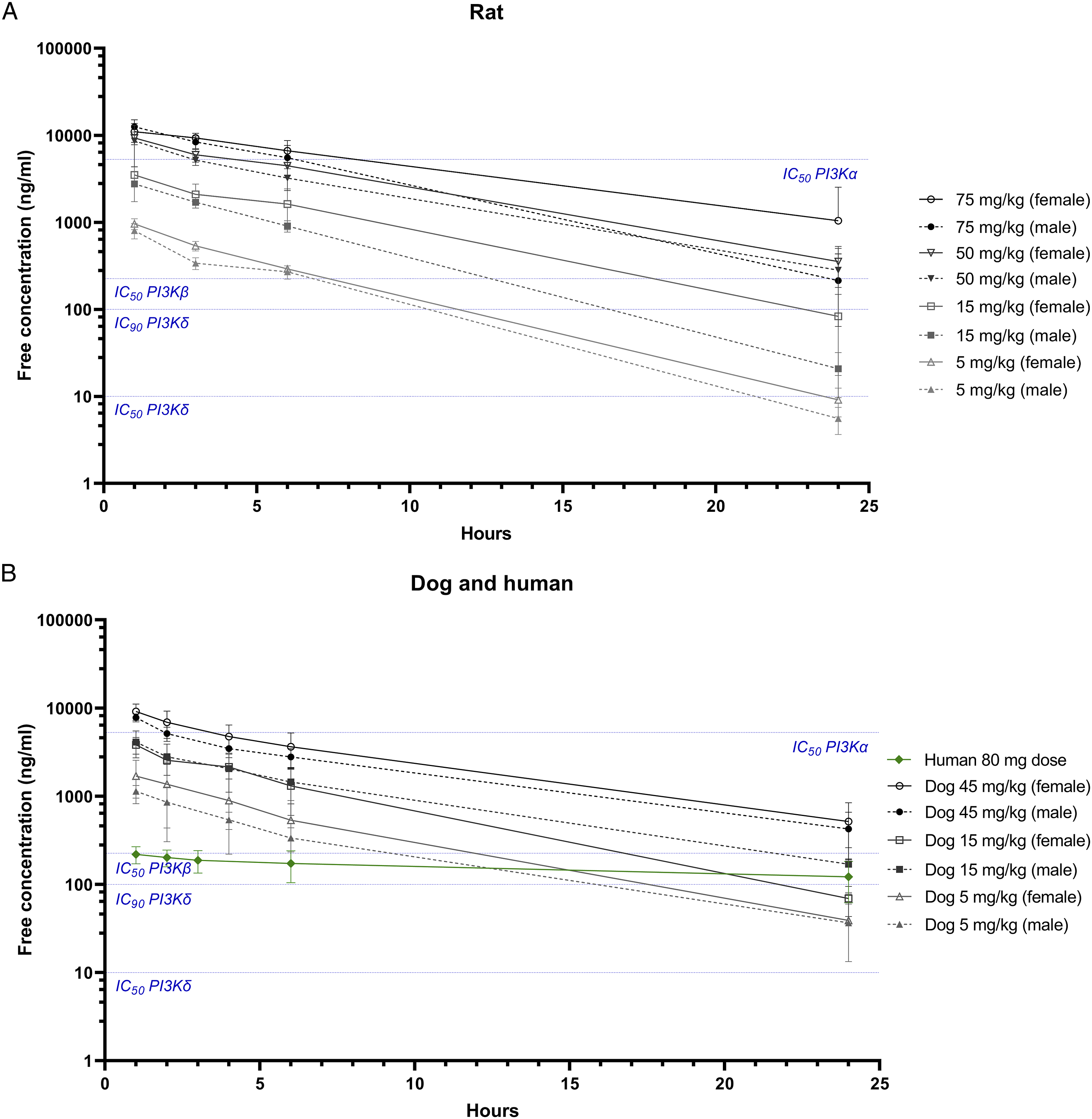
Plasma Protein Binding, pAkt Whole Blood Potency, and Predicted Clearance of Roginolisib in Mouse, Rat, Dog, and Human.
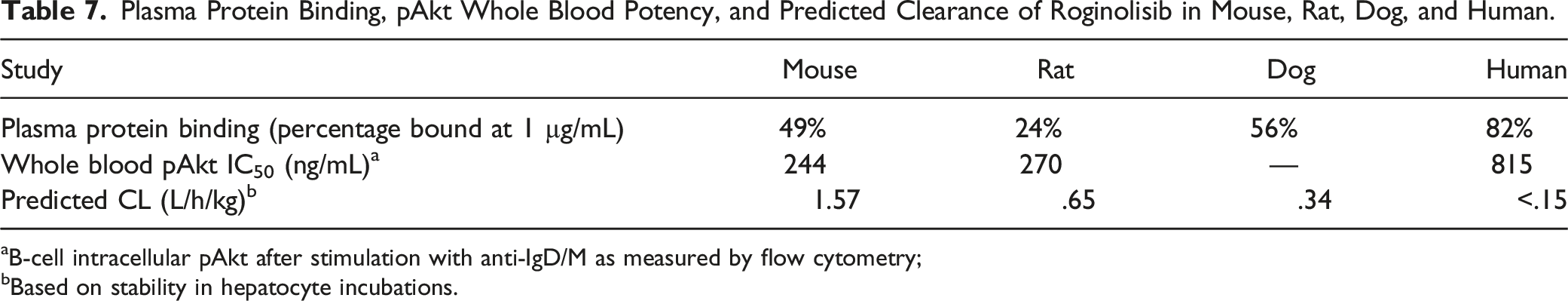
aB-cell intracellular pAkt after stimulation with anti-IgD/M as measured by flow cytometry;
bBased on stability in hepatocyte incubations.
Conclusion
Roginolisib has a toxicology profile similar to other PI3Kδ inhibitors: the compound is well-tolerated in rats, but is associated with skin and gastrointestinal toxicities in dogs. Though dose-dependent toxicities were observed, especially at doses resulting in concomitant inhibition of PI3Kα and PI3Kβ, these effects were mostly reversible. Our data indicate that doses that do not selectively inhibit PI3Kδ and result in inhibition of additional PI3K isoforms are associated with adverse effects. Based on this concept, doses for human investigation were chosen not to exceed a daily oral dose of 200 mg to prevent any inhibition of PI3Kα at Cmax and the optimal dose range for selective PI3Kδ inhibition was deemed to be in the range of 20 mg to 100 mg. In addition, roginolisib shows no apparent risk for metabolite generation (unlike idelalisib 36 ) and a predictable PK profile in human. All these properties make it a candidate for future clinical investigation, and current clinical data 35 confirm this anticipated profile of a well-tolerated PI3Kδ inhibitor.
Footnotes
Acknowledgements
The authors would like to thank the Peter Donoghue, a medical writer at Niche Science and Technology Ltd for assistance with the preparation of the manuscript and support with its submission.
Author Contributions
Van der Veen, L. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Schmitt, M. contributed to design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Deken, M.A. contributed to design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Lahn, M. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Marcel Deken, Michael Lahn, and Lars van der Veen are employees of iOnctura. Michael Schmitt is an employee of Merck Healthcare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by iOnctura SA and Merck Healthcare KGaA.
