Abstract
Enarodustat (JTZ-951) is an oral hypoxia-inducible factor-prolyl hydroxylase (HIF-PH) inhibitor for the treatment of anemia with chronic kidney disease. Carcinogenicity of enarodustat was evaluated in a 26-week repeated oral dose study in Transgenic rasH2 (Tg.rasH2) mice and a 2-year repeated oral dose study in Sprague-Dawley (SD) rats. The highest dose levels were set at 6 mg/kg in the Tg.rasH2 mouse study and at 1 mg/kg in the SD rat study based on the maximum tolerated doses in the 3-month and 6-month dose-range finding studies, respectively. Enarodustat did not increase the incidence of any tumors or affect survival in these carcinogenicity studies. Pharmacology-related findings including increases in blood RBC parameters were observed at the highest dose levels for each study. The AUC-based exposure margins as protein-unbound drug base are 16.3-/26.0-fold multiple (males/females) for Tg.rasH2 mice and 1.6-/1.1-fold multiple for SD rats when compared with the estimated exposure in human with chronic kidney disease at 8 mg/day (maximum recommended human dose). In conclusion, enarodustat was considered to have no carcinogenic potential at the clinical dose.
Introduction
Hypoxia-inducible factor-prolyl hydroxylase (HIF-PH) inhibitors are a class of agents for the treatment of renal anemia.1,2 Small-molecule inhibitors of HIF-PH enzymes induce endogenous erythropoietin (EPO) production via HIF stabilization. HIF-PH inhibitors mimic physiological responses to hypoxia, stabilizing HIF by regulating its degradation and leading to the expression of HIF-target genes involved in EPO production and iron metabolism.3,4 At least 6 HIF-PH inhibitors have been developed: roxadustat, daprodustat, vadadustat, enarodustat, molidustat, and desidustat, all of which competitively inhibit binding of 2-oxoglutarate, an essential co-substrate of the HIF-PH enzymes. 5 Enarodustat (JTZ-951, CAS No.: 1262132-81-9, ENAROY®) is a once-daily, orally administered HIF-PH inhibitor developed by Japan Tobacco Inc. It was approved in September 2020 in Japan, in November 2022 in South Korea and in June 2023 in China for the treatment of anemia associated with chronic kidney disease (CKD).6–8
The pharmacological action of enarodustat is to enhance EPO production through a HIF-PH inhibitory action, resulting enhanced erythrocytosis (increases in reticulocytes and RBC-related parameters). All of the findings observed in general toxicity studies of enarodustat in mice, rats, dogs and monkeys were considered to be directly or secondarily related to enhanced erythropoiesis and resultant polycythemia or the HIF-PH inhibition caused by enarodustat. There were no target organ toxicities due to the intrinsic properties of the enarodustat molecule itself in any animal species. Most of the observed changes disappeared or tended to disappear on the withdrawal of dosing and were considered to be reversible.
The genotoxicity of enarodustat was investigated in a standard battery of in vitro and in vivo tests under the GLP regulations (data not shown). Enarodustat had no potential to induce reverse mutations in a bacterial reverse mutation test. There were no aberrations in an in vitro chromosomal aberration test using human peripheral blood lymphocytes. In addition, there was no potential to induce chromosomal aberrations or DNA-damage in the in vivo chromosomal aberration test using rat bone marrow cells or in the in vivo comet assay using rat liver and peripheral blood lymphocytes.
Carcinogenicity assessments of small-molecule pharmaceuticals have been generally conducted in 2-year carcinogenicity studies in rats and mice or using an alternative model such as short-term or medium-term carcinogenicity studies in transgenic mice. The carcinogenic potential of enarodustat was evaluated in a traditional 2-year repeated oral dose study in rats and a 26-week repeated oral dose study in Tg.rasH2 mice.
Tg.rasH2 mice express the human protooncogene c-Ha-ras and develop a high incidence of pulmonary tumors and hemangiosarcomas. 9 This mouse strain has been well-studied and is sensitive to both genotoxic and nongenotoxic carcinogens. A 26-week carcinogenicity assay using Tg.rasH2 mice is an acceptable alternative to the traditional 2-year mouse carcinogenicity assay as described in the ICH S1B document.9,10 The U.S. Food and Drug Administration (FDA) states that the Tg.rasH2 mouse model has been adequately evaluated as an alternative model for carcinogenicity testing of pharmaceutical candidates. 9 Due to the low incidence of spontaneous tumors and lethal degenerative changes as commonly observed in long-term rodent studies, the Tg.rasH2 mouse has been used for most short-term carcinogenicity studies of pharmaceuticals in recent years.11-13
Carcinogenicity assessments of other HIF-PH inhibitors, namely, roxadustat, daprodustat and vadadustat, have been already published.14-16 Two-year traditional carcinogenicity studies were conducted in mice and rats for roxadustat and daprodustat, and a short-term carcinogenicity study using Tg.rasH2 mouse was conducted as an alternative model to the traditional 2-year mouse carcinogenicity assay for vadadustat, the same as enarodustat.
Materials and Methods
The 26-week Tg.rasH2 mouse study was conducted at the former BioReliance, Merck's BioReliance testing services (Rockville, MD, USA, now Millipore Sigma, a subgroup of Merck KGaA, Darmstadt, Germany) and the 2-year rat study was conducted in BoZo Research Center Inc. (Shizuoka, Japan). Toxicokinetic analysis for these studies was conducted by the Toxicology Research Laboratories Central Pharmaceutical Institute Japan Tobacco (Kanagawa, Japan). The present carcinogenicity studies were designed in accordance with ICH guidelines, to fulfill the requirements of regulatory authorities, for the toxicity testing of new drugs and were conducted in accordance with the OECD Principles of Good Laboratory Practice as accepted by Regulatory Authorities throughout the European Union, USA, and Japan. Study protocols of the carcinogenicity studies including the dose levels were reviewed and agreed on special protocol assessment by the FDA’s Carcinogenicity Assessment Committee (CAC).
The present studies were conducted in facilities accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC) according to the Guide for the Care and Use of Laboratory Animals.17-19 Study design, the number of animals and animal procedures for these studies were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) in each test facility.
Test Articles and Vehicle and Positive Control formulations
Enarodustat (JTZ-951, Lot Number: T) was manufactured by Hamari Chemicals Ltd. (Osaka, Japan) and supplied by Japan Tobacco, Inc., stored at 2-8°C, and protected from light. The purity of this lot was 99.8% enarodustat. Dose formulations were prepared at concentrations of .06, .20, and .60 mg/mL for the Tg.rasH2 mouse study and .02, .06, and .2 mg/mL for the rat study. In both carcinogenicity studies, enarodustat suspended in a .5% methylcellulose aqueous solution, and was administered at a dosing volume of 10 mL/kg for the mouse study and of 5 mL/kg for the rat study by gavage. The animals in the negative control groups were administered the vehicle in each study at the same volume. Individual dose volumes were calculated based on each animal’s most recently recorded body weight.
In the Tg.rasH2 mouse study, urethane in .9% sodium chloride was used as the positive control compound and was administered as intraperitoneal injections of urethane at 1000 mg/kg on Days 1, 3, and 5.
Animals
26-Week Tg.rasH2 Mouse Study
A 26-week carcinogenicity study using Tg.rasH2 mice is an acceptable alternative to the traditional 2-year mouse carcinogenicity assay as described in the ICH guidance. CByB6F1 mice are the wild-type littermates of the Tg.rasH2 mice and thus have the same genetic background, except for the omission of the Tg element. Therefore, the wild-type littermates were considered acceptable for use in the toxicokinetic (TK) cohort.9,10 In the present study, male and female Tg.rasH2 [CByB6F1-Tg (HRAS)2Jic (+/−hemizygous c-Ha-ras)] mice (n = 25/sex/group) were obtained from Taconic Biosciences (Germantown, NY, USA). These animals at 6 weeks of age were assigned to the main cohort groups to assess carcinogenicity. Male and female wild-type CByB6F1 mice [CByB6F1-Tg (HRAS)2Jic (−/−homozygous c-Ha-ras)] (n = 11/sex/group for enarodustat-treated groups and n = 8/sex/group for vehicle control group) at 6 weeks of age were assigned to the TK groups of enarodustat. The body weights on Day 1 ranged from 19.6 to 26.3 g (main cohort males), from 15.5 to 21.0 g (main cohort females), from 22.3 to 29.1 g (TK cohort males) and from 17.5 to 23.3 g (TK cohort females). The animals were housed under the following conditions: temperature at 21.9°C to 22.8°C, relative humidity at 36.6 to 55.1%, air ventilation with at least 10 times per hour and twelve-hour illumination per day. The animals were housed as 2 animals per cage prior to allocation and 1 animal per cage following allocation and through the remainder of the study. Cages and feeders were changed at least weekly. Animals were transferred to clean racks at least once every other week. The animals were housed in polycarbonate cages (190 × 115 × 152 mm) with Sani-Chip Hardwood bedding (P.J. Murphy Forest Products, USA). TEKLAD Global Diet #2018CM (Certified 18% Protein Rodent Diet, Envigo, Madison, Wisconsin, USA) was provided to all animals in stainless-steel rodent feeders ad libitum from arrival until termination of the study. The animals were provided ad libitum access to drinking water (Washington Suburban Sanitary Commission Potomac Plant, Potomac, MD, USA) by an automatic watering system from arrival until termination. Supplemental water, Napa Nectar™ (Systems Engineering, Napa, CA, USA), was provided as needed. The water met U.S. EPA drinking water standards. Environmental enrichments were provided to the animals during the period of the animal experiment.
2-Year Rat Study
Male and female Sprague-Dawley rats (Crl:CD (SD) (n = 65/sex/group) were obtained from Charles River Laboratories Japan, Inc. (Atsugi Breeding Center, Kanagawa, Japan). These animals at 5 weeks of age were assigned to the main cohort groups to assess carcinogenicity (260 animals of each sex for the main group and 20 animals of each sex for the satellite group). The animals were housed in a separate animal room for each sex under the following conditions: temperature at 21 to 24°C, relative humidity at 42 to 62%, air ventilation at 10 to 15 times per hour and 12-hour illumination per day. Two rats of the same sex were housed in a plastic solid floor cage (440 × 275 × 180 m) with bedding (ALPHA-dri: Shepherd specialty papers Inc., USA). Cages were relocated in cage racks once every 6 months to avoid the bias of cage disposition between the groups. Pelleted diet (irradiation-sterilized CR-LPF, Oriental Yeast Co. Ltd., Tokyo, Japan, in a stainless-steel feeder) and drinking water (Gotemba City Water, via an automatic water-supply system) were provided ad libitum. A few animals with teeth malocclusion/fracture were given triturated diet (CR-LPF) in a stainless-steel feeder to enable them to feed. Environmental enrichments were provided to animals and exchanged with the cage once a week during the period of the animal experiment.
Dose Justification
26-Week Tg.rasH2 Mouse Study
In a 13-week oral dose dose-range finding study using Tg.rasH2 wild-type mice (dose levels: 6, 10, and 20 mg/kg/day), 5 males and 2 females were found dead at 20 mg/kg. Spleen weight was increased at 10 mg/kg in males and at 6 mg/kg and above in females due to congestion and extramedullary hematopoiesis related to the pharmacological action of enarodustat. Spleen weights at 10 mg/kg were 1.8 times and 2.6 times higher than those of the control groups in males and females, respectively. Therefore, the dose levels of 10 mg/kg and above which could induce severe polycythemia were considered to have the potential to reduce the lifespan of the mice and so were unsuitable for use in a 26-week carcinogenicity study in mice. Thus, the dose level of 6 mg/kg was considered to be the estimated maximum tolerated dose (MTD) in a 26-week mouse carcinogenicity study. Therefore, the highest dose level in this study was set at 6 mg/kg and the mid and low dose levels are set at 2 and .6 mg/kg, respectively.
Study Design of the 26-Week Carcinogenicity Study of Enarodustat in Tg.rasH2 Mice.

*: The positive control animals were administered a total of 3 i.p. injections of urethane on Days 1, 3, and 5 and necropsied in Week 11.
2-Year Rat Study
Study Design of the 2-Year Carcinogenicity Study of Enarodustat in Sprague-Dawley Rats.

In Life Observations, Hematology
26-Week Tg.rasH2 Mouse Study
Clinical signs, body weights and food consumption were assessed for the main cohort animals. Blood samples for hematology were collected from the retro-orbital sinus under anesthesia from the first 10 surviving animals for each group prior to terminal necropsy into blood collection tubes containing EDTA-2K. The following parameters were measured or calculated using the Siemens ADVIA 120 hematology: erythrocyte count (RBC), hemoglobin (HGB), hematocrit (HCT), reticulocyte count (RET), mean corpuscular hemoglobin concentration (MCHC), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), platelet count (PLT), white blood cell count (WBC), and Differential WBC count.
2-Year Rat Study
Clinical signs, body weights and food consumption were assessed for the main cohort animals. At the time of scheduled necropsy at the end of the dosing period, all surviving animals (nonfasted) were subjected to abdominal incision under isoflurane anesthesia. Blood samples for hematology were collected from the abdominal aorta into blood collection tubes containing EDTA-2K. The following parameters were determined: RBC, HGB, HCT, RET count, PLT count, and WBC/Differential WBC count. In the rat study, hematology examination was also conducted in Week 32 on all satellite animals only for the control and high dose groups for the check of the increased hematopoiesis due to pharmacology of enarodustat.
Toxicokinetics
26-Week Tg.rasH2 Mouse Study
Blood samples were collected from the retro-orbital sinus of mice under anesthesia by 70% CO2/30% O2 into sodium heparin-treated tubes and the samples were maintained on wet ice. They were centrifuged (8000× g for 5 minutes at 4°C) to obtain plasma (>200 μL). The plasma concentrations of enarodustat were determined at .5 and 2 hours after dosing on Days 1 and 177 of the dosing period, after oral doses of enarodustat at the dose levels of .6, 2, and 6 mg/kg, using liquid chromatography-tandem mass spectrometry (LC/MS/MS).
The aim of monitoring the plasma concentration of enarodustat was just to confirm that the exposure level was equivalent to that of the previous studies (13-week dose-range finding study and preliminary single oral dose TK study); therefore, TK sampling point was limited to 2 time points (.5 and 2 hours after dosing) for each day of sampling.
2-Year Rat Study
Blood samples at each time point were collected from the jugular vein at .5, 1, 2, 4, 8, and 24 hours after dosing (6 time points) on Day 1, and before dosing and at .5, 1, 2, 4, 8, and 24 hours after dosing (7 time points) in Week 26 (Day 176). Blood samples were collected into centrifuge tubes treated with heparin sodium and placed on wet ice. Blood samples were centrifuged (6000× g for 1 minute at 4°C) to obtain plasma (>100 μL). The plasma concentrations of enarodustat were determined by the LC/MS/MS method.
Pathology
26-Week Tg.rasH2 Mouse Study
Tissues and Organs Collected at Necropsy for Histopathologic Examination.

2-Year Rat Study
All animals were euthanized by exsanguination from the abdominal aorta under isoflurane anesthesia. Moribund animals were euthanized by exsanguination and necropsied after collecting blood samples when possible, and animals found dead were subjected to necropsy soon after discovery. Following detailed external and internal examinations, the organs/tissues listed in Table 3 except for the eyeballs including optic nerves were fixed and preserved in 10% NBF. The eyeballs and optic nerves were fixed with a mixture consisting of 3% glutaraldehyde and 2.5% formalin, and then preserved in 10% NBF. All organs/tissues listed below from all the animals were embedded in paraffin, sectioned, stained with H&E and examined histopathologically. Paired organs were collected and examined bilaterally. A pathological peer review was conducted by a neutral senior pathologist (John Curtis Seely, EPL, Inc.).
Statistical Analysis
26-Week Tg.rasH2 Mouse Study
Body weight, body weights gain, food consumption, hematology and organ weight data were analyzed by Dunnett's test. Analyses for the test article groups compared to the vehicle control group was conducted separately. Statistical analysis of body weight, food consumption and clinical observations from positive control animals was not conducted. All required statistics were based on a significance value of P < .05.
Tumor pathology and mortality data were transferred to SAS transport format. Statistical analysis of survival and tumor data was conducted by BioSTAT Consultants, Inc (Portage, MI, USA) using methods outlined in references.20-22 Kaplan-Meier estimates of group survival rates were calculated by sex. The generalized Wilcoxon test for survival was used to compare the homogeneity of survival rates across the vehicle control and test article groups by sex. If the survival rates were significantly different (P < .05), the generalized Wilcoxon test was used to make pairwise comparisons of each test article group with the vehicle control group. The positive control group was compared to the vehicle control group using the generalized Wilcoxon test. Survival times in which the status of the animal’s death is classified as an accidental death, planned interim sacrifice or terminal sacrifice were considered censored values for the purpose of the Kaplan-Meier estimates and survival rate analyses.
In the statistical analysis of tumor data for vehicle and test article groups, the incidence of tumors was analyzed by Peto’s mortality-prevalence method, without continuity correction, incorporating the context (incidental, fatal, or mortality independent) in which tumors were observed. The following fixed intervals were used for incidental tumor analyses: weeks 1 through end of study (not including scheduled terminal sacrifice) and scheduled terminal sacrifice. All tumors in the scheduled terminal sacrifice interval were considered incidental for the purpose of statistical analysis. Tumors classified as mortality-independent were analyzed with Peto’s mortality-independent method incorporating the day of detection. Each diagnosed tumor type was analyzed separately and, at the discretion of the Study Director, analysis of combined tumor types and/or organs was performed. All metastases and invasive tumors were considered secondary and not included in the analyses. A 1-sided comparison of each test article group with the vehicle control was performed. An exact permutation test was conducted for all analyses. Findings were evaluated for statistical significance at both the .01 and .05 levels.
Tumor incidence in the positive control group was compared to the vehicle control group with a 1-sided Fisher’s exact test at both the .01 and .05 significance level (only for alveolar-bronchiolar adenoma and alveolar-bronchiolar carcinoma).
2-Year Rat Study
Body weights, body weight gain, food consumption and hematology were analyzed according to the following procedure. RBC-related parameters (red blood cells, hemoglobin, hematocrit, and reticulocytes) were analyzed by a one-tailed test from the perspective of pharmacology. Data from moribund animals for hematology were excluded from the statistical analysis. First, homogeneity of the variance in each group was analyzed by Bartlett’s test. For homogeneous data (P > .01), the group mean differences between the control and each dose group were analyzed by Dunnett’s test (P ≤ .05, .01, two-tailed levels). For heterogeneous data (P ≤ .01), the mean rank differences between the vehicle control and each dose group were analyzed by Steel’s test (P ≤ .05, .01, two-tailed levels). All analyses were performed by the integrated statistical package SAS Release 9.1.3 (SAS Institute Inc., USA).
The survival function of each test group was estimated by Kaplan-Meier’s method. The positive trend of the survival rate to dose level was analyzed by a Tarone-type method, and then the difference between the control group and each dose group was compared using a log-rank test. The level of significance was .05. The above analyses were performed by the integrated statistical package Stat View Version 5.0 (SAS Institute Inc., USA).
For the tumors that occurred at high incidence (incidence of 10/sex or more: common tumor), positive trend to dose level and pairwise comparison between the control group and each dose group were analyzed by the survival-adjusted Peto’s method. 23 For the tumors that occurred at low incidence (incidence of less than 10/sex: rare tumors), positive trend to dose level and pairwise comparison between the control group and each dose group were analyzed by Peto’s exact test. Incidental tumors were analyzed in the following partitions: Week 1 to Week 52, Week 53 to Week 78, Week 79 to Week 92, Week 93 to Week 104, and during the terminal necropsy period. Analysis of positive trends in the incidence was conducted at the significance levels of .005 (one-tailed level) for common tumors and .025 (one-tailed level) for rare tumors. Pairwise comparison was conducted at the significance levels of .01 (one-tailed level) for common tumors and .05 (one-tailed level) for rare tumors. Common tumors were defined as those with a historical incidence in controls at BoZo Research Center Inc. exceeding 1% (>1%) and rare tumors as 1% or less (≤1%). The above analyses were performed by the integrated statistical package SAS Release 9.1.3 (SAS Institute Inc., USA) and EXSUS Version 7.6 (CAC Croit Corporation, USA).
Results
26-Week Tg.rasH2 Mouse Carcinogenicity Study
Survival
Summary of Early Deaths/Moribund Condition in the 26-Week Carcinogenicity Study of Enarodustat in Tg.rasH2 Mice.

*: Urethane 1000 mg/kg.
Clinical Observations
There were no treatment-related clinical signs in either sex at any dose level. The positive control animals exhibited urethane-related toxicity including ataxia and prostrate posture during the treatment period (data not shown).
In Life data (Body Weights and Food Consumption)
Statistically significant increases in mean body weights in females at 6 mg/kg were consistently observed from Day 36 onward but the increases relative to the vehicle control were slight (4% to 8%) (data not shown). These differences appeared to be related to polycythemia due to enarodustat treatment. The positive control group demonstrated a decrease in body weights during the first week of the study, which was expected related to the toxicity of the urethane. The increase in body weights also associated with a large, statistically significant increase in spleen weights in females at 6 mg/kg, which was considered to be due to the exaggerated pharmacology of enarodustat.
Hematology
Hematology Examination in the 26-Week Carcinogenicity Study of Enarodustat in Tg.rasH2 Mice (Percent Difference From the Vehicle Control).
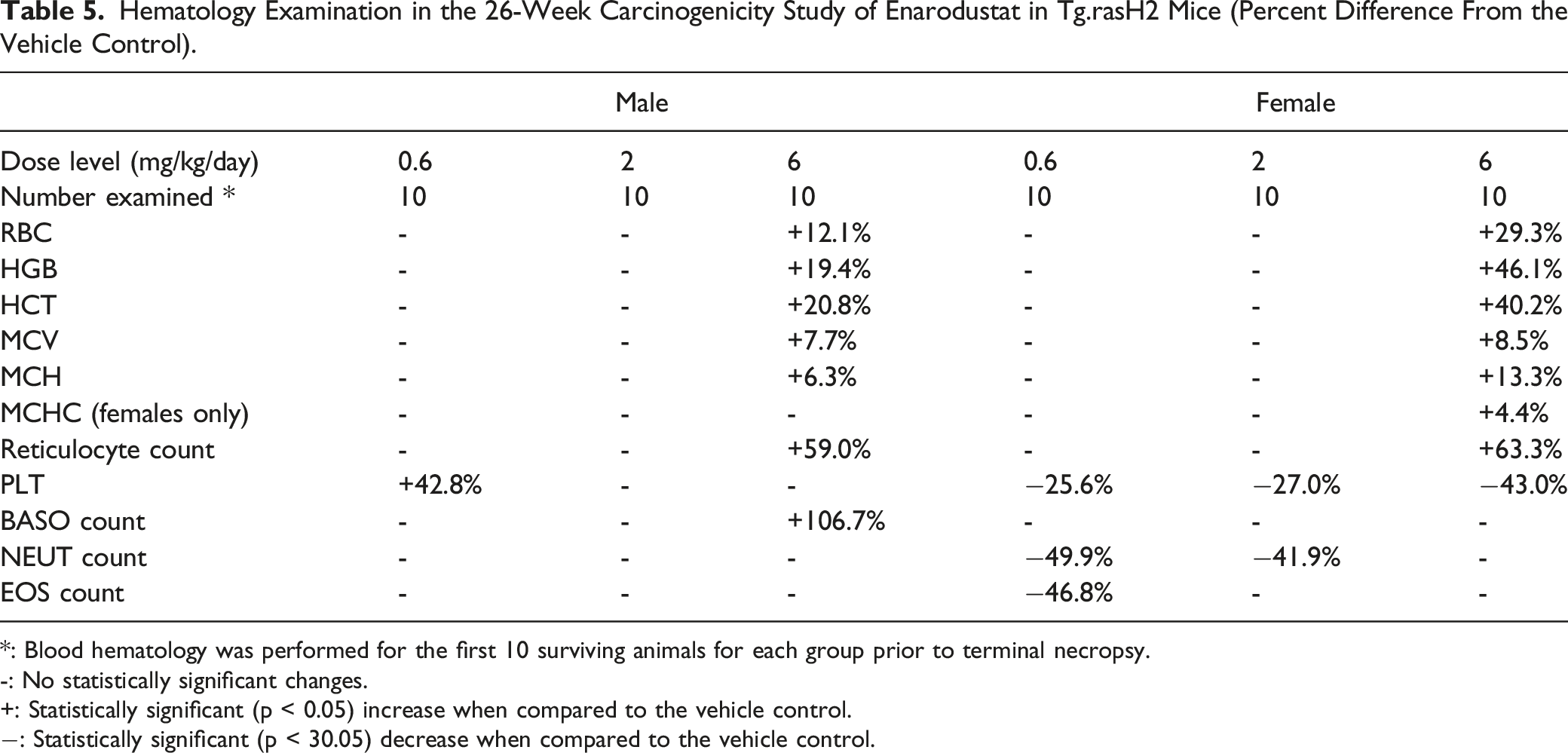
*: Blood hematology was performed for the first 10 surviving animals for each group prior to terminal necropsy.
-: No statistically significant changes.
+: Statistically significant (p < 0.05) increase when compared to the vehicle control.
−: Statistically significant (p < 30.05) decrease when compared to the vehicle control.
Necropsy and Organ Weights
The enlargement of the spleen was observed in the enarodustat-treated groups (data not shown). The pulmonary lesions in the positive control group were expected and related to tumor formation by the positive control (urethane). Statistically significant increases in the spleen weights were observed in both sexes at 6 mg/kg and increases in the kidney weights were observed in males at 2 mg/kg and above and in females at 6 mg/kg. The percent change of the absolute spleen weights at 6 mg/kg were +27.9% in males and 72.8% in females when compared to that in the vehicle control group, respectively. Statistically significant increases in the terminal body weight were noted in females at 6 mg/kg (+5.2% vs vehicle control). The increases in weights of kidney and spleen were attributed to renal congestion and extramedullary hematopoiesis, respectively, which were considered to be pharmacology-related and similar findings were observed in the previously conducted general toxicity studies of enarodustat in mice. Increases in body weights in females at 6 mg/kg were also attributed to the exaggerated pharmacology of enarodustat, polycythemia.
Neoplastic Microscopic Findings
Neoplastic Microscopic Findings in the 26-Week Carcinogenicity Study of Enarodustat in Tg.rasH2 Mice (Male).
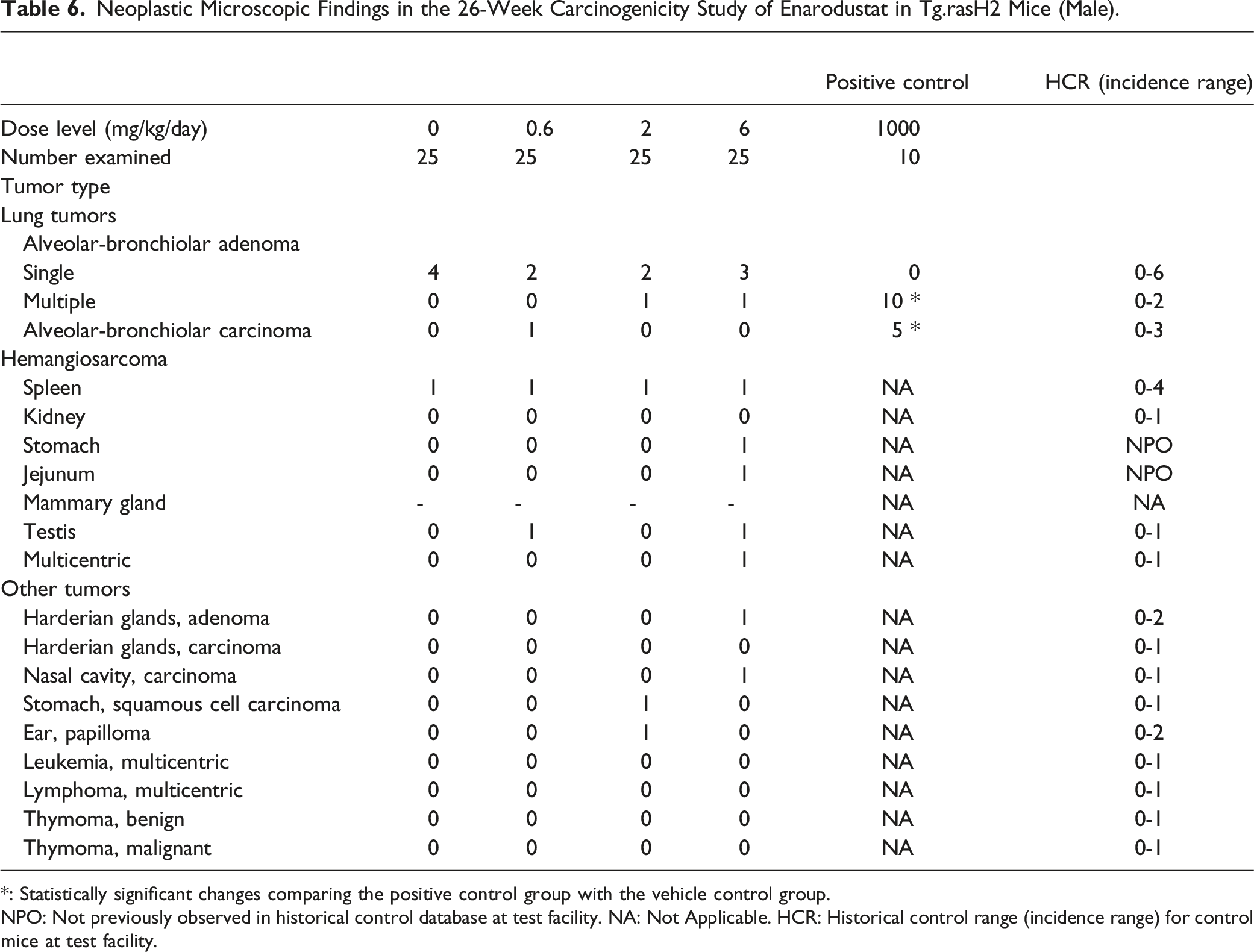
*: Statistically significant changes comparing the positive control group with the vehicle control group.
NPO: Not previously observed in historical control database at test facility. NA: Not Applicable. HCR: Historical control range (incidence range) for control mice at test facility.
Neoplastic Microscopic Findings in the 26-Week Carcinogenicity Study of Enarodustat in Tg.rasH2 Mice (Female).
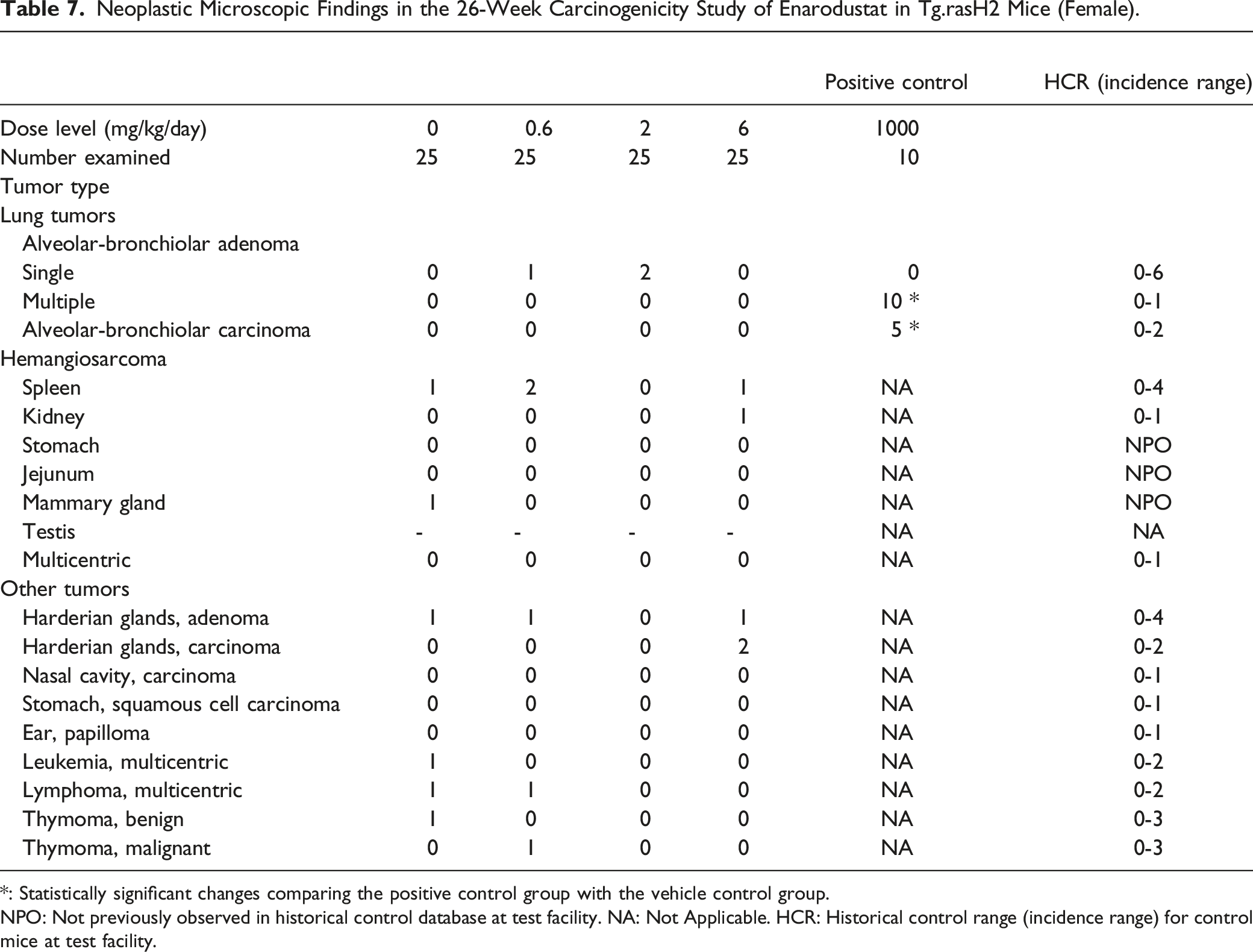
*: Statistically significant changes comparing the positive control group with the vehicle control group.
NPO: Not previously observed in historical control database at test facility. NA: Not Applicable. HCR: Historical control range (incidence range) for control mice at test facility.
Lung Tumors
All positive control mice in both sexes had alveolar-bronchiolar adenomas and there was a statistically significant increase (P < .01) in the incidence of pulmonary tumors when compared with the vehicle control group, proving the validity of the test assay. When the incidence of each type of tumor (single vs multiple adenoma, and carcinoma) was compared between vehicle control and test article treated groups, the incidence fell within the historical control ranges at the test facility and there were no statistically significant differences in the incidence of tumors.
Hemangiosarcomas
When the incidence of hemangiosarcomas was compared between vehicle control and test article treated groups, the incidence fell within the historical control ranges established at the test facility and there were no statistically significant differences in the incidence of tumors. The incidence of hemangiosarcomas in the spleen fell within the historical control ranges at the test facility and there were no statistically significant differences in the incidence of tumors, when compared between vehicle control and test article treated groups. The incidence of all hemangiosarcomas in multiple organs (including the spleen) was low, involved isolated organs and was without any tendency to involve a particular organ or organ system. When the incidence of hemangiosarcomas was compared to the historical control database, they all fell within the historical control range except for hemangiosarcoma in the mammary gland, jejunum and stomach, which had not been previously observed in other 26-week studies using Tg.rasH2 mice at the test facility. However, there was no statistically significant difference in tumor incidence between the vehicle control and test article treated groups.
Other Tumors
When the incidence of all other tumors (non-vascular and non-pulmonary) was compared between the vehicle control and the test article treated groups, there was no evidence of an increase in a statistically significant manner for any of the tumors. The occurrence of these tumors was low and there was no tendency for involvement of any particular organ or organ system.
Non-Neoplastic Microscopic Findings
Selected Non-Neoplastic Microscopic Findings in the 26-Week Carcinogenicity Study of Enarodustat in Tg.rasH2 Mice.
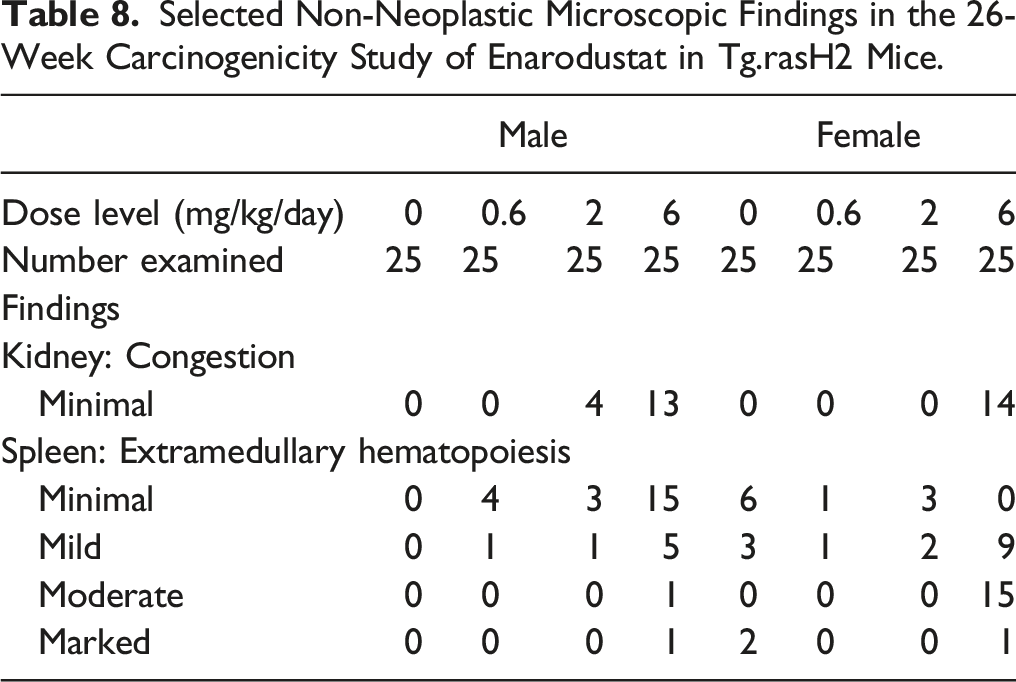
Treatment-related findings were observed in the kidneys and spleen. In males, there was an increase in the incidence of extramedullary hematopoiesis in the spleen at 6 mg/kg and there was also tendency of increase in the low and mid dose groups, which was not dose-dependent. In females, a higher incidence of extramedullary hematopoiesis is commonly observed even in the non-treatment animals. The incidence of extramedullary hematopoiesis in females at .6 mg/kg and 2 mg/kg remains lower than the vehicle control but was higher at 6 mg/kg. These findings at 6 mg/kg were considered to be due to the pharmacological action of enarodustat.
There were a variety of other histopathological findings in this study (data not shown). Their incidence was similar across the treatment groups; therefore, these findings were considered to be spontaneous and/or background lesions.
Systemic Exposure to Enarodustat
Systemic Exposure to Enarodustat in Carcinogenicity Studies.
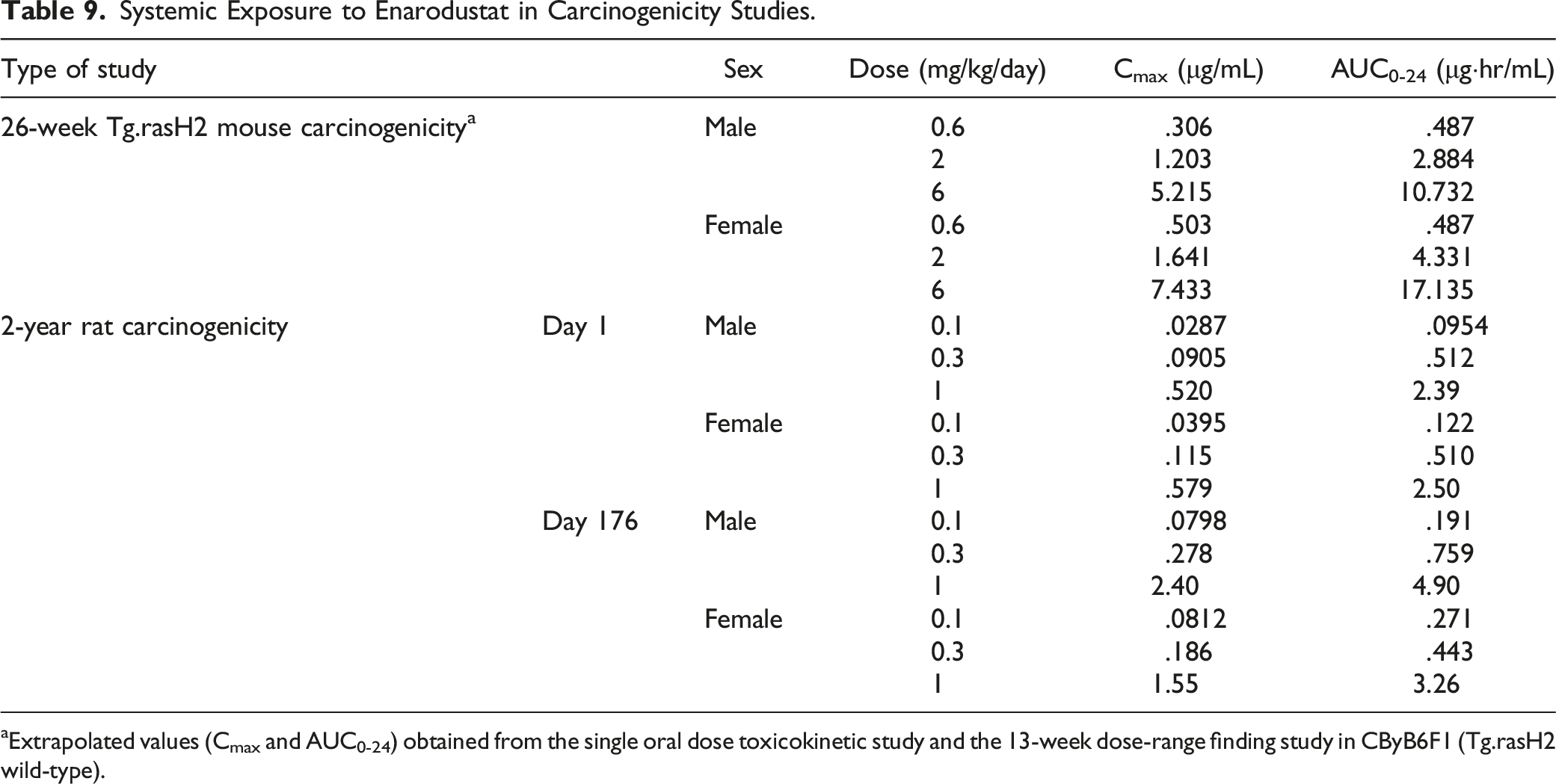
aExtrapolated values (Cmax and AUC0-24) obtained from the single oral dose toxicokinetic study and the 13-week dose-range finding study in CByB6F1 (Tg.rasH2 wild-type).
Comparison of Systemic Exposure Between the Maximum Recommended Human Dose and the Highest Dose in the Carcinogenicity Studies of Enarodustat.
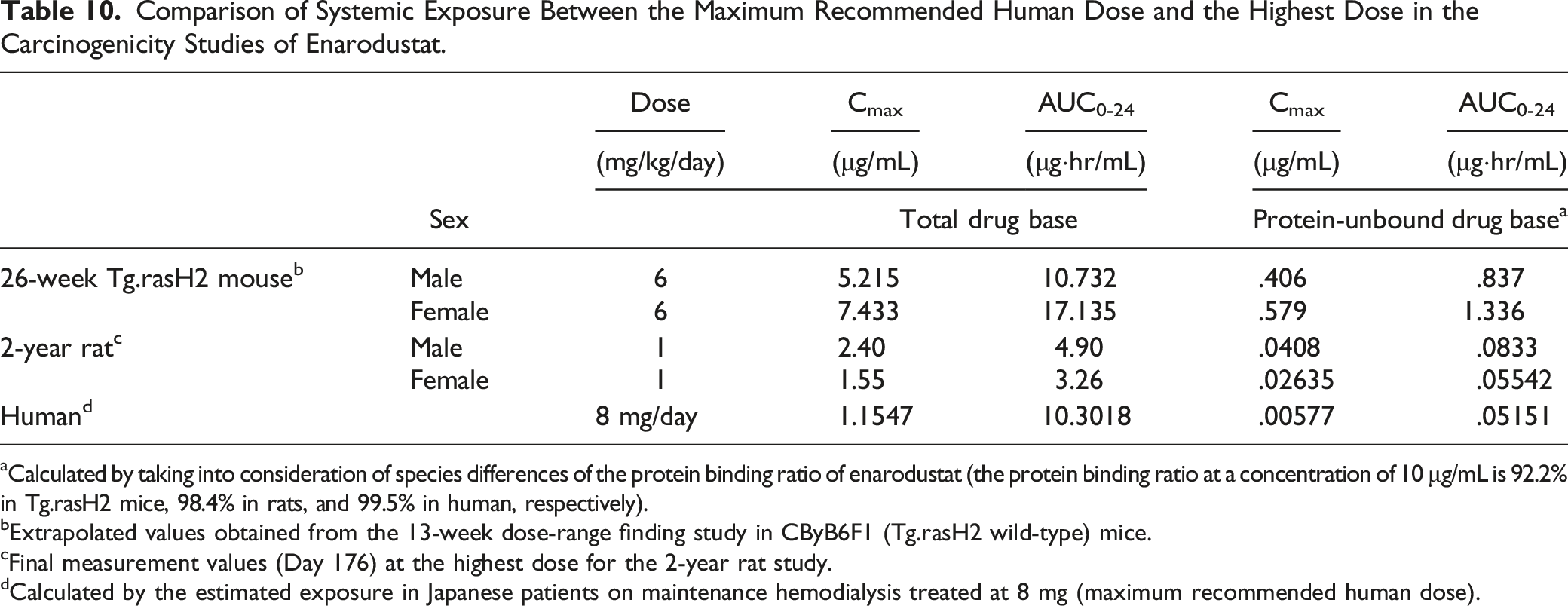
aCalculated by taking into consideration of species differences of the protein binding ratio of enarodustat (the protein binding ratio at a concentration of 10 μg/mL is 92.2% in Tg.rasH2 mice, 98.4% in rats, and 99.5% in human, respectively).
bExtrapolated values obtained from the 13-week dose-range finding study in CByB6F1 (Tg.rasH2 wild-type) mice.
cFinal measurement values (Day 176) at the highest dose for the 2-year rat study.
dCalculated by the estimated exposure in Japanese patients on maintenance hemodialysis treated at 8 mg (maximum recommended human dose).
2-Year Rat Carcinogenicity Study
Survival
Cause of Early Death/Moribund in the 2-Year Carcinogenicity Study of Enarodustat in Sprague-Dawley Rats.
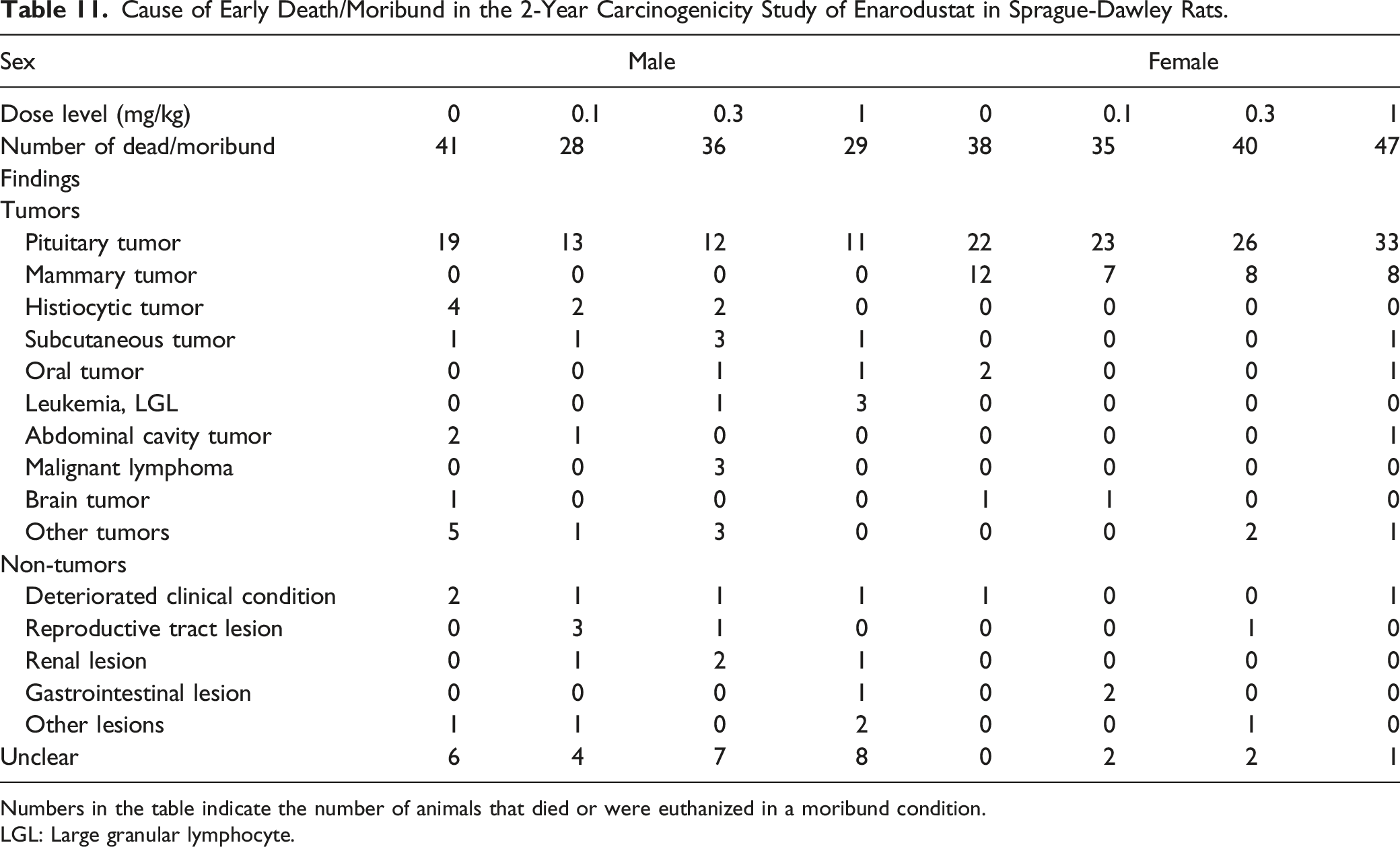
Numbers in the table indicate the number of animals that died or were euthanized in a moribund condition.
LGL: Large granular lymphocyte.
Body Weights, Food Consumption, and Clinical Observations
There were no treatment-related changes in either sex at any dose level (data not shown).
Hematology
Hematology was performed at Week 32 on 5 rats of each sex only for the control and 1 mg/kg groups in the satellite group. A slight increase in erythrocyte-related parameters was observed in some animals at 1 mg/kg as follows: hemoglobin concentrations in some males, hematocrit values in some animals of both sexes, and reticulocyte counts in some females (data not shown).
In the main groups at the end of the dosing period, a statistically significant increase in HGB concentrations was observed in males at 1 mg/kg. In addition, slight increases in RBC counts and HCT values were observed in several animals of both sexes at 1 mg/kg, and a slight increase in HGB concentrations was observed in several females at 1 mg/kg. These changes in RBC-related parameters in hematology were attributable to the pharmacological action of enarodustat. Although a statistically significant increase in neutrophil counts in males at .3 mg/kg and decreases in RBC, HGB, and HCT were noted in females at .3 mg/kg, they were considered to be incidental because there was no dose-dependency (data not shown).
Necropsy
There were no treatment-related macroscopic findings in either sex at any dose level. Numerous spontaneous findings noted as nodule in the pituitary, pancreas, and subcutaneous tissue were observed in all the groups including the controls.
Neoplastic Microscopic Findings
Neoplastic Microscopic Findings in the 2-Year Carcinogenicity Study of Enarodustat in Sprague-Dawley Rats (Male).
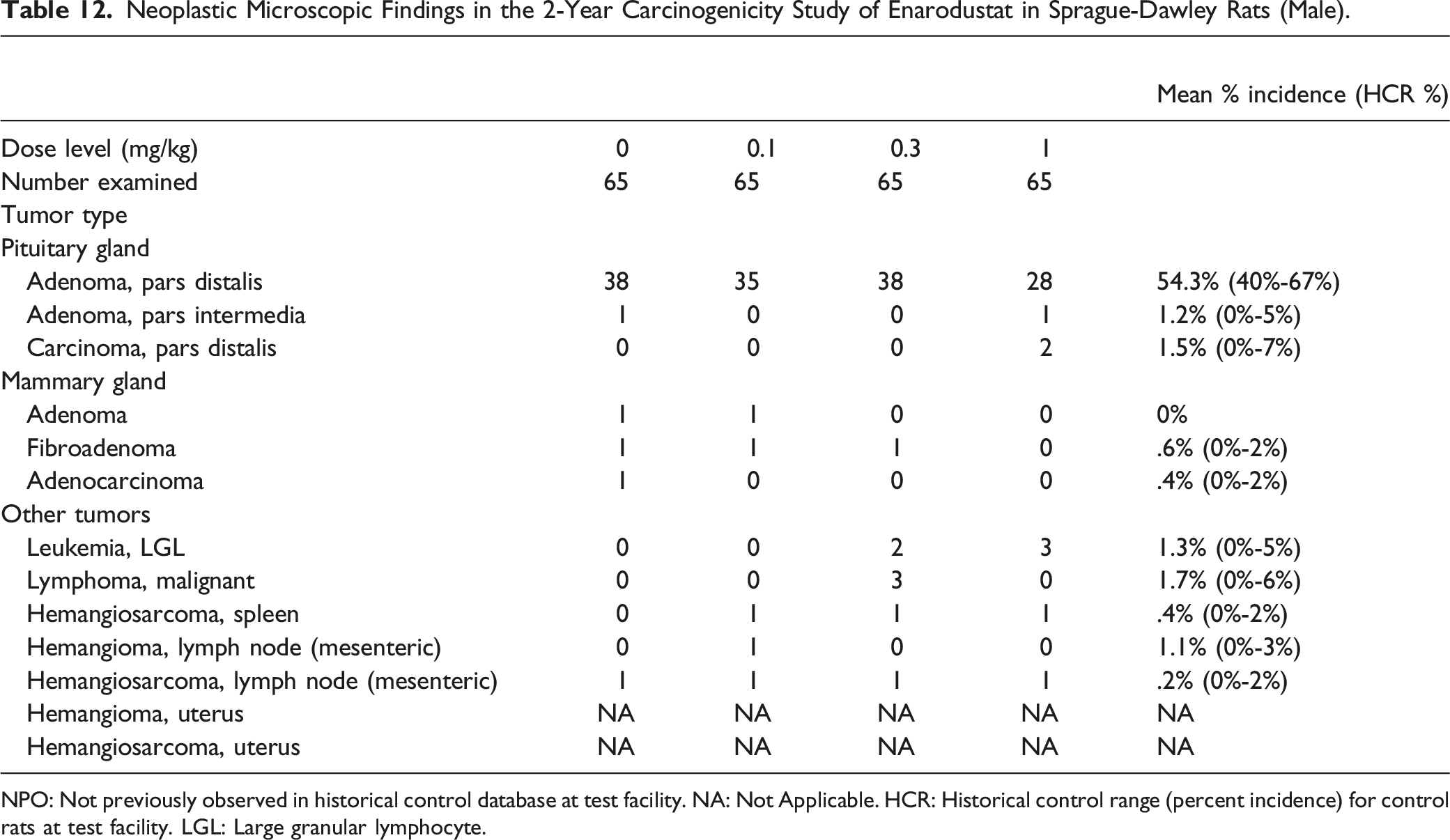
NPO: Not previously observed in historical control database at test facility. NA: Not Applicable. HCR: Historical control range (percent incidence) for control rats at test facility. LGL: Large granular lymphocyte.
Neoplastic Microscopic Findings in the 2-Year Carcinogenicity Study of Enarodustat in Sprague-Dawley Rats (Female).
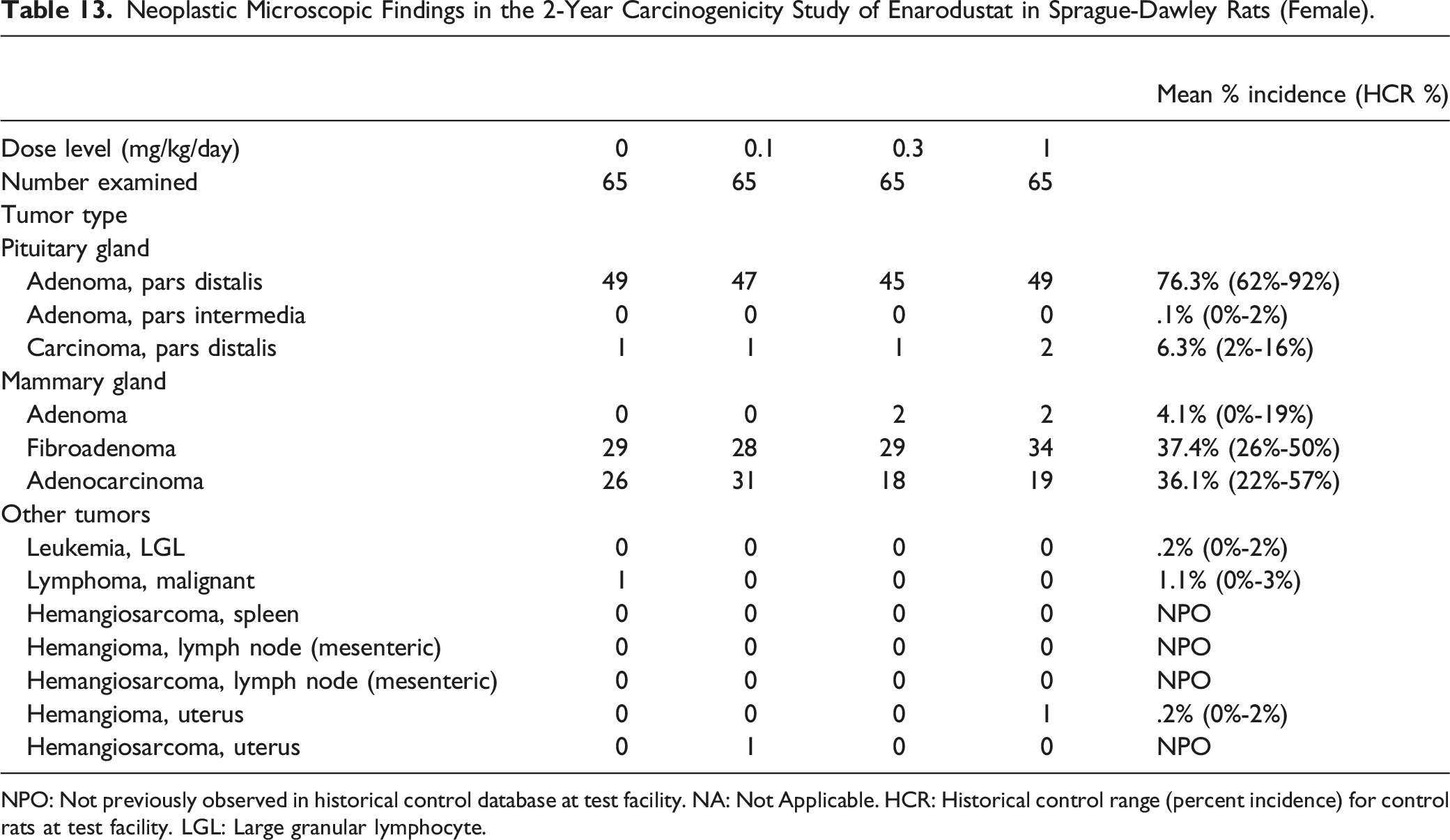
NPO: Not previously observed in historical control database at test facility. NA: Not Applicable. HCR: Historical control range (percent incidence) for control rats at test facility. LGL: Large granular lymphocyte.
Non-Neoplastic Microscopic Findings
There were no treatment-related findings in either sex at any dose level. The lesions observed in the treated animals including non-neoplastic proliferative lesions were considered to be incidental/spontaneous and commonly observed in aged rats of this strain (data not shown).
Systemic Exposure to Enarodustat
The results are shown in Table 9. The systemic exposure (Cmax and AUC0-24h) to enarodustat increased dose-dependently in both sexes up to the highest dose level at 1 mg/kg. There were no gender differences in the systemic exposure to enarodustat at any dose level. The systemic exposure at the highest dose level in the carcinogenicity studies and the estimated exposure at maximum recommended human dose (8 mg/day) are shown in Table 10. Systemic exposure (AUC0-24) to enarodustat at 1 mg/kg was 4.90 μg·hr/mL and 3.26 μg·hr/mL in males and females, respectively. The AUC-based exposure margins in males/females after dosing enarodustat are .5-/0.3-fold multiple as total drug base and are 1.6-/1.1-fold multiple as protein-unbound drug base when compared with the estimated exposure in human with CKD at 8 mg/day.
Discussion
In the 26-week repeated oral dose carcinogenicity study of enarodustat at .6, 2, and 6 mg/kg/day using Tg.rasH2 mice [CByB6F1-Tg (HRAS)2Jic (+/− hemizygous cHa-ras)], there was no increased enarodustat-related mortality in either sex and no increase in the incidence of any type of tumor or evidence of leukemia in any of the enarodustat-treated groups. This carcinogenicity study was valid based on the expected response of lung tumors in the positive control (urethane) animals and the high survival rates in all groups. Higher mean body weights and/or body weight gains due to increased spleen weight were observed in females at 6 mg/kg. Increased RBC-related parameters and organ weights in the kidneys and spleen were considered due to the pharmacological action of enarodustat. Splenic enlargement was noted grossly at terminal necropsy at 6 mg/kg. Congestion in the kidneys and extramedullary hematopoiesis in the spleen were observed in most enarodustat-treated groups and attributed to the exaggerated pharmacology of enarodustat.
In the 2-year repeated oral dose carcinogenicity study of enarodustat at .1, .3, and 1 mg/kg in SD rats, there were no treatment-related effects on the survival rate in either sex. Enarodustat did not increase the number of deaths attributable to specific tumors or non-tumor lesions. There were no treatment-related increases in the incidences of any tumor in any organ/tissue in either sex. At 1 mg/kg, there was a trend toward increases in RBC-related parameters (RBC count, Hb concentration, and Hct value) related to the pharmacology of enarodustat.
Several HIF-PH inhibitors have been approved and widely used for improvement of renal anemia.3,5 Of the carcinogenicity evaluation of other HIF-PH inhibitors, those for roxadustat, daprodustat, and vadadustat have been already published.14-16 The absence of carcinogenic potential in the present studies of enarodustat is in agreement with the results of the carcinogenicity studies of these other HIF-PH inhibitors.
Table 10 shows the systemic exposure at the highest dose in the carcinogenicity studies and the estimated exposure in Japanese patients on maintenance hemodialysis receiving a single oral administration at MRHD (8 mg/day), the highest clinical dose (AUC0-24: 10.3 μg·hr/mL; Cmax: 1.15 μg/mL). The AUC-based exposure margins in males/females after dosing enarodustat are 1.0-/1.7-fold multiple for Tg.rasH2 mice and .5-/0.3-fold multiple for SD rats as total drug (protein-bound drug plus protein-unbound drug) base. There are species differences in the protein binding ratios of enarodustat (the protein binding ratio at a concentration of 10 μg/mL is 92.2% in Tg.rasH2 mice, 98.4% in rats, and 99.5% in humans, respectively). Based on the protein-unbound concentration of enarodustat, the AUC-based exposure margins as protein-unbound drug base are 16.3-/26.0-fold multiple for Tg.rasH2 mice and 1.6-/1.1-fold multiple for SD rats when compared with the estimated exposure in human with CKD at 8 mg/day. Therefore, systemic exposures to enarodustat in the carcinogenicity studies are considered to exceed that of the MRHD in Tg.rasH2 mice and to be equivalent to MRHD in SD rats. In addition, the human equivalent dose (HED) calculated using the HED conversion factor 24 were 29.3 mg/man in mice (6 mg/kg) and 9.7 mg/man in rats (1 mg/kg), respectively.
The mechanism of action of HIF-PH inhibitors may raise theoretical safety concerns about tumorigenesis. 25 It is well documented that certain gene expressions induced by HIF activation lead to tumor formation and progression. 25 HIF-α has a potential to increase the expression of vascular endothelial growth factor (VEGF), a vascular endothelial cell-specific factor that increases vascular permeability and induces angiogenesis.26,27 HIF-PH inhibitors may increase these risks through their effects on VEGF expression and increased VEGF levels in cells and/or tissues and this could possibly be a contributing factor to develop vascular tumors including hemangiosarcoma. Since hemangiosarcomas occur spontaneously in Tg.rasH2 mice, it has been a concern that enarodustat could have a potential to induce VEGF expression leading to increase in the incidence of vascular tumors. In the present Tg.rasH2 mouse carcinogenicity study, there was no increase in the incidence of vascular tumors including hemangiosarcoma. A lot of conflicting nonclinical and clinical data associating increased HIF stabilization with tumor progression, metastasis and poor prognosis indicate that the role of HIF in cancer biology is highly complicated. Clinical studies using HIF-PH inhibitors had no serious adverse events related to increased systemic VEGF levels.28,29 The potential detrimental effect on VEGF production and function from enarodustat were assessed in other nonclinical studies. Plasma VEGF concentrations were increased by enarodustat-administration in rats at a >10-fold higher dose level of enarodustat than that required for endogenous EPO-mediated erythropoiesis. 8 Additionally, in a study using a colorectal cancer cell-inoculated mouse xenograft model, there were no effects of enarodustat on tumor growth at the dose level at which the blood VEGF level was increased. 8 These results indicate that enarodustat has a potential to induce VEGF at a high dose level without affecting tumorigenesis. In the 2-year rodent carcinogenicity studies of daprodustat (a HIF-PH inhibitor), very slight increase in VEGF mRNA was observed in lungs of the daprodustat-treated mice; however, no treatment-related neoplastic findings were noted. 15
In conclusion, in the 26-week carcinogenicity study in Tg.rasH2 mice and the 2-year carcinogenicity study in rats, the enarodustat-treatment did not increase the incidence of any tumor. Therefore, enarodustat was considered to have no carcinogenic potential under the conditions of these studies.
Footnotes
Acknowledgements
The authors would like to thank Dr John Curtis Seely (Experimental Pathology Laboratories Inc.), for histopathological peer review of both carcinogenicity studies in mice and rats.
Author Contributions
Y.K., K.T., T.I., Y.Y., and T.S. contributed to conception or design, contributed to acquisition, analysis, or interpretation of data, drafted the manuscript, and critically revised the manuscript for important intellectual content. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring integrity and accuracy.
Declaration of Conflicting Interests
The authors declare the following potential conflicts of interest with respect to the research, authorship, and publication for this article: Yusuke Kemmochi, Kaoru Toyoda, Tomio Ishida, Yuzo Yasui, and Toshiyuki Shoda are employees of Japan Tobacco Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
