Abstract
Transformed follicular lymphoma (t-FL) is an aggressive malignancy that is refractory and rapidly progressing with poor prognosis. There is currently no effective treatment. High-throughput screening (HTS) platforms are used to profile the sensitivity or toxicity of hundreds of drug molecules, and this approach is applied to identify potential effective treatments for t-FL. We randomly selected a compound panel from the School of Pharmaceutical Sciences Xiamen University, tested the effects of the panel on the activity of t-FL cell lines using HTS and the CCK-8 assay, and identified compounds showing synergistic anti-proliferative activity with the Bcl-2 inhibitor venetoclax (ABT-199). Bioinformatics tools were used to analyze the potential synergistic mechanisms. The single-concentration compound library demonstrated varying degrees of activity across the t-FL cell lines evaluated, of which the Karpas422 cells were the most sensitive, but it was the cell line with the least synergy with ABT-199. We computationally identified 30 drugs with synergistic effects in all cell lines. Molecularly, we found that the targets of these 30 drugs didn’t directly regulate Bcl-2 and identified 13 medications with high evidence value above .9 of coordination with ABT-199, further confirming TP53 may play the largest role in the synergistic effect. Collectively, these findings identified the combined regimens of ABT-199 and further suggested that the mechanism is far from directly targeting Bcl-2, but rather through the regulation and synergistic action of p53 and Bcl-2. This study intended to reveal the best synergistic scheme of ABT-199 through HTS to more quickly inform the treatment of t-FL.
Introduction
Follicular lymphoma (FL) is the most common indolent non-Hodgkin lymphoma (NHL) and accounts for 20% of all NHL cases.1,2 Translocation t (14; 18) (q32; q21) places the antiapoptotic Bcl-2 oncogene under the control of the immunoglobulin (Ig) heavy-chain enhancer and is a genetic hallmark of FL.3,4 Although the disease follows a progression-free course over a long period, progression and transformation still occur.2,5 Among them, the transformation into more aggressive forms is called transformed follicular lymphoma (t-FL), and the incidence of this aggressive form of lymphoma is 2-3% per year. 4 Patients with t-FL have rapid progression, treatment resistance, poor prognosis, and outcomes that are unfortunate and far inferior to those of FL patients whose disease did not undergo transformation. Despite the improvement in the efficacy of chemoimmunotherapy regimens, most patients remain incurable due to the genetic diversity and the high heterogeneity in the clinical presentation of t-FL.4–7 Therefore, further updated therapy regimens are urgently needed to impede progression, ameliorate prognosis, and achieve personalized treatment.
Bcl-2 is a member of the Bcl-2 family, which regulates cell apoptosis; it was first discovered by its involvement in the t (14; 18) chromosomal translocations commonly found in FL.8,9 Bcl-2, classified as an oncogene, has been identified as a cause for the onset of lymphomas, confers a survival advantage on B cells by inhibiting apoptosis, and more generally may block a common cell death pathway induced by chemotherapy, conferring clinical drug resistance on cancer cells overexpressing Bcl-2 protein.10,11 Therefore, many strategies have been developed to block or modulate the expression of Bcl-2. The first genuine BH3 mimetic and a specific antagonist of the antiapoptotic proteins Bcl-2, Bcl-xL, and Bcl-w, ABT-737 has attracted the attention of numerous biologists. However, the obvious targeted thrombocytopenia (through Bcl-xL inhibition) observed in clinical trials of this agent limited its clinical utility.12,13
Venetoclax (ABT-199), a highly selective Bcl-2 inhibitor, is currently approved by the U.S. Food and Drug Administration (FDA) for the treatment of patients with relapsed chronic lymphocytic leukemia (CLL) with a 17p deletion. 14 Research has demonstrated that venetoclax has potent activity against FL, diffuse large B-cell lymphoma (DLBCL), and mantle cell lymphoma (MCL) cell lines, as well as in a t (14; 18)-carrying xenograft model.15,16 Although venetoclax is active as a single agent in NHL, the clinical benefit varies depending on the histologic subtype. The combination of venetoclax with chemotherapy, monoclonal antibodies, and B-cell receptor signaling inhibitors suggests that venetoclax would be a logical partner in a range of combination regimens.16,17 Given the high heterogeneity of t-FL, the search for the optimal combination of venetoclax will enable targeted inhibition of Bcl-2 to have a greater clinical impact.
Currently, multitargeted drug combinations are evaluated in vitro and preclinically to establish anticancer efficacy and potential standards of care for different types of malignancies. 18 The further clinical development of venetoclax in B-cell malignancies, especially t-FL, will be aided by establishing venetoclax combination treatments with maximum synergistic effects. High-throughput screening (HTS) is often used to profile the sensitivity or toxicity of hundreds of drug molecules to identify a few hit compounds with desired response profiles for further development.19,20 We designed a drug HTS platform to systematically screen for combinations of venetoclax with approved therapeutics, emerging drugs, and well-characterized molecular probes targeting different aspects of the MAPK and PI3K signaling pathways known to be relevant in the pathogenesis of cancer. Compared to target-based drug discovery, which starts from the individual disease-related protein Bcl-2 in lymphoma, phenotype-based drug discovery assays profile the complex cellular system as a whole. The goal of the present study was to identify a venetoclax combination regimen that causes apoptosis in t-FL cell lines.
Material and Methods
Cell Culture
The t-FL cell line RL was obtained from the Institute of Hematology, Xiamen University School of Medicine (Xiamen, Fujian, China). The t-FL cell lines SU-DHL4 (CBP60628) and Karpas422 (CBP60629) were purchased from Nanjing Cobioer Biotechnology Co., Ltd. (Nanjing, Jiangsu, China), and SC-1 cells were generously donated by the Department of Hematology, Jiangsu Province Hospital (Nanjing, Jiangsu, China). All these cell lines were cultured in RPMI-1640 (Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (FBS, Gibco, CA, USA), 100 units/ml penicillin, and 100 μg/mL streptomycin (Invitrogen, Carlsbad, CA, USA). The cells were maintained in a 37°C incubator with a 5% CO2 environment.
The Automated Drug Screening System
The plate and handler automated screening system of PerkinElmer Co., Ltd. (PerkinElmer, Waltham, MA, USA) was used to conduct experiments through the Core Facility of Biomedical Sciences, Xiamen University (Xiamen, Fujian, China). The system is based on the plate and handler Flex multiaxis robotic arms, integrates various equipment such as a JANUS liquid workstation, a Nivo multifunction microplate reader, and an incubator, and has the function of high-throughput automated drug screening.
Compound Library
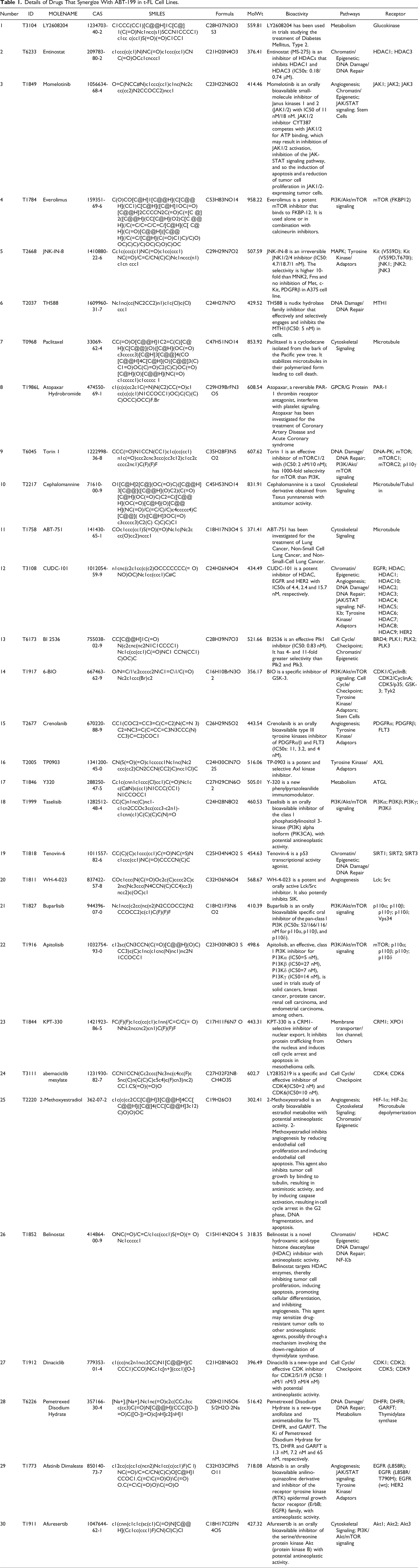
Venetoclax (ABT-199; GDC-0199) was provided by Topscience Co. Ltd. (Shanghai, China) and dissolved in sterile DMSO (Sigma, MO, USA) to produce a 5 mM stock solution stored at −20°C. Venetoclax was combined with a compound library obtained from the School of Pharmaceutical Sciences Xiamen University (Xiamen, Fujian, China). All drugs were approved by the FDA and dissolved in sterile DMSO to produce a 2.5 mM stock solution stored at −80°C. The layout of the compound library 384-well plate in the experiment can be seen in Supplementary Table 1, and the details of the bioactive compound library can be confirmed in Supplementary Table 2. All compounds are currently approved therapies for malignancies.
Anti-Proliferation Assay
All t-FL cell lines were seeded in 384-well plates (Nest Life Technology Co., Ltd., 761 001, Wuxi, Jiangsu, China) at 3000 cells per well in 50 μL of the respective medium using a Multidrop Combi automatic dispenser (Thermo Fisher Scientific, Waltham, MA, USA) with or without venetoclax (20 nM). Then, single-dose drugs of the test compound plates were thawed completely, and .1 μL of the respective drug volume (5 μM) was added to each well using a plate and handler automated instrument (PerkinElmer). Cells were treated for 24 h followed by an assessment of cell viability utilizing the Cell Counting Kit-8 (CCK-8, Zeta Life, CA, USA) assay and incubated for an additional 2 h, after which absorbance at 450 nm (OD value) was detected on a Nivo multifunction microplate reader (PerkinElmer). Cell viability × (%) = [OD value (drug)-OD value (blank)]/[ OD value (DMSO)-OD value (blank)] ×100. Experiments were conducted in triplicate.
Synergy Assessment
Due to there being only one concentration of all drugs in the compound library, we chose a method with higher specificity rather than sensitivity in this synergistic drug screening. To identify antiproliferative combination effects at on-target concentrations, the single-agent dose axis for ABT-199 was limited to a concentration of 20 nM. With this method, a cell viability of less than 80% compared to ABT-199 (which is considered a control) was considered an effect. If the medication is effective, and the value of medication monotherapy subtracted from the value after the combined ABT-199 is greater than 0, we defined the drug to be synergistic with ABT-199.
Network Construction and Pathway Analysis
The interaction network of proteins and the involved pathways were analyzed through the STRING database (https://string-db.org/). Information on drug targets was provided by Topscience Co., Ltd. (Supplementary Table 2). All the interactive relationships were constructed for the network by Cytoscape (Version 3.9.0, MD, USA).
Results
t-FL Cell Lines Exhibited Individual Sensitivities to 320 Drugs
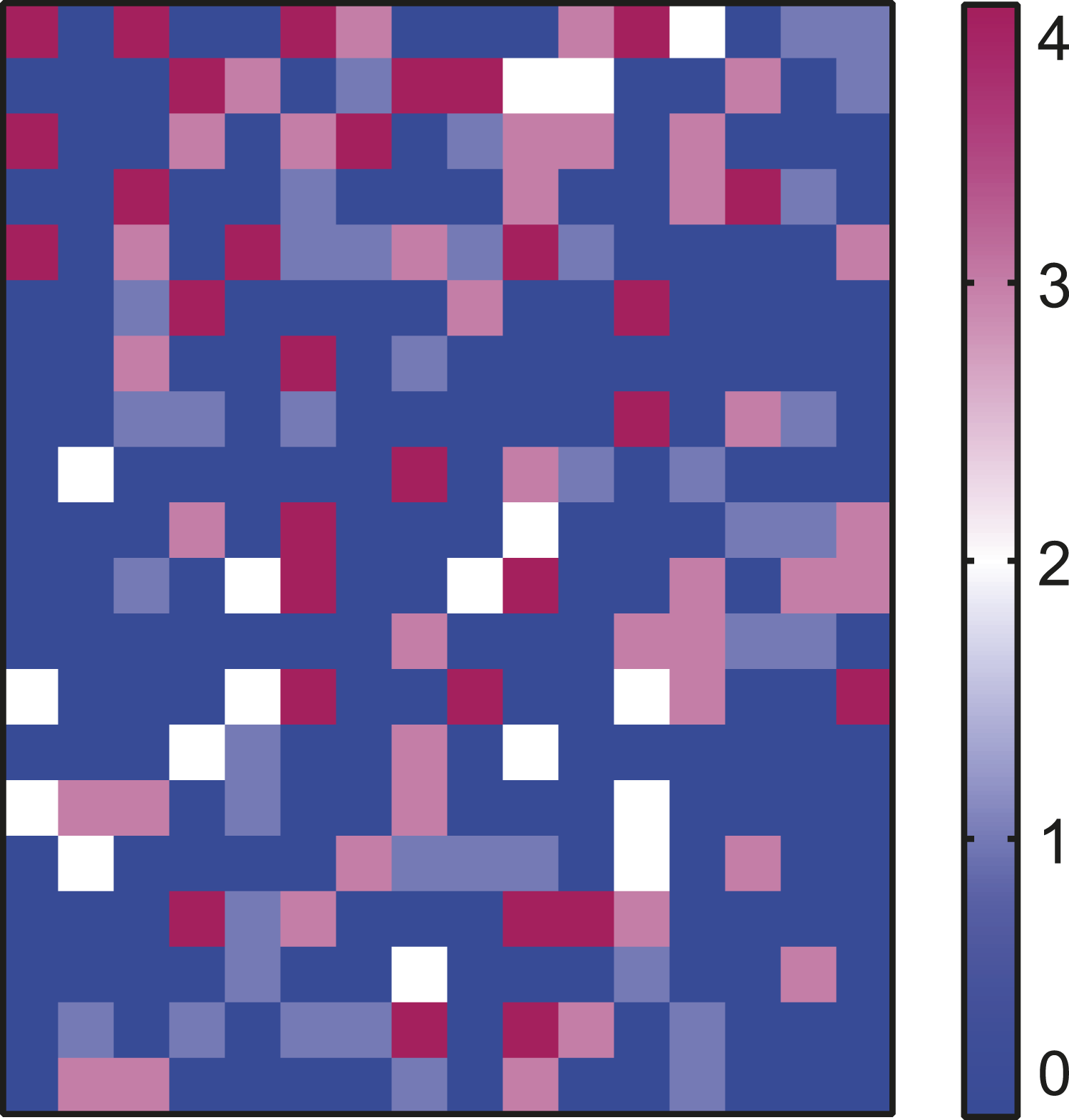
To determine the sensitivity of transformed follicular lymphoma to various drugs, a compound library plate was randomly selected from the School of Pharmaceutical Sciences Xiamen University, which contained 320 compounds in a 384-well plate. Four t-FL cell lines, SU-DHL4, Karpas422, RL, and SC-1, were treated with this compound library (the final concentration was 5 μM) under the drug screening platform. As shown in Figure 1, the four cell lines exhibited completely different sensitivities to the 320 drugs at 24 h. In addition, Karpas422 cells were determined as the most sensitive cell line under the action of most agents. To obtain a more effective treatment strategy for t-FL, this compound plate was combined with the Bcl-2 targeting drug ABT-199, attempting to better gain the coordinated lethality of these 320 drugs and ABT-199. Cell viability assay in t-FL cell lines treated with a compound library. The cell viability percentage of (A) SU-DHL4, (B) Karpas422, (C) RL, and (D) SC-1 cells was measured after treatment with 320 drugs compared to equal volume DMSO. Heat maps were drawn by the GraphPad Prism software according to cell viability percentage.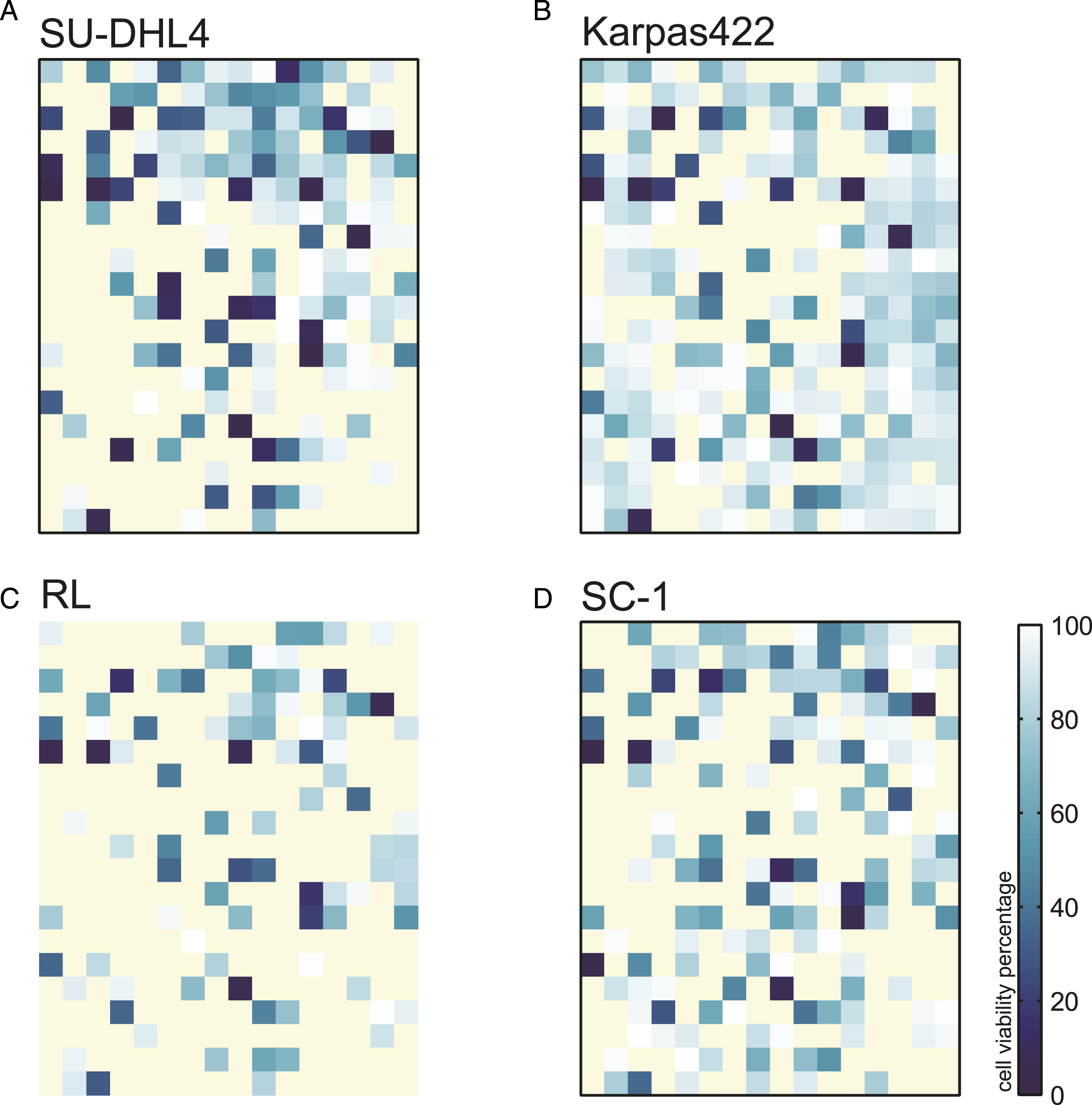
ABT-199 Alters the Effect of the Compound Library on t-FL Cell Lines
To investigate the synergistic drugs for ABT-199, the concentration of ABT-199 was fixed at 20 nM and cotreated for four cell lines with the compound library for 24 h. Then the value of cell viability with a combined treatment regimen was divided by the cell viability with ABT-199 treatment alone to accurately define drugs with an efficacious effect on t-FL cells. Drug percentages above 80% would be deemed to lack cytotoxicity of the t-FL cell line. As expected, it was found that many drugs showed different effects than the original single drugs by combination therapy (Figure 2), suggesting that their target molecules may coordinate with Bcl-2, which is the target molecule of ABT-199. Cell viability of t-FL cell lines treated with ABT-199 combined with compound plates. The combination of ABT-199 (20 nM) with special drugs was applied (A-D) to four t-FL cell lines to obtain the percentage of cell viability relative to ABT-199 treatment alone. The blue color of some squares means the percentage was more than 100%.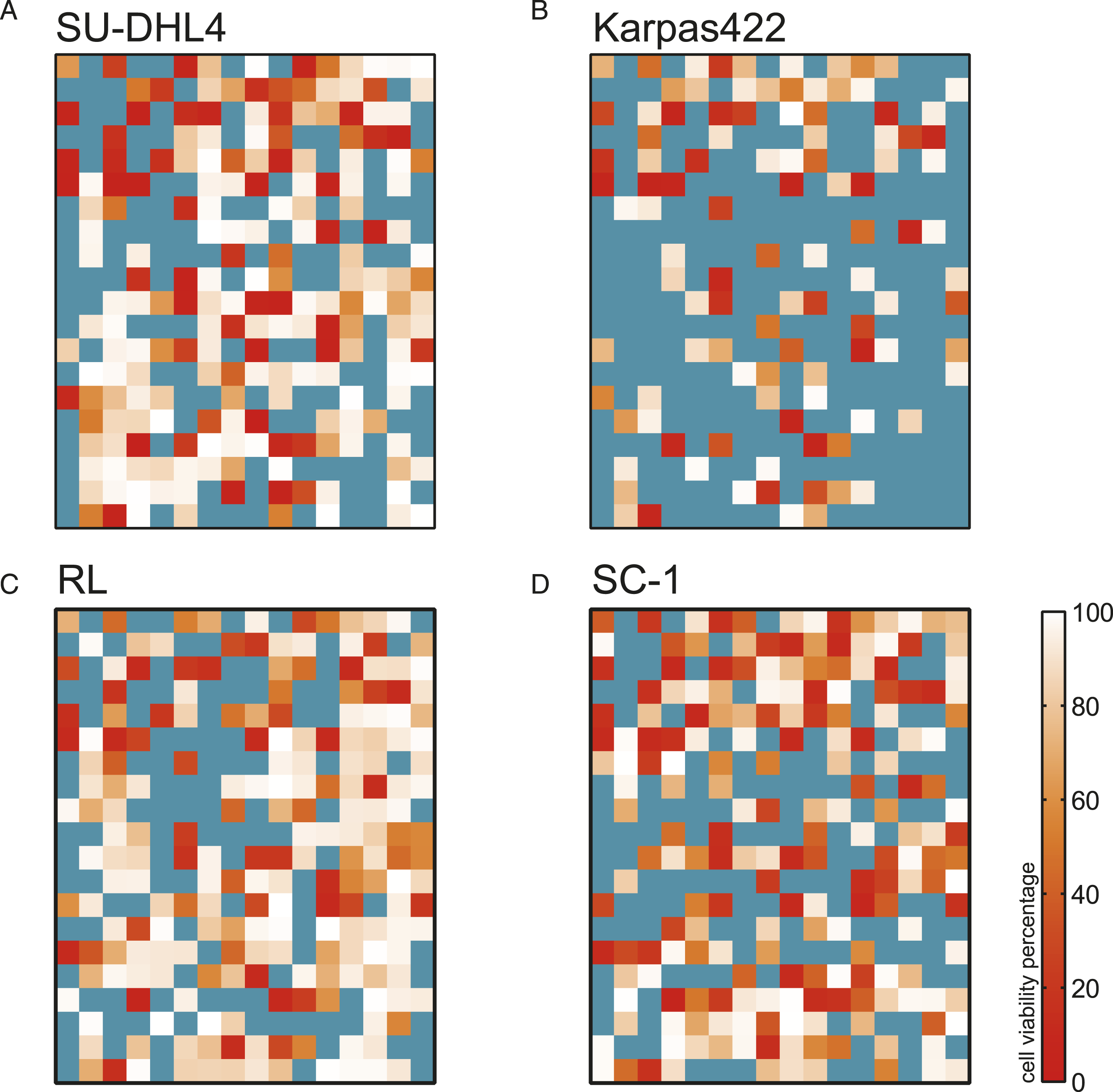
In cases where these combination treatments were effectively lethal to t-FL cell lines, the combined percentages were subtracted from the original percentages of these drugs to define the drugs for which ABT-199 contributed to their effects. As shown in Figure 3, despite being sensitive to most drugs in the library, Karpas422 cells showed the least promotion of drug effects by ABT-199. The effects of ABT-199 on this compound library. (A-D) The difference values of the combined regimen from the original percentage in four t-FL cell lines, in which the value of treatment with ABT-199 alone was subtracted. The blue color in some squares means the value is less than 0.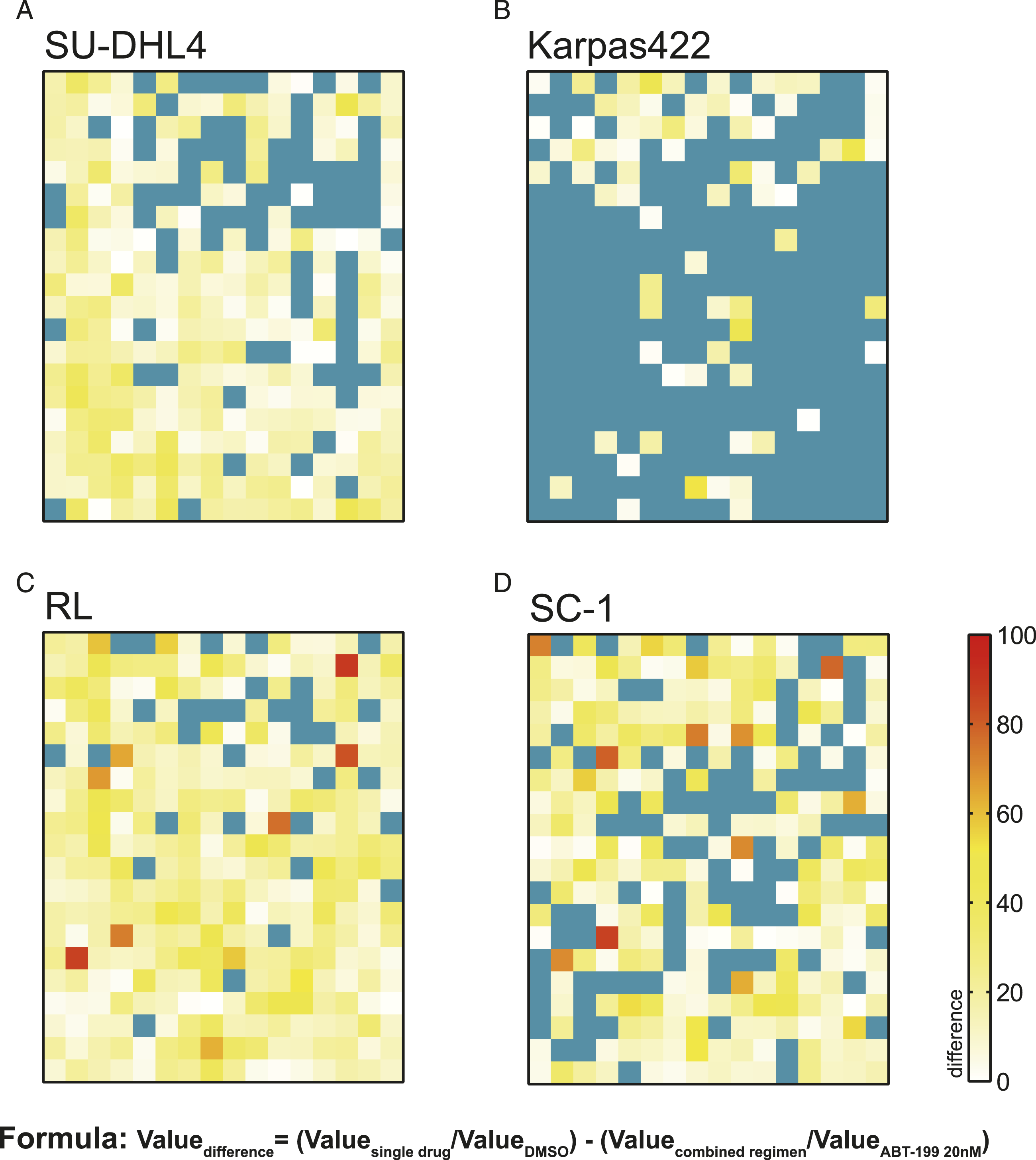
Certain Drugs Show Synergism With ABT-199 in t-FL Cell Lines
To eliminate random errors, a percentage of less than 80% was interpreted to mean that the drug was effective for t-FL. If a drug combined with ABT-199 was effective and ABT-199 promoted the efficacy of this agent, it was judged that the drug and ABT-199 have a synergistic effect on the treatment of t-FL. This approach avoids most combinatorial strategies that exert no cooperation or an antagonistic effect.
As shown in Figure 4, certain drugs were demonstrated to synergize with ABT-199, and in Karpas422 cells, there were only 43 different types of medications that work in synergy with ABT-199. All drugs were subsequently statistically analyzed that synergized with ABT-199 in all four cell lines, and the results are shown in Figure 5. Ignoring less synergistic or antagonistic drugs, 30 drugs were interpreted to be synergistic with ABT-199 in four cell lines (Table 1). Coordination of compound library plates with ABT-199 in t-FL. The association of 320 drugs with ABT-199 demonstrated in (A) SU-DHL4, (B) Karpas422, (C) RL, and (D) SC-1 cells. The yellow color in the square means that the specific drugs are synergistic with ABT-199. The whiteness or darkness indicates that the drug combined with ABT-199 has no effect or antagonism. The compound library exhibited synergism with ABT-199 in all cell lines. The color changes from blue to white to pink, which means that the drug’s synergism with ABT-199 ranges from a single cell line to all cell lines in t-FL. Details of Drugs That Synergize With ABT-199 in t-FL Cell Lines.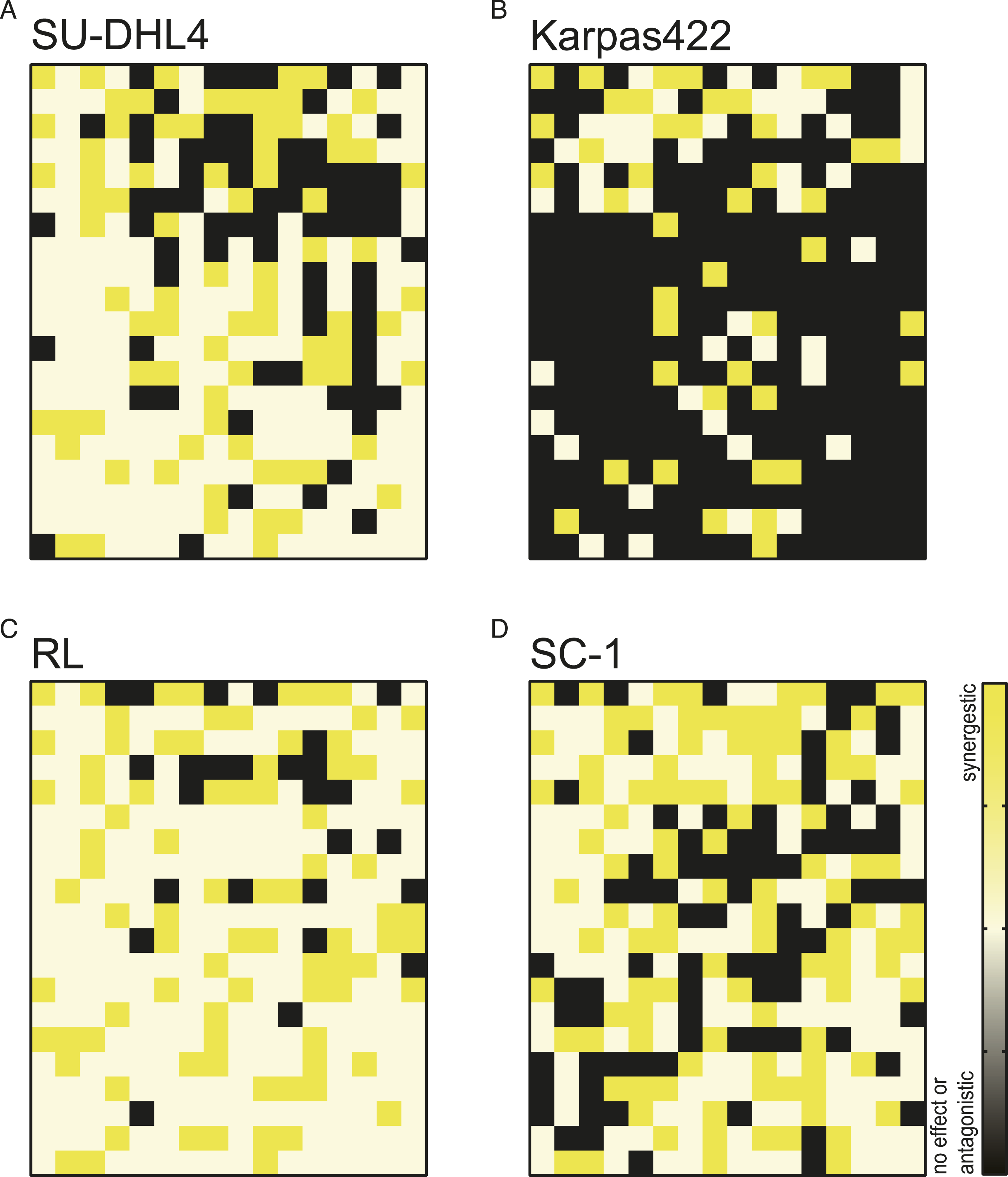
Drugs Synergistic With ABT-199 Target Various Molecules
Due to the noticeable results discovered above, the potential mechanism of synergism was further investigated. Considering that Bcl-2 is the most effective target of ABT-199, how many proteins may be affected by ABT-199 were first identified. Through calculations on the STRING database (https://cn.string-db.org/), proteins that are directly regulated by Bcl-2 were searched for. The 10 proteins most directly regulated by Bcl-2 were identified, including BID, BIK, and TP53 (Figure 6A). Proteins directly affected by Bcl-2 and targets of the compound library plate. (A) Bcl-2 directly affects 10 proteins. (B) The targets of 30 drugs synergistic with ABT-199 are plated in rounds filled with red color, and the drugs in the green square.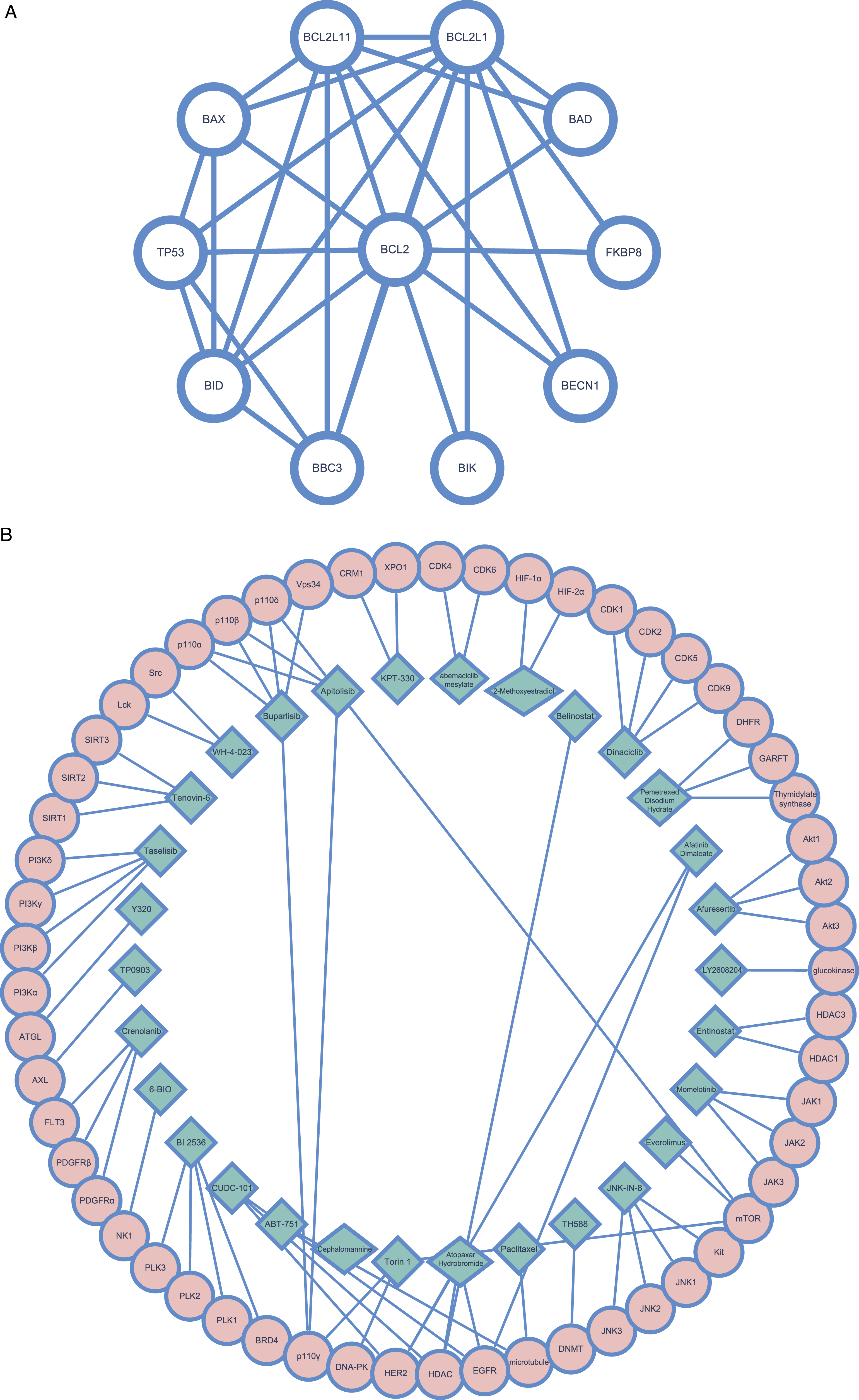
However, these 10 proteins were not present in the target library of a defined set of 30 drugs that synergized with ABT-199. Therefore, all direct targets of the 30 defined agents were found (Figure 6B), including CDK2, JAK, AKT. These results indicate that the mechanism of combination therapy is more complex than the direct interaction with Bcl-2.
The Synergistic Effect of ABT-199 With Each Drug Arises Through a Complex Interaction Network and Pathway
To describe the interaction between ABT-199 and 30 defined drugs, their target molecules were entered into the STRING database to complete their interaction network (Figure 7). Medications with evidence values of less than .9 were discarded to obtain more specific results, resulting in 13 medications with high evidence of coordination with ABT-199 (Table 2). Notably, TP53 is at the center of this network crosslinking map. Interaction regulation of Bcl-2-related networks with 13 drug targets. The interaction network around Bcl-2 shows the drugs taking part in Bcl-2 regulation. Details of 13 Drugs With High Evidence of Coordination With ABT-199.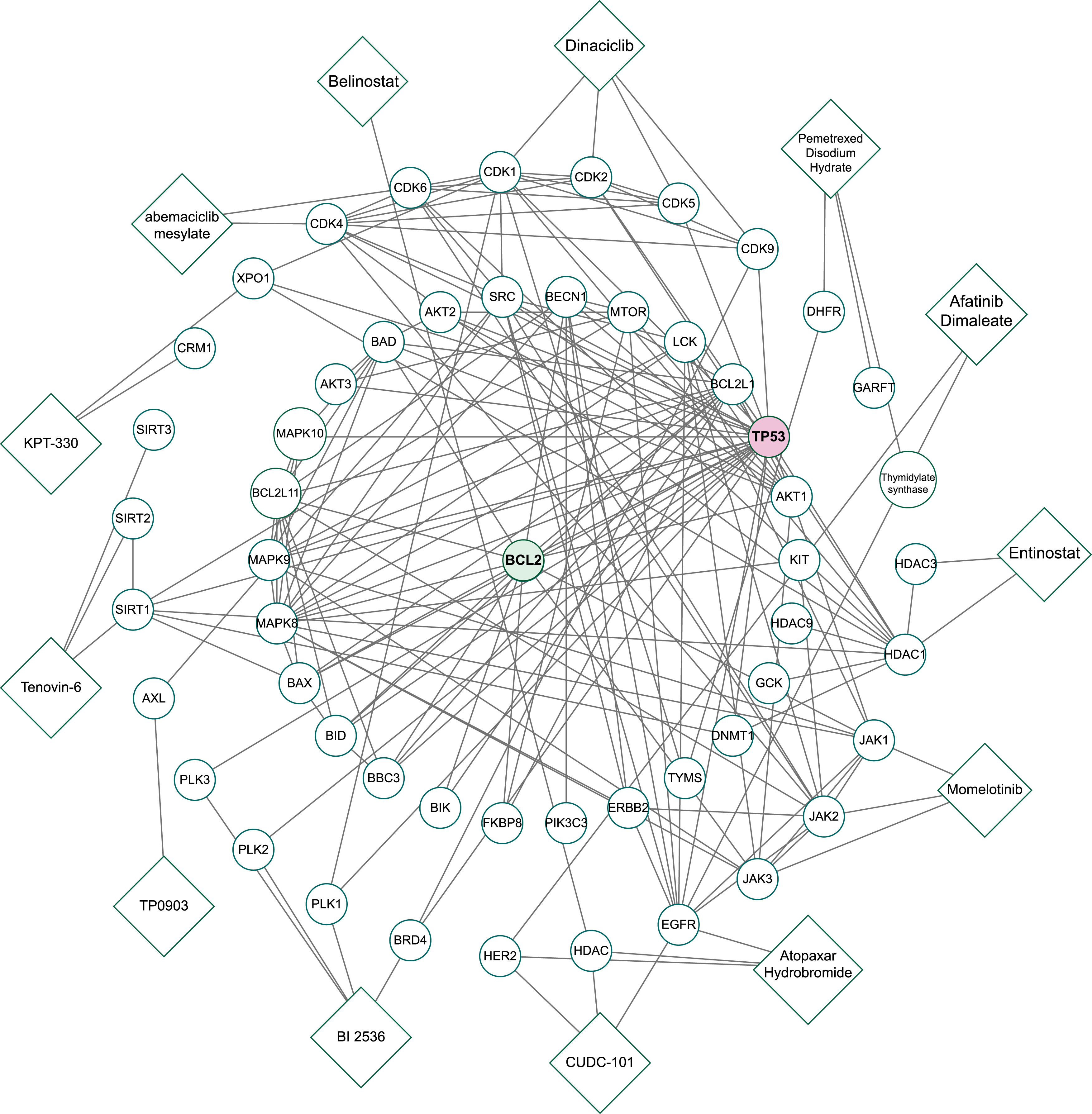
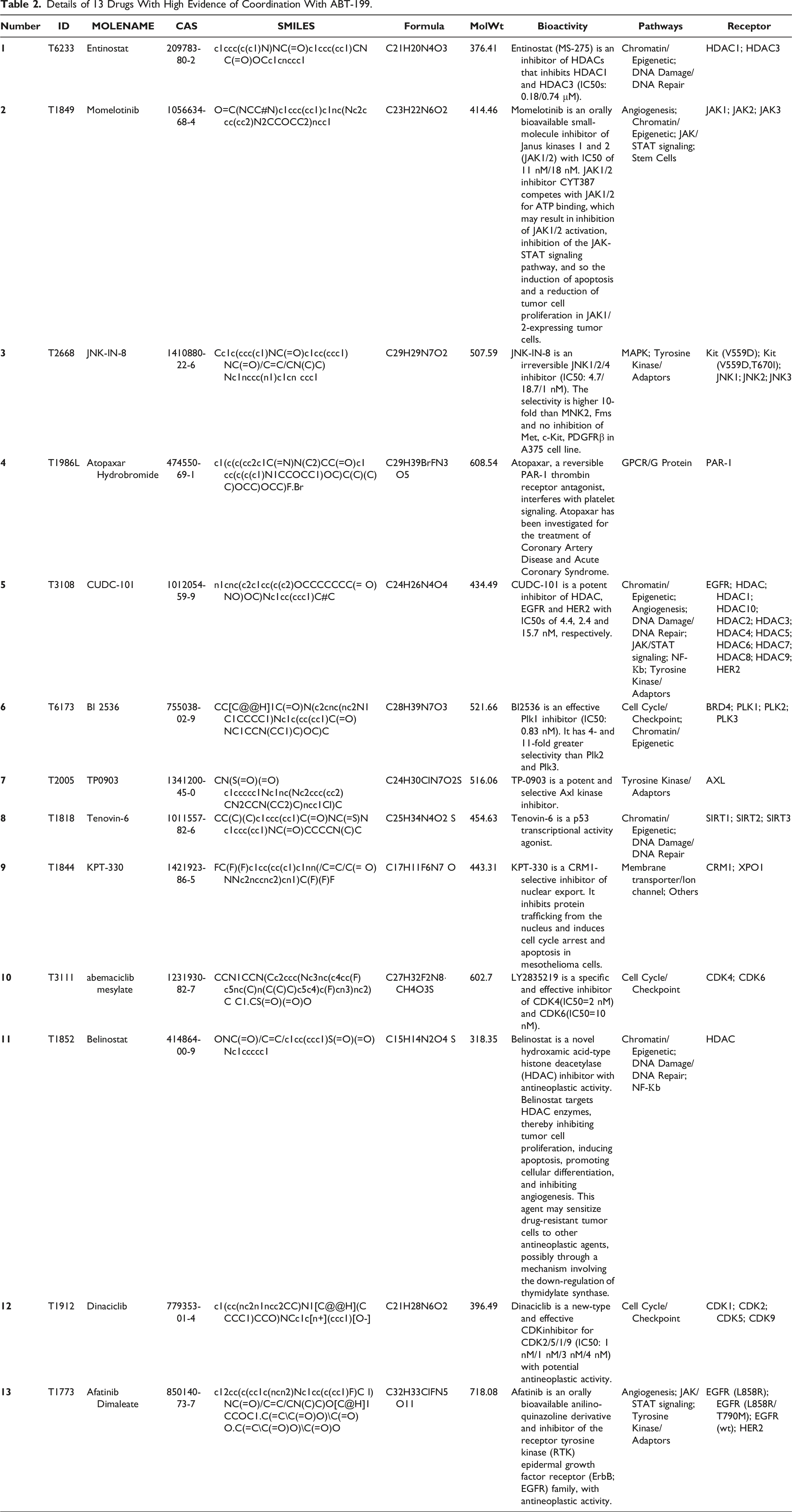
Bcl-2 was placed at the center of the network for a better scan of molecules that interact with ABT-199 targets (Figure 7). None of the drug targets were found to directly interact with Bcl-2, again implying that the mechanism of synergism is more complex. Some proteins present in the network that coordinate directly with Bcl-2 mediate the interactions between some drugs and Bcl-2, such as TP53, BIK, and BID. These proteins may be significant for the synergism of ABT-199 in some pathways.
Finally, the top 20 GO-MF and KEGG pathways were used to describe the processes in which the 30 drugs may be involved (Figure 8). Notably, among them, pathways such as “phosphatidylinositol 3-kinase (PI3K) binding”, “JUN kinase activity”, “p53 signaling pathway” and “EGFR tyrosine kinase inhibitor resistance” have been extensively studied in malignancy, which also implies that their changes may be instructive for a collaborative regimen with ABT-199. Functional analysis based on 30 drug targets. The top 20 GO-MF and KEGG pathways involve targets of 30 drugs synergistic with ABT-199.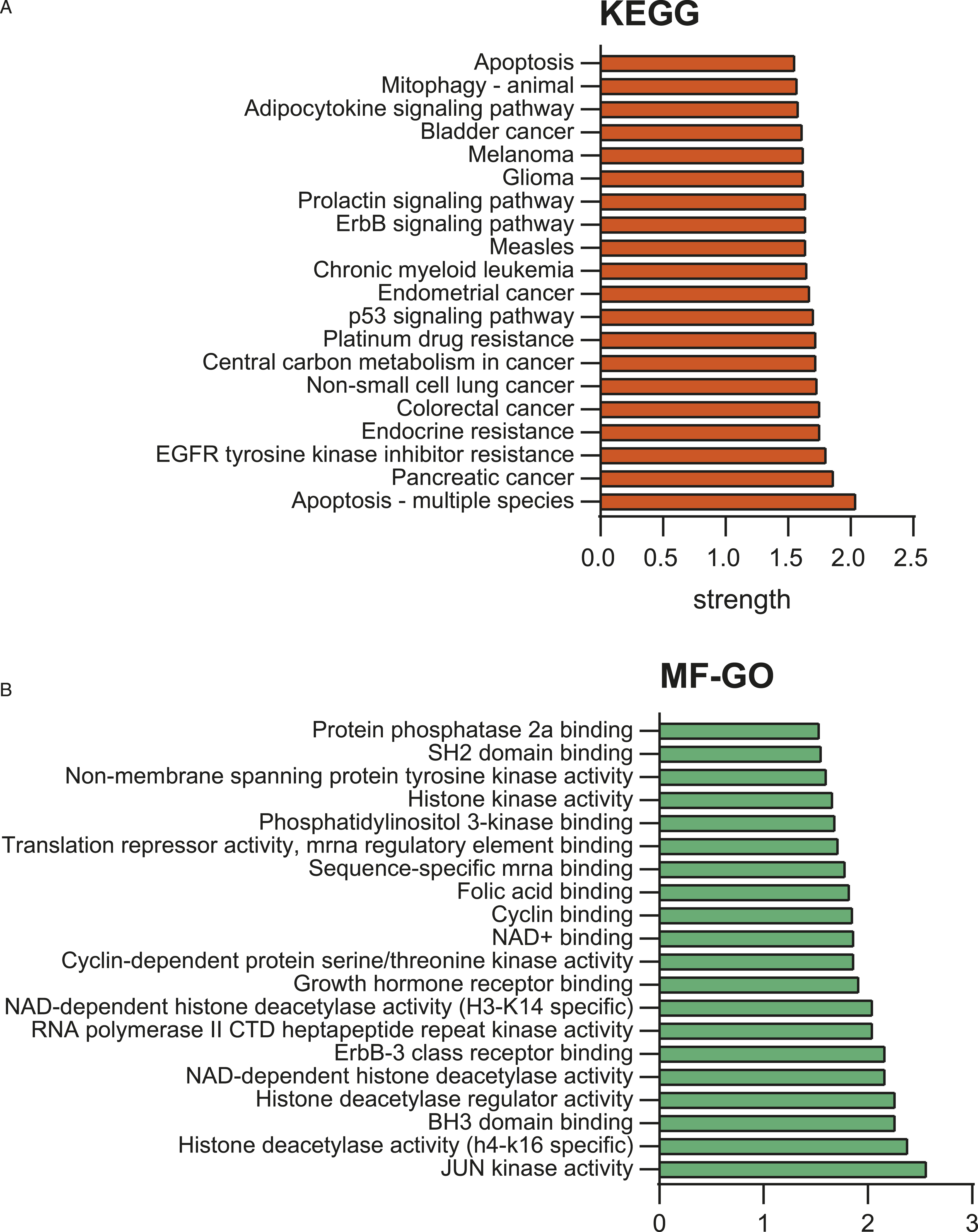
Discussion
t-FL is a devastating malignancy because of its association with treatment-refractory disease and dismal outcomes. 5 This study, for the first time, determined the feasibility of HTS as a precision medicine tool to identify effective targeted drug combinations for the preclinical treatment of patients with t-FL. We randomly selected a compound panel containing 320 U.S. FDA-approved drugs and found that they exhibited different degrees of cytotoxicity on four t-FL cell lines, of which Karpas422 was the most sensitive. Monotherapy is often unable to control the progression of transformed diseases. Some studies have reported that ABT-199 alone rarely exhibits notable efficacy in other B-cell malignancies except chronic lymphocytic leukemia (CLL),21,22 and significant drug resistance has also emerged. 23 This study aimed to identify an effective combination regimen with ABT-199.
Unexpectedly, the most effective cell line during compound library screening, Karpas422, proved to be the least synergistic cells following ABT-199 was administered. We used a computational approach to screen 30 drugs in four cell lines and revealed the specific molecular targets of those 30 drugs, most of which involve HDAC inhibitors, the MAPK/JNK signaling pathway, the PI3K/AKT/mTOR signaling pathway, the cell cycle, and death regulation, which are consistent with the results of combination therapies reported by most of the current studies.24–26 However, the underlying cellular response patterns and target mechanisms of the screened drugs often remain unknown in HTS experiments. Thus, we analyzed whether the targets of these 30 agents have some effect on the ABT-199 target Bcl-2 but found that they are not directly related, indicating that the potential mechanism of the combined regimen is far more than the direct effect on the Bcl-2 protein, and is largely involved in the intersection of multiple pathways or histone modification of intermediate proteins.
Resistance to ABT-199 is a potential concern, as with other targeted therapeutics. Although the initial response might be promising, resistance is often inevitable due to various compensatory mechanisms mediated by complex cancer signaling networks.27,28 We further sought the cross-linking network between the targets of these 30 drugs and Bcl-2 and found that TP53 was located in the middle of this network. Evasion of apoptosis is a hallmark of cancer, and Bcl-2 and p53 represent two important nodes in apoptosis signaling pathways, 29 implying that the combined effect of these drugs lies mainly in modulating their relationship.
Mutations in TP53 are frequent in cancer and hematologic malignancies, where they correlate with unfavorable prognosis and chemotherapy resistance.30,31 TP53mut is generally not detected at diagnosis in t-FL, but mutations may be detected at follow-up and are usually associated with transformation.32,33 Through the Cellosaurus database, we found that in addition to the SC-1-cell line, the other three cell lines have been confirmed to have TP53 mutations; we further verified that transformation is closely related to TP53 mutations. Preclinical studies have suggested that TP53mut confers intrinsic resistance to ABT-199 through the perturbation of mitochondrial homeostasis and cellular metabolism, including increased oxidative phosphorylation. 34 Among the 30 drugs screened in this study, the p53 transcriptional activity agonist Tenovin-6 exerts an antitumor effect by inhibiting the protein deacetylation activity of SirT1 and SirT2. 35 In addition, the activation of p53 also leads to proteasomal degradation of Mcl-1 through activation of GSK3, 29 perhaps inhibiting any Mcl-1 upregulation that may occur as a consequence of exposure to ABT-199 and may further confer the synergy seen in these experiments.
Ultimately, our drug screening results also confirmed the synergistic effect of inhibitors of the PI3K/AKT/mTOR signaling pathway with ABT-199. Considering that most targeted compounds focus on upstream nodes of cancer signaling pathways, we mainly discussed the PI3K kinase here. Studies have confirmed that the delta subunit of class IA PI3K is mainly expressed in hematopoietic cells and is involved in signal transduction, development, and survival of B cells. 36 Excessive activation of PI3Kδ promotes the development of B-cell malignancies. 37 Downstream of PI3Kδ lie several effector molecules, including AKT. This is especially relevant for the study of p53, as AKT phosphorylates MDM2, enhancing its ability to ubiquitinate p53 and target it for proteasomal degradation. 38 Thus, in addition to blocking the upregulation of the antiapoptotic protein Mcl-1, inhibition of PI3Kδ may partly restore p53 activity by stabilizing p53. 39 This is also consistent with our findings.
Conclusion
In conclusion, the goal of this study was to evaluate the cytotoxicity of a combined regimen containing ABT-199 on t-FL utilizing high-throughput drug screening and to further reveal that TP53 plays a role in the combination regimen based on ABT-199. However, there are some limitations to our experiment. First, the HTS analysis is limited by the utilization of a single concentration of the compound library plate. Second, the study was performed on tumor cell lines; further efficacy evaluation and toxicity testing on patient samples are required in the future. Our data complement ABT-199 monotherapy and highlight the need for ABT-199-based combination therapy to improve outcomes in patients with TP53mut t-FL. This approach may uncover new drug combinations that have not been evaluated before, which could motivate the design of future clinical trials.
Supplemental Material
Supplemental Material - High-Throughput Drug Screen for Potential Combinations With Venetoclax Guides the Treatment of Transformed Follicular Lymphoma
Supplemental Material for High-Throughput Drug Screen for Potential Combinations With Venetoclax Guides the Treatment of Transformed Follicular Lymphoma by Zhifeng Li, Guangchao Pan, Mengya Zhong, Li Zhang, Xingxing Yu, Jie Zha, and Bing Xu in International Journal of Toxicology.
Footnotes
Author Contributions
Zhifeng Li contributed to conception and design, contributed to acquisition, and critically revised manuscript; Guangchao Pan contributed to conception and design, contributed to analysis, and drafted manuscript; Mengya Zhong contributed to acquisition, analysis, and interpretation and critically revised manuscript; Li Zhang contributed to acquisition and critically revised manuscript; Xingxing Yu contributed to interpretation and drafted manuscript; Jie Zha contributed to conception and design, contributed to acquisition, and critically revised manuscript; Bing Xu contributed to conception and design, contributed to acquisition, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (U22A20290, 82170180, 82100204), the Natural Science Foundation of Fujian Province (2020J011246, 2021J011359), the Backbone Talent Training Project of Fujian Provincial Health(2020GGB054), and the Xiamen Municipal Bureau of Science and Technology (3502Z20209003, 3502Z20209008).
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
