Abstract
Lead acetate (300 mg/L) and/or cadmium chloride (50 mg/L) were administered as drinking water to Sprague-Dawley rats for 9 weeks to investigate the effects of concurrent exposure to lead and cadmium on the distribution patterns of five essential trace elements. Inductively coupled plasma mass spectrometry was used to determine the concentrations of zinc, copper, manganese, selenium and iron in the urine at different exposure times, as well as their levels in the renal cortex and serum at the end of treatment. Compared with the control group, exposure to lead and/or cadmium resulted in a significant increase in the urinary excretion of these five elements during the experiment, whereas significant decreased levels of these elements were found in kidney and serum. In conclusion, increased urinary loss of antioxidant trace elements due to lead and/or cadmium exposure induced the deficiency of antioxidants in the body, which could result in further oxidative damage. Moreover, there was an obvious synergistic effect of lead combined with cadmium on the distribution patterns of these essential trace elements, which may be related to the severity of co-exposure to these two metals.
Introduction
Along with the rapid development of modern industry, environmental contamination by heavy metals has increased drastically. Some of these metals are lead (Pb) and cadmium (Cd), which are highly toxic, and their levels have increased substantially during the last few years. Industrial contamination of soil, atmosphere and water, foods and plants with Pb and Cd causes their incorporation into the food chain, which can entail a wide variety of adverse effects on animals and humans due to contaminant accumulation. 1 In reality, Pb and Cd are often released simultaneously in the environment from a number of natural and man-made sources, adverse health effects caused by combined exposure to lead and cadmium has provoked a significant public health concern.2–4
It is now well established that kidneys represent the major targets of lead and/or cadmium toxicity.5–8 The nephrotoxicity induced by lead and/or cadmium has been extensively studied and widely reported in many literatures. The majority of these reports were conducted with single lead or cadmium treatment, while only a few studies concentrated on the concomitant exposure. Moreover, in vitro and in vivo studies regarding the toxic mechanism indicated that oxidative stress plays an important role in the nephrotoxicity induced by lead and/or cadmium exposure.6,8–11 Pb and/or Cd can induce oxidative stress conditions in organ targets not only by generating reactive oxygen species but also by reducing cellular antioxidant defenses. 6 While essential trace elements such as zinc (Zn), copper (Cu), manganese (Mn), selenium (Se) and iron (Fe) are intimately associated with oxidant and/or antioxidant processes, they can directly or indirectly protect the mammalian cells from oxidative damage.11,12 However, little information is available on the interactions between mixed metal (Pb + Cd) exposures and the metabolism of these five essential trace elements in the body. Meanwhile, the combined effect of Pb and Cd on the excretion of these trace elements was not well documented. Therefore, the present study was undertaken to determine the effects of co-exposure to Pb and Cd on the homeostasis of trace elements in the body of rats. As ingestion is the most important route of human exposure, we chose for an exposure via the drinking water. In addition, we have used an experimental model of rats treated with Pb and Cd at relatively high levels (300 mg PbAc2/L and 50 mg CdCl2/L) for 9 weeks as a model of Pb/Cd-induced chronic nephropathy.
Materials and methods
Animals and experimental design
This study was approved by the Institutional Ethical Committee on Animal Experimentation, Shandong University Hospital, Faculty of Medicine, Jinan, China. It was carried out on inbred female 30-day-old Sprague-Dawley (SD) rats of initial body weight 90–95 g. Thirty-two rats were housed individually in conventional conditions at a temperature of 22 ± 1°C, with a relative humidity of 50 ± 10% and a 12-h/12-h light/dark cycle. They had free access to drinking water and commercial standard pellet diet (metal contents, in mg/[kg dry weight], Cu 12.2, Mn 82.23, Fe 200.6, Zn 85.6, Se 0.21, Cd 0.018, Pb 0.116).
After a 1-week adaptation period, rats were allocated randomly to four experimental groups of eight animals each, maintained under the supervision of a licensed veterinarian in accordance with the principles set forth in the NIH guide for the care and use of laboratory animals. The experimental period was 9 weeks. (1) Control group: rats consumed distilled water as drinking water. (2) Lead group: rats consumed a solution of lead acetate (PbAc2, 300 mg/L) as drinking water. (3) Cadmium group: rats consumed a solution of cadmium chloride (CdCl2, 50 mg/L) as drinking water. (4) Lead–cadmium group: these animals consumed a solution of Pb and Cd ([300 mg PbAc2 + 50 mg CdCl2]/L) as drinking water during the entire experimental period. Metal solutions were prepared in distilled water and replaced daily to minimize the precipitation of lead and/or cadmium, which were analyzed by inductively coupled plasma mass spectrometry (ICP-MS) method (ELAN-6000 model, Perkin–Elmer, Sciex, Toronto, Canada) to verify their concentrations. Control drinking water was also analyzed to determine the absence of metals. During the experimental period, water consumption and weight gain were measured everyday.
Collection of rat urine
On the day before the experiment (W0) and at the end of 3, 6 and 9 weeks of treatment (W3, W6, W9), rats were kept in individual metabolic cages for 24-h urine collection. The rats were provided with water but no feed was given. The collected urine was free from fecal contamination. Urine samples were centrifuged at 1000 g for 10 min and the aliquots were separated. One portion was used for the determination of various trace elements level immediately. The remaining was frozen at −70°C for the assessment of other indices.
Collection of rat tissues
After the exposure period (9 weeks), rats were fasted overnight and killed by cervical decapitation under ether anesthesia. Blood was collected from the heart to obtain the serum by centrifugation at 2000 g for 15 min. Then the kidneys were removed, weighted, dissected out and washed in ice-cold saline for renal cortex pieces isolation. Some pieces were used for the determination of various trace elements level. The remaining was frozen at −70°C for the assessment of other indices.
Determination of trace elements in the serum, urine and renal cortex
The collected serum and fresh urine samples were digested using HNO3 and H2O2 (Guaranteed reagents) in Teflon PFA microwave digestion vessels. Samples of the renal cortex (about 400 mg wet weight) were dried to constant weight (dry weight) at 80°C for 48 h. Then the dried samples were weighted and digested using HNO3 and H2O2 in Teflon PFA microwave digestion vessels. All digested samples were diluted with deionised water so that the analyte was within the calibration range. Concentrations of trace elements in the serum, urine and renal cortex were determined by ICP-MS method according to the manufacturer’s recommendation. Quality control was strictly carried out using standard reference materials (SRM 1598, NIST).
Statistical analysis
Experimental groups were compared using a two-way analysis of variance (ANOVA) followed by Scheffe’s test when the data were normally distributed and by the Kruskal–Wallis test when they were not normally distributed. All the data were expressed as mean ± SD of number of experiments (n = 8). The statistical significance was evaluated by using SPSS Version 13.0 (SPSS, Cary, NC, USA) and p values < 0.05 were considered to be significant.
Results
Effects of lead and/or cadmium treatment on the performance of rats
In this experiment, the amounts of Pd and/or Cd ingested during the exposure were calculated using the formula mL water consumed/day × metal concentration in the drinking water (mg/mL)/rat weight (kg), as shown in Table 1. As we expected, there was no significant difference in the amounts of Pb or Cd consumed by the rats between the Pb/Cd-treated group and (Pb + Cd)-treated group. Likewise, there was no significant difference in the concentrations of metals in the serum between the different groups (p > 0.05). Also, heavy metal treatment had no effect on weight gain (Table 2) or on water and food consumption in rats (data not shown). Likewise, there were no specific signs attributable to the treatment with Pb and/or Cd. But exposure to Pb and/or Cd induced a significant increase in urinary flow (UF; Table 2).
Metal consumption in treated rats and metal levels in the serum of the experimental rats a

aControl rats showed 4.26 ± 1.32 μg Pb/L in the serum, whereas Cd was not detected in their blood. Results are expressed as mean ± SD (n = 8).
Results of the body weight (BW) and urinary flow (UF) in rats (n = 8) a
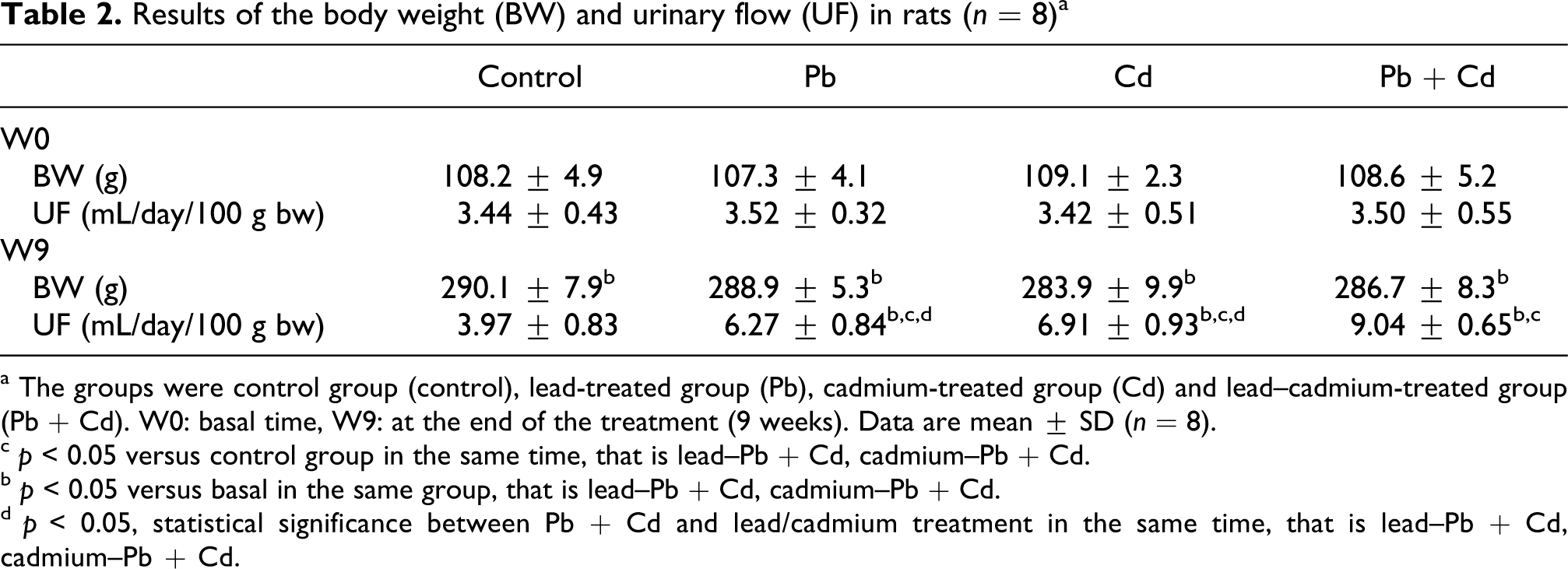
a The groups were control group (control), lead-treated group (Pb), cadmium-treated group (Cd) and lead–cadmium-treated group (Pb + Cd). W0: basal time, W9: at the end of the treatment (9 weeks). Data are mean ± SD (n = 8).
c p < 0.05 versus control group in the same time, that is lead–Pb + Cd, cadmium–Pb + Cd.
b p < 0.05 versus basal in the same group, that is lead–Pb + Cd, cadmium–Pb + Cd.
d p < 0.05, statistical significance between Pb + Cd and lead/cadmium treatment in the same time, that is lead–Pb + Cd, cadmium–Pb + Cd.
Effect of heavy metal treatment on the urinary trace elements excretion
As shown in Figure 1, exposure to Pb and/or Cd for 9 weeks induced a time-dependent increase in urinary trace elements excretion. After 3 weeks' exposure (W3), urinary Zn, Cu, Mn and Se levels in the rats exposed to Pb or Cd were significantly (p < 0.05) higher than those in controls, but urinary Fe was not significantly different (p > 0.05). Moreover, the combined group (Pb + Cd) significantly (p < 0.01) increased urinary Zn, Cu, Mn, Se and Fe levels. Among the five trace elements, only urinary Zn levels between the single treatment (Pb or Cd) and the combined group (Pb + Cd) reached statistical significance after 3 weeks of treatment. After 6 weeks' exposure (W6), urinary Zn, Cu, Mn, Se and Fe levels in the rats exposed to Pb and/or Cd were all significantly (p < 0.01) higher than those of the control group, respectively. Urinary trace elements excretion caused by the combined group was always significantly (p < 0.05) higher than that by the corresponding Pb treatment or Cd treatment after 6 weeks' exposure, respectively. At the end of the treatment (W9), the urinary Zn levels were 157%, 168% and 209% of the control value after exposure to Pb, Cd and Pb + Cd, respectively; the urinary Cu levels were 153%, 158% and 188% of the control, respectively; the urinary Mn levels were 174%, 181% and 236% of the control, respectively; the urinary Se levels were 145%, 173% and 245% of the control, respectively; the urinary Fe levels were 138%, 139% and 158% of the control, respectively. Compared to the single metal exposure, Pb combined with Cd induced an obvious synergistic effect on the urinary trace elements excretion.

Changes in the concentrations of Zn (A), Cu (B), Mn (C), Se (D) and Fe (E) in urine during the experiment. The groups were control group (control), lead-treated group (Pb), cadmium-treated group (Cd) and lead–cadmium-treated group (Pb + Cd). W0: basal time, W3, W6, W9: at the end of 3, 6, 9 weeks of treatment. Data are mean ± SD (n = 8), +p < 0.05 versus basal value (W0) in the same group, #p < 0.05 versus control group in the same time; *p < 0.05, statistical significance between Pb + Cd and lead/cadmium treatment in the same time, that is lead–Pb + Cd, cadmium–Pb + Cd.
Contents of trace elements in rat kidney cortex
The total contents of Zn, Cu, Mn, Se and Fe in the renal cortex of the four experimental groups are shown in Figure 2. Administration with Pb and/or Cd for 9 weeks had a significant (p < 0.01) influence on these five trace elements level in kidney. With the exception of Fe, concentrations of Zn, Cu, Mn and Se in kidney of the (Pb + Cd) group were significantly (p < 0.01) lower than those of the single Pb treatment or Cd exposure, respectively (p < 0.01), that is the Pb–Cd mixture exhibited a synergistic effect on these four trace elements level in kidney. Compared with the control values, renal Zn levels (Figure 2A) were reduced by 10.4%, 12.8% and 26.9%; renal Cu levels (Figure 2B) were reduced by 11.2%, 15.6% and 27.6%; renal Mn levels (Figure 2C) were decreased by 10.1%, 12.6% and 25.2%; renal Se levels (Figure 2D) were decreased by 13.8%, 16.9% and 28.4%; renal Fe levels (Figure 2E) were decreased by 9.49%, 11.8% and 16.9%, exposed to Pb, Cd and Pb + Cd, respectively.

Contents of Zn (A), Cu (B), Mn (C), Se (D) and Fe (E) in the renal cortex of rats at the end of the treatment (9 weeks). The groups were control group (control), lead-treated group (Pb), cadmium-treated group (Cd) and lead–cadmium-treated group (Pb + Cd). Data are mean ± SD (n = 8), #p < 0.05, statistical significance between control and heavy metal treatment, that is control–lead, control–cadmium, control–Pb + Cd; *p < 0.05, **p < 0.01, statistical significance between Pb + Cd and lead/cadmium treatment, that is lead–Pb + Cd, cadmium–Pb + Cd.
Distribution of trace elements in the serum
Compared with the control group, the levels of Zn, Cu, Mn, Se and Fe in the serum decreased significantly (p < 0.01) after 9 weeks of heavy metal exposure, both singly and combined (Figure 3). Furthermore, five serumal trace elements level in the (Pb + Cd) group were all significantly (p < 0.01) lower than those of the Pb group or Cd group, respectively. When exposed to Pb, Cd and Pb + Cd for 9 weeks, serumal Zn levels (Figure 3A) were decreased by 13.5%, 14.0% and 27.3%; serumal Cu levels (Figure 3B) were decreased by 13.8%, 14.1% and 27.6%; serumal Mn levels (Figure 3C) were decreased by 12.7%, 13.7% and 25.2%; serumal Se levels (Figure 3D) were decreased by 13.9%, 15.3% and 31.0%; serumal Fe levels (Figure 3E) were decreased by 9.20%, 11.7% and 20.7%, compared with the control values, respectively. Likewise, the combined treatments of Pb and Cd had a synergistic effect on these five serumal trace elements level.
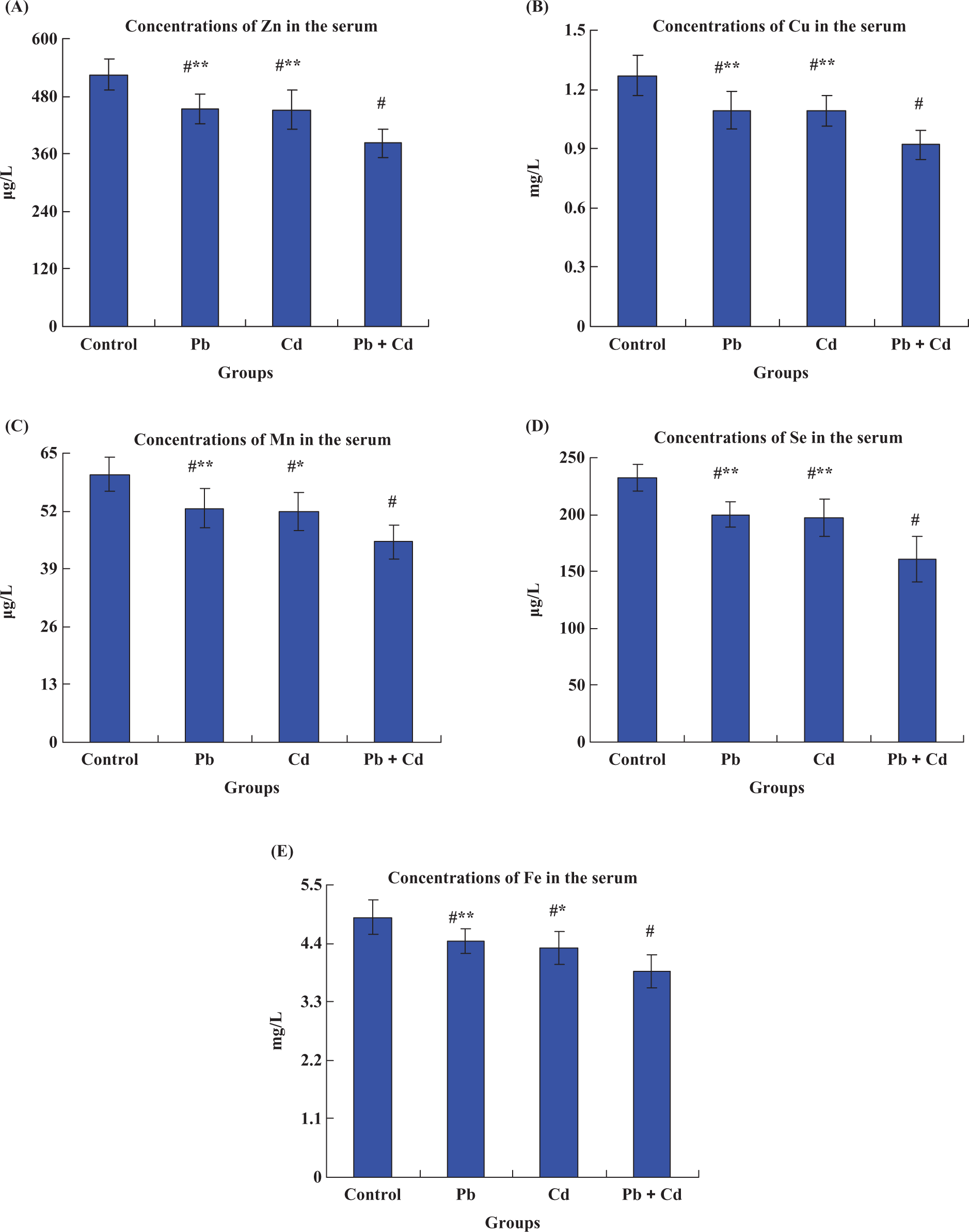
Effects of lead and/or cadmium on the levels of Zn (A), Cu (B), Mn (C), Se (D) and Fe (E) in the serum at the end of treatment (9 weeks). The groups were control group (control), lead-treated group (Pb), cadmium-treated group (Cd) and lead–cadmium-treated group (Pb + Cd). Data are mean ± SD (n = 8), #p < 0.01, statistical significance between control and heavy metal treatment, that is control–lead, control–cadmium, control–Pb + Cd; *p < 0.05, **p < 0.01, statistical significance between Pb + Cd and lead/cadmium treatment, i.e., lead–Pb + Cd, cadmium–Pb + Cd.
Discussion
Pb and Cd are known nephrotoxicants. Both Pb and Cd can generate reactive oxygen species (ROS); moreover, oxidative stress contributes to the pathogenesis of Pb or Cd nephrotoxicity for disrupting the delicate prooxidant/antioxidant balance that exists within mammalian cells.8,13–15 Several previous studies demonstrated that these two metals can enhance lipid peroxidation in kidneys of rats and induce the reduction in the renal activities of superoxide dismutase (SOD), glutathione peroxidase (GSH-Px) and catalase (CAT), since these antioxidant enzymes protect the tissue from oxidative damage via elimination of ROS.8,11,13,14,16 In addition, the activities of these antioxidant enzymes depend on a sufficient supply of the corresponding trace elements, such as Zn, Cu, Mn, Se and Fe.11,14,16–18 Furthermore, our recent published results indicated that there was an obvious synergistic oxidative damage effect of Pb combined with Cd on rat kidneys. 8 This led us to wonder about the simultaneous administration of Pb and Cd interfering with the distribution of trace elements related to oxidative stress (Zn, Cu, Mn, Se and Fe). Herein, it was considered of interest to investigate whether the combined treatment with Pb and Cd makes more adverse effects than that provided by either of them alone in altering the distribution of these five trace elements.
It is known that trace elements play an important role in a number of biological processes. These include the activation or inhibition of enzymatic reactions, competition between elements and metal proteins for binding positions, and modifications in the permeability of cellular membranes. 19 Some essential trace elements, such as Zn, Cu, Mn, Se and Fe, are of physiological importance, which have four major functions as stabilizers, elements of structure, essential elements for hormonal function and enzyme cofactors. 19 Also these five trace elements are essential for normal homeostasis, especially in maintaining the antioxidant defense systems.11,14 For example, Zn acts as an antioxidant, since it is involved in cell membrane stabilization, copper/zinc superoxide dismutase (Cu/Zn SOD) structure, and metallothionein induction. Zn deficiency can be a compromised oxidant defense system, which in turn can be reflected by evidence of cellular or tissue oxidative damage. 20 Cu is involved at the active site of several enzymes. Examples are the largely distributed Cu/Zn SOD, complex IV of the mitochondrial respiratory chain, multi-copper oxidases, such as ceruloplasmin and tyrosinase, and the secreted lysyloxidase. 11 Mn could participate in mitochondrial antioxidant defense via Mn–superoxide dismutase (Mn–SOD). 11 Se is a vital antioxidant which, in mammals, exerts its most important function via selenium-dependent GSH-Px. Se deficiency is usually associated with increased lipid peroxidation which alters the integrity of cell membranes and, consequently, affects cell functions. 21 Since Fe acts as the active center of CAT, Fe deficiency in the tissue could result in the decreased activity of CAT. 18 Hence, it can be concluded that a deficiency of these trace metals as cofactors of antioxidative enzymes could impair the host’s resistance against oxidative stress.
Additionally, it has been indicated that these trace elements can interact with the metabolism of Pb or Cd. Some of them play important roles in alleviating adverse effects of Pb or Cd on the body. Zn and Se are common antagonists to Pb or Cd and exogenous supplementation of Zn and/or Se can compete for and effectively reduce the availability of binding sites for Pb or Cd uptake.13,14,22,23 Fe deficiency can increase the absorption of Pb and Cd to aggravate the biotoxicity of these two metals.24,25 In addition, individuals with several deficiencies of essential trace elements may be at a higher risk for heavy metal-induced adverse health effects. 24 Renal excretion is one of the most important routes of loss in essential trace elements. 26 Hence, concentrations of urinary Zn, Cu, Mn, Se and Fe during the treatment were detected to assess the effects of Pb and/or Cd on the excretion status of them (Figure 1). The urinary element levels were significantly increased in a time-dependent manner after being exposed to Pb and/or Cd. Concerning the single-metal exposure, urinary Zn, Cu, Mn and Se levels are significantly higher than the respective control values following 3 weeks' exposure (W3), with the exception of urinary Fe; whereas five elements level in the Pb + Cd group were significantly increased in the urine. At the end of the treatment (W9), five urinary elements level in the combined group were significantly higher than that in the single Pb or Cd group, respectively, which suggests that synergistic effect lies in the co-administration of both metals. Moreover, exposure to Pb and/or Cd induced a significant increase in urinary volume (UV, i.e., 24-h urine collection) and the UV in the Pb + Cd group was higher than the single metal group (data not shown). Increased urinary excretion of essential trace elements may be due to the impaired tubular capacity for the reabsorption and decrease of glomerular filtration caused by Pb or Cd exposure.16,27 On the other hand, Pb/Cd can interact with the essential trace element to limit its absorption.13,14,22,23,28 Regarding the amount of urinary excretion of five elements, different changes occurred to three exposed groups, that is Mn > Zn > Cu > Se > Fe for Pb exposure, Mn > Se > Zn > Cu > Fe for Cd exposure, Se > Mn > Zn > Cu > Fe for Pb–Cd mixture. This may be related to different binding sites for Pb and/or Cd nephrotoxicity, which will be worthy of further investigation.
Chronic exposure to Pb or Cd predominantly leads to proximal tubular damage. 29 Moreover, the proximal tubule is mainly located in the renal cortex. Our previous study suggested that concurrent exposure to Pb and Cd induced severe lipid peroxidation in the rat kidney cortex, which exhibited a synergistic oxidative damage effect. 8 Herein, renal cortex of rats was collected for the analysis of antioxidant trace elements concentration. As shown in Figure 2, exposure to Pb and/or Cd for 9 weeks (W9), singly and in combination, caused a significant decrease in renal cortex Zn, Cu, Mn, Se and Fe levels (p < 0.05). Furthermore, combination of Pb and Cd produced a synergistic decrease in the concentrations of Zn, Cu, Mn and Se. As far as the decreased rates are concerned, the same variation occurred to Pb, Cd and Pb + Cd, that is Se > Cu > Zn > Mn > Fe. Similarly, significant decrease of these five trace elements level (p < 0.01) in the serum after a 9-week exposure to Pb and/or Cd was shown in Figure 3. Compared to the single metal treatment, combined exposure to Pb and Cd caused a synergistic decrease in the concentrations of Zn, Cu, Mn, Se and Fe (p < 0.05 or p < 0.01). Regarding the decreased rates of these elements level, the same change occurred to Pb, Cd and Pb + Cd, that is Se > Cu > Zn > Mn > Fe. Combined with the results in Figure 1, we can see that increased urinary excretion was intimately related to the deficiency of antioxidant trace elements in the renal cortex and serum. Given their antioxidative properties,11,18–21,23 an insufficiency of these trace elements can directly result in the decreased antioxidative ability in the regional organ (kidney) and whole body (serum). Likewise, this would give an indication whether supplementation with the optimal ratio of these five elements will have an efficient protection against Pb and/or Cd toxicity, which will be worthy of further investigation.
In summary, exposure to Pb and/or Cd induced the increased urinary loss of Zn, Cu, Mn, Se and Fe. Excessive urinary excretion of these antioxidant trace elements led to the insufficiency of antioxidants in the regional soft tissue (kidney) and the whole body, which could result in further oxidative damage. Of course, urinary excretion was only one route of essential trace elements loss due to Pb and/or Cd exposure. The other loss routes need to be explored further. Significant biochemical changes demonstrate the severity of co-exposure to Pb and Cd.
Footnotes
Lin Wang and Xuelei Zhou contributed equally to this work.
This work was supported by the Shandong Provincial Natural Science Foundation, China (No. ZR2010CQ014). Also, this work was supported by Science & Technology Innovation foundation for young scholars of Shandong Agricultural University, China (No.23690).
The authors declared no conflicts of interest.
