Abstract
Agents that act at the N-methyl-D-aspartate receptor (NMDAR), such as ketamine, have gained increasing attention as rapid-acting antidepressants; however, their use has been limited by potential neurotoxicity. Recent FDA guidance requires a demonstration of safety on histologic parameters prior to the initiation of human studies. D-cycloserine (DCS) is a partial NMDA agonist that, along with lurasidone, is being investigated as a treatment for depression. The current study was designed to investigate the neurologic safety profile of DCS. To this end, female Sprague Dawley rats (n = 106) were randomly divided into 8 study groups. Ketamine was administered via tail vein infusion. DCS and lurasidone were administered via oral gavage in escalating doses to a maximum of 2000 mg/kg DCS. To ascertain toxicity, dose escalation with 3 different doses of D-cycloserine/lurasidone was given in combination with ketamine. MK-801, a known neurotoxic NMDA antagonist, was administered as a positive control. Brain tissue was sectioned and stained with H&E, silver, and Fluoro-Jade B stains. No fatalities were observed in any group. No microscopic abnormalities were found in the brain of animal subjects given ketamine, ketamine followed by DCS/lurasidone, or DCS/lurasidone alone. Neuronal necrosis, as expected, was seen in the MK-801 (positive control) group. We conclude that NRX-101, a fixed-dose combination of DCS/lurasidone, when administered with or without prior infusion of IV ketamine was tolerated and did not induce neurotoxicity, even at supratherapeutic doses of DCS.
Introduction
Drugs that act at the NMDAR are gaining interest for the treatment of depression, PTSD, chronic pain, and other conditions. Ketamine, an NMDAR antagonist, was serendipitously discovered to have potent and rapidly acting antidepressant effects.1,2 Indeed, ketamine is one of the longest used and best-known NMDA antagonist drugs. While single infusions of ketamine commonly used as anesthesia do not induce neurotoxicity, chronic administration was shown to cause neuronal apoptotic lesions under experimental conditions. 3 NMDAR antagonists including phencyclidine, MK-801, and ketamine cause vacuolization lesions in the posterior cingulate and retrosplenial cortices (Olney’s lesions) in rodents peaking at 3-4 days after adminstration.4,5 Because of the side effects, potential neurotoxicity, and habituation, ketamine is not deemed suitable for chronic use in the treatment of depression. There is a need for safer, orally bioavailable drugs that mimic or at least prolong the ketamine antidepressant effect.
D-cycloserine (DCS) is a broad spectrum anti-infective used to treat tuberculosis and urinary tract infections since the 1950s. DCS has subsequently been identified as a partial agonist of the NMDAR and appears to act via allosteric modification of the glycine-binding site. 6 As a partial agonist at NMDAR, DCS acts like an agonist at low doses but has antagonistic features with high doses. 7 DCS has been shown to raise Glx in the brain in a manner comparable to ketamine with single oral administration. 8 The antidepressant effect of DCS appears to be linked to its propensity to raise glutamate + glutamine (Glx) in the anterior cingulate cortex. Subnormal levels of Glx have been associated with depression in multiple case/control studies. 9
NRX-101 is a fixed-dose combination of D-cycloserine (DCS) and lurasidone that has been awarded Fast Track designation, Breakthrough Therapy Designation, a Special Protocol Agreement, and a Biomarker Letter of Support by the US Food and Drug Administration (FDA) for the treatment of Severe Bipolar Depression with Acute Suicidal Ideation or Behavior. The drug is now entering Phase 3 clinical trials. The components of NRX-101 are well-understood compounds that have been used safely in patients for decades. NRX-101 was developed as a novel oral antidepressant based on a fixed-dose combination of DCS and lurasidone. The latter component is a D2/5HT2A receptor inhibitor that appears to block the hallucinatory side effects which might otherwise be seen with administration of DCS at high doses. While lurasidone’s precise mechanism of action is unknown, its efficacy suggests involvement in the mediation of central dopamine type 2 and serotonin type 2 (5HT-2A) receptor antagonism.
Because of the potential for agents that act at the NMDAR to cause excitatory neurotoxicity, the FDA has issued guidance that requires NMDAR therapeutics be shown to lack histologic neurotoxicity prior to human studies. 10 In one clinical application, a single infusion of ketamine is followed by chronic administration of NRX-101. Thus, this study reports the clinical safety of NRX-101 administered by or following ketamine infusion. MK-801 (dizocilpine; INN), an uncompetitive antagonist of NMDA receptor known to produce Olney’s lesions in test rats, was used as a positive control. This study is designed as an IND-enabling study to demonstrate the neurological safety of increasing doses of DCS when combined with lurasidone by itself or when preceded by an IV infusion of ketamine.
Materials and Methods
Ethics and Regulatory Compliance
National Institutes of Health guide for the care and use of Laboratory animals (NIH Publications No. 8023, revised 1978) was strictly observed in this study. All portions of this study and histopathology evaluation were conducted in compliance with OECD Principles of Good Laboratory Practice, as revised in 1997 and adopted November 26th, 1997, by decision of OECD Council (C 97) 186/Final and US FDA Good Laboratory Practice Regulations 21 CFR 58, effective June 20, 1979, as amended 52 FR 33780, September 4, 1987, and subsequent amendments. The study was performed under the sponsorship of NRx Pharma, Inc. (Wilmington, DE), according to a protocol reviewed and accepted in advance by the US Food and Drug Administration. All experimental subjects were treated in accordance with the standards set forth in the eighth edition of “Guide for the Care and Use of Laboratory Animals” and their ethical treatment was overseen by the animal rights committee of WuXi AppTec, which operates in full accord with US and international law. The study protocol was approved by the Animal Research Ethics Committee of China in accordance with China experimental animal administrative regulations.
Animals
Study Drug Doses for Ketamine, DCS/Lurasidone, and MK-801.

Study Drug Administration
Drug administration was performed by WuXi AppTec (Shanghai, CN) under the supervision of NRx Pharma, Inc. Ketamine, DCS/lurasidone, and MK-801 were administered by IV infusion (40 minutes), oral gavage, and subcutaneous injection, respectively. The group assignment and dosing regimen are summarized in Table 1. Animals in Groups 3 through 5 were given 12.5 mg/kg ketamine on Day 1, followed by 500/35 mg/kg, 1000/69 mg/kg, or 2000/139 mg/kg DCS/lurasidone on Day 2 (approximately 24 hours after ketamine infusion). Animals in Group 7 were administered 2000/139 mg/kg DCS/lurasidone only on Day 2. Animals in Groups 2 and 8 were administered 1 and 5 mg/kg MK-801 on Day 1, respectively. Initially, ketamine was dosed to 6 TK rats at 35 mg/kg via an IV bolus injection. Due to unscheduled deaths observed following this dose, the ketamine dose was reduced to 12.5 mg/kg and the dosing route was changed to slow IV infusion (40 minutes) to mimic the clinical dosing route. Animals were reassigned to replace those already dosed with 35 mg/kg ketamine. The day of ketamine administration was designated as Day 1 for all groups.
Study Endpoints
Parameters evaluated during the study included mortality, clinical observations, body weights, food consumption, ophthalmology examinations, clinical pathology (hematology, coagulation, serum chemistry, and urinalysis), gross pathology, histopathology of brain and spleen, and toxicokinetics (TK).
Toxicokinetics
Blood samples were collected from TK animals to evaluate systemic exposure of ketamine and DCS/lurasidone. Toxicokinetics for ketamine were assessed at baseline, 15 min, 1 hour, 4 hours, and 24 hours post IV infusion. Toxicokinetics for NRX-101 were assessed at baseline, 0.5 hours, 1, 2, 4, 8, and 24 hours post oral gavage. At each time point, approximately .3 mL of blood was collected from animals from the jugular vein. Blood was collected into appropriately labeled tubes containing K2EDTA as the anticoagulant. The tubes were gently inverted several times to ensure mixing and immediately placed on ice. The quantification of ketamine, D-cycloserine, and lurasidone concentrations was conducted at the Testing Facility using validated liquid chromatographic triple quadrupole mass spectrometric (LC-MS/MS) methods (methods no. 409-0013-M for D-cycloserine and 409-0015-M for ketamine and lurasidone).
Pathology
All animals were anesthetized by sodium pentobarbital and perfused with 10% neutral buffered formalin to clear erythrocytes and preserve tissues. A single section of hematoxylin and eosin (H&E)-stained spleen was evaluated for each animal. The brain was trimmed into 8 sections as a modification of the procedure described by Bolon et al (2013) and Carson and Cappellano (2015) and placed in paraffin blocks. The paraffin blocks containing posterior cingulate and retrosplenial cortices were subjected to at least 3 non-serial sections for each level about 50-100 microns apart. Thus, a total of approximately 14 planes of section were evaluated microscopically from each brain with a focus on assessing potential Olney lesions.4,11 Three microscopic sections prepared from each plane of section: H&E, silver, and Fluoro-Jade B (FJB). Neurodegeneration was characterized by nuclear pyknosis and condensed, brightly eosinophilic cytoplasm by H&E. The Sevier-Munger modification of Bielschowsky’s silver method was used to visualize neurites and neuronal processes. FJB was used to identify degenerating/necrotic neurons, which were brightly fluorescent. 12 Using Provantis™ v9.3.1.1 pathology software, microscopic findings were given a severity score of minimal, mild, moderate, and marked. At the discretion of the study pathologist (WJ), epifluorescent evaluation of H&E-stained microscopic sections was conducted to aid in interpretation of potential neurodegeneration, a procedure described by Jordan and Hall. 13
Results
Toxicokinetics of Ketamine, DCS, and Lurasidone.
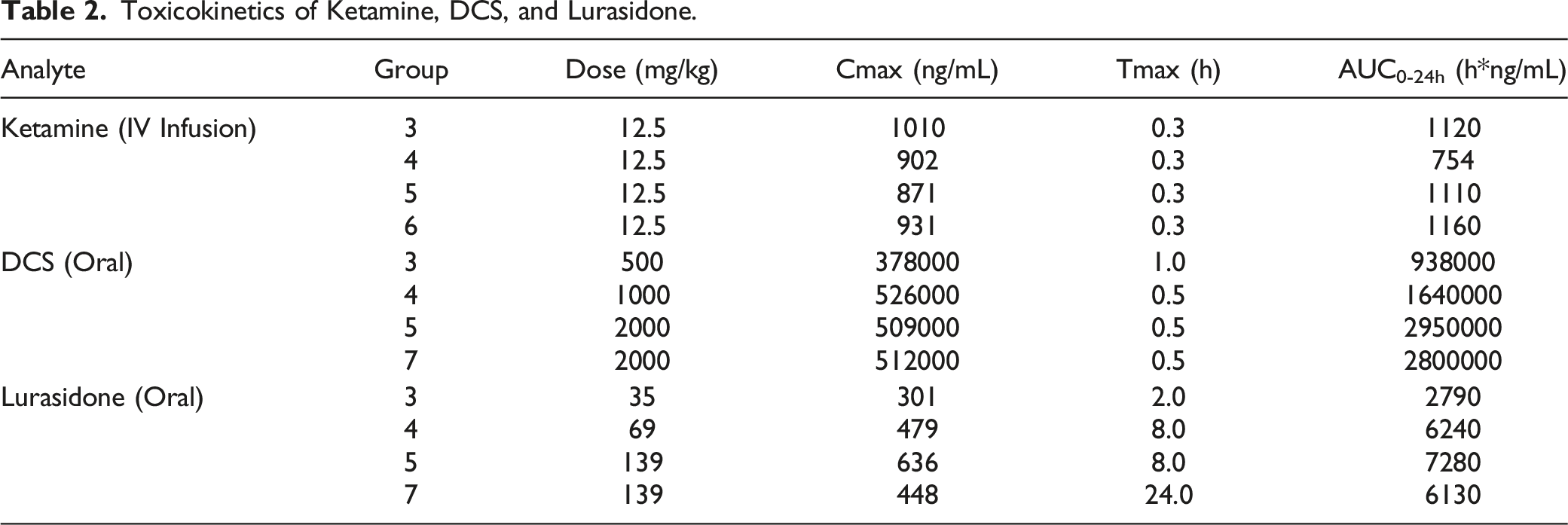
Ketamine did not appreciably change the toxicokinetics of DCS [Group 5 (with pre-dose ketamine): Cmax 509000 ng/mL, Tmax 0.5 h, AUC0-24h 2950000 h*ng/mL; Group 7 (no pre-dose ketamine): Cmax 512000 ng/mL, Tmax 0.5 h, AUC0-24h 280000 h*ng/mL]. However, ketamine did tend to increase the Cmax and AUC0-24h of lurasidone and reduce the Tmax (Group 5 (with pre-dose ketamine): Cmax 636 ng/mL, Tmax 8.0 h, AUC0-24h 7280 h*ng/mL; Group 7 (no pre-dose ketamine): Cmax 448 ng/mL, Tmax 24.0 h, AUC0-24h 6130 h*ng/mL). Thus, the systemic exposure to lurasidone was slightly higher with ketamine pretreatment.
Clinical Findings
Following oral dosing of DCS/lurasidone, dose-responsive test article-related clinical signs included soiled coat at ≥500/35 mg/kg, ocular and/or nasal discharge at ≥1000/69 mg/kg, decreased activity and material around nose at 2000/139 mg/kg in animals dosed with or without ketamine, and prostration, atonia, and closed eyes in animals dosed 2000/139 mg/kg DCS/lurasidone alone. Clinical signs observed in the positive control groups (MK-801) included prostration, atonia, decreased activity, abnormal gait, salivation, ocular discharge, and material around nose at ≥1 mg/kg.
Decreased body weight of 5 to 8% (relative to start weight) was noted following oral dosing of DCS/lurasidone at ≥1000/69 mg/kg, which was accompanied by reduced food consumption of 31 to 77% (relative to concurrent control value) noted at ≥500/35 mg/kg (Figure 1(A)). Decreased body weight (10 to 16%) and food consumption (more than 95%) were also noted in the positive control (MK-801) groups (Figure 1(B)). There were no test article-related morphological changes in ophthalmology examinations. Consistent with the clinical observations, red or brown ocular discharge and excessive lacrimation were principally noted in rats given 2000/139 mg/kg DCS/lurasidone with or without ketamine. Similar ocular findings were also observed in the positive control groups. (A) Mean body weight prior to and after test article administration. (B) Mean food consumption prior to and after test article administration.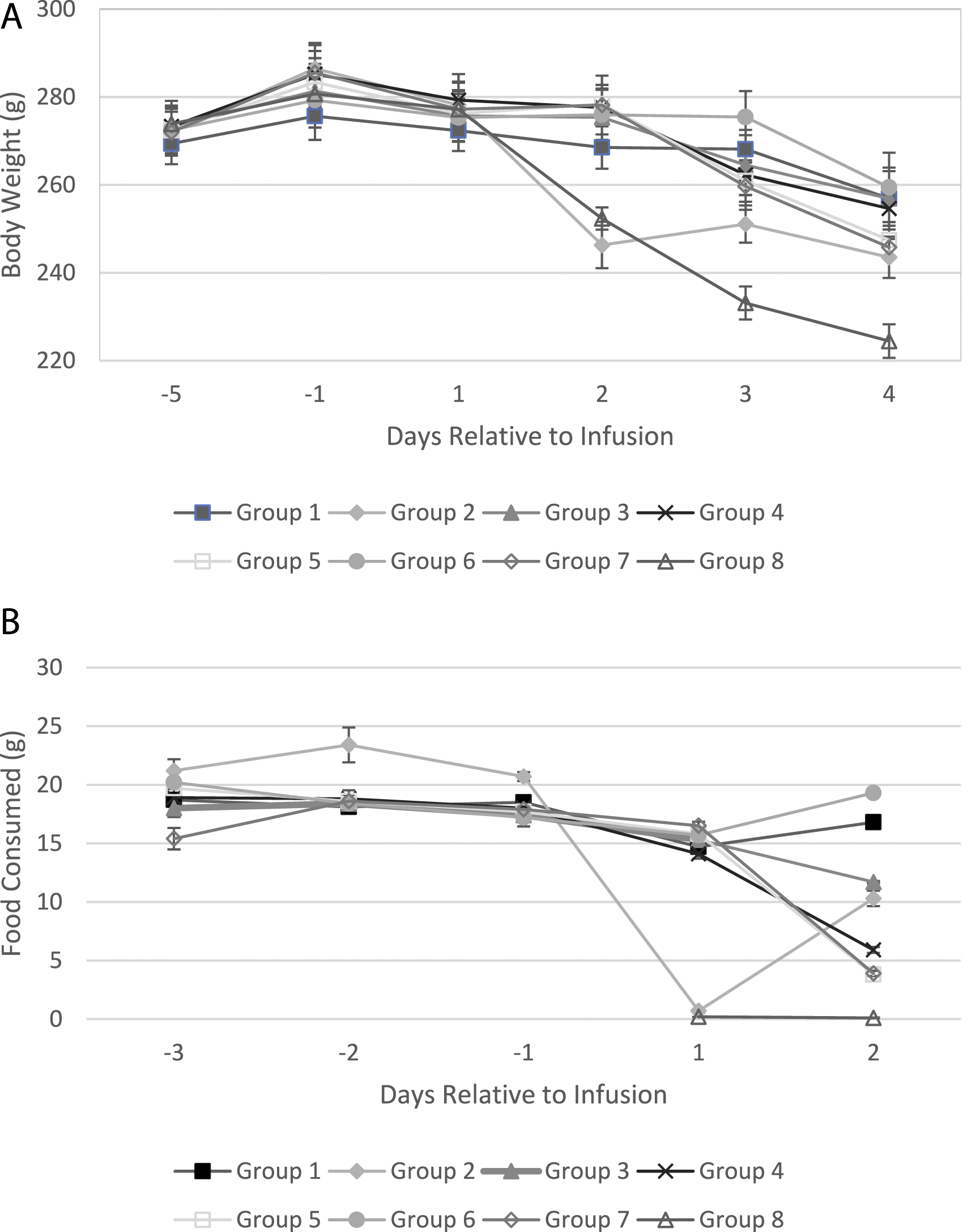
Test article-related hematology changes included increased neutrophils and decreased lymphocytes at ≥500/35 mg/kg DCS/lurasidone, and decreased reticulocyte count, increased eosinophils at 2000/139 mg/kg DCS/lurasidone with or without ketamine, and possibly increased platelets at ≥500/35 mg/kg DCS/lurasidone. Increased neutrophils and decreased lymphocytes were likely direct effects of the test article or related to a stress response. Decreased reticulocyte count was likely related to decreased food consumption. Increased neutrophils and decreased reticulocytes were also noted in the positive control groups. Test article-related coagulation changes were restricted to decreased prothrombin time noted at ≥500/35 mg/kg DCS/lurasidone. Decreased prothrombin time and increased fibrinogen were noted in the 5 mg/kg MK-801 dose group.
Test article-related serum chemistry changes included increased serum total bilirubin at doses ≥1000/69 mg/kg of DCS/lurasidone, increased serum urea in all DCS/lurasidone dose groups, increased serum triglyceride at 2000/139 mg/kg DCS/lurasidone with or without ketamine, and decreased serum alanine aminotransferase at ≥1000/69 mg/kg DCS/lurasidone. Increased total bilirubin and triglyceride may suggest effects on the bile duct. Increased urea may suggest renal nephropathy. Decreased alanine aminotransferase was not considered of toxicological significance. Test article-related changes in urinalysis included increased urine protein, urine urobilinogen, and urine glucose at ≥1000/69 mg/kg DCS/lurasidone, which may suggest renal nephropathy. Increased urine protein and urine glucose were also noted in the 5 mg/kg MK-801 dose group. No visible lesions were detected in any of the kidney samples.
Histopathology
Microscopic abnormalities were limited to the expected findings in rats administered MK-801 using Recommended Neuroanatomical Sampling Practices for Comprehensive Brain Evaluation in Nonclinical Safety Studies.
14
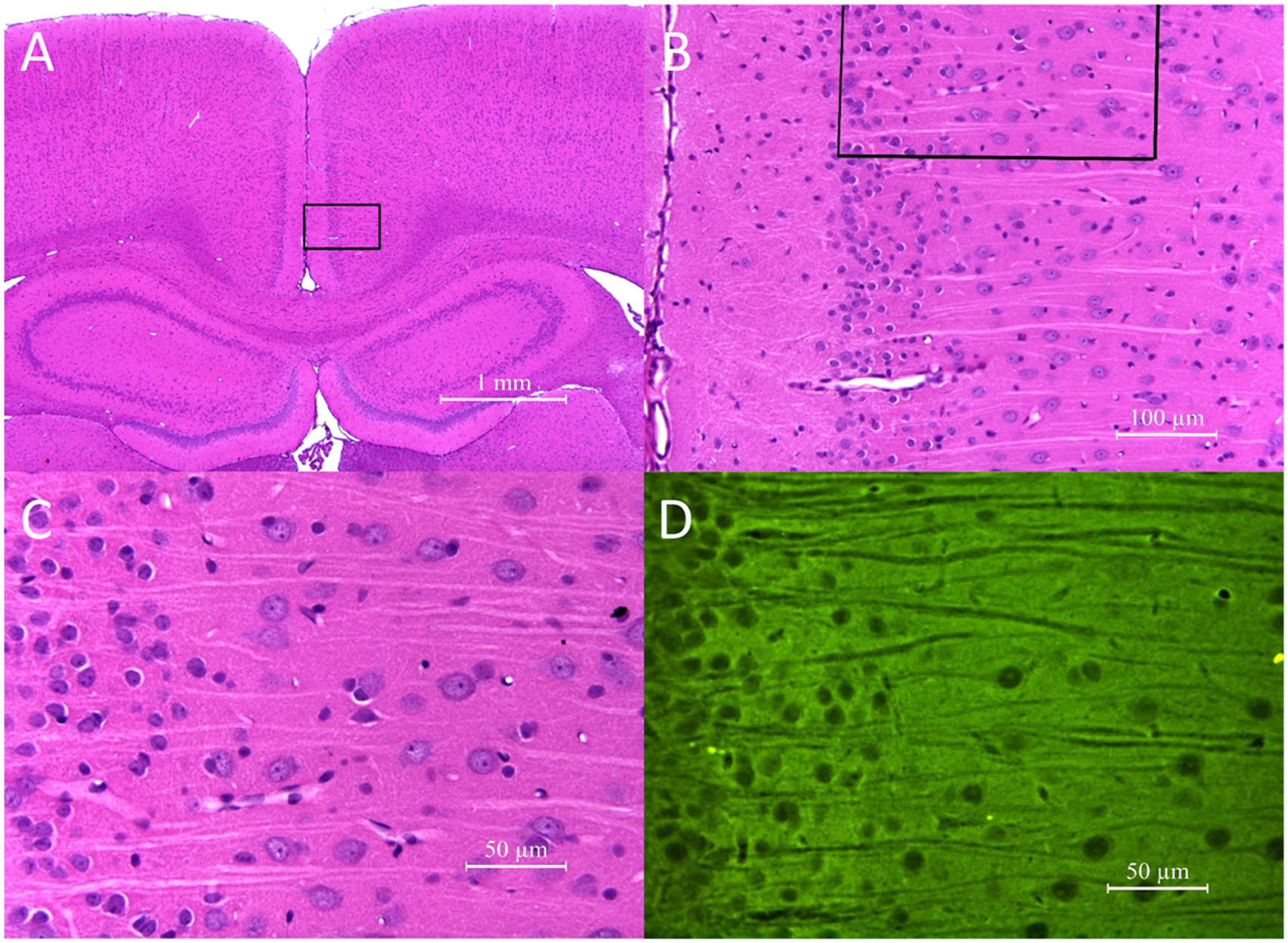
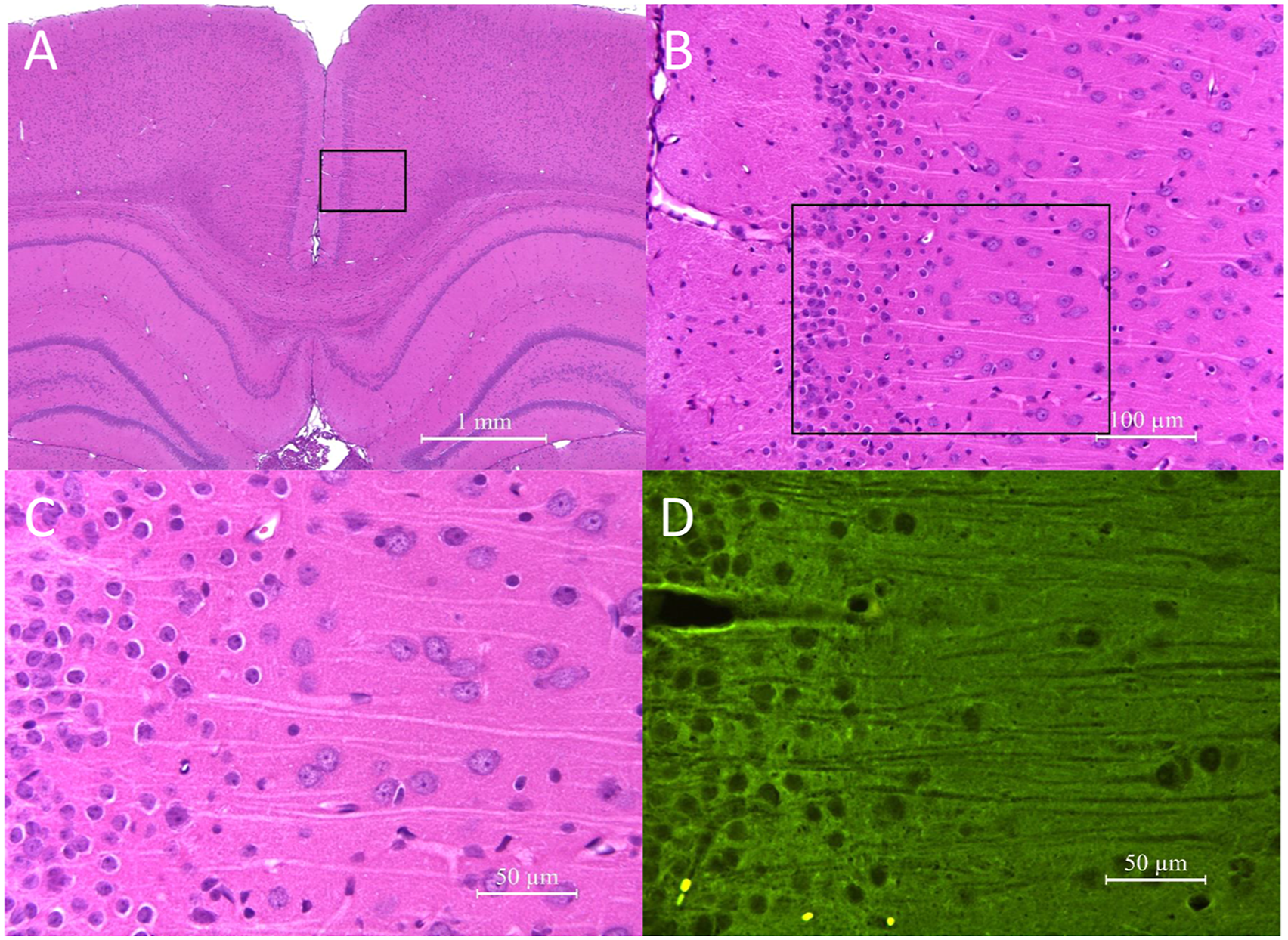
Neuronal degeneration/necrosis, characterized by neuronal nuclear pyknosis, cytoplasmic condensation, and cytoplasmic hypereosinophilia, occurred in the posterior cingulate and retrosplenial cortices of all animals administered MK-801. The sensitivity of the testing method was demonstrated by the presence of dose-responsive severity and distribution of neuronal degeneration/necrosis by FJB in Layers III and IV of the posterior cingulate and retrosplenial cortices and in other expected sub-anatomic brain locations (e.g., piriform cortex) of MK-801 positive control rats (Figure 2). All animals treated with 1 mg/kg MK-801 had “minimal” degeneration and those treated with 5 mg/kg MK-801 had “mild” neurodegeneration. Consistent with observations in the FJB-stained slides, decreased neuronal processes were identified in the silver-stained sections, but degenerating neurons were difficult to differentiate from normal neurons in the silver-stained sections. No microscopic abnormalities were observed in the brain of subjects given any dose of ketamine, DCS/lurasidone alone (Figure 3), or ketamine followed by DCS/lurasidone (Figure 4). The figures depict representative histology for the 2000 mg/kg DCS with 139 mg/kg lurasidone (Figure 3) and for 12.5 mg ketamine with 2000 mg/kg DCS with 139 mg/kg lurasidone (Figure 4). At 40× magnification using stains FJB and HE, the combination of DCS/lurasidone with or without ketamine revealed normal neuronal structures and no evidence of neurotoxicity as shown by absence of degenerating neurons. In the Group 8 positive control rats, neurodegeneration was detected in the retrosplenial cortex at 72 hours after administration of MK-801 (5 mg/kg). Figures A-C. Neurodegeneration was characterized by nuclear pyknosis and condensed, brightly eosinophilic cytoplasm of individual neurons (at the arrows) in H&E-stained slides and, in Figure D, by green fluorescent labeling with FJB (yellow fluorescent structures in Figure D represent residual erythrocytes). The H&E labeled images represent progressively increasing magnification of the same brain location from a single rat. No microscopic abnormalities occurred in the brains of Group 7 rats administered DCS/lurasidone (2000/139 mg/kg) either Figures A-C in the H&E-stained sections or Figure D in the FJB-labeled microscopic sections. In the Group 5 rats, administration of ketamine (12.5 mg/kg) on the day prior to administration of DCS/lurasidone (2000/139 mg/kg) did not produce detectable morphological changes in Figures A-C, the H&E-stained microscopic sections, or in Figure D, the FJB-labeled sections.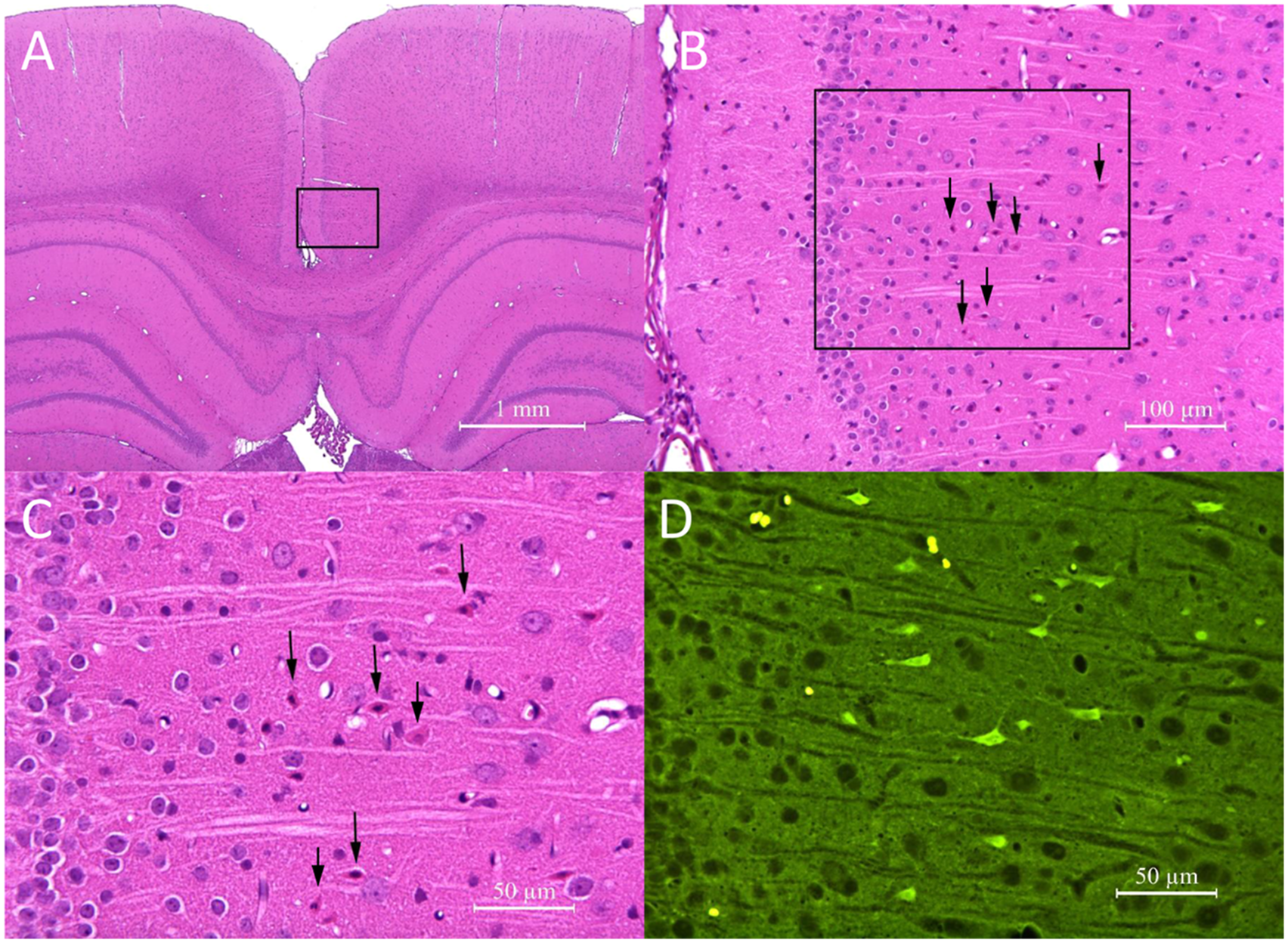
Discussion
The data presented support the FDA requirement to show agents acting at the NMDA receptor for the treatment of depression do not cause histological neurotoxicity. The full data report associated with the above study was submitted to FDA and the agency issued a corresponding “Study may proceed” letter and subsequent “Special Protocol Agreement,” for phase 2b/3 clinical trials of NRX-101.
The clinical signs observed in DCS/lurasidone-dosed animals, including decreased activity, prostration, atonia, and closed eyes were considered an extension of pharmacological effects of DCS and/or lurasidone. Other clinical signs observed, including ocular and/or nasal discharge, material around nose, and soiled coat, were considered to be related to stress. Therefore, these changes did not suggest additional neuronal toxicity of the treatment. Other findings including decreased body weight and food consumption, ocular changes, and changes in clinical pathology were also not related to neuronal toxicity of the treatment. These results, together with the key study outcome that DCS/lurasidone treatment administered with or without ketamine did not induce microscopic abnormalities in the brain, suggest the intended treatment did not result in neuronal toxicity in this study. For all endpoints and parameters evaluated, changes seen in animals treated with 12.5 mg/kg ketamine + 2000/139 mg/kg DCS/lurasidone were in general similar to those given 2000/139 mg/kg DCS/lurasidone alone, which suggests the combination treatment did not result in additional toxicity under the conditions of the study.
Equivalent Dosages of DCS/Lurasidone for Animal and Human Studies.

The findings demonstrate a substantial margin of safety as measured by histologic change associated in increasing doses of NRX-101. As doses increased from 500/35 to 2000/139 mg/kg, the systemic exposures (AUC0-24h and/or Cmax) to DCS and lurasidone increased dose-proportionally. At the same dose level of 2000/139 mg/kg DCS/lurasidone, the systemic exposures to DCS were comparable between animals dosed with or without ketamine; the systemic exposure to lurasidone was slightly lower in animals dosed without ketamine than in animals dosed with ketamine, which was likely due to a slightly extended Tmax. No obvious test article-related effects or changes were noted in animals given 12.5 mg/kg ketamine alone. In addition to the histologic safety, no clinical signs of neurotoxicity and no deaths of experimental subjects were observed.
The authors recognize that neurobehavioral testing and other techniques might in the future be required to decisively prove the lack of neurotoxicity associated with NRX-101. However, the current FDA standard is based on histologic safety.
Conclusion
D-cycloserine/lurasidone combined, when administered once at doses up to 2000/139 mg/kg (actual dose 1600/100 mg/kg) to female rats via oral gavage with or without IV infusion of 12.5 mg/kg ketamine approximately 24 hours prior to the oral dosing, was tolerated and did not induce neurotoxicity. Under the conditions of the study, the no-observed-adverse-effect level (NOAEL) for neurotoxicity was considered to be 2000/139 mg/kg. The corresponding AUC0-24 h and Cmax of DCS and lurasidone at the NOAEL were 2950000 h*ng/mL and 509000 ng/mL for DCS, and 7280 h*ng/mL and 636 ng/mL for lurasidone, respectively.
Footnotes
Author Contributions
Jordan, W. contributed to acquisition, analysis, and interpretation and critically revised manuscript; Sapko, M. contributed to analysis and interpretation and critically revised manuscript; Seigel, R. contributed to conception and design, drafted manuscript, and critically revised manuscript; Javitt, J. contributed to conception and design, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: State: - Statement of proprietary interest: Drs Siegel and Javitt are employees and shareholders of NeuroRx, Inc. All data collection and interpretation were conducted by individuals with no propriety interest in the outcome of the research.
Funding
This research was sponsored by NRx Pharma Inc. and did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Correction (July 2023):
There was a typo in the email address. The typo has been corrected in the article.
Statement of Proprietary Interest
Drs Siegel and Javitt are employees and shareholders of NRx Pharma Inc. All data collection and interpretation were conducted by individuals with no propriety interest in the outcome of the research.
