Abstract
Species selection plays a pivotal part during non-clinical safety assessment in drug development. If possible, use of non-human primates (NHPs) should be avoided due to ethical considerations. However, limiting factors as lack of pharmacologic activity in other species could necessitate use of NHPs. LAI-PCSK9i is a bi-functional molecule combining a long-acting insulin analogue with a PCSK9 inhibitor peptide aiming to provide glycaemic control and to reduce plasma LDL concentrations. The NHP was chosen for the safety assessment of LAI-PCSK9i being the most relevant species with basal levels and plasma lipid composition closest to humans, while the dog and initially also the minipig were deemed irrelevant due to lack of pharmacologic activity on LDL-lowering and biological differences in lipid profiles. An in vivo tolerability and toxicokinetic study of LAI-PCSK9i in NHPs showed recurrent and severe hypoglycaemia at very low doses. Therefore, the minipig was re-evaluated and a follow-up study thoroughly assessing blood glucose and cholesterol levels and clinical signs illustrated that minipigs dosed with LAI-PCSK9i, tolerated the compound and LAI-PCSK9i decreased glucose and LDL over time. This work underlines that careful consideration is required when selecting species during safety assessment in drug development. The tolerability issue in NHPs led to the subsequent selection of the minipig for safety evaluation of LAI-PCSK9i although as a suboptimal alternative, which unexpectedly had a measurable pharmacologic response on LDL lowering. In conclusion, the NHPs may be unsuitable as test species for safety assessment of long-acting insulin analogues due to high sensitivity to recurring hypoglycaemic episodes.
Keywords
Introduction
Species selection plays a pivotal role during non-clinical safety assessment of new drugs in development and depends on a range of factors including test item experience in the specific species, amount of available historical data, as well as important considerations regarding similarities to humans in relation to the specific target and/or pathways involved to ensure that the species is susceptible to the intended pharmacological effect(s) and to increase predictability of the human response. 1 The selection is further challenged when the drug has more pharmacologic targets that should be balanced when selecting the appropriate species. Use of the non-human primates (NHPs) is only permitted when no other alternative species or methods are available.2,3 However, limiting factors such as low homology of a target receptor or major differences to human biology could necessitate the use of NHPs rather than other possible non-rodent species eg dogs or minipigs.
Long-acting insulin proprotein convertase subtilisin/kexin type 9 inhibitor (LAI-PCSK9i) is a bi-functional molecule acylated with a fatty acid side chain for increased half-life and display both long-term glucose and low-density lipoprotein (LDL) lowering properties (proprietary structure). Biological differences in plasma lipid fractions and cholesterol metabolism across non-rodent species (ie, dog, minipig and NHP) played a main role in the choice of non-rodent species for the non-clinical safety assessment of LAI-PCSK9i for this bi-functional molecule.
The human biological relevance of the pharmacological effect of LAI-PCSK9i on LDL cholesterol is governed by the distribution of lipoprotein particles (high-density lipoprotein (HDL), LDL, very low-density lipoprotein (VLDL)) and the presence of cholesteryl ester transfer protein (CETP), a key enzyme involved in plasma cholesterol transport that transfers cholesteryl ester from HDL to lipoproteins such as LDL and VLDL. The minipig and dog have low LDL levels compared to humans, cholesterol in dogs is mainly contained in the HDL fraction, and pigs and dogs have no or little CETP.4,5 The main non-rodent toxicology models of choice for testing insulin effects are typically the dog and alternatively pig/minipig (Development programs for eg, Insulin Degludec, Insulin Glargine, Insulin Aspart), whereas there are only few reports on administration of insulin to NHPs. 6 However, the pig was initially deemed irrelevant based on lack of significant LDL-cholesterol or total cholesterol lowering effects observed in a single dose pharmacokinetic study (data not shown) and the dog was ruled out due to lack of expression of the target PCSK9.
The NHP is considered the animal species most similar to humans in terms of the basal levels and composition of plasma lipids. 7 Approximately 50% lowering of LDL for up 10 days was observed in the NHP dosed with a peptide containing an amino acid sequence close (but not identical) to the PCSK9i peptide moity included in LAI-PCSK9i (unpublished data). Additionally, there is high homology between human and cynomolgus monkey insulin/insulin receptor/Insulin-Like Growth Factor I receptor binding sequence (Sequence alignment of publicly available sequences) and single-dosing of human insulin to cynomolgus monkeys reduces blood glucose levels; 8 Thus, this species was expected to be equally responsive to insulin as man and as other non-rodent species eg minipigs or dogs. Hence, the NHP (cynomolgus monkey) was considered the only appropriate non-rodent species when assessing safety of an insulin molecule with PCSK9 inhibiting properties.
Two maximum tolerated dose (MTD) in vivo studies were performed to assess the tolerability and toxicokinetics of the systemic toxic potential of LAI-PCSK9i in non-rodent species. First, a MTD study was performed in cynomolgus monkeys. Following results from this study, the NHP proved to be irrelevant due to being highly sensitive towards the blood glucose lowering effect of LAI-PCSK9i. This high sensitivity confounded data interpretation and made the NHP a less ideal model for non-clinical safety assessment of LAI-PCSK9i. Therefore, a second MTD study was performed in Göttingen minipigs® to re-evaluate the potential of this suboptimal species for testing LAI-PCSK9i. Here, we present results from these MTD studies and provide learnings for non-rodent species selection of a bifunctional compound with insulin and low-density lipoprotein properties and provide insights into non-rodent species selection of a bifunctional molecule.
Materials and Methods
Animals and Study Design
Study I: Cynomolgus Monkeys Dosed With LAI-PCSK9i
The objective of the study was to assess the tolerability and toxicokinetics of LAI-PCSK9i in cynomolgus monkeys and make a preliminary evaluation of the systemic toxic potential of LAI-PCSK9i after once daily subcutaneous (s.c.) administration.
Animals
Three male and three female cynomolgus (Macaca fascicularis) monkeys (∼27 months old) were supplied from Vietnam (Purpose bred; Nafovanny, Vietnam). Animals were screened for Tuberculosis (Mantoux test), Simian Herpes B Virus and general health by a veterinarian and acclimatized to the local environment (16-25°C, 40-70% humidity, 12 hours light cycle) for a minimum of 6-8 weeks prior to start of dosing and randomly allocated to groups prior to Day 1 (first day of dosing). They were housed in stainless steel cages, designed for non-human primates, in trios of the same sex, except for 1-2 hours in connection with dosing or due to ill-health. The cages were incorporated in the animal room walls and floor, which were coated in slip resistant epoxy floor coating and covered with a wood granulate litter (environmental enrichment). They contained polypropylene dividing walls and shelves, a food hopper and a wooden or polypropylene platform. The animals were offered a daily commercial diet in the morning pre-dosing (Teklad 2050, 100 g), a daily food supplement (two biscuits) given with fresh fruit produce at least 1 hour post dosing and water ad libitum. In addition, they were given a small treat after dosing. In case of hypoglycaemia related clinical signs (Supplementary Table S1) or decreased food consumption, a carbohydrate rich food supplement was given and, if deemed necessary, oral glucose solution was given. Animals were observed at least twice daily for clinical signs of ill-health or reactions to treatment (hypoglycaemia), body weight was recorded daily, prior to feeding, throughout the study (acclimatisation to necropsy) and food consumption was measured for each trio of animals throughout the study.
Study Design
The subcutaneous route of administration was chosen to simulate the clinical route of administration; the first day of dosing was designated as Day 1. The study was planned, as a standard MTD study, to include two groups receiving different dosing regimens and an initial toxicokinetic phase (single dose of 4 nmol/kg/day) was planned on Day 1 and termination end of study (Figure 1A). The initial dose of 4 nmol/kg was based on previous pharmacokinetic single dose studies with LAI-PCSK9i in pigs and dogs (unpublished data). The dose volume was calculated based on the most recent body weight of the animal. Study design of the monkey study. A) In the planned study design, an initial toxicokinetic phase consisting of a single-dose of 4 nmol/kg/day (in green) was planned to measure LAI-PCSK9i exposure during the first 96 h after dosing, followed by escalating doses from Day 8, starting with 4 nmol/kg/day, determining subsequent doses based on the response. B) In the final design, doses were as follows: 4 nmol/kg on Day 1 (in green), 1.0 nmol/kg on Day 11, 0.5 nmol/kg on Day 12, and 0.05 nmol/kg on Day 20 and 21. Dosed animals were terminated on Day 43. Stars indicate clinical signs of hypoglycaemia, solid stars: male, open stars: female. TK, toxicokinetic. TBD, to be determined. †, termination.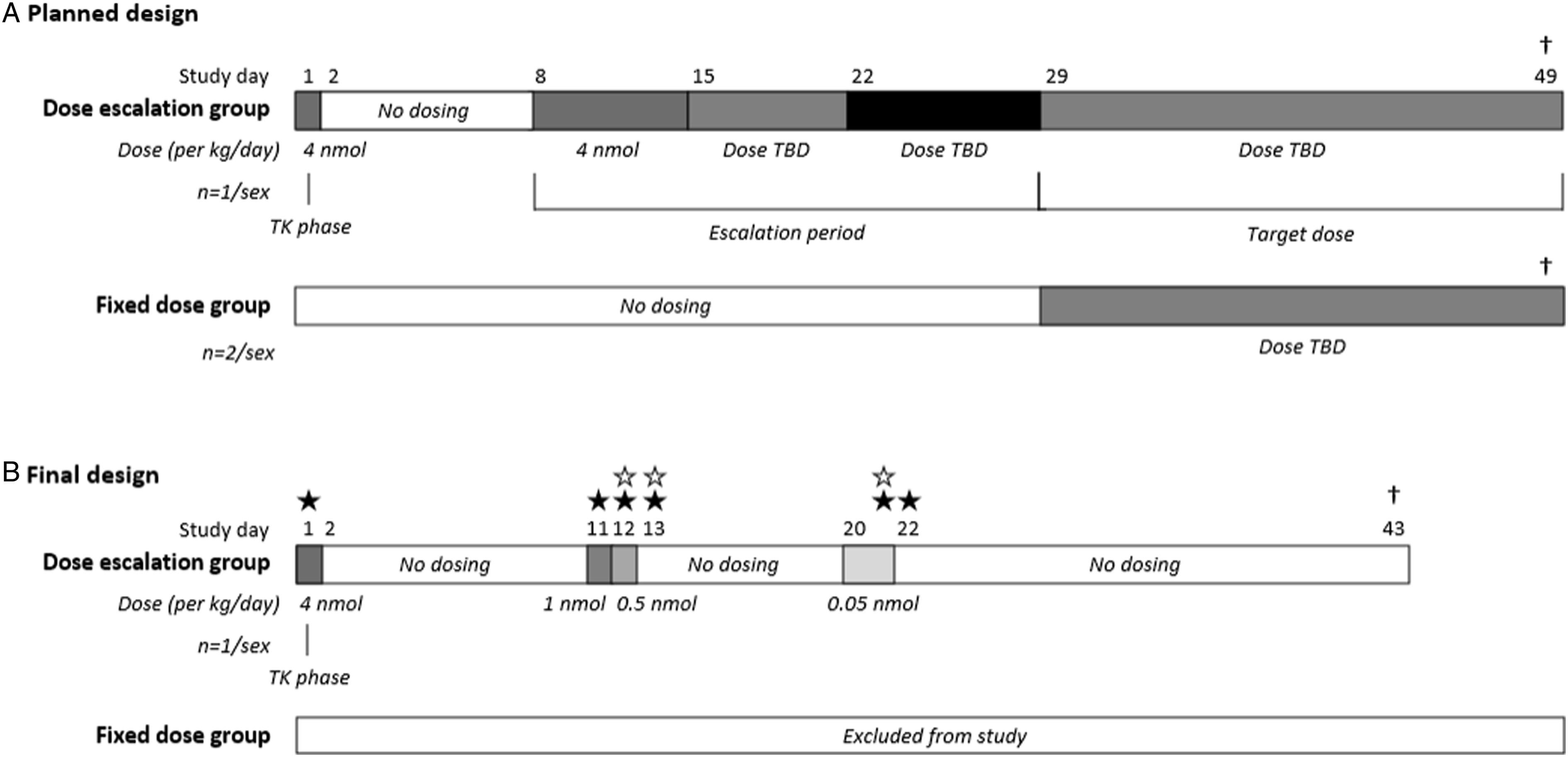
Due to development of clinical signs of hypoglycaemia in the male after the first dose of 4 nmol/kg on Day 1, the study design in the dose escalation group was revised (described below, depicted in Figure 1B); furthermore, due to the high sensitivity and development of clinical signs of hypoglycaemia even at very low dose levels in the dose escalation group (see Figure 1B, details described in result section), no dosing was initiated in the fixed dosed group, and these animals were excluded from the study. After dosing on Day 1, days of dosing were revised, and doses reduced to 1.0, 0.5, 0.05, and 0.05 nmol/kg/day on Day 11, 12, 20, and 21, respectively. Animals were terminated on Day 43 by sedation with ketamine prior to an intravenous injection of sodium pentobarbitone solution (200 mg/mL) followed by exsanguination. A full macroscopic examination was performed.
Study II: Göttingen minipigs dosed with LAI-PCSK9i
The objective of this study was to assess the MTD in an alternative non-rodent species to NHPs. The purpose of the fixed dose phase was to assess tolerability following 3 weeks of dosing and the toxicokinetics of LAI-PCSK9i following once daily subcutaneous administration to minipigs.
Animals
Three male and three female Göttingen SPF (specific pathogen free) minipigs (4-5 months old, 10.4-11.4 kg at arrival) were supplied from Ellegaard Minipigs A/S, Denmark. Animals were randomised into two groups (see below) and received a 5 day acclimatisation period and were housed in an animal room with filtered air and controlled conditions (21 ± 3°C, approximately 30 ± 10% humidity, 10 air changes per hour, 12 hour light cycle from 06
Study Design
The subcutaneous route of administration was chosen to simulate the clinical route of administration; the first day of dosing was designated Day 1. The study included two groups (see Figure 2A), 1 group (dose escalation group, n = 1/sex) subjected to a three-week dose escalation period to establish the target dose (MTD) of LAI-PCSK9i, followed by a two-week period of dosing with the target dose (fixed dose phase), and a second parallel group (fixed dose group, n = 2/sex) receiving no dosing during the first three weeks of the study and hereafter dosed with the same fixed target dose as in the dose escalation group at this time. A 14-day dosing period and subsequent termination (Day 35) was originally planned. Both groups had a pre-dosing period of 17 days prior to Day 1 (the fixed dose group had an additional 21 days after Day 1). The starting dose of 2 nmol/kg/day in the dose escalation group was based on previous single dose studies in pigs given LAI-PCSK9i (unpublished data). Selection of the next dose level during escalation was based on blood glucose levels, clinical signs, body weight and food consumption effects. For the second level of the dose escalation phase, a dose of 8 nmol/kg/day was selected since there did not appear to be any clinical signs of hypoglycaemia or lowering of blood glucose level at 2 nmol/kg/day. Due to lowering of blood glucose level in both animals and uncertain clinical signs (described below in result section) in the female at 8 nmol/kg/day, the dose was reduced to 6 nmol/kg/day during the third week of dose escalation (Figure 2B). In addition, the female animal was off dose for the last two days at 8 nmol/kg/day and for the first day at 6 nmol/kg/day. Study design of minipig study. A) Planned study design with escalating doses based on response to the starting dose of 2 nmol/kg/day. B) Final study design, the planned study design was revised based on the occurrence of clinical signs of hypoglycaemia. Stars indicate clinical signs of hypoglycaemia, solid stars: male, open star: female. TK (toxicokinetics) signifies samples for measurements of LAI-PCSK9Ii exposure. TBD, to be determined. †, termination.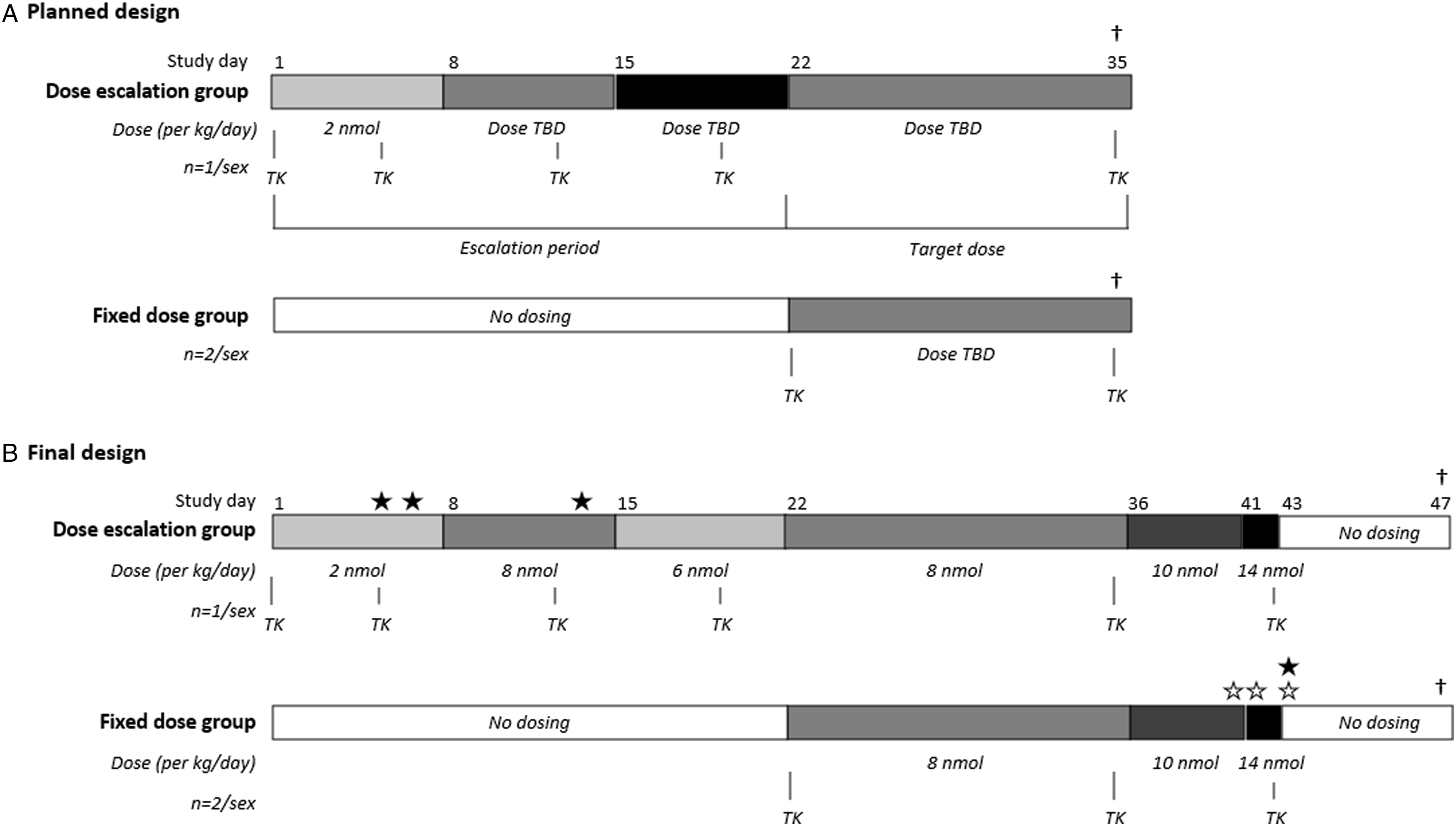
As there were no clinical signs or significant effects on blood glucose in any of the animals at 6 nmol/kg/day, 8 nmol/kg/day was chosen as the target dose during the fixed dose phase starting Day 22 in both groups (Figure 2B). As no clinical signs were observed at 8 nmol/kg/day after the planned 14 days of dosing, the dose was increased to 10 nmol/kg/day from Day 36 and the study extended by 1 week. The dose was further increased to 14 nmol/kg/day on Day 41 due to uncertainty of clinical signs. On Day 43, dosing was discontinued due to clinical signs of hypoglycaemia and low blood glucose level and animals terminated on Day 47.
Implantation of ear vein catheters for blood sampling: See Supplementary Files (Materials and Methods) for description of procedure.
Study I and II: Live Animal Procedure Licenses
Procedures involving live animals were performed under the Project Licence authorized by the United Kingdom Secretary of State and according to the applicable sections of the United Kingdom Animals (Scientific Procedures) Act 1986, Amendment Regulations 2012 (the Act) as well as Covance CRS Limited (now LabCorp). In addition, they were performed in accordance with EU directive 2010/63/EU on the protection of animals used for scientific purposes, 3 as well as Scantox A/S and Novo Nordisk A/S company policies on the care and use of laboratory animals, and under the Project Licence number 2015−15−0201−00713 authorized by the Danish Animal Ethics Council.
Compound
LAI-PCSK9i was delivered as a 600 nmol/mL stock solution in a vehicle buffer (19.6 mg/mL glycerol, 16 mM phenol, 16 mM m-cresol, 1.4 mM CaCl2, 0.23 mM ZnAc, 10 mM Tris, pH 7.4), diluted with a vehicle (19.6 mg/mL glycerol, 16 mM phenol, 16 mM m-cresol, 10 mM Tris, pH 7.4) to obtain the desired formulation (Novo Nordisk A/S, Maaloev, Denmark).
Blood Glucose Measurements
Study I: Cynomolgus Monkeys Dosed With LAI-PCSK9i
Monitoring of whole blood glucose levels included profiles (Day 1 and 21) for evaluation of changes over time following dosing and single time-point daily glucose assessments for overall monitoring. All samples were taken in unfasted animals (See section 2.1 for feeding schedule). Profiles: Measurements were performed on Day 1 at the following time-points: pre-dose, 10 min and 30 min, as well as 1, 2, 4, 8, 16, 24, 48, 72, and 96 hours after dosing. Additionally, measurements were performed on Day 21: 32, 40, 48, 56, 64, 72, 84 and 96 hours after dosing. Single time-point monitoring: Blood was sampled (0.05 mL, superficial and/or femoral vein, no anaesthesia) bi-daily from Day-14, at time of anticipated dosing on Day 1, and daily from Day 1 just prior to dosing (or at similar times during non-dosing days) and 8 hours post dosing, except on Day 1-5 and Day 22 (0 hour sampling performed)-25 (where samples for glucose profiles were drawn), and glucose levels snap-measured using an Accu-Chek® glucometer.
Study II: Göttingen Minipigs Dosed With LAI-PCSK9i
Two types of blood glucose measurements were included: Serum for glucose profiling and whole blood for continuous single time-point assessment. Serum profiles: In the dose escalation group, blood samples (150 µL/sample taken from samples taken for serum LAI-PCSK9i quantification, see below), were taken on Day 1, 5, 12 and 19 (escalation period) and in the fixed dose group on the first day of dosing (Day 22); all animals were sampled during the fixed dose phase on Day 35 and 42. Samples were taken at 7 timepoints: pre-dosing, 30 min., and 1, 2, 4, 8 and 24 hours post-dosing, moreover, on Day 42 also at 48, 72, 96 and 120 hours post-dosing. Pre-dose samples were taken at a maximum of 30 min before dosing. Serum was separated, 150 µL allocated for glucose quantification and the remaining sample for determination of serum LAI-PCSK9i concentration. The samples were stored frozen at −18°C or below within 90 min after collection until further analysis. Glucose levels were quantified using a Cobas® 6000 analyzer (Roche Diagnostics, Burgess Hill, West Sussex, UK). Whole blood: Measurements were performed (1 drop, through ear vein catheter, when possible, for the male in the dose Escalation Group, remaining animals: dorsal side of the ear or jugular vein/bijugular trunk, no anaesthesia) every other day from Day-9 of the pre-treatment period as late in the day as possible. During the dosing period (excluding day of blood sampling for serum profiles) samples were taken 8 hours post dosing in the escalation period and prior to feeding and dosing in the fixed dose phase. Measurements were performed using a glucometer (Accu-Chek® Aviva blood glucose monitoring system, Roche Diagnostics).
Systemic LAI-PCSK9i Concentration and Toxicokinetics
Study I: Cynomolgus Monkeys Dosed With LAI-PCSK9i
On Day 1 and Day 21, blood samples (0.3 mL) were taken at the same nominal time-points and conditions, as samples for the blood glucose profile described above. Samples were stored at −10°C to −30°C until analysis.
Study II: Göttingen Minipigs Dosed With LAI-PCSK9i
Blood samples were taken with samples for quantification of serum glucose levels (see above).
Study I and II
LAI-PCSK9i Levels in Serum
Quantification was performed using an internally developed luminescent oxygen channeling imunoassay (LOCI) (details can be given upon request). The lower limit of quantification (LLOQ) was 100 pmol/L. Results were reported as the mean of duplicate determinations.
Toxicokinetic Analysis
The concentration vs time data was evaluated by non-compartmental analysis using PhoenixTM WinNonlin® version 8.1 (Pharsight®, Certara). The following parameters were calculated for each individual profile: tmax (time at maximum plasma/serum concentration), Cmax (maximum observed plasma/serum concentration), AUC0-24hr (AUC from time 0 to 24 hr). In addition, the following toxicokinetic parameters were calculated for profiles with sampling schedule longer than 24 hours: AUC0-∞ (AUC from time 0 to infinity), AUCextrap (percentage of AUC extrapolated from the last concentration to infinity), and T½ (terminal half-life). The AUCs were calculated using the “Linear Up Log Down” method, ie the linear trapezoidal rule when the concentration increased, and the logarithmic trapezoidal rule when the concentration decreased. T½ was calculated using “individual best fit” of the log-linear regression of concentrations vs time (ie points at and prior to Cmax were not used, at least three data points were included and if R2 was within 0.0001 of the largest possible R2 value, the regression with the larger number of points was used). For non-compartmental analysis, the nominal sampling times were used, and the pre-dose sample was set to zero. In addition, concentrations below LLOQ were excluded or set to zero for pre-dose samples.
Quantification of Cholesterol
Study I: Cynomolgus Monkeys Dosed With LAI-PCSK9i
Quantification of plasma cholesterol at regular intervals during the dosing periods was planned, however, due to the unexpected revision of the study design, blood was only sampled pre-dosing (nominally 0.3 mL) and measurements therefore not performed.
Study II: Göttingen Minipigs Dosed With LAI-PCSK9i.
Once weekly approximately 3 mL blood was sampled prior to first feeding and pre-dosing (no anticoagulant) for quantification of serum cholesterol, including HDL, and LDL levels. For the dose escalation group, blood was sampled before start of the dosing period on Study Day-7 and on Day 5, 12, 19, 35, and 42 (ie the days before increase in dose, Figure 2B). For the fixed dose group, blood was sampled on Day-7, as well as 35 and 42 (days before increase in dose). Samples were analysed on a Cobas® 6000 analyzer.
Results
Animals
Study I: Cynomolgus Monkeys Dosed With LAI-PCSK9i.
Clinical Signs Related to Treatment
Both monkeys displayed clinical signs that were consistent with hypoglycaemia (hunched posture, tremors, decreased activity, fasciculations, chin rubbing and/or piloerection), which in some cases coincided with reduced blood glucose levels, occurring at all doses and there appeared to be decreased tolerability after repeated administration (study days with clinical signs indicated in Figure 1B). Animals recovered after oral glucose administration and access to sugary food supplements. Details on timing of clinical signs and related blood glucose levels are shown in Supplementary Table S1.
At end of study, no macroscopic abnormalities were observed at termination on Day 43.
Body Weight and Food Consumption
Body weight was not affected by dosing, however both animals showed minor fluctuations attributed to normal day-to-day variation (data not shown). Food consumption was measured on a cage basis, as both dosed (dose escalation group) and non-dosed (fixed dose group) animals were housed together, it was not possible to assess if dosing with LAI-PCSK9i had any effect on eating patterns.
Study II: Göttingen Minipigs Dosed With LAI-PCSK9i.
Clinical Signs Related to Treatment
Clinical signs of hypoglycaemia (abnormal gait/ataxia, increased activity, salivation, nervous behaviour and muscle shivering) were observed at dose levels of 2 (incidental), 8, 10 and 14 nmol/kg/day and, in general, occurred before feeding. Other clinical signs, including watery diarrhoea, reduced appetite, increased body temperature, and decreased activity/subdued behaviour, were not attributed to treatment due to their occurrence in undosed animals. Details on timing of treatment related clinical signs of hypoglycaemia and related blood glucose levels are shown in Supplementary Table S2.
At the end of study, no macroscopic abnormalities were observed at termination.
Body Weight and Food Consumption
All animals displayed normal weight gain during the study (data not shown). One dose escalation group female and 1 fixed dose group male (naïve) were observed with food remains on Day 12-16 and Day 13-15, respectively, coinciding with clinical observations made in these animals.
Blood Glucose Levels
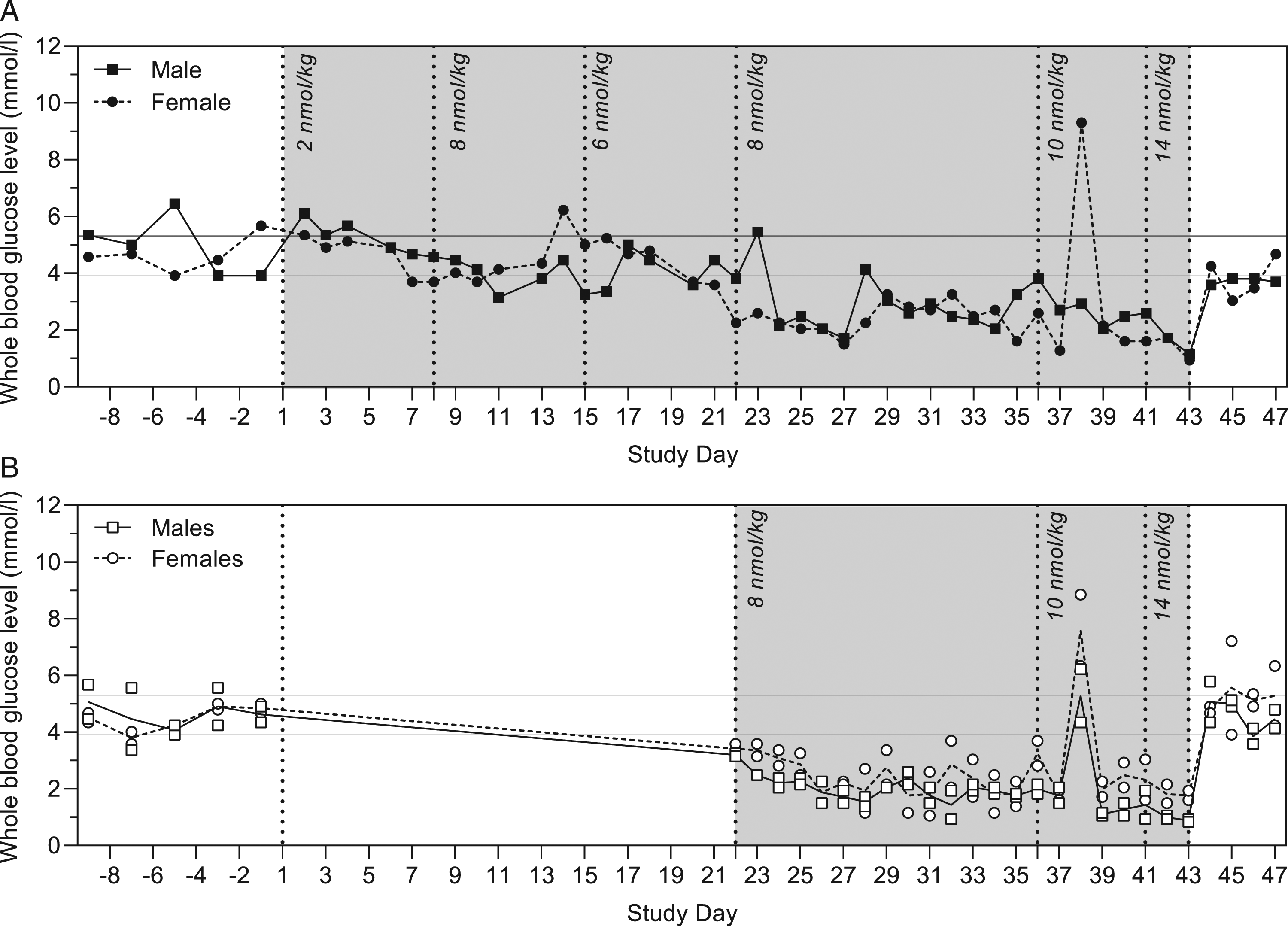
Study I: Cynomolgus Monkeys Dosed With LAI-PCSK9i
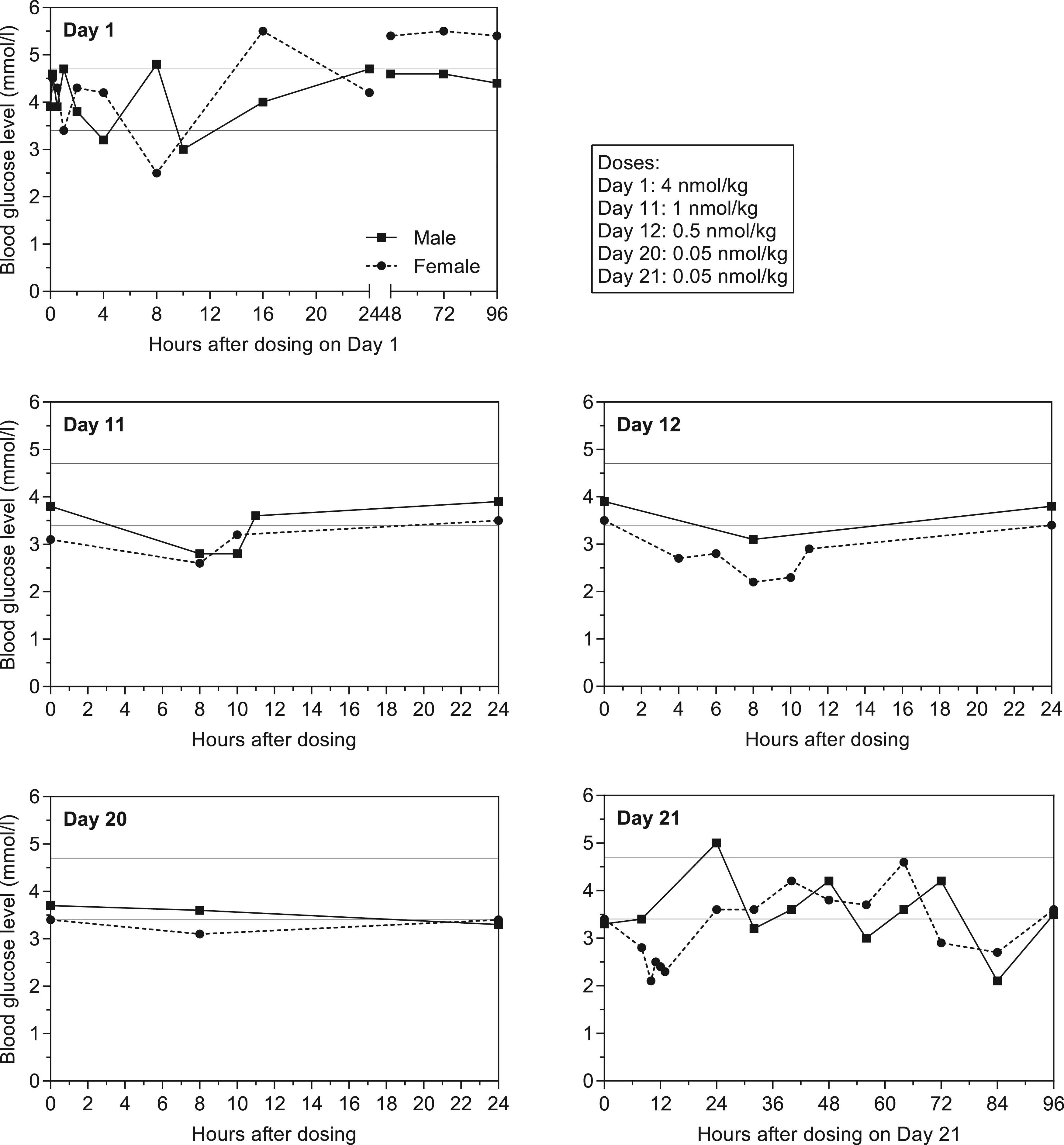
Whole blood glucose levels over time throughout the study (8 hours post dosing on dosing days) are presented in Figure 3 and glucose profiles on dosing days in Figure 4. Overall, pre-dose whole blood glucose levels were within the range measured from Day-14 prior to study start (3.1 to 5.1 mmol/L) on dosing days (Figure 3). Overall, a blood glucose lowering effect of dosing with LAI-PCSK9i was seen 4-24 hours after dosing with a similar maximal effect at all dose levels, except for males at 0.05 nmol/kg. On Day 11, oral glucose was administered in both animals 10 hours after dosing. On Day 20, following a 7 day dosing free period, only a slight decrease in blood glucose level was seen in the female 8 hour after dosing (0.05 nmol/kg), however, on Day 21, levels were reduced in the female at 8 hours after dosing (0.05 nmol/kg), decreasing further between 10 and 13 hours after dosing despite of administration of oral glucose at 8 and 10 hours after dosing to abate the clinical signs of hypoglycaemia observed in this animal. Glucose levels had returned to normal after 24 hours. There was a reduction of blood glucose in both animals at 84 hours after the second 0.05 nmol/kg dose for unknown reasons (Figure 4, Day 21). The animals were given access to sugary treats and the level was normal by the 96 hour time-point. Whole blood glucose level over time in Cynomolgus monkeys. Grey area indicates dosing-periods, vertical dotted lines indicate changes in dose, and horizontal grey lines show normal mean upper and lower blood glucose level (mean + SD and mean-SD, respectively, corresponding to 3.4 and 4.7 mmol/L) across all animals and from Day -14 to -2 (ie before dose-start). Measurements were performed 8 h post-dosing on dosing days, on days without dosing, measurements were performed at the time corresponding to pre-dose on dosing days. Note: Measurement for the female on Day 12 (2.2 nmol/L) was taken ≤2 h after oral glucose administration. Whole blood glucose profiles in Cynomolgus monkeys. 96 h blood glucose profiles taken on Day 1 and 21. For the remaining dosing days, pre- and post-dose measurements are also included, 8 h measurements are also included in Figure 3. 0 h measurements were performed pre-dose. Horizontal grey lines show normal mean upper and lower blood glucose level (mean + SD and mean-SD, respectively, corresponding to 3.4 and 4.7 mmol/L) across all animals and from Day -14 to -2 (ie before dose-start). Additional measurements performed during episodes with clinical signs of hypoglycaemia are included for Day 1 (male, approximately 10 h post-dosing), Day 11 (female, 10 h post-dosing; male, 10 and 11 h post-dosing), Day 12 (female, 4, 6, 10, and 11 h post-dosing), and Day 21 (female, 10, 11, 12, and 13 h post-dosing). All additional measurements, except for at 10 h on Day 1, were taken 1-2 h after oral glucose administration.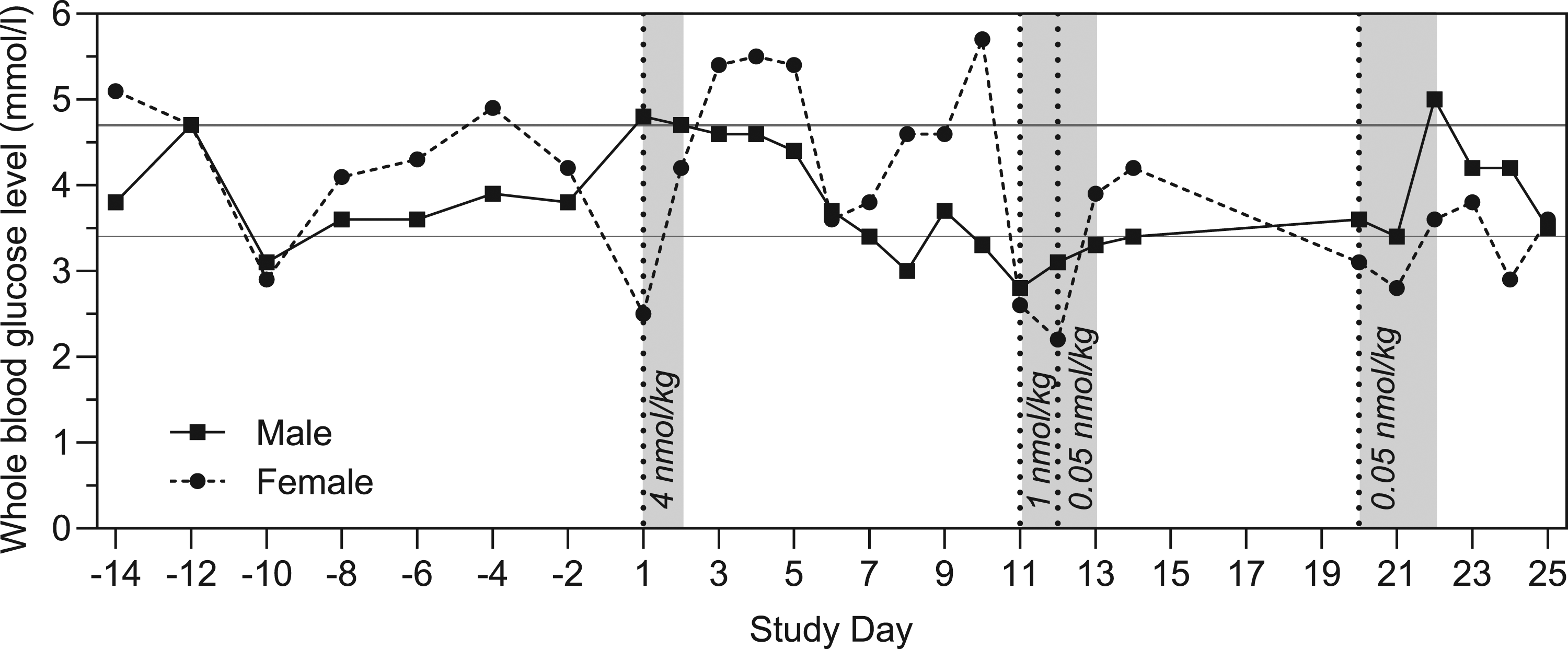
Study II: Göttingen Minipigs Dosed With LAI-PCSK9i
No blood glucose lowering effect was seen on first day of dosing and Day 5 at 2 nmol/kg/day, or at 6 nmol/kg/day on Day 19 (dose escalation group, Figure 5A-B and D and 6A). At 8, 10, and 14 nmol/kg/day, a dose-dependent glucose lowering effect was observed, primarily at 4 to 24 hours post-dosing (both groups, Figure 5C and E-I, and 6A and B). After cessation of dosing on Day 43, whole glucose levels had normalised within 48 hours. and blood glucose levels were normal at end of study on Day 47 (Figure 6 and Supplementary Figure S1). Generally, serum blood glucose levels were increased at 0.5 to 1 hour post-dosing, normalising within two hours, except for Day 42, where increases were also seen at 48 to 72 hours (Figure 5). The increase in whole blood glucose levels on Day 38 was caused by mistakenly feeding the animals prior to sampling (Figure 6A and B). In general, whole blood glucose values were slightly lower compared to serum levels measured on the same day. Serum glucose profiles in minipigs. A-F) Dose escalation group, G-I) Fixed dose group, means and individual values. A) Day 1, 2 nmol/kg/day; B) Day 5, 2 nmol/kg/day; C) Day 12 (5th day on 8 nmol/kg/day); D) Day 19 (5th day on 6 nmol/kg/day); E) Day 35 (14th day on 8 nmol/kg/day); F) Day 42 (2nd day on 14 nmol/kg/day); G) Day 22, first day of dosing in the Fixed dose group; H) Day 35 (14th day on 8 nmol/kg/day); I) Day 42 (2nd day on 14 nmol/kg/day). NOTE: Day 35 and 42 (E, F, H, I) have different y-axes from the remaining days and Day 42 results are 96 h profiles (F and I). Whole blood glucose levels over time in minipigs. A) Dose escalation group, B) Fixed dose group, individual and means. Grey area indicates dosing-period, vertical dotted lines indicate changes in dose, and horizontal grey lines show normal mean upper and lower blood glucose level (mean + SD and mean-SD, respectively) across all animals and days from Day -8 to -2. The increase in blood glucose levels at Day 38 was caused by mistakenly feeding the animals prior to sampling.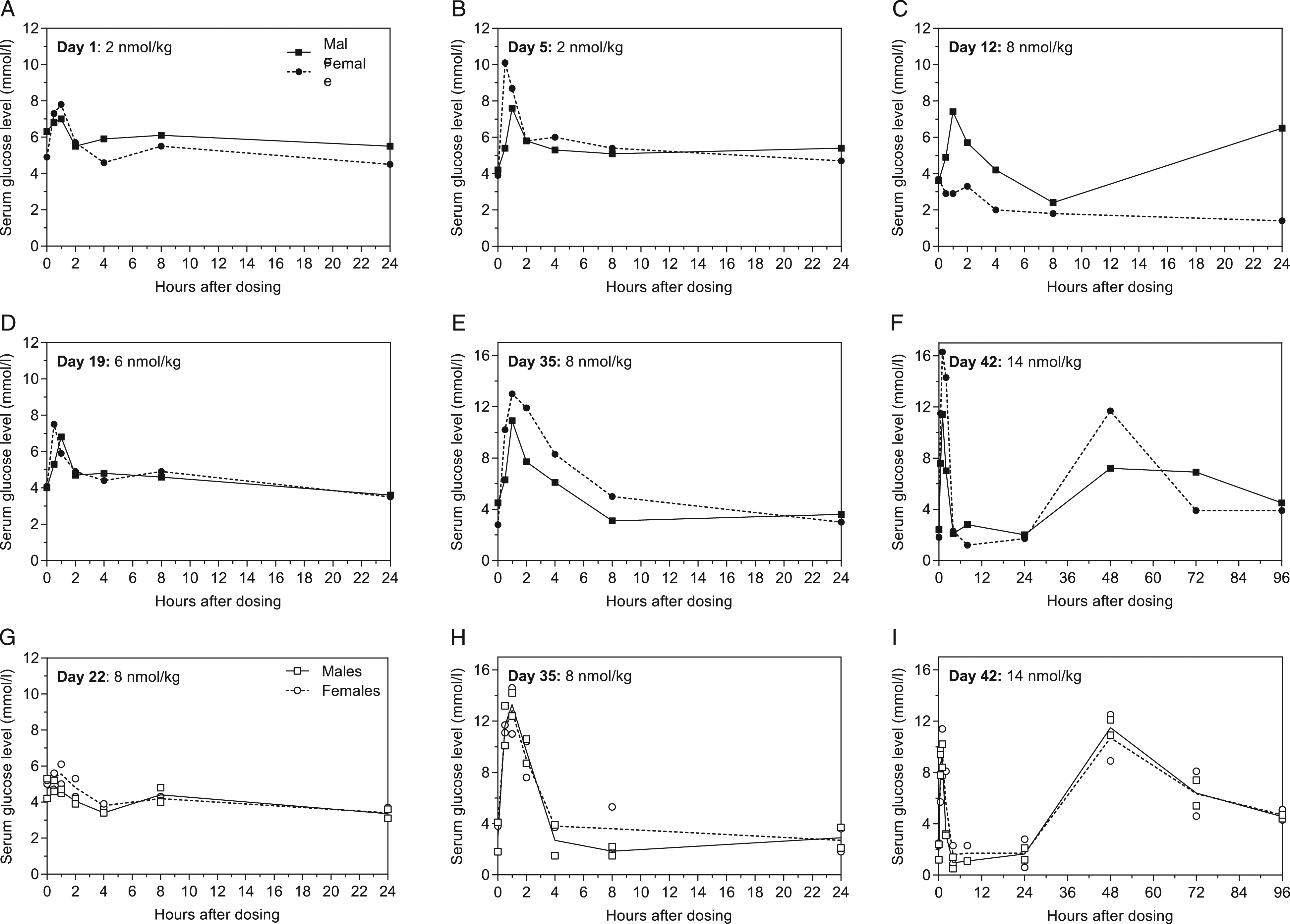
Systemic LAI-PCSK9i Concentration and Toxicokinetics
Study I: Cynomolgus Monkeys Dosed With LAI-PCSK9i
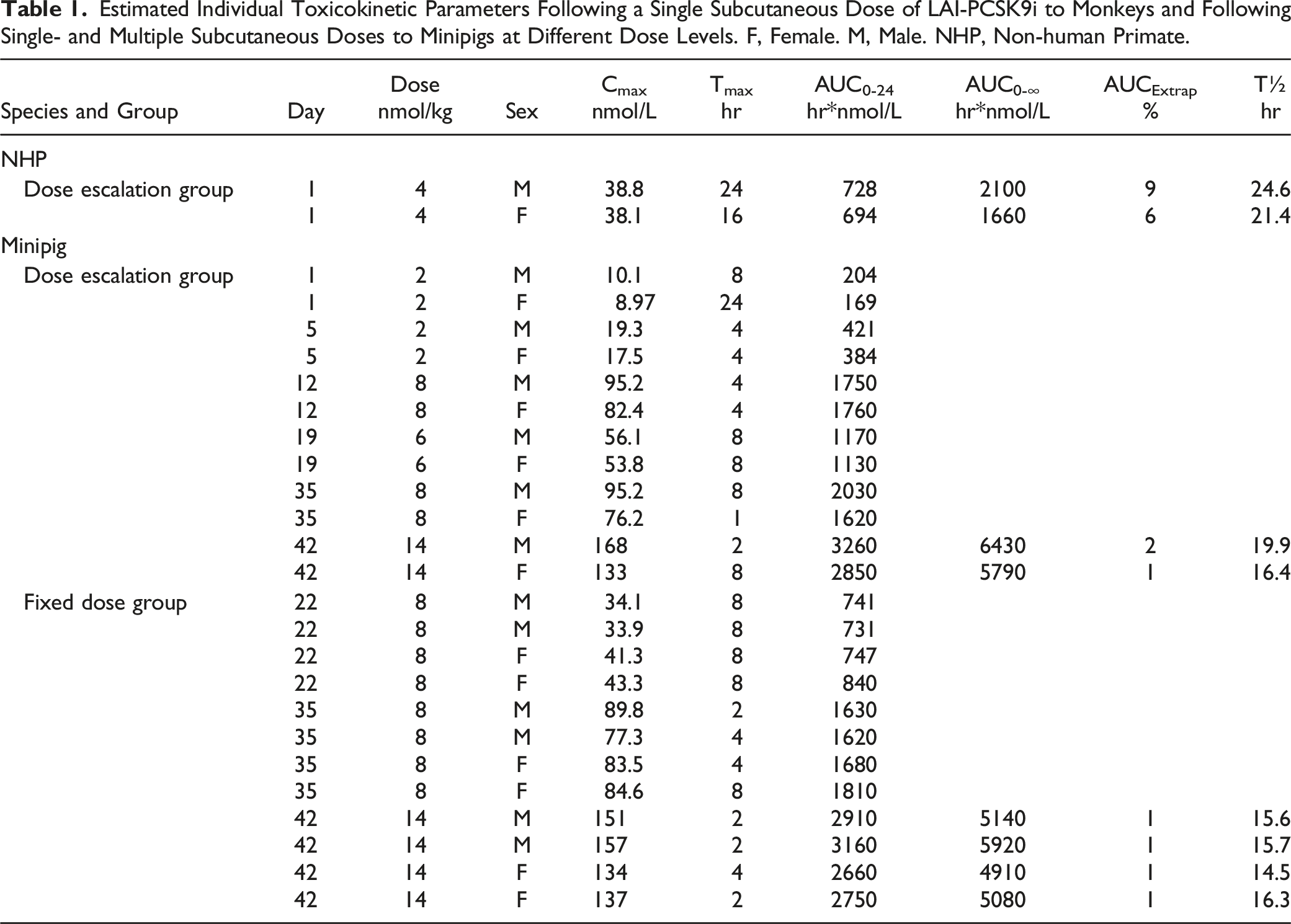
Both animals were systemically exposed to LAI-PCSK9i on toxicokinetic sampling days (Figure 7) and LAI-PCSK9i concentrations were below the LLOQ (100 pmol/L) in all pre-dose samples. Individual plasma concentration vs time profiles are presented in Figure 7 and toxicokinetic parameters are listed in Table 1. In summary, the tmax after a single dose of 4 nmol/kg was 24 and 16 hours in the male and female, respectively, and the tlast was 96 hours post-dose for both animals. A single dose of 4 nmol/kg resulted in a LAI-PCSK9i Cmax of 38.1-38.8 nmol/L and an AUC0-∞ of 1660-2100 hr*nmol/L. The terminal T½ was found to be 24.6 and 21.4 hours in the male and female, respectively. LAI-PCSK9i plasma concentration vs time profiles following a single dose in Cynomolgus monkeys. Dose escalation group only. Levels reported as being < LLOQ omitted (Day 1: pre-dose samples, Day 21: from 72 h after dosing in the female). Estimated Individual Toxicokinetic Parameters Following a Single Subcutaneous Dose of LAI-PCSK9i to Monkeys and Following Single- and Multiple Subcutaneous Doses to Minipigs at Different Dose Levels. F, Female. M, Male. NHP, Non-human Primate.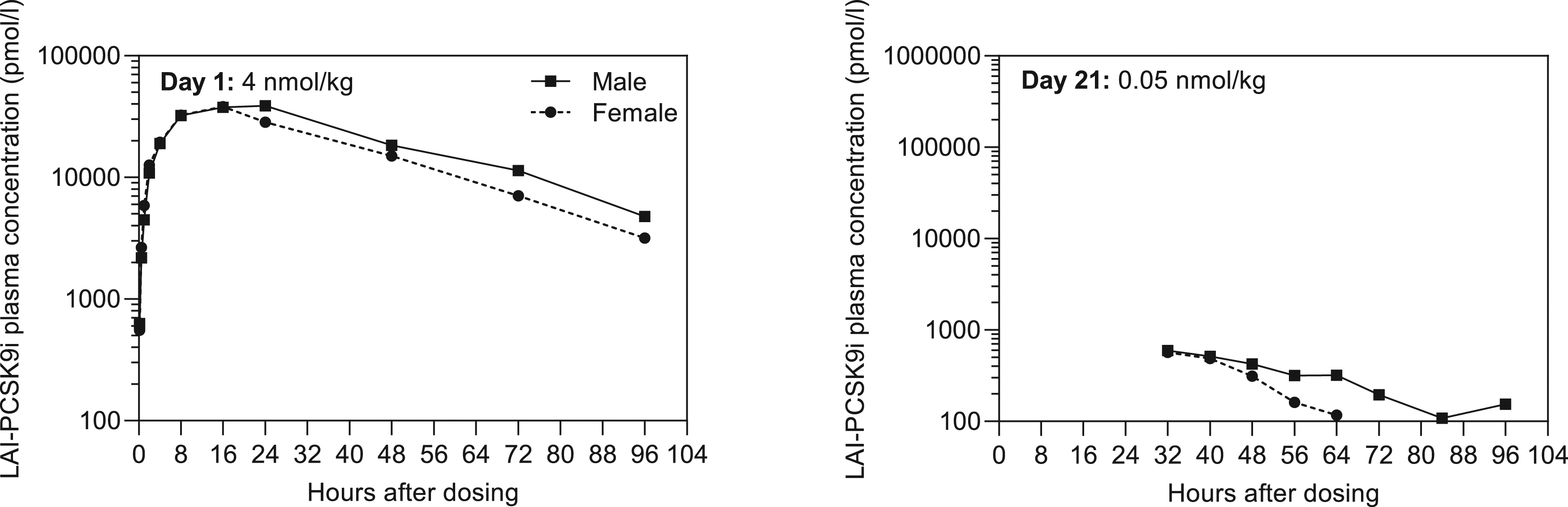
Study II: Göttingen Minipigs Dosed With LAI-PCSK9i
All animals were systemically exposed to LAI-PCSK9i on toxicokinetic sampling days and LAI-PCSK9i exposure was below LLOQ (100 pmol/L) in all pre-dose samples. Toxicokinetic evaluation was performed following single and multiple doses to Göttingen minipigs. Individual serum concentration vs time profiles are presented in Figure 8 and toxicokinetic parameters are listed in Table 1. In the minipig dose escalation group, Cmax was in the range of 8.97-168 nmol/L and AUC0-24hr in the range of 169-3260 hr*nmol/L, dependent on the escalation step from 2-14 nmol/kg. In the fixed dose group, Cmax was 33.9-157 nmol/L dependent on escalation from 8-14 nmol/kg and AUC0-24hr was in the range of 4910-5920 hr/nmol/L on Day 42 where the dose of 14 nmol/kg was reached. LAI-PCSK9i plasma concentration vs time profiles in minipigs. Levels reported as being < LLOQ omitted (Pre-dose samples on Day 1 and Day 22 in the Dose escalation and Fixed dose groups, respectively). ESC, Dose escalation group. FIX, Fixed dose group.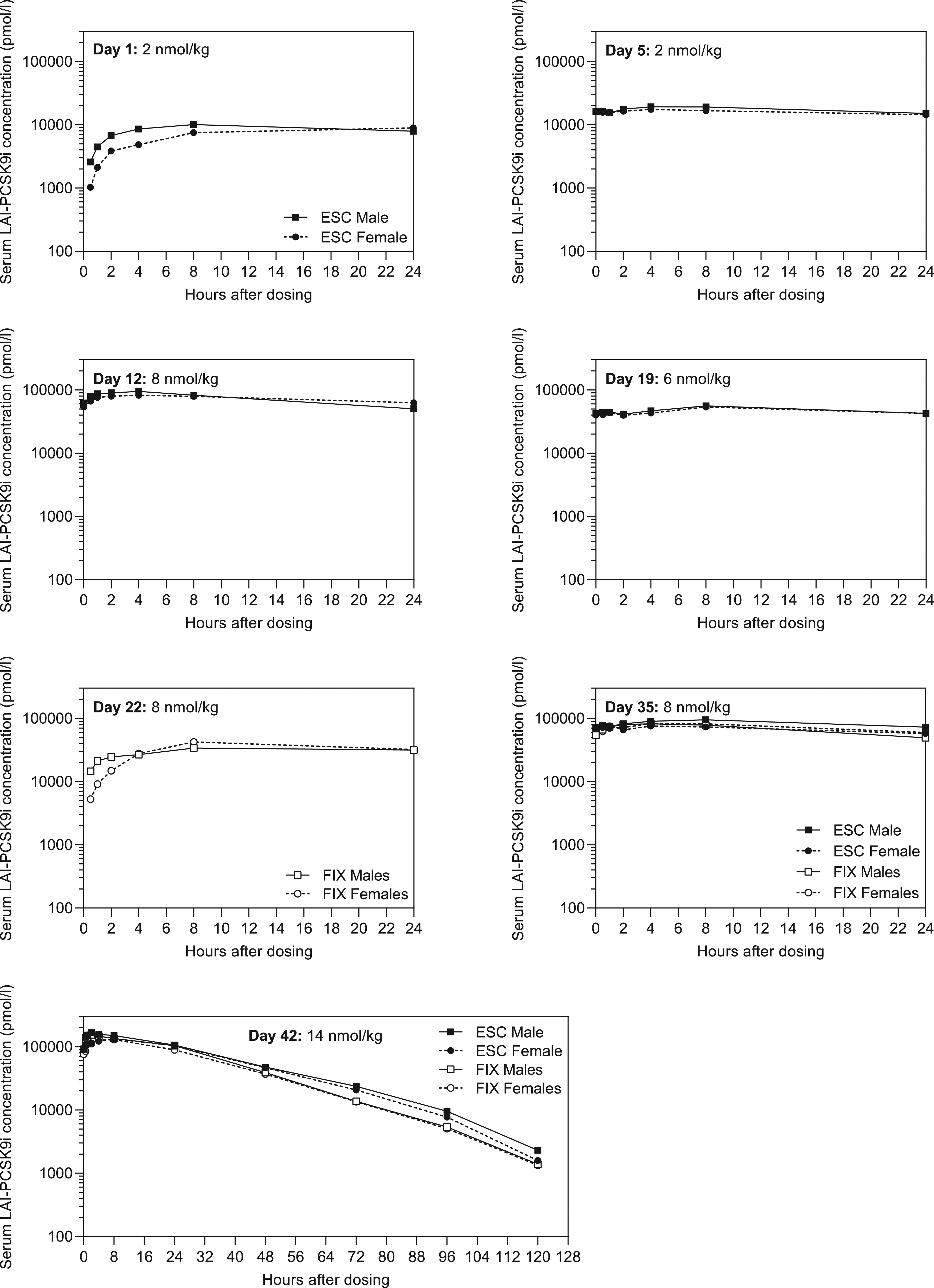
Study I and II
There were no indications of sex differences and in the minipig where several dose levels were evaluated, Cmax and AUC parameters seemed to increase at increasing dose.
Serum Cholesterol Levels
Study II: Göttingen Minipigs Dosed With LAI-PCSK9i
Results are depicted in Figure 9. A slight decrease in cholesterol and HDL was observed in some of the animals and a decrease in LDL was observed for all animals between pre-treatment (Day-7) and Day 42 (36% and 54% in the Escalation group male and female, respectively, 26-65% and 29-42% in fixed dose group males and females, respectively). Serum cholesterol levels over time in minipigs, individual values. Left panel: Dose escalation group, right panel: Fixed dose group. Vertical dotted line indicates dose start.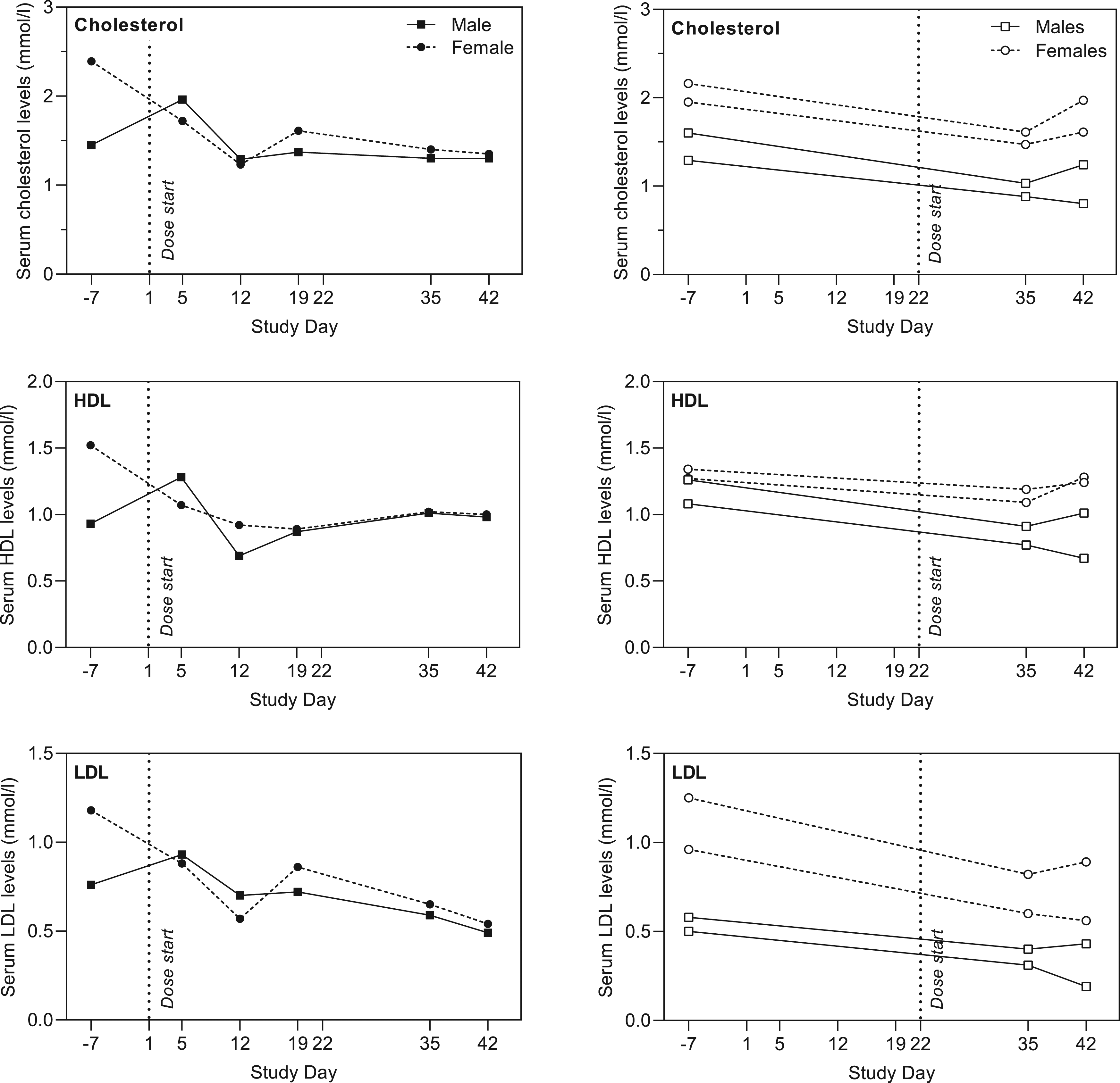
Discussion
Selecting the best species for the non-clinical safety assessment model is critical to the success of the non-clinical safety package. Careful considerations are required when selecting the most relevant species and this process is challenged when developing bi-functional molecules where different pharmacologic effects need to be balanced. In this paper in vivo data are presented that guided the non-rodent species selection for a long-acting insulin with PCSK9 inhibiting properties (LAI-PCSK9i), a bi-functional molecule with both long-term glucose and low-density lipoprotein (LDL) lowering properties.
The NHP is considered the animal species most similar to humans in terms of basal levels and composition of plasma lipids. Therefore, the NHP was initially considered the only relevant species during safety assessment of the bi-functional compound, LAI-PCSK9i, with cholesterol lowering properties. However, the monkeys proved to be highly sensitive towards the prolonged blood glucose lowering effect of LAI-PCSK9i and thus became an irrelevant non-rodent species for safety assessment due to this overt sensitivity to the blood glucose lowering ability of the test item. Therefore, the minipig was re-evaluated and successfully used in a MTD study as the non-rodent species. The dog was not re-evaluated due to lack of expression of PCSK9.
During the study in NHPs, the animals displayed recurrent episodes with clinical signs of hypoglycaemia despite significant reduction of the doses (from 4 nmol/kg/day eventually down to 0.05 nmol/kg/day at the end of the dosing period) resulting in exposure levels below clinical relevance and clinical observations requiring clinical intervention. Thus, NHPs seem to be highly responsive to the pharmacological effect of insulin ie lowering of blood glucose levels. This is supported by other laboratories, where a low single dose of human insulin to normal healthy cynomolgus monkeys induced clear effects on blood glucose levels. An i.v. dose of 3 nmol/kg decreased blood glucose levels by about 35-70% within 15-20 min and a s.c. dose of 0.9 nmol/kg by about 50% within 60 minutes in males and females.6,8,9 This illustrates that doses should be selected carefully as small increases in dose may be intolerable (ie, a steep dose response curve). In humans it is known that frequent hypoglycaemic episodes weaken the counter-regulatory responses and may therefore increase the incidence and severity of such episodes over time.10,11 Therefore, it could be speculated, that the risk of a hypoglycaemic episode may increase for each hypoglycaemic recurrence also in NHPs and may, at least in part, explain why the episodes continued at lower doses. LAI-PCSK9i Cmax was in the range of 77-95 nmol/L following 35 days of dosing of the tolerable dose level of 8 nmol/kg/day in the minipig. As for comparison, the intolerable single dose of 4 nmol/kg given to the NHP produced a LAI-PCSK9i Cmax in the range of 38-39 nmol/L. The maximum tolerable plasma LAI-PCSK9i concentration was not identified in the monkey, but the results indicate that at the given experimental design, the cynomolgus monkey tolerated less than half the LAI-PCSK9i exposure compared to the minipig.
LAI-PCSK9i resulted in similar glucose lowering and clinical observations in minipigs and rats, as other long-acting insulins with similar potency previously developed by Novo Nordisk (Unpublished data). Hence, the low tolerability to LAI-PCSK9i observed in NHPs may not be unique to this particular long-acting insulin analogue. The increased sensitivity could possibly be related to the low basal blood glucose levels in NHPs. In cynomolgus monkeys, the normal serum/plasma glucose level is about 3-4 mmol/L,6,8,9,12 as compared to about 3.5-5 mmol/L in Göttingen minipigs, 4-6 mmol/L in Beagle dogs, 8-10 mmol/L in rats and 3.9-5.6 mmol/L in humans.13–18 Therefore, relatively small absolute decrements in blood glucose level will correspond to a high percentage decrease; here, for instance, the fall in blood glucose level by 1.4 mM seen in the female after the first dose corresponded to a 36% decrease. In addition, the NHP was shown to have a higher basal insulin secretion, measured by C-peptide, compared to humans. 12 Thus, a low baseline blood glucose and a higher basal secretion of insulin may make NHPs prone to develop severe hypoglycaemia. Another predisposing factor could be the age of the animals in the present study, where 27-month-old juvenile animals were included. A study by others in cynomolgus monkeys showed an age difference in the blood glucose lowering effect of a single dose of human insulin in females, where young adults (6-7 years) displayed a decrease of about 70% as compared to 50% and 40% in middle-aged (11-13 years) and old (17-18 years) adult monkeys. 6 Thus, younger animals may be more insulin sensitive. Furthermore, others found a ≥10-fold higher baseline of endogenous fasting insulin levels in two 8–9-year-old as compared to 1 5-year-old cynomolgus monkeys, which could indicate compensatory increases caused by decreased insulin sensitivity with increasing age. 8 NHPs are also very lean which may increase insulin sensitivity; In humans, it is established that body mass index is positively correlated with insulin resistance. 19
An additional factor, which may make the NHPs more susceptible to development of severe hypoglycaemia could be species differences in the counter-regulatory response of acutely increasing food intake. Whereas rats are known to increase food intake in response to hypoglycaemia, this has to our knowledge not been documented in monkeys and it has been suggested that food intake in NHPs is not particularly responsive to excursions in blood glucose levels because they tend to eat small and variable amounts during the day and not large quantities at once.17,20 Unfortunately, food consumption could not be assessed in the present study, as the insulin-dosed monkeys were housed with naïve animals that share food. The group housing with naïve animals as well as the group hierarchy could also have influenced food intake of the dosed animals. These factors could also be important reasons for the difference in insulin sensitivity between NHPs and minipigs.
In minipigs, LAI-PCSK9i performed similarly to other long-acting insulin analogues (unpublished data) and was generally well tolerated, while also displaying cholesterol (LDL) lowering effects and thus proved to be an acceptable non-rodent species for safety assessment of LAI-PCSK9i, at least based on the few animals included in this study. This suggests that the minipig could be an alternative species of choice to the NHP in non-clinical development with cholesterol lowering drugs. This is relevant as the use of NHPs should be avoided if possible due to ethical considerations. However, this should be assessed on a case-by-case basis, due to the differences in blood lipid profiles and cholesterol metabolism in the minipig compared to humans, and effects would likely depend on the mechanism of action. Also, it may be challenging to detect discrete changes to cholesterol levels due to the lower baseline in minipigs vs NHPs, which is further dependent on the detection limit of assays used for measuring cholesterol levels.
Species selection for nonclinical safety evaluation of bi-functional molecules in general may be more complex compared to single-target drugs, since the species will have to be comparable to humans for pharmacological activity for both functionalities, normal ranges and responses to change and the drug may address distinct different biological pathways as is the case here. It may therefore be tempting to choose the non-human primate for bi-functional or co-agonist molecules since the likelihood of suitability is higher than for other toxicological species. However, it is important to look for alternative species to NHPs and a species may be deemed suitable even if it only shows some degree of activity on both targets and may not have to show the same level of activity as expected in humans. In summary, the present work underlines that careful consideration is required when selecting non-clinical toxicity species to assessing the tolerability of new drugs in development, particularly with more complex pharmacology such as bi-functional compounds, where more than one pharmacologic response needs to be balanced when selecting the appropriate species. Here, the NHP was the obvious and scientifically justifiable choice based on a clinically relevant lipid profile and similar homology to the human insulin receptor. However, the monkeys proved to be highly sensitive to the blood glucose lowering effect of LAI-PCSK9i.
In conclusion, this suggests that the NHP may be unsuitable as test species for safety assessment of long-acting insulin analogues due to high sensitivity and recurring hypoglycaemic episodes.
Supplemental Material
Supplemental Material - The Non-Human Primate in Safety Assessment of a Bifunctional Long-Acting Insulin Analogue
Supplemental Material for The Non-Human Primate in Safety Assessment of a Bifunctional Long-Acting Insulin Analogue by Vivi FH Jensen, Nikolai K Jensen, Line H Schefe, Jens Sigh, Akinyemi Akintomide, Kari Kaaber, Sophia G Moesgaard and Mona H Pedersen in International Journal of Toxicology
Footnotes
Acknowledgments
The authors thank contributing scientist Lene Andersen and laboratory technicians Katja Sundstrup and Tina K Andersen (LAI-PCSK9i levels in plasma) from Novo Nordisk A/S as well as study director Marianne K. Bracken from Scantox A/S.
Author Contributions
Jensen, V. contributed to conception, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Jensen, N. contributed to conception, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Schefe, L. contributed to design, contributed to analysis and interpretation, and critically revised manuscript; Sigh, J. contributed to acquisition, analysis, and interpretation and critically revised manuscript; Kaaber, K. contributed to interpretation and critically revised manuscript; Akintomide, A. contributed to design, contributed to acquisition and interpretation, and critically revised manuscript; Moesgaard, S. contributed to design, contributed to interpretation, and critically revised manuscript; Pedersen, M. contributed to design, contributed to interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work, ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VFHJ, NKJ, LHS, JS, SGM, and MHP are or have all been employees at Novo Nordisk A/S.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
