Abstract
Cisplatin is a chemotherapeutic drug used to treat a great variety of solid tumors. Its dose is commonly limited by its nephrotoxicity, manifested as acute kidney injury (AKI). Erythropoietin (Epo) is a glycoprotein hormone that regulates the production of red blood cells. This study was performed to evaluate the presence of endogenous Epo in male Wistar rat urine and to analyse changes in urinary Epo levels in response to cisplatin- induced AKI. Dose-dependent studies and time-dependent experiments were performed to evaluate changes in urea nitrogen and creatinine in plasma as well as Epo, neutrophil gelatinase-associated lipocalin (NGAL), alkaline phosphatase (AP) activity, creatinine and total proteins in urine at 2 days post-dosing. Rats received 2, 5 or 10 mg/kg b.w., i.p. of cisplatin. At 5 mg/kg b.w., i.p. cisplatin, significant increases in urinary Epo were detected. Significant increases in urea nitrogen and creatinine in plasma, NGAL, AP, proteins, and Epo were observed in urine from rats that received 10 mg/kg b.w., i.p. of cisplatin. In the time-dependent experiments, rats were injected with a dose of 5 mg/kg b.w., i.p. of cisplatin, and sampling occurred 2, 4, and 14 days post-dosing. In these animals, there were significant increases in urea nitrogen and creatinine in plasma and total proteins, AP activity, Epo, and NGAL in urine on day 4. Urinary Epo was also detected on day 2. Taken together, these findings provide weight of evidence for urinary Epo as a promising early biomarker of cisplatin-induced AKI in male rats.
Introduction
Acute kidney injury (AKI) is a clinical syndrome with multiple etiologies and risk factors. It is characterized by a sudden loss of kidney function with an abrupt decrease in glomerular filtration rate over hours to days. 1 The prevention of AKI development and/or its early diagnosis is essential, since AKI is related to high-short and long-term morbidity and mortality rates, and also to high health costs. In addition, AKI can progress to chronic kidney disease (CKD), which could lead to kidney failure, requiring dialysis or kidney transplantation to maintain life.2,3 Drug-induced nephrotoxicity has been considered one of the major pathogenic factors of AKI.3,4
Cisplatin (cis-diamminedichloroplatinum II) is a commonly used chemotherapeutic drug to treat a great variety of solid tumors. Its dose and its antitumor efficacy are commonly limited by its nephrotoxicity. Cisplatin renal toxicity may arise as a severe presentation of AKI in 20-30% of patients, and this may lead to discontinuation of chemotherapy with this agent. 5 Therefore, early diagnosis of cisplatin-induced nephrotoxicity is essential, above all, in the prevention of underlying injuries and further complications.
The accurate diagnosis of drug-induced nephrotoxicity at early stages is difficult to achieve, especially because slight renal damage is frequently unrecognized. This situation leads to a delay in detection, with an associated change in renal function, and with the consequent loss of an early therapeutic opportunity. 6
Kidney function has been assessed by traditional markers such as circulating creatinine or blood urea nitrogen levels and urine output, but it has been extensively demonstrated that they have low diagnostic performance. They do not have sufficient sensitivity since their values are detected outside the normal ranges after a considerable loss of kidney function. 4 For all these reasons, in the past decade, intense efforts have been undertaken to find promising biomarkers for early and accurate detection and diagnosis of AKI. The more promising early diagnosis biomarkers that count with more supporting results are neutrophil gelatinase-associated lipocalin (NGAL), N-acetyl-β-D- glucosaminidase (NAG), kidney injury molecule-1 (Kim-1), interleukin-18 (IL-18), and cystatin C. They can detect subclinical AKI, which in many cases can be characterized by the elevation of biomarkers levels without evident loss of renal function. Thus, they can provide more refined prognostic information than the renal markers currently used, circulating levels of creatinine and urea.2,7
Erythropoietin (Epo) is a glycoprotein hormone composed of 165 amino acids that regulates the production of red blood cells by promoting erythroid progenitors’ proliferation and differentiation. In this way, Epo helps to keep the blood haemoglobin concentration constant under normal conditions. It is well documented that inadequate concentrations of Epo may results in anaemia. This hormone is principally synthesized in the renal cortex by peritubular fibroblasts and is excreted in the urine.8,9 In the past years, few works have been conducted about the detection of endogenous Epo in urine and its possible modifications after a kidney injury.10,11 Almost all works were focused on purification and detection methods of Epo in urine to detect and control the abuse of recombinant Human Epo (rHuEpo) in endurance athletes as an anti-doping test.12-14
Our group has been working in the last years in the detection of different proteins in urine, and has proposed them as novel early biomarkers of AKI of different etiologies. Among them, we can name the Organic Anion Transporter 5 (Oat5), Aquaporin-2 (AQP2), Na-K-2Cl-cotransporter (NKCC2), Caveolin-2 (Cav2), Sodium-dependent Dicarboxylate Cotransporter 1 (NaDC1), and Organic Anion Transporting Polypeptide 1 (Oatp1).15-29 Therefore, based on our previous experience in urinary biomarkers of kidney damage, and taking into account the little information available about the role of endogenous Epo in urine, the aim of this study was to evaluate the presence of endogenous Epo in rat urine and to analyse possible changes in urinary Epo levels in response to nephrotoxic injury induced by cisplatin.
Materials & Methods
Materials
The antibodies used in this work were commercial: mouse antibody against erythropoietin (Epo) was purchased from R&D Systems Inc. (Minneapolis, MN, USA, AE75 A, Cat. Number MAB 2871) and mouse monoclonal antibody against neutrophil gelatinase-associated lipocalin (NGAL) was purchased from Thermo Fisher Scientific, Invitrogen (Waltham, MA, USA, Cat. Number: ABS 039-32-02). The Kaleidoscope Prestained Standards of molecular mass were purchased from Bio Rad Laboratories (Hercules, CA, USA, Cat. Number: 161-0324). Pure chemicals of analytical grade as well as cisplatin (99,99% purity) were purchased from Sigma-Aldrich (St Louis, MO, USA).
Experimental Animals
The experiments carried out with animals were performed under principles and guidelines for the care and use of laboratory animals, following the recommendations of the Guide for the Care and Use of Laboratory Animals: Eighth Edition. 30 The experimental procedures were approved by the Institutional Animal Care and Use Committee, Faculty of Biochemical and Pharmaceutical Sciences, National University of Rosario (Res. N° 385/2017).
A total of 36 male Wistar rats 130 days old were used in this work. Rats were maintained in an environment under controlled constant temperature and humidity, with regular light cycles of 12 h, and free access to food and tap water. The animals were acclimated for three days before being randomly divided into different experimental groups.
To carry out cisplatin dose-dependent experiments, rats were injected with cisplatin (Cp), at doses of 2 (Cp2 2d, n = 4), 5 (Cp5 2d, n = 4), and 10 (Cp10 2d, n = 4) mg/kg of body weight (b.w.), intraperitoneally (i.p.). The experiments were carried out 2 days after the injection. The cisplatin doses were selected considering their relation to doses currently used in clinical chemotherapy and to the nephrotoxicity they produce, and also based on previous studies.18-20,31
Another set of experimental animals was used to perform time-dependent experiments. Based on previous studies,18-20,28 the 5 mg/kg dose of cisplatin was selected to achieve these studies (this dose is related to clinical doses used in chemotherapy and shows nephrotoxicity). Rats were injected with a single dose of cisplatin of 5 mg/kg b.w., i.p., and studies were performed at 4 (n = 4) and 14 (n = 4) days after the injections (Cp5 4d, n = 4, and Cp5 14d, n = 4, respectively). Cp5 2d group from the previous dose- dependent experiments was included in these time-dependent experiments. Control groups were injected with vehicle (1 mL saline/kg b.w., i.p.) for each time point evaluated (2 (n = 4), 4 (n = 6), and 14 (n = 6) days). As all the tested parameters in the control groups (2 (n = 4), 4 (n = 6), and 14 (n = 6) days after vehicle administration) showed no significant differences between them, we decided to consider them as one group “Control group” (C, n = 16), to facilitate the analysis of the results as also previously reported.19,28
One day before necropsy, rats were individually placed in metabolic cages to collect the urine (under cold temperature refrigerated condition) when voided over an 16- 24 h period. In this instance, they had unrestricted access to water. The animals were euthanized at different cisplatin post-treatment time points (2, 4 or 14 days after injections, as appropriate). They were anesthetized with sodium thiopental (70 mg/kg b.w., i.p.), blood samples were collected with heparin by cardiac puncture, and immediately after that, the animals were euthanized with anesthetic overdose and thoracotomy.
Biochemical Determinations
Urine samples were centrifuged for 10 min at 1000×g, to remove cell debris. Blood samples were also centrifuged for 10 min at 1000 × g to obtain plasma. In urine samples, alkaline phosphatase (AP) activity, creatinine, and total proteins levels were measured, and in plasma samples, the urea and creatinine levels were assessed. Determinations were carried out using commercial reagent kits (Wiener Laboratory, Rosario, Argentina), by spectrophotometric methods.
Electrophoresis and Immunoblotting
Epo and NGAL abundances were analysed in rat urines from different experimental groups. Briefly, urine samples (10 μL) were boiled for 3 min in 1% 2- mercaptoethanol, 2% sodium dodecyl sulphate (SDS) and were subjected to separation by a 12% polyacrylamide gel (SDS-PAGE). Then, they were electroblotted to nitrocellulose membranes. Ponceau Red was employed to verify equal protein loading and transfer between lanes as was broadly described.18-29,32 The membranes were later incubated for 1 h with 5% non-fat dry milk (5% in phosphate-buffered saline containing 1% Tween 20) and after that, incubated overnight at 4°C with a commercial mouse monoclonal antibody against rat and human Epo (1/800) or with a commercial mouse monoclonal antibody against rat NGAL (1/4000), as appropriate. Subsequently, the membranes were incubated with a peroxidase coupled sheet anti-mouse IgG (Amersham, Buckinghamshire, UK) for 1 h. To detect the signal, the blots were processed with a commercial kit (ECL enhanced chemiluminescence system, Amersham, Buckinghamshire, UK). The western blot signal intensity of membranes was densitometrically quantified using specific software (Gel-Pro Analyzer Media Cybernetics). Samples from rats treated with different doses of cisplatin or treated with 5 mg/kg b.w. of cisplatin at different times after injections, as appropriate, were run on each gel with corresponding control samples. To correct for variations in urine production, Epo and NGAL abundances in urine were expressed as optical density (O.D., arbitrary units) related to urinary creatinine concentration (Cru) using the equation [O.D./Vu/Cru] (Vu: volume of urine sample used in the electrophoresis, 10 uL in this work), as previously reported for different urinary proteins.18-29 The value obtained was finally expressed as a percentage of the mean value from the control group. As molecular mass indicators, Kaleidoscope Prestained Standards were employed (Bio-Rad Laboratories, Hercules, CA, USA). A negative sample (using 10 μL of a saline solution) showed no signal.
Statistical Analysis
Statistical analyses were performed using GraphPad software. A Student’s t-test for unpaired data was used to compare two experimental groups; Welch’s correction was employed when variances were not homogeneous. One-way ANOVA followed by the Newman–Keuls test was performed to compare more than two groups. P values of less than .05 were considered significant. The results are presented as the means ± standard error (SE).
Results
Multiple studies on the presence of Epo in urine emphasize the search and refinement of methods for detecting rHuEpo in the urine of athletes as an anti-doping test,12-14 but very little is known about the presence and the role of endogenous Epo in urine. Firstly, we wondered if endogenous Epo could be detected in rat urine by the direct Western blotting technique that has been used in our laboratory for years, without any previous purification step. The urine of four control rats was subjected to Western blotting, employing a commercial antibody, the clone AE7A5 (from R & D Systems, Cat. number MAB2871) that has been previously used in many studies and has shown to recognize effectively Epo protein in the urine.12-14,33,34 As it can be observed in Figure 1, a clear and relatively broad band, of about 30-36 kDa was detected in the urine of four control rats, corresponding to the molecular weight described to the glycosylated form of Epo.8,12,14,34-37 Immunoblot for Epo in urine from four control male Wistar rats. Each lane represents negative sample (lane 1) and different urine samples from four control animals (lanes 2-5) that were separated by SDS-PAGE and blotted onto nitrocellulose membranes exposed to a commercial anti-Epo antibody. Kaleidoscope Prestained Standards of molecular mass (Molecular rule, Mr) corresponding to β-galactosidase (117.593 kDa), BSA (80.845 kDa), Carbonic anhydrase (38.262 kDa), Soybean trypsin inhibitor (30.131 kDa), Lysosyme (15.727 kDa), and Aprotinin (6.099 kDa) were employed. Carbonic anhydrase (38.262 kDa) and Soybean trypsin inhibitor (30.131 kDa) are indicated in the right of the figure. Negative sample (using 10 μL of a saline solution) showed no signal.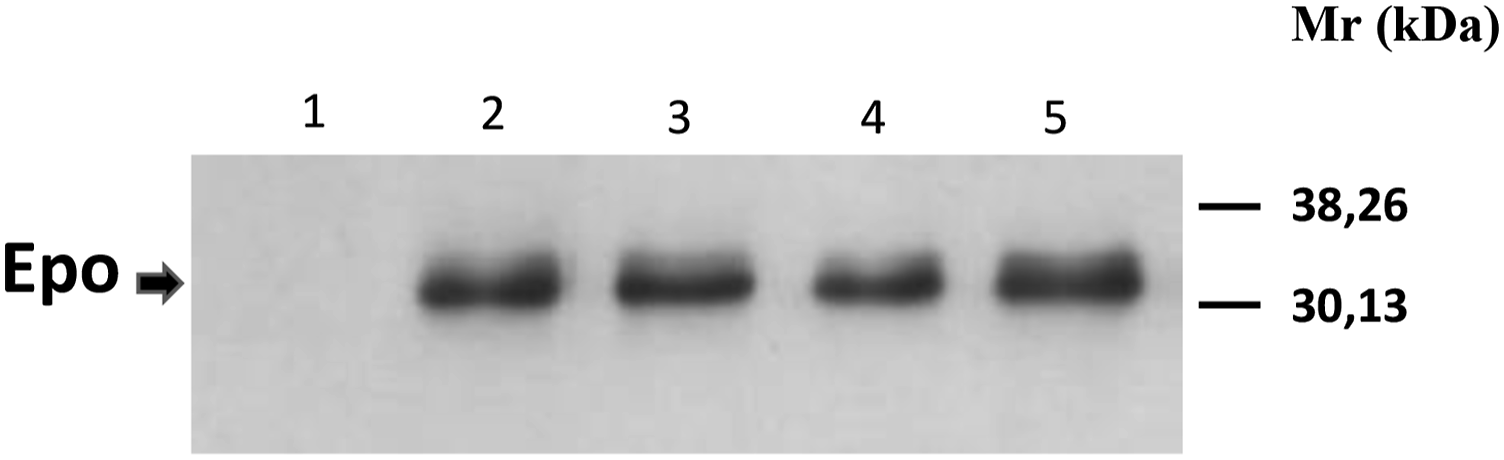
Next, we asked what happened with the urinary excretion of endogenous Epo after AKI. Therefore, we evaluated it in an AKI model induced by cisplatin administration (10 mg/kg b.w., i.p., Cp10 2d, a well-established model of nephrotoxic AKI that has been studied in the past years,18-20,28,31). As observed in Figure 2 cisplatin caused the renal impairment, denoted by a significant increase in urea and creatinine plasma levels,18-20,28,31 and Epo in urine was significantly increased, as compared with control animals. These results demonstrated that Epo urinary levels are modified in response to renal injury caused by cisplatin administration. Urea and creatinine plasma levels, and Epo urinary levels from control (C) and treated male Wistar rats with 10 mg/kg b.w., i.p. of cisplatin, after two days of treatment (Cp10 2d). Densitometric quantification of Epo Western blotting from urine is expressed as arbitrary units related to urinary creatinine concentration to correct for variations in urine production. The mean of the control value was set as 100%. The results are expressed as mean ± SE. (*) P < .05.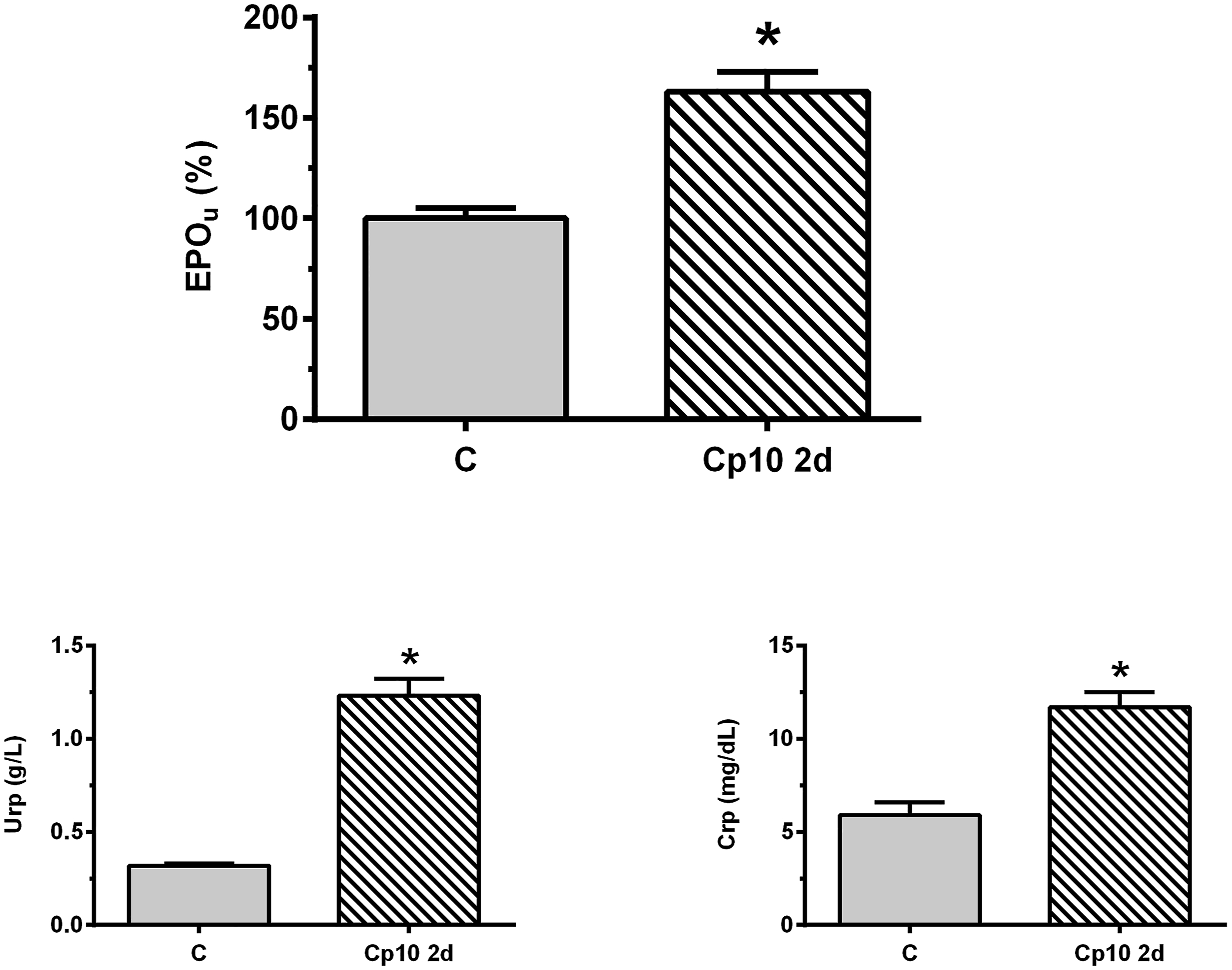
To evaluate the Epo urinary levels in response to AKI in depth, two experimental protocols were conducted in parallel: a dose-dependent study, to evaluate the influence of the degree of nephrotoxicity caused by increasing doses of cisplatin, and on the other hand, a time-dependent study, to evaluate how the urinary levels of Epo behave in the different stages of a cisplatin-induced AKI.
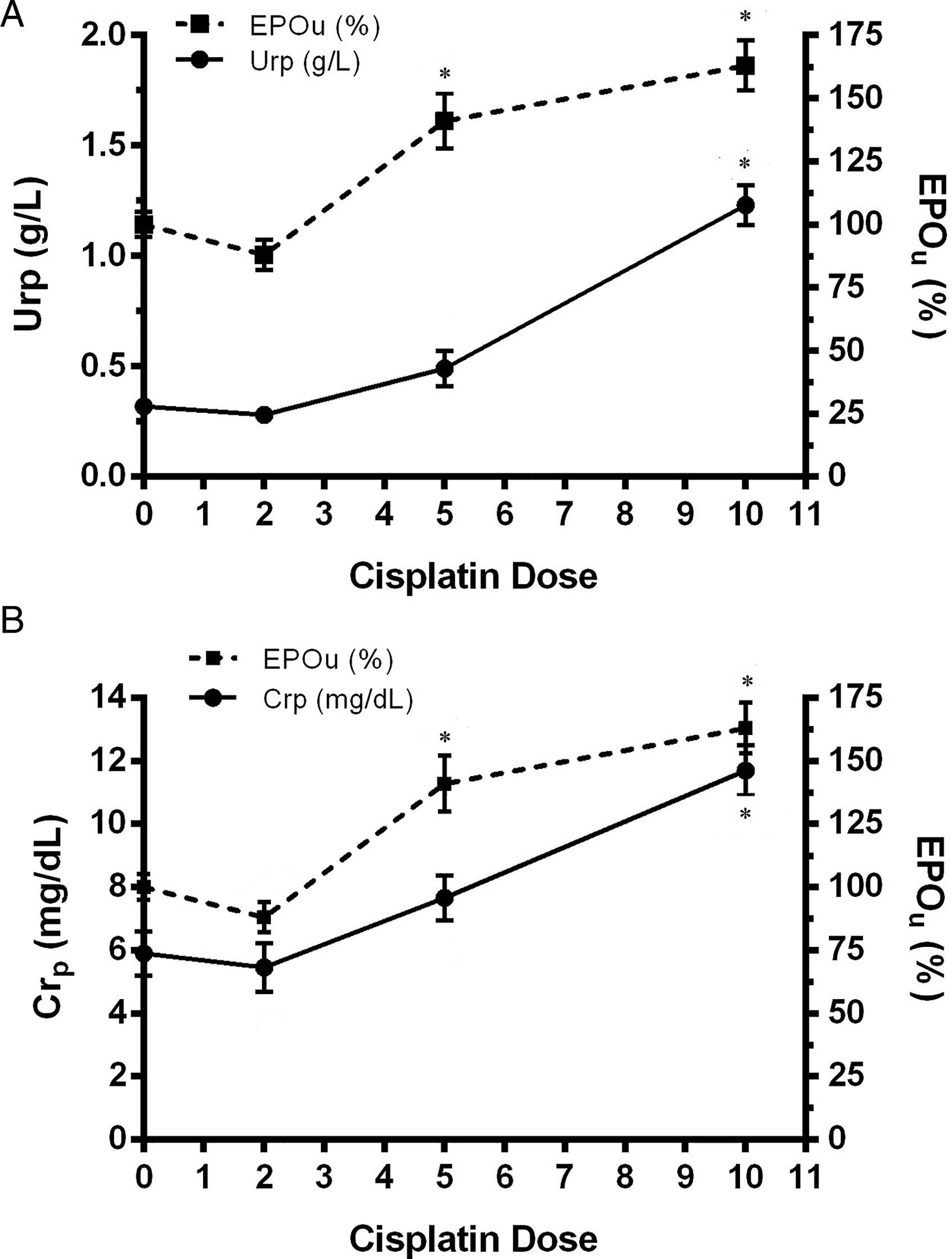
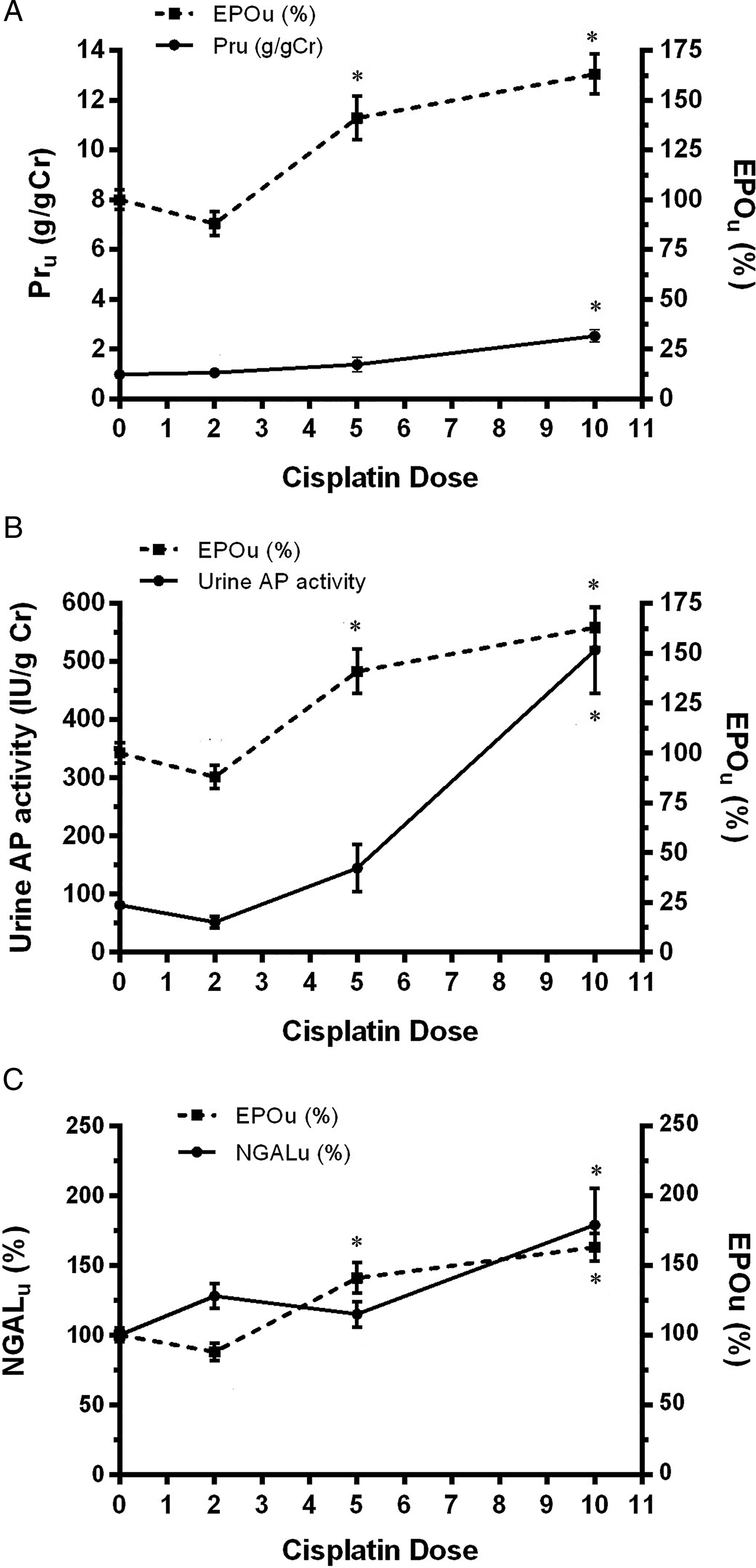
In the dose-dependent studies, Epo urinary levels were analysed after 2 days of 2, 5, and 10 mg/kg b.w., i.p. of cisplatin administration, and compared to traditional (urea and creatinine plasma levels, total proteins in urine levels and AP activity in urine) and novel (NGAL levels in urine) kidney injury biomarkers. Epo in urine was statistically increased, as compared to control values, in the Cp5 2d group, a behaviour that was also observed in the Cp10 2d treated group (Figure 3). In addition, urea and creatinine plasma levels (Figure 4A and B), and total proteins in urine and AP urinary activity (Figure 5A and B) were compared to Epo urinary levels in all experimental groups. All these parameters were statistically increased only at the highest dose of cisplatin, 10 mg/kg b.w., i.p. (Cp10 2d group). These results indicate that Epo in urine was modified at a lower dose of cisplatin than the dose where traditional parameters of kidney injury were altered. Also, NGAL levels in urine were analysed in the different experimental groups and compared with Epo urinary levels (Figure 5C). NGAL in urine was only significantly modified in the Cp10 2d group, at a higher dose of cisplatin than the dose at which Epo in urine started to evidence an increase as compared to control values. Epo abundance in urine from control (C) and treated male Wistar rats with 2, 5, and 10 mg/kg b.w., i.p. of cisplatin after 2 days of treatment (Cp2 2d, Cp5 2d, and Cp10 2d, respectively). Densitometric quantification of Epo Western blotting from urine is expressed as arbitrary units related to urinary creatinine concentration to correct for variations in urine production. The mean of the control value was set as 100%. The experiments were carried out in four different samples for each experimental group. The results are expressed as mean ± SE. (*) P < .05 vs C. (A) Urea plasma levels (g/L) and Epo urinary levels (%), and (B) creatinine plasma levels (mg/L) and Epo urinary levels (%) vs the cisplatin dose (mg/kg b.w., i.p.) employed to treat the male Wistar rats, after two days of treatment. The results are expressed as mean ± SE. (*) P < .05 vs C. (A) Total proteins urinary levels (Pru, g/g Cr) and Epo urinary levels, (B) urinary alkaline phosphatase (AP) activity (IU/g Cr) and Epo urinary levels (%), and (C) NGAL and Epo urinary levels (%) vs the cisplatin dose (mg/kg b.w., i.p.) employed to treat the male Wistar rats, after two days of treatment. NGAL and Epo urinary levels were related to urinary creatinine concentration to correct for variations in urine production. The results are expressed as mean ± SE. (*) P < .05 vs C.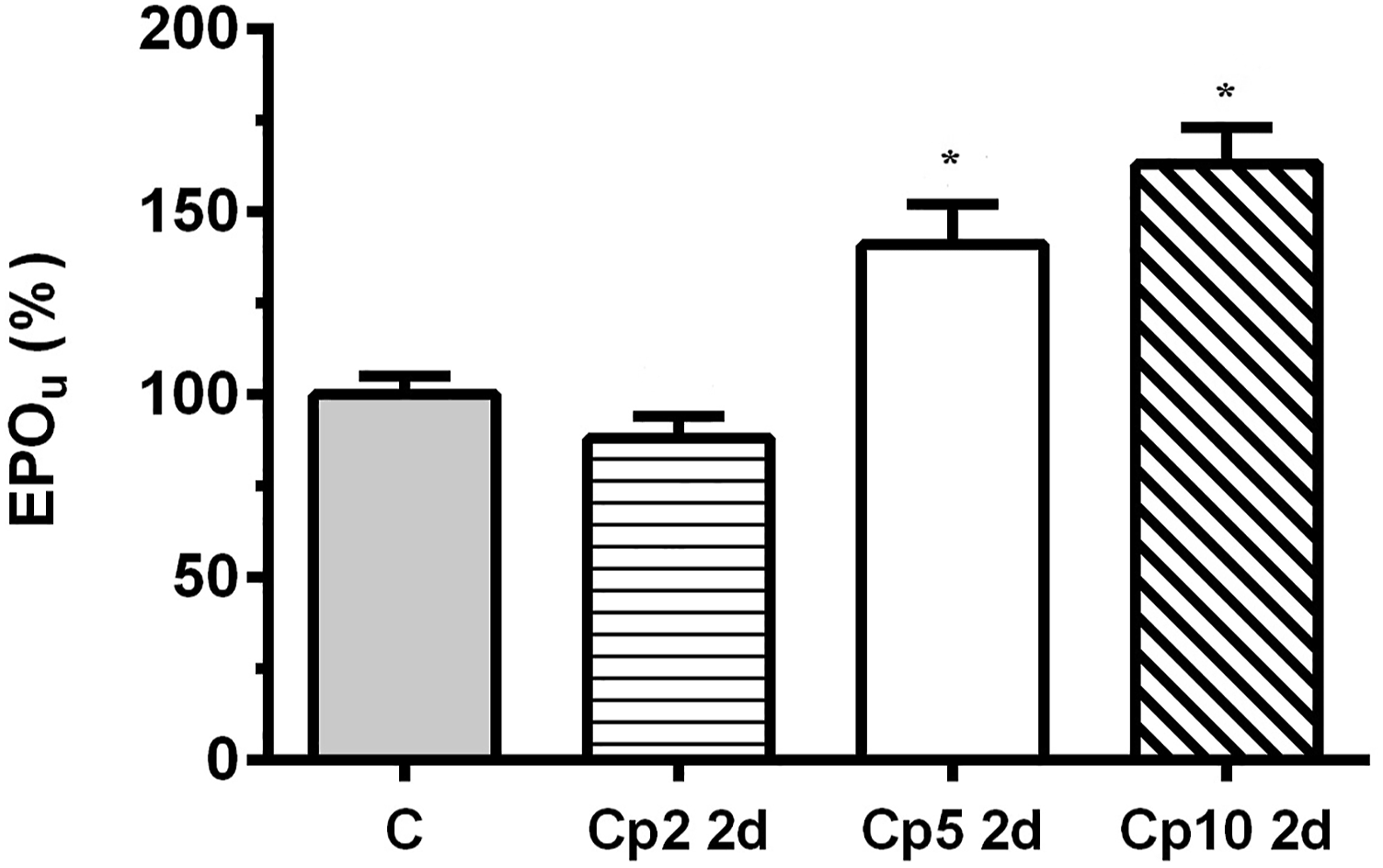
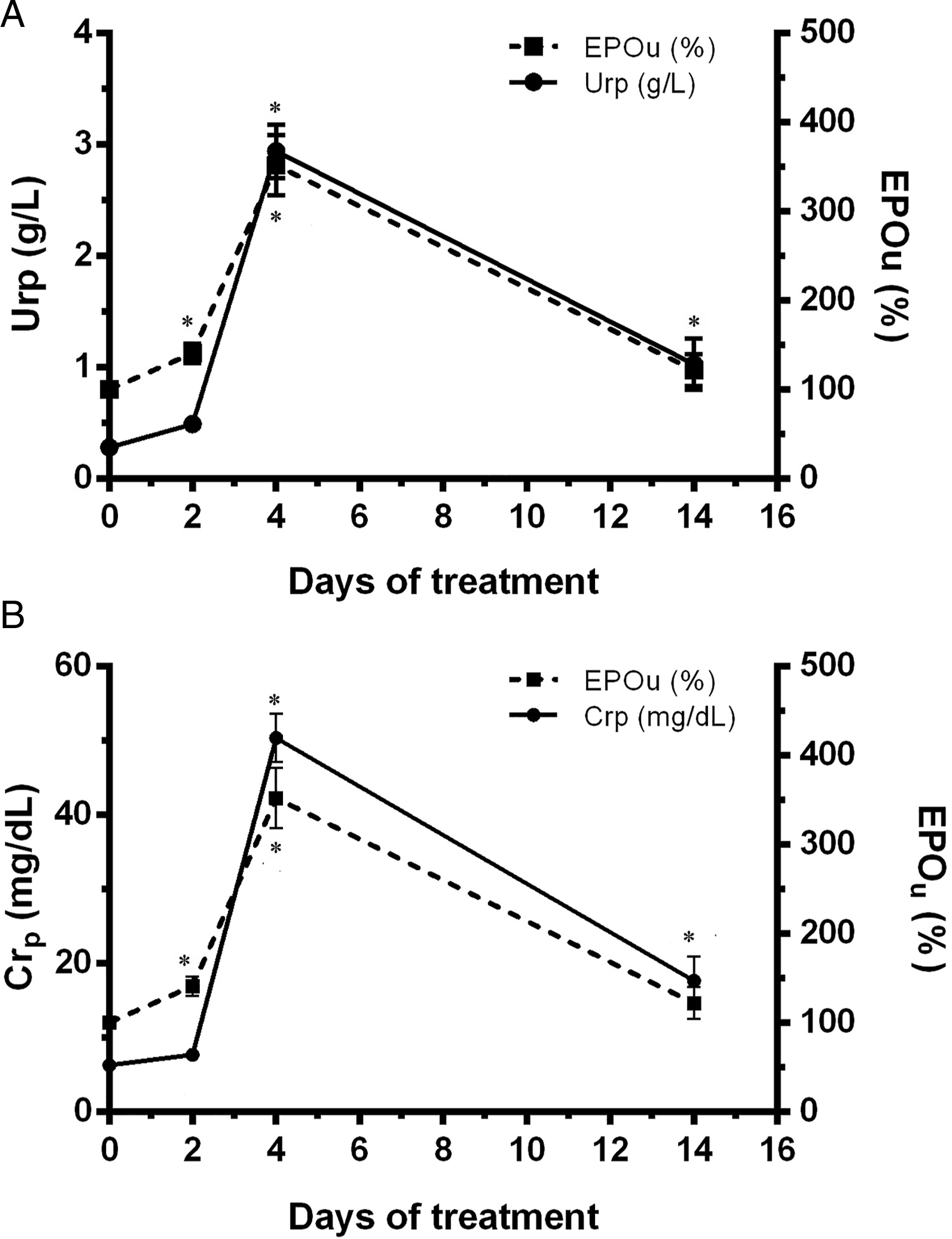
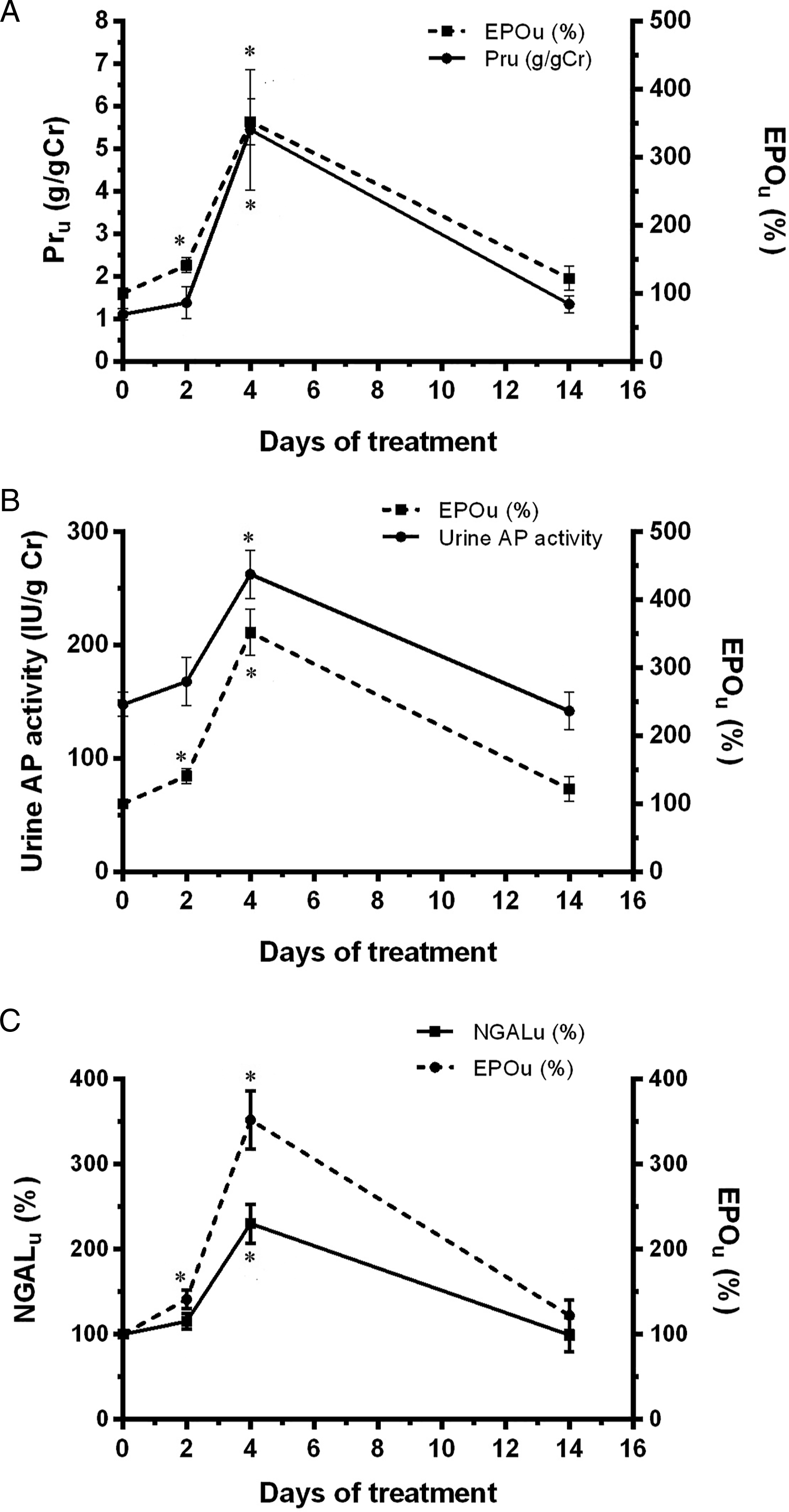
In the time-dependent studies, Epo levels in urine as well as different parameters of kidney injury were evaluated after 2, 4, and 14 days of 5 mg/kg b.w.,i.p. of cisplatin administration. Epo urinary levels were increased 2 days after cisplatin administration, continued increasing by the fourth day of treatment and returned to its basal value by the day 14 (Figure 6). Figures 7 and 8 show the time profile of plasma and urinary kidney injury biomarkers, respectively, and their comparison with the time profile of Epo levels in urine. Urea and creatinine plasma levels were increased by the 4th day of treatment, indicating the induction of renal damage, and then they gradually returned toward their basal value on the day 14, in the renal recovery phase (Figure 7A and B). Urinary levels of total proteins, urinary AP activity, and NGAL urinary levels were also significantly increased on day 4; and returned to their basal values on day 14 (Figure 8A-C Epo abundance in urine from control (C) and treated male Wistar rats after 2, 4, and 14 days of treatment with 5 mg/kg b.w., i.p. of cisplatin (Cp5 2d, Cp5 4d, and Cp5 14d, respectively). Densitometric quantification of Epo Western blotting from urine is expressed as arbitrary units related to urinary creatinine concentration to correct for variations in urine production. The mean of the control value was set as 100%. The experiments were carried out in four different samples for each experimental group. The results are expressed as mean ± SE. (*) P < .05 vs C. (A) Urea plasma levels (g/L) and Epo urinary levels (%), and (B) creatinine plasma levels (mg/L) and Epo urinary levels (%) vs days of treatment with 5 mg/kg b.w., i.p. of cisplatin. The results are expressed as mean ± SE. (*) P < .05 vs C. (A) Total proteins urinary levels (g/g Cr) and Epo urinary levels (%), (B) urinary alkaline phosphatase (AP) activity (IU/g Cr) and Epo urinary levels (%), and (C) NGAL and Epo urinary levels (%) vs days of treatment with 5 mg/kg b.w., i.p. of cisplatin. NGAL and Epo urinary values were related to urinary creatinine concentration to correct for variations in urine production. The results are expressed as mean ± SE. (*) P < .05 vs C.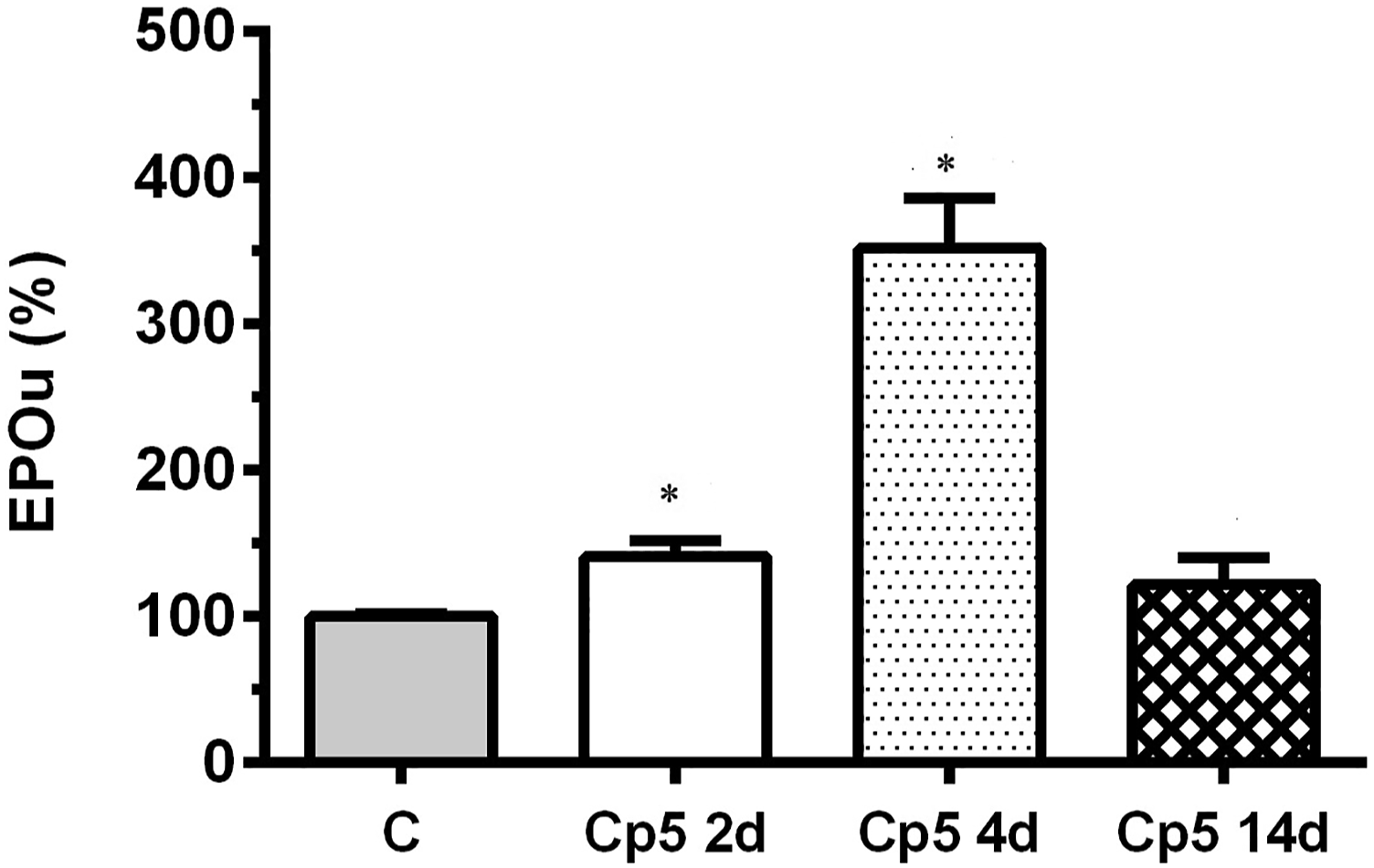
Discussion
In the present work, dose-dependent and time-profile studies of Epo urinary levels in a cisplatin-induced AKI model were performed. In the first study, the animals were treated with 2, 5, and 10 mg/kg b.w., i.p. of cisplatin, and renal injury was assessed 2 days after cisplatin administration by traditional markers such as urea and creatinine in plasma, total proteins in urine, and AP urinary activity. None of those parameters were significantly altered up to the highest dose of cisplatin employed, 10 mg/kg b.w., i.p. where the renal injury was established. In this connection, we have also previously reported renal histopathological alterations, such as tubular dilatation, tubular desquamation cells, and disrupted tubular basement membranes, in rats 2 days post- receiving 10 mg/kg b.w., i.p. of cisplatin. 18 On the other hand, in the present work, Epo levels in urine were found significantly increased at a lower dose, 5 mg/kg b.w., i.p. allowing predicting renal perturbation when no modifications of traditional markers of renal injury were still observed and no histopathological damage was previously reported.18,19
NGAL is expressed in numerous organs and cell types, and it is also found in urine at low levels under physiological conditions. Several experimental and clinical studies have shown that NGAL urinary levels increase significantly in AKI. It is considered one of the earliest and most sensitive novel biomarkers for AKI diagnosis, and also it is one of the novel AKI biomarkers that count with more studies.38,39 In this work, we also analysed NGAL urinary levels in rats treated with increasing doses of cisplatin and compared it with Epo urinary levels. Epo levels in urine were increased at a lower dose of cisplatin than NGAL urinary levels. Taking into account the results obtained so far, Epo urinary levels behaved as a more sensitive biomarker than traditional and novel biomarkers of renal injury in rats.
In the time-dependent study, Epo urinary levels were evaluated in the time course of AKI induced by the administration of 5 mg/kg b.w., i.p. of cisplatin and compared to conventional biomarkers of renal injury, and also with urinary NGAL. Two days after cisplatin administration, Epo urinary levels were significantly increased meanwhile no modifications in hemodynamic (plasma urea and creatinine levels) nor tubular parameters (total proteins in urine and urinary AP activity) were still observed, and no histopathological damage was previously reported.18,19 At this time point, no alterations were found either in NGAL urinary levels. Four days after treatment, traditional renal injury markers, NGAL, and Epo urinary levels were found significantly increased. At this time and with the same dose, impairment in tubular architecture, tubular dilatation, tubular desquamation cells, and epithelial cell detachment from basement membranes were previously observed in our laboratory. 19 Hemodynamic parameters, tubular parameters, and NGAL and Epo urinary levels tended to return to their basal values after 14 days of treatment as it is shown in Figures 7 and 8, in connection with an evident improvement of renal morphology that was also previously reported by Bulacio et al. 19 Thus, urinary Epo was modified earlier in the time-course of cisplatin-induced AKI than traditional and novel biomarkers of renal injury.
Thus, taking into account the results obtained from both studies, we propose urinary Epo as a novel and early biomarker of cisplatin-induced AKI. It is important to highlight that endogenous Epo was detected in rat urines by a simple Western Blot technique, with no pre-purification or pre-concentration method needed resulting a rapid and convenient method of Epo detection in urine, that allow the evaluation of relative differences in Epo concentrations.
Epo is a glycoprotein hormone. Around 90% of systemic Epo in adults is produced in the kidney by peritubular interstitial fibroblasts in the renal cortex and outer medulla. In addition, it has been reported that proximal renal tubular cells are also responsible for Epo production. A feedback mechanism involving oxygen delivery to the tissues regulates Epo production.37,40 In the bone marrow, Epo promotes survival, proliferation, and differentiation of erythrocytic progenitors, particularly the forming units of their colonies. This hormone is metabolized in the liver and undergoes renal excretion. 8 In CKD, impaired Epo production by renal cells causes anaemia. Since the 1980s, rHuEpo has been widely used to treat the anaemia observed in CKD and also to treat anaemia in cancer patients receiving chemotherapy.8,33 Moreover, Epo has been misused as a doping agent for endurance athletes to improve their aerobic performances. Therefore, in the last years, several studies have been conducted to improve rHuEpo detection in urine as an anti-doping test.12-14 Severin et al 41 have recently observed that Epo increases the renal protein expression of the transporters Oat1, Oat3, and Mrp2 which results in alteration in the pharmacokinetics of organic anion compounds. Epo protective effects in different organ injuries have also been reported.8,36,42 Parallel to injury, the kidney develops defensive responses to restrict cellular damage and promote repair, one of which is associated with Epo. Thus, Epo has been proposed as a therapeutic option to improve the outcome of AKI.42-44 Accumulated evidence suggests that the Erythropoietin Receptor/β Common Receptor (EPOR/βcR) mediates the tissue protective effects of Epo. 42 The swift upregulation of EPOR/βcR in the kidney after AKI induced by ischemia-reperfusion suggested that this heterodimer might be a potential early biomarker of this pathology. However, its detection in plasma and urine has not still been reported. 42 Yamashita et al. 45 observed an increase in plasma Epo levels from patients with AKI, but they did not assess urinary Epo. Yasuoka et al. 34 showed that kidney nephrons produce Epo under control conditions and that kidney interstitial cells produce Epo in response to severe hypoxia or anaemia and they assayed Epo levels in plasma and urine samples. Nevertheless, at present, very little is known about the urinary excretion of endogenous Epo in response to kidney injury. Li et al. 11 have observed higher Epo urinary levels during the early postnatal period in critically ill neonates, associated with kidney injury, and Vaziri et al. 10 found increased Epo urinary excretion in patients with Nephrotic Syndrome. Therefore, taking all of the above into account, our work on the behaviour of endogenous Epo in urine in response to kidney injury would also provide new insights into renal pathophysiology.
In the AKI process, numerous alterations take place at cellular and molecular levels that finally result in structural damage and renal dysfunction. In the case of cisplatin, its conversion to a nephrotoxic metabolite in the proximal tubule is required for cell injury. It is conjugated to glutathione and then metabolized through a γ- glutamyltranspeptidase and a cysteine-S-conjugate β-lyase dependent pathway to a reactive thiol, which is a potent nephrotoxin.5,46,47 Cisplatin-induced nephrotoxicity involves increased inflammatory reactions, oxidative stress, fibrogenesis, and tubular cell apoptosis. Hypoxia and mitochondrial injury are also involved in this nephrotoxic mechanism.5,46,47 These renal injuries are manifested as glomerular dysfunction and tubulointerstitial inflammation, leading to AKI.5,46,47 In this connection, it has been reported that Epo production may be increased in some renal pathologies as polycystic kidney disease, renal artery stenosis, hypernephroma, and renal transplantation. 12 Moreover, it has been reported that kidney interstitial cells produce Epo in response to severe hypoxia. 34 The rate of Epo production is regulated by oxygen tension as determined by oxygen supply and demand within the Epo-producing cell. A reduction in oxygen supply associated with hypoxia stimulates Epo production.8,9,12 Thus, the oxidative stress and the hypoxia involved in cisplatin toxicity might increase Epo levels in renal tissue with the consequent Epo excretion in urine. Current experiments are being performed in our laboratory to evaluate the exact molecular mechanisms involved in the increase of urinary Epo after cisplatin-induced AKI. In the present work, Epo in urine is proposed as an early biomarker of nephrotoxic AKI produced by cisplatin. In addition, it would also be of great interest to evaluate urinary Epo as a potential biomarker in AKI of other etiologies.
It is well-known that an early diagnosis of AKI is essential for the establishment of adequate treatment. Over recent years, several promising biomarkers of this pathology have been proposed through cooperation between pharmaceutical and biotechnology industries and regulatory bodies, such as Food and Drug Administration. Studies performed in our laboratory using animal experimental models of renal pathologies have proposed different proteins as urinary AKI biomarkers.15-29 In cisplatin-induced AKI (the experimental model employed in the present work), urinary Oat5 was proposed as a diagnostic, as an early biomarker, and as a biomarker for treatment monitorization, and urinary Cav2 was claimed to be useful as a biomarker of renal recovery.18-20,28 In this connection, urinary Epo has similar behaviour to Oat5 as a diagnostic and as an early biomarker of cisplatin nephrotoxicity, while its application as a biomarker for treatment monitorization remains to be studied. Thus, urinary Oat5, Cav2 and Epo could, in the future, be part of a panel of biomarkers for early detection, diagnosis, and evaluation of disease progression in cisplatin-induced AKI.
Footnotes
Acknowledgments
The authors thank Wiener Lab Argentina for the analytical kits.
Author Contributions
Bulacio, R. contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Torres, A. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript. Both authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the following grants: Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT) (PICT 2017-N°0936), Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) (PIP 2015-2017-N°0460), and Universidad Nacional de Rosario (UNR) (PID 2020-2021).
