Abstract
Acute myeloid leukemia (AML) is the most common subtype of leukemia, accounting for 62% of all leukemia fatalities. As a polyphenol glycoside, hesperidin triggers the apoptotic pathway, which might positively affect combating cancer cells. In this study, we investigated the pro-apoptotic effects of hesperidin in KG1a cells. The MTT assay was used to determine the IC50 of hesperidin in KG1a cell lines. For the apoptotic cell morphology study, we used Hoechst 33 258 staining. Activation of the caspase-3 enzyme was evaluated by the caspase-3 assay and spectrophotometry. Cell cycle distribution was analyzed by propidium iodide staining and flow cytometry. Moreover, p21, survivin, Bax, and Bcl2 gene expression was investigated by real-time PCR. Hesperidin decreased the viability of KG1a leukemic cell4s, but not that of HFF2, a non-cancer cell line. Apoptotic cell morphological alterations and increase in caspase-3 activity were observed after hesperidin treatment. Our results revealed that the expression of anti-apoptotic genes survivin and Bcl2 significantly decreased with hesperidin treatment, and pro-apoptotic gene Bax and cell cycle regulator p21 increased compared to the control group. These findings revealed that hesperidin may be an effective factor in initiating the intrinsic pathway of apoptosis and may be good candidate for the treatment of AML.
Introduction
Acute myeloid leukemia (AML) is characterized by a malignant clonal proliferation of progenitor cells and a halt in differentiation.
1
It is responsible for 1% of cancer deaths worldwide.
2
The disease’s etiology is influenced by the interaction of many environmental variables and genetic backgrounds. Radiation, benzene exposure, obesity, smoking, and chemical carcinogen exposure are all environmental risk factors.3,4 Also, significant chromosomal translocations, point mutations, amplifications, and deletions are genetic factors that might contribute to carcinogenesis.
5
The process of mitochondrial outer membrane permeabilization (MOMP), which is brought on by pores mediated by Bax and Bak (proapoptotic members of the BCL-2 family), causes intrinsic apoptosis.
6
Hesperidin (7-rutinoside) is a flavonoid in citrus fruits with anti-cancer properties. This polyphenolic glycoside triggers the apoptotic pathway, which might positively affect combating cancer cells. Recent study revealed that hesperidin could prevent calcium/calmodulin-dependent protein kinase IV (CAMKIV) activity, thereby inhibitingthe caspase-3-dependent intrinsic apoptotic pathway through the up-regulation of Bax (BCL2 associated X, apoptosis regulator) as a pro-apoptotic protein.
7
Moreover, based on previous studies, p21 over-expression caused cell cycle arrest in cancer cells.
8
Considering the growth of the global statistics of deaths related to leukemia and also the desire of modern science to use the products available in nature to fight diseases, we will examine the cytotoxicity, cell cycle and apoptotic effects of hesperidin (Figure 1A) in acute myeloid KG1a leukemia cells. Hesperidin and its cytotoxicity on KG1a leukemia cell lines. (A) The chemical structure of hesperidin (CAS No. 520-26-3). (B) Cytotoxicity assessment on KG1a and HFF2 cell lines. Results showed that 50% of the KG1A cells died at the concentration of 125 μM of hesperidin. After 72 hrs of exposure, the cytotoxicity of hesperidin against normal human skin fibroblasts cells (HFF2) was measured at different concentrations (5-250 μM). The results are indicated as the mean ± SD of 3 independent experiments. P-values less than 0.01 was considered significant (*P < 0.01, **P < 0.001, ***P < 0.0001) (n = 3).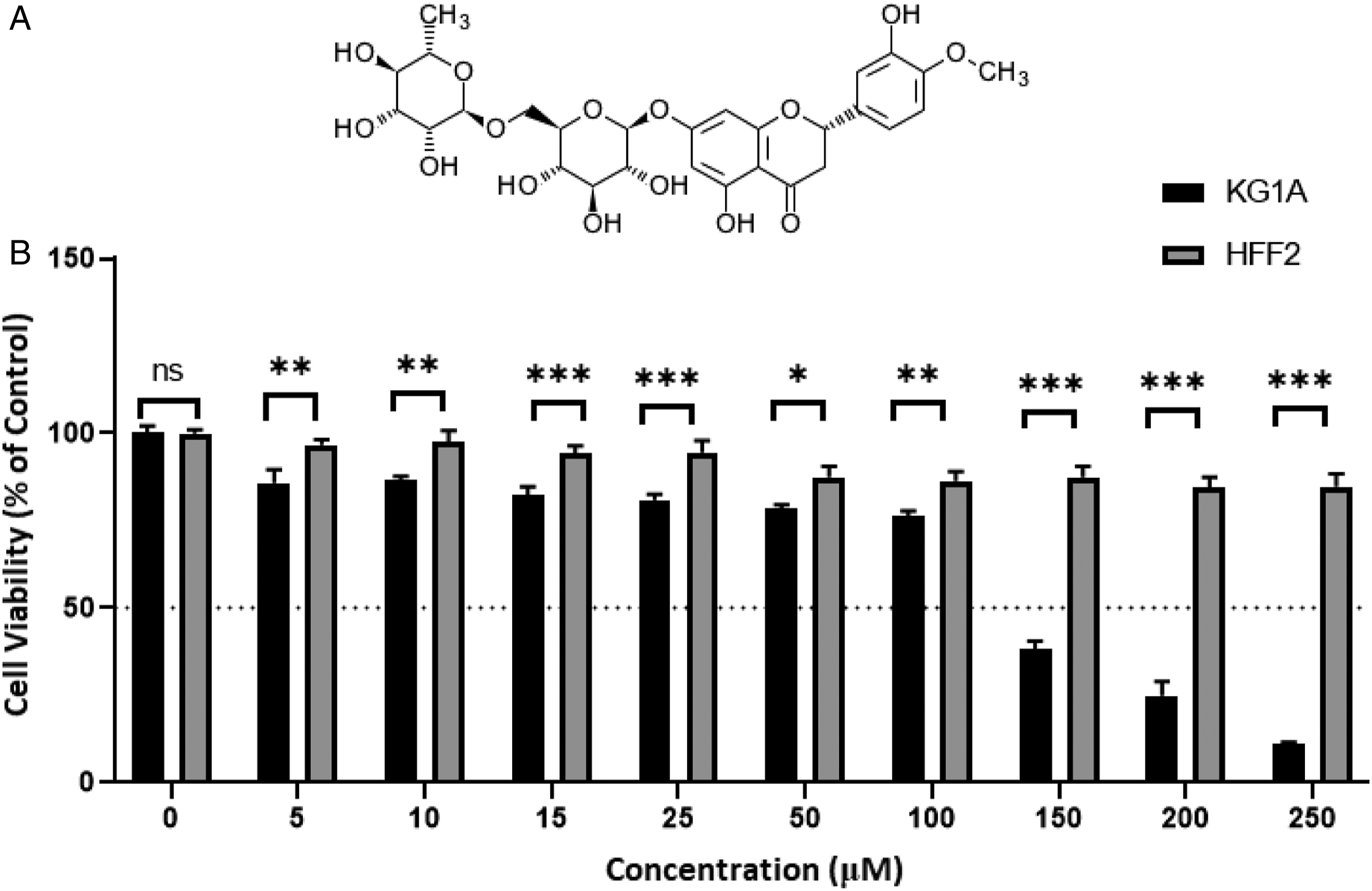
Material and Methods
Materials
KG1a and HFF2 cell lines were obtained from the Pasteur Institute of Iran (Tehran, Iran). The cell culture medium RPMI-1640 was provided by Gibco (Life Technologies, USA). Fetal bovine serum (FBS) was procured from PAN Biowest (France). The cells were cultured in plates (SPL, South Korea). Hesperidin (CAS-Number 520-26-3) with ≥80% purity, was obtained from Sigma-Aldrich (Germany). The cell cycle test was performed using propidium iodide (PI) and for the MTT assay, dimethyl sulfoxide (DMSO), 3-(4, 5-dimethyl-2-thiazolyl)-2, 5-diphenyl-2-tetrazolium bromide, and Hoechst 33 342, were procured from Sigma Aldrich (St Louis, Missouri, USA). The caspase-3 assay kit was purchased from BD Biosciences Pharmingen (San Diego, CA, USA). RNA isolation Kit purchased from Sinaclon (Iran). Real Q Plus 2×Master Mix Green High ROX™ for performing Real-Time PCR was acquired from AmpliQon (AmpliQon, Denmark). This study used the Prime Script™ RT reagent kit (Takara, Japan) for cDNA synthesis.
Cell Culture
Normal human skin fibroblasts (HFF2) and the human acute myeloid leukemia (AML) KG1-a cells were cultured in RPMI 1640 medium in 25 cm2 culture flasks, consisting of 15% fetal bovine serum (FBS) and penicillin/streptomycin (10 000 U/ml penicillin and 10 000 μg/mL streptomycin). Cells were cultured in standard conditions, including humified atmosphere and 5% CO2 at 37°C.
Cytotoxicity Assessment by MTT Assay
The MTT colorimetric assay was carried out to study the effect of hesperidin on cell viability and to calculate the IC50 value in the cells. KG1a and HFF2 cells (2 × 104) were seeded in each well of 96-well plates and treated with hesperidin concentrations from 5 to 250 μM for 72 hrs. Next, 20 μL of MTT (5 mg/mL) was added to each well and incubated for 4 hrs at 37°C. After draining the contents of each well, 100 μL of DMSO was added. The plate was slowly shaken for 15 minutes then the absorbance was read at 570 nM with a multi-well plate reader (Quant BioTek Instruments, USA).9,10 The IC50 values were calculated from the sigmoidal dose-response graph using Graph Pad Prism 6.0.2 software (Graph Pad Software, CA, USA).
Apoptotic Cells Morphological Study With Hoechst 33 258 Staining
Hoechst staining was performed to evaluate the morphologic evidence of apoptosis in untreated and treated cells. Twelve-well plates were seeded with 5 × 105 KG1a cells, and treated with hesperidin for 72°hrs. The cells were then harvested and rinsed with cold phosphate-buffered saline (PBS). Then, Hoechst 33 258 solution (1 mg/mL ddH2O) was added to the cells (final concentration of 100 μg/mL). Fluorescence microscopy was used to examine cellular morphology (EUM-5000 FLCD, Labex Instrument).
Evaluation of Caspase-3 Enzyme Activity
KG1a cells (2 × 10⁵) were seeded in each well of 96-well plates and treated at the IC50 concentration of hesperidin. The test procedure was carried out according to the instructions provided by the manufacturer (caspase-3 assay kit, BD Biosciences, USA). 11 The assessment measures the chromogenic substrate (Ac- DEVD-pNA) cleavage, with caspase-3 activation. The released pNA, which has a direct association with caspase-3 activity, was assessed by a microplate spectrophotometer (Epoch™, BioTek® Instruments, Inc, USA) at 450 nm.
Cell Cycle Distribution Analysis by PI Staining and Flow Cytometry
PI staining method was used to calculate the percentage of cell distribution in different cell cycle phases. In this way, 5 × 10⁵ cells were cultured for each control and treatment (125 μM hesperidin) group. After 72°hrs of treatment, the cells were incubated with PI in a dark room for 30°min (50 μg/mL PI, 100 μg/mL RNase A and 0.5% Triton X-100). The stained cells were analyzed by flow cytometry (BD Biosciences, San Jose, CA, USA). The distributions of cells in the sub-G1, G0/G1, S, and G2/M phases were revealed as DNA histograms. Cell cycle dispersal was evaluated by FlowJo software (Ashland, OR, USA).
Gene Expression Investigation by Real-Time PCR
Sequences of Primers Used in q-PCR.

Statistical Analyses
Tests were done in triplicates and depicted as the mean ± SD. Graph Pad Prism software version 6.02 (GraphPad Software, CA, USA) was used to do the IC50 calculations. Also, data were analyzed by independent sample t-test or one-way ANOVA followed by Tukey’s post hoc test via SPSS 23.0 software (SPSS Inc., Chicago, IL, USA). At the P-value of < 0.05(*), < 0.01(**), and < 0.0001 (***), the results were declared statistically significant.
Results
Hesperidin Decreased the Viability of KG1a Leukemic Cells
HFF2 and the KG1a cells were treated with concentrations of hesperidin ranging from 5 to 250 μM. These cells demonstrated different growth rates after exposure to hesperidin (Figure 1B). The data revealed that KG1a viability decreased with increasing hesperidin dose, and at the concentration of 250 μM of hesperidin for 72°h, the maximum level of cytotoxicity was observed. The concentration of 125 μM was used as IC50 in subsequent analyses. HFF2 cell growth and viability were not reduced in dosage or time-dependent manner by hesperidin treatment.
Apoptotic Cell Morphological Alterations
To confirm that the sub-G1 fraction results from apoptosis, nuclear condensation assay was performed with Hoechst 33 342 staining as described in the experimental procedures. KG1a cells were treated with an IC50 concentration of 125 μM of hesperidin for 72°h.
The treatment caused a time-dependent increase in the nuclear fragmentation and the chromatin condensation without the necrosis induction. These observations verified the results of the cell cycle assay. Chromatin condensation and nuclear fragmentation assay were executed with Hoechst 33 258 staining as described in the methods to survey morphological changes on treated cells (Figure 2A). The untreated KG1-a cells revealed round nuclei with normal and spindle-shaped cell membranes (Figure 2B). The morphological alterations of KG1a cells after treatment with hesperidin. (A) Test group treated with 125 μM hesperidin (72 hr). Hoechst 33 342 staining was used to detect cell apoptosis. Chromatin condensation, nuclear fragmentation, and apoptotic bodies were observed in apoptotic cells (arrows). (B) Control group with no apoptosis.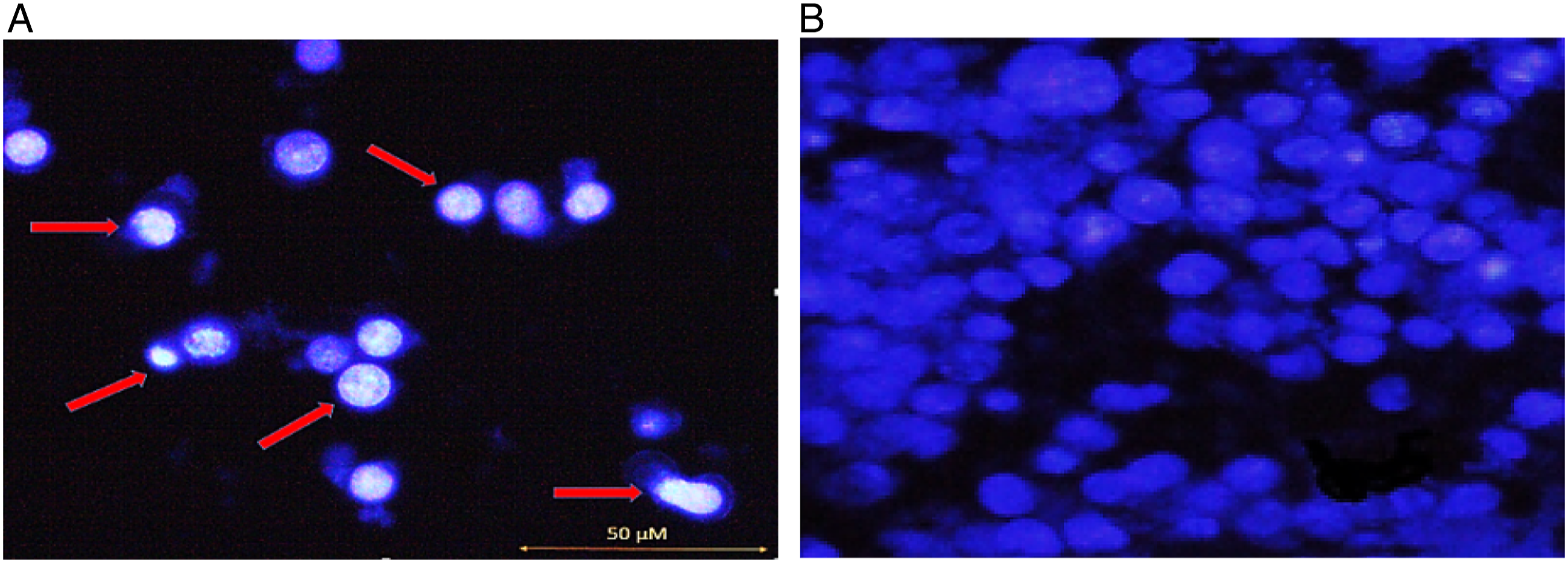
Assessment of Caspase-3 Enzyme Activity
The enzymatic activity of caspase-3 as a marker of apoptosis was measured using the spectrophotometric approach to validate the occurrence of apoptosis. Compared to the untreated control cells, treatment of the KG1a cells with 125°μΜ hesperidin resulted in a time-dependent increase in caspase-3 activity. After 72°hrs of exposure, the reactivity was 1.33 times higher. These outcomes confirmed the quantitative and qualitative results of the cell cycle and Hoechst staining. (Figure 3). The fold differences in caspase-3 activity between hesperidin-treated (125 μM for 72 hr) and vehicle treated KG1a cells. Data are shown as mean ± SD. ANOVA used to compare the treated and vehicle treated Groups (*P < 0.1) (n = 3).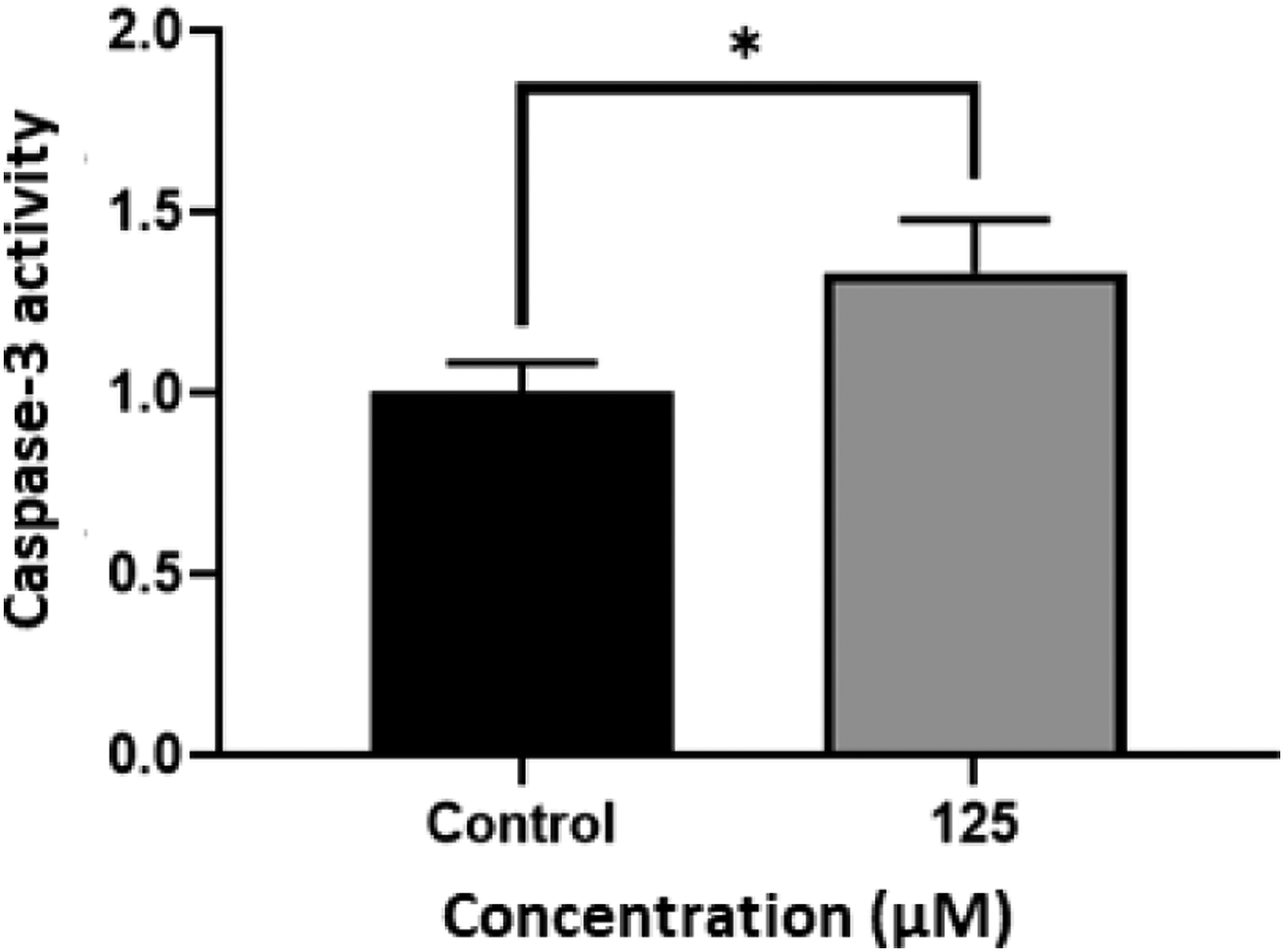
Hesperidin Might Cause an Arrest in S Phase of the Cell Cycle
Cell cycle analysis was performed using flow cytometry. According to our findings, control cells were distributed almost 0.92, 43.84, 8.57, and 8.78% across sub-G1, G0/G1, S, and G2/M phases, respectively. The cell cycle distribution altered to 3.26, 41.02, 11.03, and 8.95% at sub-G1, G0/G1, S, and G2/M phases, after a 72-hour treatment with 125°μΜ hesperidin. (Figure 4A-C). Cell cycle analysis by flow cytometry in the vehicle treated and hesperidin treated KG1a cells. (A) Cell cycle distribution in KG1a control cells; (B) Cell cycle distribution in KG1a cells after incubation with 125 μM hesperidin for 72 h (n = 3). Flow cytometry was used to examine the proportion of untreated and treated KG1a cells in each cell cycle phase. (C) The graph shows the statistical quantification of results following hesperidin exposure. The diagram depicts the statistical quantification of data following hesperidin treatment. Increase in S phase of cell cycle indicated S phase cell cycle arrest after 72 h. The results were analyzed by FlowJo software.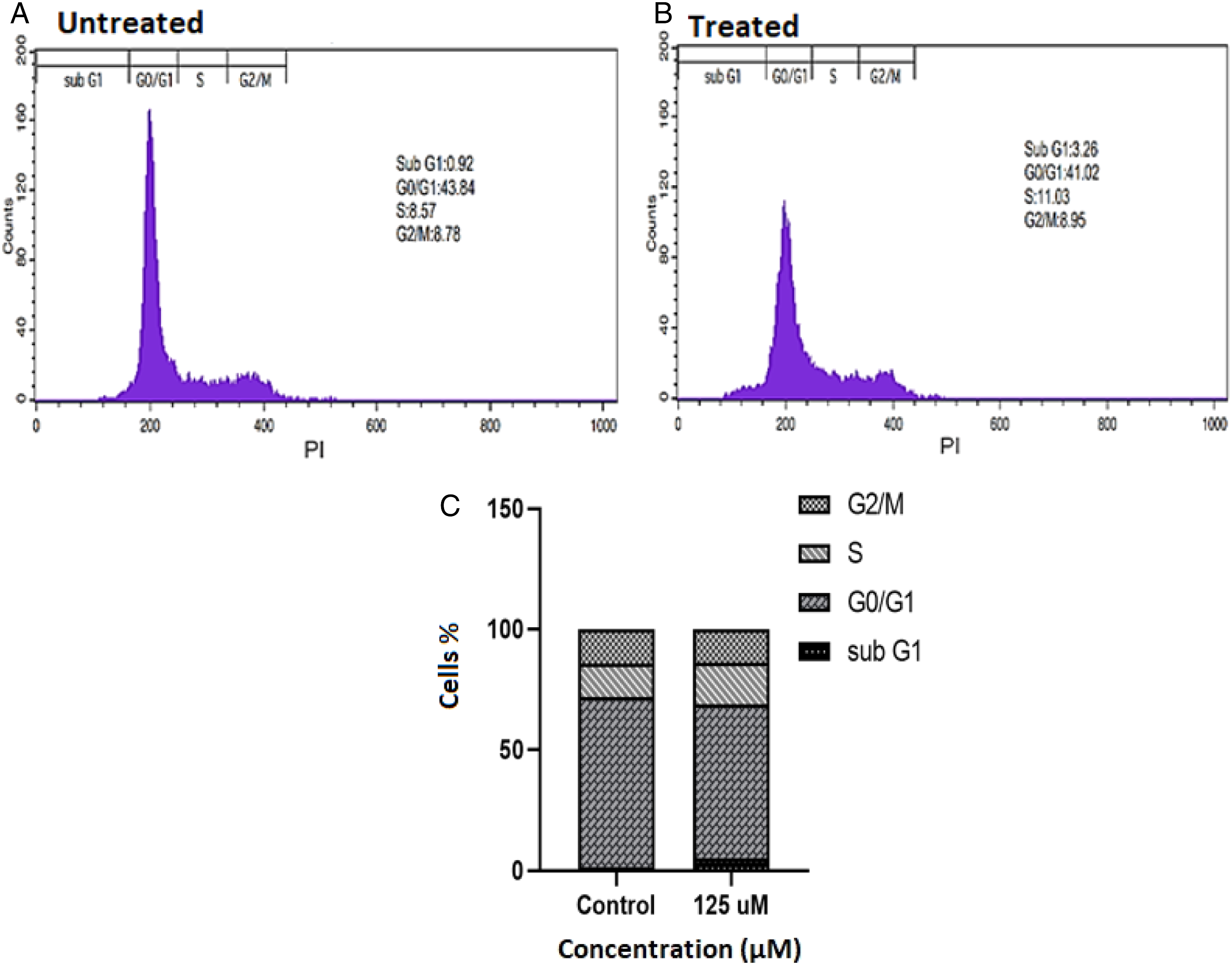
Assessment of Gene Expression Levels by Real-Time PCR
After the KG1a cells were exposure to the hesperidin, the transcription levels of Bax, Bcl2, p21, and survivin were measured (Figure 4). Based on our findings, after 72 hrs incubation of KG1a cells with IC50 concentration (125 μM) of hesperidin, the expression of Bax was significantly increased by about 19.53-fold (**P < 0.001) (Figure 5A). Furthermore, Bcl2 gene expression was reduced in the treatment group by 5-fold, compared to the control group (**P < 0.001) (Figure 5B). The gene expression level of p21 was 5.08-fold higher in the treated group in comparison with the control group (**P < 0.001) (Figure 5C). The results also revealed that survivin showed a reduction ( Measurement of mRNA expression levels after 72 h incubation with hesperidin. q-PCR was used to evaluate variations in transcription levels of Bax (A), Bcl2 (B), p21 (C) and Survivin (D) genes. The results are expressed as mean ± SD; Treated and vehicle treated groups were compared by ANOVA (Tukey’s post hoc test). (**P < 0.001) (n = 3).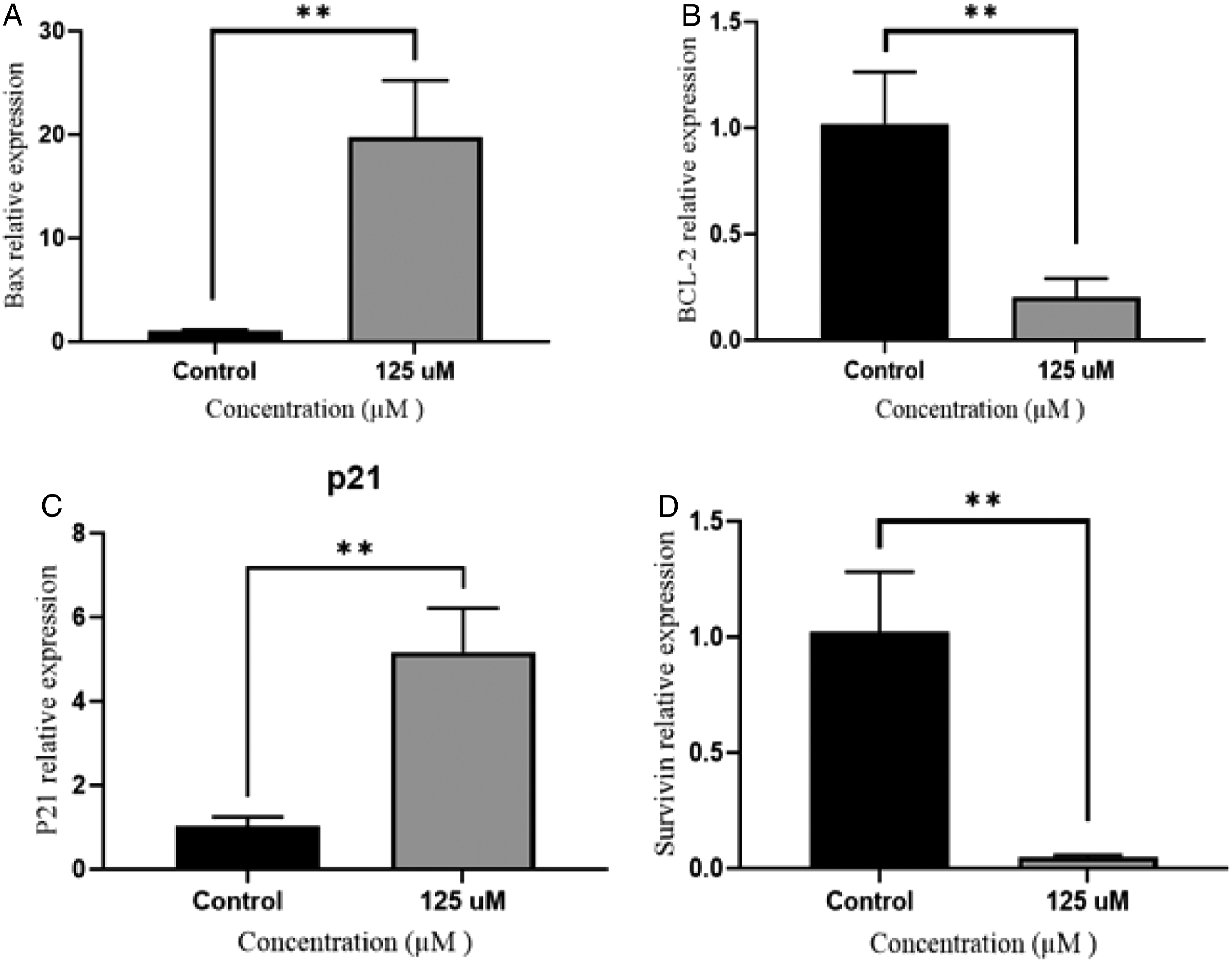
Discussion
In this study, according to the results of the MTT assay and Hoechst staining, apoptosis was induced by 125 μM of hesperidin in KG1a cells after 72°hrs (Figure 1 and Figure 2). Also DNA condensationand fragmentation was observed in KG1a cell line. We showed that hesperidin increased cell death in KG1a cells but not in the non-cancer HFF2 cell line. In this study, we also examined the activity of the caspase-3 enzyme. The results of this experiment showed an increase in the activity of caspase-3 due to the treatment of KG1a cells with hesperidin (Figure 3). According to a study on MCF-7 breast cancer cells, treating the cells with the concentration of 80 μM of hesperidin trigged shrinkage of the cells. It created membrane blebs and cell disintegration, and an increase in the amount of caspase-3 enzyme, eventually leading to apoptotic cell death. 13 In this study, by performing real-time PCR on genes related to the cell death pathway, the expression level of Bax and p21 mRNA in the untreated control cells were revealed to be lower than in the treated KG1a cells. This result showed the pro-apoptotic effect of hesperidin on KG1a leukemia cells. Similar to our research, another study by Ismail et al. showed that hesperidin increased the level of ROS in HCT11b colon cancer cells, and an increase in Bax and p21 gene expression induced apoptosis in this cell line. 14 Based on the findings of Banjerdpongchai et al. in hesperidin-treated HepG2 cells, Bax protein increased and apoptosis occurred through the mitochondrial and death receptor pathway. 15 In a study by Byun et al., hesperidin, which was modified to increase its effectiveness by gamma rays, reduced the expression of Bax and cytochrome C genes in melanoma cancer cells (B16BL6). It also reduced metastasis in mice with lung cancer. 16 According to another study, concentrations of 50 and 100 mg/kg of hesperidin in gamma-ray-affected mice increased the Bax/Bcl2 ratio in peripheral blood lymphocytes. 17 Kyung et al showed that a concentration of hesperidin (152.3 μL) increased the accumulation of MSTO.211H cells in the sub-G1 phase and the cell death was induced by increasing the mRNA expression of Bax and Caspase-3 genes. 18 In a study by Park et al., a 100 μM concentration of the hesperidin reduced growth of intestinal cancer cells (SNU-C4) by 65% compared to control cells. It also decreased Bcl2 mRNA expression level and increased caspase-3 activity in this cell line. 19 Based on the results of real-time PCR, when KG1a cells were treated with 125 μM of hesperidin, a significant increase in the expression of Bax and p21 genes, as well as a decrease in Bcl2 and survivin gene expression led to apoptotic cell death (Figure 5). Similar to our study, a study on lung cancer A549 cells treated with hesperidin showed that down-regulation of Bcl2 associated with up-regulation of caspase-3 and -9 can induce apoptosis. 20 These findings were consistent with our results and indicated that hesperidin contributes to the initiation of the intrinsic pathway of apoptosis. Another study by Cincin et al, indicated that increase in the expression of Bax pro-apoptotic gene and decrease in Bcl2 anti-apoptotic gene can promote apoptosis cell death in endometrial cancer cells (ECC-1) through the ERK/MAPK signaling pathway. 20
In this study, we also examined the cell cycle in leukemia KG1a cells treated with hesperidin. We observed S-phase arrest in the cells exposed to hesperidin (Figure 4). According to the findings of Pandey et al, a concentration of 200 μM hesperidin for 24°hrs increased caspase-3 activity and in contrast with our research, they showed an arrest in G2/M phase in gallbladder cancer cells. 21 Zhu et al also showed that over expression of the p21 might be related to S phase-arrest and finally cell death. 22 According to our results, by treating KG1a cells with hesperidin, the expression of the p21 gene increased and cell cycle arrest happened in S phase. In another study, the effect of hesperidin on glioblastoma, as a highly invasive brain tumor cells, increase in Bax and decrease in Bcl2 gene expression was observed due to induction of the p38/MAPK pathway. 23 Looking at the results of survivin gene expression after treatment of leukemia KG1a cells with hesperidin, we found that hesperidin induces the apoptosis process by decreasing the survivin gene expression level. Another study of malignant pleural mesothelial cells showed that MSTO-211H cells were subjected to death by reducing survivin gene expression when exposed to a 152.3 μM concentration of hesperidin for 48°hrs. Moreover, activation of caspase-3, as well as up-regulation of Bax, p21, and cyclin D1 contribute to apoptosis induction in the cells. 18 The Rauch et al. findings also showed that overexpression of p21 causes S phase cell cycle arrest in colorectal cancer cells and a reduction in survivin mRNA expression level leads to cell death. 24
Conclusion
Our findings revealed that a 125°μΜ concentration of hesperidin causes S phase cell cycle arrest in KG1a cells. The cell cycle arrest was associated with a reduction in survivin and Bcl2 mRNA expression and an increase in Bax and p21 gene expression following by increased caspase-3 activity and induction of apoptosis. Based on alteration in the expression of Bax, Bcl2 and survivin which play an important role in intrinsic pathway of apoptosis, so we can conclude that hesperidin contributes to the initiation of the intrinsic pathway of apoptosis.
Footnotes
Acknowledgments
The authors appreciate the support of this investigation by the research council of the University of Tabriz, Tabriz, Iran. Also we would like to thank Dr Morteza KosariNasab for providing the hesperidin.
Author Contributions
Tannaz Novinbahador contributed to acquisition, analysis, and interpretation and drafted manuscript; Mostafa Araj-Khodaei contributed to conception and drafted manuscript; Majid Mahdavi contributed to conception and design and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
