Abstract
Next-generation urinary protein biomarkers have been qualified to enable monitoring for drug-induced kidney injury in toxicology studies conducted in rats. However, there is limited literature on the utility of these biomarkers in dogs. To add to the existing body of knowledge on the utility of the next-generation drug-induced kidney injury (DIKI) biomarkers, we evaluated the value of these biomarkers for the early detection of DIKI in Beagle dogs using a differentiated nephrotoxicant, Amphotericin B (AmpB). In dogs with AmpB-induced kidney injury, we monitored the response of urinary albumin, total protein, clusterin, kidney injury molecule 1, neutrophil gelatinase-associated lipocalin and N-acetyl-beta-D-glucosaminidase. We also measured blood urea nitrogen, serum creatinine and cystatin C. The results showed that urinary clusterin (up to ∼ 112x) was much more sensitive to AmpB-induced kidney injury relative to other biomarkers. Moreover, other than urinary clusterin and to a much lesser extent urinary albumin and total protein, none of the other biomarkers analyzed in this study were more sensitive than blood urea nitrogen and serum creatinine. The AmpB related tubular alterations were characterized by minimal to mild, multifocal necrosis, degeneration, regeneration, dilatation and mineralization. The mild nature of these histopathologic findings further attested to the sensitivity of urinary clusterin to AmpB-induced kidney injury in dogs. These results will help drug developers make informed decisions when selecting urinary biomarkers for monitoring DIKI in dogs for toxicology studies.
Introduction
Blood urea nitrogen (BUN) and serum creatinine (sCr) are the traditional biomarkers of nephrotoxicity; however, they have been considered to have relatively low sensitivity in preclinical safety studies. 1 This is due to the fact that elevation of BUN and sCr are usually not observed until approximately two-thirds to three-fourths of the nephrons are non-functional. 2 Non-renal factors such as species and gender may influence the value of BUN and/or sCr when used to detect nephrotoxicity. 3 The Critical Path Institute’s (C-Path) Predictive Safety Testing Consortium’s (PSTC) Nephrotoxicity Working Group (NWG)4,5; and the Health and Environmental Sciences Institute (HESI) Committee for Nephrotoxicity Biomarkers 6 have methodically evaluated eight next-generation drug-induced kidney injury (DIKI) biomarkers. Urinary total protein (Ur TP), albumin (Ur ALB), clusterin (Ur CLU), kidney injury molecule 1 (Ur KIM-1), N-acetyl-beta-D-glucosaminidase (Ur NAG), β-2-microglobulin (Ur β2M), cystatin C (Ur CYS C), trefoil factor 3 (Ur TFF3), and renal papillary antigen 1 (Ur RPA-1) have been qualified for preclinical use by the U.S. Food and Drug Administration (US FDA), the European Medicines Agency (EMA) and the Pharmaceutical and Medical Devices Agency, Japan (PMDA) to enable monitoring for drug-induced kidney injury (DIKI) according to specified “context of use” (CoU) in toxicology studies conducted in rats4-6; The CoU is a concise description of the biomarker’s intended use in drug development and includes the BEST (Biomarkers, EndpointS and other Tools) category7,8; The aforementioned qualified rat kidney safety biomarkers are intended to be utilized in conjunction with traditional biomarkers, BUN and sCr. 5 Based on the evidence from breakthrough studies conducted in rats by the PSTC NWG, 9 the US FDA 10 and the EMA 11 have issued a “Letter of Support” for two additional biomarkers, osteopontin (Ur OPN) and neutrophil gelatinase-associated lipocalin (Ur NGAL), as potential translational DIKI biomarkers; however, additional prospective nonclinical and exploratory clinical studies are warranted12,13;
While DIKI biomarkers have proven useful for screening compounds in early drug development in rats, 14 similar utilities in dogs (Canis familiaris) following the administration of potential new molecular entities, remain less understood. 15 Data from exploratory studies with prototypical nephrotoxicants suggests that DIKI biomarkers may be utilized when monitoring DIKI in dogs, which is one of the most commonly used non-rodent species in regulatory toxicology studies. 16 However, there is limited literature regarding the performance of the DIKI biomarkers in dogs.17-20 In the quest for detecting early reductions in glomerular filtration rate (GFR) in dogs, systematic evaluations intended to better understand the COU for newer biomarkers of nephrotoxicity in this model are needed. 21 If next-generation urinary protein biomarkers can detect DIKI and/or alterations in kidney function in dogs with more sensitivity and specificity than traditional, non-specific biomarkers, the drug development process would be remarkably enhanced, particularly for drugs with potential nephrotoxic liabilities in humans. 22
Studies evaluating the utility of the next-generation DIKI biomarkers in Beagle dogs have been previously conducted with nephrotoxicants that have distinct mechanisms of nephrotoxicity including gentamicin and cisplatin.16,20,23,24 To build on the existing body of knowledge, we evaluated the value of these biomarkers for the early detection of drug-induced kidney injury (DIKI) in dogs dosed with another nephrotoxicant with a differentiated mechanism of nephrotoxicity. As such, we used the antifungal agent, amphotericin B (AmpB), which is known to induce dose-limiting nephrotoxicity in multiple species.25,26 We believe the results from this study will add to the existing body of knowledge on the utility of these next-generation DIKI biomarkers in Beagle dogs used in toxicology studies.
Materials and Methods
Ethical Statement
All procedures were conducted under an Institutional Animal Care and Use Committee approved protocol at an Association for Assessment and Accreditation of Laboratory Animal Care International accredited facility which conformed to the current recommendations from the Guide for the Care and Use of Laboratory Animals. 27 This work was conducted in accordance with the 3Rs (Replacement, Reduction and Refinement) policy. The Beagle dogs were first utilized to evaluate selected toxicokinetic (TK) parameters following single low dose administration of a proprietary compound which was structurally unrelated to AmpB. All dogs underwent an extensive wash-out period of at least 1 month prior to the animal selection phase for the exploratory study with AmpB.
Animal Selection and Biofluid Samples Collection
One year old male and female beagle dogs were purchased from Marshall Farms, North Rose, NY, USA. For the purpose of this study, animals were individually housed. Animals were randomly assigned to either a control group or a test article group (6 per group; 3 males and 3 females in each group). Water, food, and enrichment were provided. All animals had access to water ad libitum by an automatic watering system and/or water bowls. Animals were provided approximately 300 g/day of Advanced Protocol™ Non-Certified High-Density Canine Diet 5L18 (PMI™ Nutrition International, Richmond, IN, USA) for ad libitum consumption via a food bowl. The first day of dosing was considered study day 1 (D1). Food was provided post dose. Any food remaining from the previous day was discarded prior to dosing. Cage side examinations were conducted daily at 1 hr and 6 hr post-dose to identify clinical signs of toxicity, moribundity and/or mortality. Food consumption was assessed at pre-dose (Day −3) and once daily thereafter.
Using a pan catch method, representative urine samples were collected on wet ice for an 18-hr period overnight pre-dose (on Day −5 or D-5); and for an 18-hr period (6-24 hr post-dose) overnight after daily dosing. Urine collected overnight on ice after the first dose (but before the second dose was administered) are referred to as Day 1 (D1). Similarly, urine overnight collection after second and fourth doses have been referred to as Day 2 (D2) and Day 4 (D4), respectively. Total urine volumes (Ur Vol) were recorded and immediately placed on wet ice for up to 1 h prior to centrifugation at 800 x g for at least 10 min at 4°C. Urine supernatants were prepared as aliquots and stored at −80°C until time of urine chemistry parameter measurements. Where relevant, the concentrations of urine-based markers were normalized to concurrent urine creatinine (Ur Cr) concentrations. Whole blood samples were collected from unfasted animals by lateral saphenous venipuncture into serum separator tubes and maintained at room temperature (RT) for up to 1 hr prior to centrifugation at 2400 x g for at least 10 min (min) at 4°C. Serum fractions were prepared as aliquots and stored at −80°C until time of serum chemistry parameter measurements. Serum was collected pre-dose and during treatment (24 hr post-dose).
All animals selected for the in-life study phase showed baseline serum biochemistry and urinalysis parameter values that were within the normal reference ranges for healthy, adult male and female beagle dogs, in the laboratory where the samples were collected and analyzed.
Test Article Administration
Non-naïve male and female beagle dogs (n = 3/sex/group) were treated with 0.3 mg/kg of AmpB for Injection, USP (X-Gen Pharmaceuticals, Inc., Big Flats, NY, USA) which was formulated in vehicle (5% dextrose solution for injection, USP, Hospira, Inc., Lake Forest, IL, USA). The dose of 0.3 mg/kg/day was selected and was expected to cause mild/moderate nephrotoxicity in dogs. The dosing volume of 2 mL/kg was administered by intravenous infusion at a rate of 1 mL/min once daily for 4 consecutive days. Individual body weights were measured at pre-dose and prior to necropsy. The dose, route of administration, and duration of dosing were selected based on literature26,28-30 and initial pilot dose range finding study conducted in dogs.
Serum-Based Biomarker Measurements
BUN, sCr and sCys C were measured in serum samples by utilizing Siemen’s reagents validated for dogs on a Siemens Advia 1800 automated chemistry system (Siemens Corporation, Washington, DC, USA).
Urine-Based Biomarker Measurements
All urine samples were examined for characterization of color and clarity. Urinary pH, glucose, occult blood, specific gravity (SP GR), urobilinogen, bilirubin, and ketones were assessed with dipstick using a Mission U120 Urine Analyzer (ACON Laboratories, Inc., San Diego, CA, USA). Urinary concentrations of N acetyl beta D glucosaminidase (Ur NAG), albumin total protein (Ur TP) and Ur Cr were measured utilizing Siemen’s reagents on a Siemens Advia 1800 automated chemistry system and reagents (Siemens Corporation, Washington, DC, USA). Various novel urinary biomarkers were measured using singleplex immunoassay kits according to the manufacturer’s instructions. Concentrations of Ur CLU were quantified using a canine-specific ELISA kit (BioVendor, L.L.C., Ashville, NC, USA); Ur KIM-1 was quantified using a canine-specific ELISA kit (Immunology Consultants Laboratory, Portland, OR, USA); and Ur NGAL was measured using a canine-specific ELISA kit (BioPorto, Copenhagen, Denmark).
Toxicokinetics
The systemic exposure to AmpB was assessed for all dogs on D1 (first day of dosing) and D4 (fourth day of dosing) at pre-dose, 0.25, 1, 3, 6, and 24 hr post-dose. At each time point, blood was collected via a taped-on butterfly catheter from the jugular vein into K2EDTA-treated microtubes and placed on wet ice before being processed to prepare plasma samples. AmpB plasma concentration was measured using a proprietary liquid chromatographic-triple quadrupole mass spectrometric method.
Kidney Histopathology
After 4 days of dosing, animals were euthanized, and representative kidney tissues were collected and immersed in 10% neutral-buffered formalin fixative. The formalin-fixed kidney (left and right) samples were processed, embedded in paraffin, sectioned at 4-μm, mounted on glass slides, de-paraffinized, and stained with hematoxylin and eosin for subsequent microscopic evaluation in a blinded-fashion by a pathologist. Histopathology lesion grading followed the recommended histopathology best practices in the context of kidney safety biomarker assessment in dogs 16 which represent adapted approaches as were described previously for exploratory DIKI biomarker studies in rats. 9 Severity grading was assigned based on the classification of microscopic evidence of lesions which was characterized as minimal, mild (slight), moderate, marked or severe.
KIM-1 Immunohistochemistry Staining
After 4 days of dosing, representative formalin-fixed left and right kidney tissues were immunostained for KIM-1 as described previously. 16 Briefly, kidney paraffin blocks were sectioned at 4 μm, mounted on glass slides, deparaffinized, and hydrated in phosphate buffered solution (PBS). Endogenous peroxide was blocked using 3% hydrogen peroxide solution. To avoid nonspecific reaction with primary antibody, slides were pretreated with 10% normal donkey serum. Slides were incubated for 30 min at 4°C with 10% normal rabbit serum before incubation for 2 hours at 4°C with primary rabbit monoclonal antibody to canine KIM-1/TIM-1/HACVR1 (Sino Biological, Inc., 70001-R202) at 1:50. Normal rabbit IgG (Millipore, Billerica, MA, 12 - 370) was used as a negative control. Donkey anti-rabbit IgG Antibody, HRP conjugate, Species Adsorbed (Millipore, AP182P) was used as the secondary antibody at 1:2000 dilution. The immunoreactivities were visualized using VECTASTAIN Elite ABC Systems reagents (Vector Labs, Inc., Burlingame, CA, PK6100) and Liquid DAB + Substrate Chromagen System reagents (DAKO North America, Inc., Carpinteria, CA, K3468) followed by counterstaining with hematoxylin. Slides were scanned at 20× magnification using the NanoZoomer 2.0 HT Digital Pathology system scanner (Hamamatsu Photonics, Bridgewater, NJ).
Data Analysis
Fold changes in biomarker concentration were expressed versus baseline.
Results
Toxicokinetics Showed Adequate Exposure
Exposure in male and female AmpB-treated dogs was comparable and showed slight accumulation on D4 as compared to D1. Mean C0 and AUC0-24 h values were 1.2- to 1.5-fold greater on D4 (Figure 1 and Supplementary Table 1). Mean plasma concentrations (ng/mL) of AmpB at pre-dose, 0.25, 1, 3, 6, and 24 hr post-dose in male dogs, Days 1 (closed circle) and 4 (open square); and mean plasma concentrations (ng/mL) of AmpB at pre-dose, 0.25, 1, 3, 6, and 24 hr post-dose in female dogs, Days 1 (closed triangle) and 4 (open triangle) following single or multiple intravenous dosing of AmpB (0.3 mg/kg/day) by intravenous infusion for 4 consecutive days.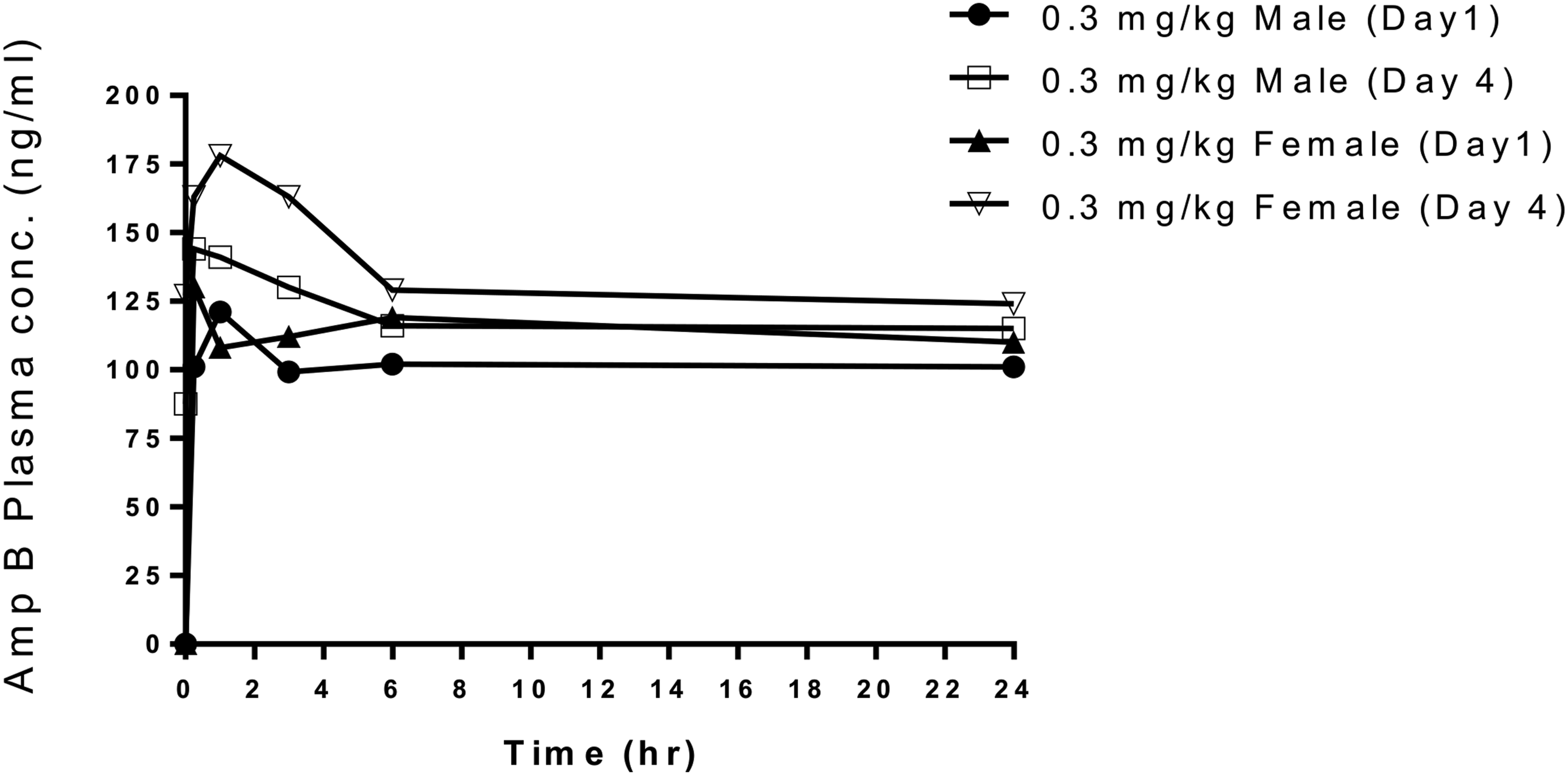
Histopathology Demonstrated AmpB-Induced Renal Tubular Injury
Incidence of AmpB-Related Histopathological Observations in the Kidney of Male and Female Dogs.
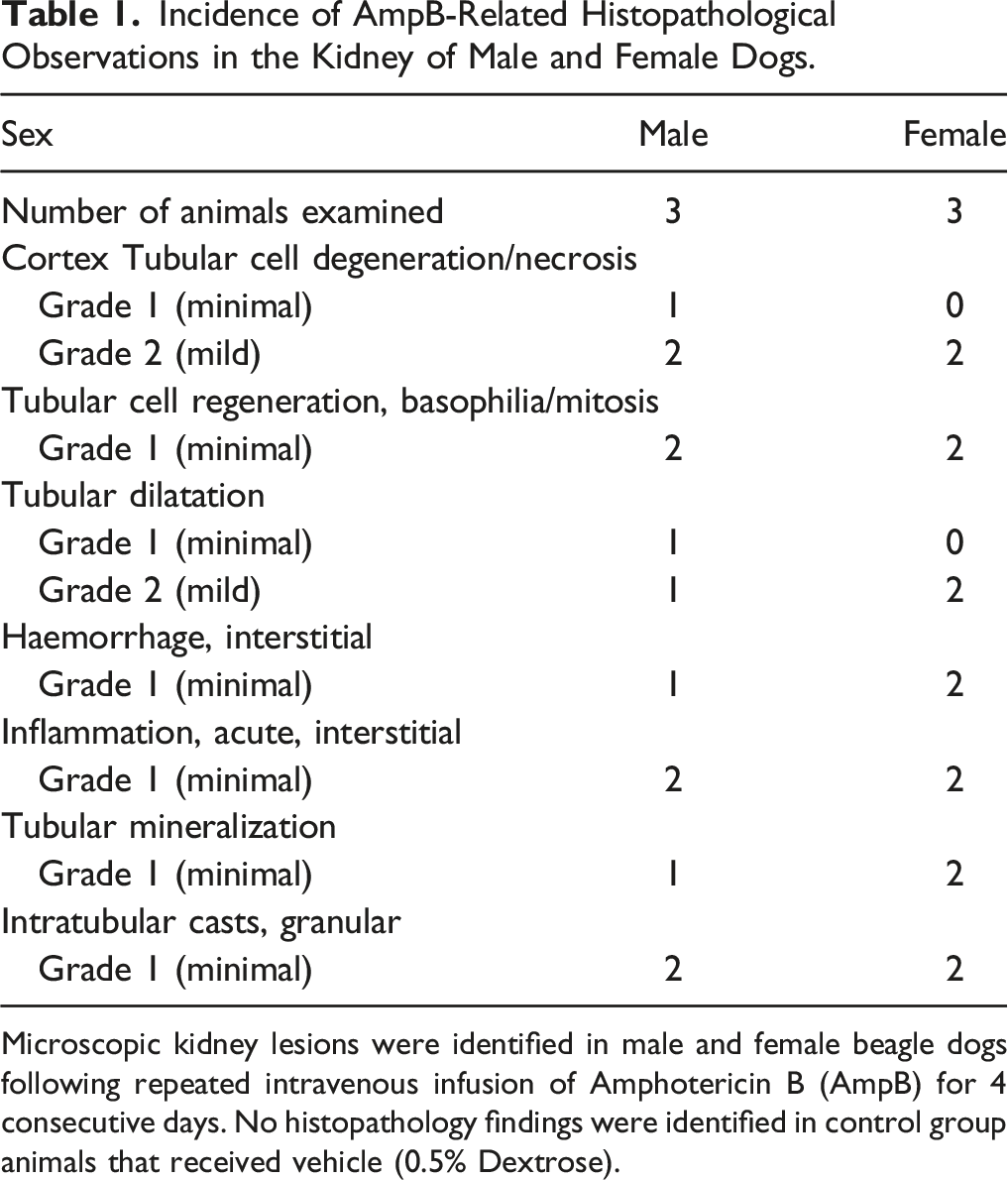
Microscopic kidney lesions were identified in male and female beagle dogs following repeated intravenous infusion of Amphotericin B (AmpB) for 4 consecutive days. No histopathology findings were identified in control group animals that received vehicle (0.5% Dextrose).
Urinary Clusterin Was Highly Sensitive to AmpB-Induced Kidney Injury
Urine and blood samples were collected on D-5 (prior to dosing), D1, D2 and D4 of AmpB administration. For the urinary biomarkers, marked increases in Ur CLU were observed on D1, D2 and D4, with the average increases ranging from 47- to 112-fold increases (Figure 2(A)). Other than one animal with markedly increased urinary ALB on D1 (mean increases of 9.6-fold), urinary ALB levels were generally minimally to mildly increased on D1, D2 and D4 (Figure 2(B)). Similar increases in urinary TP were also observed on D1 (mean increases of 4.7-fold) with lesser increases observed on D2 and D4 (Figure 2(C)). Minimal increases in Ur KIM-1 (1.3 to 1.5-fold versus pre-dose) were observed throughout the dosing period of 4 days (Figure 2(D)). It is to be noted that these changes in Ur KIM-1 were attributed to one male and one female. No changes were observed in urinary NGAL (Figure 2(E)) and NAG levels (data not shown). Urinary biomarkers’ concentrations of individual animals over time and the fold changes of group mean over time relative to the predose values for: (A) Ur CLU, (B) Ur ALB (C) Ur TP, (D) Ur KIM-1 and (E) Ur NGAL in both sexes (n = 6/group) administered AmpB (0.3 mg/kg/day, orange square) or vehicle (0.9% Saline, blue circle) by intravenous infusion for four consecutive days.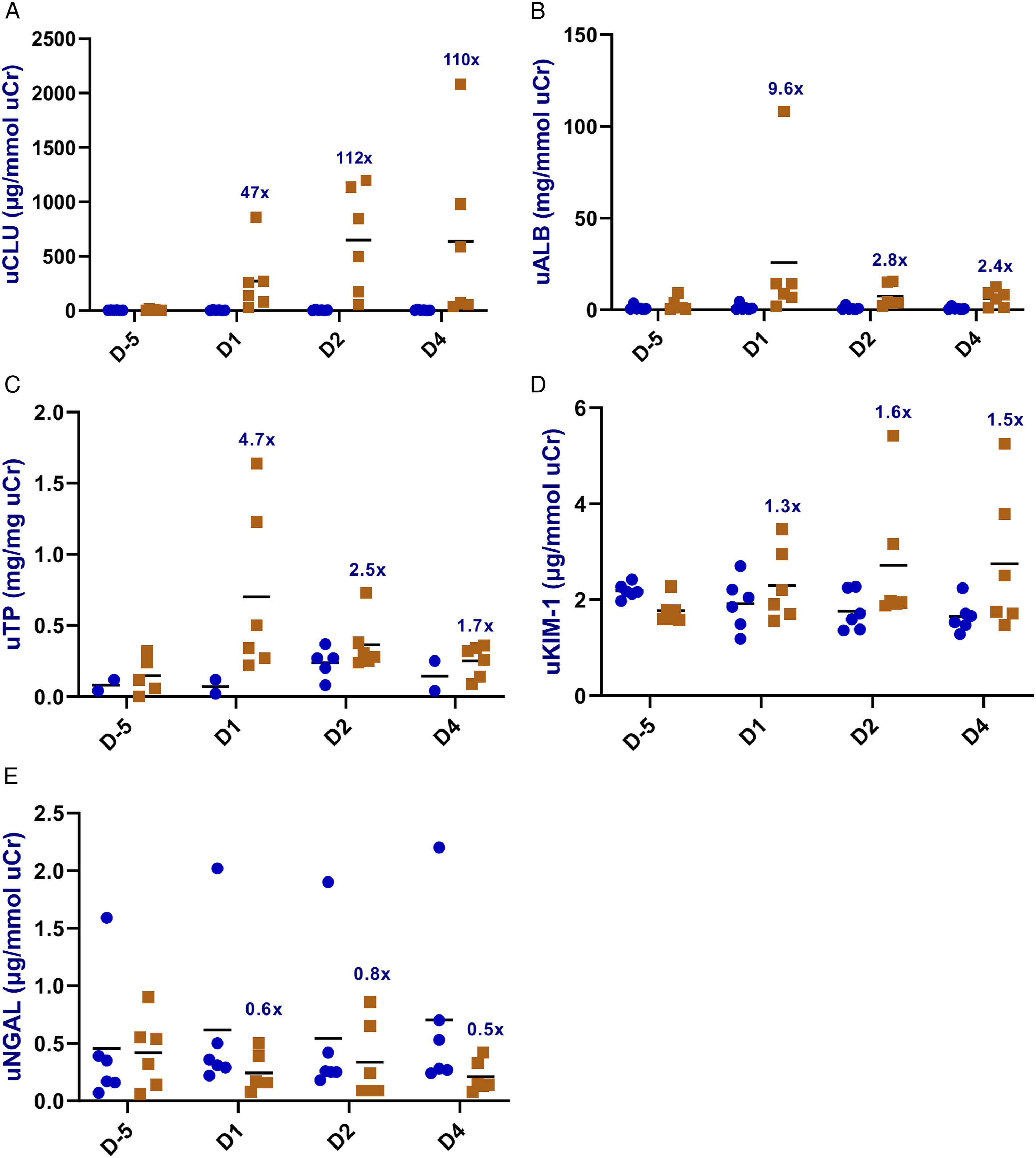
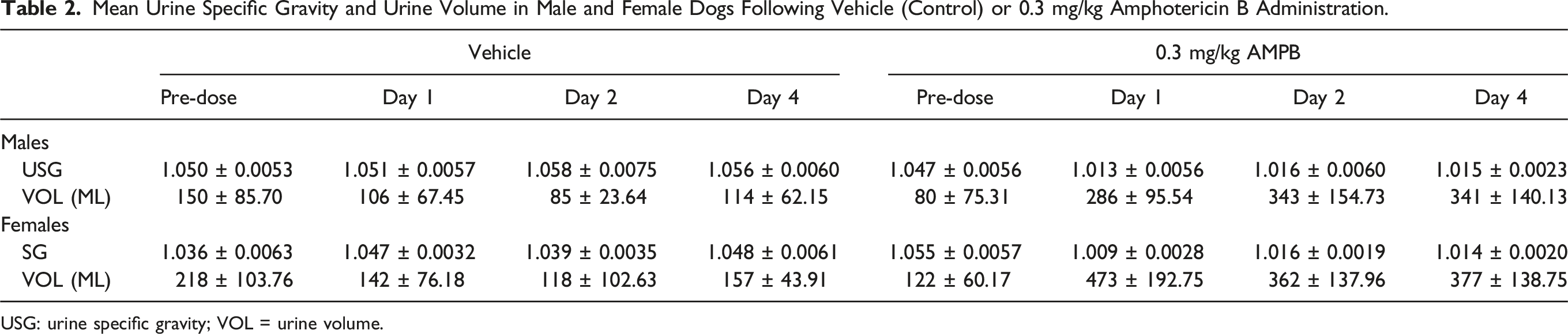
For the serum biomarkers, AmpB-related minimal increases in BUN (1.3- to 1.5-fold versus pre-dose; Figure 3(A)) and sCr (1.2- to 1.3-fold; Figure 3(B)) were observed on D1, D2 and D4. However, these increases were mainly observed in female dogs. Similarly, sCys C levels were minimally increased over baseline (1.2 to 1.3-fold; Figure 3(C)) on D1, D2 and D4. Despite the minimal changes in the serum biomarkers, substantial increases in urine volume and reductions in urine specific gravity were observed in both sexes when compared to the baseline values (Table 2), thereby suggestive of renal functional impairment. Overall, besides the increases in urine volume and reductions in urine specific gravity, Ur CLU was the most sensitive marker for renal tubular injury in this study. Serum biomarkers’ concentrations of individual animals over time and the fold changes of group mean over time relative to the predose values for: (A) BUN, (B) sCr, and (C) sCys-C in both sexes (n = 6/group) administered AmpB (0.3 mg/kg/day, orange square) or vehicle (0.9% Saline, blue circle) by intravenous infusion for four consecutive days. Mean Urine Specific Gravity and Urine Volume in Male and Female Dogs Following Vehicle (Control) or 0.3 mg/kg Amphotericin B Administration. USG: urine specific gravity; VOL = urine volume.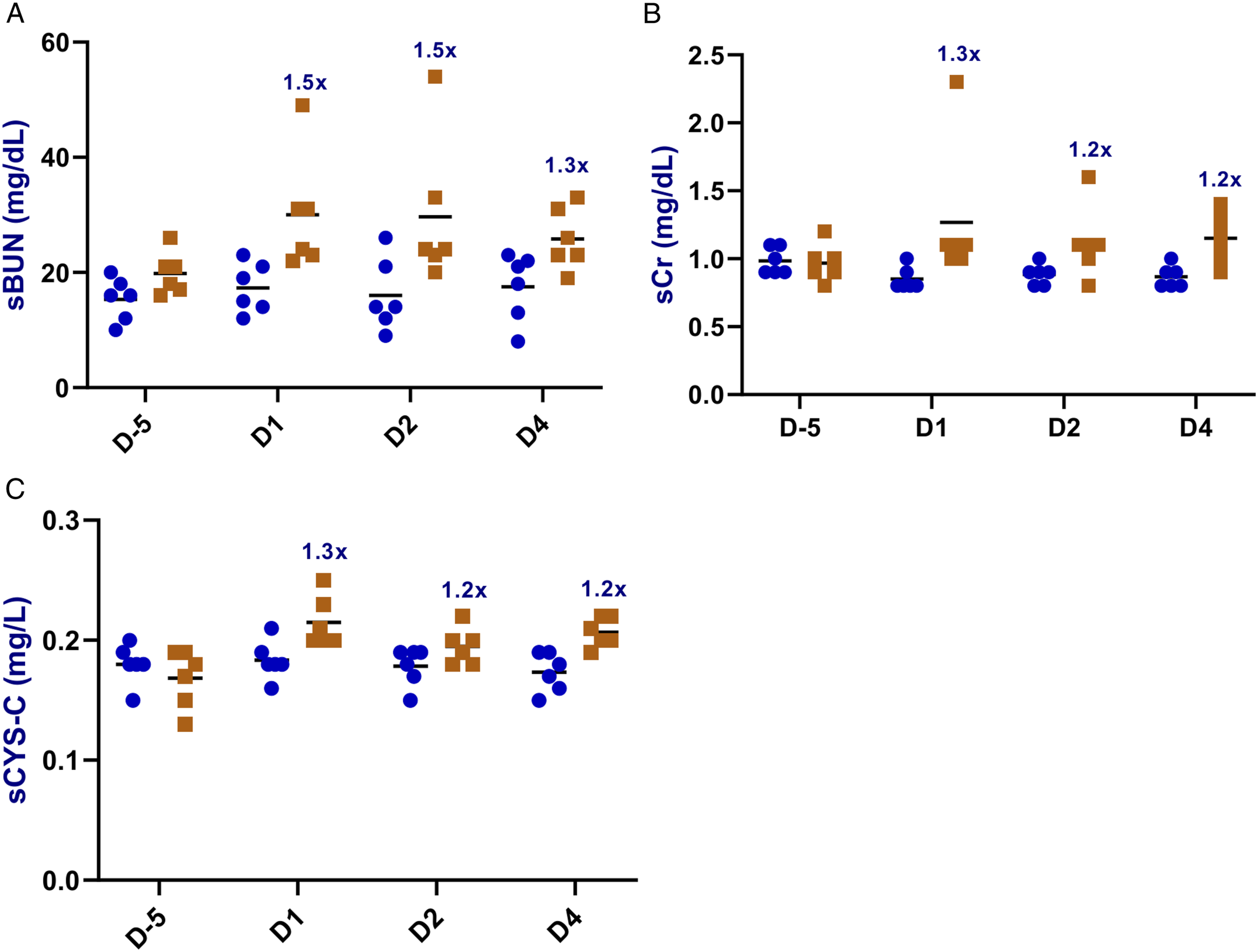
KIM-1 Expression Was Detected in Kidneys of Animals Dosed With AmpB
Generally, increases in Ur KIM-1 signify shedding of the ectodomain of KIM-1 into the urine from tubular epithelium. The localization of positive KIM-1 immunostaining was evaluated to better understand the potential relevance of the minimal fold increases in AmpB-induced increases in Ur KIM-1 detected in this study. Constitutive KIM-1 expression was not detected in kidneys from control (vehicle)-treated male and female dogs (Figure 4 A, C). However, after 4 consecutive days of dosing, the AmpB-treated male and female dogs showed mild to moderate positive KIM-1 immunostaining in the cortical and medullary epithelial cells of the kidneys (Figures 4(B) and (D), respectively). KIM-1 staining was mainly observed in cortical tubular epithelial cells of the kidneys, with more KIM-1 staining in the females (Figure 4(D)) than in the males (Figure 4(B)). Representative immunohistochemistry photomicrographs of KIM-1 expression in kidneys from beagle dogs (n = 6/group) administered the vehicle, 0.9% Saline (A, males; and C, females); or 0.3 mg/kg/day of AmpB (B, males; and D, females) by intravenous infusion for four consecutive days. No KIM-1 positive immunostaining in the renal epithelial cells of the cortex/medulla in the control male (A) or control female (C) dogs. Mild to moderate KIM-1 immunostaining was observed in the cortical and medullary epithelial cells of the kidneys in the AmpB-treated male (B) and female (D) dogs. ×20 magnification.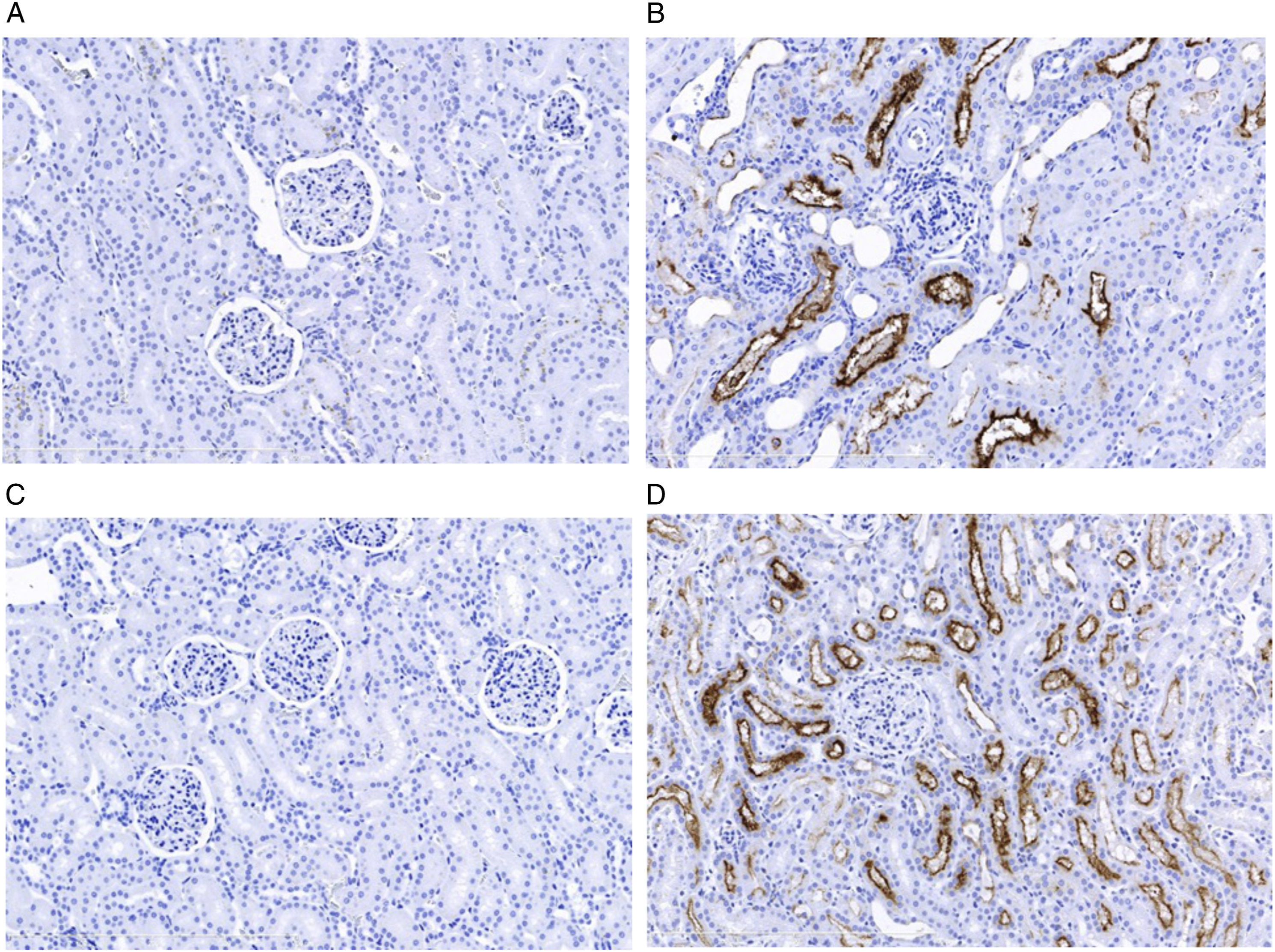
Discussion
Ur CLU has been qualified as a rat urinary renal biomarker for the prediction of compound-induced renal dysfunction, tissue injury response and tissue leakage. 4 The most important observation in this study was the marked increases in Ur CLU in response to the acute tubular injury by AmpB. Ur CLU as a biomarker increased progressively suggesting its temporal profile parallels the progression of the tissue injury. Gentamicin-induced acute kidney injury (AKI) in Beagle dogs also showed marked increases in Ur CLU as early as after three doses (80 mg/kg/day given intramuscularly for 9 days), and correlated well with the severity of the kidney injury. 20 Moreover, in a cisplatin-induced kidney injury study in Beagle dogs (0.9 mg/kg/day given intravenously for 5 days and then monitored for up to 10 days prior to necropsy), increases in Ur CLU were observed at time points where sCr and BUN remained within baseline values. 16 In the same study, immunohistochemistry revealed moderate expression of CLU in cisplatin-induced damaged tubular epithelium in kidneys relative to the control. In the present study, Ur CLU levels were significantly increased throughout exposure to AmpB. Given the substantial increases in Ur CLU in three independent studies with three different nephrotoxicants, we strongly believe Ur CLU is a highly sensitive biomarker for acute/progressive tubular injury in dogs.
Urinary ALB has been qualified as a rat urinary kidney safety biomarker for the early detection of nephrotoxicant-induced renal dysfunction and kidney tissue injury (leakage) response. 4 In the present study, other than one animal on D1, Ur ALB values were marginally increased over the course of the study period. This result is consistent with what was observed in another DIKI dog study conducted with cisplatin, 16 further suggesting Ur ALB is not a highly sensitive biomarker for detecting proximal tubular injury in dogs compare to rodents. Previous studies have shown much more significant increases in Ur ALB in response to cisplatin-and AmpB-induced proximal tubular injury in rodents.31,32 From the authors’ experience, in the absence of any kidney injury, these interspecies differences may be due to the possibility that rodents filter a relatively larger amount of microalbumin through their glomeruli relative to dogs. Therefore, rodents will generally have more albumin available for resorption at the proximal tubule compared to dogs. As such, any interference to the ability of the proximal tubules to reabsorb the filtered microalbumin would easily result in a more significant albuminuria in rodents compared to dogs. This may explain the reason why Ur ALB is a much more sensitive biomarker for drug-induced tubular injury in rodents relative to dogs.
Urinary KIM-1 has also been qualified as a rat urinary renal biomarker for proximal tubular injury1,4,5; however, little is known about its value for monitoring DIKI in dogs. In one study, increased Ur KIM-1 mRNA was observed in dogs following treatment with gentamicin. This correlated with the multifocal KIM-1 immunostaining in damaged cortico-medullary tubular epithelium. 16 Additionally, dogs treated with cisplatin (0.9 mg/kg/day for 5 days and then monitored for up to 10 days prior to necropsy) had slight reductions in mean Ur KIM-1 levels on D3 and D5. 16 However, for two dogs that survived the entire study period, minimal increases in Ur KIM-1 were detected on days 10 and 15, which correlated with the minimal to moderate increases in KIM-1 immunolabelling in the cortico-medullary tubular epithelial cells. 16 Similarly, in this study, minimal increases in Ur KIM-1 correlated with mild to moderate KIM-1 labeling in the cortical and medullary epithelial cells of the kidneys. Even though moderate KIM-1 protein expression was localized in the damaged kidneys, the magnitude (fold changes) of KIM-1 protein released into the urine on treatment with AmpB was not substantial, suggesting a limited utility for Ur KIM-1 as a non-invasive marker to support early identification of DIKI in dogs. The lack of widely-accepted dog-specific KIM-1 immunoassays may also contribute to comparatively low performances for Ur KIM-1 in dog nephrotoxicity biomarker studies as compared to readily applied species-specific immunoassay kits for rodents and humans.
Increases in Ur NGAL have been associated with AKI in rats 9 and dogs17,20 with kidney injury primarily localized to proximal tubules. In this study, AmpB caused tubular lesions with absence of remarkable changes in Ur NGAL suggesting possible differences in mechanism. In dogs treated with cisplatin (0.9 mg/kg/day for 5 days and then monitored for up to 10 days prior to necropsy), microscopic corticomedullary lesions were evident; however, only one moribund dog that was euthanized early on D7 showed insignificant increases in Ur NGAL on D5. 16 Urinary NGAL has shown promise as a diagnostic tool for detection of acute proximal tubular injury in dogs that received gentamicin and polymyxin B.14,17,19,20 Additional studies are warranted to further understand and better characterize changes in Ur NGAL in response to nephrotoxicants encountered during drug development.
Urinary NAG concentrations did not change following AmpB treatment. NAG is a lysosomal brush border enzyme of proximal renal tubular cells and expected to increase in urine after acute proximal tubular injury/dysfunction. 20 The data from this study suggests that histopathological kidney lesions of minimal or mild severity are not associated with changes in Ur NAG in dogs.
The kidney microscopic lesions observed in this study were primarily characterized by acute tubular injury which was in agreement with previously reported findings from dogs exposed to nephrotoxic doses of AmpB. 26 Amphotericin B nephrotoxicity was detected by traditional biomarkers indicative of decreased glomerular filtration rate, including toxicologically relevant increases in BUN and/or sCr; however, BUN and sCr do not signify nephron segment specificity for the apparent tubular damage/dysfunction. This further attests to the limitation of the traditional biomarkers for DIKI. That said, other than Ur CLU and to a much lesser extent Ur ALB and Ur TP, none of the other biomarkers analyzed in this study were more sensitive than BUN and sCr.
While this study was limited in scope especially with regards to the number of animals used, the consistency in the response of some of the biomarkers with previous nephrotoxicity studies in dogs (eg marked elevation of Ur CLU,16,20 and non-remarkable changes in Ur ALB, Ur KIM-1, Ur NGAL and Ur NAG 16 ) makes this observation convincing. Moreover, with the advancement and advocacy for 3Rs (replacement, reduction and refinement) in drug development, if similar conclusions will be attained with lesser number of animals, then adding more animals to a study should not be encouraged.
In summary, intravenous infusions of AmpB at 0.3 mg/kg/day for 4 days, resulted in clinical pathology and histopathology changes in the urine and kidney respectively, in male and female beagle dogs. Significant increases in novel DIKI biomarkers, particularly Ur CLU and to a much lesser extent Ur ALB appeared as potentially promising non-invasive biomarkers to enable monitoring for acute/progressive kidney injury in exploratory studies in dog during drug development programs. The PSTC, under the umbrella of the Critical Path Institute, will continue to engage the participating organizations to develop and potentially validate these kidney injury biomarkers in other non-rodent species (eg, cynomolgus monkeys).
Supplemental Material
Supplemental Material - Evaluation of Novel Urinary Biomarkers in Beagle Dogs With Amphotericin B-Induced Kidney Injury
Supplemental Material for Evaluation of Novel Urinary Biomarkers in Beagle Dogs With Amphotericin B-Induced Kidney Injury by Adeyemi O. Adedeji, Manisha Sonee, Yafei Chen, Karen Lynch, Katrina Peron, Nicholas King, James E. McDuffie, and Petra Vinken in International Journal of Toxicology
Footnotes
Acknowledgments
This material is based upon work supported by Critical Path Institute’s (C-Path) Predictive Safety Testing Consortium (PSTC), Nephrotoxicity Working Group (NWG). The authors would like to acknowledge and thank the members of PSTC for their scientific, financial, and in-kind contributions that supported these research activities, as well as the input from US FDA and EMA scientists who served as advisors. We would like to thank Charles Johnson, Stewart Bryant, and Jing-Ying Ma for their scientific contributions and technical assistance. We would also like to thank Lila Ramaiah, Shashi Ramaiah and Ragu Ramanathan for their thorough review of the manuscript.
Author Contributions
Adedeji, A. O., Sonee, M., McDuffie, J. E. and Vinken, P. contributed to conception, design, data analysis and interpretation, drafted and critically revised the manuscript. Chen, Y. contributed to data acquisition, analysis and critically revised the manuscript. Lynch, K. contributed to conception, design, data acquisition and interpretation, and critically revised the manuscript. Peron, K. and King, N. contributed to conception, data acquisition and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All work was supported by Janssen Research & Development, LLC, GlaxoSmithKline, and Genentech, a member of the Roche group. In addition, Critical Path Institute is supported by the Food and Drug Administration (FDA) of the Department of Health and Human Services (HHS) and is 55% funded by the FDA/HHS, totaling $17,612,250, and 45% funded by non-government source(s), totaling $14,203,111. The contents are those of the author(s) and do not necessarily represent the official views of, nor an endorsement by, FDA/HHS or the U.S. Government.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
