Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) re-reviewed the safety of Malic Acid and Sodium Malate in cosmetics. Malic Acid is reported to function in cosmetics as a fragrance ingredient and a pH adjuster and Sodium Malate functions as a skin-conditioning agent - humectant. The Panel reviewed the available data to determine the safety of these ingredients. The Panel concluded that Malic Acid and Sodium Malate are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Introduction
The Panel published a Final Report on the Safety Assessment of Malic Acid and Sodium Malate in 2001 and concluded that Malic Acid and Sodium Malate are safe for use as pH adjusters in cosmetic formulations; however, the Panel determined that the data were insufficient to determine the safety of these ingredients for any other functions. The data needs, based on the reported function of Sodium Malate (skin conditioning agent – humectant), were concentration of use data, dermal irritation and sensitization data, and ocular irritation data. In accordance with its procedures, the Panel evaluates the conclusions of previously-issued reports every 15 yr, and it has been at least 15 yr since this assessment was issued. Because the number of uses and concentrations of use increased for Malic Acid since the original assessment, the Panel reopened the Safety Assessment of Malic Acid and Sodium Malate in 2017 to amend the original conclusion. The conclusion of this report supersedes the 1 found in the 2001 report.
According to the web-based International Cosmetic Ingredient Dictionary and Handbook (Dictionary), Malic Acid is reported to function in cosmetics as a fragrance ingredient and a pH adjuster, while Sodium Malate is reported to function in cosmetics as a skin-conditioning agent – humectant.1,2 These functions are similar to what was reported in the 2001 assessment, except at that time Malic Acid was only reported to function as a pH adjuster.
Malic Acid (or malate) is an intermediate in the citric acid cycle (also known as the tricarboxylic acid (TCA) cycle or Krebs cycle) and is formed during the hydration reaction of fumarate (or fumaric acid) with the enzyme fumarase. 3 Fumarate is formed by the oxidation reaction of succinate (succinic acid) and coenzyme Q (ubiquinone) with succinic dehydrogenase. The Panel published the safety assessments of Fumaric Acid (with related salts and esters) in 2009 and Succinic Acid and Sodium Succinate (as part of the report on dicarboxylic acids) in 2012, and concluded that these ingredients, which have the same functions as Malic Acid, are safe as used in cosmetics.4,5
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) Web site (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties. The original report that was published in 2001 is available on the CIR Web site (http://www.cir-safety.org/ingredients).
Chemistry
Definition and Structure
The Dictionary defines Malic Acid as an organic carboxylic acid, the molecular formula of which is C4H6O5 and the stereoisomers of which are depicted below (Figure 1).
1
Malic Acid (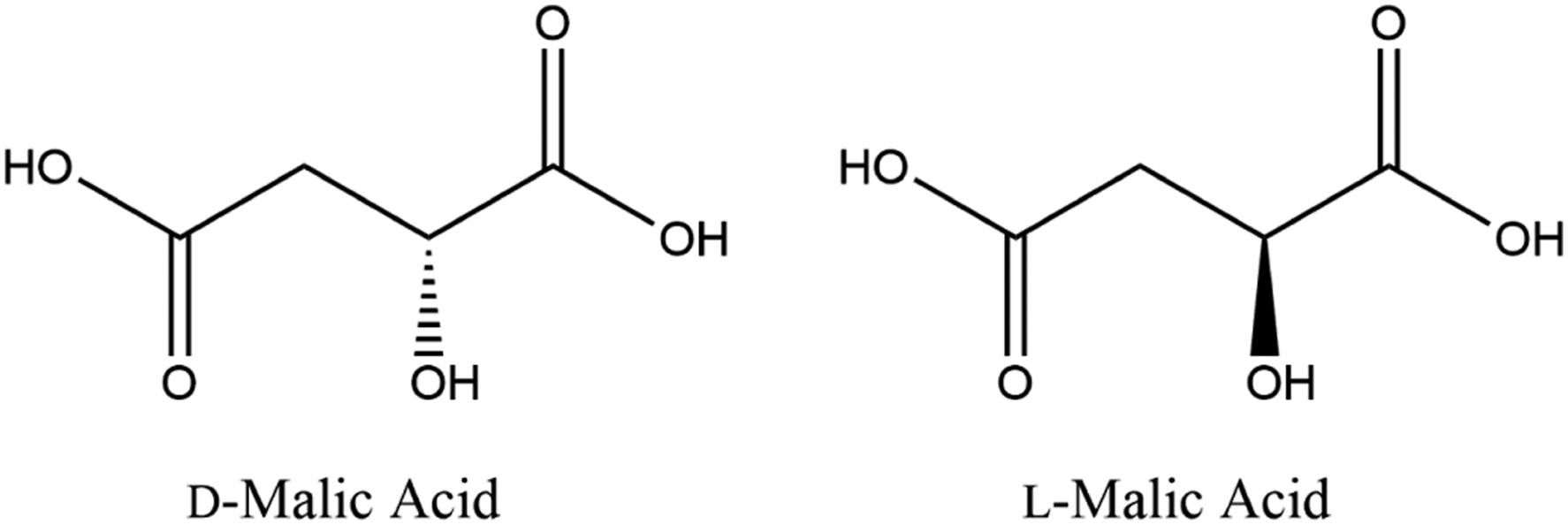
Sodium Malate is the sodium salt of Malic Acid. It conforms to the formula shown below (Figure 2).
2
With 2 carboxylic acid functional groups, Sodium Malate is available as the mono- or di-sodium salt. Sodium Malate (monosodium and disodium, CAS No. 676-46-0 (disodium salt)).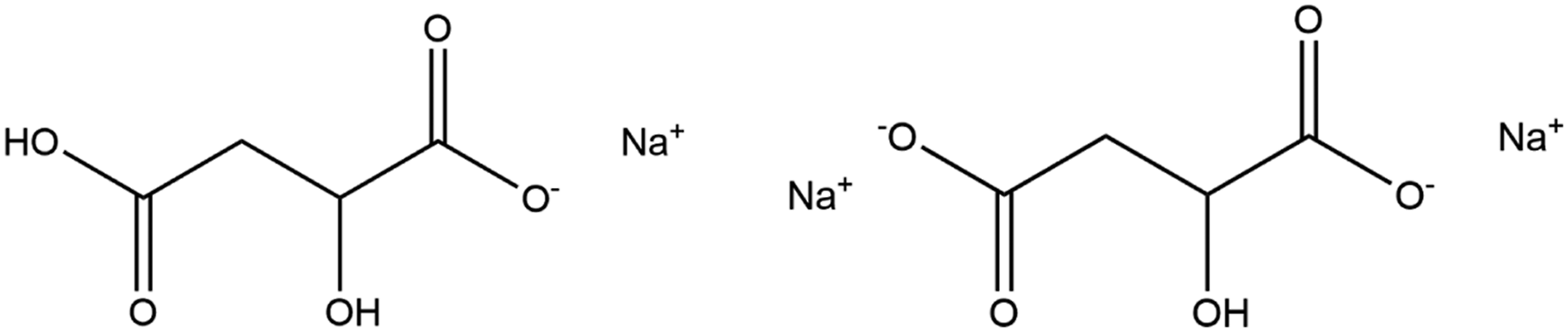
Malic Acid and Sodium Malate are α-monohydroxy succinic acid ingredients. These ingredients have 1 stereocenter, and thereby 2 stereoisomers, the configuration of which is most commonly denoted by
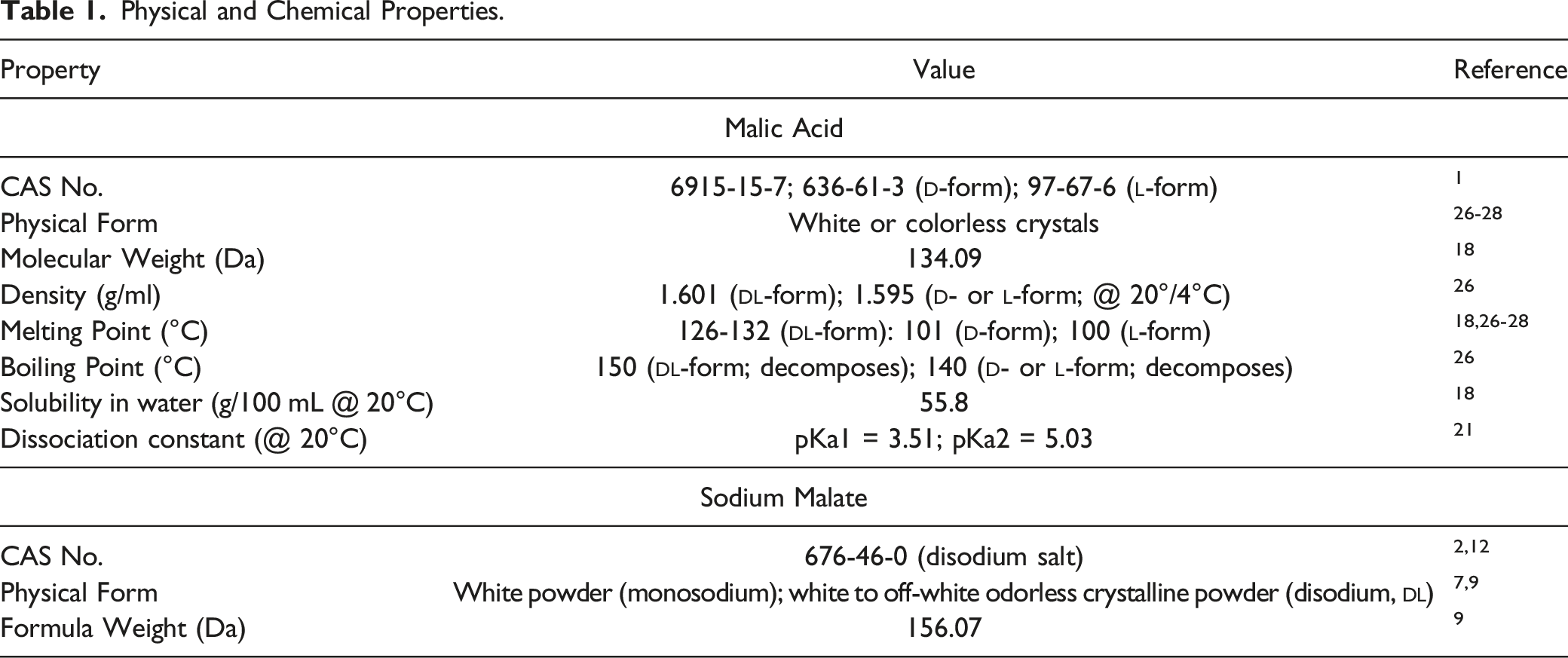
Physical and Chemical Properties
Physical and Chemical Properties.
Methods of Manufacture
Sodium Malate (reported as
Natural Occurrence
The
Impurities
Maleic and fumaric acids are by-products of the manufacture of Malic Acid. 6 Malic Acid is generally purified until the amounts of fumaric and maleic acid are 7.5 and <500 ppm, respectively.
As stated in the United States Pharmacopeia (USP), Malic Acid for food use should be 99 - 100.5% pure with no more than 1% fumaric acid and no more than 0.05% maleic acid.
8
According to the Food Chemicals Codex, Sodium Malate (disodium,
Use
Cosmetic
The safety of the cosmetic ingredients addressed in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of this ingredient in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetics industry in response to a survey, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
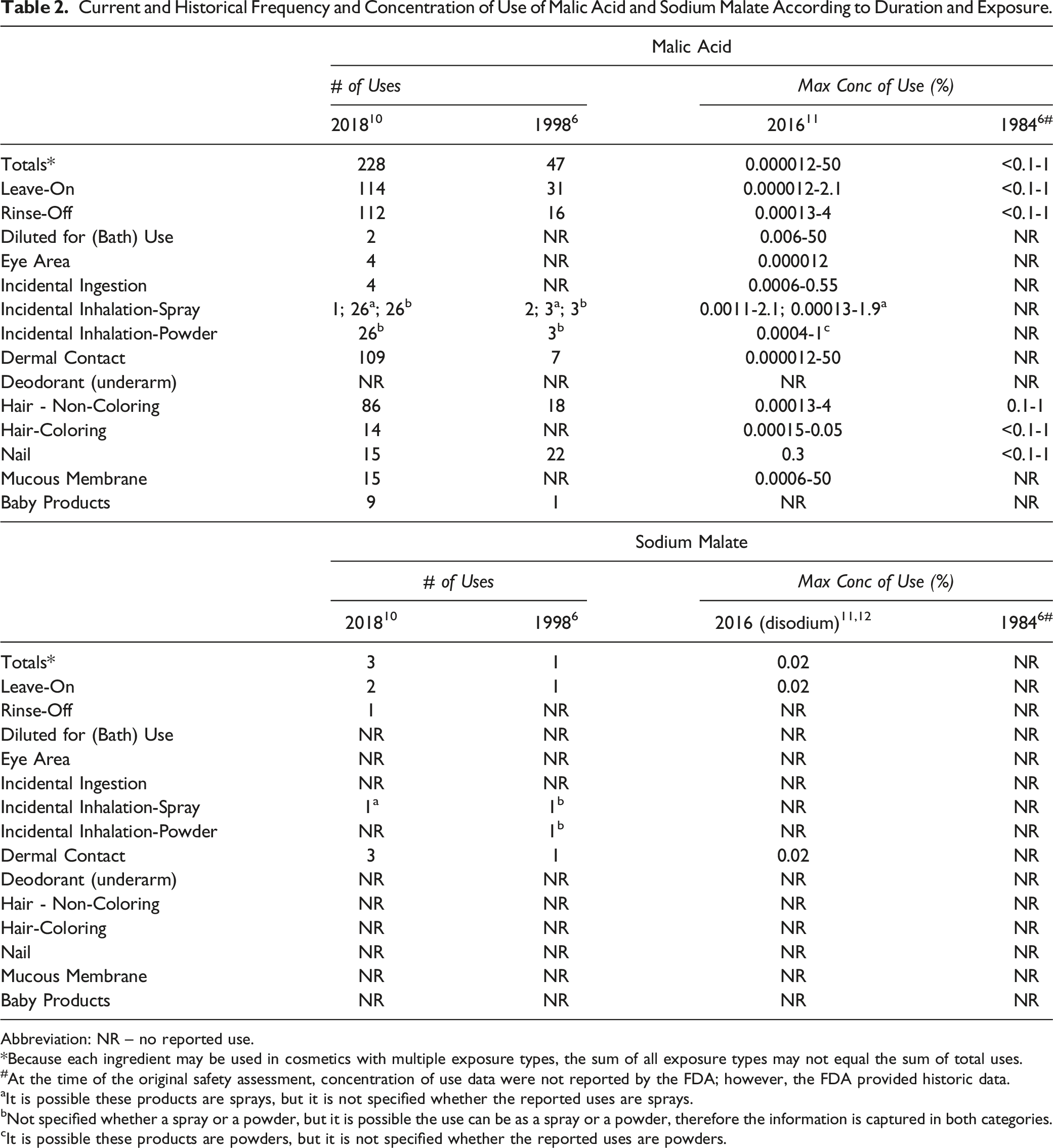
Current and Historical Frequency and Concentration of Use of Malic Acid and Sodium Malate According to Duration and Exposure.
Abbreviation: NR – no reported use.
*Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
#At the time of the original safety assessment, concentration of use data were not reported by the FDA; however, the FDA provided historic data.
aIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
bNot specified whether a spray or a powder, but it is possible the use can be as a spray or a powder, therefore the information is captured in both categories.
cIt is possible these products are powders, but it is not specified whether the reported uses are powders.
The frequency of use for Sodium Malate has also increased since the original review, from 1 reported use in 1998 6 to 3 reported uses in 2018. 10 Current uses of Sodium Malate are reported in coloring hair care products and skin care preparations. No concentration of use for Sodium Malate was reported in the 2001 safety assessment. 6 The Council in 2016 reported that Sodium Malate (disodium) is used at 0.02% in “other” skin care preparations.11,12
Malic Acid is used in products that are used near the eye at a maximum concentration of 0.000012% (in eyeliners) and in those that can come in contact with mucous membranes at maximum concentrations up to 50% (in bath oils, tablets and salts); no concentrations of use were reported for these categories in the original assessment. 11 Additionally, Malic Acid is used in body and hand products and pump hair spray formulations at concentrations up to 2.1%; these product-types could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared with pump sprays.13,14 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (ie, they would not enter the lungs) to any appreciable amount.15,16
Malic Acid and Sodium Malate are not restricted from use in any way under the rules governing cosmetic products in the European Union (EU). 17
Non-Cosmetic
Malic Acid is an intermediate in chemical synthesis.
18
It is a chelating and buffering agent. In foods, it is a flavoring agent, a flavor enhancer, and an acidulant (a substance that gives food a tart, sour, or acidic flavor). The Food Chemicals Codex reports that Malic Acid (
Toxicokinetics Studies
Absorption, Distribution, Metabolism, and Excretion
Most of the radioactivity from 2.5 mg/kg U-[14C]-
Skin Penetration
The ability for Malic Acid to penetrate the skin, as used in rinse-off personal care products, was assessed in an in vitro study.
20
A shampoo with radiolabeled Malic Acid (U-[14C]-
Toxicological Studies
Acute Toxicity Studies
The oral LD50 values of Malic Acid for mice, rats, and rabbits ranged from 2.66 to greater than 3.2, 1.60 - 3.5, and 3 - 5 g/kg, respectively. 6 The acute LD50 of Malic Acid given intravenously was 2.4 g/kg for rabbits, and the i.p. LD50 for mice and rats were 0.05 to 0.1 and 0.1 – 0.2 g/kg, respectively.
Chronic Toxicity Studies
In a chronic oral study in rats, Malic Acid at concentrations up to 50,000 ppm (5.0%) in feed for 104 wk resulted in decreases in body weight gains and feed consumption, but compound-related lesions were not observed. 6 No significant changes or lesions were observed when dogs were fed Malic Acid at concentrations up to 50,000 ppm for 104 wk.
Developmental and Reproductive Toxicity (Dart) Studies
Oral dosing of Malic Acid did not cause developmental toxicity in mice (at up to 266 mg/kg), rats (at up to 350 mg/kg), or rabbits (at up to 300 mg/kg). 6 In a multigenerational oral DART study, no significant adverse effects were observed in rats that received up to 10,000 ppm Malic Acid.
Genotoxicity Studies
In Vitro
Malic Acid was not mutagenic in Ames tests or a mammalian cell chromosomal assay. 6 In 1 bacterial cell study, pyrolyzates of Malic Acid were not mutagenic, but in another bacterial cell study they were. Products formed from treatment of Malic Acid with aqueous solutions of chlorine were considered mutagenic.
Carcinogenicity Studies
No published carcinogenicity studies on Malic Acid or Sodium Malate were discovered, and no unpublished data were submitted since the 2001 safety assessment.
Dermal Irritation and Sensitization Studies
Animal Studies
Malic Acid was moderately irritating to rabbit skin (500 mg for 24 h) and was a strong irritant to guinea pigs (concentration not reported). 6
Human Studies
In a test determining subjective skin irritation potential, the average irritation scores over a 15-minute period were 39.4, 37.1, and 23.1 for 1 M Malic Acid at pH 3, 5, and 7, respectively (ie, mildly irritating). 6
Human Repeat Insult Patch Tests.

Abbreviation: HRIPTs, human repeat insult patch tests.
Ocular Irritation Studies
In Vitro Studies
The ocular irritation potential of Malic Acid was tested in formulation in chorioallantoic membrane vascular assays (CAMVA) and bovine corneal opacity and permeability tests (BCOP). 22 Malic Acid at 2.2725% was tested in a hair styler and a hair shampoo at pH 3.6 and pH 3.0, respectively. The assays predicted that the formulation with Malic Acid at pH 3.6 would be a severe ocular irritant and the formulation with Malic Acid at pH 3.0 would be an ocular irritant.
Animal Studies
Malic Acid (750 µg) caused severe ocular irritation in rabbit eyes. 6
Clinical Studies
In predictive testing in patients with atopic dermatitis, 18 of 34 patients reacted to a diet high in Malic Acid and citric acid, and 6 reacted to a diet high in Malic Acid. 6 Reactions included immediate (seasonal allergic rhinitis and urticarial) and delayed (contact dermatitis) responses. In assessing the effect of Malic Acid on cell renewal, an 18%, 10%, and 5% increase was observed at pH 3, 5, and 7, respectively. Malic Acid (200 mg) was not toxic in a clinical efficacy and safety test.
The cumulative irritation potential of Malic Acid with other fruit acids was tested in 20 healthy volunteers. 25 The volunteers were exposed twice daily for 4 days to 2% Malic Acid (pH 2 and pH 4), either alone or in tandem with 0.5% sodium lauryl sulfate (SLS). Positive and negative controls were 0.5% SLS and distilled water, respectively. Approximately 50 µl of the test materials were applied to each test area on the paravertebral mid back by occlusive patches (Finn Chambers on Scanpor, 12 mm diameter). The patches were removed after 30 min, rinsed with ∼10 mL of tap water, and dried with tissue paper without rubbing. Irritant cutaneous reactions were quantified by visual scoring, transepidermal water loss, and skin color reflectance. The twice daily application of Malic Acid (pH 2 or pH 4) alone did not induce significant irritant reactions and were comparable to the negative control. Combined exposures to Malic Acid and SLS caused marked barrier disruption, but the effect was less than that observed from combined exposure to SLS and water, which indicated a protective effect by Malic Acid. The authors of the study concluded that Malic Acid did not significantly contribute to the occurrence of irritant contact dermatitis or increase susceptibility to SLS-induced irritation.
Summary
The Panel published a Final Report on the Safety Assessment of Malic Acid and Sodium Malate in 2001 and concluded that Malic Acid and Sodium Malate are safe for use as pH adjusters in cosmetic formulations; however, the Panel determined that the data were insufficient to determine the safety of these ingredients for any other functions. In accordance with its procedures, the Panel evaluates the conclusions of previously-issued reports every 15 yr, and it has been at least 15 yr since this assessment has been issued. Because the number of uses and concentrations of use increased since the original assessment, the Panel reopened the Safety Assessment of Malic Acid and Sodium Malate in 2017 to amend the original conclusion.
Malic Acid is reported to function in cosmetics as a fragrance ingredient and a pH adjuster, while Sodium Malate is reported to function in cosmetics as a skin-conditioning agent – humectant. These functions are similar to what was reported in the 2001 assessment except at that time Malic Acid was only reported to function as a pH adjuster.
Malic Acid (or malate), an intermediate in chemical synthesis including the citric acid cycle (also known as TCA cycle or Krebs cycle), is formed during the hydration reaction of fumarate (or fumaric acid) with the enzyme fumarase. Malic Acid is a chelating and buffering agent. In foods, it is a flavoring agent, a flavor enhancer, and an acidulant.
The frequency of use of Malic Acid has increased since safety was originally reviewed, from 47 reported uses in 1998 to 228 reported uses in 2018. Notably, the number of uses near the eye area and mucous membranes increased from no reported uses to 4 and 15, respectively. The reported maximum concentration of use has increased; the maximum leave-on concentration of use reported was 1% (in multiple formulation types) in 1984, and the results of the survey conducted by the Council in 2016 report a maximum leave-on use concentration of 2.1% (in a hair spray). It is used at up to 50% in products diluted for baths.
The frequency of use for Sodium Malate has also increased since the original review, from 1 reported use in 1998 to 3 reported uses in 2018. Uses of Sodium Malate include coloring hair care products and skin care preparations. No concentration of use for Sodium Malate was reported in the 2001 safety assessment. The Council, in 2016, reported that Sodium Malate is used at 0.02% in “other” skin care preparations.
In an in vitro study, epidermal penetration of <1% radiolabeled Malic Acid (pH 5.0 - 7.0) in a shampoo was considered negligible, with >99% removed by rinsing. The actual skin dose for Malic Acid was 2.69 µg/cm2, the total absorbable dose was 0.003% and the total dose delivered was 0.000067 µg/cm2.
Malic Acid at up to 1% in formulation was not a significant skin irritant and did not induce allergic contact dermatitis in HRIPTs. Malic Acid (2%, pH 2 and pH 4) did not significantly contribute to the occurrence of irritant contact dermatitis or increase susceptibility to SLS-induced irritation in a cumulative irritation study. Malic Acid in formulations at 2.2725% was predicted to be an ocular irritant was tested in vitro.
Discussion
In accordance with its procedures, the Panel evaluates the conclusions of previously-issued reports every 15 yr, and it has been at least 15 yr since this assessment has been issued. Because the number of uses and concentrations of use increased since the original assessment, the Panel reopened the Safety Assessment of Malic Acid and Sodium Malate in 2017 to amend the original conclusion. The conclusion of this report supersedes the 1 found in the 2001 report.
Overall, the Panel considered that the available data, including the role of Malic Acid in normal metabolism and animal toxicity data, were adequate to assess the safety of these ingredients as used in cosmetics. The Panel noted that in formulation, a pH dependent equilibrium exists between Malic Acid and its salts, thus the safety profile between Sodium Malate would not differ from Malic Acid. The Panel also noted that there are no sensitization data for Malic Acid at the maximum leave-on use concentration of 2.1%. Based on the negative results of a HRIPT of Malic Acid at 1%, the experience of the clinicians on the Panel, and considering the fact that Malic Acid and Sodium Malate are common chemicals in human biology, the Panel concluded that these ingredients would not induce sensitization at use concentrations.
The Panel noted that the only significant toxic effect of Malic Acid was irritation to the skin and eyes, which would be predicted based on its pH. Since Malic Acid is used as a pH adjuster in cosmetics, the irritating property of the acid would be minimized in formulated products. The Panel also noted that use of Malic Acid in a hair spray has been reported. The Panel thus advises consumers to minimize incidental ocular exposure of hair sprays containing Malic Acid.
The Panel discussed the issue of incidental inhalation exposure in body and hand products and in pump hair sprays. There were no inhalation toxicity data available. The Panel considered other pertinent data indicating that incidental inhalation exposures to Malic Acid and Sodium Malate in such cosmetic products would not cause adverse health effects, including data characterizing the potential for these ingredients to cause acute and chronic toxicity, developmental and reproductive toxicity, genotoxicity, and ocular or dermal irritation or sensitization. These ingredients are reportedly used at concentrations up to 2.1% in cosmetic products that may be aerosolized. The Panel noted that 95% – 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. The potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that Malic Acid and Sodium Malate are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Footnotes
Author Contributions
Burnett, C. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., and Snyder, P. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. Heldreth, B. contributed to design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
