Abstract
The fluorescent tracer, MB-102, has been designed for the direct, real-time measurement of glomerular filtration rate. Previous studies, both in vitro and in vivo (rats, rabbits and dogs), were conducted to assess potential toxicity including single dose toxicity, mutation assay, chromosomal aberration assay, phototoxicity, local tolerance study, micronuclease assay, hERG channel changes, CNS and cardiovascular safety. The results of these studies led to a safety/toxicology profile for this agent deemed sufficient by the FDA to conduct Phase I and Phase II human clinical studies. In this paper we report on maternal toxicity and the potential effects on embryo-fetal development and the toxicokinetics of MB-102 administered daily via intravenous (bolus) injection into pregnant rabbits during the period of organogenesis gestation day 7-19. Assessment of toxicity was based on mortality, clinical observations, body weight, food consumption, reproductive performance and necropsy and cesarean section findings. Blood samples were collected for toxicokinetic evaluation. No test article findings were noted in any of these studies. The only clinical findings observed were the discoloration of skin, eyes or pelage in the 2 higher dose groups, which were considered related to the color and fluorescent properties of MB-102 and were deemed non-adverse. Exposure, as assessed by Cmax and AUC(0-6) increased in a dose dependent manner from 4.5 to 113 mg/kg/day. No accumulation of the test article was noted after multiple doses were administered. Thus, intravenous administration of MB-102 was not associated with any adverse developmental or reproductive toxicities in pregnant rabbits.
Keywords
Introduction
Determination of glomerular filtration rate (GFR) is considered the most reliable measure of renal function. 1 Nevertheless, current common clinical practice does not directly measure GFR. A measurement of serum creatinine is performed and an estimated GFR (eGFR) is obtained from empirically derived equations.2-4 However up to 50% of kidney function can be lost before an abnormal level of serum creatinine is measured, and a change in serum creatinine level due to kidney insult is time-delayed and may not be apparent for 24 to 72 hours post-insult. 5 Furthermore factors not related to renal function including age, hydration, muscle mass, and diet often significantly affect serum creatinine values.
The use of exogenous fluorescent tracer agents has been explored6-8 for real-time point-of-care GFR measurement. MB-102 is a novel fluorescent tracer agent that has previously exhibited characteristics essential for accurate real-time measurement of GFR in several animal models9,10 and has been clinically demonstrated to be a GFR agent in subjects with normal renal function. 11 In first-in-human clinical studies, this agent has been shown to be well-tolerated in subjects over the range of renal function from normal to Stage 4 chronic kidney disease.11,12
Previously, MB-102 was evaluated in a first-tier and next-tier battery of in vitro and in vivo safety/toxicity studies. The sum-total of these studies13,14 resulted in negligible demonstrable pathological test article concerns, leading to a safety/toxicity profile that was subsequently deemed sufficient by the FDA to begin a first-in-human clinical study, and subsequently for Phase I and Phase II human clinical trials.
MB-102 was then evaluated for reproductive and developmental toxicity in pregnant rats. 15 No significant findings were noted in any endpoints. This report assesses maternal toxicity and potential effects on embryo-fetal development in pregnant rabbits following the intravenous administration of MB-102.
Materials and Methods
Fluorescent Tracer Agent
MB-102 is a compound in the pyrazine class, with a molecular weight of 372.3 (chemical name 3,6-diamino-2,5-bis{N-[(1R)-1-carboxy-2-hydroxyethyl]carbamoyl}pyrazine, Figure 1). It has light absorption and emission peaks at 445 nm and 560 nm respectively.
10
Structure of 3,6-diamino-2,5-bis{N-[(1R)-1-carboxy-2 hydroxyethyl]carbamoyl}pyrazine, MB-102.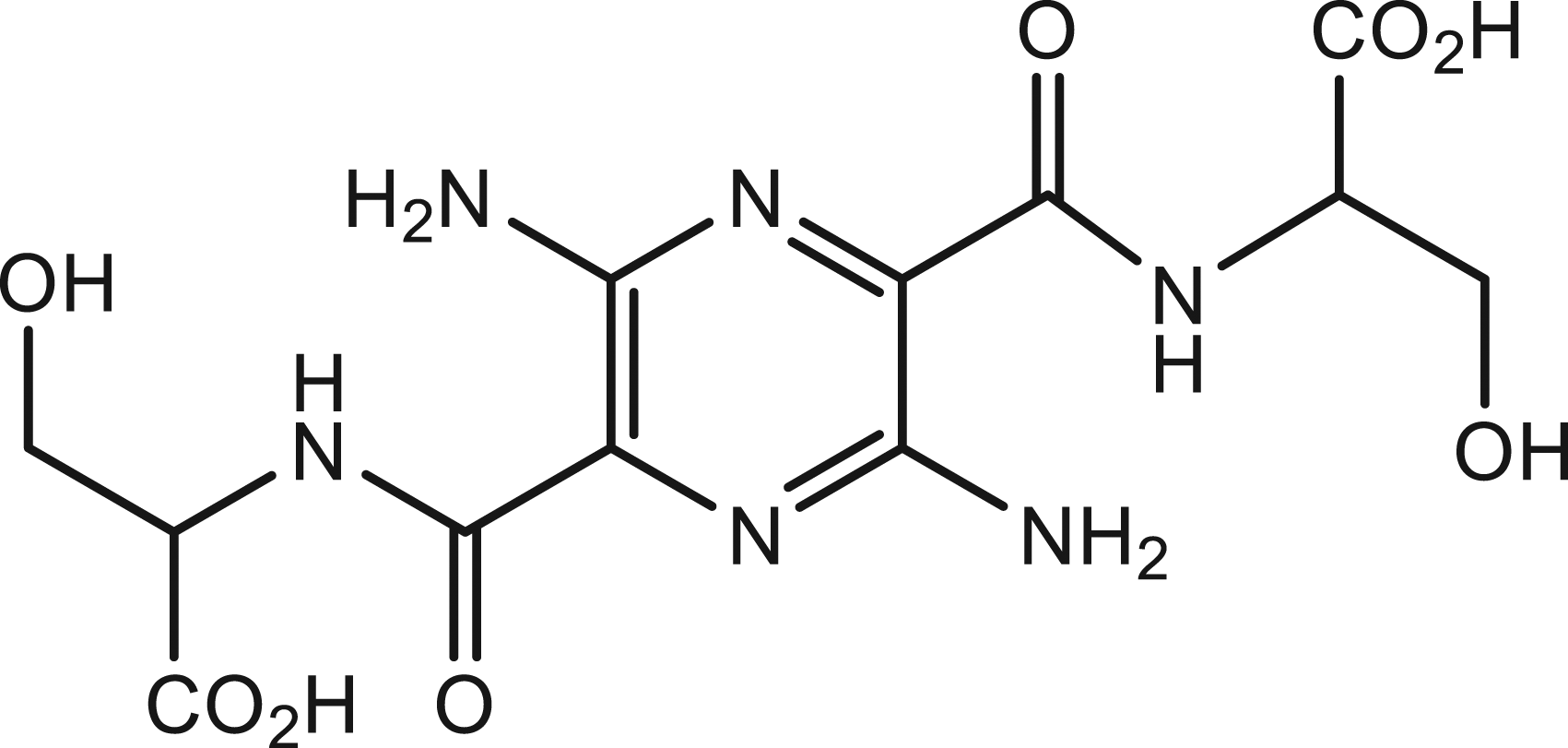
MB-102 is formulated with phosphate-buffered saline for intravenous administration. Properties of MB-102 and its clinical formulation have been recently published. 16
Study Design: Dosing Schedule and Toxicokinetic Evaluation
Dosing Schedule.
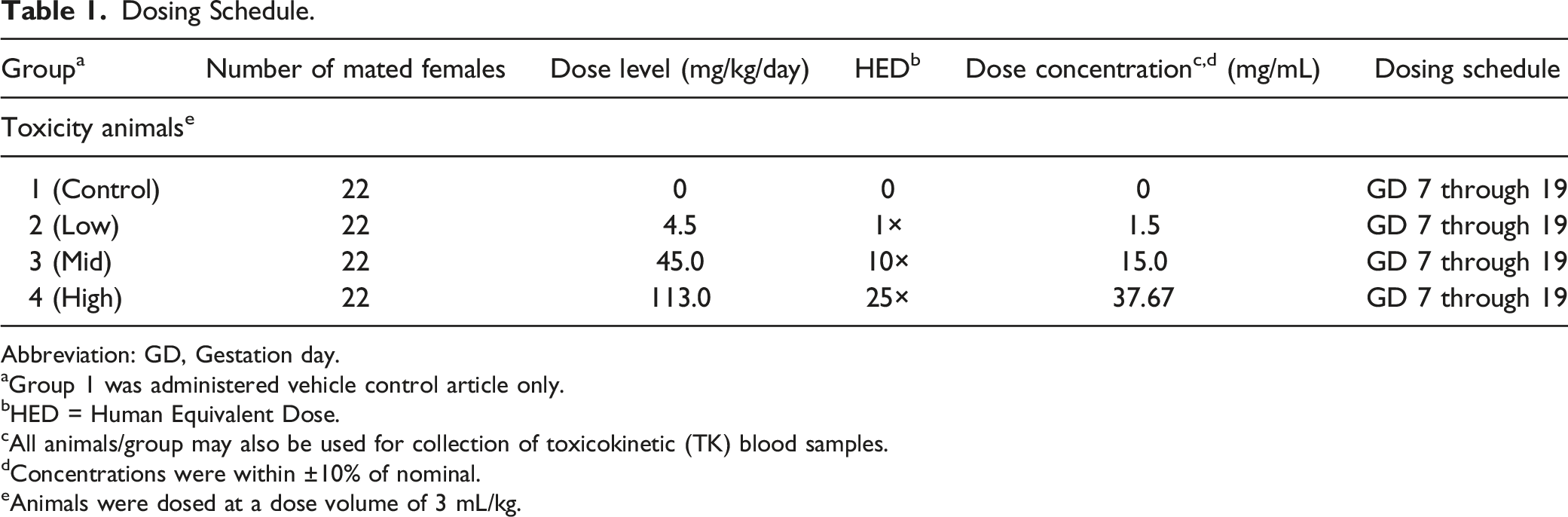
Abbreviation: GD, Gestation day.
aGroup 1 was administered vehicle control article only.
bHED = Human Equivalent Dose.
cAll animals/group may also be used for collection of toxicokinetic (TK) blood samples.
dConcentrations were within ±10% of nominal.
eAnimals were dosed at a dose volume of 3 mL/kg.
Study Design for Toxicokinetics.
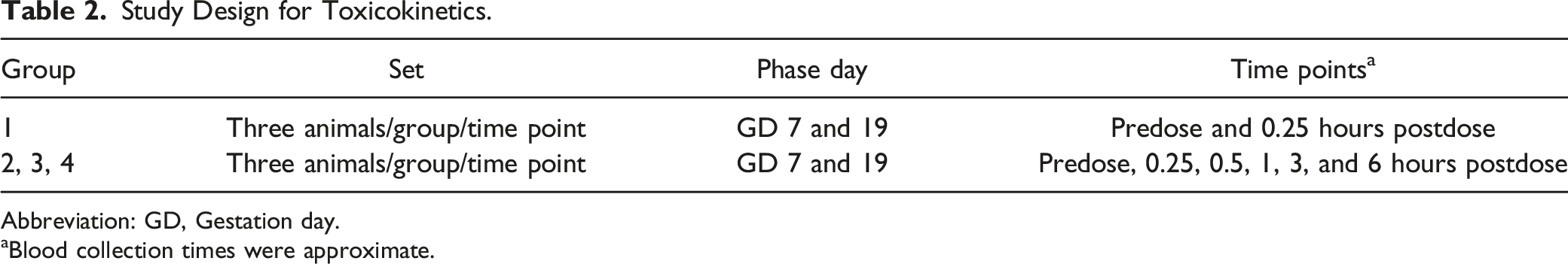
Abbreviation: GD, Gestation day.
aBlood collection times were approximate.
Toxicokinetic Parameters.

Animal Selection and Dose Administration Rationale
Rabbits were chosen for this study due to their sensitivity to a number of agents known to cause reproductive or developmental toxicity. Rabbits historically have been used as the second species in safety evaluation studies of this type (evaluating potentially teratogenic agents) and are recommended by appropriate regulatory agencies. The New Zealand White rabbit was selected based on availability of historical control data and susceptibility to known developmental toxicants.
The doses were chosen on 1-, 10-, 25-fold the planned human equivalent dose (HED) of 1.5 mg/kg, in accordance with the FDA Guidance for Industry “Developing Medical Imaging Drug and Biological Products Part 1: Conducting Safety Assessments” (2004).
Mating of the animals was performed by the supplier (Envigo Global Services Inc., Greenfield, IN). Gestation day zero (GD 0) was designated as the day mating was confirmed. All animals were shipped prior to GD 4. Animals were individual housed in suspended stainless steel cases. Water was provided ad libitum. Animals were fed certified rabbit diet number 2030C (Envigo). Beginning on GD 4 food consumption was measured daily. Environmental controls were set to maintain a temperature range of 16 to 22°C; a relative humidity range of 30 to 70%; 10 or greater air changes per hour and a 12 hour light/12 hour dark cycle.
Animals were checked twice daily (a.m. and p.m.) for mortality, abnormalities, and signs of pain or distress. Abnormal observations were recorded as they were observed. The supplier provided documentation as to whether mated females appeared normal on GD 0. Detailed observations were made on GD 4, 7, 10, 13, 17, 20, 24, and 29. Any abnormal findings or an indication of normal were recorded.
Cage-side observations were made once daily during the dosing interval (GD 7 through 19), approximately 1 hour post-dose, based on the last animal dosed for each group. Abnormal findings and any unscheduled observations were recorded as they were observed. Animals were weighed on GD 0, 4, 7 through 20, 22, 24, 27, and 29. The animal supplier provided GD 0 body weights.
Beginning on GD 4, food consumption was measured daily. Spilled feeders were recorded in the data and reported as Not Weighed.
All procedures in the protocol complied with the Animal Welfare Act, the Guide for the Care and Use of Laboratory Animals, and the Office of Laboratory Animal Welfare (Covance Laboratories, Salt Lake City, Utah) and were approved by the local Institutional Animal Care and Use Committee (IACUC).
Maternal Necropsy
Cesarean sections were performed on GD 29 for all animals. Animals were sacrificed via an appropriate barbiturate, followed by exsanguination. Pregnancy status was determined, uterine contents were examined, and the number of implantation sites was recorded, when present. When implantation scars were not apparent, the uterus was pressed between glass plates. The uterus from each pregnant animal was excised, weighed, and examined for the number and placement of live and dead fetuses, the number of early or late resorptions, and any abnormalities. The right and left ovaries from each pregnant female were examined for the number of corpora lutea. Each animal was examined macroscopically for abnormalities of the external features of the carcass; external body orifices; cervical, abdominal, and thoracic viscera; organs; and tissues; macroscopic abnormalities were noted.
Fetal Examinations
Each fetus was weighed and examined for external abnormalities. Live fetuses were sacrificed via injection with an appropriate barbiturate. A mid-coronal slice was made to the head of each fetus to evaluate the contents of the cranium. Internal organs of the thoracic and abdominal cavities were examined in the fresh state using modified Staples' technique. 17 At that time, the sex of each fetus was determined, and viscera were removed and discarded.
Using the Alizarin Red S staining method,18,19 carcasses were eviscerated and processed for skeletal evaluation which included examination of the skull, vertebral column, rib cage, pectoral and pelvic girdles, long bones, and extremities. Bone alignment and degree of ossification was assessed. Fetuses processed for skeletal evaluation were retained in glycerin, with thymol added as a preservative.
Any findings in the fetal external, visceral, and skeletal examinations were classified as either malformations or variations. Malformations are developmental deviations which are of gross structural changes, incompatible with life, or may affect the quality of life. Variations are structural or ossification deviations believed to have essentially no effect on body conformity or the well-being of an animal.
Data Evaluation and Statistical Analysis
Various models of calculators, computers, and computer programs were used to analyze data in this study. Values in some tables (eg, means, standard deviations, or individual values) may differ slightly from those in other tables, from individually calculated data, or from statistical analysis data because different models round off or truncate numbers differently. Neither the integrity nor the interpretation of the data was affected by these differences.
Except where otherwise stated, tests were performed using a two-sided risk at the 5% level of significance. Tables supporting statistical evaluations were generated using the Pristima preclinical software.
Mean values and standard deviations were calculated, as appropriate. Data from non-pregnant animals were excluded from summary calculations, as appropriate. For food consumption, uterine weight (including carcass weights), body weight, and body weight change data, Levene’s test was conducted to test for equality of variances between groups. Where Levene’s test was not significant (P > .05), a statistical comparison across test article-treated and control groups was conducted using a one-way analysis of variance (ANOVA). If the group effect of the ANOVA was not significant (P > .05), no further analyses were conducted. If the ANOVA was significant (P ≤ .05), Dunnett’s test was used for pairwise comparisons between each test article-treated group and the control group. If Levene’s test was significant (P ≤ .05), a Kruskal-Wallis nonparametric ANOVA was conducted. If the Kruskal-Wallis test was significant (P ≤ .05), pairwise comparisons of each test article-treated group with the control group were made using the Wilcoxon Rank Sum Test.
Cesarean section data (excluding uterine and carcass weights), percent male data, and fetal defect data (mean litter percent) were evaluated using a Kruskal-Wallis nonparametric ANOVA and the Wilcoxon Rank Sum Test, as described previously. Fetal defect data (proportion of litters affected) were evaluated using Fisher’s Exact Test (one-sided, increasing).
Mean live fetal weights (males, females, and combined sexes) were analyzed using analysis of covariance (ANCOVA). The litter size (live and dead fetuses) was used as the covariate. Where the group effect from the ANCOVA was significant (P ≤ .05), Dunnett’s test was used for pairwise comparisons between each test article-treated group and the control group.
Toxicokinetic statistical analysis was limited to the calculation of means and standard deviations.
Results
Clinical Observations
Non-adverse test article-related clinical observations following dose administration were noted in all MB-102-treated groups. All animals administered 45 and 113 mg/kg/day were observed with discolored urine (yellow and/or orange), discolored ears (yellow and/or orange), and discolored periorbital region (yellow, orange, and/or white). All animals administered 113 mg/kg/day were also observed with the entire body discolored (yellow) and discolored eyes (yellow). Additional clinical observations of discolored eyelids (yellow or orange) at 45 mg/kg/day and discolored mouth, nose, and/or pelage (yellow) were noted at 113 mg/kg/day. Discolored urine and ears (yellow) were also observed in animals administered 4.5 mg/kg/day, however, at lesser incidence. Based on the nature of the findings (yellow/orange discoloration) and the color and fluorescent properties of the test article, these observations in all MB-102-treated groups were considered test article-related but non-adverse.
All other observations were single incidence and/or transient.
Toxicokinetics
Summary of the MB-102 Toxicokinetic Parameters in the Plasma of Pregnant Rabbits.
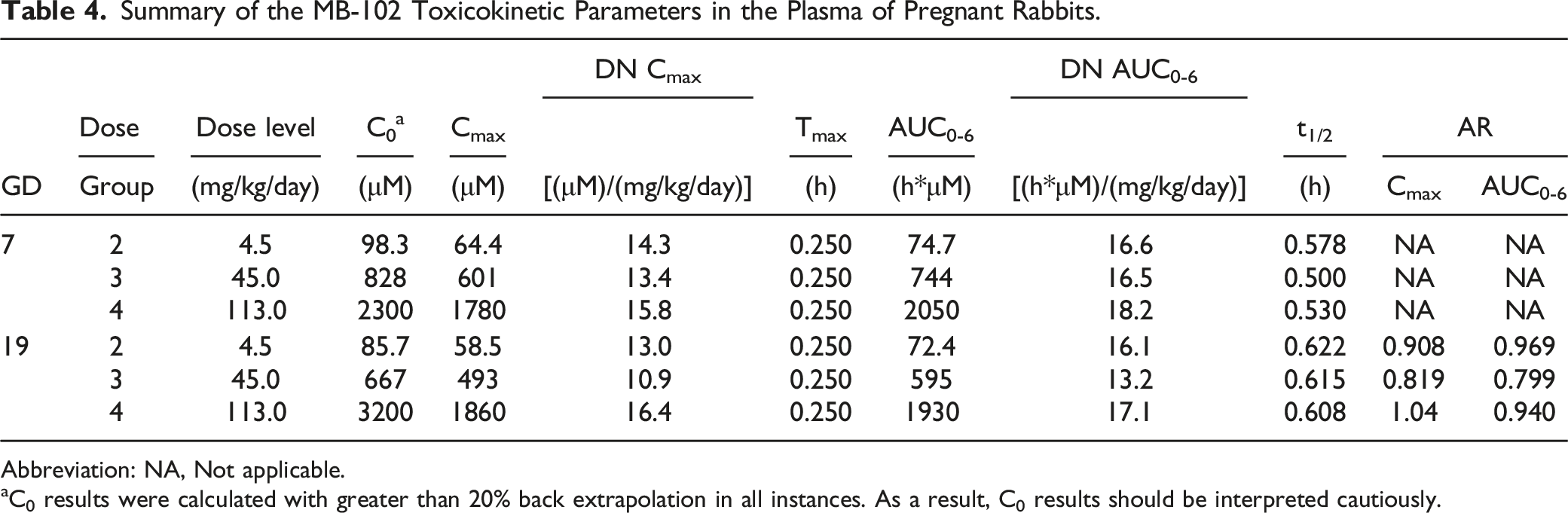
Abbreviation: NA, Not applicable.
aC0 results were calculated with greater than 20% back extrapolation in all instances. As a result, C0 results should be interpreted cautiously.
Body Weights
Summary of Body Weight Gain.
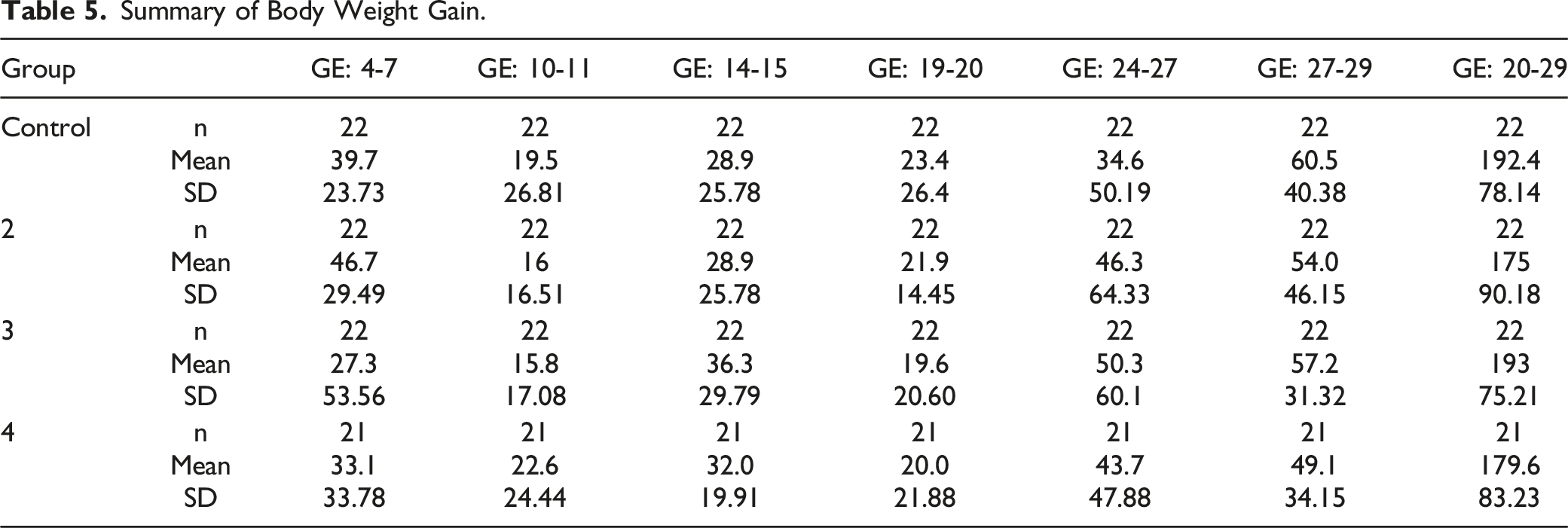
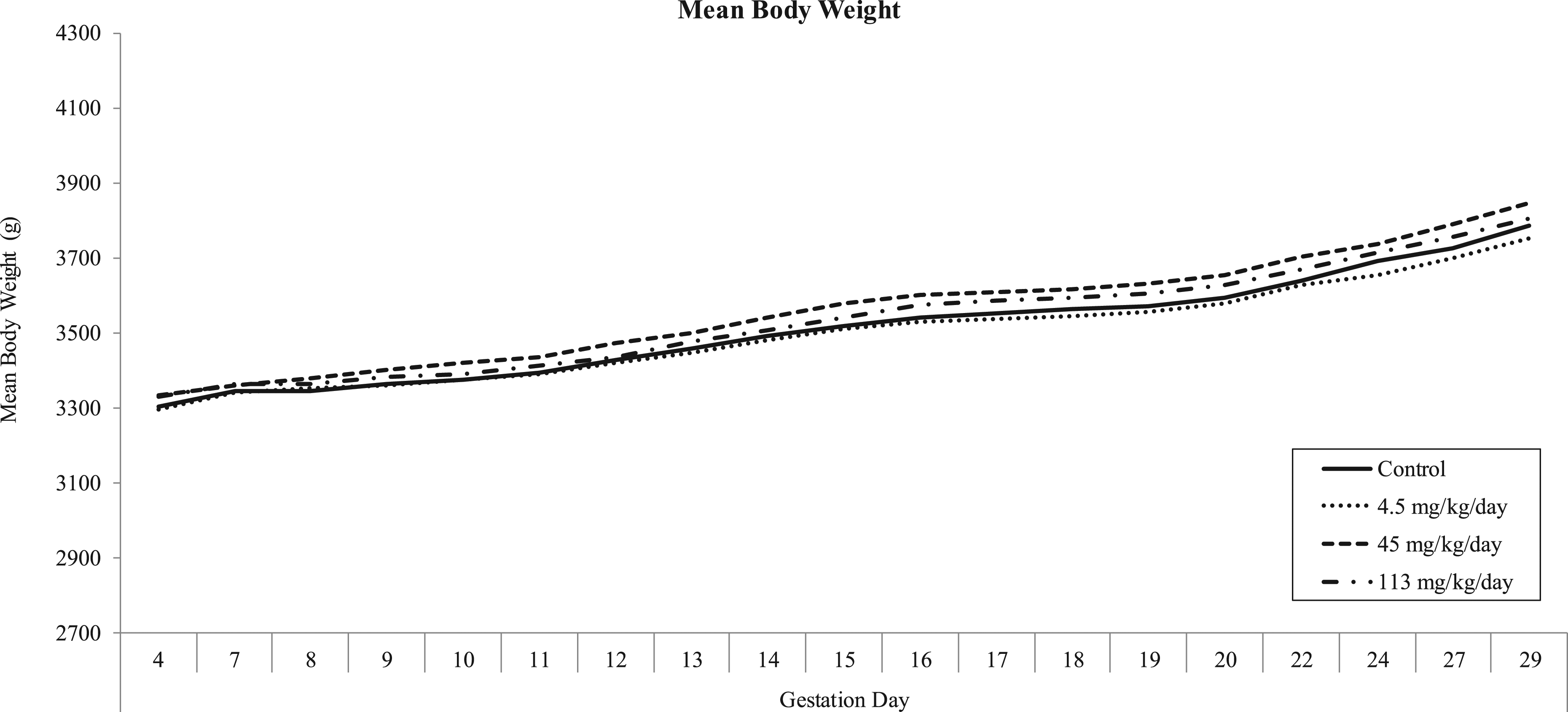
Mean body weights.
No test article-related effect on mean body weight values was observed for any MB-102 dose level. All intervals were comparable with controls.
Food Consumption
Food consumption data are depicted graphically in Figure 3 and summarized in Table 6. Summary of mean food consumption. Summary of Mean Food Consumption.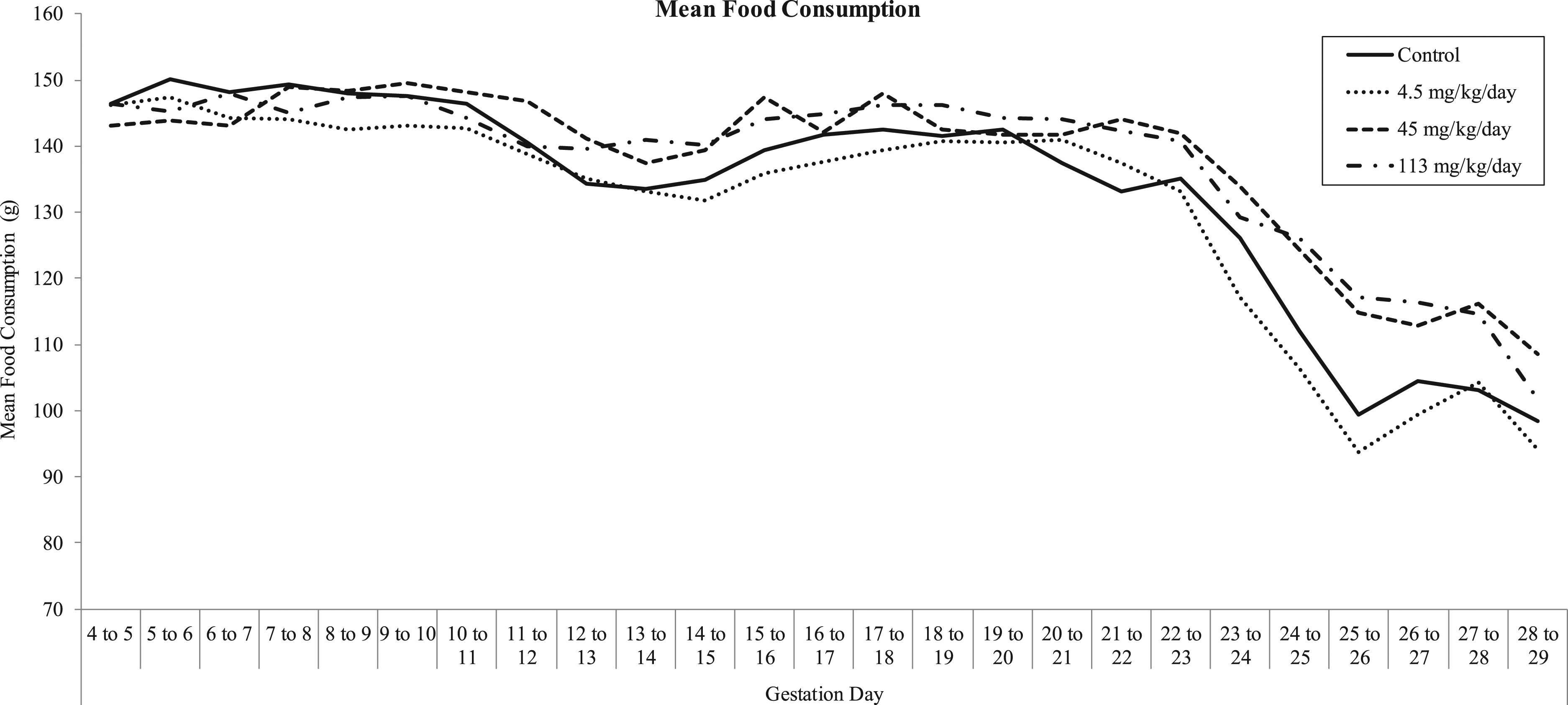
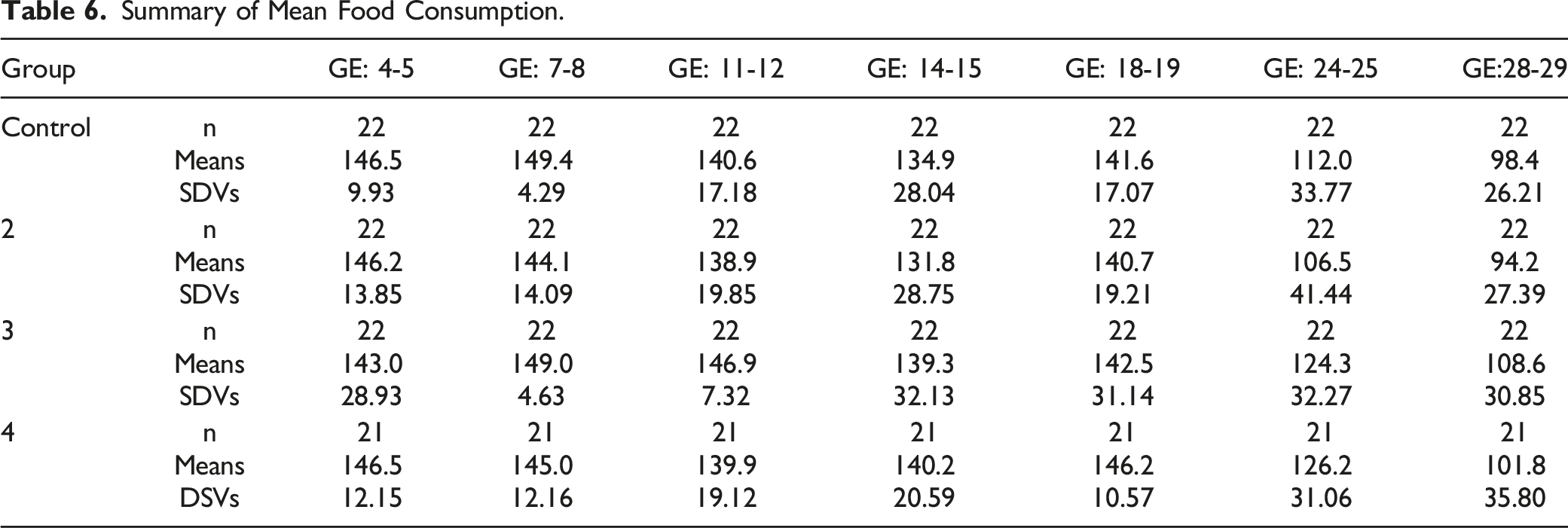
No test article-related effect on mean food consumption was observed for any MB-102 dose level. All intervals were comparable with controls.
Reproductive Performance
Summary of Reproductive Performance.
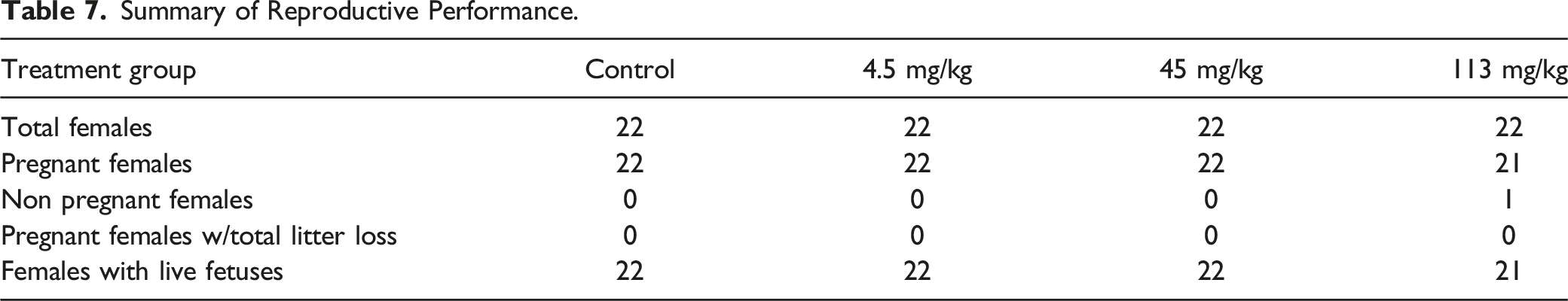
The pregnancy rate was 100, 100, 100 and 95% for animals administered vehicle control article or 4.5, 45, and 113 mg/kg/day, respectively.
Cesarean Section Analyses-Maternal Necropsy
Summary of Cesarean Section Results.
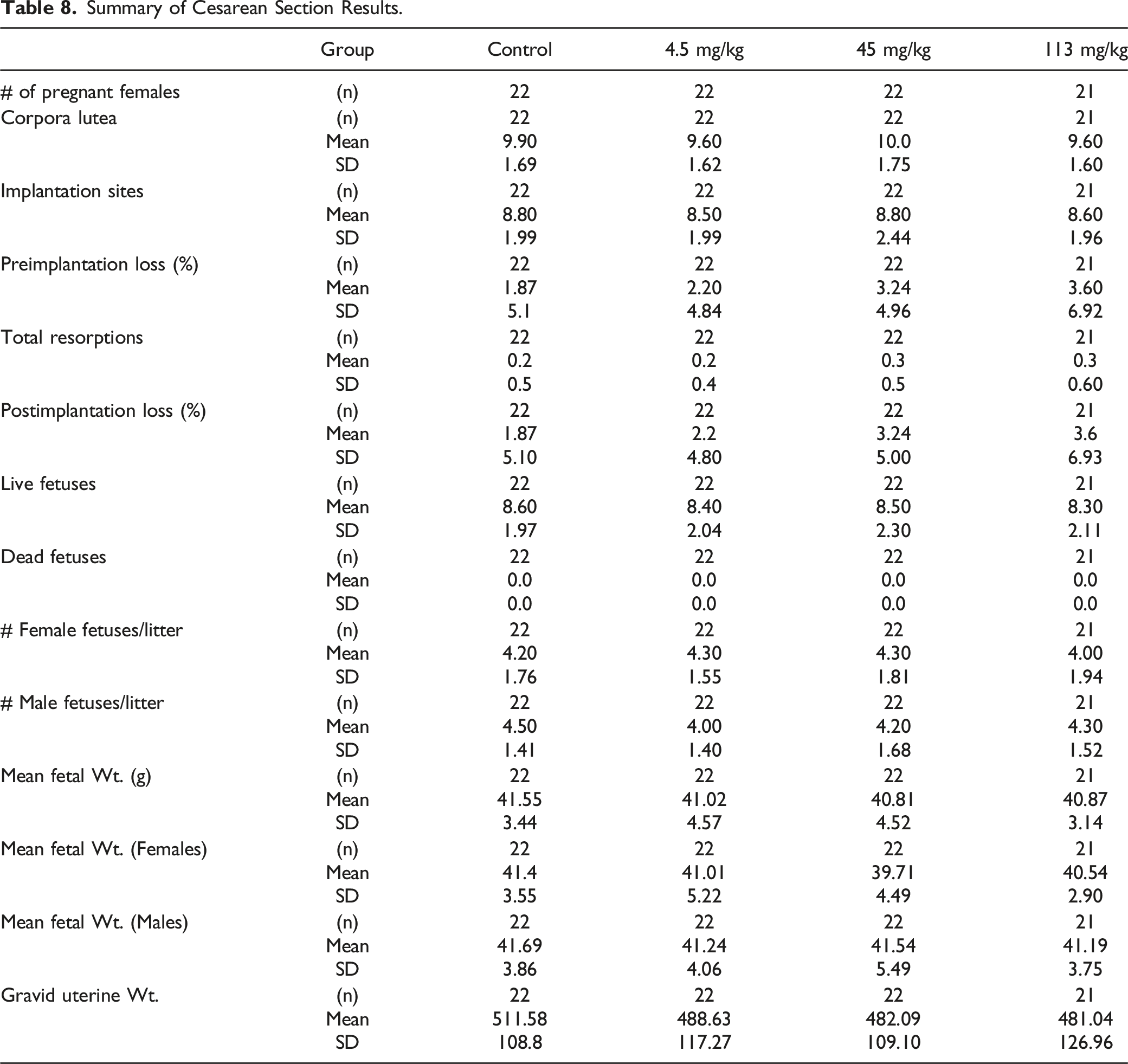
Summary of Fetal Evaluations
No test article-related fetal external anomalies were observed for any dose level.
Omphalocele (malformation) was noted in a single fetus from an animal administered 113 mg/kg/day. This was a single occurrence, and the incidence was within the Covance DART Historical Control database. Protruding tongue (malformation) was noted in a single fetus from an animal administered 4.5 mg/kg/day. This was also a single occurrence, only in the low dose group. Due to the single occurrence of these anomalies, they were considered not test article-related.
Fresh Visceral Evaluations
No test article-related fetal visceral anomalies were observed for any dose level.
The observed visceral malformations included retroesophageal subclavian artery, absent gall bladder, cardiomegaly, ventricular septum defect, and malpositioned kidney. As all the malformations were single incidence, also noted in controls, and/or were within the DART Historical Control database, they were considered not test article-related.
All visceral variations were single incidence within a litter or group, also noted in controls, lacked dose responsiveness, and/or were within the DART Historical Control database, and thus were considered not test article related.
Summary of Skeletal Evaluations
No test article-related fetal skeletal anomalies were observed for any dose level.
The observed skeletal malformations included absent or fused ribs, fused mandible/zygomatic arch, fused or misaligned sternebra, supernumary cervical arch, fused cervical centrum, absent or hemivertebra lumbar vertebra, absent or fused thoracic centrum, and hemivertebra thoracic vertebra. All malformations were single incidence, also noted in controls, lacked dose response, limited to single litters, and/or were within the DART Historical Control database and thus, were considered not test article related.
Two fetuses from 2 litters administered 113 mg/kg/day were observed with misaligned caudal vertebra centra. Although the incidence of this variation was slightly outside of the DART Historical Control database (% fetus and % litter incidence of 1.55 and 10%, respectively, in this study; vs incidence of 0.93 and 6%, respectively, in the database), this variation was limited to single fetuses in 2 litters. In the absence of maternal and fetal toxicity and fetal dysmorphogenesis in this study, the occurrence of this variation was considered incidental and thus considered not test article related.
Discussion
Previously reported safety and toxicity studies of MB-102 demonstrated negligible safety concerns and were critical to the FDA clearance of this agent to advance to Phase II human clinical trials. The studies included herein further supplement the overall safety and toxicity profile of this novel optical tracer. The focus of these studies was specifically designed to assess maternal toxicity and potential effects on embryo-fetal development in pregnant rabbits following the intravenous administration of MB-102. Included in this series of studies were mortality, food consumption, body weight gain, reproductive indices, Cesarean section parameters, fetal mortality, and overall clinical observations. A toxicokinetic profile of this agent was also determined. All animals survived to the conclusion of the study. With regards to food consumption and body weight, gain there were no significant differences between the untreated control group and the 3 groups of animals dosed with MB-102. Clinical observations were limited to discoloration of the skin, eyes and/or pelage at the 2 higher doses administered. These findings are consistent with this fluorescent tracer and were considered nonadverse. No test article-related fetal external abnormalities were observed for any MB-102 dose level, and no test article-related fetal skeletal abnormalities were observed for any MB-102 dose level.
Exposure, as assessed by MB-102 Cmax and AUC0-6, increased with the increase in dose level from 4.5 to 113 mg/kg/day. The increases in Cmax and AUC0-6 values were generally dose proportional. No accumulation of MB-102 was observed after multiple doses in pregnant rabbits. Based on these data, the no observed adverse-effect level (NOAEL) for maternal and fetal toxicity was 113 mg/kg/day (GD 7 Cmax and AUC0-6 values of 1780 μM and 2050 h*μM, respectively; GD 19 Cmax and AUC0-6 values of 1860 μM and 1930 h*μM, respectively).
In conclusion, intravenous administration of MB-102 was not associated with any test article related effects on mortality, food consumption, body weight gain, Cesarean section parameters, macroscopic observations, reproductive performance, and developmental toxicity including teratogenic potential in pregnant rabbits.
Footnotes
Acknowledgments
The authors wish to thank Lisa M. Balbes, PhD, of Balbes Consultants LLC for editorial support.
Author Contributions
Richard Dorshow contributed to conception and design, contributed to acquisition, analysis, and interpretation, critically revised manuscript, gave final approval and agrees to be accountable for all aspects of work ensuring integrity and accuracy; Joseph Bugaj contributed to conception, contributed to interpretation, drafted manuscript, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RBD is an employee of MediBeacon Inc. and JEB is a paid consultant of MediBeacon Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by MediBeacon Inc.
Ethical Approval
The study was conducted at Covance Laboratories Inc., Madison, WI.
