Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of 16 bovine milk proteins and protein-derived ingredients, which function mainly as skin and hair conditioning agents in personal care products. The Panel reviewed relevant data provided in this safety assessment, and concluded that these ingredients are safe in the present practices of use and concentration.
Keywords
Introduction
The safety of various hydrolyzed proteins as used in cosmetics has been reviewed by the Panel in several previous assessments. The Panel concluded that Hydrolyzed Keratin (finalized in 2016), Hydrolyzed Collagen (published in 1985, re-review published in 2006) Hydrolyzed Soy Protein (finalized in 2015), Hydrolyzed Silk (finalized in 2015), Hydrolyzed Rice Protein (published in 2006), and Hydrolyzed Corn Protein (published in 2011) are safe for use in cosmetics.4-10 Additionally, the Panel concluded that Hydrolyzed Wheat Gluten and Hydrolyzed Wheat Protein are safe for use in cosmetics when formulated to restrict peptides to a weight-average molecular weight (MW) of 3500 Daltons (Da) or less. 11 The Panel concurrently reviewed the safety of plant-derived proteins and peptides and tissue-derived proteins and peptides, in separate reports.
While relevant data on the cosmetic ingredient Hydrolyzed Lactalbumin could not be identified in the published literature, information on the unprocessed protein, lactalbumin, was discovered and has been incorporated into this report to aid in the review of safety.
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) Website (http://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; http://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Chemistry
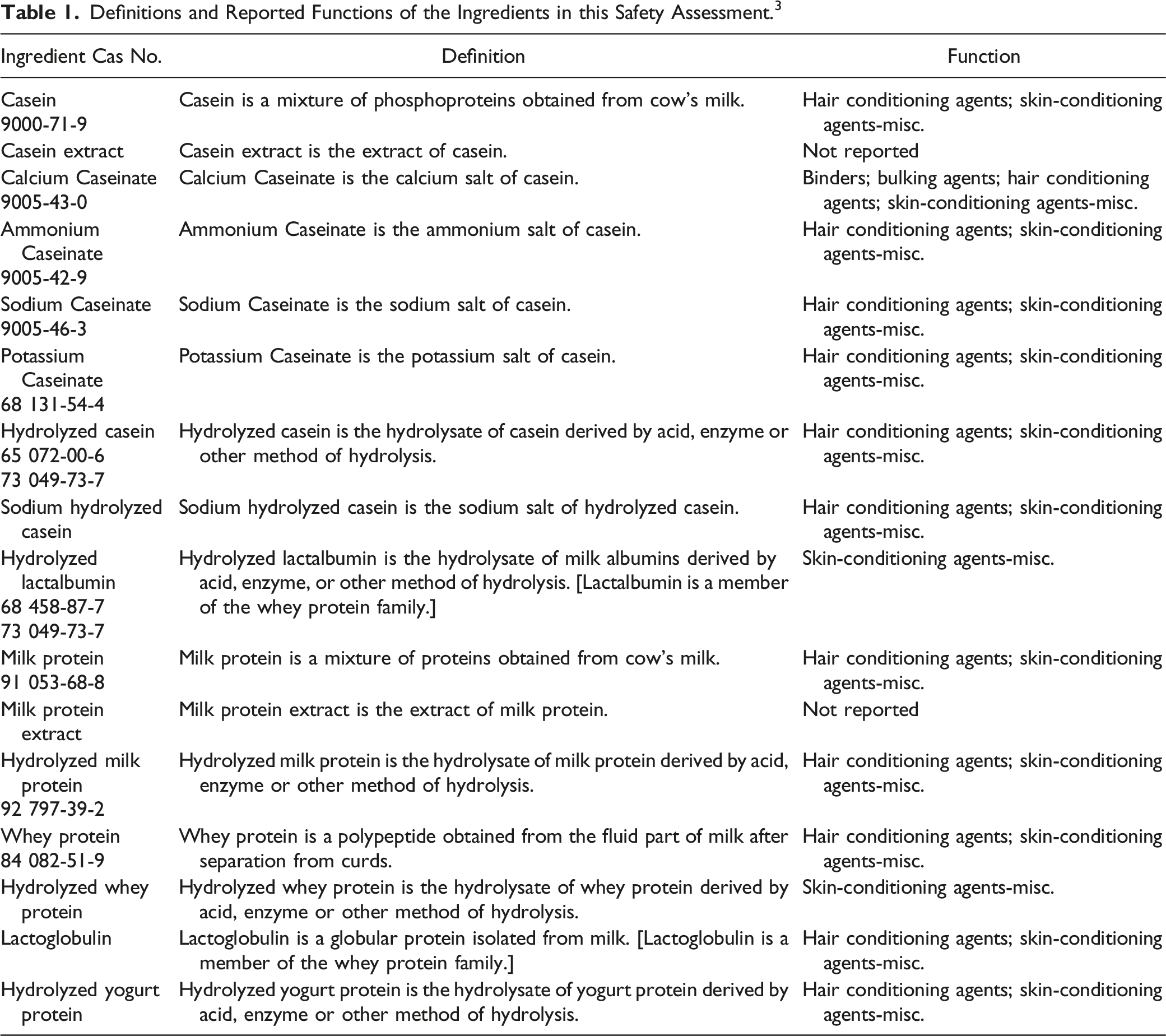
Definition
Definitions and Reported Functions of the Ingredients in this Safety Assessment. 3
Protein hydrolysates can be prepared via acid hydrolysis, enzymatic hydrolysis, or other methodologies. The methodology selected and the conditions and duration of the hydrolysis may profoundly affect the size and reactivity of the hydrolysates. Most of the ingredients in this report, even those without “hydrolyzed” in the name, are hydrolyzed to some degree as necessary for extraction or solubilization. Further steps towards solubilization of these macromolecules commonly include reaction with an alkaline substance to produce a protein salt (e.g., Calcium Caseinate).
Milk proteins in yogurt are partially hydrolyzed by proteolytic enzymes in lactic acid bacteria during fermentation. 14 However, the levels of hydrolyzed bacterial proteins in yogurt are expected to be insignificant compared to the levels of hydrolyzed milk protein after processing to produce hydrolyzed yogurt protein.
Physical and Chemical Properties
Casein and Caseinate Salts
Casein is described as an off-white to cream-colored granular or fine powder. It is insoluble in water and alcohol, but can be dissolved by aqueous alkalis to form caseinate salts. 15 Caseinate salts are white to cream-colored granules or powders that are soluble or dispersible in water. The amino acid sequence of β-casein contains 209 residues with an approximate MW of 23 600 Da. 16
Hydrolyzed Casein
A supplier has reported that the MW of a Hydrolyzed Casein product is approximately 600 Da. 17
Hydrolyzed Milk Protein
A Hydrolyzed Milk Protein product was described as a cream colored powder with a slight, characteristic odor and a pH of 5.0 to 7.0. 18
A supplier has reported that the MW of Hydrolyzed Milk Protein is ∼1000 Da. 19 Another supplier has reported the MW distribution of 3 batches of Hydrolyzed Milk Protein yielded 58.4% of the MW to be below 5000 Da and 41.4% of the MW to be greater than 5000 Da and less than 30 000 Da. 20
At 25°C, Hydrolyzed Milk Protein is soluble in water, partially soluble in 75/25 and 50/50 water/ethanol, and insoluble in 25/75 water/ethanol, 200 proof ethanol, mineral oil, glycerin, and propylene glycol. 18
Hydrolyzed Lactalbumin
α-Lactalbumin (non-hydrolyzed) is described as a homogenous, free-flowing, semi-hygroscopic, light cream-colored powder. 15 Physical and chemical properties on Hydrolyzed Lactalbumin were not found.
Method of Manufacturing
Methods used to manufacture protein hydrolysates typically yield broad MW distributions of peptides, ranging from 500 to 30 000 Da. 21 However, certain enzymes, such as papain, can routinely produce narrower distributions of 500 to 10 000 Da. For example, if the average MW of an amino acid is 135 Da, then, under the broader distribution figures (i.e., 500 to 30 000 Da), these ingredients are approximately 4 to 220 amino acids in length (and approximately 4 to 74 amino acids in length under the narrower distribution, i.e., 500 to 10 000 Da). 22
Casein
Commercial casein is derived from the coagulum formed by treating skim milk with a food-grade acid (acid casein), enzyme (rennet casein), or other food-grade precipitating agent. 15 After precipitation, Casein is separated from the soluble milk fraction, washed, and dried. Casein is a mixture of at least 20 electrophoretically distinct phosphoproteins, with the main fractions being α-casein, β-casein, and κ-casein.
Hydrolyzed Casein
A supplier reported that a Hydrolyzed Casein product (MW = 600 Da; 30% solution in water) is prepared by acidic, alkaline, and/or enzymatic hydrolysis of bovine milk until the MW reached the target range. 17
Hydrolyzed Lactalbumin
α-Lactalbumin (non-hydrolyzed) is isolated from either bovine milk or from whey. 15 A method of manufacture for the hydrolysis of lactalbumin (specifically) to Hydrolyzed Lactalbumin was not found.
Hydrolyzed Milk Protein
A supplier reported that Hydrolyzed Milk Protein is produced from milk intended for human consumption. 23 The milk solids are separated and hydrolyzed with a protease for 2 h. When the target MW is achieved, the enzyme is inactivated by heating the solution to 140°C for 30 min. The inactivation step is repeated if gelatin mixed with a sample loses viscosity, indicating the presence of active protease.
Another supplier reported that Hydrolyzed Milk Protein is manufactured by enzymatic hydrolysis for a specific duration and at an elevated temperature (details not provided). 24 The resultant hydrolyzed proteins have MWs in the 2000 - 4000 Da range and all contain di- and tri-peptides.
Whey Protein
Whey is the liquid obtained by separating the coagulum from milk, cream, and/or skim milk (usually in cheese making). 15 Acid-type whey is produced by converting a significant amount of lactose to lactic acid or by direct acidification of milk. Sweet-type whey is derived from a process in which there is insignificant conversion of lactose to lactic acid. Whey protein concentrate is a liquid or dry product that is obtained by the removal of sufficient non-protein constituents from whey so that the finished dry product contains not less than 25.0% protein, while whey protein isolate is a liquid or dry product that is obtained by removing sufficient non-protein constituents from whey so that the finished dry product contains not less than 90% protein. Whey protein concentrate and whey protein isolate are produced by physical separation techniques such as precipitation, filtration, dialysis and/or ion exchange.
Composition
Casein
Casein is reported to have all the amino acids considered to be essential for human nutrition. 15
Hydrolyzed Milk Protein
Amino Acid Distribution for a Hydrolyzed Milk Protein Produced by Enzymatic Hydrolysis. 18

Impurities
The ingredients in this safety assessment are bovine sourced; however, the US FDA does not consider milk or processed milk ingredients as risk materials for transmission of infectious agents (i.e. bovine spongiform encephalopathy) in cosmetic products (21 CFR §700.27).
The World Organization for Animal Health (OIE) recommends that “when authorizing import or transit of [milk and milk products] and any products made from these commodities and containing no other tissues from cattle, veterinary authorities should not require any BSE related conditions [i.e. restrictions], regardless of the BSE risk status of the cattle population of the exporting country, zone, or compartment.” 26
The Food Chemicals Codex, a compendium of internationally recognized standards published by the United States Pharmacopeia (USP) for the purity and identity of food ingredients, states that the acceptable lead limit for Casein and caseinate salts is no more than 1 mg/kg. 15 Acid casein should contain not less than 90% protein calculated on a dry basis. The acceptable lead limit in α-lactalbumin (non-hydrolyzed form of Hydrolyzed Lactalbumin) is no more than .5 mg/kg on the dried basis, and the acceptable phosphorus limit is no more than 700 µg/g. α-Lactalbumin may also contain β-lactoglobulin (no more than 6.5% calculated on total protein basis), lactose (no more than 1.0%), and lipids (no more than 1.0%). Whey, whey protein concentrate, and whey protein isolate may contain no more than .5 mg/kg lead calculated on the dried basis. Whey protein isolate should contain not less than 90% protein calculated on a dry basis.
Hydrolyzed Casein
A supplier reported that a Hydrolyzed Casein product (MW = 600 Da, 30% solution in water) did not contain more than 5 ppm heavy metals and not more than .5 ppm arsenic. 17
Hydrolyzed Milk Protein
A Hydrolyzed Milk Protein product was reported to have a maximum microbiological count of 500 organisms per gram (opg), with yeast and molds being < 100 opg. 18
United States Pharmacopeia
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the FDA and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by Industry in response to surveys conducted by the Personal Care Products Council (Council) of maximum reported use concentrations by product category.
NR = Not reported.
Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
aIt is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
bNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
cIt is possible these products may be powders, but it is not specified whether the reported uses are powders.
dUse reported in an aerosol deodorant.
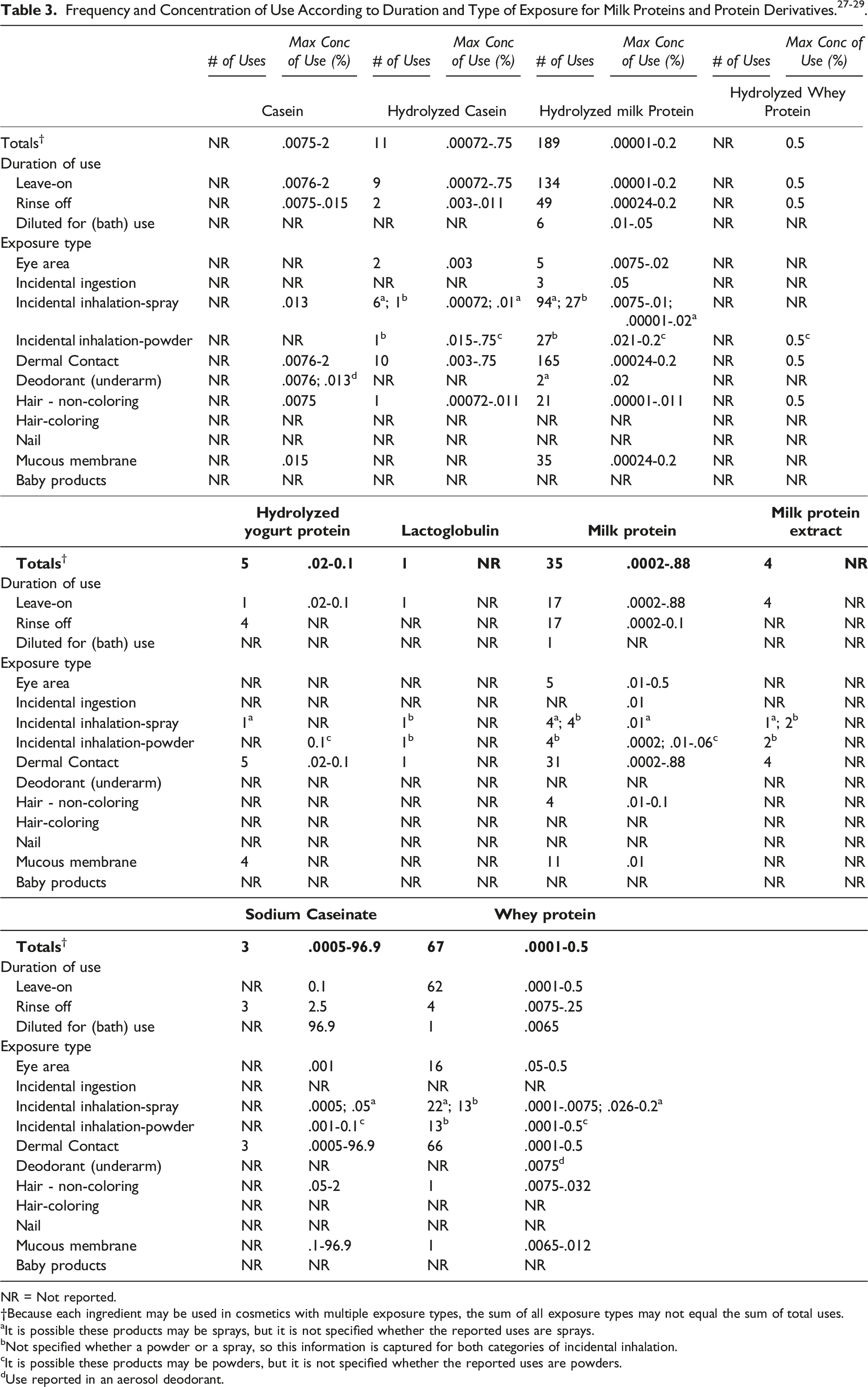
In some cases, reports of use were received from the VCRP, but no concentration of use data were provided. For example, Milk Protein Extract is reported to be used in 4 formulations, but no use concentration data were provided. In other cases, no uses were reported to the VCRP, but a maximum use concentration was provided in the industry survey. For example, Casein was not reported in the VCRP database to be in use, but the industry survey indicated that it is used at concentrations up to 2% in makeup preparations. It should be presumed that Casein is used in at least 1 cosmetic formulation for each category for which it is reported to be used.
Some of these ingredients may be used in products that can come into contact with mucous membranes and the eyes. For example, Sodium Caseinate is used in bath oils, tablets, and salts at up to 96.9% and Milk Protein is used in eye makeup preparations at up to .5%. 28 Additionally, some of these ingredients were reported to be used in spray deodorants, hair sprays, face powders, face and neck sprays, body and hand sprays, and fragrances and could possibly be inhaled. For example, Casein was reported to be used in a spray deodorant at .013% and Milk Protein was reported to be used in face powders at .0002%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays.30-33 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.30,32 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 32 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.34-36
The milk protein and protein-derived ingredients described in this safety assessment are not restricted from use in any way under the rules governing cosmetic products in the European Union. 37
Non-Cosmetic
According to the FDA, bovine milk is considered generally recognized as safe (GRAS) as it is a substance used in food prior to January 1, 1958, through experience based on common use in food (21 CFR§170.30). The FDA has also determined that the use of peptones as direct food substances is GRAS. These GRAS peptones are defined as “the variable mixture of polypeptides, oligopeptides, and amino acids that are produced by partial hydrolysis of casein…or lactalbumin (whey protein) (21 CFR §184.1553). Additionally, Casein is GRAS as substances migrating to food from paper and paperboard products (21CFR §182.90). Sodium Caseinate is GRAS for human and animal consumption (21CFR§182.1748, 21CFR§582.1748). Whey is GRAS for human consumption (21CFR§184.1979). Labeling requirements for milk-related ingredients and hydrolyzed proteins in food that is GRAS for human consumption are defined in 21CFR101.4 and 21CFR102.22.
Calcium Caseinate and Sodium Caseinate are used in over the counter (OTC) weight control drug products, but these active ingredients do not have adequate data available to be generally recognized as safe and effective for these specified uses (21 CFR§ 310.545). These casein salts and whey protein, in mixtures with other substances, are also being investigated for use as drug coatings and topical drug delivery systems, respectively.38-40
The FDA requires allergen labeling when 1 or more of the 8 major food allergens, such as milk, are included in food. 41
Casein and caseinate salts, α-lactalbumin, whey, whey protein concentrate, and whey protein isolate are all listed in the Food Chemicals Codex. 15 Casein and caseinate salts are described as binders, extenders, clarifying agents, emulsifiers, and stabilizers in food. α-Lactalbumin is described as a nutrient and a source of tryptophan. Whey and whey protein concentrate are described as texturizers and nutrients, with the concentrate also used as an emulsifier, water-binding aid, and gelling agent in foods. Whey protein isolate is considered a source of high-quality protein that may also be used as a gelling agent, water-binding aid, foaming or whipping aid, emulsifier, and an edible coating used as a moisture barrier.
Toxicokinetics
Hydrolyzed Milk Protein
While no experimental data were available for the dermal absorption of Hydrolyzed Milk Protein, it was noted that gastro-intestinal absorption allows for substantially greater bioavailability than dermal absorption. 42 In worst-case scenarios of oral exposures greater than 2000 mg/kg, no signs of systemic toxicity were observed and, therefore, it was concluded that no systemic toxicity would occur with cutaneous exposure.
Toxicological Studies
Bovine milk, milk proteins, and milk protein derivatives are GRAS food substances, and daily exposures from food use would result in much greater systemic exposures than those resulting from use in cosmetic products. Consequently, systemic toxicity potential for these ingredients is not addressed further in this report. The safety assessment focuses on the potential for irritation and sensitization from topical exposure to these milk ingredients.
Genotoxicity
Hydrolyzed Casein
The mutagenic potential of a Hydrolyzed Casein product (MW = 600 Da, 30% solution in water) was studied in an Ames test using Salmonella typhimurium strains TA98, TA100, TA1535, and TA1537 and Escherichia coli strain WP2uvrA, with and without S9 metabolic activation. 17 Concentrations were tested up to 5000 μg/plate. The test material did not induce reverse mutations with or without S9. It was concluded that Hydrolyzed Casein was not mutagenic.
Hydrolyzed Milk Protein
The potential of Hydrolyzed Milk Protein to induce gene mutation was studied in S. typhimurium strains TA 98, TA 100, TA 1535, and TA 1537 with and without S9 metabolic activation. 42 Concentrations were tested up to 5000 μg/plate. The test material did not induce reverse mutations with or without S9. It was concluded that Hydrolyzed Milk Protein was not mutagenic.
Carcinogenicity
Tumor Suppression
Several studies have investigated the carcinogenic effects of milk and its related proteins and protein derivatives in the diet, and the results indicated that these substances may suppress tumor formation.43-45 Review articles of the role of milk proteins and hydrolyzed proteins on cancer reported that Casein and casein peptides have antimutagenic properties, and that animal models for colon and mammary tumorigenesis (like the study described below) showed that Hydrolyzed Whey Protein suppressed tumor development.46-48 The tumor suppression observed in studies with Hydrolyzed Whey Protein has been attributed to the high content of cystine/cysteine and γ-glutamylcyst(e)ine dipeptides in the milk proteins, which are efficient substrates for synthesizing glutathione, an important cellular antioxidant.
An example of tumor suppression is the effect of milk proteins on the ability of dimethylbenzanthracene (DMBA) to induce mammary tumors in pregnant Sprague-Dawley rats. 45 The rats (number not reported) were fed diets that included 20% Casein, Hydrolyzed Casein, Whey Protein, or Hydrolyzed Whey Protein starting on gestation day 4. The offspring of these rats were fed the same diet. At 50 d, the female offspring (44 - 49 rats/group) were dosed by gavage with sesame oil containing 80 mg/kg DMBA and were killed 62 d post-treatment. The rats that were fed Hydrolyzed Whey Protein had an adenocarcinoma incidence of 17% compared to rats fed Casein (34%), Hydrolyzed Casein (33%), and Whey Protein (36%) (P < .001). The median palpable tumor latency for rats fed Hydrolyzed Whey Protein (61 d, P < .001) was greater compared to those fed Casein (44 d), Hydrolyzed Casein (42 d), or Whey Protein (45 d). When compared to rats fed Casein and Hydrolyzed Casein, tumor multiplicity was lower in rats fed Hydrolyzed Whey Protein (1.5 vs 3.0, P < .05). The authors of the study concluded that dietary intake of Hydrolyzed Whey Protein reduced DMBA-induced mammary tumor formation.
Other Relevant Studies
Type 1 Hypersensitivity
Bovine milk protein is a major food allergen that can produce Type 1 (immediate) reactions in sensitized individuals, including up to 8% of children.49,50 The allergy to bovine milk protein usually occurs in infancy and childhood and is often outgrown by age 5, but approximately 15% to 20% of allergic children remain allergic into adulthood with increased levels of immunoglobulin E (IgE), especially IgE to bovine-sourced proteins. The IgE-mediated reaction may include cutaneous, respiratory, and gastrointestinal reactions that may on rare occasions result in systemic anaphylaxis.1,49,50 Non-IgE-mediated reactions may also occur, but these are not as well characterized. 50 While the reactions may be to any of the proteins found in milk, reactions are most commonly linked to α-lactalbumin, β-lactoglobulin, and casein.
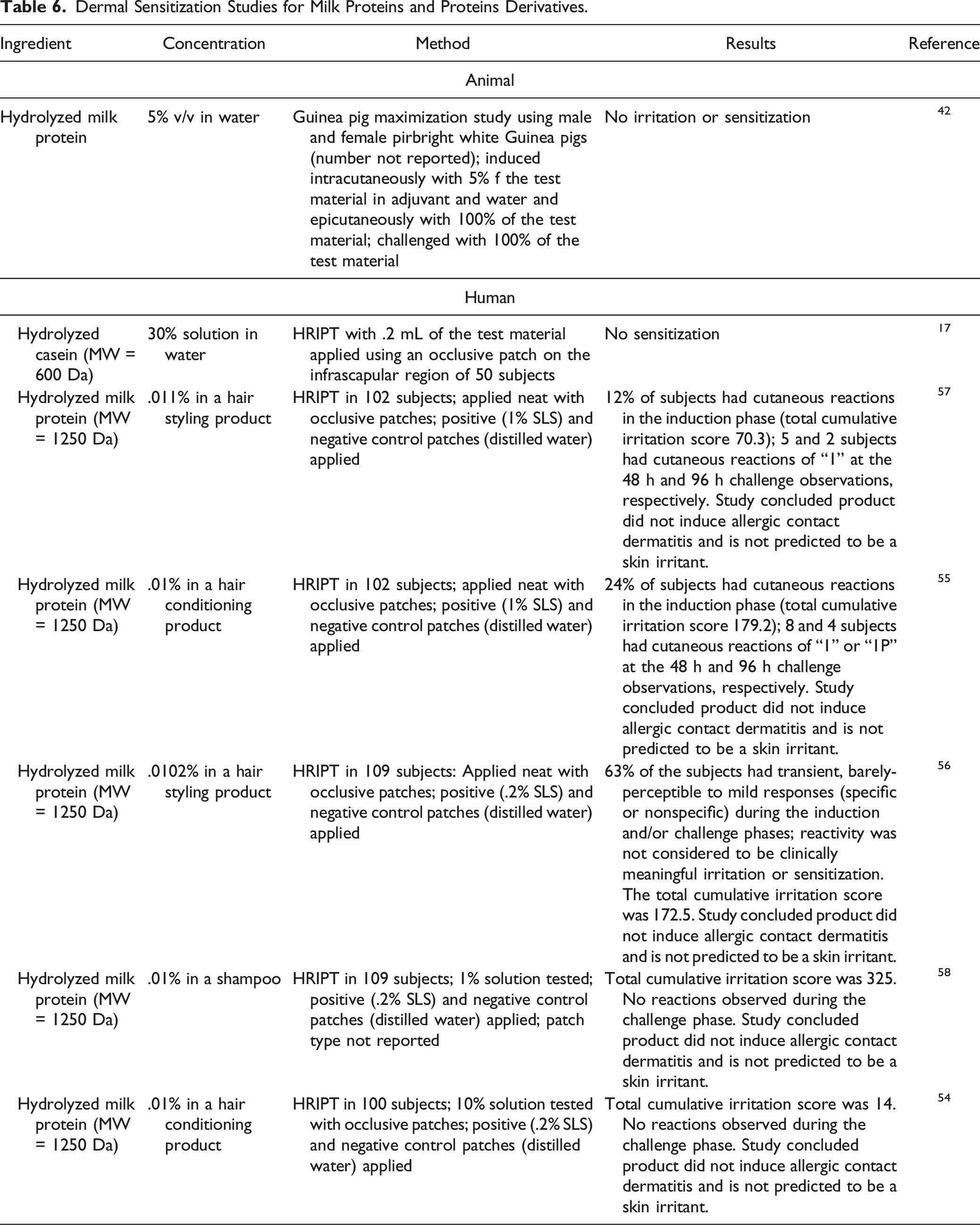
Dermal Irritation and Sensitization Studies
Irritation and Sensitization
Dermal Irritation Studies for Milk Proteins and Protein Derivatives.

*OECD = Organisation for Economic Co-operation and Development.
Dermal Sensitization Studies for Milk Proteins and Proteins Derivatives.
Phototoxicity
Ocular Irritation Studies
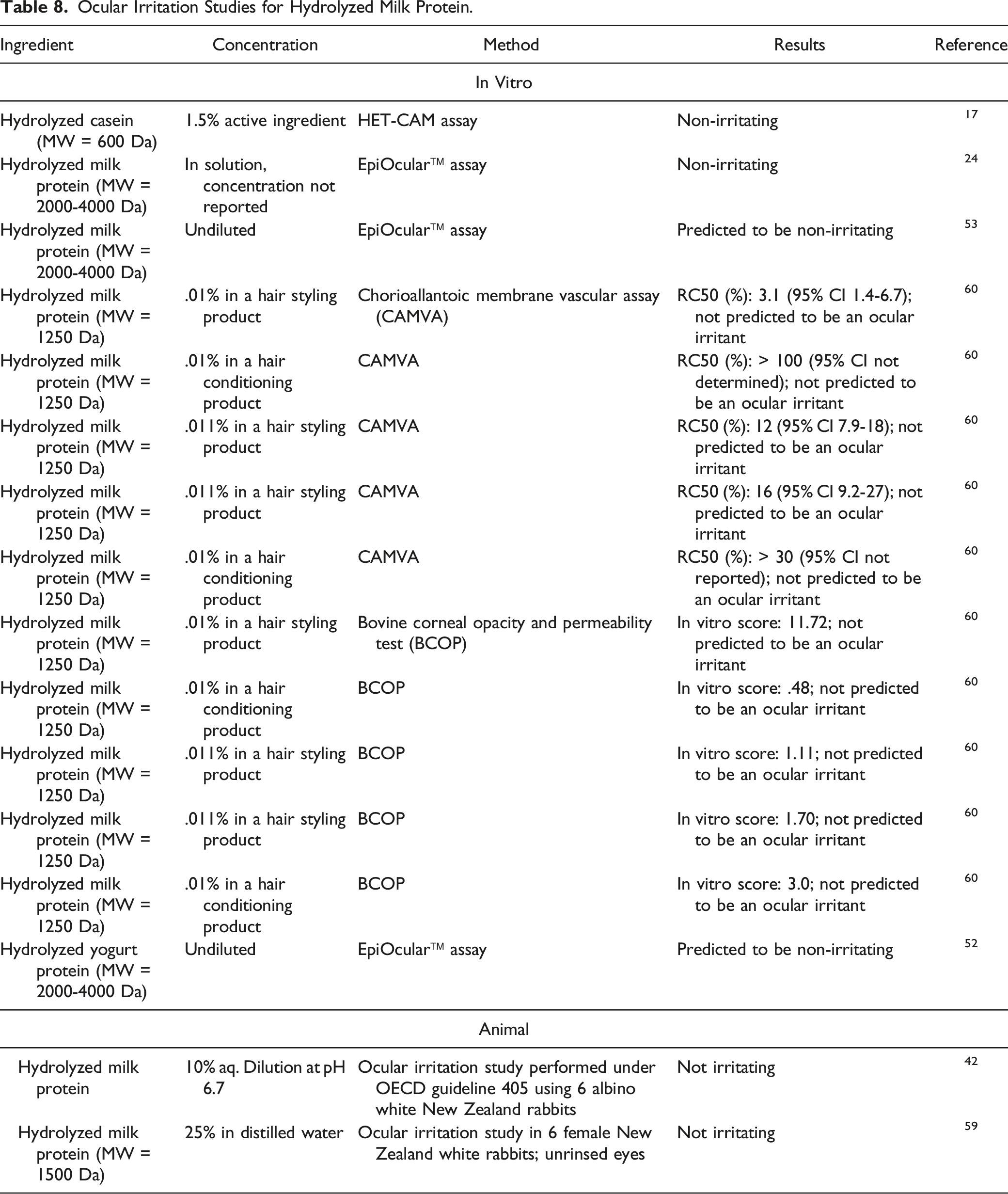
Ocular Irritation Studies for Hydrolyzed Milk Protein.
Clinical Studies
Hydrolyzed Milk Protein
A study of sensitization to protein hydrolysates in hair care products was performed in 3 groups of patients. 61 Eleven hairdressers with hand dermatitis comprised the first group, which submitted to scratch and prick tests with 22 trademarked protein hydrolysates, including Hydrolyzed Milk Protein, as well as quaternized hydrolyzed proteins. The second test group included 1260 consecutive adults with suspected allergic respiratory disease; they were subjected to skin prick tests with 1 to 3 of the protein hydrolysates (only 1232 patients in this group were tested with Hydrolyzed Milk Protein). The third group of patients included 28 adults with atopic dermatitis and was also tested with 1 to 3 protein hydrolysates via a skin prick test.
Of all 3 groups tested, positive reactions were seen in a total of 12 patients (all female with atopic dermatitis) from exposure to 3 of the 22 protein hydrolysates. All 12 had reactions to hydroxypropyl trimonium hydrolyzed collagen. One of the 12 also had a reaction to hydroxypropyl trimonium hydrolyzed milk protein (not an ingredient in this report) while 3 others had a reaction to 1 trademarked version of hydrolyzed collagen. No adverse reactions to Hydrolyzed Milk Protein were observed in the 1271 patients tested . 61
Summary
Hydrolyzed Milk Protein is used in 189 formulations; the majority of uses are in leave-on products. Whey Protein has the second greatest number of overall uses reported, with a total of 67; the majority of the uses are in leave-on formulations. Sodium Caseinate has the highest reported maximum concentration of use; it is used at up to 96.9% in bath oils, tablets and salts. The highest reported maximum concentration of use in a leave-on formulation for this ingredient is .1% in a face and neck skin care product. Casein has the highest reported maximum concentration of use in a leave-on product and is used at up to 2% in makeup preparations.
Bovine milk, milk proteins, and milk protein derivatives are GRAS, and daily exposures from food use would result in much greater systemic doses than those resulting from use in cosmetic products. The safety assessment focuses on the potential for irritation and sensitization from topical exposure to these milk ingredients.
Hydrolyzed Milk Protein and Hydrolyzed Casein were not mutagenic at concentration up to 5000 μg/plate in Ames assays.
Casein and casein peptides are reported to have antimutagenic properties, and animal models for colon and mammary tumorigenesis have shown that Whey Protein and Hydrolyzed Whey Protein suppressed tumor development. The tumor suppression observed in studies with Hydrolyzed Whey Protein have been attributed to the high content of cystine/cysteine and γ-glutamylcyst(e)ine dipeptides in the milk proteins, which are efficient substrates for synthesizing glutathione, an important cellular antioxidant.
Bovine milk protein is a major food allergen that can produce Type 1 reactions in sensitized individuals, especially children. The IgE-mediated reaction may include cutaneous, respiratory, and gastrointestinal reactions that may, on rare occasions, result in systemic anaphylaxis. While the reactions may be to any of the proteins found in milk, reactions are most commonly linked to α-lactalbumin, β-lactoglobulin, and casein.
Hydrolyzed Milk Protein (concentration not reported) was negative in an in vitro dermal irritation assay. Hydrolyzed Milk Protein was not irritating to rabbits or humans when tested at up to 25% and 5%, respectively. Hydrolyzed Casein (MW = 600 Da) was not irritating to humans when tested in a 30% solution in water.
No dermal sensitization was observed in a guinea pig maximization study of Hydrolyzed Milk Protein at up to 100%. No sensitization was observed in a study of Hydrolyzed Milk Protein in sensitized patients (concentration not reported). Hydrolyzed Casein (MW = 600 Da) was not sensitizing in a HRIPT when tested in a 30% solution in water.
Hydrolyzed Milk Protein was not a photoirritant or a photosensitizer in human subjects when tested at 5%.
No ocular irritation was predicted to Hydrolyzed Milk Protein (concentration not reported) or Hydrolyzed Casein (1.5% active ingredient) in vitro assays. Hydrolyzed Milk Protein was not irritating to rabbit eyes when tested at up to 25%.
No adverse effects from cosmetic use of milk protein or protein-derived ingredients were discovered in the published literature.
Discussion
The bovine milk protein ingredients in this assessment are found in foods, and daily exposures from the consumption of foods can be expected to yield much larger systemic exposures to these ingredients than those from use in cosmetic products. Bovine milk and bovine milk proteins are generally recognized as safe (GRAS) in foods and animal feeds. The Panel did acknowledge that bovine milk proteins are known food allergens that can elicit Type I hypersensitivity reactions when ingested by sensitized individuals. However, no relevant ocular irritation and no dermal irritation or sensitization were reported in animals or human subjects, and no reported cases of Type I hypersensitivity reactions from cosmetic use were found in the published literature. Additionally, according to their collective knowledge in treating patients with Type 1 hypersensitivity, the Panel clinicians have not experienced responses to bovine milk protein via dermal exposures. Thus, the Panel was not concerned that Type I reactions would be induced by dermal exposure to bovine milk proteins in cosmetics.
The Panel noted that Sodium Caseinate has use concentrations reported up to 96.9%; however, this concentration is in bath oils, tablets, and salts, which are diluted in water prior to use. In leave-on products, the maximum concentration of use reported in the Casein-derived ingredients is 2%. Safety test data of Hydrolyzed Casein were negative at up to 30%. Because of these factors, the Panel was not concerned with the use of Sodium Caseinate at such a high concentration in bath products that are intended to be diluted for use.
The Panel discussed the issue of incidental inhalation exposure in spray deodorants, hair sprays, face powders, face and neck sprays, body and hand sprays, and fragrances. There were no inhalation toxicity data available. Although the Panel noted that droplets/particles from spray and loose-powder cosmetic products would not be respirable to any appreciable amount, the potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at the CIR website http://www.cir-safety.org/cir-findings.
Conclusion

*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
Burnett, C.L. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Bergfeld, W.F., Belsito, D.V., Hill, R.A., Klaassen, C.D., Liebler, D.C., Marks, J.G., Shank, R.C., Slaga, T.J., and Snyder, P.W. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Heldreth, B. contributed to design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.