Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of Hydrofluorocarbon 152a, which functions as a propellant in personal care products. The Panel reviewed relevant data provided in this safety assessment, and concluded that Hydrofluorocarbon 152a is safe in the present practices of use and concentration described in this safety assessment.
Introduction
Hydrofluorocarbon 152a is a gas that functions as a propellant in personal care products, according to the International Cosmetic Dictionary and Handbook. 1 It is commonly known as 1,1-difluoroethane.
Chemistry
Definition
Hydrofluorocarbon 152a (CAS No. 75-37-6) is the halocarbon, 1,1-difluoroethane, that conforms to the formula CH3CHF2 (Figure 1).
1
Hydrofluorocarbon 152a.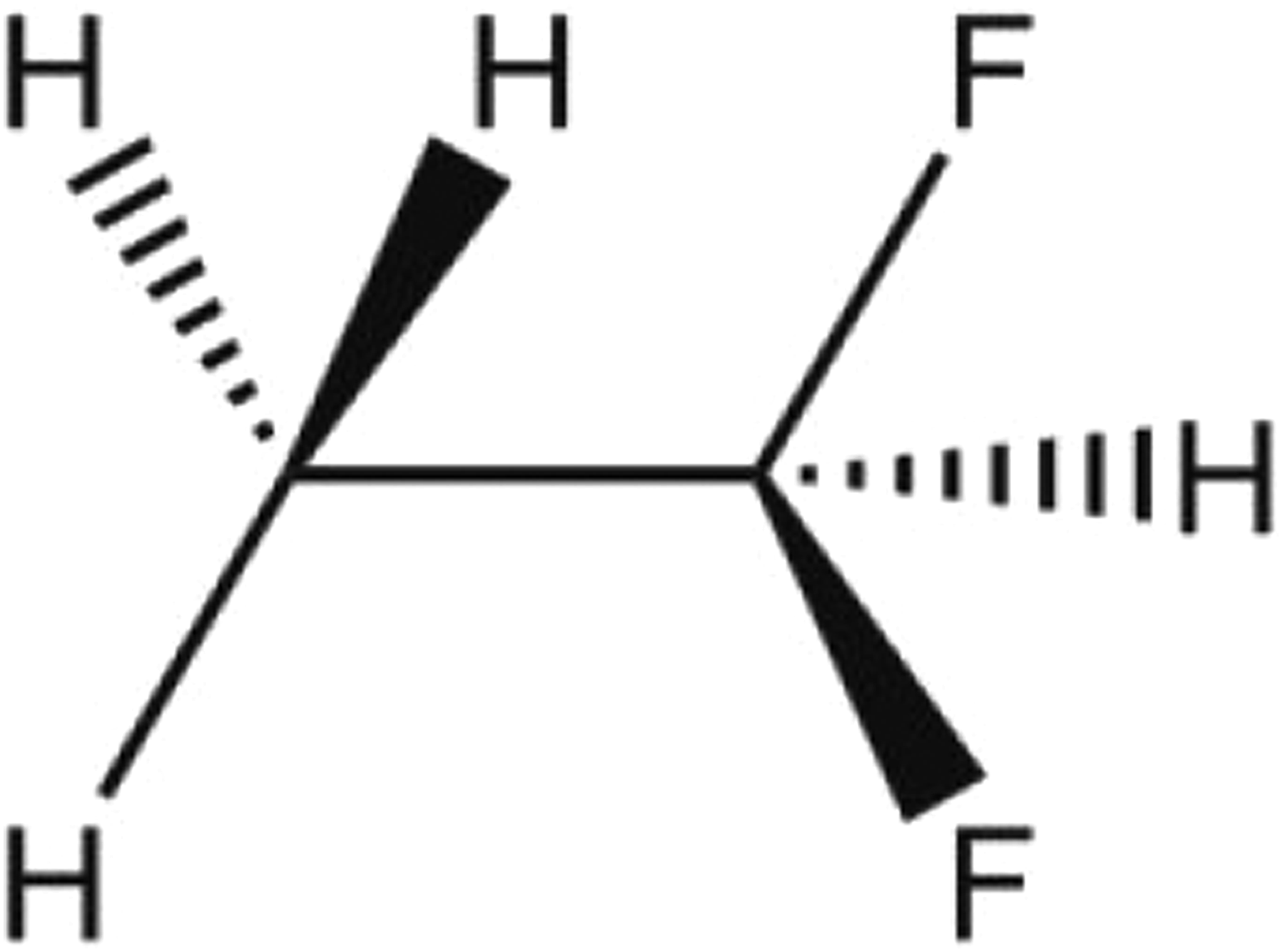
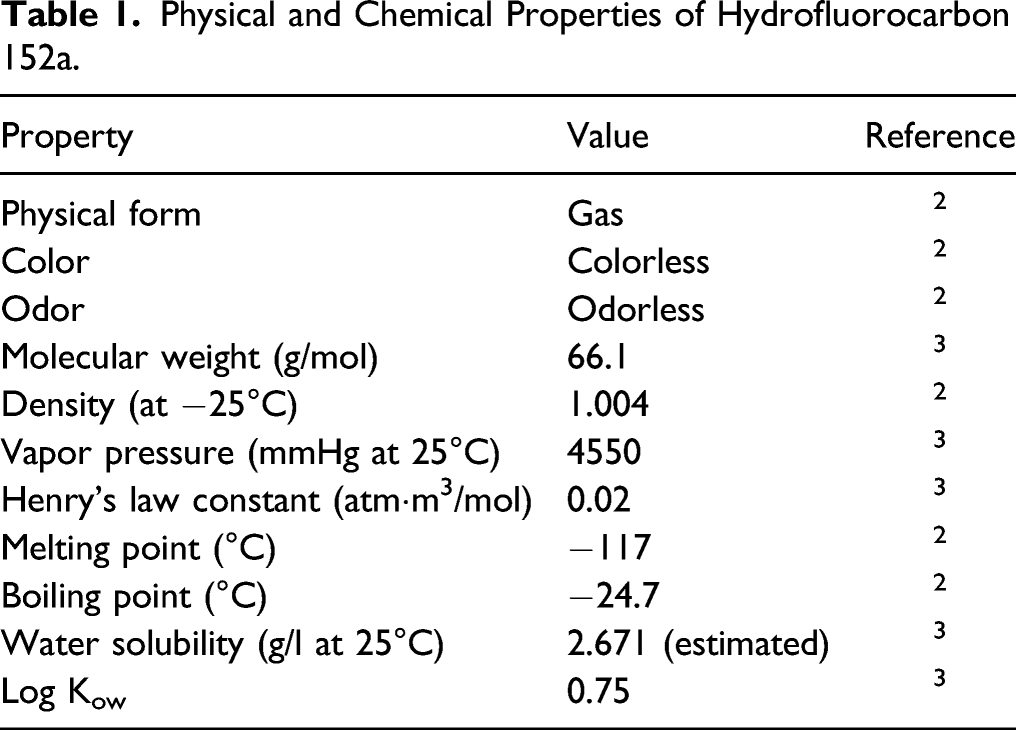
Chemical and Physical Properties
Physical and Chemical Properties of Hydrofluorocarbon 152a.
Method of Manufacturing
Hydrofluorocarbon 152a may be derived by reacting hydrogen fluoride with acetylene. 2 The material may also be produced by a catalytic reaction of vinyl chloride with hydrofluoric acid in a closed system.3
Impurities
Hydrofluorocarbon 152a is reported to be greater than 99.9% pure. 3 Impurities may include water, residual hydrochloric acid, and residual hydrofluoric acid.
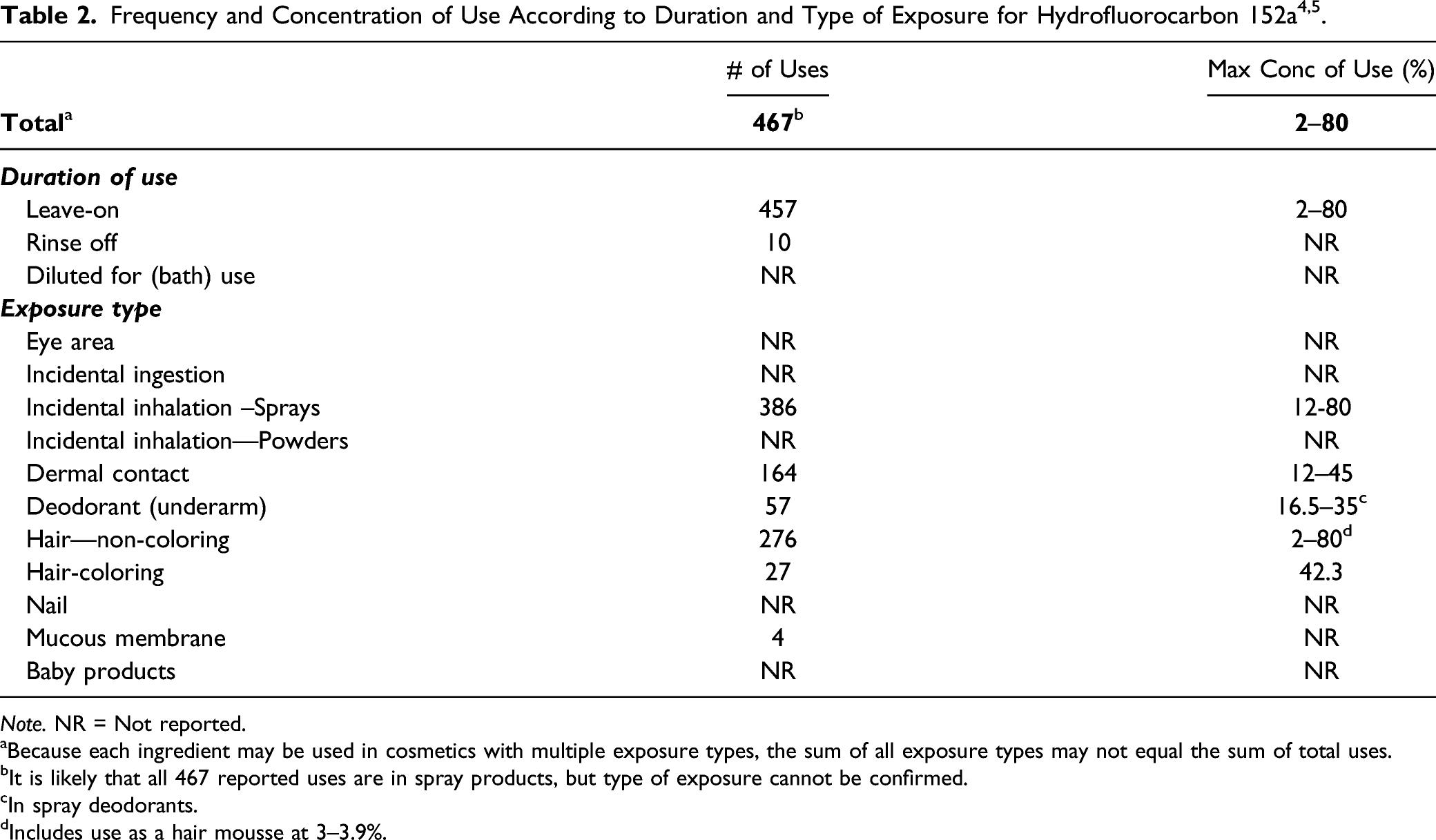
Use
Cosmetic
The safety of the cosmetic ingredient included in this assessment is evaluated based on data received from the U.S. Food and Drug Administration (FDA) and the cosmetics industry on the expected use of this ingredient in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by Industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
Note. NR = Not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIt is likely that all 467 reported uses are in spray products, but type of exposure cannot be confirmed.
cIn spray deodorants.
dIncludes use as a hair mousse at 3–3.9%.
This product is believed to be solely used in spray products, like hair spray and spray deodorants. However, because this ingredient is a gas under all exposure conditions, inhalation is possible for all product types in which it is used.
Based on environmental regulations that include a requirement that those products containing fluorocarbons be labeled, Hydrofluorocarbon 152a is not used in personal hygiene or household products in Europe.6-8
Non-Cosmetic
Hydrofluorocarbon 152a may be used as an aerosol propellant, a foam expansion agent, a refrigerant, and as a catalyst regenerator. 3
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion (ADME)
Animal
Inhalation
Metabolites included fluoride ion and a trace of acetyl fluoride in urine collected for nuclear magnetic resonance (NMR) spectroscopy analysis from male CD rats exposed via inhalation to 3000 ppm Hydrofluorocarbon 152a (see Acute Toxicity for further study details). 9 No fluoroacetate was detected. The time at which the urine was collected after exposure was not identified in the study report.
Human
Inhalation
The uptake, distribution, and elimination of Hydrofluorocarbon 152a were studied in male and female subjects. 10 6 women and 4 men completed all exposure sessions (0, 200, and 1000 ppm test material), with 1 additional male exposed to 0 and 200 ppm, another male exposed to only 200 ppm, and another male exposed only to 0 ppm (n for 0 ppm = 12, n for 200 ppm = 12, and n for 1000 ppm = 10). Subjects were exposed for 2 h on 3 separate occasions to Hydrofluorocarbon 152a, and exposures were performed during light exercise on computer-controlled ergometer bicycles in an exposure chamber (20 m3) with a controlled climate. The concentration of the test material in the chamber was checked by gas chromatography (GC) at 5 minute intervals throughout the exposure sessions. Mixed exhaled air was collected once before the exposure, 5 times during the exposure, and 7 times after the exposure. Pulmonary ventilation was recorded with an electric spirometer during every breath sampling period. Venous blood was collected from the brachial vein prior to exposure and at 3 h and 22 h after exposure for analysis of inflammatory markers, while arterialized capillary blood was collected from the subjects’ finger tips before, during, and after exposure. Urine was sampled once before exposure and at 2, 4, and 6 h after onset of exposure, as well as twice in the evening and once the following morning. The presence of Hydrofluorocarbon 152a in the blood and urine was analyzed by head-space GC. The urine was analyzed for fluoride with an ion selective electrode and for potential metabolites with NMR.
In the blood, initial increases in Hydrofluorocarbon 152a were fast, and average concentrations of 7.4 μM (for 200 ppm) and 34.3 μM (for 1000 ppm) were achieved within a few minutes of exposure. Within 4 h post-exposure, the concentration was less than 1% of the steady-state level. Blood concentrations were below detection limits 22 h post-exposure. The area under the curve (AUC) of the test material in blood was 1042 μM·min at 200 ppm and 4572 μM·min at 1000 ppm, which indicated dose-proportional kinetics. No exposure-related effects were observed in inflammatory markers in the blood plasma. Total inhaled Hydrofluorocarbon 152a was approximately 20.6 and 99.6 mmol for the 200 and 1000 ppm exposures, respectively. Post-exposure decreases of the test material in exhaled air and urine were similar to that in blood. The AUCs of the test material in urine were 190 μM·min for 200 ppm and 1271 μM·min for 1000 ppm. After exposure to 200 ppm and 1000 ppm Hydrofluorocarbon 152a, about 0.004% and 0.009%, respectively, of the total amount inhaled was excreted in the urine within 23 h. About 20 μmol excess fluoride (0.013% of inhaled) was excreted in urine following exposure to 1000 ppm test material when compared to the control. This was statistically significantly greater than the amount excreted in the urine of both the control and the 200 ppm exposed subjects (P = 0.008), and urinary fluoride excretion remained significantly elevated in the post hoc test (P < .05). Fluoride excretion rate varied; however, it was statistically significantly higher in the first 2 urine samples after exposure to 1000 ppm test material when compared to the control subjects and subjects exposed to 200 ppm (P = .0004). No fluorine-containing metabolites could be detected in the urine, indicating biotransformation of the test material in humans was very low. 10
Toxicological Studies
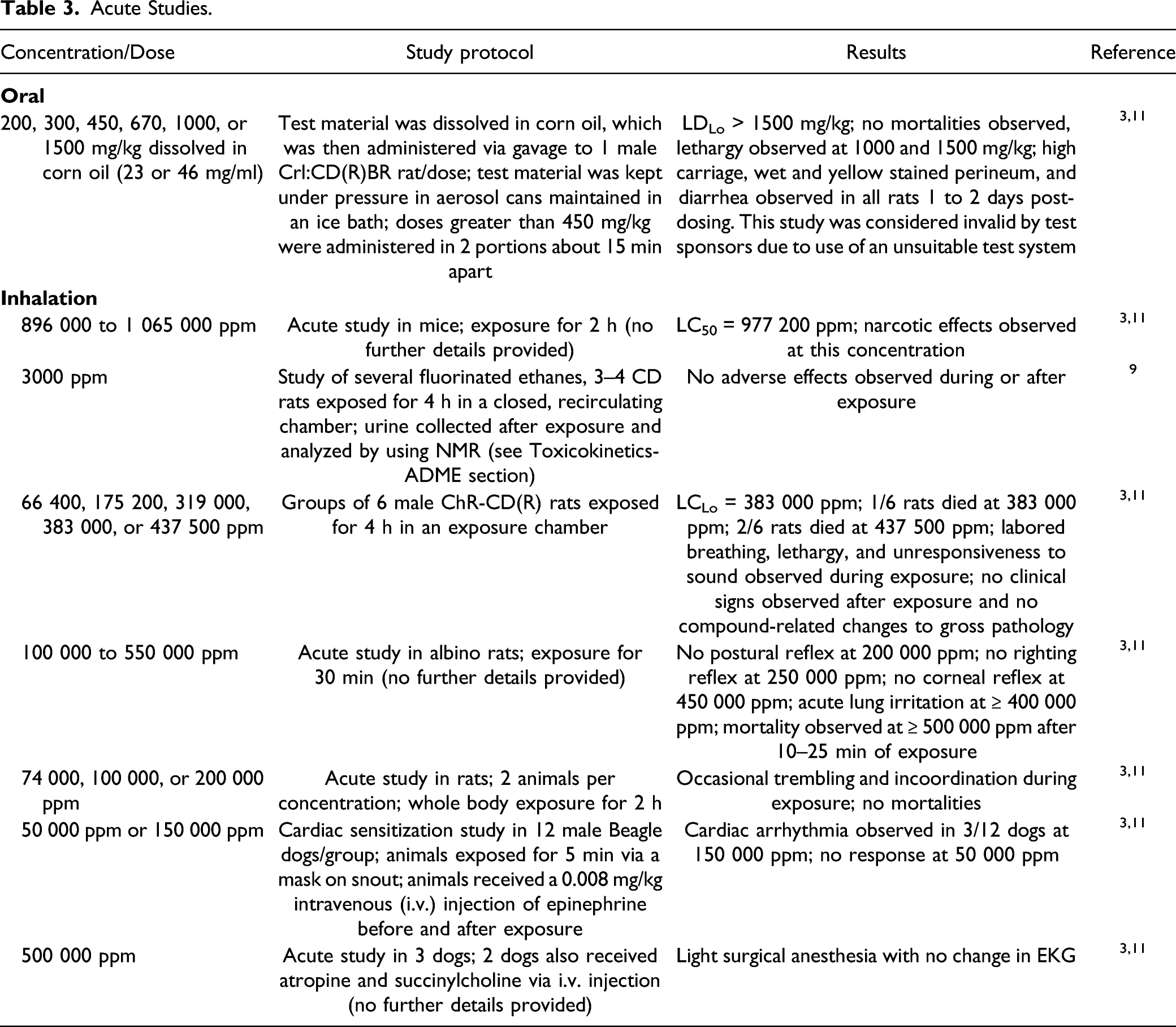
Acute Toxicity Studies
Acute Studies.
Short-Term Toxicity Studies
Inhalation
In a short-term inhalation study of Hydrofluorocarbon 152a, 10 male ChR-CD rats were exposed to 100 000 ppm of the test material in air 6 h/day, 5 days/week, for 2 weeks (exposure method not reported).3,11 Following the final exposure, 5 rats were killed for gross and histopathologic examination while the remaining rats were killed after a 14-d recovery period. Hematological, urine analytical, and biochemical indices were measured in all rats before they were killed. During exposure to the test material, the rats appeared to be anesthetized, which was indicated by sleep and unresponsiveness to sound. No other adverse effects were observed. A slight increase in urinary fluoride was observed following the final exposure.
In another short-term study, 8 albino rats (sex not specified) were exposed to 100 000 ppm Hydrofluorocarbon 152a in air 16 h/d for 2 months (exposure method not reported).3,11 At the end of the exposure period, the animals were killed and examined for gross pathological changes. Lung and liver sections were examined microscopically. During the exposure period, no clinical signs of toxicity were observed. Necropsy indicated no adverse changes. Mild diffuse infiltration of small and large round cells in the lung was observed during microscopic examination, which indicated mild chronic irritation.
Chronic Toxicity Studies
See Carcinogenicity section below.
Developmental and Reproductive Toxicity Studies
The maternal and developmental toxicity of Hydrofluorocarbon 152a was investigated in mated CD rats.3,11 Groups of 27 female rats were exposed 6 h/d on gestation days 6–15 via whole body inhalation to 0, 5000, or 50 000 ppm. The dams were exposed to the test material in 1.4 m3 stainless steel and glass chambers under dynamic airflow conditions. The animals were observed daily for signs of toxicity and weighed periodically throughout the study. The dams were killed on gestation day 21, and organs of the thoracic and abdominal cavities and the fetuses were examined.
No treatment-related clinical signs of toxicity or body weight changes were observed in the dams. No statistically significant differences were observed in the numbers of corpora lutea, implantations, resorptions or live fetuses, fetal weight, or crown-rump length between the treatment groups and the controls. The pregnancy ratios at 0, 5000, and 50 000 ppm were 22/27, 21/27, and 19/27, respectively. No gross pathological abnormalities were observed in ovaries, uterine horns, vital organs, or tissues of the treated animals. External, skeletal, and internal examinations of fetuses revealed no evidence of teratogenicity. The no observed effect level (NOEL) for maternal and developmental toxicity in rats was 50 000 ppm, which was the highest concentration tested in this study.3,11
Genotoxicity Studies
Genotoxicity Studies.
Carcinogenicity Studies
In a 2-year inhalation study, male and female Crl:CD(R)Br rats were exposed to 0, 2000, 10 000, or 25 000 ppm Hydrofluorocarbon 152a in air 6 h/d, 5 d/wk.3,11 There were 30 rats/sex in each exposure group, and rats were 54 d old at the first exposure. Rats were exposed whole body to the test material in chambers. Body weights were recorded twice monthly for the first 14 weeks and then once a month for the rest of the study. Animals were observed for clinical signs of toxicity twice daily during the work week while animals were observed daily for mortality on the weekends and holidays. Ten rats/sex/dose underwent clinical pathology evaluation at 1, 3, 6, 12, 18, and 24 month, which included hematology and clinical chemistry studies. Urine was collected and analyzed the day prior to blood collection. Ten rats/sex/dose were killed and necropsied at 3 and 12 months and all remaining surviving animals were killed and necropsied at 24 mo. Gross examinations were performed on all rats and select tissues underwent microscopic examination. Organ weights were recorded and histopathological examinations were conducted on the control and high-dose groups and on any animals that died during the study. Kidney and nasal tissues at the 3 and 24 months killings were evaluated from all low- and mid-dose groups.
During the study, no statistically significant differences in body weights or body weight gains were observed. Clinical signs of toxicity observed included ocular/nasal discharge, wet/stained perinea, stained body/face, and/or swollen ears. These clinical signs were also observed in some control animals. Clinical chemistry effects included increased mean corpuscular volumes, increased serum bilirubin, increased hematocrits, and/or increased urobilinogen. Because there were no abnormalities in hematopoietic tissues or red blood cells or changes in serum bilirubin, there was no conclusive evidence of a hemolytic effect. A decrease in peripheral circulating eosinophils and/or monocytes was observed. A dose-dependent increase was observed in urinary fluoride concentration, but there was no evidence of fluorosis. An increase in serum creatinine and urine volume and a decrease in urine osmolality were observed in female rats. Upon study conclusion, no treatment-related differences in organ weights were observed in male rats, but significant increases in absolute and relative lung weights, absolute and relative stomach weights, relative heart weight, and relative liver weight were observed in female rats at all concentrations. The biological significance of these observations in the female rats is unknown. No treatment-related tumors were observed in male and female rats. The authors of the study concluded that Hydrofluorocarbon 152a was not carcinogenic and did not produce life-shortening toxic effects in rats in this 2-year inhalation study.3,11
Dermal Irritation and Sensitization
Dermal Irritation
No relevant published dermal irritation studies on Hydrofluorocarbon 152a were identified in a literature search for this ingredient, and no unpublished data were submitted. These studies are considered technically not feasible for gases. 11
Dermal Sensitization
No relevant published dermal sensitization studies on Hydrofluorocarbon 152a were identified in a literature search for this ingredient, and no unpublished data were submitted. These studies are considered technically not feasible for gases. 11
Ocular Irritation Studies
A hair spray containing 80% Hydrofluorocarbon 152a was considered not irritating to the eye in a bovine corneal permeability (BCOP) assay. 13 The assay was performed based on methods described in Organization for Economic Co-Operation and Development (OECD) test guideline 437. The test material, the positive control (ethanol), and the negative control (sterile deionized water) were applied via aerosol sprays at 1 second burst from a distance of 10 cm. The mean amount of the test material sprayed on each cornea was 0.9 ± 0.19 g. Four to five corneas each were treated with each test article. The in vitro score was 2.3 for the test material while it was 48.4 for the positive control.
In the two-year inhalation study in rats described above, clinical signs of toxicity included ocular/nasal discharge.3,11 No further details were provided.
Clinical Studies
Numerous case reports of adverse events from abusive inhalation of products containing Hydrofluorocarbon 152a have been described in the literature. Adverse events include death, cardiomyopathy, cardiac arrhythmia and other cardiac and respiratory effects, rhabdomyolysis, fulminant hepatitis, acute kidney injury, angioedema, frostbite, chemical burns, and even thermal burns.14-18
Risk Assessment
The American Industrial Hygiene Association (AIHA) 8 hour workplace environmental exposure limit for Hydrofluorocarbon 152a is 1000 ppm. 19
The U.S. Environmental Protection Agency’s (EPA) Integrated Risk Information System (IRIS) has estimated the reference concentration for chronic inhalation exposure (RfC) for Hydrofluorocarbon 152a to be 40 mg/m3, which was calculated based on an uncertainty factor of 300, a modifying factor of 1, and a no observed adverse effect level (NOAEL) value of 67 500 mg/m3 (25 000 ppm) based on the value obtained from the carcinogenicity study described above. 20
Summary
Hydrofluorocarbon 152a, commonly known as 1,1-difluoroethane, is a gas that functions as a propellant in personal care products.
According to 2017 VCRP data, Hydrofluorocarbon 152a is used in 467 formulations; the majority of uses are in leave-on hair care products. The results of the concentration of use survey conducted in 2015 by the Council indicate the highest reported maximum concentration of use to be 80% in hair sprays.
Hydrofluorocarbon 152a may be used as an aerosol propellant, a foam expansion agent, a refrigerant, and as a catalyst regenerator.
Metabolites identified in urine collected from male rats exposed via inhalation to 3000 ppm Hydrofluorocarbon 152a included fluoride ion and a trace of acetyl fluoride.
In an uptake, distribution, and elimination inhalation study of Hydrofluorocarbon 152a in human subjects exposed to 0, 200, or 1000 ppm, initial increases of Hydrofluorocarbon 152a in the blood were fast, and within 4 h post-exposure, the concentration was less than 1% of the steady-state level. Blood concentrations were below detection limits 22 h post-exposure. Total inhaled Hydrofluorocarbon 152a was approximately 20.6 and 99.6 mmol for the 200 and 1000 ppm exposures, respectively. Post-exposure decreases of the test material in exhaled air and urine were similar to that in blood. After exposure to 200 ppm and 1000 ppm Hydrofluorocarbon 152a, about 0.004% and 0.009%, respectively, of the total amount inhaled was excreted in the urine within 23 h. Fluoride excretion rate was varied; however, it was significantly higher in the first 2 urine samples after exposure to 1000 ppm test material when compared to the control and 200 ppm exposure. No fluorine-containing metabolites could be detected in the urine, indicating biotransformation of the test material in humans was very low.
In a rat oral dose study, the LDLo for Hydrofluorocarbon 152a was reported to be greater than 1500 mg/kg (this study was considered invalid by test sponsors because an unsuitable test system was used). The reported LC50 of animals in a mouse inhalation study was 977 200 ppm. Cardiac arrhythmia was observed in dogs exposed for 5 min to 150 000 ppm Hydrofluorocarbon 152a in an acute inhalation study.
In a two-week inhalation study of 100 000 ppm Hydrofluorocarbon 152a, rats that received the test material 6 h/d for 5 d/wk appeared to be anesthetized, which was indicated by sleep and unresponsiveness to sound. No other adverse effects were observed. A slight increase in urinary fluoride was observed following the final exposure. In another short-term inhalation study, rats exposed to 100 000 ppm Hydrofluorocarbon 152a for 16 h/d had no clinical signs of toxicity during 2 months of exposure. Necropsy indicated no adverse changes. Mild diffuse infiltration of small and large round cells in the lung was observed during microscopic examination, which indicated mild chronic irritation.
No treatment-related clinical signs of toxicity or body weight changes were observed in the dams (rats) of a maternal and developmental toxicity study that were exposed to up to 50 000 ppm Hydrofluorocarbon 152a in air 6 h/d on gestation days 6–15 in rats. No statistically significant differences between the control group and test groups were observed in pregnancy or fetal parameters. The NOEL for maternal and developmental toxicity in rats was 50 000 ppm.
Hydrofluorocarbon 152a was not mutagenic in Ames tests at concentrations up to 75%, but it was weakly clastogenic at 60% and 70% (19 h exposure without metabolic activation) in a chromosomal aberration test in human lymphocytes. In a rat micronucleus assay, Hydrofluorocarbon 152a was not genotoxic at concentrations up to 19 500 ppm.
The authors of a two-year inhalation study of rats exposed to concentrations up 25 000 ppm Hydrofluorocarbon 152a for 6 h/d, 5 d/wk concluded that this chemical was not carcinogenic and did not produce life-shortening toxic effects.
A hair spray containing 80% Hydrofluorocarbon 152a was considered not irritating to the eye in a BCOP assay.
Numerous case reports of adverse events from abusive inhalation of products containing Hydrofluorocarbon 152a have been described in the literature. Adverse events include death, cardiomyopathy, cardiac arrhythmia and other cardiac and respiratory effects, rhabdomyolysis, fulminant hepatitis, acute kidney injury, angioedema, frostbite, chemical burns, and even thermal burns.
The AIHA 8 hour workplace environmental exposure limit for Hydrofluorocarbon 152a is 1000 ppm. The EPA’s IRIS has estimated the RfC for chronic inhalation exposure for Hydrofluorocarbon 152a to be 40 mg/m3.
Dermal irritation and sensitization studies are considered technically not feasible for gases, including Hydrofluorocarbon 152a.
Discussion
Hydrofluorocarbon 152a is a largely inert gas that is rapidly volatilized and dispersed upon application and, if incidentally inhaled and absorbed into the blood stream, is quickly cleared from the body by exhalation. Significant dermal exposures to this ingredient and potential effects during normal use of cosmetic spray products are unlikely because of the chemical and physical properties of the ingredient. The available evidence indicates that Hydrofluorocarbon 152a is not metabolized in the body to any significant extent. Extensive inhalation exposure studies of Hydrofluorocarbon 152a indicate that this ingredient in cosmetic spray products will not cause adverse health effects when these products are used as intended. These studies include acute, subchronic, and chronic studies at concentrations orders of magnitude greater than the occupational exposure limit for this compound and greater still than concentrations that can reasonably be expected during cosmetic use. The Panel found the overall safety profile of this ingredient to be favorable.
The Panel noted that the European Union has issued regulations restricting the use of fluorinated gases in personal care and household products. The regulations are directed toward protection of the global environment, which falls outside of the Panel’s purview of personal use safety.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that Hydrofluorocarbon 152a is safe in the present practices of use and concentration described in this safety assessment.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contribution
Burnett, C. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., Snyder, P., and Gill, L.J. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Heldreth, B. contributed to design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.