Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of 12 Rosa canina-derived ingredients, which are reported to function as skin conditioning agents, fragrance ingredients, cosmetic astringents, anti-acne agents, abrasives, humectants, and exfoliants in cosmetic products. Because final product formulations may contain multiple botanicals, each containing the same constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching cumulative levels that may be hazardous to consumers. The Panel reviewed relevant data relating to the safety of these ingredients and concluded that these ingredients are safe in the present practices of use and concentration described in this safety assessment when formulated to be non-irritating and non-sensitizing.
Keywords
Introduction
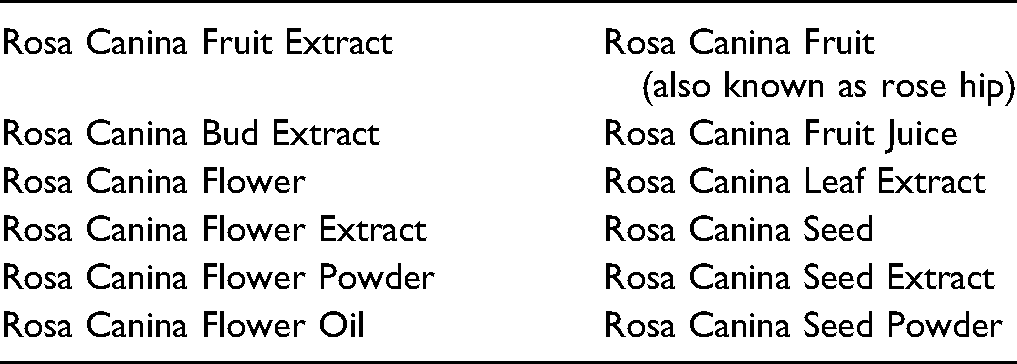
Definitions and Reported Functions of the Ingredients in This Safety Assessment. 1
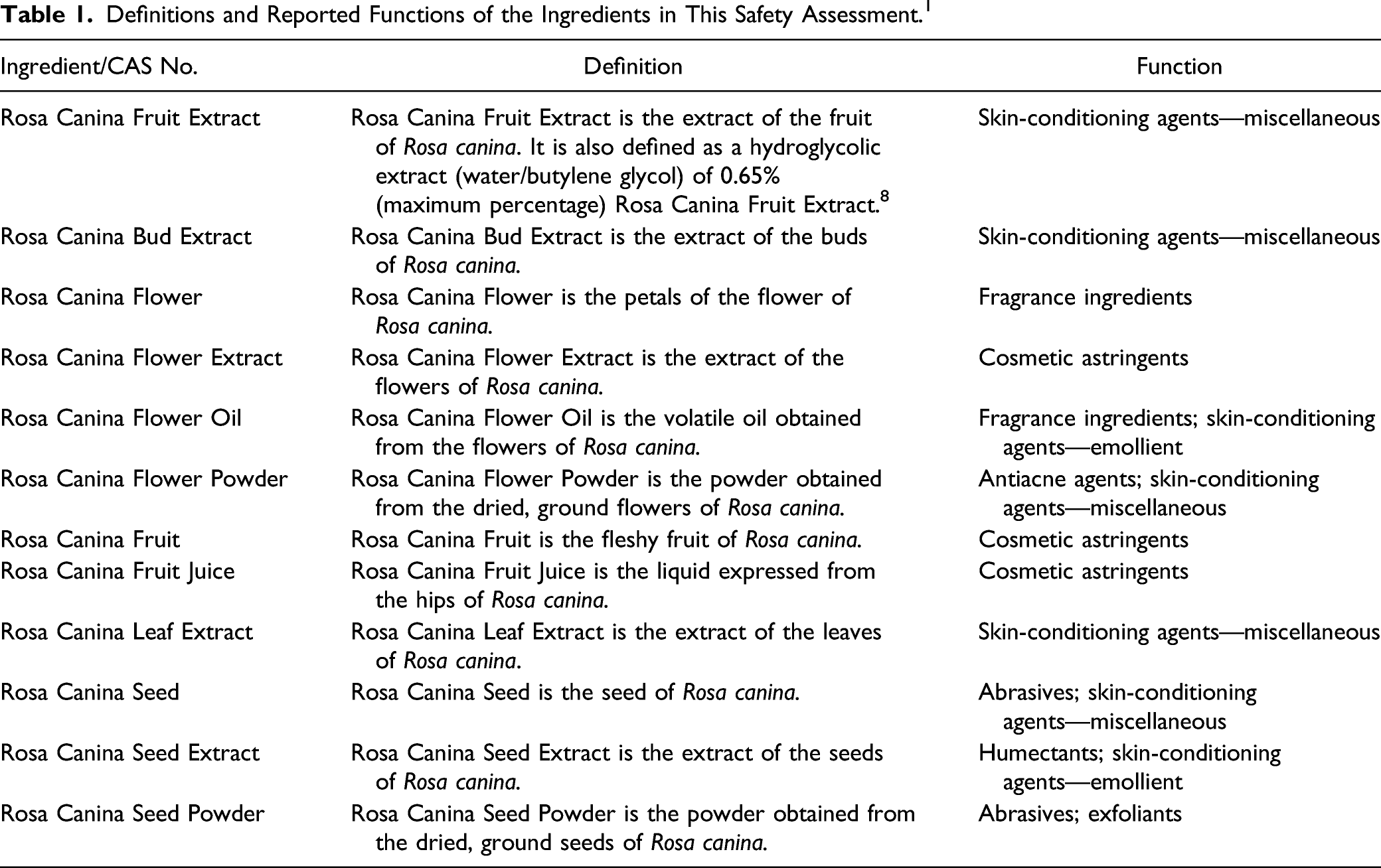
The Panel evaluated the safety of Rosa Canina Fruit Oil and other plant-derived fatty acid oils in cosmetics, and issued a final report in 2011 with the conclusion that these oils are safe in the present practices of use and concentration. 2 The Panel has also evaluated the safety of butylene glycol, which can be a major component of Rosa Canina Fruit Extract, in cosmetics and issued a final report in 1985 with the conclusion that butylene glycol, hexylene glycol, ethoxydiglycol, and dipropylene glycol are safe as presently used in cosmetics. 3 This conclusion was reaffirmed by the Panel in a 2006 publication. 4
Chemistry
Plant Identification
Rosa canina (also known as dog rose) is an herb that belongs to the Rosaceae family, and is among the plants growing in Northeastern Portugal and in the Hadim, Taskent, and Ermenek regions of Turkey.5,6 Rose hip is a common name for the dried fruit of Rosa canina. The definitions of Rosa canina-derived ingredients are presented in Table 1.
Chemical and Physical Properties
Rosa Canina Fruit Extract
Using ultraviolet spectrophotometry, the λmax for Rosa Canina Fruit Extract (ethanol extract) has been reported at ∼ 280 nm. 7
Method of Manufacture
Rosa Canina Fruit Extract
The part of the Rosa canina plant that is used to manufacture Rosa Canina Fruit Extract is the fruit without achene. Key steps in the manufacture of Rosa Canina Fruit Extract include: (1) solubilization of Rosa canina powder produced from the fruit without achene in a mixture of water and butylene glycol, (2) separation of soluble and insoluble phases, (3) clarification by filtration, (4) decoloration, and (5) filtrations and sterilizing filtration. 8
The method of manufacture of Rosa Canina Fruit Extract (ethanol extract) has been described as follows:
7
Dried raw material → extract with 50 vol% ethanolic solution → concentration → adjustment → sedimentation → filtrate → adjustment → packaging A description of the method of manufacture of Rosa Canina Fruit Extract (butylene glycol extract) is included below.
7
Dried raw material → extract with 1,3-butylene glycol → filtrate → sedimentation → filtrate → adjustment → packaging
Further details relating to this method of manufacture were not provided.
Composition
Rosa Canina Fruit Extract
According to one manufacturer, a tradename mixture comprising in part, Rosa Canina Fruit Extract, consists of 0.65% (maximum percentage) Rosa Canina Fruit Extract. 8 Composition data on this tradename mixture are as follows: Rosa Canina Fruit Extract (maximum percentages: 0.45% to 0.65%), butylene glycol (maximum percentages: 76.50% to 93.50%), and water (maximum percentages: 5.85% to 23.05%). Additional data relating to the composition of the dried matter of Rosa Canina Fruit Extract are: sugars (90%), mineral ashes (9% as residue following pyrolysis), and polyphenols (1%).
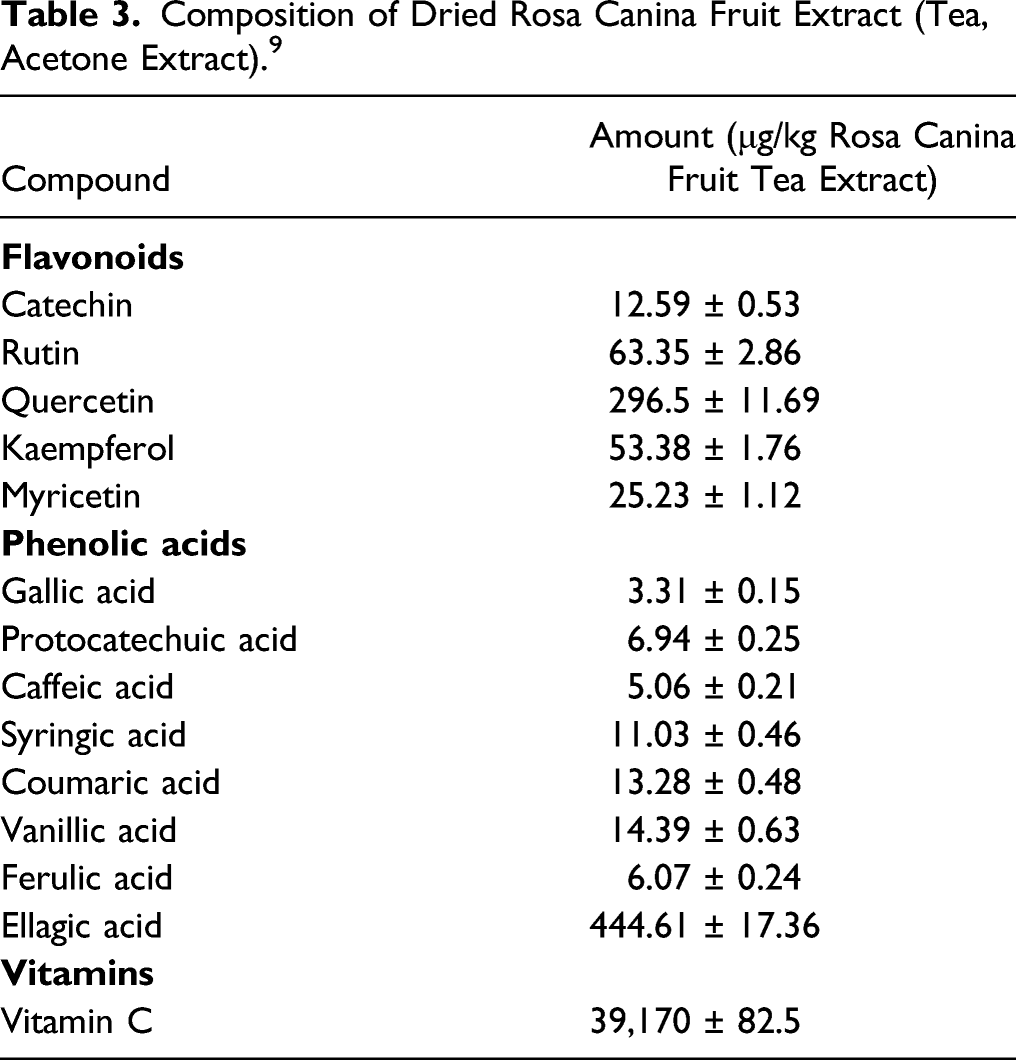
Rosa Canina Fruit Extract (ethanol extract or butylene glycol extract) has flavonoid and tannin components, most prominent of which is the glycoside formed from the flavonoid quercetin, namely quercetrin. 7 The highest concentration phenolic acid found in Rosa Canina Fruit Extract is ellagic acid. 9
Content of Some Phenolic Acids and Flavonoids in Rosa Canina Fruit Extracts. 10
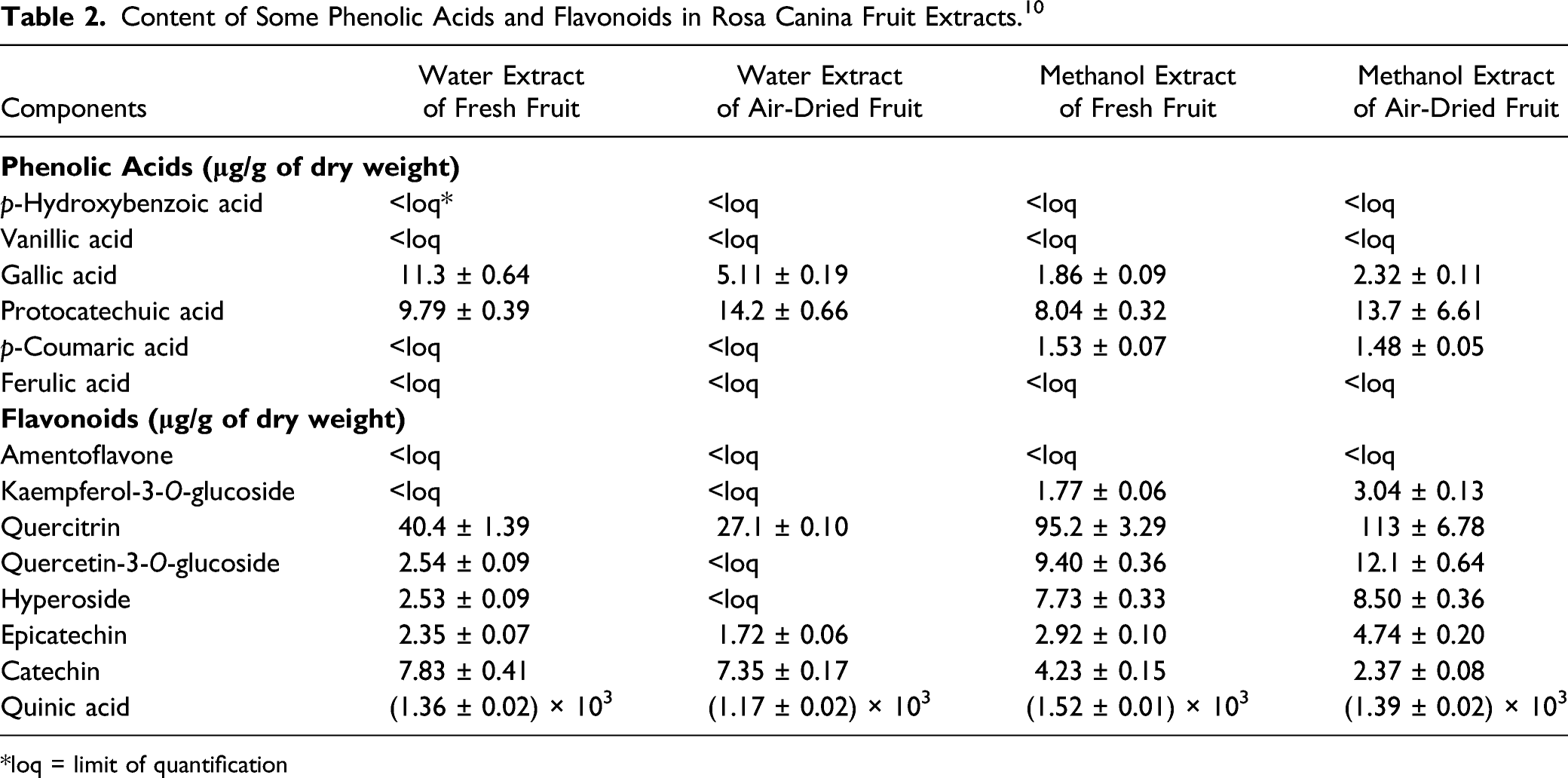
*loq = limit of quantification
Composition of Dried Rosa Canina Fruit Extract (Tea, Acetone Extract). 9
Rosa Canina Fruit
The fruits of Rosa canina contain phenolic acids, proanthocyanidins, tannins, flavonoids, fatty acids, pectins, carotenoids, and fruit acids (ascorbic acid, malic acid, and citric acid). 11 (+)-Catechin, a flavonoid, has been identified as the most abundant flavan-3-ol (3.59 mg/100 g) in Rosa Canina Fruit, 5 and the abundance of ascorbic acid (vitamin C, 880 mg/100 mL) in Rosa Canina Fruit has also been noted.12,13
In addition to vitamin C, the following other nutrients in Rosa Canina Fruit have been reported: carotenoids, tocopherol, bioflavonoids, tannins, pectin, sugars, organic acids, amino acids, essential oils, phosphorus (P, 4860 ppm), potassium (K: 5467 ppm), calcium (Ca: 2867 ppm), magnesium (Mg: 1254 ppm), iron (Fe: 27 ppm), copper (Cu: 27 ppm), manganese (Mn: 56 ppm), and zinc (Zn: 30 ppm). 12 According to another source, the following 6 main carotenoids have been identified in Rosa Canina Fruit: epimers of neochrome, lutein, zeaxanthin, rubixanthin, lycopene, and β,β-carotene. 14
Nutritional Composition of Wild Rosa Canina Fruit. 15

Rosa Canina Bud Extract
Flavonols such as glycosides of quercetin and kaempferol, hydroxycinnamic acids, and ellagitannins were detected in samples of Rosa Canina Bud Extract, with gallotannins being the main components (up to 1.7 g/L). 17
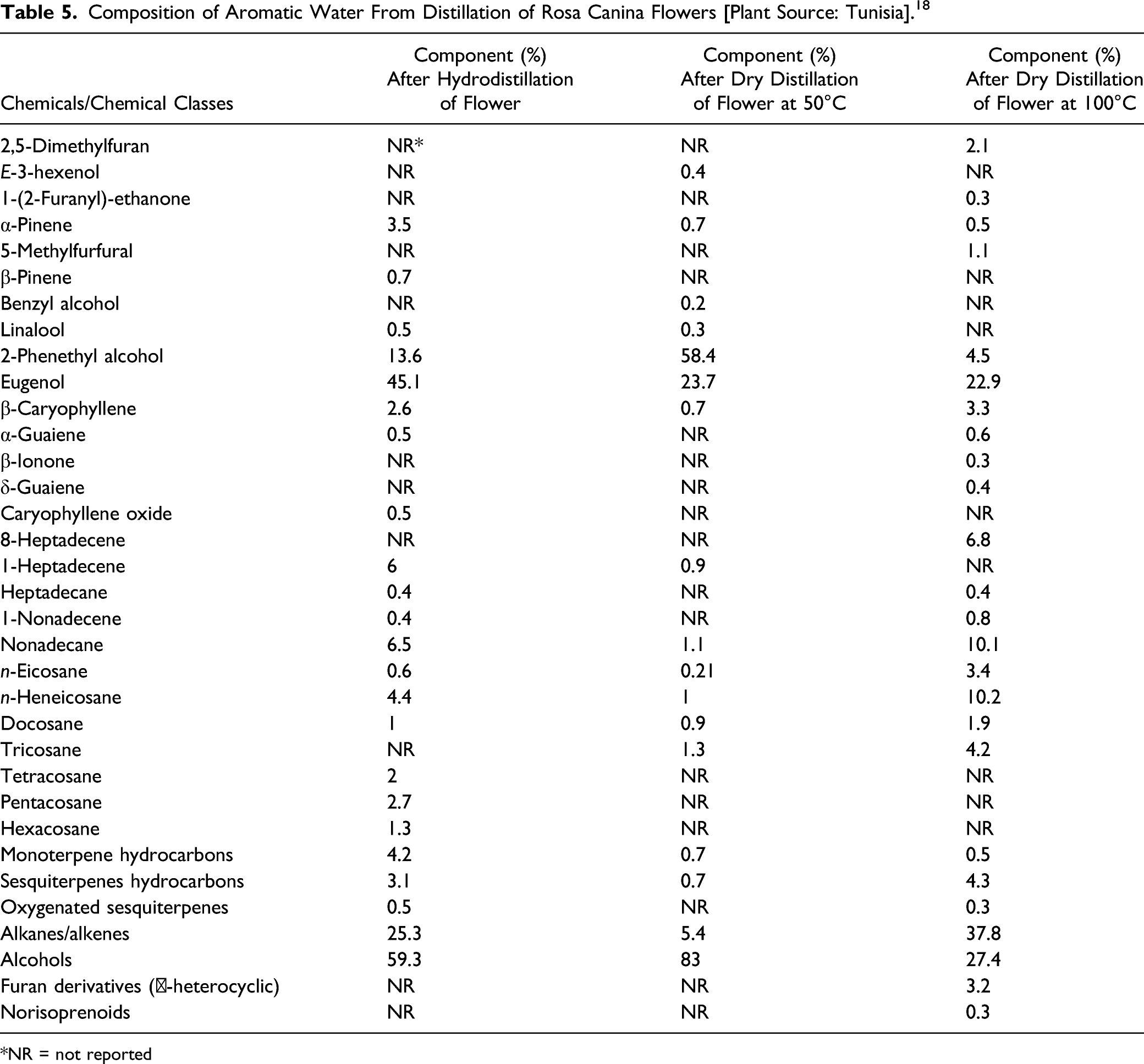
Rosa Canina Flower Extract
Composition of Aromatic Water From Distillation of Rosa Canina Flowers [Plant Source: Tunisia]. 18
*NR = not reported
Rosa Canina Leaf Extract
Composition of Essential Oils From Rosa Canina Leaves in Two Areas of Tunisia. 20

Rosa Canina Seed
*Typical fatty acid composition in seed (%).
Impurities
Rosa Canina Fruit Extract
An impurities analysis of Rosa Canina Fruit Extract for the following components was performed: allergens (26 listed in European Regulation 1223/2009), alkaloids, aflatoxins (B1, B2, G1, and G2), and pesticides. These impurities were not detected; that is, all concentrations were lower than the threshold sensitivity of the method (not specified). 8 A heavy metals analysis of Rosa Canina Fruit Extract indicated no traces of the following: cadmium, chromium, cobalt, mercury, and vanadium. However, traces of antimony, arsenic, nickel, lead, and selenium were found; less than 2 ppm of heavy metals was reported. According to another source, Rosa Canina Fruit Extract (ethanol extract or butylene glycol extract) contains heavy metals (not more than 20 ppm) and arsenic (not more than 2 ppm). 7
Rosa Canina Fruit
Three different brands of tea bag containing dried rose hip were mixed and pulverized and 0.5 g was obtained to determine the presence of various elements. 22 Each of the following 14 elements (mean ± standard deviation mg/kg; n = 5) was present in rose hip powder: calcium (17,596 ± 150), magnesium (1909.2 ± 90), iron (267.2 ± 30), aluminum (157 ± 12), manganese (244 ± 13.8), zinc (21.9 ± 1.2), copper (4.94 ± 0.76), strontium (58.8 ± 2.98), barium (46.8 ± 2.4), nickel (2.90 ± 0.78), chromium (0.92 ± 0.07), cobalt (0.40 ± 0.01), lead (0.34 ± 0.15), and cadmium (0.07 ± 0.01). The following 11 elements (% = percent of mineral originally found in dried rose hips) were present in the rose hip powder infusion (prepared from 0.5 g rose hip in 25 mL water for 30 minutes at 95°C): calcium (5.5%), magnesium (72%), iron (14.4%), aluminum (3.6%), manganese (20%), zinc (28%), copper (60%), strontium (52%), barium (25%), nickel (66%), chromium (27%). Cobalt, lead, and cadmium were not detected in the infusion.
Use
Cosmetic

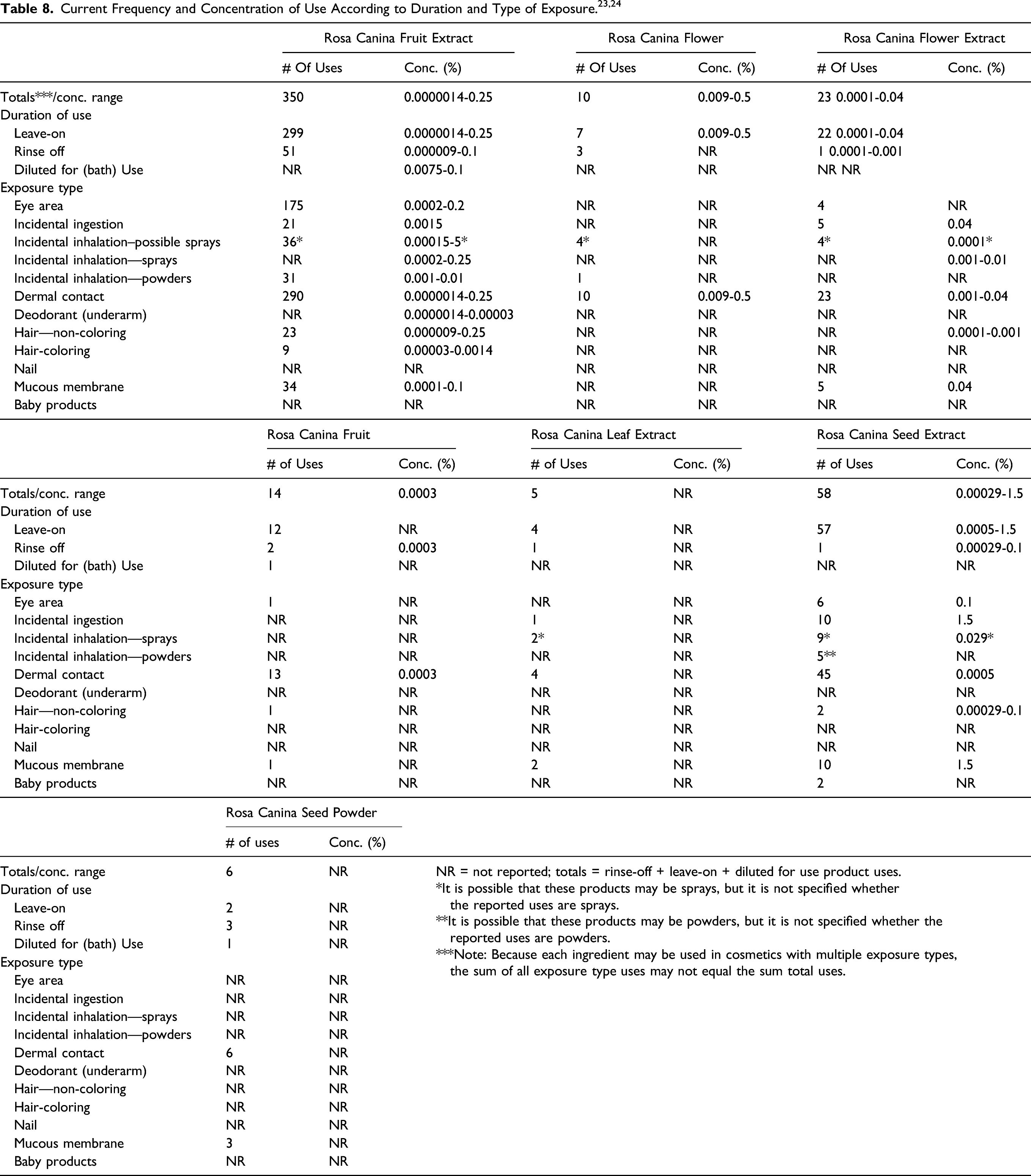
According to 2017 VCRP data, the greatest reported use frequency is for Rosa Canina Fruit Extract (350 formulations, mostly leave-on products), followed by Rosa Canina Seed Extract (58 formulations, mostly leave-on products) (Table 8). 23 The results of a concentration of use survey conducted in 2016 indicate that Rosa Canina Seed Extract has the highest maximum concentration of use; it is used at concentrations up to 1.5% in leave-on products (lipstick) (Table 8). 24 In some cases, reported uses appear in the VCRP database, but concentrations of use data were not provided. For example, according to the VCRP, Rosa Canina Leaf Extract and Rosa Canina Seed Powder are being used in 5 and 6 cosmetic products, respectively; however, use concentration data on these ingredients were not provided in the concentration of use survey.
Cosmetic products containing Rosa canina-derived ingredients may be applied to the skin and hair or, incidentally, may come in contact with the eyes (e.g., Rosa Canina Fruit Extract at maximum use concentrations up to 0.2% in eye area cosmetics) and mucous membranes (e.g., Rosa Canina Seed Extract at maximum use concentrations up to 1.5% in lipstick). Additionally, some of these ingredients are being used in products that may result in incidental ingestion. For example, Rosa Canina Seed Extract is being used in lipstick at maximum use concentrations up to 1.5%, Rosa Canina Flower Extract is being used in lipstick at maximum use concentrations up to 0.04%, and Rosa Canina Fruit Extract is being used in lipstick at maximum use concentrations up to 0.0015%. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
Rosa Canina Fruit Extract is used in aerosol hair sprays at maximum use concentrations up to 0.0002% and in pump hair sprays at concentrations up to 0.25%; Rosa Canina Flower Extract is being used in pump hair sprays at maximum use concentrations up to 0.001% and, in perfumes, at maximum use concentrations up to 0.01%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles below 10 μm, compared with pump sprays.25-28 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.25,26 Rosa Canina Fruit Extract is also being used in powders (dusting and talcum) at maximum use concentrations up to 0.01%, and in face powders at maximum use concentrations up to 0.002%. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.29-31
The International Fragrance Association (IFRA) has established limitations for eugenol and linalool (hydroperoxide content) in cosmetic products. 32 The limit for eugenol is dependent upon the product type and ranges from 0.2% (lip products and deodorants/antiperspirants) to 4.3% (mouthwash). The limit for the hydroperoxide content of linalool is 20 mmol/l.
Noncosmetic
According to the FDA, rose fruit (hips) is generally recognized as safe for use in food for human consumption. 33
In traditional folk medicine, the petals, fruit, and leaves of Rosa canina are used in the treatment of various diseases/conditions, such as, nephritis, common cold, flu, coughing, bronchitis, eczema, itching, and biliary diseases. 11 Rosa Canina Fruit contains a wide range of bioactive compounds, including GOPO, vitamin C, phenolics, lycopene, lutein, zeaxanthin, and other carotenoids. 15
According to another source, a standardized powder of Rosa Canina Fruit is marketed as an herbal remedy for the treatment of pain in patients with osteoarthritis. 34 Among the components of this powder is a mixture of 3 triterpene acids (oleanolic, ursolic, and betulinic acids).34,35
Toxicokinetic Studies
No relevant published toxicokinetics studies on Rosa canina-derived ingredients were identified in a literature search for these ingredients, and no unpublished data were submitted. Toxicokinetics data were not expected to be found because each botanical ingredient is a mixture of many constituents.
Toxicological Studies
Acute Toxicity Studies
Intraperitoneal
Rosa Canina Leaf Extract
The acute intraperitoneal (i.p.) toxicity of Rosa Canina Leaf Extract (methanol extract) was evaluated using groups of 5 albino mice. 19 An estimated acute i. p. LD50 of 455.19 ± 23 mg/kg was reported. The animals exhibited toxic signs at doses greater than the LD50.
Short-Term Toxicity Studies
Oral
Animal
Rosa Canina Fruit Extract
Rosa Canina Fruit Extract (aqueous extract diluted to 10% w/v, 500 mg/kg body weight/d) was administered orally to 12 female brownish guinea pigs daily for 35 d. 36 The vehicle control group (12 guinea pigs) received water. The general condition and behavior of all animals were described as normal, and body weight and food consumption in both groups were approximately the same.
Developmental and Reproductive Toxicity (DART) Studies
Data on the reproductive and developmental toxicity of Rosa canina-derived ingredients were not found in the published literature, and unpublished data were not submitted.
Genotoxicity Studies
In vitro
Rosa Canina Fruit Extract
The genotoxicity of a product containing a maximum concentration of 0.65% Rosa Canina Fruit Extract was evaluated in the Ames test (Organization for Economic Co-operation and Development (OECD) Test Guideline (TG) 471) using Salmonella typhimurium strains (strains not stated). 8 The product was evaluated at doses up to 5,000 μg/plate, with and without metabolic activation. It was concluded that the product did not have mutagenic or pro-mutagenic activity in this assay.
Ames test results for Rosa Canina Fruit Extract (butylene glycol extract) were negative. 7 The test concentrations/doses and bacterial strains tested were not stated. Rosa Canina Fruit Extract (butylene glycol extract) also was not genotoxic in the chromosome aberration test using the Chinese hamster lung cell line (CHL/IU). Details relating to the test protocol were not provided.
Rosa Canina Fruit Juice, Rosa Canina Leaf, and Rosa Canina Seed
Rosa Canina Fruit (unclear if Rosae pseudofructus cum or Rosae pseudofructus sine fructibus, R canina L., Rosaceae) was boiled at 100°C, stewed for 10 min, and then evaluated for genotoxicity in the Ames test.22,37 Raw, boiled juice, boiled leaves, and dried seeds (concentration of each not stated) were not mutagenic in S. typhimurium strain TA 100.
Anti-Genotoxicity Studies
In vitro
Rosa Canina Fruit
In an anti-genotoxicity assay, Rosa Canina Fruit (raw, concentration not stated) decreased the genotoxicity of sodium azide by 44%.22,37
Rosa Canina Fruit Extract
The micronucleus test was used to evaluate the genotoxic effects of cypermethrin and fenvalerate (both insecticides); the effect of the water and ethanol extracts of Rosa Canina Fruit on the genotoxicity of these insecticides was also determined in this study. 38
Using human peripheral lymphocyte cultures in vitro, cypermethrin was tested at concentrations of 20, 30, 40, and 50 ppm, and fenvalerate was tested at concentrations of 25, 50, 75, and 100 ppm. Rosa Canina Fruit extracts were tested at a concentration of 100 ppm. The negative control was dimethyl sulfoxide (DMSO, 1%), and ethyl methanesulfonate (1 mM) served as the positive control. The Duncan test was used for statistical evaluation. For cypermethrin, the micronucleus frequency was 1.275 at the highest test concentration, and the micronucleus frequency for fenvalerate was 1.6 at the highest test concentration. Micronucleus frequencies were 0.725 and 2.7 for negative and positive controls, respectively. These differences between the experimental and DMSO control groups were statistically significant (P < 0.05). In the genotoxicity tests with Rosa Canina Fruit Extracts, the micronucleus frequencies were as follows: 1.0 (cypermethin + water extract), 1.075 (cypermethin + ethanol extract), 1.225 (fenvalerate + water extract), and 1.275 (fenvalerate + ethanol extract). Both extracts (ethanol and water) of Rosa Canina Fruit caused statistically significant reductions (P < 0.05) in the micronucleus frequencies that were associated with insecticide exposure. It was concluded that the water and ethanol extracts of Rosa Canina Fruit reduced the genotoxicity of both insecticides.
Carcinogenicity Studies
Data on the carcinogenicity of Rosa canina-derived ingredients were not found in the published literature, and unpublished data were not submitted.
Other Relevant Studies
Cytotoxicity
Rosa Canina Seed Extract
Dried Rosa Canina Seed (100 g) was extracted with petroleum ether, 95% ethanol, or water, with yields of 0.3%, 5.9%, and 10%, respectively.22,39 The aqueous Rosa Canina Seed Extract had little cytotoxic effect on Yoshida ascites sarcoma cells (LC50 > 10 mg/mL). However, the ethanol and petroleum ether extracts had a substantial cytotoxic effect on these cells, with LC50s of 3.9 and 1.2 mg/mL, respectively. The authors noted that these results indicated a possible anti-carcinogenic effect. However, this study did not involve testing to determine whether or not Rosa Canina Seed Extract (ethanol and petroleum ether extracts) is cytotoxic to normal cells.
Effect on Skin Pigmentation
Animal
Rosa Canina Fruit Extract
Rosa Canina Fruit Extract (500 mg/kg body weight/d as aqueous extract, diluted to 10% w/v) was administered orally to 12 female brownish guinea pigs daily for 35 d. 36 The vehicle control group (12 guinea pigs) received water. To develop pigmentation, a 4 cm2 area of shaved skin was irradiated with 0.384 J/cm2 (0.8 mw/cm2 x 8 min) using a short wave ultraviolet (290 to 320 nm; UVB) lamp on days 8, 10, and 12. The animals were killed on day 36. The skin lightening effect of Rosa Canina Fruit Extract was determined by measuring the “L*”value (lightness) with a reflectance spectrophotometer, and was evaluated quantitatively by determining the change in the L* value during the 35-d oral dosing period. Though the L* value of the irradiated area in the vehicle control group decreased substantially due to UVB-induced pigmentation, the L* value in the experimental group was statistically significantly higher (compared to control) at each time point after irradiation. UVB-induced skin pigmentation was reduced after dosing with Rosa Canina Fruit; thus, the oral administration of Rosa Canina Fruit Extract to brown guinea pigs caused inhibition of skin pigmentation. Proanthocyanidins in Rosa Canina Fruit Extract was found to be the active principle responsible for the inhibitory effect on pigmentation of guinea pig skin. It should be noted that, according to another study (in vitro), proanthocyanidins from grape seeds had no effect on the expression of tyrosinase protein in normal human melanocytes. 40
In vitro
Rosa Canina Fruit Extract
The effects of compounds isolated from a methanolic extract of Rosa Canina Fruit on melanin biosynthesis in B16 mouse melanoma cells was investigated. 41 Quercetin, one of the components isolated from Rosa Canina Fruit, was added to the culture medium at concentrations of 10 μm, 20 μm, and 40 μm; the melanin content was reduced (compared to untreated control cells) in a dose-dependent manner to 64%, 34.5%, and 1%, respectively. It should be noted that, according to another study, the enhancement of melanogenesis by quercetin has been observed in human melanoma cells (20 μM quercetin) and normal epidermal melanocytes (1 μM quercetin). 42
Rosa Canina Fruit Extract (aqueous extract) was added to B16 mouse melanoma cell cultures in vitro at concentrations of 250 μg/ml, 500 μg/ml, and 1000 μg/ml to confirm its melanogenesis-inhibitory effect. Untreated cultures served as negative controls. Additionally, arbutin (known inhibitor of melanogenesis) served as the positive control. Rosa Canina Fruit Extract had an inhibitory effect on melanogenesis in mouse melanoma cells, having caused the following concentration-dependent reduction in melanin content when compared to negative control cultures: 65.6% at 250 μg/ml, 37.8% at 500 μg/ml, and 19% at 1000 μg/ml. The reduction in melanin content occurred without any significant cytotoxicity. 36
Immune System Effects
Animal
Rosa Canina Fruit Extract
A study was performed to investigate the potential for Rosa Canina Fruit Extract (hydro-alcoholic extract) to induce immunomodulatory activity using 45 rats (3 groups of 15). 43 The 3 groups received normal saline (10 mg/kg), Rosa Canina Fruit Extract (250 mg/kg), and Rosa Canina Fruit Extract (500 mg/kg) orally, by gavage, daily for a period of 4 wk.
At Rosa Canina Fruit Extract doses of 250 mg/kg and 500 mg/kg, the gamma globulin level, neutrotrophil and monocyte counts, and phagocyte activity increased statistically significantly, when compared to the normal saline group. Lymphocyte percentages were statistically significantly decreased in treatment groups at weeks 2 and 3. On days 14 and 21, neutrophil levels increased in the 250 mg dose group. The phagocytic activity in both test groups was significantly higher, compared to the control group, during all days of the study. There was no statistically significant difference in alanine aminotransferase (ALT), aspartate aminotransferase (AST), or alkaline phosphatases (ALP) when compared to the control group. However, Rosa Canina Fruit Extract (both doses) statistically significantly increased thiobarbituric acid reactive substances (TBARS) and also decreased glutathione (GSH) levels when compared to the control group on day 28. It was concluded that Rosa Canina Fruit Extract might have immunomodulatory effects.
Dermal Irritation and Sensitzation Studies
Irritation and Sensitization
Animal
Rosa Canina Fruit Extract
In a skin irritation test involving 3 rabbits (strain not stated), results for the butylene glycol extract of Rosa Canina Fruit (0.3% solids—100% of the butylene glycol extract) were negative. 7 Further details relating to the test protocol were not provided.
The skin sensitization potential of Rosa Canina Fruit Extract (butylene glycol extract) was evaluated using 10 guinea pigs (strain not stated). 7 The following concentrations of Rosa Canina Fruit Extract were tested: 4% and 20% of the original solution (0.3% solids) (first induction), 20% of the original solution (second induction), and 4% and 20% of the original solution (challenge). Additional details relating to the test protocol were not presented. Test results were classified as negative.
Human
Rosa Canina Fruit Extract
The skin irritation potential of a cosmetic product diluted to a maximum concentration of 0.0975% Rosa Canina Fruit Extract was evaluated using 10 adult subjects. 8 The product was applied and left in place under an occlusive patch for 48 h. Neither the location of the test site on the bodies of the subjects nor the concentration/dose per cm2 of the exposed skin was stated. The product was classified as non-irritating.
In another study, the skin sensitization potential of the diluted product tested in the preceding study was evaluated using 110 normal volunteers in accordance with the method of Marzulli and Maibach. 8 The product was applied to the back using an occlusive patch with filter paper. The concentration/dose per cm2 of the exposed skin was not stated. The 3-week induction phase was followed by a 2-wk non-treatment period and then a 1-wk challenge phase. The product was classified as non-irritating and non-sensitizing.
Rosa Canina Flower Extract
The skin irritation and sensitization potential of a lip balm containing 0.04% Rosa Canina Flower Extract was evaluated using 106 male and female healthy subjects. 44 Approximately 0.2 g of the test substance was applied to the upper back (between the scapulae) using a 1" × 1" semi-occlusive patch, which remained in place for 24 h. Reactions were scored at the time of patch removal and just prior to application of the next patch. The patches were applied 3 times per week for a total of 9 induction applications. After a 2-week (approximately) non-treatment period, a challenge patch was applied for 24 h to a new test site. Reactions were scored at 24 h and 72 h (or 120 h) post-application. The lip balm did not have skin irritation or sensitization potential in this study.
In another study, the skin sensitization potential of a lip liner containing 0.018% Rosa Canina Flower Extract was studied using 202 healthy male and female subjects. 45 The product (0.2 g) was applied to the infrascapular area of the back using an occlusive patch or a semi-occlusive patch (each 2 cm x 2 cm). The test procedure was similar to that stated in the preceding study, with the exceptions that induction reactions were scored at 48 h (or 72 h) post-application of the 24-h induction patch, and challenge reactions were scored at 48 h and 72 h post-application. No adverse events were reported in this study, and the authors concluded that, under occlusive and semi-occlusive conditions, there was no evidence of sensitization to the lip liner containing 0.018% Rosa Canina Flower Extract.
Ocular Irritation Studies
Data on the ocular irritation potential of Rosa canina-derived ingredients were not found in the published literature, and unpublished data were not submitted.
Clinical Studies
Case Reports
Rosa Canina Fruit and Rosa Canina Fruit Extract
An anaphylactic reaction was observed in a male patient, sensitized to Rosaceae (without related pollinosis), after consumption of a tea containing Rosa Canina Fruit. 46 The tea also contained hibiscus, apple, orange peel, and elderberry. The patient had no history of asthma or rhinitis, but had presented with an oral allergy syndrome to peach and almonds and had also experienced an anaphylactic reaction after eating cherries. Prick test results were positive for Rosa Canina Fruit Extract. The presence of specific IgE against the Rosa Canina Fruit in the tea was also demonstrated, using in vitro and in vivo methods, suggesting that Rosa Canina Fruit caused the anaphylactic reaction. Cutaneous tests involving other ingredients in the tea were negative.
Thirteen workers with respiratory symptoms (asthma [9 subjects]; rhinitis [5 subjects]; urticaria [1 subject]) related to occupational exposure to powdered Rosa Canina Fruit were evaluated. 47 Based on the results of positive skin prick tests, 7 of the workers were found to have evidence of immunoglobulin E (IgE) specific to Rosa Canina Fruit (1 mg/mL). Four workers with histories of work-related asthma underwent bronchopulmonary challenges with Rosa Canina Fruit, and 2 of the workers had positive challenges with greater than 20% declines in forced expiratory volume (FEV1) measurements. It was concluded that Rosa Canina Fruit is an occupational allergen that is capable of producing asthma.
In-Use Test
Rosa Canina Flower Extract
The cutaneous acceptability of a cosmetic investigational product (night cream) containing approximately 0.005% Rosa Canina Flower Extract was studied using 48 female subjects. 48 Twenty-five subjects (52% of the subjects tested) had “sensitive” skin and 6% of the subjects (3 subjects) had a history of atopy. During 4 wk, the product was applied once per day (in the evening) to the face and neck (including the eye contours). The following reactions were observed in 7 subjects: discomfort (4 subjects—prickling in particular), irritation + discomfort and palpebral swelling (2 subjects), and “small pimples” + discomfort (1 subject). Only the reactions observed in 2 subjects (irritation + discomfort and palpebral swelling) were considered pertinent. The authors noted that no abnormal clinical sign was observed by the dermatologist after 4 wk of product use.
Other Clinical Reports
Rosa Canina Fruit Extract
A double-blind, placebo-controlled clinical trial involving 2 groups of 16 subjects was performed. 49 One group received placebo tablets (1 per subject) and the other group received tablets containing Rosa Canina Fruit Extract (100 mg + excipients, 1 per subject) once per day for 12 wk. The Rosa Canina Fruit Extract tested was an aqueous ethanol extract of Rosa Canina Fruit containing its seeds, dextrin, cyclodextrin, and not less than 0.1% tiliroside (glycosidic flavonoid). According to the physical assessments that were performed and the blood biochemical and hematological parameters evaluated, there were no clinically problematic abnormalities or findings in any of the subjects tested.
Rosa Canina Fruit
Rosa Canina Fruit (powder form) was evaluated in a double-blind, placebo-controlled clinical trial involving 44 subjects (active treatment group) and 45 subjects (placebo group). 50 The active treatment group was instructed to take 5 capsules, each containing 0.5 g Rosa Canina Fruit (powder form), daily for 6 months. The other group was treated with placebo of a similar taste according to the same procedure. The following 14 adverse effects were reported in this study: gastrointestinal disturbances, common cold/influenza, skin rash/eczema, vasculitis, elevated diuresis, back problems, swallowing problems, dizziness, urinary tract infection, headache, cyst in left breast, pain in hand, weight gain, sleeping disturbances, and elevated blood pressure. It was noted that these adverse effects were not related to dosing with Rosa Canina Fruit (powder form).
Summary
Rosa canina, the plant source of ingredients reviewed in this safety assessment, is an herb that belongs to the Rosaceae family. Rosa Canina Fruit Extract is reported to function as a skin conditioning agent in cosmetic products. 1 Functions reported for other Rosa canina-derived ingredients include: skin conditioning agent, fragrance ingredient, cosmetic astringent, anti-acne agent, abrasive, humectant, and exfoliant.
Using ultraviolet spectrophotometry, the λmax for Rosa Canina Fruit Extract (ethanol extract) has been reported at ∼280 nm (the short end of UVB).
Collectively, information supplied to FDA by industry as part of the VCRP and a survey of ingredient use concentrations conducted by the Council indicates that the following Rosa canina-derived ingredients are being used in cosmetic products: Rosa Canina Fruit Extract, Rosa Canina Flower, Rosa Canina Flower Extract, Rosa Canina Fruit, Rosa Canina Leaf Extract, Rosa Canina Seed Extract, and Rosa Canina Seed Powder. The highest use frequency is reported for Rosa Canina Fruit Extract (350 uses). The Council survey data also indicate that Rosa canina-derived ingredients are being used in cosmetics at maximum ingredient use concentrations up to 1.5% (i.e.,, Rosa Canina Seed Extract in leave-on products [lipstick]).
In traditional folk medicine, the petals, fruit, and leaves of Rosa canina are used in the treatment of various diseases/conditions. Examples of such include, nephritis, common cold, flu, coughing, bronchitis, eczema, itching, and biliary diseases.
The fruits of Rosa canina contain phenolic acids, proanthocyanidins, tannins, flavonoids, fatty acids, pectins, carotenoids, and fruit acids (ascorbic acid, malic acid, and citric acid). (+)-Catechin, a flavonoid, has been identified as the most abundant flavan-3-ol (3.59 mg/100 g) in Rosa Canina Fruits, and the abundance of ascorbic acid (vitamin C, 880 mg/100 mL) in Rosa Canina Fruit has also been noted. In addition to vitamin C, the following other nutrients in Rosa Canina Fruit have been reported: carotenoids, tocopherol, bioflavonoids, tannins, pectin, sugars, organic acids, amino acids, essential oils, phosphorus, potassium, calcium, magnesium, iron, copper, manganese, and zinc. Additionally, the following 6 main carotenoids have been identified in Rosa Canina Fruit: epimers of neochrome, lutein, zeaxanthin, rubixanthin, lycopene, and β-carotene. The chemical composition of Rosa Canina Fruit differs, depending on the cultivar, growing region, climate, maturity, cultivation practice, and storage conditions.
Flavonols such as glycosides of quercetin and kaempferol, hydroxycinnamic acids, and ellagitannins were detected in samples of Rosa Canina Bud Extract, with gallotannins being the main components. Rosa Canina Leaf Extract contains alkaloids, flavonoids, glycosides, saponins, and a volatile oil. Rosa Canina Seed contains fatty acids and various elements, some of which are common to Rosa Canina Fruit.
An acute i.p. LD50 of 455.19 ± 23 mg/kg was reported for Rosa Canina Leaf Extract (methanol extract) in a study involving groups of 5 albino mice. Toxic signs were observed at doses greater than the LD50.
Rosa Canina Fruit (500 mg/kg body weight/day, aqueous extract diluted to 10% w/v) was administered orally to 12 female guinea pigs daily for 35 d. The general condition and behavior of all animals were described as normal, and body weights and food consumption were comparable to control values.
Rosa Canina Fruit Extract (aqueous ethanol extract, 100 mg + excipients per tablet) was administered orally to 16 subjects once daily for 12 wk. The test substance was an aqueous ethanol extract of Rosa Canina Fruit containing its seeds, dextrin, cyclodextrin, and not less than 0.1% tiliroside (glycosidic flavonoid). There were no abnormalities, subjective symptoms, or findings that may have been indicative of clinical effects during the study. In a similar study, 44 subjects were instructed to take 5 capsules, each containing 0.5 g Rosa Canina Fruit (powder form) daily for 6 months. Dosing did not result in any adverse effects.
A product containing a maximum concentration of 0.65% Rosa Canina Fruit Extract did not have mutagenic or pro-mutagenic activity in Salmonella typhimurium strains when evaluated in the Ames test (with and without metabolic activation). Ames test results for Rosa Canina Fruit Extract (butylene glycol extract, test concentration not stated) were also negative. Additionally, Rosa Canina Fruit Extract (butylene glycol extract, test concentration not stated) was not genotoxic in the chromosome aberration test using the Chinese hamster lung cell line (CHL/IU).
Rosa Canina Fruit Juice, Rosa Canina Leaf, and Rosa Canina Seed (concentrations not stated), were not mutagenic to Salmonella typhimurium strain TA 100 in the Ames test.
In an anti-genotoxicity assay, Rosa Canina Fruit (raw, concentration not stated) decreased the genotoxicity of sodium azide by 44%. Rosa Canina Fruit Extract (at 100 ppm, water and ethanol extracts) reduced the genotoxicity of 2 insecticides, cypermethrin and fenvalerate, in the micronucleus test.
Dried Rosa Canina Seed (100 g) was extracted with petroleum ether, 95% ethanol, or water, with yields of 0.3%, 5.9%, and 10%, respectively. The aqueous Rosa Canina Seed Extract had a weak cytotoxic effect on Yoshida ascites sarcoma cells (LC50 > 10 mg/mL). However, the ethanol and petroleum ether extracts had a substantial cytotoxic effect on these cells, with LC50s of 3.9 and 1.2 mg/mL, respectively.
In a skin irritation test involving 3 rabbits, results for the butylene glycol extract of Rosa Canina Fruit (0.3% solids—100% of the butylene glycol extract) were negative.
The skin sensitization potential of Rosa Canina Fruit Extract (butylene glycol extract) was evaluated using 10 guinea pigs (strain not stated), and the following concentrations were tested: 4% and 20% of the original solution (0.3% solids) (first induction), 20% of the original solution (second induction), and 4% and 20% of the original solution (challenge). Test results were negative.
A lip balm containing 0.04% Rosa Canina Flower Extract was evaluated for skin irritation and sensitization potential using 106 male and female subjects. Study results were negative. In another study, the skin sensitization potential of a lip liner containing 0.018% Rosa Canina Flower Extract was studied using 202 male and female subjects. The lip liner did not induce sensitization in this study.
A cosmetic product diluted to a concentration of 0.0975% maximum Rosa Canina Fruit Extract was evaluated in a 48-h occlusive patch test using 10 adult subjects. Results were negative. The skin sensitization potential of the same product was evaluated in a repeated insult patch test using 110 normal volunteers. The product was classified as non-irritating and non-sensitizing.
In a use test, the cutaneous acceptability of a cosmetic investigational product (night cream) containing approximately 0.005% Rosa Canina Flower Extract was studied using 48 female subjects, some with a history of sensitive skin/atopy. The following reactions were observed in 7 subjects: discomfort (4 subjects—prickling in particular), irritation + discomfort and palpebral swelling (2 subjects), and “small pimples” + discomfort (1 subject). Only the reactions observed in 2 subjects (irritation + discomfort and palpebral swelling) were considered pertinent. No abnormal clinical signs were observed after 4 wk of product use.
Positive skin prick tests (1 mg/mL Rosa Canina Fruit) were reported for 7 of 9 subjects exposed to powdered Rosa Canina Fruit in the workplace.
An anaphylactic reaction was observed in a male patient after consumption of a tea containing Rosa Canina Fruit. Prick test reactions to the fruit were positive, and the presence of specific IgE against the fruit was demonstrated using in vitro and in vivo methods.
Neither toxicokinetic data nor data on the carcinogenicity and reproductive and developmental toxicity of Rosa canina-derived ingredients were identified in the published literature.
Oral dosing with Rosa Canina Fruit Extract (aqueous extract, 10% w/v) caused a reduction in UVB-induced skin pigmentation in guinea pigs. Rosa Canina Fruit Extract (aqueous extract) also caused a concentration-dependent (250, 500, and 1000 μg/ml) decrease in the melanin content of B16 mouse melanoma cell cultures in vitro. Quercetin, isolated from a methanolic extract of Rosa Canina Fruit, reduced the melanin content of B16 mouse melanoma cells in a concentration-dependent manner.
Data suggestive of immunomodulatory activity induced by Rosa Canina Fruit Extract (hydro-alcoholic extract) have been identified in the published literature.
Discussion
The Panel previously evaluated the safety of butylene glycol, a major component of Rosa Canina Fruit Extract, in cosmetics and issued a final report in 1985 with the conclusion that butylene glycol, hexylene glycol, ethoxydiglycol, and dipropylene glycol are safe as presently used in cosmetics. This conclusion was reaffirmed by the Panel in a 2006 publication. Rosa Canina Fruit Extract contains 76.5% to 93.50% butylene glycol. The Panel determined that, given the low use concentration of Rosa Canina Fruit Extract in cosmetics (≤0.25%), the concentration of butylene glycol in this ingredient is orders of magnitude lower than the maximum use concentration of butylene glycol (>50%; considered a safe use concentration) that is stated in the published final report.
An effect of Rosa Canina Fruit Extract on skin pigmentation was reported in in vitro and in vivo studies, and the quercetin and proanthocyanidins components of this ingredient were identified as the active principles for this effect. The Panel noted that use concentrations of this ingredient and, thus, the levels of these components in cosmetics, are considered below the threshold of concern for this effect.
The Panel determined that linalool and eugenol, two of the components of Rosa canina-derived ingredients, are potential constituents of concern based on their sensitization potential, and noted that IFRA has established limits relating to eugenol in finished products and the hydroperoxide content of linalool. The Panel noted that because botanical ingredients are complex mixtures, there is concern that multiple botanical ingredients may each contribute to the final concentration of a single constituent. Therefore, when formulating products, manufacturers should avoid reaching levels in the final formulation of botanical constituents that may cause sensitization or other adverse effects.
The Panel also expressed concern about pesticide residues, heavy metals, and other plant species that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit impurities.
Rosa Canina Fruit Extract is used in aerosol hair sprays at maximum use concentrations up to 0.0002% and in pump hair sprays at concentrations up to 0.25%; Rosa Canina Flower Extract is being used in pump hair sprays at maximum use concentrations up to 0.001% and, in perfumes, at maximum use concentrations up to 0.01%. Rosa Canina Fruit Extract is also being used in powders (dusting and talcum) at maximum use concentrations up to 0.01%, and in face powders at maximum use concentrations up to 0.002%. The Panel discussed the issue of incidental inhalation exposure during cosmetic use, and agreed that incidental inhalation exposures to these ingredients in such cosmetic products would not cause adverse health effects.
Conclusion

*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contribution
Johnson, W. contributed to conception and design; acquisition, analysis, and interpretation; drafted manuscript; and select item. Heldreth, B. contributed to conception and design; acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; and gave final approval. Gill, Lillian J. contributed to conception and design; analysis and interpretation; select item; critically revised manuscript; and gave final approval. Bergfeld, W. contributed to conception and design; analysis and interpretation; select item; critically revised manuscript; and gave final approval. Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., and Snyder, P. contributed to conception and design; contributed to analysis and interpretation; select item; critically revised manuscript; and gave final approval. All authors agree to be accountable for all aspects of work ensuring integrity and accuracy.