Abstract
Glyphosate-based herbicides are the most used herbicides in the world. Despite being widely used, a dispute exists whether glyphosate-based herbicides have a negative effect on human health, particularly genotoxic effects. Therefore, the aim of this study was to investigate glyphosate genotoxicity on cultured human lymphocytes. Cultured human lymphocytes were treated with different concentrations of glyphosate (20, 40, and 200 µmol/L). Four toxicity measures were examined: frequency of chromosomal aberrations (CAs), frequency of sister-chromatid exchange (SCE), production of 8-OHdG, and cell kinetics analysis. The results show that glyphosate induced significant (P < 0.05) increases in the levels of SCE at the highest used concentration (200 μmol/L). However, no significant elevation in SCE levels was observed at the lower examined concentrations (20 and 40 μmol/L). No significant changes in CA were detected at all examined concentrations (P = 0.86). Also, glyphosate did not induce changes to the normal level of 8-OHdG at all examined concentrations (P = 0.98). Last, no significant changes in either mitotic index or proliferative index were observed at any examined concentrations (P > 0.05). The results collectively indicate a lack of genotoxicity and cytotoxicity of glyphosate in cultured human lymphocytes when dealing with environmentally relevant concentrations (20 and 40 μmol/L). However, being exposed to higher concentrations (200 μmol/L) led to slightly higher level of SCE. Therefore, we recommend cautionary measures when dealing with glyphosate-based herbicides for individuals, such as farmers, who may be extensively exposed to high concentrations of these herbicides.
Introduction
Glyphosate (CAS number: 1071-83-6) is the active ingredient in many well-known herbicides, with numerous glyphosate-based formulations, such as Roundup™ (a trademark of Monsanto Technology LLC), registered in over 100 countries under a variety of brand names.1,2 In 2011 alone, about 600,000 tons of glyphosate products were used worldwide. 3 This extensive use of glyphosate-based herbicides was the result of their effectiveness and the reports deeming their use to have no human health concern. This is highlighted by the declaration of WHO (1992), which considers the available studies on glyphosate that do not indicate that it is mutagenic, carcinogenic, or teratogenic.2,4
However, in 2015, the International Agency for Research on Cancer (Lyon, France) indicated that glyphosate is toxic to humans, and should be considered “probably carcinogenic.” 5 Accordingly, many countries started banning glyphosate use, including Sri Lanka 6 and Bermuda. 7 This sparked numerous human and animal studies to be conducted to test for carcinogenicity. The results of these studies were controversial, where some of them found that glyphosate could induce genotoxicity in different cells such as human peripheral blood lymphocytes8,9 and in mice bone morrow cells. 10 However, other studies concluded that there is no genotoxic effect of glyphosate in rats, 11 mice, 12 or humans. 13
In light of the extensive use of glyphosate herbicides worldwide despite a lack of evidence to prove their safety, combined with the potential genotoxicity and the detrimental impact on human health; the aim of this study is to investigate glyphosate genotoxicity on cultured human lymphocytes through performing chromosomal aberrations assay (CA), sister-chromatid exchange assay (SCE) assays, and 8-OHdG assay. In addition, cytotoxicity of glyphosate was examined using mitotic (MI) and proliferative (PI) indices to complement previously reported genotoxicity and mutagenicity assays.14‐16
Materials and Methods
Subjects
Blood samples (12–15 mL) were obtained from 5 healthy male adults (20–30 years old) with no history of smoking or alcohol use. Blood samples were collected in sodium heparinized tubes and were used immediately following sampling. The study was ethically approved (IRB of University of Jordan, approval ID: 10120161240) and subjects were consented prior to blood sampling.
Cell Cultures
Prior to culture initiation, donated blood was mixed by inverting the tube several times to ensure homogeneity of the blood sample and that lymphocytes were seeded at the same cell density in the cultures. The cell density varies in each experiment as the number of lymphocytes varies from donor to another. The mean density of lymphocytes per ml in the cultures was 0.244 ± 0.0201 * 106. Blood was cultured for all assays in Pb-Max media (1 mL blood: 10 mL media) and incubated for 72 h under 5% CO2 as previously described. 17 Glyphosate (final concentration of 20, 40, and 200 µmol/L, prepared in dimethyl sulfoxide (DMSO)) was added to cultures in the last 24 h of incubation time. The glyphosate doses were according to a previous study conducted on cultured human lymphocytes. 8 Such concentrations were slightly higher than that reported in human exposure studies.18,19 Cultures treated with DMSO alone were used as controls. 20
Sister-Chromatid Exchange Assay
Sister-chromatid exchange analysis has come into use as a sensitive manner to monitor DNA damage by mutagenic agents.21,22 To differentially stain sister-chromatids, bromodeoxyuridine (BrdU, 20 µg/ml) was added to cultures designated for SCE assay at the culture initiation. 23 Sister-chromatid exchange cultures were kept in the dark during incubation and processing to prevent BrdU degradation. In the last 2 h of incubation, colchicine was added to arrest cultures in the metaphase stage. 24 Harvesting process involved exposure to hypotonic solution (0.075 M KCL, 20 min), cell fixation using methanol/acetic acid (ratio: 3/1) and dropping of cells onto microscopic slides. 25 Sister-chromatid exchange were visualized using florescent-plus-Gimsa procedure. 26 Sister-chromatid exchange were scored using light microscope at 1000× magnification power by an individual who was unaware of treatment conditions. For each treatment, 50 M2 metaphases were scored for the presence of exchanges. 25
Chromosomal Aberrations Assay
For CAs assay, blood was cultured as described above for SCE assay except BrdU was not included in the culture media. Harvesting steps were also as described above. Chromosomal aberrations were visualized using Giemsa stain as previously described. 27 About 500 metaphases were examined per treatment by an individual who was unaware of treatment conditions. Chromosomal aberrations were scored using light microscope with 1000× magnification power. 28
8-OHdG Assay
8-Hydroxy-2′-deoxyguanosine (8-OHdG) is one of the prevalent forms of free radical-induced oxidative lesions. As a result, 8-OHdG has been utilized as a key marker in measuring the effect of endogenous oxidative DNA damage and a factor in the initiation and promotion of carcinogenesis. 29 Cultures designated for 8-OHdG assay were grown for 72 h and then washed 5 times using RPMI media. 28 Cells were then incubated in RPMI for additional 6 hr and treated with glyphosate during the second incubation period. Cultures were then processed as previously described 30 and levels of 8-OHdG assay were assessed using a specialized kit according to instruction supplied by the producer (Kit ID: ab201734, Abcam Inc, UK).
Cell Kinetics Analysis
The MI and PI were used to determine the cytotoxic effect of glyphosate drug. The MI and PI indices were calculated as previously described. 20
Statistical Analysis
Statistical analysis was achieved using Graph pad Prism software (version 5). Measures were expressed as percentages and means depending on the variable. Groups were compared using ANOVA and Tukey test. The threshold for statistical significance was P less than or equal to .05.
Results
In this study, we investigated genotoxicity of glyphosate in cultured human lymphocytes using SCE, CA and 8-OHdG assays.
Figure 1 shows frequencies of SCE induced by treatment of cultures with different concentrations of glyphosate (20, 40, and 200 μmol/L). Glyphosate induced significant slight increases in the levels of SCE at the highest used concentration (200 μmol/L) relative to the control group (5.65 ± .156 in control vs 6.395 ± .192 in 200 μmol/L glyphosate, P = .0138). However, no significant elevation in SCE levels was observed at lower examined concentrations (20 and 40 μmol/L). As a positive control, mitomycin C at 10 ng/mL final concentration was used. Mitomycin C increased SCE by about 2-fold (5.6 ± .15 in control group vs 12.5 ± 0.23 in mitomycin C group, P < .001). Frequencies of sister-chromatid exchange induced by treatment of cultured human lymphocytes (n = 5) with different concentrations of glyphosate (20, 40, and 200 μmol/L) and mitomycin C (10 ng/mL) for 50 metaphases per treatment. * Indicates significant difference from the control group (P value <0.05).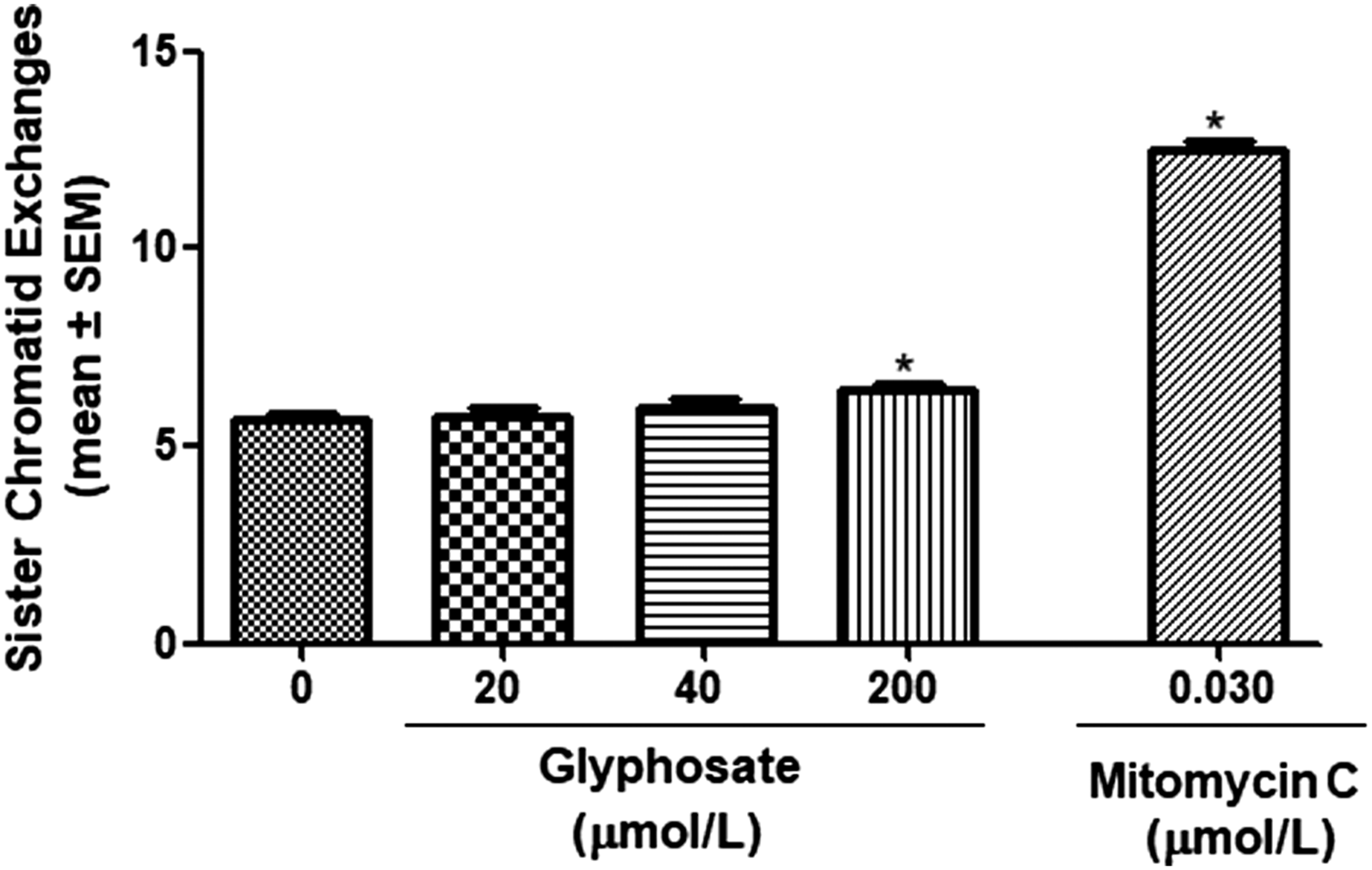
Figure 2 shows levels of CA induced by glyphosate in cultured human lymphocytes. No significant changes in CA were detected at all examined concentrations (P = .86). Treatment of cultures with mitomycin C at 10 ng per mL significantly increased CA by several folds (.018 ± .011 vs .12 ± .014, P < .001). Frequencies of CA induced by treatment of cultured human lymphocytes (n = 5) with different concentrations of glyphosate (20, 40, and 200 μmol/L) and mitomycin C (10 ng/mL) for 500 metaphases per treatment. * Indicates significant difference from the control group (P value <0.05).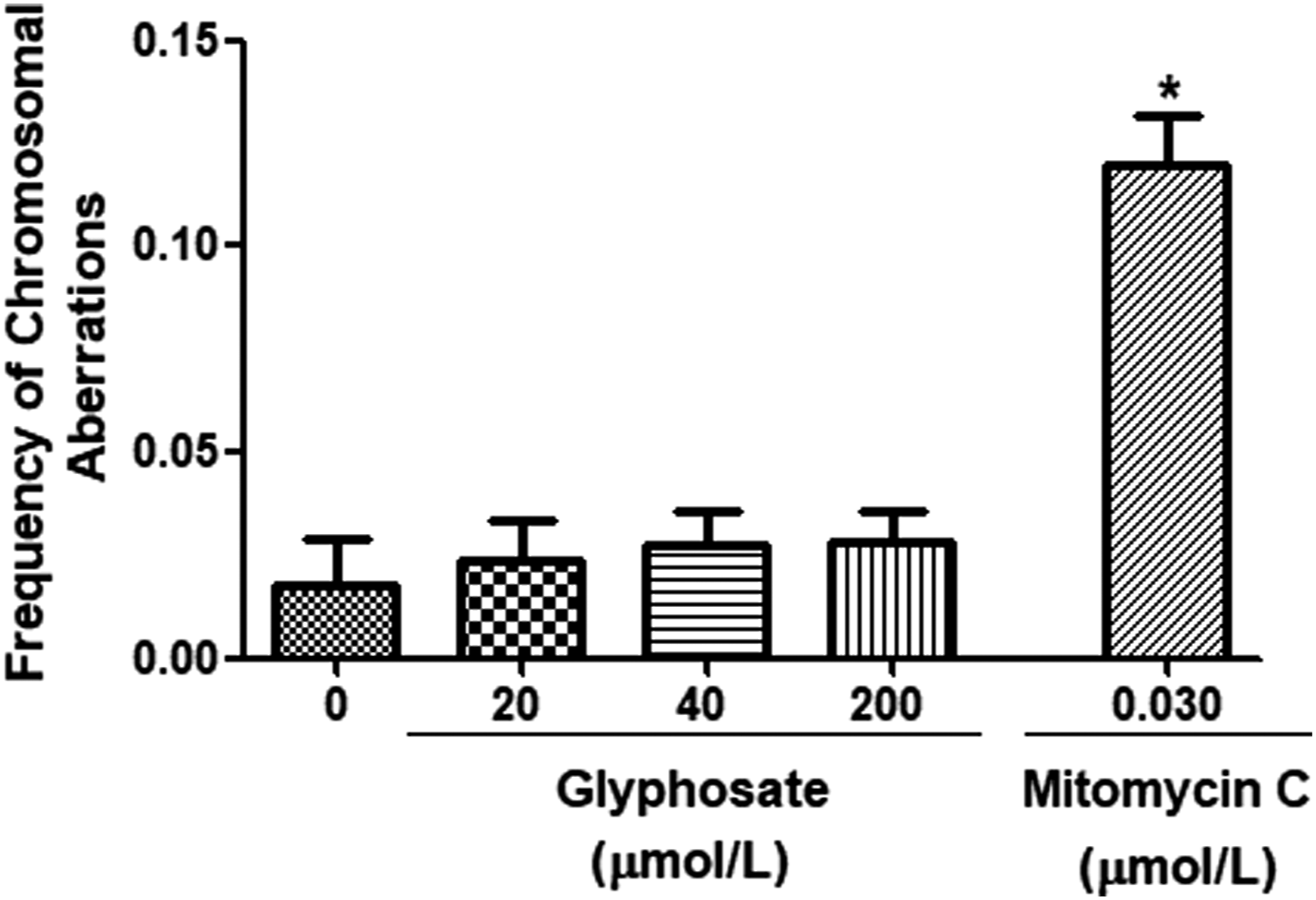
Induction of oxidative DNA damage by glyphosate was examined using 8-OHdG assay. Similar to CA, glyphosate did not cause any changes in the levels of 8-OHdG at all examined concentrations (P = .98) (Figure 3) Levels of oxidative stress biomarker (8-OHdG) induced by glyphosate in cultured human lymphocytes (n = 5) with different concentrations of glyphosate (20, 40, and 200 μmol/L).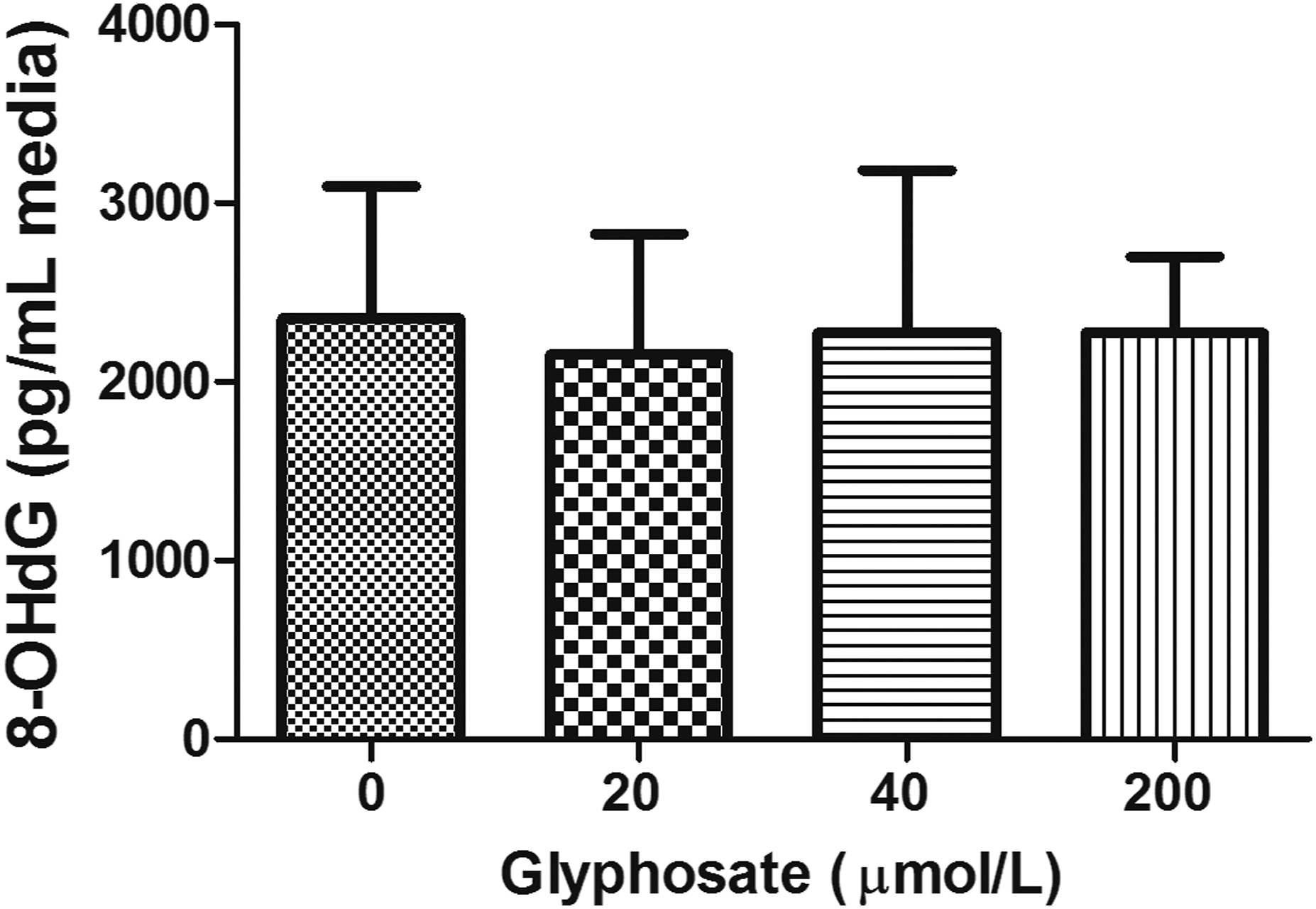
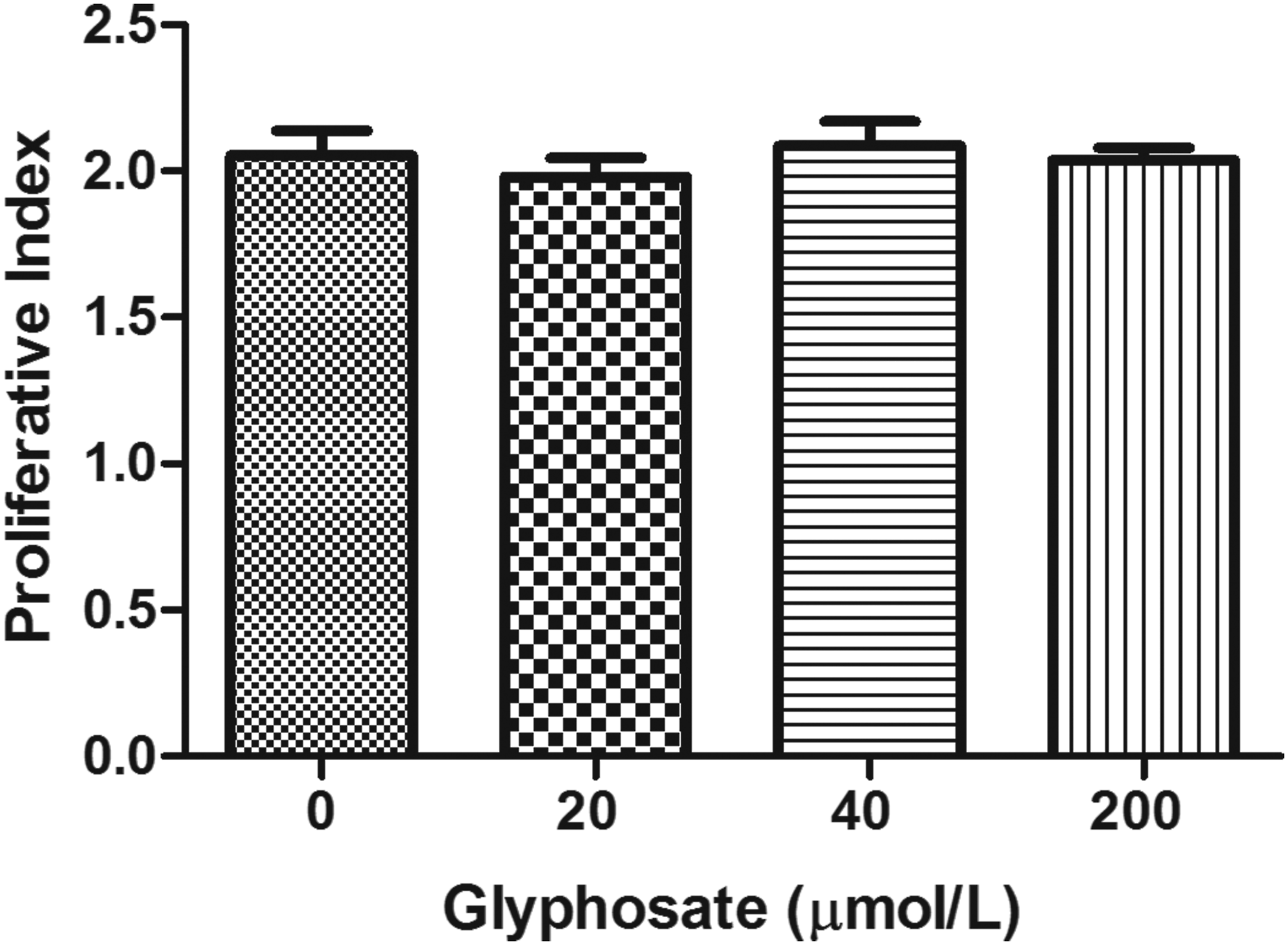
To examine cytotoxicity of glyphosate, MI and PI were evaluated (Figures 4 and 5, respectively). No significant changes in either indices were observed at all examined concentrations (P > .05). The mitotic index (MI) of cultured human lymphocytes (n = 5) after treatment with different concentrations of glyphosate (20, 40, and 200 μmol/L). The proliferative index (PI) of cultured human lymphocytes (n = 5) after treatment with different concentrations of glyphosate (20, 40, and 200 μmol/L).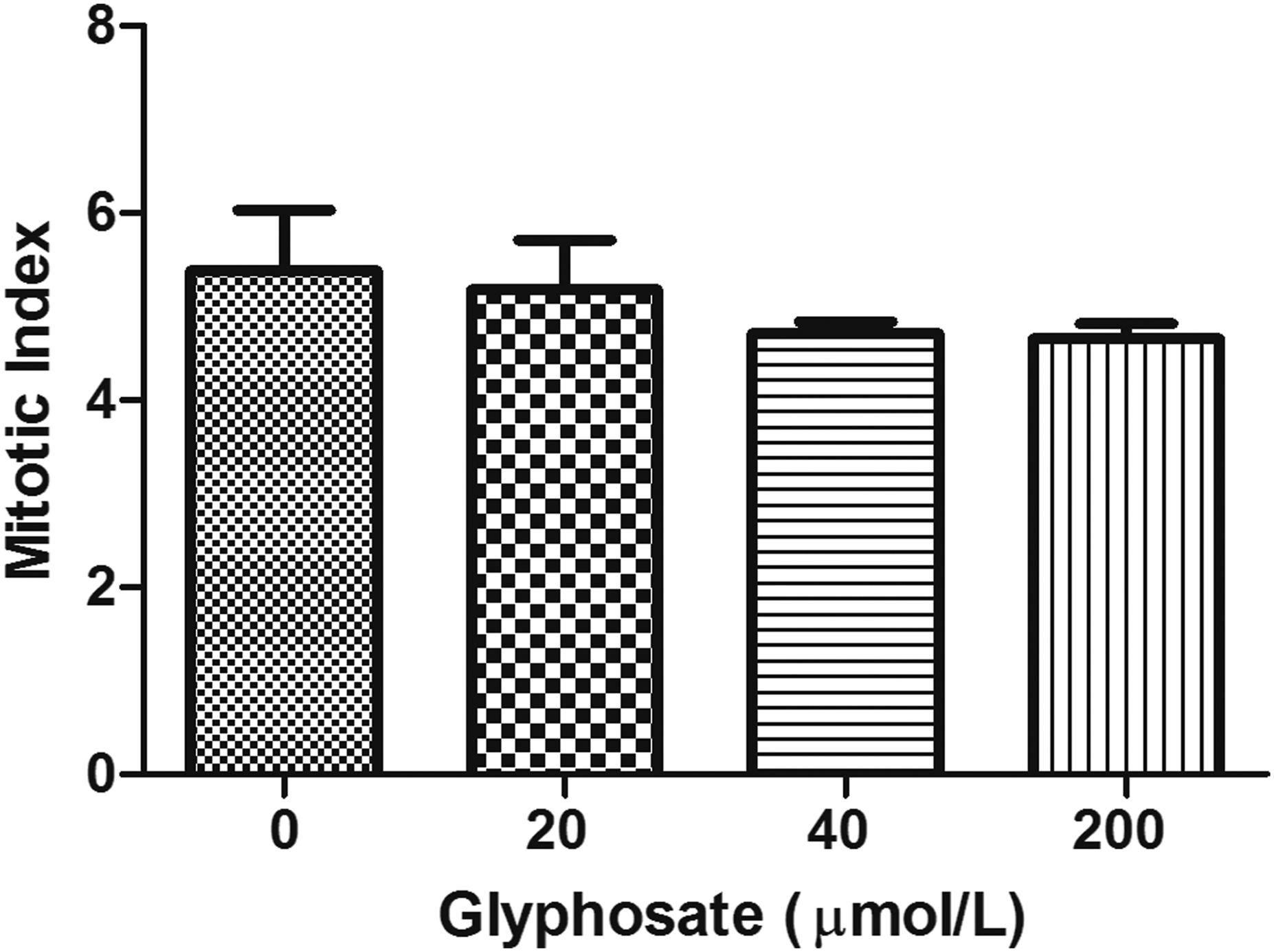
Discussion
Glyphosate consists of the amino acid glycine joined with a phosphonomethyl group [N-(phosphonomethyl) glycine]. Its herbicidal properties were discovered in 1970 by Monsanto company scientists. In plants and bacteria, glyphosate inhibits enolpyruvylshikimate phosphate synthase, the enzyme responsible for synthesizing the aromatic amino acids tryptophan, phenylalanine, and tyrosine, which results in inhibition of their growth and subsequently their death. 2 The main breakdown product of glyphosate in plants, soil, and water is amino methyl phosphoric acid. 13 No human health concerns were initially expressed regarding glyphosate product use, since there is no shikimic pathway within the human body. 31
To detect genotoxicity of glyphosate as well as cancer risk and DNA damage, CA and SCE tests are among the most frequently used cytogenetic assays. 32 Increased level of chromosomal breakage in blood lymphocytes appears to be a relevant biomarker of future cancer risk; this was based on 4 separate follow-up cohort studies which had performed CA analysis for large groups, then tracked if some of them developed different types of cancer.33‐36 Sister-chromatid exchange analysis has come into use as a sensitive manner to monitor DNA damage where increased frequency of spontaneous SCE has been reported in patients with various neoplastic diseases.21,22
In our study, glyphosate induced significantly higher levels (P < .05) of SCE at the highest used concentration (200 μmol/L). This is similar to various other studies that used glyphosate.8,9,34 Although all 3 studies concluded that it has a genotoxic effect, there was a major conflict with respect to the genotoxic concentration. Lioi, et al reported genotoxic significant effect in a range of 1.44–8.62 μg/ml (8.5–50.9 μmol/L) of glyphosate, 8 while Bolognesi, et al and Vigfusson and Vyse reported a significant genotoxic effect at concentrations higher than 250 μg/ml (1557 μmol/L).9,37 Also similar to the literature, no significant elevation in SCE levels was observed at the lower examined concentrations (20 and 40 μmol/L). This is important when considering that the level of glyphosate detected in human biological samples were found to be between 0.16 and 7.6 μg/L (0.9–44.95 μmol/L). 19 Therefore, although glyphosate does cause SCE, the concentration at which it causes it is not clinically relevant as these concentrations are higher than reported exposure levels in human.
Concerning CA, no significant changes in CA were detected at any examined concentrations (P = 0.86). While 1 study reported that glyphosate could induce CA at a concentration of 1.44 μg/ml (8.5 μmol/L), 8 and another study has found CA effect using concentrations of 100 and 200 μg/ml (622 and 1244 μmol/L), 38 numerous other reports concluded that there was no significant CA effect of glyphosate even at concentrations reaching 1250 μmol/L. 38 Using both in vivo and in vitro mammalian micronucleus gentoxicity assay, the majority of reviewed studies indicate that glyphosate is not genotoxic at a wide range of concentrations. Our study adds to the literature that even at the upper limit of our concentrations (200 μmol/L) no significant CAs occurred.
Similar to a study conducted by Santovito et al., we found no significant differences in MI or PI values, indicating that glyphosate did not impact the replicative capacity of the cells. 39 Nevertheless, a study on glyphosate effects on bovine lymphocytes observed a reduction of MI and PI at higher glyphosate concentrations 94.68 μg/mL and 189 μg/mL (560 and 1117 μmol/L). 40
Several studies have documented the ability of glyphosate to induce oxidative stress in human and animal cells. For example, glyphosate induced significant alterations to oxidative stress biomarkers in normal human prostate epithelial WPM-Y.1 cell line. 41 Similar findings were reported using mice,42,43 and C. elegans. 44 In the current study, glyphosate did not cause an increase in the induction of oxidative DNA damage, as our results show no changes in the levels of 8-OHdG at any examined concentrations.
The debate whether glyphosate causes genotoxicity even extends to observational studies. A meta-analysis on prospective cohort and case control done by Donato et al concluded no overall evidence of an increased risk for non-Hodgkin lymphoma (NHL) in subjects exposed to glyphosate during their occupation. 44 Nevertheless, a review conducted by Weisenburger concluded that glyphosate herbicides do have an association with NHL. 45 The extent of disagreement concerning the topic is evident by the fact that 2 reviews done within 1 year of each other have yielded opposing views.
To detect genotoxicity of glyphosate as well as cancer risk and DNA damage, CA and SCE tests are among the most frequently used cytogenetic assays. 32 Increased level of chromosomal breakage in blood lymphocytes appears to be a relevant biomarker of future cancer risk; this was based on 4 separate follow-up cohort studies which had performed CA analysis for large groups, then tracked if some of them developed different types of cancer.33‐36 Sister-chromatid exchange analysis has come into use as a sensitive manner to monitor DNA damage where increased frequency of spontaneous SCE has been reported in patients with various neoplastic diseases.21,22
Among the limitations of this is the range of glyphosate concentrations tested. Increasing the range and concentrations could have given insight on whether extremely high concentrations could cause even further genotoxicity. Also, adding other concentrations could have given insight into whether concentrations between 40 and 200 μmol/L lead to higher levels of SCE.
To conclude, our results collectively indicate a lack of genotoxicity and cytotoxicity of glyphosate in cultured human lymphocytes when dealing with environmentally relevant concentrations (20 and 40 μmol/L). However, being exposed to a higher concentration (200 μmol/L) led to higher level of SCE. Therefore, we recommend cautionary measures when dealing with glyphosate-based herbicides for individuals, such as farmers, who may be extensively exposed to highly concentrated solutions of these herbicides. We also recommend that further experimental and observational studies are conducted in order for health officials and agencies to be able to form informed decisions concerning placed regulations and restrictions on glyphosate-based herbicides.
Footnotes
Acknowledgments
The authors would like to thank our donors for their time and effort to donate blood to make this project possible.
Author Contributions
Abu Tarboush, N, contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Almomani, D, contributed to conception and design; contributed to acquisition and analysis; drafted manuscript; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Azzam, M, contributed to conception and design; contributed to analysis and interpretation; drafted manuscript; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Khabour, O, contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is funded by the Deanship of Academic Research—University of Jordan
