Abstract
Osteosarcoma (OS) is a malignant bone tumor characterized by poor prognosis due to its regional invasion and early metastasis. In this study, we aimed to find the role and the underlying mechanism of Cathepsin E (CTSE) in OS growth and metastasis. We found CTSE is upregulated in metastatic OS, rather than in the primary lesion, as confirmed by RT-qPCR and western blot analysis of clinical OS samples. Furthermore, both in vitro and in vivo experiments illustrated that CTSE promoted both growth and metastasis of OS cells, partially mediated through the modulation of Epithelial–Mesenchymal Transition (EMT). Bioinformatics analysis predicted that miR-185-5p downregulates CTSE via directly binding to the 3’UTR of CTSE, which was verified by luciferase reporter assay and rescue assays. This study reported for the first time that CTSE is a potential biomarker in OS tumorigenesis and metastasis, providing a promising therapeutic target for OS treatment.
Introduction
Osteosarcoma (OS) is the most common primary bone malignant tumor in children and adolescents, accounting for around 60% of primary bone malignancies in the first two decades of life. 1 Rapid development, regional invasion, and early pulmonary metastasis renders OS the second highest cause of cancer-related death in this age group.2,3 Over the past three decades, integrated therapy encompassing primary chemotherapy, tumor excision and multi-agent adjuvant chemotherapy has replaced limb amputation as the most commonly used therapeutic regimen for OS,4,5 but no further advances have been achieved during the past few years.
Cathepsin E (CTSE) is an intracellular aspartate protease belonging to the cathepsin family, which is mainly identified in the antigen-presenting immune cells, 6 including dendritic cells (DCs) and macrophages, indicating its potential role in immunological defense against cancer cells.7-9 In addition, it was found that CTSE could specifically catalyze the proteolytic release of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) from human prostate carcinoma tumor cells, leading to growth arrest and apoptosis. 10 However, it was also proposed that CTSE was involved in carcinogenesis and was highly upregulated in gastric adenocarcinoma, indicating that CTSE was a marker of both gastric differentiation and signet-ring cell carcinoma. 11 Indeed, its activity level was negatively associated with the prognosis of breast cancer. 12
These observations and limited evidence prompted us to ascertain the critical role of CTSE in OS. In the present study, we found that CTSE may be a determinant in OS metastasis. Further research showed that CTSE enhanced proliferation and migration both in vivo and in vitro under the regulation of miR-185-5p. We thereby concluded that CTSE could serve as a potential regulator of tumorigenicity and metastasis in OS.
Materials and Methods
Collection of Clinical Tissue Samples
A total of 14 pairs of primary OS and metastatic OS clinical tissue samples as well as 28 cases non-tumor tissues were obtained from the Changsha Central Hospital. All the patients were diagnosed with OS clinically and pathologically. The research subjects and their families were informed and they signed a fully-informed consent form. The research was approved by the Ethics Committees of Changsha Central Hospital.
RT-qPCR
Total RNA was isolated from the tumor cells or tissues using the TRIzol Reagent (Invitrogen, Thermo Fisher Scientific, Inc. USA), according to the manufacturer’s protocol. For the quantitative detection of mRNA expression, mRNA was reverse-transcribed into cDNA and amplified according to the manufacturer’s protocol (Biouniquer Technology, Nanjing, China). The reaction was performed under the following conditions: 37°C for 15 min and 98°C for 5 min. 2−ΔΔCt method was used for data analysis normalized to β-actin. U6 was used as endogenous control for the analysis of miR-185-5p expression. Primer sequences were as follows:CTSE forward primer 5’-AGGCATCCGTCCCTCAAGAA-3’, Reverse Primer 5’-CCTTGGCACTCTGGTCCATTG-3’; β-actin sense primer 5′-GTCCCTCACCCTCCCAAAAG-3’, and antisense primer 5′-GCTGCCTCAACACCTCAACCC-3’; miR-185-5p sense primer: 5’-GTGGAGAGAAAGGCAGTTC-3’, antisense primer:5′-CGCTTCACGAATTTGCGTGTCAT-3’; U6 sense primer: 5′-GCTTCGGCAGCACATATACTAAAAT-3, antisense primer: 5′-CGCTTCACGAATTTGCGTGTCAT-3’;
Western Blot
The tumor cells or tissues were washed with PBS 3 times and lysed in RIPA buffer (Beyotime Institute of Biotechnology, Beijing, China) with protease inhibitor and phosphoric-acid protease inhibitor (Fdbio science, Tianjing, China) at 4°C for 30 min. Then the lysates were centrifuged at 12000 r/min for 15 min at 4°C. The proteins were then separated on SDS-PAGE gel and electro-transferred to polyvinylidene fluoride membranes (EMD Millipore, Billerica, MA, USA). The membranes were blocked with 5% nonfat milk in Tris Buffered Saline with Tween 20 (TBST)(pH 7.0), then incubated with primary antibodies, including CTSE, E-cadherin, N-cadherin, and GAPDH antibodies, at 4°C overnight. An HRP (horseradish peroxidase)-conjugated anti-rabbit IgG antibody was consecutively used as the secondary antibody. Immunoreactive proteins were detected using enhanced chemiluminescence reagents (Pierce, Rockford, IL, USA). All experiments were independently performed in triplicate.
Cell Culture
Human OS cell lines HOS, U2OS, KRIB, and KHOS were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). All cell lines were grown in Dulbecco’s modified Eagle’s medium (DMEM) (GIBICO, USA) supplemented with 10% fetal bovine serum (FBS) (GIBICO, USA), which was designated as total culture medium, and incubated in a humidified atmosphere of 5% CO2 at 37°C.
Cell Transfection
CTSE overexpression plasmid, short hairpin RNA (shRNA) targeting CTSE and negative control (sh-NC) were synthesized by Shanghai Genechem (Shanghai, China). Lentivirus packing expression plasmids were co-transfected into 293T cells for 48 h to generate stable cell lines. Following this, KHOS and HOS cell lines were infected with the supernatants containing the virus for 48 h. After infection, stably infected cells were selected using puromycin (1 μg/mL) and further confirmed by RT-qPCR assay. The cells (10
6
/well??) in six-well plate were transfected with 4 μg CTSE-overexpressed or control plasmids, constructed by Vigene Bioscienses (Shangdong, China), using Lipofectamine 2000 reagent. miR-185-5p mimics, inhibitor, and corresponding controls (miR1N0000001-1-10, miR2N0000001-1-5) were synthesized by Guangzhou RiboBio Co., Ltd. (Guangzhou, China). These reagents were transfected into OS cell lines at final concentrations of 50 nM in a 6-well plate. All transfections were performed using Lipofectamine 2000 Transfection Reagent (Invitrogen) in accordance with the manufacturer’s instructions. The cell transfection was carried out according to the manufacturer’s instructions. The expression of CTSE in transfected cells was evaluated by RT-qPCR and western blot analysis. The sequences were listed in the Supplementary Tables 1 and 2
CCK8 Assay
Approximately 5000 cells per well were seeded into 96-well plates with a volume of 200 μl culture medium. Subsequently, 10 μl of CCK8 reagent (Bimake, China) mixed with 90 μl DMEM medium without FBS was added into each well and plates were incubated in a humidified atmosphere of 5% CO2 at 37°C for 1 h. Finally, the absorbance value (OD) was measured at 450 nm using a Varioskan LUX Multimode Microplate Reader (Thermofisher, USA). The observations were carried out every 24 hours for 3 days.
Colony Forming Assay
KHOS and HOS cells were plated into 12-well plates with 400 cells per well and cultured in a humidified atmosphere of 5% CO2 at 37°C for 2 weeks, and then fixed with methanol for 30 min and stained with 1% crystal violet for 1 min. All assays were independently performed in triplicate.
EdU Assay
KHOS and HOS cells were cultured in 24-well plates and added to EdU (Invitrogen, Cat. A10044, USA) as recommended dose according to the standard EdU (5-ethynyl-2’-deoxyuridine) assay protocol and imaged by fluorescence microscopy.
Wound Healing Assay
Transfected cells were first cultured in 6-well plates. Three parallel scratch wounds across each well were made using P-10 pipette tips and the culture was allowed to reach 90% confluence. Fresh medium supplemented with 2% FBS was then added, and photographs were taken at 0 and 24 h, respectively. All assays were independently performed in triplicate.
Transwell
To assess the invasive capacity of KHOS and HOS cells, serum-free DMEM was mixed with AGS and SNU16 cells in transwell chambers coated with Matrigel (BD, United States).For both migration and invasion analysis, the bottom chamber contained complete medium, and KHOS and HOS cells were maintained in it for 24 h. Next, 4% paraformaldehyde was used to fix cells, followed by staining using 1% crystal violet. Photographs were obtained and cells number were measured using a microscope (Zeiss, Germany).
Subcutaneous Xenograft and Metastasis Models
Female BALB/c nude mice (4-5-weeks-old) were obtained from Charles River Laboratories (Beijing, China) and maintained in a pathogen-free condition in accordance with relevant guidelines and regulations for the care and use of laboratory animals. About 3 × 106 KHOS cells transfected with sh-NC or sh-CTSE were subcutaneously injected into a group of six nude mice with 0.1 mL DMEM medium, respectively. Tumor volume was calculated every 7 days. The animal experiment was approved by the Institutional Animal Care and Use Committee of Changsha Central Hospital. The mice were euthanized at day 42, and excised tumor tissues were weighed and measured.
For the tumor metastasis model, 1 × 106 OS cell lines or control cell lines were injected into the tail vein of nude mice. Animals were euthanized 30 days later, lungs were removed, and hematoxylin and eosin (H&E) staining was conducted.
Immunohistochemistry (IHC)
Immunohistochemical staining was performed for the measurement of EMT-related proteins. Briefly, the primary antibody of E-Cadherin (1:2000, ab40772, abcam, UK) and N-Cadherin (1:2000, ab98952, abcam, UK) were added to the specimen slices and incubated at 4°C overnight The sections were washed with PBS and incubated with the secondary antibodies (1:8000, A0279, Beyotime, China; 1:8000,A0286, Beyotime, China) labeled with biotin for 10 min. Then, slides were incubated with 3,3’-diaminobenzidine substrate (DAB) to view immunolocalization, and hematoxylin was used to counterstain. The optical microscopy was performed for the analysis.
Luciferase Reporter Assay
Luciferase reporter assay was performed using Dual-Luciferase Reporter Assay Kit (Promega Corporation E1910, Madison, WI, USA). The 3’-UTR sequence region of CTSE was amplified by PCR using the PCR Amplification kit (Takara Biotechnology, Shanghai, China). Three luciferase reporter recombinant vectors were evaluated: wild-type 3'UTR of CTSE gene (pGL3-CTSE wt), mutated 3’UTR of CTSE gene (pGL3-CTSE mut), and a wild-type 3’UTR as control (pGL3-con). The KHOS cells were co-transfected with vectors and hsa-185-5p mimic (or miRNA mimics control, NC group) (GenePharma Co., Ltd. Suzhou, China) using Lipofectamine® 2000 (Thermo Fisher Scientific, USA).
Statistical Analysis
Statistical analysis provided in the paper was analyzed with SPSS 18.0 software (SPSS, Chicago, USA). One-way ANOVA or two-tailed Student’s t-test was used for comparisons between groups. A value of P < .05 was considered significant.
Results
CTSE Is Differentially Expressed in Primary and Metastatic OS
We first analyzed the expression level of CTSE in OS tissues and non-OS tissues and found higher CTSE expression was identified in OS than in non-tumor tissues (Figure 1A). We extended our research to clarify the impact of CTSE on OS metastasis. Based on RT-qPCR analysis of 14 pairs of clinical samples, it was observed that higher level of CTSE was detected in the metastatic OS than in the primary 1 (Figure 1B). In line with this, significant difference was illustrated even at protein level (Figures 1C and 1D). Utilizing pertinent OS cell lines, we further confirmed higher CTSE expression in KRIB and KHOS rather than HOS and U2OS, wherein the former were metastatic cell lines and the latter were non-metastatic (Figures 1(E) and (F)). These results indicated that CTSE may be a determinant in OS metastasis. CTSE is differentially expressed in primary and metastatic OS. (A) RT-qPCR was performed to analyze the expression of CTSE in osteosarcoma samples and non-tumor tissues.(B) RT-qPCR was performed to analyze the expression of CTSE in 14 pairs of clinical human osteosarcoma samples involving both primary and metastatic origin. (C) and (D) Western blot was performed to analyze the expression of CTSE in clinical human osteosarcoma samples involving paired primary and metastatic origin. M, metastatic; P, primary. (E) and (F) Western blot and RT-qPCR were performed to analyze the expression of CTSE in metastatic osteosarcoma cell lines and non-metastatic osteosarcoma cell lines. P < .05, **P < 0.01. OS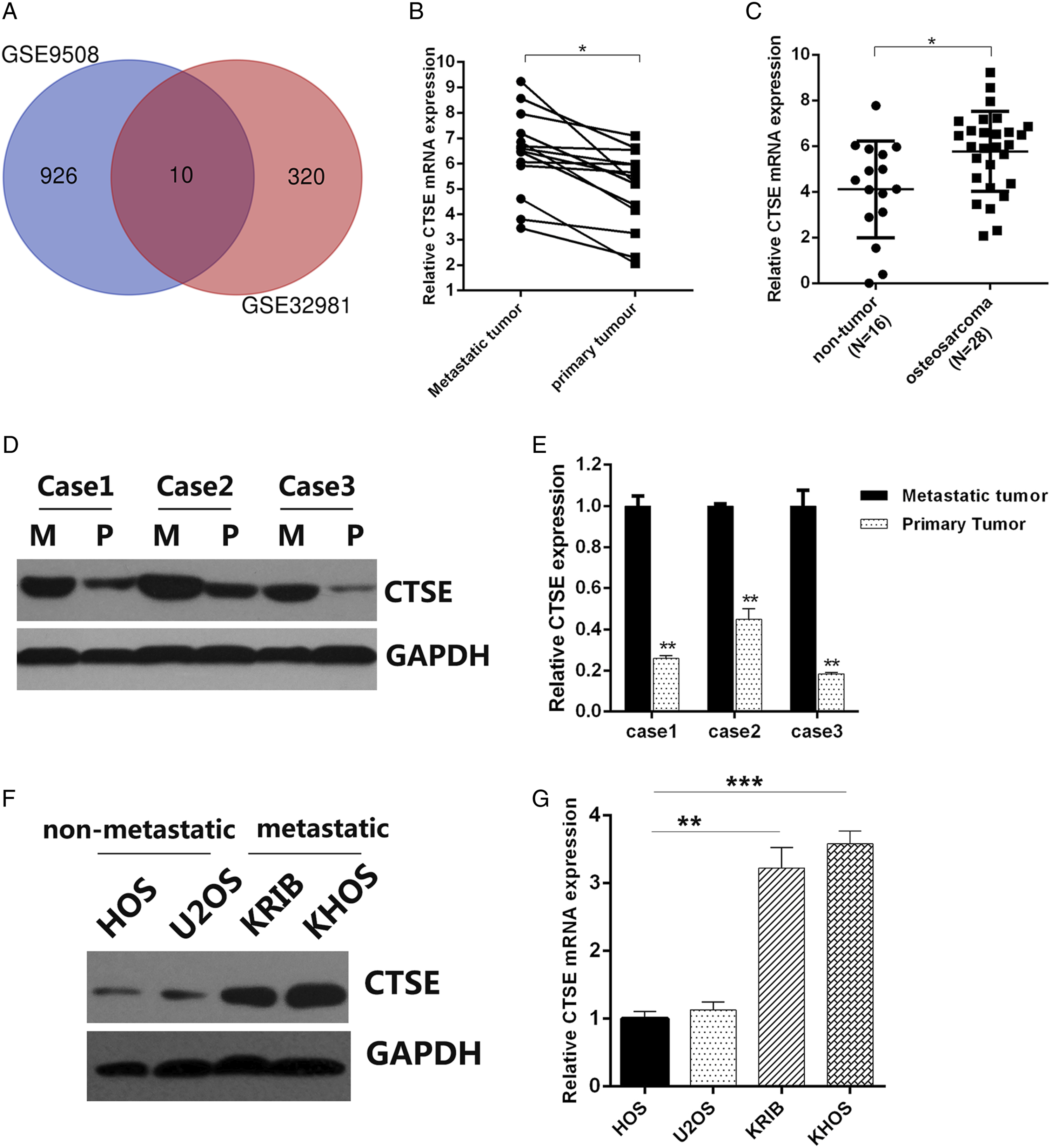
CTSE Promotes Proliferation of OS Cell Lines
Considering the different CTSE levels seen between metastatic and non-metastatic OS cell lines, KHOS and HOS were chosen for further experiments. sh-RNA targeting CTSE and CTSE overexpressing plasmid were established and were transfected into KHOS and HOS, respectively. Western blotting confirmed that sh-CTSE significantly abolished CTSE expression in KHOS (Figure 2A and 2B), and CTSE plasmid successfully overexpressed CTSE in HOS (Figure 2C and 2D). The impact of CTSE on proliferation was then elucidated. Knockdown of CTSE led to inhibition of proliferation (Figure 2E), while overexpression of CTSE led to enhanced proliferation (Figure 2F). In addition, colony formation assay showed that in comparison to the counterpart NC or vector group, inhibition of CTSE resulted in diminished colony forming ability (Figure 2G), and increased CTSE expression resulted in higher colony forming ability (Figure 2H). In addition, the promoted effect of CTSE on OS proliferation was also confirmed by EdU assays (Figure 2I and J). CTSE promotes proliferation of OS cell lines. (A) and (B) Western blot and RT-qPCR were performed to confirm the inhibition effect of CTSE by shRNA in KHOS cells. (C) and (D) Western blot and RT-qPCR were performed to confirm the overexpression of CTSE by transfection of CTSE plasmids in HOS cells. (E) and (F) CCK8 assay was performed to analyze the growth rates of transfected KHOS and HOS cells, respectively. (G) and (H) Colony forming assay was performed to assess the colony forming ability in transfected KHOS (G) cell and HOS (H) cells. (I) and (J) EdU assay was performed to assess the proliferaion in transfected KHOS (I) cell and HOS (J) cells P* < .05, **P < .01. OS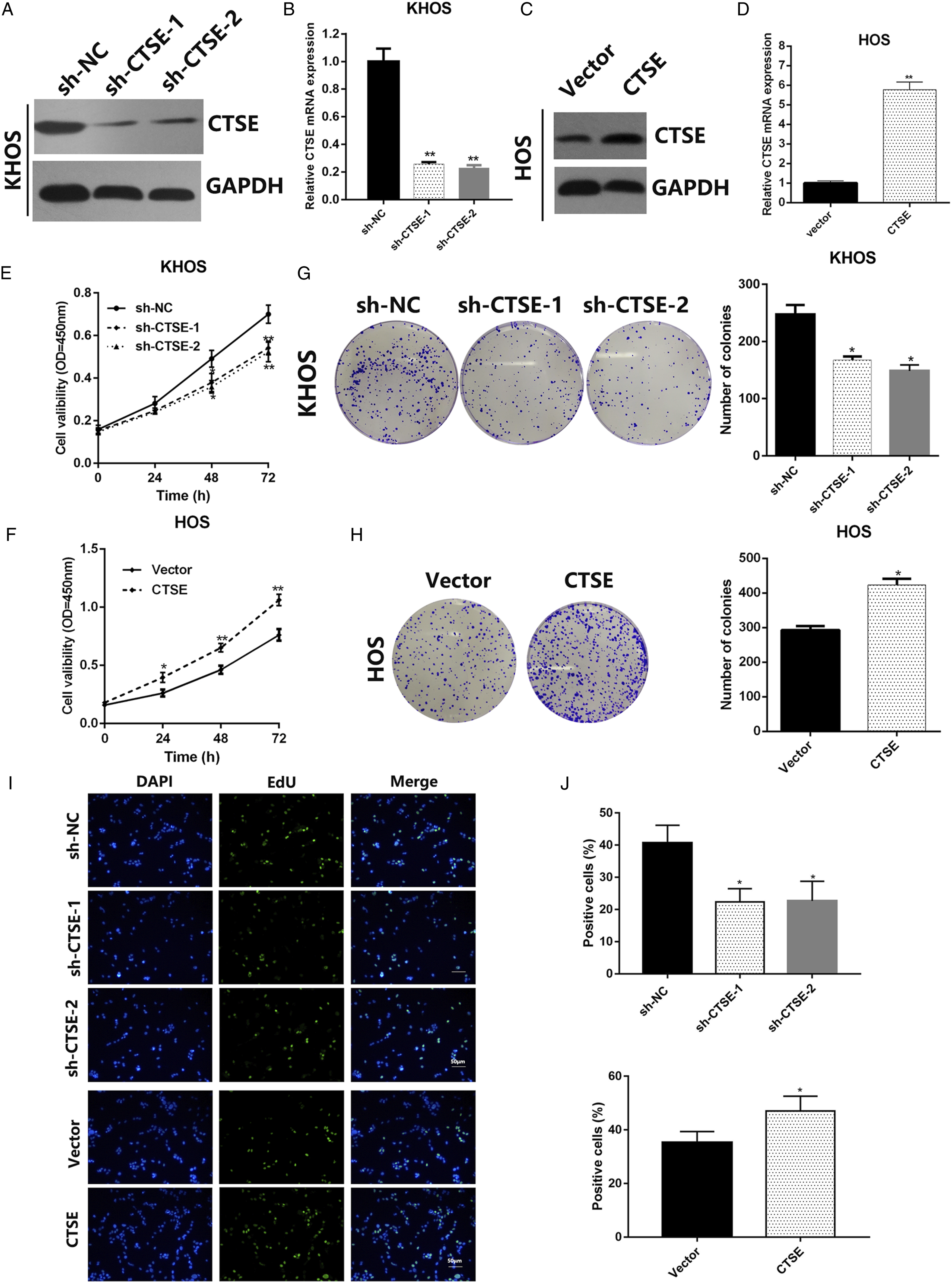
CTSE Promotes Migration of OS Cell Lines
In the next step, we further investigated whether CTSE was involved in migration of tumor cells. In the transwell assay and wound healing assay, knockdown of CTSE resulted in decrease in the number of migration and invasion of tumor cells (Figure 3A and B). In contrast, transfection of CTSE overexpressing plasmid promoted migration and invasion of OS cells (Figure 3C and D). Furthermore, EMT-related proteins were also detected by western blot, CTSE overexpression promoted the epithelial–mesenchymal transition process (Figure 3E and F), which might have led to the increased migration of OS. CTSE promotes migration of OS cell lines. (A) Transwell assay was performed to assess the migration ability in transfected KHOS cells. (B) Wound healing assay was performed to assess the migration ability in transfected KHOS cells. (C) Transwell assay was performed to assess the migration ability in transfected HOS cells. (D) Wound healing assay was performed to assess the migration ability in transfected HOS cells. (E) and (F) Western blot was performed to detect the E-cadherin and N-cadherin in transfected KHOS and HOS cell lines. P* < .05, **P < .01. OS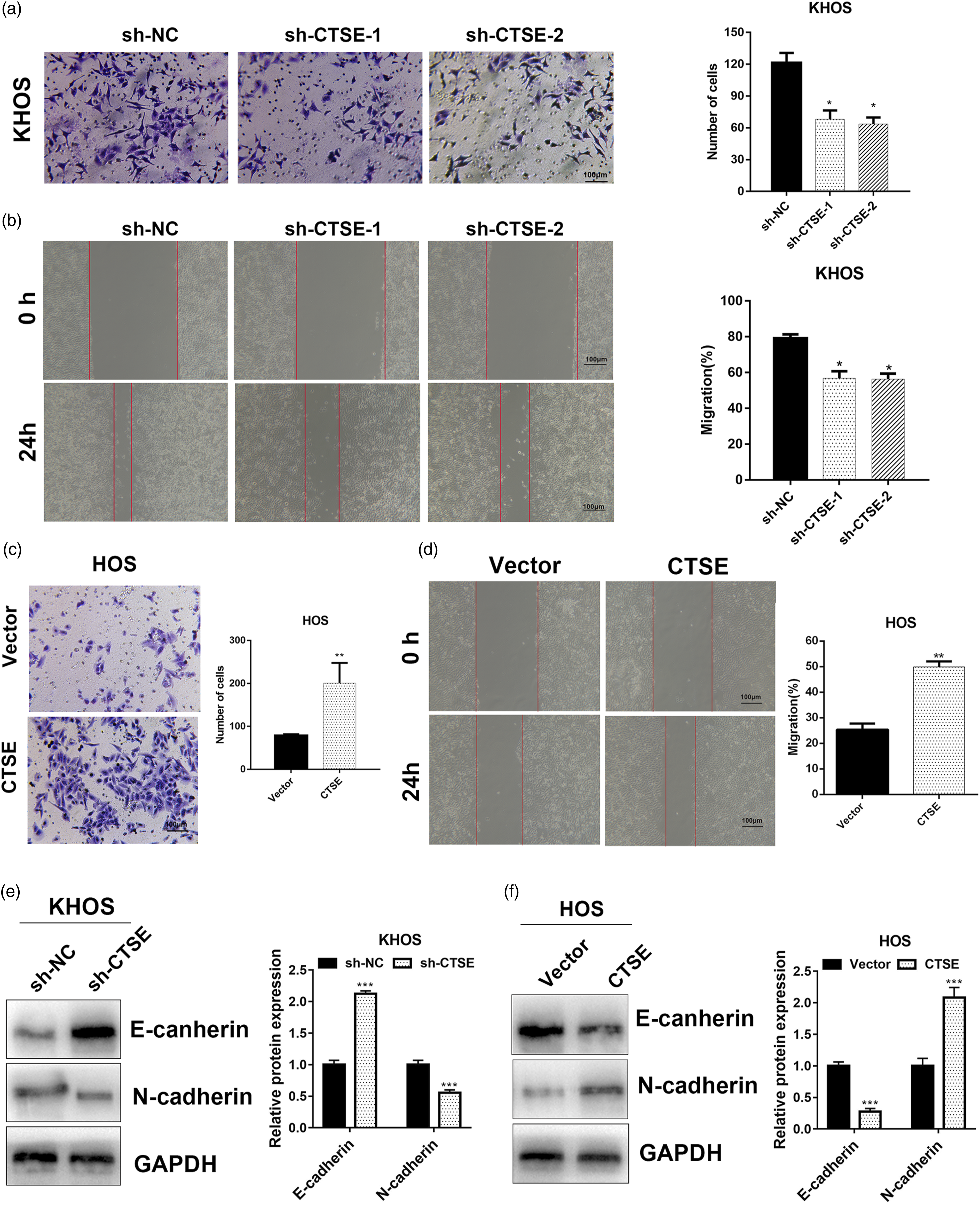
CTSE Promotes Tumorigenesis and Metastasis of OS in Vivo
We also implemented the xenograft model to characterize the influence of CTSE on OS. Nude mice were divided into NC group and sh-CTSE group comprising of 6 mice per group. Subcutaneous xenograft model was established using KHOS cells transfected with sh-NC or sh-CTSE, respectively. In line with previous results, CTSE knockdown decreased the tumorigenicity of KHOS cells (Figure 4A), as confirmed by consecutive measurement of the tumor volume every 7 days after transplantation (Figure 4B). Both the weight and volume of the sh-NC group were significantly higher than those of the sh-CTSE group (Figure 4C). To further uncover the probable molecular changes, the expression of EMT-related proteins was examined in the xenograft tumors using IHC and Western blot. In the sh-CTSE group, CTSE levels and N-cadherin levels were significantly decreased, whereas the expression of E-cadherin was upregulated, indicating that CTSE could promote tumorigenesis and metastasis in OS (Figure 4E). Moreover, we explored the role of CTSE in tumor metastasis in vivo via a lung metastasis model. The results showed CTSE knocking down significantly decreased the number of metastatic nodules, as confirmed by stained lung sections (Figures 4F and G). CTSE promotes migration of OS cell lines. (A) Subcutaneous xenograft model was established using KHOS cells transfected with sh-CTSE or sh-NC. (B) The tumor volume of the xenograft was measured every 7 days until day 42. (C) The weight and volume of the xenograft tumor was measured at day 42. (D) The expression of E-cadherin and N-cadherin in the xenograft tumors. (E) Western blot was performed to analyze the expression of CTSE, E-cadherin and N-cadherin in 3 samples from each xenograft group, respectively. (F) and (G) Representative images of metastatic lungs (F), representative HE images (G) and numbers of metastatic foci per lung (H) injected with KHOS cells stably silencing CTSE or empty vector (n = 5). P* < .05, **P < .01. OS, Osteosarcoma; CTSE, Cathepsin E.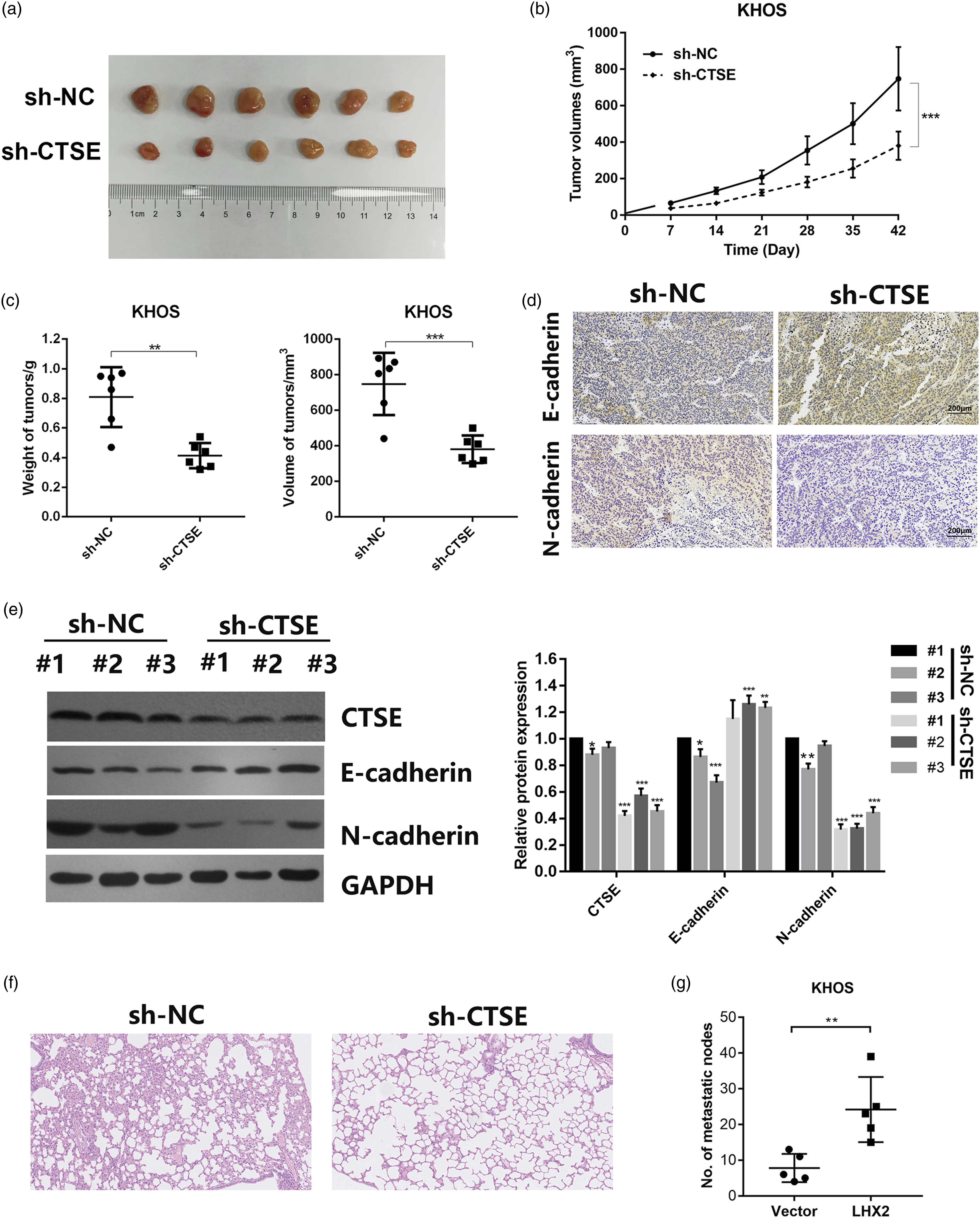
miR-185-5p Targets CTSE in the Regulation of Osteosarcoma Progression
To better understand the regulating mechanism of CTSE in OS, we used the TargetScan online platform to identify the potential upstream miRNAs which might target CTSE. miR-185-5p, reported as a suppressor gene in various cancers, was identified as an promising candidate. Western blot and RT-qPCR demonstrated that transfection of miR-185-5p mimics significantly decreased CTSE level (P < .05), while the administration of miR-185-5p inhibitor significantly upregulated CTSE level (P < .01) (Figures 5A to C). Luciferase reporter assay indicated that miR-185-5p mimics significantly decreased the luciferase activity of wild-type CTSE reporter, while no statistic difference was found on the mutant CTSE reporter (Figure 5D and E). Furthermore, a negative correlation between miR-185-5p levels and CTSE mRNA levels in OS was confirmed (Figure 5F). Next, we applied rescue experiments to verify the role of miR-185-5p in the regulation of CTSE during OS progression. miR-185-5p mimics greatly weakened the proliferative ability of KHOS cells while the extra administration of CTSE plasmid partially reversed this effect (Figure 5G). In the wound healing and Transwell assay, miR-185-5p overexpression significantly harmed the cellular migration ability in KHOS cell lines, whereas upon co-transfection with CTSE, the migration ability was found to return (Figure 5H and I). In summary, these results revealed miR-185-5p inhibits the metastasis process in OS by regulating CTSE. miR-185-5p targets CTSE in the regulation of Osteosarcoma progression. (A) RT-qPCR were performed to confirm the expression of miR-185-5p in KHOS cells after transfection of miR-185-5p mimics and inhibitor. (B) and (C) Western blot and RT-qPCR were performed to detect the mRNA level of CTSE in HKOS cells transfected with miR-185-5p mimics or inhibitor. (D) Schematic diagram of the miR-185-5p putative binding site and mutant site in 3’ UTR of CTSE. (E) Dual-luciferase assay was used to detect the luciferase activity in KHOS cells transfected with pGL3-con, pGL3-CTSE wt or pGL3-CTSE mut in the presence of mimics-NC or miR-185-5p mimics.(F) The correlation between the expression of miR-185-5p and CTSE mRNA in Osteosarcoma samples. (G) CCK8 assay was performed to detect the proliferation rate of KHOS cells transfected with miR-185-5p alone or co-transfected with CTSE plasmid. (H) Transwell assay was performed to evaluate the metastasis ability of KHOS cells transfected with miR-185-5p alone or co-transfected with CTSE plasmid. (I) Wound healing assay was performed to evaluate the metastasis ability of KHOS cells transfected with miR-185-5p alone or co-transfected with CTSE plasmid. P* < 0.05, **P < 0.01. CTSE, Cathepsin E.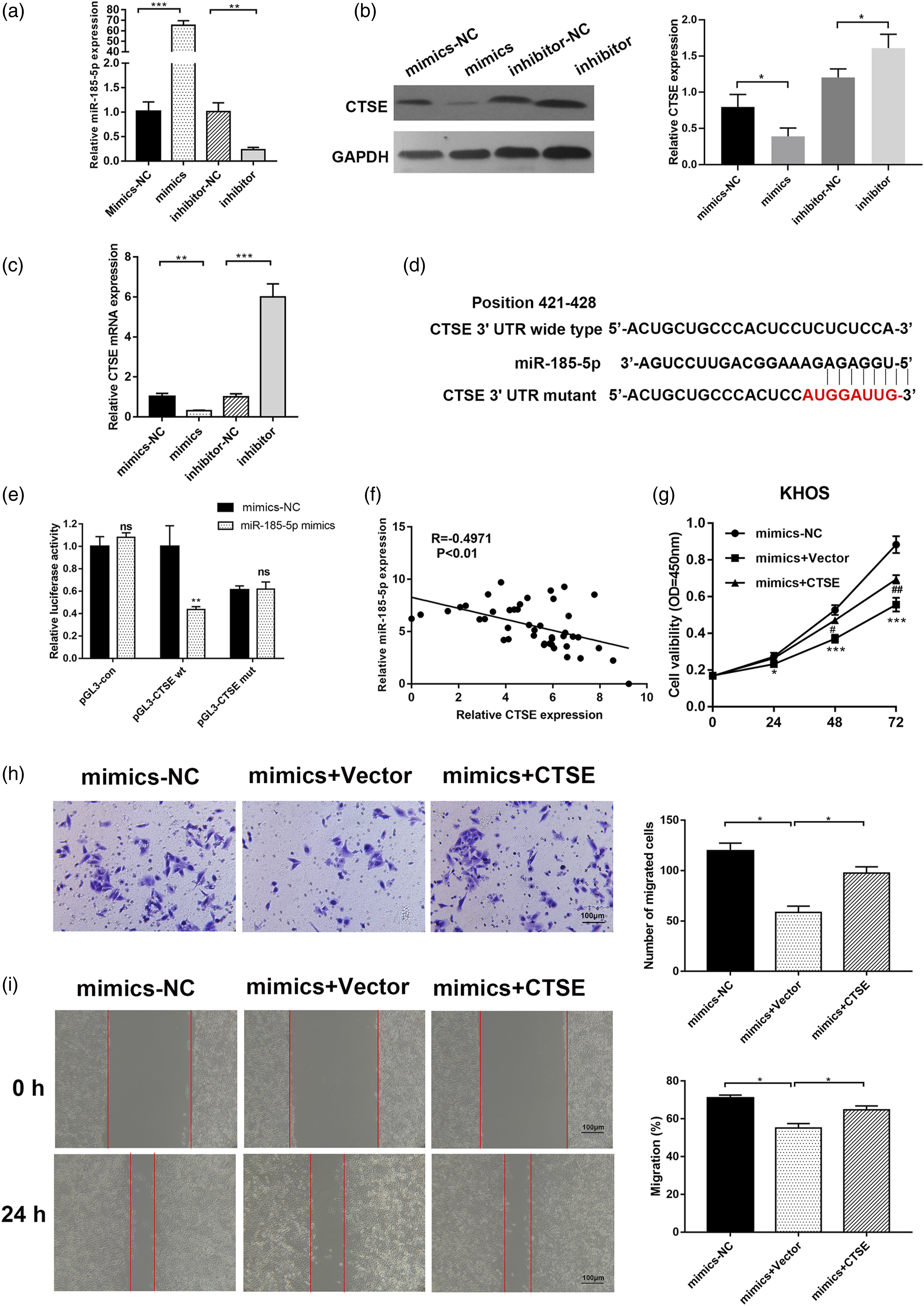
Discussion
In this study, we aimed to reveal the pivotal molecular changes between primary and metastatic OS. Utilizing gene expression profiling data from GEO, we proposed that CTSE was upregulated in metastatic OS rather than in primary OS. This result was first determined the expression of CTSE in clinical OS tissues and pertinent OS cell lines by western blot and RT-qPCR analysis. We then extended our research to confirm the effect of CTSE on biological characteristics of OS including proliferation and migration in both metastatic and non-metastatic cell lines. It was illustrated that CTSE could promote both proliferation and migration in OS in vitro, and subcutaneous xenograft and metastasis models further confirmed that knockdown of CTSE could reverse the EMT process and diminish the tumorigenicity of OS cell lines. In theory, miR-185-5p could target CTSE via binding to its 3’UTR region, thus regulating the progression of OS.
CTSE has emerged as a novel tumor biomarker in previous studies.13,14 CTSE expression was mainly identified in gastrointestinal superficial epithelial cells, and it is considered to be an effective marker for pancreatic ductal adenocarcinoma diagnosis.15-17 While CTSE was significantly associated with the progression of non-muscle invasive bladder cancer, 18 it could induce apoptosis and cell cycle arrest by inducing the release of TRAIL, and a combination of CTSE and doxorubicin could effectively inhibit the proliferation of chemo-resistant prostate cancer cells. 19 However, the role of CTSE in OS remained elusive, and our study fills this gap showing that CTSE was a potential biomarker in OS and could promote its malignancy partially through the modulation of EMT process.
Moreover, CTSE was previously reported to be involved in antigen processing and innate immunity,20-22 indicating that CTSE could be a probable target to enhance immune response. Immunotherapy is becoming a promising therapeutic regimen for several malignant tumors, such as melanoma, myeloma, and breast cancer.23-25 Though the data about OS remains limited, compromised antigen-presenting activity was found in chemo-resistant OS. 26 Further study focusing on the innate immune system found several promising targets. Dendritic-cells-fused tumor vaccine was proved to both stimulate the proliferation of T lymphocytes as well as induce their tumor-cytotoxic activity. 27 Activated natural killer cells were also proven to target OS tumor initiating cells and eliminate the tumor. 28 Moreover, antibodies targeting CTLA-4 and PD-1 have emerged as an effective treatment for OS.29,30 Nevertheless, conclusive studies considering CTSE as an immunotherapy target for OS are still limited.
While searching for the upstream regulator of CTSE in OS progression, we examined Targetscan (http://www.targetscan.org/vert_72/) and miRDB (http://mirdb.org/), online miRNA-mRNA prediction tools, and found that miR-185-5p could bind to the 3’ UTR of CTSE. miR-185-5p was reported to be downregulated in various cancers, and overexpression of miR-185-5p suppressed the malignant behavior of cancers.31-33 miR‐185‐5p was found to be downregulated in glioma tissue and was closely correlated with poor prognosis in glioma patients. Increasing miR-185-5p via transfecting mimics resulted in decreased cell proliferation and migration in glioma cells. 32 miR-185-5p suppressed human papillomavirus (HBV) replication by targeting ETS Transcription Factor ELK1 (ELK1), thus decreasing the morbidity of HBV-related liver cancer. 34 Long non-coding RNA RNCR3 promoted the development of prostate cancer by negative regulation of miR-185-5p. 33 In the present study, miR-185-5p proved to be a regulator of CTSE, and miR-185-5p mimics reversed the stimulation effect of CTSE in the proliferation and migration of OS. Our findings are helpful in understanding the pathogenesis of OS and in devising novel strategies for treatment of OS.
In summary, we concluded that CTSE is involved in OS metastasis and could prove vital in diagnosis of OS. CTSE appears to promote the proliferation and migration of OS cells under regulation of miR-185-5p. Further delineation of the molecular mechanisms by which CTSE regulates OS metastasis will impart a better understanding of OS and provide a promising therapeutic target for this malignancy.
Supplemental Material
sj-pdf-1-ijt-10.1177_10915818211069270 – Supplemental Material for miR-185-5p Represses Cells Growth and Metastasis of Osteosarcoma via Targeting Cathepsin E
Supplemental Material, sj-pdf-1-ijt-10.1177_10915818211069270 for miR-185-5p Represses Cells Growth and Metastasis of Osteosarcoma via Targeting Cathepsin E by Yue Wu, Weili Zhou, Zhijun Yang, Jinping Li and Yi Jin in International Journal of Toxicology
Supplemental Material
sj-pdf-2-ijt-10.1177_10915818211069270 – Supplemental Material for miR-185-5p Represses Cells Growth and Metastasis of Osteosarcoma via Targeting Cathepsin E
Supplemental Material, sj-pdf-2-ijt-10.1177_10915818211069270 for miR-185-5p Represses Cells Growth and Metastasis of Osteosarcoma via Targeting Cathepsin E by Yue Wu, Weili Zhou, Zhijun Yang, Jinping Li and Yi Jin in International Journal of Toxicology
Footnotes
Author Contributions
Wu, Y. contributed to conception and design and drafted manuscript; Zhou, W.L. contributed to acquisition, analysis, and interpretation; Yang, Z.J. contributed to acquisition, analysis, and interpretation; Li, J.P. contributed to acquisition, analysis, and interpretation; Jin, Y. contributed to conception and design and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplement material for this article is available in online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
