Abstract
Drug powder composition analysis is of particular interest in forensic investigations to identify illicit substance content, cutting agents and impurities. Powder profiling is difficult to implement due to multiple analytical methods requirement and remains a challenge for forensic toxicology laboratories. Furthermore, visualization tools allowing seizure products identification appear to be under-used to date. The aim of this study is to present the utility of molecular networking for the composition establishment of natural origin drugs. A powder suspected to contain heroin and three powders suspected to contain cocaine obtained from law enforcement agency seizures were analyzed using untargeted screening by liquid chromatography coupled with high-resolution mass spectrometry (LC-HRMS/MS). Molecular networking and metabolite annotation applied to suspected heroin sample allowed rapid confirmation of its illicit content (heroin), the identification of structurally related major impurities (6-monoacetylmorphine, 6-monoacetylcodeine, noscapine, and papaverine), as well as cutting agents (acetaminophen and caffeine). The cocaine powder profiling allowed the comparison of its constituents in a semi-quantitative manner (cocaine, benzoylecgonine, trans/cis-cinnamoylcocaine, trimethoxycocaine, hexanoylecgonine methylester, caffeine, hydroxyzine, levamisole, and phenacetin), bringing additional information for their identification, including geographically sourcing of natural product and their putative place in the supply chain. Although this approach does not replace the profiling techniques used by forensic laboratories, the use of molecular networks provides a visual overview of structurally related constituents which aids the comparison and investigation of seizure powders. Molecular networks offers here an ideal way to depict structurally related and unrelated compounds in these often complex mixtures of chemicals.
Introduction
In 2018, total drug seizures in the United States amounted over $64M mainly with cocaine, heroin, marijuana, methamphetamine, and fentanyl. 1 Drug origins can be diverse, involving various manufacturing methods and therefore different compositions. Highlighting illicit substances nature and presence in powders makes it possible to establish legal sanctions. Moreover, drug sample composition is of interest in the context of judicial investigations; the analysis of contaminants in these powders contributes to their origin and provides scientific evidence that can help in their manufacture localization.
In the case of natural drugs origin, we can distinguish 2 cases: extraction or hemisynthesis. Cocaine is a tropane alkaloid obtained from several species of Erythroxylum shrub leaves.2,3 Since the solvents used for cocaine extraction lead to only a few traces in the final product after evaporation, it is difficult to rely on their analysis for powder study. Conversely, cutting agents analysis such as levamisole, caffeine, lidocaine, acetaminophen, or phenacetin provides more information on the methods used by traffickers and helps to discriminate between powders based upon their origin, purity, and therefore their place in the supply chain.4,5 Other illicit substances are produced by hemisynthesis, such as heroin. Therefore, the finished product can potentially contain substances from the plant extraction stage, contaminants from the hemisynthesis stage, as well as cutting products. These compounds can greatly vary in composition or in proportion, and therefore constitute elements of high value for profiling purposes. In the case of heroin, obtained by diacetylation of morphine extracted from Papaver somniferum, it is difficult to differentiate the hemi-synthesis methods used. 6 For this reason, the study of the cutting agents remains essential for powder discrimination.
Drug profiling is not routinely performed in forensic toxicology laboratories and innovative data visualization solutions thus remain poorly used in such contexts. Molecular networking (MN) is a computational strategy that may help visualization and interpretation of the complex data arising from MS analysis. Initially developed within the field of natural products research, this tool is particularly useful for complex biological matrices analysis at the molecular level.7-10 Data reprocessing using open source software such as MzMine 2 or Cytoscape allows visualization of the acquired data by non-targeted screening.7,11 MN is able to identify compounds and potential similarities among all MS/MS spectra within the dataset and to propagate annotation to unknown but related molecules. Also, it facilitates the comparison of different matrices in a semi-quantitative manner.12,13
Recently, our laboratory reported a case of fatal poisoning by an unknown powder ingestion (incorrectly labeled as Tabernanthe iboga) and applied MN to identify the plant involved in a woman’s death. 14 Here, in order to compare powders composition suspected to contain illicit substances, we applied this tool to law enforcement agencies seizures products. This study illustrates the utility of molecular networks to efficiently determine the nature and number of compounds structurally related to the main expected component in the frame of illicit drug seizures products analysis.
Material and Methods
Sample Preparation
Powder samples analyzed in this study were collected from law enforcement agency seizures. The sample X, a beige powder sample soluble in methanol, was sold as heroin powder. The sample Y1, a white powder soluble in methanol, and the samples Y2 and Y3, white powders soluble in water, were sold as cocaine powders. The four samples were powdered to weigh 30 mg on a precision balance, dissolved in 3 mL of appropriate solvent, and diluted 1000 times for analysis.
Drug Powder Analysis
Samples were analyzed on an Orbitrap Q Exactive™ mass spectrometer coupled to an Accela 1250 pump (both from Thermo Scientific, San Jose, CA) using a heated electrospray ionization source (HESI-II). Data acquisition, calibration and instrument control were managed using Xcalibur® 2.1 software (Thermo Scientific).
Quantitative assays of illicit drugs were performed using a LC-HRMS method. The mobile phases were composed of ammonium formate at 10 mM and formic acid 0.1% in water (phase A) and acetonitrile and formic acid 0.1% (phase B). LC was performed on a Hypersil Gold column (5 mm × 2.1 mm, 3 μm) (Thermo Scientific, San Jose, CA). Heroin and cocaine were quantified on calibration curves using heroin-D9 and cocaine-D3 as internal standards, respectively (LOQ = 5 ng/mL, range: 10–1000 ng/mL, r2 > 0.999). Calibration solutions were purchased from LGC (Teddington, UK).
In order to achieve MN, untargeted screening LC-HRMS/MS analyses were performed according to Allard et al (2019). 12 Briefly, LC separation was performed on a Hypersil GOLD PFP column (150 × 2.1 mm, 5 μm) (Thermo Scientific, San Jose, USA). The mobile phases were composed of 10 mM ammonium acetate and 0.1% formic acid in water (A), and 0.1% formic acid in acetonitrile (B) at a flow rate of 200 μL/min. Detection was performed by data dependent untargeted screening as previously described. 12
Generation of Molecular Networks
Molecular networks were generated according to Le Daré et al (2020). 15 Raw spectral data files were converted from.raw format into the.mzXML open mass format using Proteowizard software. 16 Spectra were converted from profile to centroid mode in the positive ionization mode. Preprocessing of the data was required for the generation of multi-sample molecular networks, and the use of precursor ion abundance for relative quantification. Such processing additionally allows to take into account the retention time and thus to separate isomers into individual nodes in the resulting molecular networks. For this, the Feature Based Molecular Network process was followed. 17
Preprocessed and aligned spectral file were saved as a single.mgf file using the integrated Global Natural Products Social Molecular Networking (GNPS) export module of MZmine 2. 17 The spectral file.mgf was uploaded to the GNPS platform (http://gnps.ucsd.edu). Due to the use of HR data, the basic parameters were set to 0.02 m/z for the mass tolerance of precursor and fragment ions used for MS/MS spectral library searching, and 0.02 m/z for the mass tolerance of fragment ions used for MN. Molecular networks and MS1- and MS2-level spectral annotations were viewed offline with Cytoscape 3.8.0 (http://www.cytoscape.org).
Results
Powder X Analysis
To identify compounds in a drug seizure suspected to contain heroin, we generated a molecular network that displayed the MS/MS data acquired during analysis (Figure 1). We annotated the nodes with the precursor ion mass, and we labeled the edges with the mass shift between the two connected nodes. During the generation of the network, nodes are linked together in clusters according to their MS2 spectral similarities. The size of each node is proportional to the peak area of each compound, allowing semi-quantitative analysis. We performed node annotation on the basis of their chromatographic retention time (if available), exact mass, and the match between the fragmentation spectrum and the GNPS and/or mzCloud (https://www.mzcloud.org, HighChem LLC, Slovakia) spectral databases. We also used spectral in-house database and bibliography information for annotation. The overall molecular network, composed of nodes from the sample powder X. RT: retention time.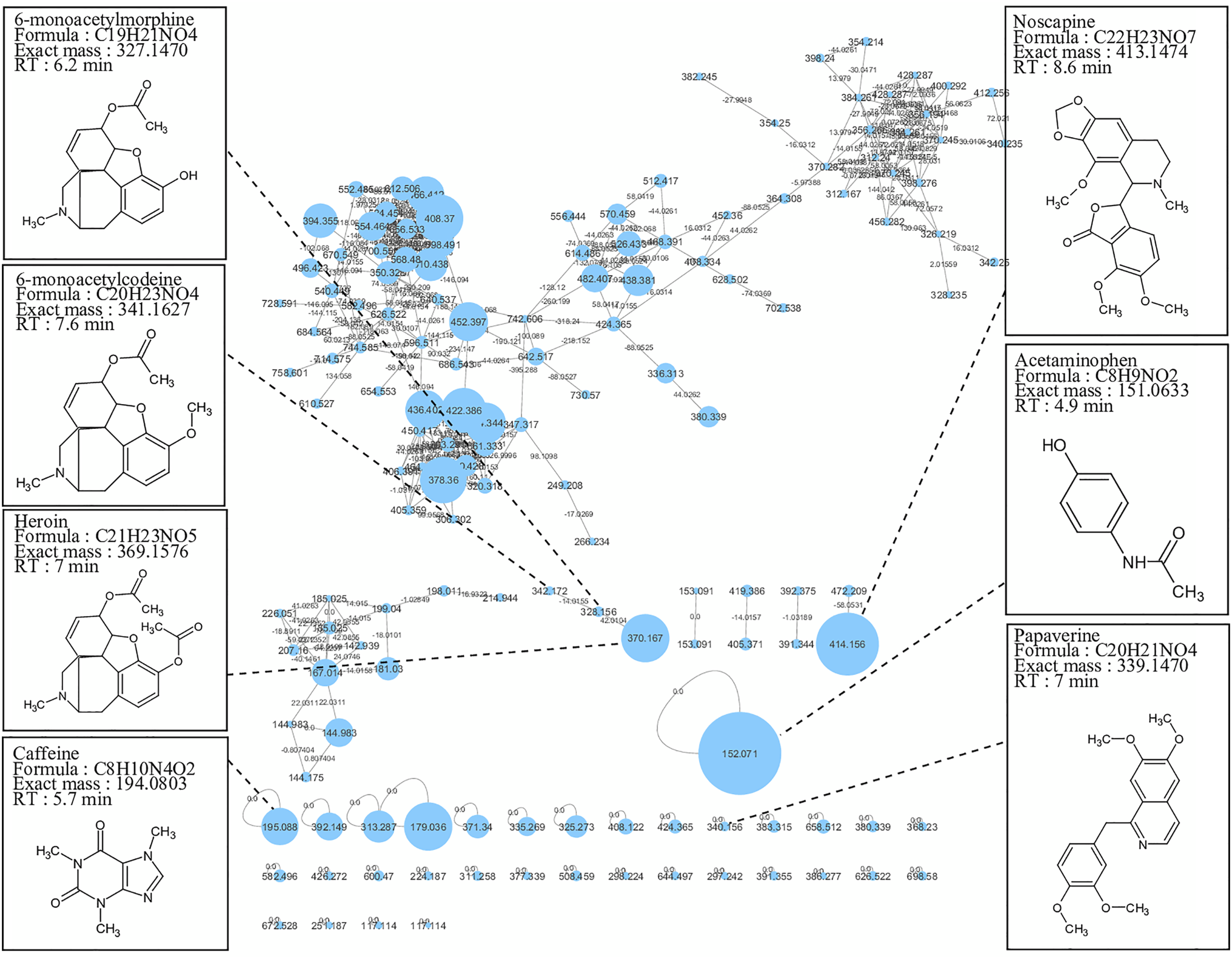
Visual analysis of the molecular network allows us to raise several observations. First, the annotation step using spectral match revealed the presence of heroin (m/z 370.167), confirming illicit compound content. Its derivative 6-monoacetylmorphine (6-MAM) (m/z 328.156) was identified, linked to the heroin node by a mass shift of −42.0104 (typical of an acetyl group). Second, caffeine (m/z 195.088), acetaminophen (m/z 152.071), noscapine (m/z 414.156), papaverine (m/z 340.156), and 6-monoacetylcodeine (m/z 342.172) were also detected as impurities (from synthesis or from plant origin) or cutting agents (Figure 1). Taken together, these results suggest that MN allows to quickly visualize groups of structurally similar compounds and thus to quickly differentiate cutting products from structurally related metabolites (coextracted compounds or synthetic impurities), with identification of illicit compounds.
The powder X quantitative analysis using LC-HRMS evaluated heroin content at 48 μg/mg of powder, corresponding to around 5% purity.
Products Y Analysis
Three powders suspected to contain cocaine were compared using MN. In this sample-to-sample comparison network, we colored the nodes with a pie chart assigning a specific color to each sample. In addition, the size of each colored section of the chart was proportional to the corresponding peak area in each powder. A cocaine-containing cluster was identified in the three powders (Figure 2). The semi-quantitative approach suggests that the Y1 (orange) powder contains more cocaine than the two others (turquoise and red) (Figure 2(a)). Other nodes linked to cocaine (m/z 304.156) were identified with a mass shift of +26.0155, putatively annotated as trans and cis-cinnamoylcocaine (m/z 330.172); with a mass shift of −14.0156 giving rise to benzoylecgonine (m/z 290.141); with a mass shift of +5.953 giving rise to hexanoylecgonine methylester (m/z 298.303) and finally with a mass shift of +90.031 giving rise to trimethoxycocaine. Visual analysis also revealed a higher proportion of these cocaine structurally related compounds in Y1 powder (orange). (a) The cocaine-containing cluster, composed of nodes from the sample Y1 (orange), Y2 (turquoise), and Y3 (red). The node size is mapped to the peak area of cocaine. (b) Main isolated nodes found in the multi-sample molecular network.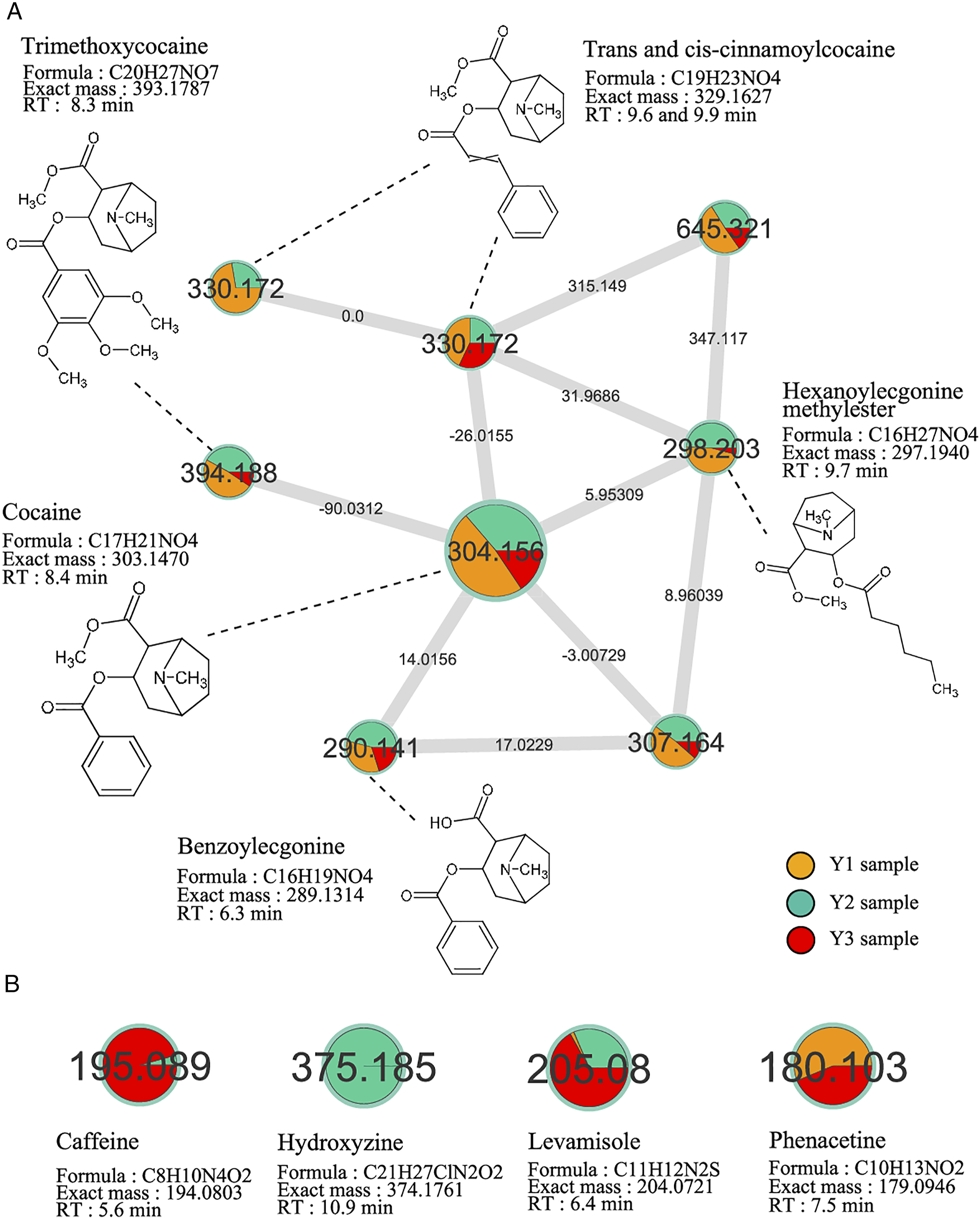
Interestingly, other isolated nodes annotated as phenacetin, levamisole, and caffeine were found in powder Y3 (red); hydroxyzine, levamisole, and caffeine in powder Y2 (turquoise); and phenacetin and levamisole in powder Y1 (orange). These compounds were found in different proportions according to the powders. Taken together, these results revealed differences in composition among these three powders, showing higher cocaine purity in the Y1 sample.
Quantitative analysis of the three powders (Y1, Y2, and Y3) using LC-HRMS confirmed the presence of cocaine in different proportions. Y1, Y2, and Y3 powder samples contained cocaine ratios at 572 μg/mg, 397 μg/mg, and 168 μg/mg of powder, corresponding to purity around 57%, 40%, and 17%, respectively.
Discussion
Highlighting the presence of illicit substances in seized powders is mandatory to establish legal sanctions. In addition, comparison of multiple drug samples by their composition is of definite interest in judicial investigations. The aim of this study is to illustrate the utility of MN as a tool for (i) drug powder composition determination, helping in their profiling and (ii) comparison of various powder sample profiles likely to contain illicit compounds. For these purposes, we analyzed powders likely to contain either heroin or cocaine.
In the first example concerning a seized powder suspected of containing heroin (powder X), molecular network analysis allowed us to organize numerous chemical species detected by LC-HRMS/MS. The LC-HRMS quantitative method using standard calibration curve revealed 5% purity. Since heroin is produced by morphine diacetylation, it is expected that synthetic by-products would be present. We found 6-monoacetylmorphine (m/z 328.1562) and 6-monoacetylcodeine (m/z 342.1717), showing incomplete heroin synthesis and codeine acetylation, respectively.18,19 According to literature data, it is nowadays difficult to differentiate an hemisynthesis using acetic anhydride or a trifluoroacetic anhydride/acetic acid mixture. However, specific techniques to detect in particular bis-trifluoroacetylmorphine, trifluoroacetylcodeine, and 3-acetyl-6-trifluoroacetylmorphine can orient towards a synthetic pathway using trifluoroacetic anhydride. 6 Since we did not detect these substances in our sample, we suggest that this heroin sample was not likely produced by this latter method. We also found other alkaloids of Papaver somniferum, namely, noscapine and papaverine, reflecting the morphine extraction stage. 20 Given that noscapine can also be used as a heroin adulterant, the high proportion found in our sample (regarding to the node size) oriented us toward this hypothesis. 21 In a study analyzing 3476 heroin samples, Morelato et al (2019) reported that heroin adulteration consisted almost exclusively in caffeine and acetaminophen. 22 This observation was consistent with previous reports, highlighting these two substances as the most frequent cutting products found in European heroin seizures since the beginning of the 1900s. 23 Accordingly, we detected these two substances in this powder. Therefore, MN allowed us to organize and visualize illicit content and major structurally related impurities, giving a good understanding of powder composition.
As a second example, three powders suspected to contain cocaine (Y1, Y2, and Y3 powders) were compared to further investigate the utility of molecular networks for drug seizure comparison. Since cocaine is reported to be adulterated by a diverse combination of cutting agents, we believe that such an approach is of particular relevance. Conversely to heroin, cocaine is a naturally present alkaloid, which can be directly extracted and thus requires no hemisynthesis. In addition, the diversity of cocaine powder compositions is known to reflect the origin of the market, which is more complex and dynamic for cocaine than for heroin. 22 In our experiments, MN provided information for profile comparisons, allowing discrimination between sample powders. At first glance, the cocaine node confirmed the illicit content, and a purity analysis can be initiated by the semi-quantitative approach. Higher cocaine content was found in powder Y1 compared with the two others, which was confirmed with LC-HRMS quantitative method using standard calibration curve (57% cocaine). Other alkaloids from Erythroxylum genus shrub leaves (namely, hexanoylecgonine methylester, trimethoxycocaine, and trans/cis-cinnamoylcocaine) were also found in a higher proportion in Y1 powder, corroborating this observation.24-26 Four varieties of Erythroxylum are well known to be used by coca farmers for illicit cocaine production: E novogranatense var novogranatense, E novogranatense var truxillense, E coca var. ipadu and E coca var. coca. Interestingly, trimethoxycocaine is only found in native E coca var. coca and E coca var. ipadu, historically cultivated by coca farmers in Peru/Bolivia and Colombia, respectively.3,27 Moreover, Malette et al (2016) showed that the combinations of alkaloids present in cocaine powders made it possible to carry out geographically sourcing, targeting more precisely the regions of origin of the plants used. As a result, they determined that the majority of the trimetoxycocaine producing shrubs came from eastern colombia, bringing valuable information for our samples. 28 However, new cultigens of cocaine-bearing Erythroxylum have been propagated by Colombian coca farmers in recent years, making geographic determination more difficult.3,27 According to Lukaszewski and Jeffery (1980), benzoylecgonine is commonly encountered in illicit cocaine samples, mostly because of cocaine hydrolysis degradation. 29 Here, benzoylecgonine was found in our three cocaine samples. Besides the semi-quantitative analysis of the alkaloids contained in these powders, cutting products analysis also provides valuable elements for their discrimination. Phenacetin, levamisole, lidocaine, caffeine, diltiazem, hydroxyzine, procaine, tetracaine, acetaminophen, creatine, and benzocaine are among the most widespread cutting agents in Europe.22,30 Consistent with these literature data, MN allows us to find levamisole and phenacetin in powder Y1; caffeine, hydroxyzine, and levamisole in powder Y2; and caffeine, levamisole, and phenacetin in powder Y3. Interestingly, adulterant nature can also bring valuable information regarding the supply chain. For instance, it is assumed that hydroxyzine is usually added at an early stage of the supply chain, while phenacetin and caffeine can be added at different levels, from distribution to consumption level. Lastly, levamisole adulteration, widely used in South America, seems to take place immediately after production or just before exportation. 22 Here, MN helped in sample-to-sample comparison, and appeared to be of particular interest in major component profile screening. Furthermore, all cocaine derivatives were grouped in a unique cluster, allowing to quickly discriminate cocaine-related ions from adulterants.
Illicit drug powder profiling usually corresponds to a combination of complex, expensive and time-consuming analytical and mathematical methods hyphenated to adequate databases.22,31 However, visualization tools, such as MN, appear to be under-used. Guéniat and Esseiva (2005) reported that powder comparison is usually based on GC-MS analysis, searching for major compounds (e.g., benzoic acid for cocaine). Minor compounds (e.g., meconine for heroin) and inorganic compounds (e.g., zinc or iron) are not included in this comparison, due to the high inter-sample variability. 32 Although different, this method remains complementary to the molecular network approaches. In this study, the use of MN allowed a simple and efficient visualization of the different drug design stages, including main compounds identification, organization and comparison. Therefore, it brings many complementary elements to quantitative assays, without expensive external standards requirement. In recent years, the use of MN has diversified in many scientific disciplines. Multiple applications have been described in the literature such as metabolomics, 10 drug discovery, 33 identification of known metabolites or de-replication, 34 toxicological screening, 12 or xenobiotic metabolism exploration, either of natural or synthetic origin.12-14,33 Recently, Vincenti et al (2020) reported the use of MN to analyze new psychoactive substances in two seizures collected by the Italian Department of Scientific Investigation of Carabinieri allowing the identification of fentanyl analogs and synthetic cannabinoids, and associated impurities or synthetic byprodcuts. 35 Here, we further extend the use of this tool for the analysis of drugs of natural origin. These data reinforce the utility of MN in molecular mapping, and show that this tool can provide extremely accurate visual drug fingerprints facilitating legal investigations.
In further studies, it would be interesting to investigate the potential of information propagation of quantitative results via molecular networks under the following hypothesis: since molecular clusters imply spectral similarity, it could be assumed that the presence within a given cluster implies potentially comparable ionization responses. This ionization response could thus be propagated within a cluster from properly quantified standards to unknown but related analogues allowing the semi-quantitative approach to be refined and expended. These perspectives could be of interest in analytical toxicology, showing then a triple utility of the molecular network: grouping spectrally similar compounds and propagating both qualitative (type of structure) and quantitative information (expected similarity in the ionization response).
Conclusion
This is the first application of a molecular network approach for the analysis and comparison of illicit drug seizures of natural origin. Here, organization of data using MN brings a major benefit compared to classical approaches. It provides visual modeling that facilitates the interpretation and understanding of toxicological test results by forensic scientists and magistrates in the context of law enforcement agencies seizures analysis. When used in conjunction with spectral databases, it thus provides valuable information on the product manufacturing, from synthesis to final shaping. Further investigation and exploitation of the spectral similarities evidenced by MN in the frame of illicit drugs profiling will undoubtedly indicate such visualization approaches as necessary assets in the toolbox of the legal toxicologists.
Footnotes
Author Contributions
Brendan Le Daré contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Sophie Allard contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Aurélien Couette contributed to conception and design, drafted manuscript, and critically revised manuscript; Pierre-Marie Allard contributed to conception and design and critically revised manuscript; Isabelle Morel contributed to conception and design and critically revised manuscript; Thomas Gicquel contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of Data and Material
Full data processed through the GNPS platform are accessible with these links: https://gnps.ucsd.edu/ProteoSAFe/status.jsp?task=d85ac138c806487aabab29d0b7be17cf (Figure 1 – Heroin powder); https://gnps.ucsd.edu/ProteoSAFe/status.jsp?task=eaadf7631b6d4e269dfb99a371b1d490 (![]() – Cocaine powders).
– Cocaine powders).
