Abstract
With shrapnel injuries, the metal fragment is usually left in place to reduce the risk of morbidity extensive surgery might bring. This means the individual may retain those metals for the remainder of their life. Often the long-term health effects of the embedded metal are not known, especially with respect to protein damage and perturbations of muscle repair pathways. In this study, using homogenates of rat gastrocnemius muscle implanted with pellets of military-relevant metals, we investigated expression of iNOS and eNOS, enzymes involved in nitric oxide production, as well as MMP-2 and MMP-9, matrix metalloproteinases associated with muscle repair. In addition, hydroxynonenal-modified proteins were investigated to assess metal-induced oxidative damage and metal levels in the gastrocnemius determined. Metals were implanted for up to 12 months in order to determine the long-term effects on the expression of muscle-associated proteins. With the exception of iron and cobalt at 1-month post-implantation, there were no significant differences in metal levels in the gastrocnemius in any of the cohorts. Protein expression analysis showed significant decreases in iNOS and eNOS in the 6-month and 12-month lead and depleted uranium groups. Hydroxynonenal-modified proteins were also significantly increased in the iron, copper, lead, and depleted uranium groups. These results suggest that some embedded metals can induce long-term oxidative damage, as well as affect enzyme systems involved in signal transduction.
Introduction
Injuries containing embedded metal fragments are an unfortunate result of armed conflicts. However, advances in protective equipment and medical responses have greatly increased the survival rate from these types of wounds. Since most embedded metals were originally considered inert, standard surgical guidance was to leave the fragment in place except for certain circumstances. 1 Consequently, many individuals now carry retained metal fragments in their bodies, possibly for the rest of their lives. There were occasional case reports in the scientific literature describing issues related to embedded fragment injuries,2-8 with most of these wounds suffered many years prior to the appearance of the adverse health-related issue. In almost all cases, these injuries were suffered during wartime, with a few resulting from industrial accidents.9,10 Because of significant advancements in weapon and armor design the list of metals potentially found in embedded fragment wounds is extensive. Further, in many cases the toxicological properties of these metals when embedded are not well known. This issue came to the forefront during the First Persian Gulf War in 1990–1991 which saw the first combat use of depleted uranium (DU) munitions. Regrettably, due to friendly-fire incidents, several U.S. military personnel were wounded by DU fragments. There was some concern over leaving the shrapnel in place because of the unique chemical and radiological properties of DU. In addition, because of apprehension over the potential medical and environmental effects of DU munitions, substitute materials were sought as replacements with tungsten-based compositions showing the most promise from a ballistic standpoint. However, when tungsten/nickel/cobalt was tested in a rodent model system, it was found that this material induced a malignant rhabdomyosarcoma at the implantation site.11,12 To our surprise, a composition of tungsten/nickel/iron did not result in tumor formation. 13 These discoveries further magnified our lack of knowledge in regards to the short- and long-term health effects of embedded metal fragments. As a result of these findings, both the U.S. Department of Defense and Department of Veterans Affairs established a list of “metals of concern” with respect to embedded fragments.14,15
Since surgical guidelines still recommend leaving fragments in place in an attempt to balance potential long-term health risks with the morbidity risk extensive surgery entails, the localized response of the injured muscle to the embedded metal and its ability to repair metal-induced damage will be critical to the long-term health and well-being of the wounded individual. The findings described in this manuscript are from a multi-center collaborative project with colleagues at the Department of Veterans Affairs, the University of Maryland School of Medicine, the University of Kentucky, and the United States Food and Drug Administration. The overall goal of this effort was to study the potential long-term health effects of embedded fragments using a rodent model system alongside a human investigation of military personnel wounded with embedded metal fragments. To address this aim, in this particular study, we investigated the response of rat gastrocnemius muscle implanted with metals for up to 12 months with respect to expression of proteins involved in nitric oxide production (iNOS, eNOS) and muscle repair and regeneration (MMP-2, MMP-9). In addition, metal-induced oxidative damage of muscle was investigated by assessing hydroxynonenal-modified proteins. Lastly, metal concentration in the gastrocnemius immediately surrounding the pellet was determined to evaluate metal fragment breakdown.
Materials and Methods
Animals
Prior to initiation, all animal research associated with this project was approved by the Armed Forces Radiobiology Research Institute (AFRRI) Institutional Animal Care and Use Committee (IACUC) under protocol number 2016-05-006. All procedures were conducted in compliance with animal research guidelines as found in the Guide for the Care and Use of Laboratory Animals 16 in an Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)-accredited facility. Male Sprague Dawley (Rattus norvegicus) rats, approximately 30 days old and 75–100 g body weight, were purchased from Envigo (Barrier 208A, Frederick, MD, USA). Animals were allowed to acclimate for at least 2 weeks after arriving at the AFRRI vivarium and were pair-housed throughout the study in plastic microisolator cages (23.8 x 45.4 cm) with filter tops. Teklab Sani-Chips (Envigo) were used as bedding and was changed 2–3 times per week. Vivarium rooms were maintained at 21 ± 2°C with 30-70% humidity. A 12:12 h light:dark cycle was maintained with lights on at 0600. Rats were fed a standard rodent chow (Teklad Global Rodent Diet 8604, Envigo) with water available ad libitum.
Experimental Design
Previous research in our Institute developed a rodent model for studying the health effects of embedded metal fragments, such as those suffered in a shrapnel wound. 17 We used that model system in this study to investigate the effects of eight military-relevant metals including tungsten (W), nickel (Ni), cobalt (Co), iron (Fe), copper (Cu), aluminum (Al), lead (Pb), and depleted uranium (DU). Tantalum (Ta) was used as a control for any changes resulting from the surgical procedure or due to the presence of a foreign material in the muscle. Tantalum is considered inert and has been used for implanted prosthetic devices.18-20 Previous studies have shown no differences between naïve and tantalum-implanted rats.11,21,22 Therefore, the total number of rats needed for the study was reduced and the ARRIVE Guidelines met. 23 Animals were randomly assigned to 1 of the 9 metal implantation groups with an n = 8 per metal. The metal groups were conducted in 4 different cohorts: 1, 3, 6, or 12 months post-implantation surgery (i.e., “Time from Implant”). A total of 288 rats (n = 8 x 9 metal groups x 4 cohorts) were used in the study.
Metal pellets (1 mm diameter x 2 mm length) were purchased from Alfa Aesar (Ward Hill, MA, USA) with the exception of DU which was purchased from Aerojet Ordnance (Jonesboro, TN, USA). Prior to implantation, pellets were cleaned and chemically sterilized as previously described. 13
Pellet Implantation Procedures
Metal pellets were surgically implanted in the gastrocnemius muscle of rats as previously described.24,25 Briefly, animals were initially anesthetized in an induction chamber using isoflurane (Baxter Health care, Deerfield, IL, USA) and then maintained throughout the surgical period using a nose cone with a scavenger/recapture system. The surgical sites were clipped, swabbed with 70% 2-propanol, and finally cleansed with betadine (Purdue Pharma LP, Stamford, CT, USA). Prior to surgery, a prophylactic dose of an analgesic (buprenorphine, 0.05–0.1 mg/kg, s.c., Rickitt and Colman, Hull UK) was administered. Two identification methods were used. A small transponder (Electronic Lab Animal Monitoring System, Bio-Medic Data Systems, Seaford, DE, USA) was injected subcutaneously in the mid-dorsal thoracic region. Each transponder was programmed with a unique animal identification number that can be read with a low-power radio frequency scanner. An ear punch system was used as the second backup identification system in case of transponder failure. Using aseptic technique, a small incision approximately 5 mm in length was made through the skin of each hind leg to expose the gastrocnemius muscle. Each gastrocnemius muscle was implanted with two sterile pellets spaced approximately 1.5 mm apart on the lateral side of the muscle by placing the sterile pellet in a 14–16 gage needle. Using a specially designed plunger placed inside the needle, the needle was inserted into the gastrocnemius and the plunger depressed forcing the pellet into the muscle. Tissue adhesive (VetBond; 3M Corporation, St Paul, MN, USA) was used to seal the incision. After surgery, rats were closely monitored until ambulatory. The surgery sites were examined daily for 2 weeks to assess for signs of inflammation, infection, and local metal toxicity, and after that time, weekly, for the duration of the study.
Euthanasia and Tissue Collection
Upon reaching their experimental end point or when indicated by guidelines approved by the AFRRI IACUC, rats were humanely euthanized under deep isoflurane by exsanguination and confirmatory pneumothorax as per the guidelines of the American Veterinary Medical Association. 26 After euthanasia, a complete gross pathology examination was conducted. A variety of tissues were collected for further examination as has been described in earlier publications.24,25 For this study, the gastrocnemius was isolated, snap-frozen in liquid nitrogen within a piece of aluminum foil and stored at −80°C until needed. Gastrocnemius samples for protein expression analysis were prepared by taking the −80°C-stored tissue and placing on a Plexiglas slab chilled over dry ice. The pellet implantation site in the muscle was located and the tissue around the pellet site dissected with a chilled razor blade. The tissue was placed in a chilled 1.5 mL centrifuge tube and processed as described below. In cases where tumor formation impinged on the pellet implantation site, the muscle sample was taken 3–5 mm away from the tumor.
Protein Expression in Gastrocnemius Tissue
Tissue samples were suspended in RIPA buffer (Thermo, Waltham, MA, USA) plus Halt Protease and Phosphatase Inhibitor Cocktail (Thermo) and homogenized in a Bullet Blender (Next Advance, Troy, NY, USA) with 1.0 mm zirconium oxide beads (NextAdvance) (settings speed 6, 5 min x 3 runs) and then centrifuged at 1340 x g for 10 min. Total protein from each sample supernatant was measured by Bio-Rad Protein Assay (Bio-Rad Laboratories, Hercules, CA, USA), in triplicate, against a BSA standard curve, on a spectrophotometer (BioTek Synergy Model H1M Multimodal Plate Reader with GEN5 Software, BioTek Instruments, Winooski, VT), and read at 595 nm. Proteins of interest were quantified using an automated capillary-based size sorting chemiluminescent system “WES” from ProteinSimple (San Jose, CA, USA). All procedures were performed with manufacturer’s reagents (12-235 kDa kit) according to the user manual with some adjustments: samples are aliquoted to 1 μg protein/μl before mixing 4 μL with 1 μL fluorescent master mix, then denatured at 94°F (34.4°C) for 4 min, given a quick spin, and loaded on the plate at 4 μL per well. In run settings, stacking time was changed to 18 sec, separation time to 31 min, and antibody diluent time to 30 min. Antibody information, including source and concentrations used, is listed in Table S1, along with any additional changes specific to a particular antibody. Peak values were determined using Compass Software (ProteinSimple). Results are in arbitrary units and presented as a ratio of the target protein expression normalized to the system control antibody added following manufacturer instructions. While this is a technical loading control and not a biological loading control, it was chosen because β-actin expression was not detected in the gastrocnemius samples, and as we present later, the muscle marker desmin was affected by implanted metals and would not serve as a reliable normalizing marker. The ProteinSimple Wes system uses capillary size separation and chemilumiscent detection, and an example of the raw data as well as a traditional Western blot image derived from the raw data is presented in our previous work with brains of the same animals. 24
Metal Analysis by Inductively Coupled Plasma-Mass Spectrometry
All reagents used were of the highest grade available. Plasticware and other disposables were obtained from Thermo Fisher (Pittsburgh, PA, USA). Gastrocnemius homogenates were dissolved in ultrapure nitric acid (Opitma Grade, Fisher Scientific, Newark, DE, USA) and metal content determined using an inductively coupled plasma-mass spectrometer (Thermo Fisher iCAP RQ/Q with Quantara software, Madison, WI, USA) equipped with a Cetac ASX-560 Autosampler (Cetac Technologies, Omaha, NE, USA). High-pressure liquid argon, 99.997%, was used for the plasma gas. Helium, 99.999%, was used as the CCT gas. Calibration was accomplished with external standards of the appropriate metal standard (SPEX CertiPrep, Methuchen, NJ, USA) in 2% nitric acid. The sample probe was washed with a constant flow of 2% nitric acid to prevent carryover. Quantitative analysis was obtained by reference to the slope of the calibration curve (counts per second/ng per liter) as well as an internal standard. Limit of Detection (LoD)/Limit of Quantitation (LoQ), in ppb, are as follows: Ta - 0.012/0.015; W - 0.002/0.010; Ni - 0.016/0.042; Co - 0.001/0.002; Fe - 0.541/1.226; Cu 0.026/0.106; Al - 0.629/1.716; Pb - 0.001/0.009; U - 0.001/0.003.
Statistical Analysis
Metal concentrations or protein expression were analyzed by two-way ANOVA using the variables of time from implant and implanted metal followed by Sidak’s multiple comparisons test where each metal implant group within a time from implant is compared back to the corresponding Ta-implanted group value. Analyses were performed using GraphPad Prism Software (version 8.0.1, La Jolla, CA, USA). In all cases P values < 0.05 were considered significant.
Results
In this study, male Sprague–Dawley rats were surgically implanted in the gastrocnemius muscle with pellets of military-relevant metals selected from the U.S. Department of Defense’s list of “metals of concern”. At 1, 3, 6, and 12 M post-implantation, the rats were humanely euthanized and the gastrocnemius isolated and analyzed for metal content and expression of proteins involved in metal-induced oxidative damage, signal transduction, and muscle repair.
Metal Analysis of Gastrocnemius
ICP-MS Operating Conditions and Parameters.
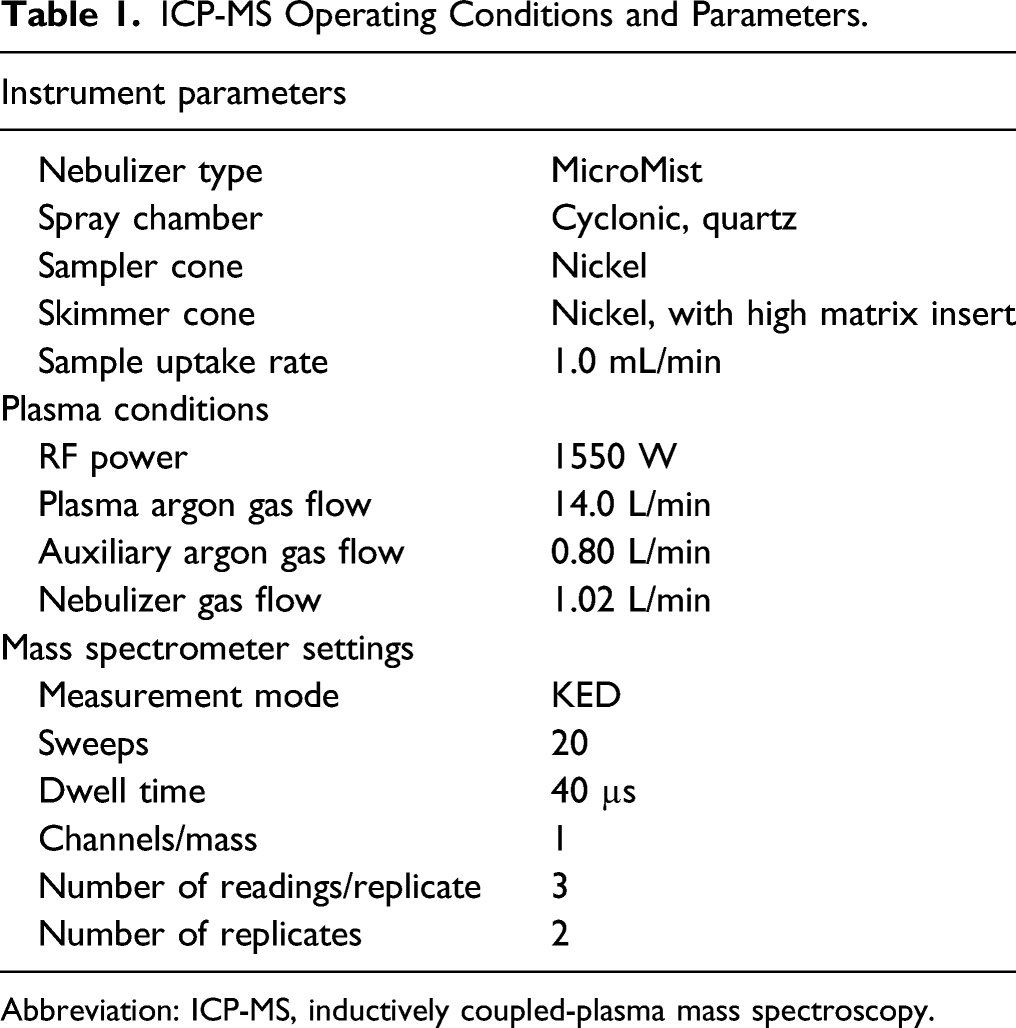
Abbreviation: ICP-MS, inductively coupled-plasma mass spectroscopy.
Figure 1 shows the metal concentrations in the gastrocnemius homogentes from the various implanted cohorts: A) tungsten: Fmetal (1,50) = 28.30, P < 0.0001; B) nickel: Fmetal (1, 52) = 2.773, P = 0.1019; C) cobalt: Fmetal (1, 51) = 6.086, P = 0.0170; D) iron: Fmetal (1, 54) = 5.982, P = 0.0177; E) copper: Fmetal (1, 55) = 1.985, P = 0.1645; F) aluminum: Fmetal (1, 55) = 6.669, P = 0.0125; G) lead: Fmetal (1, 51) = 23.11, P < 0.0001; and H) depleted uranium: Fmetal (1, 51) = 1.133, P = 0.2920. With the exception of the 1M cobalt and iron groups, all other metal levels showed no significant differences from the control group. The 1 M cobalt and iron gastrocnemius levels were significantly higher than control suggesting that these 2 metals can rapidly solubilize and diffuse into the surrounding tissue. Other metal levels in the gastroc homogenates were generally very low, except for aluminum. We attribute these elevated levels in both control and implanted groups to minute pieces of aluminum foil from sample collection adhered to the frozen tissue during homogenization.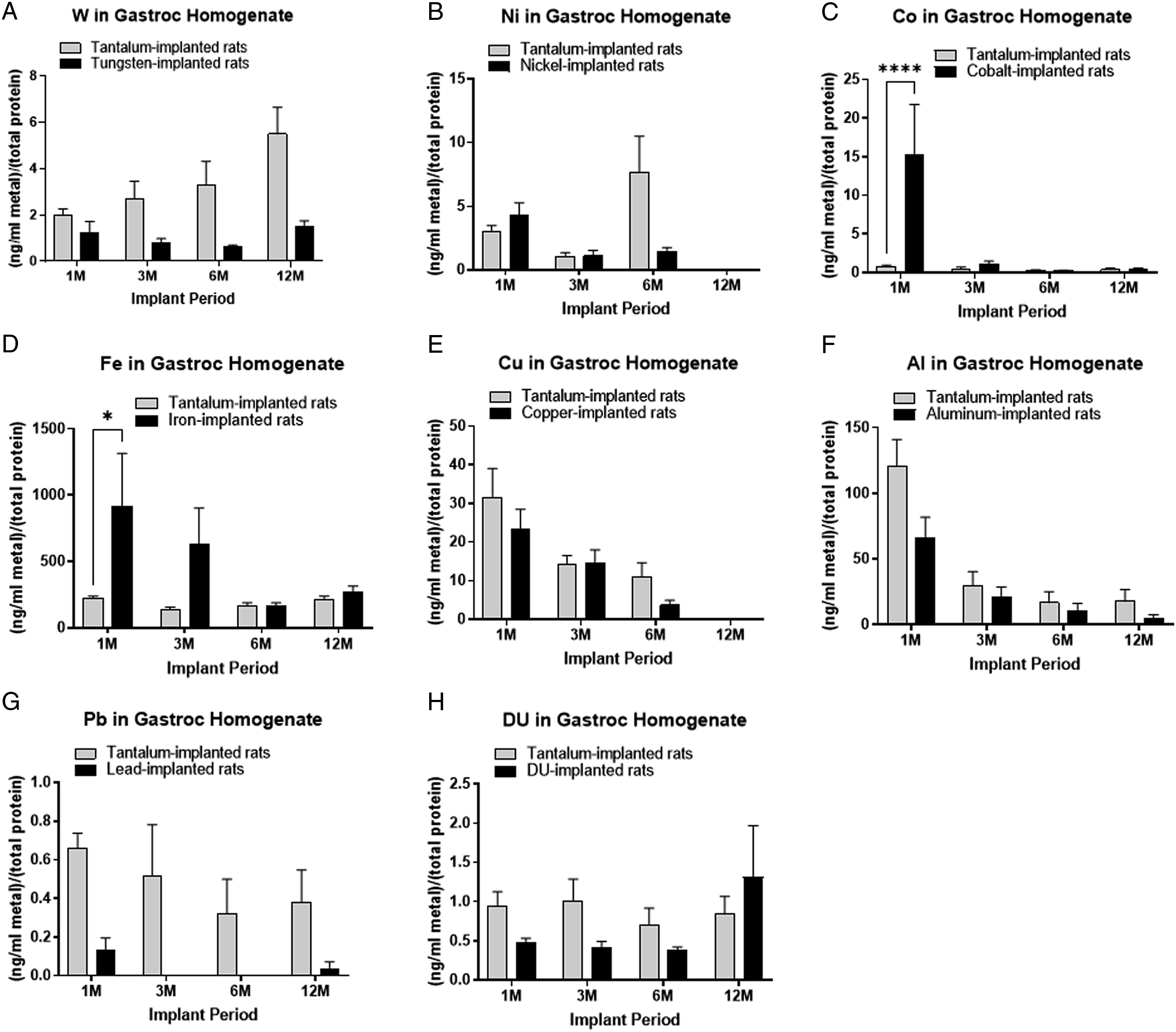
Protein Expression in Gastrocnemius Muscle
The Protein Simple Wes system was used to investigate the expression of several proteins in the gastrocnemius homogenate. The system uses capillary size separation and chemiluminescent detection. We were unable to detect β-actin in the gastrocnemius homogenate and therefore used the Protein Simple system control antibody for normalization.
Desmin Expression
Figure 2 shows the expression of the muscle protein desmin in the gastrocnemius tissue immediately surrounding the implanted metal pellet: A) tungsten, Fmetal (1, 56) = 0.9427, P = 0.3358; B) nickel, Fmetal (1, 55) = 0.4478, P = 0.5062; C) cobalt, Fmetal (1, 55) = 1.516, P = 0.2235; D) iron, Fmetal (1, 55) = 2.924, P = 0.0929; E) copper, Fmetal (1, 53) = 13.99, P = 0.0005; F) aluminum, Fmetal (1, 56) = 0.4672, P = 0.4971; G) lead, Fmetal (1, 56) = 0.4331, P = 0.5132; and H) DU, Fmetal (1, 55) = 0.0274, P = 0.8692. Desmin was increased in the 3M implant group in Co- (*P = 0.0068), Fe- (*P = 0.0101), and Cu-implanted animals (*P < 0.0001) compared with Ta-implanted animals. For this reason, desmin was not used as the muscle marker for normalization as originally planned, since we did not expect desmin expression to be affected by implanted metals.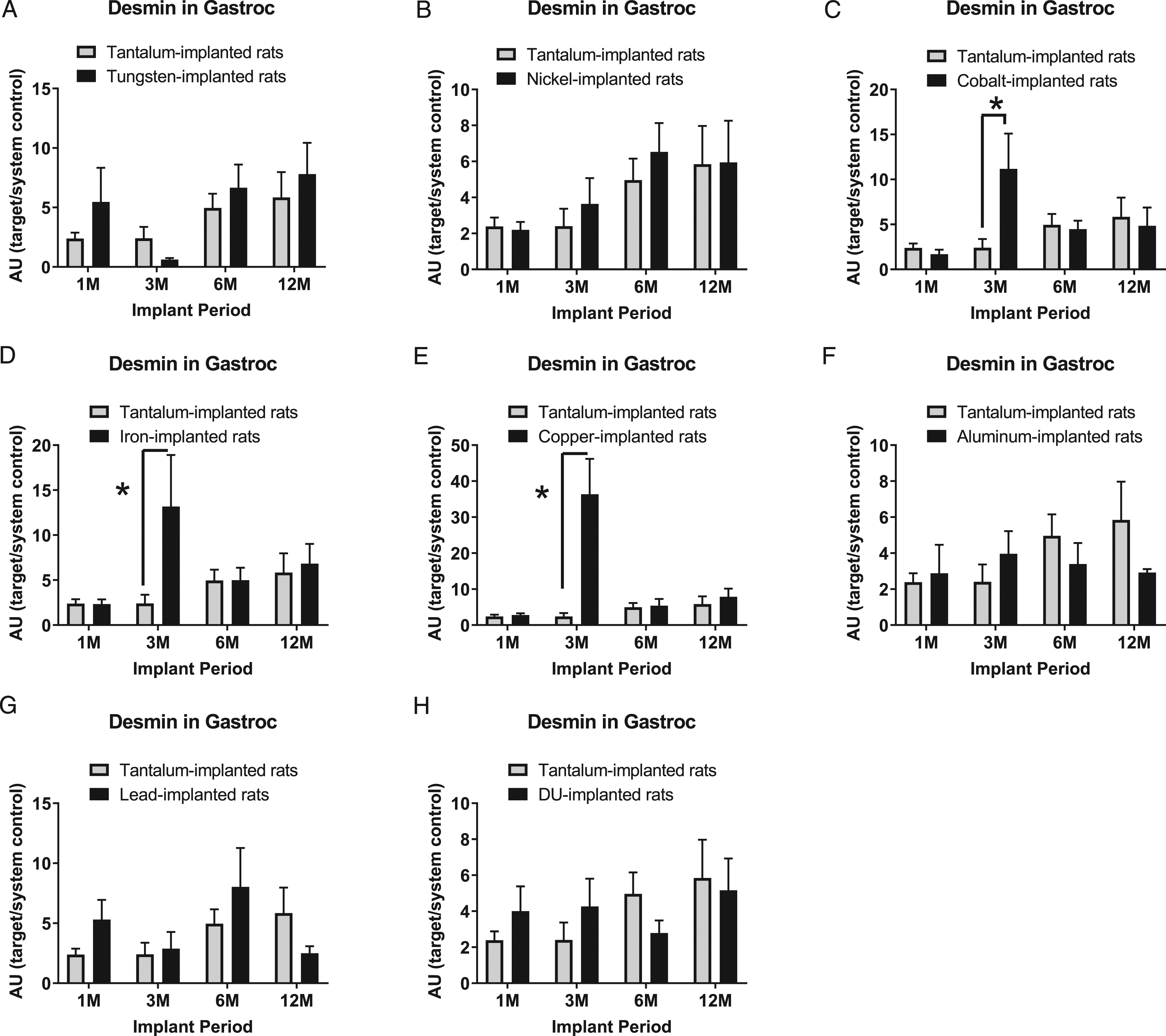
Endothelial Nitric Oxide Synthase Expression
Endothelial nitric oxide synthase expression levels in gastrocnemius samples are presented in Figure 3: A) tungsten, Fmetal (1, 56) = 1.290, P=0.2869; B) nickel, Fmetal (1, 56) = 0.1295, P=0.7203; C) cobalt, Fmetal (1, 54) = 0.5784, P = 0.4502; D) iron, Fmetal (1, 53) = 1.305, P = 0.2584; E) copper, Fmetal (1, 54) = 1.141, P = 0.2903; F) aluminum, Fmetal (1, 56) = 0.0184, P = 0.8927; G) lead, Fmetal (1, 56) = 0.9541, P = 0.3329; and H) DU, Fmetal (1, 51) = 0.6911, P = 0.4097. eNOS was decreased in the 6M implant group in both Pb- (*P = 0.0197) and DU-implanted animals (*P = 0.0486) compared with Ta-implanted animals.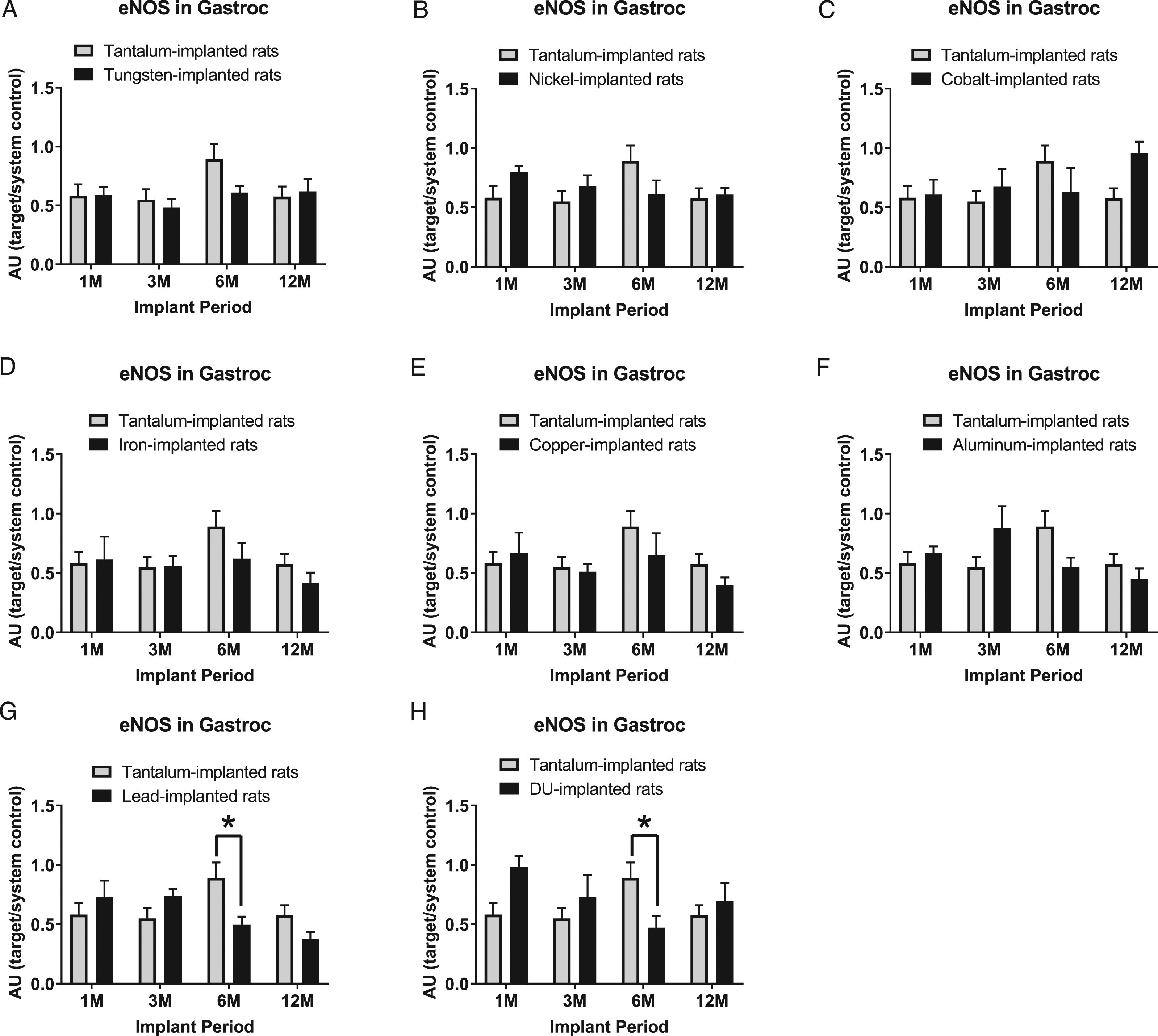
Inducible Nitric Oxide Synthase Expression
Inducible nitric oxide synthase expression levels in gastrocnemius samples are presented in Figure 4: A) tungsten, Fmetal (1, 56) = 2.158, P = 0.1475; B) nickel, Fmetal (1, 56) = 1.027, P = 0.3153; C) cobalt, Fmetal (1, 54) = 1.582, P = 0.2138; D) iron, Fmetal (1, 51) = 0.0348, P = 0.8527; E) copper, Fmetal (1, 52) = 2.448, P = 0.1237; F) aluminum, Fmetal (1, 56) = 7.063, P = 0.0102; G) lead, Fmetal (1, 56) = 19.28, P < 0.0001; and H) DU, F metal1, 56) = 15.75, P = 0.0002. In the 6M group, iNOS was higher in Co-implanted animals (*P < 0.0001) and Cu-implanted animals (*P = 0.0003) compared to Ta-implanted animals but was lower in Al-implanted animals in the 6M group (*P=0.0227). iNOS was lower in Pb-implanted animals than Ta-implanted animals in the 6M and 12M groups (*P = 0.0002 and 0.0449, respectively). iNOS was also lower in DU-implanted animals than Ta-implanted animals in the 6M and 12M groups (*P = 0.0002 and *P = 0.0044, respectively).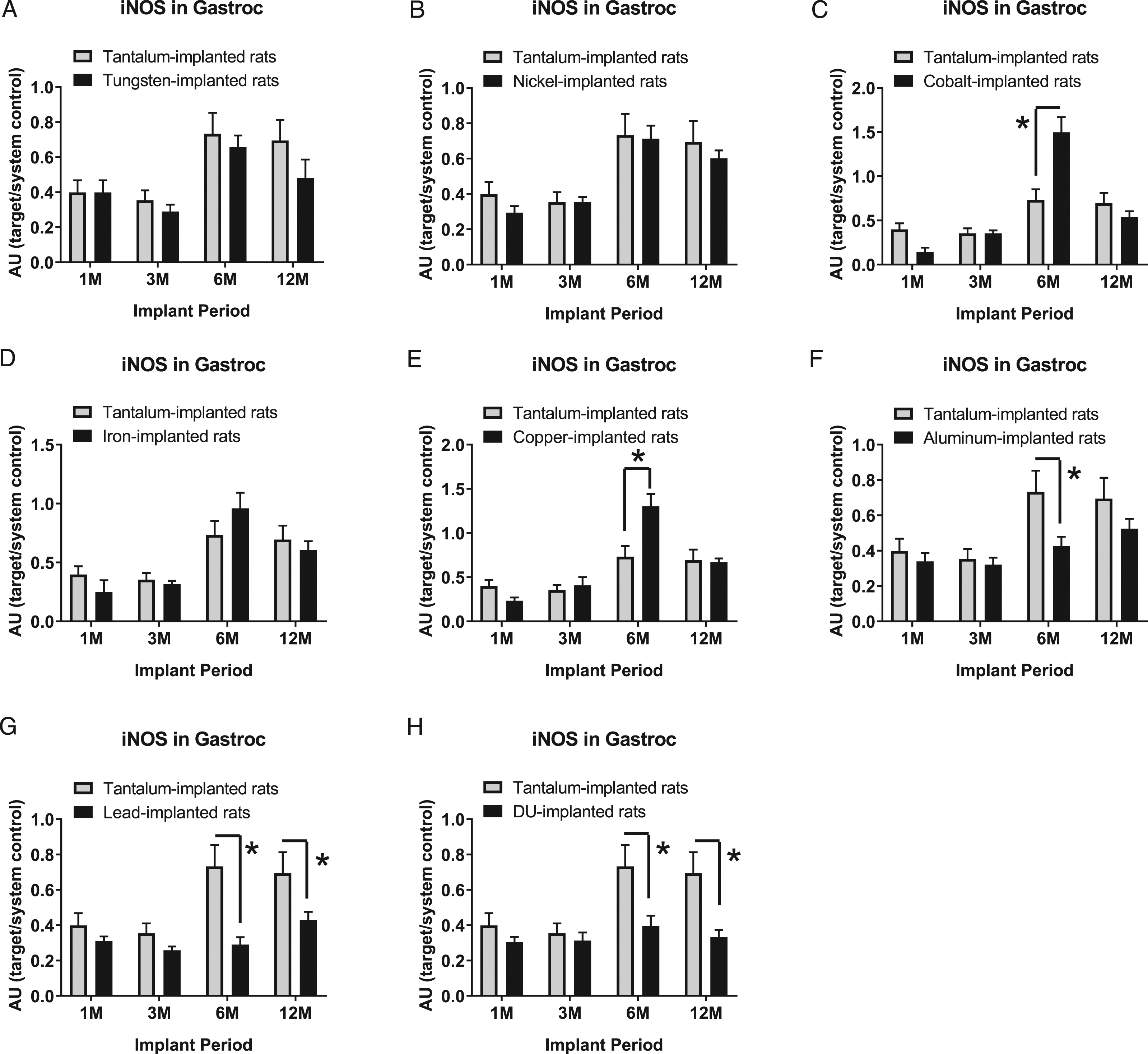
Matrix Metalloproteinase-2 Expression
Matrix metalloproteinase-2 showed up in full form at 93 kDa, and two cleaved forms, at 47 kDa and 58 kDa. The full size MMP-2 band (93 kDa) expression level in gastrocnemius samples are presented in Figure 5: A) tungsten, Fmetal (1, 56) = 2.519, P = 0.1181; B) nickel, Fmetal (1, 56) = 0.0898, P = 0.7656; C) cobalt, Fmetal (1, 56) = 3.559, P = 0.0644; D) iron, Fmetal (1, 55) = 9.861, P = 0.0027; E) copper, Fmetal (1, 54) = 4.264, P = 0.0442; F) aluminum, Fmetal (1, 56) = 2.919, P = 0.0931; G) lead, Fmetal (1, 56) = 10.87, P = 0.0017; and H) DU, Fmetal (1, 56) = 8.453, P = 0.0052. Interestingly, in the 6M implant group, MMP-2 was lower in both the Pb-implanted animals and in the DU-implanted animals compared to Ta-implanted animals (*P = 0.0190 and *P = 0.0449, respectively).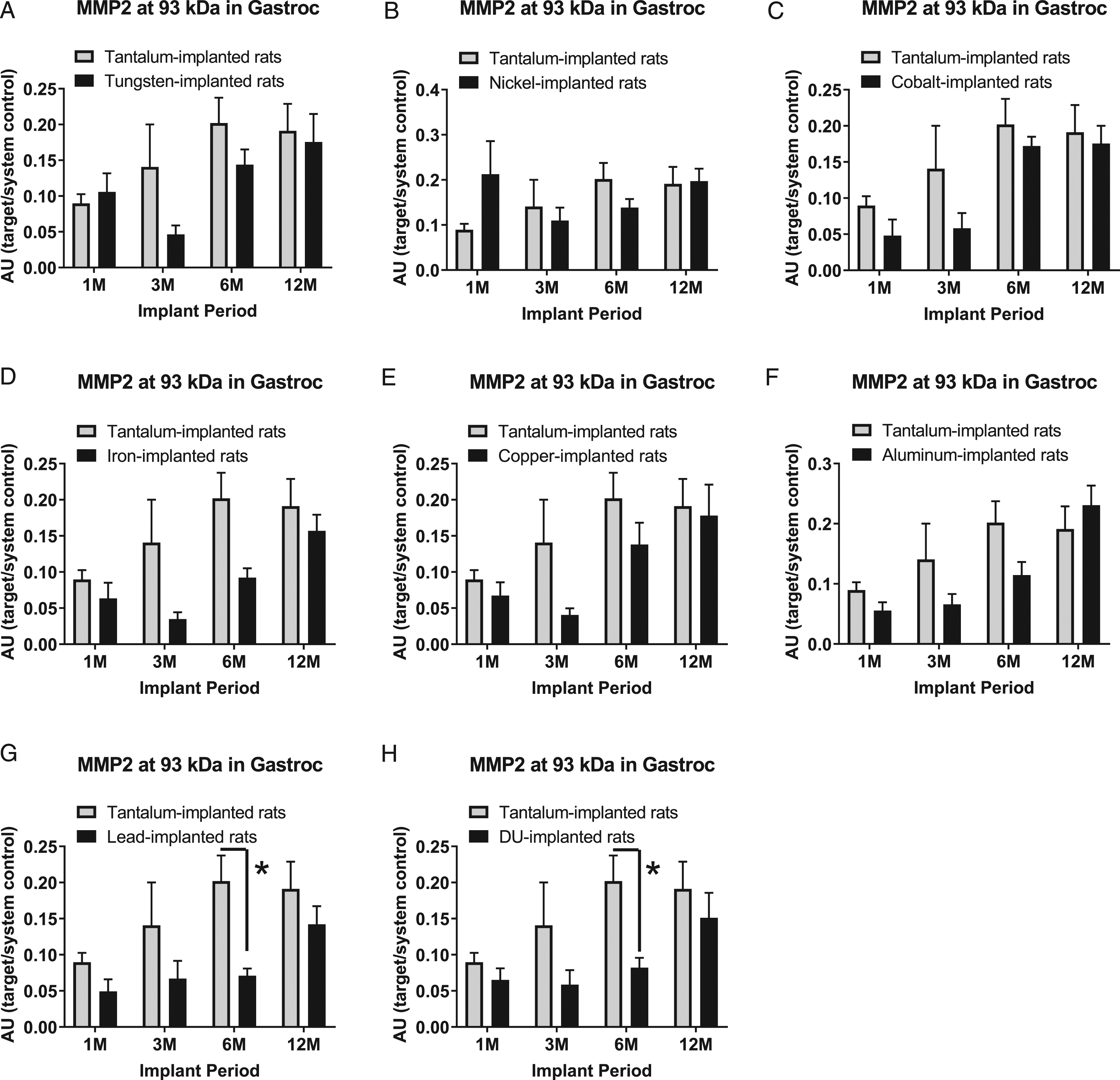
The 58 kDa MMP-2 band expression level in gastrocnemius samples are presented in Figure 6: A) tungsten, Fmetal (1, 56) = 0.2616, P = 0.6110; B) nickel, Fmetal (1, 56) = 1.998, P = 0.1630; C) cobalt, Fmetal (1, 56) = 6.429, P = 0.0140; D) iron, Fmetal (1, 55) = 4.483, P = 0.0388; E) copper, Fmetal (1, 54) = 0.1208, P = 0.7295; F) aluminum, Fmetal (1, 56) = 0.0002, P = 0.9887; G) lead, Fmetal (1, 56) = 1.449, P = 0.2337; and H) DU, Fmetal (1, 56) = 0.3558, P = 0.5532. The only significant difference was in the 1M group, where the 58 kDa MMP-2 was higher in Fe-implanted animals than Ta-implanted animals.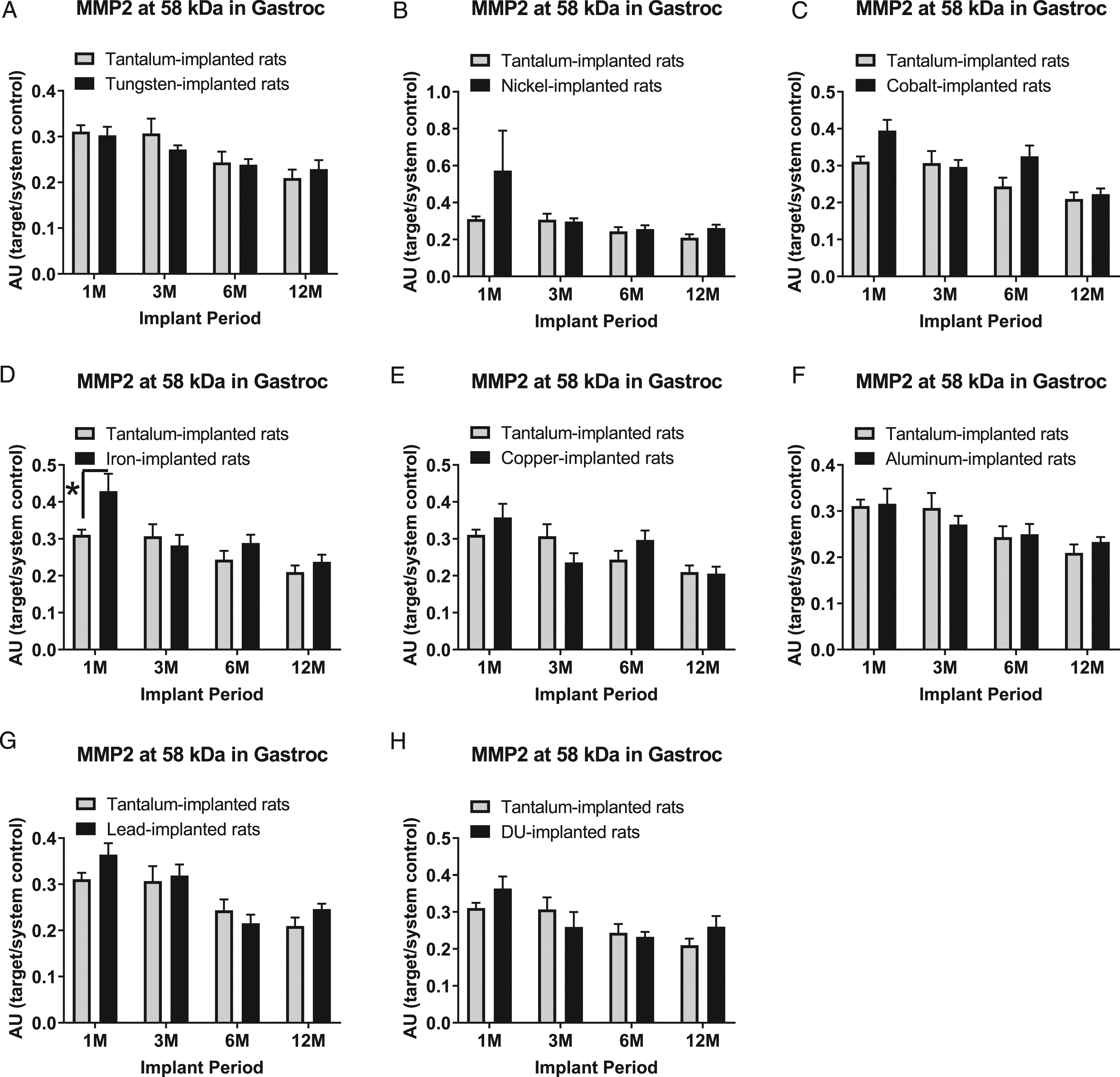
The 47 kDa MMP-2 band expression levels in gastrocnemius samples are presented in Figure 7: A) tungsten, Fmetal (1, 56) = 1.884, P = 0.1753; B) nickel, Fmetal (1, 56) = 3.059, P = 0.0858; C) cobalt, Fmetal (1, 56) = 0.8924, P = 0.3489; D) iron, Fmetal (1, 55) = 0.0013, P = 0.9709; E) copper, Fmetal (1, 54) = 2.620, P = 0.1114; F) aluminum, Fmetal (1, 56) = 0.6302, P = 0.4306; G) lead, Fmetal (1, 56) = 3.224, P = 0.0780; and H) DU, Fmetal (1, 56) = 1.330, P = 0.2538. In the 1M group, the 47 kDa MMP-2 was higher in both the Fe-implanted animals (*P = 0.0203) and Cu-implanted animals (*P = 0.0269) than Ta-implanted animals. In the 12M group, 47 kDa was higher in both the Al-implanted animals (*P = 0.0044) as well as the DU-implanted animals (*P = 0.0085) when compared to Ta-implanted animals. Surprisingly, 47 kDa MMP-2 was lower in Pb-implanted animals than Ta-implanted animals in the 6M group (*P = 0.0202).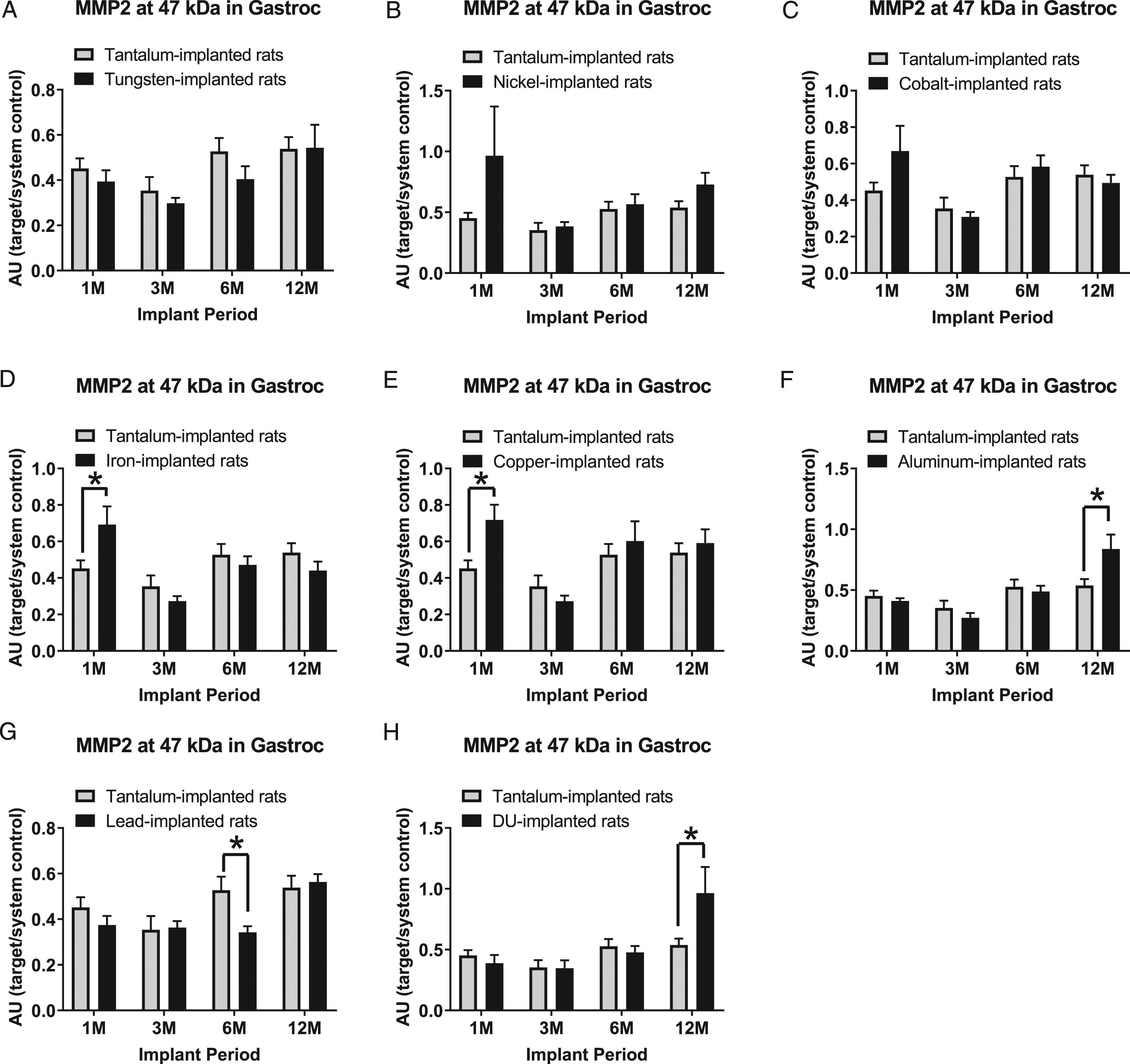
Matrix Metalloproteinase-9 Expression
Matrix metalloproteinase-9 expression levels in gastrocnemius samples are presented in Figure 8: A) tungsten, Fmetal (1, 55) = 1.481, P = 0.2287; B) nickel, Fmetal (1, 53) = 4.116, P = 0.0475; C) cobalt, Fmetal (1, 55) = 0.9078, P = 0.3449; D) iron, Fmetal (1, 54) = 0.5798, P = 0.4497; E) copper, Fmetal (1, 51) = 0.9623, P = 0.3312; F) aluminum, Fmetal (1, 54) = 0.0421, P = 0.8383; G) lead, Fmetal (1, 54) = 0.2721, P = 0.6041; and H) DU, Fmetal (1, 52) = 1.745, P = 0.1923. MMP-9 was higher in W-implanted animals in the 3M implant group (*P=0.0429) and in Ni-implanted animals in the 12M implant group (*P = 0.0302) when compared to the corresponding Ta-implanted animals.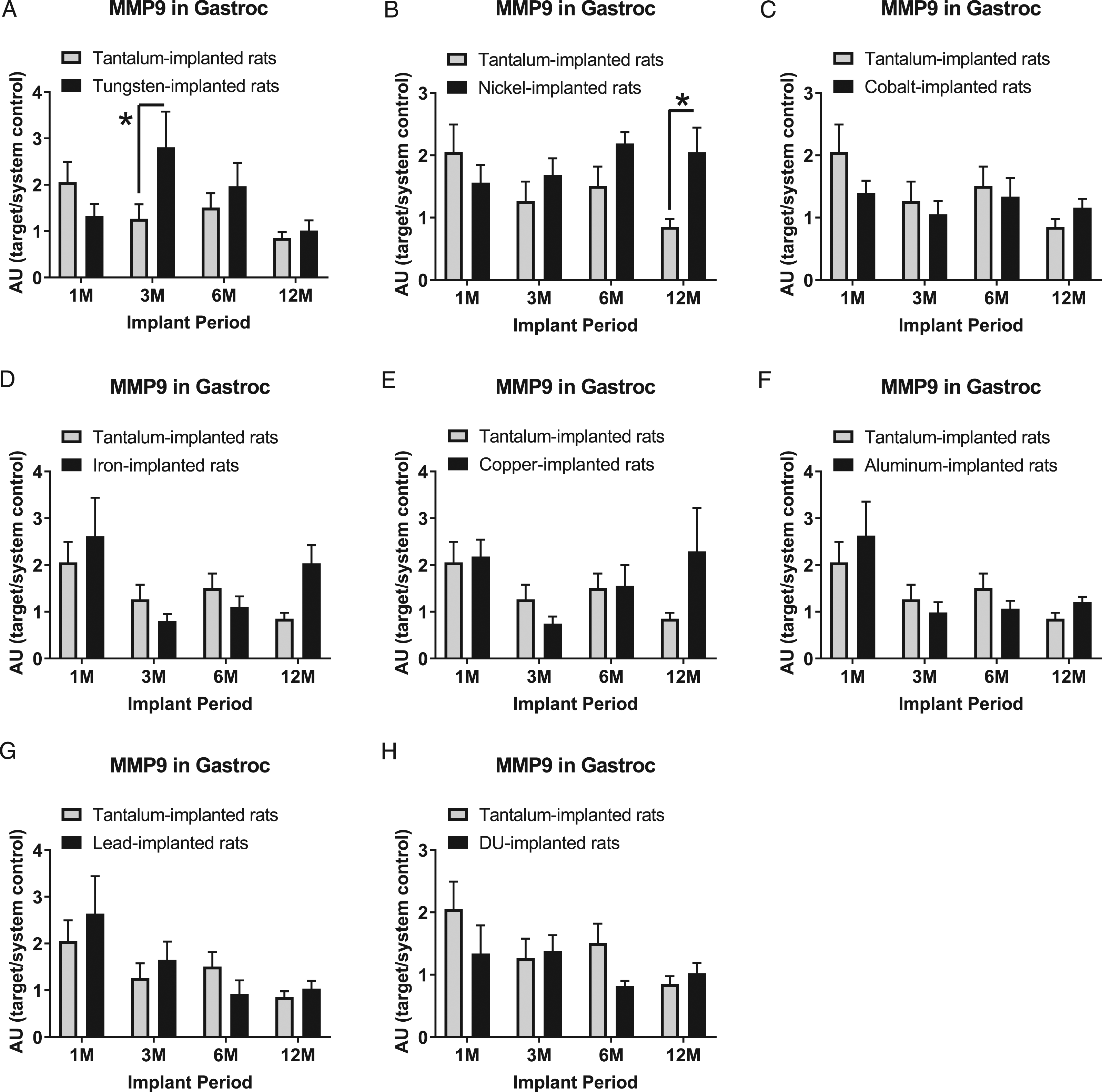
Hydroxynonenal-Modified Proteins
4-Hydroxynonenal (HNE) is a highly reactive by-product of lipid peroxidation in cells and is considered a marker of oxidative stress. HNE-modified protein expression appears at multiple different bands in these samples. Six of the most prominent bands were assessed for metal-induced effects. At 35 kDa, expression levels in gastrocnemius samples are presented in Figure 9: A) tungsten, Fmetal (1, 54) = 0.1276, P=0.7223; B) nickel, Fmetal (1, 53) = 0.3034, P=0.5840; C) cobalt, Fmetal (1, 55) = 2.185, P=0.1451; D) iron, Fmetal (1, 54) = 2.016, P=0.1614; E) copper, Fmetal (1, 53) = 0.7512, P=0.3900; F) aluminum, Fmetal (1, 55) = 2.274, P=0.1373; G) lead, Fmetal (1, 55) = 1.646, P=0.2049; and H) DU, Fmetal (1, 55) = 5.815, P=0.0193. In the 12M group, HNE (35 kDa) was increased in Co-implanted animals compared with Ta-implanted animals (*P=0.0191). With the Cu-implanted animals, HNE (35 kDa) was decreased compared to Ta-implanted animals in the 1M implant group (*P=0.0097) but increased at the 12M implant period (*P=0.0093). HNE (35 kDa) was also increased in the DU-implanted animals compared with the Ta-implanted animals at the 6M implant period (*P=0.0497).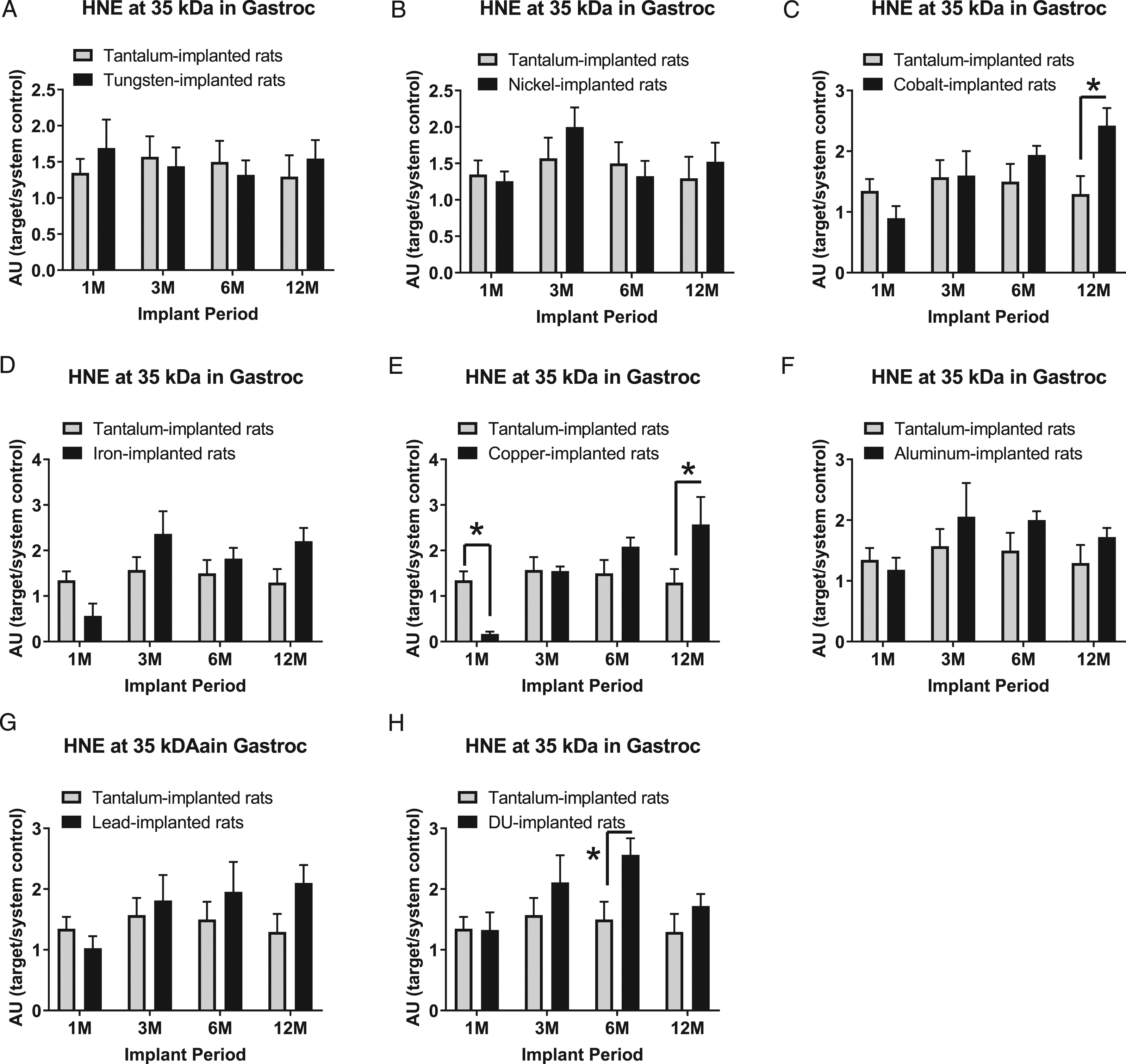
Hydroxynonenal-modified protein expression at the 40 kDa band in gastrocnemius samples are presented in Figure 10: A) tungsten, Fmetal (1, 56) = 1.709, P=0.1965; B) nickel, Fmetal (1, 54) = 0.8218, P = 0.3687; C) cobalt, Fmetal (1, 56) = 0.6520, P = 0.4228; D) iron, Fmetal (1, 55) = 20.13, P < 0.0001; E) copper, Fmetal (1, 54) = 1.766, P = 0.1895; F) aluminum, Fmetal (1, 56) = 0.5764, P = 0.4509; G) lead, Fmetal (1, 56) = 4.040, P = 0.0493; and H) DU, Fmetal (1, 56) = 0.5795, P = 0.4497. HNE (40 kDa) was higher in the Fe-implanted animals compared to Ta-implanted animals at both the 6M and 12M implant periods (*P = 0.0498 and *P < 0.0001, respectively). Also, in the 12M implant group, HNE (40 kDa) was higher in both the Pb-implanted (*P = 0.0126) and DU-implanted animals (*P = 0.0361) compared with Ta-implanted animals.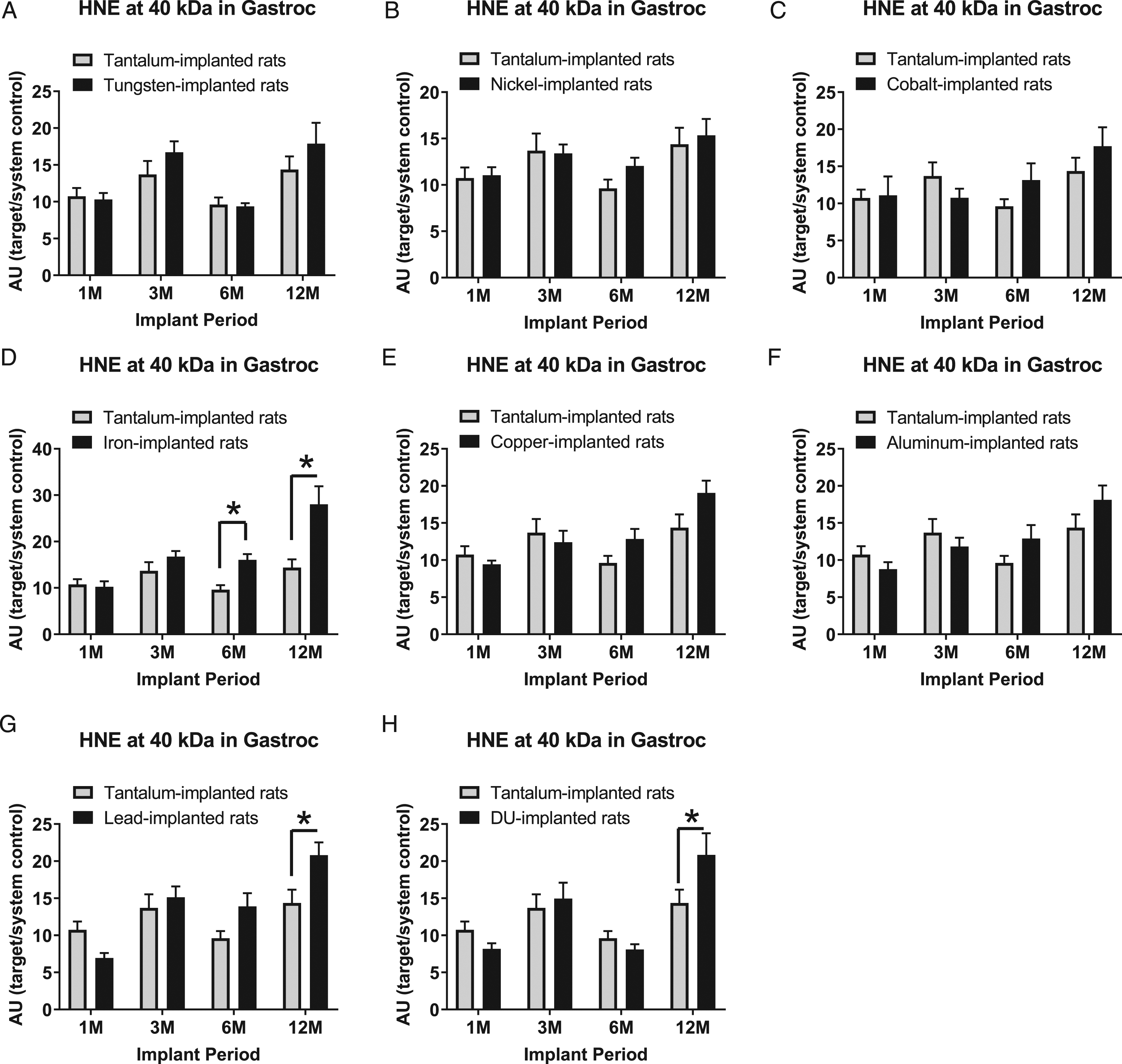
Expression of HNE-modified proteins at the 55 kDa band in gastrocnemius samples are presented in Figure 11: A) tungsten, Fmetal (1, 56) = 0.0367, P = 0.8489; B) nickel, Fmetal (1, 54) = 3.497, P = 0.0669; C) cobalt, Fmetal (1, 56) = 5.441, P = 0.0233; D) iron, Fmetal (1, 55) = 11.83, P = 0.0011; E) copper, Fmetal (1, 54) = 21.06, P < 0.0001; F) aluminum, Fmetal (1, 56) = 9.352, P = 0.0034; G) lead, Fmetal (1, 56) = 5.540, P = 0.0221; and H) DU, Fmetal (1, 56) = 5.296, P = 0.0251. HNE (55 kDa) was higher in Co-implanted animals than Ta-implanted animals in the 6M implant group (*P = 0.0433). It was also higher in Fe-implanted animals than Ta-implanted animals in the 3M and 12M implant groups (*P = 0.0349 and *P = 0.0021, respectively). Hydroxynonenal (55 kDa) was higher in Cu-implanted (*P < 0.0001) and Al-implanted (*P < 0.0055) animals compared to Ta-implanted animals in the 12M implant group. In the 6M group, HNE (55 kDa) was higher in the DU-implanted animals than the Ta-implanted animals (*P = 0.0383).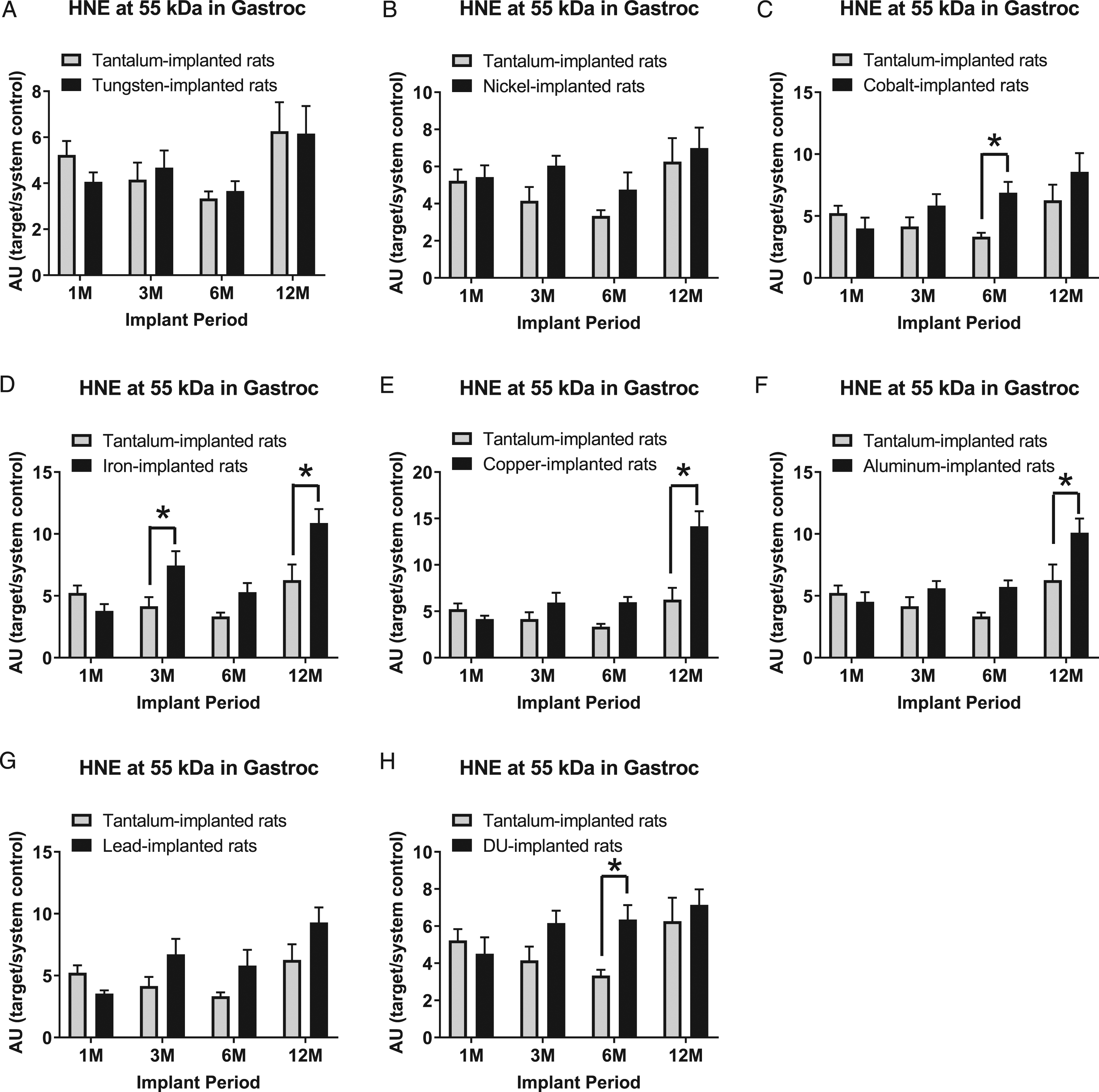
Hydroxynonenal-modified protein expression at the 62 kDa band in gastrocnemius samples are presented in Figure 12: A) tungsten, Fmetal (1, 56) = 1.696, P = 0.8991; B) nickel, Fmetal (1, 54) = 4.850, P = 0.0319; C) cobalt, Fmetal (1, 56) = 3.628, P = 0.0620; D) iron, Fmetal (1, 55) = 3.904, P = 0.0532; E) copper, Fmetal (1, 54) = 5.154, P = 0.0272; F) aluminum, Fmetal (1, 56) = 1.030, P = 0.3146; G) lead, Fmetal (1, 55) = 0.6447, P = 0.0221; and H) DU, Fmetal (1, 56) = 1.024, P = 0.3158. In the 12M groups, HNE (62 kDa) was higher in the Cu-implanted animals (*P = 0.0367) and Pb-implanted animals (*P = 0.0065) than Ta-implanted animals.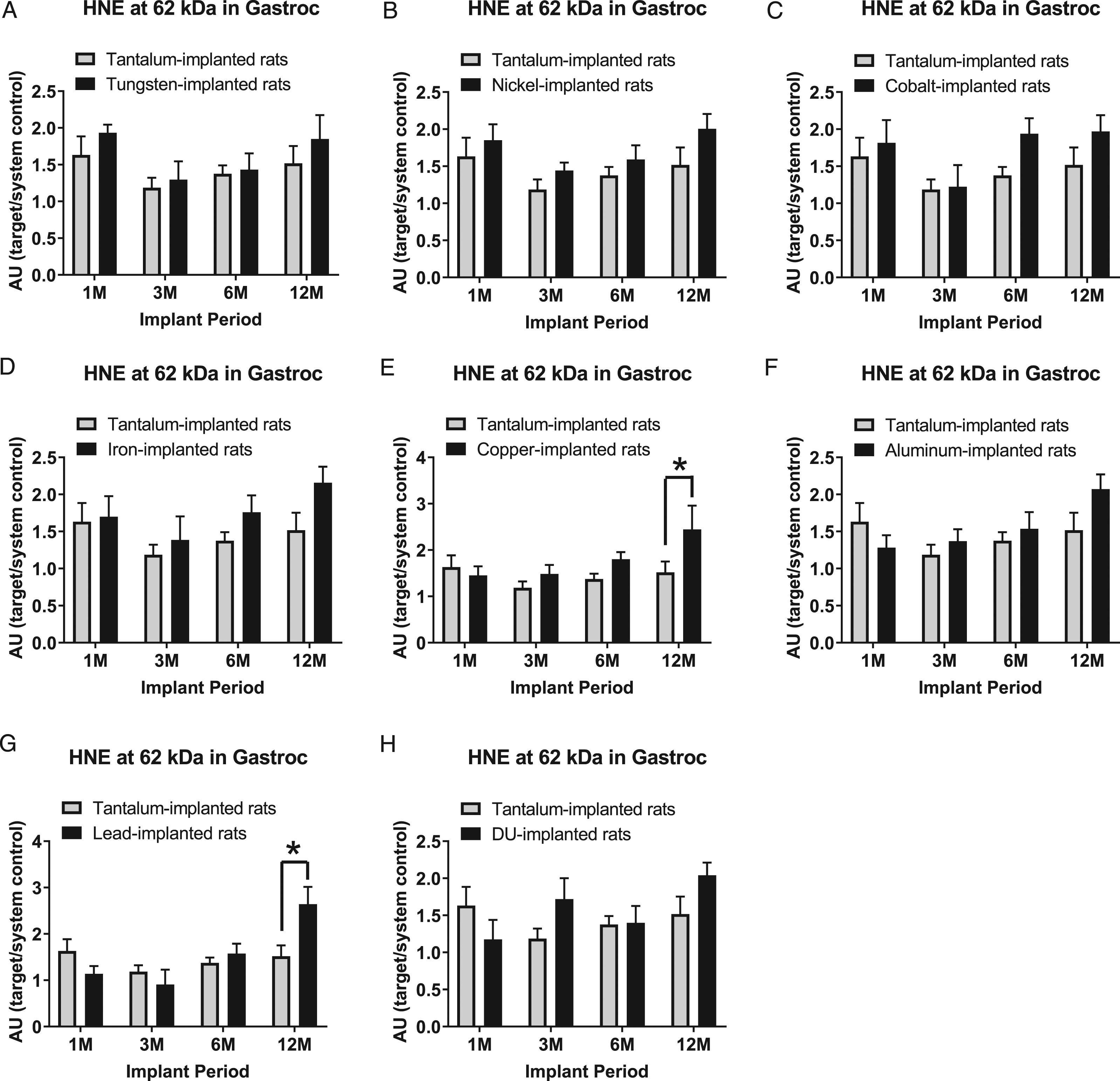
Expression of HNE-modified proteins at the 95 kDa band in gastrocnemius samples are presented in Figure 13: A) tungsten, Fmetal (1, 56) = 3.140, P = 0.0818; B) nickel, Fmetal (1, 54) = 7.475, P = 0.0084; C) cobalt, Fmetal (1, 56) = 0.4350, P = 0.5123; D) iron, Fmetal (1, 55) = 29.39, P < 0.0001; E) copper, Fmetal (1, 54) = 0.4396, P = 0.5101; F) aluminum, Fmetal (1, 56) = 0.1945, P = 0.6609; G) lead, Fmetal (1, 56) = 11.17, P = 0.0015; and H) DU, Fmetal (1, 56) = 0.1182, P=0.7323. HNE (95 kDa) was higher in Fe-implanted animals than Ta-implanted animals in the 3M, 6M, and 12M groups (*P = 0.0296, *P = 0.0299, and *P < 0.0001, respectively). It was also higher in Pb-implanted animals in the 12M group (*P = 0.0017) and in DU-implanted animals in the 3M group (*P = 0.0395) when compared to Ta-implanted animals.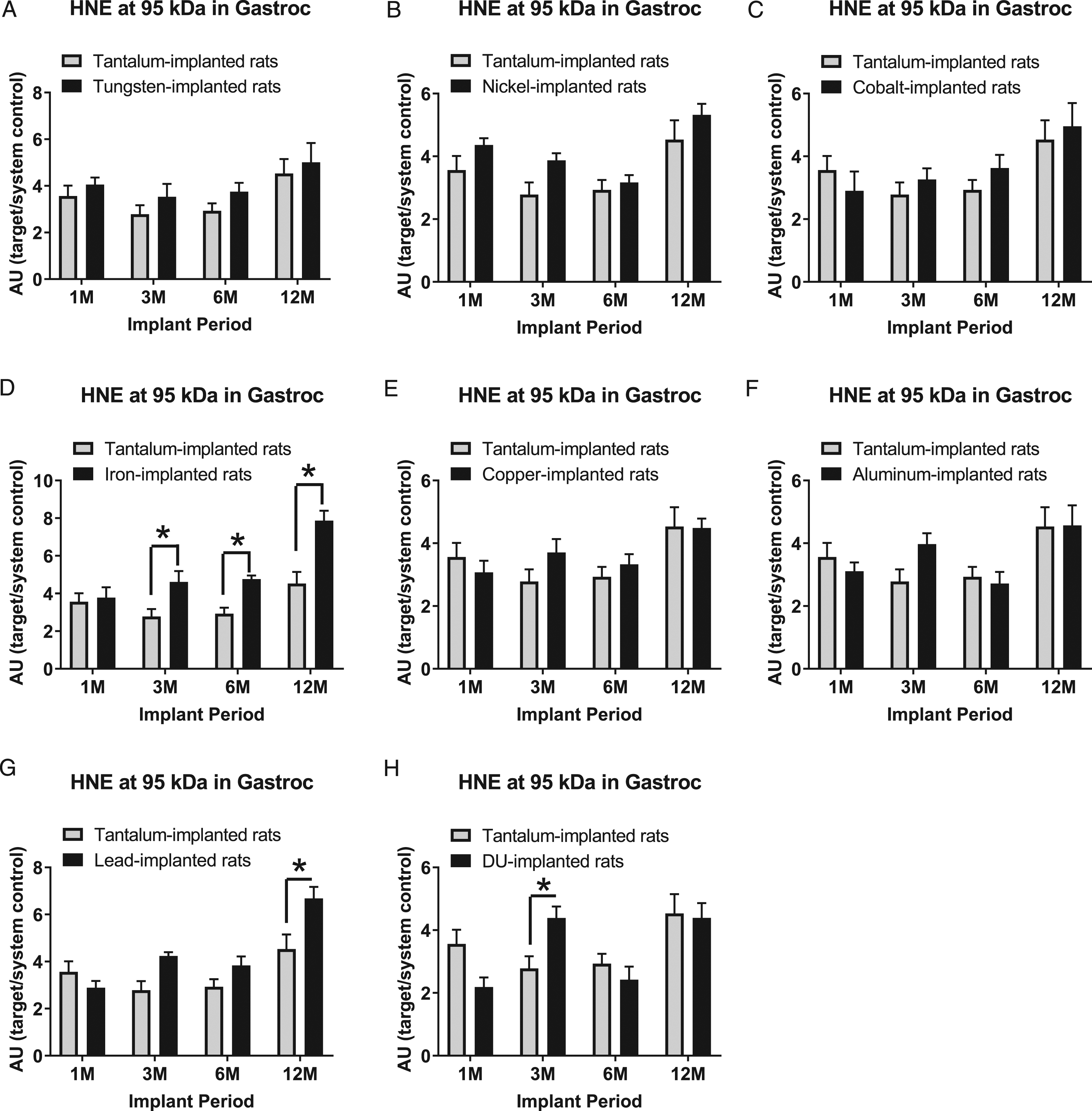
Hydroxynonenal-modified protein expression at the 144 kDa band in gastrocnemius samples are presented in Figure 14: A) tungsten, Fmetal (1, 56) = 3.581, P = 0.0636; B) nickel, Fmetal (1, 54) = 4.810, P = 0.0326; C) cobalt, Fmetal (1, 56) = 1.200, P = 0.2779; D) iron, Fmetal (1, 55) = 37.71, P<0.0001; E) copper, Fmetal (1, 54) = 0.7126, P = 0.4023; F) aluminum, Fmetal (1, 56) = 0.0319, P = 0.8590; G) lead, Fmetal (1, 56) = 18.20, P < 0.0001; and H) DU, Fmetal (1, 56) = 0.0003, P=0.9870. HNE (144 kDa) was higher in Fe-implanted animals than Ta-implanted animals in the 3M and 12M groups (*P = 0.0006, *P < 0.0001), respectively. Similarly, HNE (144 kDa) was higher in Pb-implanted animals than Ta-implanted animals in the 3M and 12M groups (*P = 0.0068 and *P = 0.0038, respectively).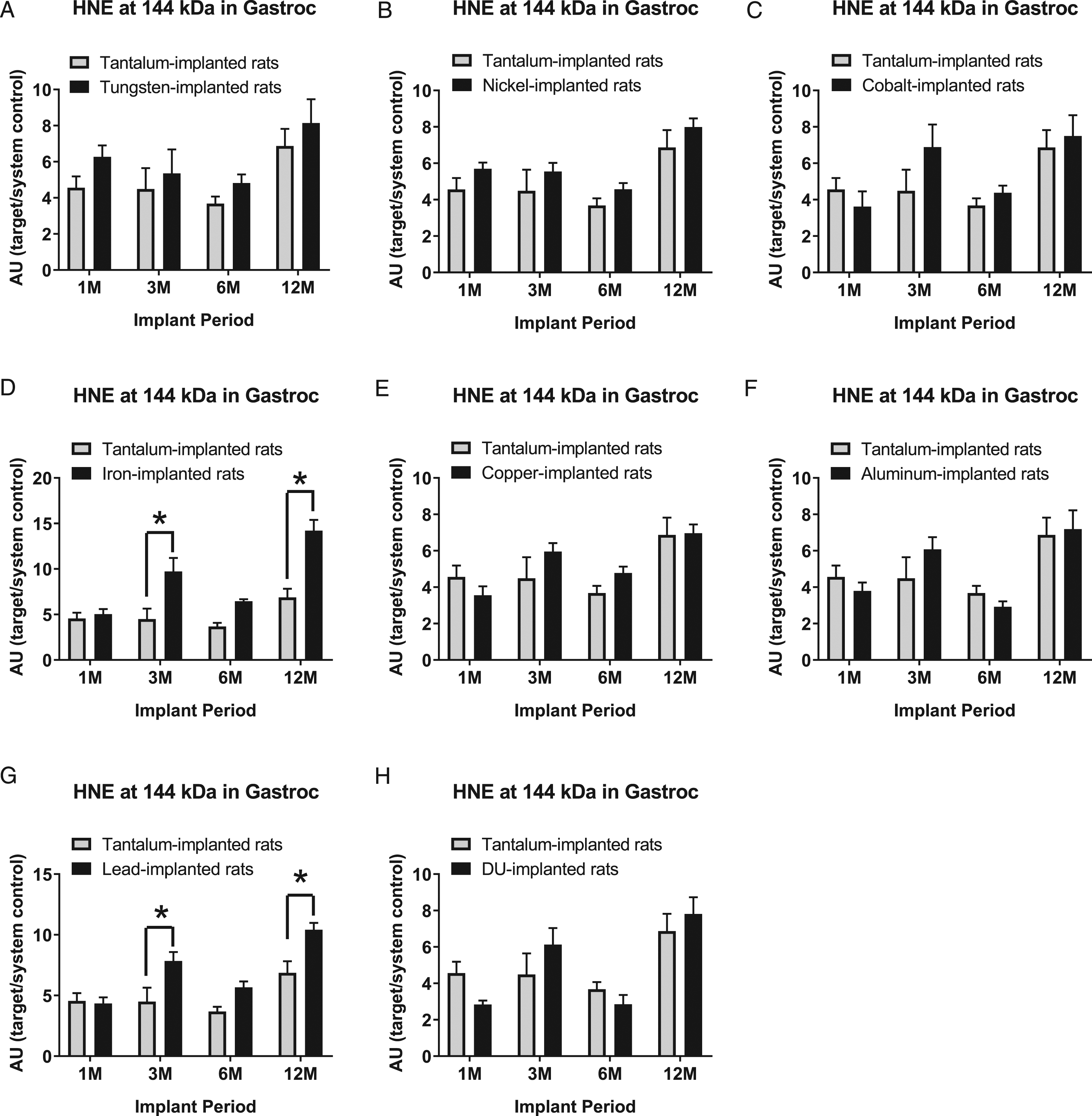
Summary of Protein Expression Changes in Metal-Implanted Gastrocnemius.

Where there is a significant post-hoc value difference of metal-implanted vs tantalum, green = increased over Ta-implanted animals, magenta = decreased over Ta-implanted animals.
Discussion
Standard surgical guidance is to leave embedded metal fragments in place unless they can be easily removed, are in a joint space, or likely to cause adverse health effects in the future. Since the toxicological and carcinogenic properties of most materials when embedded are not known, extensive surgery is usually avoided due to the risk of morbidity. This means that a wounded individual may carry these embedded metals for the rest of their lives. The appearance of uncommon metals and metal mixtures munitions, as well as armor, on today’s battlefields increases the risk of embedded metal fragment injuries. Recent results have shown that embedded metal fragments, once thought to be medically inert, are anything but.11-13,22 This increases the need for a more extensive knowledge base with respect to the health effects of embedded metals, especially considering the length of time they may be left in the body. The results here describe the localized muscle effects of embedded pellets of military relevant metals in a rat model system for up to 12 months.
Most embedded metals evidentially solubilize and are excreted in the urine and are deposited in target tissues.11,28-31 This mobilization can result from the solubilization of the metal itself as well as removal and transport of metal particles by the tissue macrophages. In this study, gastrocnemius muscle homogenates from Fe- and Co-implanted rats showed significantly elevated metal levels as compared to control suggesting that these metals rapidly solubilize once implanted. Metal levels returned to control levels by 3M post-implantation. The gastrocnemius homogenate levels of the other implanted metals were no different from control. This suggests that mobilization is occurring by another mechanism as all these implanted metals are eventually found in the urine of these rats. 25
In an earlier publication from our group on mRNA changes in metal-implanted gastrocnemius from this cohort, we also reported on histological changes in the muscle. 27 Histopathology of isolated gastrocnemius muscle from metal-implanted rats showed a variety of effects depending upon the metal. Both the Ni- and Co-implanted rats developed tumors at the pellet implantation sites, 27 with tumor incidence in the Ni-implanted rats at 100%. In addition, the severity of the tumor necessitated that humane early euthanasia of the 12M cohort. The early development onset of the Ni-induced tumors may have negated any changes in the parameters measured in this study, as the only Ni-associated change was an increase in MMP-9 expression in the 12 month cohort. While the Co-implanted rats also developed tumors, they were not as aggressive and therefore, based upon our IACUC guidelines, no early euthanasia was needed for this group. In the Ta-, W-, Cu-, Al-, and Pb-implanted groups, minimal inflammation was observed at the 1- and 3-month post-implantation time points but had resolved by 6 months. Examination of the gastrocnemius from Fe-implanted rats also showed some inflammation as well as some pigmented macrophages, suggesting that Fe particulates may be internalized by these scavenger cells. Although no tumors were observed in the DU-implanted rats, inflammation was identified at all time points.
We had originally intended to use β-actin to normalize our protein expression data. However, in our hands, β-actin was not detected in the gastrocnemius homogenates. The intermediate filament, desmin, was proposed as a normalization alternate. While desmin expression is affected by age, increasing in the gastrocnemius as the rat reaches old age (24 months), there is no indication that it would be affected at the age of the rats in this study. 32 There were no differences in desmin expression between any of the metal-implanted groups and control except for Co-, Fe-, and Cu-implanted rats at the 3-month time point. Interestingly, significant metal levels in the gastrocnemius homogenate also occur at this time point. It is not known at this time what role, if any, the increased gastrocnemius metal levels played in the increased desmin expression. However, because of these findings, target protein expression was normalized to the Wes system loading control antibody.
Matrix metalloproteinases play a major role in the repair of damaged skeletal muscle by regulating the extracellular matrix and contributing to the migration, proliferation, and differentiation of muscle stem cells.33,34 Two of the key matrix metalloproteinases in this process are MMP-2 and MMP-9. While constitutively expressed in muscle cells, they are upregulated after injury 35 with each playing a specific role in repair. Although we expect to see increased expression after metal implantation, MMP-9 expression was unchanged, with only a transient increase in expression at 3 months post-implantation for W and 12 months post-implantation for Ni. Several protein bands were identified by the primary antibody used for MMP-2 detection. The full-sized MMP-2 was found, as reported, at 93 kDa and there were also strong signals at 47 and 58 kDa as well. Thus, we analyzed all three for metal-induced protein changes. There was increased expression of the 47 and 58 kDa entities for the 1 month Fe and the 47 kDa for 1 month Cu, as well as increased expression of the 47 kDa fragment for the 12-month DU and Al groups. At 6 months post-implantation, a decrease in expression for the full-length (93 kDa) MMP-2 was seen for the Pb and DU groups, and for the 47 kDa fragment for the Pb group. These effects were transient as levels in the 12 month Pb group were no different than control. The lack of consistent metal-induced effects is difficult to interpret. However, two potential reasons are plausible. First, implantation of two pellets per gastrocnemius does not cause sufficient muscle damage to result in extended activation of repair processes. Second, any tissue damage occurring as a result of metal pellet implantation is repaired prior to the first experimental time point at 1 month. This seems to be the likely scenario as it has been reported that after skeletal muscle damage MMP-2 mRNA increases sharply up to day 3 post-injury and has returned to normal values by day 21. 36 If the same response timeline holds for metal-induced injury, we would have missed expression changes with the first experimental time point at 30 days post-implantation.
Nitric oxide (NO) is a small diffusible molecule involved in the regulation of numerous cell functions. It is synthesized by a variety of cells, including skeletal muscle cells, by the action of nitric oxide synthase (NOS) on L-arginine.37-39 There are three NOS isoforms: endothelial NOS (eNOS), neuronal NOS (nNOS), and inducible NOS (iNOS). eNOS and nNOS are constitutively expressed while iNOS is inducible depending upon cellular conditions. 40 Both eNOS and iNOS have been shown to have a role in the repair of muscle injury.41-43 Repair can loosely be divided into three distinct but overlapping stages: the destruction stage, the repair stage, and the remodeling stage. 44 NO appears to play a role in all three stages. Protein expression analysis of iNOS and eNOS showed no significant differences from control levels at the 1- and 3-month time points. However, at 6 months, iNOS levels were elevated in the Co- and Cu-implanted groups. Conversely, iNOS and eNOS levels in the Al-, Pb, and DU-implanted 6 month groups were decreased compared to control and levels stayed decreased for iNOS in the 12 month Pb- and DU-implanted groups. Once again, although beyond the scope of this study, it would be informative to assess iNOS and eNOS levels in the 0 to 14 day post-implantation period.
4-Hydroxynonenal (HNE) is one of the major aldehydic end-products resulting from the peroxidation of ω-6 polyunsaturated fatty acids such as linoleic and arachidonic acids. 45 This highly reactive intermediate has been shown to form adducts with proteins, lipids, and nucleic acids, 46 potentially contributing to a wide range of pathological conditions.47,48 It has also been reported that metals capable of catalyzing the formation of reactive oxygen and nitrogen species can enhance the formation of HNE in cells. 49 Because of the possibility that the implanted metals could induce localized oxidative stress and thus result in the formation of HNE, we investigated the number of HNE-modified proteins in the gastrocnemius homogenates from metal-implanted rats. The six protein bands ranging from 35 to 144 kDa that exhibited the greatest amount of HNE modification were analyzed for metal-induced changes. There were no significant changes in HNE-modified protein levels until the 3-month post-implantation time point and then only for Fe, Pb, and DU. The number of HNE-modified protein bands for these metals increased over time up to the final experimental end point at 12 months post-implantation. Not all metal exposures enhanced the formation of HNE-modified proteins, but the metals that did have been identified as those that undergo redox-cycling reactions (e.g., Fe, Co, Cu, and U).50-52 In addition, lead and aluminum have been reported to induce oxidative stress-induced damage in several model systems.53,54 The absence of HNE-modified proteins at the early post-implantation time points suggests that the normal antioxidant defense systems are capable of preventing any metal-induced oxidative damage such as HNE production. However, over time and especially as the animal ages, these defenses lose their ability to inhibit the production of radical oxygen and nitrogen species and ameliorate any oxidative damage. At this point, oxidative damage accumulates and, in this instance, is expressed as HNE-modified proteins. The specific identity of these HNE-modified proteins was beyond the scope of this study. However, using gastrocnemius tissue from the same set of experimental animals, we demonstrated that metal-induced transcriptional changes occur in the gastrocnemius immediately adjacent to the implanted pellet. 27 Work continues to elucidate the various pathways involved in the tissue response to embedded metals.
Conclusions
Although standard surgical guidance is to leave embedded metal fragments in place, the decision to do so should not be taken lightly. Advances in weapon design and the use of improvised explosive devices open the possibility of injuries to both combatants and civilians with metals not usually encountered. The overall goal of this multi-institute study is to identify, at an early time point, when an embedded metal could result in adverse health effects. Such information would be crucial in assessing whether the embedded metal could be safely left in place or more extensive surgical removal required. To attempt to address this question, in this study, male Sprague-Dawley rats were implanted in the gastrocnemius muscle with pellets of military relevant metals for times up to 12 months. Using homogenates of the tissue surrounding the implanted pellet, no significant increase in metal levels were found with the exception of Fe and Co at the 1-month post-implantation time point. This suggests that mobilization of embedded metals can occur via multiple mechanisms. Expression of iNOS and eNOS was no different than control in the earlier time points but was elevated at 6 months in Co- and Cu-implanted rats and depressed in Pb- and DU-implanted animals at both the 6- and 12 month points. The reasons behind these differences are unknown at this time but may reflect a difference in metal-induced gene regulation pathways. Expression of the matrix metalloproteinases involved in muscle repair and regeneration (MMP-2 and MMP-9) showed no significant changes in expression suggesting that earlier times after pellet implantation may need to be investigated. Starting at 3 months post-implantation, there were significant increases in hydroxynonenal-modified proteins in the Fe, Co, Cu, Pb, and DU groups indicating metal-induced oxidative damage of proteins. Further investigations into the localized antioxidant defenses around the embedded pellet, as well as the identity of the HNE-modified proteins await further study.
Supplemental Material
sj-pdf-1-ijt-10.1177_10915818211062878 – Supplemental Material for Protein Expression in the Gastrocnemius Muscle of a Rodent Shrapnel-Injury Model
Supplemental Material, sj-pdf-1-ijt-10.1177_10915818211062878 for Protein Expression in the Gastrocnemius Muscle of a Rodent Shrapnel-Injury Model by Jessica F. Hoffman, Vernieda B. Vergara and John F. Kalinich in International Journal of Toxicology
Footnotes
Acknowledgments
The authors would like to thank Raisa Marshall, Anya Fan, and William Danchanko for their expertise in the pellet implantation surgeries, animal welfare checks, and tissue collection. The authors would also like to thank W. Louis Wilkins for histopathology support.
Author Contributions
Conceptualization, JFK.; methodology, JFH, VBV, and JFK; software, JFH, VBV, and JFK; validation, JFH, VBV, and JFK; formal analysis, JFH and VBV; investigation, JFH, VBV, and JFK.; resources, JFK; data curation, JFH, VBV, and JFK; writing—original draft preparation, JFK; writing—review and editing, JFH, VBV, and JFK; visualization, JFH, VBV, and JFK; supervision, JFK.; project administration, JFK; funding acquisition, JFK. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project described was supported by the grant Health Effects of Blast Injuries and Embedded Metal Fragments (W81XWH-16-2-0058) from the Congressionally Directed Medical Research Program (CDMRP) Peer-reviewed Medical Research Program.
Institutional Review Board Statement
All procedures involving animals were (a) conducted with maximal possible well-being of the rats, (b) approved by the AFRRI Institutional Animal Care and Use Committee (IACUC) prior to the start of the study under protocol 2016-05-006, and (c) performed in compliance with the guidelines set forth in the Guide for the Care and Use of Laboratory Animals in an Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)-accredited facility.
Data Availability
All data supporting the results described are provided within the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
