Abstract
Drug-induced thrombocytopenia (DITP) can be triggered by antibiotics; however, the details remain unclear. Here, we evaluated the expression profiles of DITP using the Japanese Adverse Drug Event Report (JADER) database. We analyzed reports of DITP between April 2004 and January 2021 from the JADER database. The reporting odds ratio (ROR) and 95% confidence interval (CI) were used to detect DITP signals. Factors thought to affect DITP, such as male sex and an age of at least 60 years, were added as covariates. We evaluated the time-to-onset profile and hazard type using the Weibull shape parameter. The JADER database contained 1,048,576 reports. Twelve of 60 antibiotics showed signals for DITP; the RORs (95% CIs) for ampicillin/sulbactam, ceftazidime, cefozopran, ciprofloxacin, fluconazole, fos-fluconazole, linezolid, pazufloxacin, piperacillin/tazobactam, teicoplanin, trimethoprim/sulfamethoxazole, and voriconazole were 1.75 (1.41-2.16), 1.77 (1.42-2.18), 1.35 (1.06-1.72), 2.56 (2.19-2.98), 1.93 (1.67-2.23), 2.08 (1.76-2.46), 5.29 (2.73-9.60), 1.92 (1.51-2.41), 1.54 (1.05-2.19), 1.47 (1.16-1.84), 1.92 (1.73-2.14), and 2.32 (1.59-3.30), respectively. In multiple logistic regression analysis, 7 and 6 antibiotics were detected for the factors age and male sex, respectively. The median times-to-onset of DITP for ciprofloxacin (oral treatment), fluconazole, linezolid, piperacillin/tazobactam, and trimethoprim/sulfamethoxazole were 91, 91, 11.5, 10, and 9 days, respectively. Furthermore, the 95% CI of the Weibull shape parameter β for these antibiotics was above and excluded 1, indicating that the antibiotics were the wear out failure type. We revealed the expression profiles of DITP following treatment with 12 antibiotics.
Keywords
Introduction
Thrombocytopenia typically appears suddenly, is often severe, and can cause major bleeding and death. 1 Drug-induced thrombocytopenia (DITP) is triggered by an immune response in which a drug-dependent antiplatelet antibody targets glycoproteins on the cell membrane of the platelets, such as glycoprotein Ib/IX and glycoprotein GPIIb/IIIa. 2 DITP can be caused by a wide range of medications, including antibiotics 3 and nonsteroidal anti-inflammatory drugs. 4 In particular, antibiotic-induced thrombocytopenia is a serious life-threatening adverse event (AE), similar in severity to septic shock and endocarditis. Bernstein et al reported a patient with Staphylococcus epidermidis endocarditis who developed thrombocytopenia after 7 days of linezolid (LZD) administration, resulting in death. 5 However, the mechanisms, onset time, and risk factors of antibiotic-induced thrombocytopenia have not been elucidated, and further clinical research is required.
Although antibiotic-induced thrombocytopenia has been reported in several case reports,6-9 it has still considered a rare AE. Therefore, it may be difficult to carry out clinical studies owing to small sample sizes. Spontaneous reporting systems (SRSs) are useful for the detection of rare AEs and have been used as primary tools in post-marketing surveillance. In Japan, individual AEs are reported by healthcare workers to the Pharmaceuticals and Medical Devices Agency (PMDA), as is required by the Pharmaceutical and Medical Devices Affairs Law. The Japanese Adverse Drug Event Report (JADER) database, which is available online, was established in April 2004 by the PMDA and reflects the realities of clinical practice. 10 Therefore, data mining using the JADER database can be used as a screening tool for rare AEs. Recently, analysis of time-to-onset data has been conducted for detection of signals in JADER.11,12 However, to the best of our knowledge, no reports have described the details of antibiotic-induced thrombocytopenia.
Accordingly, in this study, we aimed to evaluate the expression profiles of antibiotic-induced thrombocytopenia using the JADER database.
Materials and Methods
Data Source
We downloaded the JADER database from the PMDA website (https://www.pmda.go.jp/safety/info-services/drugs/adr-info/suspected-adr/0004.html) for data from April 2004 to January 2021. The JADER database included 4 data tables, as follows: (1) demo (patients’ demographic information, including sex, age, and weight); (2) drug (drug name, causality, etc.); (3) reac (AEs, outcomes, etc.); and (4) hist (medical history, primary illness, etc.). We built relational database from the 4 data tables according to patient identification number using Microsoft Access 2019 (Microsoft, Redmond, WA, USA). Because duplicate reports in the SRS database may affect the evaluation from the relational database, we extracted only the latest of AE reports based on the demo file. Data with inaccurate dates were excluded.
Drugs and AE Names
The 60 antibiotics reported in the JADER database were analyzed as follows: anti-methicillin-resistant Staphylococcus aureus (MRSA) agents (arbekacin, LZD, teicoplanin [TEIC], and vancomycin), antifungal agents (amphotericin B, fluconazole [FLCZ], flucytosine, fos-fluconazole [F-FLCZ], itraconazole, ketoconazole, micafungin, miconazole, and voriconazole [VRCZ]), aminoglycosides (amikacin, dibekacin, gentamicin, kanamycin, and streptomycin), quinolones (ciprofloxacin [CPFX], gatifloxacin, levofloxacin, norfloxacin, pazufloxacin [PZFX], and tosufloxacin), macrolides (azithromycin, clarithromycin, and erythromycin), carbapenems (imipenem/cilastatin, doripenem, biapenem, and meropenem), tetracyclines (doxycycline and minocycline), antiviral agents (acyclovir and valaciclovir), penicillins (amoxicillin, amoxicillin/clavulanate, ampicillin, ampicillin/sulbactam [ABPC/SBT], benzylpenicillin, faropenem, piperacillin, and piperacillin/tazobactam [PIPC/TAZ]), cephalosporin (cefaclor, cefazolin, cefcapene, cefditoren, cefepime, cefmetazole, cefoperazone/sulbactam, cefotaxime, cefotiam, cefozopran [CZOP], ceftazidime [CAZ], cefteram, ceftriaxone, cephalexin, and flomoxef), clindamycin, and trimethoprim/sulfamethoxazole (TMP/SMX).
Thrombocytopenia, the target AE in the current study, was defined using 2 preferred terms (PTs) provided by the Medical Dictionary for Regulatory Activities version 23.1 J, including thrombocytopenia (PT code: 10043554) and platelet count decreased (PT code: 10035528). A flowchart for the construction of the data analysis is shown in Figure 1. Flowchart for the construction of data analysis tables from the Japanese Adverse Drug Event Report database.
Data Analysis
In the current study, we selected the reporting odds ratio (ROR) for the detection of signals associated with antibiotic-induced thrombocytopenia. As shown Figure 2, ROR and 95% confidence interval (95% CI) were calculated from a two-by-two contingency table. Detection of signal was defined as follows: ROR greater than 1, lower limit of the corresponding 95% CI greater than 1, and case number greater than 2.13,14 Conversely, the condition of ROR less than 1 was considered no exposure-event association. Subsequently, the ROR was adjusted using multivariable logistic regression analysis with covariates. Tsuji et al. reported that age may affect thrombocytopenia induced by LZD.
15
Therefore, age greater than or equal to 60 years and male sex were added as covariates in the current study. LZD was excluded from multivariable logistic regression analysis owing to the small number of reports. Multivariable logistic regression analysis was conducted with SPSS Statistics version 27 (IBM Japan, Tokyo, Japan), and the level of significance was set at P < 0.05. Two-by-two contingency table for the calculation of reporting odds ratios.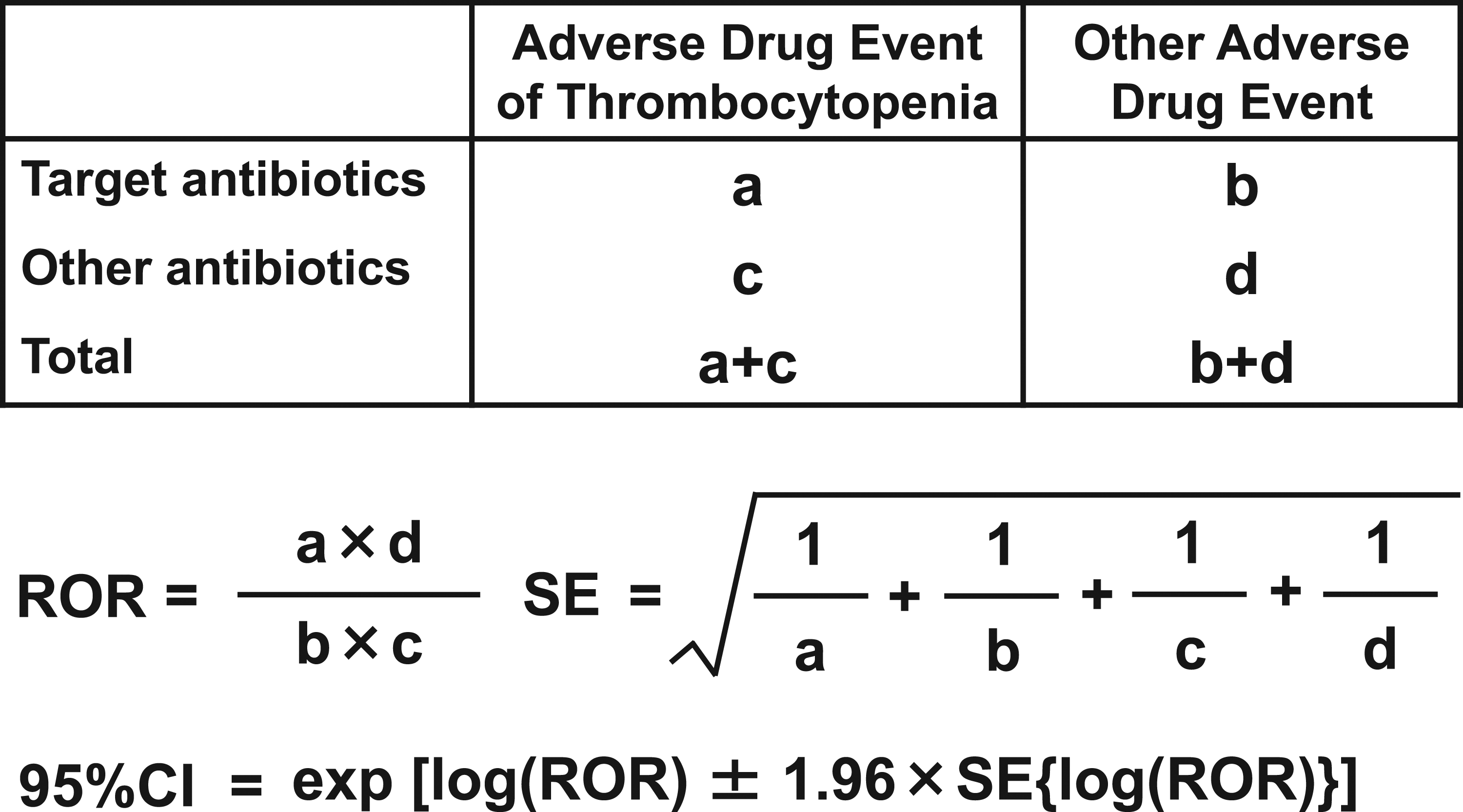
The median of the period until DITP onset from the time of first administration for each patient was evaluated using the Weibull shape parameter (WSP), which does not require a reference population. 16 The elapsed time from the first administration was calculated by subtracting the first administration date from the DITP expression date. Two hundred eighty-five cases were excluded because the date of first administration or onset of DITP was unknown (Figure 1). The combination of WSP β and 95% CI could be interpreted as follows: when β was equal to 1 (random failure) and 95% CI of β included the 1, the hazard was estimated to be constant over time; if β was greater than 1 (wear out failure) and the 95% CI (lower) of β exceeded 1, the hazard was considered to increase over time; if β was less than 1 (early failure) and the 95% CI (upper) of β was less than 1, the hazard was considered to increase at an early stage, but then decrease. Cases of oral or intravenous administration were examined to clarify the effect of the administration route on the DITP expression profile. In this study, CPFX, FLCZ, and TMP/SMX, which were available for both administration routes, were used. Time-to-onset analyses were performed using JMP version 15 software (SAS Institute, Cary, NC, USA).
Ethics Approval Statement
Ethics approval and consent to participate were not applicable to this study because we used publicly available voluntarily provided reports.
Results
Number of Reports and Reporting Odds Ratios of Thrombocytopenia Associated With Antibiotics.
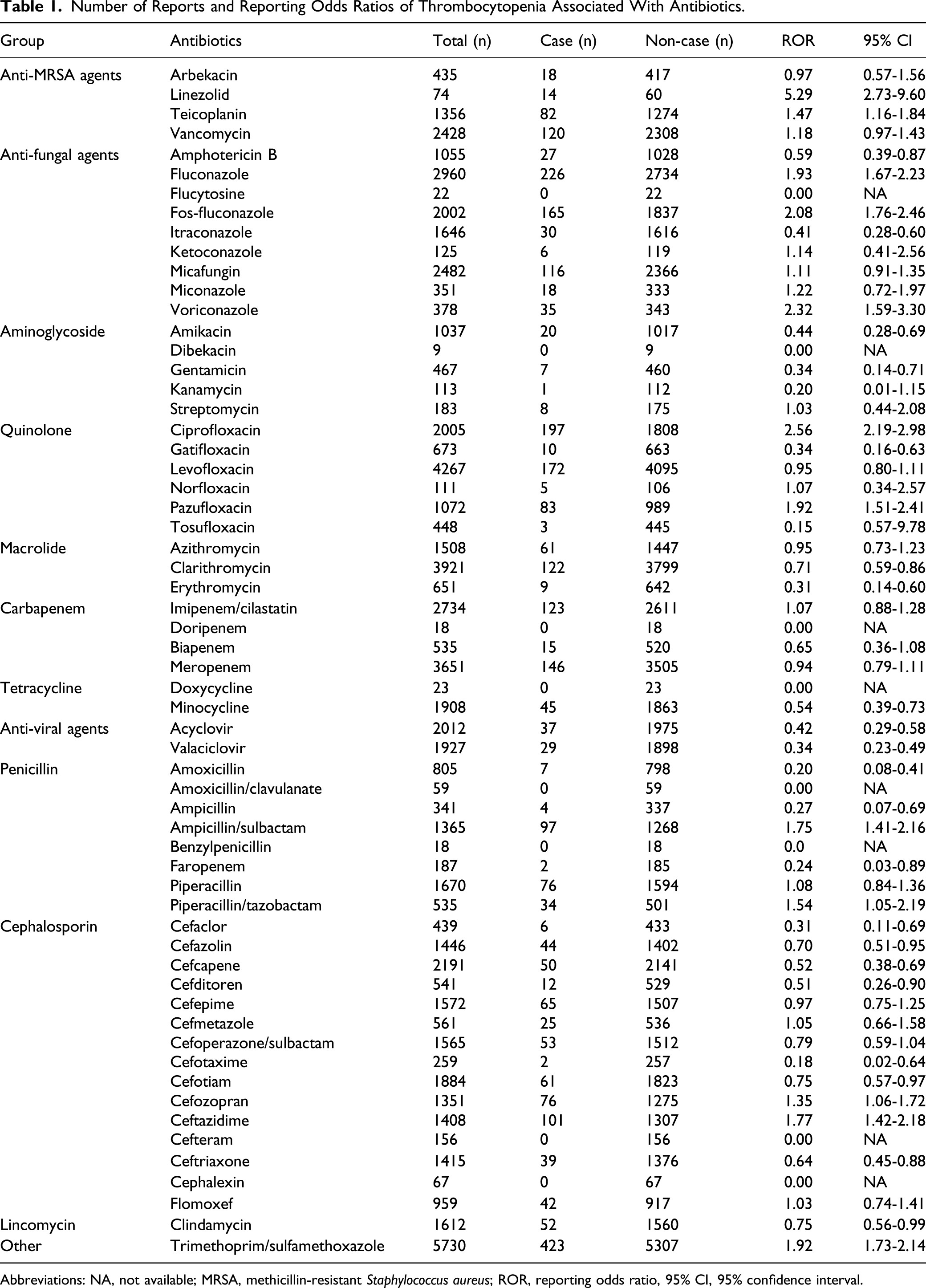
Abbreviations: NA, not available; MRSA, methicillin‐resistant Staphylococcus aureus; ROR, reporting odds ratio, 95% CI, 95% confidence interval.
Factors Affecting the Incidence of DITP According to a Multivariable Logistic Regression Model.
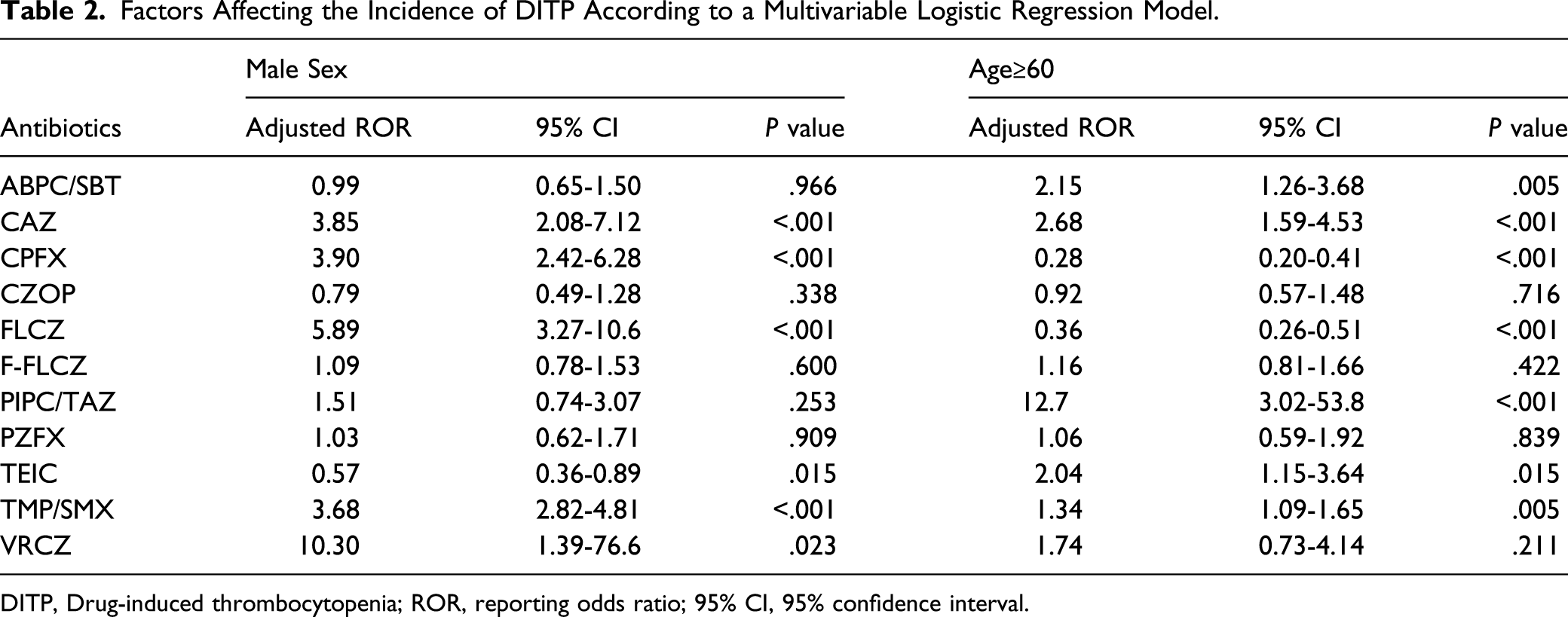
DITP, Drug-induced thrombocytopenia; ROR, reporting odds ratio; 95% CI, 95% confidence interval.
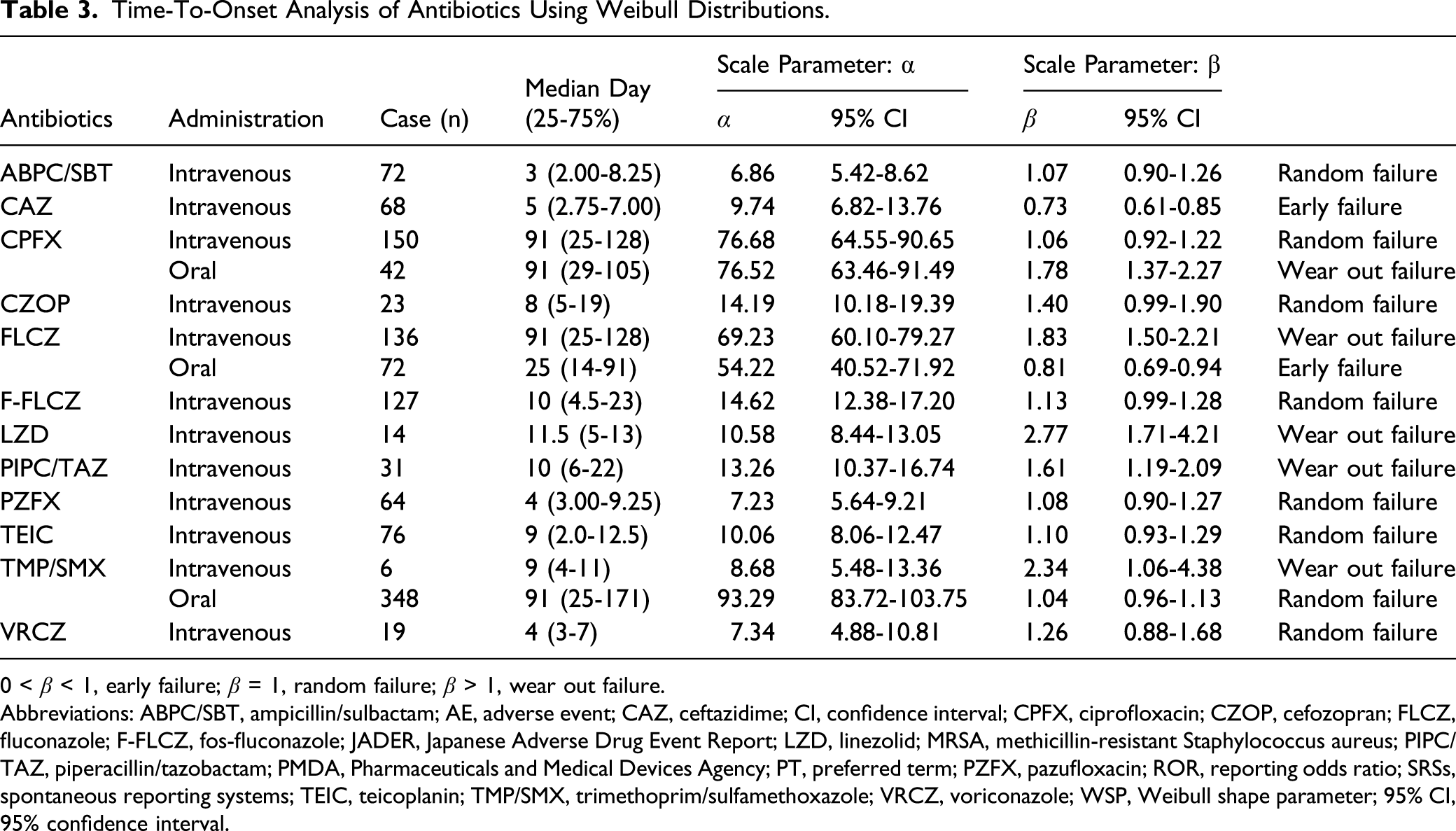
For the time-to-onset analysis, we extracted 1,248 cases for the 12 antibiotics with signal detection (n = 1533). Figure 3 shows a box plot of the number of DITP cases from the first administration date. The median durations (interquartile ranges) of DITP onset by intravenous administration of ABPC/SBT, CAZ, PZFX, and VRCZ were within 1 week of the first treatment (Table 3). The median times-to-onset of DITP for CPFX (oral treatment), FLCZ (intravenous treatment), LZD, PIPC/TAZ, and TMP/SMX (intravenous treatment) was 91, 91, 11.5, 10, and 9 days, respectively, and the 95% CIs of WSP β for these antibiotics were above and excluded 1, indicating a wear out failure type. The profiles of CAZ and FLCZ (oral treatment) were observed as early failure type. Histogram and Weibull shape parameter for antibiotic-induced thrombocytopenia. Time-To-Onset Analysis of Antibiotics Using Weibull Distributions. 0 < β < 1, early failure; β = 1, random failure; β > 1, wear out failure. Abbreviations: ABPC/SBT, ampicillin/sulbactam; AE, adverse event; CAZ, ceftazidime; CI, confidence interval; CPFX, ciprofloxacin; CZOP, cefozopran; FLCZ, fluconazole; F-FLCZ, fos-fluconazole; JADER, Japanese Adverse Drug Event Report; LZD, linezolid; MRSA, methicillin-resistant Staphylococcus aureus; PIPC/TAZ, piperacillin/tazobactam; PMDA, Pharmaceuticals and Medical Devices Agency; PT, preferred term; PZFX, pazufloxacin; ROR, reporting odds ratio; SRSs, spontaneous reporting systems; TEIC, teicoplanin; TMP/SMX, trimethoprim/sulfamethoxazole; VRCZ, voriconazole; WSP, Weibull shape parameter; 95% CI, 95% confidence interval.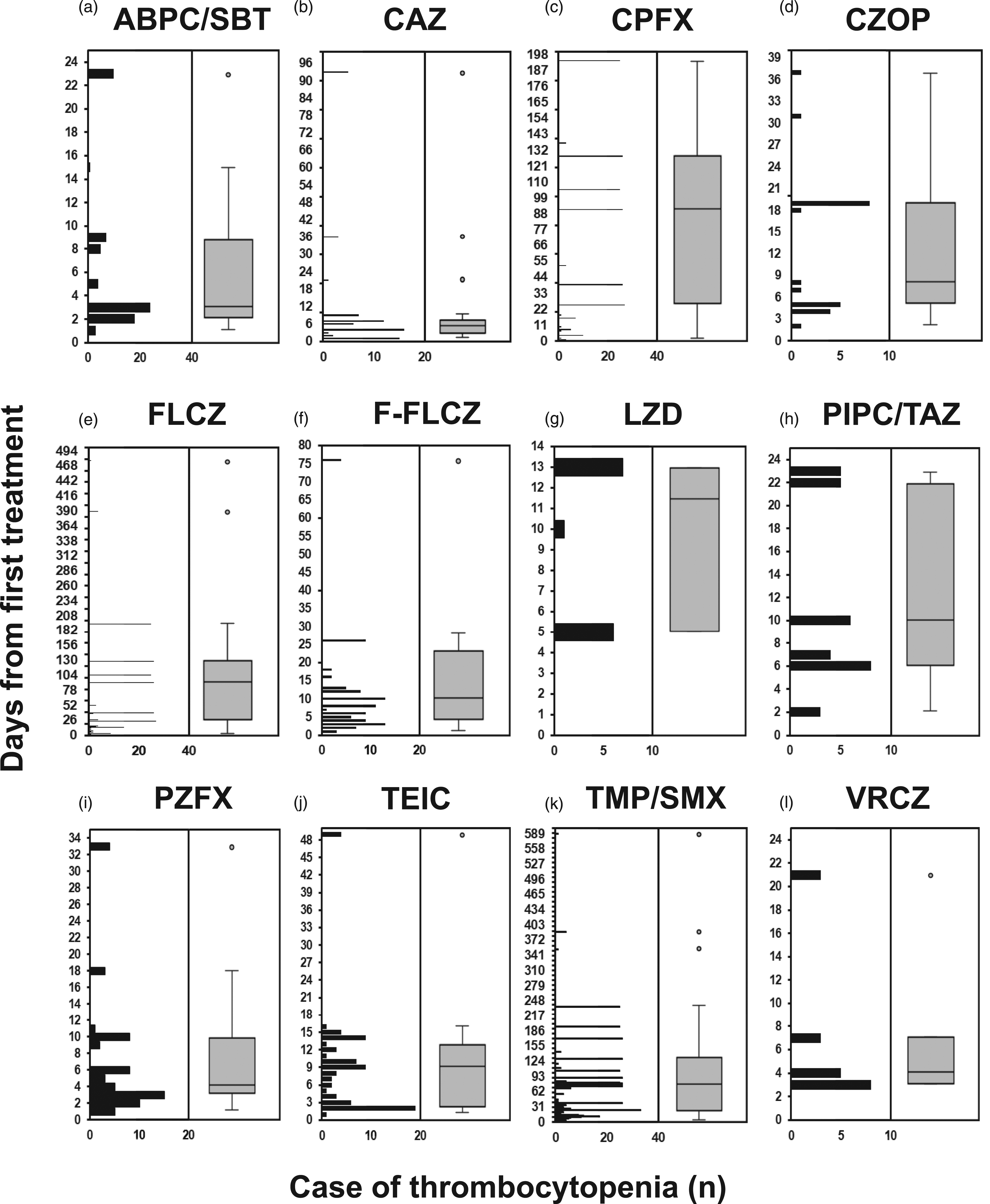
Discussion
Drug-induced thrombocytopenia is a rare AE with an expected incidence rate of .001%, but is potentially fatal. 17 Accordingly, in this study, we used RORs to evaluate the AE profiles of antibiotic-induced thrombocytopenia.
Platelets are produced in the bone marrow; therefore, we speculated that antibiotics with high cerebrospinal fluid transferability may show high expression rates of DITP before data mining. However, signals were not detected for antibiotics with high cerebrospinal fluid transferability, such as ceftriaxone 17 and cefotaxime, 18 whereas TEIC, 19 an antibiotic with poor cerebrospinal fluid transferability, was detected. Therefore, it may be difficult to explain the risk of developing DITP based on cerebrospinal fluid transferability.
Thrombocytopenia has been shown to be induced by LZD, an anti-MRSA agent, and several retrospective studies have been conducted to identify the risk factors for this condition.15,20,21 Tsuji et al. revealed that age-related renal dysfunction reduces LZD clearance-induced myelosuppression. Because all cases of LZD-induced thrombocytopenia in the current study were in elderly individuals, analysis by JADER data mining did not contradict the findings of this previous study. However, there may be a bias toward LZD because this antibiotic tends to be used more frequently in elderly individuals owing to high rates of MRSA infection in patients 70 years old and older. 22 A previous study showed that an LZD trough concentration range of 2-8 μg/mL may be beneficial for preventing treatment failure and promoting recovery from exposure-dependent thrombocytopenia, 23 suggesting that therapeutic drug monitoring for LZD may be necessary in patients who are at high risk of thrombocytopenia, including those with renal dysfunction.
Although β-lactam/β-lactamase inhibitors, such as ABPC/SBT and PIPC/TAZ, were detected, piperacillin and ampicillin were not, suggesting that tazobactam and sulbactam assessed in this study could cause thrombocytopenia. Further basic research needs to be performed to confirm this hypothesis. According to multivariate analysis, although sex differences in ABPC/SBT- and PIPC/TAZ-induced thrombocytopenia did not exist, there were age differences. The high adjusted ROR of PIPC/TAZ may reflect that PIPC/TAZ is frequently used for elderly individuals because PIPC/TAZ has an antibacterial spectrum against Pseudomonas aeruginosa, which is a common causative bacterium of pneumonia in the elderly. 24
Azole antifungals, such as FLCZ, F-FLCZ, and VRCZ, were detected in the current study. These antifungal agents are typical inhibitors of cytochrome P450 3A4, a drug-metabolizing enzyme, 25 indicating that FLCZ, F-FLCZ, and VRCZ may often exhibit drug-drug interactions. Many anticancer drugs have been shown to be metabolized by cytochrome P450 3A4. 26 Moreover, the anticancer agent gemcitabine, a substrate of cytochrome P450 3A4, 27 often causes thrombocytopenia. 28 Combination with azole antifungals may increase the risk of thrombocytopenia owing to increased blood concentrations of gemcitabine. In addition, because FLCZ, F-FLCZ, and VRCZ themselves can also cause thrombocytopenia, the risk of thrombocytopenia is potentially very high under this combination therapy, implying that more careful platelet monitoring may be required for anticancer drugs, which are metabolized mainly by cytochrome P450 3A4, 26 in combination with FLCZ, F-FLCZ, and VRCZ.
In recent reports of SRS data mining, WSP analysis has been conducted to evaluate the time-to-onset data for target AEs.11,12 Our findings in this study showed that the median time-of-onset of DITP varied for different antibiotics. A retrospective study showed that the median time of LZD-induced thrombocytopenia was 7.15 ± 5.18 days, 21 similar to that in the current study. In contrast, intravenous CAZ and oral FLCZ treatment is likely to induce thrombocytopenia relatively sooner, and it is possible that the risk of thrombocytopenia following treatment with CPFX (oral treatment), FLCZ (intravenous treatment), LZD, PIPC/TAZ, and TMP/SMX (intravenous treatment) may increase over time. Accordingly, time-to-onset analysis using WSP is likely to be a useful tool for determination of the specific safety monitoring period for DITP and rare AEs. Only intravenous TMP/SMX administration expressed DITP earlier than oral administration among the antibiotics administered intravenously or orally, implying that thrombocytopenia does not always develop earlier in intravenous administration than in oral administration. The mechanisms of DITP involve immune responses toward drug-dependent antiplatelet antibodies or drugs themselves, 29 suggesting that the differences in the time-of-onset of DITP may occur through different mechanisms.
Data mining using the JADER database has some limitations that should to recognize because the JADER database has several biases and confounders. First, because the JADER database does not contain information for control patients, the intensity of RORs cannot be quantified and compared among drugs. Furthermore, the frequency of thrombocytopenia could not be calculated in this study. On the other hand, the results of research on thrombocytopenia incidence using medical receipt data as “real world data” can be compared with those of clinical trials, which warrant further research. Second, there are also some reporting biases, such as under-reporting and lack of data. Third, since thrombocytopenia may be induced by severe infections, the thrombocytopenia reported by SRS may not be caused by administration of antibiotics.
In conclusion, this comprehensive analysis was the first report evaluating the incidences of antibiotic-induced thrombocytopenia. Despite the various limitations of using the JADER database, we identified 12 antibiotics that may be associated with high risk of thrombocytopenia, suggesting that platelet counts in patients taking these drugs should be monitored carefully in the clinical setting. If some antibiotics are shown to cause thrombocytopenia, it may be appropriate to switch to other antibiotics that have not shown DITP signals in the current study. Finally, further clinical studies are needed to verify the mechanisms through which ABPC/SBT, CAZ, CPFX, CZOP, FLCZ, F-FLCZ, LZD, PIPC/TAZ, PZFX, TEIC, TMP/SMX, and VRCZ may induce thrombocytopenia.
Footnotes
Author Contributions
Yuki, A. contributed to conception and design and contributed to acquisition, analysis, and interpretation; Takanori, Y. contributed to conception and contributed to acquisition; Yasuharu, A. contributed to conception and design and contributed to analysis. All authors drafted manuscript, critically revised manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The authors declare that they have no competing interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
