Abstract
This is a safety assessment of 40 saccharide ester ingredients as used in cosmetics. The saccharide esters are reported to function in cosmetics as emollients, skin-conditioning agents, fragrance ingredients, and emulsion stabilizers. The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the relevant data for these ingredients. The Panel concluded that the saccharide esters are safe in cosmetics in the present practices of use and concentrations described in this safety assessment.
Keywords
Introduction
This safety assessment includes the 40 saccharide esters listed below. Maltitol Laurate has been reviewed previously by the Expert Panel for Cosmetic Ingredient Safety (Panel); in 2008, the Panel concluded that this ingredient is safe as used in cosmetics. 1 Neither safety test were data available for Maltitol Laurate nor was it used in cosmetics, at the time it was previously reviewed; however, available Maltitol and Lauric Acid safety test data were used by the Panel to infer safety for Maltitol Laurate.
The saccharide esters below are listed in alphabetical order; they are also shown in Table 1, ordered by subgroups according to chain length.
Saccharide Esters Subgroups Ordered by Chain Length.

* Carbon chain length is indicated in parentheses; the number of double bonds or the double bonded hydroxyl group (in structures where these exist) within the chain is preceded by a colon.
** In the FDA VCRP data, both names are listed, but they refer to the same ingredient.
FDA = Food and Drug Administration; VCRP = Voluntary Cosmetic Registration Program.
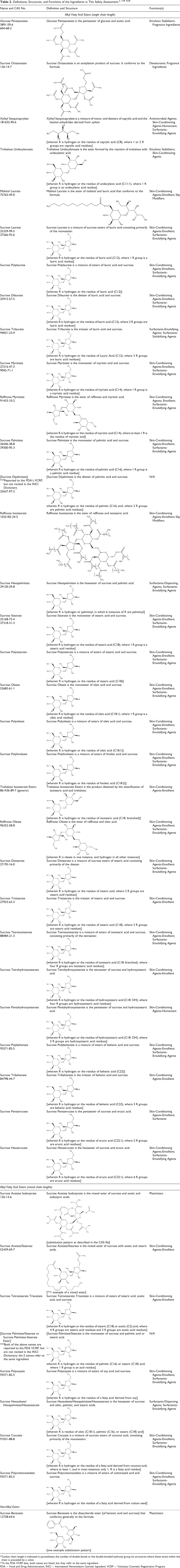
Sucrose Palmitate/Stearate, Sucrose Dipalmitate, and Sucrose Stearate-Palmitate Ester are not found in the International Cosmetic Ingredient Dictionary and Handbook (INCI; Dictionary), 2 but they are included in the US Food and Drug Administration (FDA) Voluntary Cosmetic Registration Program (VCRP) as ingredients used in cosmetic products. 3 Thus, they are included in this safety assessment. The VCRP provides data on Sucrose Palmitate/Stearate and Sucrose Stearate-Palmitate Ester as separate ingredients, however structurally they are considered to have the same definition; thus, they are presented in the Definitions and Functions table (Table 2) as 1 entry under both names.
Definitions, Structures, and Functions of the Ingredients in This Safety Assessment.2, CIR Staff
* Carbon chain length is indicated in parentheses; the number of double bonds or the double bonded hydroxyl group (in structures where these exist) within the chain is preceded by a colon.
** In the FDA VCRP data, both names are listed, but they refer to the same ingredient.
FDA = Food and Drug Administration; INCI = International Nomenclature Cosmetic Ingredient; VCRP = Voluntary Cosmetic Registration Program.
The saccharide esters have various reported functions in cosmetics, including use as emollients, skin-conditioning agents, fragrance ingredients, emulsion stabilizers, and plasticizers. 2 Xylityl Sesquicaprylate is used as an antimicrobial agent, humectant, skin-conditioning agent, and surfactant. Trehalose Undecylenoate is used as a hair-conditioning agent and surfactant. Functions reported for each ingredient are listed in Table 2.
The saccharide ester ingredients in this report are structurally related carboxylic acid esters of simple saccharides. Most of these carboxylic acids are fatty acids or mixtures of fatty acids from plant sources. All of the saccharide moieties of these ingredients, except Raffinose and Xylitol, have been evaluated by the Panel (2014) and found to be safe as used in cosmetics; the conclusions for these saccharide moieties are presented in Table 3.1,4-15 A safety assessment of Decyl Glucoside and other alkyl glucosides (differing from the glucose ester ingredients of this report only in the number of glucose equivalents) was completed (2013) with the conclusion of safe as used in cosmetics when formulated to be nonirritating. 16 Several of the constituent acids that are used to synthesize some of the saccharide esters in this report have been reviewed previously by the Panel and found to be safe as used in cosmetics; summaries of those safety conclusions are also presented in Table 3.1,4,15
Constituent Sugars, Alcohols, and Acids With Panel Conclusions.
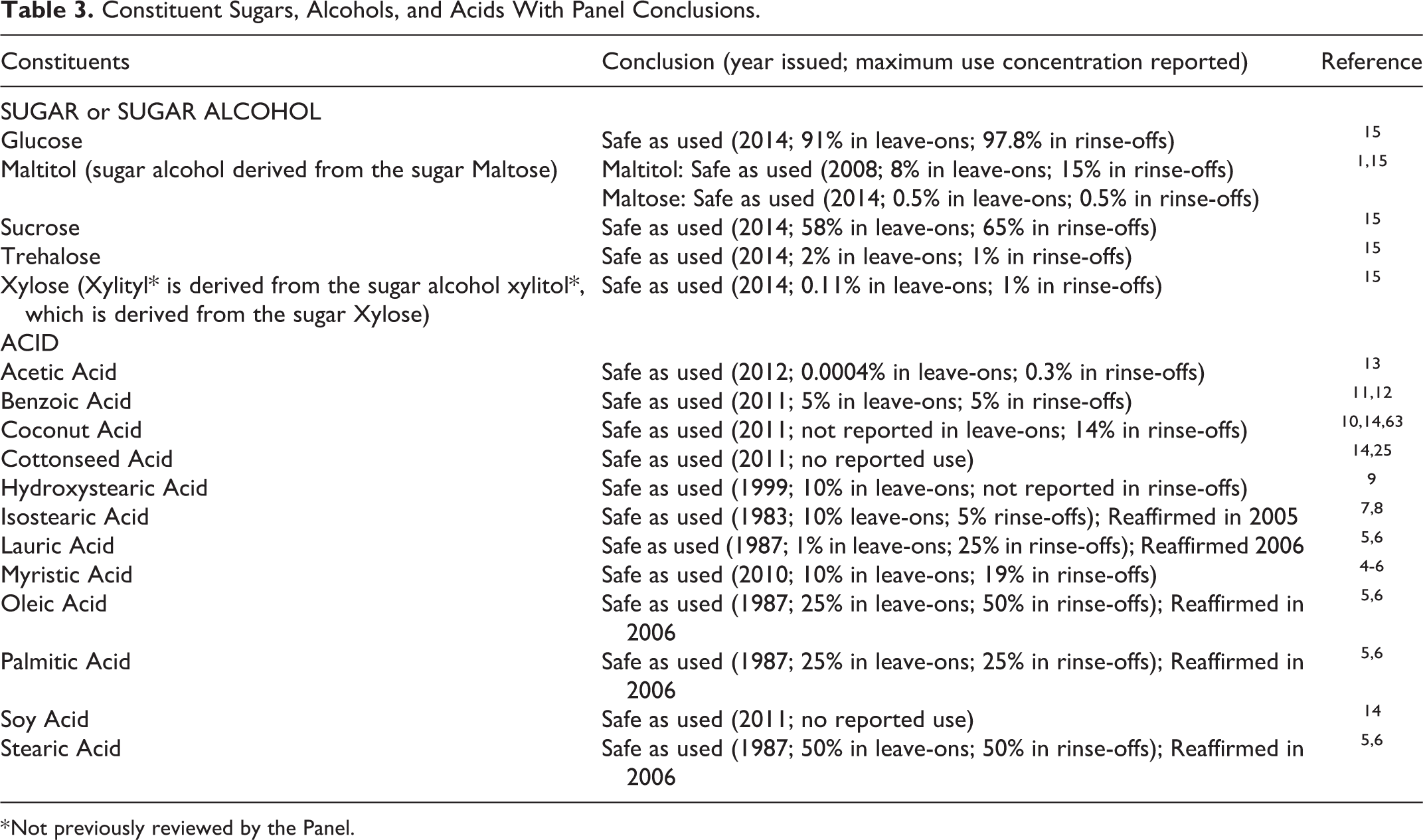
* Not previously reviewed by the Panel.
Sucrose Acetate Isobutyrate is generally recognized as safe (GRAS) for use as a direct food additive in the United States. 17 Given its GRAS status, the focus of this assessment for Sucrose Acetate Isobutyrate will be on assessing the potential for local effects, primarily dermal irritation and sensitization.
This safety assessment includes relevant published and unpublished data that are available for each end point that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the end points that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Study reports and unpublished data included in this safety assessment were found on the European Chemicals Agency (ECHA) website, 18 on the Australian Government Department of Health’s National Industrial Chemicals Notification and Assessment Scheme (NICNAS) website,19,20 and in a report published by the World Health Organization (WHO). 21 The ECHA and NICNAS websites provide data summaries from industry. The WHO report is cited when industry data submitted to the WHO are included in this safety assessment.
Chemistry
Definition and Structure
The ingredients in this report are all carboxylic esters of small saccharides. These synthetic ingredients are the end products of the esterification of simple saccharides with a carboxylic acid, such as acetic acid or a fatty acid. The sugar entity that comprises the saccharide esters is glucose (monosaccharide), sucrose (disaccharide composed of glucose and fructose), the sugar alcohol maltitol derived from maltose (a disaccharide composed of 2 glucose molecules, α-1,4 bond), trehalose (disaccharide composed of 2 glucose molecules, α-1,1 bond), raffinose (trisaccharide composed of galactose, glucose, and fructose), or xylityl derived from the sugar alcohol xylitol, which is derived from xylose (monosaccharide). While the names and definitions of some of these ingredients imply single, discrete chemical entities, it is more likely that all are mixtures of saccharide esters varying in chain length, degree of esterification, and/or regiospecificity of substitution. For example, Maltitol Laurate contains a monoester of maltitol and lauric acid but, without further specification, it is unknown whether it also contains (1) other chain-length fatty acid residues (eg, myristate); (2) di-, tri-, or tetraesters; or (3) esterification at a different active site (free hydroxyl group; Figure 1).
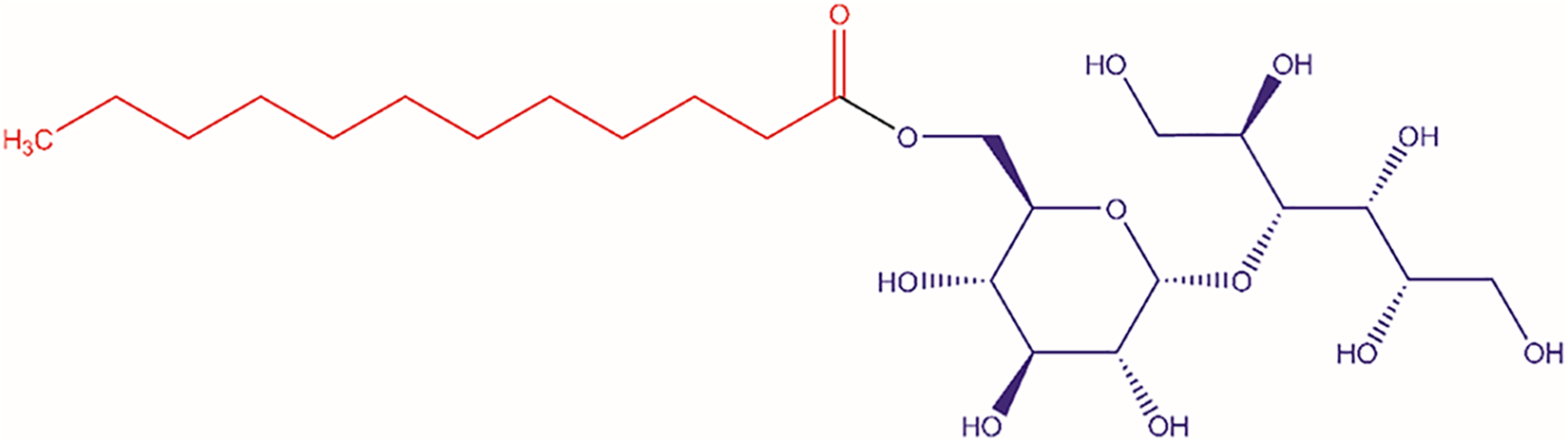
Maltitol Laurate, a saccharide ester.
The wide variation in hydrophilic-lipophilic balance (HLB), which is characteristic of small-saccharide (sugar) esters, allows these chemicals to function as oil-in-water (high HLB values) and water-in-oil emulsifiers (low HLB values). 22 The less-highly substituted esters (eg, mono-, di-, and tri-) are used for water-in-oil or oil-in-water emulsions, depending on the degree of esterification. More highly substituted sucrose esters have lower HLB values (increased lipophilicity); less highly substituted esters have higher HLB values (increased hydrophilicity). Secondary to the extent of substitution is the influence of the fatty acid chain length; the shorter the chain, the higher the HLB value.
The ingredients included in this safety assessment are defined (in order by subgroups according to chain length) in Table 2, and structures and functions in cosmetics, as presented in the Dictionary, are also provided.
Chemical and Physical Properties
Sucrose fatty acid esters (eg, Sucrose Laurate) may be stiff gels, soft solids, or white/slightly gray powders (Table 4). 23 Generally, they are sparingly soluble in water, depending on the percentage of mono esters, and soluble in ethanol. For instance, Sucrose Trilaurate has an estimated water solubility of 1.35 × 10−12 mg/L, while Sucrose Laurate (ie, 2 fewer fatty acyl chains) has an estimated water solubility of 42.37 mg/L. 24
Chemical and Physical Properties.
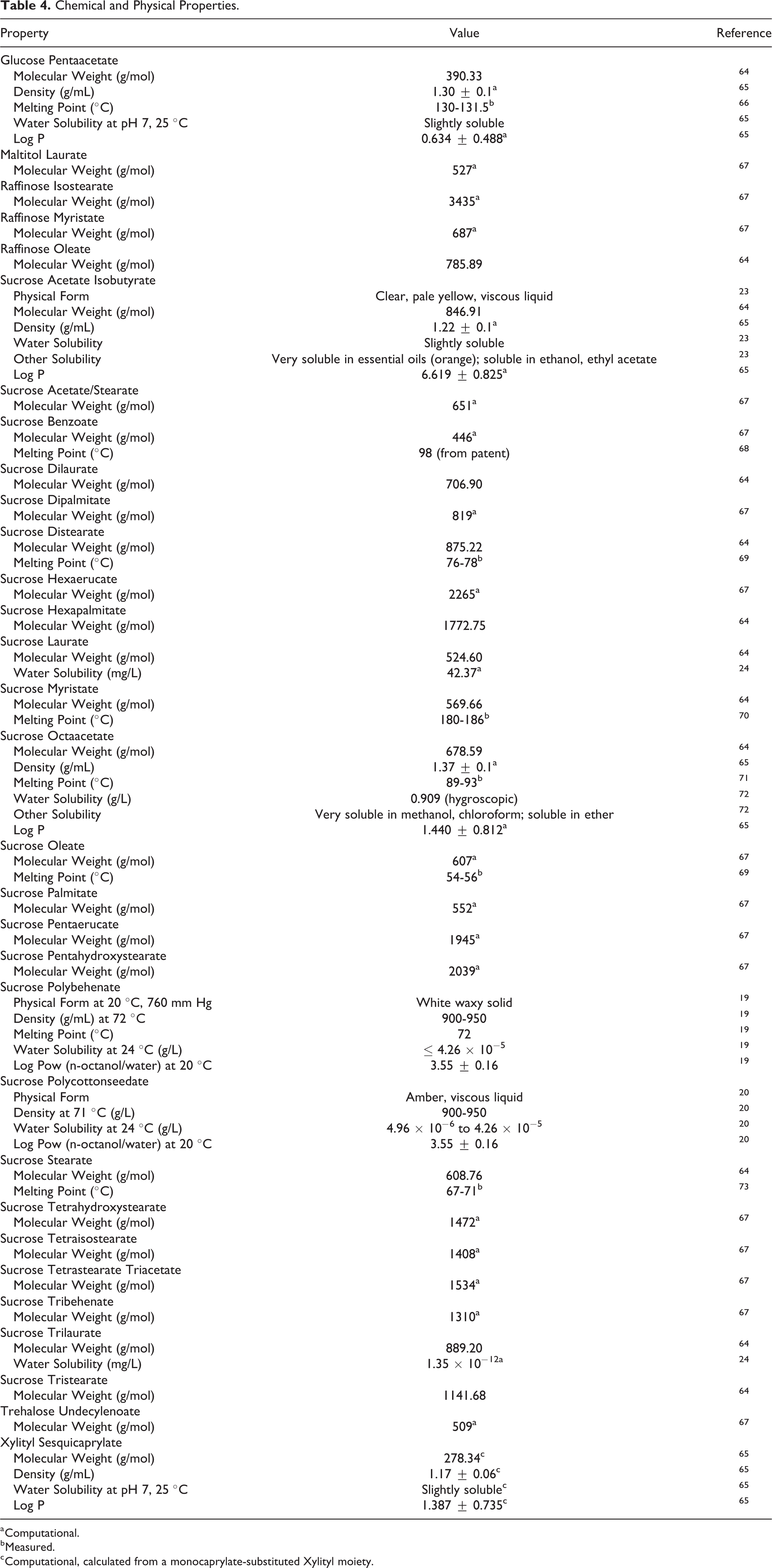
a Computational.
b Measured.
c Computational, calculated from a monocaprylate-substituted Xylityl moiety.
Method of Manufacture
Sucrose fatty acid esters (eg, Sucrose Laurate) that are used in food may be prepared from sucrose, transesterified with the methyl or ethyl esters of edible fatty acids. 23 Sucrose may also be transesterified with edible, naturally occurring vegetable oils (fatty esters of glycerol). In either type of preparation food-grade solvents, such as ethyl acetate, methyl ethyl ketone, dimethyl sulfoxide, or isobutanol may be used.
Impurities
Lead impurities are acceptable at not more than (NMT) ≤1 mg/kg (1 ppm) in Sucrose Acetate Isobutyrate used in food (based on atomic absorption spectrophotometric method), with a Sucrose Acetate Isobutyrate purity of not less than ≥98.8% and ≤101.9%, according to the Food Chemicals Codex. 23 The saponification value of Sucrose Acetate Isobutyrate used in food is acceptable at ≥ 524 and ≤540 (acid value acceptable at ≤0.2). The following acceptance criteria apply to sucrose fatty acid esters (purity ≥ 80.0% of combined mono-, di-, and triesters of sucrose) in food: ≤ 2 mg/kg (2 ppm) lead (based on atomic absorption spectrophotometric method); ≤2 mg/kg (2 ppm) dimethyl sulfoxide (based on gas chromatography/flame photometric detection method); ≤350 mg/kg (350 ppm) ethyl acetate, ≤10 mg/kg (10 ppm) isobutanol, ≤10 mg/kg (10 ppm) methanol, and ≤10 mg/kg (10 ppm) methyl ethyl ketone (based on gas chromatography/flame ionization method); ≤6 acid value; ≤ 5.0% free sucrose (based on high-performance liquid chromatography/refractive index method); and ≤2.0% sulfated ash residue on ignition.
Sucrose Polycottonseedate
Sucrose Polycottonseedate contains mixtures of cottonseed acid esters. 2 Cottonseed Acid is derived from Cottonseed Oil; impurities that may be found in cottonseed oils, and are known to be toxic, include gossypol, aflatoxin, cyclopropenoid fatty acids, heavy metals, polychlorinated biphenyls, and pesticide residues. 25 In a Panel safety assessment published in 2001, 25 the Panel concluded that Hydrogenated Cottonseed Oil, Cottonseed (Gossypium) Oil, Cottonseed Acid, Cottonseed Glyceride, and Hydrogenated Cottonseed Glyceride are safe as used in cosmetic products, with the stipulation that established limits on gossypol (< 450 ppm), heavy metals (lead ≤ 0.1 mg/kg; arsenic ≤ 3 ppm; mercury ≤ 1 ppm), and pesticide concentrations (≤ 3 ppm with NMT 1 ppm for any specific residue) are not exceeded.
Use
Cosmetic
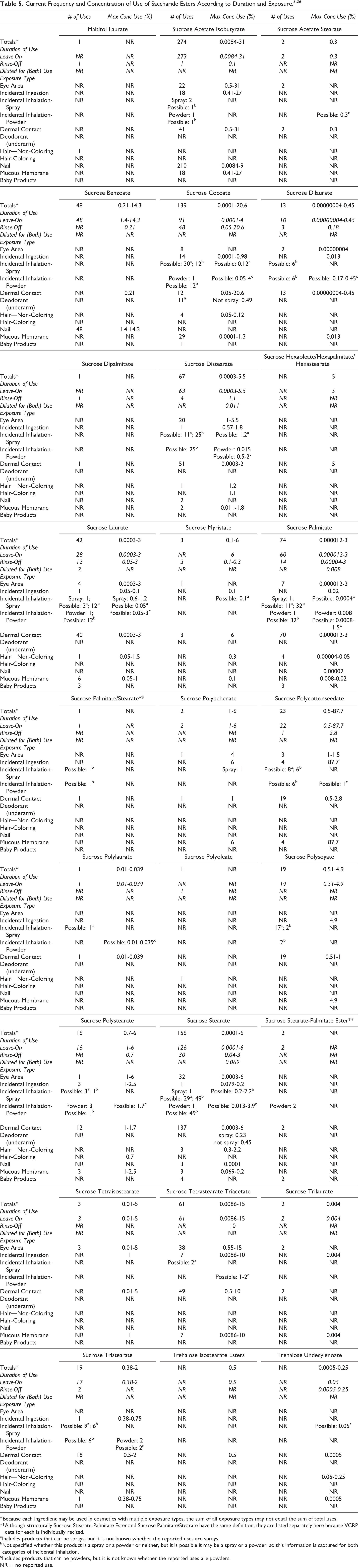
The Panel evaluated the safety of the cosmetic ingredients included in this assessment based on the expected use of and potential exposure to the ingredients in cosmetics. The data received from the FDA are collected from manufacturers through the FDA VCRP and include the use of individual ingredients in cosmetics by cosmetic product category. The data received from the cosmetic industry are collected by the Personal Care Products Council (Council) in response to a survey of the maximum reported use concentrations by product category. The VCRP data obtained from the FDA in 2016 3 indicated that the ingredients most frequently used are Sucrose Acetate Isobutyrate (274 reported uses), Sucrose Stearate (156 reported uses), and Sucrose Cocoate (139 reported uses; Table 5). The 2015 to 2016 concentration of use survey data 26 indicated that the highest maximum reported concentrations of use are as follows: 87.7% Sucrose Polycottonseedate (in lipstick); 31% Sucrose Acetate Isobutyrate (up to 31% in eye shadow and foundation; up to 27% in lipstick); 20.6% Sucrose Cocoate (in shaving soap); 15% Sucrose Tetrastearate Triacetate (up to 10% in lipstick; up to 15% in mascara); and 14.3% Sucrose Benzoate (in nail polish and enamel).
* Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
** Although structurally Sucrose Stearate-Palmitate Ester and Sucrose Palmitate/Stearate have the same definition, they are listed separately here because VCRP data for each is individually recited.
a Includes products that can be sprays, but it is not known whether the reported uses are sprays.
b Not specified whether this product is a spray or a powder or neither, but it is possible it may be a spray or a powder, so this information is captured for both categories of incidental inhalation.
c Includes products that can be powders, but it is not known whether the reported uses are powders.
NR = no reported use.
The frequency and concentration of use data are summarized, alphabetically by ingredient, in Table 5. Following the Council’s industry survey, there were no concentrations of use reported for Sucrose Dipalmitate and Sucrose Palmitate/Stearate or Sucrose Stearate-Palmitate Ester. Although listed together in Tables 1 and 2, Sucrose Palmitate/Stearate and Sucrose Stearate-Palmitate Ester are listed separately in Table 5 because the data for each are reported separately by the VCRP. 3 Only 1 ingredient (Maltitol Laurate) has been reviewed previously by the Panel, but there were no frequency or concentration of use data for this ingredient presented in the 2008 report. 1 According to 2016 VCRP data, there is 1 reported use of this ingredient in shampoos non-coloring. 3
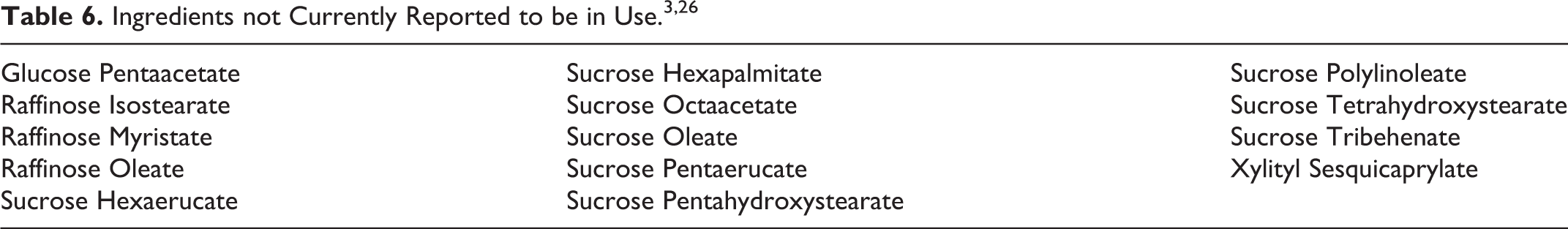
The 14 saccharide esters that are included in this safety assessment, but are not currently in use according to the VCRP and Council industry survey, are presented in Table 6.
In some cases, reported uses of saccharide esters were available in the VCRP data, but concentration of use data was not provided. For example, Maltitol Laurate is reported to be used in 1 cosmetic formulation, but no use concentration data were reported. 3 Conversely, there were instances in which no reported uses were indicated in the VCRP data, but a use concentration was provided for the ingredient in the industry survey. For example, Trehalose Undecylenoate was not reported in the VCRP data, but the industry survey indicated that it is used in leave-on formulations at up to 0.05% (in tonics, dressings, and other hair grooming aids) and in rinse-off formulations at up to 0.25%. 26 It should be presumed in these cases that there is at least one use in every category for which a concentration of use is reported.
Saccharide esters were reported to be used in perfumes, hair sprays, and deodorant sprays, and therefore, could possibly be inhaled. As examples, Sucrose Laurate was reportedly used in pump hair sprays at concentrations up to 1.2%; Sucrose Stearate was reportedly used in aerosol deodorant sprays at concentrations up to 0.23%. 26 In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters > 10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays.27-30 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount.27,28 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 28 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays. Sucrose Tristearate was reported to be used in face powders at concentrations up to 2% 26 and could possibly be inhaled. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.31-33
Saccharide esters were reported to be used in cosmetic formulations indicative of potential eye exposure (Sucrose Acetate Isobutyrate up to 31% in eye shadow), possible mucous membrane exposure (Sucrose Polycottonseedate up to 87.7% in lipstick), and possible ingestion (Sucrose Polycottonseedate up to 87.7% in lipstick).3,26 These ingredients are also incorporated into various baby products (eg, Sucrose Stearate is reported to be used in 4 baby products, but no concentration of use was reported). 3
None of the saccharide esters named in this report are restricted from use in any way under the rules governing cosmetic products in the European Union. 34
Non-Cosmetic
The non-cosmetic uses of the saccharide esters (Table 7) consist largely of either direct or indirect food additives, as specified in the Code of Federal Regulations (CFR) Title 21. Sucrose fatty acid esters are listed as direct food additives (21CFR172.859). Sucrose Acetate Isobutyrate is GRAS for use as a direct food additive in the United States. 17 Sucrose Octaacetate has been present in over the counter (OTC) drugs as nail-biting and thumb-sucking deterrents. However, the FDA has stated that Sucrose Octaacetate cannot be GRAS and effective in this application because the available safety data are not adequate to assess the safety of the use of Sucrose Octaacetate for this purpose (21CFR310.536):
Non-Cosmetic Uses.
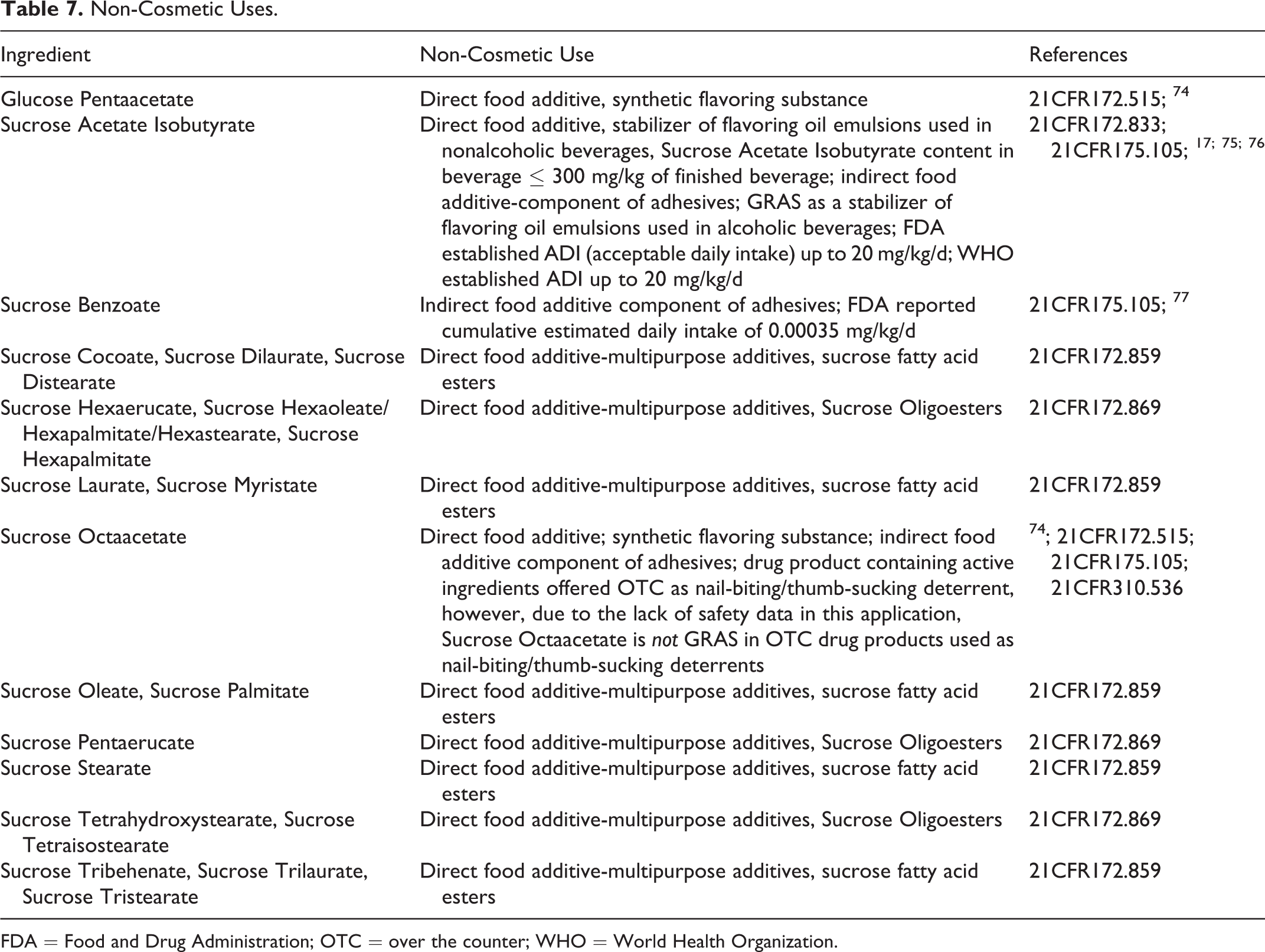
FDA = Food and Drug Administration; OTC = over the counter; WHO = World Health Organization.
Any OTC drug product that is labeled, represented, and promoted as a nail biting or thumb sucking deterrent is regarded as a new drug within the meaning of section 201(p) of the Federal Food, Drug, and Cosmetic Act (the act) for which an approved application or abbreviated application…is required for marketing. In the absence of an approved new drug application or abbreviated new drug application, such product is also misbranded under section 502 of the act. Clinical investigations designed to obtain evidence that any drug product labeled, represented, or promoted for OTC use as a nail biting or thumb sucking deterrent is safe and effective for the purpose intended must comply with the requirements and procedures governing the use of investigational new drugs…(21CFR310.536)
The following saccharide esters are listed as inactive ingredients in FDA-approved drug products: Sucrose Laurate at 30 mg in a gelatin-coated capsule for oral administration; Sucrose Palmitate at 10 mg in a powder for suspension intended for oral administration; Sucrose Stearate/Sucrose Distearate at 5% (w/w) in a topical emulsion cream; Sucrose Stearate at up to 44.5% in a sustained-action capsule or an extended-release tablet for oral administration. 35
Toxicokinetic Studies
Dermal Penetration
Human
Sucrose Laurate
Tape-stripping studies were conducted on the transport of elastic vesicles into human skin to understand the effects of occlusion and the duration and volume of application. 36 Vesicles have been used to increase the transport rate of drugs through the skin, although in tests reported here drugs were not used in the vesicle formulations. Elastic liquid-state vesicles (100-120 nm and composed of molar ratio 50:50:5; Sucrose Laurate: micelle-forming surfactant PEG-8-L: sulfosuccinate stabilizer) containing Sucrose Laurate (30% mono-, 40% di-, and 30% triesters) were evaluated in human subjects (n ≥ 3). In all of the tests, a 0.05 M citrate buffer solution (pH 5.0) was used as a control applied to the skin; no vesicles without Sucrose Laurate were used as a control. Electron micrographs corresponding to tape strip 1 (skin surface) and either 9 or 15 (deeper layers of stratum corneum) were reported for all the test conditions. In the duration test, the vesicles were applied nonocclusively (20 µL to a 1 cm 2 skin surface area), and tape-stripping was performed 1 and 4 hours after the solution had dried (evaporation of vesicle solutions was necessary to establish an osmotic gradient thought to facilitate transport into the skin). After the 1 and 4-hour treatments, vesicles were observed up to the 9th and 15th strips, respectively, with extensive vesicle fusion (multiple vesicles forming conglomerates at the skin surface and in stratum corneum) in the 4-hour treatment. In the volume test, 20 µL and 100 µL of vesicle formulation were applied non occlusively, and tape-stripping was performed 1 hour after the solution had dried. The skin surface showed no difference, based on comparison of skin structure as seen in electron micrographs, between the 20 µL and 100 µL volumes; however, in the stratum corneum, the 100 µL volume, as compared to 20 µL, increased the amount of intact vesicles (maintaining individual vesicle formation as depicted in electron micrographs) and fused vesicle material in the 9th strip. The presence of intact and fused vesicles was also noted in the electron micrographs of the first tape strips with the 20 µL and 100 µL treatments; little vesicle material was observed in the 15th tape strips at either volume tested.
The effect of occlusion was evaluated in a test performed by applying 100 µL of vesicle formulation, both occlusively and for comparison, nonocclusively, for 1 hour, after which the skin surface was wiped off and tape-stripping performed. Results from the occlusion test were: the skin surface contained vesicles (intact and fused, similar to controls) for occluded and nonoccluded samples and lipid plaques (the authors postulated that lipid plaques were the dispersal of vesicle fragments or components into the stratum corneum) in the occluded samples; unlike in nonoccluded samples, the occluded samples showed that the stratum corneum had few intact vesicles in the 9th strip, and lipid plaques were found in the 9th and 15th strips; lipid plaques may have enhanced skin permeability by disrupting intercellular skin structure organization; very few intact vesicles were present in the deeper layers of the stratum corneum during occlusion. Fast penetration of vesicles into the stratum corneum for nonocclusive treatment was noted. The researchers indicated that nonocclusive conditions facilitated the elastic vesicle incorporation into the skin by establishing a transepidermal osmotic gradient.
Penetration Enhancement
A synopsis of penetration enhancement experiments is provided below; details are provided in Table 8.
Penetration Enhancement Studies.

IV = intravenous.
In vitro
Penetration enhancement tests in vitro showed Sucrose Laurate (1.5%) evaluated in the pH range of 6 to 8 in mouse skin to be a potent percutaneous absorption enhancer for the drug lidocaine, 37 and in rat skin Sucrose Laurate (30% aqueous solution) was found to be a penetration enhancer for the drug cyclosporine. 38 Experiments in micropig skin, evaluating polyphenols in oil-in-water microemulsions (25:19:5:60, Sucrose Laurate: ethanol: isopropyl myristate: water), showed rapid distribution from the microemulsion vehicle to the epidermis, but slower dispersion from epidermis to dermis; hydrophilic polyphenols were distributed slightly more to the epidermis, and hydrophobic (small molecular weight) polyphenols distributed mainly to the dermis. 39 Sucrose Laurate and Sucrose Myristate (variable between 0.1 and 3 mg/mL) showed a concentration-dependent enhancement of paracellular permeability of a fluorescein isothiocyanate-labeled dextran marker in human nasal epithelial cells. 40 In a nanoemulsion, Sucrose Stearate (1%) was a permeation enhancer for progesterone in an in vitro porcine skin test. 41
Animal
Animal tests revealed that Sucrose Laurate (5% in a hydrogel) increased skin hydration and penetration of the drug ibuprofen in a mouse tape-stripping experiment 42 ; in rabbits, Sucrose Laurate (5% and 15% in a hydrophilic gel) increased epidermal skin-fold thickness and was a percutaneous absorption enhancer of the drug estradiol. 43 In an experiment in which rats were exposed to the drug sumatriptan succinate and 0.5% Sucrose Laurate by intranasal administration, results showed that Sucrose Laurate enhanced the effect of intranasal absorption of sumatriptan succinate. 44 Sucrose Cocoate (0.5%), when exposed via nasal administration using a pipette or by ocular installation, was found to increase the absorption of drugs (insulin and calcitonin) 9-fold (nasal) and 4-fold (ocular) in rats. 45
Human
Sucrose Palmitate (2%) and Sucrose Stearate (0.5%) in a tape-stripping (12×) experiment in human subjects increased skin absorption of the drug aceclofenac, which was then detected at all depths of the stratum corneum. 46 Sucrose Oleate and Sucrose Laurate, both tested at 2% and 10% in human subjects, increased skin penetration of the drug 4-hydroxy-benzonitrile. 47
Absorption, Distribution, Metabolism, Excretion
A brief summary of absorption, distribution, metabolism, and excretion studies is provided below; details are provided in Table 9.
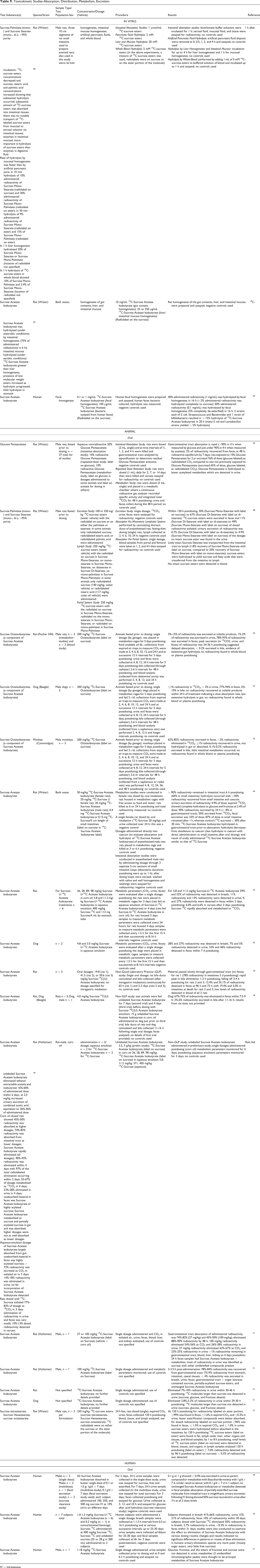
Toxicokinetic Studies-Absorption, Distribution, Metabolism, Excretion.
IV = intravenous.
In vitro
Experiments showed that a mixture of 1 µmol/mL Sucrose Palmitate and Sucrose Stearate ([ 14 C] radiolabels on either the sucrose or ester portion) was not transported from mucosal to serosal solution in everted intestinal sacs of rats; hydrolysis by mucosal homogenates from the rats was 10% to 30%, compared with little hydrolysis in whole blood. 48 Results from another rat test indicated that up to 250 µg/mL [ 14 C]Sucrose Acetate Isobutyrate (label on sucrose) was 75% hydrolyzed by intestinal homogenates in 6 hours; less hydrolysis occurred in the stomach and liver. 49 A study, in which human fecal homogenates were incubated with 1 mg/mL and 0.1 mg/mL [ 14 C]Sucrose Acetate Isobutyrate (label on sucrose), resulted in 40% and 60% hydrolysis, respectively, in 16 hours. 49
Animal
In oral studies in rats, Glucose Pentaacetate (20% aqueous solution, no radioactive label used) was rapidly absorbed (> 90% in 4 hours), 50 and excretion occurred mostly in the feces after administration of a [ 14 C]Sucrose Palmitate and Sucrose Stearate mixture (details on radiolabeling are specified in Table 9) 48 ; a mixture of sucrose esters (250 mg/kg, radiolabels on either sucrose or ester portion, see Table 9 for details), including Sucrose Hexastearate, was hydrolyzed prior to intestinal absorption (less esterified compounds were better absorbed) and largely excreted in feces (>96%-99% of radioactivity) at 120 hours postdosing 51 ; and 200 mg/kg of Sucrose Octaisobutyrate (a component of Sucrose Acetate Isobutyrate, 14 C label on sucrose) was excreted in feces (78%-93% of dosed radioactivity), excreted as a volatile product (3%-15% of dosed radioactivity), and eliminated in urine (1% to 2% of dosed radioactivity). 52 In dogs and monkeys orally administered 200 mg/kg of Sucrose Octaisobutyrate ([ 14 C] label on sucrose), no dosed radioactivity was detected in whole blood or plasma and excretion in feces was 77% to 94% of dosed radioactivity and 62% to 85% of dosed radioactivity, respectively. 52 In dogs, Sucrose Octaisobutyrate was slowly absorbed, with less extensive hydrolysis in the gut, as compared to rats; in monkeys it was not absorbed or hydrolyzed in the gut. 52
Human
In single-dose (0.1 g or 1.0 g Sucrose Acetate Isobutyrate) and multidose (1 g/d Sucrose Acetate Isobutyrate for 7 d) oral exposure studies, results indicated that < 0.4% were excreted in urine as the parent compound or metabolite with a disaccharide moiety. 49 In a fecal excretion study, 0.1 g/d Sucrose Acetate Isobutyrate was administered to 1 subject for 7 days and no unchanged Sucrose Acetate Isobutyrate or metabolites were detected in fecal samples. The absorption of partially esterified sucrose molecules from the intestinal tract was insignificant. In a different test, human subjects were administered a single, oral dosage of 1.0 to 1.2 mg/kg Sucrose Acetate Isobutyrate (radiolabel on sucrose) and exhaled 41% to 66% of dosed radioactivity in the breath within 30 days postadministration; 15% to 21% of dosed radioactivity was eliminated in urine and 10% of dosed radioactivity was excreted in feces. 53
Toxicological Studies
Acute Toxicity Studies
Provided below is a synopsis of the acute (single exposure) toxicity studies that are presented in detail in Table 10.
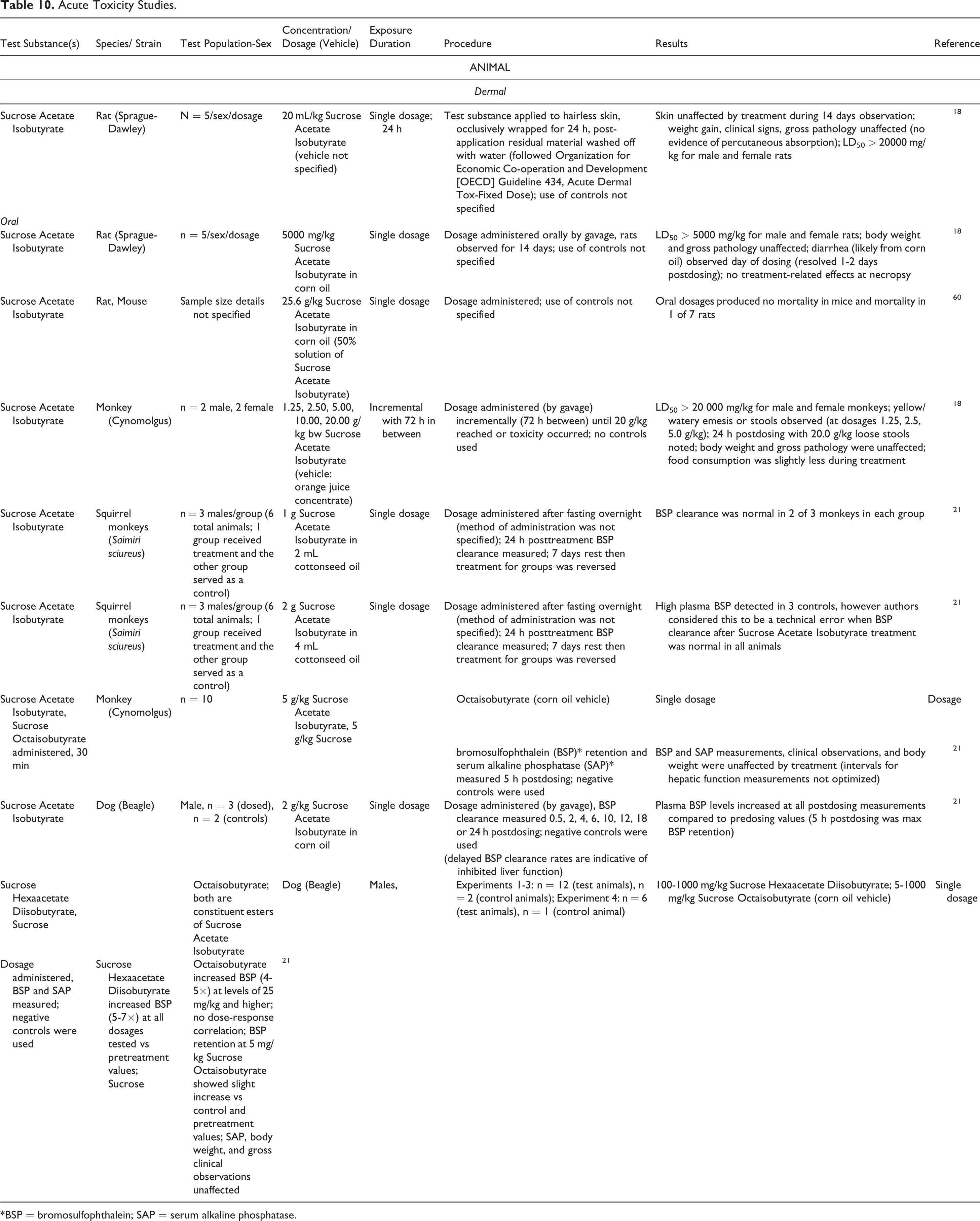
Acute Toxicity Studies.
* BSP = bromosulfophthalein; SAP = serum alkaline phosphatase.
The LD50 was reported to be > 20 g/kg in a study in which a single dosage of Sucrose Acetate Isobutyrate was dermally applied to rats. 18 For rats and monkeys orally administered single dosages of Sucrose Acetate Isobutyrate, the LD50 was reported to be > 5 g/kg and > 20 g/kg, respectively. 18 In dogs orally administered a single dosage of 2 g/kg Sucrose Acetate Isobutyrate, an increase in plasma bromosulfophthalein (BSP) levels was reported. 21 Constituent esters of Sucrose Acetate Isobutyrate, namely Sucrose Hexaacetate Diisobutyrate and Sucrose Octaisobutyrate, were both shown to increase BSP levels in dogs when single dosages up to 1 g/kg were orally administered; however body weight and gross clinical observations were unaffected by the treatment. Overall, single, high dosages of Sucrose Acetate Isobutyrate, administered through dermal and oral exposure, were well-tolerated in animals.
Short-Term Toxicity Studies
Below is a synopsis of the short-term toxicity studies that are presented in detail in Table 11.
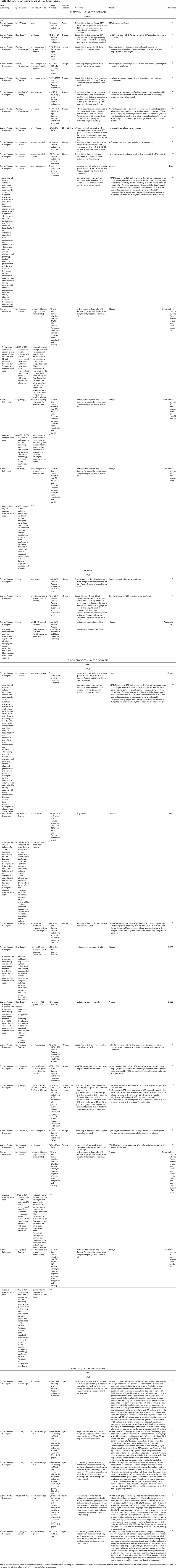
Short-Term, Subchronic, and Chronic Toxicity Studies.
BSP = bromosulfophthalein; ICG = indocyanine Green plasma clearance rates indicating liver functionality; NOAEC = no-observed-adverse-effect concentration; NOEC = no-observed-effect concentration; NOAEL = no-observed-adverse-effect level; SAP = serum alkaline phosphatase.
Animal
Sucrose Acetate Isobutyrate was well-tolerated in orally exposed animals. In rats, a no-observed-adverse-effect concentration (NOAEC) was reported for doses up to 10% daily in the diet for 6 weeks (decrease in mean heart weight for treated males was noted) 54 ; monkeys dosed up to 10 g/kg/d in the diet (for 15 days) showed no change in body weight, food consumption, or clinical parameters 21 ; mice dosed up to 5 g/kg/d in the diet (for 4 weeks) were unaffected by the treatment 55 ; in a 2-week study in dogs dosed up to 0.5% daily in the diet, BSP retention was reported at 0.3% and 0.5%. 21 Sucrose Polysoyate was orally administered to rats and dogs. In rats dosed for 28 days, a no-observed-effect concentration (NOEC) of 15% was reported; softer feces, lower growth rates (dose-related), and a dose-dependent heart weight decrease were observed.19,20 Two studies in dogs reported an NOAEC and (NOEC) of 15% daily in the diet for 28 days; results showed a higher food consumption in treated animals compared to controls, yet hematology, urine, and organs were unaffected by treatment.19,20
Human
In 3 different studies conducted in human subjects orally administered up to 0.02 g/kg/d Sucrose Acetate Isobutyrate for 14 days, results showed no blood chemistry or hematological abnormalities. 21
Subchronic Toxicity Studies
Below is a synopsis of the subchronic toxicity studies that are presented in detail in Table 11.
An NOAEL of 10% daily in the diet was reported in a 12-week study in rats orally administered Sucrose Acetate Isobutyrate; a decrease in mean heart weights in all treated males was observed. 54 In 13-week studies in rats dosed with up to 9% Sucrose Acetate Isobutyrate daily in the diet, slight diarrhea was reported, but no toxic effects were observed.18,21 Dogs orally administered up to 5% Sucrose Acetate Isobutyrate daily in the diet for 91 days showed a moderate elevation in serum alkaline phosphatase (SAP) liver enzyme, heavier liver weights compared to controls, and a functional effect on the liver (reversible when Sucrose Acetate Isobutyrate was removed from the diet). 18 In rats orally administered Sucrose Polysoyate for approximately 90 days, an NOEC of 15% daily in the diet was reported; softer feces and lower growth rates were noted, an increase in food consumption was seen with increasing doses, however no toxicity was observed.19,20
Chronic Toxicity
Provided below is a summary of the chronic toxicity studies that are presented in detail in Table 11.
Sucrose Acetate Isobutyrate was evaluated in monkeys in a 1-year study in which an NOAEL of 2.4 g/kg/d (the highest dosage rate tested) was reported. 56 Although statistically significant changes in hematological parameters were reported at the higher dosage rates (1.45-2.4 g/kg/d), overall the treatment was well-tolerated. Reported from a 1-year rat study testing Sucrose Acetate Isobutyrate was an NOAEL of 2 g/kg/d (highest dosage rate tested). 55 Body weight gain decreases were observed in males and females (2 g/kg/d); 1 female death (0.5 g/kg/d) was noted, 1 female was killed in a moribund condition (2 g/kg/d), and 1 control and 2 treated rats died during blood collection. Small, but statistically significant hematology differences between control and treated groups occurred at varying dosage rates and time points in the study; however all resolved by 54 weeks. The same researchers conducted 2-year toxicity studies (including evaluations for carcinogenicity; see Carcinogenicity section in-text) in rats and mice. Survival rates for rats were 46% to 78% and for mice were 66% to 80%. The NOAELs reported for rats and mice were 2 g/kg bw/d (highest dosage rate tested) and 2.5 g/kg/d, respectively. In mice, the NOAEL did not include the highest dosage rate tested (5 g/kg/d) because at that dosage rate, there was a treatment-related decrease in mean absolute and relative kidney weights observed at necropsy in males, compared to controls. Another 2-year toxicity study (carcinogenicity evaluations were performed; see Carcinogenicity section in-text) was conducted in rats (concentration up to 9.38% in diet). 21 A dose-related increase in absolute and relative kidney weights was noted; however the organ weight findings were deemed inconclusive because of discrepancies in male body weights, as compared to controls, and low survival numbers. Within 10 weeks of the study, 4 males dosed with 9.38% died (massive hemorrhages in multiple organs were reported), but the deaths were not attributed to treatment (no further details specified).
Developmental and Reproductive Toxicity Studies
Provided below is a summary of the developmental and reproductive toxicity (DART) studies that are presented in detail in Table 12.
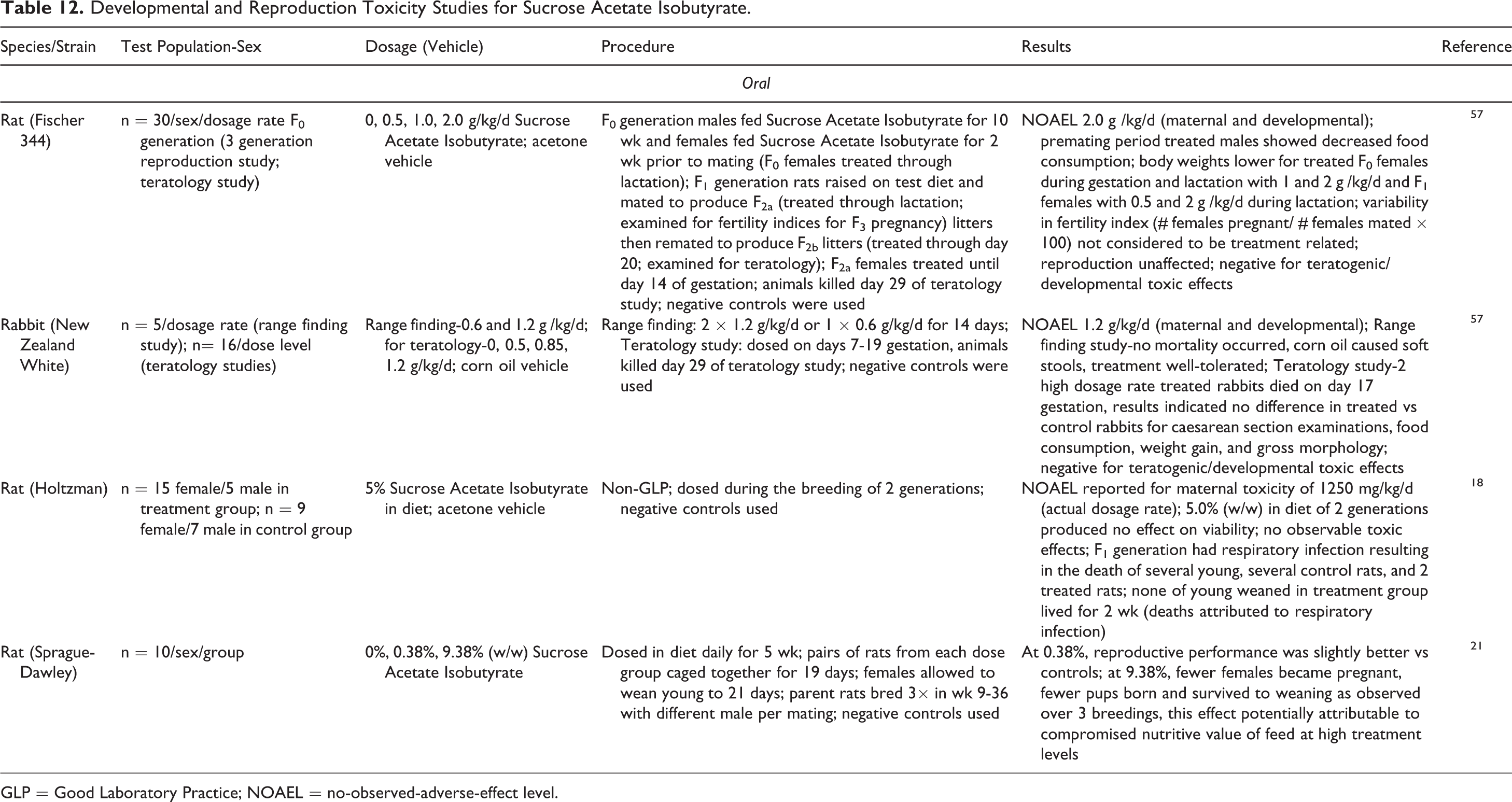
Developmental and Reproduction Toxicity Studies for Sucrose Acetate Isobutyrate.
GLP = Good Laboratory Practice; NOAEL = no-observed-adverse-effect level.
Four oral-exposure DART studies are reported for Sucrose Acetate Isobutyrate in rats and rabbits. In a 3-generation dietary study in which male rats were dosed daily for 10 weeks and female rats for 2 weeks prior to mating, teratogenic and developmental toxic effects were not observed and an NOAEL of 2 g/kg/d, the highest dosage rate tested, was reported. 57 In another dietary study, rats fed 9.38% in the diet for 5 weeks (rats bred 3× during weeks 9-36) showed fewer pregnancies and fewer pup births with survival to weaning. However, this was attributed to the potentially reduced nutritive value of the diet. 21 In a study in which rabbits were dosed on days 7 to 19 of gestation by gavage, an NOAEL of 1.2 g/kg/d was reported; 2 of 16 rabbits dosed at this level died on day 17 of gestation, but teratogenic or developmental toxic effects were not observed. 42
Genotoxicity Studies
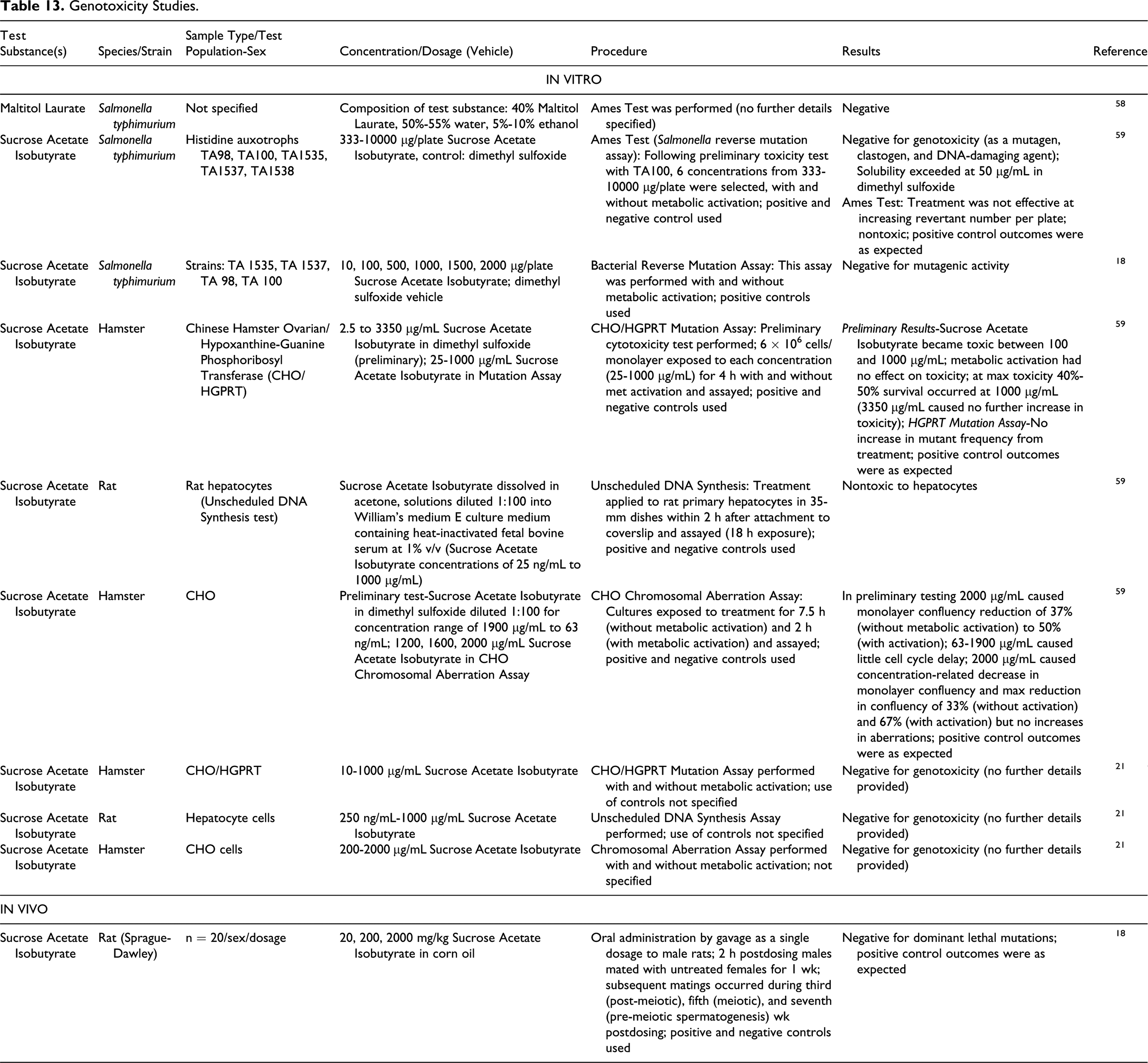
Below is a synopsis of the genotoxicity studies that are presented in detail in Table 13.
Genotoxicity Studies.
In Vitro
Maltitol Laurate and Sucrose Acetate Isobutyrate were evaluated in vitro. Maltitol Laurate (40%) was negative in an Ames test performed using Salmonella typhimurium. 58 An Ames test conducted in S typhimurium cells showed that Sucrose Acetate Isobutyrate was negative for genotoxicity as a mutagen, clastogen, and DNA-damaging agent at concentrations up to 10000 µg/plate (nontoxic; no increase in number of revertants). 59 A mutation assay in Chinese Hamster Ovarian/Hypoxanthine-Guanine Phosphoribosyl Transferase (CHO/HGPRT) cells showed no increase in mutation frequency up to 1000 µg/mL Sucrose Acetate Isobutyrate. A chromosomal aberration assay in CHO cells showed no increase in aberrations up to 2000 µg/mL Sucrose Acetate Isobutyrate. An unscheduled DNA synthesis assay in rat hepatocytes was nontoxic for test substance Sucrose Acetate Isobutyrate at concentrations up to 10000 µg/mL.
In Vivo
A study in which male rats were administered a single dosage (2000 mg/kg) of Sucrose Acetate Isobutyrate by gavage and subsequently mated with untreated females several times during the 7 weeks postdosing yielded negative results for dominant lethal mutations. 18
Carcinogenicity Studies
Animal
Sucrose Acetate Isobutyrate
In a 2-year chronic study (Table 11) that also evaluated carcinogenicity, F344 rats (n = 50/sex/dosage rate) were fed a diet containing Sucrose Acetate Isobutyrate. 55 The nominal dosage rates were 0 (control group 1), 0 (control group 2), 0.5, 1.0, and 2.0 g/kg/d. The highest tested concentration of Sucrose Acetate Isobutyrate in the diet (less than 5%) was not expected to cause nutritional deficiencies in this long-term study. Sucrose Acetate Isobutyrate was not carcinogenic; no treatment-related tumors were found, only tumors typical of those that occur spontaneously in the F344 rat were noted.
A 2-year chronic dietary study (Table 11), also evaluating carcinogenicity, was conducted in B6C3F1 mice (n = 50/sex/dosage rate). 55 A 4-week range finding study (Table 11) was conducted at 0, 0.625, 1.25, 2.5, and 5.0 g/kg/d Sucrose Acetate Isobutyrate (n = 10/sex/dosage rate). Results indicated that Sucrose Acetate Isobutyrate was well-tolerated. Dosage rates selected for the 2-year carcinogenicity study were 0, 0, 1.25, 2.5, and 5.0 g/kg/d Sucrose Acetate Isobutyrate (highest dietary concentration of Sucrose Acetate Isobutyrate at 4.4%). Sucrose Acetate Isobutyrate was not carcinogenic; tumors found were typical of those that occur spontaneously in the B6C3F1 mouse and were not treatment-related.
Carcinogenicity in Sprague-Dawley rats was evaluated (n = 10/sex/dose level) at 0%, 0.38%, and 9.38% Sucrose Acetate Isobutyrate in the diet for 2 years in a chronic study (Table 11). 21 No Sucrose Acetate Isobutyrate treatment-related lesions were found upon histological examination, therefore the study results were negative for carcinogenicity.
Other Relevant Studies
Cytotoxicity Studies
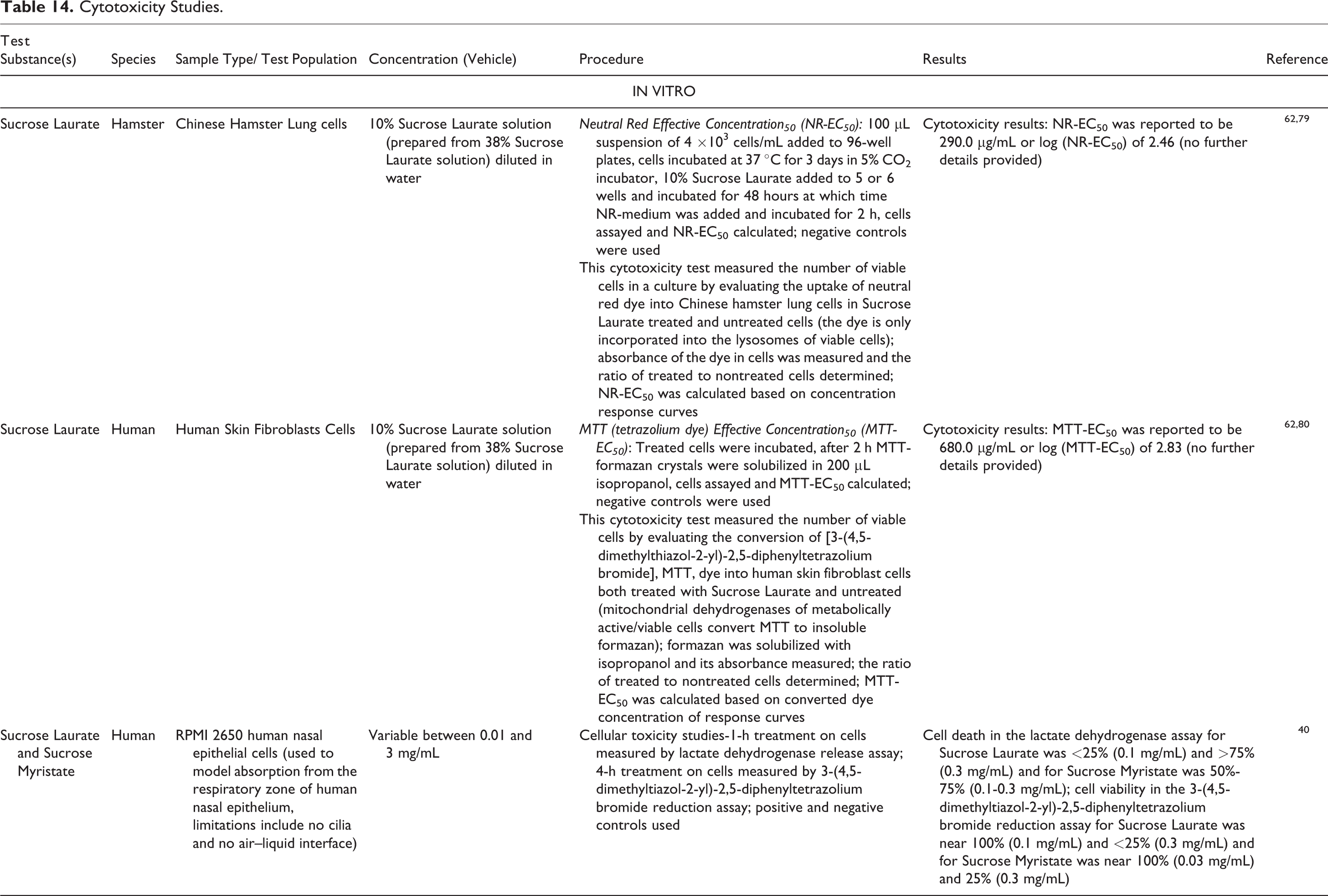
Provided below is a synopsis of cytotoxicity studies presented in detail in Table 14.
Cytotoxicity Studies.
In vitro
Tests on various cell cultures were conducted to evaluate the cytotoxicity of Sucrose Laurate and/or Sucrose Myristate. Human nasal epithelial cell death in the lactate dehydrogenase assay of Sucrose Laurate was < 25% at 0.1 mg/mL and of Sucrose Myristate was 50% to 75% at 0.1 to 0.3 mg/mL; the 3-(4,5-dimethyltiazol-2-yl)-2,5-diphenyltetrazolium bromide reduction assay test indicated that cell viability was 100% with 0.1 mg/mL Sucrose Laurate and 100% with 0.03 mg/mL Sucrose Myristate. 40
Dermal Irritation and Sensitization Studies
Irritation
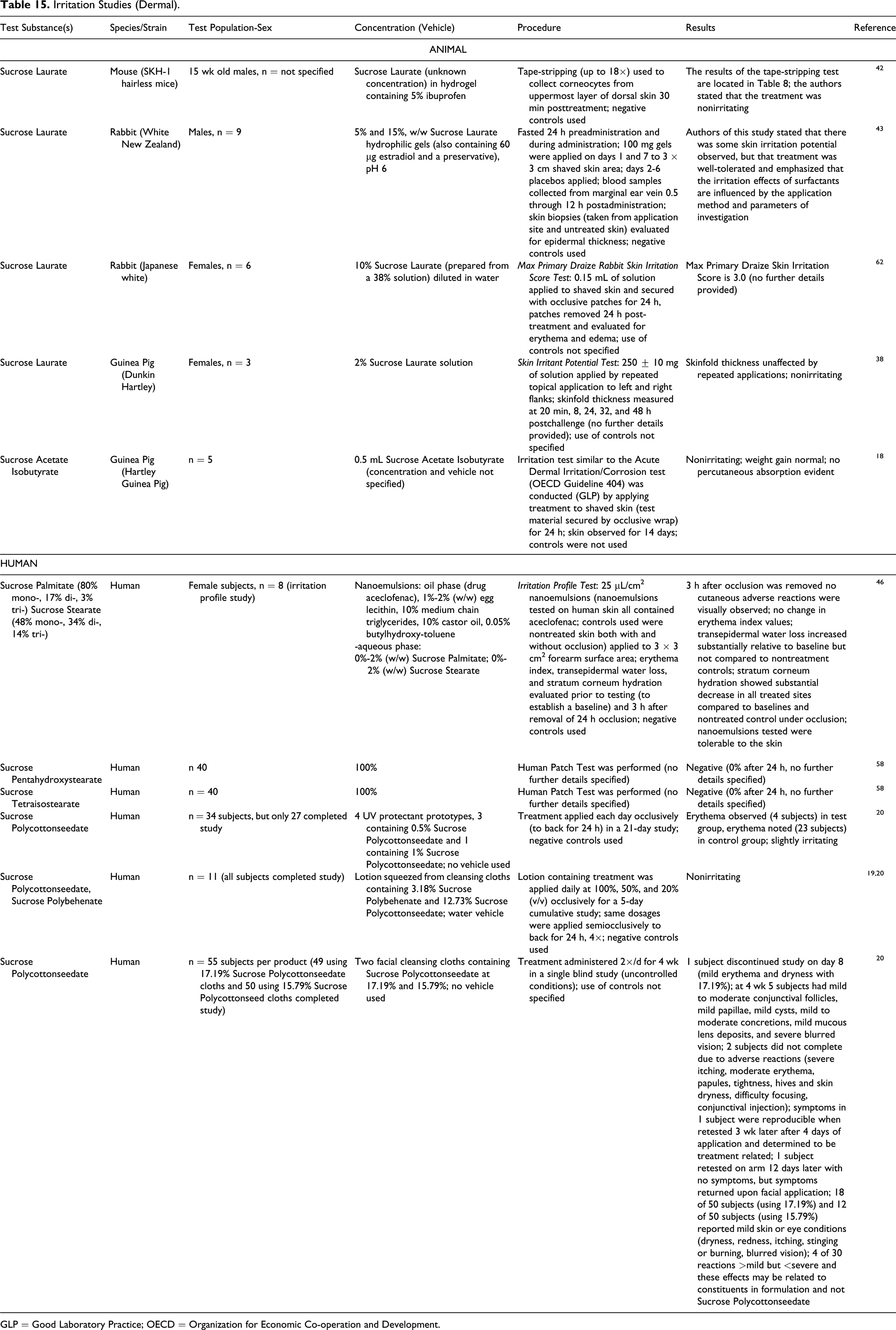
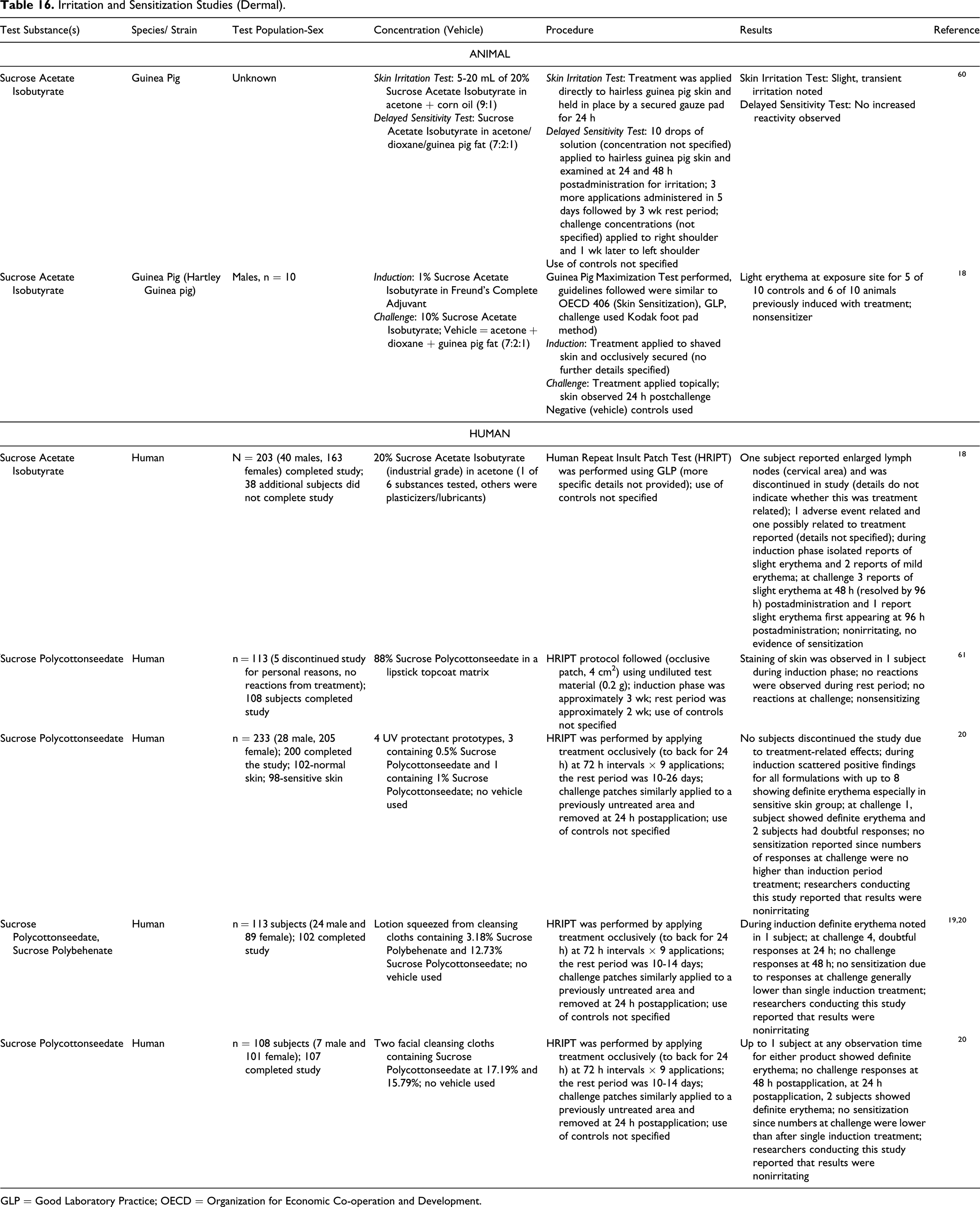
Details of dermal irritation and sensitization studies found in Tables 15 and Table 16 are summarized below.
Irritation Studies (Dermal).
GLP = Good Laboratory Practice; OECD = Organization for Economic Co-operation and Development.
Irritation and Sensitization Studies (Dermal).
GLP = Good Laboratory Practice; OECD = Organization for Economic Co-operation and Development.
Animal
Skin irritation testing of Sucrose Laurate in animals resulted in the following: a hydrogel formulation (concentration of Sucrose Laurate unknown) containing 5% ibuprofen was nonirritating to mouse skin 42 ; 5% and 15% hydrophilic gel formulations (also containing the drug estradiol) yielded increased epidermal thickness and some irritation potential when tested in rabbits 43 ; a 2% solution was nonirritating when tested in guinea pigs. 38 Sucrose Acetate Isobutyrate (0.5 mL applied directly to shaved skin with an occlusive covering for 24 hours) was nonirritating to guinea pig skin. 18 The saccharide esters evaluated were generally nonirritating, with sporadic occurrences of slight irritation.
Human
Sucrose Stearate and Sucrose Palmitate (up to 2% in a nanoemulsion containing the drug aceclofenac), evaluated for irritation potential in a 24-hour occlusive patch test in human subjects, produced a decrease in stratum corneum hydration. However, no adverse skin reactions were visible and the treatment was tolerable to the skin. 46 Human patch tests evaluating Sucrose Pentahydroxystearate (100%) and Sucrose Tetraisostearate (100%) for irritation were negative (0% after 24 hours, no further details specified). 58 Sucrose Polycottonseedate (up to 1% in formulation) was slightly irritating in a 21-day occlusive patch test in human subjects. 20 Sucrose Polycottonseedate (up to 13% solution) and Sucrose Polybehenate (up to 3% solution) were nonirritating in a 5-day occlusive patch test in human subjects.19,20 Moderate skin irritation observed in 4 of 30 humans subjects dermally exposed to cleansing cloths containing Sucrose Polycottonseedate (up to 17.19%) was thought to be caused by constituents in the formulation other than the saccharide ester. 20 Overall, the saccharide esters were characterized as nonirritating to human skin in the studies summarized in this safety assessment; for the intermittent irritation noted in a few of the tests (see Tables 15 and 16), it was reported to be slight to mild in a relatively small percentage of the total population of subjects evaluated.
Sensitization
Animal
Slight, transient irritation was noted in a study testing Sucrose Acetate Isobutyrate (20% solution in 9:1, acetone: corn oil) for 24 hours on hairless guinea pig skin. 60 A delayed sensitization experiment was also conducted in guinea pigs (induction and challenge phase concentrations were not specified) and no sensitization was reported. In a Guinea Pig Maximization Test evaluating Sucrose Acetate Isobutyrate (1% solution at induction; 10% solution at challenge), no sensitization was observed. 18 Sucrose Acetate Isobutyrate was nonsensitizing in animals.
Human
Sucrose Acetate Isobutyrate (20% solution) in a human repeat insult patch test (HRIPT) was nonirritating and nonsensitizing. 18 In an HRIPT, Sucrose Polybehenate (∼3%) and Sucrose Polycottonseedate (∼13%) were nonirritating and nonsensitizing19,20 Sucrose Polycottonseedate (∼16%-17% in facial cleansing cloths) was nonirritating and nonsensitizing in an HRIPT. 20 Sucrose Polycottonseedate (88% in a lipstick topcoat matrix) was nonsensitizing in human subjects and the only reaction reported was skin staining in 1 subject. 61 The saccharide esters evaluated were nonsensitizing in human subjects.
Ocular Irritation
Animal
Sucrose Laurate
A study in Japanese white female rabbits was conducted to evaluate the effects of Sucrose Laurate on rabbit eyes. 62 A Maximum Draize Rabbit Eye Score (MDES) test was performed by instilling 0.1 mL of 10% Sucrose Laurate solution, prepared from a 38% Sucrose Laurate solution, into the conjunctival sac of the left eye (right eye served as untreated control) of each of 3 rabbits. There was no eye washing postapplication. Observations were made at 1, 3, 6, 24, 48, 72, and 96 hours post-treatment. The observed MDES score reported for Sucrose Laurate was 21 (no further irritation results provided). The threshold score of around 20 was considered by the authors to be the value below which corneal damage was not observed.
Sucrose Acetate Isobutyrate
A study evaluating the irritation of Sucrose Acetate Isobutyrate in New Zealand White rabbit eyes was conducted. 18 The guidelines followed in this study were similar to OECD 405 (Acute Eye Irritation/Corrosion), using good laboratory practice. To all of the eyes of 3 rabbits, 0.1 mL of a 50% Sucrose Acetate Isobutyrate dilution in corn oil was instilled into the conjunctival sac (3 washed and 3 unwashed eyes, following application). To another unwashed eye, 0.1 mL of corn oil was instilled into the conjunctival sac for use as a control. Observations were noted for 72 hours postapplication. The control performed as expected. Moderate erythema of conjunctivae and nictitating membranes were noted in all unwashed eyes 1 hour postapplication; slight erythema of conjunctivae and nictitating membranes observed in 2 of 3 unwashed eyes 24 hours postapplication. At 48 hours postapplication, 2 of 3 unwashed eyes were normal and at 72 hours postapplication, all 3 unwashed eyes were normal. There was slight (in 2 of 3 eyes) to moderate (in 1 of 3 eyes) erythema of conjunctivae and nictitating membranes in washed eyes 1 hour postapplication. All washed eyes were normal at 24 hours postapplication (no corneal or adnexal staining seen when eyes were examined with fluorescein dye). Sucrose Acetate Isobutyrate was slightly irritating to rabbit eyes.
Summary
The 40 saccharide esters included in this safety assessment have a variety of reported functions in cosmetics, that is, surfactants, humectants, emulsion stabilizers, emollients, and skin-conditioning agents.
The VCRP data obtained from the FDA in 2016 indicated that Sucrose Acetate Isobutyrate has the highest reported number of uses (274) with the next highest reported for Sucrose Stearate (156) and Sucrose Cocoate (139). Concentration of use industry survey data obtained by the Council in 2015 to 2016 indicated that the highest maximum reported concentrations of use are for Sucrose Polycottonseedate (87.7% in lipstick), Sucrose Acetate Isobutyrate (31% in eye shadow and foundation; 27% in lipstick), Sucrose Cocoate (20.6% in shaving soap), Sucrose Tetrastearate Triacetate (15% in mascara and 10% in lipstick), and Sucrose Benzoate (14.3% in nail polish and enamel). There were no concentrations of use reported in a Council industry survey for Sucrose Dipalmitate and Sucrose Palmitate/Stearate or Sucrose Stearate-Palmitate Ester. There are 14 saccharide esters included in this safety assessment that are not reported to be in use according to the 2016 VCRP data and the 2015 to 2016 Council concentration of use industry survey.
Saccharide esters are used as penetration enhancers in pharmaceutical applications. They are also incorporated into foods as direct and indirect food additives (ie, flavoring substances and emulsion stabilizers). Sucrose Acetate Isobutyrate is a GRAS direct food additive. Sucrose Laurate, Sucrose Palmitate, Sucrose Stearate, and Sucrose Distearate are listed as inactive ingredients in FDA-approved drug products.
Human dermal penetration studies showed at 1 hour postdermal (nonocclusive) application of elastic vesicles (containing a molar ratio of 50:50:5; Sucrose Laurate: micelle-forming surfactant; PEG-8-L: stabilizer sulfosuccinate) that Sucrose Laurate was observed up to the ninth tape strip and after 4 hours up to the 15th strip, suggesting that Sucrose Laurate permeated the stratum corneum.
The in vitro penetration enhancement studies demonstrated that Sucrose Laurate was a percutaneous absorption enhancer for the drug lidocaine at pH 6, in mice. Micropig experiments (in vitro) showed that Sucrose Laurate enhanced skin incorporation of polyphenols with accumulation of hydrophilic polyphenols occurring more in the epidermis and accumulation of lower molecular weight hydrophobic polyphenols more in the dermis. Sucrose Laurate, in in vitro rat skin tests, exhibited effective skin penetration-enhancing properties for dermal hydrophilic drug (cyclosporine A) delivery. Sucrose Laurate and Sucrose Myristate (variable between 0.1 and 3 mg/mL) showed a concentration-dependent enhancement of paracellular permeability of a fluorescein isothiocyanate-labeled dextran marker in human nasal epithelial cells. Sucrose Stearate was shown to be an emulsifier and dermal drug (eg, fluconazole) penetration enhancer in pig skin (in vitro).
Penetration enhancement studies (in vivo) testing Sucrose Laurate in mice showed increased skin hydration and penetration of ibuprofen and facilitated the absorption of lipophilic hydrocarbon components of the hydrogel (vehicle containing Sucrose Laurate) in the stratum corneum. Sucrose Laurate was a good intranasal absorption enhancer for the drug sumatriptan in rats. Experiments conducted in rabbits in vivo showed that Sucrose Laurate increased dermally administered drug (estradiol) bioavailability by 15%; Sucrose Laurate was a percutaneous absorption enhancer in single dose drug (estradiol) applications, but less effective after multiple applications. Skin biopsies from the application sites of rabbits treated with 5% and 15% Sucrose Laurate exhibited substantially greater thickness. Sucrose Cocoate, when exposed via nasal administration using a pipette or by ocular installation, was found to increase the absorption of drugs (insulin and calcitonin) 9-fold (nasal) and 4-fold (ocular) in rats. In human subjects, Sucrose Palmitate (2%) and Sucrose Stearate (0.5%) were found to be absorption enhancers of the dermally applied drug aceclofenac, which was subsequently detected at all depths of the stratum corneum.
Toxicokinetic studies in vitro showed that a mixture of Sucrose Palmitate and Sucrose Stearate (1 µmol/mL, [ 14 C] labels on either sucrose or ester portion) was not transported from mucosal to serosal solution in everted intestinal sacs of rats; hydrolysis by mucosal homogenates from the rats was 10% to 30%, compared with little hydrolysis in whole blood. Results from another rat test indicated that up to 250 µg/mL [ 14 C]Sucrose Acetate Isobutyrate (label on sucrose) was 75% hydrolyzed by intestinal mucosa in 6 hours; less hydrolysis occurred in the stomach and liver. A study in which human fecal homogenates were incubated with 1 mg/mL or 0.1 mg/mL [ 14 C]Sucrose Acetate Isobutyrate (label on sucrose) resulted in 40% and 60% hydrolysis, respectively, in 16 hours.
Toxicokinetic tests conducted in rats (oral exposure) revealed the following: Glucose Pentaacetate (20% aqueous solution, no radioactive label used) was rapidly absorbed (>90%) in 4 hours; a mixture of Sucrose Palmitate and Sucrose Stearate (up to 250 mg/kg) was excreted in feces (30%-67% of dosed radioactivity), exhaled (11% to 49% of dosed radioactivity), and not detected in urine or blood at 120 hours postdosing; a mixture of sucrose esters (250 mg/kg), including Sucrose Hexastearate, were hydrolyzed prior to intestinal absorption (less esterified compounds were better absorbed) and largely excreted in feces (> 95% of dosed radioactivity) at 120 hours postdosing; 200 mg/kg of Sucrose Octaisobutyrate (a component of Sucrose Acetate Isobutyrate) was excreted in feces (78%-93% of the dose of radioactivity), excreted as a volatile product (3%-15% of the dose of radioactivity), and eliminated in urine (1%-2% of the dose of radioactivity). In dogs and monkeys orally administered 200 mg/kg of Sucrose Octaisobutyrate, no radioactivity was detected in whole blood or plasma and excretion in feces was 77% to 94% of dose of radioactivity and 62% to 85% of the dose of radioactivity, respectively. In dogs, Sucrose Octaisobutyrate was slowly absorbed with less extensive hydrolysis in the gut, compared to rats; in monkeys it was not absorbed or hydrolyzed in the gut.
In single-dose (0.1 g or 1.0 g Sucrose Acetate Isobutyrate) and multidose (1 g/d Sucrose Acetate Isobutyrate for 7 days) oral-exposure toxicokinetic studies in human subjects, results indicated that <0.4% were excreted in urine as the parent compound or metabolite with a disaccharide moiety. In a fecal excretion study, 0.1 g/d Sucrose Acetate Isobutyrate was administered to 1 subject for 7 day and no unchanged Sucrose Acetate Isobutyrate or metabolites were detected in fecal samples. The absorption of partially esterified sucrose molecules from the intestinal tract was insignificant. In a different study in human subjects who were administered a single, oral dosage of 1.0 to 1.2 mg/kg Sucrose Acetate Isobutyrate exhaled 41% to 66% of the oral dose of radioactivity in the breath within 30 days postadministration; 15% to 21% of the dose of radioactivity was eliminated in urine and 10% was excreted in feces.
The acute toxicity studies in rats evaluated dermal and oral exposure to Sucrose Acetate Isobutyrate for which LD50 > 20 g/kg and LD50 > 5 g/kg were reported, respectively. In 1 study, rats and mice were orally dosed with 25.6 g/kg of Sucrose Acetate Isobutyrate. No mortality was observed for the mice and 1 of the 7 rats died. Sucrose Acetate Isobutyrate (5 g/kg) and Sucrose Octaisobutyrate (5 g/kg) were orally administered to monkeys in a study which found that liver metabolism parameters were unaffected by the treatment. However, in dogs that were orally administered 2 g/kg Sucrose Acetate Isobutyrate, measured plasma concentrations of BSP were found to be elevated. In another test in monkeys, the LD50 was reported to be > 20 g/kg for oral administration of Sucrose Acetate Isobutyrate.
In short-term studies, Sucrose Acetate Isobutyrate was well-tolerated in orally exposed animals. In rats, an NOAEC was reported for doses up to 10% daily in the diet for 6 weeks; monkeys dosed up to 10 g/kg/d in the diet (15 days) showed no change in body weight, food consumption, or clinical parameters; mice dosed up to 5 g/kg/d in the diet (4 weeks) were unaffected by the treatment; in a 2-week study in dogs dosed up to 0.5% daily in the diet, BSP retention was reported at 0.3% and 0.5%. In rats dosed with Sucrose Polysoyate in the diet for 28 days, an NOEC of 15% was reported; softer feces, lower growth rates (dose-related), and a dose-dependent heart weight decrease were observed. Two studies in dogs reported an NOAEC and an NOEC of 15% daily in the diet for 28 days for Sucrose Polysoyate; results showed a higher food consumption in treated animals compared to controls, yet hematology, urine, and organs were unaffected by treatment.
In a 12-week subchronic study in rats orally administered Sucrose Acetate Isobutyrate, an NOAEC of 10% daily in the diet was reported; a decrease in mean heart weights in all treated males was observed. In 13-week studies in rats dosed with up to 9% Sucrose Acetate Isobutyrate daily in the diet, slight diarrhea was reported, but no toxic effects were observed. Dogs orally administered up to 5% of Sucrose Acetate Isobutyrate daily in the diet for 91 days showed a moderate elevation in SAP liver enzyme, heavier liver weights compared to controls, and a functional effect on the liver (reversible when Sucrose Acetate Isobutyrate was removed from the diet). In rats orally administered Sucrose Polysoyate for approximately 90 days, an NOEC of 15% daily in the diet was reported; softer feces and lower growth rates were noted, an increase in food consumption was seen with increasing doses, however no toxicity was observed.
Chronic toxicity studies testing Sucrose Acetate Isobutyrate orally administered to animals reported an NOAEL of 2 g/kg/d for 1 year in rats and an NOAEL of 2.4 g/kg/d for 1 year in monkeys. In the rat study, body weight gain decreases were observed in males and females (2 g/kg/d); 1 female death (0.5 g/kg/d) was noted, 1 female was killed in a moribund condition (2 g/kg/d), and 1 control and 2 treated rats died during blood collection. In 2-year studies conducted in animals, the NOAELs reported for rats and mice were 2 g/kg/d (highest dosage rate tested) and 2.5 g/kg/d, respectively. In mice, the NOAEL did not include the highest dosage rate tested (5 g/kg/d) because at that dosage rate a treatment-related decrease in mean absolute and relative kidney weights was observed at necropsy in males compared to controls. Another 2-year study conducted in rats (dosing up to 9.38% in diet) showed a dose-related increase in absolute and relative kidney weights, however the organ weight findings were deemed inconclusive because of discrepancies in male body weights compared to controls and low survival numbers. Within 10 weeks of the study, 4 males dosed with 9.38% died (massive hemorrhages in multiple organs were reported), but the deaths were not attributed to treatment (no further details specified).
Developmental and reproductive toxicity studies reported an NOAEL of 2 g/kg/d Sucrose Acetate Isobutyrate in rats (dosed daily in the diet for 10 weeks, males, and for 2 weeks, females, prior to mating) and an NOAEL of 1.2 g/kg/d Sucrose Acetate Isobutyrate in rabbits (dosed on days 7-19 of gestation by gavage). Sucrose Acetate Isobutyrate was not found to impair reproduction or produce toxic teratogenic/developmental effects in rats and rabbits. Rats fed 9.38% Sucrose Acetate Isobutyrate for 5 weeks (rats bred 3× in weeks 9-36) resulted in decreased pregnancies and decreased number of pups surviving to weaning, but this may have been attributed to compromised nutritional value of the diet at high Sucrose Acetate Isobutyrate concentrations.
Maltitol Laurate (40%) was negative in an Ames test performed using S typhimurium. An Ames test conducted in S typhimurium cells showed that Sucrose Acetate Isobutyrate was negative for genotoxicity as a mutagen, clastogen, and DNA-damaging agent at concentrations up to 10000 µg/plate (nontoxic; no increase in number of revertants). A mutation assay in CHO/HGPRT cells showed no increase in mutation frequency up to 1000 µg/mL Sucrose Acetate Isobutyrate. A chromosomal aberration assay in CHO cells showed no increase in aberrations up to 2000 µg/mL Sucrose Acetate Isobutyrate. An unscheduled DNA synthesis assay in rat hepatocytes was nontoxic for test substance Sucrose Acetate Isobutyrate at concentrations up to 10000 µg/mL. An in vivo animal study in rats tested for dominant lethal mutations showed negative results up to 2000 mg/kg Sucrose Acetate Isobutyrate (male rats were dosed once by gavage 2 hours prior to mating with untreated females; males were mated several times with untreated females during the 7 weeks postdosing).
Carcinogenicity bioassays, of 2-year duration, were conducted in rats (up to 2 g/kg/d Sucrose Acetate Isobutyrate) and mice (up to 5 g/kg/d Sucrose Acetate Isobutyrate); study results were negative for carcinogenicity. Another test in rats dosed up to 9.38% Sucrose Acetate Isobutyrate in the diet for 2 years indicated no treatment-related lesions.
Cytotoxicity tests evaluating Sucrose Laurate and Sucrose Myristate on human nasal epithelial cells showed cell death in a lactate dehydrogenase assay was < 25% for Sucrose Laurate (0.1 mg/mL) and was 50% to 75% for Sucrose Myristate (0.1 to 0.3 mg/mL); the 3 (4,5-dimethyltiazol-2-yl)-2,5-diphenyltetrazolium bromide reduction assay test indicated that cell viability was 100% for Sucrose Laurate (0.1 mg/mL) and 100% for Sucrose Myristate (0.03 mg/mL).
Dermal exposure studies in test animals indicated that Sucrose Laurate (unknown concentration) in a hydrogel was nonirritating, and 5% to 15% had some irritation potential but was well-tolerated. A 20% Sucrose Acetate Isobutyrate solution caused slight, transient irritation. In humans, Sucrose Palmitate and Sucrose Stearate (up to 2% in oil/water nanoemulsions) substantially decreased hydration in the stratum corneum during an occlusive irritation profile test, however no adverse skin reactions were visually observed, and the treatment was tolerable to the skin. In human patch tests evaluating irritation, Sucrose Pentahydroxystearate (100%) and Sucrose Tetraisostearate (100%) were negative (0% after 24 hours, no further details); Sucrose Polycottonseedate at 0.5% to 1% was slightly irritating. Moderate skin irritation observed in 4 of 30 humans subjects dermally exposed to cleansing cloths containing Sucrose Polycottonseedate (up to ∼17%) was thought to be caused by constituents in the formulation and not the saccharide ester.
A 1% solution (at induction) of Sucrose Acetate Isobutyrate was nonsensitizing in a Guinea Pig Maximization Test. In humans, Sucrose Acetate Isobutyrate (20% in an HRIPT) was found to be nonirritating and nonsensitizing; in an HRIPT, Sucrose Polybehenate (∼3%) and Sucrose Polycottonseedate (∼13%) were nonirritating and nonsensitizing; an HRIPT evaluating Sucrose Polycottonseedate (up to ∼17%) was deemed to be nonirritating and nonsensitizing; Sucrose Polycottonseedate (88% in a lipstick topcoat matrix) in an HRIPT was found to be nonsensitizing and the only reaction reported was skin staining in 1 subject.
A 10% Sucrose Laurate solution and a 50% Sucrose Acetate Isobutyrate solution were slightly irritating to rabbit eyes.
Discussion
The Panel considered relevant systemic toxicity, reproductive and developmental toxicity, genotoxicity, carcinogenicity, and irritation and sensitization data to assess the safety of the saccharide esters. They noted an absence of systemic toxicity at high dosages of Sucrose Acetate Isobutyrate in acute dermal (LD50 > 20 g/kg) and oral (LD50 > 5 g/kg) exposure studies in rats, in a 2-week oral study in humans (0.02 g/kg/d Sucrose Acetate Isobutyrate), and in chronic, oral administration animal studies (2-year duration, NOAELs of 2-2.5 g/kg/d). Sucrose Acetate Isobutyrate was nontoxic in developmental and reproductive tests, showed an absence of genotoxic potential in an Ames test and genetic mutation experiments, and was noncarcinogenic in chronic studies. Sucrose Laurate (10%) and Sucrose Acetate Isobutyrate (50%) were slightly irritating to rabbit eyes; however the doses applied were considerably higher than the concentrations at which those ingredients are reported to be used in cosmetics. The saccharide esters are metabolized to products that are common, physiologic intermediates and nutrients, thus supporting a safe toxicity profile. The data presented in this safety assessment affirm the lack of toxicity of saccharide esters for use in cosmetics.
The Panel noted gaps in the data available to address some end points for some of the saccharide esters in this safety assessment. However, structural similarity and similarities in reported functions and concentrations of use in cosmetics of the ingredient groups enable the read-across of the data available for some of the ingredients to the ingredients with data gaps to support the safety of the entire group. For instance, the Panel applied read-across to the single-chain length subgroup of these ingredients to assess the potential for dermal irritation and sensitization of these ingredients based on the data available for Sucrose Polybehenate to address these end points. For the mixed-chain length subgroup, read-across was applied using multiple-end point safety data for Sucrose Acetate Isobutyrate and dermal irritation and sensitization safety data for Sucrose Polycottonseedate. The Panel noted that Sucrose Acetate Isobutyrate has a lower molecular weight, and therefore, a greater potential for oral and dermal absorption and thus would have a greater potential to exert biological effects than most of the ingredients for which it was used as a read-across analog.
As part of the safety determination, the Panel referred to the GRAS status of Sucrose Acetate Isobutyrate, for use as a direct food additive, and the use of several saccharide esters as direct and indirect food additives.
In animal and human studies evaluating several saccharide esters, dermal irritation was reported to be none, in most cases, and slight to mild in sporadic occurrences. In 1 animal study, up to 15% Sucrose Laurate yielded some evidence of irritation potential, but was noted to be generally well-tolerated. In humans, Sucrose Polycottonseedate was slightly irritating (up to 1%) in 1 study, but in 2 other studies (∼13% and ∼16%-17%) it was not irritating; tested at 88% (lipstick topcoat, n = 108 subjects) in an HRIPT, Sucrose Polycottonseedate was nonsensitizing, and the only reaction reported was one instance of skin staining during induction. Moderate irritation responses were reported in 1 human study (up to 17% Sucrose Polycottonseedate). However, the researchers conducting the study suspected the irritation was related to constituent ingredients in the formulation and not to Sucrose Polycottonseedate. Therefore, the Panel considered the potential for saccharide esters to cause dermal irritation to be low and not of concern. The sensitization data reported in this safety assessment indicated that saccharide esters are nonsensitizing.
The Panel recognized that saccharide esters can enhance the penetration of other ingredients through the skin. The Panel cautioned that care should be taken when formulating cosmetic products that may contain these ingredients in combination with any ingredients for which safety was based on data supporting a lack of dermal absorption, or for which dermal absorption was a concern.
The Panel expressed concern about pesticide residues and heavy metals that may be present in botanical ingredients. They emphasized that the cosmetics industry should continue to use current good manufacturing practices to limit impurities. Aflatoxins have been detected in Cottonseed Oil. The Panel believes that aflatoxins will not be present at levels of toxicological concern in Cottonseed Oil or Cottonseed Oil derivatives (eg, Cottonseed Acid), used in the esterification reaction with sucrose to produce Sucrose Polycottonseedate. The Panel recognized the US Department of Agriculture designation of ≤ 15 ppb as corresponding to ″negative″ aflatoxin content.
The Panel discussed the issue of incidental inhalation exposure from perfumes, hair sprays, deodorant sprays, and face powders. There were no inhalation toxicity data available. Sucrose Laurate is reportedly used at concentrations up to 1.2% in cosmetic products that may be aerosolized and Sucrose Tristearate is used up to 2% in face powder that may become airborne. The Panel noted that droplets/particles produced in cosmetic aerosols and loose-powder cosmetic products would not be respirable to any appreciable amount. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Panel concluded that the following 40 ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment:

* Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contribution
Scott, L. contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., Snyder, P., and Gill, L. contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. Heldreth, B. contributed to design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.