Abstract
Downregulation of the apoptotic protein DNA fragmentation factor 40 (DFF40) is correlated with poor overall survival in some malignancies, including melanoma. In this study, DFF40 gene expression driven by survivin promoter, a tumor-specific promoter, was used to selectively induce cytotoxicity in melanoma cells. The activity and strength of survivin promoter were examined in B16F10 murine melanoma, and L929 murine normal fibroblast cell lines using enhanced green fluorescent protein reporter assay and reverse transcription polymerase chain reaction. The effect of expression of DFF40 under the control of cytomegalovirus (CMV) or survivin promoter on viability of cancerous and normal cells was determined by MTT [3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide] assay. Apoptosis induction by expression of DFF40 was evaluated using Annexin-V/propidium iodide staining. Our findings showed high activity of survivin promoter comparable to the control promoter (ie, CMV) in melanoma cells, while survivin activity in normal cells was negligible. Survivin promoter-derived DFF40 gene expression led to selective inhibition of cell viability and induction of apoptosis in cancerous cells. Low and sublethal concentrations of a chemotherapeutic drug, dacarbazine, significantly enhanced the growth inhibitory effect of DFF40 gene therapy. Combination of survivin-driven gene therapy and chemotherapy could be considered as a potential therapeutic treatment for melanoma and possibly other malignancies with similar features.
Introduction
Cancer is a complex disease considered as one of the major cause of mortality over the world especially in developed countries. Progress in genomic science during last 2 decades has increased cancer researchers’ interest in the development of therapeutic approaches based on genetic manipulation or gene therapy. Specific and selective targeting of cancer cells are the most critical points for a safe and effective gene therapy approach. Tissue- or tumor-specific promoters have been applied for targeted gene therapy. Yet some of these promoters can be used only in some specific cancer types and this fact limits their clinical applications. 1
Survivin is a member of the apoptosis inhibitors family (AIP) whose expression and activity increase in 60% to 70% of cancers but not in adult normal tissues. Recent studies suggest that overexpression of survivin is associated with resistance to chemotherapy, hormone therapy, and radiotherapy in many types of cancers.2-4 Furthermore, upregulation of survivin expression contributes to metastasis of many kinds of cancers.5,6 Accordingly, survivin as a potential biomarker or therapeutic target has been the focus of recent research in cancer treatment. 7 For example, some studies have used survivin promoter, a tumor-specific promoter, to drive specifically the expression of target gene in cancerous cells. Zhao et al reported successful applications of radionuclide imaging and therapy after specifically driving expression of sodium iodide symporter (ie, a glycoprotein involved in iodide uptake by thyroid follicular cells) under the control of survivin promoter in prostate, liver, lung, and skin cancer cells xenografted models. 8
One of the most promising gene therapy approaches is application of tumor-specific promoters for expression of cytotoxic or apoptotic genes in cancer cells.9,10 DNA fragmentation factor 40 (DFF40) is an apoptotic protein which is activated by caspase-3 or caspase-7, also called caspases activated DNAse. 11 DFF40 enzyme is responsible for fragmentation of DNA, chromatin condensation, and cell apoptosis. Downregulation of DFF40 is correlated with worse overall survival in some kind of malignancies; therefore, DFF40 could be used as a potential anticancer agent.12,13 Mathew et al reported effective application of a fusion protein containing DFF40, as the toxic moiety, and granulocyte-macrophage colony-stimulating factor (GM-CSF0, as the targeting moiety, to eradicate acute myeloid leukemia cells. GM-CSF-DFF40 successfully induced release of cytochrome c, activation of caspases, DNA fragmentation, and formation of apoptotic bodies. 14 These findings indicate that overexpression of DFF40 through gene therapy could promote cancer apoptosis and inhibit tumor cell proliferation. However, there are few reports on this research area.
Here, for the first time we report construction of a plasmid containing DFF40 gene under the control of survivin promoter for gene therapy of cancer. Furthermore, we examined the effect of this construct on apoptosis and viability of mouse melanoma and fibroblast cell lines. We also evaluated the effect of combining the gene and chemotherapeutic agents on survival of melanoma cells.
Materials and Methods
Cell Culture
The murine melanoma cell line (B16F10 cells) and the murine fibroblast cell line (L929 cells) were obtained from the Pasteur Institute of Iran, Tehran. The cells were maintained in a humidified atmosphere containing 5% CO2 at 37 °C. They were fed with high glucose Dulbecco Modified Eagle medium supplemented with 10% (v/v) fetal bovine serum and antibiotic (100 IU/mL penicillin and 100 μg/mL streptomycin).
Construction of Vectors Expressing Transgenes Under the Control of Survivin or Cytomegalovirus Promoter
A pcDNA3.1 (+) plasmid backbone was used to construct pcDNA3.1-Sur-DFF40 (encoding DFF40 gene under the control of survivin promoter), pcDNA3.1-CMV-DFF40 (encoding DFF40 gene under the control of cytomegalovirus [CMV] promoter), pcDNA3.1-Sur-EGFP (encoding enhanced green fluorescent protein [EGFP] gene under the control of survivin promoter), and pcDNA3.1-CMV-EGFP (encoding EGFP gene under the control of CMV promoter) expression vectors. The Sur-DFF40 gene containing a human codon optimized DFF40 gene and a 267 base pair (bp) human survivin promoter were chemically synthesized (Biomatik). This fragment contains BglII (5′) and XhoI (3′) restriction sites (Figure 1), a BamHI restriction site between survivin promoter and DFF40 gene, a Kozack sequence, and a stop codon. The pcDNA3.1-Sur-DFF40 was constructed by ligation of Sur-DFF40 fragment into BglII and XhoI sites of pcDNA3.1 vector (Invitrogen). The pcDNA3.1-CMV-DFF40 was generated by cloning of DFF40 fragment into BamHI and XhoI sites of pcDNA3.1 vector. The EGFP fragment encoding a reporter gene that exhibits bright green fluorescence was cut from pEGFP-N1 vector (Pasteur) and subcloned into pcDNA3.1-Sur-DFF40 pcDNA3.1 and pcDNA3.1-CMV-DFF40 vectors at BamHI and XbaI sites to generate pcDNA3.1-CMV-EGFP and pcDNA3.1-CMV-EGFP, respectively. These 4 constructed plasmids were propagated in Escherichia coli DH5α and purified by large scale plasmid preparation (Endofree MaxiPrep) which yields clean transfectable DNA.
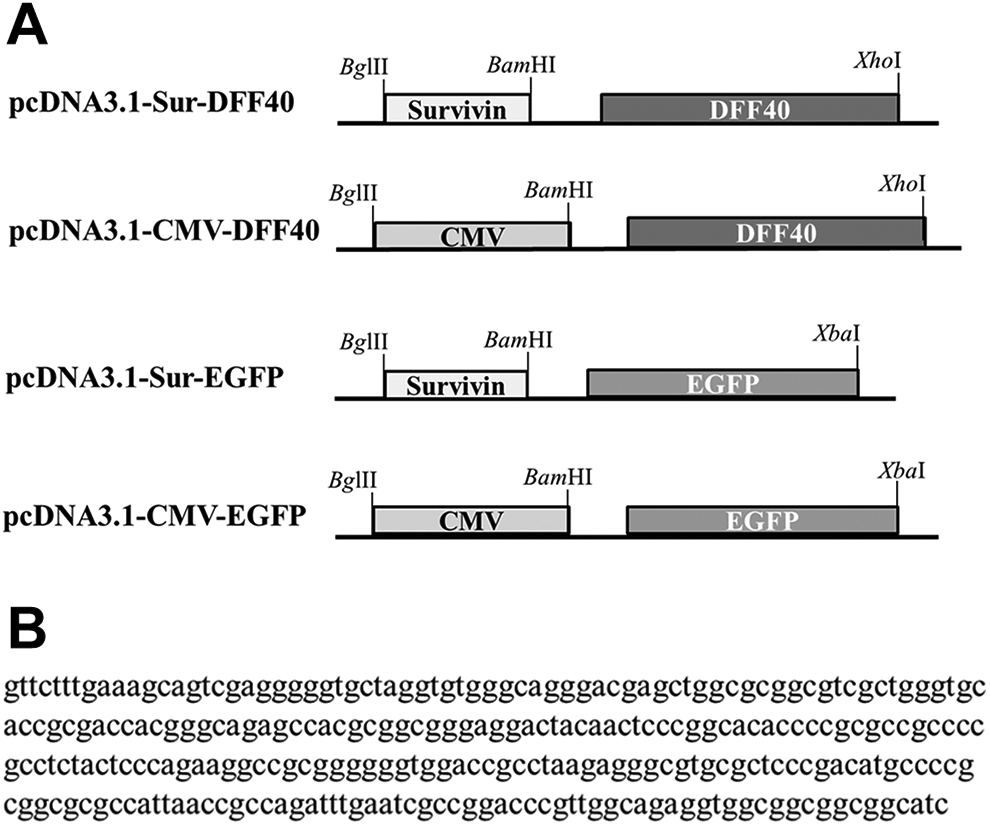
A, Schematic illustration of recombinant vectors constructed in this work. The main plasmid pcDNA3.1-Sur-DFF40 was constructed by inserting the gene encoding DFF40 under the control of survivin promoter in BamHI and XhoI restriction sites. pcDNA3.1-CMV-DFF40 was generated by inserting CMV promoter in the main plasmid by BglII and BamHI restriction sites. The control vectors, pcDNA3.1-Suv-EGFP and pcDNA3.1-CMV-EGFP, were similar to the previous 2 constructed vectors but instead of the DFF40 gene, the EGFP gene was inserted by BamHI and XbaI restriction sites. B, The nucleic acid sequence of the 267 bp survivin promoter was used in this work. CMV indicates cytomegalovirus; DFF40, DNA fragmentation factor 40; EGFP, enhanced green fluorescent protein.
Enhanced Green Fluorescent Protein Reporter Assay
To evaluate the activity and strength of survivin promoter, B16F10 and L929 cells were plated into 6-well plates (3 × 105 cells/well) 18 hours before transfection. The cells were transfected with 2 µg/well of pcDNA3.1-Sur-EGFP or pcDNA3.1-CMV-EGFP plasmid using Lipofectamine 2000 (Invitrogen) according to the manufacturer instructions. The level of EGFP expression in transfected B16F10 and L929 cells with different constructs was evaluated to measure the activity of the promoter. The EGFP florescence was analyzed using a FACSCalibur flow cytometer (BD Biosciences), and the cells were also evaluated by florescent and optical microscope on 1 to 3 days.
Semiquantitative Reverse Transcription Polymerase Chain Reaction
B16F10 and L929 cells were transfected with pcDNA3.1-Sur-DFF40 or pcDNA3.1-CMV-DFF40 plasmid as described above. Twenty-four hours later, total RNA was isolated using BioFACT Total RNA Prep Kit. The integrity of the RNA was evaluated by denaturing agarose gel electrophoresis, and the concentration of total RNA was measured by determination of optical density at 260 nm. Complementary DNA (cDNA) was generated by reverse transcription of total RNA using the RevertAid First Strand cDNA Synthesis kit (Thermo Scientific) and then the cDNAs were used in the quantitative polymerase chain reaction (PCR) reactions using specific DFF40 and GAPDH primers (Table 1). After gel electrophoresis, the messenger RNA (mRNA) expression was evaluated by quantification of the intensity of the PCR bands using ImageJ software (version 1.52, National Institutes of Health). The quantification of DFF40 was normalized to GAPDH as the reference gene or internal control.
Primers Used in the Present Study.

Cell Viability Assay
Cells were transfected with 0.25 µg/well of pcDNA3.1-Sur-DFF40, pcDNA3.1-CMV-DFF40, or pcDNA3.1 plasmid (empty vector) using Lipofectamine 2000 according to the manufacturer instructions. The cells were incubated for 24, 48, and 72 hours, and then cells were incubated with MTT solution (5 mg/mL) at 37 °C for 3 hours. Dimethyl sulfoxide was added to dissolve MTT-formazan crystals, and the absorbance was measured at 570 nm by a plate reader. The percentage of cell viability was determined according to the following equation:
% Cell survival = (Absorbance in treated wells [cells were transfected with the recombinant vector] − Absorbance in blank wells)/(Absorbance in negative control wells [cells were transfected with the empty vector] − Absorbance in blank wells)
Additionally, synergistic effect of gene therapy and chemotherapy for treatment of melanoma was evaluated. The cell transfection was performed as described above and 24 hours after transfection the chemotherapeutic agent dacarbazine (2.2 µM) was added to each well. After 48 hours of incubation with drug, the viability of cells was evaluated by MTT assay as described above.
Cell Death Detection Using Annexin-V/Propidium Iodide Staining
After 24 hours of transfection, cells were costained with fluorescein isothiocyanate conjugated Annexin V (Annexin-V) and propidium iodide (PI) using Apoptosis Detection Kit (Annexin V-FLUOS Staining kit). To quantify apoptosis, fluorescence intensity of cells was determined by FACSCalibur flow cytometer (BD Biosciences), and the cells were also evaluated by florescent and optical microscope.
Statistical Analysis
All statistics analysis was performed using Statistical Package for the Social Sciences software version 16 (SPSS Inc). Significant differences between groups were determined using analysis of variance followed by a Tukey post Hoc test. P values less than 0.05 were considered as a criteria for significant differences. Data are presented as mean 3 separate experiments ± standard deviation.
Results
Enhanced Green Fluorescent Protein Reporter Assay
To assess the strength and specificity of survivin promoter in the 2 cell lines, we carried out EGFP reporter assay. Recombinant vectors carrying EGFP reporter gene driven by CMV or survivin promoters were generated, then transiently transfected into B16F10 and L929 cells (transfection efficiency was about 50%). The level of expression of EGFP in the transfected cells was evaluated by flow cytometry and fluorescence microscopy (Figure 2A) to estimate the relative promoter activity based on the mean fluorescence intensity of pcDNA3.1-CMV-EGFP-transfected cells. The results indicated that 2 promoters generated moderate EGFP signal intensity in tumor cells, B16F10 cell line, with CMV being slightly stronger than survivin. However, survivin promoter was very weak in normal cells, L929 cell line, displaying remarkably less green fluorescence (6-fold lower activity) compared to CMV promoter (P < 0.001). These findings suggest that the 267-bp survivin promoter might retain sufficient activity and specificity for targeted cancer gene therapy.
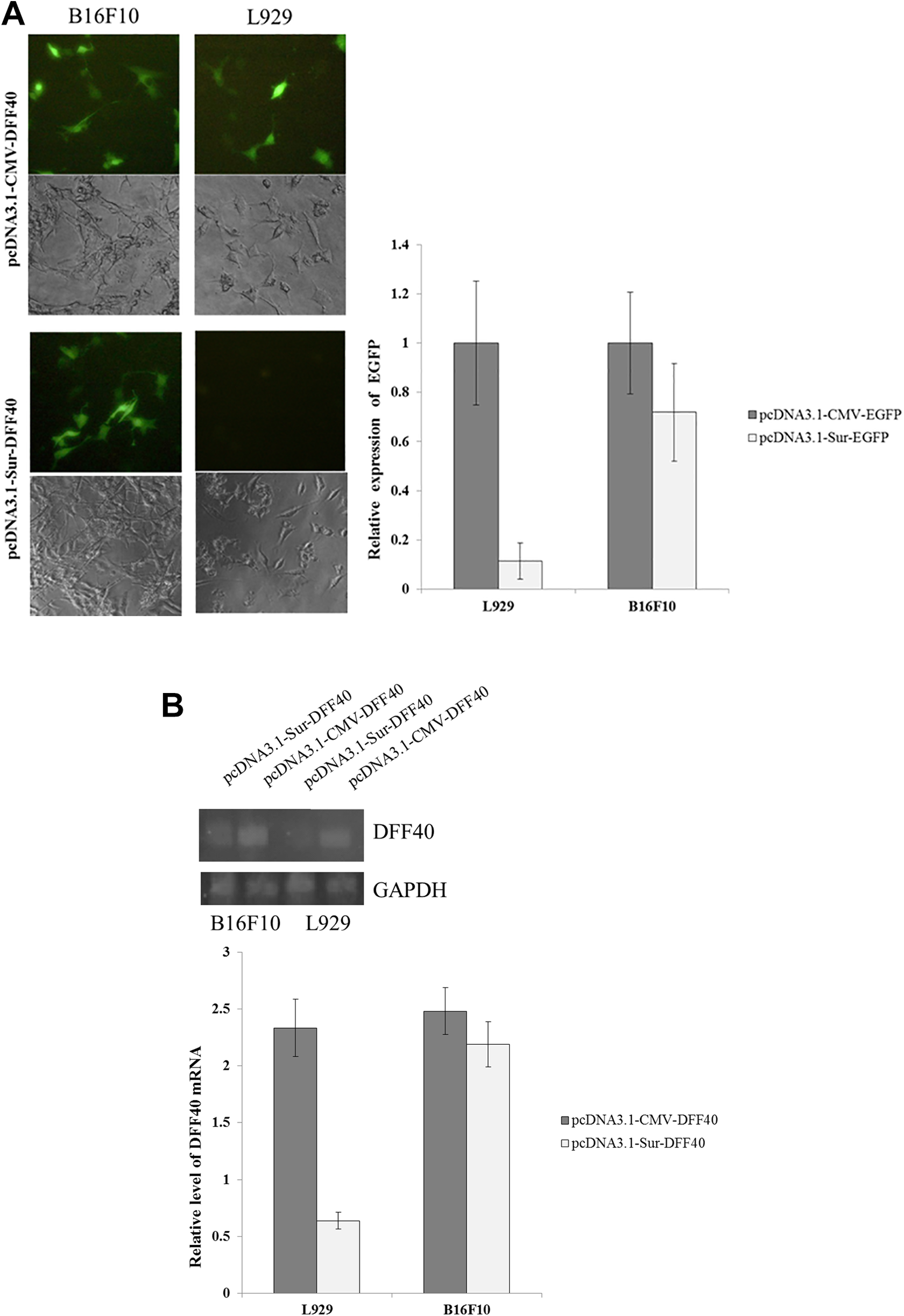
Comparison of the strength and the specificity of the survivin promoter with a viral promoter (CMV). B16F10 and L929 cells were transfected with pcDNA3.1-Suv-EGFP and pcDNA3.1-CMV-EGFP for 24 hours. A, The level of expression of EGFP in transfected cells was evaluated by fluorescence microscopy and flow cytometry after 24 hours. The relative promoter activity was calculated based on the mean fluorescence intensity of pcDNA3.1-CMV-EGFP-transfected cells. B, Semiquantitative analysis of DFF40 expression by RT-PCR. Expression profile normalized by GAPDH gene as the internal control in cancerous or normal cells (mean ± SD, n = 3). CMV indicates cytomegalovirus; DFF40, DNA fragmentation factor 40; EGFP, enhanced green fluorescent protein; RT-PCR, reverse transcription polymerase chain reaction.
Evaluation of DFF40 mRNA Expression by Semiquantitative Reverse Transcription Polymerase Chain Reaction
B16F10 and L929 cells were transfected with pcDNA3.1-Sur-DFF40 and pcDNA3.1-CMV-DFF40, following which the mRNA expression level of DFF40 was evaluated by reverse transcription polymerase chain reaction (Figure 2B). Results were presented as the relative level of DFF40 mRNA, a ratio of the intensity mean of DFF40 bands to the intensity mean of GAPDH bands. High levels of expression mRNA DFF40 driven by CMV promoter were observed in both normal and tumor cells. As expected, DFF40 mRNA expression driven by survivin promoter was considerably lower in normal cells, L929, compared with tumor cells, B16F10, consistent with the results from EGFP reporter assay studies.
Evaluation of Cell Viability
The MTT assay was performed to evaluate the in vitro cytotoxicity of recombinant vectors against normal and cancerous cells. As illustrated in Figure 3A, expression of DFF40 under the control of CMV promoter exhibited comparable cytotoxicity (P > 0.05) against normal and cancerous cells (29% and 35% cell survival inhibition for B16F10 and L929, respectively). However, expression of DFF40 under the control of survivin promoter showed more significant decrease (P < 0.05) in cell viability of cancerous cells in comparison with normal cells (30% and 13% decrease for B16F10 and L929, respectively).
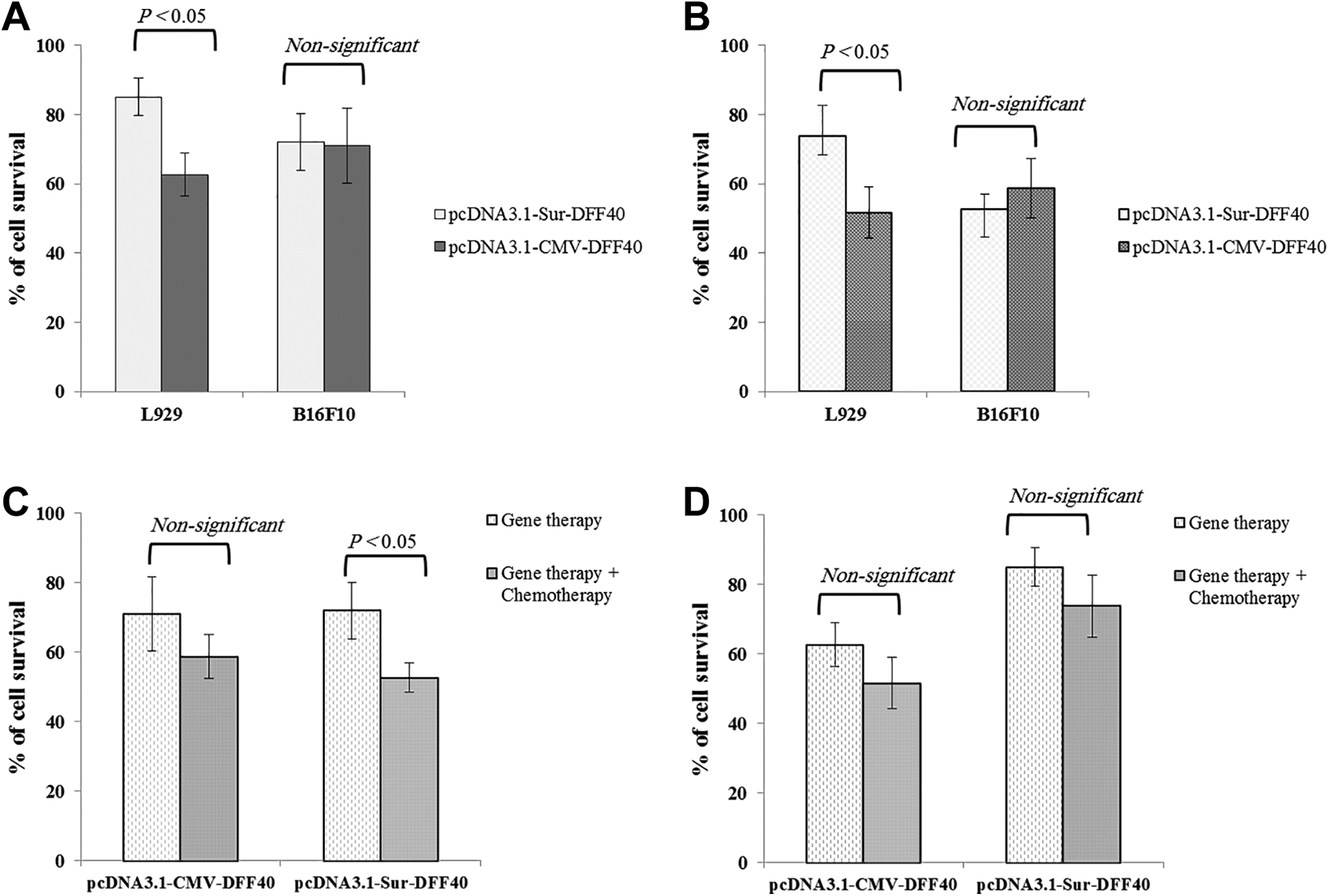
A, Effect of DFF40 expression on cell viability. After 72 hours of transfection, cell survival was evaluated by the MTT assay. B, Effect of combination DFF40 expression and dacarbazine treatment on cell viability. C, Effect of gene therapy alone or in combination with chemotherapy on viability of B16F10 cells. D, Effect of gene therapy alone or in combination with chemotherapy on viability of L929 cells. After 24 hours of transfection, cells were treated with the drug for 48 hours, and the viability of cells was determined by MTT assay. Data were presented as percentage value relative to the negative control group. P < 0.05 indicates statistically significant difference between 2 columns (mean ± SD, n = 3). DFF40 indicates DNA fragmentation factor 40; SD, standard deviation.
To evaluate the combination of gene therapy and chemotherapy, a subtoxic concentration of dacarbazine was used (more than 90% of cells were viable). As presented in Figure 3B, combination of gene therapy and chemotherapy led to a higher cytotoxicity (51% and 45% cell viability inhibition for cells transfected with pcDNA3.1-Sur-DFF40 and pcDNA3.1-CMV-DFF40, respectively) in melanoma cells when compared with gene therapy or chemotherapy alone. However, this synergistic effect was significantly lower in normal cell, L929, transfected with pcDNA3.1-Sur-DFF40 compared with pcDNA3.1-CMV-DFF40 group (26% vs 50% cell survival inhibition; Figure 3C and D).
Evaluation of Cell Death Using Annexin-V/PI Staining
After 24 hours of transfection, normal and cancerous cells were double stained with Annexin V-PI and then were analyzed using flow cytometry and also observed under fluorescent microscopy to evaluate the effect of DFF40 expression on cellular apoptosis. As presented in Figure 4A, expression of DFF40 under control of survivin or CMV promoter caused significant apoptotic effect against melanoma cells when compared to cell transfected with pcDNA3.1. Following expression of DFF40 under control of survivin or CMV promoter, increase in annexin V or annexin V/PI positive B16F10 cells (26% and 23%) was significant (P >0.05) in comparison to the empty vector (1.5%). However, significant amount of cells was annexin V positive and cell percentage at early apoptosis was more significant (P > 0.01).
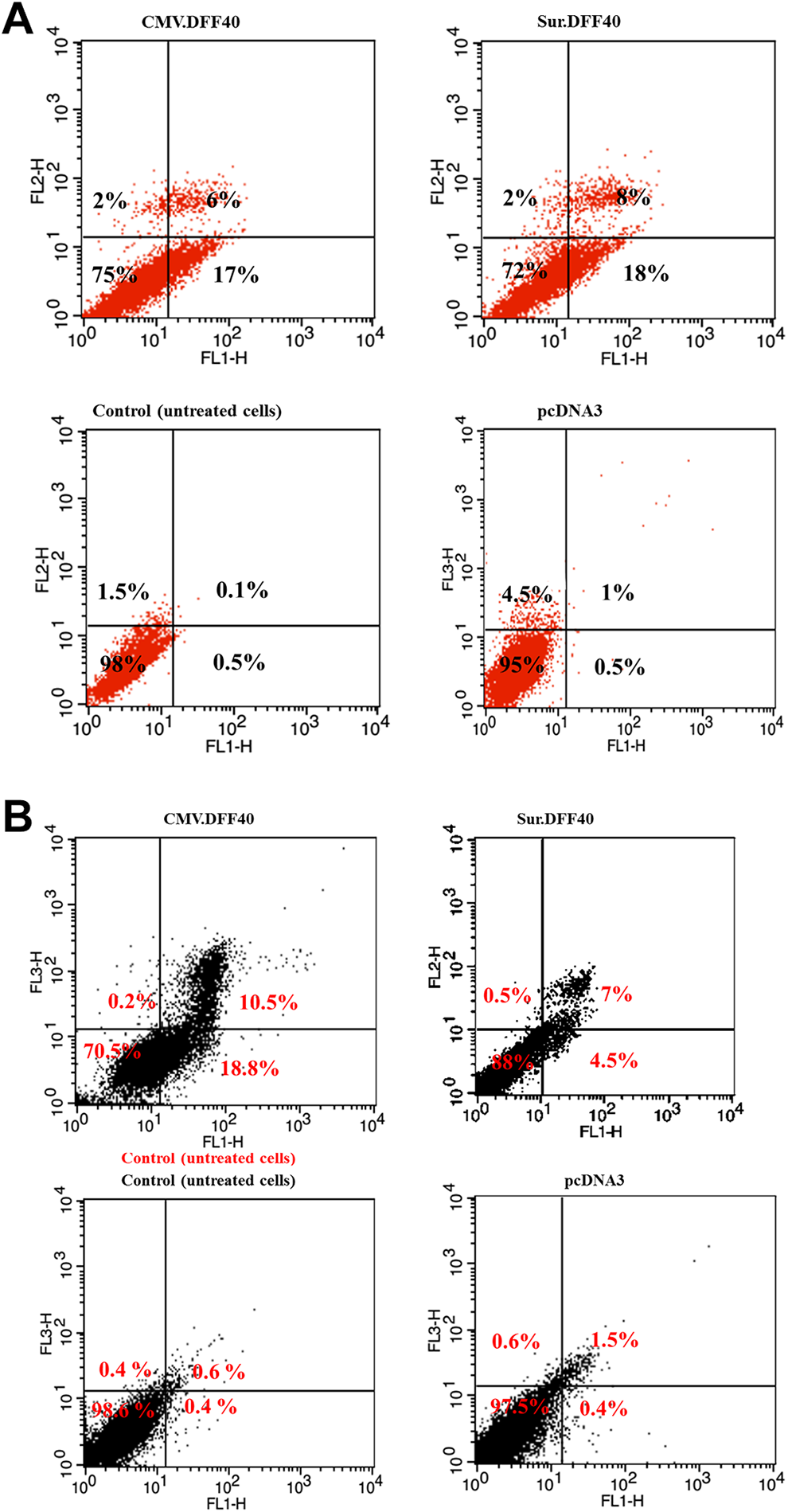
Evaluation of cell death using Annexin-V/PI staining. After 24 hours of transfection, B16F10 (A) and L929 (B) cells were double stained with Annexin V and PI and evaluated using flow cytometry. The percentages indicated in the lower left, the lower right, the upper right, and the upper left quadrants refer to viable cells, early apoptotic cells, late apoptotic cells, or secondary necrotic cells and primary necrotic, respectively. PI indicates propidium iodide.
As shown in Figure 4B, expression of DFF40 under the control of CMV promoter caused more apoptotic effect against fibroblast cells when compared to cell transfected with pcDNA3.1-Sur-DFF40 (29.3% vs 11.5%).
Discussion
Despite advancements in surgical techniques and discovery of more powerful chemotherapeutic agents, treatment of malignant melanoma remains a challenge, and development of novel and efficient therapeutic strategies (eg, gene therapy) has attracted much attention. 15 Easy and direct access to the tumors through skin, better understanding of its genetic and molecular characteristics, and identification of tumor-specific antigens have made melanoma as an excellent candidate for gene therapy approaches.16-18 One of the most essential features for successful gene therapy is restricting the transgene expression to cancerous cells without affecting the neighboring healthy and normal cells. 19 This could be achieved by the expression of target gene under the control of tumor-specific promoters which are active specifically in tumor cells. 20
In the present study, the expression of an apoptotic protein, DFF40, was driven by survivin, a tumor-specific promoter. Different lengths of survivin promoter were used in previous studies. Yang et al. generated different constructs of luciferase consisting 269, 1,430, or 6,270 nucleotides of proximal region of survivin gene. 21 They reported that activity of 269 bp promoter was similar or even higher than activity of other promoter lengths. Here, we used a 267 bp length survivin promoter with sufficient activity and specificity for cancer gene therapy. The EGFP reporter assay revealed that 267 bp survivin promoter was selectively more active in melanoma cells compared with fibroblast cells. In agreement with our results, reported that 260 bp survivin promoter had high activity to drive expression of luciferase reporter gene in cancer cells; while its activity in normal cells (ie, human fibroblast and mammary epithelial cells) was negligible. 22 Furthermore, we found that expression level of EGFP under the control of the survivin promoter in melanoma cells was comparable (ie, slightly lower) to that of CMV promoter. Consistent with our findings, a pervious study reported 269 bp promoter exhibited the same activity as a viral promoter, SV-40, in breast cancer and pancreatic cancer cell lines. 21
There are different types of transgene used for melanoma gene therapy including genes encoding cytokines or immune booster agents, suicide genes, tumor suppressor genes, or apoptosis inducers.23-25 Our results showed expression of DFF40 as an apoptotic protein under the control of survivin led to specific growth inhibition of cancerous cells. In agreement with our findings, Ben-Yehudah et al reported a fusion protein containing DFF40 as a toxic moiety and gonadotropin-releasing hormone as a targeting moiety induced apoptosis in colon adenocarcinoma cells and led to significant decrease in tumor size of xenograft mouse models. 26
We further demonstrated that combination of chemotherapy and gene therapy could significantly inhibit growth of tumor cells. Chemotherapy is one the most common approaches for treatment of advanced and metastatic melanoma. 27 Dacarbazine is the only Food and Drug Administration approved chemotherapeutic drug for melanoma but response rates are very low (only 5%-15%). 28 The exact mechanism of action of dacarbazine has not been completely understood. The drug can exert its cytotoxic effect through its action as an alkylating agent and DNA synthesis inhibition mostly in cancer cells due to their rapid rate of proliferation. 29 Our results showed that even sublethal concentrations of this drug enhanced cell death induced by gene therapy. Previous studies proposed that some stresses like hypoxia and radiation can increase activity of survivin promoters.21,30 Naoum et al 30 and Yang et al 21 proposed that some regulatory elements responsive to radiation and hypoxia might exist in the sequence of survivin promoter. However, according to the best of our knowledge this is the first report on synergistic effect of survivin-driven gene therapy and chemotherapy. This synergistic effect might be explained based on the fact that DNA damage caused by chemotherapy increases cell sensitivity to DFF40. Similarly, Bagheri et al reported that stable DFF40 expression in breast cancer cells improves the anticancer activity of doxorubicin by increasing apoptosis. 31 However, we showed that even transient expression of DFF40 led to increased chemosensitivity of cancer cells. Therefore, survivin-driven DFF40 gene therapy could be applied in combination with other chemotherapeutic agents, practically those that induce DNA damage (eg, alkylating agents, topoisomerase inhibitors, antimetabolites) to improve their efficacy and reduce side effects.
However, more investigations are still needed to elucidate the effectiveness and safety of this approach in in vivo melanoma model. Syngeneic models applying the B16F10 cell line have been considered as the most common platform for evaluation of melanoma behavior (eg, invasion and metastasis). In contrast to patient-derived tumor xenografts which use immune-deficient mice, these models can provide the possibility to evaluate the immune system effects on therapeutic response (eg, inflammatory killing of tumor cells). 32 Here, we used B16F10 cell line, a murine melanoma cells, to in vitro evaluate the activity and selectivity of survivin-driven DFF40 gene therapy which may be helpful for design of future in vivo experiments. Considering the in vitro synergistic effect of survivin-DFF40 gene therapy and chemotherapy toward melanoma cancer cells has been shown in this study, it is also suggested that this approach be evaluated in vitro and in vivo for treatment of other malignancies with similar features (eg, downregulation of DFF40).
Conclusion
In this study, we reported construction of a novel recombinant vector containing DFF40 gene under the control of 267 bp survivin promoter. The EGFP reporter assay showed high activity of survivin promoter comparable to a viral promoter in melanoma cells; while survivin activity in normal cells (ie, fibroblast) was negligible. Furthermore, this recombinant vector selectively inhibited cell viability and induced apoptosis in cancerous cells. Our findings revealed that nontoxic concentrations of a chemotherapeutic drug, dacarbazine, significantly improved the growth inhibitory effect of expression of DFF40 under the control of tumor-specific survivin promoter. Therefore, survivin-driven gene therapy could be considered as a potential therapeutic treatment for melanoma and probably other malignancies with similar features.
Footnotes
Acknowledgments
We thank Mrs Fatemeh Moazen for her excellent technical assistance.
Author Contributions
Minaiyan contributed to conception and design and contributed to acquisition; Shafiee contributed to conception and design and contributed to acquisition and analysis; Akbari contributed to conception and design and contributed to acquisition, analysis, and interpretation. All authors drafted the manuscript, critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was financially supported by the National Institute for Medical Research Development (NIMAD) of Iran (Grant No: 957782) and the Isfahan University of Medical Sciences (Grant No: 295201).
