Abstract
Phosphatidylinositol 3-kinases (PI3Ks) regulate intracellular signaling events for multiple cell surface receptors. Phosphatidylinositol 3-kinase δ, 1 of 4 class I PI3K isoforms, is primarily found in leukocytes and regulates immune cell functions. Here, we report changes in the immune and digestive systems that were associated with AMG2519493, a highly selective small-molecule PI3Kδ inhibitor. Following 1- or 3-month oral repeat dosing in the cynomolgus monkey, changes were observed in circulating B cells, lymphoid tissues (spleen, lymph nodes, gut-associated lymphoid tissue, tonsil), and the digestive tract. Decreased circulating B cells and lymphoid cellularity in B cell-rich zones in lymphoid tissues were attributed to the intended pharmacologic activity of AMG2519493. Dose- and duration-dependent digestive system toxicity was characterized by inflammation in the large intestine and secondary opportunistic infections restricted to the digestive tract. Digestive tract changes were associated with moribundity and mortality at high-dose levels, and the effect level decreased with increased duration of exposure. These observations demonstrate the role of PI3Kδ in regulation of the immune system and of host resistance to opportunistic infections of the digestive tract.
Introduction
Phosphatidylinositol 3-kinases (PI3Ks) are lipid kinases that catalyze the phosphorylation of phosphatidylinositol 4,5-bisphosphate (PI(4,5)P2) to biologically active phosphatidylinositol 3,4,5-triphosphate (PI(3,4,5)P3 or PIP3). Generally, PI3Ks are activated by receptor-coupled tyrosine kinases, Ras-related GTPases, or heterotrimeric G proteins. 1 Ultimately, PI3K signaling drives a multitude of cellular responses, including proliferation, migration, growth, and survival, and has important roles in normal physiology and disease.
The PI3K family is divided into 3 classes based on structural features and lipid substrate preferences. Class I PI3Ks are heterodimers consisting of 1 of 4 catalytic isoforms (p110α, β, γ, or δ) for which they are named (PI3Kα, PI3Kβ, PI3Kγ, and PI3Kδ, respectively), and a regulatory subunit that modulates activity and subcellular location. 1,2 Although p110α and β isoforms are ubiquitously expressed and considered targets for various oncologic indications, p110γ and δ isoforms are primarily restricted to immune cells and activation generally confers immunostimulatory functions. Although Class I PI3Ks have been extensively characterized and reviewed, 1 -3 less in known about the regulation and roles of class II and III PI3Ks.
Complex signaling pathways mediate multiple downstream effectors of class I PI3K activity. 1 Following phosphorylation of PI(4,5)P2, membrane bound PIP3 accumulates and serves as a second messenger to recruit and activate a variety of cytoplasmic effector proteins. AKT, a subfamily of the AGC serine/threonine kinases, is an important signaling node frequently activated by PI3K, and phosphorylation of AKT serves as a surrogate in vitro assay indicator of class I PI3K activation. Downstream of activated AKT lies the rich biology of mammalian target of rapamycin (mTOR)and its important role in multiple cellular functions, including proliferation, differentiation, and trafficking. 4
Phosphatidylinositol 3-kinase δ plays critical roles in the immune system making it an attractive target for pharmaceutical intervention of inflammatory, autoimmune disease, and oncology indications. Phosphatidylinositol 3-kinase δ is required for B-cell receptor–mediated activation, differentiation, and immunoglobulin secretion, and promotes various functions for T cells, natural killer (NK) cells, basophils, mast cells, and neutrophils. 5 Inhibition of PI3Kδ has been shown to be effective in multiple inflammatory disease animal models, including inflammatory arthritis, asthma, and obstructive respiratory disease. 6,7 In addition, PI3Kδ inhibitors, such as idelalisib (Zydelig), are approved for the treatment of human B-cell malignancies, such as chronic lymphocytic leukemia. 8 Treatment in humans with idelalisib may be associated with severe or fatal hepatotoxicity, diarrhea, colitis, pneumonitis, and intestinal perforation, for which United States (US) prescribing information contains a black box warning. 9
AMG2519493, a highly selective and potent small-molecule inhibitor of PI3Kδ, was in clinical development for the treatment of systemic lupus erythematosus and rheumatoid arthritis; however, the development program was recently terminated. The AMG2519493-mediated inhibitory activity of recombinant human PI3K isoforms was measured by luminescence detection of ATP in a luciferase-coupled assay. 10 In this assay, AMG2519493 exhibited selectivity of approximately 2400-fold, 1500-fold, and 400-fold over human PI3kinases α, β, and γ, respectively (unpublished data). AMG2519493 was a potent inhibitor of human B cell proliferation in in vitro and whole blood functional assays, and efficacious in mouse models of systemic lupus erythematosus and collagen-induced arthritis. In addition, AMG2519493 was inactive against a broad panel of 402 kinases (Ambit Biosciences Kinome Scan Panel).
The nonclinical safety profile of AMG2519493 was evaluated in 1- and 3-month repeat-dose toxicology studies. Here, we report AMG2519493-related immunomodulation, digestive tract inflammation, and secondary opportunistic infections in the cynomolgus monkey toxicology studies. These observations demonstrate the role for PI3Kδ in regulating the cynomolgus monkey immune system and the consequences for host defense following inhibition of this important pathway.
Materials and Methods
Test and Control Article
AMG2519493, a highly selective and potent small-molecule inhibitor of PI3Kδ, was synthesized at Amgen Inc. The chemical name for AMG2519493 is (S)-4-amino-6-((1-(8-fluoro-2-(2-(methylsulfonyl)phenyl)quinolin-3-yl)ethyl)amino)pyrimidine-5-carbonitrile. The vehicle control article was 0.5% (wt/vol) hydroxypropyl methylcellulose/1% (wt/vol) Pluronic F68 in reverse osmosis water, pH adjusted to 2.0 ± 0.1 with dilute hydrochloric acid. AMG2519493 and the vehicle control articles were provided to the test facility by Amgen Inc and stored at room temperature protected from light. Prior to dosing, AMG2519493 was suspended with the vehicle control at appropriate concentrations for the oral toxicology studies. Stability of the test article was confirmed for the duration of the toxicology studies, and all dose formulations were homogeneous and met concentration verification criteria.
Animals
Animals were housed at Association for Assessment and Accreditation of Laboratory Animal Care international-accredited facilities, and all research protocols were approved by their respective institutional animal care and use committees.
The cynomolgus monkey (Macaca fascicularis) was selected as the nonrodent species for toxicology studies based on the similarity of in vitro metabolic profiles with human, favorable pharmacokinetic parameters, and the availability of reagents for detailed immunophenotyping of circulating lymphocytes. Moreover, the amino acid identities of the human and cynomolgus monkey PI3 kinase isoforms is ≥98.8% (Supplementary Table 1). 11 -13 Animals of Chinese origin were group housed (up to 4 animals/sex/cage) in stainless steel cages and offered Certified Primate Diet #2055C (Harlan Laboratories, Inc) 1 to 2 times daily and water ad libitum unless fasted for study procedures. At initiation of dosing in the 1- and 3-month toxicology studies, animals were 5.6 to 8.5 and 4 to 8 years old, respectively. Environmental controls for the animal room were set to maintain 18 °C to 26 °C, a relative humidity of 30% to 70%, >10 air changes/hour, and a 12:12 hour light:dark cycle. Animals were given fruits, vegetables, or additional supplements and various forms of environmental enrichment. Prior to relocation to testing sites, all animals were reported as seronegative for simian immunodeficiency virus, simian retroviruses, filoviruses, simian T-lymphotropic virus, and herpes B virus, fecal culture negative for Salmonella sp. and Shigella sp., and skin-tested negative for tuberculosis.
Study Design
In the 1-month repeat-dose toxicology study, AMG2519493 was administered daily by oral gavage at 0 (vehicle control), 0.1, 1, 8, or 200 mg/kg (n = 3-5/sex/group). The lowest dose of 0.1 mg/kg was expected to be a subtherapeutic dose and was included to fully characterize the dose–response of pharmacologically mediated changes. Scheduled necropsies were conducted at the end of the 28-day dosing phase (n = 2-3/sex/group) and following a 1-month recovery (n = 2/sex in vehicle or 200 mg/kg group).
In the 3-month repeat-dose toxicology study, AMG2519493 was administered daily by oral gavage at 0 (vehicle control), 1, 8, or 100 mg/kg (n = 4-6/sex/group). The intended duration of dosing was 3 months with a 2-month recovery phase; however, early deaths occurred at ≥8 mg/kg. Due to signs of moribundity and early deaths at 100 mg/kg, dosing at 100 mg/kg was discontinued on study day 49. After dose discontinuation at 100 mg/kg, 2 animals/sex at 0 and 100 mg/kg entered the 2-month recovery phase and the remaining animals at 100 mg/kg were necropsied. Scheduled necropsies were conducted on study day 91 and recovery phase necropsies (not including early deaths) were conducted on study day 106.
Both toxicology studies were conducted at the same contract research organization and in compliance with the Food and Drug Administration Good Laboratory Practice (GLP) Regulations, 21 CFR Part 58. Study end points included clinical observations, body weight, qualitative food consumption, toxicokinetics, ophthalmic evaluation, clinical pathology, organ weights, macroscopic observation, and light microscopic evaluation of a full set of tissues. Vital signs (body temperature and respiration rate), electrocardiographic examination, and fecal smears were also included. Fecal examination and culture (Meriter Laboratories) was performed for the animals euthanized moribund as well as 2 concomitantly euthanized control females in the 3-month monkey study. Treatments to address fecal abnormalities and body weight loss included fiber bites, high calorie food supplements (yams, yogurt, and peanut butter), and antibiotics (enrofloxacin at 5-10 mg/kg twice daily; or erythromycin at 75 mg/kg twice daily).
Flow cytometry analysis of peripheral blood lymphocytes was conducted in the 1-month monkey study using routine methods. Blood was collected twice prior to initiation of dosing, on dosing phase days 7 and 28, and on recovery phase day 29 (study day 57). Total lymphocytes were identified using CD45+ staining and side scatter characteristics. Additionally, the following lymphocyte subsets were identified as follows: total T lymphocytes (CD3+); helper T lymphocytes (CD3+, CD4+, CD8−); cytotoxic T lymphocytes (CD3+, CD4−, CD8+), B cells (CD3−, CD20+), and NK cells (CD3−, CD16+). The percent relative and absolute numbers of each lymphocyte phenotype were enumerated.
Animals euthanized for unscheduled or scheduled necropsies were anesthetized with sodium pentobarbital, exsanguinated, and necropsied. The necropsy was routine and consisted of an examination of external features, body cavities, organs, and tissues. Weighed organs and tissues evaluated by light microscopy included a full list of tissues and organs consistent with those recommend for nonrodent toxicology studies. 14 Tissues and organs were preserved in 10% neutral buffered formalin, with the exception of eyes, optic nerves, and testes, which were preserved in modified Davidson’s fixative. After fixation, tissues were routinely processed for histologic examination and stained with hematoxylin and eosin. Representative sections of stomach from the 1-month toxicology study were stained with Warthin-Starry stain to enhance visualization of Helicobacter sp. Light microscopic changes were graded on a 5-point severity scale (minimal, slight, moderate, marked, and severe) in the 1-month study, and on a 4-point severity scale (minimal, mild, moderate, and marked) in the 3-month study. Light microscopic evaluation was performed by an American College of Veterinary Pathology (ACVP) board-certified pathologist at a contract research organization and peer reviewed by at least one additional ACVP board-certified pathologist. Only AMG2519493-related light microscopic changes in the lymphoid tissues and digestive tract are reported here; independent changes in the reproductive tract are not discussed.
Results
Toxicokinetics
In the 1-month study, AMG2519493 systemic exposure (Cmax and AUC0-24h) increased with ascending dose in a generally dose-dependent manner (Figure 1 and Supplementary Table 2). There was no sex-related difference in exposure, and no drug accumulation, except for minor accumulation at 200 mg/kg (day 28 exposure/day 1 exposure = ∼3.5-fold). At 0.1 mg/kg, the lowest dose tested, mean exposures were greater than the half-maximal inhibitory concentration (IC50) for human PI3Kδ isoform for approximately 4 hours. In contrast, exposures at 1 to 200 mg/kg doses were greater than or approximately equal to the human PI3Kδ IC50 for the entire 24-hour sampling period. Although AMG2519493 is considered highly selective for PI3Kδ, the high exposure levels at 8 and 200 mg/kg occasionally equaled or exceeded the human IC50 values for the α, β, and γ isoforms as shown in Figure 1.
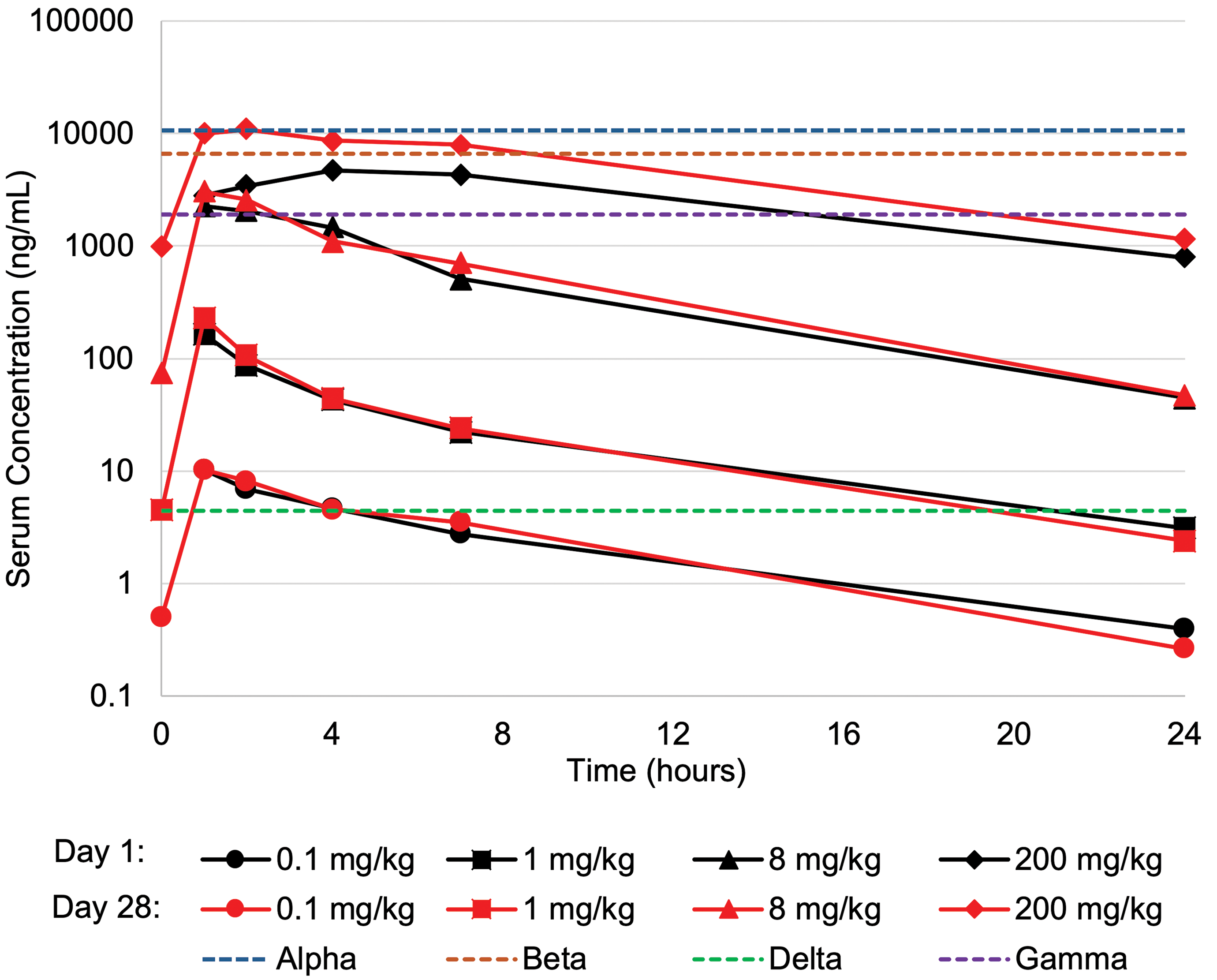
AMG2519493 concentration time course in the 1-month toxicology study in the cynomolgus monkey. AMG2519493 concentration was measured 1, 2, 4, 7, and 24 hours postdose on days 1 and 28 and predose on day 28. The enzymatic activity of AMG2519493 on recombinant human phosphatidylinositol 3-kinase (PI3K) isoforms was measured by luminescence detection of ATP in a luciferase-coupled assay. The IC50 values (corrected for protein binding) for human α, β, γ, and δ (dotted lines) PI3 kinase isoforms are overlaid for comparison to exposure in the toxicology study.
Systemic exposure of AMG2519493 in the 3-month study was similar to the 1-month study (Supplementary Table 2). After the first dose, exposure levels were within 2-fold of levels observed in the 1-month study at 1 and 8 mg/kg, the only dose levels used in both studies. Similar to the 1-month study, there were no sex-related differences in exposure, and exposure increased with dose level. Moribundity and early deaths confounded the comparison of the toxicokinetics following the last dose.
Moribundity and Mortality
In the 1-month study, one male at 200 mg/kg was euthanized on study day 5 due to clinical signs attributed to an enlarged retropharyngeal mass (see Light Microscopy section). Clinical pathology parameter changes from samples taken prior to necropsy were consistent with an inflammatory response (eg, increased neutrophils and fibrinogen). The remaining animals survived to the scheduled dosing phase necropsy; however, 2 females at 200 mg/kg were in poor clinical condition. In both cases, the poor condition was characterized by severe body weight loss (approximately 18%) and hypoactivity that was attributed to inflammation in the large intestines. In addition, one of the females had a spectrum of unique changes that was consistent with fatal fasting syndrome, which is associated with negative energy balance and rapid body weight loss in overweight animals. 15 -17 The lack of tolerability at 200 mg/kg informed the dose selection for the subsequent 3-month toxicology study, in which the high-dose level was reduced to 100 mg/kg.
Compared to the 1-month study, there was an increased incidence of unscheduled euthanasia in the 3-month study (Table 1). Dosing in 2 males and 2 females at 8 mg/kg and all animals at 100 mg/kg was discontinued on study day 49 due to poor clinical condition, abnormal feces, and body weight loss; 2 animals/sex at 0 and 100 mg/kg were subsequently designated to the recovery phase of the study, and the remaining moribund animals were submitted for necropsy. Due to progression of clinical signs, the 2 recovery phase females at 100 mg/kg were euthanized on recovery days 2 and 5 (study days 51 and 54, respectively), and the 2 concurrent vehicle control females were euthanized on recovery day 5. The poor clinical condition was attributed to inflammation in the large intestines. Animals in poor clinical condition also had secondary changes attributed to negative energy balance, dehydration, and/or physiologic stress. Recovery necropsy was conducted for 2 males at 0 and 100 mg/kg at the end of the 2-month recovery phase (study day 106).
Incidence of Unscheduled, Scheduled, and Recovery Necropsies in the 3 Month Cynomolgus Monkey Study.

Abbreviations: F, female; M, male.
a For animals at 100 mg/kg, the dosing phase was terminated on study day 49 and 2 animals/sex at 0 and 100 mg/kg were placed into the recovery phase early on study day 50. Due to continued clinical signs, recovery phase females at 100 mg/kg were euthanized on recovery days 2 and 5, and the concurrent recovery phase control females were euthanized on recovery day 5. Recovery phase males survived until the scheduled recovery phase necropsy on study day 106.
Clinical Observations
There were clinical observations consistent with gastrointestinal (GI) toxicity at the highest dose tested (200 mg/kg) during the in-life portion of the 1-month study. AMG2519493-related GI clinical signs included an increased incidence (the number of affected animals) and frequency (the number of days with observation) of red and/or mucoid feces in 8 of 10 animals at 200 mg/kg. Although some fecal observations could be attributed to individual animals, group housing occasionally prohibited determination of the affected individual, which made assessment of progression challenging. In general, abnormal feces were initially observed after the first week of dosing with mucoid feces preceding red feces and both persisting intermittently until the end of the dosing phase. Fecal samples were sporadically submitted for Salmonella sp and Shigella sp culture, which were negative. A 7-day course of the antibiotic enrofloxacin was administered to animals with fecal abnormalities, and partial or full resolution followed treatment; no other treatments were considered necessary by the veterinary staff. Of the 4 animals at 200 mg/kg assigned to the recovery phase, 3 had red and/or mucoid feces during the dosing phase. Two of these 3 recovery phase animals did not have red and/or mucoid feces during the recovery phase and one animal had persistent fecal abnormalities until the end of the recovery phase, albeit at a reduced frequency. Although not completely recovered in one animal, the reduced incidence and frequency of fecal abnormalities in the recovery phase animals demonstrated that these observations are reversible. Fecal abnormalities in animals at 200 mg/kg were associated with an approximately 10% mean body weight loss compared to prestudy values, without any associated change in food consumption. In the recovery phase, 3 of 4 animals at 200 mg/kg gained weight, demonstrating that body weight loss was also a reversible change.
In the 3-month study, similar but more severe GI-related clinical signs were observed (Figure 2). As observed in the 1-month study, group housing (4/sex/cage) prohibited determination of the affected individual for some fecal observations. At 1 mg/kg, there was a minor increased incidence of fecal abnormalities (ie, diarrhea) and low food consumption associated with a decrease in body weight gain compared to concurrent controls (males: 0.4 kg gain vs 1.0 kg gain; females: 0.1 kg gain vs 0.4 kg gain). These clinical observations occurred mostly in the latter half of the dosing phase. In contrast, animals at 8 and 100 mg/kg had increased incidence and more severe GI-related clinical signs, which tended to occur earlier and persist intermittently throughout the dosing and recovery phases. At these dose levels, the clinical observations included diarrhea, decreased food consumption, and body weight loss (−2% to −34.3%), as well as other nonspecific clinical signs of poor clinical condition, such as hypoactivity, recumbency, and coldness to touch. The clinical observations of poor condition and decreases in body weight at ≥8 mg/kg precipitated early unscheduled necropsies for several animals as shown in Table 1.
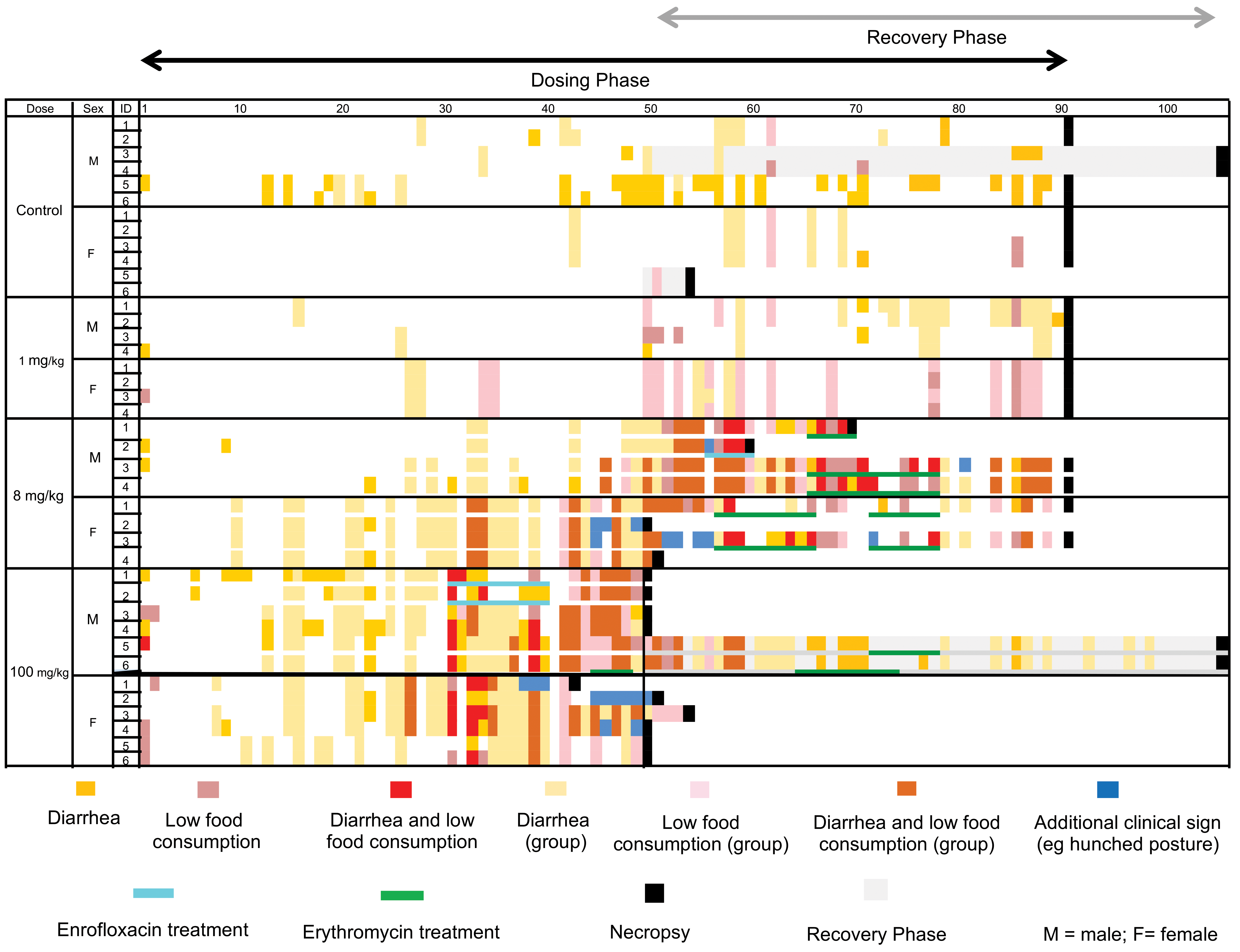
Time course of individual and cage group clinical observations related to digestive tract toxicity and antibiotic treatment in the 3-month cynomolgus monkey study. Due to group housing (4/sex/cage), some observations of abnormal feces or low food consumption could not be assigned to an individual animal. In this case, the observation was assigned as a “group” observation. When both individual animal and group observations were observed on the same day, only individual data are shown for that animal. When additional clinical signs of poor clinical condition (eg, hunched posture) were observed, only additional clinical signs (blue color) are shown for that animal irrespective of observations of diarrhea or low food consumption. Severe clinical signs at ≥8 mg/kg resulted in early euthanasia and necropsy, and termination of the dosing phase at 100 mg/kg. Administration of antibiotics to monkeys with severe clinical signs was occasionally associated with improved clinical condition.
Although high calorie food supplements were provided in the 3-month study, these did not improve the fecal abnormalities. Antibiotic administration to animals with severe clinical signs was associated with mixed outcomes. Most of the animals euthanized by day 54 did not receive antibiotic treatment, with the exception of 2 males at 100 mg/kg that received enrofloxacin but were subsequently euthanized due to deteriorating condition. After early euthanasia up to day 54, several remaining animals (3 males and 2 females at 8 mg/kg and 2 males at 100 mg/kg) received erythromycin antibiotics. All erythromycin-treated animals (except for 1 animal at 8 mg/kg) responded well to treatment and those animals remained on study without further deterioration. Fecal examinations, including bacterial cultures, were conducted for animals with severe GI clinical signs, and Campylobacter spp. was the most prevalent infectious agent identified. Moderate to heavy growth of this bacterium was identified in all 12 animals tested at 8 and 100 mg/kg, while there was no such observation in culture of feces from 2 recovery phase control females. Fecal examination also identified Cryptosporidium and Giardia lamblia at 100 mg/kg, each in one animal.
As previously indicated, 2 males at 100 mg/kg were dosed for 49 days and survived to the scheduled recovery phase necropsy after a 2-month treatment-free period. During the first 2 weeks of the recovery phase, these animals had persistent diarrhea and decreased food consumption, and one animal had additional body weight loss (11%). After 2 weeks in recovery, the clinical observations spontaneously resolved in one animal, whereas the clinical observations for the other animal responded to a course of antibiotics. For both animals, body weight at the end of the recovery phase was similar to or greater than prestudy body weight.
Clinical Pathology and Immunophenotyping
There were test article–related changes in clinical pathology parameters at 200 mg/kg in the 1-month study that were attributed to inflammation in the large intestines. These changes included a minimal to mild increase in neutrophils, a mild to marked increase in fibrinogen, and a mild decrease in albumin. Other changes, such as a minimal decrease in red blood cell (RBC) mass (RBC count, hematocrit, and hemoglobin) and a mild decrease in reticulocytes were considered secondary effects of inflammation on RBC production. All test article–related clinical pathology parameter changes were fully recovered after the 1-month recovery phase.
In the 3-month study, animals at 1 or 8 mg/kg that survived to the scheduled dosing phase necropsy had clinical pathology parameter changes that were similar to animals in the 1-month study (eg, increased neutrophils, decreased RBC mass, and decreased albumin). Animals at 8 or 100 mg/kg and euthanized early at an unscheduled time point during the dosing or recovery phases had more pronounced clinical pathology changes consistent with inflammation and generalized poor clinical condition. In 2 males at 100 mg/kg assigned to the 2-month recovery phase, all clinical pathology parameters were normal, except for albumin, which exhibited partial recovery.
In the 1-month monkey study, immunophenotyping of peripheral blood lymphocytes using flow cytometry demonstrated a dose-dependent decrease in circulating B cells (absolute counts and % of total lymphocytes) at ≥1 mg/kg (Figure 3). Although a minor decrease in mean B cells was evident at 0.1 mg/kg, this difference was considered within normal limits based on evaluation of the variation in individual animal data. There were no AMG2519493-related changes in other subpopulations, including total T cells, CD4+ T cells, CD8+ T cells, and NK cells (data not shown). After the 1-month recovery phase, B cell number fully recovered in 3 animals at 200 mg/kg and partially recovered in 1 animal at 200 mg/kg. Immunophenotyping of peripheral blood lymphocytes was not conducted in the 3-month study because recovery of the pharmacologic effect on B cell number was demonstrated in the 1-month study.
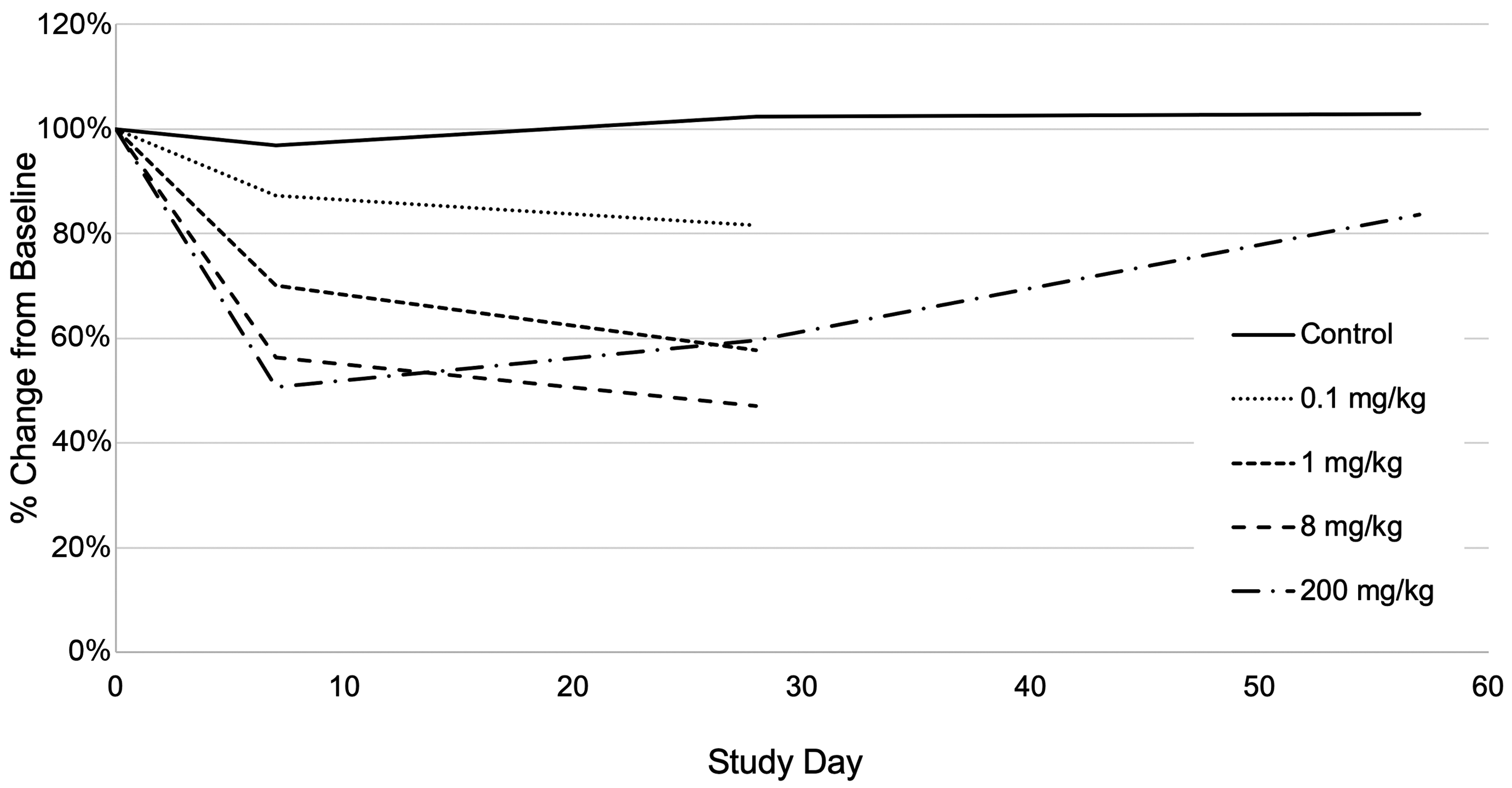
Reversible AMG2519493-related decrease in mean absolute B cell numbers in the 1-month toxicology study in the cynomolgus monkey. Flow cytometry analysis was performed on study days 1 (predose), 7, 29 (dosing phase necropsy 24 hours after the last dose), and 57 (recovery phase necropsy 29 days after the last dose). Mean values represent the percent change from baseline on study day 1.
Macroscopic Observations and Organ Weight Changes
There were a limited number of macroscopic observations and organ weight changes in the immune system and digestive tract organs and tissues. In the 1-month study, there was thickened or discolored gastric mucosa at 1 (one animal) and 200 (2 animals) mg/kg. At 200 mg/kg, decreased thymus weight in the 2 females in poor clinical condition at the end of the dosing phase was attributed to physiologic stress. In animals that survived to scheduled dosing phase necropsy in the 3-month study, there were no test article–related macroscopic observations or organ weight changes in the immune system or digestive tract organs and tissues.
Light Microscopy
Immune system
At all dose levels tested, except for 0.1 mg/kg in the 1-month study, dose-dependent light microscopic changes were observed in all lymphoid organs (spleen, mesenteric lymph node, axillary lymph node, gut-associated lymphoid tissue [GALT], and tonsil) examined in the 1- and 3-month monkey studies (Tables 2 and 3). In the spleen, lymph nodes, and GALT, there was a diffuse decrease in lymphocytes in B cell-rich areas, including the lymphoid follicles in all tissues and the marginal zone in the spleen (Figure 4). Affected lymphoid follicles were small and often lacked germinal centers, while in the spleen, the marginal zones had fewer cells and were thin. The tonsils had a generalized decrease in lymphocytes without a clear predilection for specific tissue compartments. In the 1-month study, the severity of the AMG2519493-related changes in the lymphoid tissues was minimal to marked. In the 3-month study, the severity was generally minimal in animals that survived to scheduled dosing phase necropsy, and in animals that were euthanized early due to moribundity, the change was generally more severe (minimal to moderate), likely due to higher AMG2519493 exposure and added physiologic stress. Despite the high incidence and the diffuse distribution of the decrease in lymphocytes, there was no associated change in spleen weight, which may be due to the greater inherent variability in normal monkey spleen weights and low animal numbers used in the study. 18 The lymphoid organs changes correlated with decreased circulating B cells in the 1-month study and were attributed to the intended pharmacologic activity of AMG2519493. After the recovery phase, the changes in lymphoid organs either fully recovered or showed evidence of reversibility.
AMG2519493-Related Immune System Light Microscopic Changes in the 1-Month Monkey Study.
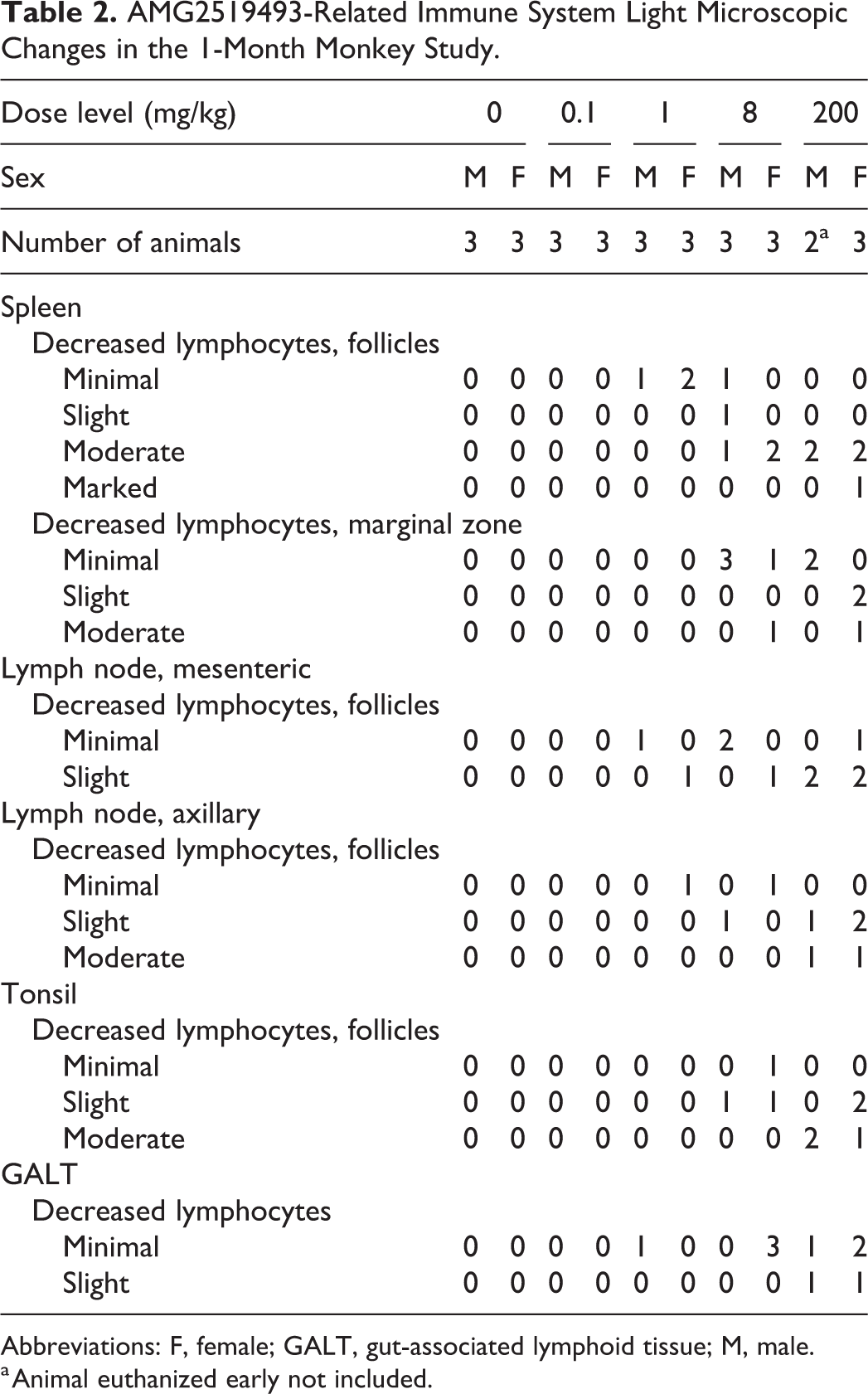
Abbreviations: F, female; GALT, gut-associated lymphoid tissue; M, male.
a Animal euthanized early not included.
AMG2519493-Related Immune System Light Microscopic Changes in the 3-Month Monkey Study.
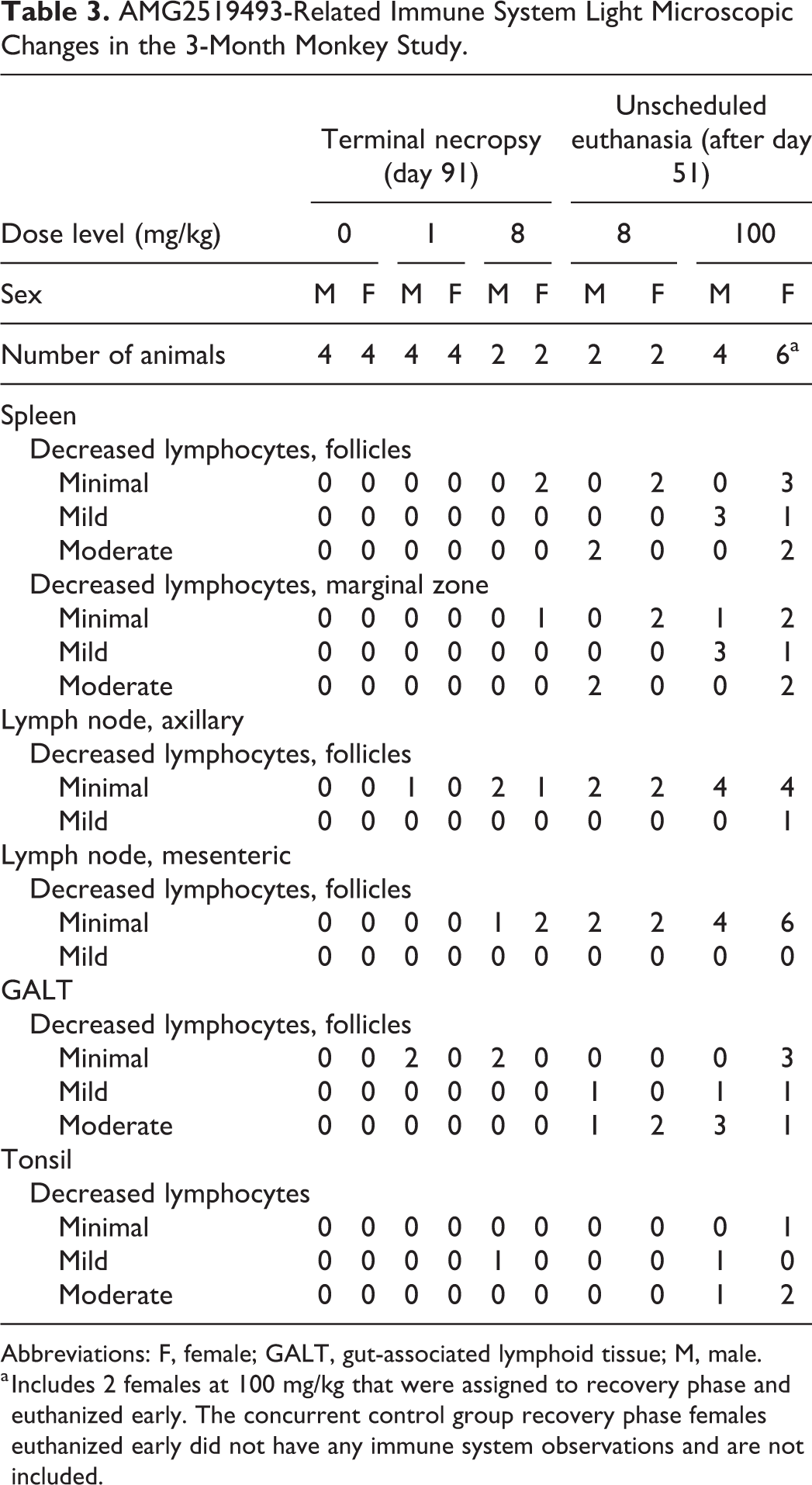
Abbreviations: F, female; GALT, gut-associated lymphoid tissue; M, male.
a Includes 2 females at 100 mg/kg that were assigned to recovery phase and euthanized early. The concurrent control group recovery phase females euthanized early did not have any immune system observations and are not included.
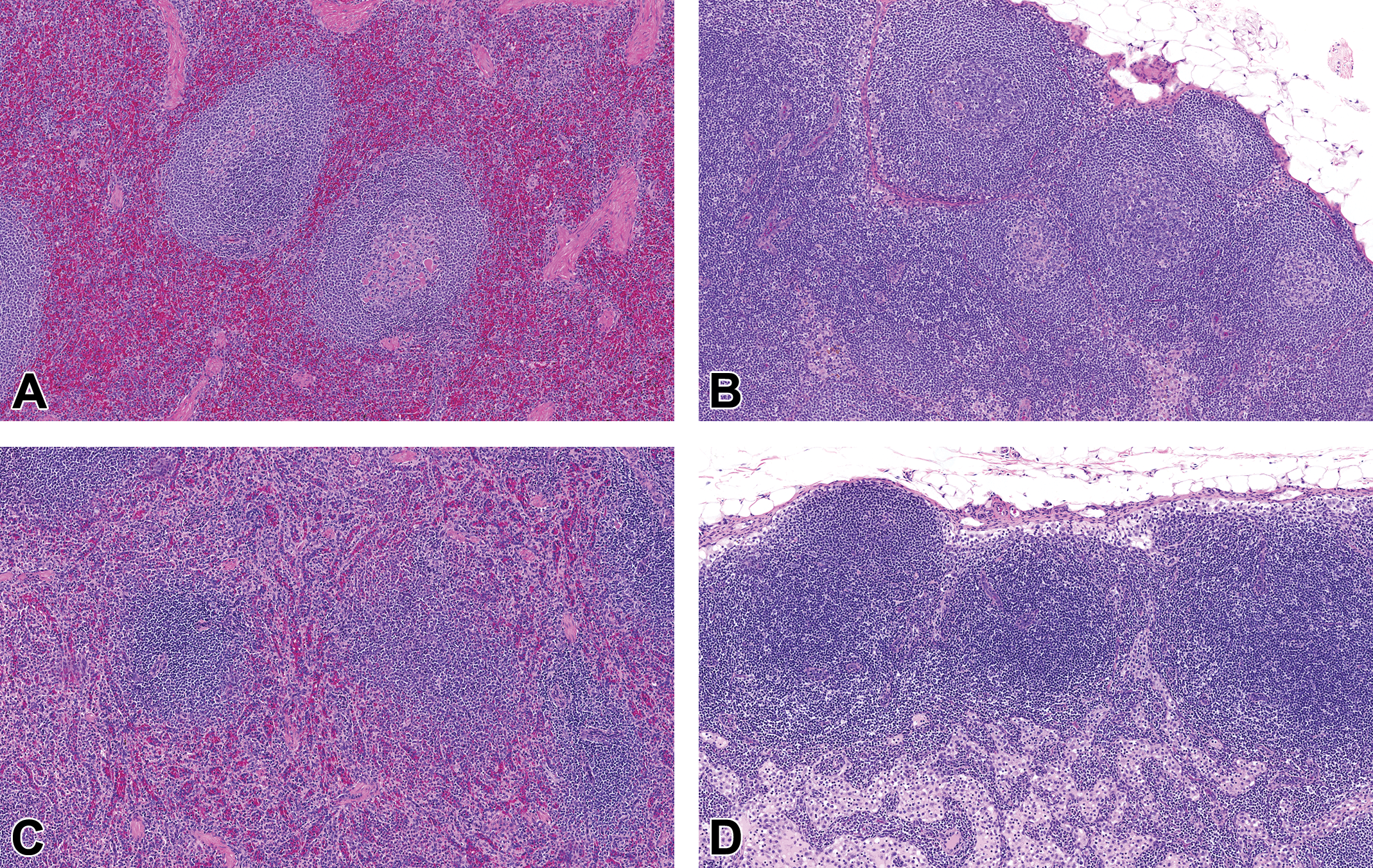
Representative photomicrographs of spleen and mesenteric lymph node from a normal control cynomolgus monkey (A and B) and a monkey administered 8 mg/kg AMG2519493 (C and D) in the 3-month toxicology study. Images demonstrate minimal AMG2519493-related decrease in lymphocytes in lymphoid follicles associated with the absence of germinal centers and reduced thickness of the splenic marginal zone. Hematoxylin and eosin, original objective ×10.
Inflammation and opportunistic infection of the digestive tract
In the 1- and 3-month toxicology studies, there were light microscopic changes in the digestive tract, including the oral cavity, esophagus, stomach, cecum, and colon. These changes were generally dose-dependent and attributed to AMG2519493-related immunomodulation and secondary alteration of resistance to infection.
One-month study
As previously described, one animal at 200 mg/kg was euthanized on day 5 due to clinical signs attributed to an enlarged retropharyngeal mass. By light microscopy, this mass was a large abscess involving a local lymph node with an inflammatory response that extended to the adjacent tonsils and thyroid glands. Inflammation surrounding the lymph node was characterized by large numbers of neutrophils (many of which were degenerating) admixed with abundant bacteria and associated with an early fibrotic response. Based on the light microscopic characteristics and the location, the retropharyngeal abscess was considered to have been initiated either by gavage trauma from the oral dosing procedure or by a preexisting nidus of bacteria that was exacerbated by AMG2519493-related immunomodulation.
In the stomach of all animals, including controls, there were light microscopic changes consistent with those reported for Helicobacter sp. infection. 19 These changes were most pronounced in the gastric antrum and included acute inflammation, epithelial hyperplasia, infiltrates of lymphocytes, focal erosions, and the presence of spiral-shaped, intraluminal and intracellular, Warthin-Starry positive bacteria (Figure 5). The light microscopic changes correlated with macroscopic observations at necropsy including thickened and red discoloration of the mucosa. Acute inflammation was characterized by locally extensive neutrophil infiltration of the lamina propria often with multifocal loss of gastric glands. Acute inflammation was often associated with increased thickness of the gastric mucosa and elongated gastric glands with increased epithelial cell basophilia. Infiltrates of lymphocytes were observed in all animals and consisted of variable numbers of lymphocytes and plasma cells within the lamina propria. Except for the lymphocytic infiltrates, the incidence and/or severity of these findings were slightly higher in animals administered 1 or 200 mg/kg AMG2519493 (Table 4).
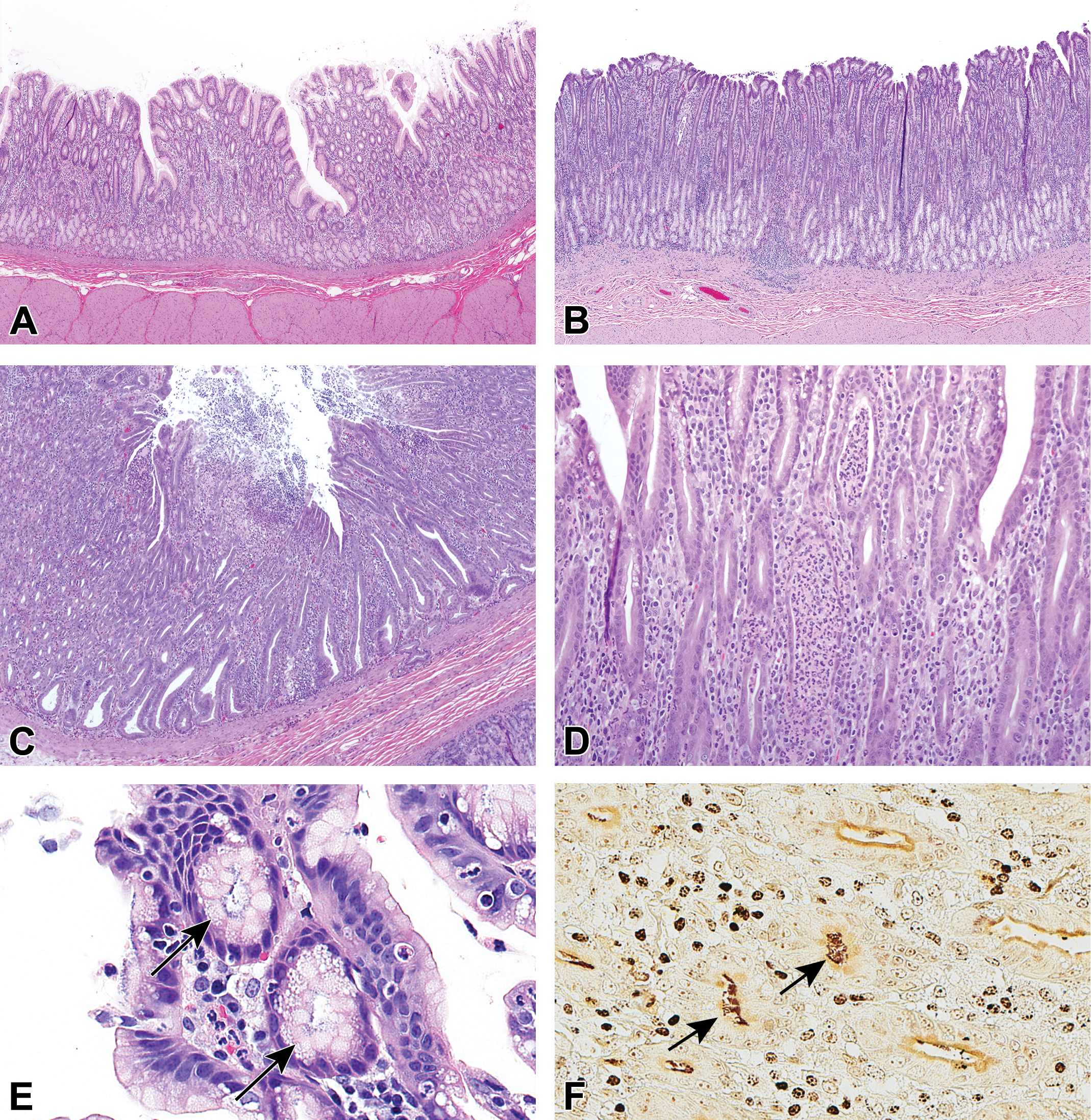
Representative photomicrographs showing morphologic changes suggestive of Helicobacter sp-induced infection in the stomach of cynomolgus monkeys in the 1-month toxicology study. Normal gastric antrum from a control monkey (A; original objective ×4) and gastric antrum from a monkey at 200 mg/kg with slight increased mucosal thickness due to epithelial hyperplasia (B; original objective ×4). A focal minimal erosion in the gastric mucosa of a monkey at 1 mg/kg (C; original objective ×4). Slight acute inflammation in the mucosa of a monkey at 200 mg/kg with neutrophil infiltration (D; original objective ×20). Small, spiral shaped, intraluminal, and intracellular, Warthin-Starry positive bacteria (arrows) in the gastric mucosa of a monkey at 200 mg/kg (E and F; original objective ×40). Hematoxylin and eosin (A-E) and Warthin-Starry (F).
Incidence and Severity of Light Microscopic Helicobacter sp.–Related Changes in the Stomach in the 1-Month Monkey Study.
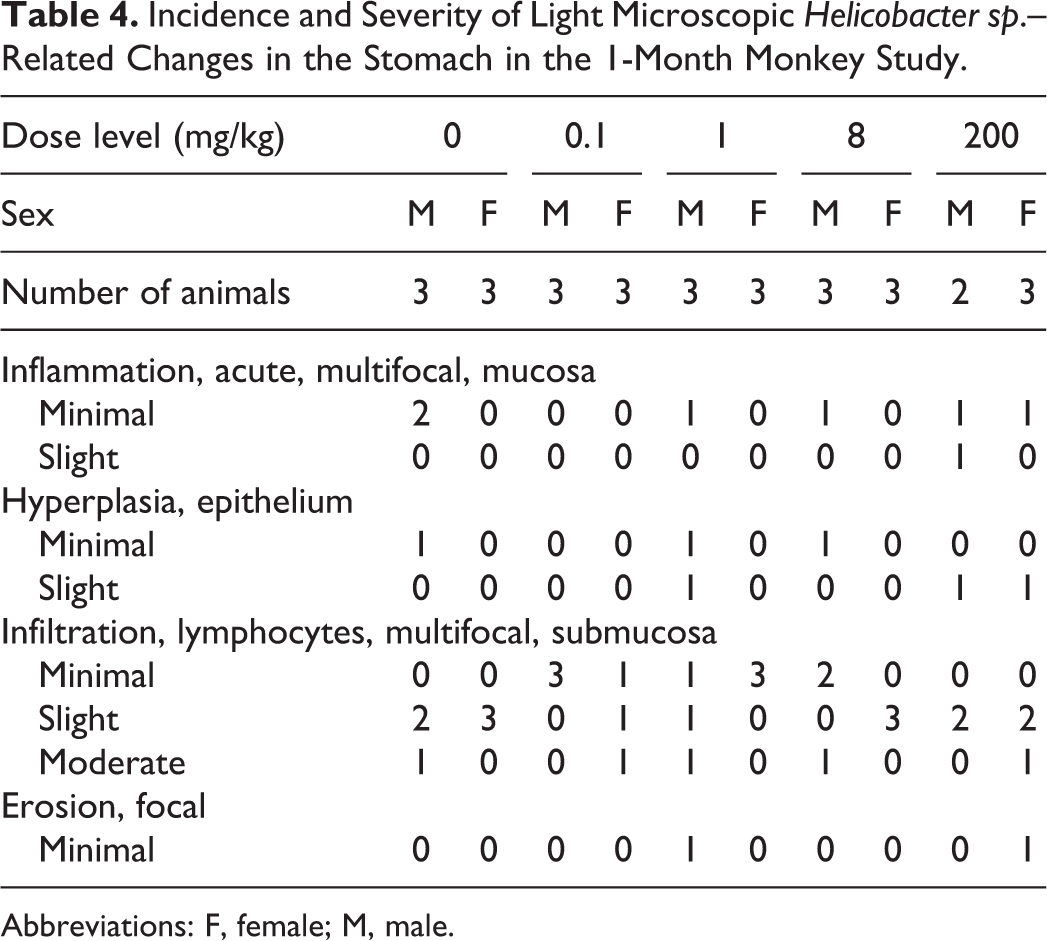
Abbreviations: F, female; M, male.
In the cecum and/or colon of some animals at all dose levels, there was AMG2519493-related inflammation, epithelial hyperplasia, and/or focal ulcers (Figure 6). Minimal inflammation was characterized by a multifocal neutrophilic infiltrate of the mucosal glands (crypts), often associated with epithelial degeneration and necrosis, or loss of glands. Slight to moderate inflammation had more extensive areas of affected mucosa with an increased neutrophilic infiltrate, more severe epithelial degeneration and necrosis, and distortion of gland architecture. Increased severity of inflammation correlated with clinical pathology parameter changes consistent with inflammation at 200 mg/kg. Hyperplasia of the colonic epithelium was generally diffuse and consisted of elongated glands, resulting in increased thickness of the mucosa.
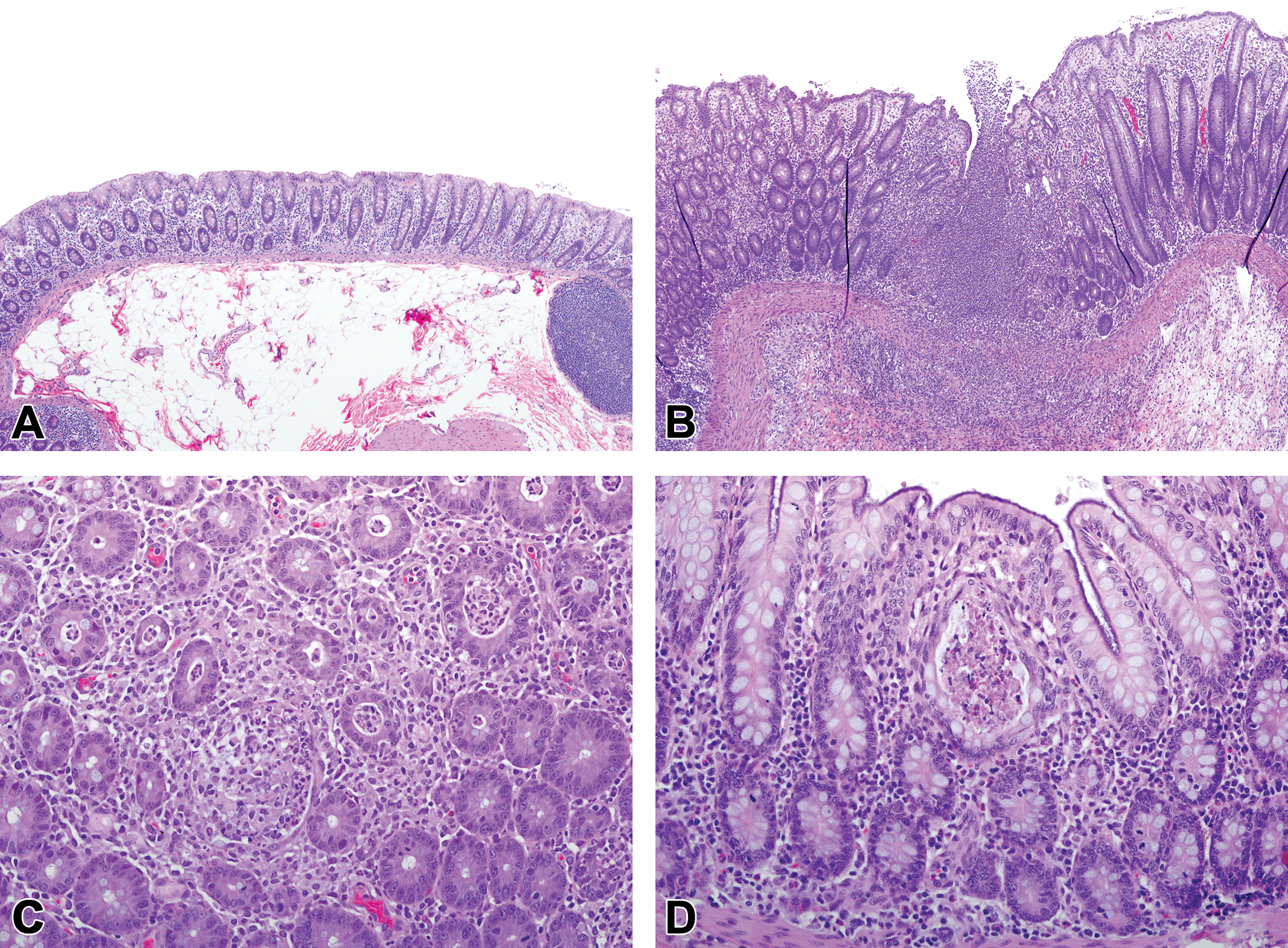
Representative photomicrographs of the colon from a normal control cynomolgus monkey (A; original objective ×4) and monkeys administered 200 mg/kg AMG2519493 (B through D) in the 1-month toxicology study. Moderate mixed cell inflammation was characterized by an extensive neutrophilic inflammatory infiltrate of the mucosa, often associated with epithelial hyperplasia, focal mucosal ulcers (B; original objective ×4), epithelial degeneration and necrosis, and distortion of gland architecture (C; original objective ×20). After a 1-month recovery phase, the inflammatory process had resolved and minimal residual dilated colonic glands containing cellular debris were observed (D; original objective ×20). Hematoxylin and eosin.
At 0.1 to 8 mg/kg, the combined incidence and severity of the light microscopic changes in the cecum and colon was low and just above the threshold of normal variation in the cynomolgus monkey (Table 5). Animals at these dose levels did not have abnormal clinical observations or body weight loss, which correlates with the low incidence and severity of these changes. However, at 200 mg/kg, all animals had inflammation, which was more severe and correlated with clinical signs of GI toxicity, including abnormal feces and decreased body weight. In addition, inflammation in these animals was associated with more prominent epithelial hyperplasia, and multiple ulcers in one animal. The changes in the large intestine at 200 mg/kg were considered the primary cause of the lack of tolerability at this dose level, including the 2 females that were in poor condition at the end of the dosing phase.
Combineda Incidence and Severity of Light Microscopic Changes in the Large Intestine in the 1-Month Monkey Study.

Abbreviations: F, female; M, male.
a The incidence is based on the combined observations in the cecum, proximal colon, and distal colon for each animal, and the severity is based on the highest severity from those 3 tissues.
One female at 200 mg/kg that was in poor condition had small numbers of basophilic intranuclear viral inclusions consistent with adenovirus 20 in the epithelium lining the jejunum and ileum (Figure 7A). Intranuclear inclusions were located at the tips or sides of villi, and many of the cells in the vicinity had lost normal basal polarity, which was detectable at low magnification (×4 to ×10 objective). In the ileum of one male at 200 mg/kg, small (3-4 µm), round, acid-fast positive protozoa consistent with Cryptosporidium sp. 21 were located within the lumen and attached to the apical surface of the epithelium (Figure 7B).
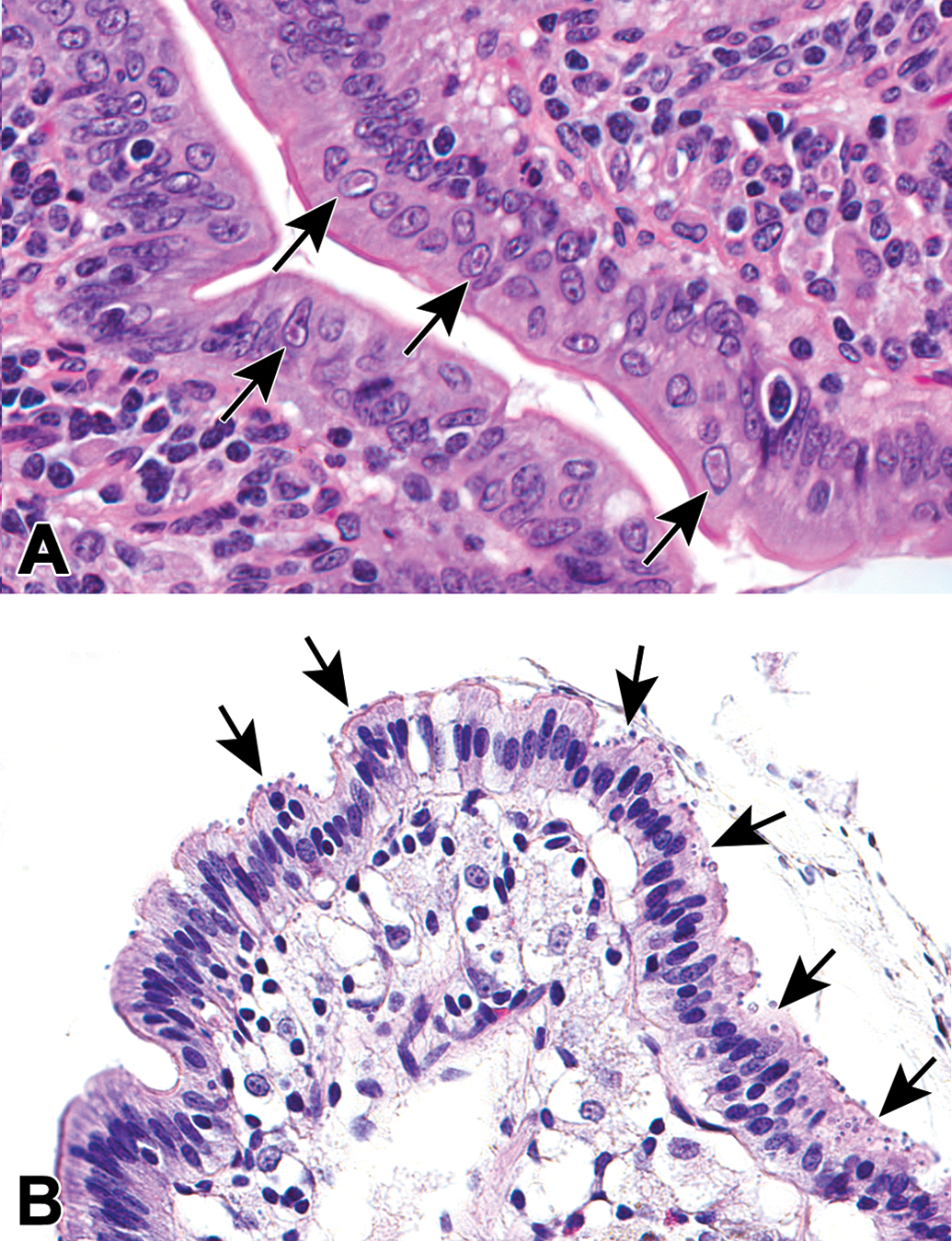
Representative photomicrographs of viral inclusions, and protozoa in the small intestine of cynomolgus monkeys administered AMG2519493 in the 1-month toxicology study. In the small intestine of one monkey administered 200 mg/kg AMG2519493, there were intranuclear viral inclusions (arrows) consistent with adenovirus within epithelial cells at the villus tips, often in cells that had lost basal polarity (A). The adjacent epithelium was frequently disorganized. In the small intestine of one monkey administered 200 mg/kg AMG2519493, there were numerous, small (3-4 µm), round protozoa (arrows) consistent with Cryptosporidium sp. on apical surface of the epithelium (B). Hematoxylin and eosin (A and B). Original objective ×40.
After a 1-month recovery period, inflammation in the stomach and large intestine had completely reversed and there was no evidence of infectious agents. In the colon and cecum of a few animals, there was minimal to slight dilatation of the glands lined by intact epithelium and associated with intraluminal cellular debris (Figure 6D). The dilated glands were considered part of the resolution process following the cessation of the inflammation, and it was expected that a return to normal mucosal morphology would occur with a longer recovery phase.
Three-month study
Compared to the 1-month study, inflammation and opportunistic infection of the digestive tract was more severe in the 3-month study. Animals at 1 and 8 mg/kg that survived to terminal necropsy had minimal inflammation in the large intestine, as well as occasional dilated crypts with cellular debris, in the cecum and colon (Table 6). In contrast, animals at 8 and 200 mg/kg that did not tolerate administration of AMG2519493 had inflammation that was minimal to moderate, more diffuse, and associated with ulceration, crypt microabscesses, and/or crypt necrosis. Inflammation of the large intestine was considered the primary cause of moribundity and mortality at 8 and 100 mg/kg dose levels. Similar to the 1-month study, the large intestinal mucosa was thickened due to diffuse hyperplasia of the epithelium; however, this change was not diagnosed separately because it was considered a secondary reactive response to the inflammatory process. Two animals had focal, minimal to mild, acute neutrophilic inflammation in the small intestine; the cause of the inflammation was not identified, and it was considered minor and not to have contributed to clinical signs. Changes attributed to Helicobacter sp. were not observed in the 3-month study.
Combineda Incidence and Severity of AMG2519493-Related Inflammation in the Large Intestine in the 3-Month Monkey Study.

Abbreviations: F, female; M, male.
a The incidence is based on the combined observations in the cecum and colon for each animal, and the severity is based on the highest severity in those 2 tissues.
In 11 of 14 animals terminated early in the 3-month study, bacterial, protozoal, viral, or fungal infectious agents were identified in histologic sections of tissues. These opportunistic infections were attributed to AMG2519493-related immunomodulation, as well as physiologic stress and poor condition. One female at 100 mg/kg had fungal invasion of the tongue, tonsil, and esophagus (Figure 8A and B). The fungus invaded the stratum corneum and formed yeast and pseudohyphal forms consistent with Candida sp. 22 The distal esophagus of 6 animals had changes suggestive of infection with lymphocryptovirus (LCV) infection, including a thickened epithelial layer, ballooning degeneration of surface epithelial cells, and intranuclear viral inclusions in the stratum granulosum (Figure 8C and D). 23 One animal had intraluminal protozoa in the cecum consistent with Giardia sp. (Figure 9A), 24 and 7 animals had minimal to moderate overgrowth and occasional tissue invasion of Balantidium coli in the cecum and/or colon (Figure 9B-D). 24 Two animals had viral inclusions consistent with adenovirus and associated epithelial changes similar to that observed in the 1-month study.
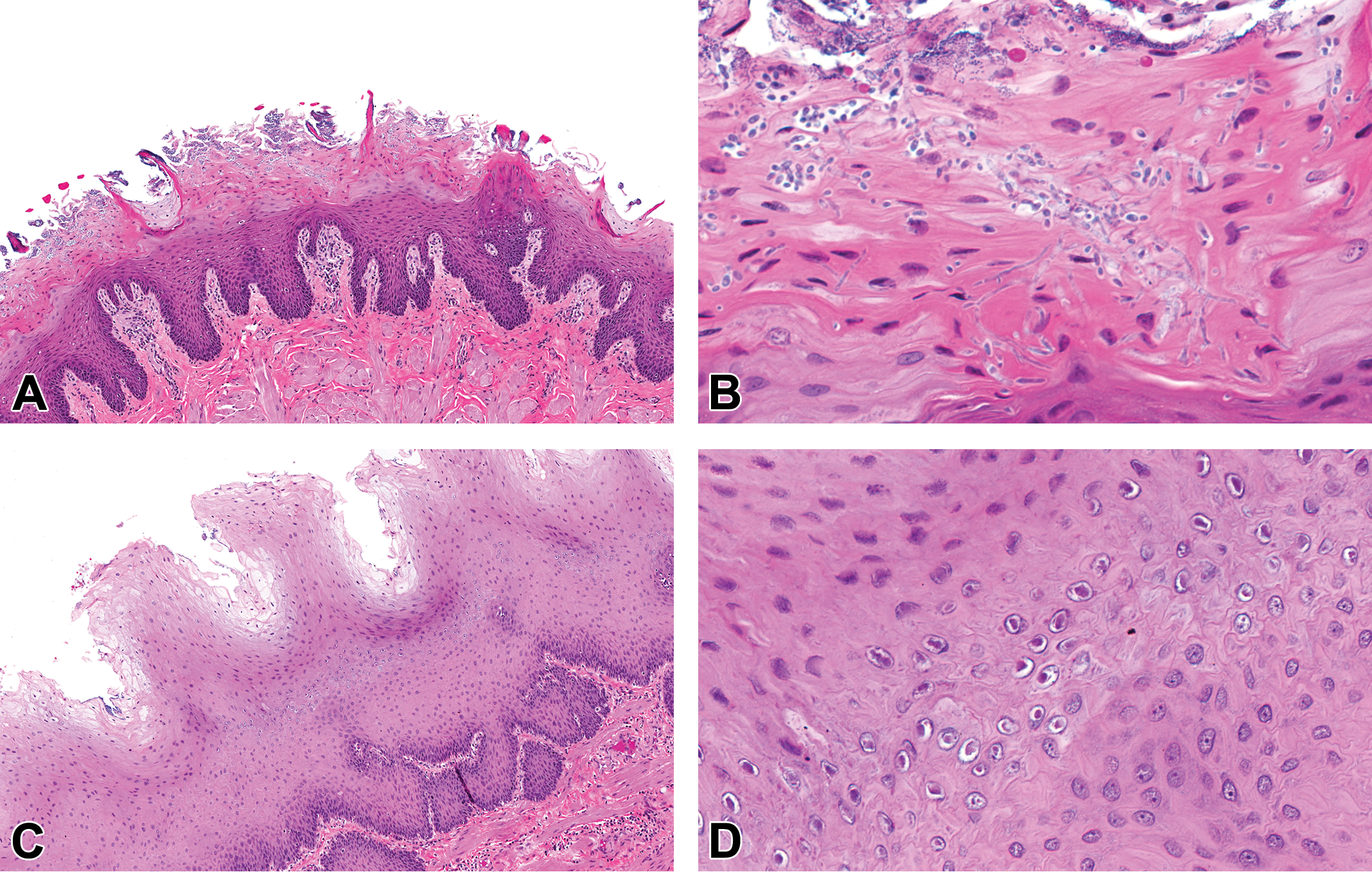
Representative low (original objective ×10) and high (original objective ×40) magnification photomicrographs of fungal invasion of the tongue and intranuclear viral inclusions in the esophagus of cynomolgus monkeys administered AMG2519493 in the 3-month toxicology study. In the tongue (A and B), the fungus invaded the stratum corneum and was characterized by yeast and pseudohyphal forms consistent with Candida sp. In the esophagus (C and D), the epithelium was thickened and composed of widespread ballooning degeneration of surface epithelial cells and frequent intranuclear viral inclusions within the stratum granulosum. The morphologic features of the esophageal epithelium and the intranuclear viral inclusions are suggestive of infection with lymphocryptovirus. Hematoxylin and eosin.
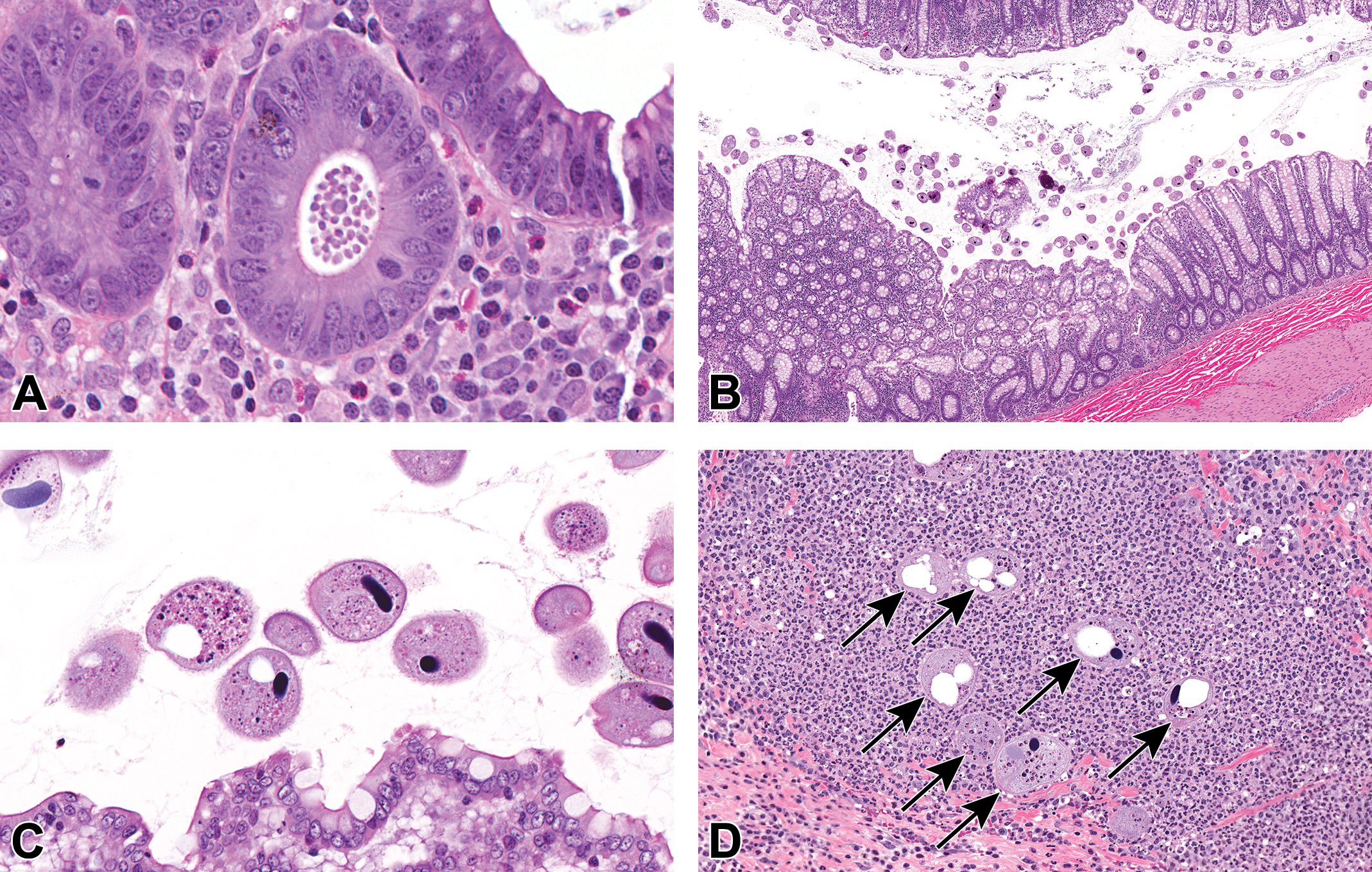
Representative photomicrographs of protozoa in the large intestine of cynomolgus monkeys administered AMG2519493 in the 3-month toxicology study. Within the cecum of one monkey at 200 mg/kg, there were aggregates of protozoal organisms consistent with Giardia sp. in the gland lumens (A, original objective ×40). Increased numbers of intraluminal protozoa consistent with Balantidium coli (B-D; original objectives ×5 and ×40) were observed in the large intestine of several monkeys, occasionally invading the submucosa (arrows; D). Hematoxylin and eosin.
Due to early termination of most of the high-dose group animals, recovery of the digestive tract inflammation and infection could only be assessed in 2 males at 100 mg/kg AMG2519493 for 49 days. Consistent with the steadily improved clinical condition throughout the recovery phase, there was no light microscopic evidence of inflammation in these 2 animals, indicating that the 2-month recovery phase was sufficient for full reversibility. Furthermore, no infectious organisms were identified in the recovery phase animals; however, it is uncertain if either animal had infectious agents at the end of the dosing phase.
Discussion
AMG2519493, a highly selective and potent small-molecule inhibitor of PI3Kδ, was in clinical development for the treatment of human inflammatory diseases; however, the development program was recently terminated. Toxicology studies in the cynomolgus monkey identified the immune system (spleen, lymph nodes, GALT, tonsil) and digestive tract as primary targets of AMG2519493-related pharmacologic effects and secondary toxicity due to anticipated immunomodulation. In general, the toxicity was dose- and duration-dependent and dose levels ≤1 mg/kg were well tolerated. In the immune system, AMG2519493-related changes included a decrease in circulating B cells and light microscopic evidence of a diffuse decrease in lymphocytes in B cell rich regions of the spleen, lymph nodes, and GALT. The changes in the immune system were consistent with the consequences of PI3Kδ inhibition, which is the intended pharmacologic activity of the test article. In the digestive tract, AMG2519493-related inflammation in the cecum and colon was associated with abnormal fecal observations, decreased food consumption, and other clinical signs of poor tolerability, resulting in early unscheduled euthanasia or dose group termination at ≥8 mg/kg in the 3-month toxicology study. The morphologic features of the inflammatory infiltrate, the Campylobacter spp. overgrowth in fecal cultures, and the partial response to antibiotic suggest that enteric bacteria played a role in the inflammation in the large intestine. In addition, pharmacologically mediated immunosuppression was associated with secondary opportunistic infection restricted to the digestive tract. After a treatment-free recovery phase, the AMG2519493-related immune and digestive tract toxicity demonstrated evidence of reversibility and there was no evidence opportunistic infection. Compared to toxicity profiles for approved kinase inhibitors, the AMG2519493-related immune system and digestive tract toxicities described here are uncommon. 25
Systemic exposure of AMG2519493 increased with increasing dose and included potentially subtherapeutic, PI3Kδ-specific, and nonspecific PI3K exposure levels. The absence of in vitro potency and selectivity data for cynomolgus monkey PI3K isoforms warrants caution in comparing exposures in these toxicology studies to in vitro human isoform inhibition and potential translation to the clinic. Despite that caution, available evidence suggests the comparisons are reasonable. There is high amino acid sequence identity between human and cynomolgus monkey PI3 kinase isoforms (99.6% for PI3Kδ; Supplementary Table 1). Moreover, the intended pharmacologic activity of the test article (eg, decreased B cell numbers) was observed at low-dose levels in rat and monkey toxicology studies and in studies using mouse models of autoimmune diseases (systemic lupus erythematosus and collagen-induced arthritis; data not shown) suggesting that it is active against a wide species range of PI3Kδ isoforms.
The lowest dose level tested (0.1 mg/kg) in the toxicology studies exceeded the IC50 for human PI3Kδ for only a few hours. Not surprisingly, there were no detectable pharmacologic effects or toxicity at this dose level. Systemic exposure at the mid dose (1 mg/kg) exceeded the IC50 for human PI3Kδ for most of the dosing interval but did not approach the IC50 for the other human PI3K isoforms. This dose level was well tolerated and associated with the intended pharmacologic effect on the immune system and only minor changes in the digestive tract, including diarrhea and minimal inflammation of the large intestine; there was no evidence of opportunistic infection.
The observations at low exposures are consistent with the role of PI3Kδ in regulating the immune system. Phosphatidylinositol 3-kinase δ is required for B cell receptor–mediated activation, differentiation, and immunoglobulin secretion, and PI3Kδ inhibition leads to a selective depletion of antigen-activated B cells. 5 In addition, genetically engineered mice expressing a catalytically inactive form of PI3Kδ (p110δD910A/D910A mice) have defects in B- and T-cell signaling, maturation, and humoral immune responses, and lack germinal centers in the spleen, lymph nodes, and Peyer’s patches. 26,27 Phosphatidylinositol 3-kinase δ inhibition also partially impairs other leukocyte functions, 7 including cytokine and chemokine production by T cells, NK cells, basophils, mast cells, and neutrophils. Although these immune cell subset numbers were normal in the 1-month toxicology study, functional activity of these cells was not evaluated.
Phosphatidylinositol 3-kinase δ also plays a critical role in intestinal homeostasis in models of experimental inflammatory bowel disease. p110δD910A/D910A mice develop spontaneous inflammation in the large intestine, characterized by mucosal hyperplasia, crypt microabscesses, and mixed inflammatory cell infiltrates, as well as regenerative changes in the glandular epithelium. 26 These genetically engineered mice have reduced regulatory T cells with attenuated suppressor function that were nonprotective in an adoptive colitis model. 28 Phosphatidylinositol 3-kinase δ expression is increased when wild-type mice are transferred from germ-free conditions to normal microbiota, and enteric microbiota are required to drive the inflammation in the large intestine in PI3Kδ deficient mice. Although similar AMG2519493 exposure and immune system changes were observed in the companion 1- and 3-month GLP toxicology studies in the Sprague Dawley rat, no digestive tract toxicity was observed (Supplementary Table 3). Taken together, the literature and our own experience indicate that differences in normal enteric microbiota and genetic background can influence the toxicity profiles of PI3Kδ inhibitors in nonclinical studies.
In the clinic, pharmacologic inhibition of PI3Kδ is associated with adverse events that share some similarities to the AMG2519493-related toxicities in the monkey studies. Idelalisib (Zydelig), a selective inhibitor of PI3Kδ (>100-fold selectivity over other isoforms), is marketed for the treatment of B cell malignancies, such as chronic lymphocytic leukemia. In humans, adverse events associated with idelalisib include fatal or severe hepatotoxicity, diarrhea, colitis, pneumonitis, and intestinal perforation, for which US prescribing information contains a black box warning. 9 In addition, drug-related deaths presumptively due to inhibition of PI3Kδ and secondary opportunistic infection with Pneumocystis jirovecii and cytomegalovirus have been observed. 29,30 In the dog, idelalisib-related changes that were similar to AMG2519493-related changes reported here include mucoid feces, decreased body weight, lymphoid depletion in multiple immune system organs (primarily B cell regions), and inflammation in the intestines with crypt dilatation. 31 In contrast, no idelalisib-related infections were reported in the dog. Idelalisib-related toxicities in humans are largely considered to be on-target, based on the similarity to observations in genetically engineered mice with inactive PI3Kδ and in humans with loss-of-function mutations. 32 -34
AMG2519493 at ≥ 8 mg/kg was not well tolerated and was associated with greater severity of immune system and digestive tract toxicity, as well as the emergence of secondary opportunistic infections. Systemic exposure at ≥8 mg/kg exceeded the IC50 for human PI3Kδ for the entire 24-hour dosing internal and approached (8 mg/kg) or exceeded (100 and 200 mg/kg) the IC50 of human PI3Kγ. Phosphatidylinositol 3-kinase γ is primarily involved in the innate immune system, including roles in chemotaxis, and the recruitment and activation of myeloid cells in tumors. 2 Although the effectors and biological responses associated with PI3K isoforms are mostly unique, a complex interplay of effector functions has been associated with activation of multiple PI3K isoforms under physiologic conditions. For example, PI3Kγ and δ activation has been associated with neutrophil respiratory burst, 35 and B-cell development and maintenance. 36 Genetically engineered mice lacking PI3Kγ and δ activity are reported to have a defect in thymocyte development and markedly reduced peripheral T cell numbers with compensatory homeostatic expansion of the T cell population, presumably including autoreactive T cell clones that drive widespread tissue inflammation mediated by Th2 cytokines. 37 Therefore, inhibition of PI3Kγ may have contributed to the increased toxicity at higher dose levels; although the authors acknowledge that this is speculative without supporting data.
Although systemic exposure approached the IC50 of PI3Kα or β at 200 mg/kg, there was no evidence of inhibition of these isoforms. Phosphatidylinositol 3-kinase α is a key effector molecule in insulin and insulin-like growth factor (IGF-1) signaling and inactivation of p110α in mice leads to insulin resistance and glucose intolerance. 2,38,39 Moreover, administration of PI3Kα inhibitors in the clinic for the treatment of cancer is frequently associated with insulin resistance, hyperglycemia, and hyperinsulinemia. 40 Although animals at the highest dose in the 3-month toxicology study were often in poor health, there was no evidence of dysfunctional glucose homeostasis, such as elevated glucose levels. Although all class 1 PI3K isoforms are expressed in platelets, the kinase activity of PI3Kβ isoform plays a critical role in regulating platelet responses initiated by several platelet receptors, particularly at high shear stress during arterial thrombosis. 41 In the toxicology studies described here, there was no evidence of an effect on platelets, such as thrombosis, embolism, or infarcts.
Opportunistic pathogens are defined as infectious agents that are usually not clinically apparent in the immunocompetent host but are capable of causing disease in the immunocompromised host. 42 Based on the multiple roles of the PI3K isoforms in regulating the immune system and the lack of tolerability at high-dose levels in the toxicology studies, it was not surprising to observe opportunistic infection in animals at nontolerated dose levels. The mix of bacterial, viral, fungal, and parasitic infections suggests broad-based AMG2519493-related modulation of the humoral and cell-mediated immune responses. 42
Multiple bacterial infections were identified in this study, including Helicobacter sp. in the stomach, heavy overgrowth of Campylobacter spp. in fecal cultures, and a retropharyngeal bacterial abscess. As spontaneous infectious agents in immunocompetent animals, Campylobacter spp. are commonly implicated in outbreaks of diarrhea and inflammation in cynomolgus macaque colonies, 42,43 and they likely contributed to the GI clinical signs and colon inflammation in this study. Natural infection with Helicobacter spp. in the cynomolgus macaque stomach is generally asymptomatic but can be associated with epithelial hyperplasia, lymphoplasmacytic, and neutrophilic infiltrates, erosions in the stomach, and argyrophilic bacteria, 19 changes which were observed in both controls and treated animals in the 1-month toxicology study. The presence of Helicobacter-related changes in the control animals confounded the interpretation in this study; however, the increased incidence and/or severity in animals administered AMG2519493, especially in the context of other study findings, is notable and may be secondary to AMG2519493-related immunomodulation.
Adenovirus and LCV have high natural seroprevalence in macaques, but infection is clinically silent in immunocompetent adult animals. 21 In immunosuppressed animals, adenovirus infection can involve the liver, GI tract, lung, and pancreas. 20,44 Although the intestinal epithelium associated with adenoviral inclusions was slightly disorganized (eg, loss of basal polarity) in this study, it is unlikely that this change contributed to the GI clinical signs because it was subtle and focal. Lymphocryptovirus, a member of the Gammaherpesvirinae subfamily and closely related to human Epstein-Barr virus, is associated with lymphoma and esophageal epithelial proliferation (oral hairy leukoplakia) in immunosuppressed macaques. 45 Although the LCV-mediated esophageal change was observed in these studies, it appears to have been clinically asymptomatic.
Primary fungal infections in immunocompetent macaques are rare, but several fungal species can infect immunosuppressed animals. 42 One animal in the 3-month toxicology study had widespread fungal invasion of the upper digestive tract, including the tongue, tonsil, and esophagus. The morphologic features of the intralesional fungi were consistent with Candida albicans, a saprophytic agent that colonizes the mucosal surfaces shortly after birth and is the causative agent of thrush in multiple species. 21
Parasitic opportunistic pathogens observed included Cryptosporidium sp., B coli, and Giardia sp. Seroprevalence to Cryptosporidium sp. in laboratory-housed cynomolgus monkeys is reportedly high by 2 to 3 years of age, and the organism is resilient in the environment, which contributes to cross-contamination. 21 In immunocompromised animals, the organism can spread to the liver, pancreas, and respiratory tract, causing mild inflammation. The presence of low numbers of B coli in the cecum and colon of cynomolgus macaques is not an uncommon observation in toxicology studies. However, overgrowth and/or tissue invasion, as observed in these studies, is less common and a high incidence of overgrowth is consistent with immunosuppression. Despite the absence of significant tissue response, the presence of these agents is consistent with the immunocompromised state and they may have contributed to the GI clinical signs, including abnormal feces and body weight loss.
In conclusion, cynomolgus monkey toxicology studies with AMG2519493, a highly selective PI3Kδ-specific inhibitor, identified the immune system and digestive tract as primary targets of AMG2519493-related pharmacologic activity and toxicity. The AMG2519493-related changes in the cynomolgus monkey are consistent with the known biological roles of PI3Kδ in the immune system, the loss-of-function phenotype in humans, and with PI3Kδ inhibitors in the clinic, indicating that the cynomolgus monkey is suitable model for future investigation of PI3Kδ inhibitors. These observations demonstrate the role of PI3Kδ in regulation of the immune system and of host resistance to opportunistic infections of the digestive tract.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320966238 - Phosphatidylinositol 3-Kinase δ Inhibitor-Induced Immunomodulation and Secondary Opportunistic Infection in the Cynomolgus Monkey (Macaca fascicularis)
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320966238 for Phosphatidylinositol 3-Kinase δ Inhibitor-Induced Immunomodulation and Secondary Opportunistic Infection in the Cynomolgus Monkey (Macaca fascicularis) by Jonathan A. Werner, Katsu Ishida, John Wisler, Christine Karbowski, Jackson Kalanzi, Jeanine Bussiere and Thomas M. Monticello in Toxicologic Pathology
Footnotes
Acknowledgments
The authors thank the following toxicology study personnel: Mingyi Trimble (study director, 1-month study), Shana Dalton (study director, 3-month study), Laura Healy (study anatomic pathologist, 1-month study), and Kenneth Shafer (study pathologist, 3-month study). In addition, the authors thank the following Amgen scientists: Sarah Sun (special stains of histologic sections), and Oliver Homann (genetic sequences for PI3K isoforms).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are or were employees of and own stock in Amgen Inc.
Funding
The author(s) received financial support from Amgen Inc for the research, authorship, and publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
