Abstract
Good Laboratory Practices (GLP) is a well-established global system that encompass a set of principles or a framework for defining how laboratory studies are planned, performed, monitored, recorded, reported, and stored for future reference. It is important that compliance with the principles of GLP continues to be maintained. Coronavirus disease 2019 (COVID-19) pandemic lockdowns in various countries, including India, have been sudden and over an extended duration. Although every GLP laboratory has Standard Operating Procedure for disaster management, the sudden lockdown due to COVID-19 created specific emergency procedures related to this situation such as travel bans, safe distancing, and work from home notifications. Good Laboratory Practice compliances in the context of animal experimentation during and post lockdown period need effective managerial responses that are not just flexible and innovative but can ensure they are well-calibrated to the challenges of business continuity and maintenance of health directives. On-the-ground realities suggest there may still be practical challenges to compliance, and guidelines may not always be complied with. This article discusses the issues that may be encountered due to COVID-19 that could potentially impact the GLP status of a study and suggests ways to manage them so as to minimize or prevent infection with COVID-19. We propose an MMM (Man, Material, and Medium) strategy to ensure compliance with health directives and guidelines that will help staff to keep themselves and others safe in the workplace while endeavoring to comply with GLP requirements.
Introduction
Managing the response to the coronavirus disease 2019 (COVID-19) pandemic requires a dynamic understanding of the situation on the ground. Resulting lockdowns that have been enforced worldwide have created a number of challenges for businesses in general. Essentially these challenges revolve around factors such as shrinking markets, fractured supply chains, credit squeezes, and staff shortages, among others.
In short, business continuity is a worry that keeps most entrepreneurs and business leaders burning the midnight oil. Individual business sectors also face additional unique challenges as a consequence of standard practices and regulations that are specific to them. The research laboratories face unique challenges, specifically those that follow Good Laboratory Practice (GLP)–related regulatory compliance aspects during these periods of lockdown. 1
Good Laboratory Practice is a well-established global system that encompasses a set of principles and a framework for defining how laboratory (those laboratories that meant to support the safety of certain products) studies are planned, performed, monitored, recorded, reported, and archived for future reference. The main goal of GLP is to encourage scientists to organize and perform their studies in an environment that will ensure quality, validity, traceability, and integrity of test data and obtain results that are reliable, repeatable, auditable, and recognized by the global scientific and health care community.
Good Laboratory Practice compliance is underpinned by the need for a lot of interactive processes involving laboratory personnel, data records, and study test items. When this is viewed in the light of overarching aim of all the health directives and guidelines during and post-lockdowns as enabling the minimization or prevention infection by COVID-19, the need for innovation to manage the practical problems become very important. 1
One of such innovative approaches is the Man, Material, and Medium (MMM) strategy, which is based on 3 critical entities that are at the center of all potential spread of infection and noncompliances. In nutshell, there will be interaction between employees (man) while performing experiments and exchanging documents (material) at work place including sharing of instruments, glassware, and work tables (medium). All ground realities will in some way or the other involve interactions between any or all of the
This article attempts to identify challenges faced by laboratories conducting preclinical studies toward regulatory compliances including GLP, during lockdown periods, and suggest some measures by which they can be managed well, based on experiences from our laboratory. We also make a case for adoption of the MMM strategy. If the process of interactions of Man, Material, and Medium are understood and controlled, the probability of spreading infection at a test facility can be reduced to a minimum during situations such as those arising from COVID-19 lockdowns.
Good Laboratory Practice Compliance During and After Lockdowns
Why GLP Compliance for Preclinical Laboratories Can Be a Challenge?
Preclinical research laboratories seek and maintain GLP certification in order to safeguard the quality and the integrity of the preclinical studies that they undertake for safety testing of drugs, either for in-house purpose or for third parties. The purpose of GLP compliance is the mutual acceptance of data among countries. GLP compliance is therefore critical not just for business continuity but also for growth of a preclinical laboratory. It assures that the preclinical laboratory study has utilized standard procedures, equipment, and methods to conduct and report an experiment, that the data submitted to the regulatory authorities are in conjunction to what was found in the actual study, and that safeguards against any fraudulent activities. Finally, it promotes international acceptance of test results, thereby promoting international business.
During the period of lockdown and as per government advisories such as safe-distancing and personal hygiene being mandated as a response to a pandemic such as COVID-19, most of the GLP activities that are required to provide assurances on a consistent basis suffer a breakdown. The reasons for such a breakdown may be different for different activities but usually they involve Nonavailability of personnel Supply-chain disruptions Working capital flow interruptions Delay in maintenance of equipment and instruments
Viktor E Frankl an Austrian neurologist and psychiatrist in his book Man’s Search for Meaning said, “When we no longer can change a situation, we are challenged to change ourselves.” 2 COVID-19 demonstrates this very well.
Managing GLP Compliance During Lockdowns
Every organization has its own emergency preparedness plans, where safety of the employees is central; however, what works for one organization may not work for another. Therefore, the Test Facility Management, evaluating current lockdown stipulations in their respective area, as well as anticipated post-lockdown scenarios, needs to flexibly adopt and adapt several innovative approaches to ensure that GLP compliance is managed effectively. Toward this, they need to use their best judgment and make all possible efforts at maintaining fundamental aspects as mentioned in Handbook of GLP (2009)
3,4
and such as:
Effective management of each the above-listed fundamental aspects of GLP compliance during periods of business uncertainties calls for an adaptive approach, such as the one that is depicted in Figure 1 and outlined below.
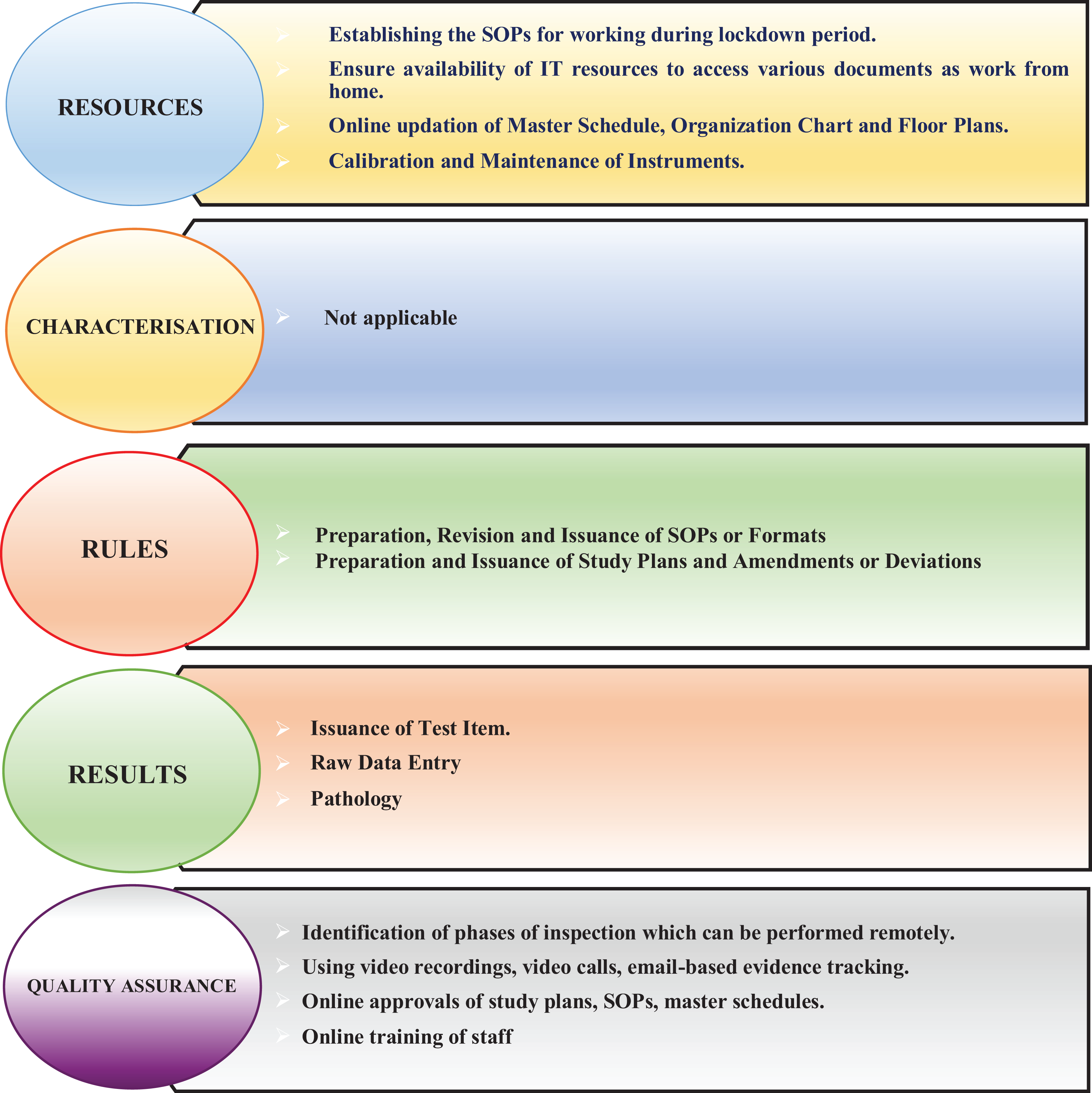
Fundamental aspects of GLP compliance—an adaptive approach.
Managing Resources
Test Facility Management needs to focus on key or vital resource aspects such as Information Technology, Organization Charts, Master Schedules, Floor Plans, Personnel Records, Instruments, study directors, and study personnel. Revising the SOP of disaster management to include changes in the activities, if any, that are being affected due to lockdown should be considered. 5 The following illustrates the nature of adaptations that will be required.
For those instruments where periodic maintenance is due, the responsible person shall communicate with the service engineer and check if the maintenance can be performed remotely using video call. The records of such communication shall be maintained, and online certificate for the maintenance service completion shall be sought and procured.
For calibration of instruments due during lockdown period, the performance can be checked by running quality or standard samples or weights and generate calibration report documenting that instrument is performing satisfactorily or up to the standard. 7
In case of emergency or breakdown, service engineer shall be requested to visit facility after getting necessary permissions.
In case it is not possible to do any of the above steps, then a note shall be generated by the person responsible for calibration and maintenance stating that the maintenance or calibration will be performed after lockdown period.
All such documents generated shall be communicated to the Quality Assurance Unit and Test Facility Management for the approval. Quality Assurance Unit can consider such documents as a part of facility inspection.
Experiments in planning stage may be delayed; study directors should generate an amendment for the changes, if any, and communicate the same to Quality Assurance Unit and the sponsor. The reviews and approvals for the changes can be performed electronically. It should be ensured that the documents shared electronically are password protected and accessed by authorized persons only. Overall, electronic security measures need to be in place (Organization for Economic Cooperation and Development, Document No 17, Guidance Document Part 11). 8,9
Experiments which are in live phases need proper planning considering lockdown situation. Study Directors shall revisit the schedule on daily basis, and in consultation with Quality Assurance Unit and Test Facility Management, they shall prioritize the critical phases. If Study Director cannot be present physically, then the person in-charge should coordinate with the staff physically present in the facility and train them appropriately to complete the respective phases. Those phases that are not having severe impact on the outcomes may be delayed after issuing the amendment or deviation for the same.
Experiments that are in terminal stages, including activities such as blood collection, urinalysis, clinical chemistry analysis, necropsy, gross pathology, and histopathology, need more staff (Such activities may vary with type of experiment). All such activities are very important from GLP perspective toward arriving at final results. The Study Director needs to plan in advance for arrangement of personnel following lockdown rules laid down by local authorities. Considering the possible impact, the activities can either be segregated or scheduled for different days to complete; or they can be conducted following social distancing norms. The data can be recorded using phone or photographs or by other possible ways by using the systems which are tested previously and compliant with GLP.
Overall during lockdown period, Study Director shall ensure that the work from home strategies implemented are in compliance with GLP toward technical conduct of the study, including interpretation, analysis, documentation, and reporting of the results. 10
Study Personnel must strictly follow health precautions, including use of appropriate clothing and Personal Protective Equipment to keep themselves healthy, to avoid cross contamination, and to report to their immediate supervisor or administration department about health or medical condition, if any.
Managing Rules
The SOPs and Study plans are the standardized, consistent, prescriptive documents that reduce test-to-test variability, and these are the governing rules that provide the framework of assurance. Therefore, it is vital that these rules pertaining to preparation and revision of SOPs, their issuance, and the study plans are managed effectively during lockdowns.
As per GLP, current SOPs relevant to the activities should be available in the respective functional area; however, due to sudden lockdown, availability of hard copies of SOP may be difficult to obtain. Electronic versions can be made available, with appropriate controls of distribution being put in place. A few key points are discussed further:
Managing Results Aspects
It is very important to ensure integrity of test items and reference items and to keep records of how results are collected, reported, and archived. Hence, the Study Director must be vigilant during all the phases of the studies and manage the activities that are conducted during lockdown period.
The activities such as raw data entry, Quality Control, and statistical analysis can be completed remotely as part of work from home. It is the responsibility of the Study Director to monitor these activities.
Managing the Quality Assurance Process
Many organizations follow processes for approving paper documents with “wet ink” signatures; however, due to the lockdown and remote working, it may be difficult to maintain paper-based processes of printing the documents, physically reviewing them in the office, and signing using “wet ink” signatures. The adequacy and effectiveness of all measures that need to be in place for ensuring GLP compliance necessitate a sound Quality Assurance process. 6,11
Managing such a Quality Assurance process during lockdowns therefore becomes vital. Some of these measures include the following:
Ensure that Quality Assurance department personnel are designated as essential workers so that they are able to get the necessary civic permissions to travel.
Quality Assurance Unit head shall identify phases of study process and facility-based inspections that can be done remotely. For conducting the inspection remotely, use of photographs, videos, and/or video calls can be considered as objective evidence. Respective study directors or the auditee shall arrange to share proof of evidence to Quality Assurance Unit and Test Facility Management regarding the inspection.
Based on the objective evidences received, Quality Assurance Unit shall prepare the inspection report and share with auditee and Test Facility Management for acknowledgment. After receiving the answers for comments from auditee, the report can be submitted for approval from Test Facility Management. 13
Review procedures of Study plans, raw data, reports, master schedule, organization chart, and any other documents shall be performed using electronic media.
The Quality Assurance Unit shall ensure that methods of document distribution and approval are defined so that the risk of error due to misunderstanding of what is being reviewed/approved will be minimized.
The Quality Assurance Unit shall also ensure that a signature performed remotely should be equivalent to the “wet ink” signature of the signatory and the security of the electronic signature, that is, so that it can only be applied by the “owner” of that signature. 6,11
Ensure that all required associated data is available to a remote reviewer that would have been available to them if they were performing the review at an office before lockdown.
All the records of the online activities shall be maintained in online archives.
Support of Regulators
How Can Regulators and Certifying Bodies Help?
As a result of the impact of COVID-19 on staffing, restrictions on movement of people, and response to Government advice, there are a number of scenarios where an existing study may need to be amended, and new studies may be required to be planned on fast-track to support clinical trials. Although preclinical research laboratories attempt to ensure GLP compliance in the face of such varied challenges, they are getting a real boost from regulators, and certifying bodies and guidance are now available for different scenarios which will certainly help.
Following information is available on US Food and Drug Administration (USFDA) and MHRA websites may help them in the following ways.
✓ Various guidance documents are available for sponsors, sites, and researchers who need to make changes to an existing study to manage the impact of COVID-19 on MHRA websites. 6,10 -12
✓ Regulatory authorities have initiated active communication with GLP laboratories that are coming up for surveillance reviews and published guidance documents for office-based inspections. 14
✓ For some organizations finding difficulties in following processes for approving paper documents with “wet ink” signatures, the guidance published by MHRA will enable organizations to consider alternative methods while maintaining basic control of documents.
✓ Various regulatory authorities such as MHRA, USFDA, South African National Accreditation System, 13 a Society of Quality Assurance, 15 National GLP Compliance Monitoring Authority, India, 14 have identified the challenges of remote auditing and offered strategies and published white papers, best practices for overcoming them throughout each phase of the assessment process: planning, document review, process review, facility review, interviews, and closing meetings. Although many of them are related to good manufacturing practices, they will be useful for GLP compliance.
Sharing Best Practices and Preparing for the Future
Laboratories themselves should proactively share the best practices and seek advice from each other on any specific activity toward maintaining the GLP compliance. Although it is the responsibility of Test Facility Management to implement possible measures to comply with norms of GLP such as possible audit trail, reconstruction of activity, each and every individual should act with responsibility to mold themselves to such situations, becoming multitaskers and complying with GLP.
With regard to animal experimentation and GLP, a lockdown period is a great source of useful lessons that can help GLP laboratories to prepare for future. The need to learn, adapt, and adopt so many things with humility that the circumstance demands can be beneficial in various ways, such as critical evaluation of how effectively time is managed, assessing the logic behind prioritizing the various work elements and identifying critical phases for maintaining the integrity of a study, among others. These can help to prepare for a future that may mandate alternatives for animal use. alternatives for animal use.
Man–Material–Medium Strategy
While the above discussion focused on managing the fundamental aspects of GLP compliance, it is also equally important that efforts to minimize the risks of being infected are also managed successfully. The MMM Strategy is one that can be impactful in this context.
Man–Material–Medium Strategy tries to explain the possible ground-reality situations inside the test facility where there is possibility of spread of the infection. Various example interactions given in the Table 1 will help to understand situations of possible means of transmission of infection. If the process of such interactions is rule-based and environmentally controlled, the probability of noncompliance with all directives and guidelines for prevention of infection at test facility can be reduced to minimum.
Illustrative Instances for Man Material Medium Interactions.

Abbreviation: MMM, Man–Material–Medium.
It is recommended that if all the staff identify and list all such interactions between MMM in their respective working areas and then follow some simple environmental controls (ie, physical boundary controls) as well as rules (SOPs), this will ensure that safe distancing and personal hygiene are always maintained, and there will be zero instances of noncompliance and thereby keeping oneself and others safe from infection.
Some suggestions for such environmental controls and rules are listed below. These can be further adapted and implemented as the final analysis of the MMM Strategy may dictate.
Suggestion 1: Mini Quarantine Stations (MQS)
These are especially useful where the man–man, man–material, and man–medium interactions are expected to be very difficult to practically control in a normal situation. The following schematics explain the logic using illustrative examples.
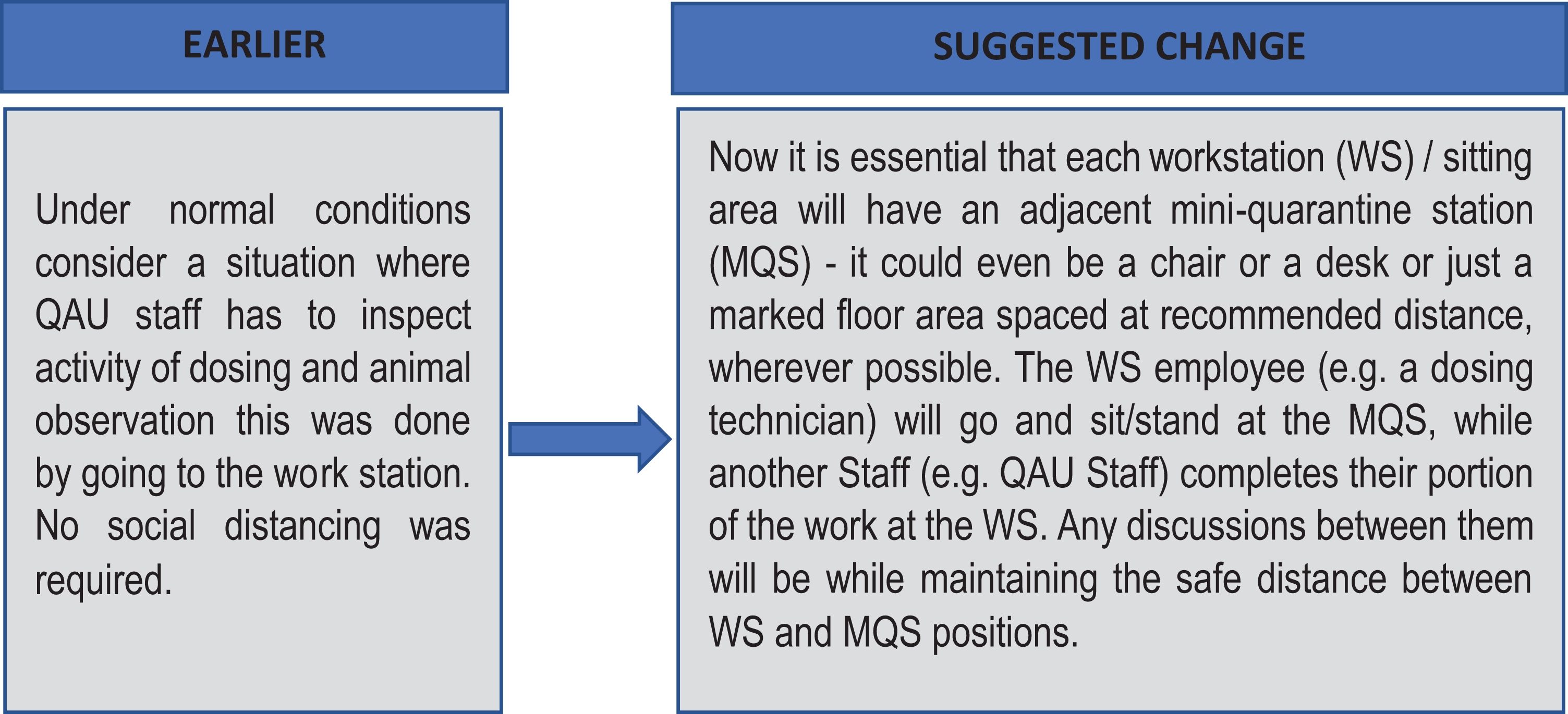
Mini Quarantine Station (MQS) (Figure 2) may be designed for various situations and departmental work where such unplanned interactions are possible. MQS may be just a clearly ear-marked floor space; they could be a chair or a desk that is clearly identified as MQS and disinfected regularly and immediately before and after use. MQS could also be a separate room depending on the need (material–medium or medium–medium instances perhaps).
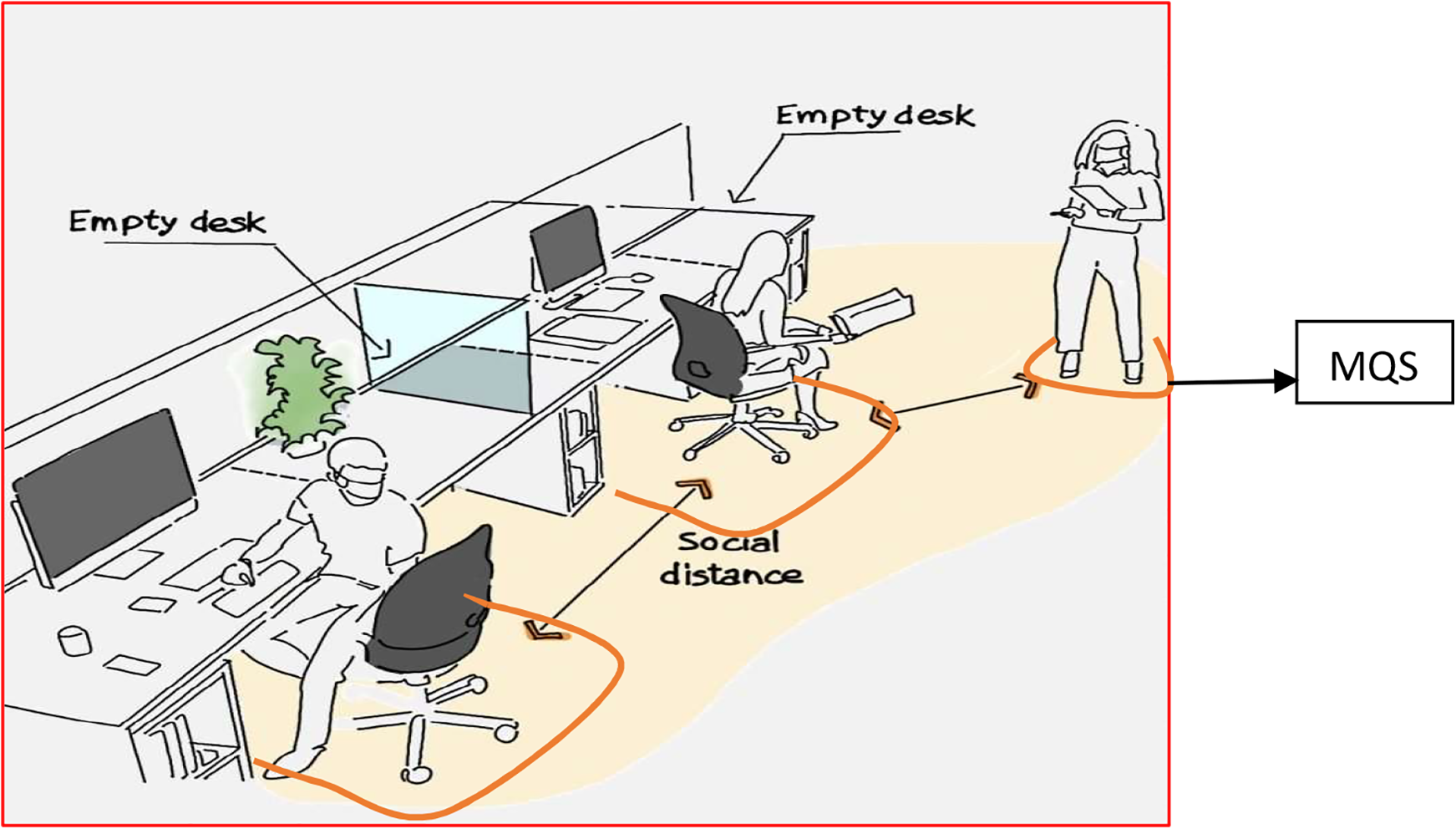
Mini quarantine station.
The general rule should be that whenever such interactions need to take place, the use of the nearest MQS should be considered to ensure that safe distancing and personal hygiene conditions can be complied with easily.
Suggestion 2: Personal Protection Rules
It is very important that a new culture of using personal protection measures by each employee must be followed compulsorily; some of these rules will be known as personal protection rules (PPR). These include:
Those who are traveling from another town, city, or village and would like to enter the office must first get a health certificate from general physician about their fitness considering the COVID-19 infection. It is responsibility of individual to ensure the requirement of quarantine after traveling as per government rules.
Staff traveling from home to the facility can use an extra apron or anything like wind breaker or rain coat. At entrance of office, the outside apron/coat can be soaked in the detergent water or spread with disinfectant and left for drying. The outside shoes shall be disinfected by spraying with disinfectant or shall be left outside at designated places and facility footwear can be used. Alternatively, at the entrance before entering office area, foot wash can be used.
Vehicles entering the facility, if any, must be disinfected before parking them inside the premises.
At entrance, temperature and oxygen levels of each employee shall be checked, and security persons shall be authorized to restrict the entry into the facility if anyone has high temperature (above 37 °C degrees).
Floor space markings shall be used while interacting with each other. Each employee will assert their right not to respond to any question or remark by another colleague if a safe distance is not maintained between them. Everyone can instruct accordingly to others.
Lunch and Break periods will be staggered. It is suggested that everyone who want to use the canteen/cafeteria area should observe and wait until the time that safe distancing will be maintained. It is responsibility of everyone to avoid taking a break at same time.
All the staff shall always be with masks and disposable gloves while working except during lunch and break periods. Staff shall use the hand sanitizers as and when required.
Overall, it is the responsibility of every individual to follow the rules, SOPs, and guidelines and keep themselves away from infection.
It is also recommended that all staff shall download any such app which helps them check their safety status, and records of the same shall be maintained.
Personal protection rule will help ensure personal hygiene and safe distancing is maintained and thereby keeping everyone healthy while performing the routine work. The use of MQS and PPRs are especially useful where the man–man, man–material, and man–medium interactions are expected to be very difficult to practically control in a normal situation.
It is possible that similar situations may arise in future where the laboratories need to be ready to handle it smoothly. Hope this article will help you to some extent to maintain GLP compliance at your laboratory.
Conclusions
Managing GLP compliance during challenging times such as COVID-19 lockdowns certainly poses challenges, but with the right approach to identifying and understanding the challenges, adequate commitment from top management to devote the necessary resources, and the total involvement of each staff member to adapt and adopt to the changes in operational procedures, it is entirely possible for GLP laboratories to ensure there are no noncompliances. Innovations such as the MMM Strategy for preventing infections will aid and benefit the laboratories to easily turn around a challenging problem.
Footnotes
Author Contributions
Wangikar and Giridhar contributed to conception and design; acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript. Prakash contributed to design and contributed to interpretation. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
