Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of polyenes, which are reported to function in cosmetics primarily as film formers and viscosity increasing agents. The Panel reviewed relevant data related to these ingredients, not inggaps in the available safety data for some of the polyenes in this safety assessment. The data available for many of the ingredients are sufficient and can be extrapolated to support the safety of the entire group because of the similarities in the chemical structures, chemical properties, use concentrations, and reported functions across the group. The Panel concluded that polyenes were safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Keywords
Introduction
The 26 ingredients listed below are simple polyolefins that are the polymerization products of vinyl-type monomers and are reported to primarily function as film formers and/or viscosity increasing agents—nonaqueous in cosmetic products. Although the molecular weights of the polyenes reviewed in this report vary over a wide range, structurally, these ingredients have many similarities, including (1) each is the product of the same vinyl-type polymerization methodologies; (2) each is manufactured from very similar starting materials (ie, olefin/alkene monomers); (3) each has similar, simple hydrocarbon structures without functional groups other than alkanes or alkenes; and (4) many are of sufficient molecular size to significantly decrease the chance for dermal penetration.
Polybutene (published in 1982), Polyethylene (published in 2007), Polyisobutene (published in 2008), and Hydrogenated Polyisobutene (published in 2008) have previously been reviewed by the Expert Panel for Cosmetic Ingredient Safety (Panel), which concluded that these ingredients are safe as cosmetic ingredients in the practices of use and concentration as described in each safety assessment. 1 -4 Information from these safety assessments are summarized in appropriate sections of this report; the complete reports are available on the Cosmetic Ingredient Review (CIR) website (http://www.cir-safety.org/ingredients).
Some chemical and toxicological data on Hydrogenated Polydecene and Polybutene included in this safety assessment were obtained from robust summaries of data submitted to the European Chemical Agency (ECHA) by companies as part of the REACH chemical registration process. These data summaries are available on the ECHA website. 5,6 The ECHA data summaries include information on analogs (eg, diisobutylene, di-n-butene, tributene, triisobutylene, and tetrabutene for Polybutene; hydrogenated decene dimer and trimer for Hydrogenated Polydecene; and hydrogenated dodecene trimer for Hydrogenated Polydodecene) for read-across purposes. Where deemed appropriate, information from the summaries has been included in this report.
Chemistry
The definitions and CAS registry numbers, where available, of the polyene ingredients are presented in Table 1.
Definitions, Idealized Structures, and Functions of the Ingredients in this Safety Assessment.a , 48
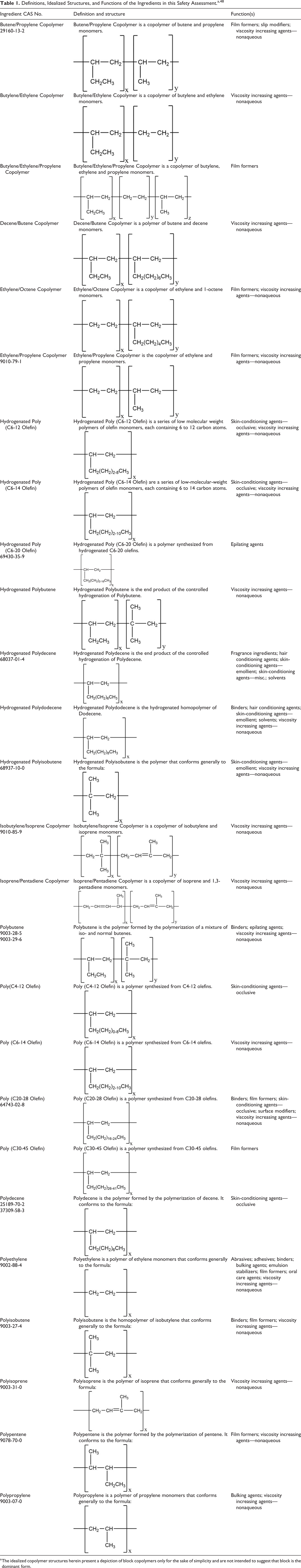
a The idealized copolymer structures herein present a depiction of block copolymers only for the sake of simplicity and are not intended to suggest that block is the dominant form.
Polyenes are the polymerization products of vinyl-type monomers (ie, alkenes or olefins; eg, Figure 1). These polyolefins are either homopolymers (eg, Polybutene) or vinyl-type copolymers of 2 or more monomers (eg, butene/propene copolymers). The term “vinyl-type copolymers” means that all the monomers utilized to make these polymer ingredients have in common an ethylene unit whose pi electrons are directly involved in the polymerization process. Typically, a catalyst is utilized to initiate the polymerization. 7 There are a large number of relevant initiating catalysts, ranging from ultraviolet (UV) light to Ziegler-Natta-type catalysts, which can result in a range of varied characteristics, such as crystallinity (and resultant hardness). The synthesis of these ingredients is typically conducted in one or more organic solvents in the presence of one or more of these catalysts.
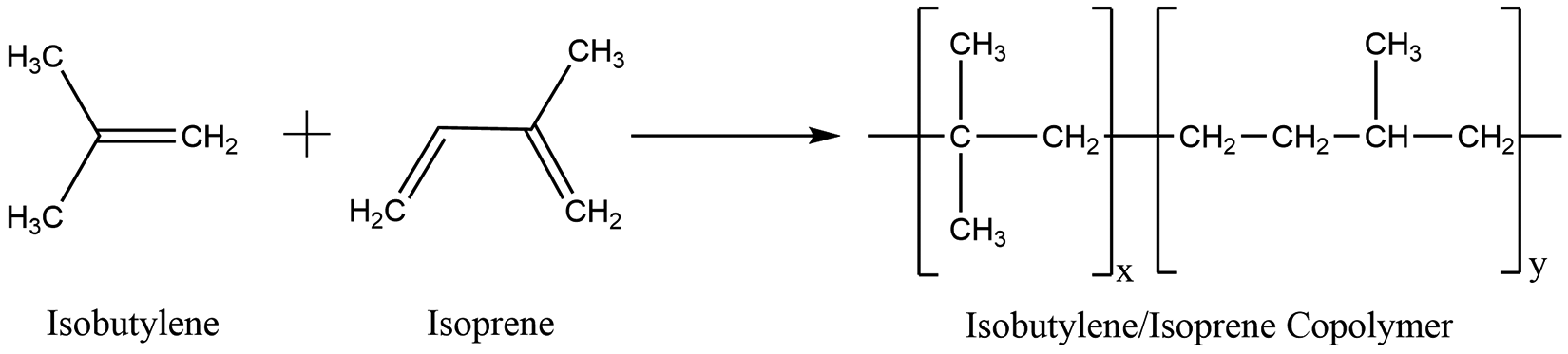
An example of polyene synthesis (Isobutylene/Isoprene Copolymer).
For example, formation of Polyisoprene occurs by reacting the isoprene monomer in the presence of catalyst in a hydrocarbon solution, usually hexane. 8 The process is stopped with the addition of a terminating reagent. The in situ stabilization of the polymer is often enhanced with the addition of an antioxidant. Subsequent steps in the process include stripping of the solvent, water washing of the polymer to remove catalyst and reagent residues, and finally pressing and formation of a granular product (for nonliquid polyenes).
Chemical Properties
Table 2 summarizes available data on chemical properties, including information from the original Panel safety assessments of Polybutene, Polyethylene, Polyisobutene, and Hydrogenated Polyisobutene. Further chemical data on these previously reviewed ingredients can be found in those reports. 1 -3
Physical and Chemical Properties of Polyenes.
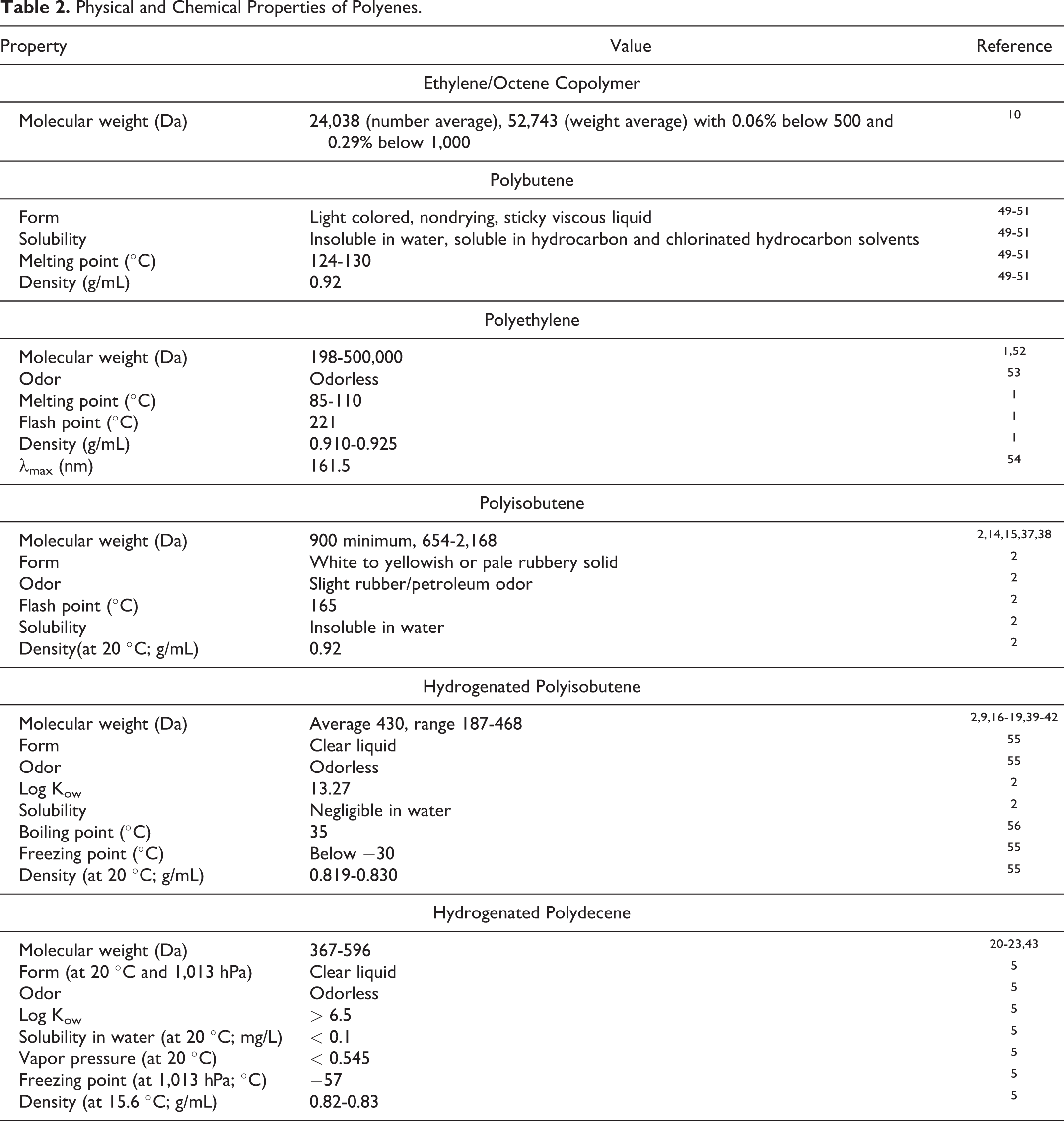
Many of these polyene ingredients are high-molecular-weight, large, inert polymers. The smaller, liquid ingredients in this group have a simple hydrocarbon structure, without functional groups other than alkanes or alkenes. These ingredients are completely insoluble in aqueous solutions or organic solvents but may be swellable in certain organic solvents.
Method of Manufacturing
Hydrogenated Polyisobutene
According to a supplier, Hydrogenated Polyisobutene is produced from the polymerization of isobutene, which is then hydrogenated, purified, and super refined before yielding the final product. 9
Composition and Impurities
Ethylene/Octene Copolymer
A supplier reported that a trade name mixture comprised in part of Ethylene/Octene Copolymer contains 14% to 16% Ethylene/Octene Copolymer and 84% to 86% C14-22 alkane. 10 Residual monomer levels are 2 ppm octene and 0 ppm ethylene. Ethylene oxide, 1,4-dioxane, and heavy metals were reported to be below the detection limit of 0.1 ppm. 11
A second trade name mixture was reported to contain 30% to 50% Ethylene/Octene Copolymer and ethylene/sodium acrylate copolymer, and 50% to 70% water. 10 The residual monomer levels were reported to be less than 165 ppm acrylic acid, less than 5 ppm ethylene, and less than 52 ppm octene. A heavy metals analysis reported arsenic was not detected (limited of detection, 27 ppb); however, lead and mercury levels were 22 ppb and 52 ppb, respectively (limits of detection for each were 5 ppb). 12
Ethylene/Propylene Copolymer
A redox titration of Ethylene/Propylene Copolymer measured 0.8 ppm of the starting material residue in the final product. 13
Polybutene
Impurities of Polybutene include isoparaffins, vinylidene and terminal vinyl structures, chloride, and sulfur-containing compounds. 3
Polyisobutene
A supplier reported that Polyisobutene does not contain detectable levels of residual solvents or monomers and has heavy metal specifications of lead < 10 ppm, arsenic < 2 ppm, and mercury < 1 ppm. 14,15
Hydrogenated Polyisobutene
A supplier reported that Hydrogenated Polyisobutene does not contain detectable levels of residual solvents or monomers and has heavy metal specifications of lead < 10 ppm, arsenic < 2 ppm, and mercury < 1 ppm. 16 -19 An anonymous source reported that Hydrogenated Polyisobutene may contain a maximum of 10 ppm n hexane as residual solvent. 9
Hydrogenated Polydecene
A supplier reported that Hydrogenated Polydecene does not contain residual solvents, has a residual monomer specification (decene) of < 10 ppm, and has heavy metal specifications of lead < 10 ppm, arsenic < 2 ppm, and mercury < 1 ppm. 20 -23
Use
Cosmetic
The safety of the cosmetic ingredients included in this safety assessment is evaluated based on the expected use in cosmetics. The Panel utilizes data received from the US Food and Drug Administration (FDA) and the cosmetics industry in determining the expected cosmetic use. The data received from the FDA are those it collects from manufacturers on the use of individual ingredients in cosmetics by cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP), and those from the cosmetic industry are submitted in response to a survey of the maximum reported use concentrations by category conducted by the Personal Care Products Council (Council).
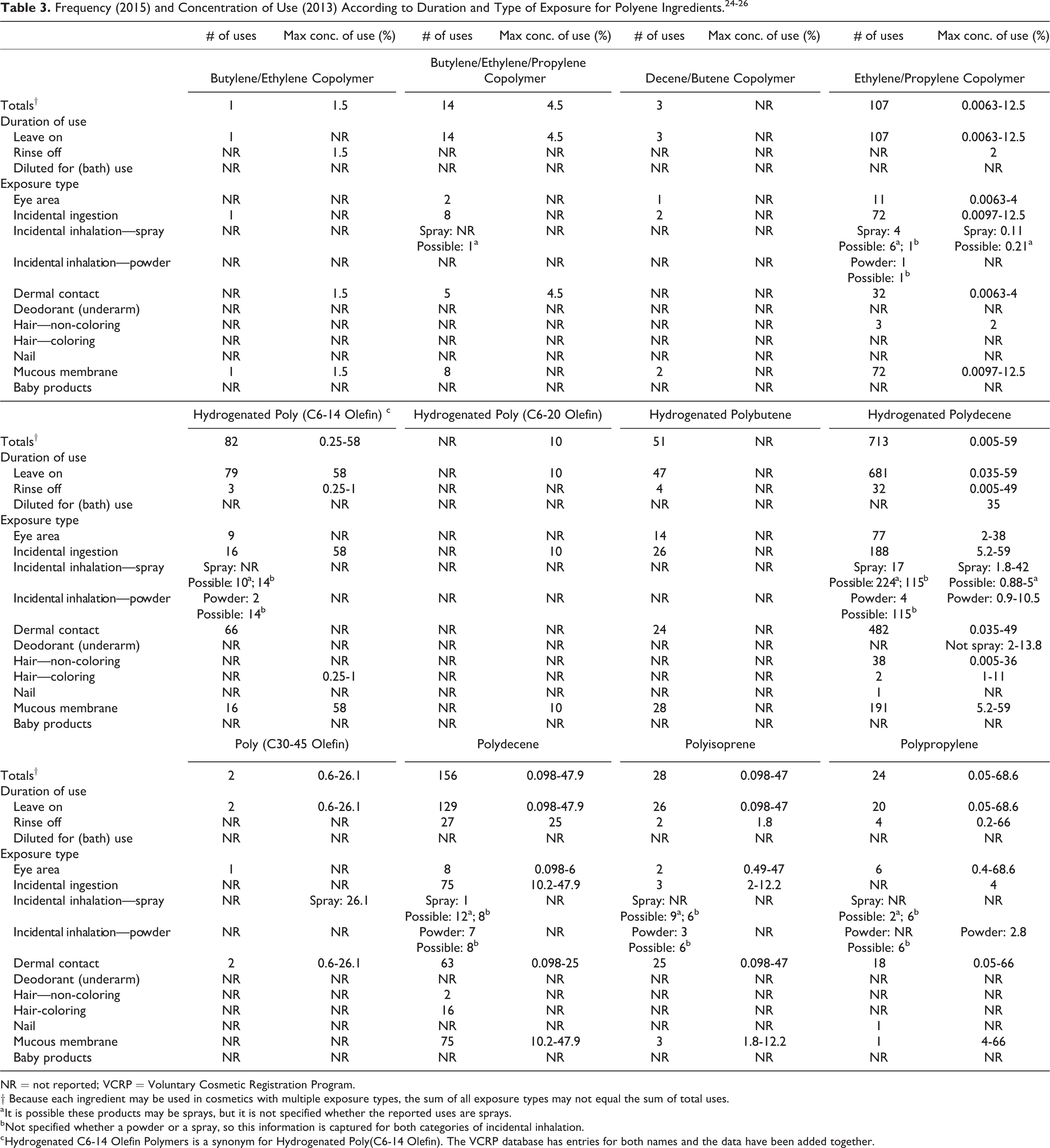
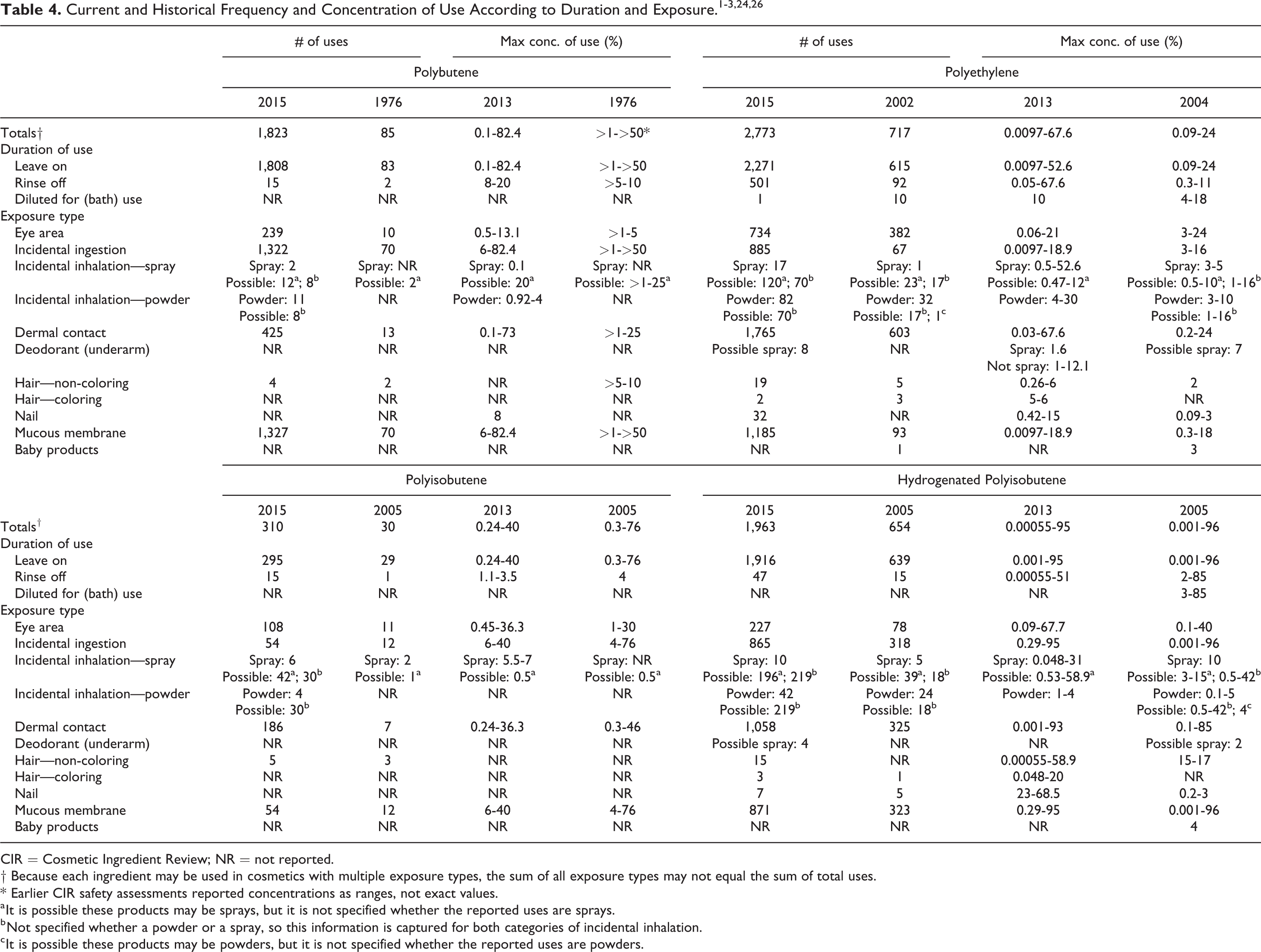
According to the 2015 VCRP data, Polyethylene is reported to be used in 2,773 formulations; the single category with the most reported uses was lipstick with 885 (Tables 3 and 4). 24 Hydrogenated Polyisobutene is reported to be used in 1,963 formulations; the single category with the most reported uses was lipstick with 865. Most of the other in-use ingredients are primarily used in leave-on products and lipsticks. The results of the concentration of use survey conducted in 2013 and 2014 by the Council indicate Hydrogenated Polyisobutene has the highest reported maximum concentration of use; it is used at up to 95% in lipsticks. 25,26
NR = not reported; VCRP = Voluntary Cosmetic Registration Program.
† Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
a It is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
b Not specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
c Hydrogenated C6-14 Olefin Polymers is a synonym for Hydrogenated Poly(C6-14 Olefin). The VCRP database has entries for both names and the data have been added together.
CIR = Cosmetic Ingredient Review; NR = not reported.
† Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
* Earlier CIR safety assessments reported concentrations as ranges, not exact values.
a It is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
b Not specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
c It is possible these products may be powders, but it is not specified whether the reported uses are powders.
Both historical and current use data for Polybutene, Polyethylene, Polyisobutene, and Hydrogenated Polyisobutene are provided in Table 4. Concentrations of use for Polybutene and Hydrogenated Polyisobutene have remained about the same, with the highest maximum use concentration of Hydrogenated Polyisobutene at 95% in lip products. The highest maximum use concentration for Polyethylene has increased from 24% (eye shadow) to 67.6% (skin cleansing agents), while the highest maximum use concentration for Polyisobutene has decreased from 76% to 40% (both concentrations in lip products). Uses for all 4 ingredients have increased by several fold since their original reviews. The ingredients not in use according to the VCRP data and industry survey are listed in Table 5.
In some cases, reports of uses were received from the VCRP, but concentration of use data was not provided. For example, Hydrogenated Polybutene is reported to be used in 51 formulations, but no use concentration data were reported. In other cases, no uses were reported in the VCRP, but concentration of use data were received from industry. Hydrogenated Poly (C6-20 Olefin) had no reported uses in the VCRP, but a use concentration in a lipstick was provided in the industry survey. Therefore, it should be presumed there is at least one use in every category for which a concentration is reported.
Some of these ingredients were reported to be used in pump and aerosol hair sprays, underarm deodorant sprays, face and neck sprays, body and handsprays, and aerosol suntan products and could be incidentally inhaled. For example, Hydrogenated Polyisobutene was reported to be used in face and neck sprays at a maximum concentration of 8.5%, and Polyethylene was reported to be used in aerosol deodorants at a maximum concentration of 1.6%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. 27 -30 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 28,29 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 29 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays. The polyene ingredients in this safety assessment currently are not restricted from use in any way under the rules governing cosmetic products in the European Union (EU). 31
Non-Cosmetic
Many of the polyene ingredients have been approved by the US FDA for use as indirect food additives and in medical devices. Additionally, Isobutylene/Isoprene Copolymer, Polyethylene, and Polyisobutene are approved direct food additives for chewing gum bases.
Polyethylene and Polypropylene are used as negative control materials for International Organization for Standardization (ISO 10993-6) international standard biological evaluation of medical devices. 32 Ultra-high-molecular-weight Polyethylene is the most used biomaterial for the articulating surface of total joint replacements. 33 Polyisobutene is used in transdermal drug delivery patches and patch adhesives. 34,35 Polyisoprene (trans-1,4) is widely used as a component of root canal filling material. 36 Table 6 lists regulated uses in foods and medical devices.
FDA-Approved Uses of Polyenes.
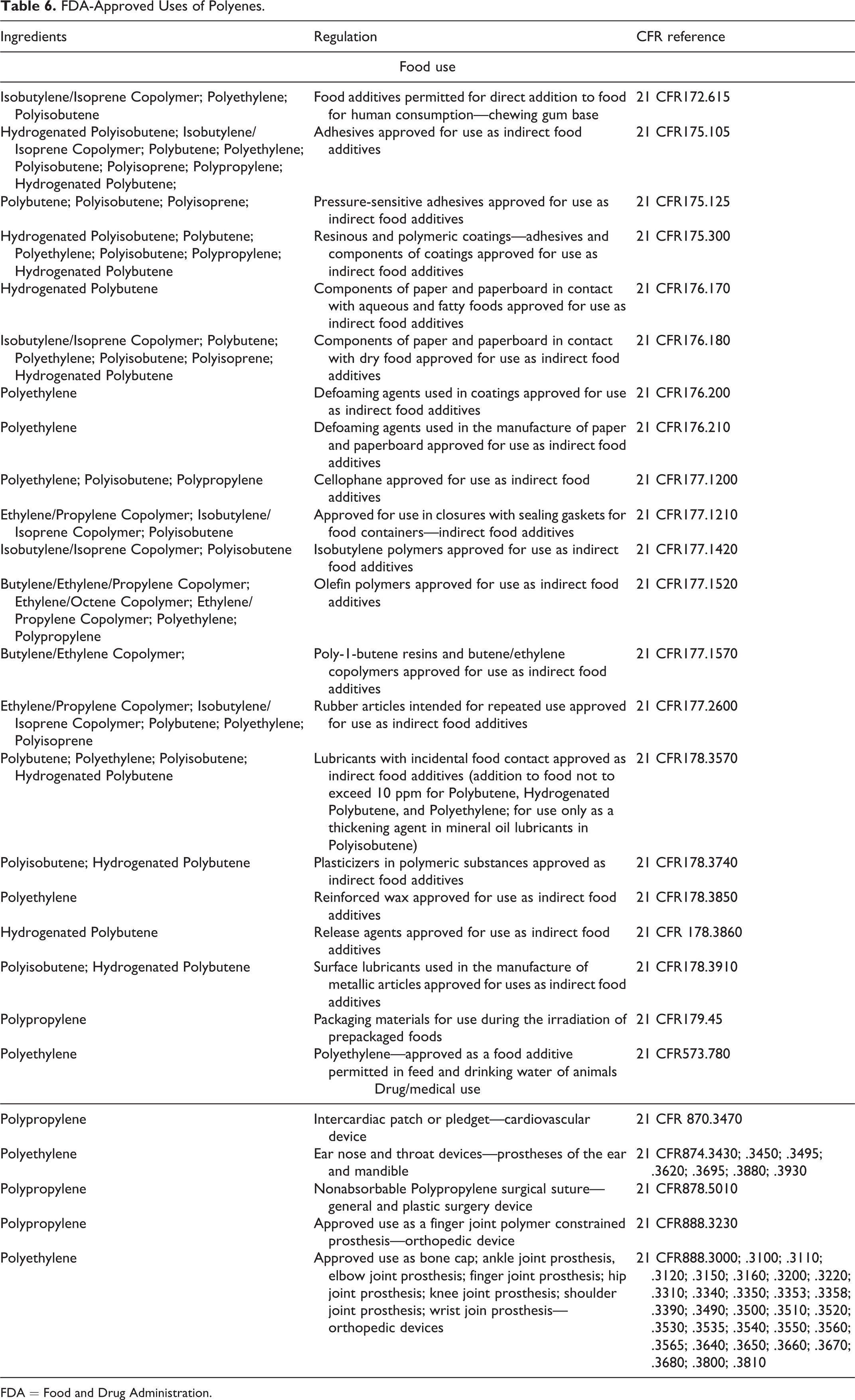
FDA = Food and Drug Administration.
Toxicokinetics
Although many of these polyene ingredients are high-molecular-weight, large, inert polymers, the smaller, liquid ingredients in this group each comprise simple hydrocarbon structure without functional groups other than alkanes or alkenes. Thus, dermal penetration is limited for the large and small polymers in this group. These ingredients are completely insoluble in aqueous solutions or organic solvents but may be swellable in certain organic solvents.
Absorption
Hydrogenated Polydecene
In one study, the absorption potential of undiluted Hydrogenated Polydecene was assessed in male Fischer rats. 5 Groups of 3 rats/time point received a single or daily (for 15 days) oral gavage dose of 30, 210, or 1,500 mg [3 H]-Hydrogenated Polydecene. Tissues and body fluids were sampled at 0.08, 0.25, 0.5, 1, 2, 4, 8, 24, 48, 72, 120, and/or 168 h post-dosing. With all 3 dose levels, very little of the administered dose was absorbed. What was absorbed was found in the liver, fat, lymph nodes, kidney, and spleen. The majority of the test compound was excreted in the feces (> 92%). Urinary excretion was low (< 1%), and very little of the dose was recovered in the bile (0.01%).
Biocompatibility
Polyethylene
Cellular and tissue responses to Polyethylene, determined as part of implant biocompatibility testing, include fibrous connective tissue build-up around the implant material that varies as a function of the physical form of the implant material. 1 Specific assays for osteoblast proliferation and collagen synthesis demonstrated a reduction as a function of exposure to Polyethylene particles that is inversely related to particle size. However, Polyethylene particles had a stimulatory effect on monocyte-derived macrophages, prolonging the survival of these cells in culture.
Toxicological Studies
Single Dose (Acute) Toxicity
Animal acute dose toxicity studies are presented in Table 7. 5,6,37 -43 In acute oral toxicity studies in rats, the LD50sof diisobutylene and triisobutylene were > 2,000 mg/kg/body weight (bw) each. The oral LD50 s of di-n-butene, tributene, and tetrabutene (containing 30% pentabutene) in rats were > 10,000 mg/kg each. The oral LD50 values for Ethylene/Octene Copolymer, undiluted Hydrogenated Polydecene and undiluted Hydrogenated Polydodecene, were > 5,000 mg/kg in rat studies. The LD50 of undiluted Polyisobutene was > 15,400 mg/kg in an oral rat study.
Acute Toxicity Studies in Animals.

Acute dermal studies of diisobutylene and Hydrogenated Polydodecene found the LD50 values > 2,000 mg/kg in rats. In rabbit studies, the dermal LD50 values for Ethylene/Octene Copolymer, hydrogenated decene dimer, Hydrogenated Polyisobutene, and Polyisobutene were > 5,000 mg/kg, > 3,000 mg/kg, >2,000 mg/kg, and > 25,000 mg/kg, respectively.
In acute inhalation studies, the LC50 of diisobutylene vapor in albino rats was > 4,185 ppm (19,171 mg/m3) after a 4-hour, single, whole-body exposure. The LC50 for an aerosol of Hydrogenated Polydecene was > 5.2 mg/L with a 4-hour exposure in rats. The LC50 for the dimer of hydrogenated decene was 1.17 mg/L in rats. In another acute inhalation study of the dimer of hydrogenated decene, the LC50 could not be determined in rats tested at 5 mg/L because 9/10 animals died within 3 days of administration of the test material. The LC50 for Hydrogenated Polydodecene was > 5.06 mg/L. The LC50 for 100% Hydrogenated Polyisobutene was > 5 mg/L. The oral, inhalation and dermal acute dose toxicity data that were presented in the original reviews of Polybutene, Polyethylene, and Hydrogenated Polyisobutene are summarized below and not in the tables.
Oral
Polybutene
When tested for acute oral toxicity in albino rats, concentrations of Polybutene ranging from 15% to 75% were relatively harmless (average molecular weight not specified). 3
Polyethylene
The LD50 for Polyethylene (average molecular weight of 450) in rats (201-223 g) was found to be > 2,000 mg/kg, and in Polyethylene with an average molecular weight of 655 Da, the LD50 was determined as >5.0 g/kg. 1
Hydrogenated Polyisobutene
No deaths in mice were observed in an acute oral toxicity test at a maximum dose of 89.608 g/kg of a Hydrogenated Polyisobutene mixture. 2 No deaths were observed in several oral toxicity rat studies of 5 g/kg Hydrogenated Polyisobutene; however, lethargy and wetness in the anogenital area after dosing was observed. The authors of these studies also concluded that the LD50 is greater than 5.0 g/kg bw. The average molecular weight was reported to be 900 Da in one of the studies.
Inhalation
Polybutene
Polybutene produced no abnormalities in rats during a 4-hour inhalation exposure up to concentrations of 18.5 mg/L. 3
Dermal
Polybutene
In acute dermal toxicity tests, Polybutene in formulations produced no abnormalities or irritation in rabbits. The LD50 of Polybutene in formulation was greater than 10.25 g/kg (average molecular weight not specified). 3
Repeated Dose Toxicity Studies
Repeated dose toxicity studies in animals are presented in Table 8. 5,37 -43 No treatment-related gross microscopic changes were observed following exposure to 100% Polyisobutene in a 90-day dietary study of rats and 2-year dietary studies in rats or dogs. No adverse effects were observed in oral repeated dose studies of Hydrogenated Polydecene, with the no observed adverse effect levels (NOAELs) determined to be 1,000 mg/kg/d in one 90-day rat study and over 4,000 mg/kg/d in another. In a 4-week oral repeated dose rat study, the NOAEL for Hydrogenated Polydecene was 6,245 mg/kg/d in males and 6,771 mg/kg/d in females. Gross necropsy, histopathology, and microscopic findings did not reveal any significant treatment-related findings. The NOAEL for the oral administration of the trimer of hydrogenated dodecene in 2 respective oral repeated dose toxicity studies in rats was 1,000 mg/kg/d. Treatment-related effects on mortality, clinical signs, bw, food consumption, hematology, clinical chemistry, organ weights, or gross and histologic pathology were not observed in either study. In a 4-week dermal study in rats, 100% Hydrogenated Polyisobutene produced minimal to mild dermal irritation in the majority of treated animals. Histopathologic examinations of the high-dose group found effects limited to the application site and included minimal to mild epidermal hyperplasia and hyperkeratosis with reactive hyperplasia of the underlying inguinal lymph nodes.
Repeated Dose Toxicity Studies in Animals.

The oral and dermal repeated dose toxicity data that were presented in the original reviews of Polybutene and Polyethylene are summarized below and not in the tables.
Oral
Polybutene
A 2-year chronic oral toxicity study of Polybutene (75% concentrate) in Charles River albino rats given up to 20,000 ppm Polybutene blended into their regular diets revealed no gross or microscopic pathological changes that could be correlated with Polybutene ingestion. 3 No significant differences were found after 24 months of feeding in the bws or weight of food consumption, hematological results, urology, or tumor formation between the animals fed Polybutene and those that were not. In the 20,000 ppm group, 3 of 6 males that died between weeks 17 and 24 exhibited hematuria. In a 2-year chronic oral toxicity study of Polybutene (75% concentrate) in Beagle dogs, daily oral administration of Polybutene at doses up to 1,000 mg/kg/d caused no abnormalities in bw, food consumption, survival, behavioral patterns, hematology, blood chemistry, urinalysis, liver function, gross and histopathologic examinations, or organ weights and ratios. Average molecular weights of Polybutene were not specified in these studies.
Polyethylene
Toxicity testing in rats showed no adverse effects to Polyethylene at doses of 7.95 g/kg or at 1.25%, 2.50%, or 5.00% in feed for 90 days. 1 The average molecular weight of Polyethylene was not specified in this study.
Dermal
Polybutene
Polybutene did not affect hepatic or skin enzymatic activities in rats following once daily treatments for 6 days (average molecular weight not specified). 3
Reproductive and Developmental Toxicology
Polybutene
No teratogenic effects were found when Polybutene was fed to rats at 1% or 10% in the diet for 6 months. 3 A 3-generation reproductive study in Charles River albino rats that ingested up to 20,000 ppm Polybutene demonstrated that, except for the test (F2) male parental animals that were fed 20,000 ppm Polybutene, none of the animals in successive generations differed from controls with regard to weight gains. The F2 male parental animals showed slight weight gain depression, although their growth patterns were still within the normal range. In all 3 generations, there were no significant differences between test and control animals regarding litter size, the number of stillborn, and the number of viable pups during lactation. The survival, bws, and reactions of test animals were comparable to those of controls. Average molecular weights were not specified in these studies.
Hydrogenated Polydecene
The reproductive effects of Hydrogenated Polydecene were studied in rats that received the test material via gavage (average molecular weight not specified). 5 Groups of 30 male and 30 female Sprague-Dawley rats received 0, 100, 500, or 1,000 mg/kg/bw/d Hydrogenated Polydecene in Polyethylene glycol daily for 4 weeks prior to mating and through mating. At the end of mating, males were sacrificed. Females were treated through gestation and until lactation day 21. No treatment-related effects were observed on clinical signs, bw, or gross pathology in the parental generation or in the pups through lactation day 21. There were no treatment related effects on reproduction or pup viability. The NOAELs for parental systemic effects, parental reproductive effects, and offspring effects in this one generation rat study are each 1,000 mg/kg/bw/d.
Polyisobutene
In a 3-generation reproductive toxicity study, an unreported number of Charles River rats received 0, 800, or 20,000 ppm 100% Polyisobutene in their feed (molecular weight range 654-2,168 Da). 37,38 No further details about dosing were provided. Weight gain was slightly reduced in the second generation high-dose male rats, but the changes were within normal control ranges. No other effects on bws, clinical signs, organ weights, or histopathology were observed. No treatment-related reproductive effects were noted in any of the parameters measured (no furthered details provided). No differences were observed in offspring survival, litter size, number of stillborn pups, and number of viable pups in any generation of the treated groups when compared to controls. No remarkable postmortem findings were reported.
Hydrogenated Polydodecene
The reproductive effects of the trimer of Hydrogenated Polydodecene were studied in 1 generation of rats that received the test material via gavage. 5 Groups of 24 male and 24 female Sprague-Dawley rats received 0, 50, 250, or 1,000 mg/kg/d of the test material in arachis oil daily for 20 weeks (during maturation, mating, gestation, and lactation). No treatment-related effects on offspring growth or development were observed. Litter sizes were comparable to controls in all dose groups. No adverse effects were observed during gross necropsy or histopathological examination. The NOAEL for reproductive and development toxicity in this rat study is 1,000 mg/kg/d.
Genotoxicity
In Vitro
Ethylene/Octene Copolymer
A trade name mixture containing 30% to 50% Ethylene/Octene Copolymer and sodium acrylate copolymer was not mutagenic in an Ames test or in an in vitro chromosomal aberration test (no further details provided). 12
Polyethylene
Genotoxicity testing of Polyethylene was negative in 2 bacterial studies. 1 Average molecular weights were not specified in these studies.
Polyisobutene
In a study to determine the ability of various insulating fluids to induce transformation in the Syrian hamster embryo cell transformation assay and to enhance 3-methylcholanthrene-induced transformation of C3H/10T1/2 cells, a low-viscosity Polyisobutene-based oil did not induce transformation activity and was slightly cytotoxic. 2 In the 2-stage transformation assay of C3H/10T1/2 cells, the Polyisobutene oil had promoter activity. Average molecular weights were not specified in these studies.
Hydrogenated Polydecene
Hydrogenated Polydecene was not mutagenic in an Ames test at concentrations up to 500 µg/plate (molecular weight range 367-596 Da; no further details provided). 43
Hydrogenated Polydecene (average molecular weight not specified) was not mutagenic in a reverse gene mutation assay in Salmonella typhimurium strains TA1535, TA1537, TA98, and TA100 and Escherichia coli strain WP2uvrA. 5 The test material was incorporated in emulsions with sorbitan stearate and polysorbate 60 at concentrations of 156.25; 312.5; 625; 1,250; 2,500; or 5,000 μg/plate, with and without metabolic activation using the preincubation method. The positive controls yielded expected results.
In reverse mutation assays, S typhimurium strains TA98, TA100, TA1535, and TA1537 were treated with Hydrogenated Polydecene (average molecular weight not specified) at concentrations up to 10 mg/plate. 5 The positive controls yielded expected results. Hydrogenated Polydecene was not mutagenic with or without S9 metabolic activation at all tested concentrations.
Hydrogenated Polydodecene
The genotoxic potential of the trimer of Hydrogenated Polydodecene was assayed in 2chromosome aberration experiments using human lymphocyte cultures. 5 In the first experiment, the concentrations tested were 0; 39; 78.1; 156.25; 312.5; 625; 1,250; 2,500; and 5,000 μg/mL. In the second experiment, the concentrations tested were 625, 1,250, 2,500, and 5,000μg/ml for 20 h or 1,250, 2,500, and 5,000 μg/mL for a 44-hour harvest time. All experiments were conducted in duplicate, with and without S9 metabolic activation. Cytotoxicity was not observed in a range finding test conducted prior to the main assay at concentrations ≤ 5,000 µg/mL. The test material did not induce chromosomal aberrations or polyploidy cells, with or without metabolic activation. Positive controls, ethyl methane sulfonate in the absence of S9, and cyclophosphamide in the presence of S9, yielded expected results. The authors concluded that the trimer of Hydrogenated Polydodecene was not clastogenic to human lymphocytes in vitro when tested at concentrations ≤ 5,000 µg/mL.
In a mammalian cell gene mutation assay (HGPRT locus), Chinese hamster ovary (CHO) cells cultured in vitro were exposed to the trimer of Hydrogenated Polydodecene in ethanol at concentrations of 0; 313; 625; 1,250; 2,500; or 5,000 μg/mL with and without metabolic activation for 4 hours. 5 In the range-finding test, relative cloning frequencies (RCEs) ranged from 97% to 73% for concentrations ranging from 0.5 to 5,000 µg/mL without metabolic activation. Relative cloning frequencies were 122% to 80% for the same concentration range with metabolic activation. Relative cloning frequencies in the first mutation assay were 92% to 77% and 111% to 89% for concentrations ranging 313 to 5,000 µg/mL with and without metabolic activation, respectively. The activated portion of the first mutation assay was repeated and RCE was 100% to 71% for the same dose range. In the confirmatory assay, the RCEs among the test material-treated cultures ranged from 50% to 23% and 89% to 52% for the concentrations of 313 to 5,000 µg/mL with and without metabolic activation, respectively. A significant response was observed at 625 μg/mL when compared to the solvent control data in the repeat definitive mutation assay with activation; however, the increase was not significant when it was compared to the historical, cumulative solvent control data. The same was true at 2,500 µg/mL, with activation, in the confirmatory mutation assay. The increase in the number of mutants was not significant when compared to historical, cumulative solvent control data. The response seen in the definitive mutation assay at 625 μg/mL was not reproduced in the confirmatory assay. Controls were within the historical negative control values. The trimer of Hydrogenated Polydodecene was not mutagenic in this mammalian cell gene mutation assay.
Carcinogenicity
Polyethylene
Numerous investigations on the tumor production of Polyethylene implantation have produced mixed results. 1 Polyethylene causes tumors in rats implanted with squares of the test substance; however, testing involving implanting coverslips and powdered Polyethylene suggests that tumors are caused by the physical reaction to imbedded plastic films and not the Polyethylene itself. International Agency for Research on Cancer lists Polyethylene as “not classifiable as to carcinogenicity in humans” based on no adequate human data and inadequate animal data. Average molecular weights were not specified.
Polyisobutene
In a carcinogenicity study conducted to determine the skin tumorigenicity effects of certain oils used for impregnation of paper-insulated power cables and their synthetic alternatives, including Polyisobutene oil, no evidence of a direct tumorigenic or carcinogenic effect was reported and Polyisobutene oil (average molecular weight 250 Da) appeared to reduce the number of 7,12-dimethylbenz[a]anthracene-induced tumors in mice. 2
Polyisobutene (100%) was not carcinogenic in rats (dosed up to 20,000 ppm) or dogs (dosed up to 1,000 mg/kg) in oral studies described in Table 9 (molecular weight range 654-2,168 Da). 37,38
Dermal Irritation Studies.
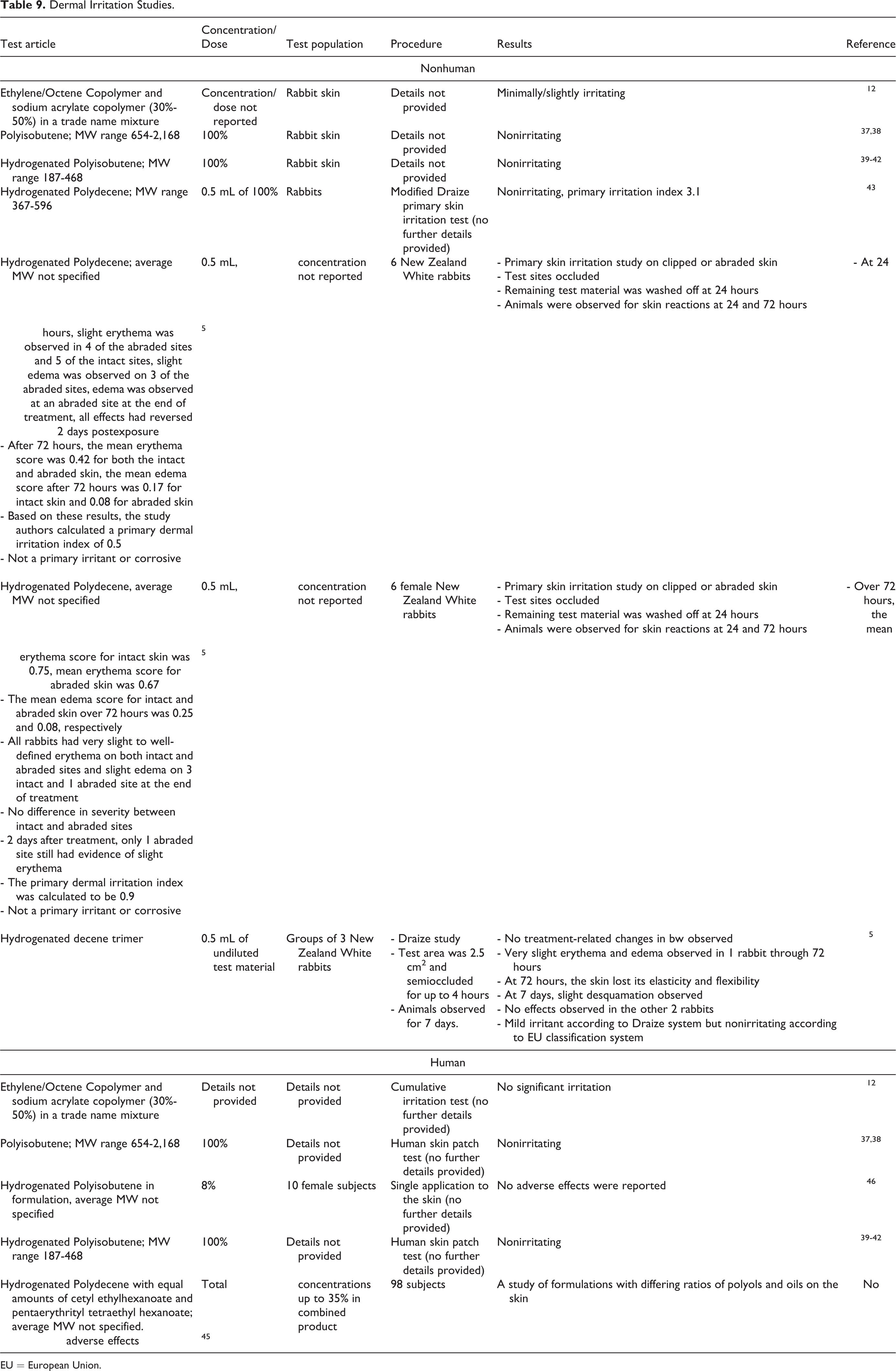
EU = European Union.
Polypropylene
International Agency for Research on Cancer determined that Polypropylene is not classifiable as to its carcinogenicity to humans (group 3) based on no adequate human data and inadequate animal data. 44
Irritation and Sensitization
Irritation
Nonhuman and human dermal irritation studies are presented in Table 9, and nonhuman ocular irritation studies are presented in Table 10. 5,12,37 -43,45,46 Ethylene/octene copolymer and sodium acrylate copolymer (30%-50% in a trade name mixture was minimally/slightly irritating to rabbit skin. Polyisobutene and Hydrogenated Polyisobutene at 100% were not irritating to rabbit skin in respective irritation studies. Hydrogenated Polydecene and the trimer of hydrogenated decene were not primary irritants or corrosives in several rabbit studies. No significant irritation was observed in human subjects in a cumulative irritation test of Ethylene/Octene Copolymer and sodium acrylate copolymer (30%-50%) in a trade name mixture. No adverse effects were reported in human subjects following irritation studies of a formulation containing 8% Hydrogenated Polyisobutene and Hydrogenated Polyisobutene at 100%. No adverse effects were reported following dermal exposure to formulations containing Hydrogenated Polydecene with equal amounts of cetyl ethylhexanoate and pentaerythrityl tetraethylhexanoate tested at total concentrations up to 35% in a study in human subjects.
Ocular Irritation Studies.
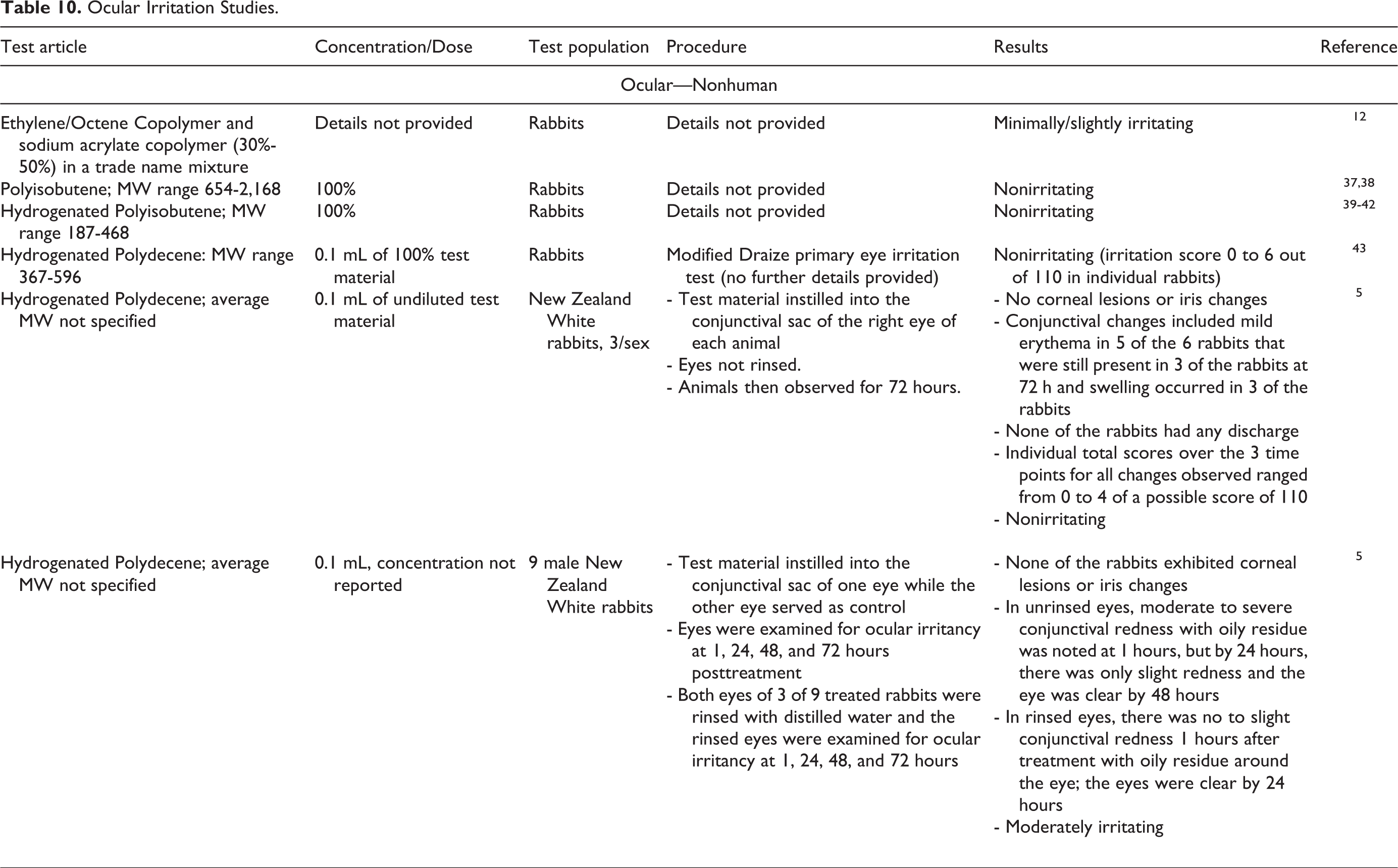
Ethylene/octene copolymer and sodium acrylate copolymer (30%-50% in a trade name mixture) was minimally/slightly irritating to rabbit eyes. Polyisobutene and Hydrogenated Polyisobutene at 100% were not irritating to rabbit eyes in respective irritation studies. Two primary eye irritation studies in rabbits found undiluted Hydrogenated Polydecene not to be an ocular irritant, while another study found the material to be moderately irritating.
The dermal, ocular, and mucous membrane irritation data that were presented in the original reviews of Polybutene, Polyethylene, Polyisobutene, and Hydrogenated Polyisobutene are summarized below and not in the tables.
Dermal
Polybutene
In primary skin irritation studies, Polybutene in formulations including lipsticks produced no abnormalities or irritation in rabbits at concentrations up to 15%; however, mild irritation was observed at concentrations greater than 15%. 3 Average molecular weights were not specified. Human primary irritation tests of a lipstick formulation containing 20% Polybutene produced no irritation. The average molecular weight was not specified.
Polyethylene
Dermal irritation studies on rabbits in which 0.5 g of Polyethylene (average molecular weight of 450 Da) was administered in 0.5 mL of water caused no irritation or corrosive effects. 1 When the same procedure was used to test Polyethylene with an average molecular weight of 655 Da, a primary irritation index score of 0.2 was found and Polyethylene was classified as a mild irritant.
Hydrogenated Polyisobutene
A skin irritation study in 6 rabbits using four patches each containing 0.5 g/patch of a Hydrogenated Polyisobutene mixture caused no reactions in any of the animals on intact or abraded skin. 2 The primary irritation index was 0.0. There was a primary irritation index score of 1.8 for rabbits treated with undiluted Hydrogenated Polyisobutene on the intact or abraded skin. Rabbits dosed dermally with 0.5 mL Hydrogenated Polyisobutene) on intact and abraded skin exhibited a primary irritation index of 0.38; not a dermal irritant. In a similar study, Hydrogenated Polyisobutene produced a primary irritation index of 0.96; also not a dermal irritant. Average molecular weights were not specified in these studies.
In humans, no primary skin irritation was produced in a 72-hour primary skin irritation patch test study with 100% Hydrogenated Polyisobutene in 25 male and female participants. 2 There was no irritancy observed in humans during a 24-hour single-insult patch test with a lip gloss containing 66.11% Hydrogenated Polyisobutene. Average molecular weights were not specified in these studies.
Ocular
Polybutene
Rabbits suffered only minimal eye irritation when Polybutene at concentrations up to 75% was instilled into the eyes with and withoutwashouts. 3 Average molecular weights were not specified.
Polyethylene
Polyethylene (molecular weight of 450 Da) was tested as a solid material (66 mg) in the eyes of rabbits. 1 The test substance caused a maximum group mean score of 11.0 and was classified as a mild irritant. All treated eyes appeared normal 48 hours after application. The same procedure, with 55 mg of Polyethylene of average molecular weight of 655, was carried out on white rabbits. The mean maximum group score produced by Polyethylene was 11.7, and it was classified as a mild irritant. All treated eyes appeared normal 72 hours after treatment. When white rabbits were tested with 13% Polyethylene beads, the maximum ocular score was 8/110 with resolution after 48 hours, and no corneal abrasions were observed.
Polyisobutene
Irritant and corrosive effects were examined following a single instillation of Polyisobutene into rabbit eyes. 2 No corneal or iridial damage was recorded in the study. One eye had irritation to the conjunctivae by 72 hours, which was present as slight hyperemia. The average molecular weight was not specified.
Hydrogenated Polyisobutene
When 0.1-mL Hydrogenated Polyisobutene was instilled into the conjunctival sac of rabbit eyes, the test material caused slight conjunctival irritation in 33% of eyes which cleared up by day 2. 2 The authors determined that Hydrogenated Polyisobutene is not an eye irritant. Another study of Hydrogenated Polyisobutene under similar test conditions produced the same results. No signs of ocular irritation were observed in a Draize study of 3 rabbits exposed to a facial lotion containing 3% Hydrogenated Polyisobutene. In a 7-day eye irritation study on rabbits, no eye irritation was observed in washed or unwashed eyes following treatment with 0.1-mL Hydrogenated Polyisobutene. An unknown concentration of Hydrogenated Polyisobutene instilled into the right eyes of 6 rabbits produced a score of 1 on the Draize scale. No other effects were observed. Average molecular weights were not specified in these studies.
In human, no adverse reactions or ocular irritation were reported in 59 subjects in a 29-day in-use study of 3different formulations of cosmetic foundations/concealer products that contained Hydrogenated Polyisobutene. 2 The concentration of Hydrogenated Polyisobutene was not specified in 2 of the 3 formulations, while the third contained 4% Hydrogenated Polyisobutene. Average molecular weights were not specified.
Mucous Membrane
Polybutene
Undiluted Polybutene produced no irritation or signs of systemic toxicity when applied to the vaginas of rabbits. 3 Average molecular weight was not specified.
Sensitization
Nonhuman and human sensitization studies are presented in Table 11. 5,11,12,43,47 Tradename mixtures containing Ethylene/Octene Copolymer were not sensitizing in a guinea pig maximization test (at 14%-16%) or in a local lymph node assay (LLNA; at 30%-50%). Hydrogenated Polydecene was not a dermal sensitizer in guinea pig maximization tests at concentrations up to 100%. The dimer of hydrogenated decene was not a dermal sensitizer in guinea pig maximization studies at 5%. The trimer of hydrogenated decene in propylene glycol was a slight sensitizer according to an LLNA. The stimulation indices were 1.56, 1.89, and 3.54 for the test concentrations of 25%, 50%, and 100%, respectively. Ethylene/octene copolymer was not a sensitizer in a human repeat insult patch test (HRIPT). A lip gloss containing 12.33% Polyisoprene was not a sensitizer according to the results of an HRIPT.
Sensitization Studies.
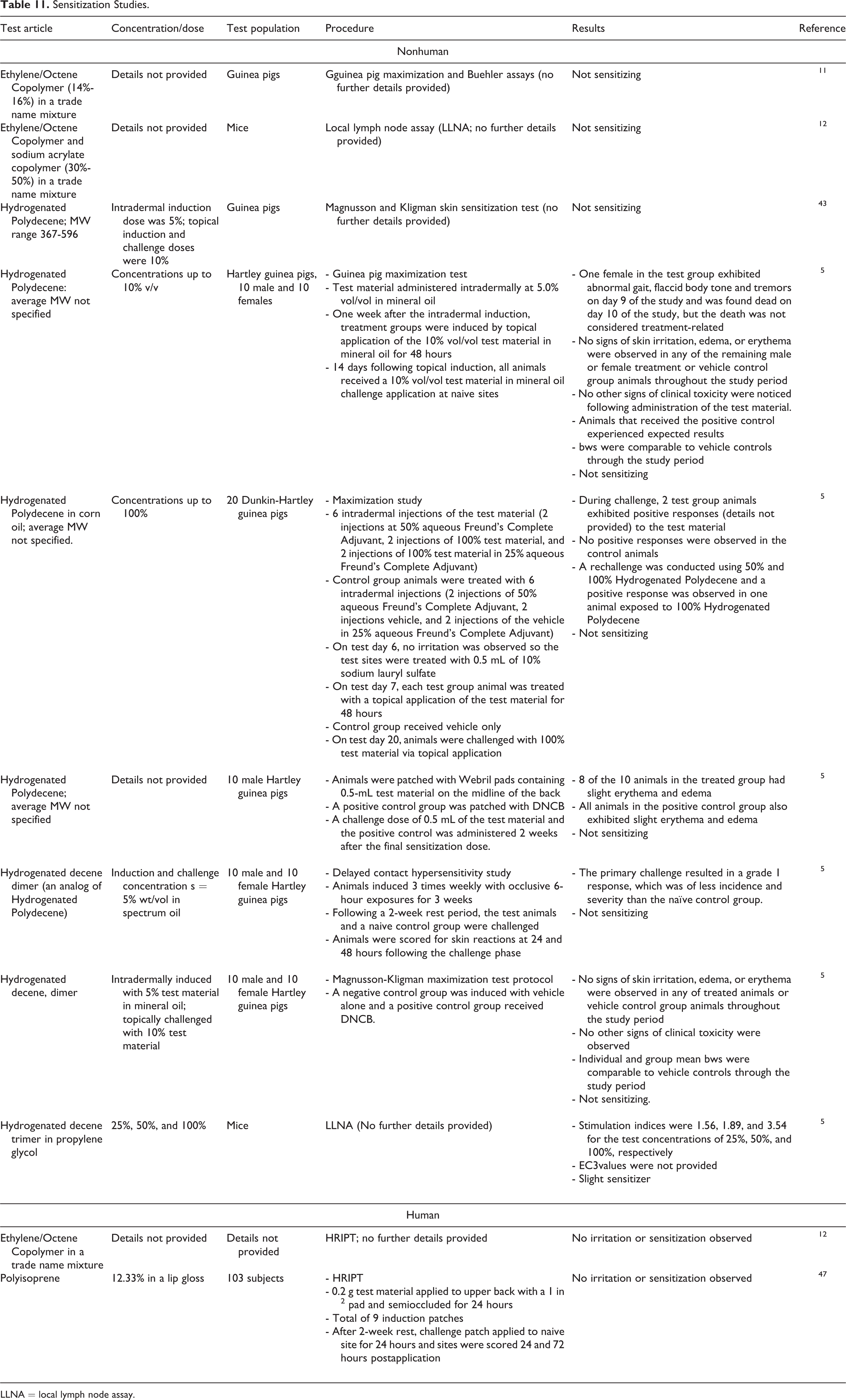
LLNA = local lymph node assay.
The sensitization data that were presented in the original reviews of Polybutene, Polyethylene, and Hydrogenated Polyisobutene are summarized below and not in the tables.
Polybutene
Repeated insult patch tests of 3.1% to 50% Polybutene in formulations produced no sensitization. 3 Average molecular weights were not specified.
Polyethylene
Polyethylene (average molecular weight of 450 Da) did not cause dermal sensitization in guinea pigs tested with 50% Polyethylene (wt/wt) in arachis oil BP. 1 In a repeat insult patch test of 201 volunteers, a product containing 13% Polyethylene beads was tested in a series of 9 consecutive administrations. There was no irritation observed with any of the induction patches. Challenge patches produced only a slight response in 1 subject, and the investigators concluded that Polyethylene has a low irritation and sensitization potential.
Hydrogenated Polyisobutene
Hydrogenated Polyisobutene was intradermally injected in an area of the skin on the back and flanks of guinea pigs. 2 Erythema and edema were observed after most inoculations, but no sensitization reactions were observed. Hydrogenated Polyisobutene injections (5%) in guinea pigs using a maximization procedure resulted in no observed reactions and an irritation index of 0.0 in both challenge phases I and II. Average molecular weights were not specified in these studies.
Repeat-insult patch tests performed to evaluate the primary irritancy/sensitization potential of formulations containing 1.44% or 4% Hydrogenated Polyisobutene in 54 male and female subjects found no reactions greater than slight erythema. 2 In a modified repeat-insult patch test under double-blind conditions, no irritation or sensitization was found in human skin patched with a makeup remover containing 51% Hydrogenated Polyisobutene. Hydrogenated Polyisobutene at up to 100% was not sensitizing in a Draize repeat insult patch in 200 subjects. Average molecular weights were not specified.
Phototoxicity
Polybutene
Photo patch tests of formulations with concentrations ranging from 15% to 50% Polybutene produced no reactions. 3 Average molecular weights were not specified.
Hydrogenated Polyisobutene
The phototoxic potential of cosmetic foundations/concealer products containing 4% Hydrogenated Polyisobutene or 1.44% Hydrogenated Polyisobutene, and a blank patch under long-wavelength UV light source (320-400 nm) was studied in 26 fair-skinned volunteers. 2 No significant reactions were reported. Formulations containing 1.44% or 4% Hydrogenated Polyisobutene were evaluated to determine their potential to induce a photoallergic reaction in the skin of 30 subjects. No response was reported at induction, rest, or challenge. Average molecular weights were not specified.
Comedogenicity
Polyisobutene
The comedogenic potential of Polyisobutene was studied using adult New Zealand White rabbits. 2 The test material was applied to the right ear of each animal daily on 5 consecutive days per week for 3 weeks. There were no signs of hyperkeratosis or comedone formation during weeks 1 and 2. By the third week, 2 treated ears exhibited signs of hyperkeratosis. The ear of the third rabbit, however, remained clear. Histological examination showed no signs of follicular hyperkeratosis on the treated, untreated, or control ears of any rabbits. The average molecular weight of Polyisobutene was not specified.
Clinical Studies
Polyethylene
There have only been a few cases of reactions to the implantation of Polyethylene in humans. 1 In the 3 published accounts, Polyethylene strips used for breast augmentation caused increased histological activity around the implant. There have also been occupational case reports on ocular irritation and systemic sclerosis in workers exposed to Polyethylene. Such workers are also exposed to other irritants. Clinical testing of intrauterine devices made of Polyethylene failed to conclusively identify statistically significant adverse effects, although squamous metaplasia was observed in treated women.
Summary
The polyene ingredients in this report are simple polyolefins that are the polymerization products of vinyl-type monomers. The polyenes reviewed in this report cover a wide range of molecular weights but have very similar structures and reaction starting materials (monomers). Polyenes function primarily as film formers and/or viscosity increasing agents—nonaqueous in cosmetic products.
According to the 2015 US FDA VCRP data, Polyethylene is reported to be used in 2,773 formulations; the single category with the most reported uses was lipstick with 885.Hydrogenated Polyisobutene is reported to be used in 1,963 formulations; the single category with the most reported uses was lipstick with 865. Most of the other in-use ingredients are primarily used in leave-on products and lipsticks. The results of the concentration of use survey conducted in 2013 and 2014 by the Council indicate Hydrogenated Polyisobutene has the highest reported maximum concentration of use; it is used at up to 95% in lipsticks.
For the ingredients that were previously reviewed by the Panel, concentrations of use for Polybutene and Hydrogenated Polyisobutene have remained about the same, with the highest maximum use concentration of 95%for Hydrogenated Polyisobutene in lip products. The highest maximum use concentration for Polyethylene has increased from 24% (eye shadow) to 67.6% (skin cleansing agents), while the highest maximum use concentration for Polyisobutene has decreased from 76% to 40% (both concentrations in lip products). Uses for all 4 ingredients have increased by several folds since their original reviews.
Many of the polyene ingredients have been approved by the FDA for use as food additives and in medical devices. An oral study that assessed the absorption potential of undiluted Hydrogenated Polydecene in rats found that most of the test compound was excreted in the feces without being absorbed (> 92%). Urinary excretion was low (< 1%), and very little of the dose was recovered in the bile (0.01%).
In acute oral toxicity studies in rats, the LD50s of diisobutylene, and triisobutylene were > 2,000 mg/kg/bw each. The oral LD50s of di-n-butene, tributene, and tetrabutene (containing 30% pentabutene) in rats were > 10,000 mg/kg each. The oral LD50 values for Ethylene/Octene Copolymer, undiluted Hydrogenated Polydecene and undiluted Hydrogenated Polydodecene were > 5,000 mg/kg in rat studies. The LD50 of undiluted Polyisobutene was > 15,400 mg/kg in an oral rat study.
Acute dermal studies of diisobutylene and Hydrogenated Polydodecene found the LD50 values > 2,000 mg/kg in rats. In rabbit studies, the dermal LD50 values for Ethylene/Octene Copolymer, hydrogenated decene dimer, Hydrogenated Polyisobutene, and Polyisobutene were > 5,000 mg/kg, > 3,000 mg/kg, >2,000 mg/kg, and >25,000 mg/kg, respectively.
In acute inhalation studies, the LC50 of diisobutylene vapor in albino rats was > 4,185 ppm (19,171 mg/m3) after a 4-hour, single, whole-body exposure. The LC50 for an aerosol of Hydrogenated Polydecene was > 5.2 mg/L in rats. The LC50 for the dimer of hydrogenated decene was 1.17 mg/L in rats. In another acute inhalation study of the dimer of hydrogenated decene, the LC50 could not be determined in rats tested at 5 mg/L because 9/10 animals died within 3 days of administration of the test material. The LC50 for Hydrogenated Polydodecene was > 5.06 mg/L. The LC50 for 100% Hydrogenated Polyisobutene was > 5 mg/L.
No treatment-related gross microscopic changes were observed following exposure to 100% Polyisobutene in a 90-day dietary study of rats and 2-year dietary studies in rats or dogs. No adverse effects were observed in oral repeated dose studies of Hydrogenated Polydecene, with the NOAELs determined to be 1,000 mg/kg/d in one 90-day rat study and over 4,000 mg/kg/d in another. In a 4-week oral repeated dose rat study, the NOAEL for Hydrogenated Polydecene was 6,245 mg/kg/d in males and 6,771 mg/kg/d in females. Gross necropsy, histopathology, and microscopic findings did not reveal any significant treatment-related findings. The NOAEL for the oral administration of the trimer of hydrogenated dodecene in 2 respective oral repeated dose toxicity studies in rats was 1,000 mg/kg/d. Treatment-related effects on mortality, clinical signs, bw, food consumption, hematology, clinical chemistry, organ weights, or gross and histologic pathology were not observed in either study. In a 4-week dermal study in rats, 100% Hydrogenated Polyisobutene produced minimal to mild dermal irritation in most treated animals. Histopathologic examinations of the high-dose group found effects limited to the application site and included minimal to mild epidermal hyperplasia and hyperkeratosis with reactive hyperplasia of the underlying inguinal lymph nodes.
In rat reproductive studies of Hydrogenated Polydecene and the trimer of Hydrogenated Polydodecene, the NOAELs for parental systemic and reproductive effects and for offspring were 1,000 mg/kg bw/d for the respective studies. No treatment-related effects were observed on clinical signs, bw, or gross pathology in the parental generation or in the pups. There were no treatment related effects on reproduction or pup viability. In a 3-generation reproductive dietary toxicity study, an unreported number of Charles River rats received 0, 800, or 20,000 ppm 100% Polyisobutene produced no treatment-related reproductive effects in any generation of the treated groups when compared to controls.
A trade name mixture containing 30% to 50% Ethylene/Octene Copolymer and sodium acrylate copolymer was not mutagenic in an Ames test or in an in vitro chromosomal aberration test Hydrogenated Polydecene at concentrations up to 10 mg/plate was not mutagenic in Ames assays, with or without metabolic activation. The trimer of Hydrogenated Polydodecene was not clastogenic to human lymphocytes nor was it mutagenic in CHO cells (HGPRT locus assay) in vitro when tested at concentrations up to 5,000 µg/mL.
International Agency for Research on Cancer determined that Polypropylene is not classifiable as to its carcinogenicity to humans (group 3). Polyisobutene (100%) was not carcinogenic in rats (dosed up to 20,000 ppm) or dogs (dosed up to 1,000 mg/kg) in oral studies.
Ethylene/octene copolymer and sodium acrylate copolymer (30%-50% in a trade name mixture) was minimally/slightly irritating to rabbit skin. Polyisobutene and Hydrogenated Polyisobutene at 100% were not irritating to rabbit skin in respective irritation studies. Hydrogenated Polydecene and the trimer of hydrogenated decene were not primary irritants or corrosives in several rabbit studies. No significant irritation was observed in human subjects in a cumulative irritation test of Ethylene/Octene Copolymer and sodium acrylate copolymer (30%-50%) in a trade name mixture. No adverse effects were reported in human subjects following irritation studies of a formulation containing 8% Hydrogenated Polyisobutene and Hydrogenated Polyisobutene at 100%. No adverse effects were reported following dermal exposure to formulations containing Hydrogenated Polydecene with equal amounts of cetyl ethylhexanoate and pentaerythrityl tetraethyl hexanoate tested at total concentrations up to 35% in a study in human subjects.
Ethylene/octene copolymer and sodium acrylate copolymer (30%-50% in a trade name mixture was minimally/slightly irritating to rabbit eyes. Polyisobutene and Hydrogenated Polyisobutene at 100% were not irritating to rabbit eyes in respective irritation studies. Two primary eye irritation studies in rabbits found undiluted Hydrogenated Polydecene not to be an ocular irritant, while another study found the material to be moderately irritating.
Ethylene/octene copolymer was not sensitizing in a guinea pig maximization test or in an LLNA. Hydrogenated Polydecene was not a dermal sensitizer in guinea pig maximization tests at concentrations up to 100%. The dimer of hydrogenated decene was not a dermal sensitizer in one guinea pig maximization study and was given a grade 1 response in another. The trimer of hydrogenated decene in propylene glycol was a slight sensitizer according to an LLNA. The stimulation indices were 1.56, 1.89, and 3.54 for the test concentrations of 25%, 50%, and 100%, respectively. Ethylene/octene copolymer was not a sensitizer in a HRIPT. Polyisoprene was not a sensitizer according to the results of a HRIPT at 12.33% in a lip gloss.
Discussion
The Panel considered the available data on polyenes, including those from the previous safety assessments on Polybutene, Polyethylene, Polyisobutene, and Hydrogenated Polyisobutene, and noted low systemic toxicity at high doses in single-dose and repeated-dose animal studies, no teratogenic effects in animal studies, and no genotoxicity in in vitro and in vivo studies. The Panel noted that use concentrations were as high as 95% in lipsticks, but a human dermal sensitization study of 100% Hydrogenated Polyisobutene in the previous safety assessment of this ingredient was negative, and no irritation or sensitization was observed in multiple tests of some of the other polyene ingredients. The Panel recognized that polyenes are approved for use in foods (directly and indirectly) and drug and medical devices.
The Panel also noted that although molecular weights are in the range that could be dermally absorbed, the lack of heteroatom functional groups dramatically limits solubility and would prevent significant absorption. The lack of functional groups also limits interactions with other biomolecules and accounts for the apparent biological inertness of these ingredients.
The Panel noted gaps in the available safety data for some of the polyenes in this safety assessment. The data available for many of the ingredients are sufficient and can be extrapolated to support the safety of the entire group because of the similarities in the chemical structures, physicochemical properties, use concentrations, and reported functions across the group.
The Panel discussed the issue of incidental inhalation exposure in pump and aerosol hair sprays, underarm deodorant sprays, face and neck sprays, body and hand sprays, and aerosol suntan products. The limited data available from inhalation studies, including acute exposure data, suggest little potential for respiratory effects at relevant doses. The Panel considered pertinent data indicating that incidental inhalation exposures to polyenes in such cosmetic products would not cause adverse health effects, including data characterizing the potential for polyenes to cause systemic toxicity, ocular or dermal irritation or sensitization, and other effects. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. The potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Panel concluded that the following polyene ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment:

* Not reported to be in current use. Where ingredients in this group not in current use to be used in the future, the expectation is that these ingredients would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author Contributions
Burnett, C. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., Snyder, P., and Gill, L. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Heldreth, B. contributed to design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Authors' Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.