Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of 2-Amino-3-Hydroxypyridine, which is reported to function as an oxidative hair dye ingredient. The Panel reviewed relevant animal and human data provided in this safety assessment, and concluded that 2-Amino-3-Hydroxypyridine is safe in the present practices of use and concentration for use in oxidative hair dye formulations.
Introduction
This report addresses the safety of 2-Amino-3-Hydroxypyridine (CAS No. 16867-03-1). This cosmetic ingredient is used as a component in hair dyes.
Chemistry
2-Amino-3-Hydroxypyridine is the heterocyclic aromatic compound that conforms to the structure shown in Figure 1. 1 Physical and chemical properties of 2-Amino-3-Hydroxypyridine are found in Table 1.
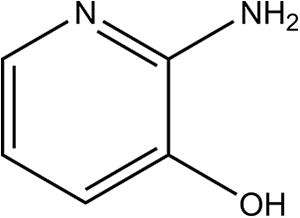
2-Amino-3-Hydroxypyridine.
Chemical Properties of 2-Amino-3-Hydroxypyridine.

Abbreviation: DMSO, dimethyl sulfoxide.
2-Amino-3-Hydroxypyridine is commonly used as a component of oxidative hair dyes. 2 This ingredient acts as a “coupler” and reacts with a “precursor.” In a typical formulation, a precursor is activated via an oxidant, such as peroxide. The resultant activated precursor proceeds to react with a coupler such as 2-Amino-3-Hydroxypyridine to form in situ a product that is purported to be the actual dye that colors hair in these types of oxidative hair dyes.
Information concerning the propensity of 2-Amino-3-Hydroxypyridineto form nitrosamines was not found in the literature or made available. 2-Amino-3-Hydroxypyridine bears a primary aryl amine. While many secondary amines and amides are readily nitrosated to form isolatable nitrosamines and nitrosamides, primary alkyl and aryl amines ultimately yield diazonium salts, instead of nitrosamines. The nitrogen atom of pyridines in general has, however, been shown to be susceptible to nitrosation, 3 but only under nonphysiologic conditions or under strictly anhydrous conditions not applicable to a hair dye product formulation. Of concern in cosmetics is the conversion (nitrosation) of nitrogen-bearing ingredients into N nitroso chemicals that may be carcinogenic. Of the approximately 209 nitrosamines tested, 85% have been shown to produce cancer in laboratory animals. 4 Nitrosation can occur under physiologic conditions. 5 Depending on the nitrosating agent and the substrate, nitrosation can occur under acidic, neutral, or alkaline conditions. Atmospheric NO2 may also participate in nitrosation in aqueous solution. 6
Impurities
Potential impurities in 2-Amino-3-Hydroxypyridine include 2,3-dihydroxypyridine and 3-hydroxy-2-pyridone. 7 Heavy metal content was described as < 20 ppm Pb, < 10 ppm Sb and Ni, < 5 ppm As and Cd, and <1 ppm Hg.
Use
Cosmetic
According to information supplied to the US Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP), 2-Amino-3-Hydroxypyridine is used in 103 hair dyes and colors and 48 hair tints for a total of 151 uses in hair coloring formulations. 8 A survey of use concentrations has reported that 2-Amino-3-Hydroxypyridine is used at a maximum concentration range of 0.1% to 1.2% in hair dyes and colors; the reported 1.2% reflects the concentration before dilution. 9 The maximum concentration as applied to the hair is 1.0%.
Europe’s Scientific Committee on Consumer Products concluded that 2-Amino-3-Hydroxypyridine would not pose a health risk to the consumer when used as an ingredient in oxidative hair dye formulations, as long as the maximum concentration applied to hair does not exceed 1.0%. 7 The European Commission has added 2-Amino-3-Hydroxypyridine to Annex III List of Substances Which Cosmetic Products Must Not Contain Except Subject to the Restrictions Laid Down; accordingly, after mixing under oxidative conditions, the maximum concentration applied to hair must not exceed 1.0%. 10
Hair dye caution statement—FDA labeling
This ingredient is considered a coal tar hair dye for which regulations require caution statements and instructions regarding patch tests in order to be exempt from certain adulteration and color additive provisions of the United States Federal Food, Drug, and Cosmetic Act. In order to be exempt, the following caution statement must be displayed on all coal tar hair dye products: Caution: This product contains ingredients which may cause skin irritation on certain individuals and a preliminary test according to accompanying directions should be made. This product must not be used for dyeing the eyelashes or eyebrows; to do so may cause blindness.
In a 2012 published report regarding such self-testing for contact sensitization to hair dyes, the authors concluded that, in its present form, the hair dye self-test has severe limitations. 11 The authors issued the warning that, if the use of a hair dye self-test to predict contact sensitization becomes widespread, there is severe risk that a tool has been marketed that may cause morbidity in European consumers. In an accompanying editorial, on behalf of the European Society of Contact Dermatitis (ESCD), the author asserted that industry is focusing on predicting the risks from exposure to hair dyes by having millions of European consumers perform a self-test prior to each hair dying and stated that it is the opinion of the ESCD that attention must be given to reducing the risks of serious allergic reactions by improving the safety of the products themselves. 12
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Dermal/percutaneous
In an in vitro percutaneous absorption study, [14C]-2-Amino-3-Hydroxypyridine at 1.04% as part of an oxidative hair dye cream formulation with hydrogen peroxide was applied to clipped, dermatomed pig skin. 13 The integrity of the skin was tested by measuring transepidermal electrical resistance prior to test material application. The test substance (20 mg/cm2) was applied to the skin samples in 2.54 cm2 glass diffusion cells for 30 minutes and then rinsed off with a detergent solution. Measurements for radioactivity in the receptor fluid were made at 0.5, 1, 2, 4, 6, 24, 29, and 48 hours after application. At experiment end, the skin was tape stripped to determine distribution of the test substance through the skin. Most of the test material was found in the rinsing solution (93.7%) at 0.5 hours. After 0.5 hours, 0.011% (0.024 μg/cm2) had penetrated the skin, and after 48 hours, the amount increased to 0.306% (0.638 μg/cm2). Most of the penetration occurred during the first hour, with a penetration rate of 0.161 μg/cm2/h. After 6 hours, the penetration rate remained relatively constant at 0.006 μg/cm2/h. The penetration rate over the 48-hour period was 0.012 μg/cm2/h. Approximately 0.152% (0.316 μg/cm2) of the applied dose was recovered from the stratum corneum, while 0.300% (0.625 μg/cm2) remained in the epidermis/dermis after tape stripping. The total amount of radiolabeled 2-Amino-3-Hydroxypyridine that was biologically available was 0.606% (1.26 μg/cm2).
Toxicological Studies
Acute Toxicity
Oral—Nonhuman
The acute oral toxicity of 2-Amino-3-Hydroxypyridinein propylene glycol was tested in Wistar rats. 14 Three females and 3 males received a single gavage dose of the test substance at 300 mg/kg body weight and 3 females received a single dose at 1000 mg/kg. The rats were observed daily for mortalities and clinical signs of toxicity for 14 days. All 3 rats of the 1000 mg/kg dose group and 1 female in the 300 mg/kg dose group died immediately after dosing. Clinical signs in the 1000 mg/kg dose group included tremor, hunched posture, abnormal gait, salivation, and chromodacryorrhea. Females in the 300 mg/kg were observed with the following clinical signs: restless, lethargy, tremor, hunched posture, uncoordinated movements, flat gait, quacking breathing, labored respiration, rales, shallow respiration, piloerection, salivation, chromodacryorrhea, pale, and ptosis. No clinical signs were observed in the 300 mg/kg dose group males. Surviving animals recovered from the symptoms between days 2 and 7, except for one female experiencing chromodacryorrhea and rales that recovered by day 15. No abnormal weight gain was observed in the males and slight weight loss or reduced body weight gain between days 8 and 15 in the females was not considered significant. At necropsy, the animals that died after the 1000 mg/kg dose had enlarged lungs with many dark red foci. In the female that died following the 300 mg/kg dose, reddish discoloration of stomach mucosa was observed. Enlarged mandibular lymph nodes were noted in the males. In this acute oral toxicity study, the median oral LD50 was greater than 300 mg/kg body weight in male Wistar rats and less than 1000 mg/kg body weight in female Wistar rats.
Repeated Dose Toxicity
Oral—Nonhuman
The potential for oral toxicity to 2-Amino-3-Hydroxypyridine in deionized water was investigated in Wistar rats. 15 Dose groups consisted of 20 animals of each sex and received 0, 30, 60, or 120 mg/kg body weight of the test material at a dose volume of 10 mL/kg body weight. The test material was administered by oral gavage once daily for 91 days in males and 92 days in females. An additional 2 groups of 5 males and 5 females received either 0 or 120 mg/kg body weight and were kept for 28 days longer without treatment to observe reversibility of any clinical signs of toxicity or persistence of test substance induced lesions. Mortalities were observed daily, and detailed clinical observations, feed consumption, and body weights were recorded weekly. Ophthalmoscopic exams were performed before treatment and on day 85 in all animals. At the end of the treatment period, complete hematology and clinical biochemistry investigations were performed. All animals were killed at the end of the treatment period. Major organs, including reproductive organs, were weighed and a detailed necropsy was performed in all animals. Histopathological examinations were performed on the organs and tissues, including the reproductive organs, of the high dose group and the negative control group and on any lesions discovered during gross examination.
During the treatment period, 2 low dose animals died due to non-test material–related circumstances. The death of 2 high dose females was attributed to the test material: edema of the lungs and subcutis were noted at necropsy with these animals. In high dose animals of both sexes, “seizure”-like abnormalities, clonic convulsions, vocalization, and salivation were observed. Chromodacryorrhea were noted in the mid and high dose groups. No irregularities were observed during the ophthalmoscopic exams. Body weights were significantly decreased in both sexes in the high dose group. Feed consumption of mid and high dose females was also decreased. Statistically significant differences in mean corpuscular hemoglobin and mean cell volume were observed, but these were not dose-dependent. In clinical biochemistry, potassium, protein, and alanine aminotransferase levels were increased in the high dose males and aspartate aminotransferase was increased and cholesterol was decreased in the high dose females.
Statistically significant differences in kidney and liver weights in high dose males and high dose females, respectively, were observed. One mid dose and 1 high dose male and 3 high dose females were noted to have focal hepatic necrosis, which was thought to be a secondary effect due to an unidentified primary lesion. No other statistically significant differences between treated and control animals were observed during the histopathological examination. In this repeated dose study, the authors concluded that the no observed effect level (NOEL) was 30 mg/kg body weight/day. 15
Reproductive and Developmental Toxicity
In a teratogenicity study, mated female Crl:(WI)BR rats received 2-Amino-3-Hydroxypyridine by gavage on days 6 to 15 of gestation. 16 The doses used were based on the results of a range finding study in rats tested with up to 480 mg/kg body weight 2-Amino-3-Hydroxypyridine. 17 In the main study, groups of 24 rats received 0, 15, 45, or 135 mg/kg body weight/day of the test material in distilled water. Clinical signs for toxicity were observed daily. Body weights were recorded on gestation days 0, 6, 11, 16, and 20, and feed consumption was measured on gestation days 0-6, 6-11, 11-16, and 16-20. Dams were killed on gestation day 20. Complete necropsy and macroscopic examination of the abdominal and thoracic organs was performed. Fetuses were removed, sexed, weighed, and examined externally. Number of corpora lutea, implantation sites, resorption sites, and fetuses were recorded. The fetuses were examined for anomalies.
Two mortalities were observed in the dams within the first 2 days of dosing of the high dose group, so the dose used was lowered to 90 mg/kg body weight on day 11. Severe convulsions and hypersalivation preceded these deaths. Hypersalivation was also observed immediately after administration in some of the remaining animals in this group. Statistically significant decreased feed consumption and body weight gain were observed in the dams when compared to controls. At necropsy, no test substance-related effects were observed in the dams. In the fetuses of the high dose group, statistically significant increases in skeletal variations and rudimentary lumbar ribs were observed when compared to the controls. No test substance-related effects were observed in dams or fetuses in the 15 or 45 mg/kg dose group. The maternal and fetal no observed adverse effect level (NOAEL) was 45 mg/kg body weight in this rat teratology study. 16
Genotoxicity
In Vitro
The potential of 2-Amino-3-Hydroxypyridine to induce gene mutation was studied in Salmonella typhimurium strains TA98, TA100, TA102, TA1535, and TA1537 using the reverse mutation assay. 18 The assay was performed with and without S9 metabolic activation at concentrations of 0, 33, 100, 333, 1000, 2500, or 5000 μg/plate. Normal background growth was observed in all strains up to the maximum concentration tested, with and without metabolic activation. No toxic effects, substantial increases in revertant colony numbers, or higher mutation rates associated with increasing concentrations were observed with any test strain at any dose level, with or without metabolic activation. Positive controls yielded expected results. It was concluded that 2-Amino-3-Hydroxypyridine was not mutagenic with and without metabolic activation in the S typhimurium strains tested.
2-Amino-3-Hydroxypyridinein deionized water was studied for cell mutation in mouse lymphoma L5178Y TK+/- cells in 1 experiment with 2 parallel cultures each. 19 Concentrations tested were 150, 300, 600, 900, and 1200 µg/mL, and the experiment was performed with and without S9 metabolic activation. Cultures were incubated with the test material for 4 hours. A substantial and reproducible dose-dependent increase in mutant colony numbers was observed in the cultures without metabolic activation. In both parallel cultures of the 1200 µg/mL dose cultures without S9, the threshold of 2-times the corresponding solvent control was reached and exceeded, and the historical range and negative and solvent controls was exceeded in one culture. The ratio of small versus large colonies was shifted toward small colonies, indicating clastogenic effects. No relevant increase of mutation frequency was observed in the cultures with metabolic activation. Positive controls yielded expected results. It was concluded that 2-Amino-3-Hydroxypyridine without metabolic activation was mutagenic in this assay.
The potential of 2-Amino-3-Hydroxypyridine to induce chromosomal aberrations was studied in Chinese hamster V79 cells. 20 The cells were treated with the test material at 0, 800, 1000, or 1100 μg/mL, with and without S9, for 4 hours. Harvest times were 18 hours after the start of treatment. In each culture, 100 metaphase plates were scored for structural chromosome aberrations. With metabolic activation, the reductions in cell numbers were 34%, 32%, and 52% at 800, 1000, and 1100 μg/mL, respectively. Without metabolic activation, the reductions in cell numbers were 14%, 32%, and 51% at 800, 1000, and 1100 μg/mL, respectively. Statistically significant increases in the number of cells with structural chromosome aberrations were observed with all 3 concentrations, with and without metabolic activation. These increases were at the same level or higher when compared to the positive control. A nonstatistically significant dose-dependent increase in polyploidy cells was observed with and without metabolic activation. Under the conditions of this study, 2-Amino-3-Hydroxypyridine was considered clastogenic.
The potential of 2-Amino-3-Hydroxypyridine in deionized water to induce gene mutations at the HPRT locus was assessed using Chinese hamster V79 cells, with and without S9 metabolic activation. 21 The assay was performed in 2 independent experiments, with the cells exposed to the test material for 4 hours in the first experiment with and without metabolic activation and for 24 hours without metabolic activation in the second experiment. In the first experiment, test concentrations were 0, 75, 150, 300, 600, or 1200 µg/mL with metabolic activation and 0, 75, 150, 300, 450, or 600 µg/mL without metabolic activation. In the second experiment, test concentrations were 0, 37.5, 75, 150, 300, or 450 µg/mL. No relevant or dose-dependent increase of mutation frequency was observed in either experiment. Positive controls yielded expected results. Under the conditions of this assay, 2-Amino-3-Hydroxypyridine did not induce gene mutations at the HPRT locus in V79 cells and, thus, was not mutagenic in this HPRT assay.
In Vivo
The genotoxic potential of 2-Amino-3-Hydroxypyridine formulated in 2.5% carboxymethyl cellulose was studied in a micronucleus test using NMRI mice. 22 Groups of 5 mice of each sex received single doses of 12.5, 25, or 50 mg/kg body weight 2 Amino-3-Hydroxypyridine intraperitoneally. Bone marrow was collected at 24 or 48 hours. At least 2000 polychromatic erythrocytes (PCE) were scored for micronuclei. The mean number of PCE was not decreased after exposure to the test material when compared to the vehicle control, which indicated that 2-Amino-3-Hydroxypyridine was not cytotoxic to the bone marrow. The validity of the test was shown by chemical analysis of blood samples of the treated mice, indicating the bioavailability of the test item. No statistically significant or biologically relevant increase in the incidence of micronucleated polychromatic cells was observed at any dose level or exposure time. The mean values of micronuclei in the treated groups were comparable with the values of the vehicle control group. Positive controls yielded expected results. Under the conditions of this study, 2-Amino-3-Hydroxypyridine was considered not mutagenic.
Irritation and Sensitization
Irritation
Dermal—Nonhuman
The irritation potential of 2-Amino-3-Hydroxypyridine was assessed in 3 male New Zealand White rabbits. 23 Approximately 0.5 g of the undiluted test substance moistened with 0.5 mL purified water was applied to a clipped, intact area (6 cm2) of skin and then semioccluded. The patches were removed after 4 hours and the remaining test substance was rinsed off with water and ethanol. No skin reactions were observed in any of the rabbits up to 72 hours after patch removal. Brownish staining was observed in the rabbits during the observation. It was concluded that 2-Amino-3-Hydroxypyridine was nonirritating to rabbit skin.
Ocular—Nonhuman
The ocular irritation potential of undiluted 2-Amino-3-Hydroxypyridine was tested in 3 male New Zealand White rabbits. 24 The conjunctival sac of one eye of each rabbit was instilled with 0.1 mL (∼60 mg sample) of the test material. The untreated eye served as a control. Both eyes were examined at 1, 24, 48, 72 hours, and 7 days posttreatment. After 24 hours, a solution of 2% fluorescein in water was instilled into both eyes in order to determine corneal epithelial damages. Immediately after the fluorescein examination, the eyes were rinsed with water. Corneal injury consisting of opacity (max grade 1) and epithelial damage (max 50% of the corneal area) was observed: these resolved within 7 days in all animals. Iridial irritation grade 1 was observed in one animal within 1 hour and 24 hours of instillation. Irritation of the conjunctivae consisting of redness, chemosis, and discharge completely resolved within 14 days in all animals. There was no evidence of ocular corrosion. Remnants of the test material were observed up to 48 hours after instillation. Under the conditions of this study, 2-Amino-3-Hydroxypyridine was considered irritating to the rabbit eye.
Sensitization
Dermal—Nonhuman
The contact hypersensitivity of 2-Amino-3-Hydroxypyridine was assessed in a local lymph node assay (LLNA). 25 CBA female mice were divided into groups of 5 and received 0%, 5%, 25%, or 50% of the test material in ethanol: water (7:3, vol/vol) on the ear surface (25 µL/ear) once daily for 3 consecutive days. Clinical signs of toxicity were assessed daily, body weights were measured on days 1 and 6, and on day 3, skin reactions were assessed. On day 6, all animals were injected intravenously with 20 µCi [3 H]-methylthymidine and the proliferation of lymphocytes in the draining lymph nodes was measured. The stimulation indices (SI) were calculated.
No clinical signs of toxicity, deaths, or skin reactions occurred during the treatment period in any dose group. Visual observations found the lymph nodes were all of equal size. The mean disintegrations per minute/animal values for each test group were 113, 332, 192, and 220 for the 0%, 5%, 25%, and 50% dose groups, respectively. The SI were 2.9, 1.7, and 1.9 for the 5%, 25%, and 50% dose groups, respectively. There was no indication that 2-Amino-3-Hydroxypyridine could elicit an SI greater than 3. It was concluded that 2-Amino-3-Hydroxypyridinewas not predicted to be a skin sensitizer in this LLNA. 25
Quantitative structure-activity relationship
A non-validated quantitative structure-activity relationship (QSAR) model was utilized to predict the sensitization potential of all hair dye ingredients registered in Europe (229 substances as of 2004). 26 The model predicted 2-Amino-3-Hydroxypyridine to be a moderate to strong sensitizer. The QSAR analysis involved calculating TOPological Substructural MOlecular DEsign (TOPS-MODE) descriptors and correlating them to unspecified sensitization data from LLNAs that were available in July 2003.
Epidemiology
2-Amino-3-Hydroxypyridine is used as a precursor in oxidative hair dyes. While the safety of individual hair dye ingredients is not addressed in epidemiology studies that seek to determine links, if any, between hair dye use and disease, such studies do provide broad information. Currently available epidemiology studies provided insufficient evidence to support a causal association between personal hair dye use and a variety of tumors and cancers. A detailed summary of the available hair dye epidemiology data is available at http://www.cir-safety.org/cir-findings.
Summary
2-Amino-3-Hydroxypyridine is used as a coupling agent in oxidative hair dyes. Per 2014 VCRP data, 2-Amino-3-Hydroxypyridine is used in a total of 151 hair coloring formulations. According to an industry survey, the maximum concentration reported is 1.2% (undiluted); the maximum “on-head” use concentration is 1%.
In an in vitro percutaneous absorption study of 1.04% radiolabeled 2-Amino-3-Hydroxypyridinein formulation with hydrogen peroxide in pig skin, most of the test material was found in the rinsing solution (93.7%) at 0.5 hours. After 0.5 hours, 0.011% (0.024 μg/cm2) had penetrated the skin, and after 48 hours, the amount increased to 0.306% (0.638 μg/cm2). Most of the penetration occurred during the first hour, with a penetration rate of 0.161 μg/cm2/h. The total amount of radiolabeled 2-Amino-3-Hydroxypyridine that was biologically available was 0.606% (1.26 μg/cm2).
An acute oral toxicity study of 2-Amino-3-Hydroxypyridine in propylene glycol in rats determined the LD50 to be 500 mg/kg body weight. The NOEL for a 90-day oral study of 2-Amino-3-Hydroxypyridine in deionized water in rats was 30 mg/kg body weight/day.
In a teratogenicity study wherein mated female rats received 2-Amino-3-Hydroxypyridine by gavage at doses of 0, 15, 45, or 135 mg/kg body weight/day of the test material in distilled water, the maternal and fetal NOAEL was 45 mg/kg body weight. Dams in the high dose group experienced hypersalivation, decreased feed consumption, decreased body weight gain, and two deaths. Fetuses of this high dose group had statistically significant increases in skeletal variations and rudimentary lumbar ribs were observed when compared to the controls, likely to be affected by the maternal toxicity found in the dams.
2-Amino-3-Hydroxypyridine was considered not mutagenic in a reverse mutation assay, an assay with the HPRT locus of Chinese hamster V79 cells, or an in vivo micronucleus test in mice. However, this ingredient was considered clastogenic without metabolic activation in mouse lymphoma L5178Y TK+/- cells, and clastogenic in a chromosomal aberration study in Chinese hamster V79 cells.
In a dermal irritation study, undiluted 2-Amino-3-Hydroxypyridine was nonirritating to rabbit skin. Undiluted 2-Amino-3-Hydroxypyridine was considered irritating to the rabbit eye. A nonvalidated QSAR model predicted that 2-Amino-3-Hydroxypyridine would be a moderate to strong sensitizer, but an LLNA study concluded that 2-Amino-3-Hydroxypyridine tested up to 50% in ethanol: water was not a skin sensitizer.
Discussion
The Panel expressed concern over in vitro genotoxicity studies of 2-Amino-3-Hydroxypyridine that had clastogenic results; however, the Panel noted that, in general, cytotoxicity of test materials tend to confound results in these types of studies. Additionally, the Panel noted the skeletal effects of fetal rodents exposed to high concentrations of 2-Amino-3-Hydroxypyridine in a teratogenicity study. Given that additional genotoxicity studies, including an in vivo study, were negative for mutagenicity, that the dams in the teratogenicity study also experienced adverse effects at high concentrations and the NOAELs for both dams and fetuses were much higher than the concentration used in hair dyes, and that dermal penetration is estimated to be relatively low, the Panel determined that none of these adverse effects would be likely at the maximum concentration of 1.0% that is applied to hair.
Although the nitrosamine content of these hair dyes has not been reported, the Panel noted this ingredient has a primary aryl amine. While many secondary amines and amides are readily nitrosated to form isolatable nitrosamines and nitrosamides, primary alkyl and aryl amines ultimately yield diazonium salts, instead of nitrosamines. The nitrogen atom of some pyridines, however, has been shown to be susceptible to nitrosation, but only under anhydrous conditions that would not be physiologically relevant or applicable to hair dye product formulations.
The Panel recognized that 2-Amino-3-Hydroxypyridine functions as a hair dye ingredient and that hair dyes containing this ingredient, as coal tar hair dye products, are exempt from certain adulteration and color additive provisions of the Federal Food, Drug, and Cosmetic Act, when the label bears a caution statement and patch test instructions for determining whether the product causes skin irritation. The Panel expects that following this procedure will prospectively identify individuals who would have an irritation/sensitization reaction and allow them to avoid significant exposures. The Panel considered concerns that such self-testing might induce sensitization, but agreed that there was not a sufficient basis for changing this advice to consumers at this time.
In considering hair dye epidemiology data, the Panel concluded that the available epidemiology studies are insufficient to scientifically support a causal relationship between hair dye use and cancer or other toxicological end points, based on lack of strength of the associations and inconsistency of findings.
The Panel noted that the use of oxidative hair dye formulations involves exposure to precursors and coupling agents as well as to their reaction products. While reaction intermediates may be formed, human exposure is to the precursors and coupling agents and to reaction products, not to reaction intermediates. The exposures to the precursors and couplers are low (they are consumed in the color forming reaction), and the exposures to reaction products are even lower (they are adsorbed onto the hair shaft itself and physically retained there). Therefore, it was the consensus of the Panel that safety assessments of oxidative hair dyes are primarily determined by the toxicological evaluation of the ingredients (ie, precursors and coupling agents), rather than by the reaction intermediates or products formed during use.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that 2-Amino-3-Hydroxypyridine is safe in the present practices of use and concentration for use in oxidative hair dye formulations.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
Burnett, C. contributed to conception, design, acquisition, analysis, interpretation, drafted the manuscript, and critically revised the manuscript; Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., Snyder, P., and Gill, L. contributed to conception, design, analysis, interpretation, and critically revised the manuscript. Heldreth, B. contributed to analysis, interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
