Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of 21 parabens as preservatives in cosmetic products. All of these ingredients are reported to function in cosmetics as preservatives; however, 5 are reported to also function as fragrance ingredients. The Panel reviewed relevant data relating to the safety of these ingredients under the reported conditions of use in cosmetic formulations. The Panel concluded that 20 of the 21 parabens included in this report are safe in cosmetics in the present practices of use and concentration described in this safety assessment when the sum of the total parabens in any given formulation does not exceed 0.8%. However, the available data are insufficient to support a conclusion of safety for benzylparaben in cosmetics.
Introduction
This is a rereview of the safety of parabens as used in cosmetics; included are the available scientific literature and unpublished data relevant to reassessing the safety of the previously reviewed ingredients and assessing other ingredients for the first time. According to the web-based
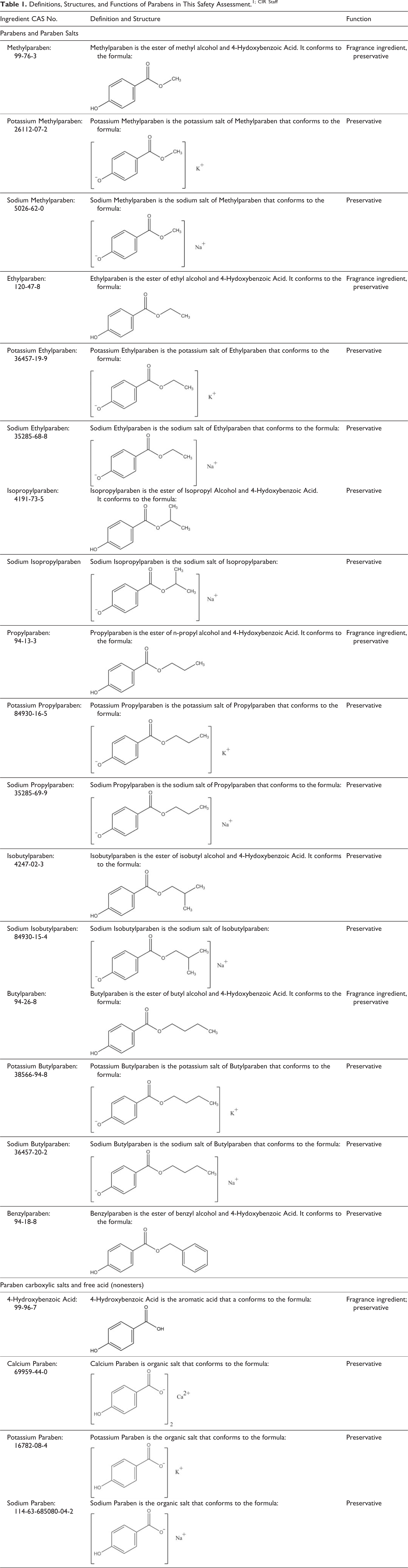
Definitions, Structures, and Functions of Parabens in This Safety Assessment.1; CIR Staff
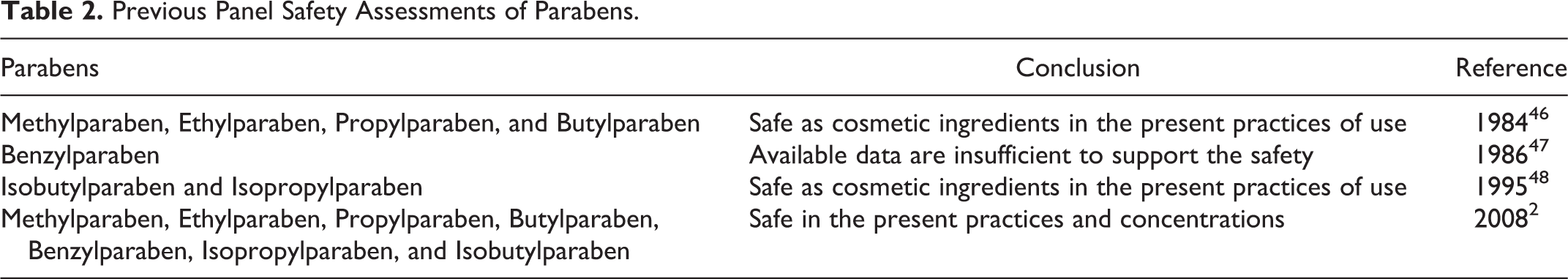
In 2017, the Expert Panel for Cosmetic Ingredient Safety (Panel) agreed to reopen the parabens report that was published in 2008 2 and to include the paraben salts and 4-Hydroxybenzoic Acid. The conclusions of all previous Panel safety assessments of parabens are summarized in Table 2. The 21 ingredients in this current assessment thus comprise the following:
Previous Panel Safety Assessments of Parabens.
Benzylparaben*
Butylparaben*
Calcium Paraben
Ethylparaben*
Isobutylparaben*
Isopropylparaben*
Methylparaben*
Potassium Butylparaben
Potassium Ethylparaben
Potassium Methylparaben
Potassium Paraben
Potassium Propylparaben
Propylparaben*
Sodium Butylparaben
Sodium Ethylparaben
Sodium Isobutylparaben
Sodium Isopropylparaben
Sodium Methylparaben
Sodium Paraben
Sodium Propylparaben
4-Hydroxybenzoic Acid
*
This rereview was initiated because some of the ingredients being reviewed for the first time had high frequencies of use (eg, Sodium Methylparaben was reported to be used in 436 cosmetic formulations at the time of prioritization). In addition, the Panel was concerned that new data from a developmental and reproductive toxicity (DART) study indicated reduced sperm counts and reduced expression of a specific enzyme and a reduction in a specific cell marker in the testes of offspring of female rats orally dosed with 10 mg/kg/d Butylparaben during gestation and lactation periods. 3 Reductions in anogenital distance (AGD) and other effects were reported at 100 mg/kg/d in this study. In comparison, the previous Panel safety assessment of parabens included the calculation of margin of safety (MOS) values for adults and infants, assuming a no observed adverse effect level (NOAEL) of 1,000 mg/kg/d from an older DART study. 2 After careful consideration of all the new data regarding endocrine activity and DART studies, the Panel determined an adequate NOAEL value of 160 mg/kg/d for Butylparaben. An MOS was recalculated accordingly, considering the different use concentrations and exposures of Butylparaben in various cosmetic product categories.
An exhaustive search of the world’s literature was conducted for new data on the safety of parabens, as well as on 4-Hydroxybenzoic Acid (a metabolite common to each of the esters herein), in preparation of this report. A few short-term toxicity studies, but no new acute, subchronic or chronic toxicity studies, were discovered. This safety assessment includes relevant published and unpublished data that are available for each end point that is evaluated. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the end points that Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data were provided by the cosmetics industry.
Pertinent data were discovered in the European Chemicals Agency database. 4 -12 Data were also discovered in reports by the Joint FAO/WHO Expert Committee on Food Additives (JECFA) 13 and the European Union’s (EU) Scientific Committee on Consumer Safety (SCCS). 14 -20
Dermal penetration, toxicokinetic, short-term toxicity, DART, endocrine activity, genotoxicity, biomonitoring, and epidemiology studies are briefly summarized in the body of the report, and in most cases, details are provided in tables. Toxicity studies conducted in animals exposed to individual parabens by subcutaneous injection are also briefly tabulated in the report; however, these studies lack relevance in assessing human exposure to parabens in cosmetics when dermal metabolism is bypassed (ie, the protective barrier of the skin is bypassed injection). In addition, toxicity tests in animals exposed to mixtures of parabens with other compounds (eg, phthalates) were not included due to their lack of relevance.
Chemistry
Definition and Structure
The ingredients in this safety assessment are paraben phenolic acids, phenolic salts, the free carboxylic acid (4-Hydroxybenzoic Acid, a known metabolite of all of the other ingredients in this report), and its salts. The basic paraben structure is provided in Figure 1, and an example of a specific paraben (Butylparaben) is provided in Figure 2.
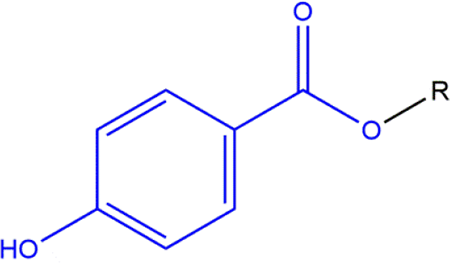
Paraben phenolic acids: a generic structure wherein R is an alkyl group from 1 to 4 carbons long or is benzyl.
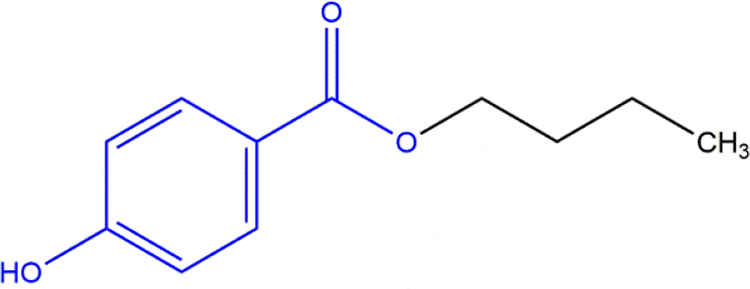
Paraben phenolic acids: an example, Butylparaben (wherein R from the generic structure in Figure 1 is an alkyl group 4 carbons long).
The salts of these phenolic acids have been included in this review of parabens. The phenolic proton is the most acidic in those parabens with an ester functional group, and the salt forms of these parabens share this same core structure (Figure 3). An example of a specific paraben salt (Potassium Butylparaben) is provided in Figure 4.
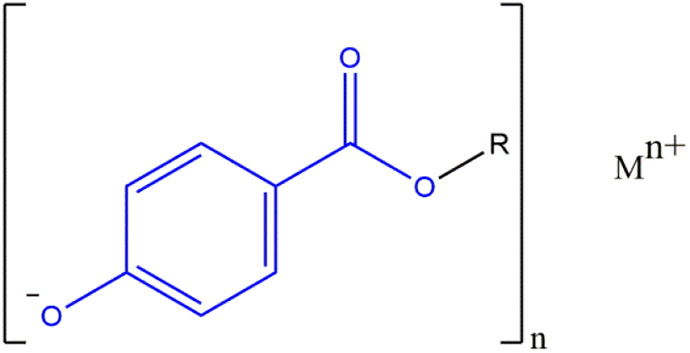
Paraben phenolic salts: generic structure wherein R is an alkyl group from 1 to 4 carbons long and M is sodium or potassium.
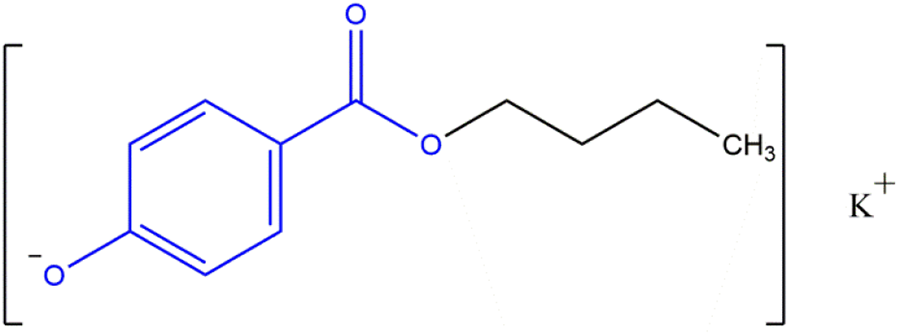
Paraben phenolic salts: an example, Potassium Butylparaben (wherein R, from the generic structure in Figure 3, is an alkyl group 4 carbons long and M is potassium).
Also included in this rereview are the free paraben carboxylic acid and its salts (ie, not esters). The carboxylic proton (of 4-Hydoxybenzoic Acid) is the most acidic in those parabens without an ester functional group, and the salt forms of these parabens share this same core structure (Figure 5). An example of a specific paraben carboxylic salt (Calcium Paraben) is provided in Figure 6.
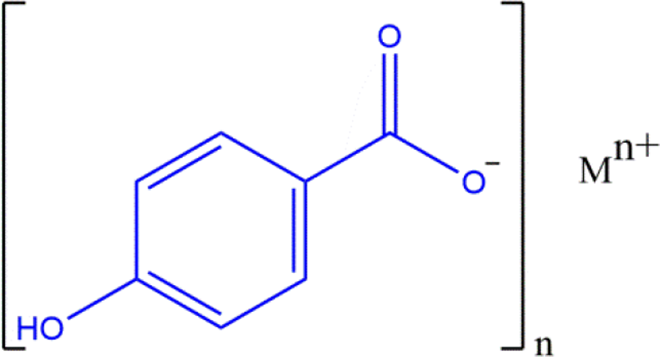
Paraben carboxylic salts: a generic structure wherein M is sodium, potassium, or calcium.
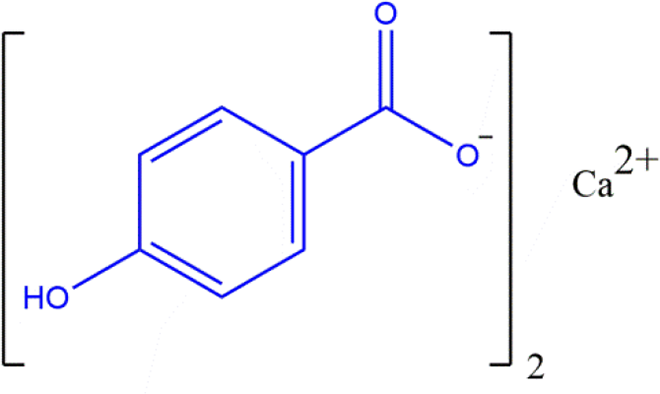
Paraben carboxylic salts: an example, Calcium Paraben (wherein M, from the generic structure in Figure 5, is calcium and n is 2).
Physical and Chemical Properties
Physical and chemical properties of parabens are presented in Table 3. Parabens form small colorless crystals or white crystalline powders with practically no odor or taste. 2 Parabens are soluble in alcohol, ether, glycerin, and propylene glycol and slightly soluble or almost insoluble in water. As the alkyl chain length increases, water solubility decreases. Parabens are hygroscopic and have a high oil/water partition coefficient. Parabens are relatively stable against hydrolysis during autoclaving and resist saponification. 21 The particle size distribution of some of the parabens included in the safety assessment is provided in Table 4. 4 -7,9,10,12,22 -24
Chemical and Physical Properties of Parabens.
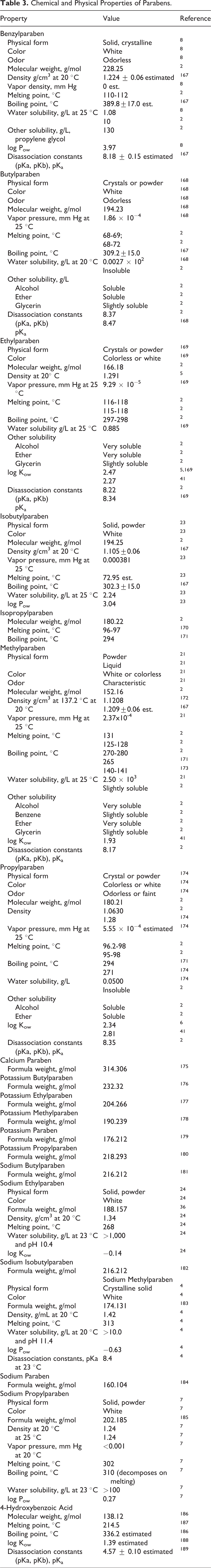
Particle Size Distribution of Paraben Raw Materials (ie, Prior to Formulation) in This Safety Assessment.
a D10 is the size below which 10% of the material is contained. Likewise, D50, D90, and D100 are the sizes at which 50%, 90%, and 100%, respectively, of the material is contained.
Method of Manufacture
Paraben phenolic acids (and salts) are prepared by esterifying 4-Hydroxybenzoic Acid with the corresponding alcohol (eg, butanol to synthesize Butylparaben) in the presence of an acid catalyst, such as sulfuric acid, and an excess of the specific alcohol. 2 The acid is then neutralized with caustic soda, and the product is crystallized by cooling, isolated by centrifugation, washed, dried under vacuum, milled, and blended. Benzylparaben can also be prepared by reacting benzyl chloride with sodium 4-Hydroxybenzoic Acid. Paraben carboxylate salts may be prepared by deprotonating 4-Hydroxybenzoic Acid with an appropriate alkaline salt (eg, sodium hydroxide could be used to prepare Sodium Paraben). 25
Use
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetic industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetic industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentration by product category.
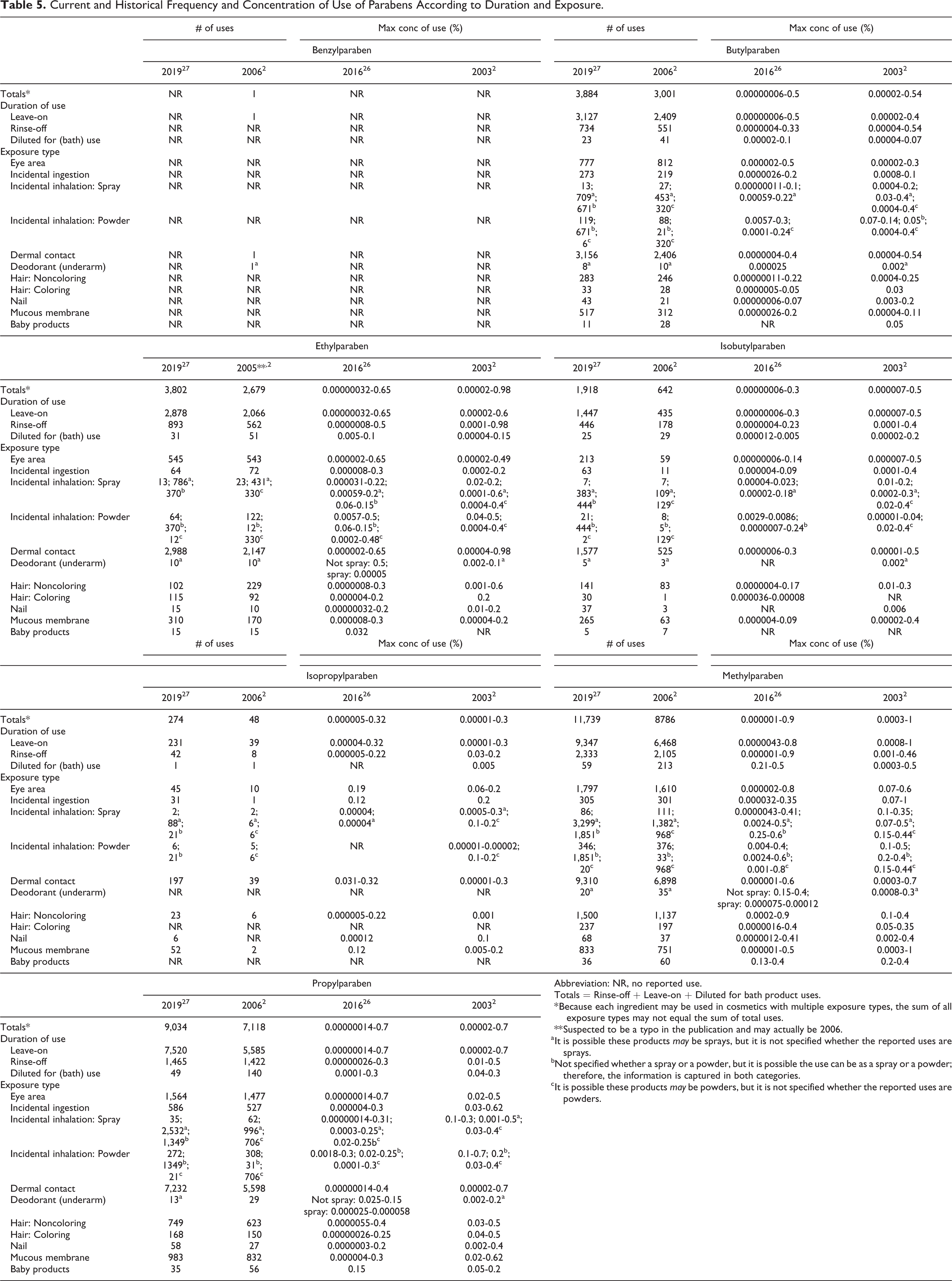
According to VCRP survey data received in 2019, Methylparaben was reported to be used in 11,739 formulations (9,347 of which are leave-on formulations); this is an increase from the 8,786 uses reported in 2006. 2,26,27 Propylparaben had the next highest number of reported uses at 9,034 (7,520 of which are leave-on formulations); this was an increase from 7,118 uses reported in 2006. All of the other previously reviewed parabens in this safety assessment increased in the number of reported uses since 2006 with the exception of Benzylparaben, which dropped from 1 reported use to none.
The results of the concentration of use survey conducted by the Council in 2016 indicate Methylparaben had the highest reported maximum concentration of use; it is used at up to 0.9% in shampoos. 2,26 The highest maximum concentration of use reported for products resulting in leave-on exposure is 0.8% Methylparaben in a mascara and for leave-on dermal exposure is 0.65% Ethylparaben in eye shadows. In 2006, Methylparaben had the highest reported maximum concentration of use at 1% in lipsticks. The maximum concentrations of use of the previously reviewed parabens have remained under 1% and the patterns of use are similar to those reported in the previous safety assessment.
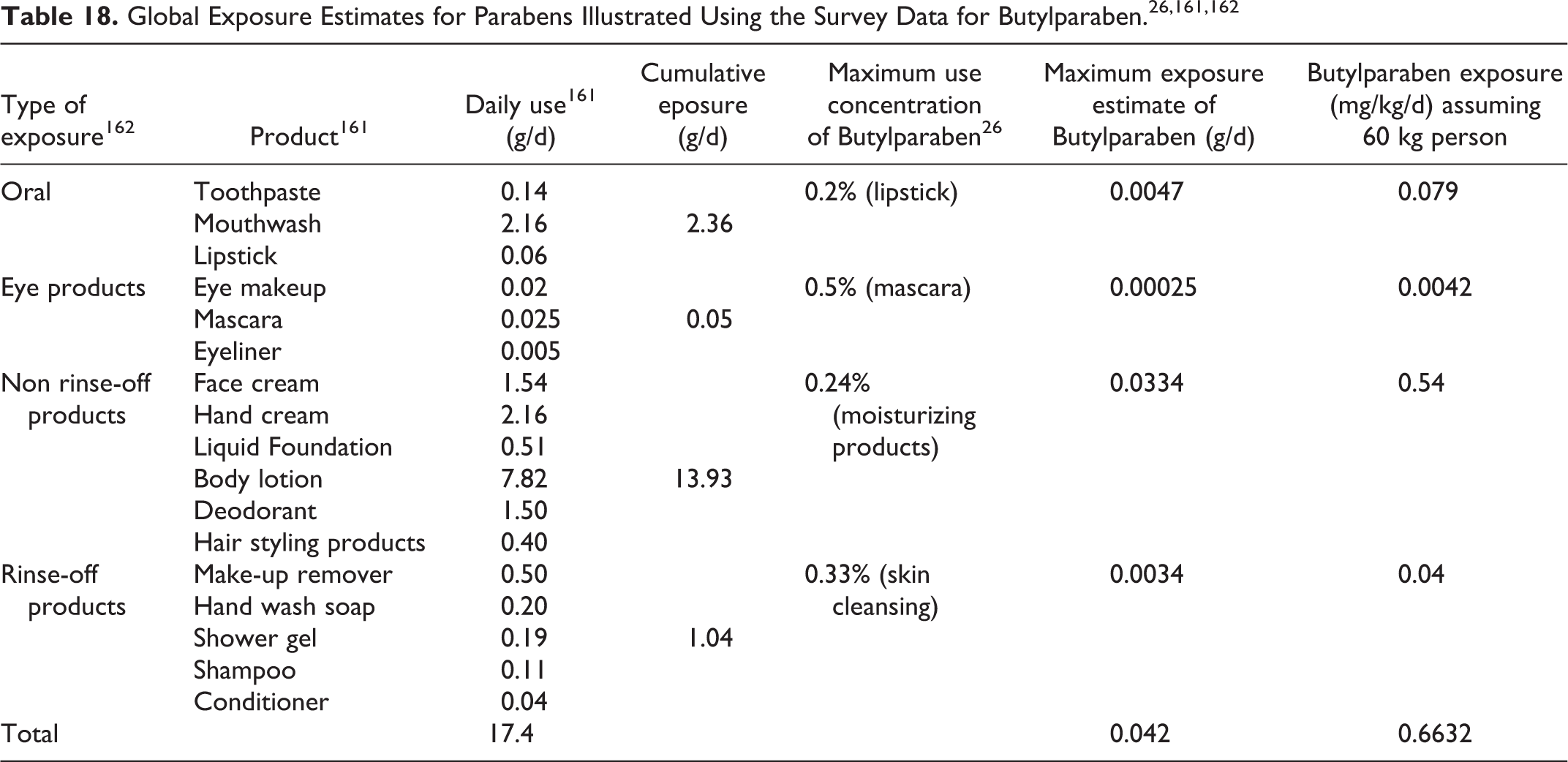
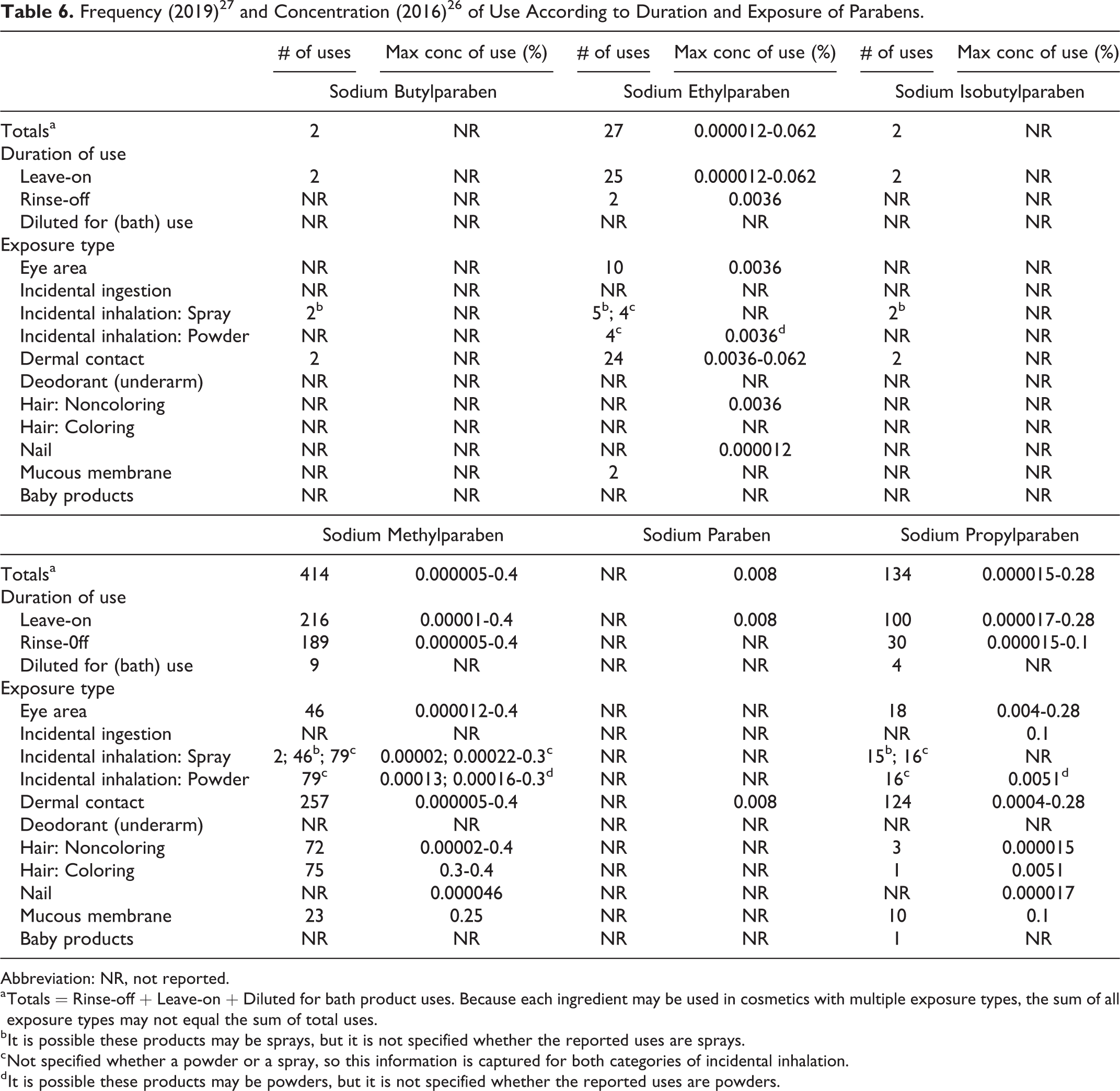
Frequency and concentration of use data for all ingredients reported to be in use are provided in Tables 5 and 6. The ingredients not in use, according to the VCRP and industry survey, are listed in Table 7.
Current and Historical Frequency and Concentration of Use of Parabens According to Duration and Exposure.
Frequency (2019) 27 and Concentration (2016) 26 of Use According to Duration and Exposure of Parabens.
Abbreviation: NR, not reported.
a Totals = Rinse-off + Leave-on + Diluted for bath product uses. Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
b It is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
c Not specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
d It is possible these products may be powders, but it is not specified whether the reported uses are powders.
Several of the parabens are reported to be used in products that can be incidentally ingested, used near the eye, come in contact with mucous membranes, or in baby products. 26,27 For example, Methylparaben is used at concentrations up to 0.35% in lipstick; 0.8% in mascara; 0.5% in bath oils, tablets, and salts; and 0.4% in baby lotions, oils, and creams.
Some of the parabens were reported to be used in cosmetic sprays (including hair sprays, hair color sprays, skin care products, moisturizing products, suntan products, deodorants, and other propellant and pump spray products) 26,27 and could possibly be inhaled. For instance, the maximum use concentration of Methylparaben in a fragrance product reported in the Council’s survey is 0.41%. Although there are reported mean diameters as small as 37.8 µm (Sodium Propylparaben) for some of these materials, as pure, raw substances, those diameters are not indicative of particle sizes in final formulations. 7 Accordingly, those raw material mean particle diameters are not relevant to cosmetic safety. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. 28 -30 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 28,30 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 28 The maximum concentration of use recorded for deodorant sprays was 0.00012% (Methylparaben). However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays. Some of the parabens were reported to be used in dusting powders and face powders (eg, Ethylparaben in face powders at up to 0.5%) and could possibly be inhaled. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1,000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace. 31 -33
The SCCS of the EU has published several opinions on parabens over the last few years (Table 8).
14
-20
The current SCCS opinion (updated on May 2013) is: The use of Butylparaben and Propylparaben as preservatives in finished cosmetic products are safe to the consumer, as long as the sum of their individual concentrations does not exceed 0.19%…With regard to Methylparaben and Ethylparaben, the previous opinion, stating that the use at the maximum authorized concentrations can be considered safe, remains unchanged…Limited to no information was submitted for the safety evaluation of isopropyl-, isobutyl-, phenyl-, benzyl- and pentylparaben. Therefore, for these compounds, the human risk cannot be evaluated. The same is true for benzylparaben…
18,20
SCCS/SCCP (Scientific Committee on Consumer Products, Predecessor of SCCS) Opinions on Parabens.

Based on SCCS opinions, the use of the different parabens is regulated by the EU Cosmetic Regulation, which has banned the use of Isopropylparaben, Isobutylparaben, phenylparaben, Benzylparaben, and pentylparaben as preservatives in cosmetic products 34 and has established maximum concentration limits of 0.4% for Methylparaben or Ethylparaben ([as acid] single esters and their salts), 0.14% for Propylparaben or Butylparaben ([as acid] single esters and their salts), and 0.8% for mixtures of the these 4 parabens, wherein the sum of the individual concentration of Butylparaben and Propylparaben and their salts does not exceed 0.14% (as acid). 34,35 In addition, “…Butylparaben and Propylparaben are prohibited in leave-on cosmetic products designed for application on the nappy area of children under 3 years of age…”
In Australia’s National Industrial Chemicals Notification and Assessment Scheme’s (NICNAS) Human Health Tier II Assessment for parabens, it was found that no critical health effects associated with these chemicals have been established; the chemicals have been shown to have weak estrogenic activity; however, there are no established adverse outcome pathways for this effect. 36 The available data do not indicate any risks associated with exposure to the chemicals in this group.
Noncosmetic
2008
The US FDA considers Methylparaben and Propylparaben to be generally recognized as safe (GRAS) as antimicrobial agents in food [21CFR184.1490; 21CFR184.1670]. Butylparaben, Ethylparaben, and Propylparaben are approved for direct addition to food for human consumption as synthetic flavoring substances and adjuvants [21CFR172.515]. Ethylparaben may be used as an indirect food additive as a component of adhesives and coatings [21CFR175.105]. Methylparaben and Propylparaben are prior sanctioned food ingredients when used as antimycotics [21CFR181.23]. Methylparaben and Propylparaben have been used in diaper rash products, but there are inadequate data to establish general recognition of the safety and effectiveness [21CFR310.545]. Methylparaben is GRAS as a chemical preservative in animal drugs, feeds, and related products at levels not to exceed 0.1% [21CFR582.3490]. Residual Methylparaben and Propylparaben are not to exceed 0.1% when used as preservatives in pesticides for food [40CFR180.930].
In many pharmaceuticals, parabens are used as excipients (inactive ingredients). In the US FDA database of inactive ingredients, Methylparaben has been approved at a maximum potency of 1.8 mg in a tablet formulation and 2.6 mg/mL in an oral solution. Ethylparaben has been approved at a maximum potency of 0.6 mg in a granule formulation and 0.6 mg/mL in an oral solution. Propylparaben has been approved for use at a maximum potency of 0.2 mg in a tablet formulation and 0.2 mg/mL in an oral solution. Butylparaben has been approved for use at a maximum potency of 0.04 mg in a sustained action tablet formulation and 0.08 mg/mL in an oral solution. 37
An evaluation by the JECFA determined that the acceptable daily intake (ADI) of the sum of the Ethylparaben and Methylparaben is up to 0 to 10 mg/kg. 38 In view of the adverse effects in male rats, Propylparaben was excluded from the ADI for use in food. 18
The NICNAS published a conclusion in 2016, indicating that “current risk management measures are considered adequate to protect public and workers’ health and safety, provided that all requirements are met under workplace health and safety, and poisons legislation as adopted by the relevant state or territory.” 36
Toxicokinetic Studies
Dermal Penetration
2008
Parabens in cosmetic formulations applied to skin penetrate the stratum corneum (SC) in inverse relation to the ester chain length. 2 Carboxylesterases present in keratinocytes and hydrolyze parabens in the skin. The extent of the breakdown to 4-Hydroxybenzoic Acid is different between rodent and human skin. In vitro studies also indicate a difference in the extent of hydrolysis to 4-Hydroxybenzoic Acid, depending on whether viable whole skin or dermatomed human skin is used, with the former having a larger extent of hydrolysis. Chemicals that disrupt the SC may increase the skin penetration of Methylparaben and possibly Ethylparaben but do not affect the penetration of parabens with longer ester chains.
In Vitro
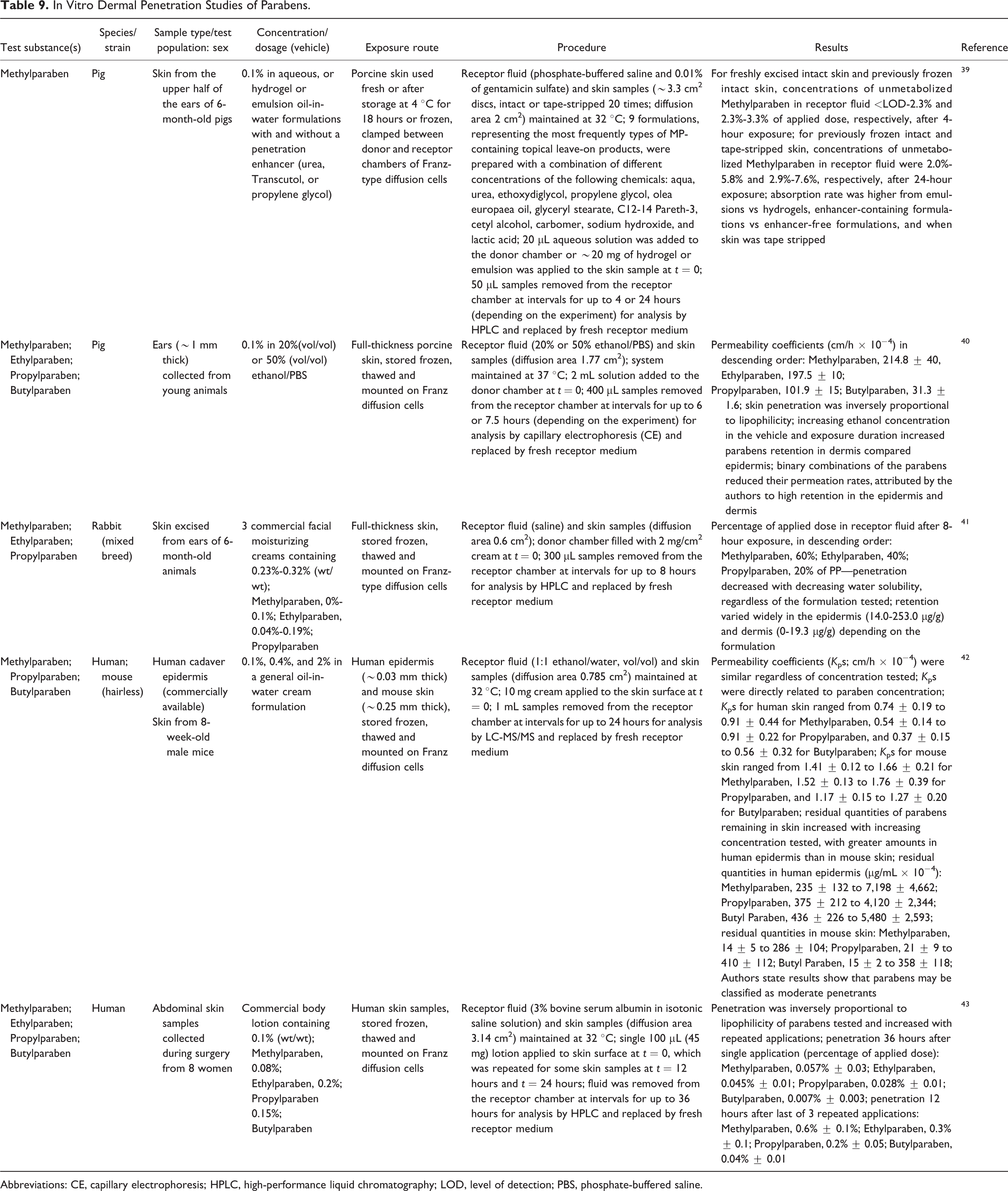
In vitro dermal penetration studies are presented in Table 9. In Franz-type diffusion cells, 2.3% to 3.3% of the applied dose of Methylparaben (0.1% in 9 different vehicles) penetrated porcine skin (intact stored frozen) in 4 hours. 39 The receptor fluid consisted of phosphate-buffered saline (PBS; pH 7.4) and 0.01% of gentamicin sulfate. In 24 hours, 2.0% to 5.8% and 2.9% to 7.6% of unmetabolized Methylparaben penetrated previously frozen intact and tape-stripped skin, respectively. In full-thickness porcine skin stored frozen, permeability coefficients ranged from 31.3 ± 1.6 cm/h × 10−4 to 214.8 ± 40 cm/h × 10−4, decreasing (Methylparaben > Ethylparaben > Propylparaben > Butylparaben) with increasing chain length. 40 Increasing the ethanol concentration in the vehicle or the exposure duration increased the retention of the parabens in the dermis relative to the epidermis. Binary combinations of the parabens reduced their permeation rates, which was attributed by the authors to high retention in the epidermis and dermis.
In Vitro Dermal Penetration Studies of Parabens.
Abbreviations: CE, capillary electrophoresis; HPLC, high-performance liquid chromatography; LOD, level of detection; PBS, phosphate-buffered saline.
In a different study, the penetration of parabens from 3 commercial facial cream formulations through rabbit ear skin ranged from 20% to 60%, after 8 hours in Franz-type diffusion cells, increasing with the water solubility of the paraben (Propylparaben < Ethylparaben < Methylparaben), regardless of the formulation tested. 41 Retention varied widely in the epidermis and dermis depending on the formulation.
Permeability coefficients estimated for Methylparaben, Propylparaben, and Butylparaben in human cadaver skin (0.37-0.91 cm/h × 10−4) and mouse skin (1.17-1.76 cm/h × 10−4) were similar regardless of concentration tested (0.1%-2%). 42 Residual quantities of parabens remaining in the skin increased as the test concentration increased, with greater amounts in the human epidermis than in mouse skin.
Human abdominal skin samples were used to determine the dermal penetration of 0.1% Methylparaben, 0.08% Ethylparaben, 0.2% Propylparaben, and 0.15% Butylparaben.
43
Previously frozen skin samples were thawed and mounted on Franz diffusion cells. A dose of 100 µL of lotion containing the test substance was applied to the skin once at
Human
Butylparaben
Dermal penetration was studied in 26 healthy Caucasian male volunteers aged 21 to 36 years old, after application of 2% (wt/wt) Butylparaben in a basic cream formulation which also contained 2% diethyl phthalate and 2% dibutyl phthalate. 44 Daily whole-body topical application of 2 mg/cm2 of the cream formulation without the test substances for 1 week (control week) was followed by daily application of the cream with the test substances for 1 week. Butylparaben serum concentrations in the blood were undetectable in most samples during the control week, with maximum concentrations not exceeding 1.0 µg/L. Butylparaben concentrations increased rapidly (mean peak concentration = 135 ± 11 µg/L in 3 hours) after the first application of cream containing the 3 test compounds. Twenty-four hours after the first application, but before the following application, the mean serum concentration was 18 ± 3 µg/L. Butylparaben could be detected in most serum samples collected throughout the second week of this study.
Penetration Enhancement
In vitro
Methylparaben
Skin samples were collected within 24 hours postmortem from the back of a 77-year-old woman and leg of a 73-year-old man and stored frozen.
45
Split thickness (∼350 µm) samples were thawed and mounted in vertical-flow Neoflon diffusion cells and exposed to a saturated aqueous solution of Methylparaben, with (saturated) and without 4-cyanophenol (CP). Receptor fluid (PBS) and skin samples (diffusion area 0.64 cm2) were maintained at 32 °C. Solutions containing one or both compounds were added to the donor chamber at
Absorption, Distribution, Metabolism, and Excretion
1984
Parabens are quickly absorbed from the blood and gastrointestinal tract, hydrolyzed to 4-Hydoxybenzoic Acid, conjugated, and the conjugate excreted in the urine. 46 Data obtained from chronic administration studies indicate that parabens do not accumulate in the body. Serum concentrations of parabens, even after intravenous administration, quickly decline and remain low. Varying amounts of parabens are passed in the feces depending upon which paraben is administered and the size of the dose. Little or no unchanged paraben is excreted in the urine. Most of an administered dose can be recovered within 5 to 72 hours as 4-Hydoxybenzoic Acid or its conjugates. Parabens appear to be rapidly absorbed through intact skin.
1986
Metabolism of Benzylparaben is by sulfate conjugation of the parent compound. 47 Excretion is in the urine. Small amounts of the ester are excreted unmetabolized or hydrolyzed to the benzyl alcohol and 4-Hydoxybenzoic Acid.
1995
When male rabbits were administered either 800 or 400 mg/kg of Isobutylparaben via a stomach tube, 77% to 85% of the ingredient was recovered as a form of 4-Hydoxybenzoic Acid; 20% was not recovered. 48
2008
Ingested parabens are quickly absorbed from the gastrointestinal tract, hydrolyzed to 4-Hydoxybenzoic Acid, conjugated, and the conjugate excreted in the urine. 2 Data obtained from chronic administration studies indicate that parabens do not accumulate in the body. Serum concentrations of parabens, even after intravenous administration, quickly decline and remain low. Varying amounts of parabens are passed in the feces depending upon which paraben is administered and the size of the dose. Little or no unchanged paraben is excreted in the urine. The Absorption, Distribution, Metabolism, and Excretion studies summarized below are presented in Table 10.
Toxicokinetic Studies: Absorption, Distribution, Metabolism, Excretion (ADME).

Abbreviations: AFP, α-Fetoprotein; Clint, intrinsic clearance; DMSO, dimethyl sulfoxide; ESI, electrospray ionization; GM, geometric mean; HHA, 4 hydroxyhippuric acid; HLC, human liver cytosol; HLM, human liver microsomes; HPLC, high-performance liquid chromatography; HSC, human skin cytosol; HSM, human skin microsomes; LC, liquid chromatography; LOQ, limit of quantification; MS/MS, tandem mass spectrometry; PBS, phosphate-buffered saline; RLC, rat liver cytosol; RLM, rat liver microsomes; RSM, rat skin microsomes; RSC, rat skin cytosol; SRM, selected reaction monitoring; UDP, uridine 5′-diphospho; UGT UDP, glucuronosyltransferase.
In Vitro
Methylparaben, Ethylparaben, and Propylparaben did not exhibit binding affinity for α-fetoprotein (AFP). 49 On the other hand, the 50% inhibitory concentration (IC50) of Benzylparaben was 0.012 µM. Butylparaben was de-esterified to 4-Hydoxybenzoic Acid in the S9 fraction of skin obtained from 5-week-old male rats, with a maximum rate at saturating concentration (Vmax) of 8.8 nmol/min/mg protein. 50
Methylparaben and Ethylparaben were stable in human plasma, but Propylparaben, Butylparaben, and Benzylparaben concentrations decreased by 50% within 24 hours. 51 All parabens tested were rapidly hydrolyzed when incubated with human liver microsomes (HLMs), with rates depending on the alkyl chain length. Parabens, but not 4-Hydroxybenzoic Acid, were actively glucuronidated by liver microsomes and human recombinant uridine-5′-diphospho-glucuronosyltransferases (UGTs).
Methylparaben, Ethylparaben, Propylparaben, and Butylparaben were hydrolyzed by rat liver microsomes (RLMs) and HLM in in vitro tests. 52 Butylparaben was most effectively hydrolyzed by the RLM, which showed relatively low hydrolytic activity toward parabens with shorter and longer alkyl side chains. In contrast to RLM, HLM showed the highest hydrolytic activity toward Methylparaben, with activity decreasing with increasing side chain length of the paraben tested. Rat small intestinal microsomes exhibited relatively higher activity toward longer side-chain parabens. Human small intestinal microsomes showed a specificity pattern similar to that of rat small intestinal microsomes.
Metabolism rates of Methylparaben, Ethylparaben, Propylparaben, and Butylparaben by HLM were inversely proportional to chain length (overall rate dominated by esterase-catalyzed hydrolysis, where the longer the alcohol moiety, the slower the hydrolysis). 53 This trend was also observed for human skin microsomes (HSMs), but at much lower rates. Paraben metabolism in HLMs was 300- to 500-fold faster than in HSM, depending on the paraben. In contrast to human tissue fractions, the rat tissue fractions tested, including skin and liver fractions, hydrolyzed the parabens at rates that increased as the ester chain length increased. Rat skin displayed 3 to 4 orders of magnitude faster hydrolysis rates than human skin.
Butylparaben was rapidly cleared in hepatocytes from rats and was cleared more slowly in hepatocytes from humans, with little or no sex difference. 54 Butylparaben was extensively hydrolyzed to 4-Hydroxybenzoic Acid as the major metabolite for both sexes and species. The other metabolite observed in the human hepatocytes was 4-hydroxyhippuric acid, which is the glycine conjugate (ie, a phase II metabolite) of 4-Hydroxybenzoic Acid.
Animal
Dermal
Nine rats were given a single dermal dose of 100 mg/kg bw 4-hydroxy [ring-U-14C]-labeled Methylparaben, Propylparaben, or Butylparaben in 60% aqueous ethanol vehicle. Cmax (≥693 and ≥614 ng Eq/g in males and females, respectively) occurred within 8 hours postapplication, and blood concentrations decreased until the last quantifiable concentration within 24 hours. 55 Most of the dosage (≥46.4%) was not absorbed, and less than 25.8% was found in the urine. About 52% and 8% of a single 10 or 100 mg/kg bw dosage, respectively, of [14C]-Butylparaben was absorbed 72 hours following application to the skin in rats. 54 Urine was the primary route of elimination. Tissues contained about 4.3% of the 10 mg/kg dosage. The kidneys contained about twice the concentration of residues found in the liver.
Oral
In rats exposed to a single oral dosage of 100 mg/kg bw [ring-U-14C]-labeled Methylparaben, Propylparaben, or Butylparaben, Cmax (≥11,432 and ≥21,040 ng Eq/g in males and female, respectively) occurred within 1 hour postgavage, and blood concentrations decreased until the last quantifiable concentration at 12 hours. 55 Radioactivity was eliminated rapidly, with averages ≥69.6% recovered in the urine during the first 24 hours. Radioactivity was excreted predominantly in urine in rats orally exposed to a single 10, 100, or 1,000 mg/kg bw/d dosage of [14C]-Butylparaben. 54 The rate of urinary excretion was similar across all dosages, with ≥66% recovered in the first 24 hours in males. Female rats excreted more Butylparaben in urine in the first 4 hours after exposure, but there was no sex difference in the total dose excreted within 24 hours.
Time-mated female SD rats were orally administered 0, 1,500, 5,000, or 15,000 ppm Butylparaben via NIH-07 feed, ad libitum, from gestation day (GD) 6 to postnatal day (PND) 28. 56 Dam plasma, amniotic fluid, and fetuses were collected on GD 18 and plasma from both the pup and dam were collected on PNDs 4, 10, 14, 21, and 28 and analyzed for free (unconjugated) and total (unconjugated and conjugated) Butylparaben. Free Butylparaben was below the limit of quantitation in fetuses (LOQ 1.91 ng Butylparaben/g fetus) and amniotic fluid (LOQ 0.17 ng Butylparaben/mL amniotic fluid) at 1,500 ppm. Analyte levels in amniotic fluid were less than 1% of maternal plasma, suggesting limited placental transfer. The total Butylparaben in PND 4 pup plasma was less than 5% of dam plasma in all exposure groups, suggesting low lactational transfer. However, at nearly all time points and exposure groups, there were higher levels of free Butylparaben in pup versus dam plasma, suggesting limited conjugation in pups. Pup conjugation of Butylparaben was age-dependent, not reaching the percentage conjugation in dams (>99%) until PNDs 21 to 28. These data illustrate low placental and lactational transfer of dietary Butylparaben and that poor conjugation in pups during early lactation results in higher exposure to free Butylparaben in pups compared to dams.
Human
Dermal
All 26 male volunteers showed increased excretion of Butylparaben following daily whole-body topical application of a cream formulation containing 2% (wt/wt) Butylparaben, 2% diethyl phthalate, and 2% dibutyl phthalate. 57 Mean total Butylparaben excreted in urine during exposure was 2.6 ± 0.1 mg/24 hours. The concentrations peaked in the urine 8 to 12 hours after application.
Oral
Free and conjugated parabens and their major, nonspecific metabolites (4-Hydroxybenzoic Acid and
Physiologically Based Pharmacokinetic Modeling
In one study, a physiologically based pharmacokinetic (PBPK) model was developed and used to estimate the plasma free paraben concentration in adults consistent with 95th percentile urine concentration reported in US National Health and Nutrition Examination Survey (NHANES) program (2009-2010 collection period). 59 For the 2009 to 2010 sampling period, the predicted plasma free concentration of Methylparaben, Propylparaben, and Butylparaben in a 70-kg male was 0.73, 0.21, and 0.052 µg/L, respectively; the predicted plasma free concentration of Methylparaben, Propylparaben, and Butylparaben in a 60-kg female was 1.19, 0.54, and 0.58 µg/L, respectively. An in vitro-based cumulative MOS was calculated by comparing the effective concentrations from an in vitro assay of estrogenicity to the predicted free plasma paraben concentrations (Methylparaben + Ethylparaben + Butylparaben). The calculated cumulative MOS for adult females was 108, whereas the cumulative MOS for males was 444.
Toxicological Studies
Acute Dose Toxicity
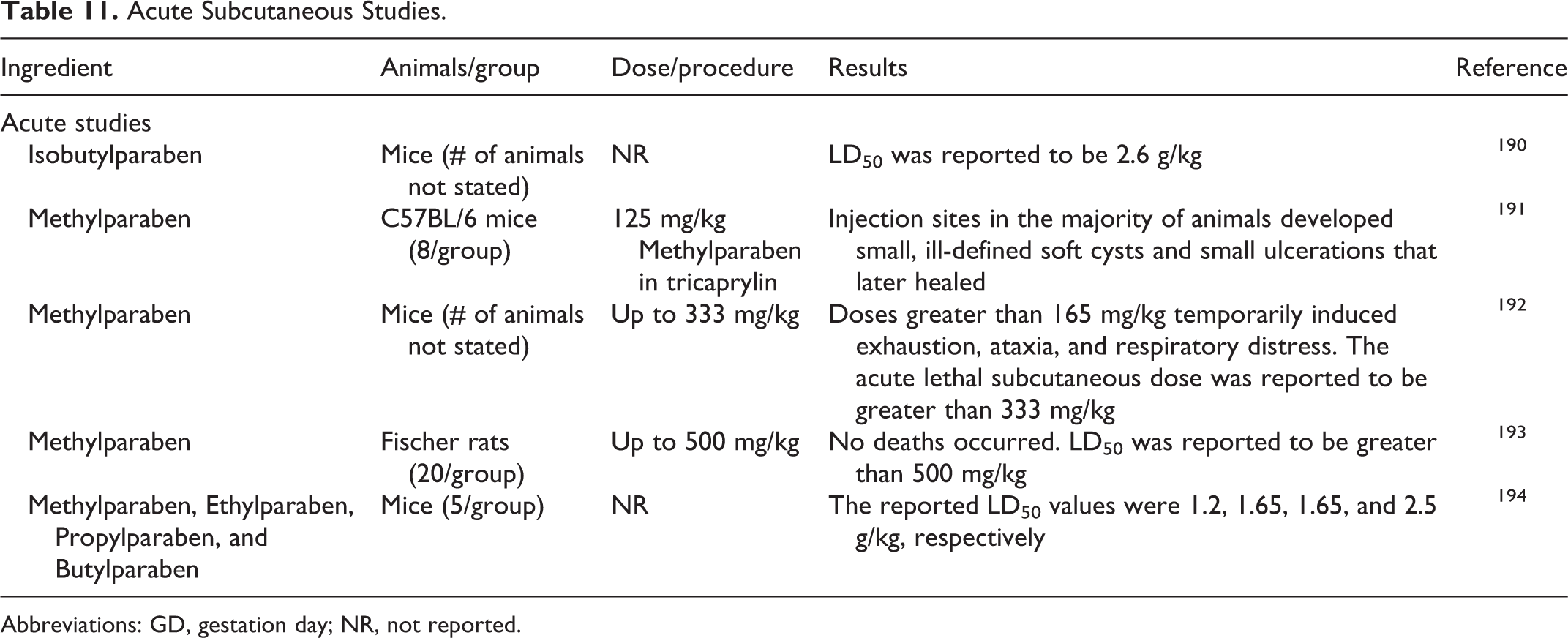
No new published oral or dermal acute toxicity studies were discovered in the published literature, and no unpublished data were submitted. Acute subcutaneous studies are summarized in Table 11.
Acute Subcutaneous Studies.
Abbreviations: GD, gestation day; NR, not reported.
1984
Acute toxicity studies in animals indicate that parabens are practically nontoxic by various routes of administration. 46
1986
Benzylparaben was not considered an acute toxic agent to mice or rats. Intravenous injections of Benzylparaben to dogs and cats caused no variation in blood sugar, circulation, and respiration. 47
1995
Isobutylparaben had a subcutaneous LD50 of 2,600 mg/kg in mice. 48
Short-Term Toxicity Studies
1995
No significant histological changes were observed in mice dosed with 0.6% Isobutylparaben in the feed for 6 weeks. Mice dosed with 1.25% had atrophy of the spleen, thymus, and lymph nodes as well as multifocal degeneration and necrosis of the hepatic parenchyma. Mice dosed with 5% and 10% Isobutylparaben died within the first 2 weeks of the study. 48
2008
Ethylparaben, Propylparaben, and Butylparaben in the diet produced cell proliferation in the forestomach of rats, with the activity directly related to chain length of the alkyl chain. 2 Fischer 344 male rats were treated by Methylparaben, Ethylparaben, Propylparaben, and Butylparaben at 4% for 9 to 27 days in the dry diet, and the magnitude of the proliferative effect in the prefundic area of the forestomach epithelium elevated as the alkyl chain length increases. The short-term toxicity studies that are summarized below are presented in Table 12.
Short-Term Toxicity Studies.
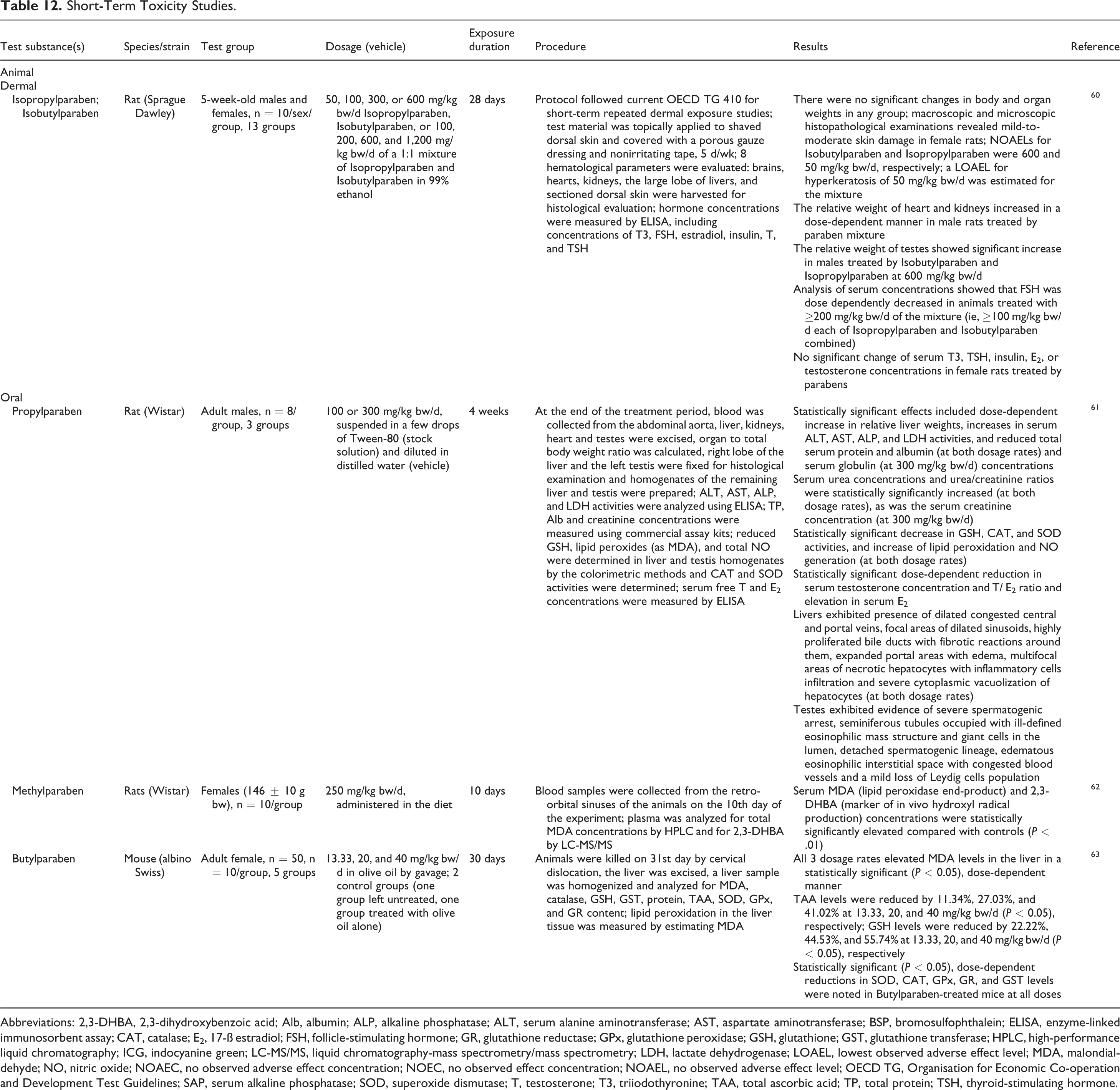
Abbreviations: 2,3-DHBA, 2,3-dihydroxybenzoic acid; Alb, albumin; ALP, alkaline phosphatase; ALT, serum alanine aminotransferase; AST, aspartate aminotransferase; BSP, bromosulfophthalein; ELISA, enzyme-linked immunosorbent assay; CAT, catalase; E2, 17-ß estradiol; FSH, follicle-stimulating hormone; GR, glutathione reductase; GPx, glutathione peroxidase; GSH, glutathione; GST, glutathione transferase; HPLC, high-performance liquid chromatography; ICG, indocyanine green; LC-MS/MS, liquid chromatography-mass spectrometry/mass spectrometry; LDH, lactate dehydrogenase; LOAEL, lowest observed adverse effect level; MDA, malondialdehyde; NO, nitric oxide; NOAEC, no observed adverse effect concentration; NOEC, no observed effect concentration; NOAEL, no observed adverse effect level; OECD TG, Organisation for Economic Co-operation and Development Test Guidelines; SAP, serum alkaline phosphatase; SOD, superoxide dismutase; T, testosterone; T3, triiodothyronine; TAA, total ascorbic acid; TP, total protein; TSH, thyroid-stimulating hormone.
Dermal
There were no significant changes in body and organ weights in any group when rats were dermally exposed to up to 600 mg/kg bw/d Isopropylparaben or Isobutylparaben for 28 days. 60 Macroscopic and microscopic examinations revealed mild to moderate skin damage in female rats treated by Isobutylparaben or Isopropylparaben at doses higher than 600 or 50 mg/kg bw/d, respectively. The weights of testes were significantly increased in male rats given a 1:1 mixture of Isobutylparaben and Isopropylparaben at doses of 600 or 1,200 mg/kg bw/d. Follicle-stimulating hormone (FSH) concentration was dose dependently decreased in males treated with a mixture of Isobutylparaben and Isopropylparaben at a dose of 100 mg/kg bw/d or higher. The NOAELs for Isobutylparaben and Isopropylparaben for female skin damage were 600 and 50 mg/kg bw/d, respectively.
Oral
At 100 and 300 mg/kg bw/d Propylparaben administered orally for 4 weeks, adult rats exhibited statistically significant increases in relative liver weights, serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and lactate dehydrogenase (LDH) activities, serum urea concentrations, lipid peroxidation and nitric oxide (NO) generation, and 17β-estradiol (E2) concentrations. 61 Statistically significant decreases in total serum protein and albumin, glutathione (GSH), catalase (CAT), and superoxide dismutase (SOD) activities, serum testosterone (T) concentrations, and T/E2 ratios were also reported. Livers of affected rats exhibited dilated congested central and portal veins, highly proliferated bile ducts with fibrotic reactions, and multifocal areas of necrotic hepatocytes, and testes exhibited evidence of severe spermatogenic arrest, among other effects. Elevations of serum markers of lipid peroxidase (ie, malondialdehyde) and hydroxyl radical production were statistically significant in rats exposed to 250 mg/kg bw/d Methylparaben. 62 Malondialdehyde levels were elevated in the liver in a statistically significant, dose-dependent manner, among other effects, in mice orally exposed to 1.33 to 40 mg/kg bw/d Butylparaben for 30 days. 63
Subchronic Toxicity Studies
No new published subchronic toxicity studies were discovered in the published literature, and no unpublished data were submitted.
1984
Subchronic oral studies indicate that parabens are practically nontoxic. 46 A subchronic oral toxicity study in humans indicated that Methylparaben was practically nontoxic at doses up to 2 g/kg/d.
Chronic Toxicity Studies
No new published chronic toxicity studies were discovered in the published literature, and no unpublished data were submitted.
1984
Chronic oral studies indicate that parabens are practically nontoxic. 46 A 60:40 mixture of the sodium salts of Propylparaben and Ethylparaben did not induce significant pathologic changes in rats treated at 1.4 g/kg bw/d for 18 months. At 2% of the diet, Methylparaben and Propylparaben exerted no toxic effect in rats after 96 weeks’ exposure. Weanling dogs treated by Methylparaben or Propylparaben at 1 g/kg bw/d for 378 to 422 days were in excellent condition throughout the experiment.
1995
Mice were orally dosed with 0.15, 0.3, and 0.6% Isobutylparaben in the feed for 102 weeks. 48 Upon necropsy, the only effect noted was amyloidosis in 58% of dosed males and 33% of dosed females surviving past 78 weeks, as compared with 25% of control males and 10% of control females.
Developmental and Reproductive Toxicity Studies
1984
Methylparaben was nonteratogenic in rabbits, rats, mice, and hamsters, and Ethylparaben was nonteratogenic in rats. 46 Pregnant animals were given orally 5.0 to 550 mg/kg bw/d (rats, mice) or 3.0 to 300 mg/kg bw/d (hamsters) Methylparaben from day 6 of gestation to day 10 (hamsters) or 15 (rats, mice). Pregnant rabbits were orally administered 3.0 to 300 mg/kg bw/d Methylparaben daily from day 6 of gestation to day 18. Pregnant rats were dosed in diet of Ethylparaben at concentrations of 0.1, 1, or 10% between GDs 8 and 15. On day 21 of pregnancy, rats were killed, and the number of fetal implantations, status of maternal visceral organs, fetal body weights, and numbers of skeletal, visceral, and external defects in fetuses were recorded. No apparent teratogenesis or toxicity was observed in 363 fetuses from rats fed up to10% Ethylparaben.
At the 10% level, cerebral hemorrhages, abnormal enlargement in the ventricles of the brain, and, in some, hydronephrosis and hypo-osteogenesis were observed in fetuses. Some fetuses at 1% Ethylparaben had no blood in the cardiac ventricle; some had intraperitoneal hemorrhages. Fetuses of rats of the 0.1% group had no significant visceral or skeletal defects.
2008
Methylparaben was nonteratogenic in rabbits, rats, mice, and hamsters, and Ethylparaben was nonteratogenic in rats. 2 Parabens, even at levels that produce maternal toxicity, do not produce terata in animal studies. One study examined the developmental toxicity of Butylparaben in rats and reported no effect on development up to an oral dose of 1,000 mg/kg bw/d, even with some maternal toxicity at that dose. The maternal toxicity NOAEL dose was 1,000 mg/kg bw/d.
Parabens have been extensively studied to evaluate male reproductive toxicity. In one in vitro study, sperm viability was eliminated by concentrations as low as 6 mg/mL Methylparaben, 8 mg/mL Ethylparaben, 3 mg/mL Propylparaben, or 1 mg/mL Butylparaben, but an in vivo study of 0.1% or 1.0% Methylparaben or Ethylparaben in the diet of mice for 8 weeks reported no spermatotoxic effects. Propylparaben did affect sperm counts at all levels from 0.01% to 1.0% (approximately 10 and 1,000 mg/kg bw/d, respectively). Epididymis and seminal vesicle weight decreases were reported in rats given a 1% oral Butylparaben dose, and decreased sperm number and motile activity in F1 offspring of rats maternally exposed to 100 mg/kg bw/d were reported. Decreased sperm numbers and activity were reported in F1 offspring of female rats exposed to Butylparaben subcutaneously at 100 or 200 mg/kg bw/d, but there were no abnormalities in the reproductive organs. The total treatment period was from GD 6 to PD 20, with a 2-day interruption at parturition.
Methylparaben was studied using male rats at levels in the diet up to 10,000 ppm (estimated mean dose of 1,141.1 mg/kg/d) with no adverse effects. Butylparaben was studied using rats at levels in the diet up to 10,000 ppm (estimated mean dose of 1,087.6 mg/kg/d) in a repeat of the study noted above, but using a larger number of animals and a staging analysis of testicular effects. Rats received Butylparaben in the diet for a minimum of 56 days. No adverse reproductive effects were found.
Butylparaben, administered subcutaneously at 2 mg/kg bw/d in male rats on PDs 2 to 18, produced only minor effects on epithelial cell height. No effect of Butylparaben on the expression of the water channel protein aquaphorin-1 (APQ-1), efferent duct distension, or rete testis morphology was seen.
Dermal
No new published dermal DART studies were discovered and no unpublished data were submitted.
Oral
The oral DART studies summarized below are described in Table 13. Time-mated rats were orally exposed to 10, 100, or 500 mg/kg bw/d of Butylparaben from GD 7 to PND 22. 3 The AGD of newborn male and female offspring was significantly reduced at 100 or 500 mg/kg bw/d. The reduced expression of the Sertoli/Leydig cell marker Nr5a1 in adult male offspring was statistically significant at 10 mg/kg bw/d or above. In male offspring, epididymal sperm count decreased 76% to 78% compared to controls at all doses from 10 to 500 mg/kg bw/d. The reduction in epididymal sperm count showed the same effect at all doses (ie, no dose–response effect was observed). Adult prostate weight reductions were statistically significant at 500 mg/kg bw/d. In prepubertal females, ovary weight reduction was statistically significant and mammary gland outgrowth was increased at 100 and 500 mg/kg bw/d. No clear effect was seen on mammary glands of adult female offspring.
Developmental and Reproduction Toxicity (DART) Studies.
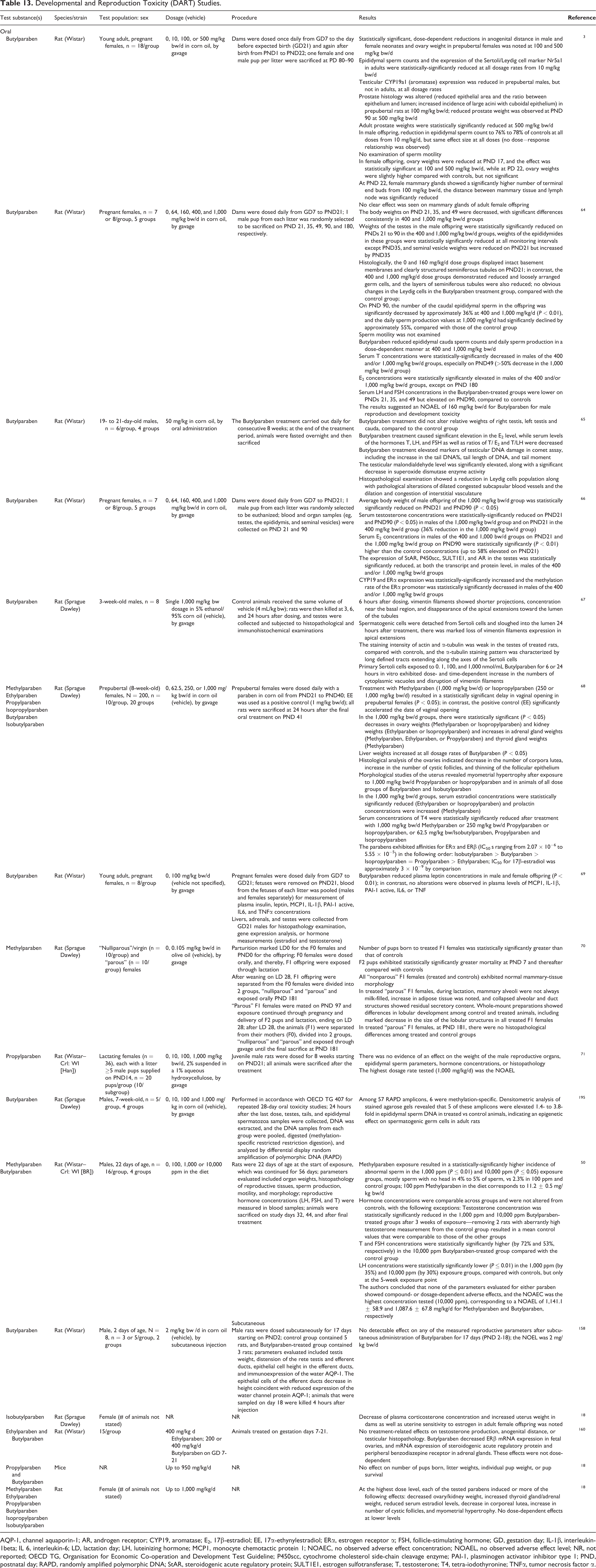
AQP-1, channel aquaporin-1; AR, androgen receptor; CYP19, aromatase; E2, 17β-estradiol; EE, 17α-ethynylestradiol; ERα, estrogen receptor α; FSH, follicle-stimulating hormone; GD, gestation day; IL-1β, interleukin-1beta; IL 6, interleukin-6; LD, lactation day; LH, luteinizing hormone; MCP1, monocyte chemotactic protein 1; NOAEC, no observed adverse effect concentration; NOAEL, no observed adverse effect level; NR, not reported; OECD TG, Organisation for Economic Co-operation and Development Test Guideline; P450scc, cytochrome cholesterol side-chain cleavage enzyme; PAI-1, plasminogen activator inhibitor type 1; PND, postnatal day; RAPD, randomly amplified polymorphic DNA; StAR, steroidogenic acute regulatory protein; SULT1E1, estrogen sulfotransferase; T, testosterone; T4, tetra-iodothyronine; TNFα, tumor necrosis factor α.
Pregnant rats were orally exposed to 64, 160, 400, or 1,000 mg/kg bw/d of Butylparaben from GD 7 to PND 21. 64 In the 400 and 1,000 mg/kg bw/d groups of male offspring, reduced AGD and delayed preputial separation (PPS) were observed; the weights of the testes were significantly reduced and serum T was reduced in a dose–response manner from PND 21 to PND 90. On PND 90, the number of the caudal epididymal sperm was significantly decreased by approximately 36% at 400 and 1,000 mg/kg bw/d, and daily sperm production values were significantly decreased. In contrast, weights of the testes, epididymal cauda sperm counts, serum T and luteinizing hormone (LH) levels, and daily sperm production in male offspring did not change at doses of 64 and 160 mg/kg bw/d.
Estradiol level was significantly elevated in weanling male rats orally exposed to Butylparaben at 50 mg/kg for 8 consecutive weeks, whereas serum levels of the hormones T, LH, and FSH, as well as ratios of T/E2 and T/LH were decreased, compared to control groups. 65 Butylparaben treatment elevated markers of testicular DNA damage in a comet assay, such as the increase in the tail DNA%, tail length of DNA, and tail moment. In addition, the testicular malondialdehyde level was significantly elevated, along with a significant decrease in CAT enzyme activity. Histopathological examination showed a reduction in Leydig cells population along with pathological alternations of dilated congested subcapsular blood vessels and the dilation and congestion of interstitial vasculature.
The increase in CYP19 and estrogen receptor (ER) α expression; the reduction in steroidogenic acute regulatory protein (StAR), cytochrome cholesterol side-chain cleavage enzyme (P450scc), estrogen sulfotransferase (SULT1E1), and testes androgen receptor (AR) expression; and the reduced methylation rate of the ERα promoter, were statistically significant in male offspring of female rats exposed to 400 or 1,000 mg/kg bw/d Butylparaben from GD 7 to GD 21. 66 Vimentin filaments showed shorter projections, concentration near the basal region, and disappearance of the apical extensions toward the lumen of the seminiferous tubules in 3-week-old rats 6 hours after a single 1,000 mg/kg bw oral dosage of Butylparaben. 67 Spermatogenic cells were detached from Sertoli cells and sloughed into the lumen 24 hours after treatment.
Prepubertal female rats were exposed orally to Methylparaben, Ethylparaben, Propylparaben, Isopropylparaben, Butylparaben, or Isobutylparaben in a dose-dependent manner (62.5, 250, and 1,000 mg/kg bw/d) on PND 21 to PND 40. Rats treated with 1,000 mg/kg bw/d Methylparaben or 250 mg/kg bw/d Isopropylparaben exhibited statistically significant delays in vaginal opening. 68 In the 1,000 mg/kg bw/d groups, there were statistically significant decreases in the weights of the ovaries (Methylparaben or Isopropylparaben) and kidneys (Ethylparaben or Isopropylparaben) and increases in the weights of the adrenal glands (Methylparaben, Ethylparaben, or Propylparaben) and thyroid glands (Methylparaben). Liver weights increased at all dosage rates of Butylparaben. Morphological studies of the uterus revealed myometrial hypertrophy after exposure to 1,000 mg/kg bw/d Propylparaben or Isopropylparaben and in animals of all dose groups of Butylparaben and Isobutylparaben. Among the statistically significant effects on serum hormone concentrations, E2 concentrations were reduced (Ethylparaben or Isopropylparaben) and prolactin concentrations were increased (Methylparaben) in the 1,000 mg/kg bw/d groups. Reduced plasma leptin concentrations were observed in male and female offspring of young adult female rats exposed orally to 100 mg/kg bw/d Butylparaben. 69
F2 pups exhibited a statistically significant greater mortality at PND 7 and thereafter, compared with controls, in a DART study in which F0 females and their F1 offspring were exposed to 0.105 mg/kg bw/d Methylparaben by gavage. 70 During lactation, treated “parous” F1 females exhibited mammary alveoli, which were not always milk-filled, collapsed alveolar and duct structures with residual secretory content, and marked decrease in the size of the lobular structures.
There was no evidence of an effect on the weight of the male reproductive organs, epididymal sperm parameters, hormone concentrations, or histopathology in juvenile male rats exposed via gavage receiving up to 1,000 mg/kg bw/d Propylparaben for 8 weeks. 71
Methylparaben was associated with a statistically significant higher incidence of abnormal sperm in rats exposed to 1,000 ppm or 10,000 ppm in the diet for 8 weeks, mostly sperm with no head in 4% to 5% of sperm, compared with 2.3% in 100 ppm and control groups. 50 Measurements of hormone concentrations were generally not altered, except that T and FSH concentrations were higher in the 10,000 ppm Butylparaben-treated group, compared with the control group. The authors concluded that the no observed adverse effect concentration was the highest concentration tested (10,000 ppm), corresponding to a NOAEL of about 1,140 and 1,100 mg/kg/d for Methylparaben and Butylparaben, respectively.
Histopathologic examination revealed progressive detachment and sloughing of spermatogenic cells into the lumen of the seminiferous tubules and reduction and/or disappearance of tubular lumen 3 hours after a single 1,000 mg/kg oral dosage of Butylparaben in rats. 72 Terminal deoxynucleotidyl transferase–mediated fluorescein-dUTP nick end labeling (TUNEL) assays revealed a substantial increase in the number of apoptotic spermatogenic cells in the treated rats; the effect was maximal at 6 hours.
Subcutaneous
Subcutaneous DART studies are also summarized in Table 13.
Aquatic
Zebrafish embryos were exposed to sublethal concentrations of Methylparaben: 0.1, 1, 10, and 100 ppb. A significant inhibition in the acetylcholinesterase activity, as well as an increase in cortisol levels, was observed in the exposed groups. 73 Alterations in developmental landmarks such as heart rate and hatching percentage were observed in embryos exposed to 10 ppb and 100 ppb of Methylparaben. Anxiety-like behavior was induced in larvae exposed to 0.1 and 1 ppb of Methylparaben.
Exposure of zebrafish embryos to Methylparaben at 200, 400, 800, and 1,000 μM for 96 hours postfertilization (hpf) resulted in decreased heart rate and hatching rate and developmental abnormalities, including pericardial edema blood cell accumulation and bent spine. 74 The 96 hpf LC50 of Methylparaben was 428 μM (0.065 mg/L) and expression of vitellogenin was significantly upregulated compared to the control group in larval zebrafish exposed to 100 μM (0.015mg/L) of Methylparaben till 96 hpf.
Genotoxicity Studies
1984
Numerous mutagenicity studies, including the Ames test, dominant lethal assay, host-mediated assay, and cytogenic assays, indicate that the Methylparaben, Ethylparaben, and Propylparaben are nonmutagenic. 46
1995
Chinese hamster fibroblast cell lines treated with 0.03% Isobutylparaben had no chromosomal aberrations after 48 hours.
48
At a concentration of 1 mg/plate, Isobutylparaben and Isopropylparaben had negative Ames tests in
2008
A number of genotoxicity studies suggest the Methylparaben, Propylparaben, Isopropylparaben, and Butylparaben are generally nonmutagenic. 2 Ethylparaben, Propylparaben, and Butylparaben induced 1% to 3% increases in polyploid cell production in an in vitro assay using Chinese hamster ovary (CHO) cells; Ethylparaben and Methylparaben were judged to induce significant chromosomal aberrations (11.0% and 15.0% increases, respectively) in the same study.
In Vitro
Methylparaben
Human spermatozoa were exposed to 13 mM Methylparaben for 2 or 5 hours. 75 Methylparaben had no significant effect on DNA fragmentation as measured by the TUNEL and the sperm chromatin dispersion assays in human spermatozoa. A statistically significant decrease in spermatozoa motility was observed after 2 and 5 hours. After 5 hours of exposure, a significant increase in the following parameters was observed in a time-dependent manner: Annexin V and fluorescently labeled inhibitor of caspase assay signals, mitochondrial and total superoxide generation, and 8-hydroxy-2′-deoxyguanosine (8OHdG) production. In contrast, Methylparaben at a concentration of 2.5 mM did not induce any significant changes to the motility, vitality, mitochondrial reactive oxygen species (ROS) production, and 8OHdG formation over the 5-hour time exposure period.
Propylparaben
Vero cells (derived from African green monkey kidney) were grown and incubated for 24 hours with 0, 50, 200, 300, 400, or 500 µM Propylparaben at 37 °C in Dulbecco’s modified Eagle medium (DMEM) supplemented with 5% fetal calf serum (FCS), 100 U/mL penicillin, 100 mg/mL streptomycin, and 2 mM
The CHO cells were grown and incubated for 1 or 3 hours with 0, 0.5, 1, 1.5, 2, or 2.5 µM Propylparaben. 77 Sister chromatid exchange (SCE), chromosome aberration (CA), and DNA strand break (comet) assays were performed. Statistically significantly elevated SCEs/cell and CAs/cell were observed in cells incubated with Propylparaben (≥1.5 µM) and Propylparaben (≥1.0 µM) for 3 hours, respectively.
Human spermatozoa were exposed to 2.5 mM Propylparaben for 2 or 5 hours. 75 A statistically significant reduction in sperm motility and stimulation of mitochondrial ROS were observed at both time points. After 2 hours, Propylparaben exposure resulted in a significant loss of mitochondrial membrane potential.
Butylparaben
The CHO cells were incubated for 1 or 3 hours with 0, 0.2, 0.4, 0.6, 0.8, or 1.0 mM or 0, 0.1, 0.25, 0.5, or 0.75 µM, respectively Butylparaben. 77 Sister chromatid exchange, CA, and DNA strand break (comet) assays were performed. Statistically significantly elevated indices of DNA fragmentation were observed in cells incubated for 1 hours with ≥0.4 µM Butylparaben. Comparatively high incidences of fragmentation were observed. Statistically significant, elevated SCEs/cell and CAs/cell were observed in cells incubated with 0.75 µM Butylparaben for 3 hours.
Methylparaben, Ethylparaben, Propylparaben, and Butylparaben
Human spermatozoa were exposed to a paraben mixture containing equal concentrations of Methylparaben, Ethylparaben, Propylparaben, and Butylparaben for 24 hours. 75 Significantly reduced motility was observed immediately after the treatment and was further exacerbated after 24 hours at concentrations of 1, 2, and 4 mM (ie, a mixture containing 250, 500, and 1,000 µM of each paraben). After 24 hours, spermatozoa that had been treated with 0.2 and 1 mM of the paraben mixture exhibited a significant increase in the generation of mitochondrial ROS, which then declined in concert with the loss of cell viability. An acute total superoxide response was also observed with dihydroethidium shortly after exposure of parabens, which became statistically significant at 2 and 4 mM. Caspase activation was observed following exposure to concentrations of parabens above 1 mM and increased still further after 24 hours.
In Vivo
No published in vivo genotoxicity studies were discovered in the published literature, and no unpublished data were submitted.
Carcinogenicity Studies
No new published dermal, oral, or inhalation carcinogenicity studies were discovered in the published literature, and no unpublished data were submitted since the 2008 CIR report.
1984
Methylparaben was noncarcinogenic when administered intravaginally in rats and was not co-carcinogenic when injected with dibenzo[a, i]pyrene subcutaneously in mice. 46 Propylparaben was noncarcinogenic in a study of transplacental carcinogenesis.
1995
No changes in either neoplasm incidence or time to neoplasm development were observed in mice dosed with 0.15, 0.3, or 0.6% Isobutylparaben in the feed for 102 weeks as compared with controls. 48
2008
Isobutylparaben and Butylparaben were noncarcinogenic when given to mice in diet at levels of 0.15%, 0.3%, and 0.6% for 102 weeks, respectively. 2
Other Relevant Studies
Endocrine Activity
2008
Butylparaben binds to ERs in isolated rat uteri, with an affinity orders of magnitude less than natural E2. 2 The estrogenic effect of parabens has been estimated by their competitive binding to the human ERs α and β. With diethylstilbestrol binding affinity set at 100, the relative binding affinity of the parabens increased as a function of chain length from not detectable for Methylparaben to 0.267 ± 0.027 for human ER-α and 0.340 ± 0.031 for human ER-β for Isobutylparaben. In a study of AR binding, Propylparaben exhibited weak competitive binding, but Methylparaben had no binding effect at all.
Methylparaben failed to produce any effect in uterotrophic assays in 2 laboratories but did produce an effect in other studies from another laboratory. The potency of Methylparaben was 1,000 to 20,000 less when compared to natural E2. The same pattern was reported for Ethylparaben, Propylparaben, and Butylparaben when potency was compared to natural E2; in positive studies, the potency of Ethylparaben was 346 to 25,000 less, the potency of Propylparaben was 1,612 to 20,000 less, and the potency of Butylparaben was 436 to 16,666 less. In 2 studies, Isobutylparaben did produce an estrogenic response in the uterotrophic assay, but the potency was 240,000 to 4,000,000 less than E2. In 1 study, Benzylparaben produced an estrogenic response in the uterotrophic assay, but the potency was 330,000 to 3,300,000 less than E2.
Estrogenic activity of parabens and 4-Hydroxybenzoic Acid was increased in human breast cancer cells in vitro, but the increases were around 4 orders of magnitude less than that of E2. Several overviews of the endocrine disruption (estrogenic and androgenic effects) generally note that any effect of parabens is weak.
Another assessment of the endocrine disrupting/estrogenic potential of parabens noted that parabens do not have genotoxic, carcinogenic, or teratogenic potential and are rapidly hydrolyzed to 4-Hydroxybenzoic Acid and excreted. This assessment noted that parabens are able to bind estrogen and ARs, activate estrogen-responsive genes, stimulate cellular proliferation, and increase levels of ER protein. To place the in vitro data in context, the assessment cited the comparisons of parabens activity with E2 and diethylstilbestrol (2 to 5 orders of magnitude lower) and phytoestrogens, including isoflavones (comparable or less). This assessment acknowledged increases or decreases in testes, epididymides, or prostate weights in male animals exposed to Butylparaben and Propylparaben and lower sperm counts in rats and mice exposed to Butylparaben and in rats exposed to Propylparaben, but discounted these effects as without pattern or dose–response. The endocrine activity studies summarized below are described in Table 14.
Endocrine Activity.
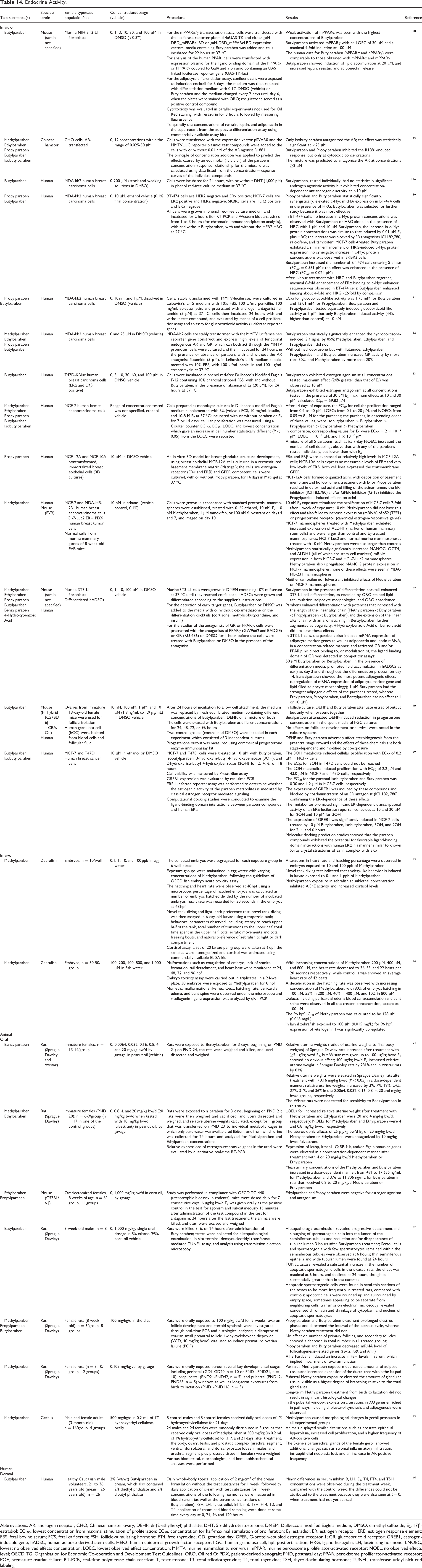
Abbreviations: AR, androgen receptor; CHO, Chinese hamster ovary; DEHP, di-(2-ethylhexyl) phthalate; DHT, 5α-dihydrotestosterone; DMEM, Dulbecco’s modified Eagle’s medium; DMSO, dimethyl sulfoxide; E2, 17β-estradiol; EC100, lowest concentration from maximal stimulation of proliferation; EC50, concentration for half-maximal stimulation of proliferation; E2: estradiol; ER, estrogen receptor; ERE, estrogen response element; FBS, fetal bovine serum; FCS, fetal calf serum; FSH, follicle-stimulating hormone; FT4, free thyroxine; GD, gestation day; GPER, G-protein-coupled estrogen receptor 1; GR, glucocorticoid receptor; GREB1, estrogen-inducible gene; hADSC, human adipose-derived stem cells; HER2, human epidermal growth factor receptor; hGC, human granulosa cell; hpf, postfertilization; HRG, ligand heregulin; LH, luteinizing hormone; LNOEC, lowest no observed effects concentration; LOEC, lowest observed effect concentration; MMTV, murine mammalian tumor virus; mPPAR, murine peroxisome proliferator-activated receptor; NOEL, no observed effects level; OECD TG, Organisation for Economic Co-operation and Development Test Guidelines; ORO, Oil red O; PDX, patient-derived xenograft; PND, postnatal day; PPAR, peroxisome proliferator-activated receptor; POF, premature ovarian failure; RT-PCR, real-time polymerase chain reaction; T, testosterone; T3, total triiodothyroxine; T4, total thyroxine; TSH, thyroid-stimulating hormone; TUNEL, transferase uridyl nick end labeling.
In vitro
Weak activation of murine peroxisome proliferator-activated receptor (mPPAR) α was seen in murine NIH-3T3-L1 cells at the highest concentrations of Butylparaben tested (100 µM). 78 Butylparaben activated mPPARγ with a lowest observed effect concentration (LOEC) of 30 µM and a maximal (4-fold) induction at 100 µM. The human data for Butylparaben (hPPARα and hPPARγ) were comparable to those obtained with mPPARα and mPPARγ, indicating a similar responsiveness.
Isobutylparaben antagonized the AR in CHO cells. The effect was statistically significant at ≥25 µM. 79 Butylparaben increased the number of BT-474 cells entering S-phase (concentration for half maximal stimulation of proliferation [EC50] = 0.551 µM); the effect was enhanced in the presence of ligand heregulin (HRG; EC50 = 0.024 µM). 80 The EC50 for glucocorticoid-like activity in MDA-kb2 cells was 1.75 mM for Butylparaben and 13.01 mM for Propylparaben. 81 Butylparaben at 25 µM statistically significantly enhanced the hydrocortisone-induced glucocorticoid receptor (GR) signal by 85%; Methylparaben, Ethylparaben, and Propylparaben did not have this effect. 82
Butylparaben exhibited estrogen agonism at all concentrations tested in T47D-KBluc cells. 83 The maximum effect was observed at 10 µM.
The EC50 for stimulating proliferation of MCF-7 cells ranged from 0.4 to 40 µM, LOECs from 0.1 to 20 µM, and no observed effects levels from 0.05 to 8 µM for the parabens tested. 84 The parabens tested, in descending order of these values, were Isobutylparaben > Butylparaben > Propylparaben > Ethylparaben > Methylparaben. In comparison, corresponding values for E2 were EC50 = 2 × 10−6 µM, LOEC = 10−6 µM, and 1 × 10−7 µM. Propylparaben at 10 µM resulted in deformed acini and filling of the acinar lumen in nontransformed MCF-12A and MCF-10A cells. 85 MCF-7 and HCI-7-Luc2 mammospheres treated with Methylparaben exhibited increased expression of ALDH1 (marker of human mammary stem cells) and were larger than control and E2-treated mammospheres. 86 Neither tamoxifen nor fulvestrant inhibited effects of Methylparaben on MCF-7 mammospheres.
Parabens enhanced differentiation of murine 3T3-L1 cells with potencies that increased with the length of the linear alkyl chain (Methylparaben < Ethylparaben < Propylparaben < Butylparaben), and the extension of the linear alkyl chain with an aromatic ring in Benzylparaben further augmented adipogenicity. 87 In the presence of differentiation media, 50 µM Butylparaben or Benzylparaben promoted lipid accumulation in human adipose-derived stem cells (hADSCs) as early as day 3 and throughout the differentiation process. Butylparaben had the strongest adipogenic effects of the parabens tested, whereas other parabens had no effect at 1 or 10 µM.
The US Environmental Protection Agency (EPA) Endocrine Disruptor Screening Program (EDSP) program conducted a series of in vitro assays to examine the estrogenic properties of parabens. 88 There are 15, 14, 11, 5, and 2 positive results out of total 18 arrays for Butylparaben, Propylparaben, Ethylparaben, Methylparaben, and 4-Hydroxybenzoic Acid, respectively, while in vitro antiandrogen studies showed negative results.
Metabolites of Butylparaben and Isobutylparaben, 3-hydroxy n-butyl 4-hydroxybenzoate (3OH) and 2-hydroxy isobutyl 4-hydroxybenzoate (2OH), exhibited estrogenic properties in MCF-7 and T47D human breast cancer cells.
89
The expression of estrogen-inducible gene (
In isolated mouse preantral follicle and human granulosa cell (hGC) cultures, Butylparaben adversely affected steroidogenesis at concentrations relevant to human exposure (100 nM), but no effects on follicular development or survival were noted in the culture systems. 90 Butylparaben attenuated di-(2-ethylhexyl) phthalate (DEHP)-induced reduction of progesterone concentrations in the spent media of hGC cultures. When present together, Butylparaben and DEHP decreased E2 production.
Animal
Longer diestrus phases and a shortened interval of the estrous cycle were observed in 8-week old rats exposed to Propylparaben or Butylparaben at a dose of 100 mg/kg/d orally for 5 weeks.
91
No effect on the number of primary follicles was observed, while secondary follicles showed a decrease in the total number in all groups treated with Methylparaben, Propylparaben, or Butylparaben. Propylparaben and Butylparaben decreased messenger RNA (mRNA) level of folliculogenesis-related genes (
Perinatal Methylparaben exposure in rats via gavage at doses mimicking human exposure (0.105 mg/kg/d) decreased amounts of adipose tissue and increased expansion of the ductal tree within the mammary fat pad. 92 Perinatal Methylparaben treatment was associated with a significant reduction in adipose tissue and more abundant glandular tissue. Long-term Methylparaben treatment from birth to lactation did not result in significant histological changes. In the pubertal window, expression alterations in 993 genes enriched in pathways including cholesterol synthesis and adipogenesis were observed.
Oral exposure to Methylparaben at 500 mg/kg/d caused morphological changes in gerbil prostates. 93 After 3, 7, and 21 days of treatment, male and female gerbils displayed similar alterations such as prostate/Skene’s paraurethral gland epithelial hyperplasia, increased cell proliferation, and a higher frequency of AR binding activity.
Relative uterine weights were elevated in immature Sprague Dawley rats after treatment with ≥0.16 mg/kg bw/d Benzylparaben via gavage on PNDs 21 to 23. 94 Lowest observed effect levels for increased relative uterine weight after treatment of immature female rats with Methylparaben or Ethylparaben on PNDs 21 to 23 were 20 and 4 mg/kg bw/d, respectively. 95 No observed effect levels (NOELs) for Methylparaben and Ethylparaben were 4 and 0.8 mg/kg bw/d, respectively. Ethylparaben and Propylparaben were negative for estrogen agonism and antagonism in ovariectomized female mice exposed to 1,000 mg/kg bw/d by gavage for 7 days. 96
Histopathologic examination revealed progressive detachment and sloughing of spermatogenic cells into the lumen of the seminiferous tubules and reduction and/or disappearance of tubular lumen 3 hours after a single 1,000 mg/kg oral dosage of Butylparaben in rats. 72 The TUNEL assays revealed a substantial increase in the number of apoptotic spermatogenic cells in the treated rats; the effect was maximal at 6 hours.
Human
In 26 healthy Caucasian males, minor differences in inhibin B, LH, E2, total thyroxine (T4), free thyroxine (FT4), and TSH concentrations were observed after daily whole-body topical application of a cream formulation containing 2% (wt/wt) Butylparaben as well as 2% diethyl phthalate and 2% dibutyl phthalate, compared to the concentrations measured before the treatment. 44 The differences could not be attributed to the treatment.
Effects on Human Breast Cells
MCF-10A nontransformed, immortalized human breast epithelial cells were exposed to 500 µM Methylparaben, 10 µM Propylparaben or Butylparaben in semisolid 2% methylcellulose suspension culture, or 1 µM Methylparaben or 0.1 µM Propylparaben or Butylparaben in monolayer culture.
97
Ethanol served as the vehicle. The cells were grown in suspension culture (nonadherent conditions) to assess colony growth after a 17-day incubation period. Cells were grown in monolayer culture (adherent conditions) to assess cellular proliferation after a 7-day incubation period. In suspension culture, MCF-10A cells produced very few colonies and only of a small size. The presence of 500 µM Methylparaben or 10 µM Propylparaben or Butylparaben resulted in greater numbers of colonies per dish (
Human high-risk donor breast epithelial cells (HRBECs) were collected from the unaffected contralateral breasts of women undergoing breast surgery with a personal or family history of breast cancer, atypical neoplastic histopathology, and/or high mammographic density.
98
The cells were incubated for 7 days with 10 nM to 1 µM (vehicle not specified) Methylparaben in phenol red-free medium supplemented with 0.2% charcoal-stripped fetal bovine serum (FBS).
98
Some cells were exposed to 10 µM 4 hydroxy tamoxifen (OHT) or 1, 10, or 100 nM rapamycin for 24 hours before functional analysis. Methylparaben substantially reduced the fraction of OHT-induced apoptotic cells in a concentration-dependent manner (
Effects on Human Trophoblast Cells
Butylparaben
Human trophoblast cells, HTR8/SVneo, were exposed to Butylparaben at 50, 100, 200, and 400 µM. 99 Butylparaben inhibited cell proliferation and induced both apoptosis and endoplasmic reticulum stress at all concentrations. Butylparaben promoted the production of intracellular ROS, increased Ca2+ concentration, and induced mitochondrial membrane depolarization. Butylparaben also inhibited the activation of PI3K/AKT pathways including AKT, ribosomal protein S6, P70 S6 kinase, and glycogen synthase kinase 3b. In addition, ERK1/2 activity was involved in Butylparaben-mediated signal transduction in HTR8/SVneo cells. The study author claimed that exposing human trophoblast cells to Butylparaben diminished normal physiological activity, leading to apoptosis and problems with early placental development.
Biomonitoring
The biomonitoring studies summarized below are described in Table 15. Biomonitoring is the direct measurement of human exposure by measuring the parabens or their metabolites in human biological fluids (eg, urine, blood), which account for both oral intake (eg, from foods and medicinal products with paraben preservatives) and dermal application of products with parabens. However, the presence of a substance in the blood or urine does not mean that it will cause effects or disease. 100 Chemical toxicity is related to its dose or concentration, in addition to a person’s individual susceptibility. Small amounts may be of no health consequence, whereas larger amounts may cause adverse health effects.
Biomonitoring Studies in Humans.
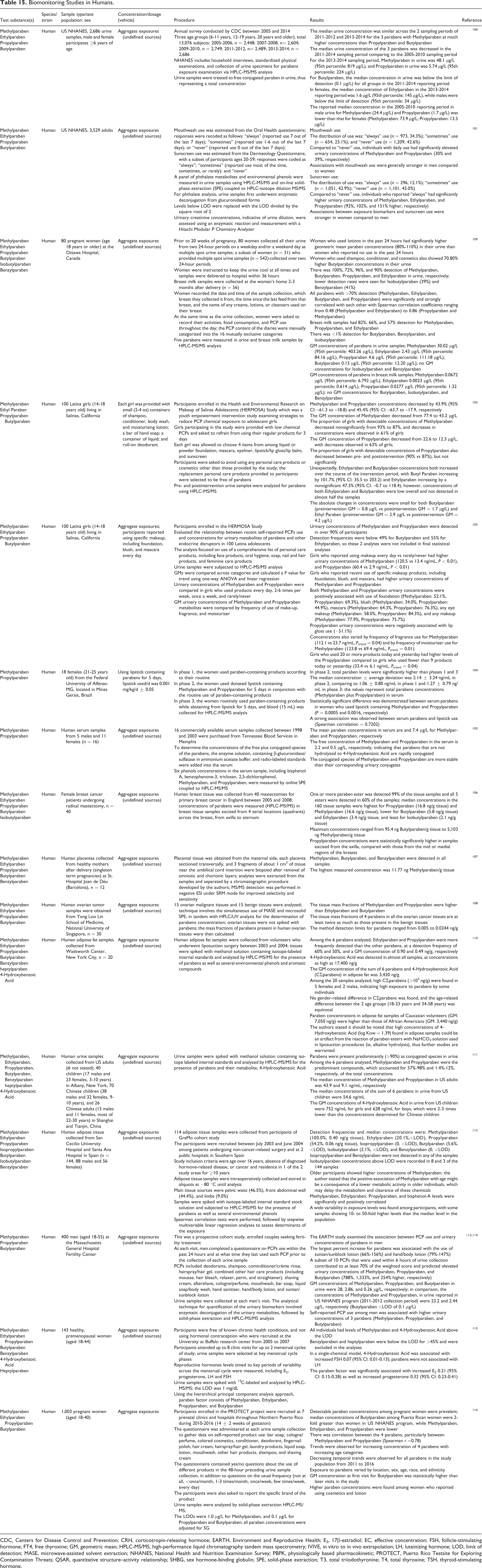
CDC, Centers for Disease Control and Prevention; CRH, corticotropin-releasing hormone; EARTH, Environment and Reproductive Health; E2, 17β-estradiol; EC, effective concentration; FSH, follicle-stimulating hormone; FT4, free thyroxine; GM, geometric mean; HPLC-MS/MS, high-performance liquid chromatography tandem mass spectrometry; IVIVE, in vitro to in vivo extrapolation; LH, luteinizing hormone; LOD, limit of detection; MASE, microwave-assisted solvent extraction; NHANES, National Health and Nutrition Examination Survey; PBPK, physiologically based pharmacokinetic; PROTECT, Puerto Rico Testsite for Exploring Contamination Threats; QSAR, quantitative structure–activity relationship; SHBG, sex hormone-binding globulin; SPE, solid-phase extraction; T3, total triiodothyronine; T4, total thyroxine; TSH, thyroid-stimulating hormone.
The US NHANES program (the Fourth National Report) provides a large data set for human spot urine levels of parabens, collected from 2005 to 2014, with 2013 to 2014 being the most recent collection period. 100 A total of 2,686 urine specimens from a representative sample of persons ≥6 years of age in the US general population was analyzed for the exposure level to Methylparaben, Ethylparaben, Propylparaben, and Butylparaben. For the 2013 to 2014 sampling period, the median concentration of Methylparaben in urine was 48.1 µg/L (95th percentile: 819 µg/L), and Propylparaben in urine was 5.74 µg/L (95th percentile: 224 µg/L). For Butylparaben, the median concentration in urine was below the limit of detection (LOD, 0.1 µg/L) for all groups (age, gender, and race/ethnicity) in the 2011 to 2014 reporting period. In females, the median concentration of Ethylparaben in the 2013 to 2014 reporting period was 1.6 µg/L (95th percentile: 145 µg/L), while concentrations in males were below the LOD (1 µg/L).
Data from the US NHANES program were also used to analyze the exposure to parabens through oral hygiene products and sunscreen use. 101 Compared to individuals who reported “never” using mouthwash, individuals who reported daily use had significantly elevated urinary concentrations of Methylparaben and Propylparaben (30% and 39% higher, respectively). Individuals who reported “always” using sunscreen had significantly higher urinary concentrations of Methylparaben, Ethylparaben, and Propylparaben (92, 102, and 151% higher, respectively) compared to “never” users of sunscreen. Associations between exposure biomarkers and sunscreen use were stronger in women compared to men, and associations with mouthwash use were generally stronger in men compared to women.
A community-based intervention study indicated that using personal care products (PCPs) that are labeled to be free of parabens, for 3 days, lowered urinary concentrations of Methylparaben and Propylparaben in 100 girls: Methylparaben and Propylparaben concentrations decreased by 43.9% (95% CI: −61.3 to −18.8) and 45.4% (95% CI: −63.7 to −17.9), respectively.
102
The geometric mean (GM) concentration of Methylparaben decreased from 77.4 to 43.2 μg/L and Propylparaben decreased from 22.6 to 12.3 μg/L. In contrast, the GM concentration of Ethylparaben increased from 2.9 to 4.2 µg/mL and Butylparaben increased from 0.8 to 1.7 µg/mL. Concentrations of both Ethylparaben and Butylparaben were low overall and not detected in almost half the samples. In the same study population of 100 adolescent girls, participants who reported using “makeup” every day versus rarely/never had higher urinary concentrations of Methylparaben (120.5 vs 13.4 ng/mL,
A statistically significant difference was observed between serum parabens in 18 women who used lipstick containing Methylparaben and Propylparaben for 5 days compared with those not using this cosmetic (
One study reported the free and total paraben concentrations in 16 human serum samples in the United States. 105 The mean total paraben concentrations in serum are 42.6 and 7.4 µg/L for Methylparaben and Propylparaben, respectively, whereas the free concentration of Methylparaben and Propylparaben in the serum is 2.2 and 0.5 µg/L, respectively, indicating that parabens that are not hydrolyzed to 4-Hydroxybenzoic Acid are rapidly conjugated.
One or more of 5 parabens (Methylparaben, Ethylparaben, Propylparaben, Butylparaben, Isobutylparaben) was detected in 99% of breast tissue samples collected from women with breast cancer, and all 5 were detected in 60% of the samples. 106 Median concentrations were highest for Propylparaben (16.8 ng/g tissue) and Methylparaben (16.6 ng/g tissue). Propylparaben concentrations were statistically significantly higher in samples excised from the axilla, compared with those from the mid or medial regions of the breasts.
Ethylparaben, Butylparaben, and Benzylparaben were detected in all placenta samples collected from healthy mothers. 107 The highest measured concentration was 11.77 ng Methylparaben/g tissue. The amount of Butylparaben, Ethylparaben, Methylparaben, and Propylparaben was studied in human ovarian tumor samples. 108 The tissue mass fractions of the 4 parabens in the malignant tissues were at least twice as much as those present in the benign tissues. The tissue mass fractions of Methylparaben and Ethylparaben were higher than Propylparaben and Butylparaben.
Thirty-one pregnant women who provided multiple spot urine samples (n = 542) collected over two 24-hour periods had their samples analyzed for Methylparaben, Propylparaben, Ethylparaben, Butylparaben, Isobutylparaben, and Benzylparaben. 109 These parabens were also measured in breast milk samples collected at approximately 3 months postpartum (n = 56 women). Women who used body and face lotions in the past 24 hours had significantly higher GM paraben concentrations (80%-110%) in their urine than women who reported no use in the past 24 hours. There was 100%, 72%, 96%, and 90% detection of Methylparaben, Butylparaben, Propylparaben, and Ethylparaben in urine, respectively. Lower detection rates were seen for Isobutylparaben (39%) and Benzylparaben (41%). Breast milk samples had 82%, 66%, and 57% detection for Methylparaben, Propylparaben, and Ethylparaben, respectively.
The conjugated or free species of 6 parabens (Methylparaben, Ethylparaben, Propylparaben, Butylparaben, Benzylparaben, and heptylparaben), or their metabolite, 4-Hydroxybenzoic Acid, were measured in human adipose fat samples collected from 20 donors who underwent liposuction surgery. 110 Ethylparaben and Propylparaben were more frequently detected than the other parabens at a detection frequency of 60% and 50% and a GM concentration of 0.90 and 0.49 ng/g, respectively. The GM concentrations of other parabens were not calculated due to their detection of lower than 50%. The GM concentration of the sum of 6 parabens and 4-Hydroxybenzoic Acid (CΣparabens) in adipose fat was 3,420 ng/g. While a positive correlation between donor’s age and CΣ parabens (75th percentile of adipose concentrations; n = 15) was observed, no significant difference in concentrations of CΣ parabens between the 2 age groups was found (18-33 years and 34-58 years). However, the authors noted that total paraben measurements may have been compromised by alkaline hydrolysis in the tissue due to the use of alkali in the liposuction procedure.
The conjugated or free species of 6 parabens (Methylparaben, Ethylparaben, Propylparaben, Butylparaben, Benzylparaben, and heptylparaben [not a cosmetic ingredient]), or their metabolite, 4-Hydroxybenzoic Acid, were measured in urine samples collected from 40 US children, 70 Chinese children, and 26 Chinese adults. 111 Parabens were present predominantly (>90%) as conjugated species in urine. Among the 6 parabens analyzed, Methylparaben and Propylparaben were the predominant compounds, which accounted for 57% to 98% and 1.4% to 12%, respectively, of the total concentrations. The median concentrations of Methylparaben and Propylparaben in US adults were 43.9 and 9.1 ng/mL, respectively. The median concentration of the sum of 6 parabens in urine from US children was 54.6 ng/mL. The GM concentrations of 4-Hydroxybenzoic Acid in urine from US children were 752 ng/mL for girls and 628 ng/mL for boys, which were 2 to 3 times lower than the concentrations determined for Chinese children.
One or more of 7 parabens (Methylparaben, Ethylparaben, Propylparaben, Isopropylparaben, Butylparaben, Isobutylparaben, and Benzylparaben) were measured in 144 human adipose tissue samples collected from patients >16 years old, who were undergoing non-cancer-related surgery, and presented no evidence of diagnosed hormone-related disease or cancer.
112
Detection frequencies and median concentrations were Methylparaben (100.0%, 0.40 ng/g tissue), Ethylparaben (20.1%, <LOD), Propylparaben (54.2%, 0.06 ng/g tissue), Butylparaben (5.6%, <LOD), and Isobutylparaben (2.1%, <LOD). Isopropylparaben and Benzylparaben were not detected in any of the samples, while Butylparaben and Isobutylparaben concentrations above LOD were only recorded in 8 and 3 of the 144 samples. Methylparaben, Ethylparaben, and Propylparaben levels were significantly and positively correlated. No statistically significant relationship between age and paraben concentrations in human adipose tissue was identified. Of the 7 parabens measured, only a positive association between age and Methylparaben concentrations was found (close to, but not statistically significant,
The Environment and Reproductive Health (EARTH) study examined the association between the use of 14 PCPs and the urinary concentrations of parabens in 400 men (18-55 year of age). 113 The largest percentage increase for parabens was associated with the use of suntan/sunblock lotion (66%-156%) and hand/body lotion (79%-147%). A subset of 10 PCPs that were used within 6 hours of urine collection contributed to at least 70% of the weighted score and predicted elevated urinary concentrations of Methylparaben, Propylparaben, and Butylparaben (788%, 1,333%, and 254% higher, respectively). The GM concentrations of Methylparaben, Propylparaben, and Butylparaben in urine were 28, 2.86, and 0.26 µg/L, respectively.
The EARTH study also showed that, among 346 infants, none of the maternal preconception paraben concentrations were associated with birth weight. 114 Maternal preconception Methylparaben concentration was associated with a decreased head circumference of 0.27 cm (95% CI: −0.54 to 0), while no associations were observed between Ethylparaben, Propylparaben, and Butylparaben concentrations and head circumference.
Six parabens (Methylparaben, Ethylparaben, Propylparaben, Butylparaben, Benzylparaben, and heptylparaben) and 4-Hydroxybenzoic Acid were measured in 143 urine samples from healthy, premenopausal women. 115 4-Hydroxybenzoic Acid was associated with increased FSH 0.07 (95% CI: 0.01-0.13) and paraben concentrations were associated with increased E2 0.21 (95% CI: 0.15-0.28) and increased progesterone 0.32 (95% CI: 0.23-0.41).
Among 1,003 pregnant women, median concentrations of Butylparaben were 2-fold greater than US women from the NHANES program, while concentrations of Methylparaben, Ethylparaben, and Propylparaben were lower.
116
There was correlation between the 4 parabens, particularly between Methylparaben and Propylparaben (Spearman
Effects on Adhesin Genes in Candida glabrata
Culture of
Dermal Irritation and Sensitization Studies
1984
Methylparaben (10% and 100%), Propylparaben (10%), and Ethylparaben (10% and 100%) were, at most, mildly irritating when applied to rabbit skin. 46
Parabens are practically nonirritating in the [human] population with normal skin. Skin irritation and sensitization tests on product formulations containing from 0.1% to 0.8% of 1 or 2 of the parabens, including Methylparaben, Ethylparaben, Propylparaben, and Butylparaben, showed no evidence of significant irritation or sensitization potential for these ingredients.
Parabens are practically nonsensitizing in the [human] population with normal skin. Practically all animal sensitization tests indicate that the parabens are nonsensitizing.
1986
Benzylparaben was not a skin irritant when tested in rabbits. 47 Sensitization to Benzylparaben has been observed in eczematous patients. A 3% mixture of Benzylparaben, Methylparaben, Ethylparaben, Propylparaben, and Butylparaben produced positive reactions ranging from 1% to 3.7%. The cross-sensitization potential of paraben esters was demonstrated in patients previously sensitized to a paraben mixture. Two-thirds of the patients sensitive to one paraben ester also reacted to one or more of the other esters.
2008
Benzylparaben applied directly (0.5 g) to rabbit skin produced no significant irritation. 2 Parabens are practically nonirritating in the population with normal skin. Skin irritation tests on product formulations containing from 0.1% to 0.8% of one or two of the parabens showed no evidence of significant irritation for these ingredients.
In Vitro
The parabens were tested individually for irritancy and sensitization potential in cocultured human keratinocytes and peripheral blood mononuclear cells (PBMCs). 118 The keratinocytes were isolated from skin received as residual material from plastic surgery; PBMCs were enriched from buffy coats by density centrifugation. The cells were cocultured in serum-free keratinocyte growth medium 2 on 12-well cell culture plates. The coculture was incubated for 48 hours with or without a paraben. The concentrations tested were not specified, but likely ranged around 1 to 1,000 µM, in dimethyl sulfoxide (vehicle). Fluorescence-activated cells sorting was used to identify and characterize dendritic cell-related cells. Categorization of compounds as potential irritants and sensitizers was based on EC50 calculated from concentration–response data for cell death (irritancy) and CD86 expression (sensitization) compared with vehicle controls. Substances with EC50 for cell death of ≤50 µM were considered to be irritating, with EC50 ranging from 50 to 1,000 µM were considered weakly irritating, and substances that did not reach the 50% threshold for cytotoxicity, or for which EC50 >1,000 μM, were considered nonirritating. Substances with an EC50 for CD86 expression of ≤12.5 µM were categorized as extreme sensitizers, >12.5 µM to <50 µM as strong sensitizers, >50 µM to <100 µM as moderate sensitizers, and >100 EC50 as nonsensitizers. Methylparaben and Ethylparaben showed no potential for irritation in this test. Propylparaben, Isopropylparaben, Butylparaben, Isobutylparaben, and Benzylparaben appeared to be weak irritants. The sensitization potential of the parabens tested was correlated with side-chain length: Methylparaben, Ethylparaben, Propylparaben, and Isopropylparaben were classified as weak sensitizers, and Butylparaben, Isobutylparaben, and Benzylparaben were strong sensitizers in this study.
Photosensitization/Phototoxicity
1984
Photocontact sensitization and phototoxicity tests on product formulations containing 0.1% to 0.8% Methylparaben, Propylparaben, and/or Butylparaben gave no evidence for significant photoreactivity. 46
In vitro
Methylparaben
Normal human keratinocytes (HaCaT cells) were exposed to 0%, 0.003%, 0.03%, and 0.3% (0, 0.197, 1.97, and 19.7 mM, respectively) Methylparaben in an ethanol vehicle.
119
The cells were grown and incubated, with or without Methylparaben, for 6 or 24 hours in DMEM supplemented with 5% FBS, 2 mM glutamine, and 100 U/mL penicillin/streptomycin at 37 °C. Methylparaben-treated and Methylparaben-untreated cells were exposed to medium-wavelength ultraviolet light (UVB; 15 or 30 mJ/cm2) after replacing the culture medium with PBS. The UVB source was a bank of 6 fluorescent sunlamps with an emission spectrum of 275 to 375 nm, mainly in the UVB range, peaking at 305 nm, and including a small amount of long-wavelength ultraviolet light (UVA) and short-wavelength ultraviolet light (UVC). After irradiation, the cells were incubated in culture medium without Methylparaben for various durations. Methylparaben reduced cell viability in a statistically significant manner within 6 hours at 0.3% and within 24 hours at 0.03%. Concurrent observation of fluorescent microscopy images and use of a fluorescent microplate reader revealed little evidence of ROS or NO production after Methylparaben exposure. UVB irradiation at 30 mJ/cm2 (but not at 15 mJ/cm2) induced small amounts of late apoptosis and necrosis. Methylparaben induced statistically significant elevation of (
Ocular Irritation Studies
1984
Methylparaben and Ethylparaben at 100% concentration were slightly irritating when instilled into the eyes of rabbits. 46 A primary eye irritation study in humans showed Methylparaben to be nonirritating at concentrations up to 0.3%.
1986
Benzylparaben was neither an eye nor skin irritant when tested in rabbits. 47
2008
A number of rabbit eye irritation studies have been conducted on products containing Methylparaben, Ethylparaben, Propylparaben, and/or Butylparaben at concentrations of 0.1% to 0.8%. Most products produced no signs of eye irritation. Other products produced slight or minimal eye irritation, with scores of 1.0 to 3.3 of 110. 2
In vitro
Methylparaben
Wong-Kilbourne-derived human conjunctival epithelial cells (WCCs) and immortalized human corneal epithelial cells (HCEs) were exposed to 0, 0.001%, 0.0025%, 0.005%, 0.0075%, 0.01%, 0.025%, 0.05%, 0.075%, and 0.1% Methylparaben.
120
The cells were cultured under standard conditions in Hank’s balanced salt solution supplemented with 10% FCS, 1%
Clinical Studies
Adverse Event Reports
1984
Industry complaint experience data showed low to moderate numbers of safety-related complaints with the incidence depending on the product. 46 Paraben sensitization has occurred, especially when paraben-containing medicaments have been applied to damaged or broken skin. Even when applied to patients with chronic dermatitis, parabens generally induce sensitization in less than 3% of such individuals. Of 27,230 patients with chronic skin problems, 2.2% were sensitized by preparations of parabens at concentrations of 1% to 30%. Many patients sensitized to paraben-containing medications can wear cosmetics containing these ingredients with no adverse effects.
Parabens were designated “nonallergen” of the year (2019) by the American Contact Dermatitis Society. 121,122 Monitoring for paraben allergy followed with studies reporting paraben testing in standard screening fashion since 1940. The frequency of allergic contact sensitization to parabens has remained low and remarkably stable for many decades despite wide use. Parabens have been considered relatively nonirritating at levels used in current formulations, as verified in extensive experience with the mix at current applied patch test concentrations.
Retrospective and multicenter studies
In 1 retrospective analysis, 1,363 cumulative irritation test studies in more than 45,000 subjects, who use-tested 151 different paraben-containing formulations (along with other ingredients), did not demonstrate parabens to be irritating in typical in-use conditions and irritation scores did not correlate with preservative concentrations. 123
Allergic contact dermatitis caused by paraben mixture was analyzed on the basis of data collected by the European Surveillance System on Contact Allergies (ESSCA) network between 2009 and 2012 from 12 European countries (Table 16). 124 Of the 52,586 tests during the study period, parabens yielded less than 1% positive reactions. Of the results obtained from 2,362 TRUE-Test, the paraben mixture yielded only 0.4% positive reactions. The allergic contact dermatitis data are summarized in Table 16.
Contact Dermatitis Studies on Paraben Mixture (Data Collected by ESSCA Between 2009 and 2012 From 12 European Countries). 124
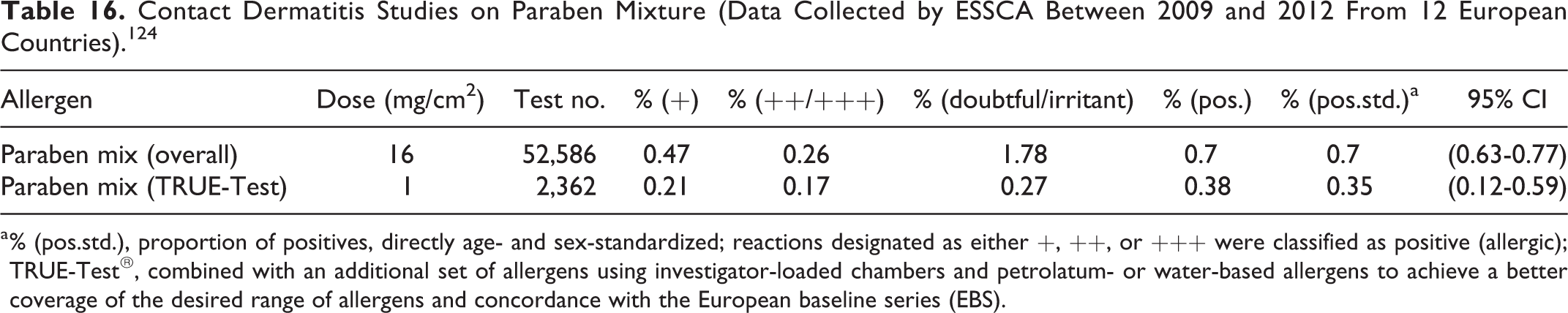
a % (pos.std.), proportion of positives, directly age- and sex-standardized; reactions designated as either +, ++, or +++ were classified as positive (allergic); TRUE-Test®, combined with an additional set of allergens using investigator-loaded chambers and petrolatum- or water-based allergens to achieve a better coverage of the desired range of allergens and concordance with the European baseline series (EBS).
Epidemiological Studies
The epidemiological studies summarized below are described in Table 17.
Epidemiological Studies of Parabens.

a Bolded text was used to highlight statistically significant increases; italicized text was used to highlight statistically significant decreases.
Abbreviations; AFC, anthral follicle count; ANOVA, analysis of variance procedures; BKMR, Bayesian kernel machine regression; BMI, body mass index; BSI, Behavioral Symptoms Index; BSID, the Bayley Scales of Infant Development; CASA, computer-aided semen analysis; CI, confidence interval; DLLME, dispersive liquid–liquid microextraction; EARTH, Environment and Reproductive Health; E2, estradiol; EDI, estimated daily intake; EDEN, Etude des Déterminants pré et postnatals du développement et de la santé de l’Enfant; EEQ, estrogen equivalency; FSH, follicle-stimulating hormone; GDM, gestational diabetes mellitus; GLT, glucose loading test; GM: geometric mean; GnRH, gonadotropin-releasing hormone; HELIX, the Human Early-Life Exposome project; HPLC-MS/MS, high-performance liquid chromatography–mass spectrometry/mass spectrometry; HR, hazard ratio; ICSI, intracytoplasmic sperm injection; IADPSG, International Association of Diabetes and Pregnancy Study Groups; IQR, interquartile range; IVF, in vitro fertilization; LOD, limit of detection; LOQ, limit of quantification; LVI-GC-MS/MS, large-volume injection gas chromatography with tandem mass spectrometry; MDI, Mental Developmental Index; MDL, method detection limit; MGH, Massachusetts General Hospital; MPC, mean percent change; NA, not applicable; NHANES, National Health and Nutrition Examination Survey; NBC, New Bedford Cohort; OR, odds ratio; OV, ovarian volume; PDI, Psychomotor Development Index; PFR, placental to birth weight ratio; Parabens, Sum molar concentrations of the parabens; PIR, poverty income ratio; PTB, preterm birth; SART, Society for Assisted Reproductive Technology; SART: Society for Assisted Reproductive Technology; SG, specific gravity; OGTTs, oral glucose tolerance tests; UPLC-MS/MS, ultra-high-performance liquid chromatography–tandem mass spectrometry; WHO, World Health Organization.
Prospective Studies
In vitro fertilization (IVF) outcomes were not associated with urinary Methylparaben, Propylparaben, or Butylparaben concentrations of women undergoing treatments for infertility. 125 No significant associations were observed between current exposure levels of Methylparaben, Ethylparaben, and Propylparaben in Chinese pregnant women and size of infants at birth. 126 One study examined 420 women undergoing IVF treatment. 125 Urinary concentrations of parabens (Methylparaben and Propylparaben) were not associated with any IVF outcome, such as endometriosis, diminished ovarian reserve, tubal, or ovulatory disorders.
Urinary Methylparaben and Propylparaben concentrations were associated with an increase in gestational age in northern Puerto Rico. 127 Methylparaben, Butylparaben, and Propylparaben were associated with a 34% to 50% decrease in the odds of small for gestational age (SGA).
Among 501 male partners of couples planning to become pregnant, urinary concentrations of Methylparaben, Ethylparaben, and Butylparaben were associated with diminished sperm count and several sperm motility parameters. 128 In contrast, hydroxylated paraben metabolites (methyl-protocatechuic acid and ethyl-protocatechuic acid) were positively associated with select semen quality parameters. The median urinary concentrations of Methylparaben, Ethylparaben, Propylparaben, Butylparaben, and Benzylparaben among 419 participants who provided both urine and semen samples are 6.51, 0.36, 1.39, 0.03, and 0.02 ng/mL, respectively. In the same study population, no associations were observed between paraben concentration in seminal plasma and 35 semen quality parameters among 339 male partners after false discovery rate adjustment. 129 In addition, seminal plasma concentrations of Ethylparaben and Benzylparaben were associated with an increased percentage of sperm motility.
Among 936 men of couples seeking infertility treatment, urinary concentrations of Methylparaben and Propylparaben remained stable over the study period between 2000 and 2017. 130 The downward trends in sperm concentration and normal morphology were not affected when including urinary paraben concentrations in linear regression models, that is, parabens did not substantially change the downward trends in semen parameters (volume, sperm concentration, count, motility, and morphology).
Among 482 pregnant women, an interquartile range increase in urinary Ethylparaben (10.4 ng/mL) was associated with a 7.7% decrease in pro-inflammatory marker interleukin (IL) 1β (95% CI: −14.1 to −0.86). 131 However, the association between Ethylparaben and IL-1β differed across pregnancy, becoming positive at the end of the study.
In Latino girls, at age 9, earlier thelarche, pubarche, and menarche were associated with urinary Methylparaben concentrations, and earlier pubarche was associated with urinary Propylparaben concentrations. 132 In boys, no prenatal parabens were associated with pubertal timing, while one association of earlier gonadarche with urinary Propylparaben concentrations was observed. However, associations of peripubertal measurements with parabens may reflect reverse causality: Children going through puberty early may be more likely to use products that expose them to parabens.
Urinary paraben concentrations (Methylparaben, Propylparaben, and Butylparaben) and pregnancy blood glucose levels during the first and/or second trimester were measured in 241 women. 133 Investigating parabens individually did not provide any significant results. However, when investigating these parabens as a mixture, positive associations of Butylparaben (eg, comparing the 4th and 1st quartiles) with glucose levels were observed for both the first trimester (adjusted difference = 12.5 mg/dL; 95% CI: 0.9-24.2) and second trimester (adjusted difference = 11.2 mg/dL; 95% CI: 0.2-22.3) and a negative association between first trimester Propylparaben and glucose (adjusted difference = −22.3 mg/dL; 95% CI: −43.2 to −1.4).
Among 1,087 pregnant women in China, 5 parabens (Methylparaben, Ethylparaben, Propylparaben, Butylparaben, and Benzylparaben) concentrations were measured in spot urine samples collected between 8 and 16 gestational weeks. 134 A total of 103 (9.5%) women were diagnosed with gestational diabetes mellitus (GDM). Urinary Ethylparaben was associated with GDM. The relative risks (RRs) = 1.12 (95% CI: 0.63-2.01) for the second quartile, RRs = 1.11 (95% CI: 0.64-1.93) for the third quartile, and RRs = 1.70 (95% CI: 1.02-2.82) for the highest quartile, compared with the lowest quartile. In contrast, there was no evidence of associations between urinary Methylparaben or Propylparaben and GDM.
Maternal urinary paraben levels of Methylparaben, Ethylparaben, Propylparaben, Butylparaben, and Benzylparaben were measured in 850 mother–infant pairs.
135
In all infants, each doubling increase in average Ethylparaben concentration was associated with −2.82% (95% CI: −5.11% to −0.53%) decrease in weight
Among 473 pregnant women in France, 4 parabens (Methylparaben, Ethylparaben, Propylparaben, and Butylparaben) were measured in spot urine samples collected between weeks 23 and 29 of gestation.
136
A positive association between the sum of parabens and placental weight was identified (β = 7.12,
Urine concentrations of 4 parabens (Methylparaben, Ethylparaben, Propylparaben, and Butylparaben) were measured in 199 pregnant Taiwanese women during their third trimester. 137 The GMs of Methylparaben, Ethylparaben, Propylparaben, and Butylparaben were 51.79, 1.26, 4.21, and 1.25 µg/g creatinine, respectively. Sex-specific associations between maternal Methylparaben levels and birth outcomes were observed.
Five parabens (Methylparaben, Ethylparaben, Propylparaben, Butylparaben, and Benzylparaben) were measured in 3 spot urine samples (in the first, second, and third trimesters) of 478 pregnant women in China. 138 Each 2-fold increase in average prenatal paraben concentration was associated with lower mental development index (MDI) scores among girls (β = −1.08, 95% CI: −2.10 to −0.06) and (β = − 1.51, 95% CI: −2.69 to −0.32) for Methylparaben and sum of combined parabens (Σparabens), respectively, but the association was not statistically significant among boys.
Among 392 women, Methylparaben, Propylparaben, and Butylparaben were measured in 2 spot urine samples collected during pregnancy. 103 T helper 1 (Th1) and T helper 2 (Th2) cells were measured in offspring blood samples at ages 2, 5, and 7; probable asthma and aeroallergies were assessed at age 7. Methylparaben was associated with lower Th1% (RR: −3.35, 95% CI: −6.58 to −0.02) and Th2% at borderline significance (RR: −4.45, 95% CI: −8.77 to 0.08). Propylparaben was associated with decreased odds of probable asthma (odds ratio [OR]: 0.86, 95% CI: 0.74-0.99).
Among 480 pregnant women, 130 cases of preterm birth (PTB) were identified, including 75 cases of spontaneous PTB and 37 cases of placental PTB. 131 Regression analyses indicated Ethylparaben was associated with increased risk for placental PTB, OR = 1.47 (95% CI: 1.14-1.91).
Of 252 adolescents participating in a new Bedford cohort (NBC) study, urine concentrations of parabens were not associated with any maladaptive behavior, for example, internalizing and externalizing behavior, Behavioral Symptoms Index, adaptive skills, and Developmental Social Disorders. 139
Among 152 pregnant women, a significant decrease in diastolic blood pressure was associated with exposure to parabens, including Methylparaben, Ethylparaben, and Butylparaben, in the second trimester (β = −0.62 mm Hg; 95% CI: −1.16 to −0.08 per doubling of Methylparaben concentrations). 140
The associations between maternal urinary parabens (Methylparaben, Ethylparaben, Propylparaben, and Butylparaben) and plasma inflammatory markers across pregnancy were examined in 130 PTB cases and 352 controls. 131 An interquartile range increase in Methylparaben (359 ng/mL) was positively associated with a 6.69% increase in IL-6 (95% CI: 0.02-13.8), while an increase in Ethylparaben (10.4 ng/mL) was associated with a 7.7% decrease in IL-1β (95% CI: −14.1 to −0.86). However, the authors stated that it is difficult to make conclusions about the magnitude by which parabens contribute toward inflammatory processes during pregnancy due to the complexity of receptor signaling in immune cells.
Urinary paraben concentration and reproductive and thyroid hormones were measured in 602 pregnant women in Puerto Rico. 141 Butylparaben, Methylparaben, and Propylparaben were associated with decreases in the sex hormone-binding globulin (SHBG) by 5.27% (95% CI: −9.4 to −1.14), 3.53% (95% CI: −7.37 to 0.31), and 3.74% (95% CI: −7.76 to 0.27), respectively. Methylparaben was associated with decreases in reproductive hormones, including an 8% decrease (95% CI: −15.4 to 0.61) in estriol, a suggestive 3% increase (95% CI: −2.95 to 9.61) in the progesterone/estriol ratio and a suggestive 6.7% decrease (95% CI: −13.13 to 0.29) in T at 16 to 20 weeks.
Retrospective Studies
Preterm birth was associated with umbilical cord blood concentrations of Butylparaben (OR = 60.77; 95% CI = 2.60-1,419.93) and Benzylparaben (OR = 0.03, 95% CI = 0.01-0.44). 142 The authors stated that the OR of 0.03 for Benzylparaben indicated a “protective effect” of Benzylparaben for PTB. Linear regression analysis indicated an association between maternal urinary concentrations and decreased gestational age and body length in newborns. No statistically significant associations were observed between Methylparaben or Ethylparaben concentrations and the outcomes evaluated (ie, body length, gestational age at birth, birth weight, head circumference). No statistically significant associations were found between prenatal or postnatal growth of male newborns and maternal urinary paraben concentrations of Methylparaben, Ethylparaben, Propylparaben, or Butylparaben. 143
The incidence of cryptorchidism and/or hypospadias, combined, was associated with placental concentrations of Methylparaben ≥1.96 ng/g (OR = 3.18; 95% CI = 0.88-11.48) and Propylparaben concentrations ≥1.16 ng/g (OR = 4.72; 95% CI = 1.08-20.65).
144
Of 436 children at 3 years of age, the median values of estimated daily intake of Methylparaben, Ethylparaben, Propylparaben, Butylparaben, and Benzylparaben were 12.10, 5.68, 4.50, 0.06, and 0.17 μg/kg bw/d, respectively.
145
Urinary Ethylparaben concentrations of boys were positively associated with weight z scores (β = 0.16, 95% CI: 0.04-0.29,
Mean percentage change (MPC) and the results of statistical tests for trends were not statistically significant in a study of urinary concentrations of Methylparaben, Propylparaben, and Butylparaben in women undergoing infertility evaluation and ovarian volume (OV) or antral follicle count (AFC) measurements. 146
No statistically significant associations were found between the urinary concentrations of Methylparaben, Propylparaben, or Butylparaben and serum hormone concentrations, semen quality parameters, and motion characteristics or all but one indicator of sperm damage in a comet assay. 147 The exception was a trend for increased tail% in comet assays of sperm DNA with increasing Butylparaben concentrations.
Cross-Sectional Studies
Among 315 men under 45 years of age who attended an infertility clinic for diagnostic purposes in Poland, urinary concentrations of Ethylparaben and Butylparaben were associated with an increase in the percentage of sperm with abnormal morphology. 148 Urinary Isobutylparaben concentrations were significantly associated with an increase in the percentage of sperm with high DNA stainability. Urinary concentrations of parabens (Methylparaben, Ethylparaben, Propylparaben, Butylparaben, and Isobutylparaben) were not associated with the level of reproductive hormones, including FSH, T, and E2. In addition, urinary concentrations of Methylparaben and Propylparaben were not related to any of the examined semen quality parameters, sperm DNA damage, or the level of reproductive hormones. The unadjusted GM urinary concentrations of Methylparaben, Ethylparaben, Propylparaben, Butylparaben, and Isobutylparaben were 14.7, 1, 4.3, 0.3, and 0.4 µg/L, respectively.
In cord plasma of 27 healthy pregnant women (37th week of pregnancy), Methylparaben, Propylparaben, and the sum of all measured parabens (Methylparaben + Ethylparaben + Propylparaben + Butylparaben) were inversely associated with T levels. 149
Urinary paraben concentrations of Methylparaben, Ethylparaben, Propylparaben, and Butylparaben were measured in 215 young healthy men (18-23 years old), 94% of whom had detectable urinary concentrations of parabens. 150 Urinary concentrations of parabens were not significantly associated with any semen parameters or any of the reproductive hormone levels, including FSH, LH, T, inhibin B, and E2. The unadjusted GM urinary concentrations of Methylparaben, Ethylparaben, and Propylparaben were 11.2, 1.1, and 0.64 ng/mL, respectively.
Among 42 male partners (36.8 ± 5.4 years old) of couples who visited a gynecology clinic in Tokyo for infertility consultation, no significant association was found between semen parameters (sperm volume, concentration and motility) and urinary paraben concentrations in regression analyses. 151 The GM urinary concentrations of Methylparaben, Ethylparaben, Propylparaben, and Butylparaben were 48.2, 1.88, 1.13, and 0.184 ng/mL, respectively.
Linear regression analyses of data from the US NHANES program indicated an association between reduced serum thyroxine (T4) concentrations and urinary concentrations of Methylparaben, Ethylparaben, Propylparaben, and Butylparaben. 152
Analysis of data from the NHANES program indicated an association between aeroallergen and food sensitization, combined, and urinary concentrations of Methylparaben (OR = 1.74; CI = 1.02-3.22), Propylparaben (OR = 2.04; CI = 1.12-3.74), and Butylparaben (OR = 1.55; CI = 1.02-2.33). 153 The results also indicated an association between urinary concentrations of Methylparaben and nonatopic asthma (OR = 0.025; CI = 0.07-0.90) and nonatopic wheeze (OR = 0.23; CI = 0.05-0.99).
One study examined the association between parabens and asthma morbidity among 450 children with asthma and with asthma prevalence among 4,023 children participating in the US NHANES program (2005-2014). 154 An increased prevalence of reporting emergency department visits were observed for every 10-fold increase in Methylparaben and Propylparaben concentrations among boys with asthma (prevalence OR = 2.61, 95% CI: 1.40-4.85 and OR = 2.18, 95% CI: 1.22-3.89, respectively). Among children in the general population, no overall associations with current asthma were observed, although there was a positive trend with Propylparaben and a current asthma diagnosis.
Urine samples were collected from 696 pregnant women in China. 155 The detection rates for the 5 parabens in the urine samples were 97.70% (Methylparaben), 71.26% (Ethylparaben), 96.55% (Propylparaben), 15.80% (Butylparaben), and 2.73% (Benzylparaben). No significant association was found between parabens and GDM among the overall population. However, significant nonlinear associations of Propylparaben and the summed estrogenic activity of parabens with GDM were found in the stratified analysis by prepregnancy body mass index (BMI) in the overweight/obese population, with adjusted ORs of 3.47 (95% CI: 1.28-9.42) and 2.87 (95% CI: 1.07-7.73) for GDM in the second tertile of urinary Propylparaben and the summed estrogen activity, respectively, when compared to the first tertile.
Among 1,693 black women aged 23 to 34 years, morbid obesity (BMI ≥ 35 kg/m2) was inversely associated with Butylparaben and Methylparaben concentrations. 156 Methylparaben concentrations were 30.7% lower for BMI ≥35 versus <25 kg/m2 (95% CI: −48.0% to −7.7%), and Butylparaben concentrations were 30.6% lower for BMI ≥35 versus <25 kg/m2 (95% CI: −49.6% to −4.6%).
Among 156 men under 45 years of age who attended the infertility clinic for diagnostic purposes with normal semen concentration, a positive association was found between urinary level of Butylparaben and XY18 disomy (
The association between urinary phenol biomarkers and breast cancer incidence was studied in 711 women with breast cancer and 598 women without breast cancer. 157 Among all women, the highest (vs lowest) quintiles of urinary Methylparaben, Propylparaben, and sum of parabens were associated with breast cancer ORs of 1.50 (95% CI = 1.03-2.18), 1.31 (95% CI = 0.90-1.90), and 1.35 (95% CI = 0.93-1.97), respectively. In the age-adjusted model, the highest quintile of urinary Methylparaben was associated with a breast cancer OR of 1.21 (95% CI = 0.86-1.72). Associations for breast cancer incidence were more pronounced among women with a BMI <25 kg/m2; however, associations for mortality were more pronounced among women with a BMI greater ≥25 kg/m2.
Risk Assessment
Margin of Safety
For the purpose of this risk assessment, the Panel determined an adequate NOAEL value of 160 mg/kg/d for Butylparaben in consideration of the new data in the category of endocrine activity and from DART studies. 3,64,70,71,158 -160 Specifically, the NOAEL has been derived from a study where pregnant rats were orally exposed to Butylparaben by gavage from GD 7 through PD 21. 64 Above a dose of 160 mg/kg/d, Butylparaben exerted adverse effects on the reproductive system in male offspring, including delayed PPS, reduced reproductive organ weights at several ages, reduced LH level, and elevated E2 and progesterone levels in serum from prepubertal male rats. Importantly, Butylparaben exposure in utero and during lactation significantly reduced epididymal cauda sperm counts, daily sperm production, and serum T in a dose-dependent manner.
In comparison, the SCCS chose an NOEL of 2 mg/kg bw/d for the calculation of the MOS of Butylparaben. The NOEL was derived from a study in which 3 neonatal male rats were exposed subcutaneously to 2 mg/kg bw/d Butylparaben from PND 2 to PND 18 (Table 13). 158 No effects on any of the measured reproductive parameters were documented, compared with the control group. The DART parameters examined in this study included testis weight, distension of the rete testis and efferent ducts, epithelial cell height in the efferent ducts, and immunoexpression of the APQ-1. However, the Panel considered that such study suffers from several critical limitations: it involves a route of subcutaneous exposure, it is not an Organisation for Economic Co-operation and Development Test Guidelines (OECD TG) study, only 1 postpartum dose was tested, and it did not examine the intergeneration toxicity and typical DART end points, such as AGD, weight of the epididymis/seminal vesicle, sperm counts, and reproductive hormone levels.
For the purposes of an MOS calculation, the Panel considered a scenario wherein a consumer would use a set of cosmetic products containing Butylparaben; aggregate exposure to 17 cosmetic products is calculated to be 17.4 g/d based on addition of deterministic values for a range of products (Table 18). 161 These 17 cosmetic products are divided into 4 main categories 162 : (1) oral products, (2) eye products, (3) non rinse-off products, and (4) rinse-off product; the global daily exposure of products for each category was estimated using the data summarized in Table 18.
The Panel also considered the different use concentrations and exposures of Butylparaben in each main cosmetic product category. For purposes of worst-case assumption, the maximum use concentration of Butylparaben was set to represent the concentrations of use across the products in that category. The Council’s concentration of use survey indicates that the maximum use concentration of Butylparaben in the category of (1) oral products, (2) eye products, (3) leave-on products, and (4) rinse-off product is 0.2%, 0.5%, 0.24%, and 0.33%, respectively 26 (Table 18).
The Panel noted that the measured extent of dermal penetration of parabens is variable, ranging from 1% to 75%; the wide range is likely due to differences in animal species used, matrix effects, and other experimental conditions. 162,163 However, methodologies used in dermal penetration studies, such as radiolabeling, may lead to false presumptions about the rate of dermal penetration of whole, unmetabolized parabens. Depending on the specific location of a radiolabel, high levels of detection of radioactivity are more likely the result of detected metabolites (eg, radiolabeled 4-Hydroxybenzoic Acid), rather than the detection of actual parabens. For purposes of calculating an MOS, the systemic availability of unmetabolized Butylparaben after topical application to human skin is of the primary concern. A human toxicokinetic study has been conducted in 26 young adult males with dermal repeated exposure to Butylparaben at a daily dose of 10 mg/kg bw/d for 5 days. 44,57 No effects of Butylparaben on serum hormonal levels were observed during the exposure time of 5 days, and about 2.1% unmetabolized Butylparaben was detected in the urine of the participants. Note that Butylparaben was applied to the whole body in this human study (10 mg/kg bw/d), while a conservative estimation indicates that daily exposure of consumers to Butylparaben is much lower (0.66 mg/kg bw/d, as shown in Table 18). In addition, the available in vitro percutaneous absorption studies using human split- or full-thickness skin suggest a conservative assumption of human dermal penetration of unmetabolized Butylparaben at 3.7% (which was used by SCCS to calculate the MOS of Butylparaben and then to derive the recommended maximum use concentration of Butylparaben in the EU). 16,18 Taking into account that dermal absorption of lower molecular weight parabens is higher, the Panel selected an estimate of a 50% dermal absorption of unmetabolized parabens in the calculation of the MOS, which thereof represents a conservative assumption.
For adults (60 kg body weight), the relevant calculations are: Global daily exposure (GDE, Butylparaben) = (2.36 g/d of oral products × 0.2% maximum use concentration) + (0.05 g/d of eye products × 0.5% maximum use concentration) + (13.93 g/d of non-rinse-off products × 0.24% maximum use concentration) + (1.04 g/d of rinse-off products × 0.33% maximum use concentration) = 0.042 g/d. Systemic exposure dose (SED, Butylparaben) = GDE/60 kg body weight × 50% dermal absorption × 1,000 mg/g conversion factor = 0.35 mg/kg/d. MOS (adult, Butylparaben) = NOAEL/SED = 160 mg/kg/d/0.35 mg/kg/d = 457.
Since alkyl parabens undergo the similar enzymatic hydrolysis to form 4-Hydroxybenzoic Acid, a conservative MOS of Butylparaben for adults could be applied to other individual alkyl parabens.
The Panel considered exposures to cosmetic products containing multiple parabens. A protective MOS level of 100 was used to calculate a maximum safe use concentration for combined paraben use in a single formulation. NOAEL/MOS (adult, multiple paraben) = SED = 160 mg/kg/d/100 = 1.6 mg/kg/d (SED × body weight)/(dermal absorption × conversion factor × GDE) = (1.6 mg/kg/d × 60 kg)/(50% × 1,000 mg/g × 17.4 g/d ) = maximum use concentration = 1.1%.
Accordingly, the Panel determined that the commonly used limitation of 0.8% for Σparabens is conservative and is safe for human health when parabens are used in combination in cosmetic products.
Estimate and Refinement of Aggregate Exposure
Estimate of aggregate exposure
In addition to cosmetic and PCPs, parabens are also widely used in drugs and foods. According to one study, considering aggregate exposure to parabens from various sources, the total combined exposure was 76 mg/d, with cosmetics and PCPs accounting for 50 mg/d; drugs, 25 mg/d; and foods, 1 mg/d. 164
The Dutch National Institute for Public Health and the Environment (RIVM) conducted an exposure assessment in consideration of the aggregated exposure to parabens via 3 major sources: PCPs, foods, and medicinal products. 163 For Methylparaben, adding exposures results in an aggregate exposure estimate of 3.0 mg/kg/d for both adults and children. The estimate for medicinal products contributes 70% to 74% of this value, while the contribution of food is less than 1%. For Propylparaben, adding the exposures results in an aggregate exposure estimate of 1.2 mg/kg/d for both children and adults; 64% to 72% of the exposure is from medicinal products and less than 1% from food. For Ethylparaben, due to the lack of use information on medicinal products, the summation of exposure via PCPs and exposure via foods will result in an aggregate exposure of 0.2 mg/kg/d for adults and children and, as with Methylparaben and Propylparaben, the contribution of foods is less than 1%. However, the authors noted that such an aggregation estimate was based on a series of studies with varying levels of information and uncertainties.
Refinement of aggregate exposure
In current risk assessments, aggregate exposure of parabens is commonly estimated by using a simplistic approach of summing the exposures from all the individual product types in which parabens are used. However, this summation will result in an unrealistic estimation because (1) the use frequency of products and the amount of product applied are overestimated, (2) parabens may not be used in all products of a given type (eg, all make-up products), (3) the extent of use factors for parabens in products is not considered, (4) individuals in the population vary in their patterns of product use including co-use and nonuse, and (5) the extent to which parabens are absorbed from the skin into the internal system warrants further studies. Use of multiple exposure models help provide realistic estimates in comparison with observational biomonitoring data. 165 A recent study indicated that approximately 60% to 90% of the model predictions from 5 implemented models were within a factor of 10 of the observed paraben exposures, while 30% to 40% of the predictions were within a factor of 3 (ie, a factor of 3 or 10, above or below the minimum observed absorbed doses). These models included 3 of the screening models (ie, RIVM ConsExpo, SCCS notes of guidance algorithms, and the Risk Assessment Identification and Ranking–Indoor and Consumer Exposure) and 2 higher tier probabilistic models (US EPA’s Stochastic Human Exposure and Dose Simulation–High Throughput and Creme Care & Cosmetics). A number of uncertainties affect interpretation of the modeled versus measured exposures, such as parabens in preservative product concentrations, dermal absorption parameters, and degree of metabolism following dermal absorption.
An approach has been developed to refine the aggregate exposure estimates using 4 of the more commonly used parabens (ie, Methylparaben, Ethylparaben, Propylparaben, and Butylparaben). 162 The relative refinement allowed co-use and nonuse data, as well as the extent of parabens use data, to be developed for 9 cosmetic and skin care products, including body lotion, body cream, facial mask, hand lotion, foundation/liquid make-up, facial moisturizer, lip color, night cream, and facial cleanser. Simple summed aggregate exposure from these 9 cosmetic and skin care products was 1.61, 0.80, 1.70, and 0.016 mg/kg/d for Methylparaben, Propylparaben, Ethylparaben, and Butylparaben, respectively. When the refining factors were applied, and a conservative dermal penetration rate of 80% was chosen, the aggregate exposure compared to the simple addition approach was reduced by 51%, 58%, 90%, and 92% for Methylparaben, Propylparaben, Butylparaben, and Ethylparaben, respectively. In comparison, estimated internal exposure based on the 95th percentile values of parabens concentration in human urine was 19.9, 8.2, 1.39, and 0.86 µg/kg/d for Methylparaben, Propylparaben, Ethylparaben, and Butylparaben, respectively. This means that in all cases the aggregate exposure estimates are significantly greater than the exposures derived from the biomonitoring data. 162 If exposure via food was included, the aggregate exposure for Methylparaben and Propylparaben, which are used extensively in foods, would only increase by 1% and 4%, respectively. That is, estimates for exposure to Methylparaben and Propylparaben via food are at least 25-fold lower than the estimates for aggregate exposure resulting from dermal exposure to cosmetic products. 162,164
Another study takes population variability of individual characteristics and behavior within the female US population into account. 166 Daily paraben intake was estimated based on skin permeation coefficient models, product use characteristics, and multipathway exposure model, that is, aqueous dermal uptake, gaseous dermal uptake, inhalation intake, and environmentally mediated intake due to disposal after use of parabens. The mean (2.5th-97.5th percentiles) modeled population intakes were 0.2 (0.003-0.8), 0.03 (0-0.2), 0.06 (0-0.3), and 0.02 (0-0.1) mg/kg/d for Methylparaben, Ethylparaben, Propylparaben, and Butylparaben, respectively. This intake estimate represents a consumer who uses the following 11 PCPs which all contain parabens: shampoo, conditioner, body lotion, facial cream, night cream, facial cleanser, deodorant, body wash, foundation, eye shadow, and lipstick. The environmentally mediated paraben intake from disposal stage was 3 to 4 orders of magnitude lower than use stage. 166
Summary
This is a safety assessment of the available scientific literature and concentration of use data relevant to assessing the safety of 20 parabens and 4-Hydroxybenzoic Acid as used in cosmetics. According to the
According to VCRP survey data received in 2019, Methylparaben was reported to be used in 11,739 formulations; this is an increase from 8,786 uses reported in 2006. Propylparaben had the next highest number of reported uses at 9,034; this was an increase from 7,118 uses reported in 2006. All of the other previously reviewed parabens in this safety assessment increased in the number of reported uses since 2006 with the exception of Benzylparaben, which dropped from 1 reported use to 0.
The results of the concentration of use survey conducted by the Council in 2016 indicate Methylparaben had the highest reported maximum concentration of use, up to 0.9% in shampoos. The highest maximum concentration of use reported for products resulting in leave-on dermal exposure is Ethylparaben in eye shadows at 0.65%. In 2006, Methylparaben had the highest reported maximum concentration of use at 1% in lipsticks. The maximum concentrations of use of the previously reviewed parabens have remained under 1%, and the patterns of use are similar to those reported in the previous safety assessment.
The US FDA considers Methylparaben and Propylparaben to be GRAS as antimicrobial agents in food. Parabens may be classified as moderate penetrants. Penetration was inversely proportional to the lipophilicity of the parabens tested (Methylparaben > Ethylparaben > Propylparaben > Butylparaben). Residual quantities of parabens remaining in the skin increased as the test concentration increased, with greater amounts in the human epidermis than in mouse skin.
After application of 2% (wt/wt) Butylparaben in cream (also contains 2% diethyl phthalate and 2% dibutyl phthalate) in 26 healthy Caucasian men, Butylparaben was detected in the serum, with maximum concentrations not exceeding 1.0 µg/L. Butylparaben concentrations increased rapidly within 3 hours after the first application of cream containing the 3 test compounds and could be detected in most serum samples collected throughout the second week of this study.
In in vitro tests, Methylparaben, Ethylparaben, and Propylparaben did not exhibit binding affinity for AFP. Conversely, the IC50 of Benzylparaben was 0.012 µM. Butylparaben was metabolized to 4-Hydoxybenzoic Acid with maximum rate at saturating concentration (Vmax) of 8.8 nmol/min/mg protein. The CP enhances skin permeation of Methylparaben primarily by increasing the solubility of Methylparaben in the SC (especially in the nonlipid regions).
Methylparaben and Ethylparaben were stable in human plasma, but Propylparaben, Butylparaben, and Benzylparaben concentrations decreased by 50% within 24 hours. All parabens tested were rapidly hydrolyzed when incubated with HLM depending on the alkyl chain length. Parabens, but not 4-Hydroxybenzoic Acid, were actively glucuronidated by liver microsomes and human recombinant UGTs.
Butylparaben was rapidly cleared in hepatocytes from rats and was cleared more slowly in hepatocytes from humans, with little or no sex difference. Butylparaben was extensively hydrolyzed to 4-Hydroxybenzoic Acid as the major metabolite for both sexes and species. Methylparaben, Ethylparaben, Propylparaben, and Butylparaben were hydrolyzed by RLM and HLM in in vitro tests. In contrast to RLM, HLM showed the highest hydrolytic activity toward Methylparaben, with activity decreasing with increasing side-chain length of the paraben tested. Rat small intestinal microsomes exhibited relatively higher activity toward longer side-chain parabens. Human small intestinal microsomes showed a specificity pattern similar to that of rat small intestinal microsomes.
Metabolism rates of Methylparaben, Ethylparaben, Propylparaben, and Butylparaben by HLM and HSM were inversely proportional to chain length. Paraben metabolism in HLM was 300- to 500-fold faster than in HSM, depending on the paraben. In contrast to human tissue fractions, all rat tissue fractions tested hydrolyzed the parabens at rates that increased as the ester chain length increased. Rat skin displayed 3 to 4 orders of magnitude faster hydrolysis rates than human skin.
Nine rats were given a single dermal dose of 100 mg/kg bw [ring-U-14C]-labeled Methylparaben, Propylparaben, or Butylparaben. Cmax (≥693 and ≥614 ng Eq/g in males and females, respectively) occurred within 8 hours postapplication, and blood concentrations decreased until the last quantifiable concentration within 24 hours. Most of the dosage (≥46.4%) was not absorbed, and less than 25.8% was found in the urine. Urine was the primary route of elimination. Tissues contained about 4.3% of the 10 mg/kg dosage. The kidneys contained about twice the concentration of residues found in liver.
In rats exposed to a single oral dosage of up to 100 mg/kg bw [ring-U-14C]-labeled Methylparaben, Propylparaben, or Butylparaben, Cmax (≥11,432 and ≥21,040 ng Eq/g in males and female, respectively) occurred within 1 hour postgavage, and blood concentrations decreased until the last quantifiable concentration at 12 hours. Radioactivity was eliminated rapidly, with averages ≥69.6% recovered in the urine during the first 24 hours. When rats were given a single oral dose of up to 1,000 mg/kg bw Butylparaben, the rate of urinary excretion was similar across all dosages, with ≥66% recovered in the first 24 hours in males.
Time-mated female SD rats were orally administered 0, 1,500, 5,000, or 15,000 ppm Butylparaben via NIH-07 feed, ad libitum, from GD 6 to PND 28. Low placental and lactational transfer of dietary Butylparaben were observed. Poor conjugation in pups during early lactation results in higher exposure to free Butylparaben in pups compared to dams.
All 26 male volunteers showed increased excretion of Butylparaben following daily whole-body topical application of a cream formulation containing 2% (wt/wt) Butylparaben. Mean total Butylparaben excreted in urine during exposure was 2.6 ± 0.1 mg/24 hours. The concentrations peaked in the urine 8 to 12 hours after application. Free and conjugated parabens and their major, nonspecific metabolites (4-Hydroxybenzoic Acid and
There were no significant changes in body and organ weights in any group when rats were dermally exposed to up to 600 mg/kg bw/d Isopropylparaben or Isobutylparaben for 28 days. Macroscopic and microscopic examinations revealed mild-to-moderate skin damage in female rats. No observed adverse effect levels for Isobutylparaben and Isopropylparaben were 600 and 50 mg/kg bw/d, respectively.
At 100 and 300 mg/kg bw/d Propylparaben administered orally, rats exhibited statistically significant increases in relative liver weights, serum ALT, AST, ALP, and LDH activities. Significant decreases in total serum protein and albumin, GSH, CAT and SOD activities, serum T concentrations, and T/E2 ratios were also reported. Livers of affected rats exhibited dilated congested central and portal veins, highly proliferated bile ducts with fibrotic reactions, and multifocal areas of necrotic hepatocytes, and testes exhibited evidence of severe spermatogenic arrest.
Elevations of serum markers of lipid peroxidase (ie, malondialdehyde) and hydroxyl radical production were statistically significant in rats exposed to 250 mg/kg bw/d Methylparaben. Malondialdehyde levels were elevated in the liver in a statistically significant, dose-dependent manner, among other effects, in mice orally exposed to 1.33 to 40 mg/kg bw/d Butylparaben for 30 days.
Time-mated rats were orally exposed to 10, 100, or 500 mg/kg bw/d of Butylparaben from GD7 to PND22. The AGD of newborn male and female offspring was significantly reduced at 100 or 500 mg/kg bw/d. The expression of the Sertoli/Leydig cell marker Nr5a1 in adult male offspring was statistically significantly reduced at 10 mg/kg bw/d or above. In male offspring, epididymal sperm count decreased from 76% to 78% compared to controls at all doses from 10 to 500 mg/kg bw/d. The reduction in epididymal sperm count showed the same effect at all doses. In prepubertal females, ovary weight reduction was statistically significant and mammary gland outgrowth was increased at 100 and 500 mg/kg bw/d.
Statistically significant, dose-dependent reductions in AGD and ovary weights were observed in offspring of female rats exposed orally to 100 or 500 mg/kg bw/d Butylparaben from GD7 to GD21.
The E2 level was elevated in male rats orally exposed to Butylparaben at 50 mg/kg for 8 weeks, whereas serum levels of the hormones T, LH, and FSH were decreased. Testicular DNA damage and a reduction in Leydig cells population were recorded in Butylparaben-treated groups.
The expression of CYP19 and ERα was significantly increased, and the expression of StAR, P450scc, SULT1E1, and AR in the testes and methylation rate of the ERα promoter were significantly reduced in male offspring of female rats exposed to 400 or 1,000 mg/kg bw/d Butylparaben from GD7 to GD21.
Weights of the testes, epididymal cauda sperm counts, and daily sperm production in male offspring were significantly reduced in the 400 and 1,000 mg/kg bw/d groups of rats orally exposed to Butylparaben on GD7 to PND21. Vimentin filaments showed shorter projections, concentration near the basal region, and disappearance of the apical extensions toward the lumen of the seminiferous tubules in 3-week-old rats 6 hours after a single 1,000 mg/kg bw oral dosage of Butylparaben.
Prepubertal female rats exposed orally to 1,000 mg/kg bw/d Methylparaben or 250 mg/kg bw/d Isopropylparaben on PND21 to PND40 exhibited statistically significant delays in vaginal opening. Decreases in the weights of the ovaries, increases in the weights of the adrenal glands, thyroid glands, and liver, as well as myometrial hypertrophy were observed in the 1,000 mg/kg bw/d groups. Reduced plasma leptin concentrations were observed in male and female offspring of young adult female rats exposed orally to 100 mg/kg bw/d Butylparaben.
F2 pups exhibited statistically significantly greater mortality at PND7 when F0 females and their F1 offspring were exposed to 0.105 mg/kg bw/d Methylparaben by gavage. During lactation, treated “parous” F1 females exhibited mammary alveoli that were not always milk-filled, collapsed alveolar and duct structures with residual secretory content and marked decrease in the size of the lobular structures. There was no evidence of an effect on the weight of the male reproductive organs, epididymal sperm parameters, hormone concentrations, or histopathology in juvenile male rats exposed via lactation from maternal rats receiving up to 1,000 mg/kg bw/d Propylparaben for 8 weeks.
Methylparaben was associated with a statistically significantly higher incidence of abnormal sperm in rats exposed to 1,000 or 10,000 ppm in the diet for 8 weeks, mostly sperm with no head in 4% to 5% of sperm, compared with 2.3% in 100 ppm and control groups. Measurements of hormone concentrations were generally not altered, except that T and FSH concentrations were higher in the 10,000 ppm Butylparaben-treated group, compared with the control group.
Zebrafish embryos exposure to Methylparaben at 10 ppb and 100 ppb caused alterations in developmental landmarks such as heart rate and hatching percentage. Anxiety-like behavior was induced in larvae exposed to 0.1 and 1 ppb of Methylparaben.
Exposure of zebrafish embryos to Methylparaben at 200, 400, 800, and 1,000 μM for 96 hpf resulted in decreased heart rate and hatching rate and developmental abnormalities. Expression of vitellogenin I was significantly upregulated in larval zebrafish exposed to 100μM of Methylparaben for 96 hpf.
Three neonatal male rats were exposed subcutaneously to 2 mg/kg bw/d Butylparaben on PND 2 to PND 18. No effects on any of the measured reproductive parameters were detected.
Human spermatozoa were exposed to 13 mM Methylparaben for 2 or 5 hours. Methylparaben had no significant effect on DNA fragmentation, while a statistically significant decrease in spermatozoa motility was observed. Methylparaben at a concentration of 2.5 mM did not induce any significant changes to the motility, vitality, mitochondrial ROS production, or 8OHdG formation.
A dose-dependent decrease in the percentage of mitotic cells was observed in Vero cells exposed to Propylparaben. Induction of DNA DSBs was also observed. Statistically significant elevations in SCEs/cell and CAs/cell were observed in cells incubated with Propylparaben (≥1.5 µM) and Propylparaben (≥1.0 µM) for 3 hours, respectively.
Statistically significant, elevated indices of DNA fragmentation were observed in CHO cells incubated for 1 hour with ≥0.4 µM Butylparaben. Elevated SCEs/cell and CAs/cell were observed in CHO cells incubated with 0.75 µM Butylparaben for 3 hours.
Human spermatozoa were exposed to a paraben mixture containing equal concentrations of Methylparaben, Ethylparaben, Propylparaben, and Butylparaben. Significantly reduced motility was observed immediately after the treatment and was further exacerbated after 24 hours at doses of 1, 2 and 4 mM. Caspase activation was observed following exposure to parabens concentrations above 1 mM and increased still further after 24 hours.
Weak activation of PPARα and PPARγ was observed in NIH-3T3-L1 cells exposed to Butylparaben. Isobutylparaben antagonized the AR in CHO cells. Butylparaben increased the number of BT-474 cells entering S-phase; the effect was enhanced in the presence of ligand HRG. Butylparaben significantly enhanced the GR signal, while Methylparaben, Ethylparaben, and Propylparaben did not have this effect.
Butylparaben exhibited estrogen agonism in T47D-KBluc cells. MCF-7 and HCI-7-Luc2 mammospheres treated with Methylparaben exhibited increased expression of ALDH1. Parabens enhanced differentiation of murine 3T3-L1 cells with potencies that increased with the length chain. Butylparaben and Benzylparaben promoted lipid accumulation in hADSCs.
The EPA’s EDSP program conducted a series of in vitro assays to examine the estrogenic properties of parabens compounds. There were 15, 14, 11, 5, and 2 positive results out of total 18 arrays for Butylparaben, Propylparaben, Ethylparaben, Methylparaben, and 4-Hydroxybenzoic Acid, respectively, while in vitro antiandrogen studies showed negative results.
Metabolites of Butylparaben and Isobutylparaben, 3OH and 2OH, exhibited estrogenic properties in MCF-7 and T47D human breast cancer cells. The expression of GREB1 was induced by 3OH and 2OH metabolites and blocked by coadministration of an ER. The estrogenic activity of the 3OH and 2OH metabolites is mediated by classical ER-mediated signaling. 3OH and 2OH metabolites showed the potential for favorable ligand-binding domain interactions with human ERα.
Longer diestrus phases and reduced intervals of the estrous cycle were observed in rats orally exposed to Propylparaben or Butylparaben at a concentration of 100 mg/kg/d for 5 weeks. Propylparaben and Butylparaben decreased mRNA level of folliculogenesis-related genes (
Perinatal Methylparaben exposure in rats at doses mimicking human exposure (0.105 mg/kg/d) decreased amounts of adipose tissue and increased expansion of the ductal tree within the fat pad. Prepubertal Methylparaben treatment in rats was associated with a significant reduction in adipose tissue and more abundant glandular tissue. Long-term Methylparaben treatment from birth to lactation did not result in significant histological changes.
Oral exposure to Methylparaben at 500 mg/kg/d caused morphological changes in gerbil prostates. Male and female gerbils displayed similar alterations such as prostate epithelial hyperplasia, increased cell proliferation, and a higher frequency of AR binding activity.
In isolated mouse preantral follicle and hGC cultures, Butylparaben adversely affected steroidogenesis at concentrations relevant to human exposure (100 nM), but no effects on follicular development or survival were noted in the culture systems. Butylparaben attenuated DEHP-induced reduction in progesterone concentrations in the spent media of hGC cultures.
The presence of 500 µM Methylparaben or 10 µM Propylparaben or Butylparaben in MCF-10A nontransformed cells resulted in significant increase in colony numbers and sizes compared with control. Concentration–response experiments showed that maximal numbers of colonies were formed at 100 µM Methylparaben or 1 µM Propylparaben or Butylparaben.
Methylparaben induced a detectable decline in endogenously accumulated ROS in HRBEC cells. Methylparaben substantially reduced the fraction of OHT-induced apoptotic cells in a concentration-dependent manner. The maintenance of S-phase in OHT-treated cells, like apoptosis evasion, was correlated with increasing concentrations of Methylparaben.
Butylparaben inhibited human HTR8/SVneo cell proliferation and induced both apoptosis and endoplasmic reticulum stress at 50, 100, 200, and 400 µM. Data from the NHANES program showed that, for the 2013 to 2014 sampling period of a representative sample of the US general population, the median concentration of Methylparaben in urine was 48.1 µg/L (95th percentile: 819 µg/L), and Propylparaben in urine was 5.74 µg/L (95th percentile: 224 µg/L). For Butylparaben, the median concentration in urine was below the LOD (0.1 µg/L). In females, the median concentration of Ethylparaben was 1.6 µg/L (95th percentile: 145 µg/L), while in males were below the LOD (1 µg/L).
Analysis of data from the NHANES program showed that compared to individuals who reported “never” using mouthwash, individuals who reported daily use had significantly elevated urinary concentrations of Methylparaben and Propylparaben (30% and 39% higher, respectively). Individuals who reported “always” using sunscreen had significantly higher urinary concentrations of Methylparaben, Ethylparaben, and Propylparaben (92%, 102%, and 151% higher, respectively) compared to “never” users of sunscreen.
Women who used body and face lotions in the past 24 hours had significantly higher paraben concentrations (80%-110%) in their urine than women who reported no use. There was 100%, 72%, 96%, and 90% detection of Methylparaben, Butylparaben, Propylparaben, and Ethylparaben in urine, respectively. Breast milk samples had 82%, 66%, and 57% detection for Methylparaben, Propylparaben, and Ethylparaben, respectively.
A community-based intervention study indicated that using PCPs that are labeled to be free of parabens for 3 days lowered some parabens urinary concentrations in 100 adolescent girls: Methylparaben and Propylparaben concentrations decreased by 43.9% and 45.4%, respectively. Girls who reported using specific makeup (eg, foundation, blush, and mascara) every day versus rarely/never had higher urinary concentrations of Methylparaben (120.5 vs 13.4 ng/mL,
A statistically significant difference was observed between serum parabens in 18 women who used lipstick containing Methylparaben and Propylparaben for 5 days compared with those not using this cosmetic (
The mean concentrations of Methylparaben and Propylparaben measured in serum of 16 human are 42.6 and 7.4 µg/L, respectively, whereas the free concentrations of Methylparaben and Propylparaben in the serum are 2.2 and 0.5 µg/L, respectively.
One or more of 5 parabens (Methylparaben, Ethylparaben, Propylparaben, Butylparaben, Isobutylparaben) was detected in 99% of breast tissue samples collected from women with breast cancer, and all 5 were detected in 60% of the samples. Median concentrations were highest for Propylparaben (16.8 ng/g tissue) and Methylparaben (16.6 ng/g tissue). Propylparaben concentrations were higher in samples excised from the axilla, compared with those from the mid or medial regions of the breasts.
Methylparaben, Butylparaben, and Benzylparaben were detected in all placenta samples collected from healthy mothers. The highest measured concentration was 11.77 ng Methylparaben/g tissue.
The amounts of Butylparaben, Ethylparaben, Methylparaben, and Propylparaben were studied in human ovarian tumor samples. The tissue mass fractions of the 4 parabens in malignant tissues were at least twice as much as those present in the benign tissues. The tissue mass fractions of Methylparaben and Ethylparaben were higher than Propylparaben and Butylparaben.
One or more of 6 parabens (Methylparaben, Ethylparaben, Propylparaben, Butylparaben, Benzylparaben, heptylparaben [not a cosmetic ingredient]) as well as 4-Hydroxybenzoic Acid were detected in 20 human adipose fat samples. Ethylparaben and Propylparaben were more frequently detected than the other parabens, at a detection frequency of 60% and 50% and a GM concentration of 0.90 and 0.49 ng/g, respectively. Paraben concentrations in adipose fat samples of Caucasian volunteers were higher than those of African Americans.
One or more of 6 parabens (Methylparaben, Ethylparaben, Propylparaben, Butylparaben, Benzylparaben, and heptylparaben [not a cosmetic ingredient]) as well as 4-Hydroxybenzoic Acid, were measured in urine samples collected from 40 US children, 70 Chinese children, and 26 Chinese adults. Parabens were present predominantly (>90%) as conjugated species in urine. The median concentrations of Methylparaben and Propylparaben in US adults were 43.9 and 9.1 ng/mL, respectively. The GM concentrations of 4-Hydroxybenzoic Acid in urine from US children were 752 ng/mL for girls and 628 ng/mL for boys, which were 2 to 3 times lower than the concentrations determined for Chinese children.
One or more of 7 parabens (Methylparaben, Ethylparaben, Propylparaben, Isopropylparaben, Butylparaben, Isobutylparaben, and Benzylparaben) were detected in 144 human adipose tissue samples. Detection frequencies and median concentrations were Methylparaben (100.0%, 0.40 ng/g tissue), Ethylparaben (20.1%, <LOD), Propylparaben (54.2%, 0.06 ng/g tissue), Butylparaben (5.6%, <LOD), and Isobutylparaben (2.1%, <LOD). Isopropylparaben and Benzylparaben were not detected in any of the samples.
The EARTH study indicated the largest percentage increase for parabens was associated with the use of suntan/sunblock lotion (66%-156%) and hand/body lotion (79%-147%). The GM concentrations of Methylparaben, Propylparaben, and Butylparaben in urine were 28, 2.86, and 0.26 µg/L, respectively. Among 346 infants, none of the maternal preconception parabens concentrations were associated with birth weight. Maternal preconception Methylparaben concentration was associated with a decreased head circumference of 0.27 cm (95% CI: −0.54 to 0).
Six parabens (Methylparaben, Ethylparaben, Propylparaben, Butylparaben, Benzylparaben, and heptylparaben) and 4-Hydroxybenzoic Acid were measured in 143 urine samples from healthy, premenopausal women. 4-Hydroxybenzoic Acid was associated with increased FSH, 0.07 (95% CI: 0.01-0.13), and paraben concentrations were associated with increased E2, 0.21 (95% CI: 0.15-0.28) and increased progesterone, 0.32 (95% CI: 0.23-0.41).
Among 1,003 Puerto Rican pregnant women, median concentrations of Butylparaben were 2-fold greater than US women from the NHANES program, while concentrations of Methylparaben, Ethylparaben, and Propylparaben were lower. Positive correlation was identified between Methylparaben and Propylparaben (Spearman
Among 420 women undergoing IVF treatment, urinary concentrations of Methylparaben and Propylparaben were not associated with IVF outcomes. Of 252 adolescents participating in NBC cohort study, urine concentrations of parabens were not associated with any maladaptive behavior.
Among 152 pregnant women, a significant decrease in diastolic blood pressure was associated with exposure to parabens including Methylparaben, Ethylparaben, and Butylparaben in the second trimester (β = −0.62 mm Hg; 95% CI: −1.16 to −0.08 per doubling of Methylparaben concentrations).
Culture of
In in vitro assay, Propylparaben, Isopropylparaben, Butylparaben, Isobutylparaben, and Benzylparaben appeared to be weak irritants. The sensitization potential of the parabens tested was correlated with side-chain length: Methylparaben, Ethylparaben, Propylparaben, and Isopropylparaben were classified as weak sensitizers, and Butylparaben, Isobutylparaben, and Benzylparaben were strong sensitizers in this study.
Methylparaben elevated UVB-induced cell death in a statistically significant manner. Methylparaben elevated measurements of ROS and NO production and lipid peroxidation and activated NFκB and AP-1 in UVB-irradiated cells. Metabolic activity/number of viable cells was reduced in WCCs and HCEs in a concentration-dependent manner after exposure to Methylparaben.
Data collected by the ESSCA network between 2009 and 2012 indicated that parabens yielded less than 1% positive actions of allergic contact dermatitis in the 52,586 tests. In prospective studies, IVF outcomes were not associated with urinary Methylparaben, Propylparaben, or Butylparaben concentrations of women undergoing treatments for infertility. No significant associations were observed of the current exposure levels of Methylparaben, Ethylparaben, and Propylparaben in Chinese pregnant women with size of infants at birth. Urinary Methylparaben and Propylparaben concentrations were associated with an increase in gestational age, and Methylparaben, Butylparaben, and Propylparaben were all associated with a 34% to 50% decrease in the odds of SGA.
The associations between maternal urinary parabens (Methylparaben, Ethylparaben, Propylparaben, and Butylparaben) and plasma inflammatory markers across pregnancy were examined in 130 PTB cases and 352 controls. An interquartile range increase in Methylparaben (359 ng/mL) was positively associated with a 6.69% increase in IL-6 (95% CI: 0.02-13.8), while increase in Ethylparaben (10.4 ng/mL) was associated with a 7.7% decrease in IL-1β (95% CI: −14.1 to −0.86).
Among 602 pregnant women in Puerto Rico, urinary Butylparaben, Methylparaben, and Propylparaben were associated with decreases in SHBG by 5.27% (95% CI: −9.4 to −1.14), 3.53% (95% CI: −7.37 to 0.31), and 3.74% (95% CI: −7.76 to 0.27), respectively. Methylparaben was associated with decreases in reproductive hormones, including an 8% decrease (95% CI: -15.4, 0.61) in estriol, a suggestive 3% increase (95% CI: −2.95 to 9.61) in the progesterone/estriol ratio, and a suggestive 6.7% decrease (95% CI: −13.13 to 0.29) in T at 16 to 20 weeks.
Among 501 male partners of couples planning to become pregnant, urinary concentrations of Methylparaben, Ethylparaben, and Butylparaben were associated with diminished sperm count and several sperm motility parameters. However, seminal plasma concentrations of Ethylparaben and Benzylparaben in 339 males were associated with an increased percentage of sperm motility.
A urinary concentration increase of parabens was associated with the use of suntan/sunblock lotion (66%-156%) and hand/body lotion (79%-147%) in 400 men who reported the use of 14 PCPs. The GM concentrations of Methylparaben, Propylparaben, and Butylparaben in urine were 28, 2.86, and 0.26 µg/L, respectively.
Among 346 infants, none of the maternal preconception paraben concentrations were associated with birth weight. 114 Maternal preconception Methylparaben concentration was associated with a decreased head circumference of 0.27 cm (95% CI: −0.54 to 0).
The downward trends in sperm concentration and normal morphology among 936 men who sought infertility treatment were not affected when including urinary paraben concentrations in linear regression models, indicating that parabens exposure was not associated with the downward trends in semen parameters.
An interquartile range increase in urinary Ethylparaben (10.4 ng/mL) was associated with a 7.7% decrease in pro-inflammatory marker IL-1β (95% CI: −14.1 to −0.86). In Latino children, peripubertal urinary Methylparaben or Propylparaben concentrations were associated with altered pubertal timing; however, the causality could not be determined.
In retrospective studies, the incidence of cryptorchidism and/or hypospadias, combined, was associated with placental concentrations of Methylparaben ≥1.96 ng/g (OR = 3.18; CI = 0.88-11.48) and Propylparaben concentrations ≥1.16 ng/g (OR = 4.72; CI = 1.08-20.65). Linear regression analyses indicated an association between urinary Ethylparaben concentrations in 3-year-old boys and their body weights and heights.
Among 241 pregnant women, urinary concentrations of Butylparaben were positively associated with blood glucose levels for both the first trimester (adjusted difference = 12.5 mg/dL; 95% CI: 0.9-24.2) and second trimester (adjusted difference = 11.2 mg/dL; 95% CI: 0.2-22.3), when assessed as a mixture with 2 other parabens, Methylparaben and Propylparaben. In contrast, a negative association was found between first trimester Propylparaben and glucose (adjusted difference = −22.3 mg/dL; 95% CI: −43.2 to −1.4).
Maternal urinary paraben levels of Methylparaben, Ethylparaben, Propylparaben, Butylparaben, and Benzylparaben were measured in 850 mother–infant pairs. In all infants, each doubling increase in average Ethylparaben was associated with −2.82% (95% CI: −5.11% to −0.53%) decrease in weight
Among 473 pregnant women, 4 parabens (Methylparaben, Ethylparaben, Propylparaben, and Butylparaben) were measured in spot urine samples collected between weeks 23 and 29 of gestation. A positive association between the sum of parabens and placental weight has been identified (β = 7.12,
Among 1,087 pregnant women in China, a total of 103 (9.5%) women were diagnosed with GDM. Urinary Ethylparaben was associated with GDM. The RRs = 1.12 (95% CI: 0.63-2.01) for the second quartile, RRs = 1.11 (95% CI: 0.64-1.93) for the third quartile, and RRs = 1.70 (95% CI: 1.02-2.82) for the highest quartile, compared with the lowest quartile.
Five parabens (Methylparaben, Ethylparaben, Propylparaben, Butylparaben, and Benzylparaben) were measured in 3 spot urine samples of 478 pregnant women in China. Each 2-fold increase in average prenatal paraben concentration was associated with lower MDI scores among girls, β = −1.08 (95% CI: −2.10 to −0.06) and β = − 1.51 (95% CI: −2.69 to −0.32) for Methylparaben and Σparabens, respectively.
Methylparaben was associated with lower Th1% (RR: −3.35, 95% CI: −6.58 to −0.02) and Th2% at borderline significance (RR: −4.45, 95% CI: −8.77 to 0.08) in their children. Propylparaben was associated with decreased odds of probable asthma (OR: 0.86, 95% CI: 0.74-0.99).
Among 480 pregnant women, 130 cases of PTB were identified. Regression analyses indicated Ethylparaben was associated with increased risk for placental PTB, OR = 1.47 (95% CI: 1.14-1.91). Urinary concentrations of Methylparaben and Propylparaben were not associated with any IVF outcomes in 420 women undergoing IVF. In a different study, urine concentrations of parabens were not associated with any maladaptive behaviors. A significant decrease in diastolic blood pressure was associated with exposure to parabens in 152 pregnant women in their second trimester.
Preterm birth was associated with umbilical cord blood concentrations of Butylparaben (OR = 60.77; CI = 2.60-1,419.93) and Benzylparaben (OR = 0.03, CI = 0.01-0.44). The authors stated that the OR of 0.03 for Benzylparaben indicated a “protective effect” of Benzylparaben for PTB. Linear regression analysis indicated an association between maternal urinary concentrations and decreased gestational age and body length in newborns.
No statistically significant associations were observed between Methylparaben or Ethylparaben concentrations and the outcomes evaluated (ie, body length, gestational age at birth, birth weight, head circumference). No statistically significant associations were found between prenatal or postnatal growth of male newborns and maternal urinary paraben concentrations of Methylparaben, Ethylparaben, Propylparaben, or Butylparaben.
Linear regression analyses of data from the US NHANES program indicated an association between reduced serum T4 concentrations and urinary concentrations of Methylparaben, Ethylparaben, Propylparaben, and Butylparaben. The MPC and the results of statistical tests for trends were not statistically significant in a study of urinary concentrations of Methylparaben, Propylparaben, and Butylparaben in women undergoing infertility evaluation and OV or AFC measurements.
Analysis of data from the US NHANES program indicated an association between aeroallergen and food sensitization, combined, and urinary concentrations of Methylparaben (OR = 1.74; CI = 1.02-3.22), Propylparaben (OR = 2.04; CI = 1.12-3.74), and Butylparaben (OR = 1.55; CI = 1.02-2.33). The results also indicated associations between urinary concentrations of Methylparaben and nonatopic asthma (OR = 0.025; CI = 0.07-0.90) and nonatopic wheeze (OR = 0.23; CI = 0.05-0.99).
Urine samples were collected from 696 pregnant women in China. No significant association was found between parabens and GDM among the overall population. However, significant nonlinear associations of Propylparaben and the summed estrogenic activity of parabens with GDM were found in the stratified analysis by prepregnancy BMI in the overweight/obese population, with adjusted ORs of 3.47 (95% CI: 1.28-9.42) and 2.87 (95% CI: 1.07-7.73) for GDM in the second tertile of urinary Propylparaben and the summed estrogen activity, respectively, when compared to the first tertile.
One study examined the association between parabens and asthma morbidity among 450 children with asthma and with asthma prevalence among 4,023 children participating in the US NHANES program (2005-2014). An increased prevalence odds of reporting emergency department visits were observed for every 10-fold increase in Methylparaben and Propylparaben concentrations among boys with asthma (prevalence OR = 2.61, 95% CI: 1.40-4.85 and OR = 2.18, 95% CI: 1.22-3.89, respectively). Among children in the general population, no overall associations with current asthma were observed, although there was a positive trend with Propylparaben and a current asthma diagnosis.
Among 1,693 black women aged 23 to 34 years, Methylparaben and Butylparaben concentrations were 30% lower for BMI ≥35 vs <25 kg/m2 (95% CI: −48.0% to −7.7% for Methylparaben and 95% CI: −49.6% to −4.6% for Butylparaben, respectively).
Of 156 men under 45 years of age who attended the infertility clinic for diagnostic purposes with normal semen concentration, a positive association was found between urinary level of Butylparaben and XY18 disomy (
The highest (vs lowest) quintiles of urinary Methylparaben, Propylparaben, and sum of parabens were associated with breast cancer, ORs of 1.50 (95% CI = 1.03-2.18), 1.31 (95% CI = 0.90-1.90), and 1.35 (95% CI = 0.93-1.97), respectively, among 711 women with breast cancer and 598 women without breast cancer.
No statistically significant associations were found between the urinary concentrations of Methylparaben, Propylparaben, or Butylparaben and serum hormone concentrations, semen quality parameters, and motion characteristics (for all but one indicator). The exception was a trend for increased tail% in comet assays of sperm DNA with increasing Butylparaben concentrations.
Urinary levels of Ethylparaben and Butylparaben were associated with an increase in the percentage of sperm with abnormal morphology. Urinary Isobutylparaben concentrations were significantly associated with an increase in the percentage of sperm, with level of Isobutylparaben increased high DNA stainability. Neither categories of urinary concentrations of parabens nor continuous concentrations of parabens were associated with the level of reproductive hormones. Urinary concentrations of Methylparaben and Propylparaben were not related to any of the examined semen quality parameters, sperm DNA damage, or the level of reproductive hormones.
Urinary paraben concentrations of Methylparaben, Ethylparaben, Propylparaben, and Butylparaben were measured in 215 young healthy men, 94% of whom had detectable urinary concentrations of parabens. Urinary concentrations of parabens were not significantly associated with any semen parameters or any of the reproductive hormone levels.
Among 42 partners of couples who visited a gynecology clinic for infertility consultation, no significant association was found between semen parameters (sperm volume, concentration, and motility) and urinary paraben concentrations in regression analyses.
In cord plasma of 27 healthy pregnant women, Methylparaben, Propylparaben, and the sum of all measured parabens (Methylparaben + Ethylparaben + Propylparaben + Butylparaben) were inversely associated with T levels.
A conservative risk assessment was performed. Therein, an NOAEL value of 160 mg/kg/d for Butylparaben was determined to be adequate in consideration of the new data in the category of endocrine activity and from DART studies. For the purposes of an MOS calculation, the Panel considered a scenario wherein a consumer would use a set of cosmetic products containing Butylparaben. Therein, an aggregate exposure to 4 main categories of products was considered: (1) oral products, (2) eye products, (3) leave-on products, and (4) rinse-off product; the global daily exposure of products for each category was estimated using the maximum use concentration of Butylparaben in each category, 0.2%, 0.5%, 0.24%, and 0.33%, respectively. The Panel noted that the available in vitro percutaneous absorption studies using human split- or full-thickness skin suggest a conservative assumption of human dermal penetration of unmetabolized Butylparaben at 3.7%, though this could vary due to differences in animal species used, matrix effects, and other experimental conditions. Considering the variables, and taking into account that dermal absorption of lower molecular weight parabens is higher, the Panel selected a 50% dermal absorption rate of unmetabolized parabens as adequately conservative for the calculation of the MOS. The MOS for adults was 457 for Butylparaben. Since alkyl parabens undergo the similar enzymatic hydrolysis to form 4-Hydroxybenzoic Acid, such a conservative MOS of Butylparaben for adults could then be inferred to other single alkyl parabens.
Since multiple parabens are commonly combined for use in a single formulation, and no use data are available for such combinations, the Panel used the above parameters to calculate a maximum combined parabens (Σparabens) use concentration, starting from an MOS of 100. Utilizing this protective MOS for Σparabens, the maximum use concentration was calculated to be 1.1%.
A human paraben PBPK model developed to predict the plasma-free paraben concentration based on 95th percentile parabens concentration in urine reported in US NHANES program (2009-2010 collection period). An in vitro-based cumulative MOS was calculated by comparing the effective concentrations from an in vitro assay of estrogenicity to the predicted free plasma paraben concentrations (Methylparaben + Ethylparaben + Butylparaben). The calculated cumulative MOS for adult females was 108, whereas the cumulative MOS for males was 444.
Considering aggregate exposure from various sources, for example, cosmetics, food, and pharmaceutical use, the total combined exposure to parabens was estimated. Refinement techniques were applied in comparison with simply summed exposures from all multiple cosmetic product types. Approximately 60% to 90% of the model predictions from 5 implemented models were within a factor of 10 of the observed paraben exposures, while 30% to 40% of the predictions were within a factor of 3. More importantly though, in all cases, aggregate exposure estimates were significantly greater than the exposures derived from experimental biomonitoring data.
Discussion
The Panel discussed the issue of incidental inhalation exposure to parabens. The Panel noted that some of the parabens were reported to be used in cosmetic powder and sprays, at very low concentrations, which may result in incidental inhalation exposure; for example, Ethylparaben is used in face powders at up to 0.5%. The Panel noted that in aerosol products that are widely applied, for example, hair sprays, 95% to 99% of droplets/particles would not be respirable to any appreciable amount. The Panel also noted that, while particle/droplet size is an important parameter, the physicochemical properties of ingredients in a spray formulation and the realistic exposure factors under in-use conditions also play significant roles in inhalation safety of parabens as spray formulation. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
The Panel noted that the EU Cosmetic Regulation has banned the use of Isopropylparaben, Isobutylparaben, phenylparaben, Benzylparaben, and pentylparaben as preservatives in cosmetic products. It is the opinion of the Panel that the scientific rationale for restricting these ingredients warrants further justification.
The Panel noted that both in vitro and in vivo studies indicate a rapid and effective metabolism of parabens by carboxylesterases after oral or dermal exposure. Parabens are further metabolized by conjugation with glucuronide, sulfate, or glycine prior to excretion. When applied to human skin, parabens are metabolized to 4-Hydroxybenzoic Acid. Whereas older studies suggested that unmetabolized parabens are not excreted, recent studies with more sensitive analytical methods have measured unmetabolized parabens and their metabolites following dermal exposures. Because each of these alkyl parabens (ie, excluding Benzylparaben) undergo extensive metabolism in a similar way, the Panel felt that safety data for one of these alkyl parabens could be used to support the safety of the other alkyl parabens.
The Panel discussed concerns about the relevance of the oral animal studies to human risk assessment in that the rapid and effective metabolism of parabens observed in rodents does not occur in humans. Species differences in the esterase affinities and activities must be carefully taken into account for deriving a safe level of exposure in humans. The Panel noted that uncertainties relate to data gaps on dermal absorption of unmetabolized parabens by human skin in vivo and in vitro. One human toxicokinetic study indicates after repeated dermal exposure to Butylparaben at a daily dose of 10 mg/kg bw/d for 5 days, about 2.1% unmetabolized Butylparaben was detected in the urine of the participants. However, the Panel noted that a conservative estimation shows that daily exposure of consumers to Butylparaben is much lower (0.66 mg/kg bw/d). While the SCCS derived the value of 3.7%, based on in vitro studies using human split- or full-thickness skin, as a worst-case assumption for the dermal absorption of unmetabolized Butylparaben, absorption may be variable due to differences in animal species used, matrix effects, other experimental conditions, and dermal absorption of lower molecular weight parabens (which is likely to be higher). In light of these facts, the Panel estimated a 50% dermal absorption rate of unmetabolized parabens in the calculation of an MOS, which represents a conservative assumption.
The Panel expressed concern about new data from DART studies that indicated lower NOAEL values than the one used in the previous Panel safety assessment of the parabens. One of these studies indicated reduced sperm counts and reduced expression of testicular CYP19a1 and a reduction in the Sertoli/Leydig cell marker Nr5a1 in the testes of offspring of female rats orally dosed with 10 mg/kg bw/d Butylparaben during the gestation and lactation periods. The Panel noted that the reduction in epididymal sperm count has shown the same effect across all doses from 10 to 500 mg/kg bw/d in this study, decreasing 76% to 78% compared to controls, whereas a dose–response relationship is expected between ER agonists exposure and sperm count decrease. The Panel also noted that wide variation exists in measuring epididymal sperm count between different laboratories and/or different experimental technicians; thus, the decline in sperm counts in this study warrants further validation by making comparisons to historical sperm count control databases. In addition, the Panel noted that the data for the DART end points of AGD, epididymal sperm count, and histological examinations did not show consistency at doses ranging from 10 to 100 mg/kg bw/d when compared to other DART studies that followed similar Butylparaben exposure scenarios. In contrast, data are more consistent at doses ranging from 160 to 1,000 mg/kg bw/d.
The Panel also discussed the conflicting data from other DART studies and agreed that many of these reports (1) are irrelevant to the routes of exposure associated with intended cosmetic use or otherwise did not account for the extensive metabolism of parabens (to metabolites with no known DART activity), (2) are the result of poorly designed studies, and (3) were not verified by other methods. Recent studies have shown that the window of greatest susceptibility is the early postnatal period. In these studies, higher free concentrations of Butylparaben were measured in the plasma of rat pups during early lactation, compared to Butylparaben levels in dams and fetuses. This attributed to poor conjugation in pups, resulting in higher exposure to free Butylparaben. Thus, after careful consideration of all the new data, the Panel determined an NOAEL of 160 mg/kg bw/d for Butylparaben. The Panel determined the different use concentrations and exposures of Butylparaben in various cosmetic product category should be considered when estimating the systemic exposure levels for the MOS calculation.
The Panel recognized that the study chosen by the SCCS for the calculation of the MOS of Butylparaben examined DART end points in male rats and involved subcutaneous instead of oral administration of Butylparaben during the lactation period. The SCCS acknowledged an NOEL of 2 mg/kg bw/d, instead of an NOAEL, for deriving the MOS of Butylparaben. In order to obtain an acceptable MOS ≥100, the SCCS recommended the maximum use concentration of Butylparaben in the finished cosmetic products be set to 0.19% (0.14% as acid). The calculation is based on the assumptions that the maximum exposure to cosmetics by an adult (60 kg body weight) is 17.4 g/d, and the human dermal penetration rate of unmetabolized Butylparaben is 3.7%.
However, the Panel considered that the study with an NOEL of 2 mg/kg bw/d suffers from several critical limitations: (1) this study involves a subcutaneous route of exposure, which may result in chemicals circumventing the physiological barriers and bypassing the portal of entry metabolism and is therefore not considered suitable for quantitative risk assessment in the context of cosmetic usage; (2) this study is not an OECD TG study (eg, the Butylparaben-treated group contained only 3 rats and the control group contained only 5 rats); (3) only 1 postpartum dose at 2 mg/kg bw/d was tested; (4) male rats were exposed to Butylparaben postnatally, which did not examine the generation toxicity (eg, a more robust study design should involve gestational exposure of paraben to pregnant rats while examining toxicity in the male offspring); and (5) typical DART end points were not covered, such as AGD, PPS, weight of the epididymis and seminal vesicle, sperm counts, reproductive hormone levels, and so on.
The Panel also recognized that these ingredients can enhance the penetration of other ingredients through the skin. The Panel cautioned that care should be taken in formulating cosmetic products that may contain these ingredients in combination with any ingredients whose safety was based on their lack of dermal absorption data or when dermal absorption was otherwise a concern.
The Panel discussed the bioaccumulation potential of parabens. The Panel noted that, as lipid-soluble chemicals, parabens may distribute to tissues despite metabolism. Recent studies have demonstrated the presence of parabens in various human tissues. However, the data are equivocal regarding cumulative storage in such tissues.
The Panel noted that paraben exposures are attributed to cosmetic products, foods, medicines, and other sources. Refined aggregate exposure models suggest that cosmetic product use is a major source of dermal exposure to parabens. However, the vast quantity of biomonitoring data indicates that systemic exposure resulting from the cosmetic use of these ingredients is low. The Panel noted that measurements of total parabens in human adipose tissue warrant further investigation with larger sample sizes and unbiased analytical methods. In one study, total paraben measurements (the sum concentration of free and conjugated parabens and their metabolite 4-Hydroxybenzoic Acid) were compromised by alkaline hydrolysis in the tissue due to the use of alkali in the liposuction procedure, that is, high concentrations of 4-Hydroxybenzoic Acid could be an artifact from the reaction of paraben esters with sodium bicarbonate solution used in liposuction procedures. In another study, while a positive, though not statistically significant, association between age and Methylparaben concentrations in human adipose tissue was observed, a positive association with age might also be a consequence of the commonly lower metabolic activity in older individuals (which may delay the metabolism and clearance of chemicals).
The Panel also reviewed data from a kinetic-based study which expands the use of human biomonitoring data in this safety assessment. As biomonitoring data integrate all routes (inhalation, dermal, and oral) and sources (cosmetics, foods, drugs, etc) of exposure, it provides valuable perspective to help evaluate aggregate exposure to parabens. The human paraben PBPK model was used to estimate the plasma-free paraben concentration in adults consistent with 95th percentile urine concentration reported in US NHANES program (2009-2010 collection period). Based on the model, the calculated cumulative MOS for adult females was 108 and for males was 444. Both cumulative MOS derived from human epidemiological survey are sufficient to ensure human safety.
The Panel also discussed the safety of parabens as used in vaginally applied cosmetic products. One published reference was submitted to the Panel, along with the assertion that these ingredients cause irreparable damage to sperm and may preclude fertilization in users. However, of the multiple end points asserted in the reference, each was either constructed around an improperly chosen/designed assay to make such assertions unequivocally and/or resulted in no significant effects. Another published reference asserted these ingredients may increase the chances of developing a vaginal yeast infection. However, the cell culture studies performed therein tested extremely high concentrations as compared to cosmetic use (ie, 15%-25% preservative in these studies vs a maximum use concentration of parabens in cosmetics of 0.8%). The Panel classified these studies as illustrations of potential, general hazards, which fail to demonstrate risks relevant to cosmetic safety in the context of concentration of use.
The Panel noted that recent epidemiology studies suggested paraben exposure association with different types of health outcomes, such as a lower mental developmental index in girls, adverse impacts on fetal and childhood growth, decreased diastolic blood pressure during pregnancy, increased risk for placental PTB, disturbance of reproductive hormone levels, and altered frequency of sperm disomy; however, these were not confirmed by subsequent or previous epidemiologic investigations. Sources of parabens exposure in these studies are broadly environmental and not specified. More importantly, paraben exposures by the study population are always coupled with other preservatives and active ingredients that are used in a wide variety of consumer products, including phthalates, bisphenol A, triclosan, and so on. Therefore, the currently available scientific evidence lacks clarity regarding any cause-and-effect relationship between parabens and human health outcomes. It remains to be determined whether the costimulatory effects require multiple exposures. Further studies in larger populations and with more repeated measures across pregnancy would be useful to confirm these findings and to better understand if the hormone changes may affect downstream maternal and infant health outcomes. The Panel also noted that several studies suggested urinary paraben concentrations were associated with glucose levels in women at high risk of GDM; however, a causal relationship cannot be established. In one study, a positive association (with Propylparaben) was identified among overweight/obese pregnant women, but not in the overall population, and importantly, evidence available in other studies indicates either no association or negative association between urinary Propylparaben concentration and GDM.
A conservative risk assessment was performed. Therein, an aggregate exposure to 4 main categories of products was considered: (1) oral products, (2) eye products, (3) leave-on products and (4) rinse-off product; the global daily exposure of products for each category was estimated using the maximum use concentration of Butylparaben in each category, that is, 0.2%, 0.5%, 0.24%, and 0.33%, respectively. The MOS for adults was 457 for Butylparaben. Since alkyl parabens undergo the similar enzymatic hydrolysis to form 4-Hydroxybenzoic Acid, such a conservative MOS of Butylparaben for adults could then be inferred to other single alkyl paraben uses.
Because multiple parabens are commonly used in any given cosmetic product, a maximum safe use concentration for combined paraben (Σparabens) use in a single formulation was calculated using a protective MOS level of 100. The maximum use concentration was calculated to be 1.1%. Accordingly, the Panel determined that the commonly used (eg, in the EU) limitation of 0.8% is conservative and is safe for human health when parabens are used in combination in a cosmetic product.
The Panel considered the potential for exposure to these ingredients to cause irritation or induce skin sensitization. The Panel noted that skin tests on product formulations containing from 0.1% to 0.8% of one, or a combination of two, of the parabens showed no evidence of significant irritation or sensitization potential for these ingredients. All animal sensitization tests indicated that the parabens are nonsensitizing. These data further support the safe combined use of parabens in a single cosmetic formulation at up to 0.8%.
However, the Panel concluded that the available data are insufficient to determine the safety of Benzylparaben. The data needed to determine the safety of this ingredient comprise an NOAEL derived from DART studies. The Panel noted that this ingredient is not reported to be in current use.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that the following 20 parabens are safe in cosmetics in the present practices of use and concentration described in the safety assessment when the sum of the combined concentration of parabens in any given formulation does not exceed 0.8%.
Butylparaben
Calcium Paraben
Ethylparaben
Isobutylparaben
Isopropylparaben
Methylparaben
Potassium Butylparaben
Potassium Ethylparaben
Potassium Methylparaben
Potassium Paraben
Potassium Propylparaben
Propylparaben
Sodium Butylparaben
Sodium Ethylparaben
Sodium Isobutylparaben
Sodium Isopropylparaben
Sodium Paraben
Sodium Methylparaben
Sodium Propylparaben
4-Hydroxybenzoic Acid
*
The Expert Panel for Cosmetic Ingredient Safety also concluded that the available data are insufficient to support a conclusion of safety for Benzylparaben in cosmetics. (This ingredient is not reported to be in current use.)
Footnotes
Authors’ Note
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Author Contributions
P. Cherian contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. J. Zhu contributed to conception and design, contributed to analysis and interpretation, and drafted the manuscript. W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, J. Marks, R. Shank, T. Slaga, and P. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. B. Heldreth contributed to design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article in this supplement was sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.